aiims-2018
AIIMS 2018
MEDICAL 2018 Previous Year
3 hDuration
180Total Marks
360Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 360 across 3 section(s); maximum marks: 180.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+1 / 0.33
Assertion (A) Separation of Zr and Hf is
difficult.
Reason (R) Zr and Hf lie in the same group
of the periodic table.
Q2.
mcq single
+1 / 0.33
The correct order of spin only magnetic moment (in BM) for $$\mathrm{Mn}^{2+}, \mathrm{Cr}^{2+}$$ and $$\mathrm{Ti}^{2+}$$ ions is
Q3.
mcq single
+1 / 0.33
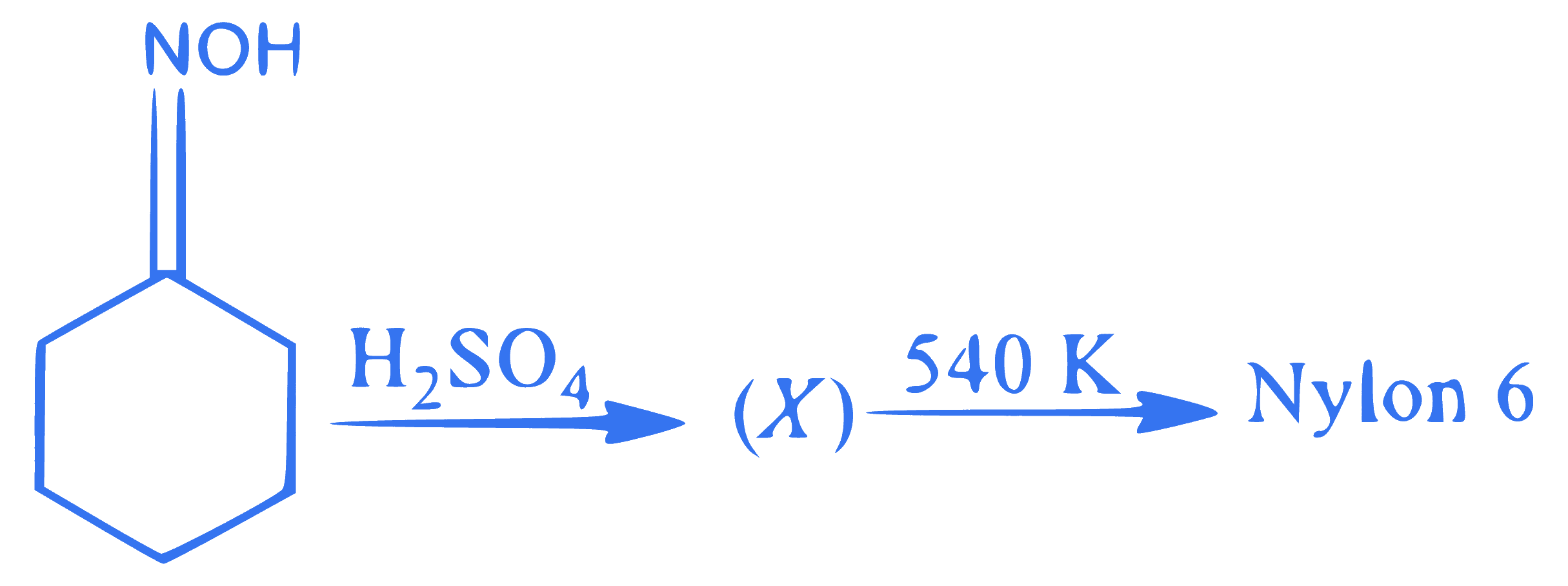 In the above reaction sequence, $$X$$ is
In the above reaction sequence, $$X$$ is
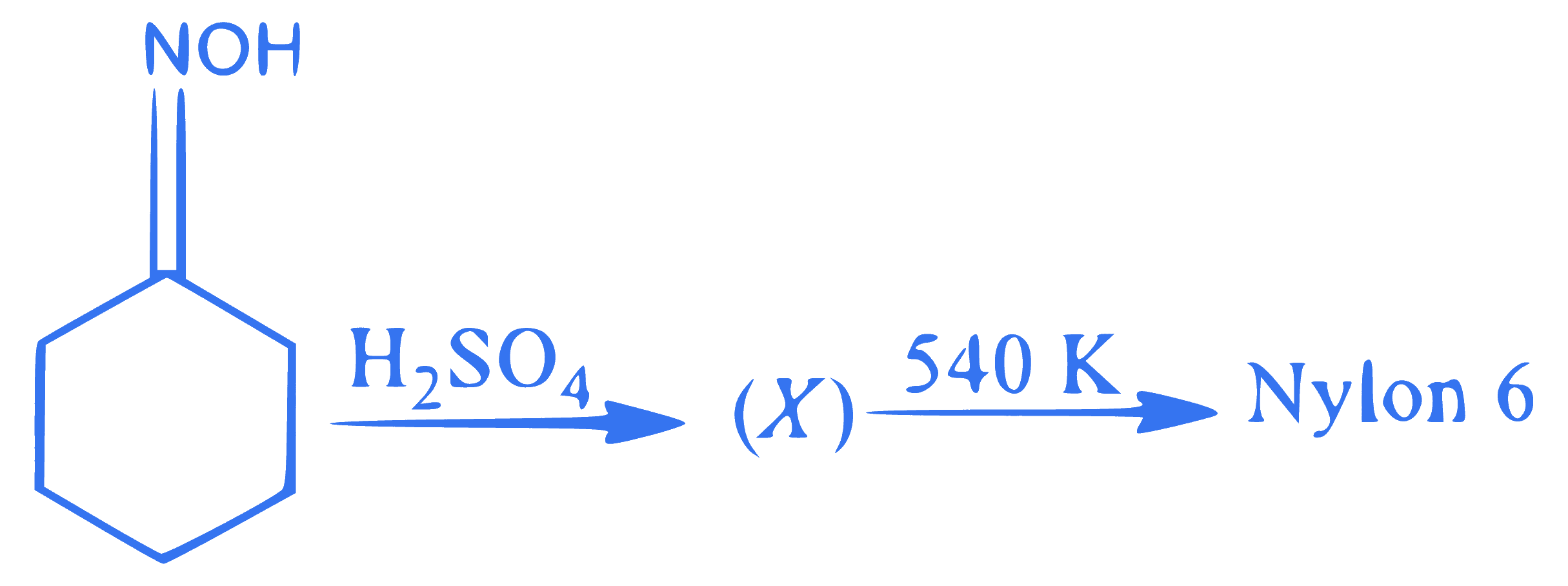 In the above reaction sequence, $$X$$ is
In the above reaction sequence, $$X$$ is
Q4.
mcq single
+1 / 0.33
Assertion (A) Most of the synthetic
polymers are not biodegradable.
Reason (R) Polymerisation process induces
toxic polymerisation.
Q5.
mcq single
+1 / 0.33
Assertion (A) The molecular mass of the
polymers cannot be calculated using the
boiling point or freezing point method.
Reason (R) The freezing point method is
used for determining the molecular mass of
small molecules.
Q6.
mcq single
+1 / 0.33
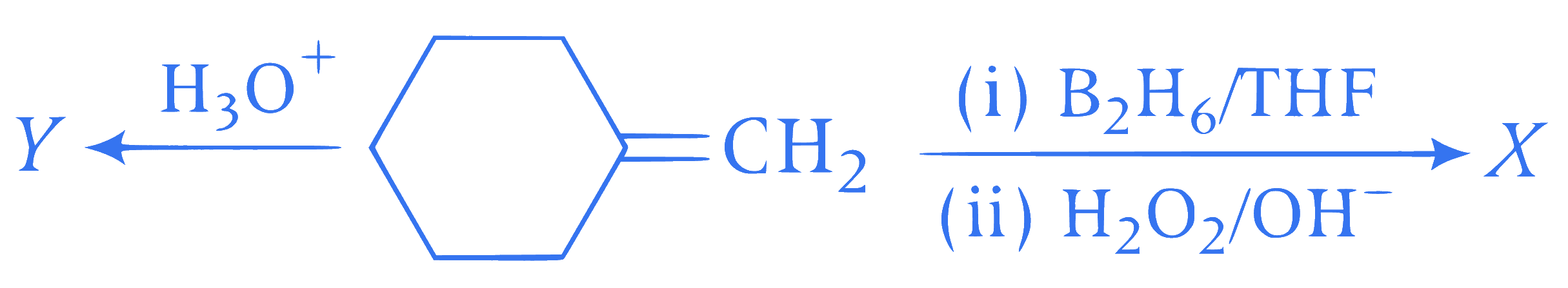 X and Y respectively are
X and Y respectively are
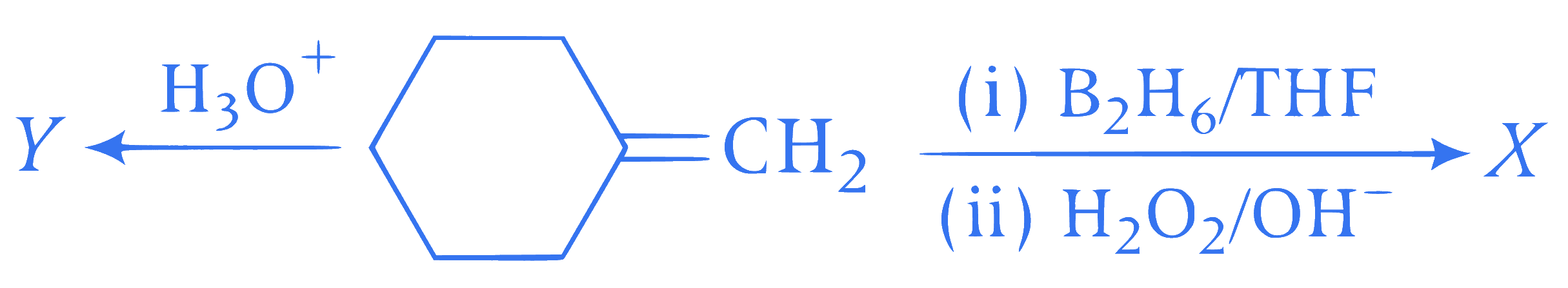 X and Y respectively are
X and Y respectively are
Q7.
mcq single
+1 / 0.33
Assertion (A) Among the two $$\mathrm{O}-\mathrm{H}$$ bonds in $$\mathrm{H}_2 \mathrm{O}$$ molecule, the energy required to break the first $$\mathrm{O}-\mathrm{H}$$ bond and the other $$\mathrm{O}-\mathrm{H}$$ bond is same.
Reason (R) This is because the electronic environment around oxygen is the same even after breakage of one $$\mathrm{O}-\mathrm{H}$$ bond.
Q8.
mcq single
+1 / 0.33
The structure of $$\mathrm{IF}_7,$$ is
Q9.
mcq single
+1 / 0.33
The solubility product of sparingly soluble salt $$A X_2$$ is $$3.2 \times 10^{-11}$$. Its solubility (in mol/L) is
Q10.
mcq single
+1 / 0.33
Assertion (A) The reaction
$$\begin{aligned}
2 \mathrm{NO}+\mathrm{O}_2 & \longrightarrow 2 \mathrm{NO}_2 \\
\text { and } \quad 2 \mathrm{CO}+\mathrm{O}_2 & \longrightarrow 2 \mathrm{CO}_2
\end{aligned}$$
proceeds at the same rate because they are similar.
Reason (R) Both the reactions have same activation energy.
Q11.
mcq single
+1 / 0.33
The rate constant for the first order
decomposition of a certain reaction is
described by the equation $$\ln k\left(\mathrm{~s}^{-1}\right)=14.34-\frac{1.25 \times 10^4 \mathrm{~K}}{T}$$. The energy of activation for this reaction is
Q12.
mcq single
+1 / 0.33
Among the following statements, the correct statement about the half-life period for a first order reaction is
Q13.
mcq single
+1 / 0.33
A gas ‘X’ is used in filling balloons for
meteorological observations. It is also used
in gas-cooled nuclear reactors. Here, the gas
X is
Q14.
mcq single
+1 / 0.33
When an excess and a very dilute aqueous
solution of KI is added to very dilute
aqueous solution of silver nitrate. The
colloidal particles of silver iodide which are
associated with the Helmholtz double layer
in the form of
Q15.
mcq single
+1 / 0.33
Assertion (A) Micelles are formed by
surfactant molecules above the critical
micellar concentration (CMC).
Reason (R) The conductivity of a solution
having surfactant molecules decreases
sharply at the CMC.
Q16.
mcq single
+1 / 0.33
Assertion (A) The boiling points of alkyl halides decrease in the order $$R \mathrm{I}>R \mathrm{Br}>\mathrm{RCl}>\mathrm{RF}$$.
Reason (R) The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
Q17.
mcq single
+1 / 0.33
A bromoalkane '$$X$$' reacts with magnesium in dry ether to form compound '$$Y$$'. The reaction of '$$Y$$' with methanol followed by hydrolysis yield an alcohol having molecular formula, $$\mathrm{C}_4 \mathrm{H}_{10} \mathrm{O}$$. The compound '$$X$$' is
Q18.
mcq single
+1 / 0.33
Which of the following represents
Wurtz-Fittig reaction?
Q19.
mcq single
+1 / 0.33
Benzoic acid undergoes dimerisation in benzene solution. The van't Hoff factor $$(i)$$ is related to the degree of association '$$x$$' of the acid as
Q20.
mcq single
+1 / 0.33
Assertion (A) $$K_p$$ can be equal to or less than or even greater than the value of $$K_C$$.
Reason (R) $$K_p=K_C(R T)^{\Delta n}$$
Relation between $$K_p$$ and $$K_C$$ depends on the change in the number of moles of gaseous reactants and products.
Q21.
mcq single
+1 / 0.33
At equilibrium, the concentration of
$$\begin{gathered}
\mathrm{N}_2=3.0 \times 10^{-3} \mathrm{M} \\
\mathrm{O}_2=4.2 \times 10^{-3} \mathrm{M} \\
\text{and} \quad \mathrm{NO}=2.8 \times 10^{-3} \mathrm{M}
\end{gathered}$$
in a sealed vessel at $$800 \mathrm{~K}$$ and $$1 \mathrm{~atm}$$ pressure. What will be $$K_p$$ for the given reaction?
$$\mathrm{N}_2(g)+\mathrm{O}_2(g) \rightleftharpoons 2 \mathrm{NO}(g)$$
Q22.
mcq single
+1 / 0.33
In which of the following arrangements, the
order is not strictly according to the property
written against it?
Q23.
mcq single
+1 / 0.33
Assertion (A) Electron gain enthalpy
becomes less negative as we go down a group.
Reason (R) Size of the atom increases on
going down the group and the added electron
would be farther away from the nucleus.
Q24.
mcq single
+1 / 0.33
The ionic radii $$(\mathop A\limits^o)$$ of $$\mathrm{C}^{4-}$$ and $$\mathrm{O}^{2-}$$ respectively are 2.60 and 1.40. The ionic radius of the isoelectronic ion $$\mathrm{N}^{3-}$$ would be
Q25.
mcq single
+1 / 0.33
$$\Delta H$$ and $$\Delta E$$ for the reaction,
$$\mathrm{Fe}_2 \mathrm{O}_3(s)+3 \mathrm{H}_2(g) \longrightarrow 2 \mathrm{Fe}(s)+\mathrm{H}_2 \mathrm{O}(l)$$
at constant temperature are related as
Q26.
mcq single
+1 / 0.33
In an adiabatic process, no transfer of heat
takes place between system and
surroundings. Choose the correct option for
free expansion of an ideal gas under
adiabatic condition from the following
Q27.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{NaCl}$$ reacts with concentrated $$\mathrm{H}_2 \mathrm{SO}_4$$ to give colourless fumes with pungent smell. But on adding $$\mathrm{MnO}_2$$ the fumes become greenish yellow.
Reason (R) $$\mathrm{MnO}_2$$ oxidises $$\mathrm{HCl}$$ to chlorine gas which is greenish yellow.
Q28.
mcq single
+1 / 0.33
A green yellow gas reacts with an alkali
metal hydroxide to form a halate which can
be used in fireworks and safety matches. The
gas and halate are, respectively
Q29.
mcq single
+1 / 0.33
Which of the following oxyacid does not contain $$\mathrm{P}-\mathrm{O}-\mathrm{P}$$ bond?
Q30.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{N}_2$$ is less reactive than $$\mathrm{P}_4$$.
Reason (R) Nitrogen has more electron gain enthalpy than phosphorus.
Q31.
mcq single
+1 / 0.33
Affinity for hydrogen decreases in the group
from fluorine to iodine. Which of the
halogen acids should have highest bond
dissociation enthalpy?
Q32.
mcq single
+1 / 0.33
Which of the following products will be obtained when copper metal is reacted with $$\mathrm{HNO}_3$$ ?
Q33.
mcq single
+1 / 0.33
$$\mathop {8N{H_3}}\limits_{(Excess)} + 3C{l_2}\buildrel {} \over
\longrightarrow X$$
$$N{H_3} + \mathop {3C{l_2}}\limits_{(Excess)} \buildrel {} \over
\longrightarrow Y$$
What is X and Y in the above reaction?
Q34.
mcq single
+1 / 0.33
The electronic configuration of central metal atom/ion in $$\left[\mathrm{Co}(\mathrm{CN})_6\right]^{3-}$$ is
Q35.
mcq single
+1 / 0.33
Which of the following compound has same
oxidation state of the central metal atom in
the cationic and anionic part?
Q36.
mcq single
+1 / 0.33
The spin only magnetic moment of $$[\mathrm{MnBr}_4]^{2-}$$ is $$5.9 \mathrm{~BM}$$. The geometry of this complex ion is
Q37.
mcq single
+1 / 0.33
The IUPAC name of the complex $$[\mathrm{Pt}\left(\mathrm{NH}_3\right)_3 \mathrm{Br}\left(\mathrm{NO}_2\right) \mathrm{Cl}] \mathrm{Cl}$$ is
Q38.
mcq single
+1 / 0.33
Among the following complex ions, the one
which shows geometrical isomerism will be
Q39.
mcq single
+1 / 0.33
Assertion (A) Toxic metal ions are removed
by the chelating ligands.
Reason (R) Chelate complexes tend to be
more stable.
Q40.
mcq single
+1 / 0.33
**Assertion (A) :** The pH of acid rain is less
than 5.6.
**Reason (R) :** Carbon dioxide present in the
atmosphere dissolves in rain water and
becomes carbonic acid.
Q41.
mcq single
+1 / 0.33
The correct order of decreasing stability of
the following carbocation is
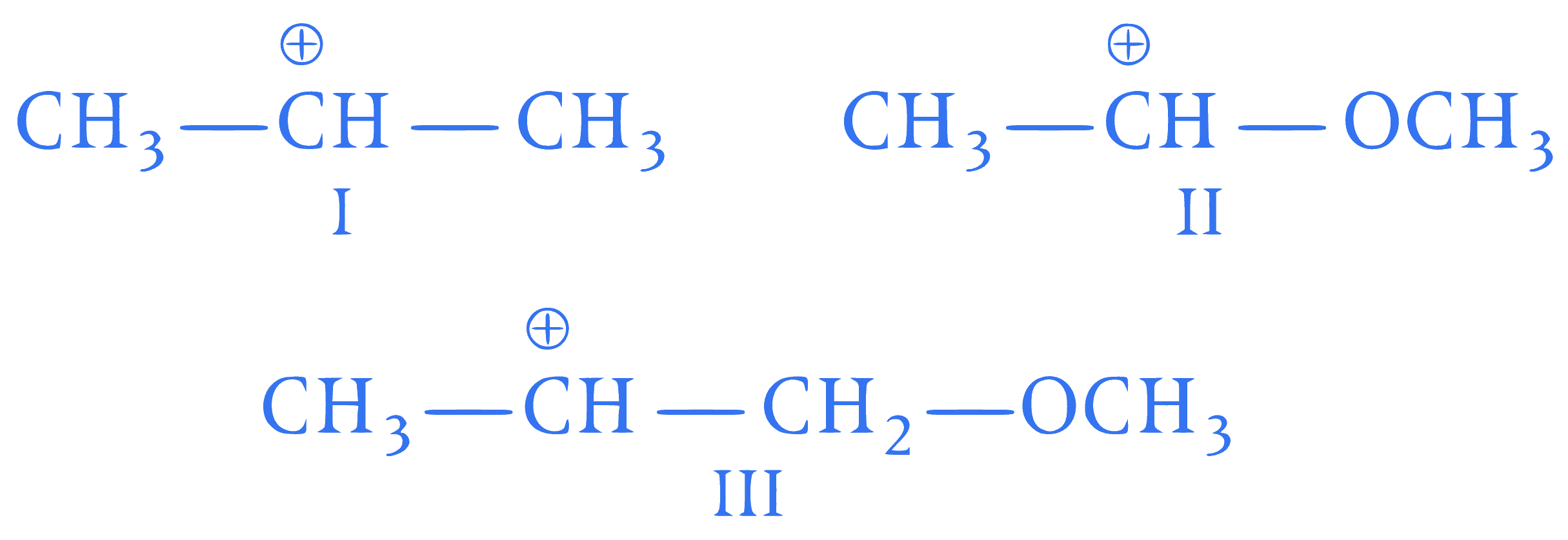

