JEE Main 2026 (Online) 22nd January Evening Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
numerical
+4 / 1
The mass of benzanilide obtained from the benzoylation reaction of 5.8 g of aniline, if yield of product is $82 \%$, is $\_\_\_\_$ g (nearest integer).
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
Q2.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The first ionization enthalpy of Cr is lower than that of Mn .
**Statement II :** The second and third ionization enthalpies of Cr are higher than those of Mn .
In the light of the above statements, choose the correct answer from the options given below :
Q3.
numerical
+4 / 1
Among the following oxides of $3 d$ elements, the number of mixed oxides are $\_\_\_\_$ .
$$ \mathrm{Ti}_2 \mathrm{O}_3, \mathrm{~V}_2 \mathrm{O}_4, \mathrm{Cr}_2 \mathrm{O}_3, \mathrm{Mn}_3 \mathrm{O}_4, \mathrm{Fe}_3 \mathrm{O}_4, \mathrm{Fe}_2 \mathrm{O}_3, \mathrm{Co}_3 \mathrm{O}_4 $$
Q4.
numerical
+4 / 1
Consider $\mathrm{A} \xrightarrow{\mathrm{k}_1} \mathrm{~B}$ and $\mathrm{C} \xrightarrow{\mathrm{k}_2} \mathrm{D}$ are two reactions. If the rate constant $\left(\mathrm{k}_1\right)$ of the $\mathrm{A} \longrightarrow \mathrm{B}$ reaction can be expressed by the following equation $\log _{10} \mathrm{k}=14.34-\frac{1.5 \times 10^4}{\mathrm{~T} / \mathrm{K}}$ and activation energy of $C \longrightarrow D$ reaction $\left(E a_2\right)$ is $\frac{1}{5}$ th of the $A \longrightarrow B$ reaction $\left(E a_1\right)$, then the value of $\left(E a_2\right)$ is
$\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (Nearest Integer)
Q5.
mcq single
+4 / 1
Correct statements regarding Arrhenius equation among the following are :
A. Factor $e^{-\mathrm{Ea} / \mathrm{RT}}$ corresponds to fraction of molecules having kinetic energy less than Ea.
B. At a given temperature, lower the Ea, faster is the reaction.
C. Increase in temperature by about $10^{\circ} \mathrm{C}$ doubles the rate of reaction.
D. Plot of $\log \mathrm{k}$ vs $\frac{1}{\mathrm{~T}}$ gives a straight line with slope $=-\frac{\mathrm{Ea}}{\mathrm{R}}$.
Choose the correct answer from the options given below :
Q6.
mcq single
+4 / 1
Among $\mathrm{H}_2 \mathrm{~S}, \mathrm{H}_2 \mathrm{O}, \mathrm{NF}_3, \mathrm{NH}_3$ and $\mathrm{CHCl}_3$, identify the molecule $(\mathrm{X})$ with lowest dipole moment value. The number of lone pairs of electrons present on the central atom of the molecule $(X)$ is :
Q7.
mcq single
+4 / 1
At $\mathrm{T}(\mathrm{K}), 100 \mathrm{~g}$ of $98 \% \mathrm{H}_2 \mathrm{SO}_4(\mathrm{w} / \mathrm{w})$ aqueous solution is mixed with 100 g of $49 \% \mathrm{H}_2 \mathrm{SO}_4(\mathrm{w} / \mathrm{w})$ aqueous solution. What is the mole fraction of $\mathrm{H}_2 \mathrm{SO}_4$ in the resultant solution?
(Given : Atomic mass $\mathrm{H}=1 \mathrm{u} ; \mathrm{S}=32 \mathrm{u} ; \mathrm{O}=16 \mathrm{u}$ ).
(Assume that temperature after mixing remains constant)
Q8.
mcq single
+4 / 1
Identify the correct statements :
A. Hydrated salts can be used as primary standard.
B. Primary standard should not undergo any reaction with air.
C. Reactions of primary standard with another substance should be instantaneous and stoichiometric.
D. Primary standard should not be soluble in water.
E. Primary standard should have low relative molar mass.
Choose the correct answer from the options given below :
Q9.
mcq single
+4 / 1
$\mathrm{A}+2 \mathrm{~B} \longrightarrow \mathrm{AB}_2$
36.0 g of 'A' (Molar mass : $60 \mathrm{~g} \mathrm{~mol}^{-1}$ ) and 56.0 g of ' B ' (Molar mass : $80 \mathrm{~g} \mathrm{~mol}^{-1}$ ) are allowed to react. Which of the following statements are correct ?
