aiims-2019
AIIMS 2019
MEDICAL 2019 Previous Year
3 hDuration
180Total Marks
360Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 360 across 3 section(s); maximum marks: 180.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+1 / 0.33
$$\mathrm{H}_2 \mathrm{O}_2$$ is obtained by which of the following?
Q2.
mcq single
+1 / 0.33
Yellow colour of chlorine water fades because of
Q3.
mcq single
+1 / 0.33
**Assertion :** Out of $$\mathrm{CrO}_3$$ and $$\mathrm{Al}_2 \mathrm{O}_3, \mathrm{CrO}_3$$ having lower melting point than $$\mathrm{Al}_2 \mathrm{O}_3$$.
**Reason :** Oxidation state of $$\mathrm{Cr}$$ in $$\mathrm{CrO}_3$$ is high.
Q4.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{Yb}^{2+}$$ is more stable in compare to $$\mathrm{Gd}^{+2}$$.
**Reason :** The electronic configuration of GD is $$[\mathrm{Xe}] 4 f^7 5 d^2 6 s^2$$.
Q5.
mcq single
+1 / 0.33
**Assertion :** $$d^5$$ configuration is more stable than $$d^4$$.
**Reason :** $$d^5$$ has more exchange energy as compared to $$d^4$$ because 10 and 6 exchanges are possible in $$d^5$$ and $$d^4$$ respectively.
Q6.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{S}_2 \mathrm{O}_7^{2-}$$ and $$\mathrm{Cr}_2 \mathrm{O}_7^{2-}$$ both exist.
**Reason :** Both have same valence electrons.
Q7.
mcq single
+1 / 0.33
Which give coloured carbonate precipitate?
Q8.
mcq single
+1 / 0.33
Which element can have oxidation state
from 4 to 6?
Q9.
mcq single
+1 / 0.33
Match the following columns.
Polymer
Monomer
(i)
Buna-S
(P)
Styrene
(ii)
Ethylene glycol
(Q)
Terylene
(iii)
Elastomer
(R)
Chloroprene
Correct set of answer :
Q10.
mcq single
+1 / 0.33
**Assertion :** Nylon-6 is condensation polymer.
**Reason :** It is polymer of caprolactum.
Q11.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{BO}_3^{-3}$$ and $$\mathrm{SO}_3^{-2}$$ are not isostructural.
**Reason :** In $$\mathrm{SO}_3^{2-}$$ sulphur has one lone pair of electron.
Q12.
mcq single
+1 / 0.33
Which of the following, number of lone pair at central atom zero $$\mathrm{XeO}_3, \mathrm{XeO}_2 \mathrm{~F}_2, \mathrm{XeO}_4, \mathrm{XeO}_3 \mathrm{~F}_2, \mathrm{XeF}_6$$ ?
Q13.
mcq single
+1 / 0.33
Which is least soluble?
Q14.
mcq single
+1 / 0.33
$$\mathrm{pH}$$ of a salt solution of weak acid $$(\mathrm{p} K_a=4)$$ and weak base $$(\mathrm{p} K_b=5)$$ at $$25^{\circ}$$ is
Q15.
mcq single
+1 / 0.33
What is the activation energy $$(\mathrm{kJ} / \mathrm{mol})$$ for a reaction if its rate constant doubles when the temperature is raised from $$300 \mathrm{~K}$$ to $$400 \mathrm{~K}$$ of these $$(R=8.314 \mathrm{~Jmol}^{-1} \mathrm{~K}^{-1})$$
Q16.
mcq single
+1 / 0.33
Find out time period of Ist order reaction. When reaction complete $$2 / 3 \mathrm{rd}$$. If the value of rate constant is $$4.3 \times 10^{-4}$$
Q17.
mcq single
+1 / 0.33
Which of the following can react with $$\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7$$ ?
Q18.
mcq single
+1 / 0.33
**Assertion :** A spherical water drops become
flaton flatter surface.
**Reason :** It become flat due to gravity.
Q19.
mcq single
+1 / 0.33
Correct order for reaction with alcoholic KOH
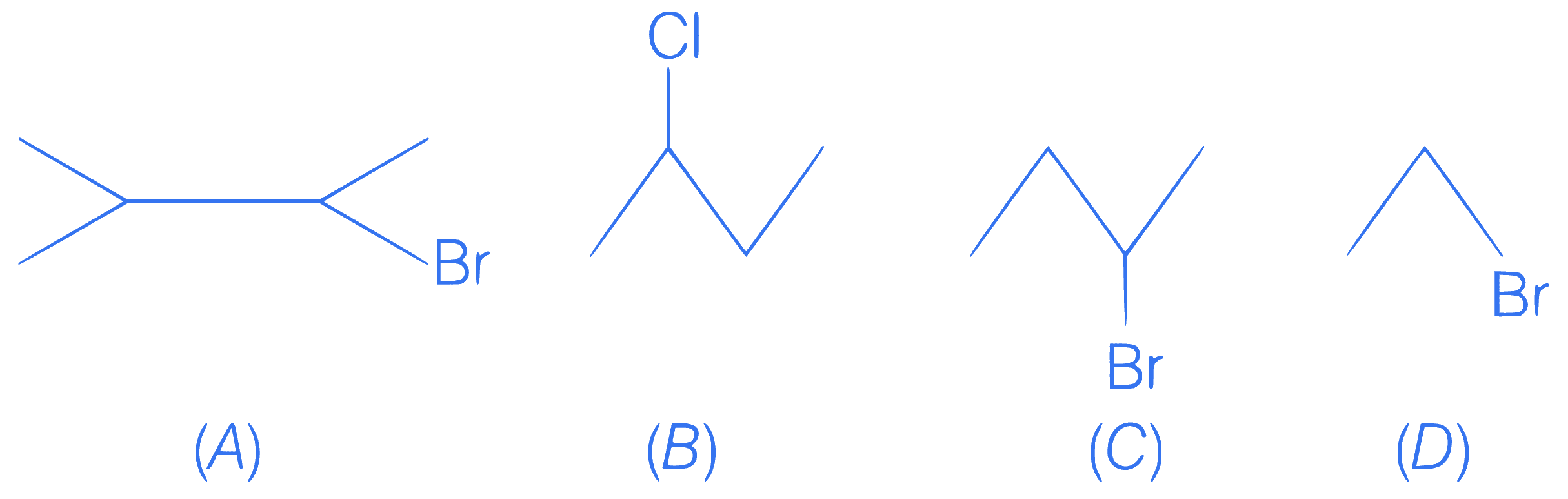

Q20.
mcq single
+1 / 0.33
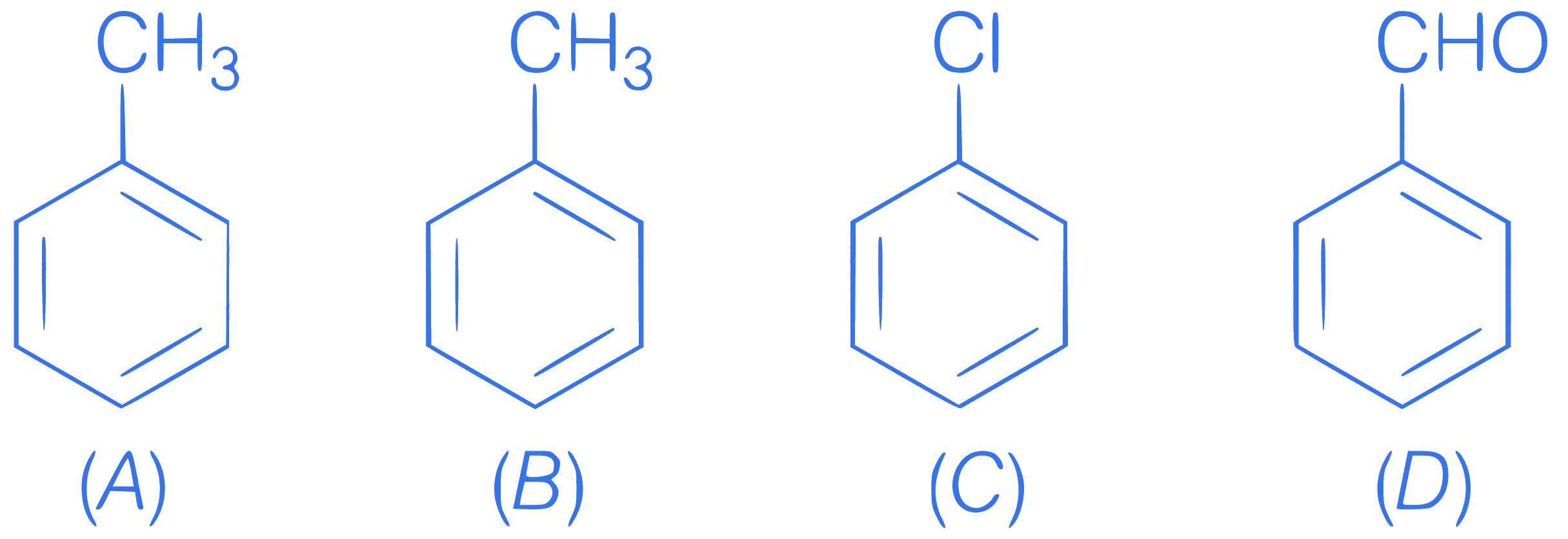
Correct order of electrophillic substitution
reaction is

Q21.
mcq single
+1 / 0.33
The vapour pressure of pure $$\mathrm{CHCl}_3$$ and $$\mathrm{CH}_2 \mathrm{Cl}_2$$ are 200 and $$41.5 \mathrm{~atm}$$ respectively. The weight of $$\mathrm{CHCl}_3$$ and $$\mathrm{CH}_2 \mathrm{Cl}_2$$ are respectively $$11.9 \mathrm{~g}$$ and $$17 \mathrm{~g}$$. The vapour pressure of solution will be
Q22.
mcq single
+1 / 0.33
If boiling point of water is $$100^{\circ} \mathrm{C}$$. How much gram of $$\mathrm{NaCl}$$ is added in $$500 \mathrm{~g}$$ of water to increase its boiling point of water by approx $$1^{\circ} \mathrm{C}\left(K_b\right)_{\mathrm{H}_2 \mathrm{O}}=0.52 \mathrm{~K} \times \mathrm{kg} /$$ mole.
Q23.
mcq single
+1 / 0.33
Find empirical formula of the compound if $$M=68 \%$$ (atomic mass $$=34$$ ) and remaining $$32 \%$$ oxygen.
Q24.
mcq single
+1 / 0.33
Correct order of bond dissociation energy
Q25.
mcq single
+1 / 0.33
Which pair of elements has maximum
electronegativity difference?
Q26.
mcq single
+1 / 0.33
Which has least covalent radius?
Q27.
mcq single
+1 / 0.33
Which have melting point below $$500^{\circ} \mathrm{C}$$ ?
Q28.
mcq single
+1 / 0.33
The correct relation is
Q29.
mcq single
+1 / 0.33
**Assertion :** $$U$$ is state function.
**Reason :** $$T$$ is an intensive property.
Q30.
mcq single
+1 / 0.33
At $$25^{\circ} \mathrm{C}, 1$$ mole of butane is heated then $$\mathrm{CO}_2$$ and $$\mathrm{H}_2 \mathrm{O}$$ liquid is formed work done is
Q31.
mcq single
+1 / 0.33
**Assertion :** The graph between $$p V$$ v/s $$\frac{1}{V}$$ is a straight line.
**Reason :** For adiabatic process, $$p \propto \frac{1}{V}$$
Q32.
mcq single
+1 / 0.33
Which of the following statement is correct
for oleum?
Q33.
mcq single
+1 / 0.33
Which of the following inert gas participate
in chemical reaction?
Q34.
mcq single
+1 / 0.33
$$\left[\mathrm{Co}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}$$ is a
Q35.
mcq single
+1 / 0.33
Which of the following complex is optically
inactive?
Q36.
mcq single
+1 / 0.33
Correct increasing order for the wavelength of absorption in the visible region for the complexes of $$\mathrm{Co}^{3+}$$ is
Q37.
mcq single
+1 / 0.33
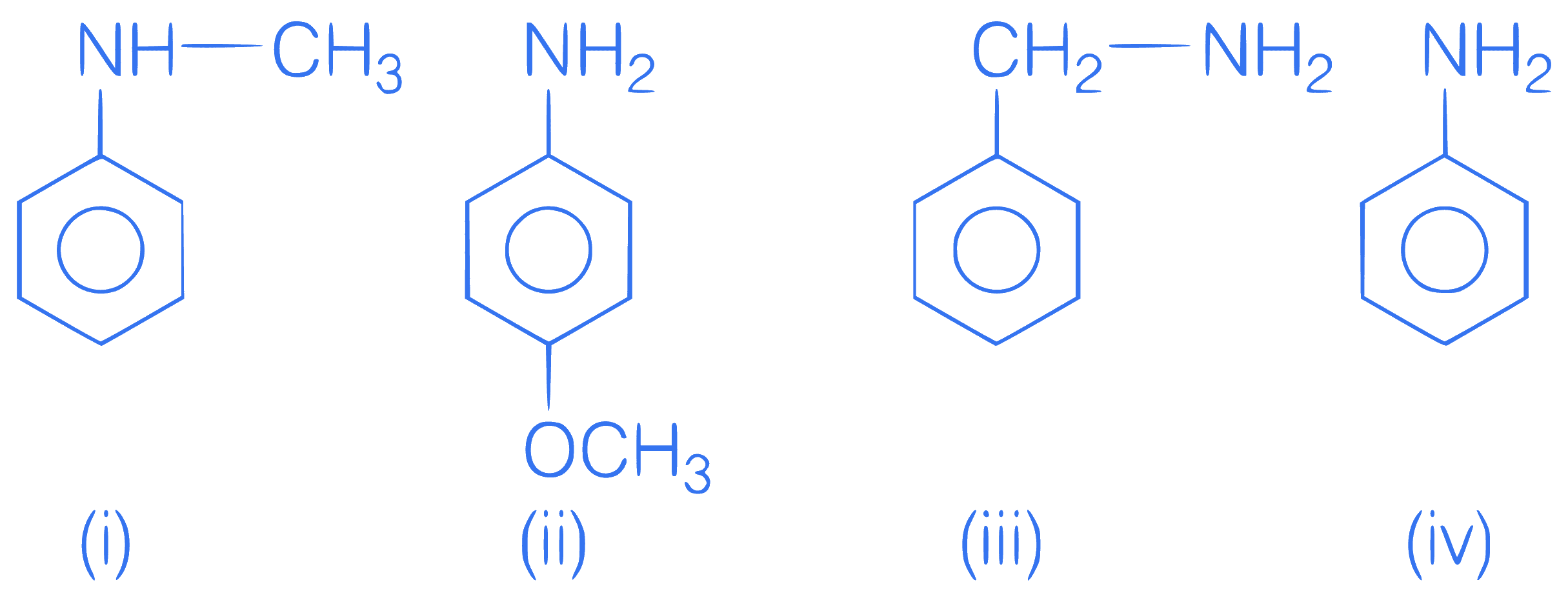 Correct order of basic strength is
Correct order of basic strength is
 Correct order of basic strength is
Correct order of basic strength is
Q38.
mcq single
+1 / 0.33
Which is most stable conformer of ethan-1,
2-diol?
Q39.
mcq single
+1 / 0.33
Compare stability of free radicals
(I) $$\mathrm{CH}_3-\mathop C\limits^ \bullet H-\mathrm{CH}_3$$
(II) 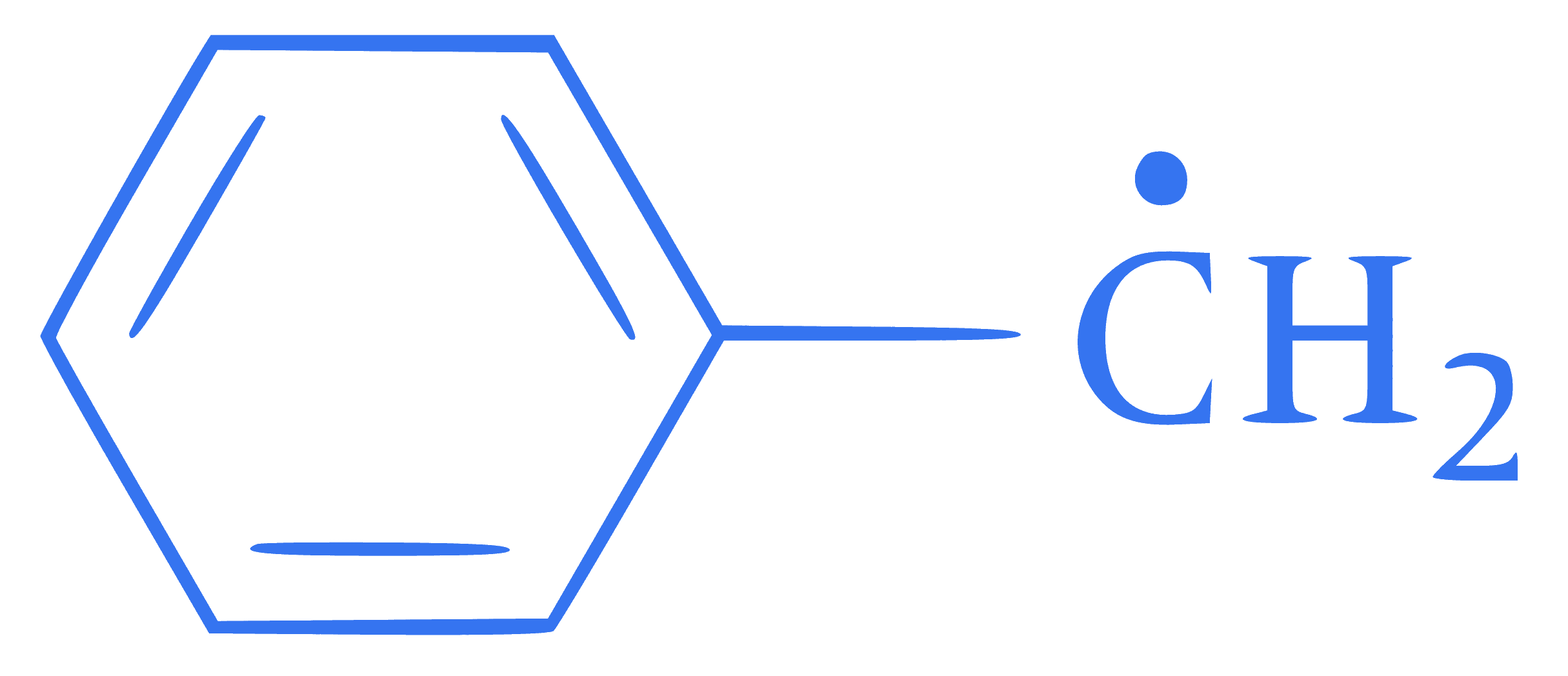 (III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
(III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
 (III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
(III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
Q40.
mcq single
+1 / 0.33
(i) $$\mathrm{F}_3 \mathrm{C}-\mathrm{COOH}$$, (ii) $$\mathrm{CH}_3 \mathrm{COOH}$$,
(iii) $$\mathrm{C}_6 \mathrm{H}_5 \mathrm{COOH}$$, (iv) $$\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{COOH}$$
Correct order of $$\mathrm{p} K_a$$ value is
Q41.
mcq single
+1 / 0.33
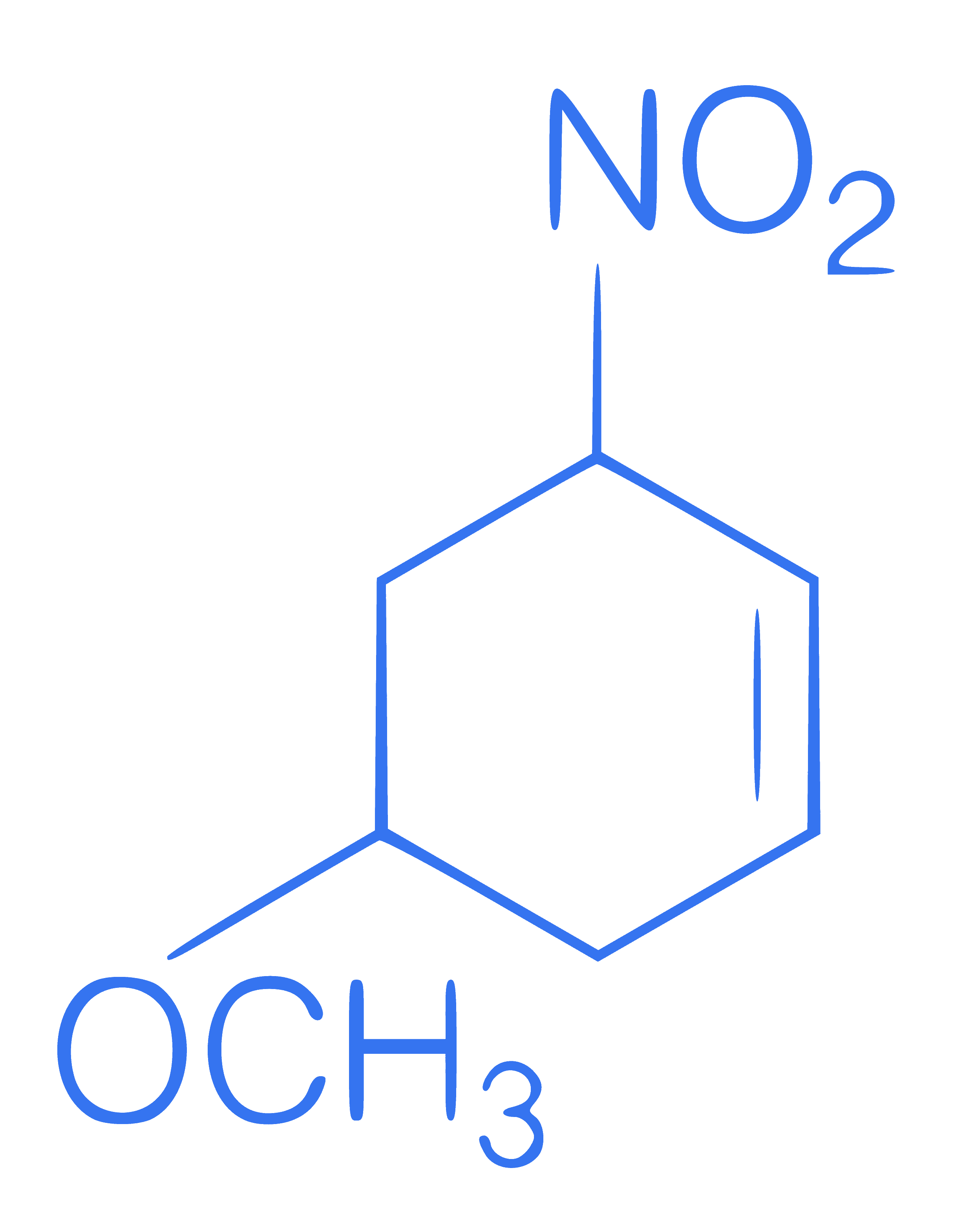 Write IUPAC name of following compound.
Write IUPAC name of following compound.
 Write IUPAC name of following compound.
Write IUPAC name of following compound.
Q42.
mcq single
+1 / 0.33
Order of acidic nature
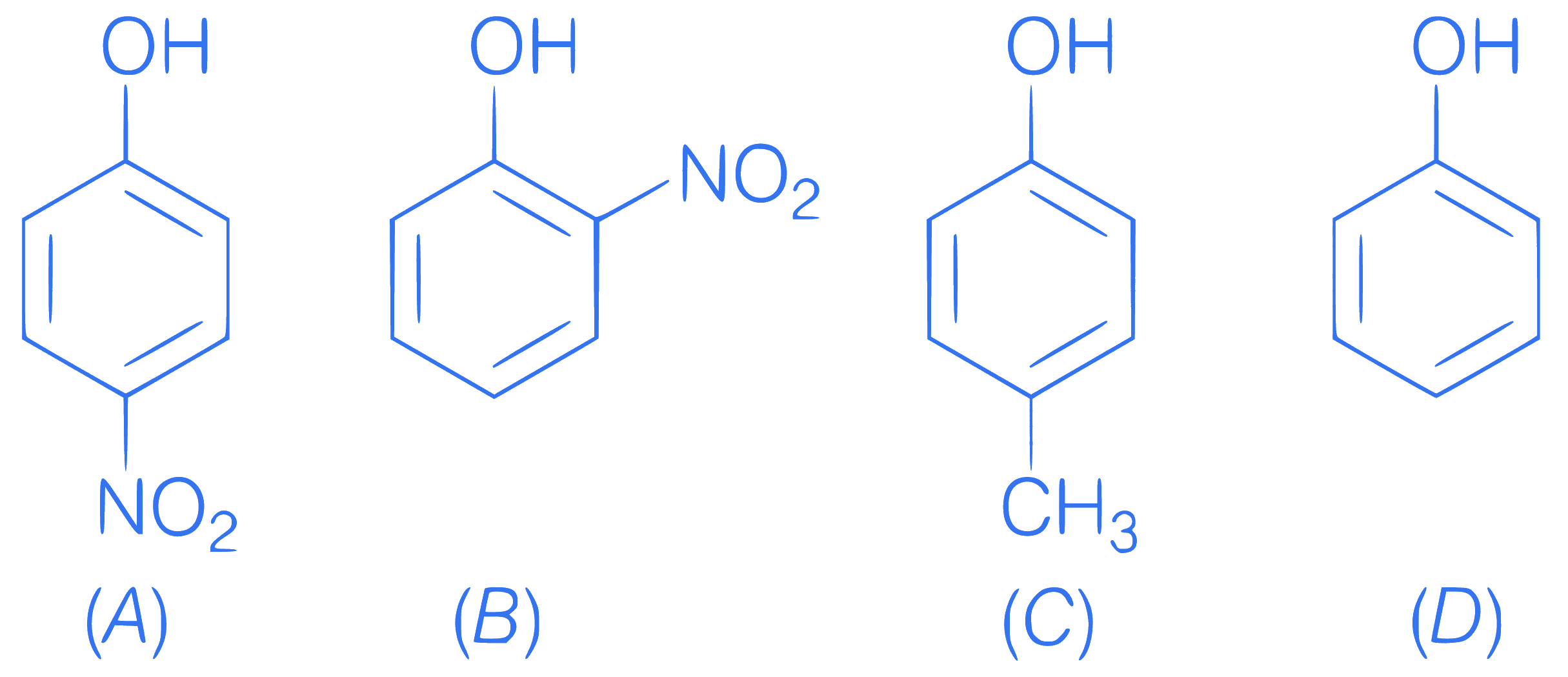

