JEE Main 2026 (Online) 21st January Morning Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
numerical
+4 / 1
Consider the following reaction sequence
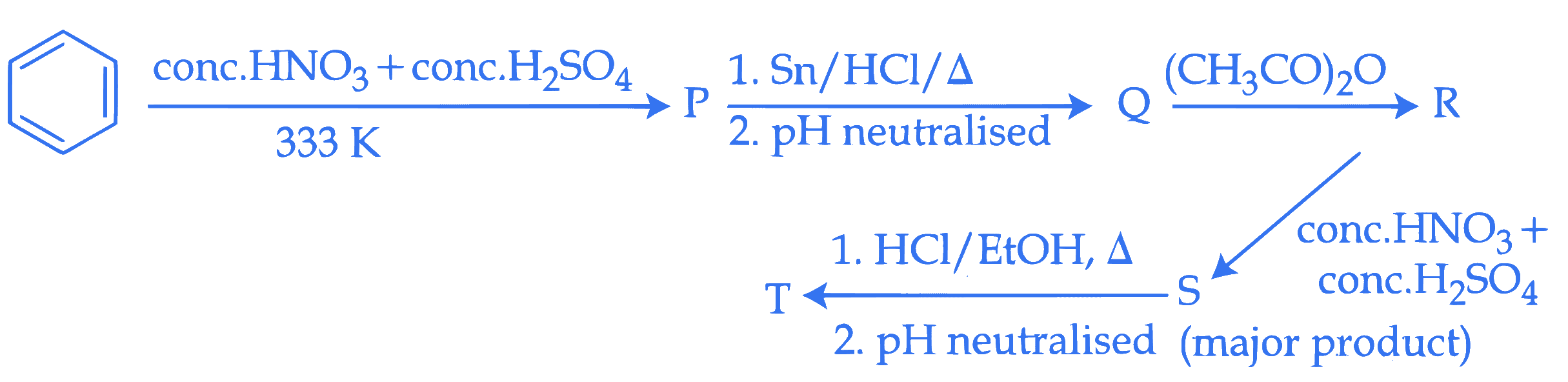 The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
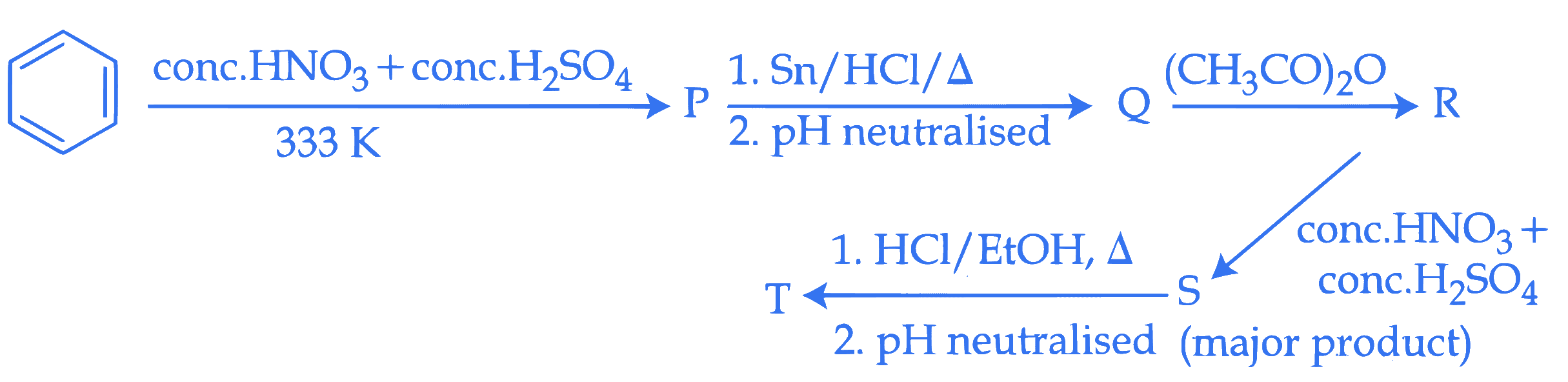 The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
Q2.
mcq single
+4 / 1
A hydrocarbon ' P ' $\left(\mathrm{C}_4 \mathrm{H}_8\right)$ on reaction with HCl gives an optically active compound ' Q ' $\left(\mathrm{C}_4 \mathrm{H}_9 \mathrm{Cl}\right)$ which on reaction with one mole of ammonia gives compound ' $\mathrm{R}^{\prime}\left(\mathrm{C}_4 \mathrm{H}_{11} \mathrm{~N}\right)$. ' $\mathrm{R}^{\prime}$ on diazotization followed by hydrolysis gives ' S '. Identify $\mathrm{P}, \mathrm{Q}, \mathrm{R}$ and S .
Q3.
mcq single
+4 / 1
An organic compound $(\mathrm{P})$ on treatment with aqueous ammonia under hot condition forms compound $(\mathrm{Q})$ which on heating with $\mathrm{Br}_2$ and KOH forms compound $(\mathrm{R})$ having molecular formula $\mathrm{C}_6 \mathrm{H}_7 \mathrm{~N}$. Names of P, Q and R respectively are.
Q4.
numerical
+4 / 1
Consider the following reactions :
$$ \begin{aligned} & \mathrm{NaCl}+\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{~A}+\mathrm{KHSO}_4+\mathrm{NaHSO}_4+\mathrm{H}_2 \mathrm{O} \\ & \mathrm{~A}+\mathrm{NaOH} \rightarrow \mathrm{~B}+\mathrm{NaCl}+\mathrm{H}_2 \mathrm{O} \\ & \mathrm{~B}+\mathrm{H}_2 \mathrm{SO}_4+\mathrm{H}_2 \mathrm{O}_2 \rightarrow \mathrm{C}+\mathrm{Na}_2 \mathrm{SO}_4+\mathrm{H}_2 \mathrm{O} \end{aligned} $$
In the product ' $C^{\prime}$, ' $X$ ' is the number of $O_2^{2-}$ units, ' $Y$ ' is the total number oxygen atoms present and ' $Z$ ' is the oxidation state of $C r$. The value of $X+Y+Z$ is $\_\_\_\_$ .
Q5.
mcq single
+4 / 1
$\mathrm{MnO}_4{ }^{2-}$, in acidic medium, disproportionates to :
Q6.
numerical
+4 / 1
Pre-exponential factors of two different reactions of same order are identical. Let activation energy of first reaction exceeds the activation energy of second reaction by $20 \mathrm{~kJ} \mathrm{~mol}^{-1}$. If $\mathrm{k}_1$ and $\mathrm{k}_2$ are the rate constants of first and second reaction respectively at 300 K , then $\ln \frac{\mathrm{k}_2}{\mathrm{k}_1}$ will be $\_\_\_\_$ . (nearest integer) $\left[\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right]$
Q7.
mcq single
+4 / 1
Given below are two statements :
Statement I :
The number of species among $\mathrm{SF}_4, \mathrm{NH}_4^{+},\left[\mathrm{NiCl}_4\right]^{2-}, \mathrm{XeF}_4,\left[\mathrm{PtCl}_4\right]^{2-}, \mathrm{SeF}_4$ and $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}$, that have tetrahedral geometry is 3 .
Statement II :
In the set [ $\mathrm{NO}_2, \mathrm{BeH}_2, \mathrm{BF}_3, \mathrm{AlCl}_3$ ], all the molecules have incomplete octet around central atom.
In the light of the above statements, choose the correct answer from the options given below:
Q8.
mcq single
+4 / 1
Elements P and Q form two types of non-volatile, non-ionizable compounds PQ and $\mathrm{PQ}_2$. When 1 g of $P Q$ is dissolved in 50 g of solvent ' $A^{\prime}, \Delta T_b$ was 1.176 K while when 1 g of $P Q_2$ is dissolved in 50 g of solvent ' $\mathrm{A}^{\prime}, \Delta \mathrm{T}_{\mathrm{b}}$ was 0.689 K . ( $\mathrm{K}_{\mathrm{b}}$ of ' $\mathrm{A}^{\prime}=5 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$ ). The molar masses of elements P and Q (in $\mathrm{g} \mathrm{mol}^{-1}$ ) respectively, are :
Q9.
mcq single
+4 / 1
14.0 g of calcium metal is allowed to react with excess HCl at 1.0 atm pressure and 273 K . Which of the following statements is incorrect?
[Given : Molar mass in $\mathrm{g} \mathrm{mol}^{-1}$ of $\mathrm{Ca}-40, \mathrm{Cl}-35.5, \mathrm{H}-1$ ]
Q10.
mcq single
+4 / 1
Which of the following represents the correct trend for the mentioned property?
**A.** F > P > S > B - First Ionization Energy
**B.** Cl > F > S > P - Electron Affinity
**C.** K > Al > Mg > B - Metallic character
**D.** K~2~O > Na~2~O > MgO > Al~2~O~3~ - Basic character
Choose the **correct** answer from the options given below :
Q11.
mcq single
+4 / 1
For the reaction, $\mathrm{N}_2 \mathrm{O}_4 \rightleftharpoons 2 \mathrm{NO}_2$, graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is $-5.40 \mathrm{~kJ} \mathrm{~mol}^{-1}$.
B. As $\Delta \mathrm{G}^{\ominus}$ in graph is positive, $\mathrm{N}_2 \mathrm{O}_4$ will not dissociate into $\mathrm{NO}_2$ at all.
C. Reverse reaction will go to completion.
D. When 1 mole of $\mathrm{N}_2 \mathrm{O}_4$ changes into equilibrium mixture, value of $\Delta \mathrm{G}^{\ominus}=-0.84 \mathrm{~kJ} \mathrm{~mol}^{-1}$
E. When 2 mole of $\mathrm{NO}_2$ changes into equilibrium mixture, $\Delta \mathrm{G}^{\ominus}$ for equilibrium mixture is $-6.24 \mathrm{~kJ} \mathrm{~mol}^{-1}$.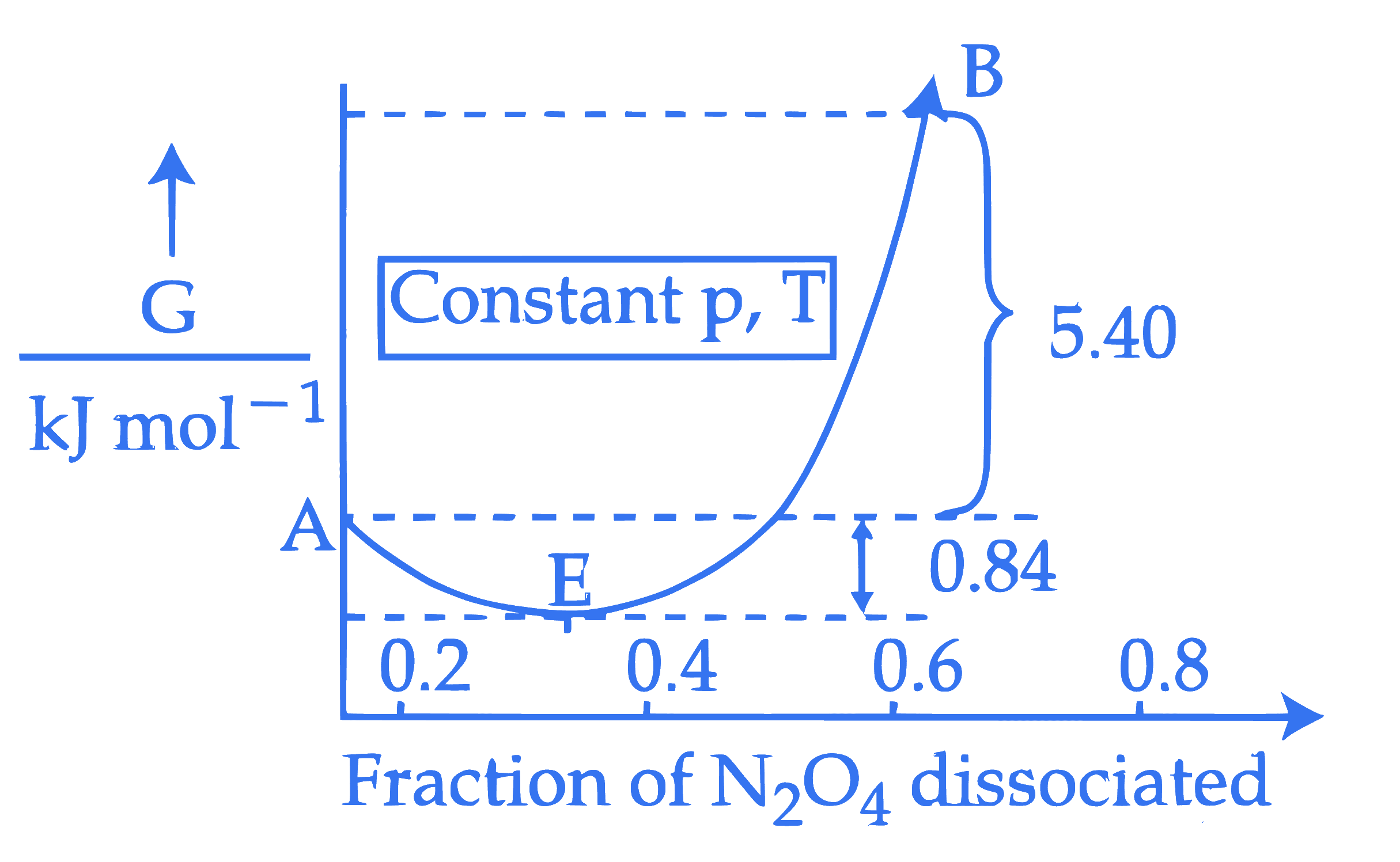 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q12.
numerical
+4 / 1
$$ \begin{aligned} &\text { Use the following data : }\\ &\begin{array}{|c|c|c|} \hline \text { Substance } & \frac{\Delta_f \mathrm{H}^{\ominus}(500 \mathrm{~K})}{\mathrm{kJ} \mathrm{~mol}^{-1}} & \frac{\mathrm{~S}^{\ominus}(500 \mathrm{~K})}{\mathrm{JK}^{-1} \mathrm{~mol}^{-1}} \\ \hline \mathrm{AB}(\mathrm{~g}) & 32 & 222 \\ \hline \mathrm{~A}_2(\mathrm{~g}) & 6 & 146 \\ \hline \mathrm{~B}_2(\mathrm{~g}) & x & 280 \\ \hline \end{array} \end{aligned} $$
One mole each of $\mathrm{A}_2(\mathrm{~g})$ and $\mathrm{B}_2(\mathrm{~g})$ are taken in a 1 L closed flask and allowed to establish the equilibrium at 500 K .
$$ \mathrm{A}_2(\mathrm{~g})+\mathrm{B}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{AB}(\mathrm{~g}) $$
The value of $x\left(\mathrm{in} \mathrm{kJ} \mathrm{mol}^{-1}\right)$ is $\_\_\_\_$ . (Nearest integer)
(Given : $\log \mathrm{K}=2.2 \quad \mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ )
Q13.
mcq single
+4 / 1
Which of the following graphs between pressure ' p ' versus volume ' V ' represents the maximum work done?
Q14.
mcq single
+4 / 1
Consider the following reactions.
$\underset{\text { (Hot solution) }}{\mathrm{PbCl}_2}+\mathrm{K}_2 \mathrm{CrO}_4 \rightarrow A+2 \mathrm{KCl}$
$$ \begin{aligned} & \mathrm{A}+\mathrm{NaOH} \rightleftharpoons \mathrm{~B}+\mathrm{Na}_2 \mathrm{CrO}_4 \\ & \mathrm{PbSO}_4+4 \mathrm{CH}_3 \mathrm{COONH}_4 \rightarrow\left(\mathrm{NH}_4\right)_2 \mathrm{SO}_4+\mathrm{X} \end{aligned} $$
In the above reactions, $\mathrm{A}, \mathrm{B}$ and X are respectively.
Q15.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of pairs among $\left[\mathrm{SiO}_2, \mathrm{CO}_2\right],\left[\mathrm{SnO}, \mathrm{SnO}_2\right],\left[\mathrm{PbO}, \mathrm{PbO}_2\right]$ and $\left[\mathrm{GeO}, \mathrm{GeO}_2\right]$, which contain oxides that are both amphoteric is 2 .
**Statement II :** $\mathrm{BF}_3$ is an electron deficient molecule, can act as a Lewis acid, forms adduct with $\mathrm{NH}_3$ and has a trigonal planar geometry.
In the light of the above statements, choose the correct answer from the options given below :
Q16.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Among $\left.\left[\mathrm{Cu}\left(\mathrm{NH}_3\right)_4\right]^{2+},\left[\mathrm{Ni}(\mathrm{en})_3\right)\right]^{2+},\left[\mathrm{Ni}\left(\mathrm{NH}_3\right)_6\right]^{2+}$ and $\left[\mathrm{Mn}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}$, $\left[\mathrm{Mn}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}$ has the maximum number of unpaired electrons.
**Statement II :** The number of pairs among $\left\{\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CO})_4\right]\right\},\left\{\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}\right\}$ and $\left\{\left[\mathrm{Ni}(\mathrm{CO})_4\right],\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}\right\}$ that contain only diamagnetic species is two.
In the light of the above statements, choose the correct answer from the options given below :
Q17.
mcq single
+4 / 1
An organic compound " P " of molecular formula $\mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_3$ gives positive Iodoform test but negative Tollen's test. When " P " is treated with dilute acid, it produces " Q ". " Q " gives positive Tollen's test and also iodoform test. The structure of " P " is :
Q18.
mcq single
+4 / 1
$$ \text { Identify A in the following reaction. } $$