Q42.
mcq single
+1 / 0.33
The correct IUPAC name of the given compound is
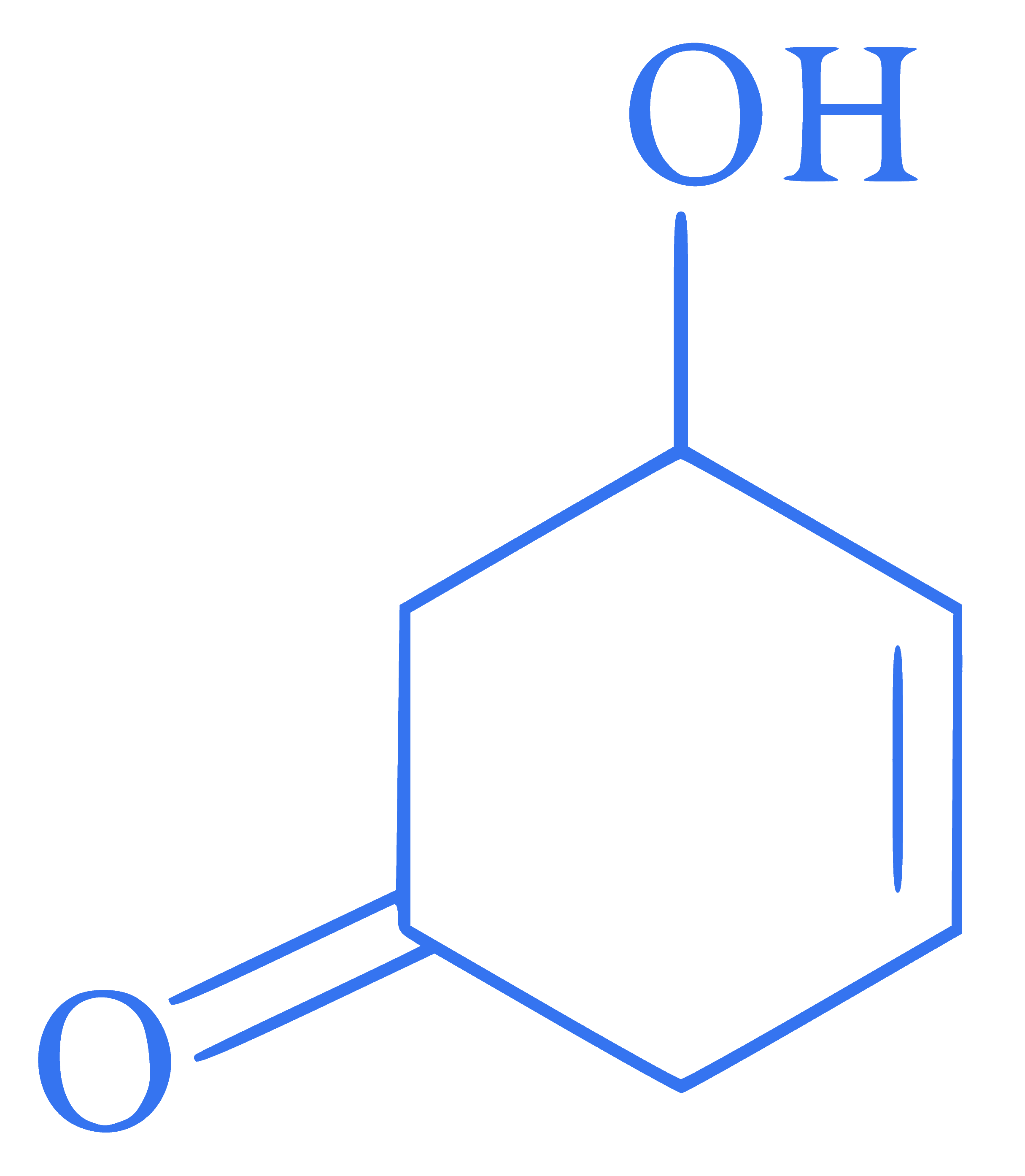

Q43.
mcq single
+1 / 0.33
Food preservatives prevent spoilage of food due to microbial growth. The most commonly used preservatives are
Q44.
mcq single
+1 / 0.33
The relative reactivities of acyl compounds
towards nucleophilic substitution are in the
order of
Q45.
mcq single
+1 / 0.33
Which of the following compounds do not
undergo aldol condensation?
Q46.
mcq single
+1 / 0.33
Assertion (A) Beryllium carbonate is kept
in the atmosphere of carbon dioxide.
Reason (R) Beryllium carbonate is unstable
and decomposes to give beryllium oxide and
carbon dioxide.
Q47.
mcq single
+1 / 0.33
Assertion (A) IUPAC name of the compound
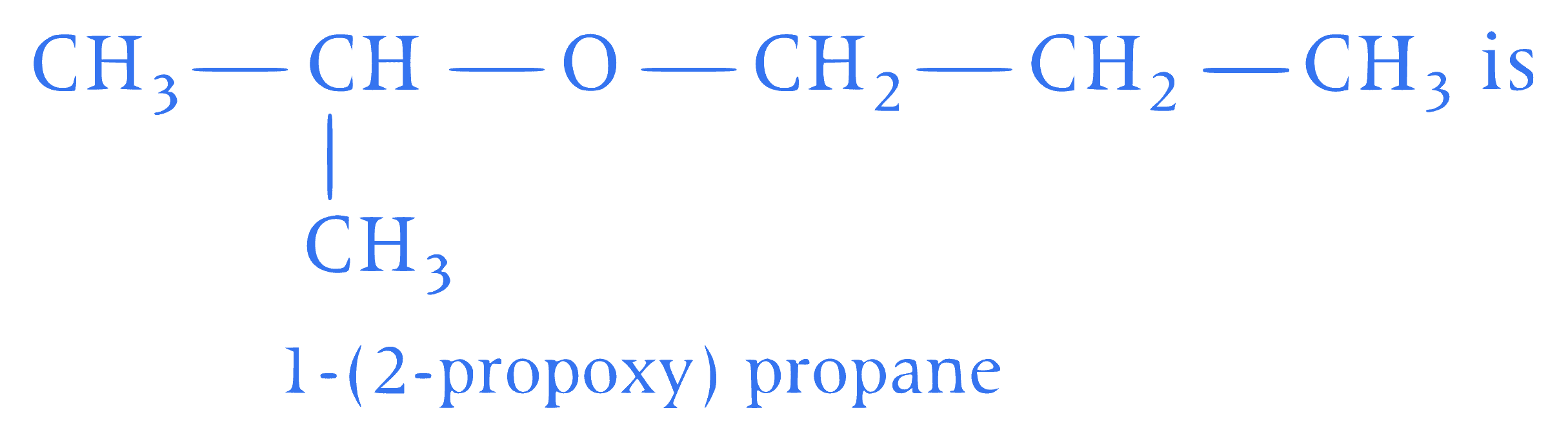 Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
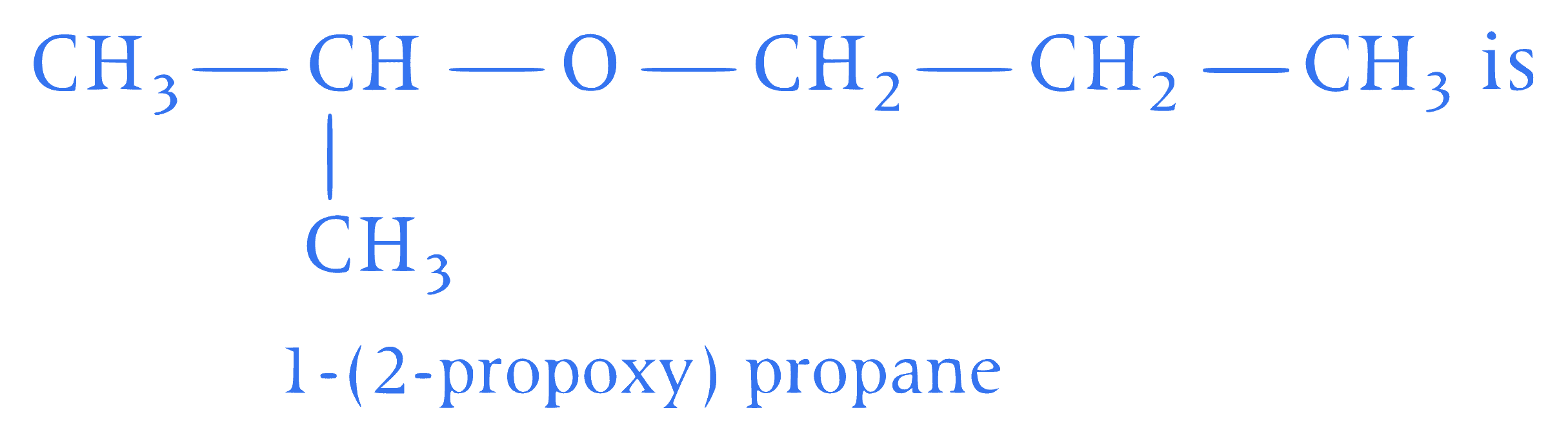 Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
Q48.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{CH}_3 \mathrm{OH}$$ and $$\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{OH}$$ can be distinguished by haloform test.
Reason (R) Haloform test is given by $$2^{\circ}$$ alcohol.
Q49.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{CH}_4$$ does not react with $$\mathrm{Cl}_2$$ in dark.
Reason (R) Chlorination of $$\mathrm{CH}_4$$ takes place in sunlight.
Q50.
mcq single
+1 / 0.33
Among the following rules, the one which is
applied in the given reaction is
$$\mathrm{CH}_3 \mathrm{CHBrCH}_2 \mathrm{CH}_3 \xrightarrow{\text { Alc. } \mathrm{KOH}}$$
I. $$\mathrm{CH}_3 \mathrm{CH}=\mathrm{CHCH}_3$$ (major product)
II. $$\mathrm{CH}_2=\mathrm{CHCH}_2 \mathrm{CH}_3$$ (minor product)
Q51.
mcq single
+1 / 0.33
Hydrolysis of sucrose with dilute aqueous
sulphuric acid yields
Q52.
mcq single
+1 / 0.33
Which of the following is an example of network solid?
Q53.
mcq single
+1 / 0.33
Assertion (A) Graphite is an example of tetragonal crystal system.
Reason (R) For a tetragonal system, $$a=b \neq c$$ and $$\alpha=\beta=90^{\circ}, \gamma=120^{\circ}$$.
Q54.
mcq single
+1 / 0.33
Niobium crystallises in body centred cubic structure. If density is $$8.55 \mathrm{~g} \mathrm{~cm}^{-3}$$, then the atomic radius of niobium is (atomic mass of niobium $$=93 u$$ )
Q55.
mcq single
+1 / 0.33
A solid has a structure in which W atoms are
located at the corners of a cubic lattice.
O atoms at the centre of edges and Na atom
at centre of the cube. The formula for the
compound is
Q56.
mcq single
+1 / 0.33
Assertion (A) For a Daniell cell $$\mathrm{Zn} / \mathrm{Zn}^{2+} \| \mathrm{Cu}^{2+} \mid \mathrm{Cu}$$ with $$E_{\text {cell }}=1.1 \mathrm{~V}$$, the application of opposite potential greater than $$1.1 \mathrm{~V}$$ results into the flow of electrons from cathode to anode.
Reason (R) Zn is deposited at zinc electrode and $$\mathrm{Cu}$$ is dissolved at copper electrode.
Q57.
mcq single
+1 / 0.33
At $$25^{\circ} \mathrm{C}$$, the molar conductance at infinite dilution for the strong electrolytes $$\mathrm{NaOH}, \mathrm{NaCl}$$ and $$\mathrm{BaCl}_2$$ are $$248 \times 10^{-4}, 126 \times 10^{-4}$$ and $$280 \times 10^{-4} \mathrm{~Sm}^2 \mathrm{~mol}^{-1}$$ respectively. $$\lambda_{\mathrm{m}}^{\circ} \mathrm{Ba}(\mathrm{OH})_2$$ in $$\mathrm{Sm}^2 \mathrm{~mol}^{-1}$$ is
Q58.
mcq single
+1 / 0.33
For a $$\mathrm{Ag}-\mathrm{Zn}$$ button cell, net reaction is
$$\begin{gathered}
\mathrm{Zn}(s)+\mathrm{Ag}_2 \mathrm{O}(s) \longrightarrow \mathrm{ZnO}(s)+2 \mathrm{Ag}(s) \\
\Delta G_f^{\circ}\left(\mathrm{Ag}_2 \mathrm{O}\right)=-11.21 \mathrm{~kJ} \mathrm{~mol}^{-1} \\
\Delta G_f^{\circ}(\mathrm{ZnO})=-318.3 \mathrm{~kJ} \mathrm{~mol}^{-1}
\end{gathered}$$
Then, $$E^{\circ}$$ cell of the button cell is
Q59.
mcq single
+1 / 0.33
**Assertion (A) :** Nitration of benzene with
nitric acid requires the use of concentrated
sulphuric acid.
**Reason (R) :** The mixture of concentrated
sulphuric acid and concentrated nitric acid
produces the electrophile, NO$$_2^+$$.
Q60.
mcq single
+1 / 0.33
The work function of a metal is $$4.2 \mathrm{~eV}$$. If radiation of 2000 $$\mathop A\limits^o$$ fall on the metal then the kinetic energy of the fastest photoelectron is
Q61.
mcq single
+1 / 0.33
Assertion (A) Separation of Zr and Hf is
difficult.
Reason (R) Zr and Hf lie in the same group
of the periodic table.
Q62.
mcq single
+1 / 0.33
The correct order of spin only magnetic moment (in BM) for $$\mathrm{Mn}^{2+}, \mathrm{Cr}^{2+}$$ and $$\mathrm{Ti}^{2+}$$ ions is
Q63.
mcq single
+1 / 0.33
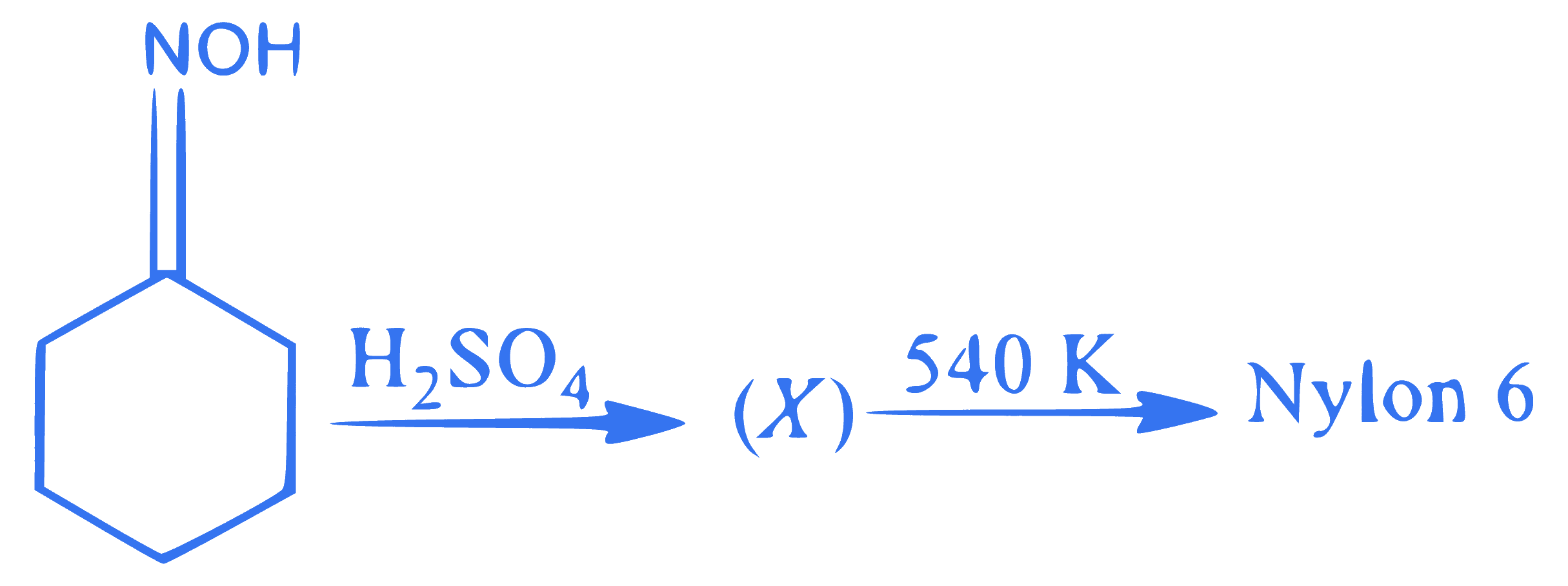 In the above reaction sequence, $$X$$ is
In the above reaction sequence, $$X$$ is
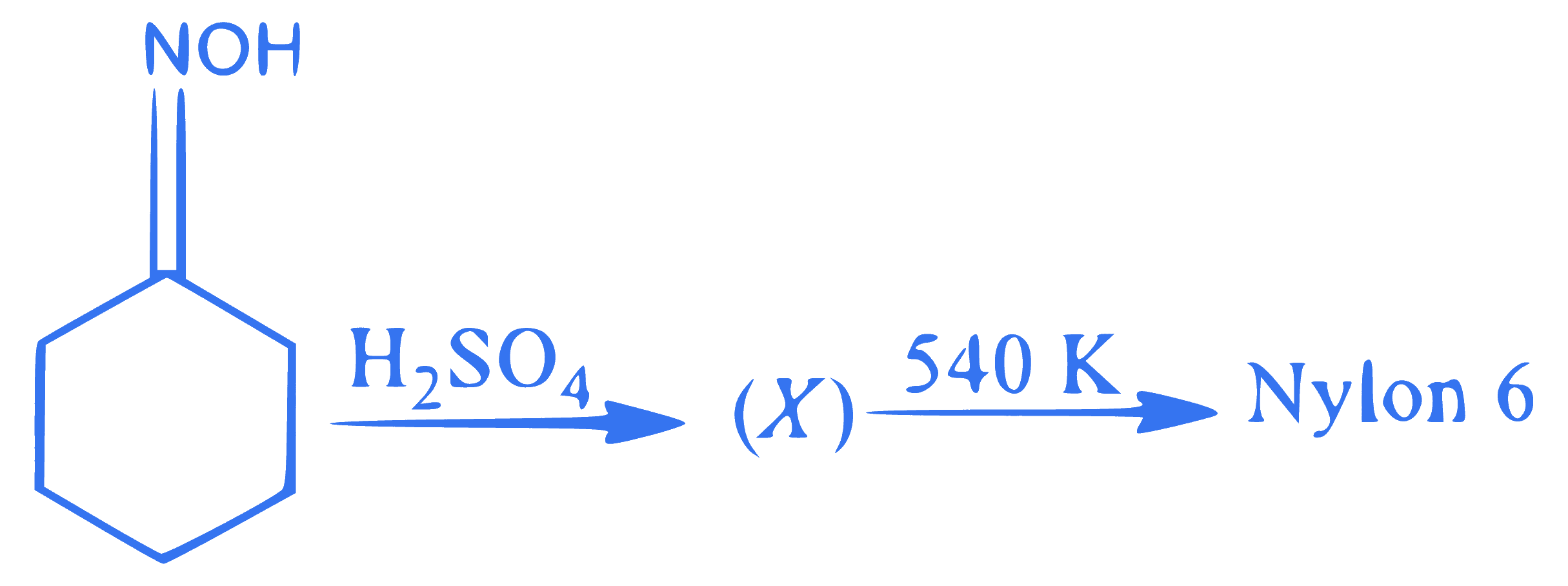 In the above reaction sequence, $$X$$ is
In the above reaction sequence, $$X$$ is
Q64.
mcq single
+1 / 0.33
Assertion (A) Most of the synthetic
polymers are not biodegradable.
Reason (R) Polymerisation process induces
toxic polymerisation.
Q65.
mcq single
+1 / 0.33
Assertion (A) The molecular mass of the
polymers cannot be calculated using the
boiling point or freezing point method.
Reason (R) The freezing point method is
used for determining the molecular mass of
small molecules.
Q66.
mcq single
+1 / 0.33
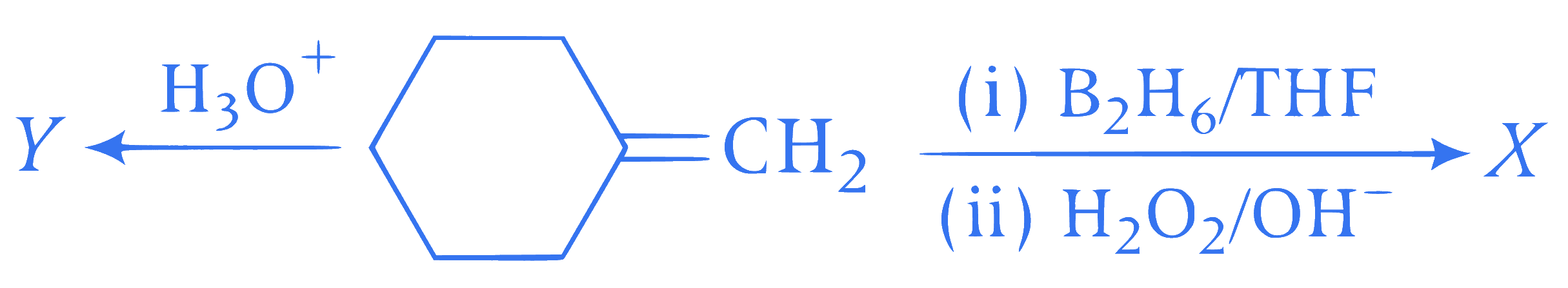 X and Y respectively are
X and Y respectively are
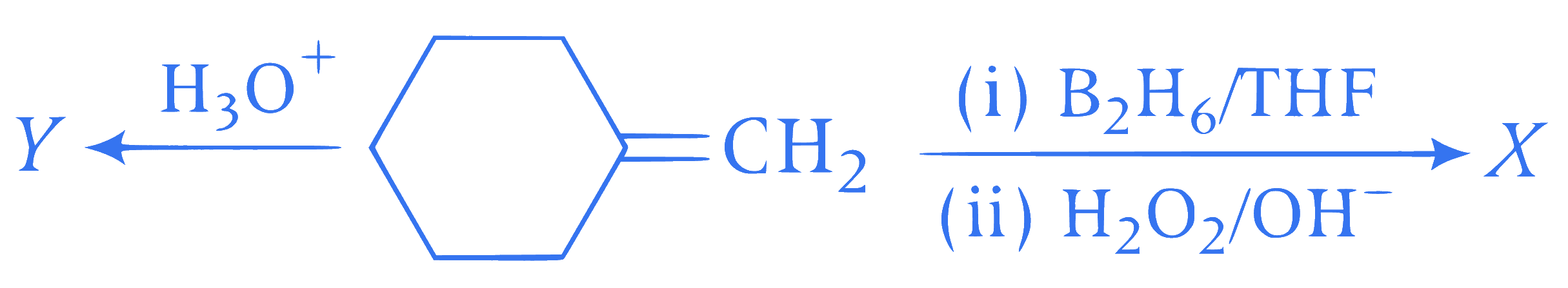 X and Y respectively are
X and Y respectively are
Q67.
mcq single
+1 / 0.33
Assertion (A) Among the two $$\mathrm{O}-\mathrm{H}$$ bonds in $$\mathrm{H}_2 \mathrm{O}$$ molecule, the energy required to break the first $$\mathrm{O}-\mathrm{H}$$ bond and the other $$\mathrm{O}-\mathrm{H}$$ bond is same.
Reason (R) This is because the electronic environment around oxygen is the same even after breakage of one $$\mathrm{O}-\mathrm{H}$$ bond.
Q68.
mcq single
+1 / 0.33
The structure of $$\mathrm{IF}_7,$$ is
Q69.
mcq single
+1 / 0.33
The solubility product of sparingly soluble salt $$A X_2$$ is $$3.2 \times 10^{-11}$$. Its solubility (in mol/L) is
Q70.
mcq single
+1 / 0.33
Assertion (A) The reaction
$$\begin{aligned}
2 \mathrm{NO}+\mathrm{O}_2 & \longrightarrow 2 \mathrm{NO}_2 \\
\text { and } \quad 2 \mathrm{CO}+\mathrm{O}_2 & \longrightarrow 2 \mathrm{CO}_2
\end{aligned}$$
proceeds at the same rate because they are similar.
Reason (R) Both the reactions have same activation energy.
Q71.
mcq single
+1 / 0.33
The rate constant for the first order
decomposition of a certain reaction is
described by the equation $$\ln k\left(\mathrm{~s}^{-1}\right)=14.34-\frac{1.25 \times 10^4 \mathrm{~K}}{T}$$. The energy of activation for this reaction is
Q72.
mcq single
+1 / 0.33
Among the following statements, the correct statement about the half-life period for a first order reaction is
Q73.
mcq single
+1 / 0.33
A gas ‘X’ is used in filling balloons for
meteorological observations. It is also used
in gas-cooled nuclear reactors. Here, the gas
X is
Q74.
mcq single
+1 / 0.33
When an excess and a very dilute aqueous
solution of KI is added to very dilute
aqueous solution of silver nitrate. The
colloidal particles of silver iodide which are
associated with the Helmholtz double layer
in the form of
Q75.
mcq single
+1 / 0.33
Assertion (A) Micelles are formed by
surfactant molecules above the critical
micellar concentration (CMC).
Reason (R) The conductivity of a solution
having surfactant molecules decreases
sharply at the CMC.
Q76.
mcq single
+1 / 0.33
Assertion (A) The boiling points of alkyl halides decrease in the order $$R \mathrm{I}>R \mathrm{Br}>\mathrm{RCl}>\mathrm{RF}$$.
Reason (R) The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
Q77.
mcq single
+1 / 0.33
A bromoalkane '$$X$$' reacts with magnesium in dry ether to form compound '$$Y$$'. The reaction of '$$Y$$' with methanol followed by hydrolysis yield an alcohol having molecular formula, $$\mathrm{C}_4 \mathrm{H}_{10} \mathrm{O}$$. The compound '$$X$$' is
Q78.
mcq single
+1 / 0.33
Which of the following represents
Wurtz-Fittig reaction?
Q79.
mcq single
+1 / 0.33
Benzoic acid undergoes dimerisation in benzene solution. The van't Hoff factor $$(i)$$ is related to the degree of association '$$x$$' of the acid as
Q80.
mcq single
+1 / 0.33
Assertion (A) $$K_p$$ can be equal to or less than or even greater than the value of $$K_C$$.
Reason (R) $$K_p=K_C(R T)^{\Delta n}$$
Relation between $$K_p$$ and $$K_C$$ depends on the change in the number of moles of gaseous reactants and products.
Q81.
mcq single
+1 / 0.33
At equilibrium, the concentration of
$$\begin{gathered}
\mathrm{N}_2=3.0 \times 10^{-3} \mathrm{M} \\
\mathrm{O}_2=4.2 \times 10^{-3} \mathrm{M} \\
\text{and} \quad \mathrm{NO}=2.8 \times 10^{-3} \mathrm{M}
\end{gathered}$$
in a sealed vessel at $$800 \mathrm{~K}$$ and $$1 \mathrm{~atm}$$ pressure. What will be $$K_p$$ for the given reaction?
$$\mathrm{N}_2(g)+\mathrm{O}_2(g) \rightleftharpoons 2 \mathrm{NO}(g)$$
Q82.
mcq single
+1 / 0.33
In which of the following arrangements, the
order is not strictly according to the property
written against it?
Q83.
mcq single
+1 / 0.33
Assertion (A) Electron gain enthalpy
becomes less negative as we go down a group.
Reason (R) Size of the atom increases on
going down the group and the added electron
would be farther away from the nucleus.
Q84.
mcq single
+1 / 0.33
The ionic radii $$(\mathop A\limits^o)$$ of $$\mathrm{C}^{4-}$$ and $$\mathrm{O}^{2-}$$ respectively are 2.60 and 1.40. The ionic radius of the isoelectronic ion $$\mathrm{N}^{3-}$$ would be
Q85.
mcq single
+1 / 0.33
$$\Delta H$$ and $$\Delta E$$ for the reaction,
$$\mathrm{Fe}_2 \mathrm{O}_3(s)+3 \mathrm{H}_2(g) \longrightarrow 2 \mathrm{Fe}(s)+\mathrm{H}_2 \mathrm{O}(l)$$
at constant temperature are related as
Q86.
mcq single
+1 / 0.33
In an adiabatic process, no transfer of heat
takes place between system and
surroundings. Choose the correct option for
free expansion of an ideal gas under
adiabatic condition from the following
Q87.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{NaCl}$$ reacts with concentrated $$\mathrm{H}_2 \mathrm{SO}_4$$ to give colourless fumes with pungent smell. But on adding $$\mathrm{MnO}_2$$ the fumes become greenish yellow.
Reason (R) $$\mathrm{MnO}_2$$ oxidises $$\mathrm{HCl}$$ to chlorine gas which is greenish yellow.
Q88.
mcq single
+1 / 0.33
A green yellow gas reacts with an alkali
metal hydroxide to form a halate which can
be used in fireworks and safety matches. The
gas and halate are, respectively
Q89.
mcq single
+1 / 0.33
Which of the following oxyacid does not contain $$\mathrm{P}-\mathrm{O}-\mathrm{P}$$ bond?
Q90.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{N}_2$$ is less reactive than $$\mathrm{P}_4$$.
Reason (R) Nitrogen has more electron gain enthalpy than phosphorus.
Q91.
mcq single
+1 / 0.33
Affinity for hydrogen decreases in the group
from fluorine to iodine. Which of the
halogen acids should have highest bond
dissociation enthalpy?
Q92.
mcq single
+1 / 0.33
Which of the following products will be obtained when copper metal is reacted with $$\mathrm{HNO}_3$$ ?
Q93.
mcq single
+1 / 0.33
$$\mathop {8N{H_3}}\limits_{(Excess)} + 3C{l_2}\buildrel {} \over
\longrightarrow X$$
$$N{H_3} + \mathop {3C{l_2}}\limits_{(Excess)} \buildrel {} \over
\longrightarrow Y$$
What is X and Y in the above reaction?
Q94.
mcq single
+1 / 0.33
The electronic configuration of central metal atom/ion in $$\left[\mathrm{Co}(\mathrm{CN})_6\right]^{3-}$$ is
Q95.
mcq single
+1 / 0.33
Which of the following compound has same
oxidation state of the central metal atom in
the cationic and anionic part?
Q96.
mcq single
+1 / 0.33
The spin only magnetic moment of $$[\mathrm{MnBr}_4]^{2-}$$ is $$5.9 \mathrm{~BM}$$. The geometry of this complex ion is
Q97.
mcq single
+1 / 0.33
The IUPAC name of the complex $$[\mathrm{Pt}\left(\mathrm{NH}_3\right)_3 \mathrm{Br}\left(\mathrm{NO}_2\right) \mathrm{Cl}] \mathrm{Cl}$$ is
Q98.
mcq single
+1 / 0.33
Among the following complex ions, the one
which shows geometrical isomerism will be
Q99.
mcq single
+1 / 0.33
Assertion (A) Toxic metal ions are removed
by the chelating ligands.
Reason (R) Chelate complexes tend to be
more stable.
Q100.
mcq single
+1 / 0.33
**Assertion (A) :** The pH of acid rain is less
than 5.6.
**Reason (R) :** Carbon dioxide present in the
atmosphere dissolves in rain water and
becomes carbonic acid.
Q101.
mcq single
+1 / 0.33
The correct order of decreasing stability of
the following carbocation is