A. 'A' is the limiting reagent.
B. $77.0 \mathrm{~g}$ of $\mathrm{AB}_2$ is formed.
C. Molar mass of $\mathrm{AB}_2$ is $140 \mathrm{~g} \mathrm{~mol}^{-1}$.
D. $15.0 \mathrm{~g}$ of A is left unreacted after the completion of reaction.
Choose the correct answer from the options given below :
Q10.
mcq single
+4 / 1
Which of the following mixture gives a buffer solution with $\mathrm{pH}=9.25$ ?
Given : $\mathrm{pK}_{\mathrm{b}}\left(\mathrm{NH}_4 \mathrm{OH}\right)=4.75$
Q11.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\mathrm{C}\mathrm{Se}>\mathrm{Te}>\mathrm{Po}>\mathrm{O}$ is the correct order in terms of the magnitude of electron gain enthalpy values.
In the light of the above statements, choose the correct answer from the options given below :
Q12.
numerical
+4 / 1
If the enthalpy of sublimation of Li is $155 \mathrm{~kJ} \mathrm{~mol}^{-1}$, enthalpy of dissociation of $\mathrm{F}_2$ is $150 \mathrm{~kJ} \mathrm{~mol}^{-1}$, ionization enthalpy of Li is $520 \mathrm{~kJ} \mathrm{~mol}^{-1}$, electron gain enthalpy of F is $-313 \mathrm{~kJ} \mathrm{~mol}^{-1}$, standard enthalpy of formation of LiF is $-594 \mathrm{~kJ} \mathrm{~mol}^{-1}$. The magnitude of lattice enthalpy of LiF is $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (Nearest Integer)
Q13.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Elements ' $X$ ' and ' $Y$ ' are the most and least electronegative elements, respectively among $\mathrm{N}, \mathrm{As}, \mathrm{Sb}$ and P . The nature of the oxides $\mathrm{X}_2 \mathrm{O}_3$ and $\mathrm{Y}_2 \mathrm{O}_3$ is acidic and amphoteric, respectively.
**Statement II :** $\mathrm{BCl}_3$ is covalent in nature and gets hydrolysed in water. It produces $\left[\mathrm{B}(\mathrm{OH})_4\right]^{-}$ and $\left[\mathrm{B}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+}$ in aqueous medium.
In the light of the above statements, choose the correct answer from the options given below :
Q14.
mcq single
+4 / 1
$\left[\mathrm{Ni}\left(\mathrm{PPh}_3\right)_2 \mathrm{Cl}_2\right]$ is a paramagnetic complex. Identify the INCORRECT statements about this complex.
A. The complex exhibits geometrical isomerism.
B. The complex is white in colour.
C. The calculated spin-only magnetic moment of the complex is 2.84 BM .
D. The calculated CFSE (Crystal Field Stabilization Energy) of Ni in this complex is $-0.8 \Delta_{\mathrm{o}}$.
E. The geometrical arrangement of ligands in this complex is similar to that in $\mathrm{Ni}(\mathrm{CO})_4$.
Choose the correct answer from the options given below :
Q15.
mcq single
+4 / 1
The compound $\mathrm{A}, \mathrm{C}_8 \mathrm{H}_8 \mathrm{O}_2$ reacts with acetophenone to form a single product via cross-Aldol condensation. The compound A on reaction with conc. NaOH forms a substituted benzyl alcohol as one of the two products. The compound A is :
Q16.
mcq single
+4 / 1
When 1 g of compound $(\mathrm{X})$ is subjected to Kjeldahl's method for estimation of nitrogen, 15 mL 1 M $\mathrm{H}_2 \mathrm{SO}_4$ was neutralized by ammonia evolved. The percentage of nitrogen in compound $(\mathrm{X})$ is :
Q17.
mcq single
+4 / 1
The dibromo compound $[\mathrm{P}]$ (molecular formula: $\mathrm{C}_9 \mathrm{H}_{10} \mathrm{Br}_2$ ) when heated with excess sodamide followed by treatment with dilute HCl gives [Q]. On warming [Q] with mercuric sulphate and dilute sulphuric acid yield $[R]$ which gives positive Iodoform test but negative Tollen's test. The compound $[\mathrm{P}]$ is :
Q18.