Q19.
mcq single
+4 / 1
In Carius method, 0.75 g of an organic compound gave 1.2 g of barium sulphate, find percentage of sulphur (molar mass $32 \mathrm{~g} \mathrm{~mol}^{-1}$ ). Molar mass of barium sulphate is $233 \mathrm{~g} \mathrm{~mol}^{-1}$.
Q20.
mcq single
+4 / 1
80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual gases after cooling to 273 K occupy 224 mL . When the system is treated with KOH solution, the volume decreases to 64 mL . The formula of the hydrocarbon is :
Q21.
mcq single
+4 / 1
Identify the correct statements.
A. Arginine and Tryptophan are essential amino acids.
B. Histidine does not contain heterocyclic ring in its structure.
C. Proline is a six membered cyclic ring amino acid.
D. Glycine does not have chiral centre.
E. Cysteine has characteristic feature of side chain as $\mathrm{MeS}-\mathrm{CH}_2-\mathrm{CH}_2-$.
Choose the correct answer from the options given below :
Q22.
mcq single
+4 / 1
Identify **correct** statements from the following :
A. Propanal and propanone are functional isomers.
B. Ethoxyethane and methoxypropane are metamers.
C. But-2-ene shows optical isomerism.
D. But-1-ene and but-2-ene are functional isomers.
E. Pentane and 2, 2-dimethyl propane are chain isomers.
Choose the **correct** answer from the options given below :
Q23.
mcq single
+4 / 1
$$ \text { From the following, the least stable structure is : } $$
Q24.
numerical
+4 / 1
The pH and conductance of a weak acid $(\mathrm{HX})$ was found to be 5 and $4 \times 10^{-5} \mathrm{~S}$, respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of $1 \mathrm{~cm}^2$ were at a distance of 15 cm apart. The value of the limiting molar conductivity is $\_\_\_\_$ $\mathrm{S} \mathrm{m}^2 \mathrm{~mol}^{-1}$. (nearest integer)
(Given : degree of dissociation of the weak acid $(\alpha) \ll 1$ )
Q25.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** When an electric discharge is passed through gaseous hydrogen, the hydrogen molecules dissociate and the energetically excited hydrogen atoms produce electromagnetic radiation of discrete frequencies.
**Statement II :** The frequency of second line of Balmer series obtained from $\mathrm{He}^{+}$is equal to that of first line of Lyman series obtained from hydrogen atom.
In the light of the above statements, choose the correct answer from the options given below :
Q26.
numerical
+4 / 1
Consider the following reaction sequence
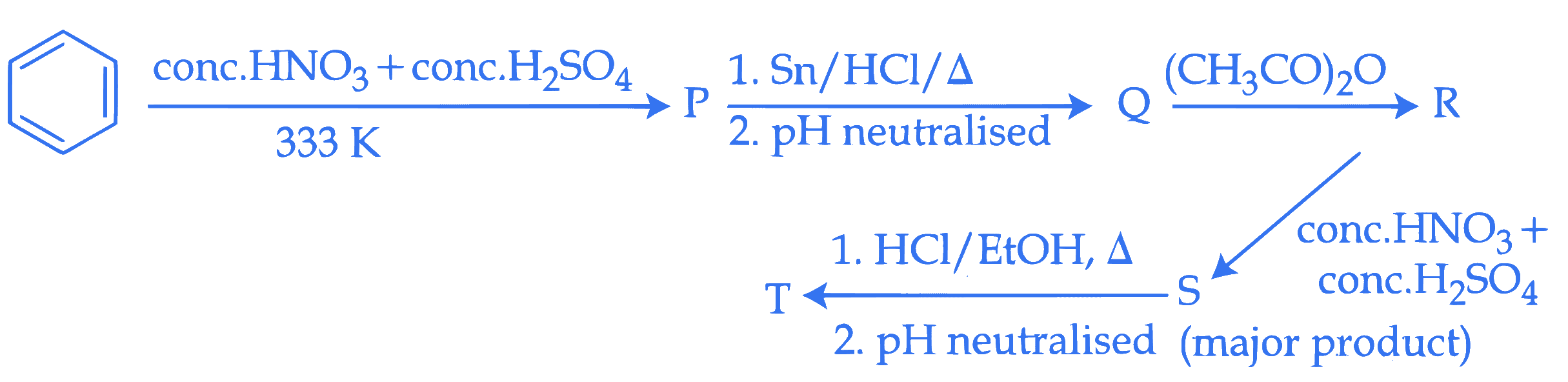 The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
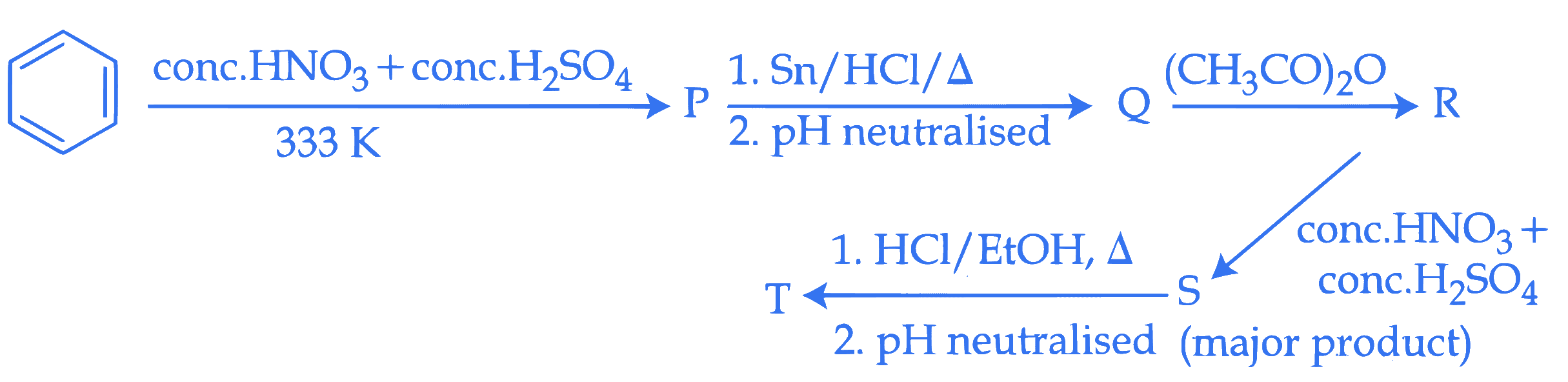 The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
The percentage of nitrogen in product ' T ' formed is $\_\_\_\_$ %. (Nearest integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
Q27.
mcq single
+4 / 1
A hydrocarbon ' P ' $\left(\mathrm{C}_4 \mathrm{H}_8\right)$ on reaction with HCl gives an optically active compound ' Q ' $\left(\mathrm{C}_4 \mathrm{H}_9 \mathrm{Cl}\right)$ which on reaction with one mole of ammonia gives compound ' $\mathrm{R}^{\prime}\left(\mathrm{C}_4 \mathrm{H}_{11} \mathrm{~N}\right)$. ' $\mathrm{R}^{\prime}$ on diazotization followed by hydrolysis gives ' S '. Identify $\mathrm{P}, \mathrm{Q}, \mathrm{R}$ and S .
Q28.
mcq single
+4 / 1
An organic compound $(\mathrm{P})$ on treatment with aqueous ammonia under hot condition forms compound $(\mathrm{Q})$ which on heating with $\mathrm{Br}_2$ and KOH forms compound $(\mathrm{R})$ having molecular formula $\mathrm{C}_6 \mathrm{H}_7 \mathrm{~N}$. Names of P, Q and R respectively are.
Q29.
numerical
+4 / 1
Consider the following reactions :
$$ \begin{aligned} & \mathrm{NaCl}+\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{~A}+\mathrm{KHSO}_4+\mathrm{NaHSO}_4+\mathrm{H}_2 \mathrm{O} \\ & \mathrm{~A}+\mathrm{NaOH} \rightarrow \mathrm{~B}+\mathrm{NaCl}+\mathrm{H}_2 \mathrm{O} \\ & \mathrm{~B}+\mathrm{H}_2 \mathrm{SO}_4+\mathrm{H}_2 \mathrm{O}_2 \rightarrow \mathrm{C}+\mathrm{Na}_2 \mathrm{SO}_4+\mathrm{H}_2 \mathrm{O} \end{aligned} $$
In the product ' $C^{\prime}$, ' $X$ ' is the number of $O_2^{2-}$ units, ' $Y$ ' is the total number oxygen atoms present and ' $Z$ ' is the oxidation state of $C r$. The value of $X+Y+Z$ is $\_\_\_\_$ .
Q30.
mcq single
+4 / 1
$\mathrm{MnO}_4{ }^{2-}$, in acidic medium, disproportionates to :
Q31.
numerical
+4 / 1
Pre-exponential factors of two different reactions of same order are identical. Let activation energy of first reaction exceeds the activation energy of second reaction by $20 \mathrm{~kJ} \mathrm{~mol}^{-1}$. If $\mathrm{k}_1$ and $\mathrm{k}_2$ are the rate constants of first and second reaction respectively at 300 K , then $\ln \frac{\mathrm{k}_2}{\mathrm{k}_1}$ will be $\_\_\_\_$ . (nearest integer) $\left[\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right]$
Q32.
mcq single
+4 / 1
Given below are two statements :
Statement I :
The number of species among $\mathrm{SF}_4, \mathrm{NH}_4^{+},\left[\mathrm{NiCl}_4\right]^{2-}, \mathrm{XeF}_4,\left[\mathrm{PtCl}_4\right]^{2-}, \mathrm{SeF}_4$ and $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}$, that have tetrahedral geometry is 3 .
Statement II :
In the set [ $\mathrm{NO}_2, \mathrm{BeH}_2, \mathrm{BF}_3, \mathrm{AlCl}_3$ ], all the molecules have incomplete octet around central atom.
In the light of the above statements, choose the correct answer from the options given below:
Q33.
mcq single
+4 / 1
Elements P and Q form two types of non-volatile, non-ionizable compounds PQ and $\mathrm{PQ}_2$. When 1 g of $P Q$ is dissolved in 50 g of solvent ' $A^{\prime}, \Delta T_b$ was 1.176 K while when 1 g of $P Q_2$ is dissolved in 50 g of solvent ' $\mathrm{A}^{\prime}, \Delta \mathrm{T}_{\mathrm{b}}$ was 0.689 K . ( $\mathrm{K}_{\mathrm{b}}$ of ' $\mathrm{A}^{\prime}=5 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$ ). The molar masses of elements P and Q (in $\mathrm{g} \mathrm{mol}^{-1}$ ) respectively, are :
Q34.
mcq single
+4 / 1
14.0 g of calcium metal is allowed to react with excess HCl at 1.0 atm pressure and 273 K . Which of the following statements is incorrect?
[Given : Molar mass in $\mathrm{g} \mathrm{mol}^{-1}$ of $\mathrm{Ca}-40, \mathrm{Cl}-35.5, \mathrm{H}-1$ ]
Q35.
mcq single
+4 / 1
Which of the following represents the correct trend for the mentioned property?
**A.** F > P > S > B - First Ionization Energy
**B.** Cl > F > S > P - Electron Affinity
**C.** K > Al > Mg > B - Metallic character
**D.** K~2~O > Na~2~O > MgO > Al~2~O~3~ - Basic character
Choose the **correct** answer from the options given below :
Q36.
mcq single
+4 / 1
For the reaction, $\mathrm{N}_2 \mathrm{O}_4 \rightleftharpoons 2 \mathrm{NO}_2$, graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is $-5.40 \mathrm{~kJ} \mathrm{~mol}^{-1}$.
B. As $\Delta \mathrm{G}^{\ominus}$ in graph is positive, $\mathrm{N}_2 \mathrm{O}_4$ will not dissociate into $\mathrm{NO}_2$ at all.
C. Reverse reaction will go to completion.
D. When 1 mole of $\mathrm{N}_2 \mathrm{O}_4$ changes into equilibrium mixture, value of $\Delta \mathrm{G}^{\ominus}=-0.84 \mathrm{~kJ} \mathrm{~mol}^{-1}$
E. When 2 mole of $\mathrm{NO}_2$ changes into equilibrium mixture, $\Delta \mathrm{G}^{\ominus}$ for equilibrium mixture is $-6.24 \mathrm{~kJ} \mathrm{~mol}^{-1}$. Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q37.
numerical
+4 / 1
$$ \begin{aligned} &\text { Use the following data : }\\ &\begin{array}{|c|c|c|} \hline \text { Substance } & \frac{\Delta_f \mathrm{H}^{\ominus}(500 \mathrm{~K})}{\mathrm{kJ} \mathrm{~mol}^{-1}} & \frac{\mathrm{~S}^{\ominus}(500 \mathrm{~K})}{\mathrm{JK}^{-1} \mathrm{~mol}^{-1}} \\ \hline \mathrm{AB}(\mathrm{~g}) & 32 & 222 \\ \hline \mathrm{~A}_2(\mathrm{~g}) & 6 & 146 \\ \hline \mathrm{~B}_2(\mathrm{~g}) & x & 280 \\ \hline \end{array} \end{aligned} $$
One mole each of $\mathrm{A}_2(\mathrm{~g})$ and $\mathrm{B}_2(\mathrm{~g})$ are taken in a 1 L closed flask and allowed to establish the equilibrium at 500 K .
$$ \mathrm{A}_2(\mathrm{~g})+\mathrm{B}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{AB}(\mathrm{~g}) $$
The value of $x\left(\mathrm{in} \mathrm{kJ} \mathrm{mol}^{-1}\right)$ is $\_\_\_\_$ . (Nearest integer)
(Given : $\log \mathrm{K}=2.2 \quad \mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ )
Q38.
mcq single
+4 / 1
Which of the following graphs between pressure ' p ' versus volume ' V ' represents the maximum work done?
Q39.
mcq single
+4 / 1
Consider the following reactions.
$\underset{\text { (Hot solution) }}{\mathrm{PbCl}_2}+\mathrm{K}_2 \mathrm{CrO}_4 \rightarrow A+2 \mathrm{KCl}$
$$ \begin{aligned} & \mathrm{A}+\mathrm{NaOH} \rightleftharpoons \mathrm{~B}+\mathrm{Na}_2 \mathrm{CrO}_4 \\ & \mathrm{PbSO}_4+4 \mathrm{CH}_3 \mathrm{COONH}_4 \rightarrow\left(\mathrm{NH}_4\right)_2 \mathrm{SO}_4+\mathrm{X} \end{aligned} $$
In the above reactions, $\mathrm{A}, \mathrm{B}$ and X are respectively.
Q40.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of pairs among $\left[\mathrm{SiO}_2, \mathrm{CO}_2\right],\left[\mathrm{SnO}, \mathrm{SnO}_2\right],\left[\mathrm{PbO}, \mathrm{PbO}_2\right]$ and $\left[\mathrm{GeO}, \mathrm{GeO}_2\right]$, which contain oxides that are both amphoteric is 2 .
**Statement II :** $\mathrm{BF}_3$ is an electron deficient molecule, can act as a Lewis acid, forms adduct with $\mathrm{NH}_3$ and has a trigonal planar geometry.
In the light of the above statements, choose the correct answer from the options given below :
Q41.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Among $\left.\left[\mathrm{Cu}\left(\mathrm{NH}_3\right)_4\right]^{2+},\left[\mathrm{Ni}(\mathrm{en})_3\right)\right]^{2+},\left[\mathrm{Ni}\left(\mathrm{NH}_3\right)_6\right]^{2+}$ and $\left[\mathrm{Mn}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}$, $\left[\mathrm{Mn}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}$ has the maximum number of unpaired electrons.
**Statement II :** The number of pairs among $\left\{\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CO})_4\right]\right\},\left\{\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}\right\}$ and $\left\{\left[\mathrm{Ni}(\mathrm{CO})_4\right],\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}\right\}$ that contain only diamagnetic species is two.
In the light of the above statements, choose the correct answer from the options given below :
Q42.
mcq single
+4 / 1
An organic compound " P " of molecular formula $\mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_3$ gives positive Iodoform test but negative Tollen's test. When " P " is treated with dilute acid, it produces " Q ". " Q " gives positive Tollen's test and also iodoform test. The structure of " P " is :
Q43.
mcq single
+4 / 1
$$ \text { Identify A in the following reaction. } $$