Q102.
mcq single
+1 / 0.33
The correct IUPAC name of the given compound is


Q103.
mcq single
+1 / 0.33
Food preservatives prevent spoilage of food due to microbial growth. The most commonly used preservatives are
Q104.
mcq single
+1 / 0.33
The relative reactivities of acyl compounds
towards nucleophilic substitution are in the
order of
Q105.
mcq single
+1 / 0.33
Which of the following compounds do not
undergo aldol condensation?
Q106.
mcq single
+1 / 0.33
Assertion (A) Beryllium carbonate is kept
in the atmosphere of carbon dioxide.
Reason (R) Beryllium carbonate is unstable
and decomposes to give beryllium oxide and
carbon dioxide.
Q107.
mcq single
+1 / 0.33
Assertion (A) IUPAC name of the compound
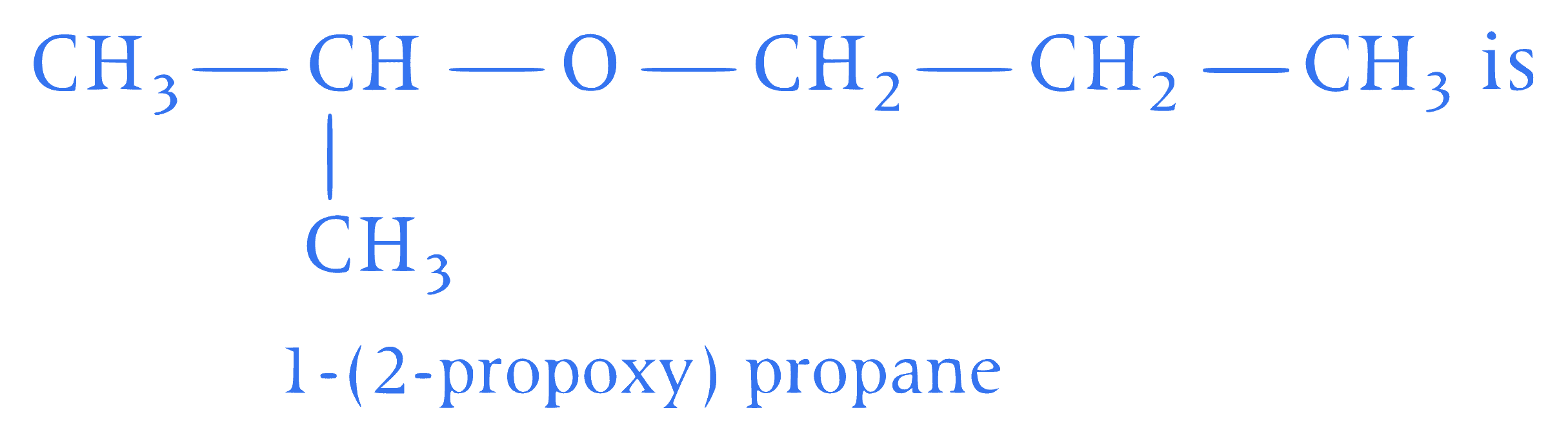 Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
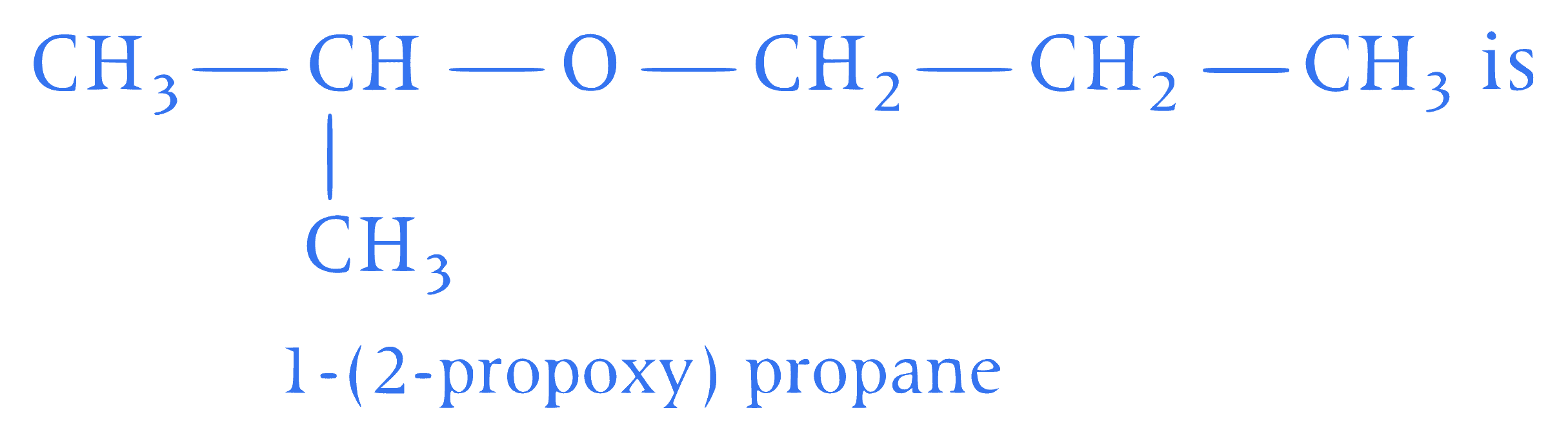 Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
Reason (R) In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by-OR or- $$\mathrm{O} A r$$ group [where, $$R=$$ alkyl group and $$A r=\operatorname{aryl}$$ group].
Q108.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{CH}_3 \mathrm{OH}$$ and $$\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{OH}$$ can be distinguished by haloform test.
Reason (R) Haloform test is given by $$2^{\circ}$$ alcohol.
Q109.
mcq single
+1 / 0.33
Assertion (A) $$\mathrm{CH}_4$$ does not react with $$\mathrm{Cl}_2$$ in dark.
Reason (R) Chlorination of $$\mathrm{CH}_4$$ takes place in sunlight.
Q110.
mcq single
+1 / 0.33
Among the following rules, the one which is
applied in the given reaction is
$$\mathrm{CH}_3 \mathrm{CHBrCH}_2 \mathrm{CH}_3 \xrightarrow{\text { Alc. } \mathrm{KOH}}$$
I. $$\mathrm{CH}_3 \mathrm{CH}=\mathrm{CHCH}_3$$ (major product)
II. $$\mathrm{CH}_2=\mathrm{CHCH}_2 \mathrm{CH}_3$$ (minor product)
Q111.
mcq single
+1 / 0.33
Hydrolysis of sucrose with dilute aqueous
sulphuric acid yields
Q112.
mcq single
+1 / 0.33
Which of the following is an example of network solid?
Q113.
mcq single
+1 / 0.33
Assertion (A) Graphite is an example of tetragonal crystal system.
Reason (R) For a tetragonal system, $$a=b \neq c$$ and $$\alpha=\beta=90^{\circ}, \gamma=120^{\circ}$$.
Q114.
mcq single
+1 / 0.33
Niobium crystallises in body centred cubic structure. If density is $$8.55 \mathrm{~g} \mathrm{~cm}^{-3}$$, then the atomic radius of niobium is (atomic mass of niobium $$=93 u$$ )
Q115.
mcq single
+1 / 0.33
A solid has a structure in which W atoms are
located at the corners of a cubic lattice.
O atoms at the centre of edges and Na atom
at centre of the cube. The formula for the
compound is
Q116.
mcq single
+1 / 0.33
Assertion (A) For a Daniell cell $$\mathrm{Zn} / \mathrm{Zn}^{2+} \| \mathrm{Cu}^{2+} \mid \mathrm{Cu}$$ with $$E_{\text {cell }}=1.1 \mathrm{~V}$$, the application of opposite potential greater than $$1.1 \mathrm{~V}$$ results into the flow of electrons from cathode to anode.
Reason (R) Zn is deposited at zinc electrode and $$\mathrm{Cu}$$ is dissolved at copper electrode.
Q117.
mcq single
+1 / 0.33
At $$25^{\circ} \mathrm{C}$$, the molar conductance at infinite dilution for the strong electrolytes $$\mathrm{NaOH}, \mathrm{NaCl}$$ and $$\mathrm{BaCl}_2$$ are $$248 \times 10^{-4}, 126 \times 10^{-4}$$ and $$280 \times 10^{-4} \mathrm{~Sm}^2 \mathrm{~mol}^{-1}$$ respectively. $$\lambda_{\mathrm{m}}^{\circ} \mathrm{Ba}(\mathrm{OH})_2$$ in $$\mathrm{Sm}^2 \mathrm{~mol}^{-1}$$ is
Q118.
mcq single
+1 / 0.33
For a $$\mathrm{Ag}-\mathrm{Zn}$$ button cell, net reaction is
$$\begin{gathered}
\mathrm{Zn}(s)+\mathrm{Ag}_2 \mathrm{O}(s) \longrightarrow \mathrm{ZnO}(s)+2 \mathrm{Ag}(s) \\
\Delta G_f^{\circ}\left(\mathrm{Ag}_2 \mathrm{O}\right)=-11.21 \mathrm{~kJ} \mathrm{~mol}^{-1} \\
\Delta G_f^{\circ}(\mathrm{ZnO})=-318.3 \mathrm{~kJ} \mathrm{~mol}^{-1}
\end{gathered}$$
Then, $$E^{\circ}$$ cell of the button cell is
Q119.
mcq single
+1 / 0.33
**Assertion (A) :** Nitration of benzene with
nitric acid requires the use of concentrated
sulphuric acid.
**Reason (R) :** The mixture of concentrated
sulphuric acid and concentrated nitric acid
produces the electrophile, NO$$_2^+$$.
Q120.
mcq single
+1 / 0.33
The work function of a metal is $$4.2 \mathrm{~eV}$$. If radiation of 2000 $$\mathop A\limits^o$$ fall on the metal then the kinetic energy of the fastest photoelectron is
Physics
Physics
Q1.
mcq single
+1 / 0.33
Red light of wavelength 5400 $$\mathop A\limits^o $$ from a
distant source falls on a slit 0.80 mm wide.
Calculate the distance between first two dark
bands on each side of central bright band in
the diffraction pattern observed on a screen
place 1.4 m from the slit.
Q2.
mcq single
+1 / 0.33
**Assertion :** If a glass slab is placed in front of
one of the slits, then fringe with will
decrease.
**Reason :** Glass slab will produce an
additional path difference.
Q3.
mcq single
+1 / 0.33
An unpolarised beam of intensity $$2 a^2$$ passes through a thin polaroid. Assuming zero absorption in the polaroid, the intensity of emergent plane polarised light is
Q4.
mcq single
+1 / 0.33
Assertion The angular momentum of
system always remain constant.
Reason For a system, $$\tau_{\mathrm{ext}}=\frac{d L}{d t}=0$$
Q5.
mcq single
+1 / 0.33
A thin horizontal circular disc is rotating about a vertical axis passing through its centre. An insect is at rest at a point near the rim of disc. The insect now moves along a diameter of the disc to reach its other end. During the journey of the insect, the angular speed of the disc
Q6.
mcq single
+1 / 0.33
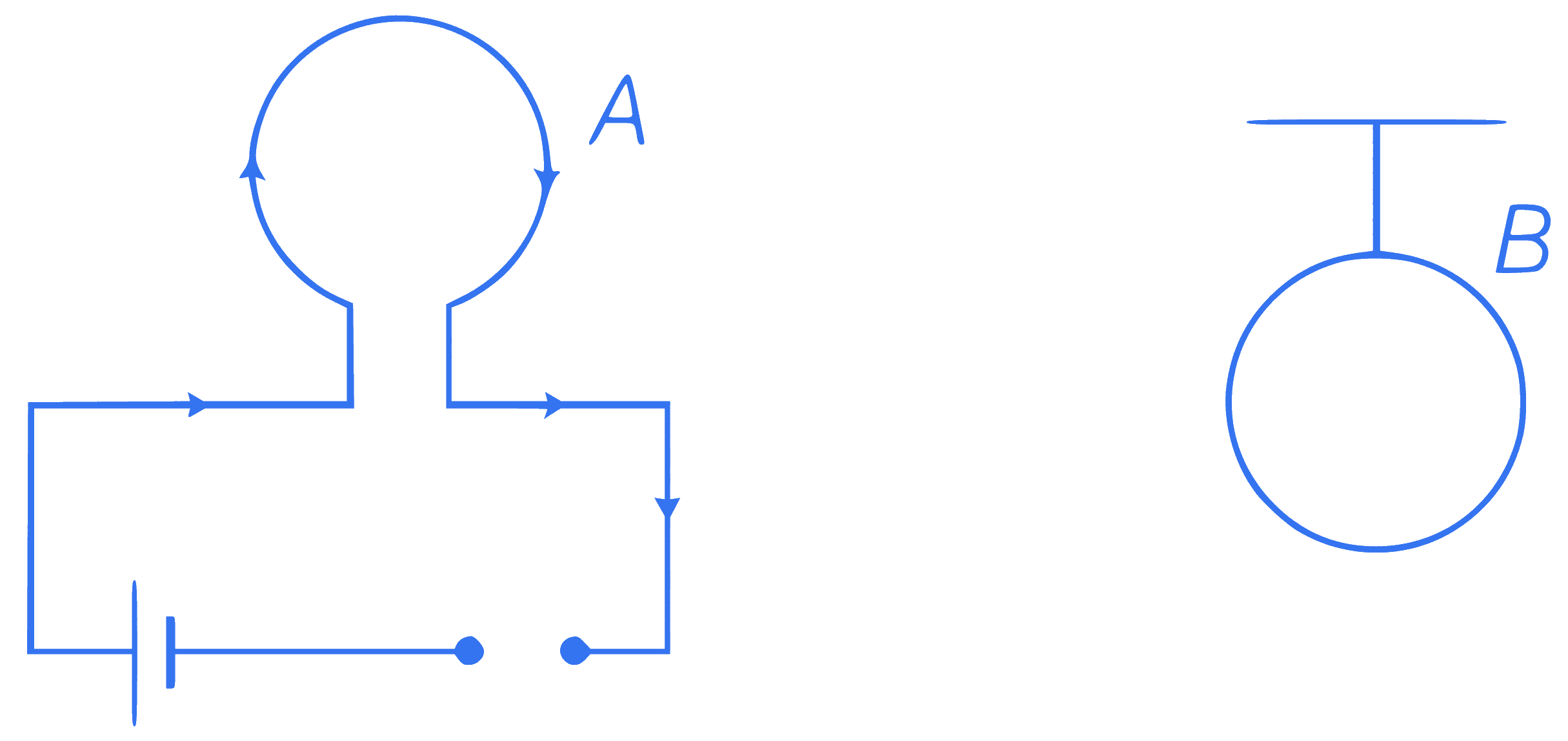
A system $$S$$ consists of two coils $$A$$ and $$B$$. The coil $$A$$ carries a steady current $$I$$. While the coil $$B$$ is suspended nearby as shown in figure. Now, if the system is heated, so as to raise the temperature of two coils steadily, then