Q43.
mcq single
+1 / 0.33
**Assertion :** For liquid dishwashing
non-ionic type of detergent are used.
**Reason :** Remove grease and oil by micelle
formation.
Q44.
mcq single
+1 / 0.33
**Assertion :** Anhydrides are more reactive than ester for nucleophilic substitution.
**Reason :** $$RCOO -$$ is better leaving group than $$\mathrm{R}-\mathrm{O}^{-}$$.
Q45.
mcq single
+1 / 0.33
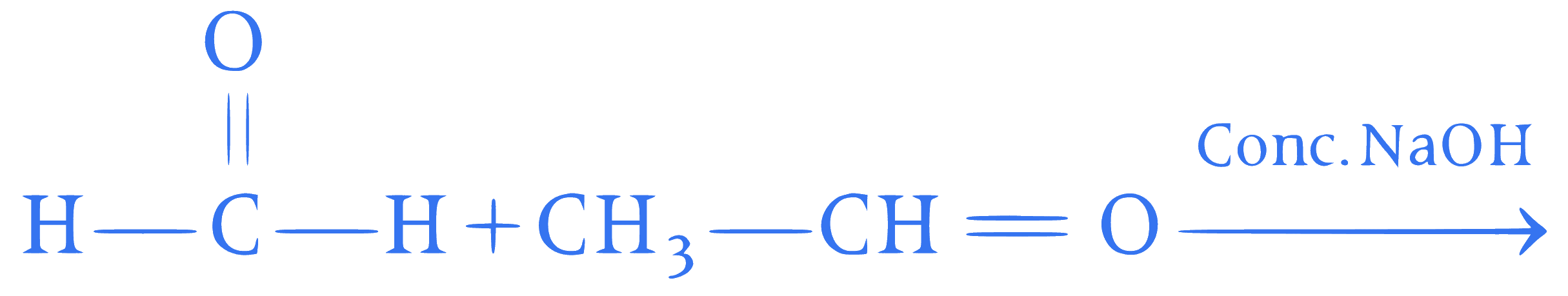 Find out the products of reaction
Find out the products of reaction
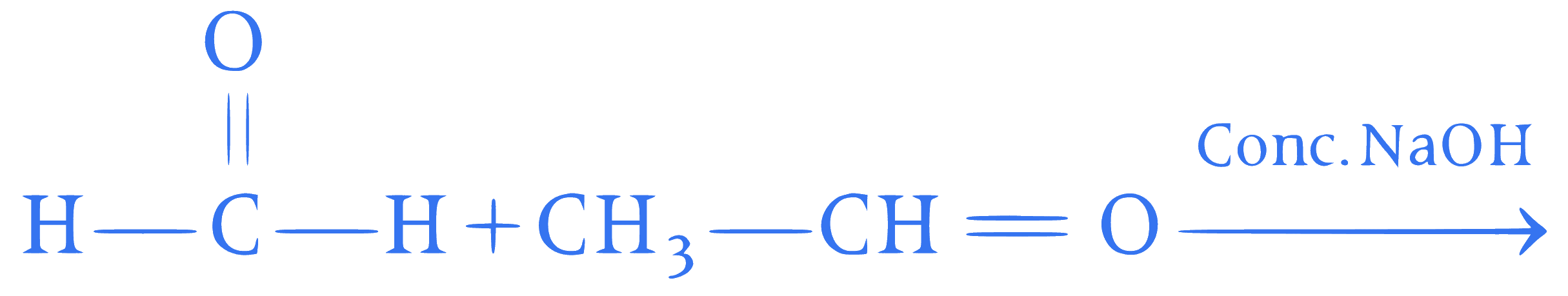 Find out the products of reaction
Find out the products of reaction
Q46.
mcq single
+1 / 0.33
Out of $$\mathrm{BeF}_2, \mathrm{MgF}_2, \mathrm{CaF}_2, \mathrm{SrF}_2$$ which has maximum solubility?
Q47.
mcq single
+1 / 0.33
Which of the following has maximum iron
content?
Q48.
mcq single
+1 / 0.33
**Assertion :** Phenol reacts with $$\mathrm{CH}_3 \mathrm{I}$$ in presence of $$\mathrm{NaOH}$$ to form methoxybenzene.
**Reason :** Phenoxide is better nucleophile than phenol.
Q49.
mcq single
+1 / 0.33
**Assertion :** Tert-butyl methyl ether react with $$\mathrm{HBr}$$ to form tert butyl, $$\left(\mathrm{CH}_3\right)_3 \mathrm{C}-\mathrm{Br}$$ and $$\mathrm{CH}_3-\mathrm{OH}$$, methanal.
**Reason :** It follows $$\mathrm{S}_{\mathrm{N}} 1$$ mechanism.
Q50.
mcq single
+1 / 0.33
**Assertion :** Propene reacts with $$\mathrm{HI}$$ in presence of peroxide give 1-iodopropane.
**Reason :** $$1^{\circ}$$ free radical is less stable than $$2^{\circ}$$ free radical.
Q51.
mcq single
+1 / 0.33
**Assertion :** Two sugar units joined by 1, 2-glycosidic bond in sucrose.
**Reason :** It contains $$\mathrm{C}_1$$-glucose and $$\mathrm{C}_2$$-fructose glycosidic bond.
Q52.
mcq single
+1 / 0.33
**Assertion :** Tryptophan is an example of
non-essential amino acids.
**Reason :** The amino acids that are not
synthesised in human body are
non-essential amino acids.
Q53.
mcq single
+1 / 0.33
**Assertion :** Glucose does not gives 2, 4- DNP
test.
**Reason :** Glucose exists in cyclic hemiacetal
form.
Q54.
mcq single
+1 / 0.33
The tetrahedral voids are present in 0.5 mole of hcp crystal structure
Q55.
mcq single
+1 / 0.33
**Assertion :** ZnO becomes yellow when it is
heated.
**Reason :** The anionic sites occupied by
unpaired electrons (due to F-centres).
Q56.
mcq single
+1 / 0.33
Which of the following is correct order of
packing efficiency?
Q57.
mcq single
+1 / 0.33
Cell notation, $$\begin{gathered}
M\left|M^{2+} \| M^{2+}\right| M. \\
0.01 \quad 0.0001
\end{gathered}$$ If value of $$E^{\circ}{ }_{\text {cell }} \text { is } 4 \text { volt ( Given } \frac{R T}{F} \text { in } 10=0.06 \text { ) }$$
Q58.
mcq single
+1 / 0.33
**Assertion :** Tert-butyl amine can be formed by Gabriel phthalimide synthesis.
**Reason :** It follow $$\mathrm{S}_{\mathrm{N}} 1$$ mechanism.
Q59.
mcq single
+1 / 0.33
**Assertion :** The chemical properties of
different isotope are same.
**Reason :** Isotopes having same number of
neutron.
Q60.
mcq single
+1 / 0.33
A bulb is emitted electromagnetic radiation of $$660 \mathrm{~nm}$$ wavelength. The total energy of radiation is $$3 \times 10^{-18} \mathrm{~J}$$. The number of emitted photon will be
$$\left(h=6.6 \times 10^{-34}\right) \mathrm{J} \times \mathrm{s}, c=3 \times 10^8 \mathrm{~m} / \mathrm{s}$$
Q61.
mcq single
+1 / 0.33
$$\mathrm{H}_2 \mathrm{O}_2$$ is obtained by which of the following?
Q62.
mcq single
+1 / 0.33
Yellow colour of chlorine water fades because of
Q63.
mcq single
+1 / 0.33
**Assertion :** Out of $$\mathrm{CrO}_3$$ and $$\mathrm{Al}_2 \mathrm{O}_3, \mathrm{CrO}_3$$ having lower melting point than $$\mathrm{Al}_2 \mathrm{O}_3$$.
**Reason :** Oxidation state of $$\mathrm{Cr}$$ in $$\mathrm{CrO}_3$$ is high.
Q64.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{Yb}^{2+}$$ is more stable in compare to $$\mathrm{Gd}^{+2}$$.
**Reason :** The electronic configuration of GD is $$[\mathrm{Xe}] 4 f^7 5 d^2 6 s^2$$.
Q65.
mcq single
+1 / 0.33
**Assertion :** $$d^5$$ configuration is more stable than $$d^4$$.
**Reason :** $$d^5$$ has more exchange energy as compared to $$d^4$$ because 10 and 6 exchanges are possible in $$d^5$$ and $$d^4$$ respectively.
Q66.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{S}_2 \mathrm{O}_7^{2-}$$ and $$\mathrm{Cr}_2 \mathrm{O}_7^{2-}$$ both exist.
**Reason :** Both have same valence electrons.
Q67.
mcq single
+1 / 0.33
Which give coloured carbonate precipitate?
Q68.
mcq single
+1 / 0.33
Which element can have oxidation state
from 4 to 6?
Q69.
mcq single
+1 / 0.33
Match the following columns.
Polymer
Monomer
(i)
Buna-S
(P)
Styrene
(ii)
Ethylene glycol
(Q)
Terylene
(iii)
Elastomer
(R)
Chloroprene
Correct set of answer :
Q70.
mcq single
+1 / 0.33
**Assertion :** Nylon-6 is condensation polymer.
**Reason :** It is polymer of caprolactum.
Q71.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{BO}_3^{-3}$$ and $$\mathrm{SO}_3^{-2}$$ are not isostructural.
**Reason :** In $$\mathrm{SO}_3^{2-}$$ sulphur has one lone pair of electron.
Q72.
mcq single
+1 / 0.33
Which of the following, number of lone pair at central atom zero $$\mathrm{XeO}_3, \mathrm{XeO}_2 \mathrm{~F}_2, \mathrm{XeO}_4, \mathrm{XeO}_3 \mathrm{~F}_2, \mathrm{XeF}_6$$ ?
Q73.
mcq single
+1 / 0.33
Which is least soluble?
Q74.
mcq single
+1 / 0.33
$$\mathrm{pH}$$ of a salt solution of weak acid $$(\mathrm{p} K_a=4)$$ and weak base $$(\mathrm{p} K_b=5)$$ at $$25^{\circ}$$ is
Q75.
mcq single
+1 / 0.33
What is the activation energy $$(\mathrm{kJ} / \mathrm{mol})$$ for a reaction if its rate constant doubles when the temperature is raised from $$300 \mathrm{~K}$$ to $$400 \mathrm{~K}$$ of these $$(R=8.314 \mathrm{~Jmol}^{-1} \mathrm{~K}^{-1})$$
Q76.
mcq single
+1 / 0.33
Find out time period of Ist order reaction. When reaction complete $$2 / 3 \mathrm{rd}$$. If the value of rate constant is $$4.3 \times 10^{-4}$$
Q77.
mcq single
+1 / 0.33
Which of the following can react with $$\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7$$ ?
Q78.
mcq single
+1 / 0.33
**Assertion :** A spherical water drops become
flaton flatter surface.
**Reason :** It become flat due to gravity.
Q79.
mcq single
+1 / 0.33
Correct order for reaction with alcoholic KOH