Q44.
mcq single
+4 / 1
In Carius method, 0.75 g of an organic compound gave 1.2 g of barium sulphate, find percentage of sulphur (molar mass $32 \mathrm{~g} \mathrm{~mol}^{-1}$ ). Molar mass of barium sulphate is $233 \mathrm{~g} \mathrm{~mol}^{-1}$.
Q45.
mcq single
+4 / 1
80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual gases after cooling to 273 K occupy 224 mL . When the system is treated with KOH solution, the volume decreases to 64 mL . The formula of the hydrocarbon is :
Q46.
mcq single
+4 / 1
Identify the correct statements.
A. Arginine and Tryptophan are essential amino acids.
B. Histidine does not contain heterocyclic ring in its structure.
C. Proline is a six membered cyclic ring amino acid.
D. Glycine does not have chiral centre.
E. Cysteine has characteristic feature of side chain as $\mathrm{MeS}-\mathrm{CH}_2-\mathrm{CH}_2-$.
Choose the correct answer from the options given below :
Q47.
mcq single
+4 / 1
Identify **correct** statements from the following :
A. Propanal and propanone are functional isomers.
B. Ethoxyethane and methoxypropane are metamers.
C. But-2-ene shows optical isomerism.
D. But-1-ene and but-2-ene are functional isomers.
E. Pentane and 2, 2-dimethyl propane are chain isomers.
Choose the **correct** answer from the options given below :
Q48.
mcq single
+4 / 1
$$ \text { From the following, the least stable structure is : } $$
Q49.
numerical
+4 / 1
The pH and conductance of a weak acid $(\mathrm{HX})$ was found to be 5 and $4 \times 10^{-5} \mathrm{~S}$, respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of $1 \mathrm{~cm}^2$ were at a distance of 15 cm apart. The value of the limiting molar conductivity is $\_\_\_\_$ $\mathrm{S} \mathrm{m}^2 \mathrm{~mol}^{-1}$. (nearest integer)
(Given : degree of dissociation of the weak acid $(\alpha) \ll 1$ )
Q50.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** When an electric discharge is passed through gaseous hydrogen, the hydrogen molecules dissociate and the energetically excited hydrogen atoms produce electromagnetic radiation of discrete frequencies.
**Statement II :** The frequency of second line of Balmer series obtained from $\mathrm{He}^{+}$is equal to that of first line of Lyman series obtained from hydrogen atom.
In the light of the above statements, choose the correct answer from the options given below :
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{c}}$ and $\overrightarrow{\mathrm{d}}$ be vectors such that $|\overrightarrow{\mathrm{c}}+\overrightarrow{\mathrm{d}}|=\sqrt{29}$ and $\overrightarrow{\mathrm{c}} \times(2 \hat{i}+3 \hat{j}+4 \hat{k})=(2 \hat{i}+3 \hat{j}+4 \hat{k}) \times \overrightarrow{\mathrm{d}}$. If $\lambda_1, \lambda_2\left(\lambda_1>\lambda_2\right)$ are the possible values of $(\vec{c}+\vec{d}) \cdot(-7 \hat{i}+2 \hat{j}+3 \hat{k})$, then the equation $\mathrm{K}^2 x^2+\left(\mathrm{K}^2-5 \mathrm{~K}+\lambda_1\right) x y+\left(3 \mathrm{~K}+\frac{\lambda_2}{2}\right) y^2-8 x+12 y+\lambda_2=0$ represents a circle, for K equal to :
Q2.
mcq single
+4 / 1
Let $(\alpha, \beta, \gamma)$ be the co-ordinates of the foot of the perpendicular drawn from the point $(5,4,2)$ on the line $\overrightarrow{\mathrm{r}}=(-\hat{i}+3 \hat{j}+\hat{k})+\lambda(2 \hat{i}+3 \hat{j}-\hat{k})$.
Then the length of the projection of the vector $\alpha \hat{i}+\beta \hat{j}+\gamma \hat{k}$ on the vector $6 \hat{i}+2 \hat{j}+3 \hat{k}$ is :
Q3.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=-\hat{i}+2 \hat{j}+2 \hat{k}, \overrightarrow{\mathrm{~b}}=8 \hat{i}+7 \hat{j}-3 \hat{k}$ and $\overrightarrow{\mathrm{c}}$ be a vector such that $\overrightarrow{\mathrm{a}} \times \overrightarrow{\mathrm{c}}=\overrightarrow{\mathrm{b}}$. If $\vec{c} \cdot(\hat{i}+\hat{j}+\hat{k})=4$, then $|\vec{a}+\vec{c}|^2$ is equal to :
Q4.
mcq single
+4 / 1
The sum of all the roots of the equation $(x-1)^2-5|x-1|+6=0$, is :
Q5.
mcq single
+4 / 1
Let the mean and variance of 7 observations $2,4,10, x, 12,14, y, x>y$, be 8 and 16 respectively. Two numbers are chosen from $\{1,2,3, x-4, y, 5\}$ one after another without replacement, then the probability, that the smaller number among the two chosen numbers is less than 4 , is :
Q6.
mcq single
+4 / 1
Let $a_1, a_2, a_3, \ldots$ be a G.P. of increasing positive terms such that $a_2 \cdot a_3 \cdot a_4=64$ and $a_1+a_3+a_5=\frac{813}{7}$. Then $a_3+a_5+a_7$ is equal to :
Q7.
numerical
+4 / 1
Let $a_1=1$ and for $n \geqslant 1, a_{n+1}=\frac{1}{2} a_n+\frac{n^2-2 n-1}{n^2(n+1)^2}$. Then $\left|\sum_{n=1}^{\infty}\left(a_n-\frac{2}{n^2}\right)\right|$ is equal to $\_\_\_\_$ .
Q8.
mcq single
+4 / 1
Let $f: \mathbf{R} \rightarrow(0, \infty)$ be a twice differentiable function such that $f(3)=18, f^{\prime}(3)=0$ and $f^{\prime \prime}(3)=4$.
Then $\lim\limits _{x \rightarrow 1}\left(\log _e\left(\frac{f(2+x)}{f(3)}\right)^{\frac{18}{(x-1)^2}}\right)$ is equal to :
Q9.
numerical
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be a twice differentiable function such that the quadratic equation $f(x) \mathrm{m}^2-2 f^{\prime}(x) \mathrm{m}+f^{\prime \prime}(x)=0$ in m , has two equal roots for every $x \in \mathbf{R}$. If $f(0)=1, f^{\prime}(0)=2$, and ( $\alpha, \beta$ ) is the largest interval in which the function $f\left(\log _{\mathrm{e}} x-x\right)$ is increasing, then $\alpha+\beta$ is equal to
$\_\_\_\_$ .
Q10.
mcq single
+4 / 1
The value of $\operatorname{cosec} 10^{\circ}-\sqrt{3} \sec 10^{\circ}$ is equal to :
Q11.
mcq single
+4 / 1
The area of the region, inside the ellipse $x^2+4 y^2=4$ and outside the region bounded by the curves $y=|x|-1$ and $y=1-|x|$, is :
Q12.
numerical
+4 / 1
For some $\alpha, \beta \in \mathbf{R}$, let $A=\left[\begin{array}{ll}\alpha & 2 \\ 1 & 2\end{array}\right]$ and $B=\left[\begin{array}{ll}1 & 1 \\ 1 & \beta\end{array}\right]$ be such that $A^2-4 A+2 I=B^2-3 B+I=O$. Then $\left(\operatorname{det}\left(\operatorname{adj}\left(A^3-B^3\right)\right)\right)^2$ is equal to $\_\_\_\_$ .
Q13.
mcq single
+4 / 1
Let O be the vertex of the parabola $x^2=4 y$ and Q be any point on it. Let the locus of the point P , which divides the line segment OQ internally in the ratio $2: 3$ be the conic C . Then the equation of the chord of $C$, which is bisected at the point $(1,2)$, is :
Q14.
mcq single
+4 / 1
If the domain of the function $f(x)=\cos ^{-1}\left(\frac{2 x-5}{11-3 x}\right)+\sin ^{-1}\left(2 x^2-3 x+1\right)$ is the interval $[\alpha, \beta]$, then $\alpha+2 \beta$ is equal to :
Q15.
mcq single
+4 / 1
If the coefficient of $x$ in the expansion of $\left(a x^2+b x+c\right)(1-2 x)^{26}$ is -56 and the coefficients of $x^2$ and $x^3$ are both zero, then $\mathrm{a}+\mathrm{b}+\mathrm{c}$ is equal to :
Q16.
mcq single
+4 / 1
Let a point A lie between the parallel lines $\mathrm{L}_1$ and $\mathrm{L}_2$ such that its distances from $\mathrm{L}_1$ and $\mathrm{L}_2$ are 6 and 3 units, respectively. Then the area (in sq. units) of the equilateral triangle ABC , where the points B and C lie on the lines $\mathrm{L}_1$ and $\mathrm{L}_2$, respectively, is :
Q17.
mcq single
+4 / 1
The number of relations, defined on the set $\{\mathrm{a}, \mathrm{b}, \mathrm{c}, \mathrm{d}\}$, which are both reflexive and symmetric, is equal to:
Q18.
mcq single
+4 / 1
If $x^2+x+1=0$, then the value of $\left(x+\frac{1}{x}\right)^4+\left(x^2+\frac{1}{x^2}\right)^4+\left(x^3+\frac{1}{x^3}\right)^4+\ldots+\left(x^{25}+\frac{1}{x^{25}}\right)^4$ is:
Q19.
mcq single
+4 / 1
Let the foci of a hyperbola coincide with the foci of the ellipse $\frac{x^2}{36}+\frac{y^2}{16}=1$. If the eccentricity of the hyperbola is 5 , then the length of its latus rectum is :
Q20.
mcq single
+4 / 1
Let $y=y(x)$ be the solution curve of the differential equation $\left(1+x^2\right) \mathrm{d} y+\left(y-\tan ^{-1} x\right) d x=0, y(0)=1$. Then the value of $y(1)$ is :
Q21.
mcq single
+4 / 1
Let PQ and MN be two straight lines touching the circle $x^2+y^2-4 x-6 y-3=0$ at the points $A$ and $B$ respectively. Let $O$ be the centre of the circle and $\angle A O B=\pi / 3$. Then the locus of the point of intersection of the lines PQ and MN is :
Q22.
mcq single
+4 / 1
The number of strictly increasing functions $f$ from the set $\{1,2,3,4,5,6\}$ to the set $\{1,2,3, \ldots ., 9\}$ such that $f(i) \neq i$ for $1 \leq i \leq 6$, is equal to :
Q23.
numerical
+4 / 1
Let $S=\{(m, n): m, n \in\{1,2,3, \ldots . ., 50\}\}$. If the number of elements $(m, n)$ in $S$ such that $6^m+9^n$ is a multiple of 5 is $p$ and the number of elements ( $m, n$ ) in $S$ such that $m+n$ is a square of a prime number is q , then $\mathrm{p}+\mathrm{q}$ is equal to $\_\_\_\_$ .
Q24.
numerical
+4 / 1
$6 \int_0^\pi|(\sin 3 x+\sin 2 x+\sin x)| d x$ is equal to $\_\_\_\_$ .
Q25.
mcq single
+4 / 1
The value of $\int\limits_{-\pi / 6}^{\pi / 6}\left(\frac{\pi+4 x^{11}}{1-\sin (|x|+\pi / 6)}\right) d x$ is equal to:
Q26.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{c}}$ and $\overrightarrow{\mathrm{d}}$ be vectors such that $|\overrightarrow{\mathrm{c}}+\overrightarrow{\mathrm{d}}|=\sqrt{29}$ and $\overrightarrow{\mathrm{c}} \times(2 \hat{i}+3 \hat{j}+4 \hat{k})=(2 \hat{i}+3 \hat{j}+4 \hat{k}) \times \overrightarrow{\mathrm{d}}$. If $\lambda_1, \lambda_2\left(\lambda_1>\lambda_2\right)$ are the possible values of $(\vec{c}+\vec{d}) \cdot(-7 \hat{i}+2 \hat{j}+3 \hat{k})$, then the equation $\mathrm{K}^2 x^2+\left(\mathrm{K}^2-5 \mathrm{~K}+\lambda_1\right) x y+\left(3 \mathrm{~K}+\frac{\lambda_2}{2}\right) y^2-8 x+12 y+\lambda_2=0$ represents a circle, for K equal to :
Q27.
mcq single
+4 / 1
Let $(\alpha, \beta, \gamma)$ be the co-ordinates of the foot of the perpendicular drawn from the point $(5,4,2)$ on the line $\overrightarrow{\mathrm{r}}=(-\hat{i}+3 \hat{j}+\hat{k})+\lambda(2 \hat{i}+3 \hat{j}-\hat{k})$.
Then the length of the projection of the vector $\alpha \hat{i}+\beta \hat{j}+\gamma \hat{k}$ on the vector $6 \hat{i}+2 \hat{j}+3 \hat{k}$ is :
Q28.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=-\hat{i}+2 \hat{j}+2 \hat{k}, \overrightarrow{\mathrm{~b}}=8 \hat{i}+7 \hat{j}-3 \hat{k}$ and $\overrightarrow{\mathrm{c}}$ be a vector such that $\overrightarrow{\mathrm{a}} \times \overrightarrow{\mathrm{c}}=\overrightarrow{\mathrm{b}}$. If $\vec{c} \cdot(\hat{i}+\hat{j}+\hat{k})=4$, then $|\vec{a}+\vec{c}|^2$ is equal to :
Q29.
mcq single
+4 / 1
The sum of all the roots of the equation $(x-1)^2-5|x-1|+6=0$, is :
Q30.
mcq single
+4 / 1
Let the mean and variance of 7 observations $2,4,10, x, 12,14, y, x>y$, be 8 and 16 respectively. Two numbers are chosen from $\{1,2,3, x-4, y, 5\}$ one after another without replacement, then the probability, that the smaller number among the two chosen numbers is less than 4 , is :
Q31.
mcq single
+4 / 1
Let $a_1, a_2, a_3, \ldots$ be a G.P. of increasing positive terms such that $a_2 \cdot a_3 \cdot a_4=64$ and $a_1+a_3+a_5=\frac{813}{7}$. Then $a_3+a_5+a_7$ is equal to :
Q32.
numerical
+4 / 1
Let $a_1=1$ and for $n \geqslant 1, a_{n+1}=\frac{1}{2} a_n+\frac{n^2-2 n-1}{n^2(n+1)^2}$. Then $\left|\sum_{n=1}^{\infty}\left(a_n-\frac{2}{n^2}\right)\right|$ is equal to $\_\_\_\_$ .
Q33.
mcq single
+4 / 1
Let $f: \mathbf{R} \rightarrow(0, \infty)$ be a twice differentiable function such that $f(3)=18, f^{\prime}(3)=0$ and $f^{\prime \prime}(3)=4$.
Then $\lim\limits _{x \rightarrow 1}\left(\log _e\left(\frac{f(2+x)}{f(3)}\right)^{\frac{18}{(x-1)^2}}\right)$ is equal to :
Q34.
numerical
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be a twice differentiable function such that the quadratic equation $f(x) \mathrm{m}^2-2 f^{\prime}(x) \mathrm{m}+f^{\prime \prime}(x)=0$ in m , has two equal roots for every $x \in \mathbf{R}$. If $f(0)=1, f^{\prime}(0)=2$, and ( $\alpha, \beta$ ) is the largest interval in which the function $f\left(\log _{\mathrm{e}} x-x\right)$ is increasing, then $\alpha+\beta$ is equal to
$\_\_\_\_$ .
Q35.
mcq single
+4 / 1
The value of $\operatorname{cosec} 10^{\circ}-\sqrt{3} \sec 10^{\circ}$ is equal to :
Q36.
mcq single
+4 / 1
The area of the region, inside the ellipse $x^2+4 y^2=4$ and outside the region bounded by the curves $y=|x|-1$ and $y=1-|x|$, is :
Q37.
numerical
+4 / 1
For some $\alpha, \beta \in \mathbf{R}$, let $A=\left[\begin{array}{ll}\alpha & 2 \\ 1 & 2\end{array}\right]$ and $B=\left[\begin{array}{ll}1 & 1 \\ 1 & \beta\end{array}\right]$ be such that $A^2-4 A+2 I=B^2-3 B+I=O$. Then $\left(\operatorname{det}\left(\operatorname{adj}\left(A^3-B^3\right)\right)\right)^2$ is equal to $\_\_\_\_$ .
Q38.
mcq single
+4 / 1
Let O be the vertex of the parabola $x^2=4 y$ and Q be any point on it. Let the locus of the point P , which divides the line segment OQ internally in the ratio $2: 3$ be the conic C . Then the equation of the chord of $C$, which is bisected at the point $(1,2)$, is :
Q39.
mcq single
+4 / 1
If the domain of the function $f(x)=\cos ^{-1}\left(\frac{2 x-5}{11-3 x}\right)+\sin ^{-1}\left(2 x^2-3 x+1\right)$ is the interval $[\alpha, \beta]$, then $\alpha+2 \beta$ is equal to :
Q40.
mcq single
+4 / 1
If the coefficient of $x$ in the expansion of $\left(a x^2+b x+c\right)(1-2 x)^{26}$ is -56 and the coefficients of $x^2$ and $x^3$ are both zero, then $\mathrm{a}+\mathrm{b}+\mathrm{c}$ is equal to :
Q41.
mcq single
+4 / 1
Let a point A lie between the parallel lines $\mathrm{L}_1$ and $\mathrm{L}_2$ such that its distances from $\mathrm{L}_1$ and $\mathrm{L}_2$ are 6 and 3 units, respectively. Then the area (in sq. units) of the equilateral triangle ABC , where the points B and C lie on the lines $\mathrm{L}_1$ and $\mathrm{L}_2$, respectively, is :
Q42.
mcq single
+4 / 1
The number of relations, defined on the set $\{\mathrm{a}, \mathrm{b}, \mathrm{c}, \mathrm{d}\}$, which are both reflexive and symmetric, is equal to:
Q43.
mcq single
+4 / 1
If $x^2+x+1=0$, then the value of $\left(x+\frac{1}{x}\right)^4+\left(x^2+\frac{1}{x^2}\right)^4+\left(x^3+\frac{1}{x^3}\right)^4+\ldots+\left(x^{25}+\frac{1}{x^{25}}\right)^4$ is:
Q44.
mcq single
+4 / 1
Let the foci of a hyperbola coincide with the foci of the ellipse $\frac{x^2}{36}+\frac{y^2}{16}=1$. If the eccentricity of the hyperbola is 5 , then the length of its latus rectum is :
Q45.
mcq single
+4 / 1
Let $y=y(x)$ be the solution curve of the differential equation $\left(1+x^2\right) \mathrm{d} y+\left(y-\tan ^{-1} x\right) d x=0, y(0)=1$. Then the value of $y(1)$ is :
Q46.
mcq single
+4 / 1
Let PQ and MN be two straight lines touching the circle $x^2+y^2-4 x-6 y-3=0$ at the points $A$ and $B$ respectively. Let $O$ be the centre of the circle and $\angle A O B=\pi / 3$. Then the locus of the point of intersection of the lines PQ and MN is :
Q47.
mcq single
+4 / 1
The number of strictly increasing functions $f$ from the set $\{1,2,3,4,5,6\}$ to the set $\{1,2,3, \ldots ., 9\}$ such that $f(i) \neq i$ for $1 \leq i \leq 6$, is equal to :
Q48.
numerical
+4 / 1
Let $S=\{(m, n): m, n \in\{1,2,3, \ldots . ., 50\}\}$. If the number of elements $(m, n)$ in $S$ such that $6^m+9^n$ is a multiple of 5 is $p$ and the number of elements ( $m, n$ ) in $S$ such that $m+n$ is a square of a prime number is q , then $\mathrm{p}+\mathrm{q}$ is equal to $\_\_\_\_$ .
Q49.
numerical
+4 / 1
$6 \int_0^\pi|(\sin 3 x+\sin 2 x+\sin x)| d x$ is equal to $\_\_\_\_$ .
Q50.
mcq single
+4 / 1
The value of $\int\limits_{-\pi / 6}^{\pi / 6}\left(\frac{\pi+4 x^{11}}{1-\sin (|x|+\pi / 6)}\right) d x$ is equal to:
Physics
Physics
Q1.
mcq single
+4 / 1
Potential energy ( $V$ ) versus distance ( $x$ ) is given by the graph. Rank various regions as per the magnitudes of the force ( $F$ ) acting on a particle from high to low.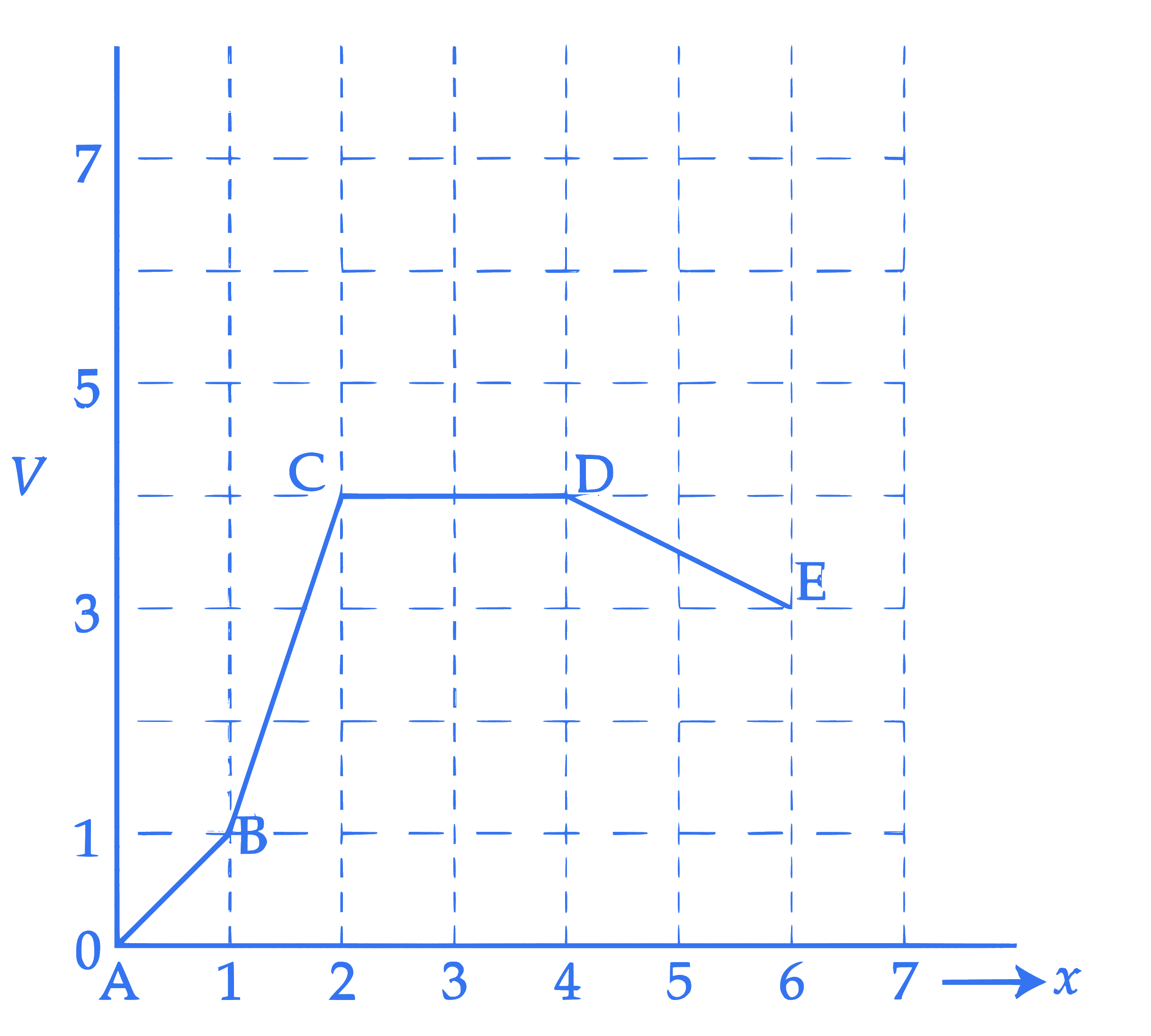