Q7.
mcq single
+1 / 0.33
A circular loop of radius 0.3 cm lies parallel
to a much bigger circular loop of radius 20 cm.
The centre of the small loop on the axis of
the bigger loop. The distance between their
centres is 15 cm. If a current of 20 A flows
through the smaller loop, then the flux
linked with bigger drop is
Q8.
mcq single
+1 / 0.33
If the formula, $$X=3 Y Z^2, X$$ and $$Z$$ have dimensions of capacitance and magnetic induction. The dimensions of $$Y$$ in MKSQ system are
Q9.
mcq single
+1 / 0.33
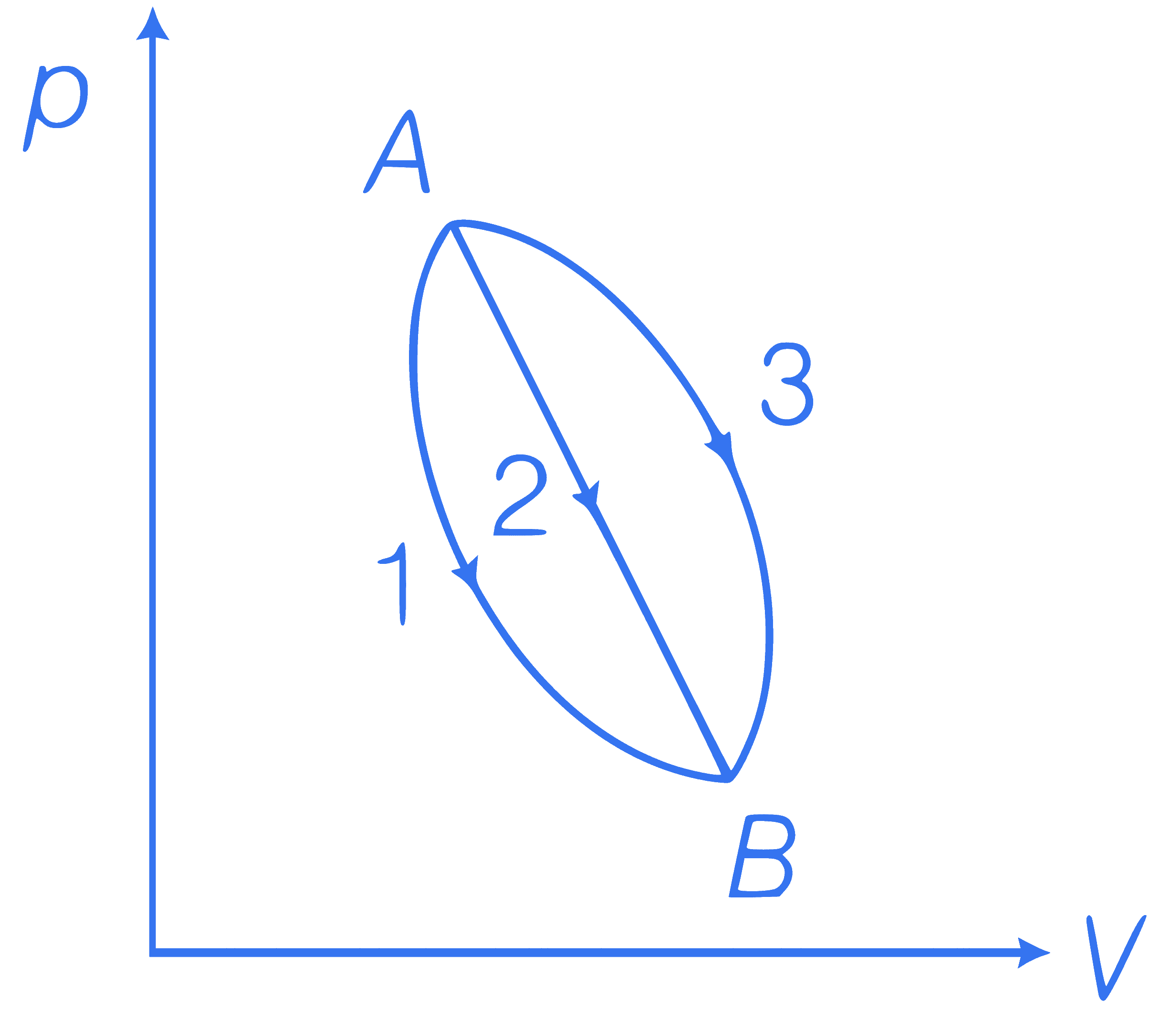 An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
 An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
Q10.
mcq single
+1 / 0.33
Assertion Internal energy of an ideal gas
does not depend on volume of gas.
Reason Internal energy depends only on
temperature of gas.
Q11.
mcq single
+1 / 0.33
A gas consisting of a rigid diatomic molecules
was initially under standard condition. Then,
gas was compressed adiabatically to
one-fifth of its initial volume. What will be
the mean kinetic energy of a rotating
molecule in the final state?
Q12.
mcq single
+1 / 0.33
Assertion In isothermal process, whole of
the heat energy supplied to the body is
converted into internal energy.
Reason According to the first law of
thermodynamics,
$$\Delta Q=\Delta U+\Delta W$$
Q13.
mcq single
+1 / 0.33
Assertion Smaller drop of water resist deformation forces better than the larger drops.
Reason Excess pressure inside drop is inversely proportional to its radius.
Q14.
mcq single
+1 / 0.33
A half ring of radius $$R$$ has a charge of $$\lambda$$ per unit length. The electric force on $$1 \mathrm{C}$$ charged placed at the centre is
Q15.
mcq single
+1 / 0.33
Assertion Mass of a body decreases slightly
when it is negatively charged.
Reason Charging is due to transfer of electrons.
Q16.
mcq single
+1 / 0.33
Positive charge $$Q$$ is distributed uniformly over a circular ring of radius $$R$$. A point particle having a mass $$(m)$$ and a negative charge $$-q$$ is placed on its axis at a distance $$x$$ from the centre. Assuming $$x < R$$, find the time period of oscillation of the particle, if it is released from there [neglect gravity].
Q17.
mcq single
+1 / 0.33
The weight of a body on the surface of the
earth is 63 N. What is the gravitational force
on it due to the earth at a height equal to
half the radius of the earth?
Q18.
mcq single
+1 / 0.33
Two satellites $$A$$ and $$B$$ revolve round the same planet in coplanar circular orbits lying in the same plane. Their periods of revolutions are $$1 \mathrm{~h}$$ and $$8 \mathrm{~h}$$, respectively. The radius of the orbit of $$A$$ is $$10^4 \mathrm{~km}$$. The speed of $$B$$ is relative to $$A$$. When they are closed in $$\mathrm{km} / \mathrm{h}$$ is
Q19.
mcq single
+1 / 0.33
A planet is revolving around the sun in a circular orbit with a radius $$r$$. The time period is $$T$$. If the force between the planet and star is proportional to $$r^{-3 / 2}$$, then the square of time period is proportional to
Q20.
mcq single
+1 / 0.33
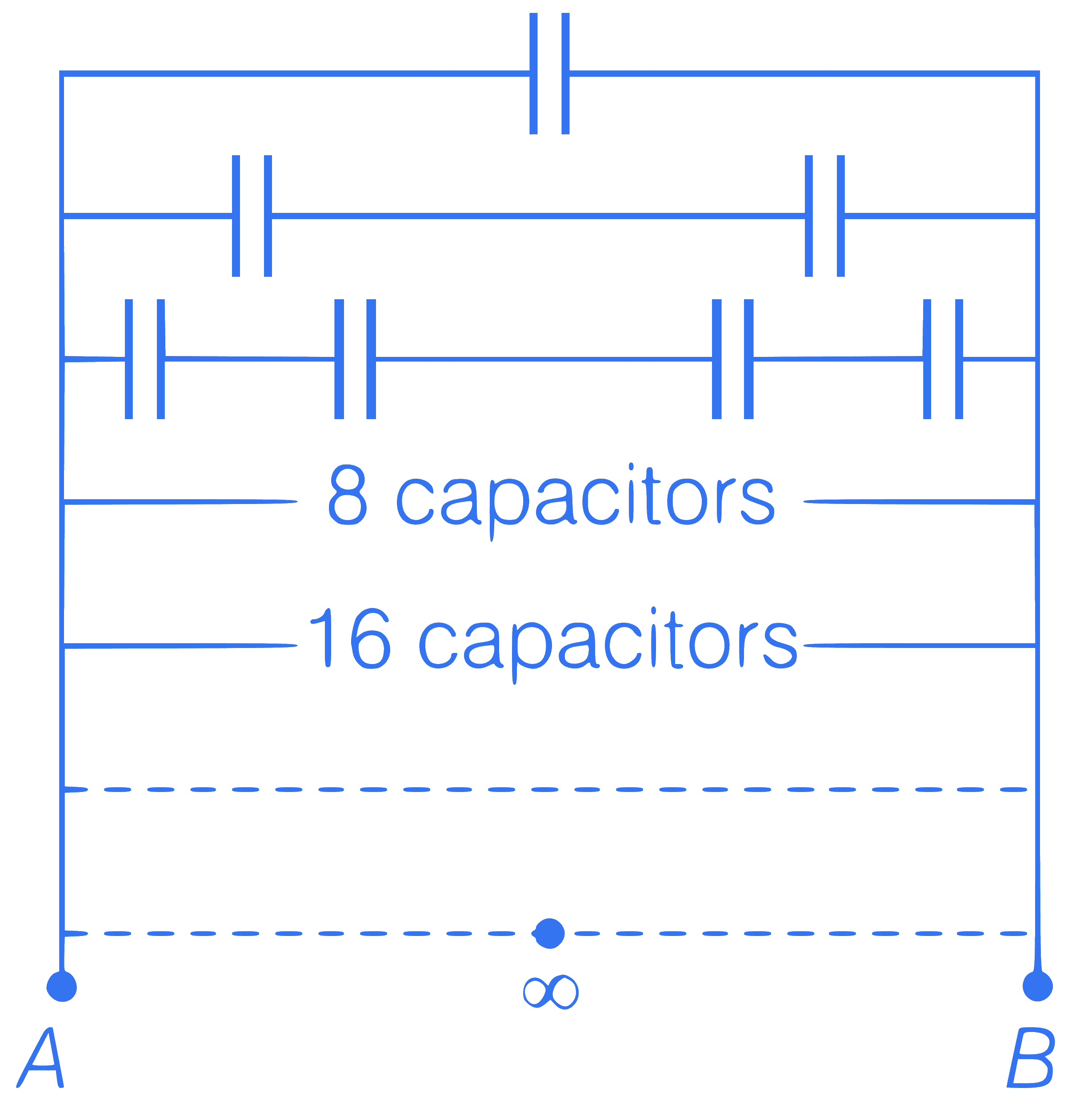
An infinite number of identical capacitors
each of capacitance 1 $$\mu$$F are connected as
shown in the figure. Then, the equivalent
capacitance between A and B is

Q21.
mcq single
+1 / 0.33
Assertion A dielectric slab is inserted
between plates of an isolated charged
capacitor which remain same.
Reason Charge on an isolated system is
conserved.
Q22.
mcq single
+1 / 0.33
Assertion A convex mirror always make a
virtual image.
Reason The ray always diverge after
reflection from the convex mirror.
Q23.
mcq single
+1 / 0.33
Assertion A pendulum is falling freely its
time period becomes zero.
Reason Freely falling body has the
acceleration equal to g.
Q24.
mcq single
+1 / 0.33
A block of rectangular size of mass $$m$$ and area of cross-section $$A$$, floats in a liquid of density $$\rho$$. If we give a small vertical displacement from equilibrium, it undergoes SHM with time period $$T$$, then
Q25.
mcq single
+1 / 0.33
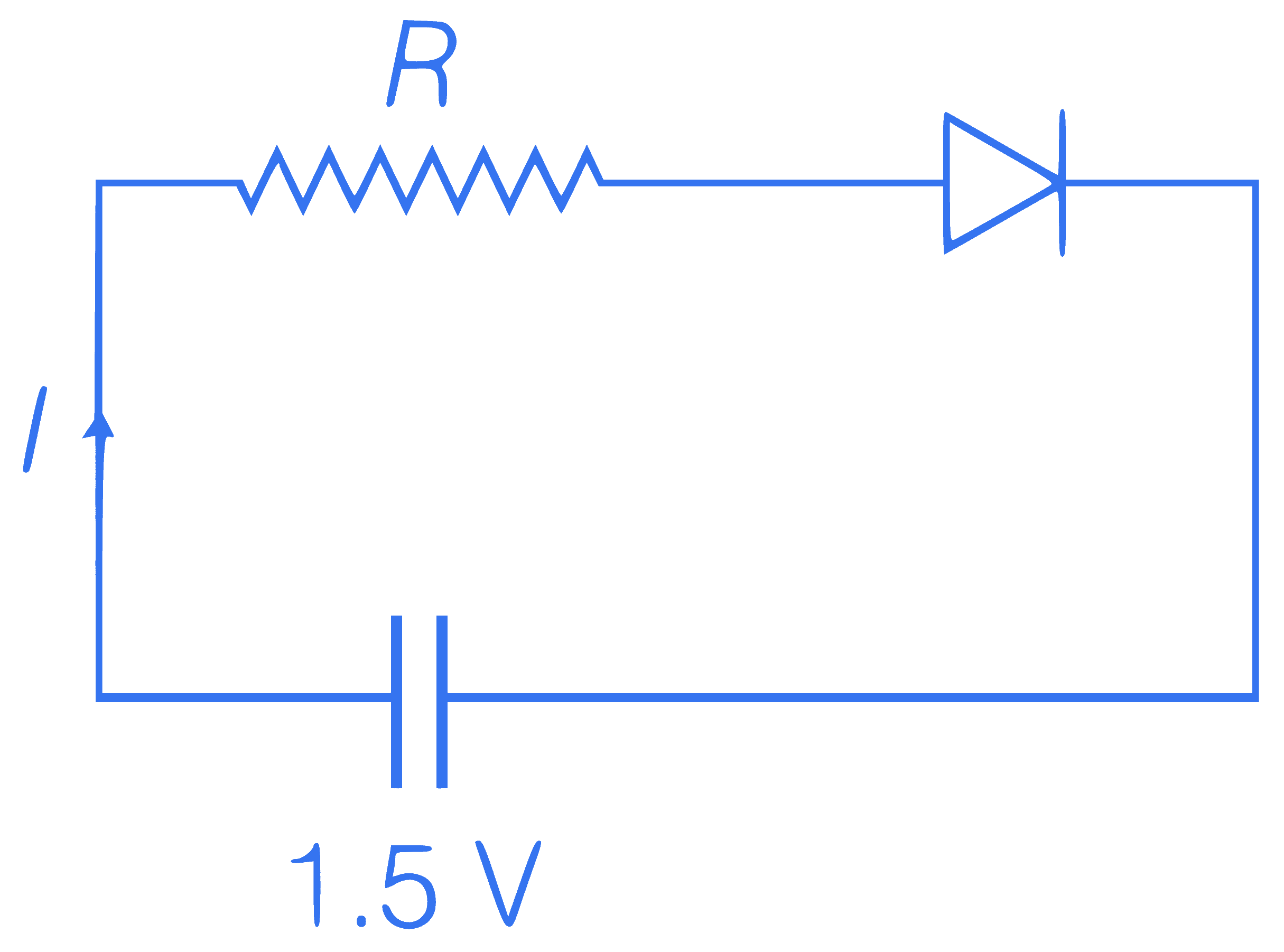 The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
 The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
Q26.
mcq single
+1 / 0.33
Assertion Thickness of depletion layer is
fixed in all semiconductor devices.
Reason No free charge carriers are available
in depletion layer.
Q27.
mcq single
+1 / 0.33
An element $$d l=d x \hat{\mathbf{i}}$$ (where, $$d x=1 \mathrm{~cm}$$ ) is placed at the origin and carries a large current $$i=10 \mathrm{~A}$$. What is the magnetic field on the $$Y$$-axis at a distance of $$0.5 \mathrm{~m}$$ ?
Q28.
mcq single
+1 / 0.33
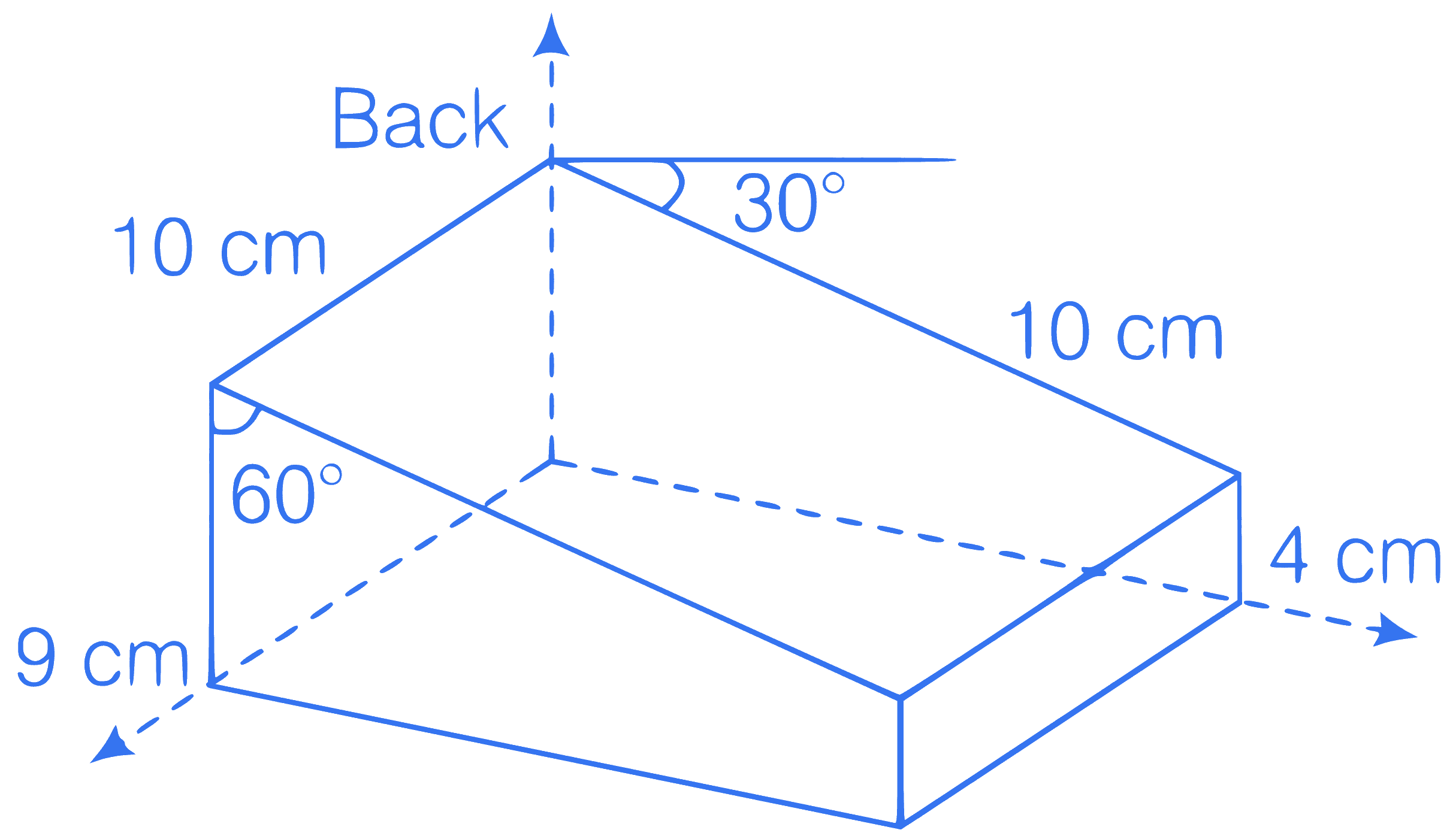
Consider the following figure, a uniform
magnetic field of 0.2 T is directed along the
positive X-axis. The magnetic flux through
top surface of the figure.