Q80.
mcq single
+1 / 0.33
Correct order of electrophillic substitution
reaction is


Q81.
mcq single
+1 / 0.33
The vapour pressure of pure $$\mathrm{CHCl}_3$$ and $$\mathrm{CH}_2 \mathrm{Cl}_2$$ are 200 and $$41.5 \mathrm{~atm}$$ respectively. The weight of $$\mathrm{CHCl}_3$$ and $$\mathrm{CH}_2 \mathrm{Cl}_2$$ are respectively $$11.9 \mathrm{~g}$$ and $$17 \mathrm{~g}$$. The vapour pressure of solution will be
Q82.
mcq single
+1 / 0.33
If boiling point of water is $$100^{\circ} \mathrm{C}$$. How much gram of $$\mathrm{NaCl}$$ is added in $$500 \mathrm{~g}$$ of water to increase its boiling point of water by approx $$1^{\circ} \mathrm{C}\left(K_b\right)_{\mathrm{H}_2 \mathrm{O}}=0.52 \mathrm{~K} \times \mathrm{kg} /$$ mole.
Q83.
mcq single
+1 / 0.33
Find empirical formula of the compound if $$M=68 \%$$ (atomic mass $$=34$$ ) and remaining $$32 \%$$ oxygen.
Q84.
mcq single
+1 / 0.33
Correct order of bond dissociation energy
Q85.
mcq single
+1 / 0.33
Which pair of elements has maximum
electronegativity difference?
Q86.
mcq single
+1 / 0.33
Which has least covalent radius?
Q87.
mcq single
+1 / 0.33
Which have melting point below $$500^{\circ} \mathrm{C}$$ ?
Q88.
mcq single
+1 / 0.33
The correct relation is
Q89.
mcq single
+1 / 0.33
**Assertion :** $$U$$ is state function.
**Reason :** $$T$$ is an intensive property.
Q90.
mcq single
+1 / 0.33
At $$25^{\circ} \mathrm{C}, 1$$ mole of butane is heated then $$\mathrm{CO}_2$$ and $$\mathrm{H}_2 \mathrm{O}$$ liquid is formed work done is
Q91.
mcq single
+1 / 0.33
**Assertion :** The graph between $$p V$$ v/s $$\frac{1}{V}$$ is a straight line.
**Reason :** For adiabatic process, $$p \propto \frac{1}{V}$$
Q92.
mcq single
+1 / 0.33
Which of the following statement is correct
for oleum?
Q93.
mcq single
+1 / 0.33
Which of the following inert gas participate
in chemical reaction?
Q94.
mcq single
+1 / 0.33
$$\left[\mathrm{Co}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}$$ is a
Q95.
mcq single
+1 / 0.33
Which of the following complex is optically
inactive?
Q96.
mcq single
+1 / 0.33
Correct increasing order for the wavelength of absorption in the visible region for the complexes of $$\mathrm{Co}^{3+}$$ is
Q97.
mcq single
+1 / 0.33
 Correct order of basic strength is
Correct order of basic strength is
 Correct order of basic strength is
Correct order of basic strength is
Q98.
mcq single
+1 / 0.33
Which is most stable conformer of ethan-1,
2-diol?
Q99.
mcq single
+1 / 0.33
Compare stability of free radicals
(I) $$\mathrm{CH}_3-\mathop C\limits^ \bullet H-\mathrm{CH}_3$$
(II)  (III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
(III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
 (III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
(III) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2$$
(IV) $$\mathop C\limits^ \bullet {H_2}-\mathrm{CH}_3$$
Q100.
mcq single
+1 / 0.33
(i) $$\mathrm{F}_3 \mathrm{C}-\mathrm{COOH}$$, (ii) $$\mathrm{CH}_3 \mathrm{COOH}$$,
(iii) $$\mathrm{C}_6 \mathrm{H}_5 \mathrm{COOH}$$, (iv) $$\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{COOH}$$
Correct order of $$\mathrm{p} K_a$$ value is
Q101.
mcq single
+1 / 0.33
 Write IUPAC name of following compound.
Write IUPAC name of following compound.
 Write IUPAC name of following compound.
Write IUPAC name of following compound.
Q102.
mcq single
+1 / 0.33
Order of acidic nature


Q103.
mcq single
+1 / 0.33
**Assertion :** For liquid dishwashing
non-ionic type of detergent are used.
**Reason :** Remove grease and oil by micelle
formation.
Q104.
mcq single
+1 / 0.33
**Assertion :** Anhydrides are more reactive than ester for nucleophilic substitution.
**Reason :** $$RCOO -$$ is better leaving group than $$\mathrm{R}-\mathrm{O}^{-}$$.
Q105.
mcq single
+1 / 0.33
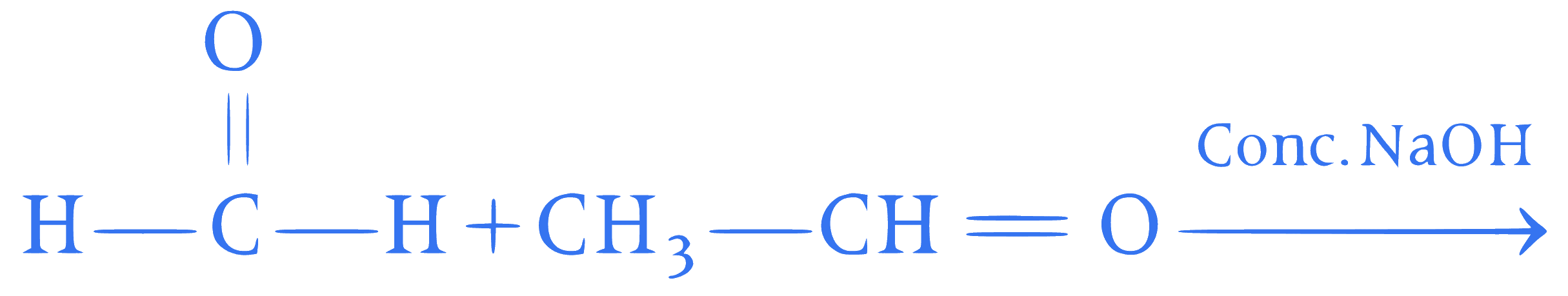 Find out the products of reaction
Find out the products of reaction
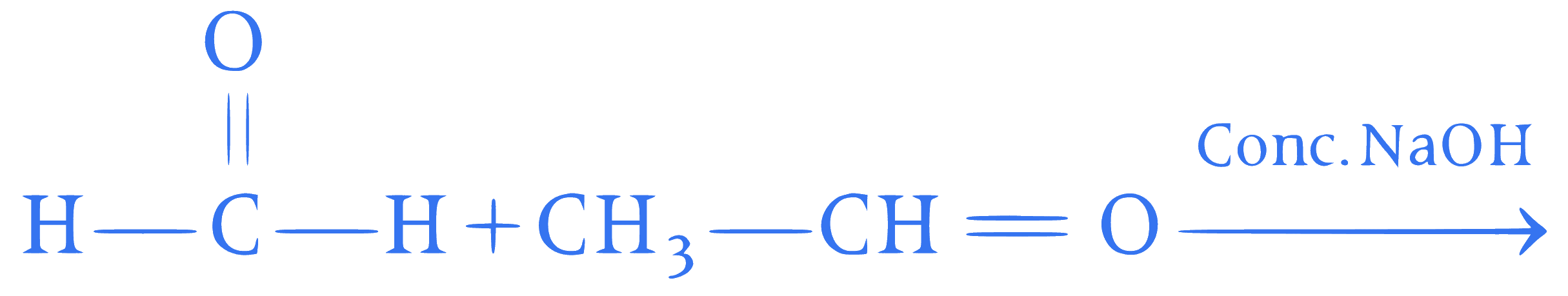 Find out the products of reaction
Find out the products of reaction
Q106.
mcq single
+1 / 0.33
Out of $$\mathrm{BeF}_2, \mathrm{MgF}_2, \mathrm{CaF}_2, \mathrm{SrF}_2$$ which has maximum solubility?
Q107.
mcq single
+1 / 0.33
Which of the following has maximum iron
content?
Q108.
mcq single
+1 / 0.33
**Assertion :** Phenol reacts with $$\mathrm{CH}_3 \mathrm{I}$$ in presence of $$\mathrm{NaOH}$$ to form methoxybenzene.
**Reason :** Phenoxide is better nucleophile than phenol.
Q109.
mcq single
+1 / 0.33
**Assertion :** Tert-butyl methyl ether react with $$\mathrm{HBr}$$ to form tert butyl, $$\left(\mathrm{CH}_3\right)_3 \mathrm{C}-\mathrm{Br}$$ and $$\mathrm{CH}_3-\mathrm{OH}$$, methanal.
**Reason :** It follows $$\mathrm{S}_{\mathrm{N}} 1$$ mechanism.
Q110.
mcq single
+1 / 0.33
**Assertion :** Propene reacts with $$\mathrm{HI}$$ in presence of peroxide give 1-iodopropane.
**Reason :** $$1^{\circ}$$ free radical is less stable than $$2^{\circ}$$ free radical.
Q111.
mcq single
+1 / 0.33
**Assertion :** Two sugar units joined by 1, 2-glycosidic bond in sucrose.
**Reason :** It contains $$\mathrm{C}_1$$-glucose and $$\mathrm{C}_2$$-fructose glycosidic bond.
Q112.
mcq single
+1 / 0.33
**Assertion :** Tryptophan is an example of
non-essential amino acids.
**Reason :** The amino acids that are not
synthesised in human body are
non-essential amino acids.
Q113.
mcq single
+1 / 0.33
**Assertion :** Glucose does not gives 2, 4- DNP
test.
**Reason :** Glucose exists in cyclic hemiacetal
form.
Q114.
mcq single
+1 / 0.33
The tetrahedral voids are present in 0.5 mole of hcp crystal structure
Q115.
mcq single
+1 / 0.33
**Assertion :** ZnO becomes yellow when it is
heated.
**Reason :** The anionic sites occupied by
unpaired electrons (due to F-centres).
Q116.
mcq single
+1 / 0.33
Which of the following is correct order of
packing efficiency?
Q117.
mcq single
+1 / 0.33
Cell notation, $$\begin{gathered}
M\left|M^{2+} \| M^{2+}\right| M. \\
0.01 \quad 0.0001
\end{gathered}$$ If value of $$E^{\circ}{ }_{\text {cell }} \text { is } 4 \text { volt ( Given } \frac{R T}{F} \text { in } 10=0.06 \text { ) }$$
Q118.
mcq single
+1 / 0.33
**Assertion :** Tert-butyl amine can be formed by Gabriel phthalimide synthesis.
**Reason :** It follow $$\mathrm{S}_{\mathrm{N}} 1$$ mechanism.
Q119.
mcq single
+1 / 0.33
**Assertion :** The chemical properties of
different isotope are same.
**Reason :** Isotopes having same number of
neutron.
Q120.
mcq single
+1 / 0.33
A bulb is emitted electromagnetic radiation of $$660 \mathrm{~nm}$$ wavelength. The total energy of radiation is $$3 \times 10^{-18} \mathrm{~J}$$. The number of emitted photon will be
$$\left(h=6.6 \times 10^{-34}\right) \mathrm{J} \times \mathrm{s}, c=3 \times 10^8 \mathrm{~m} / \mathrm{s}$$
Physics
Physics
Q1.
mcq single
+1 / 0.33
**Assertion :** Distance between position of
bright and dark fringe remain same in YDSE.
**Reason :** Fringe width, $$\beta=\frac{\lambda D}{d}$$
Q2.
mcq single
+1 / 0.33
**Assertion :** Incoming light reflected by earth
is partially polarised.
**Reason :** Atmospheric particle polarise the
light.
Q3.
mcq single
+1 / 0.33
Distance of 5th dark fringe from centre is $$4 \mathrm{~mm}$$. If $$D=2 \mathrm{~m}, \lambda=600 \mathrm{~nm}$$, then distance between slits is
Q4.
mcq single
+1 / 0.33
A light of wavelength $$500 \mathrm{~nm}$$ is incident on a Young's double slit. The distance between slit and screen is $$D=1.8 \mathrm{~m}$$ and distance between slits is $$d=0.4 \mathrm{~mm}$$. If screen moves with a speed of $$4 \mathrm{~m} / \mathrm{s}$$, then with what speed first maxima will move?
Q5.
mcq single
+1 / 0.33
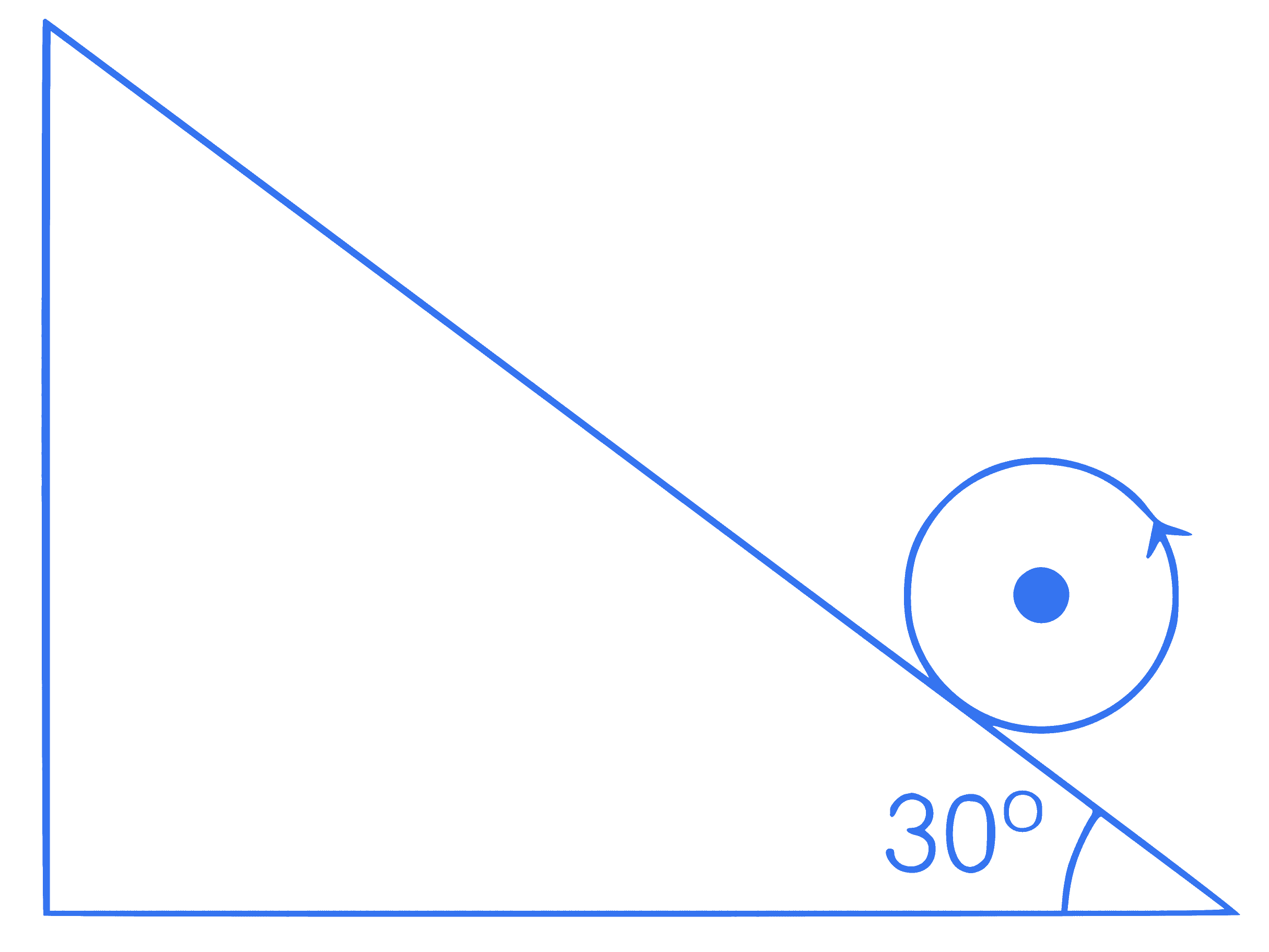
A sphere pure rolls on a rough inclined
plane with initial velocity 2.8 m/s. Find the
maximum distance on the inclined plane.