Q2.
mcq single
+4 / 1
$$ \text { The given circuit works as : } $$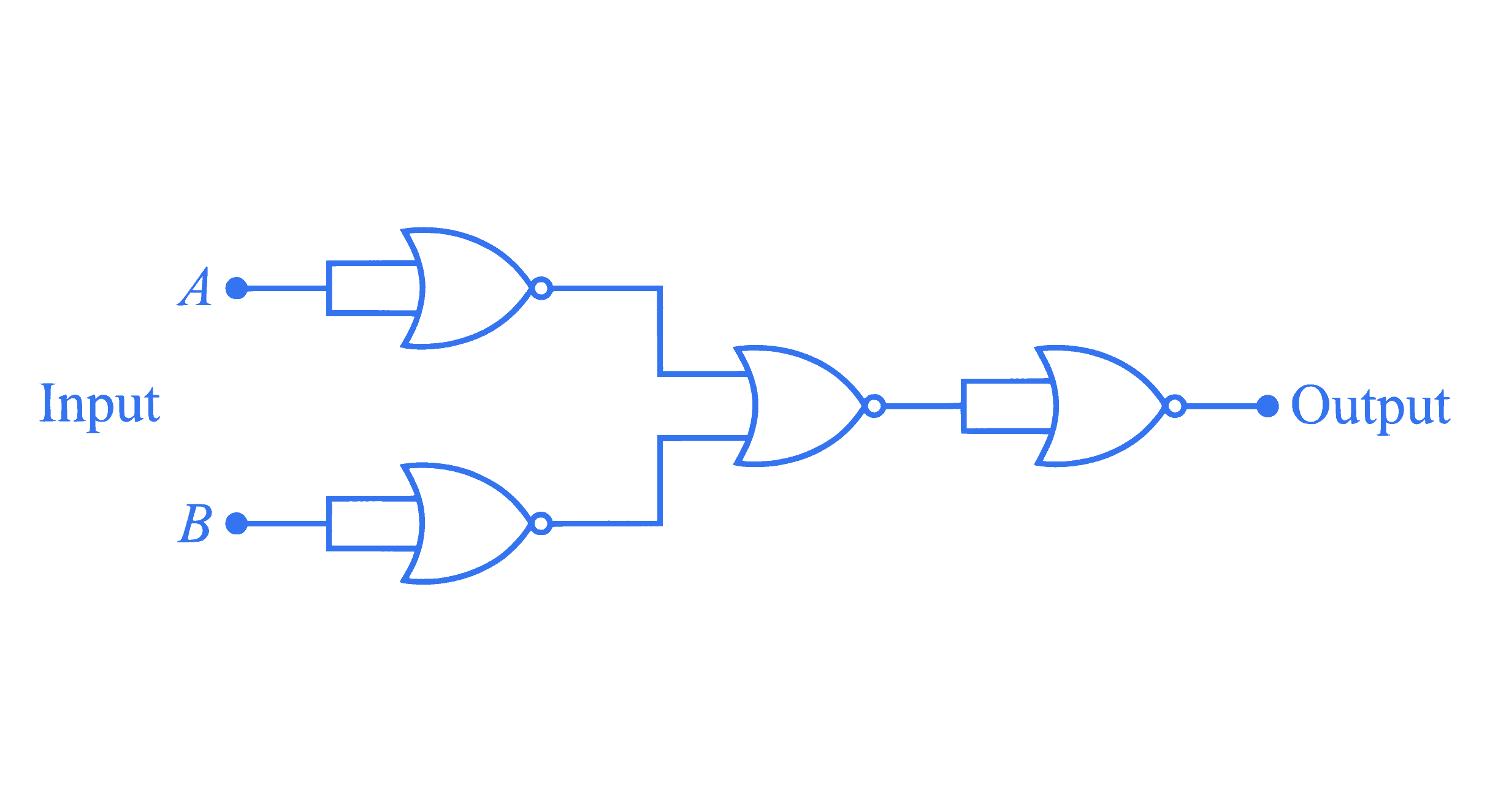