mcq single
+4 / 1
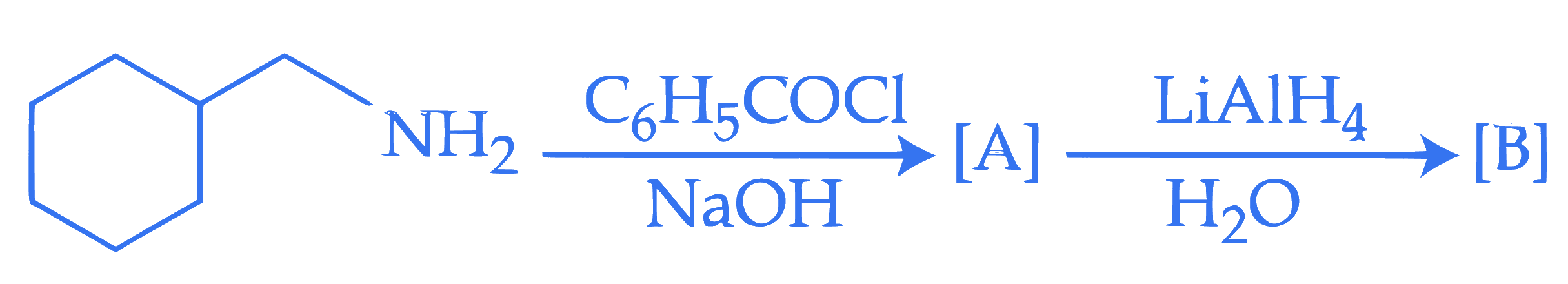 $$ \text { The final product }[\mathrm{B}] \text { is : } $$
$$ \text { The final product }[\mathrm{B}] \text { is : } $$
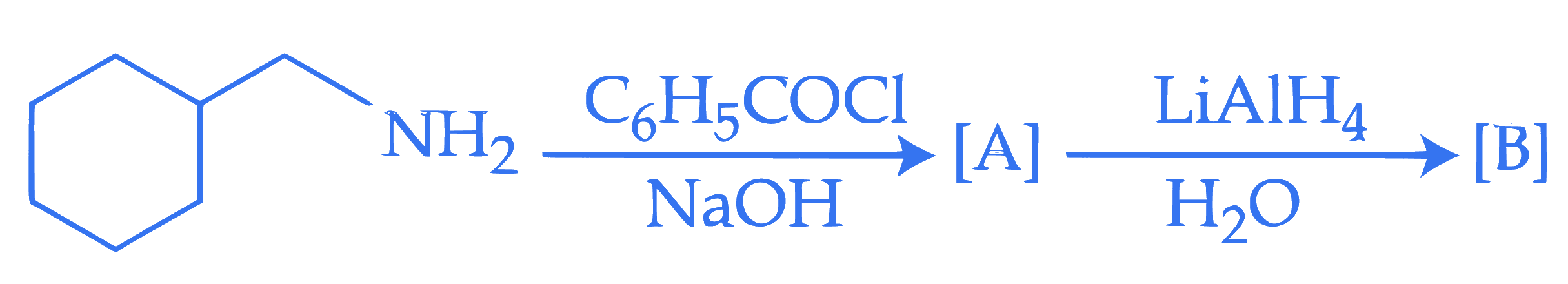 $$ \text { The final product }[\mathrm{B}] \text { is : } $$
$$ \text { The final product }[\mathrm{B}] \text { is : } $$
Q19.