Q6.
mcq single
+1 / 0.33
**Assertion :** A metallic surface is moved in
and out in magnetic field then emf is
induced in it.
**Reason :** Eddy current will be produced in a
metallic surface moving in and out of
magnetic field.
Q7.
mcq single
+1 / 0.33
An electron is moving in a circle of radius
2m with speed of 4 m/s. Find the
acceleration of the electron.
Q8.
mcq single
+1 / 0.33
Calculate the mean percentage error in five
observations,
80.0, 80.5, 81.0, 81.5, 82
Q9.
mcq single
+1 / 0.33
**Assertion :** Vibrational degree of freedom of
a di-atomic gas molecule appears at every
high temperature.
**Reason :** Di-atomic gas has two vibrational
degree of freedom in one direction.
Q10.
mcq single
+1 / 0.33
If $$7 \mathrm{~gm} \mathrm{~N}_2$$ is mixed with $$20 \mathrm{~gm} \mathrm{~Ar}$$, there $$\frac{C_p}{C_V}$$ of mixture will be
Q11.
mcq single
+1 / 0.33
Calculate radiation power for sphere whose temperature is 227$$^\circ$$C, radius 2 m and emissivity 0.8.
Q12.
mcq single
+1 / 0.33
In an isobaric process, the work done by a
di-atomic gas is 10 J, the heat given to the
gas will be
Q13.
mcq single
+1 / 0.33
**Assertion :** In adiabatic process work is
independent of the path.
**Reason : **In adiabatic process work done is
equal to negative of change in internal
energy.
Q14.
mcq single
+1 / 0.33
The temperature of food material in
refrigerator is 4$$^\circ$$C and temperature of
environment is 15$$^\circ$$C. If carnot cycle is used
in its working gas, then find its carnot
efficiency.
Q15.
mcq single
+1 / 0.33
An ideal gas initially at pressure 1 bar is being compressed from $$30 \mathrm{~m}^3$$ to $$10 \mathrm{~m}^3$$ volume and its temperature decreases from $$320 \mathrm{~K}$$ to $$280 \mathrm{~K}$$, then find final pressure of the gas.
Q16.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{NH}_3$$ is liquidities more easily than $$\mathrm{CO}_2$$.
**Reason :** Critical temperature of $$\mathrm{NH}_3$$ is more than $$\mathrm{CO}_2$$.
Q17.
mcq single
+1 / 0.33
In an isothermal process 2 water drops of radius $$1 \mathrm{~mm}$$ are combined to form a bigger drop. Find the energy change in this process if $$T=0.1 \mathrm{~N} / \mathrm{m}$$.
Q18.
mcq single
+1 / 0.33
**Assertion :** Sometimes insects can walk on
water.
**Reason :** The gravitational force on insect is
balanced by force due surface tension.
Q19.
mcq single
+1 / 0.33
**Assertion :** Water drops take spherical shape
when falling freely.
**Reason :** Water has minimum surface
tension among all liquids.
Q20.
mcq single
+1 / 0.33
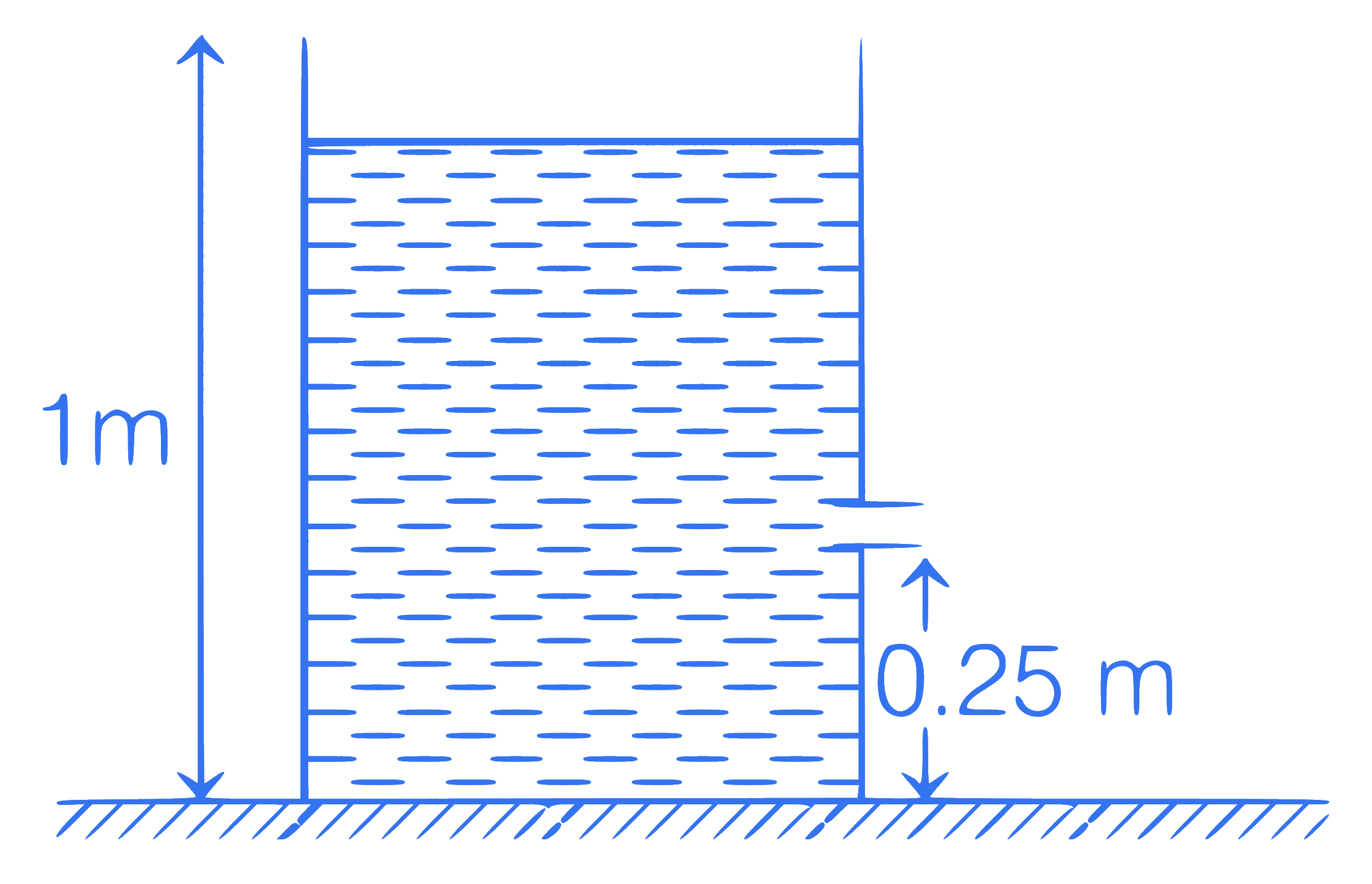
If a small orifice is made at a height of
0.25 m from the ground, the horizontal
range of water stream will be

Q21.
mcq single
+1 / 0.33
Determine the pressure difference in tube of
non-uniform cross sectional area as shown
in figure.
$$\Delta P=?, d_1=5 \mathrm{~cm}, v_1=4 \mathrm{~m} / \mathrm{s}, d_2=2 \mathrm{~cm}, v_2=?$$
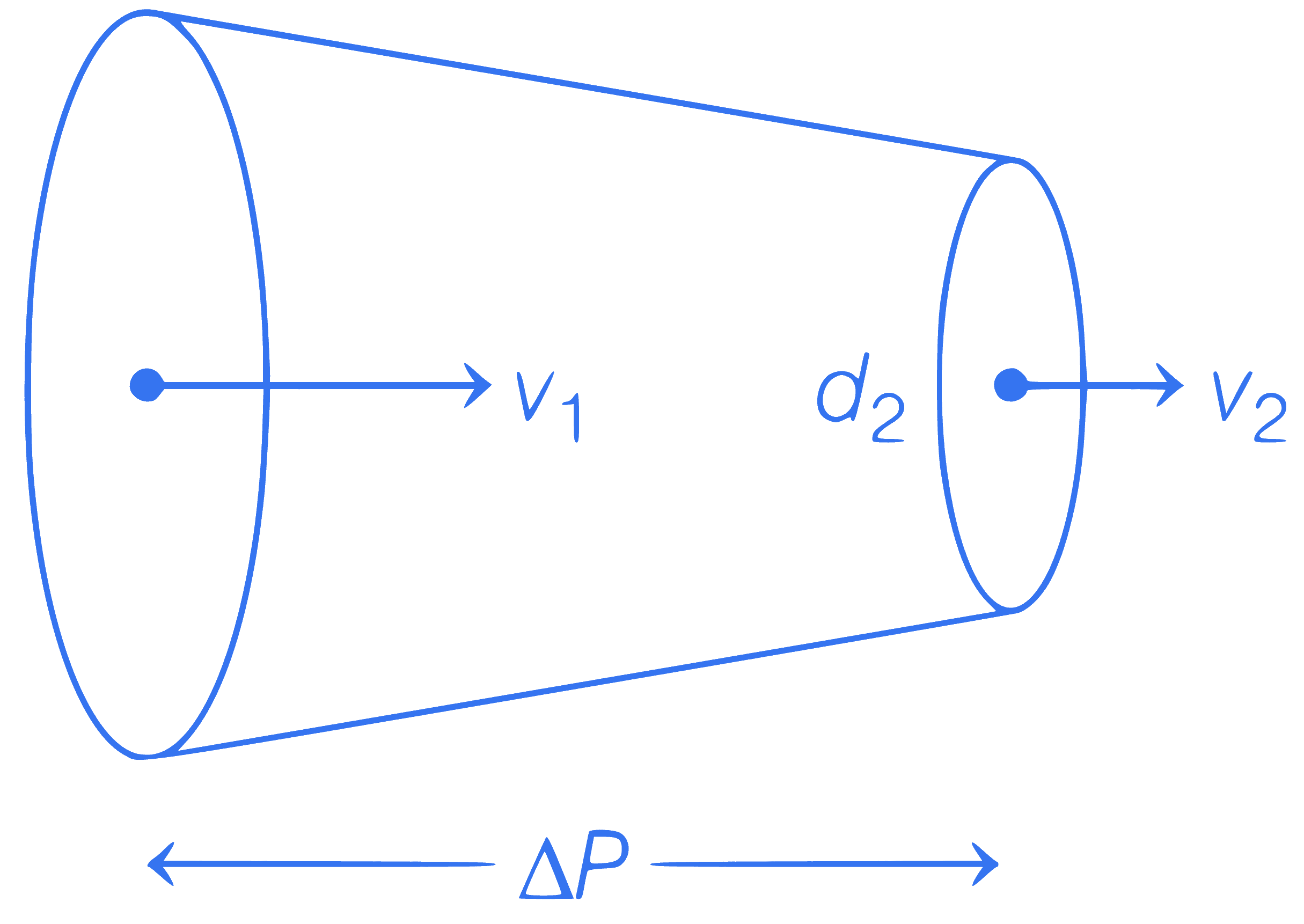

Q22.
mcq single
+1 / 0.33
Find gravitational field at a distance of $$2000 \mathrm{~km}$$ from the centre of earth. (Given
$$R_{\text {earth }}=6400 \mathrm{~km}, r=2000 \mathrm{~km} \text {, } M_{\text {earth }}=6 \times 10^{24} \mathrm{~kg} \text { ) }$$
Q23.
mcq single
+1 / 0.33
A capacitor of capacitance $$15 \mu \mathrm{F}$$ having dielectric slab of $$\varepsilon_r=2.5$$, dielectric strength $$30 \mathrm{~MV} / \mathrm{m}$$ and potential difference $$=30 \mathrm{~V}$$. Calculate the area of the plate.
Q24.
mcq single
+1 / 0.33
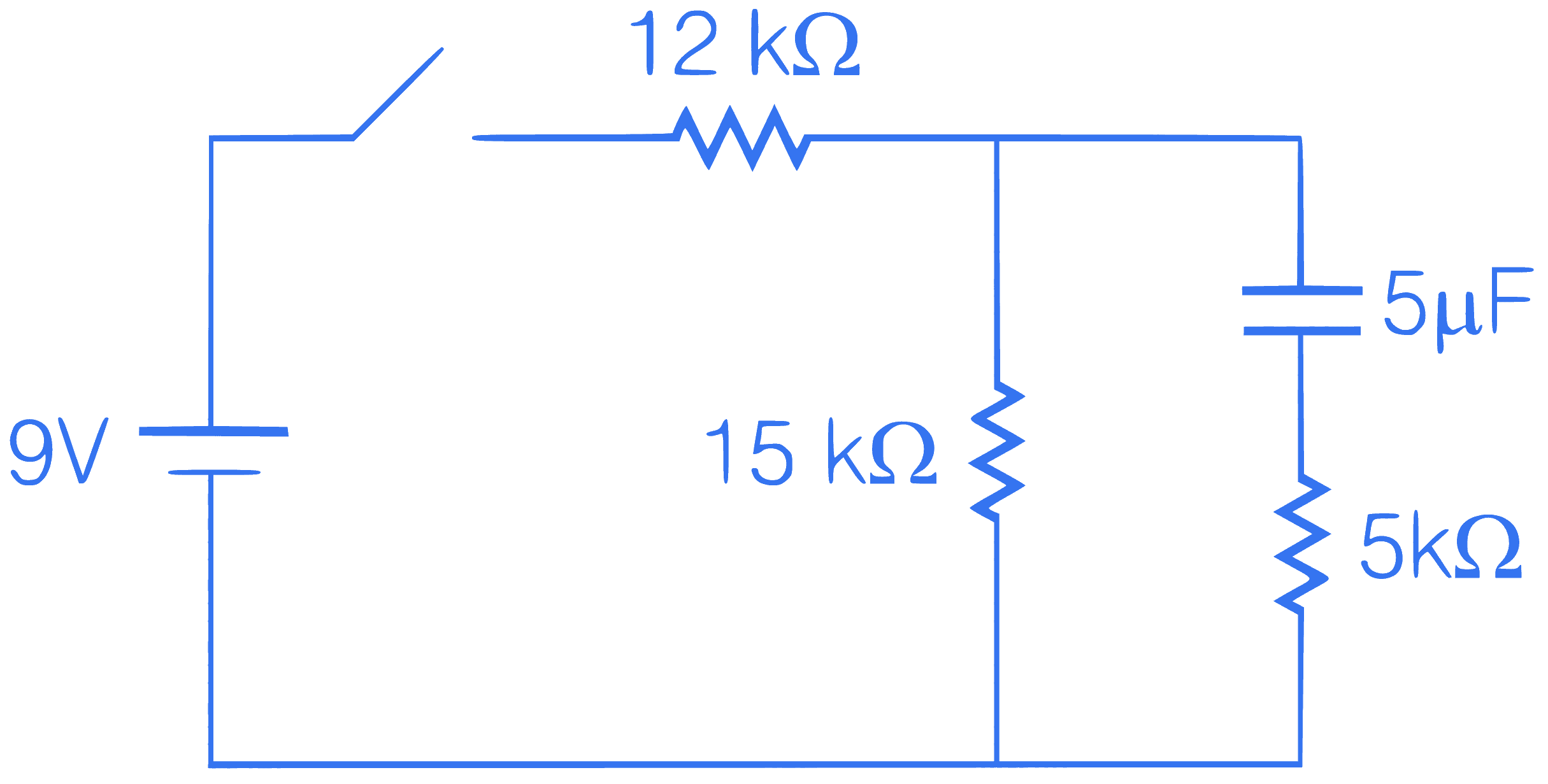 In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
 In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
Q25.
mcq single
+1 / 0.33
Calculate the focal length of given lens if the
magnification is $$-$$0 5.
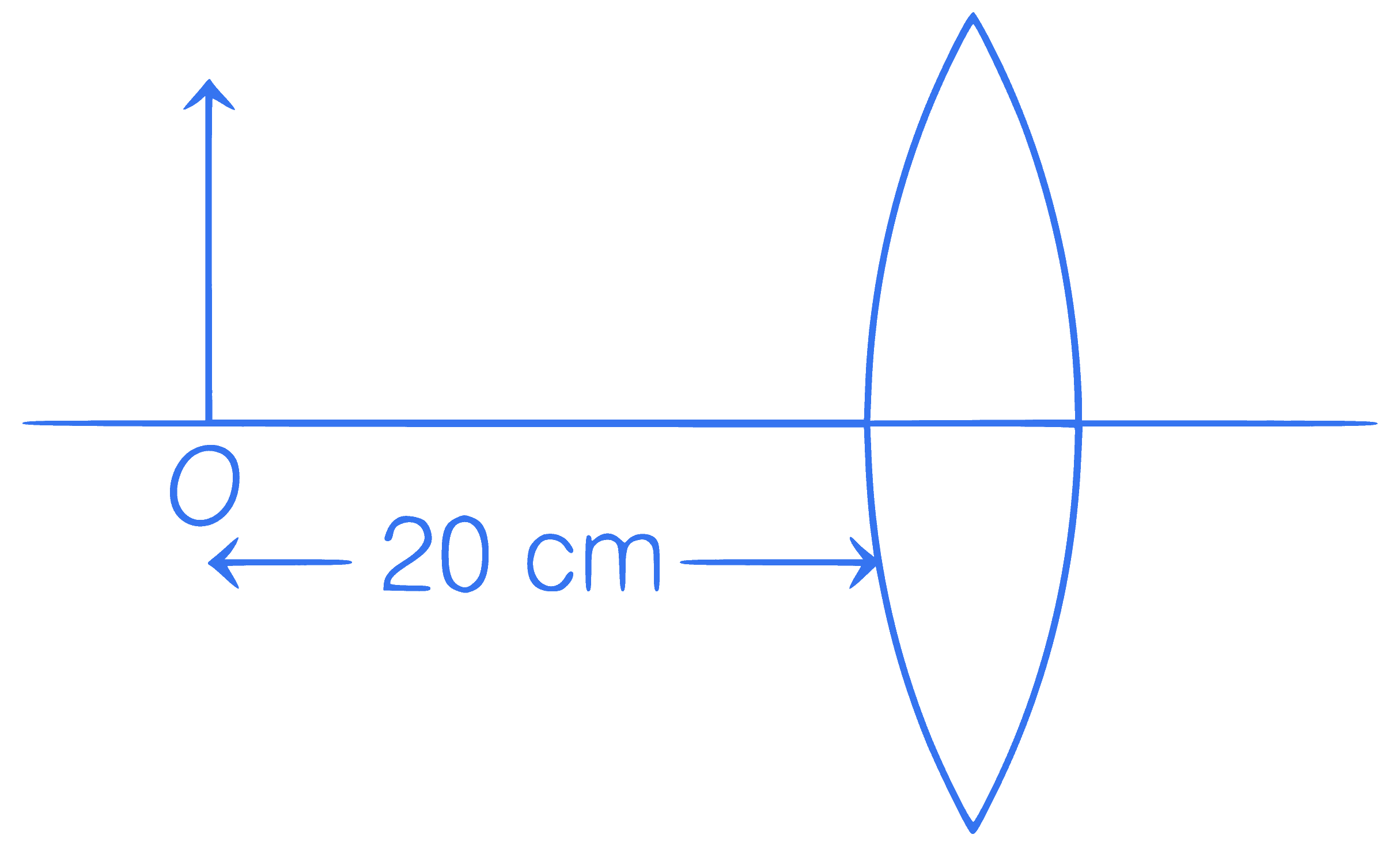