Q3.
mcq single
+4 / 1
In a double slit experiment the distance between the slits is 0.1 cm and the screen is placed at 50 cm from the slits plane. When one slit is covered with a transparent sheet having thickness $t$ and refractive index $n(=1.5)$, the central fringe shifts by 0.2 cm . The value of $t$ is
$\_\_\_\_$ cm.
Q4.
mcq single
+4 / 1
A uniform rod of mass $m$ and length $l$ suspended by means of two identical inextensible light strings as shown in figure. Tension in one string immediately after the other string is cut, is $\_\_\_\_$ . $(g$ acceleration due to gravity)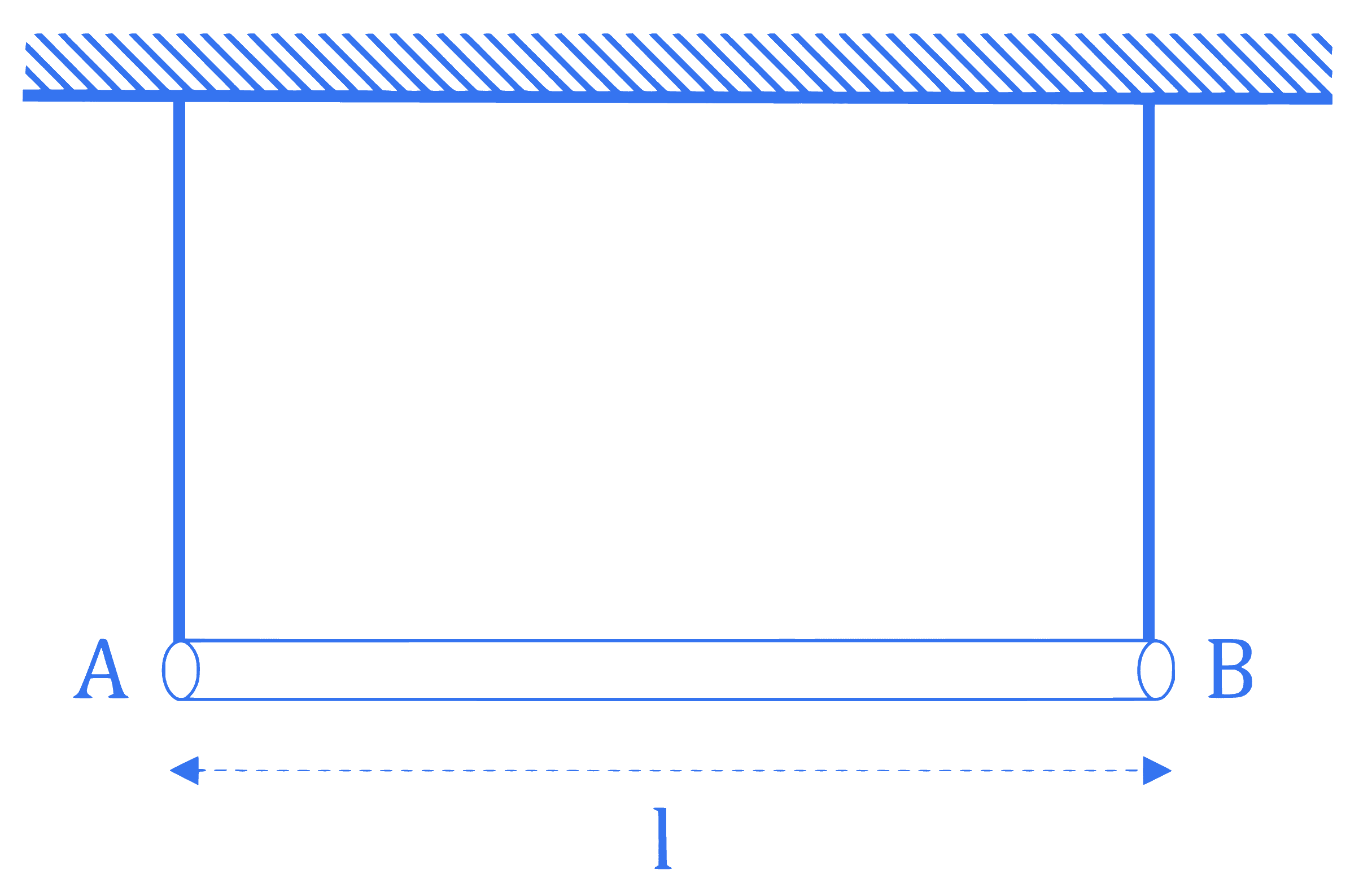