mcq single
+4 / 1
Consider the following reaction :
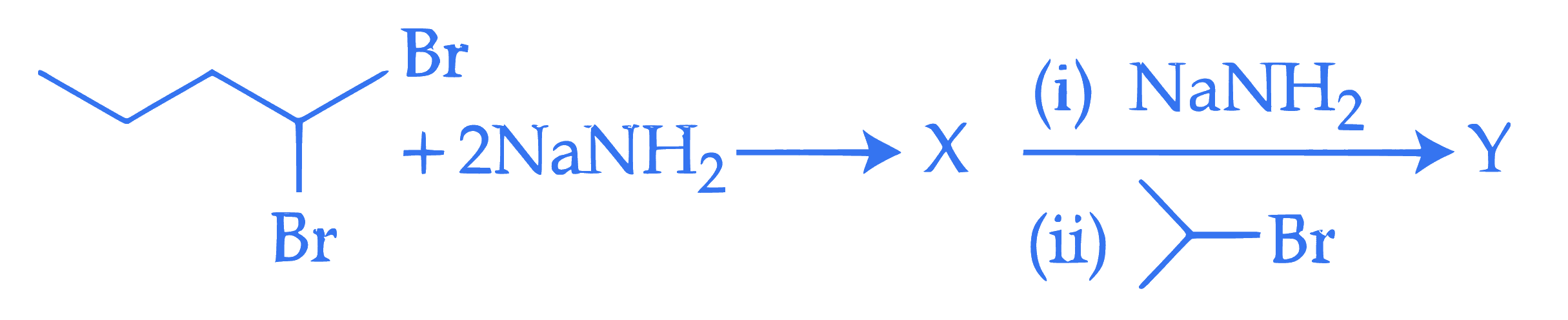 The product Y formed is :
The product Y formed is :
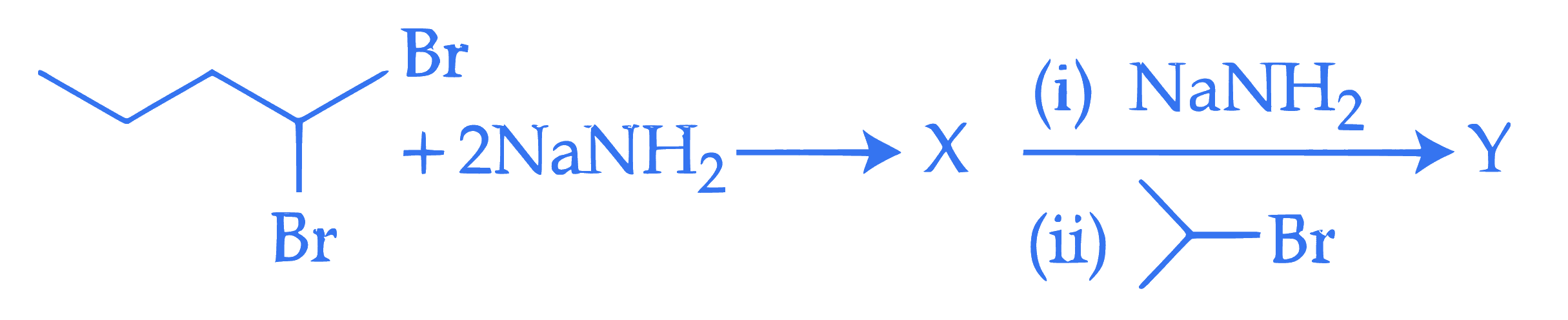 The product Y formed is :
The product Y formed is :
Q20.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Reaction of Glucose with
List - II
Product formed
A.
Hydroxylamine
I.
Gluconic acid
B.
$\mathrm{Br}_2$ water
II.
Glucose pentacetate
C.
Excess acetic anhydride
III.
Saccharic acid
D.
Concentrated $\mathrm{HNO}_3$
IV.
Glucoxime
Choose the correct answer from the options given below :
Q21.
mcq single
+4 / 1
$$ \text { The IUPAC name of the following compound is : } $$
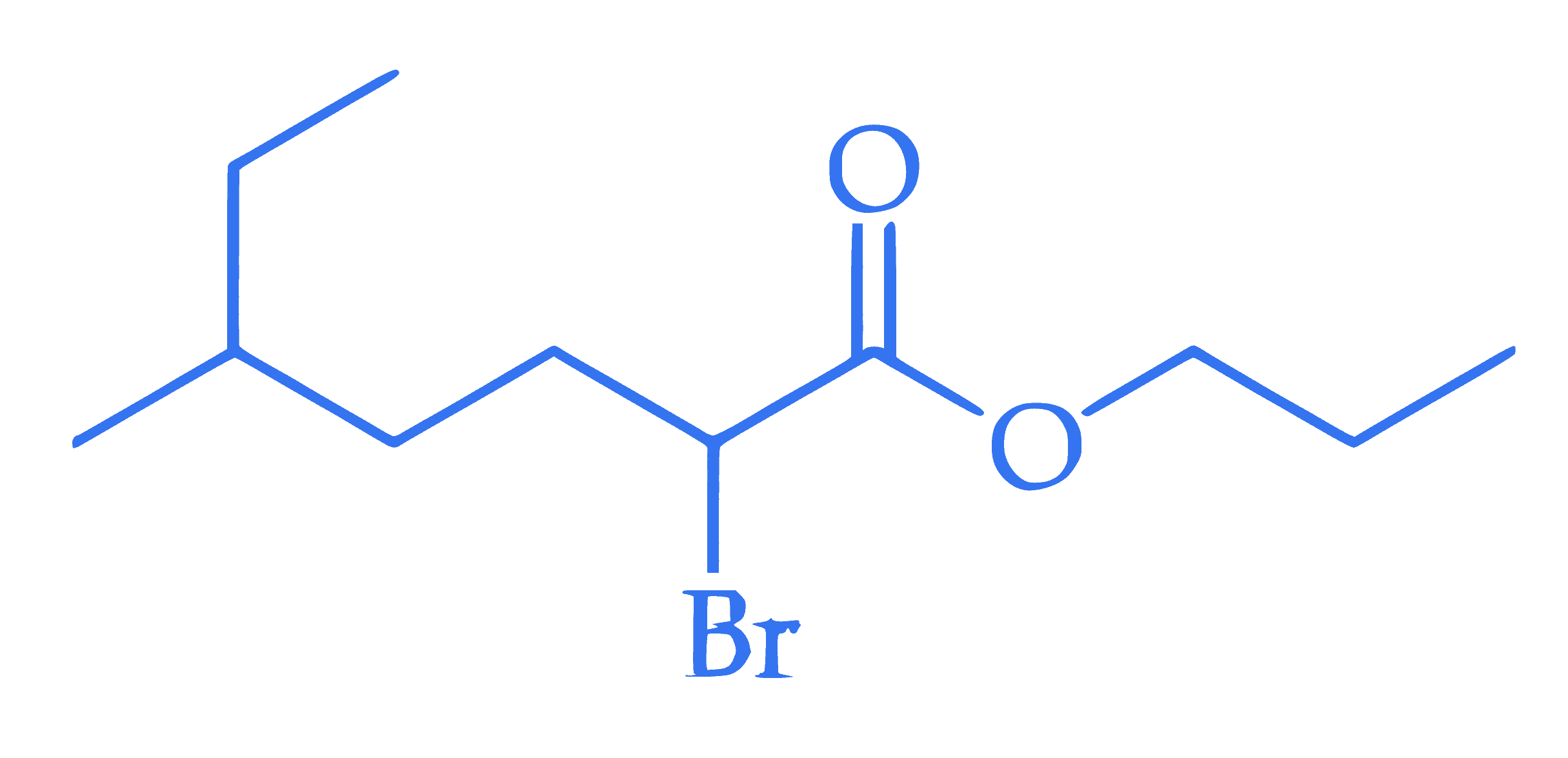

Q22.
numerical
+4 / 1
Consider the following electrochemical cell :
$$ \mathrm{Pt}\left|\mathrm{O}_2(\mathrm{~g})(1 \mathrm{bar})\right| \mathrm{HCl}(\mathrm{aq}) \| \mathrm{M}^{2+}(\mathrm{aq}, 1.0 \mathrm{M}) \mid \mathrm{M}(\mathrm{~s}) $$
The pH above which, oxygen gas would start to evolve at anode is $\_\_\_\_$ (nearest integer).