Q29.
mcq single
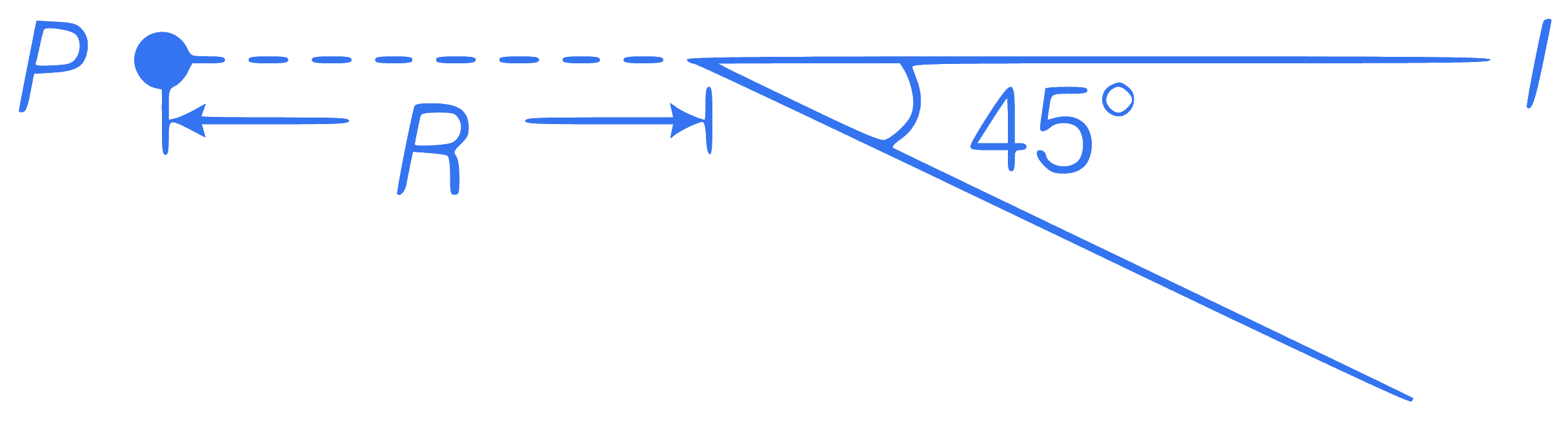
+1 / 0.33
A long straight wire, carrying current $$I$$ is bent at its mid-point to form an angle of $$45^{\circ}$$. Induction of magnetic field (in tesla) at point $$P$$, distant $$R$$ from point of bending is equal to

Q30.
mcq single
+1 / 0.33
Assertion A magnetic field interacts with a
moving charge and not with a stationary
charge.
Reason A moving charge produce a
magnetic field.
Q31.
mcq single
+1 / 0.33
A diode detector is used to detect and
amplitude modulated wave of 60%
modulation by using a condenser of capacity
250 pF in parallel with a load resistance
100 k$$\Omega$$. Find the maximum modulated
frequency which could be detected by it.
Q32.
mcq single
+1 / 0.33
The graph between the energy log E of an
electron and its de-Broglie wavelength log $$\lambda$$ will be
Q33.
mcq single
+1 / 0.33
Assertion A body is momentarily at rest at
the instant, if it reverse the direction.
Reason A body cannot have acceleration, if
its velocity is zero at a given instant of time.
Q34.
mcq single
+1 / 0.33
Assertion If electrons in an atom were
stationary, then they would fall into the
nucleus.
Reason Electrostatic force of attraction acts
between negatively charged electrons and
positive nucleus.
Q35.
mcq single
+1 / 0.33
The half-life of a radioactive substance is $$20 \mathrm{~min}$$. The approximate time interval $$\left(t_2-t_1\right)$$ between the time $$t_2$$, when $$\frac{2}{3}$$ of it has decayed and time $$t_1$$ when $$\frac{1}{3}$$ of it had decayed is
Q36.
mcq single
+1 / 0.33
Assertion Radioactive nuclei emits $$\beta^{-}$$-particles.
Reason Electrons exist inside the nucleus.
Q37.
mcq single
+1 / 0.33
Assertion The maximum height of projectile
is always 25% of the maximum range.
Reason For maximum range, projectile
should be projected at 90°.
Q38.
mcq single
+1 / 0.33
Assertion Two particles moving in the same
direction do not lose all their energy in
completely inelastic collision.
Reason Principle of conservation of
momentum holds true for all kinds of
collisions.
Q39.
mcq single
+1 / 0.33
Body of mass M is much heavier than the
other body of mass m. The heavier body with
speed v collides with the lighter body which
was at rest initially elastically. The speed of
lighter body after collision is
Q40.
mcq single
+1 / 0.33
Three bodies having masses $$5 \mathrm{~kg}, 4 \mathrm{~kg}$$ and $$2 \mathrm{~kg}$$ is moving at the speed of $$5 \mathrm{~m} / \mathrm{s}, 4 \mathrm{~m} / \mathrm{s}$$ and $$2 \mathrm{~m} / \mathrm{s}$$ respectively along $$X$$-axis. The magnitude of velocity of centre of mass is
Q41.
mcq single
+1 / 0.33
A pipe of length 85 cm is closed from one
end. Find the number of possible natural
oscillations of air column in the pipe whose
frequencies lie below 1250 Hz. The velocity
of sound in air is 340 m/s.
Q42.
mcq single
+1 / 0.33
A steel rod 100 cm long is damped at into
middle. The fundamental frequency of
longitudinal vibrations of the rod are given
to be 2.53 kHz. What is the speed of sound
in sound is steel?
Q43.
mcq single
+1 / 0.33
Assertion To hear different beats difference
of the frequencies of two sources should be
less than 10.
Reason More the number of beats more in
the confusion.
Q44.
mcq single
+1 / 0.33
Assertion Angle of repose is equal to angle
of limiting friction.
Reason When a body is just at the point of
motion, the force of friction of this stage is
called as limiting friction.
Q45.
mcq single
+1 / 0.33
A body of mass 5 kg is suspended by a spring
balance on an inclined plane as shown in
figure.
 So, force applied on spring balance is
So, force applied on spring balance is
 So, force applied on spring balance is
So, force applied on spring balance is
Q46.
mcq single
+1 / 0.33
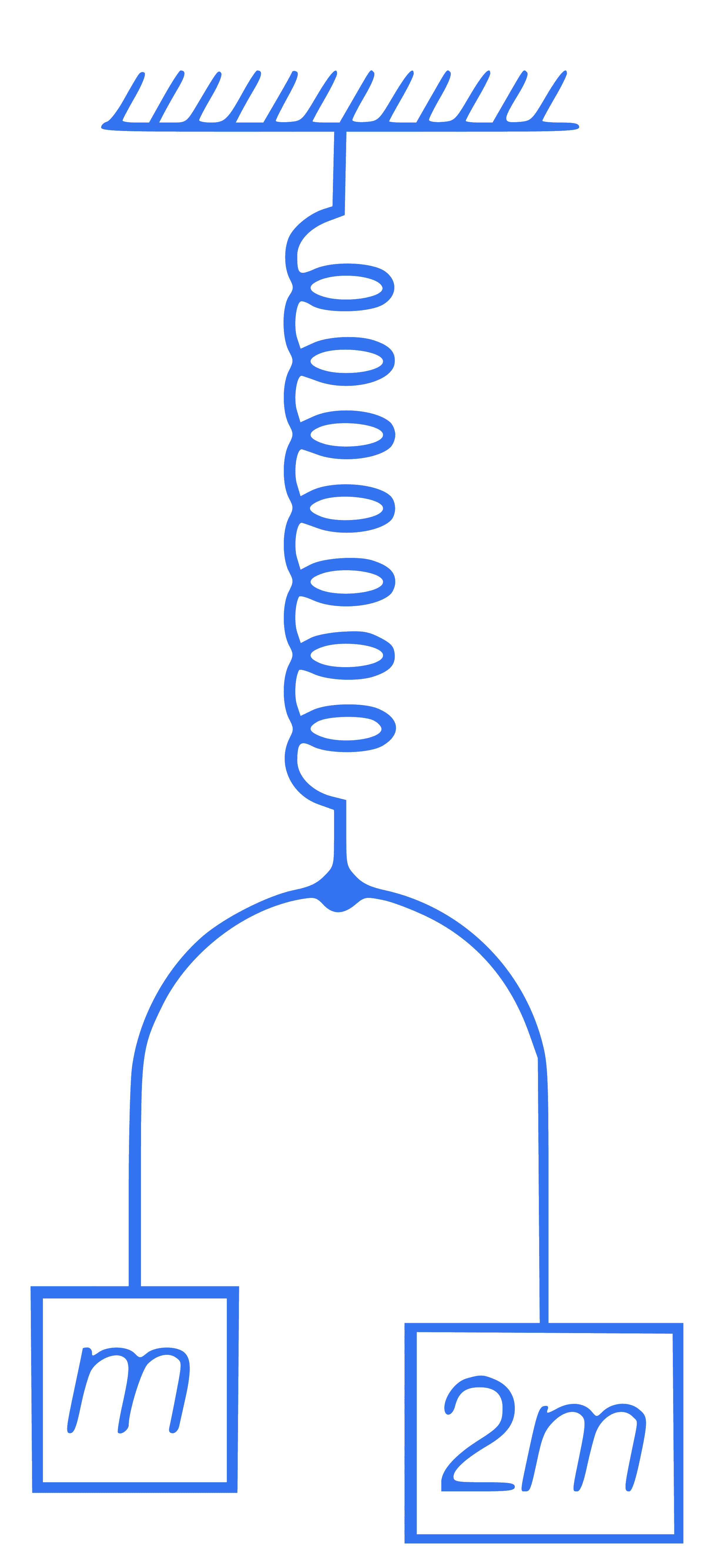 In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
 In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
Q47.
mcq single
+1 / 0.33
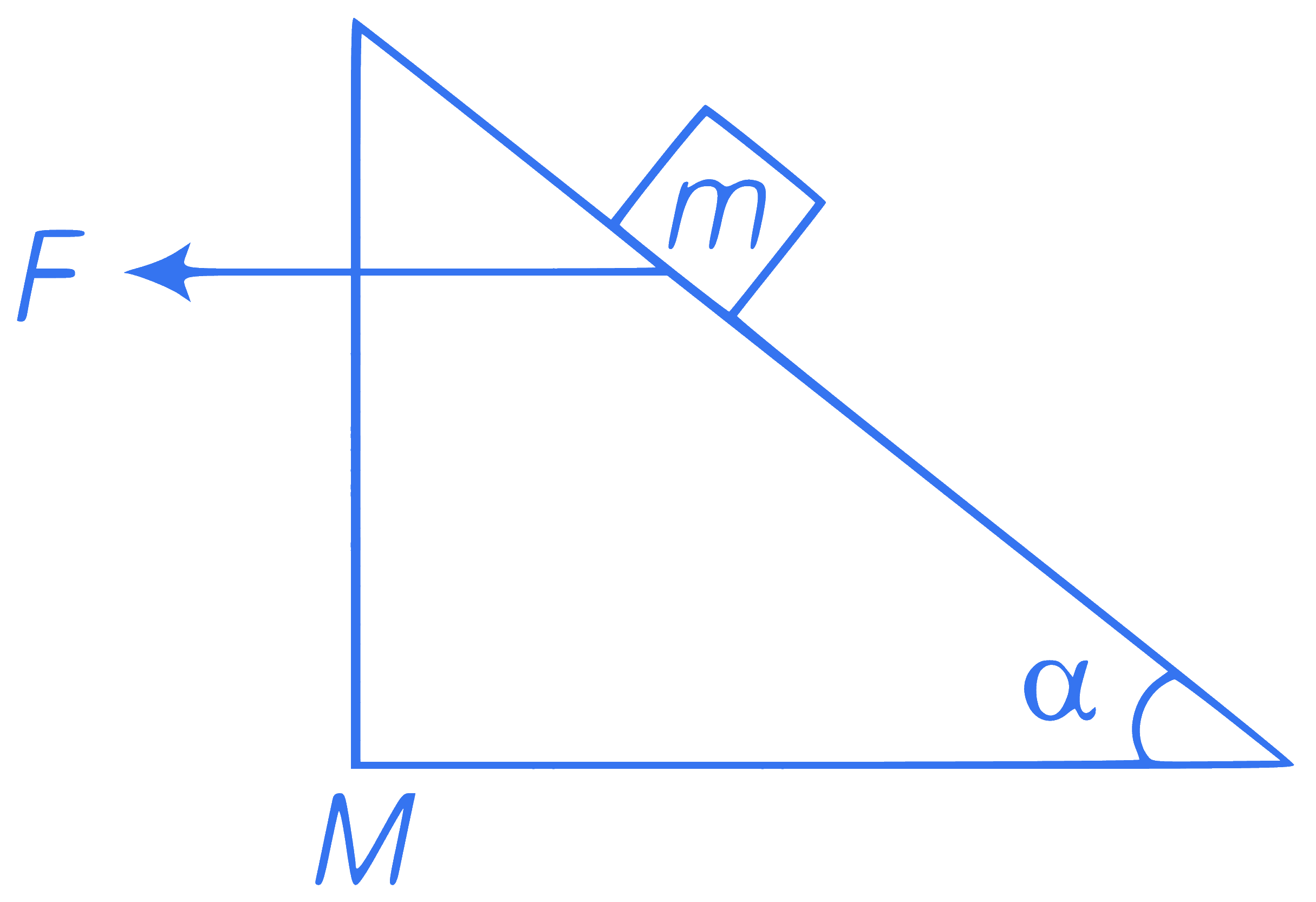
A wooden wedge of mass $$M$$ and inclination angle $$(\alpha)$$ rest on a smooth floor. A block of mass $m$ is kept on wedge. A force $$F$$ is applied on the wedge as shown in the figure such that block remains stationary with respect to wedge. So, magnitude of force $$F$$ is