Q5.
numerical
+4 / 1
Two identical thin rods of mass $M \mathrm{~kg}$ and length $L \mathrm{~m}$ are connected as shown in figure. Moment of inertia of the combined rod system about an axis passing through point $P$ and perpendicular to the plane of the rods is $\frac{x}{12} \mathrm{ML}^2 \mathrm{~kg} \mathrm{~m}^2$. The value of $x$ is $\_\_\_\_$ .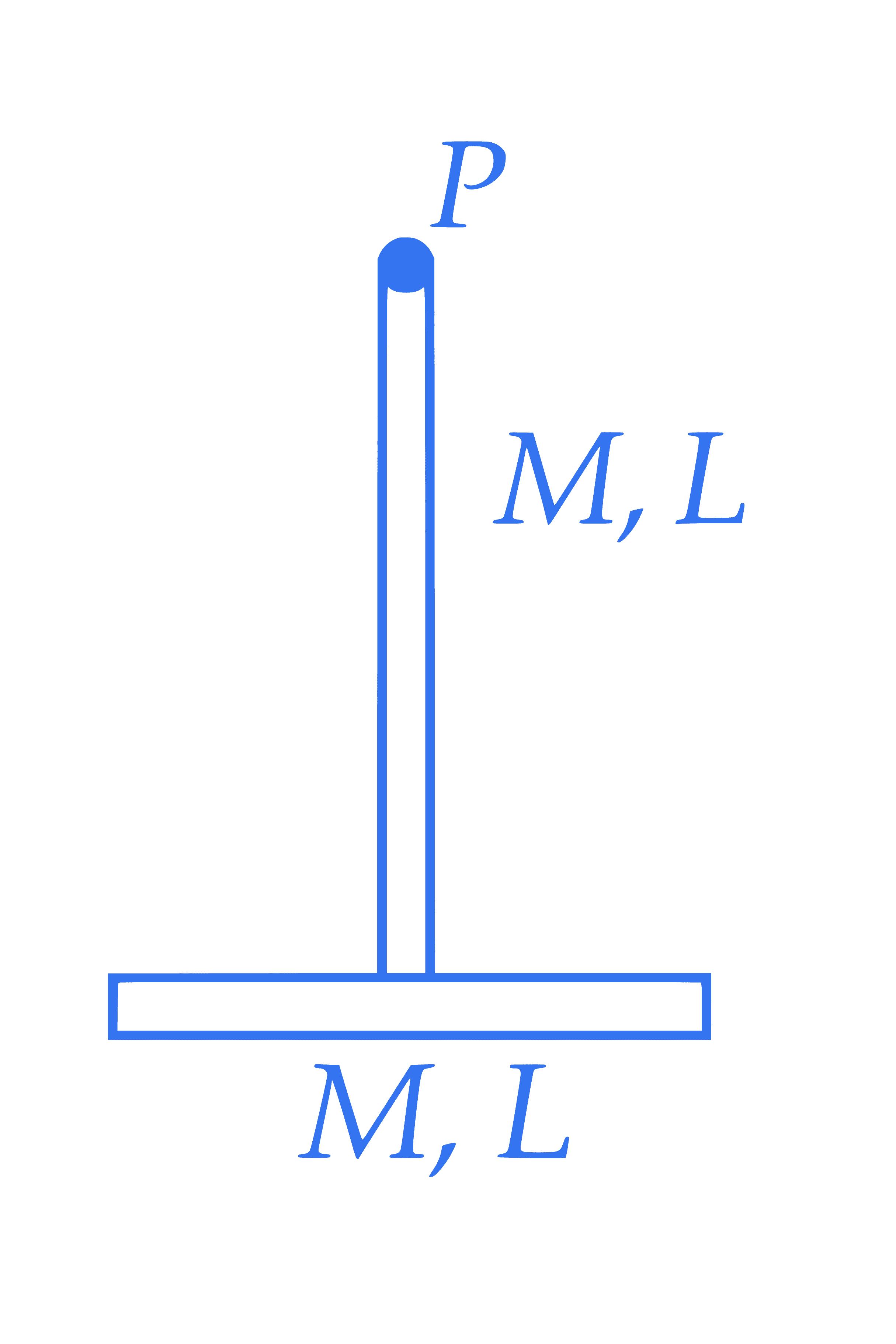

Q6.
mcq single
+4 / 1
A 1 m long metal rod AB completes the circuit as shown in figure. The area of circuit is perpendicular to the magnetic field of 0.10 T . If the resistance of the total circuit is $2 \Omega$ then the force needed to move the rod towards right with constant speed $(v)$ of $1.5 \mathrm{~m} / \mathrm{s}$ is $\_\_\_\_$ N.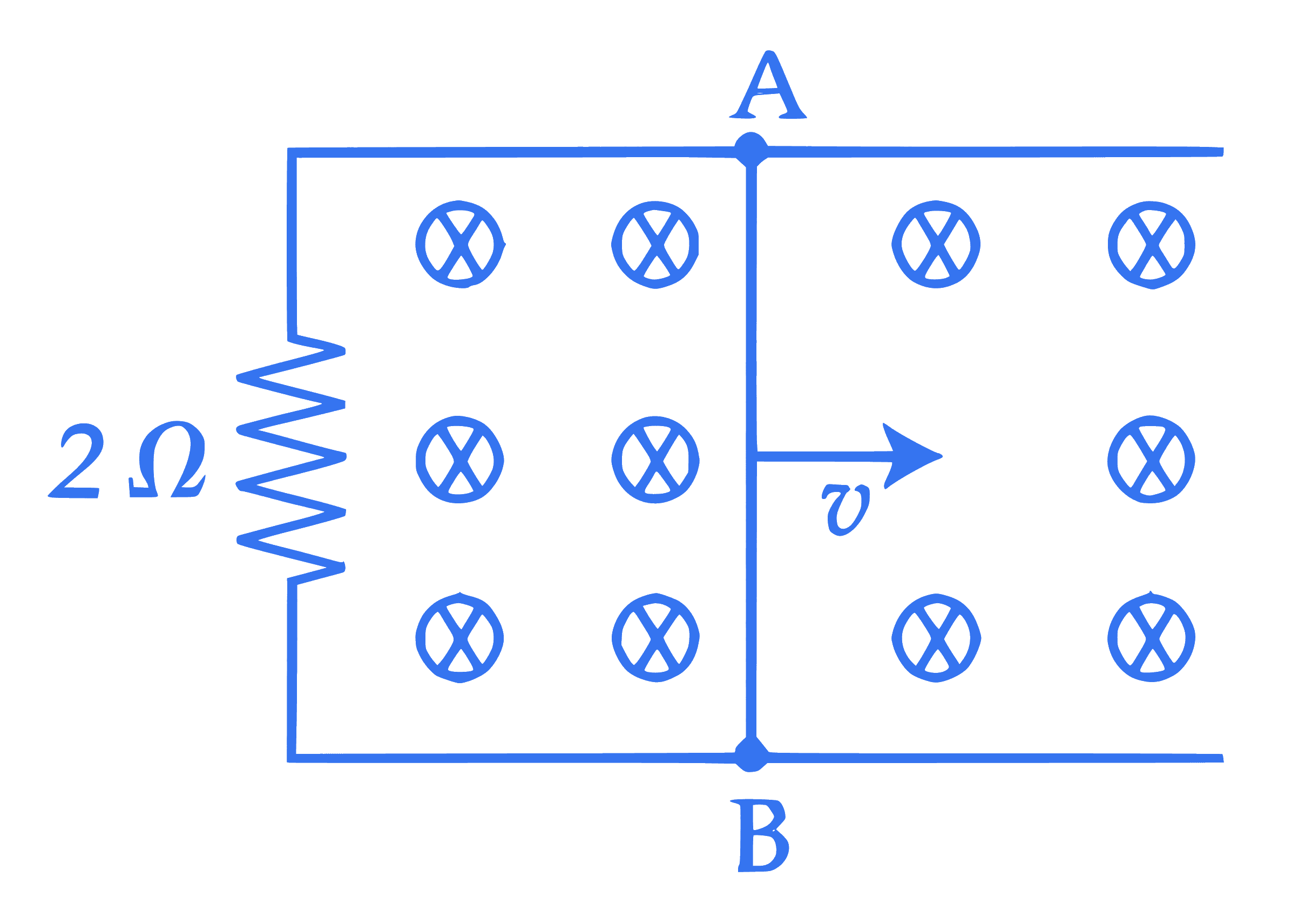