Q26.
mcq single
+1 / 0.33
If focal length of objective and eye lenses are $$10 \mathrm{~cm}$$ and $$10 \mathrm{~mm}$$ respectively and tube length is $$11 \mathrm{~cm}$$ then angular magnification of telescope is
Q27.
mcq single
+1 / 0.33
If voltage across a zener diode is 6V, then
find out the value of maximum resistance in
this condition.
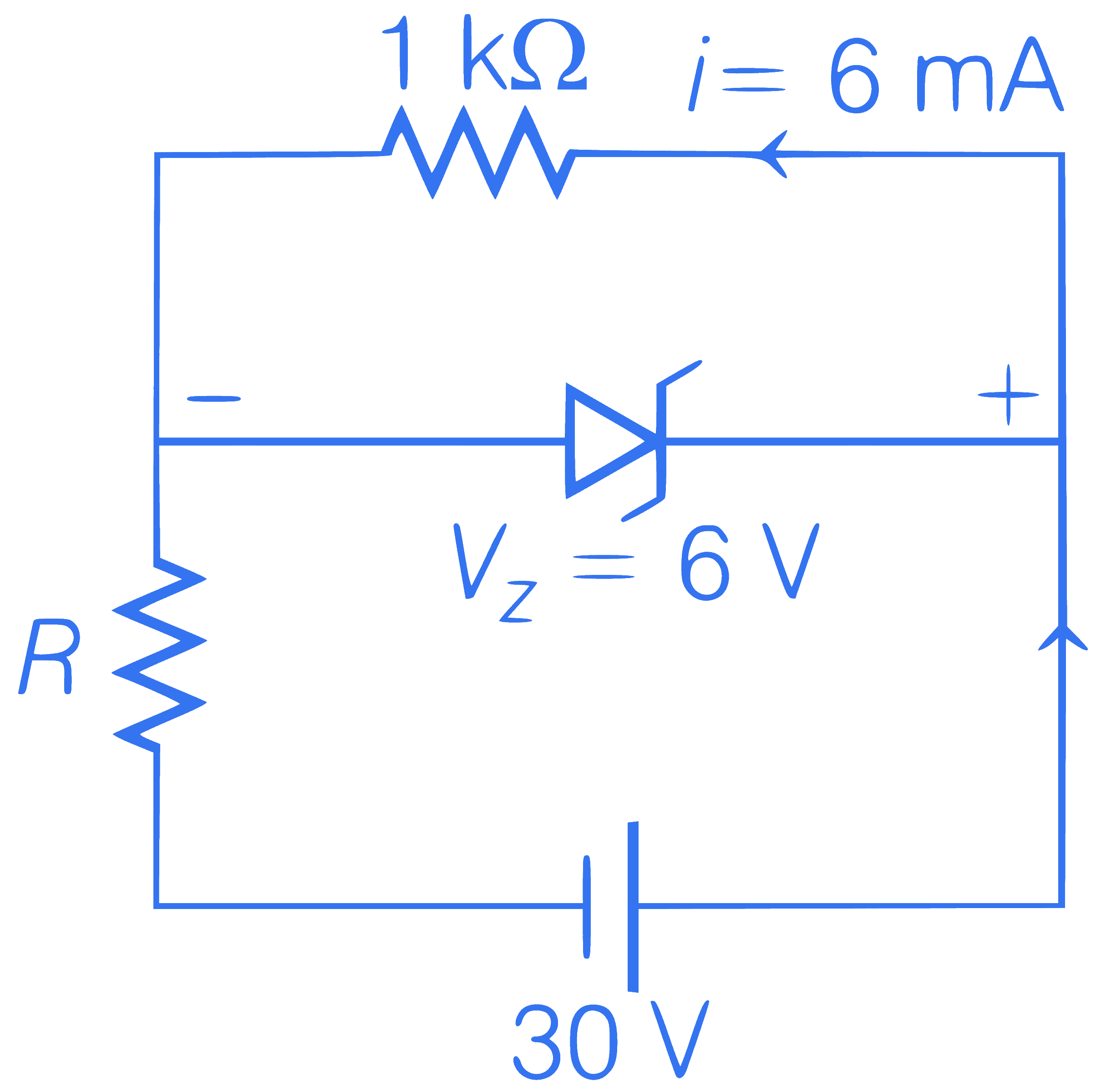

Q28.
mcq single
+1 / 0.33
The given transistor operates in saturation region then what should be the value of $$V_{B B}$$ ?
$$\begin{aligned}
& \left(R_{\text {out }}=200 \Omega, R_{\text {in }}=100 \mathrm{~k} \Omega, V_{C C}=3 \mathrm{~V},\right. \\\\
& \left.V_{B E}=0.7 \mathrm{~V}, V_{C E}=0, \beta=200\right)
\end{aligned}$$
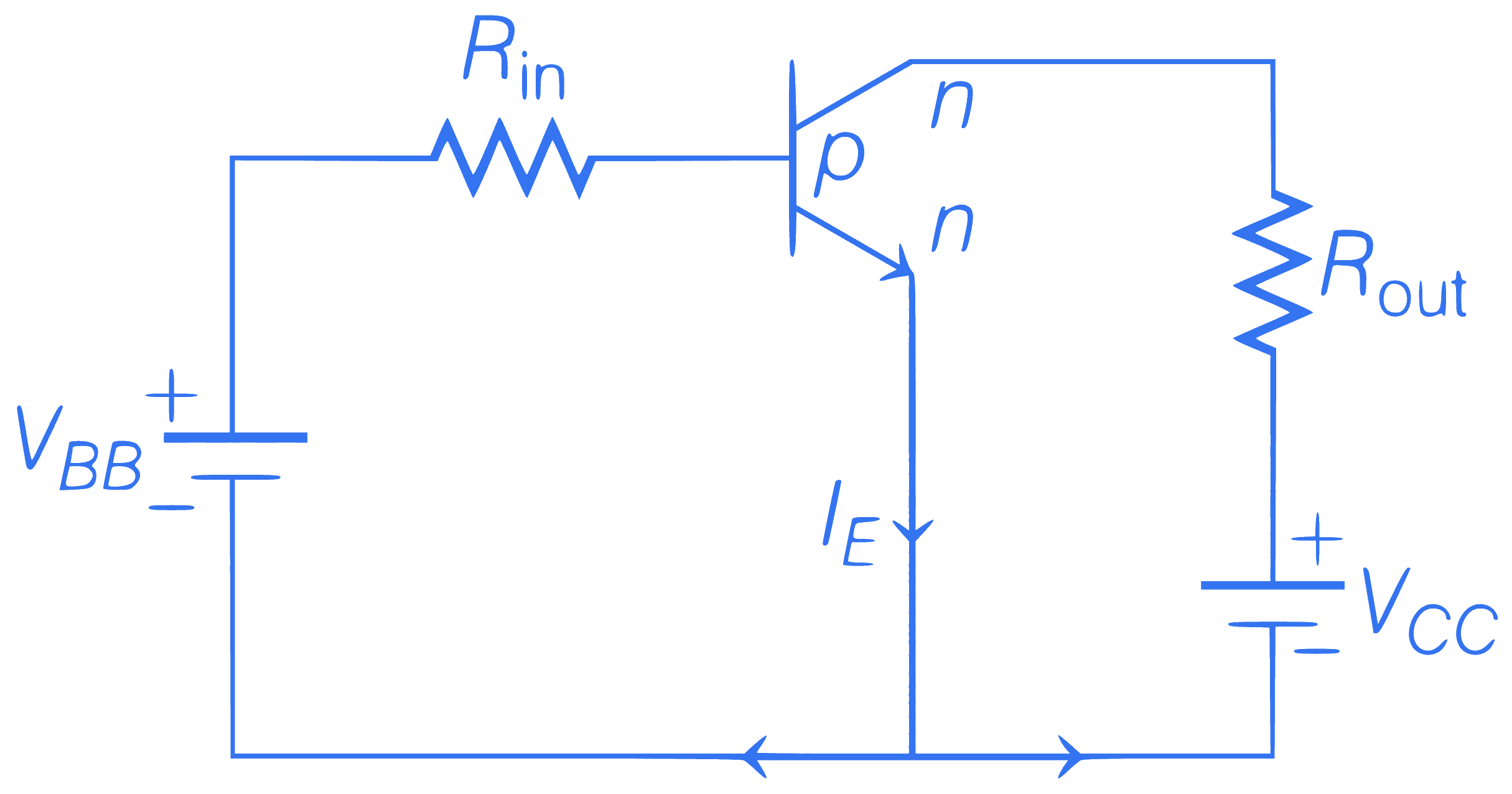

Q29.
mcq single
+1 / 0.33
**Assertion :** Photodiode and solar cell work
on same mechanism.
**Reason :** Area is large for solar cell.
Q30.
mcq single
+1 / 0.33
**Assertion :** A charge particle is released
from rest in magnetic field then it will move
in a circular path.
**Reason :** Work done by magnetic field is non
zero.
Q31.
mcq single
+1 / 0.33
A proton is projected with velocity $$\mathbf{v}=2 \hat{\mathbf{i}}$$ in a region where magnetic field $$\mathbf{B}=(\hat{\mathbf{i}}+3 \hat{\mathbf{j}}+4 \hat{\mathbf{k}}) \mu \mathrm{T}$$ and electric field $$\mathbf{E}=10 \hat{\mathbf{i}} \mu \mathrm{V} / \mathrm{m}$$. Then find out the net acceleration of proton
Q32.
mcq single
+1 / 0.33
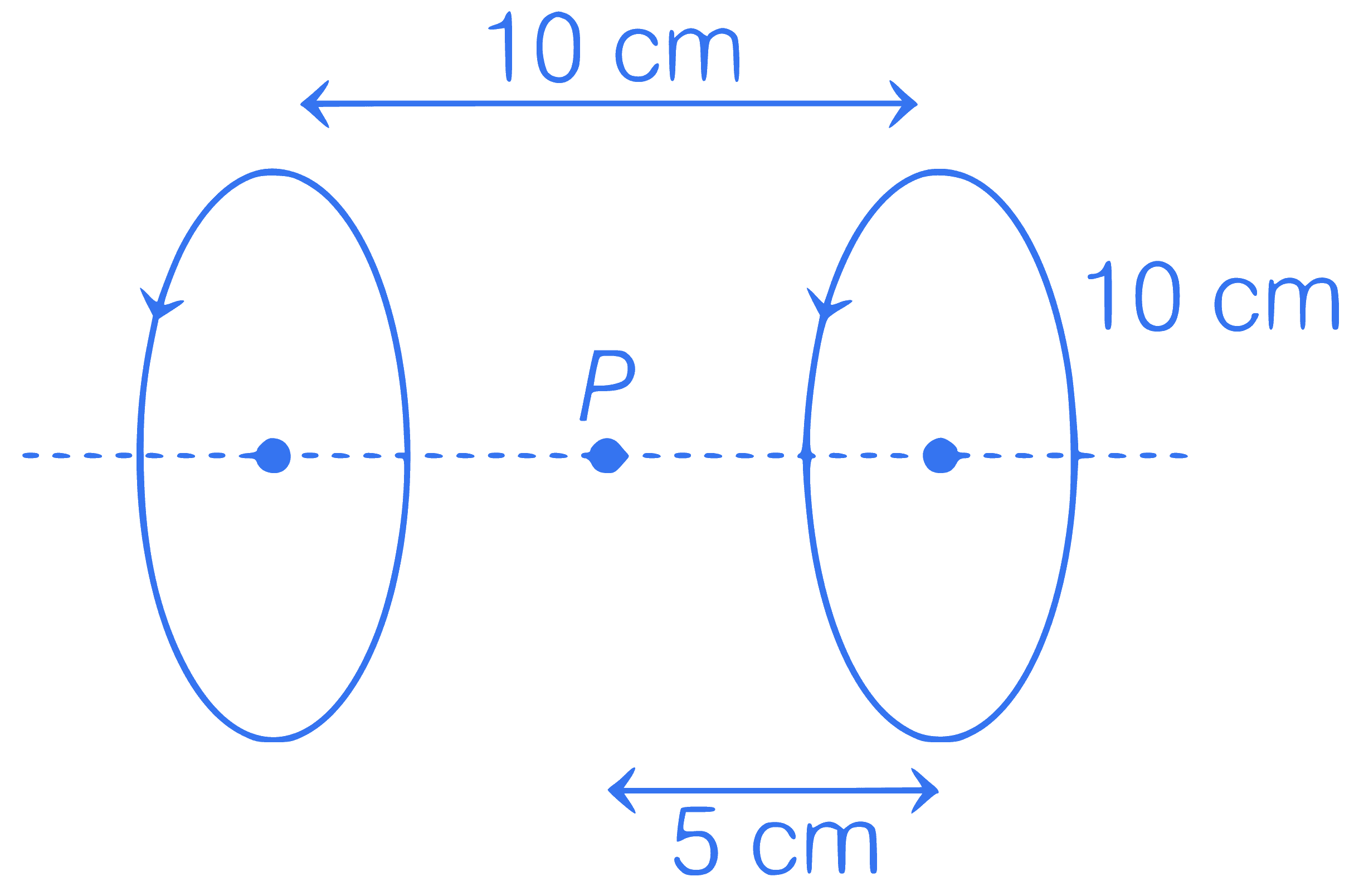
Two circular loops having same radius $$(R=10 \mathrm{~cm})$$ and same current $$\frac{7}{2} \mathrm{~A}$$ are placed along same axis as shown. If distance between their centres is $$10 \mathrm{~cm}$$, find net magnetic field at point $$P$$.

Q33.
mcq single
+1 / 0.33
If two protons are moving with speed $$v=4.5 \times 10^5 \mathrm{~m} / \mathrm{s}$$ parallel to each other then find the ratio of electrostatic and magnetic force between them
Q34.
mcq single
+1 / 0.33
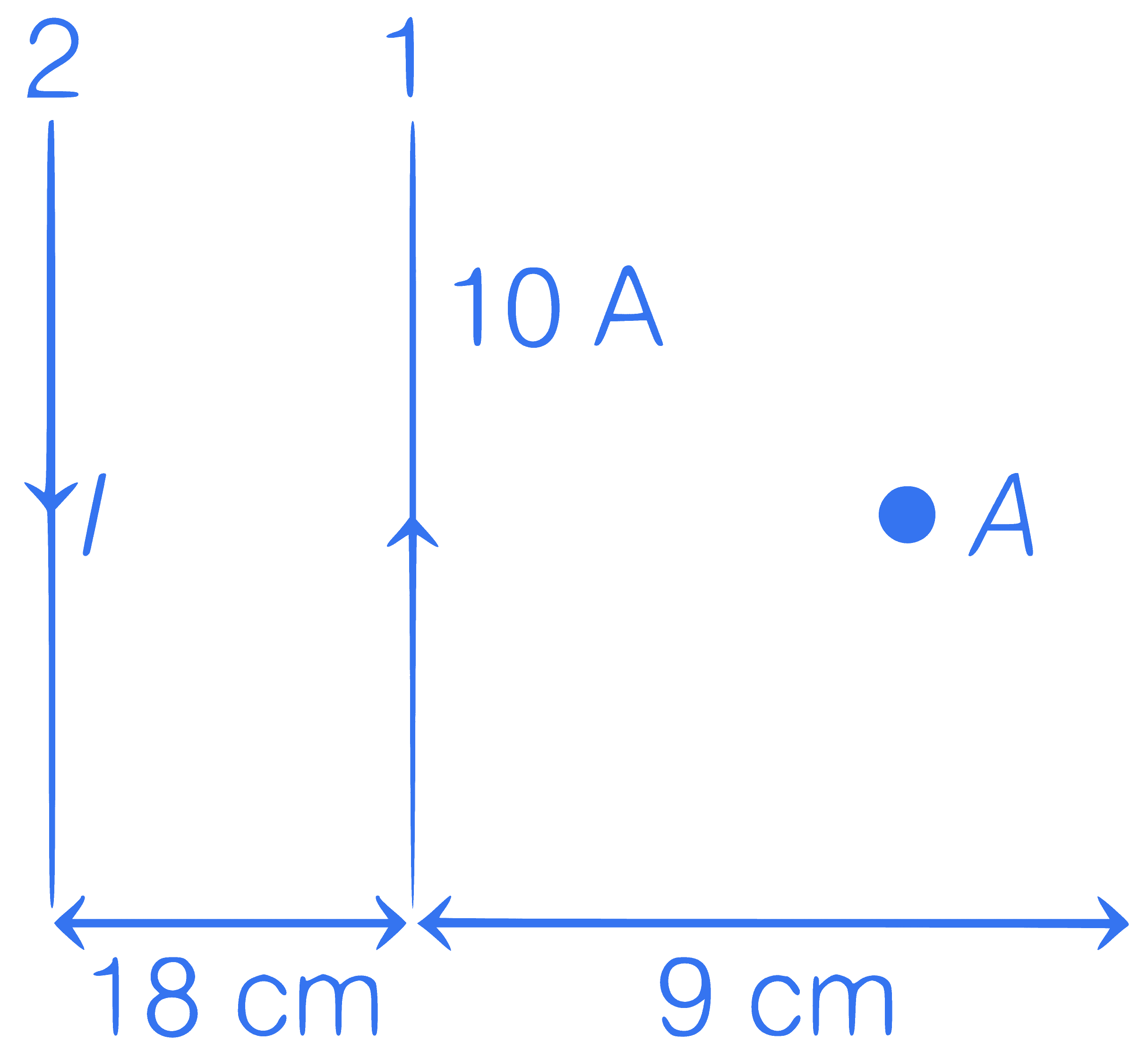
In figure, two parallel infinitely long current carrying wires are shown. If resultant magnetic field at point $$A$$ is zero. Then determine the value of current $$I$$.