Q48.
mcq single
+1 / 0.33
A piece of ice slides down a rough inclined plane at $$45^{\circ}$$ inclination in twice the time that it takes to slide down an identical but frictionless inclined plane. What is the coefficient of friction between ice and incline?
Q49.
mcq single
+1 / 0.33
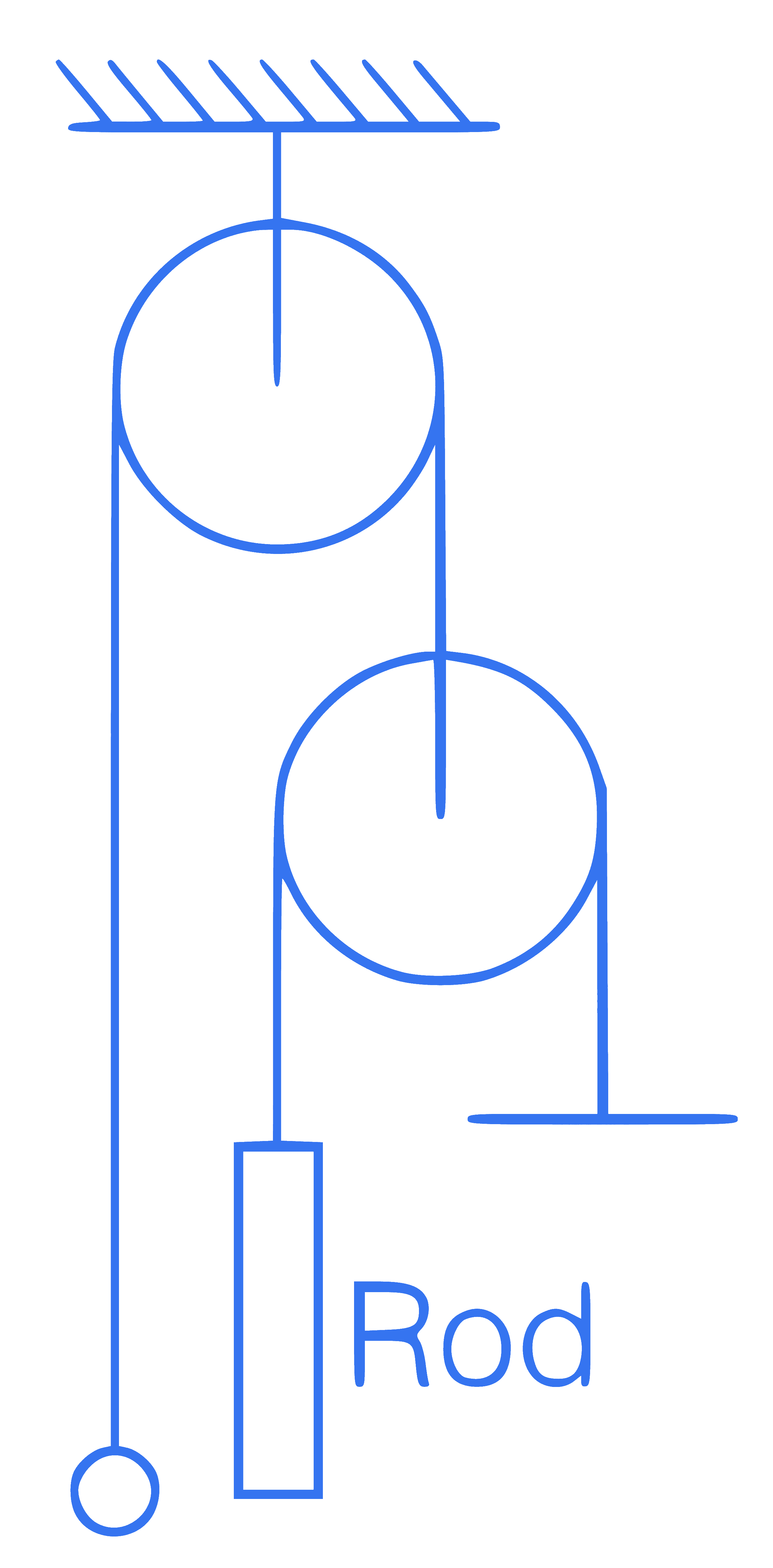 In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
 In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
Q50.
mcq single
+1 / 0.33
The horizontal component of the earth's magnetic field at any place is $$0.36 \times 10^{-4} \mathrm{~Wb} / \mathrm{m}^2$$. If the angle of dip at that place is $$60^{\circ}$$, then the value of vertical component of the earth's magnetic field will be (in $$\mathrm{Wb} / \mathrm{m}^2$$ )
Q51.
mcq single
+1 / 0.33
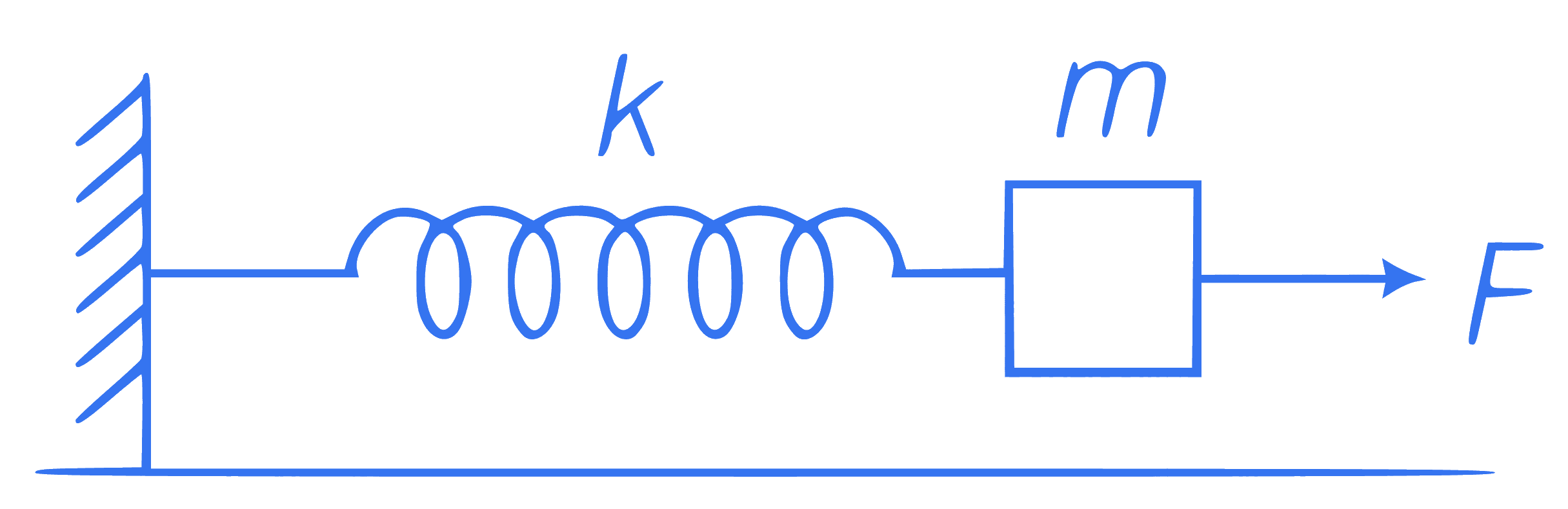
The figure shows a mass $$m$$ on a frictionless surface. It is connected to rigid wall by the mean of a massless spring of its constant $$k$$. Initially, the spring is at its natural position. If a force of constant magnitude starts acting on the block towards right, then the speed of the block when the deformation in spring is $$x$$, will be

Q52.
mcq single
+1 / 0.33
A metal wire has a resistance of $$35 \Omega$$. If its length is increased to double by drawing it, then its new resistance will be
Q53.
mcq single
+1 / 0.33
In the adjoining circuit diagram, the
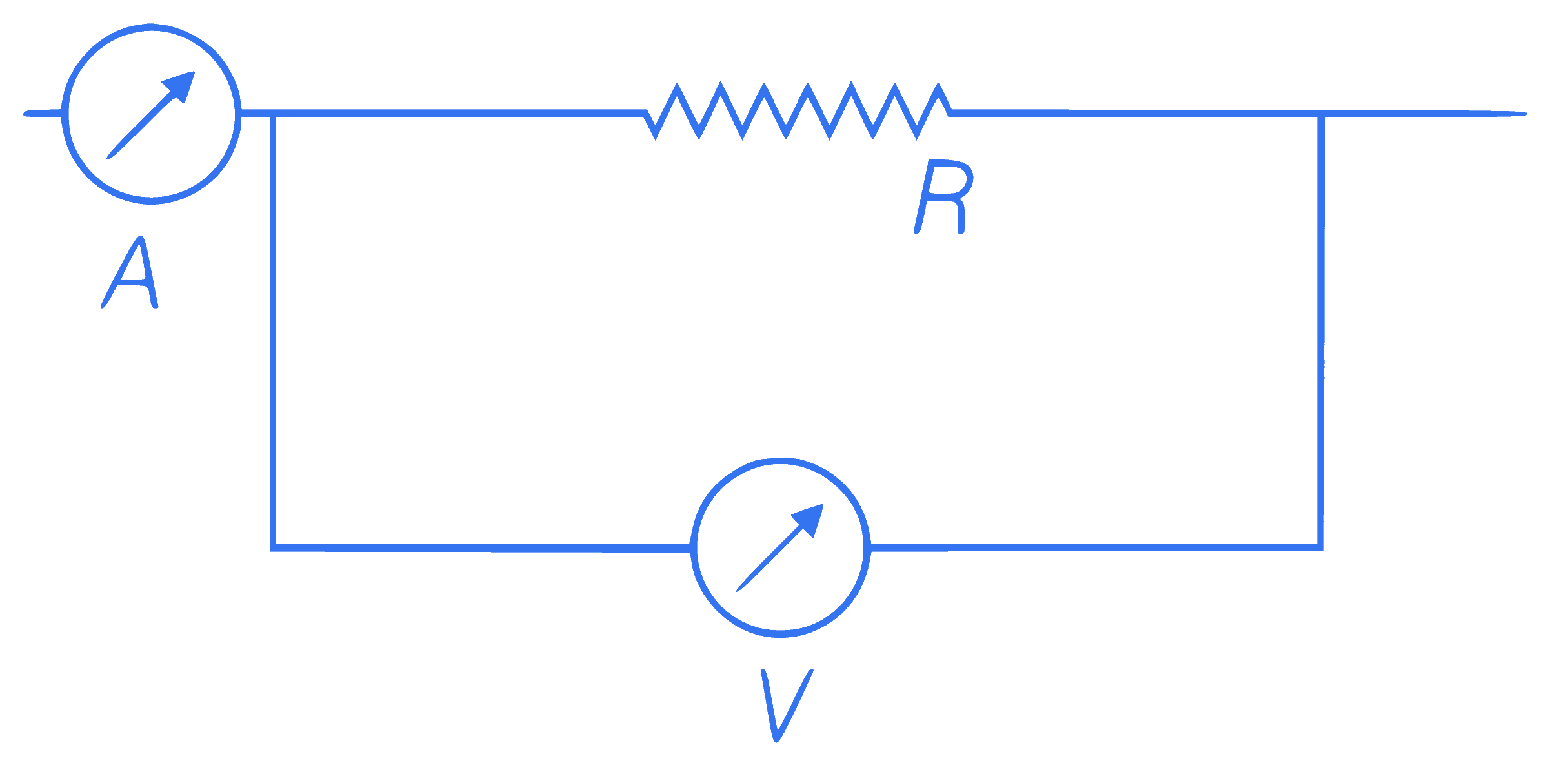
readings of ammeter and voltmeter are 2 A
and 120 V, respectively. If the value of R is
75$$\Omega$$, then the voltmeter resistance will be

Q54.
mcq single
+1 / 0.33
Assertion Terminal voltage of a cell is greater
than emf of cell during charging of the cell.
Reason The emf of a cell is always greater
than its terminal voltage.
Q55.
mcq single
+1 / 0.33
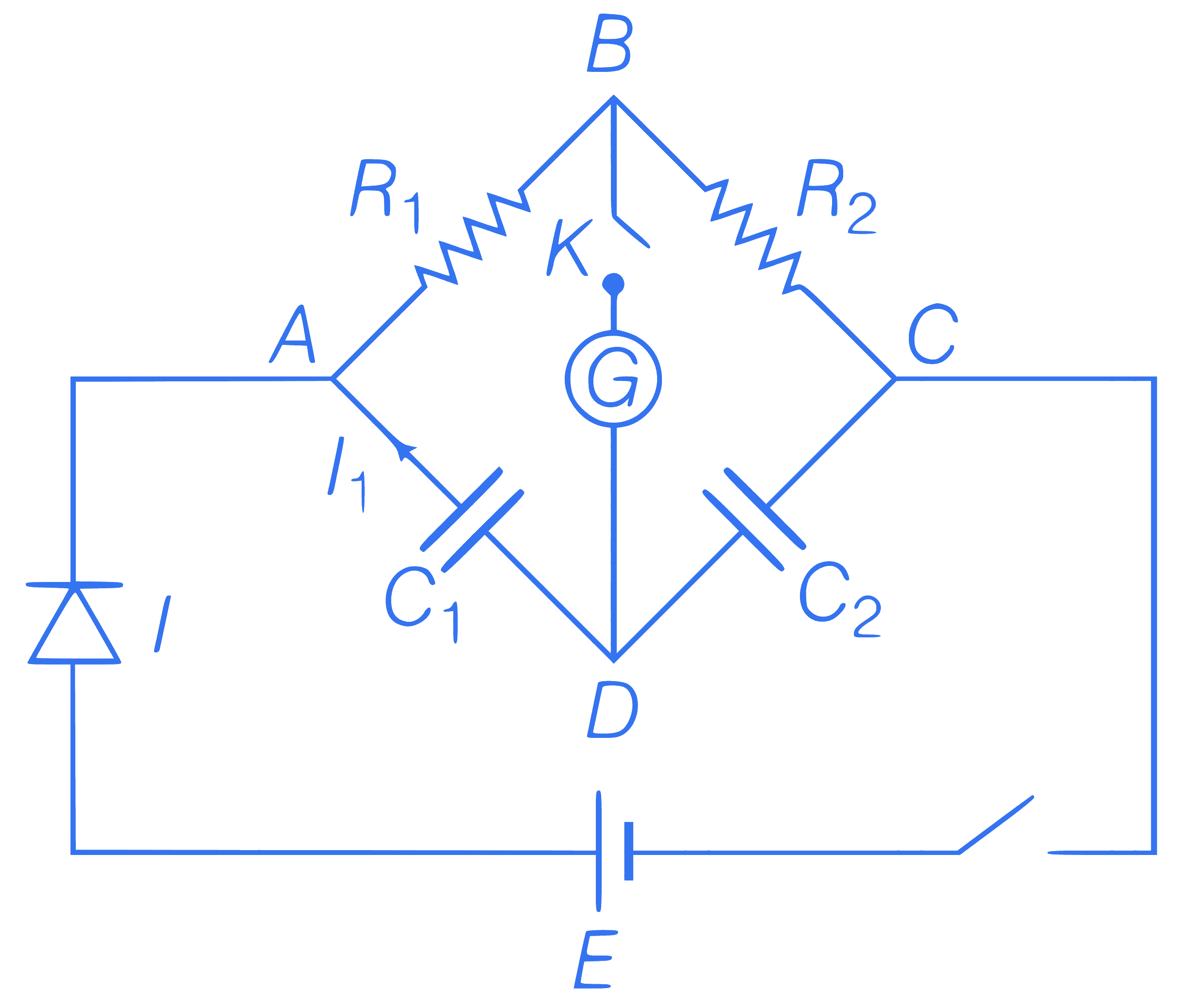
In the circuit in the figure, if no current
flows through the galvanometer when the
key K is closed, the bridge is balanced. The
balancing condition for bridge is

Q56.
mcq single
+1 / 0.33
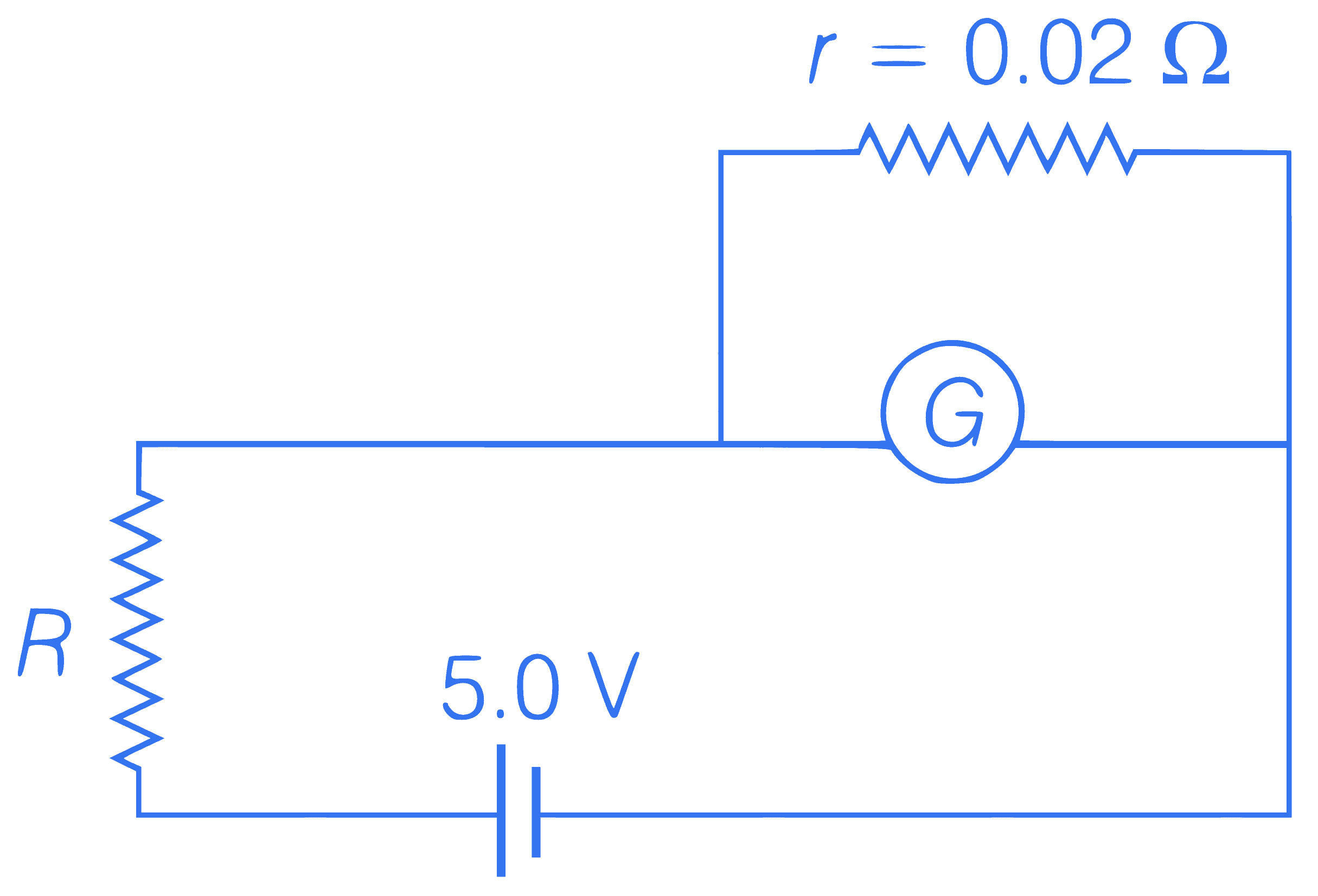
In the circuit, shown the galvanometer $$G$$ of resistance $$60 \Omega$$ is shunted by a resistance $$r=0.02 \Omega$$. The current through $$R$$ is nearly $$1 \mathrm{~A}$$. The value of resistance $$R$$ (in ohm) is nearly

Q57.
mcq single
+1 / 0.33
Assertion Bulb generally get fused when
they are switched on or off.
Reason When we switch on or off, a circuit
current changes in it rapidly.
Q58.
mcq single
+1 / 0.33
In a circuit $$L, C$$ and $$R$$ are connected in series with an alternating voltage source of frequency $$f$$. The current leads the voltage by $$45^{\circ}$$. The value of $$C$$ is
Q59.
mcq single
+1 / 0.33
An ideal coil of $$10 \mathrm{~H}$$ is connected in series with a resistance of $$5 \Omega$$ and a battery of $$5 \mathrm{~V}$$. After $$2 \mathrm{~s}$$, after the connection is made, the current flowing (in ampere) in the circuit is
Q60.
mcq single
+1 / 0.33
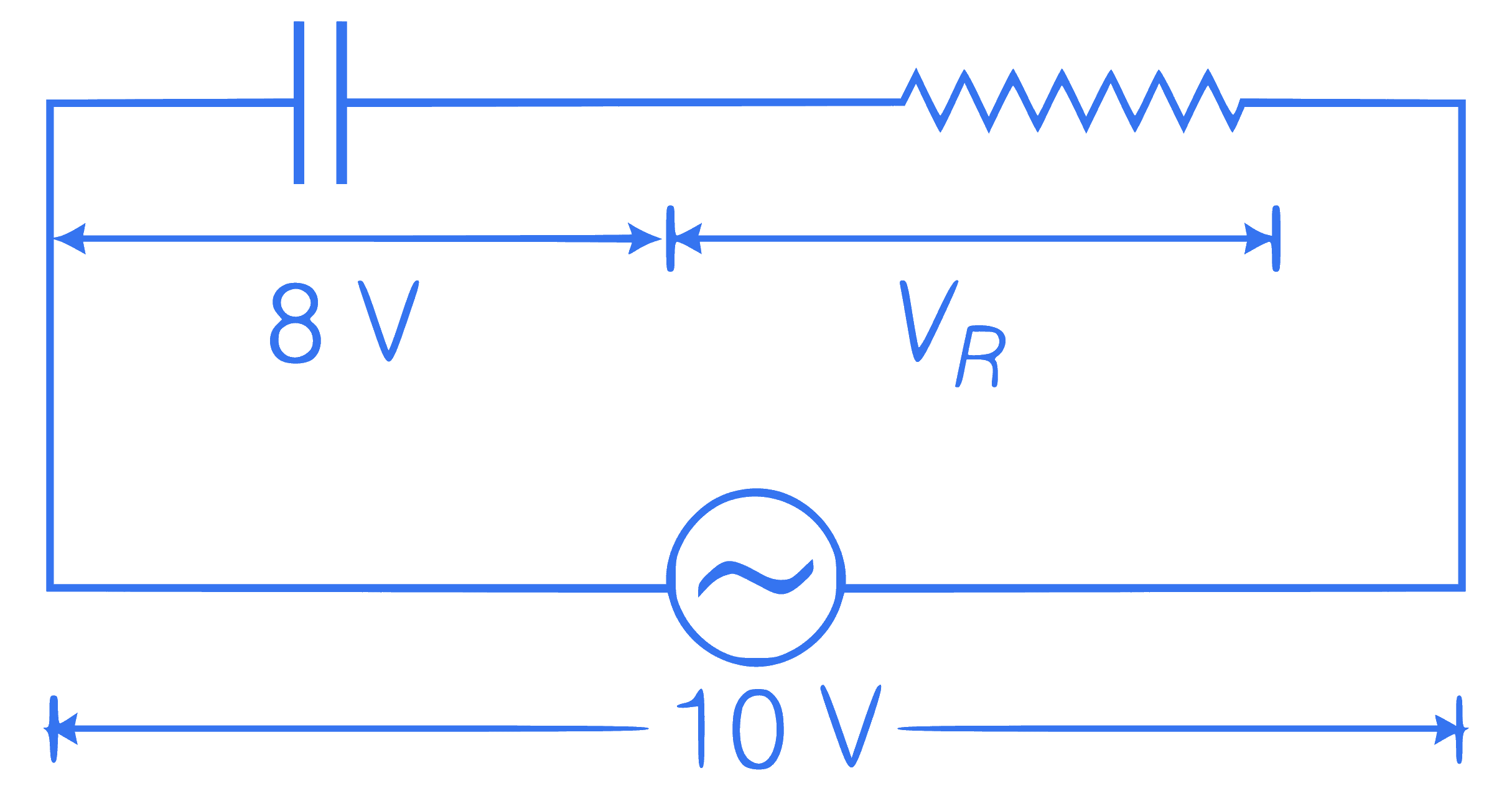
In a series R-C circuit shown in figure, the
applied voltage is 10 V and the voltage across
capacitor is found to be 8V. Then, the voltage
across R and the phase difference between
current and the applied voltage will
respectively be