Q7.
mcq single
+4 / 1
A conducting circular loop of area $1.0 \mathrm{~m}^2$ is placed perpendicular to a magnetic field which varies as $B=\sin (100 t)$ Tesla. If the resistance of the loop is $100 \Omega$, then the average thermal energy dissipated in the loop in one period is $\_\_\_\_$ J.
Q8.
numerical
+4 / 1
10 mole of oxygen is heated at constant volume from $30^{\circ} \mathrm{C}$ to $40^{\circ} \mathrm{C}$. The change in the internal energy of the gas is $\_\_\_\_$ cal. (The molecular specific heat of oxygen at constant pressure, $C_P=7 \mathrm{cal} / \mathrm{mol} .{ }^{\circ} \mathrm{C}$ and $\left.\mathrm{R}=2 \mathrm{cal} . / \mathrm{mol} .{ }^{\circ} \mathrm{C}.\right)$
Q9.
mcq single
+4 / 1
A gas based geyser heats water flowing at the rate of 5.0 litres per minute from $27^{\circ} \mathrm{C}$ to $87^{\circ} \mathrm{C}$. The rate of consumption of the gas is $\_\_\_\_$ $\mathrm{g} / \mathrm{s}$.
(Take heat of combustion of gas $=5.0 \times 10^4 \mathrm{~J} / \mathrm{g}$ ) specific heat capacity of water $=4200 \mathrm{~J} / \mathrm{kg} \cdot{ }^{\circ} \mathrm{C}$
Q10.
mcq single
+4 / 1
Water flows through a horizontal tube as shown in the figure. The difference in height between the water columns in vertical tubes is 5 cm and the area of cross-sections at $A$ and $B$ are $6 \mathrm{~cm}^2$ and $3 \mathrm{~cm}^2$ respectively. The rate of flow will be $\_\_\_\_$ $\mathrm{cm}^3 / \mathrm{s}$. (take $g=10 \mathrm{~m} / \mathrm{s}^2$ )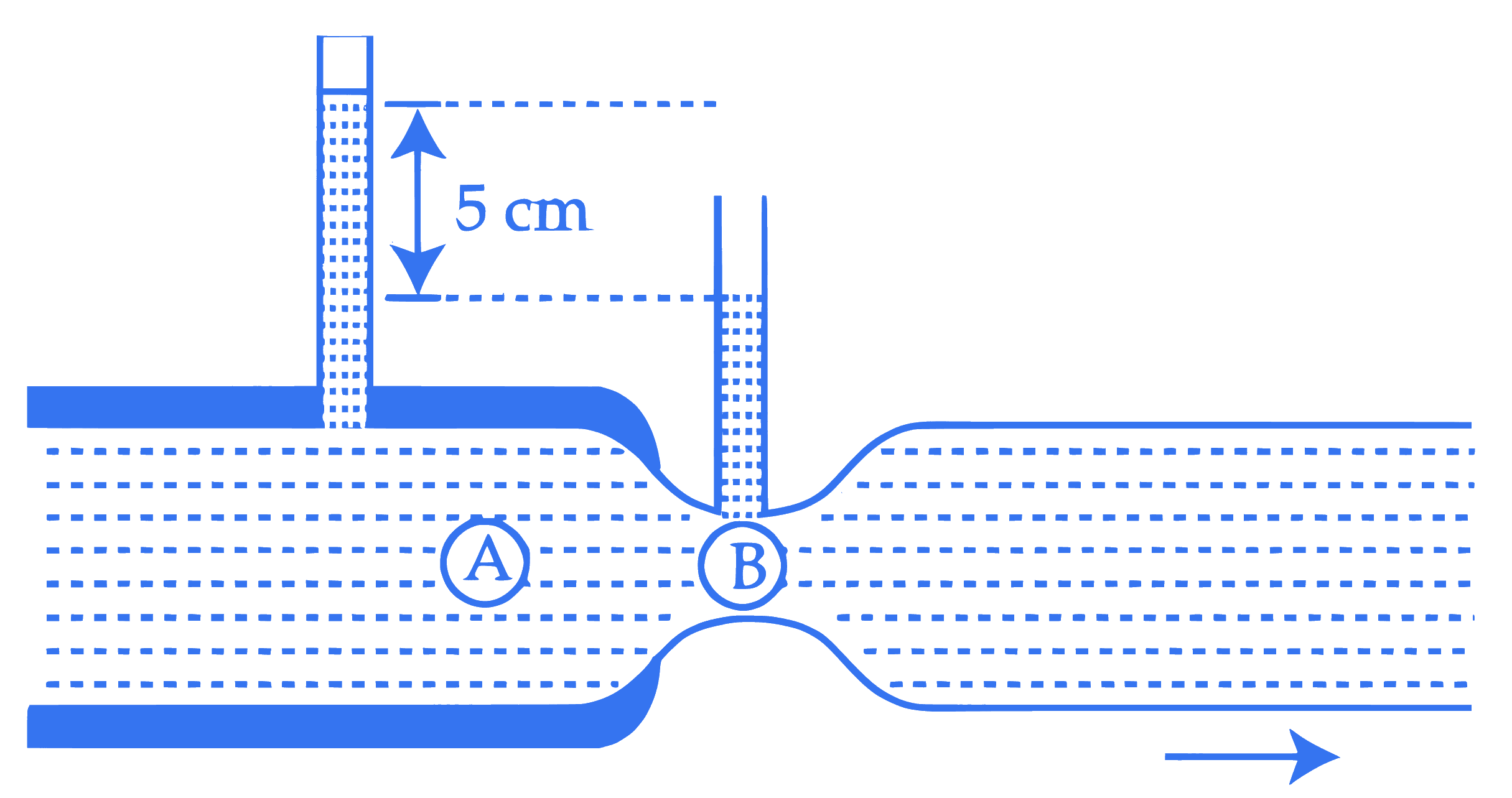

Q11.
mcq single
+4 / 1
An aluminium and steel rods having same lengths and cross-sections are joined to make total length of 120 cm at $30^{\circ} \mathrm{C}$. The coefficient of linear expansion of aluminium and steel are $24 \times 10^{-6} /{ }^{\circ} \mathrm{C}$ and $1.2 \times 10^{-5} /{ }^{\circ} \mathrm{C}$, respectively. The length of this composite rod when its temperature is raised to $100^{\circ} \mathrm{C}$, is $\_\_\_\_$ cm.
Q12.
mcq single
+4 / 1
A light wave described by $E=60\left[\sin \left(3 \times 10^{15}\right) t+\sin \left(12 \times 10^{15}\right) t\right]$ (in SI units) falls on a metal surface of work function 2.8 eV . The maximum kinetic energy of ejected photoelectron is (approximately)
$\_\_\_\_$ eV. $\left(h=6.6 \times 10^{-34}\right.$ J.s. and $\left.e=1.6 \times 10^{-19} \mathrm{C}\right)$
Q13.
mcq single
+4 / 1
A point charge of $10^{-8} \mathrm{C}$ is placed at origin. The work done in moving a point charge $2 \mu \mathrm{C}$ from point $A(4,4,2) \mathrm{m}$ to $B(2,2,1) \mathrm{m}$ is $\_\_\_\_$ J. $\left(\frac{1}{4 \pi \epsilon_{\mathrm{o}}}=9 \times 10^9\right.$ in SI units)
Q14.
mcq single
+4 / 1
Initially a satellite of 100 kg is in a circular orbit of radius $1.5 \mathrm{R}_{\mathrm{E}}$. This satellite can be moved to a circular orbit of radius $3 R_E$ by supplying $\alpha \times 10^6 \mathrm{~J}$ of energy The value of $\alpha$ is $\_\_\_\_$ .
(Take Radius of Earth $R_E=6 \times 10^6 \mathrm{~m}$ and $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q15.
mcq single
+4 / 1
A parallel plate capacitor has capacitance $C$, when there is vacuum within the parallel plates. A sheet having thickness $\left(\frac{1}{3}\right)^{\mathrm{rd}}$ of the separation between the plates and relative permittivity $K$ is introduced between the plates. The new capacitance of the system is :
Q16.
numerical
+4 / 1
In a microscope the objective is having focal length $f_o=2 \mathrm{~cm}$ and eye-piece is having focal length $f_e=4 \mathrm{~cm}$. The tube length is 32 cm . The magnification produced by this microscope for normal adjustment is $\_\_\_\_$ .
Q17.
numerical
+4 / 1
A collimated beam of light of diameter 2 mm is propagating along $x$-axis. The beam is required to be expanded in a collimated beam of diameter 14 mm using a system of two convex lenses. If first lens has focal length 40 mm , then the focal length of second lens is $\_\_\_\_$ mm.
Q18.
mcq single
+4 / 1
A current carrying solenoid is placed vertically and a particle of mass $m$ with charge $Q$ is released from rest. The particle moves along the axis of solenoid. If $g$ is acceleration due to gravity then the acceleration (a) of the charged particle will satisfy :
Q19.
mcq single
+4 / 1
Consider a modified Bernoulli equation.
$$ \left(\mathrm{P}+\frac{A}{B t^2}\right)+\rho g(h+B t)+\frac{1}{2} \rho V^2=\text { constant } $$
If $t$ has the dimension of time then the dimensions of $A$ and $B$ are $\_\_\_\_$ , $\_\_\_\_$ respectively.
Q20.
mcq single
+4 / 1
In an experiment the values of two spring constants were measured as $k_1=(10 \pm 0.2) \mathrm{N} / \mathrm{m}$ and $k_2=(20 \pm 0.3) \mathrm{N} / \mathrm{m}$. If these springs are connected in parallel, then the percentage error in equivalent spring constant is :
Q21.
mcq single
+4 / 1
If an alpha particle with energy 7.7 MeV is bombarded on a thin gold foil, the closest distance from nucleus it can reach is $\_\_\_\_$ m. (Atomic number of gold $=79$ and $\frac{1}{4 \pi \epsilon_{\mathrm{o}}}=9 \times 10^9$ in SI units)
Q22.
mcq single
+4 / 1
Two strings $(A, B)$ having linear densities $\mu_A=2 \times 10^{-4} \mathrm{~kg} / \mathrm{m}$ and, $\mu_B=4 \times 10^{-4} \mathrm{~kg} / \mathrm{m}$ and lengths $L_A=2.5 \mathrm{~m}$ and $L_B=1.5 \mathrm{~m}$ respectively are joined. Free ends of $A$ and $B$ are tied to two rigid supports $C$ and $D$, respectively creating a tension of 500 N in the wire. Two identical pulses, sent from $C$ and $D$ ends, take time $t_1$ and $t_2$, respectively, to reach the joint. The ratio $t_1 / t_2$ is:
Q23.
mcq single
+4 / 1
A 4 kg mass moves under the influence of a force $\vec{F}=\left(4 t^3 \hat{i}-3 t \hat{j}\right) \mathrm{N}$ where $t$ is the time in second. If mass starts from origin at $t=0$, the velocity and position after $t=2 \mathrm{~s}$ will be:
Q24.
numerical
+4 / 1
The heat generated in 1 minute between points $A$ and $B$ in the given circuit, when a battery of 9 V with internal resistance of $1 \Omega$ is connected across these points is $\_\_\_\_$ J.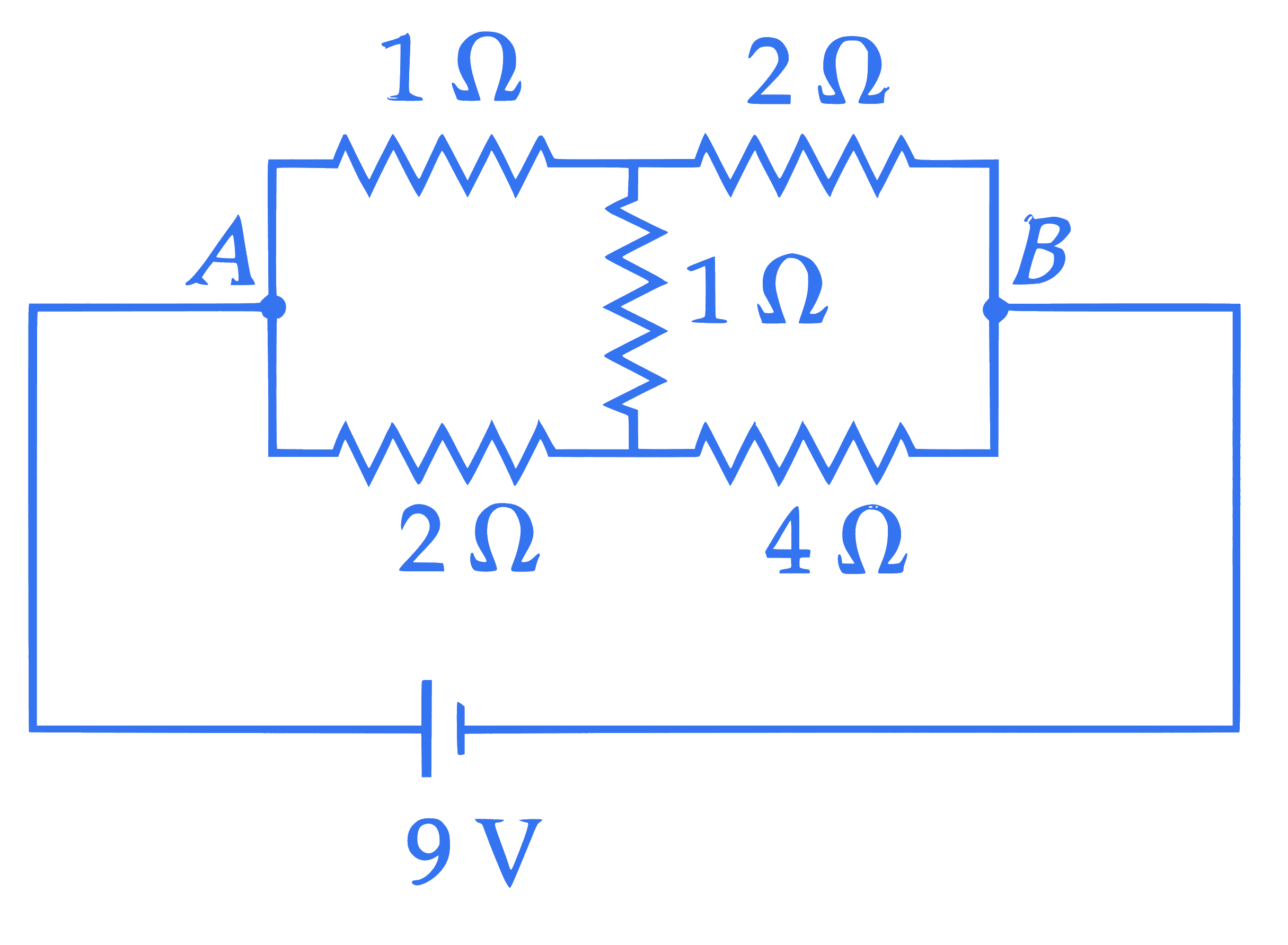