Q35.
mcq single
+1 / 0.33
**Assertion :** Electron moving perpendicular
to B will perform circular motion.
**Reason :** Force by magnetic field is
perpendicular to velocity.
Q36.
mcq single
+1 / 0.33
**Assertion :** In ionospheric reflection, phase
change does not occur with the radio wave.
**Reason :** The ionosphere reflection is similar
to the total internal reflection in miraj.
Q37.
mcq single
+1 / 0.33
**Assertion :** Amplitude modulation shows
more interference than frequency
modulation with noise.
**Reason :** Interference is function of amplitude
of modulation wave with carrier-wave.
Q38.
mcq single
+1 / 0.33
If modulation index, $$\mu=\frac{1}{2}$$ and $$V_m=2$$, then $$V_C$$ is
Q39.
mcq single
+1 / 0.33
If temperature of Sun $$=6000 \mathrm{~K}$$, radius of Sun is $$7.2 \times 10^5 \mathrm{~km}$$, radius of Earth $$=6000 \mathrm{~km}$$ and distance between Earth and Sun $$=15 \times 10^7 \mathrm{~km}$$. Find intensity of light on Earth.
Q40.
mcq single
+1 / 0.33
**Assertion :** In both radioactivity and
photoelectric effect electrons may be ejected.
**Reason :** In photoelectric effect and
radioactivity emission occurs only of
unstable elements.
Q41.
mcq single
+1 / 0.33
15 eV is given to electron in 4th orbit then
find its final energy when it comes out of
H-atom.
Q42.
mcq single
+1 / 0.33
**Assertion :** For an element generally $$N \geq Z$$ ( $$N=$$ number of neutrons, $$Z=$$ atomic number)
**Reason :** Neutrons always experience attractive nuclear force.
Q43.
mcq single
+1 / 0.33
If half life of an element is $$69.3 \mathrm{~h}$$, then how much of its percent will decay in 10th to 11th $$\mathrm{h}$$. Initial activity $$=50 ~\mu \mathrm{~Ci}$$
Q44.
mcq single
+1 / 0.33
**Assertion :** Heavy water is used to slow
neutron in nuclear reactor.
**Reason :** It does not react with slow neutron
and mass of deuterium is comparable to the
neutron.
Q45.
mcq single
+1 / 0.33
A body of mass $$5 \times 10^3 \mathrm{~kg}$$ moving with speed $$2 \mathrm{~m} / \mathrm{s}$$ collides with a body of mass $$15 \times 10^3 \mathrm{~kg}$$ inelastically and sticks to it. Then loss in $$\mathrm{KE}$$ of the system will be
Q46.
mcq single
+1 / 0.33
**Assertion :** Even though net external force
on a body is zero, momentum need not to
conserved.
**Reason :** The internal interaction between
particles of a body cancels out momentum of
each other.
Q47.
mcq single
+1 / 0.33
**Assertion :** There is no loss in energy in elastic collision.
**Reason :** Linear momentum is conserved in elastic collision.
Q48.
mcq single
+1 / 0.33
A string wave equation is given $$y=0.002 \sin (300 t-15 x)$$ and mass density is $$(\mu=0.1 \mathrm{~kg} / \mathrm{m})$$. Then find the tension force in the string.
Q49.
mcq single
+1 / 0.33
Two sources of sound S$$_1$$ and S$$_2$$ are moving
towards and away from a stationary observer
with the same speed respectively. Observer
detects 3 beats per second. Find speed of
sources (approximately).
Given, $$f_1=f_2=500$$ Hz, speed of sound in air = 330 m/s
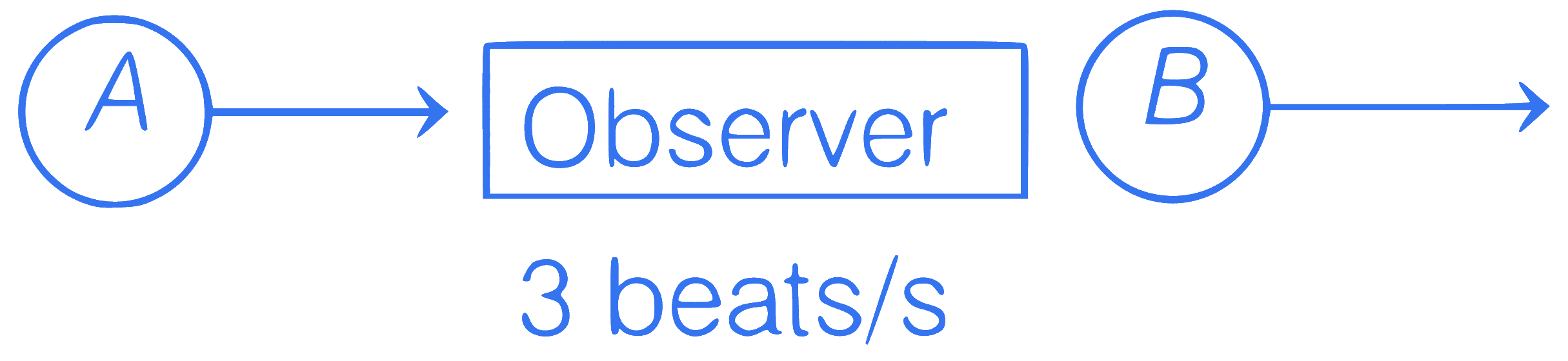
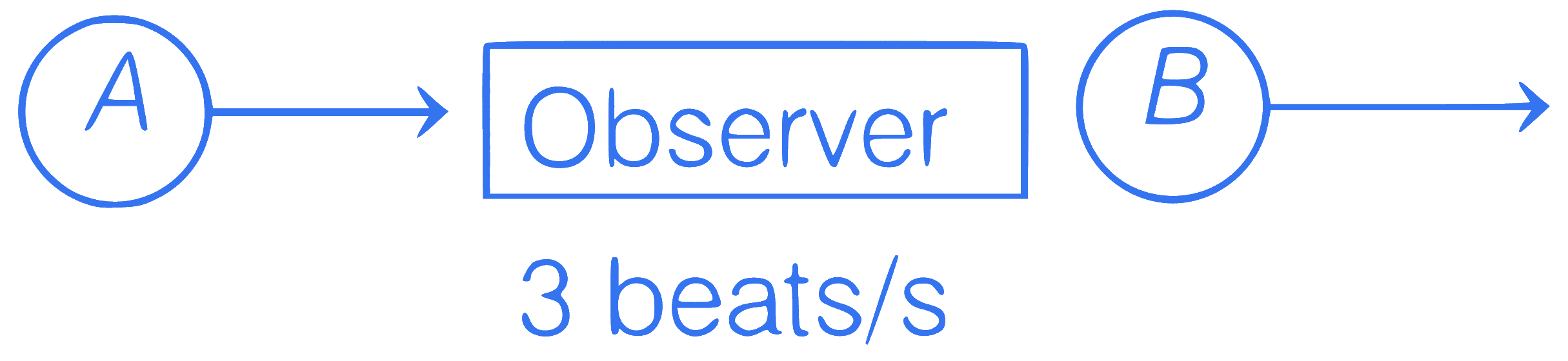
Q50.
mcq single
+1 / 0.33
If the speed of sound in air is 330 m/s then,
find the number of tones present in an open
organ pipe of length 1 m whose frequency is $$\le$$ 1000 Hz.
Q51.
mcq single
+1 / 0.33
A gun applied a force $F$ on a bullet which is given by $$F=\left(100-0.5 \times 10^5 t\right) \mathrm{N}$$. The bullet emerges out with speed $$400 \mathrm{~m} / \mathrm{s}$$. Then find out the impulse exerted till force on the bullet becomes zero.
Q52.
mcq single
+1 / 0.33
**Assertion :** A glass ball is dropped on
concrete floor can easily get broken
compared if it is dropped on wooden floor.
**Reason :** On concrete floor glass ball will take
less time to come to rest.
Q53.
mcq single
+1 / 0.33
$$\alpha$$-particle is revolving in a circular path with radius $$r$$ with speed $$v$$, then find the value of magnetic dipole moment.
Q54.
mcq single
+1 / 0.33
**Assertion :** Paramagnetic substances get
poorly attracted in magnetic field.
**Reason :** Because magnetic dipoles are
aligned along external magnetic field weakly.
Q55.
mcq single
+1 / 0.33
Initially spring in its natural
length now a block at mass 0.25 kg
is released then find out maximum
force by system on the floor.


Q56.
mcq single
+1 / 0.33
A current of $$10 \mathrm{~A}$$ is passing through a metallic wire of cross-sectional area $$4 \times 10^{-6} \mathrm{~m}^2$$. If the density of the aluminium conductor is $$2.7 \mathrm{~gm} / \mathrm{cc}$$ considering aluminium gives 1 electron per atom for conduction, then find the drift velocity of the electrons if molecular weight of aluminium is $$27 \mathrm{~gm}$$.
Q57.
mcq single
+1 / 0.33
For a wire $$\frac{R}{\ell}=\frac{1}{2}$$ and length of wire is $$\ell=5 \mathrm{~cm}$$. If potential difference of $$\mathrm{l} \mathrm{V}$$ is applied across it, then current through wire will be ($$R=$$ Resistance)
Q58.
mcq single
+1 / 0.33
If maximum energy is stored in a capacitor
at t = 0, then find the time after which
current in the circuit will be maximum.
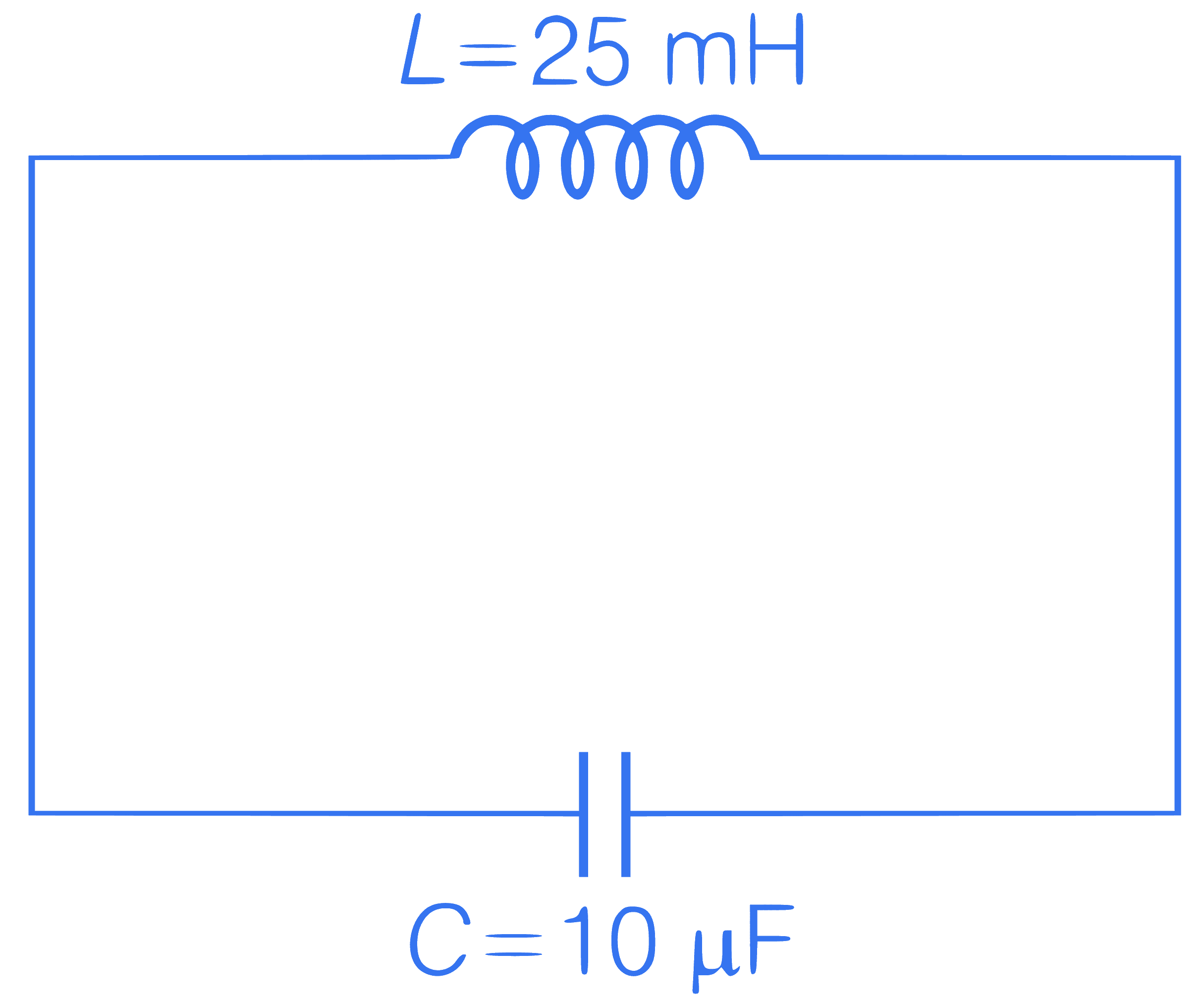

Q59.
mcq single
+1 / 0.33
In a $$L C R$$ series circuit source voltage is $$120 \mathrm{~V}$$ and voltage in inductor $$50 \mathrm{~V}$$ and resistance is $$40 \mathrm{~V}$$, then determine voltage in the capacitor
Q60.
mcq single
+1 / 0.33
A transformer with turns ratio $$\frac{N_1}{N_2}=\frac{50}{1}$$ is connected to a $$120 \mathrm{~V}$$ AC supply. If primary and secondary circuit resistances are $$1.5 \mathrm{~k} \Omega$$ and $$1 \Omega$$ respectively, then find out power of output.
Q61.
mcq single
+1 / 0.33
**Assertion :** Distance between position of
bright and dark fringe remain same in YDSE.
**Reason :** Fringe width, $$\beta=\frac{\lambda D}{d}$$
Q62.
mcq single
+1 / 0.33
**Assertion :** Incoming light reflected by earth
is partially polarised.
**Reason :** Atmospheric particle polarise the
light.
Q63.
mcq single
+1 / 0.33
Distance of 5th dark fringe from centre is $$4 \mathrm{~mm}$$. If $$D=2 \mathrm{~m}, \lambda=600 \mathrm{~nm}$$, then distance between slits is
Q64.
mcq single
+1 / 0.33
A light of wavelength $$500 \mathrm{~nm}$$ is incident on a Young's double slit. The distance between slit and screen is $$D=1.8 \mathrm{~m}$$ and distance between slits is $$d=0.4 \mathrm{~mm}$$. If screen moves with a speed of $$4 \mathrm{~m} / \mathrm{s}$$, then with what speed first maxima will move?
Q65.
mcq single
+1 / 0.33
A sphere pure rolls on a rough inclined
plane with initial velocity 2.8 m/s. Find the
maximum distance on the inclined plane.


Q66.
mcq single
+1 / 0.33
**Assertion :** A metallic surface is moved in
and out in magnetic field then emf is
induced in it.
**Reason :** Eddy current will be produced in a
metallic surface moving in and out of
magnetic field.
Q67.
mcq single
+1 / 0.33
An electron is moving in a circle of radius
2m with speed of 4 m/s. Find the
acceleration of the electron.
Q68.
mcq single
+1 / 0.33
Calculate the mean percentage error in five
observations,
80.0, 80.5, 81.0, 81.5, 82
Q69.
mcq single
+1 / 0.33
**Assertion :** Vibrational degree of freedom of
a di-atomic gas molecule appears at every
high temperature.
**Reason :** Di-atomic gas has two vibrational
degree of freedom in one direction.
Q70.
mcq single
+1 / 0.33
If $$7 \mathrm{~gm} \mathrm{~N}_2$$ is mixed with $$20 \mathrm{~gm} \mathrm{~Ar}$$, there $$\frac{C_p}{C_V}$$ of mixture will be
Q71.
mcq single
+1 / 0.33
Calculate radiation power for sphere whose temperature is 227$$^\circ$$C, radius 2 m and emissivity 0.8.
Q72.
mcq single
+1 / 0.33
In an isobaric process, the work done by a
di-atomic gas is 10 J, the heat given to the
gas will be
Q73.
mcq single
+1 / 0.33
**Assertion :** In adiabatic process work is
independent of the path.
**Reason : **In adiabatic process work done is
equal to negative of change in internal
energy.
Q74.
mcq single
+1 / 0.33
The temperature of food material in
refrigerator is 4$$^\circ$$C and temperature of
environment is 15$$^\circ$$C. If carnot cycle is used
in its working gas, then find its carnot
efficiency.
Q75.
mcq single
+1 / 0.33
An ideal gas initially at pressure 1 bar is being compressed from $$30 \mathrm{~m}^3$$ to $$10 \mathrm{~m}^3$$ volume and its temperature decreases from $$320 \mathrm{~K}$$ to $$280 \mathrm{~K}$$, then find final pressure of the gas.
Q76.
mcq single
+1 / 0.33
**Assertion :** $$\mathrm{NH}_3$$ is liquidities more easily than $$\mathrm{CO}_2$$.
**Reason :** Critical temperature of $$\mathrm{NH}_3$$ is more than $$\mathrm{CO}_2$$.
Q77.
mcq single
+1 / 0.33
In an isothermal process 2 water drops of radius $$1 \mathrm{~mm}$$ are combined to form a bigger drop. Find the energy change in this process if $$T=0.1 \mathrm{~N} / \mathrm{m}$$.
Q78.
mcq single
+1 / 0.33
**Assertion :** Sometimes insects can walk on
water.
**Reason :** The gravitational force on insect is
balanced by force due surface tension.
Q79.
mcq single
+1 / 0.33
**Assertion :** Water drops take spherical shape
when falling freely.
**Reason :** Water has minimum surface
tension among all liquids.
Q80.
mcq single
+1 / 0.33
If a small orifice is made at a height of
0.25 m from the ground, the horizontal
range of water stream will be


Q81.
mcq single
+1 / 0.33
Determine the pressure difference in tube of
non-uniform cross sectional area as shown
in figure.
$$\Delta P=?, d_1=5 \mathrm{~cm}, v_1=4 \mathrm{~m} / \mathrm{s}, d_2=2 \mathrm{~cm}, v_2=?$$


Q82.
mcq single
+1 / 0.33
Find gravitational field at a distance of $$2000 \mathrm{~km}$$ from the centre of earth. (Given
$$R_{\text {earth }}=6400 \mathrm{~km}, r=2000 \mathrm{~km} \text {, } M_{\text {earth }}=6 \times 10^{24} \mathrm{~kg} \text { ) }$$
Q83.
mcq single
+1 / 0.33
A capacitor of capacitance $$15 \mu \mathrm{F}$$ having dielectric slab of $$\varepsilon_r=2.5$$, dielectric strength $$30 \mathrm{~MV} / \mathrm{m}$$ and potential difference $$=30 \mathrm{~V}$$. Calculate the area of the plate.
Q84.
mcq single
+1 / 0.33
 In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
 In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
In the given circuit, find charge on capacitor after 1s of opening the switch at $$t=\infty$$.
Q85.
mcq single
+1 / 0.33
Calculate the focal length of given lens if the
magnification is $$-$$0 5.