Q61.
mcq single
+1 / 0.33
Red light of wavelength 5400 $$\mathop A\limits^o $$ from a
distant source falls on a slit 0.80 mm wide.
Calculate the distance between first two dark
bands on each side of central bright band in
the diffraction pattern observed on a screen
place 1.4 m from the slit.
Q62.
mcq single
+1 / 0.33
**Assertion :** If a glass slab is placed in front of
one of the slits, then fringe with will
decrease.
**Reason :** Glass slab will produce an
additional path difference.
Q63.
mcq single
+1 / 0.33
An unpolarised beam of intensity $$2 a^2$$ passes through a thin polaroid. Assuming zero absorption in the polaroid, the intensity of emergent plane polarised light is
Q64.
mcq single
+1 / 0.33
Assertion The angular momentum of
system always remain constant.
Reason For a system, $$\tau_{\mathrm{ext}}=\frac{d L}{d t}=0$$
Q65.
mcq single
+1 / 0.33
A thin horizontal circular disc is rotating about a vertical axis passing through its centre. An insect is at rest at a point near the rim of disc. The insect now moves along a diameter of the disc to reach its other end. During the journey of the insect, the angular speed of the disc
Q66.
mcq single
+1 / 0.33
A system $$S$$ consists of two coils $$A$$ and $$B$$. The coil $$A$$ carries a steady current $$I$$. While the coil $$B$$ is suspended nearby as shown in figure. Now, if the system is heated, so as to raise the temperature of two coils steadily, then


Q67.
mcq single
+1 / 0.33
A circular loop of radius 0.3 cm lies parallel
to a much bigger circular loop of radius 20 cm.
The centre of the small loop on the axis of
the bigger loop. The distance between their
centres is 15 cm. If a current of 20 A flows
through the smaller loop, then the flux
linked with bigger drop is
Q68.
mcq single
+1 / 0.33
If the formula, $$X=3 Y Z^2, X$$ and $$Z$$ have dimensions of capacitance and magnetic induction. The dimensions of $$Y$$ in MKSQ system are
Q69.
mcq single
+1 / 0.33
 An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
 An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
An ideal gas of mass $$m$$ in a state $$A$$ goes to another state $$B$$ via three different processes as shown in figure. If $$Q_1, Q_2$$ and $$Q_3$$ denote the heat absorbed by the gas along the three paths, then
Q70.
mcq single
+1 / 0.33
Assertion Internal energy of an ideal gas
does not depend on volume of gas.
Reason Internal energy depends only on
temperature of gas.
Q71.
mcq single
+1 / 0.33
A gas consisting of a rigid diatomic molecules
was initially under standard condition. Then,
gas was compressed adiabatically to
one-fifth of its initial volume. What will be
the mean kinetic energy of a rotating
molecule in the final state?
Q72.
mcq single
+1 / 0.33
Assertion In isothermal process, whole of
the heat energy supplied to the body is
converted into internal energy.
Reason According to the first law of
thermodynamics,
$$\Delta Q=\Delta U+\Delta W$$
Q73.
mcq single
+1 / 0.33
Assertion Smaller drop of water resist deformation forces better than the larger drops.
Reason Excess pressure inside drop is inversely proportional to its radius.
Q74.
mcq single
+1 / 0.33
A half ring of radius $$R$$ has a charge of $$\lambda$$ per unit length. The electric force on $$1 \mathrm{C}$$ charged placed at the centre is
Q75.
mcq single
+1 / 0.33
Assertion Mass of a body decreases slightly
when it is negatively charged.
Reason Charging is due to transfer of electrons.
Q76.
mcq single
+1 / 0.33
Positive charge $$Q$$ is distributed uniformly over a circular ring of radius $$R$$. A point particle having a mass $$(m)$$ and a negative charge $$-q$$ is placed on its axis at a distance $$x$$ from the centre. Assuming $$x < R$$, find the time period of oscillation of the particle, if it is released from there [neglect gravity].
Q77.
mcq single
+1 / 0.33
The weight of a body on the surface of the
earth is 63 N. What is the gravitational force
on it due to the earth at a height equal to
half the radius of the earth?
Q78.
mcq single
+1 / 0.33
Two satellites $$A$$ and $$B$$ revolve round the same planet in coplanar circular orbits lying in the same plane. Their periods of revolutions are $$1 \mathrm{~h}$$ and $$8 \mathrm{~h}$$, respectively. The radius of the orbit of $$A$$ is $$10^4 \mathrm{~km}$$. The speed of $$B$$ is relative to $$A$$. When they are closed in $$\mathrm{km} / \mathrm{h}$$ is
Q79.
mcq single
+1 / 0.33
A planet is revolving around the sun in a circular orbit with a radius $$r$$. The time period is $$T$$. If the force between the planet and star is proportional to $$r^{-3 / 2}$$, then the square of time period is proportional to
Q80.
mcq single
+1 / 0.33
An infinite number of identical capacitors
each of capacitance 1 $$\mu$$F are connected as
shown in the figure. Then, the equivalent
capacitance between A and B is


Q81.
mcq single
+1 / 0.33
Assertion A dielectric slab is inserted
between plates of an isolated charged
capacitor which remain same.
Reason Charge on an isolated system is
conserved.
Q82.
mcq single
+1 / 0.33
Assertion A convex mirror always make a
virtual image.
Reason The ray always diverge after
reflection from the convex mirror.
Q83.
mcq single
+1 / 0.33
Assertion A pendulum is falling freely its
time period becomes zero.
Reason Freely falling body has the
acceleration equal to g.
Q84.
mcq single
+1 / 0.33
A block of rectangular size of mass $$m$$ and area of cross-section $$A$$, floats in a liquid of density $$\rho$$. If we give a small vertical displacement from equilibrium, it undergoes SHM with time period $$T$$, then
Q85.
mcq single
+1 / 0.33
 The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
 The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
The diode used at a
constant potential drop
of 0.5 V at all currents
and maximum power
rating of 100 mW. What
resistance must be
connected in series diode, so that current in
circuit is maximum?
Q86.
mcq single
+1 / 0.33
Assertion Thickness of depletion layer is
fixed in all semiconductor devices.
Reason No free charge carriers are available
in depletion layer.
Q87.
mcq single
+1 / 0.33
An element $$d l=d x \hat{\mathbf{i}}$$ (where, $$d x=1 \mathrm{~cm}$$ ) is placed at the origin and carries a large current $$i=10 \mathrm{~A}$$. What is the magnetic field on the $$Y$$-axis at a distance of $$0.5 \mathrm{~m}$$ ?
Q88.
mcq single
+1 / 0.33
Consider the following figure, a uniform
magnetic field of 0.2 T is directed along the
positive X-axis. The magnetic flux through
top surface of the figure.


Q89.
mcq single
+1 / 0.33
A long straight wire, carrying current $$I$$ is bent at its mid-point to form an angle of $$45^{\circ}$$. Induction of magnetic field (in tesla) at point $$P$$, distant $$R$$ from point of bending is equal to


Q90.
mcq single
+1 / 0.33
Assertion A magnetic field interacts with a
moving charge and not with a stationary
charge.
Reason A moving charge produce a
magnetic field.
Q91.
mcq single
+1 / 0.33
A diode detector is used to detect and
amplitude modulated wave of 60%
modulation by using a condenser of capacity
250 pF in parallel with a load resistance
100 k$$\Omega$$. Find the maximum modulated
frequency which could be detected by it.
Q92.
mcq single
+1 / 0.33
The graph between the energy log E of an
electron and its de-Broglie wavelength log $$\lambda$$ will be
Q93.
mcq single
+1 / 0.33
Assertion A body is momentarily at rest at
the instant, if it reverse the direction.
Reason A body cannot have acceleration, if
its velocity is zero at a given instant of time.
Q94.
mcq single
+1 / 0.33
Assertion If electrons in an atom were
stationary, then they would fall into the
nucleus.
Reason Electrostatic force of attraction acts
between negatively charged electrons and
positive nucleus.
Q95.
mcq single
+1 / 0.33
The half-life of a radioactive substance is $$20 \mathrm{~min}$$. The approximate time interval $$\left(t_2-t_1\right)$$ between the time $$t_2$$, when $$\frac{2}{3}$$ of it has decayed and time $$t_1$$ when $$\frac{1}{3}$$ of it had decayed is
Q96.
mcq single
+1 / 0.33
Assertion Radioactive nuclei emits $$\beta^{-}$$-particles.
Reason Electrons exist inside the nucleus.
Q97.
mcq single
+1 / 0.33
Assertion The maximum height of projectile
is always 25% of the maximum range.
Reason For maximum range, projectile
should be projected at 90°.
Q98.
mcq single
+1 / 0.33
Assertion Two particles moving in the same
direction do not lose all their energy in
completely inelastic collision.
Reason Principle of conservation of
momentum holds true for all kinds of
collisions.
Q99.
mcq single
+1 / 0.33
Body of mass M is much heavier than the
other body of mass m. The heavier body with
speed v collides with the lighter body which
was at rest initially elastically. The speed of
lighter body after collision is
Q100.
mcq single
+1 / 0.33
Three bodies having masses $$5 \mathrm{~kg}, 4 \mathrm{~kg}$$ and $$2 \mathrm{~kg}$$ is moving at the speed of $$5 \mathrm{~m} / \mathrm{s}, 4 \mathrm{~m} / \mathrm{s}$$ and $$2 \mathrm{~m} / \mathrm{s}$$ respectively along $$X$$-axis. The magnitude of velocity of centre of mass is
Q101.
mcq single
+1 / 0.33
A pipe of length 85 cm is closed from one
end. Find the number of possible natural
oscillations of air column in the pipe whose
frequencies lie below 1250 Hz. The velocity
of sound in air is 340 m/s.
Q102.
mcq single
+1 / 0.33
A steel rod 100 cm long is damped at into
middle. The fundamental frequency of
longitudinal vibrations of the rod are given
to be 2.53 kHz. What is the speed of sound
in sound is steel?
Q103.
mcq single
+1 / 0.33
Assertion To hear different beats difference
of the frequencies of two sources should be
less than 10.
Reason More the number of beats more in
the confusion.
Q104.
mcq single
+1 / 0.33
Assertion Angle of repose is equal to angle
of limiting friction.
Reason When a body is just at the point of
motion, the force of friction of this stage is
called as limiting friction.
Q105.
mcq single
+1 / 0.33
A body of mass 5 kg is suspended by a spring
balance on an inclined plane as shown in
figure.
 So, force applied on spring balance is
So, force applied on spring balance is
 So, force applied on spring balance is
So, force applied on spring balance is
Q106.
mcq single
+1 / 0.33
 In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
 In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
In the figure, blocks A and B of
masses 2m and m are connected
with a string and system is hanged
vertically with the help of a spring.
Spring has negligible mass. Find
out magnitude of acceleration of
masses 2m and m just after the
instant when the string is cut
Q107.
mcq single
+1 / 0.33
A wooden wedge of mass $$M$$ and inclination angle $$(\alpha)$$ rest on a smooth floor. A block of mass $m$ is kept on wedge. A force $$F$$ is applied on the wedge as shown in the figure such that block remains stationary with respect to wedge. So, magnitude of force $$F$$ is


Q108.
mcq single
+1 / 0.33
A piece of ice slides down a rough inclined plane at $$45^{\circ}$$ inclination in twice the time that it takes to slide down an identical but frictionless inclined plane. What is the coefficient of friction between ice and incline?
Q109.
mcq single
+1 / 0.33
 In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
 In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
In the figure, mass of a ball is $$\frac{9}{5}$$ times mass of the rod. Length of rod is $$1 \mathrm{~m}$$. The level of ball is same as rod level. Find out time taken by the ball to reach at upper end of rod.
Q110.
mcq single
+1 / 0.33
The horizontal component of the earth's magnetic field at any place is $$0.36 \times 10^{-4} \mathrm{~Wb} / \mathrm{m}^2$$. If the angle of dip at that place is $$60^{\circ}$$, then the value of vertical component of the earth's magnetic field will be (in $$\mathrm{Wb} / \mathrm{m}^2$$ )
Q111.
mcq single
+1 / 0.33
The figure shows a mass $$m$$ on a frictionless surface. It is connected to rigid wall by the mean of a massless spring of its constant $$k$$. Initially, the spring is at its natural position. If a force of constant magnitude starts acting on the block towards right, then the speed of the block when the deformation in spring is $$x$$, will be


Q112.
mcq single
+1 / 0.33
A metal wire has a resistance of $$35 \Omega$$. If its length is increased to double by drawing it, then its new resistance will be
Q113.
mcq single
+1 / 0.33
In the adjoining circuit diagram, the
readings of ammeter and voltmeter are 2 A
and 120 V, respectively. If the value of R is
75$$\Omega$$, then the voltmeter resistance will be


Q114.
mcq single
+1 / 0.33
Assertion Terminal voltage of a cell is greater
than emf of cell during charging of the cell.
Reason The emf of a cell is always greater
than its terminal voltage.
Q115.
mcq single
+1 / 0.33
In the circuit in the figure, if no current
flows through the galvanometer when the
key K is closed, the bridge is balanced. The
balancing condition for bridge is


Q116.
mcq single
+1 / 0.33
In the circuit, shown the galvanometer $$G$$ of resistance $$60 \Omega$$ is shunted by a resistance $$r=0.02 \Omega$$. The current through $$R$$ is nearly $$1 \mathrm{~A}$$. The value of resistance $$R$$ (in ohm) is nearly


Q117.
mcq single
+1 / 0.33
Assertion Bulb generally get fused when
they are switched on or off.
Reason When we switch on or off, a circuit
current changes in it rapidly.
Q118.
mcq single
+1 / 0.33
In a circuit $$L, C$$ and $$R$$ are connected in series with an alternating voltage source of frequency $$f$$. The current leads the voltage by $$45^{\circ}$$. The value of $$C$$ is
Q119.
mcq single
+1 / 0.33
An ideal coil of $$10 \mathrm{~H}$$ is connected in series with a resistance of $$5 \Omega$$ and a battery of $$5 \mathrm{~V}$$. After $$2 \mathrm{~s}$$, after the connection is made, the current flowing (in ampere) in the circuit is
Q120.
mcq single
+1 / 0.33
In a series R-C circuit shown in figure, the
applied voltage is 10 V and the voltage across
capacitor is found to be 8V. Then, the voltage
across R and the phase difference between
current and the applied voltage will
respectively be


Biology
Biology
Q1.
mcq single
+1 / 0.33
Match Column I (Antibiotic) with Column II
(Source) and choose the correct option from
the codes given below.
Column I
Column II
A.
Fumagillin
1.
Gliocladium virens
B.
Bacitracin
2.
Streptomyces griseus
C.
Streptomycin
3.
Bacillus polymyxa
D.
Viridin
4.
Aspergillus fumigatus
E.
Polymixin
5.
Bacillus licheniformis
Q2.
mcq single
+1 / 0.33
Match the organisms given in Column I to
their functions given in Column II and
choose the correct option.
Column I
Column II
A.
Thiobacillus
1.
Free-living nitrogen-fixing cyanobacteria
B.
Nitrosomonas
2.
Denitrification
C.
Nostoc
3.
Free-living aerobic nitrogen-fixing bacteria
D.
Azotobactor
4.
$$\mathrm{NH_3}$$ to Nitrite
Q3.
mcq single
+1 / 0.33
Select the incorrect match.
I. Sedimentary nutrient cycle-Nitrogen cycle
II. Pioneer species - Lichens
III. Secondary succession - Burned forests
IV. Pyramid of biomass in sea - Upright
Q4.
mcq single
+1 / 0.33
Assertion The non-allelic genes for red hair
and prickles are usually inherited together.
Reason The genes for red hair and prickles
are located on the same chromosome in close
association.
Q5.
mcq single
+1 / 0.33
Select the incorrect statement(s) from the options given below with respect to dihybrid cross.
I. Tightly linked genes on the same chromosome show higher recombinations.
II. Genes far apart on the same chromosome show very few recombinations.
III. Genes loosely linked on the same chromosome show similar recombinations.
Q6.
mcq single
+1 / 0.33
Which one of the following conditions correctly describes the manner of determining the sex?
Q7.
mcq single
+1 / 0.33
A normal woman whose father was colourblind, marries a normal man. What kinds of children can be expected and in what proportion?
Q8.
mcq single
+1 / 0.33
Assertion Phenylketonuria is recessive
hereditary disease caused by body’s failure to
oxidise an amino acid phenylalanine to
tyrosine, because of defective enzyme.
Reason It is characterised by in the
presence of phenylalanine acid in urine.
Q9.
mcq single
+1 / 0.33
$$\mathrm{C}_4$$ pathway is advantageous over $$\mathrm{C}_3$$ pathway in plants, because it
Q10.
mcq single
+1 / 0.33
Assertion Non-cyclic photophosphorylation
occurs in the stroma of chloroplasts.
Reason There is discontinuous flow of
electrons in this process.
Q11.
mcq single
+1 / 0.33
Assertion Taenia solium and Dugesia belong
to Platyhelminthes.
Reason : Platyhelminthes are coelomates.
Q12.
mcq single
+1 / 0.33
cry II Ab and cry I Ab produce toxins that control
Q13.
mcq single
+1 / 0.33
Find the correct statements from the following
I. Gene therapy is a genetic engineering technique used to treat diseases at molecular level.
II. Calcitonin is medically useful recombinant product in the treatment of infertility.
III. Bt toxin is a biodegradable insecticide obtained from Bacillus thuringiensis.
Q14.
mcq single
+1 / 0.33
Diagram of large intestine is given below.
Identify the parts A, B, C, D, E and F.