Q25.
mcq single
+4 / 1
The electric field in a plane electromagnetic wave is given by :
$$ E_y=69 \sin \left[0.6 \times 10^3 x-1.8 \times 10^{11} t\right] \mathrm{V} / \mathrm{m} . $$
The expression for magnetic field associated with this electromagnetic wave is $\_\_\_\_$ T.
Q26.
mcq single
+4 / 1
Potential energy ( $V$ ) versus distance ( $x$ ) is given by the graph. Rank various regions as per the magnitudes of the force ( $F$ ) acting on a particle from high to low.

Q27.
mcq single
+4 / 1
$$ \text { The given circuit works as : } $$

Q28.
mcq single
+4 / 1
In a double slit experiment the distance between the slits is 0.1 cm and the screen is placed at 50 cm from the slits plane. When one slit is covered with a transparent sheet having thickness $t$ and refractive index $n(=1.5)$, the central fringe shifts by 0.2 cm . The value of $t$ is
$\_\_\_\_$ cm.
Q29.
mcq single
+4 / 1
A uniform rod of mass $m$ and length $l$ suspended by means of two identical inextensible light strings as shown in figure. Tension in one string immediately after the other string is cut, is $\_\_\_\_$ . $(g$ acceleration due to gravity)

Q30.
numerical
+4 / 1
Two identical thin rods of mass $M \mathrm{~kg}$ and length $L \mathrm{~m}$ are connected as shown in figure. Moment of inertia of the combined rod system about an axis passing through point $P$ and perpendicular to the plane of the rods is $\frac{x}{12} \mathrm{ML}^2 \mathrm{~kg} \mathrm{~m}^2$. The value of $x$ is $\_\_\_\_$ .

Q31.
mcq single
+4 / 1
A 1 m long metal rod AB completes the circuit as shown in figure. The area of circuit is perpendicular to the magnetic field of 0.10 T . If the resistance of the total circuit is $2 \Omega$ then the force needed to move the rod towards right with constant speed $(v)$ of $1.5 \mathrm{~m} / \mathrm{s}$ is $\_\_\_\_$ N.

Q32.
mcq single
+4 / 1
A conducting circular loop of area $1.0 \mathrm{~m}^2$ is placed perpendicular to a magnetic field which varies as $B=\sin (100 t)$ Tesla. If the resistance of the loop is $100 \Omega$, then the average thermal energy dissipated in the loop in one period is $\_\_\_\_$ J.
Q33.
numerical
+4 / 1
10 mole of oxygen is heated at constant volume from $30^{\circ} \mathrm{C}$ to $40^{\circ} \mathrm{C}$. The change in the internal energy of the gas is $\_\_\_\_$ cal. (The molecular specific heat of oxygen at constant pressure, $C_P=7 \mathrm{cal} / \mathrm{mol} .{ }^{\circ} \mathrm{C}$ and $\left.\mathrm{R}=2 \mathrm{cal} . / \mathrm{mol} .{ }^{\circ} \mathrm{C}.\right)$
Q34.
mcq single
+4 / 1
A gas based geyser heats water flowing at the rate of 5.0 litres per minute from $27^{\circ} \mathrm{C}$ to $87^{\circ} \mathrm{C}$. The rate of consumption of the gas is $\_\_\_\_$ $\mathrm{g} / \mathrm{s}$.
(Take heat of combustion of gas $=5.0 \times 10^4 \mathrm{~J} / \mathrm{g}$ ) specific heat capacity of water $=4200 \mathrm{~J} / \mathrm{kg} \cdot{ }^{\circ} \mathrm{C}$
Q35.
mcq single
+4 / 1
Water flows through a horizontal tube as shown in the figure. The difference in height between the water columns in vertical tubes is 5 cm and the area of cross-sections at $A$ and $B$ are $6 \mathrm{~cm}^2$ and $3 \mathrm{~cm}^2$ respectively. The rate of flow will be $\_\_\_\_$ $\mathrm{cm}^3 / \mathrm{s}$. (take $g=10 \mathrm{~m} / \mathrm{s}^2$ )

Q36.
mcq single
+4 / 1
An aluminium and steel rods having same lengths and cross-sections are joined to make total length of 120 cm at $30^{\circ} \mathrm{C}$. The coefficient of linear expansion of aluminium and steel are $24 \times 10^{-6} /{ }^{\circ} \mathrm{C}$ and $1.2 \times 10^{-5} /{ }^{\circ} \mathrm{C}$, respectively. The length of this composite rod when its temperature is raised to $100^{\circ} \mathrm{C}$, is $\_\_\_\_$ cm.
Q37.
mcq single
+4 / 1
A light wave described by $E=60\left[\sin \left(3 \times 10^{15}\right) t+\sin \left(12 \times 10^{15}\right) t\right]$ (in SI units) falls on a metal surface of work function 2.8 eV . The maximum kinetic energy of ejected photoelectron is (approximately)
$\_\_\_\_$ eV. $\left(h=6.6 \times 10^{-34}\right.$ J.s. and $\left.e=1.6 \times 10^{-19} \mathrm{C}\right)$
Q38.
mcq single
+4 / 1
A point charge of $10^{-8} \mathrm{C}$ is placed at origin. The work done in moving a point charge $2 \mu \mathrm{C}$ from point $A(4,4,2) \mathrm{m}$ to $B(2,2,1) \mathrm{m}$ is $\_\_\_\_$ J. $\left(\frac{1}{4 \pi \epsilon_{\mathrm{o}}}=9 \times 10^9\right.$ in SI units)
Q39.
mcq single
+4 / 1
Initially a satellite of 100 kg is in a circular orbit of radius $1.5 \mathrm{R}_{\mathrm{E}}$. This satellite can be moved to a circular orbit of radius $3 R_E$ by supplying $\alpha \times 10^6 \mathrm{~J}$ of energy The value of $\alpha$ is $\_\_\_\_$ .
(Take Radius of Earth $R_E=6 \times 10^6 \mathrm{~m}$ and $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q40.
mcq single
+4 / 1
A parallel plate capacitor has capacitance $C$, when there is vacuum within the parallel plates. A sheet having thickness $\left(\frac{1}{3}\right)^{\mathrm{rd}}$ of the separation between the plates and relative permittivity $K$ is introduced between the plates. The new capacitance of the system is :
Q41.
numerical
+4 / 1
In a microscope the objective is having focal length $f_o=2 \mathrm{~cm}$ and eye-piece is having focal length $f_e=4 \mathrm{~cm}$. The tube length is 32 cm . The magnification produced by this microscope for normal adjustment is $\_\_\_\_$ .
Q42.
numerical
+4 / 1
A collimated beam of light of diameter 2 mm is propagating along $x$-axis. The beam is required to be expanded in a collimated beam of diameter 14 mm using a system of two convex lenses. If first lens has focal length 40 mm , then the focal length of second lens is $\_\_\_\_$ mm.
Q43.
mcq single
+4 / 1
A current carrying solenoid is placed vertically and a particle of mass $m$ with charge $Q$ is released from rest. The particle moves along the axis of solenoid. If $g$ is acceleration due to gravity then the acceleration (a) of the charged particle will satisfy :
Q44.
mcq single
+4 / 1
Consider a modified Bernoulli equation.
$$ \left(\mathrm{P}+\frac{A}{B t^2}\right)+\rho g(h+B t)+\frac{1}{2} \rho V^2=\text { constant } $$
If $t$ has the dimension of time then the dimensions of $A$ and $B$ are $\_\_\_\_$ , $\_\_\_\_$ respectively.
Q45.
mcq single
+4 / 1
In an experiment the values of two spring constants were measured as $k_1=(10 \pm 0.2) \mathrm{N} / \mathrm{m}$ and $k_2=(20 \pm 0.3) \mathrm{N} / \mathrm{m}$. If these springs are connected in parallel, then the percentage error in equivalent spring constant is :
Q46.
mcq single
+4 / 1
If an alpha particle with energy 7.7 MeV is bombarded on a thin gold foil, the closest distance from nucleus it can reach is $\_\_\_\_$ m. (Atomic number of gold $=79$ and $\frac{1}{4 \pi \epsilon_{\mathrm{o}}}=9 \times 10^9$ in SI units)
Q47.
mcq single
+4 / 1
Two strings $(A, B)$ having linear densities $\mu_A=2 \times 10^{-4} \mathrm{~kg} / \mathrm{m}$ and, $\mu_B=4 \times 10^{-4} \mathrm{~kg} / \mathrm{m}$ and lengths $L_A=2.5 \mathrm{~m}$ and $L_B=1.5 \mathrm{~m}$ respectively are joined. Free ends of $A$ and $B$ are tied to two rigid supports $C$ and $D$, respectively creating a tension of 500 N in the wire. Two identical pulses, sent from $C$ and $D$ ends, take time $t_1$ and $t_2$, respectively, to reach the joint. The ratio $t_1 / t_2$ is:
Q48.
mcq single
+4 / 1
A 4 kg mass moves under the influence of a force $\vec{F}=\left(4 t^3 \hat{i}-3 t \hat{j}\right) \mathrm{N}$ where $t$ is the time in second. If mass starts from origin at $t=0$, the velocity and position after $t=2 \mathrm{~s}$ will be:
Q49.
numerical
+4 / 1
The heat generated in 1 minute between points $A$ and $B$ in the given circuit, when a battery of 9 V with internal resistance of $1 \Omega$ is connected across these points is $\_\_\_\_$ J.

Q50.
mcq single
+4 / 1
The electric field in a plane electromagnetic wave is given by :
$$ E_y=69 \sin \left[0.6 \times 10^3 x-1.8 \times 10^{11} t\right] \mathrm{V} / \mathrm{m} . $$
The expression for magnetic field associated with this electromagnetic wave is $\_\_\_\_$ T.