Q86.
mcq single
+1 / 0.33
If focal length of objective and eye lenses are $$10 \mathrm{~cm}$$ and $$10 \mathrm{~mm}$$ respectively and tube length is $$11 \mathrm{~cm}$$ then angular magnification of telescope is
Q87.
mcq single
+1 / 0.33
If voltage across a zener diode is 6V, then
find out the value of maximum resistance in
this condition.


Q88.
mcq single
+1 / 0.33
The given transistor operates in saturation region then what should be the value of $$V_{B B}$$ ?
$$\begin{aligned}
& \left(R_{\text {out }}=200 \Omega, R_{\text {in }}=100 \mathrm{~k} \Omega, V_{C C}=3 \mathrm{~V},\right. \\\\
& \left.V_{B E}=0.7 \mathrm{~V}, V_{C E}=0, \beta=200\right)
\end{aligned}$$


Q89.
mcq single
+1 / 0.33
**Assertion :** Photodiode and solar cell work
on same mechanism.
**Reason :** Area is large for solar cell.
Q90.
mcq single
+1 / 0.33
**Assertion :** A charge particle is released
from rest in magnetic field then it will move
in a circular path.
**Reason :** Work done by magnetic field is non
zero.
Q91.
mcq single
+1 / 0.33
A proton is projected with velocity $$\mathbf{v}=2 \hat{\mathbf{i}}$$ in a region where magnetic field $$\mathbf{B}=(\hat{\mathbf{i}}+3 \hat{\mathbf{j}}+4 \hat{\mathbf{k}}) \mu \mathrm{T}$$ and electric field $$\mathbf{E}=10 \hat{\mathbf{i}} \mu \mathrm{V} / \mathrm{m}$$. Then find out the net acceleration of proton
Q92.
mcq single
+1 / 0.33
Two circular loops having same radius $$(R=10 \mathrm{~cm})$$ and same current $$\frac{7}{2} \mathrm{~A}$$ are placed along same axis as shown. If distance between their centres is $$10 \mathrm{~cm}$$, find net magnetic field at point $$P$$.


Q93.
mcq single
+1 / 0.33
If two protons are moving with speed $$v=4.5 \times 10^5 \mathrm{~m} / \mathrm{s}$$ parallel to each other then find the ratio of electrostatic and magnetic force between them
Q94.
mcq single
+1 / 0.33
In figure, two parallel infinitely long current carrying wires are shown. If resultant magnetic field at point $$A$$ is zero. Then determine the value of current $$I$$.


Q95.
mcq single
+1 / 0.33
**Assertion :** Electron moving perpendicular
to B will perform circular motion.
**Reason :** Force by magnetic field is
perpendicular to velocity.
Q96.
mcq single
+1 / 0.33
**Assertion :** In ionospheric reflection, phase
change does not occur with the radio wave.
**Reason :** The ionosphere reflection is similar
to the total internal reflection in miraj.
Q97.
mcq single
+1 / 0.33
**Assertion :** Amplitude modulation shows
more interference than frequency
modulation with noise.
**Reason :** Interference is function of amplitude
of modulation wave with carrier-wave.
Q98.
mcq single
+1 / 0.33
If modulation index, $$\mu=\frac{1}{2}$$ and $$V_m=2$$, then $$V_C$$ is
Q99.
mcq single
+1 / 0.33
If temperature of Sun $$=6000 \mathrm{~K}$$, radius of Sun is $$7.2 \times 10^5 \mathrm{~km}$$, radius of Earth $$=6000 \mathrm{~km}$$ and distance between Earth and Sun $$=15 \times 10^7 \mathrm{~km}$$. Find intensity of light on Earth.
Q100.
mcq single
+1 / 0.33
**Assertion :** In both radioactivity and
photoelectric effect electrons may be ejected.
**Reason :** In photoelectric effect and
radioactivity emission occurs only of
unstable elements.
Q101.
mcq single
+1 / 0.33
15 eV is given to electron in 4th orbit then
find its final energy when it comes out of
H-atom.
Q102.
mcq single
+1 / 0.33
**Assertion :** For an element generally $$N \geq Z$$ ( $$N=$$ number of neutrons, $$Z=$$ atomic number)
**Reason :** Neutrons always experience attractive nuclear force.
Q103.
mcq single
+1 / 0.33
If half life of an element is $$69.3 \mathrm{~h}$$, then how much of its percent will decay in 10th to 11th $$\mathrm{h}$$. Initial activity $$=50 ~\mu \mathrm{~Ci}$$
Q104.
mcq single
+1 / 0.33
**Assertion :** Heavy water is used to slow
neutron in nuclear reactor.
**Reason :** It does not react with slow neutron
and mass of deuterium is comparable to the
neutron.
Q105.
mcq single
+1 / 0.33
A body of mass $$5 \times 10^3 \mathrm{~kg}$$ moving with speed $$2 \mathrm{~m} / \mathrm{s}$$ collides with a body of mass $$15 \times 10^3 \mathrm{~kg}$$ inelastically and sticks to it. Then loss in $$\mathrm{KE}$$ of the system will be
Q106.
mcq single
+1 / 0.33
**Assertion :** Even though net external force
on a body is zero, momentum need not to
conserved.
**Reason :** The internal interaction between
particles of a body cancels out momentum of
each other.
Q107.
mcq single
+1 / 0.33
**Assertion :** There is no loss in energy in elastic collision.
**Reason :** Linear momentum is conserved in elastic collision.
Q108.
mcq single
+1 / 0.33
A string wave equation is given $$y=0.002 \sin (300 t-15 x)$$ and mass density is $$(\mu=0.1 \mathrm{~kg} / \mathrm{m})$$. Then find the tension force in the string.
Q109.
mcq single
+1 / 0.33
Two sources of sound S$$_1$$ and S$$_2$$ are moving
towards and away from a stationary observer
with the same speed respectively. Observer
detects 3 beats per second. Find speed of
sources (approximately).
Given, $$f_1=f_2=500$$ Hz, speed of sound in air = 330 m/s
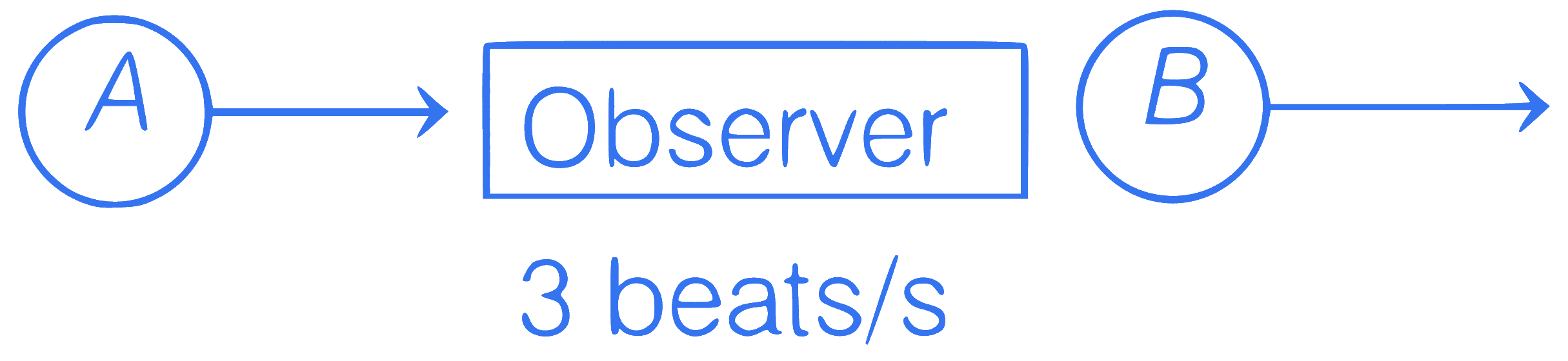
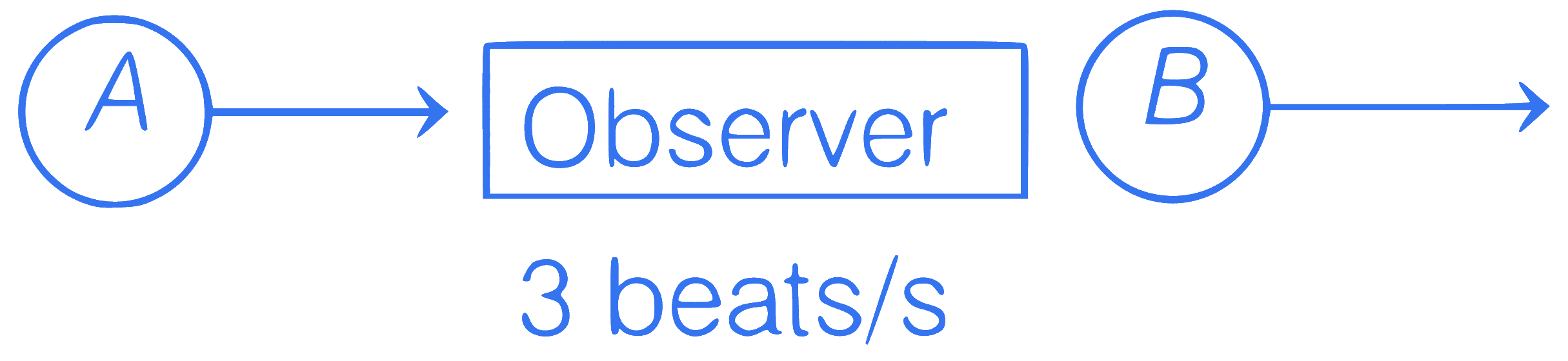
Q110.
mcq single
+1 / 0.33
If the speed of sound in air is 330 m/s then,
find the number of tones present in an open
organ pipe of length 1 m whose frequency is $$\le$$ 1000 Hz.
Q111.
mcq single
+1 / 0.33
A gun applied a force $F$ on a bullet which is given by $$F=\left(100-0.5 \times 10^5 t\right) \mathrm{N}$$. The bullet emerges out with speed $$400 \mathrm{~m} / \mathrm{s}$$. Then find out the impulse exerted till force on the bullet becomes zero.
Q112.
mcq single
+1 / 0.33
**Assertion :** A glass ball is dropped on
concrete floor can easily get broken
compared if it is dropped on wooden floor.
**Reason :** On concrete floor glass ball will take
less time to come to rest.
Q113.
mcq single
+1 / 0.33
$$\alpha$$-particle is revolving in a circular path with radius $$r$$ with speed $$v$$, then find the value of magnetic dipole moment.
Q114.
mcq single
+1 / 0.33
**Assertion :** Paramagnetic substances get
poorly attracted in magnetic field.
**Reason :** Because magnetic dipoles are
aligned along external magnetic field weakly.
Q115.
mcq single
+1 / 0.33
Initially spring in its natural
length now a block at mass 0.25 kg
is released then find out maximum
force by system on the floor.


Q116.
mcq single
+1 / 0.33
A current of $$10 \mathrm{~A}$$ is passing through a metallic wire of cross-sectional area $$4 \times 10^{-6} \mathrm{~m}^2$$. If the density of the aluminium conductor is $$2.7 \mathrm{~gm} / \mathrm{cc}$$ considering aluminium gives 1 electron per atom for conduction, then find the drift velocity of the electrons if molecular weight of aluminium is $$27 \mathrm{~gm}$$.
Q117.
mcq single
+1 / 0.33
For a wire $$\frac{R}{\ell}=\frac{1}{2}$$ and length of wire is $$\ell=5 \mathrm{~cm}$$. If potential difference of $$\mathrm{l} \mathrm{V}$$ is applied across it, then current through wire will be ($$R=$$ Resistance)
Q118.
mcq single
+1 / 0.33
If maximum energy is stored in a capacitor
at t = 0, then find the time after which
current in the circuit will be maximum.


Q119.
mcq single
+1 / 0.33
In a $$L C R$$ series circuit source voltage is $$120 \mathrm{~V}$$ and voltage in inductor $$50 \mathrm{~V}$$ and resistance is $$40 \mathrm{~V}$$, then determine voltage in the capacitor
Q120.
mcq single
+1 / 0.33
A transformer with turns ratio $$\frac{N_1}{N_2}=\frac{50}{1}$$ is connected to a $$120 \mathrm{~V}$$ AC supply. If primary and secondary circuit resistances are $$1.5 \mathrm{~k} \Omega$$ and $$1 \Omega$$ respectively, then find out power of output.
Biology
Biology
Q1.
mcq single
+1 / 0.33
Mark the incorrect statement for inbreeding.
Q2.
mcq single
+1 / 0.33
Blood group of the father is ‘A’ and blood
group of mother is ‘B’. Then predict the
blood group of the progeny
Q3.
mcq single
+1 / 0.33
**Assertion :** Down’s syndrome, Klinefelter’s
syndrome and Turner’s syndrome are
chromosomal disorders.
**Reason :** In Klinefelter’s syndrome females
are sterile.
Q4.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
Pleiotropic gene
(i)
Both alleles express equally
B.
Co-dominance
(ii)
Change in nucleotides
C.
Epistasis
(iii)
One gene shows multiple phenotypic expression
D.
Mutation
(iv)
Non-allelic gene inheritance
Q5.
mcq single
+1 / 0.33
Characteristic of female cockroach
Q6.
mcq single
+1 / 0.33
Which cell is found in mucus secreting organs?
Q7.
mcq single
+1 / 0.33
Identify the given diagram of tissue
performing secretion and absorption.
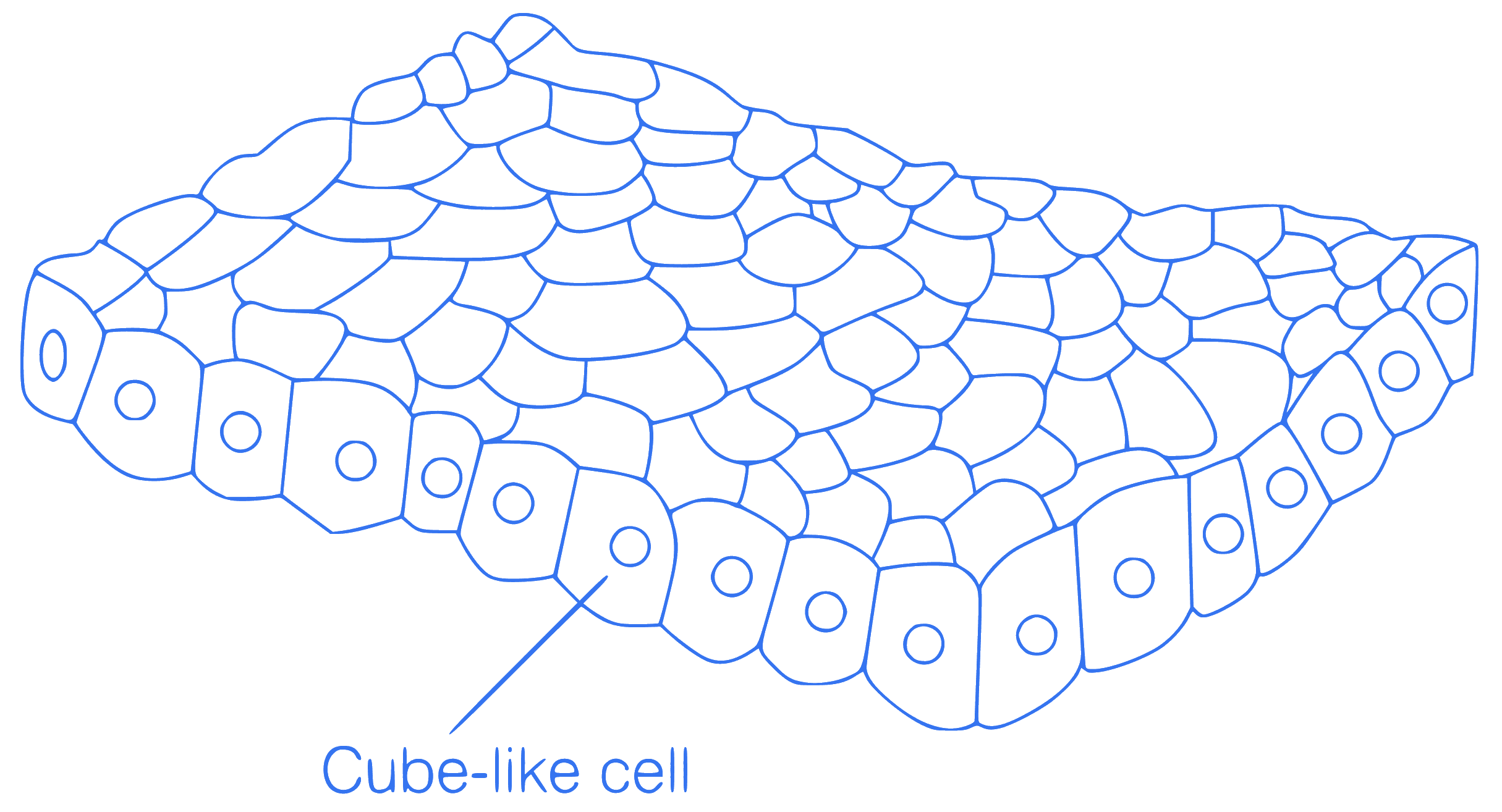