Q15.
mcq single
+1 / 0.33
**Assertion :** The collenchyma is a thick-walled
living tissue.
**Reason :** The collenchyma is thickened due
to deposition of pectin.
Q16.
mcq single
+1 / 0.33
Identify the permanent tissues shown in the
following figures


Q17.
mcq single
+1 / 0.33
Codons of glycine are
Q18.
mcq single
+1 / 0.33
Adults of Wuchereria bancrofti attack
Q19.
mcq single
+1 / 0.33
Read the following statements
I. Colostrum is recommended for the new borns because it is rich in antigens.
II. Chikungunya is caused by a Gram negative bacterium.
III. Tissue culture has proved useful in obtaining virus-free plants.
IV. Beer is manufactured by distillation of fermented grapes.
How many of the statement(s) is/are correct?
Q20.
mcq single
+1 / 0.33
Characteristics of cancer are
Q21.
mcq single
+1 / 0.33
Which of the following plants are used to treat bone fractures?
Q22.
mcq single
+1 / 0.33
Assertion Alcoholics may show deficiency
symptoms of Wernicke’s and Korsakoff’s
syndromes.
Reason Alcohol acts as depressant.
Q23.
mcq single
+1 / 0.33
Person 'A' cannot step out of his house. He has to spend his entire life in sterile isolation otherwise, he would quickly contract a fatal infection. This person has almost no effective immune system. This disease is also called as baby in a bubble syndrome. Identify the disease, this person 'A' is suffering from
Q24.
mcq single
+1 / 0.33
Assertion Pork should be properly cooked to avoid Taenia infection.
Reason Pork of pig contains hexacanth and cysticercus larvae.
Q25.
mcq single
+1 / 0.33
Which of the following pathways occurs through cell wall?
Q26.
mcq single
+1 / 0.33
A mutant plant is unable to produce materials or precursors that form Casparian strip. This plant would be
Q27.
mcq single
+1 / 0.33
Cell A has osmotic pressure of $$-$$20 bars and pressure potential of 5 bars, whereas cell B has osmotic pressure of $$-$$18 bars and pressure potential of 2 bars.
The direction of flow of water will be
Q28.
mcq single
+1 / 0.33
**Assertion :** BOD is an indicator of pollution
in water.
**Reason :** High BOD is observed in highly
polluted water.
Q29.
mcq single
+1 / 0.33
Biomagnification can be defined as
Q30.
mcq single
+1 / 0.33
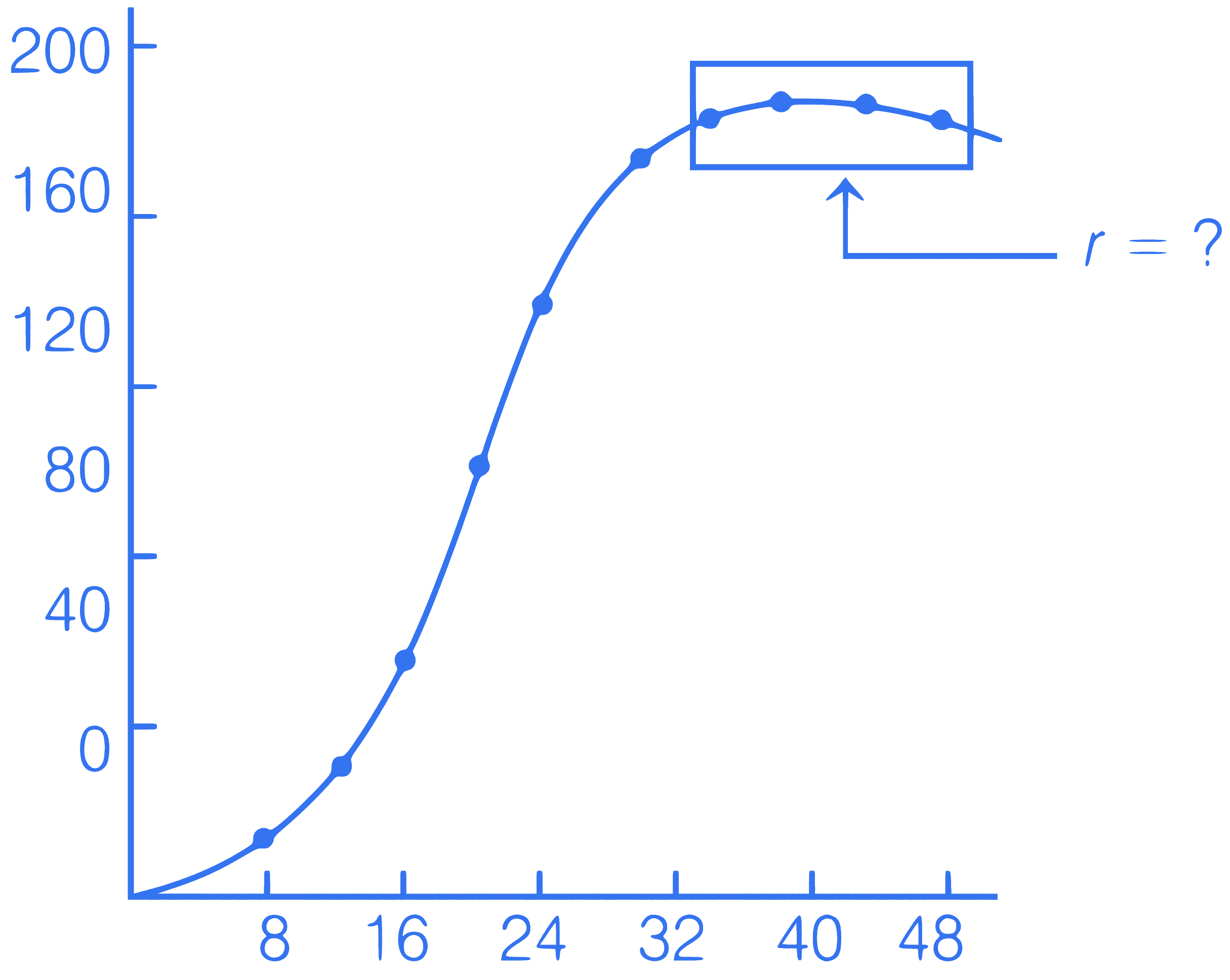
From the graph of population growth, select the correct option having correct value of '$$r$$' and bar graph.

Q31.
mcq single
+1 / 0.33
Match the stages of meiosis in column I to
their characteristic features in Column II and
select the correct option using the codes
given below.
Column I
Column II
A.
Diakinesis
1.
Crossing over takes place
B.
Pachytene
2.
Terminalisation of chiasmata
C.
Zygotene
3.
Chromosomes align at equatorial place
D.
Metaphase
4.
Pairing of homologous chromosomes
Q32.
mcq single
+1 / 0.33
Assertion In mitosis, two identical cells are
produced from a single cell and karyokinesis
is followed by cytokinesis.
Reason Cytokinesis is of two types, i.e. by
cell-furrow method and cell-plate method.
Q33.
mcq single
+1 / 0.33
Assertion Meiotic division occurs in
reproductive cells.
Reason Synapsis occurs during zygotene of
meiosis.
Q34.
mcq single
+1 / 0.33
Assertion In females, parturition is the act
of giving birth to a baby.
Reason Signals for parturition originate
from a fully developed foetus.
Q35.
mcq single
+1 / 0.33
Assertion Peptide and polypeptide
hormones directly pass across the lipid
bilayer of plasma membrane.
Reason Oxytocin hormone can pass across
the plasma membrane.
Q36.
mcq single
+1 / 0.33
Triticale is the first man-made cereal crop. Mention the type of hybridisation through which it was produced.
Q37.
mcq single
+1 / 0.33
A scion is grafted to a stock. The quality of
fruits produced will be determined by the
genotype of
Q38.
mcq single
+1 / 0.33
Consider the following four measures (I-IV) that could be taken to successfully grow chickpea in an area where bacterial blight is common.
I. Spray with Bordeaux mixture.
II. Control of the insect vector of the disease pathogen.
III. Use of disease-free seeds only.
IV. Use of varieties resistant to the disease.
Which of the above measures can control the diseases?
Q39.
mcq single
+1 / 0.33
Read the following statements
I. Species diversity decreases as we move away from the equator towards the poles.
II. Stellar's sea cow and passenger pigeon got extinct due to overexploitation by man.
III. Lantana and Eichhornia are invasive weed in India.
IV. The historic convention on biological diversity was held in 1992.
Choose the option containing correct statements.
Q40.
mcq single
+1 / 0.33
Apical dominance is caused by
Q41.
mcq single
+1 / 0.33
Assertion Photomodulation of flower is
phytochrome regulated process.
Reason Active form of phytochrome (Pfr)
directly induces floral induction in shoot
buds.
Q42.
mcq single
+1 / 0.33
**Assertion :** The quiescent centre acts as a
reservoir of relatively resistant cells, which
constitute a permanent source of active initials.
**Reason :** The cells of the inactive region of
quiescent centre become active, when the
previous active initials get damaged.
Q43.
mcq single
+1 / 0.33
Assertion Cytochrome oxidase enzyme
contains copper.
Reason Cyanide combines with copper of
cytochrome oxide and prevents oxygen to
combine with it.
Q44.
mcq single
+1 / 0.33
Assertion Two turns of Krebs’ cycle occur
per glucose molecule used.
Reason Each turn of Krebs’ cycle produces
3 NADH, 1FADH$$_2$$ and 1 ATP molecule.
Q45.
mcq single
+1 / 0.33
Match the following columns.
Column I
Column II
A.
Hinge
1.
Atlas and axis
B.
Saddle
2.
Frontal and parietal
C.
Pivot
3.
Carpal and metacarpal of humans
D.
Ball and socket
4.
Knee
5.
Humerus and pectoral girdle
Q46.
mcq single
+1 / 0.33
Which one of the following organisms do not evolve oxygen during photosynthesis?
Q47.
mcq single
+1 / 0.33
What is true about the isolated small tribal populations?
Q48.
mcq single
+1 / 0.33
The peppered moth (Biston betularia), the black-coloured form becomes dominant over the light-coloured form of moth in England during industrial revolution. This is an example of
Q49.
mcq single
+1 / 0.33
Choose the correct statements with reference to organic evolution.
I. Flippers of whale and wings of bat exhibit analogy.
II. Wings of butterfly and wings of bird exhibit homology.
III. Organs with dissimilar structure are called analogous organs.
IV. Organs with similar structure and origin are called homologous organs.
Q50.
mcq single
+1 / 0.33
Assertion In apomixis, the plants of new
genetic sequence are produced.
Reason In apomixis, two organisms of
same genetic sequence meet.
Q51.
mcq single
+1 / 0.33
Assertion Caryopsis fruits differ from
typical achenes with respect to the fusion of
pericarp with the seed-coat (testa).
Reason Caryopsis fruits commonly occur in
the members of family–Poaceae.
Q52.
mcq single
+1 / 0.33
A plant has a butterfly-shaped flower with
one standard, two wing like and two keel
petals. To which family, this plant belongs?
Q53.
mcq single
+1 / 0.33
Leaf tendrils are found in
Q54.
mcq single
+1 / 0.33
Arrange in correct order according to the
given figures.

Q55.
mcq single
+1 / 0.33
Which of the following biomolecules is common to respiration-mediated breakdown of fats, carbohydrates and proteins?
Q56.
mcq single
+1 / 0.33
If a recombinant DNA bearing gene for ampicillin resistance is transferred into E. coli cells and the host cells are spread on agar plates containing ampicillin, then
Q57.
mcq single
+1 / 0.33
Production of human protein in bacteria by genetic engineering is possible because
Q58.
mcq single
+1 / 0.33
Azolla is used as a biofertiliser because it
Q59.
mcq single
+1 / 0.33
Assertion Magnesium is important in photosynthesis and carbohydrate metabolism.
Reason $$\mathrm{Mg}^{2+}$$ is involved in the synthesis of nucleic acids.
Q60.
mcq single
+1 / 0.33
Assertion Nitrogen-fixing bacteria of
legume root nodules survive in oxygen
depleted cells.
Reason Leghaemoglobin completely
removes oxygen from nodule cells.
Q61.
mcq single
+1 / 0.33
Match Column I (Antibiotic) with Column II
(Source) and choose the correct option from
the codes given below.
Column I
Column II
A.
Fumagillin
1.
Gliocladium virens
B.
Bacitracin
2.
Streptomyces griseus
C.
Streptomycin
3.
Bacillus polymyxa
D.
Viridin
4.
Aspergillus fumigatus
E.
Polymixin
5.
Bacillus licheniformis
Q62.
mcq single
+1 / 0.33
Match the organisms given in Column I to
their functions given in Column II and
choose the correct option.
Column I
Column II
A.
Thiobacillus
1.
Free-living nitrogen-fixing cyanobacteria
B.
Nitrosomonas
2.
Denitrification
C.
Nostoc
3.
Free-living aerobic nitrogen-fixing bacteria
D.
Azotobactor
4.
$$\mathrm{NH_3}$$ to Nitrite
Q63.
mcq single
+1 / 0.33
Select the incorrect match.
I. Sedimentary nutrient cycle-Nitrogen cycle
II. Pioneer species - Lichens
III. Secondary succession - Burned forests
IV. Pyramid of biomass in sea - Upright
Q64.
mcq single
+1 / 0.33
Assertion The non-allelic genes for red hair
and prickles are usually inherited together.
Reason The genes for red hair and prickles
are located on the same chromosome in close
association.
Q65.
mcq single
+1 / 0.33
Select the incorrect statement(s) from the options given below with respect to dihybrid cross.
I. Tightly linked genes on the same chromosome show higher recombinations.
II. Genes far apart on the same chromosome show very few recombinations.
III. Genes loosely linked on the same chromosome show similar recombinations.
Q66.
mcq single
+1 / 0.33
Which one of the following conditions correctly describes the manner of determining the sex?
Q67.
mcq single
+1 / 0.33
A normal woman whose father was colourblind, marries a normal man. What kinds of children can be expected and in what proportion?
Q68.
mcq single
+1 / 0.33
Assertion Phenylketonuria is recessive
hereditary disease caused by body’s failure to
oxidise an amino acid phenylalanine to
tyrosine, because of defective enzyme.
Reason It is characterised by in the
presence of phenylalanine acid in urine.
Q69.
mcq single
+1 / 0.33
$$\mathrm{C}_4$$ pathway is advantageous over $$\mathrm{C}_3$$ pathway in plants, because it
Q70.
mcq single
+1 / 0.33
Assertion Non-cyclic photophosphorylation
occurs in the stroma of chloroplasts.
Reason There is discontinuous flow of
electrons in this process.
Q71.
mcq single
+1 / 0.33
Assertion Taenia solium and Dugesia belong
to Platyhelminthes.
Reason : Platyhelminthes are coelomates.
Q72.
mcq single
+1 / 0.33
cry II Ab and cry I Ab produce toxins that control
Q73.
mcq single
+1 / 0.33
Find the correct statements from the following
I. Gene therapy is a genetic engineering technique used to treat diseases at molecular level.
II. Calcitonin is medically useful recombinant product in the treatment of infertility.
III. Bt toxin is a biodegradable insecticide obtained from Bacillus thuringiensis.
Q74.
mcq single
+1 / 0.33
Diagram of large intestine is given below.
Identify the parts A, B, C, D, E and F.


Q75.
mcq single
+1 / 0.33
**Assertion :** The collenchyma is a thick-walled
living tissue.
**Reason :** The collenchyma is thickened due
to deposition of pectin.
Q76.
mcq single
+1 / 0.33
Identify the permanent tissues shown in the
following figures


Q77.
mcq single
+1 / 0.33
Codons of glycine are
Q78.
mcq single
+1 / 0.33
Adults of Wuchereria bancrofti attack
Q79.
mcq single
+1 / 0.33
Read the following statements
I. Colostrum is recommended for the new borns because it is rich in antigens.
II. Chikungunya is caused by a Gram negative bacterium.
III. Tissue culture has proved useful in obtaining virus-free plants.
IV. Beer is manufactured by distillation of fermented grapes.
How many of the statement(s) is/are correct?
Q80.
mcq single
+1 / 0.33
Characteristics of cancer are
Q81.
mcq single
+1 / 0.33
Which of the following plants are used to treat bone fractures?
Q82.
mcq single
+1 / 0.33
Assertion Alcoholics may show deficiency
symptoms of Wernicke’s and Korsakoff’s
syndromes.
Reason Alcohol acts as depressant.
Q83.
mcq single
+1 / 0.33
Person 'A' cannot step out of his house. He has to spend his entire life in sterile isolation otherwise, he would quickly contract a fatal infection. This person has almost no effective immune system. This disease is also called as baby in a bubble syndrome. Identify the disease, this person 'A' is suffering from
Q84.
mcq single
+1 / 0.33
Assertion Pork should be properly cooked to avoid Taenia infection.
Reason Pork of pig contains hexacanth and cysticercus larvae.
Q85.
mcq single
+1 / 0.33
Which of the following pathways occurs through cell wall?
Q86.
mcq single
+1 / 0.33
A mutant plant is unable to produce materials or precursors that form Casparian strip. This plant would be
Q87.
mcq single
+1 / 0.33
Cell A has osmotic pressure of $$-$$20 bars and pressure potential of 5 bars, whereas cell B has osmotic pressure of $$-$$18 bars and pressure potential of 2 bars.
The direction of flow of water will be
Q88.
mcq single
+1 / 0.33
**Assertion :** BOD is an indicator of pollution
in water.
**Reason :** High BOD is observed in highly
polluted water.
Q89.
mcq single
+1 / 0.33
Biomagnification can be defined as
Q90.
mcq single
+1 / 0.33
From the graph of population growth, select the correct option having correct value of '$$r$$' and bar graph.


Q91.
mcq single
+1 / 0.33
Match the stages of meiosis in column I to
their characteristic features in Column II and
select the correct option using the codes
given below.
Column I
Column II
A.
Diakinesis
1.
Crossing over takes place
B.
Pachytene
2.
Terminalisation of chiasmata
C.
Zygotene
3.
Chromosomes align at equatorial place
D.
Metaphase
4.
Pairing of homologous chromosomes
Q92.
mcq single
+1 / 0.33
Assertion In mitosis, two identical cells are
produced from a single cell and karyokinesis
is followed by cytokinesis.
Reason Cytokinesis is of two types, i.e. by
cell-furrow method and cell-plate method.
Q93.
mcq single
+1 / 0.33
Assertion Meiotic division occurs in
reproductive cells.
Reason Synapsis occurs during zygotene of
meiosis.
Q94.
mcq single
+1 / 0.33
Assertion In females, parturition is the act
of giving birth to a baby.
Reason Signals for parturition originate
from a fully developed foetus.
Q95.
mcq single
+1 / 0.33
Assertion Peptide and polypeptide
hormones directly pass across the lipid
bilayer of plasma membrane.
Reason Oxytocin hormone can pass across
the plasma membrane.
Q96.
mcq single
+1 / 0.33
Triticale is the first man-made cereal crop. Mention the type of hybridisation through which it was produced.
Q97.
mcq single
+1 / 0.33
A scion is grafted to a stock. The quality of
fruits produced will be determined by the
genotype of
Q98.
mcq single
+1 / 0.33
Consider the following four measures (I-IV) that could be taken to successfully grow chickpea in an area where bacterial blight is common.
I. Spray with Bordeaux mixture.
II. Control of the insect vector of the disease pathogen.
III. Use of disease-free seeds only.
IV. Use of varieties resistant to the disease.
Which of the above measures can control the diseases?
Q99.
mcq single
+1 / 0.33
Read the following statements
I. Species diversity decreases as we move away from the equator towards the poles.
II. Stellar's sea cow and passenger pigeon got extinct due to overexploitation by man.
III. Lantana and Eichhornia are invasive weed in India.
IV. The historic convention on biological diversity was held in 1992.
Choose the option containing correct statements.
Q100.
mcq single
+1 / 0.33
Apical dominance is caused by
Q101.
mcq single
+1 / 0.33
Assertion Photomodulation of flower is
phytochrome regulated process.
Reason Active form of phytochrome (Pfr)
directly induces floral induction in shoot
buds.
Q102.
mcq single
+1 / 0.33
**Assertion :** The quiescent centre acts as a
reservoir of relatively resistant cells, which
constitute a permanent source of active initials.
**Reason :** The cells of the inactive region of
quiescent centre become active, when the
previous active initials get damaged.
Q103.
mcq single
+1 / 0.33
Assertion Cytochrome oxidase enzyme
contains copper.
Reason Cyanide combines with copper of
cytochrome oxide and prevents oxygen to
combine with it.
Q104.
mcq single
+1 / 0.33
Assertion Two turns of Krebs’ cycle occur
per glucose molecule used.
Reason Each turn of Krebs’ cycle produces
3 NADH, 1FADH$$_2$$ and 1 ATP molecule.
Q105.
mcq single
+1 / 0.33
Match the following columns.
Column I
Column II
A.
Hinge
1.
Atlas and axis
B.
Saddle
2.
Frontal and parietal
C.
Pivot
3.
Carpal and metacarpal of humans
D.
Ball and socket
4.
Knee
5.
Humerus and pectoral girdle
Q106.
mcq single
+1 / 0.33
Which one of the following organisms do not evolve oxygen during photosynthesis?
Q107.
mcq single
+1 / 0.33
What is true about the isolated small tribal populations?
Q108.
mcq single
+1 / 0.33
The peppered moth (Biston betularia), the black-coloured form becomes dominant over the light-coloured form of moth in England during industrial revolution. This is an example of
Q109.
mcq single
+1 / 0.33
Choose the correct statements with reference to organic evolution.
I. Flippers of whale and wings of bat exhibit analogy.
II. Wings of butterfly and wings of bird exhibit homology.
III. Organs with dissimilar structure are called analogous organs.
IV. Organs with similar structure and origin are called homologous organs.
Q110.
mcq single
+1 / 0.33
Assertion In apomixis, the plants of new
genetic sequence are produced.
Reason In apomixis, two organisms of
same genetic sequence meet.
Q111.
mcq single
+1 / 0.33
Assertion Caryopsis fruits differ from
typical achenes with respect to the fusion of
pericarp with the seed-coat (testa).
Reason Caryopsis fruits commonly occur in
the members of family–Poaceae.
Q112.
mcq single
+1 / 0.33
A plant has a butterfly-shaped flower with
one standard, two wing like and two keel
petals. To which family, this plant belongs?
Q113.
mcq single
+1 / 0.33
Leaf tendrils are found in
Q114.
mcq single
+1 / 0.33
Arrange in correct order according to the
given figures.


Q115.
mcq single
+1 / 0.33
Which of the following biomolecules is common to respiration-mediated breakdown of fats, carbohydrates and proteins?
Q116.
mcq single
+1 / 0.33
If a recombinant DNA bearing gene for ampicillin resistance is transferred into E. coli cells and the host cells are spread on agar plates containing ampicillin, then
Q117.
mcq single
+1 / 0.33
Production of human protein in bacteria by genetic engineering is possible because
Q118.
mcq single
+1 / 0.33
Azolla is used as a biofertiliser because it
Q119.
mcq single
+1 / 0.33
Assertion Magnesium is important in photosynthesis and carbohydrate metabolism.
Reason $$\mathrm{Mg}^{2+}$$ is involved in the synthesis of nucleic acids.
Q120.
mcq single
+1 / 0.33
Assertion Nitrogen-fixing bacteria of
legume root nodules survive in oxygen
depleted cells.
Reason Leghaemoglobin completely
removes oxygen from nodule cells.