Q8.
mcq single
+1 / 0.33
What is the site of $$C_3$$ cycle in $$C_3$$ and $$C_4$$ plants?
Q9.
mcq single
+1 / 0.33
**Assertion :** In $$\mathrm{C}_3$$ cycle, the first stable compound is $$3 \mathrm{C}$$ compound.
**Reason :** In $$\mathrm{C}_4$$ plants, Calvin cycle is absent.
Q10.
mcq single
+1 / 0.33
Animal of which phylum have hooks and
suckers and are endoparasite on other animals?
Q11.
mcq single
+1 / 0.33
**Assertion :** Heterospory and retention of
female gametophyte are responsible for
origin of seed habit in Selaginella.
**Reason :** Psilotum is a living fossil.
Q12.
mcq single
+1 / 0.33
Which of the following are homosporous?
Q13.
mcq single
+1 / 0.33
**Assertion :** Selaginella and Salvinia are
homosporous.
**Reason :** In pteridophyte, Lycopodium is
precursor of seed habit.
Q14.
mcq single
+1 / 0.33
**Assertion :** Gastrin is a hormone that is
released from the gastrointestinal tract and
helps in digestion.
**Reason :** It promotes secretion of HCl and
trypsinogen.
Q15.
mcq single
+1 / 0.33
**Assertion :** Cholecystokinin is released by
duodenum.
**Reason :** It activates pepsinogen and bile
juice.
Q16.
mcq single
+1 / 0.33

 Identify A, B and C
Identify A, B and C

 Identify A, B and C
Identify A, B and C
Q17.
mcq single
+1 / 0.33
Select the wrong statement from the following.
Q18.
mcq single
+1 / 0.33
The genetic codes of arginine are
Q19.
mcq single
+1 / 0.33
**Assertion :** In eukaryotes, transcription
occurs in nucleus.
**Reason :** In bacteria, transcription and
translation occurs in cytoplasm.
Q20.
mcq single
+1 / 0.33
Gene library or DNA library has the collection of
Q21.
mcq single
+1 / 0.33
**Assertion :** Deficiency of an element may
lead to scurvy.
**Reason :** Daily requirement of ascorbic acid is
5 mg/day.
Q22.
mcq single
+1 / 0.33
**Assertion :** Cannabinoids are drugs of abuse.
**Reason :** They affect cardiovascular system
and central nervous system activity.
Q23.
mcq single
+1 / 0.33
Immunity tolerance developed by
Q24.
mcq single
+1 / 0.33
**Assertion :** Type-I diabetes is caused by
destruction of $$\beta$$-cells of islets of Langerhans.
**Reason :** Insulin can be taken as pills.
Q25.
mcq single
+1 / 0.33
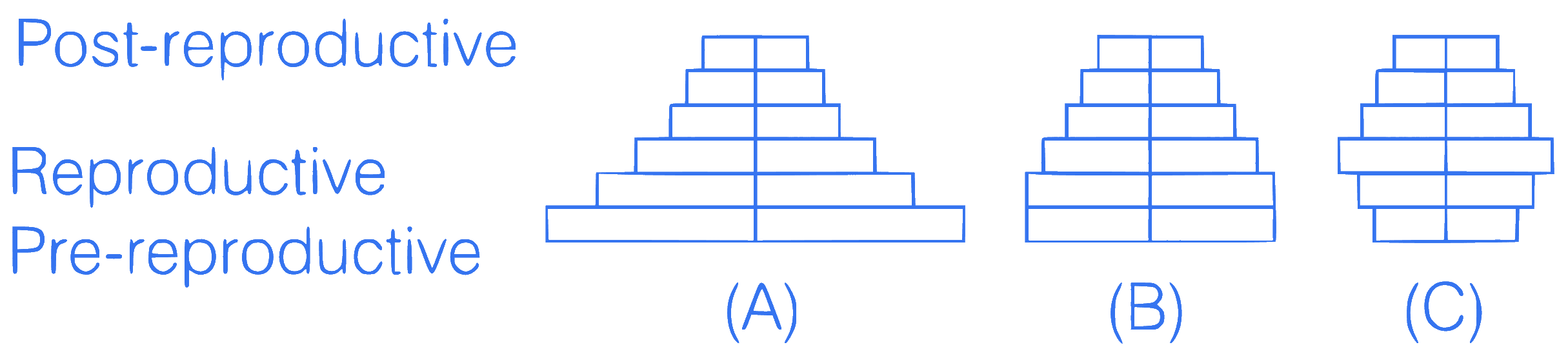 Select the correct option w.r.t. age pyramids.
Select the correct option w.r.t. age pyramids.
 Select the correct option w.r.t. age pyramids.
Select the correct option w.r.t. age pyramids.
Q26.
mcq single
+1 / 0.33
**Assertion :** In commensalism, one organism
is benefitted and other is unaffected.
**Reason :** Cattle egret bird and cattle is an
example of commensalism.
Q27.
mcq single
+1 / 0.33
**Assertion :** SA node malfunctioning leads to
disturbance of heart rate.
**Reason :** SA node is the pacemaker of heart
producing electric impulse for heart
contraction.
Q28.
mcq single
+1 / 0.33
What is the function of Bowman's capsule and glomerulus?
Q29.
mcq single
+1 / 0.33
**Assertion :** Pituitary gland releases a
hormone which is helpful in childbirth.
**Reason :** Pituitary gland releases vasopressin
and anti-diuretic hormone which helps in
childbirth.
Q30.
mcq single
+1 / 0.33
Choose the correct option.


Q31.
mcq single
+1 / 0.33
Oxytocin and ADH are produced by hypothalamus and released from
Q32.
mcq single
+1 / 0.33
**Assertion :** Biofortification is used to
increase nutrient value of crops.
**Reason :** Meristem culture is used to obtain
virus resistant plants.
Q33.
mcq single
+1 / 0.33
King of spices is
Q34.
mcq single
+1 / 0.33
**Assertion :** Biofortified crop is a source of
higher proteins, minerals and healthier fats.
**Reason :** Azolla is biofertiliser.
Q35.
mcq single
+1 / 0.33
**Assertion :** Gibberellin is useful in early seed
production in conifers.
**Reason :** Ethephon is responsible for early
ripening in tomato and apple.
Q36.
mcq single
+1 / 0.33
If mitochondria is absent in mature RBC,
what will be the source of energy?
Q37.
mcq single
+1 / 0.33
ATP formation occurs through which of the following?
Q38.
mcq single
+1 / 0.33
**Assertion :** Respiratory pathway is
considered as an amphibolic pathway.
**Reason :** It involves both anabolism and
catabolism.
Q39.
mcq single
+1 / 0.33
Cervical vertebrae differ from other vertebrae in having
Q40.
mcq single
+1 / 0.33
**Assertion :** Calcium required for skeletal
muscle contraction.
**Reason :** Calcium influx releases
acetylcholine at neuromuscular junction.
Q41.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
Chrysophyte
(i)
Gonyaulax
B.
Dinoflagellate
(ii)
Euglena
C.
Euglenoids
(iii)
Diatom
D.
Slime moulds
(iv)
Plasmodium
Q42.
mcq single
+1 / 0.33
Which statement is correct?
Q43.
mcq single
+1 / 0.33
**Assertion :** Malpighian tubules are excretory
organs in most of the insects.
**Reason :** These help in excretion of urea and
creatinine.
Q44.
mcq single
+1 / 0.33
The process of removal of anther from the
flower bud before it dehisces is called as
Q45.
mcq single
+1 / 0.33
**Assertion :** Parthenocarpy involves the
formation of seedless fruit.
**Reason :** Apomixis occurs without
fertilisation.
Q46.
mcq single
+1 / 0.33
Which of the following is correct?
Q47.
mcq single
+1 / 0.33
Which of the following represent zygomorphic symmetry?
Q48.
mcq single
+1 / 0.33
Which of the following is incorrect?
Q49.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
Tap root
(i)
Sweet potato
B.
Adventitious root
(ii)
Turnip
C.
Stem
(iii)
Wheat
D.
Fibrous root
(iv)
Potato
Q50.
mcq single
+1 / 0.33
Which of the following linkage is found in sucrose?
Q51.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
(Substrate)
Column II
(Enzyme)
(A)
Ribonucleotide
(i)
Chitinase
(B)
Chitin
(ii)
Cellulose
(C)
Cellulose
(iii)
Ribonuclease
Q52.
mcq single
+1 / 0.33
Glucose on reacting with Benedict's solution may give the following precipitates except
Q53.
mcq single
+1 / 0.33
Which of the following are all nucleotides?
Q54.
mcq single
+1 / 0.33
Protein on reaction with which yields Ruhemann's purple?
Q55.
mcq single
+1 / 0.33
Virus free plants can be formed by
Q56.
mcq single
+1 / 0.33
Hairy root disease of dicot plants is caused by
Q57.
mcq single
+1 / 0.33
What are the requirements in tissue culture?
Q58.
mcq single
+1 / 0.33
Chimeric DNA is
Q59.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
KC mehta
(i)
Fluid mosaic model
B.
P Maheshwari
(ii)
First recombinant plasmid
C.
Cohen and Boyer
(iii)
Haploid culture
D.
Singer and Nicolson
(iv)
Rust disease
Q60.
mcq single
+1 / 0.33
In Ti-plasmid, which of the following is removed?
Q61.
mcq single
+1 / 0.33
Mark the incorrect statement for inbreeding.
Q62.
mcq single
+1 / 0.33
Blood group of the father is ‘A’ and blood
group of mother is ‘B’. Then predict the
blood group of the progeny
Q63.
mcq single
+1 / 0.33
**Assertion :** Down’s syndrome, Klinefelter’s
syndrome and Turner’s syndrome are
chromosomal disorders.
**Reason :** In Klinefelter’s syndrome females
are sterile.
Q64.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
Pleiotropic gene
(i)
Both alleles express equally
B.
Co-dominance
(ii)
Change in nucleotides
C.
Epistasis
(iii)
One gene shows multiple phenotypic expression
D.
Mutation
(iv)
Non-allelic gene inheritance
Q65.
mcq single
+1 / 0.33
Characteristic of female cockroach
Q66.
mcq single
+1 / 0.33
Which cell is found in mucus secreting organs?
Q67.
mcq single
+1 / 0.33
Identify the given diagram of tissue
performing secretion and absorption.


Q68.
mcq single
+1 / 0.33
What is the site of $$C_3$$ cycle in $$C_3$$ and $$C_4$$ plants?
Q69.
mcq single
+1 / 0.33
**Assertion :** In $$\mathrm{C}_3$$ cycle, the first stable compound is $$3 \mathrm{C}$$ compound.
**Reason :** In $$\mathrm{C}_4$$ plants, Calvin cycle is absent.
Q70.
mcq single
+1 / 0.33
Animal of which phylum have hooks and
suckers and are endoparasite on other animals?
Q71.
mcq single
+1 / 0.33
**Assertion :** Heterospory and retention of
female gametophyte are responsible for
origin of seed habit in Selaginella.
**Reason :** Psilotum is a living fossil.
Q72.
mcq single
+1 / 0.33
Which of the following are homosporous?
Q73.
mcq single
+1 / 0.33
**Assertion :** Selaginella and Salvinia are
homosporous.
**Reason :** In pteridophyte, Lycopodium is
precursor of seed habit.
Q74.
mcq single
+1 / 0.33
**Assertion :** Gastrin is a hormone that is
released from the gastrointestinal tract and
helps in digestion.
**Reason :** It promotes secretion of HCl and
trypsinogen.
Q75.
mcq single
+1 / 0.33
**Assertion :** Cholecystokinin is released by
duodenum.
**Reason :** It activates pepsinogen and bile
juice.
Q76.
mcq single
+1 / 0.33

 Identify A, B and C
Identify A, B and C

 Identify A, B and C
Identify A, B and C
Q77.
mcq single
+1 / 0.33
Select the wrong statement from the following.
Q78.
mcq single
+1 / 0.33
The genetic codes of arginine are
Q79.
mcq single
+1 / 0.33
**Assertion :** In eukaryotes, transcription
occurs in nucleus.
**Reason :** In bacteria, transcription and
translation occurs in cytoplasm.
Q80.
mcq single
+1 / 0.33
Gene library or DNA library has the collection of
Q81.
mcq single
+1 / 0.33
**Assertion :** Deficiency of an element may
lead to scurvy.
**Reason :** Daily requirement of ascorbic acid is
5 mg/day.
Q82.
mcq single
+1 / 0.33
**Assertion :** Cannabinoids are drugs of abuse.
**Reason :** They affect cardiovascular system
and central nervous system activity.
Q83.
mcq single
+1 / 0.33
Immunity tolerance developed by
Q84.
mcq single
+1 / 0.33
**Assertion :** Type-I diabetes is caused by
destruction of $$\beta$$-cells of islets of Langerhans.
**Reason :** Insulin can be taken as pills.
Q85.
mcq single
+1 / 0.33
 Select the correct option w.r.t. age pyramids.
Select the correct option w.r.t. age pyramids.
 Select the correct option w.r.t. age pyramids.
Select the correct option w.r.t. age pyramids.
Q86.
mcq single
+1 / 0.33
**Assertion :** In commensalism, one organism
is benefitted and other is unaffected.
**Reason :** Cattle egret bird and cattle is an
example of commensalism.
Q87.
mcq single
+1 / 0.33
**Assertion :** SA node malfunctioning leads to
disturbance of heart rate.
**Reason :** SA node is the pacemaker of heart
producing electric impulse for heart
contraction.
Q88.
mcq single
+1 / 0.33
What is the function of Bowman's capsule and glomerulus?
Q89.
mcq single
+1 / 0.33
**Assertion :** Pituitary gland releases a
hormone which is helpful in childbirth.
**Reason :** Pituitary gland releases vasopressin
and anti-diuretic hormone which helps in
childbirth.
Q90.
mcq single
+1 / 0.33
Choose the correct option.


Q91.
mcq single
+1 / 0.33
Oxytocin and ADH are produced by hypothalamus and released from
Q92.
mcq single
+1 / 0.33
**Assertion :** Biofortification is used to
increase nutrient value of crops.
**Reason :** Meristem culture is used to obtain
virus resistant plants.
Q93.
mcq single
+1 / 0.33
King of spices is
Q94.
mcq single
+1 / 0.33
**Assertion :** Biofortified crop is a source of
higher proteins, minerals and healthier fats.
**Reason :** Azolla is biofertiliser.
Q95.
mcq single
+1 / 0.33
**Assertion :** Gibberellin is useful in early seed
production in conifers.
**Reason :** Ethephon is responsible for early
ripening in tomato and apple.
Q96.
mcq single
+1 / 0.33
If mitochondria is absent in mature RBC,
what will be the source of energy?
Q97.
mcq single
+1 / 0.33
ATP formation occurs through which of the following?
Q98.
mcq single
+1 / 0.33
**Assertion :** Respiratory pathway is
considered as an amphibolic pathway.
**Reason :** It involves both anabolism and
catabolism.
Q99.
mcq single
+1 / 0.33
Cervical vertebrae differ from other vertebrae in having
Q100.
mcq single
+1 / 0.33
**Assertion :** Calcium required for skeletal
muscle contraction.
**Reason :** Calcium influx releases
acetylcholine at neuromuscular junction.
Q101.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
Chrysophyte
(i)
Gonyaulax
B.
Dinoflagellate
(ii)
Euglena
C.
Euglenoids
(iii)
Diatom
D.
Slime moulds
(iv)
Plasmodium
Q102.
mcq single
+1 / 0.33
Which statement is correct?
Q103.
mcq single
+1 / 0.33
**Assertion :** Malpighian tubules are excretory
organs in most of the insects.
**Reason :** These help in excretion of urea and
creatinine.
Q104.
mcq single
+1 / 0.33
The process of removal of anther from the
flower bud before it dehisces is called as
Q105.
mcq single
+1 / 0.33
**Assertion :** Parthenocarpy involves the
formation of seedless fruit.
**Reason :** Apomixis occurs without
fertilisation.
Q106.
mcq single
+1 / 0.33
Which of the following is correct?
Q107.
mcq single
+1 / 0.33
Which of the following represent zygomorphic symmetry?
Q108.
mcq single
+1 / 0.33
Which of the following is incorrect?
Q109.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
Tap root
(i)
Sweet potato
B.
Adventitious root
(ii)
Turnip
C.
Stem
(iii)
Wheat
D.
Fibrous root
(iv)
Potato
Q110.
mcq single
+1 / 0.33
Which of the following linkage is found in sucrose?
Q111.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
(Substrate)
Column II
(Enzyme)
(A)
Ribonucleotide
(i)
Chitinase
(B)
Chitin
(ii)
Cellulose
(C)
Cellulose
(iii)
Ribonuclease
Q112.
mcq single
+1 / 0.33
Glucose on reacting with Benedict's solution may give the following precipitates except
Q113.
mcq single
+1 / 0.33
Which of the following are all nucleotides?
Q114.
mcq single
+1 / 0.33
Protein on reaction with which yields Ruhemann's purple?
Q115.
mcq single
+1 / 0.33
Virus free plants can be formed by
Q116.
mcq single
+1 / 0.33
Hairy root disease of dicot plants is caused by
Q117.
mcq single
+1 / 0.33
What are the requirements in tissue culture?
Q118.
mcq single
+1 / 0.33
Chimeric DNA is
Q119.
mcq single
+1 / 0.33
Match the following columns and choose the
correct option from the codes given below.
Column I
Column II
A.
KC mehta
(i)
Fluid mosaic model
B.
P Maheshwari
(ii)
First recombinant plasmid
C.
Cohen and Boyer
(iii)
Haploid culture
D.
Singer and Nicolson
(iv)
Rust disease
Q120.
mcq single
+1 / 0.33
In Ti-plasmid, which of the following is removed?