$$ \left.\left[\begin{array}{ll} \text { Given : } & \mathrm{E}_{\mathrm{M}^{2+} / \mathrm{M}}^{\mathrm{o}}=0.994 \mathrm{~V} \\ & \mathrm{E}_{\mathrm{O}_2 / \mathrm{H}_2 \mathrm{O}}^{\mathrm{o}}=1.23 \mathrm{~V} \end{array}\right\} \text { standard reduction potential }
\\ \text {and} \frac{\mathrm{RT}}{\mathrm{F}}(2.303)=0.059 \mathrm{~V} \text {at the given condition}\right] $$
Q23.
mcq single
+4 / 1
Consider the following reduction processes :
$$ \begin{aligned} & \mathrm{Al}^{3+}+3 \mathrm{e}^{-} \longrightarrow \mathrm{Al}(\mathrm{~s}), \mathrm{E}^0=-1.66 \mathrm{~V} \\ & \mathrm{Fe}^{3+}+\mathrm{e}^{-} \longrightarrow \mathrm{Fe}^{2+}, \mathrm{E}^0=+0.77 \mathrm{~V} \\ & \mathrm{Co}^{3+}+\mathrm{e}^{-} \longrightarrow \mathrm{Co}^{2+}, \mathrm{E}^0=+1.81 \mathrm{~V} \\ & \mathrm{Cr}^{3+}+3 \mathrm{e}^{-} \longrightarrow \mathrm{Cr}(\mathrm{~s}), \mathrm{E}^0=-0.74 \mathrm{~V} \end{aligned} $$
The tendency to act as reducing agent decreases in the order :
Q24.
mcq single
+4 / 1
$$ \text { 3, 3-Dimethyl-2-butanol cannot be prepared by : } $$
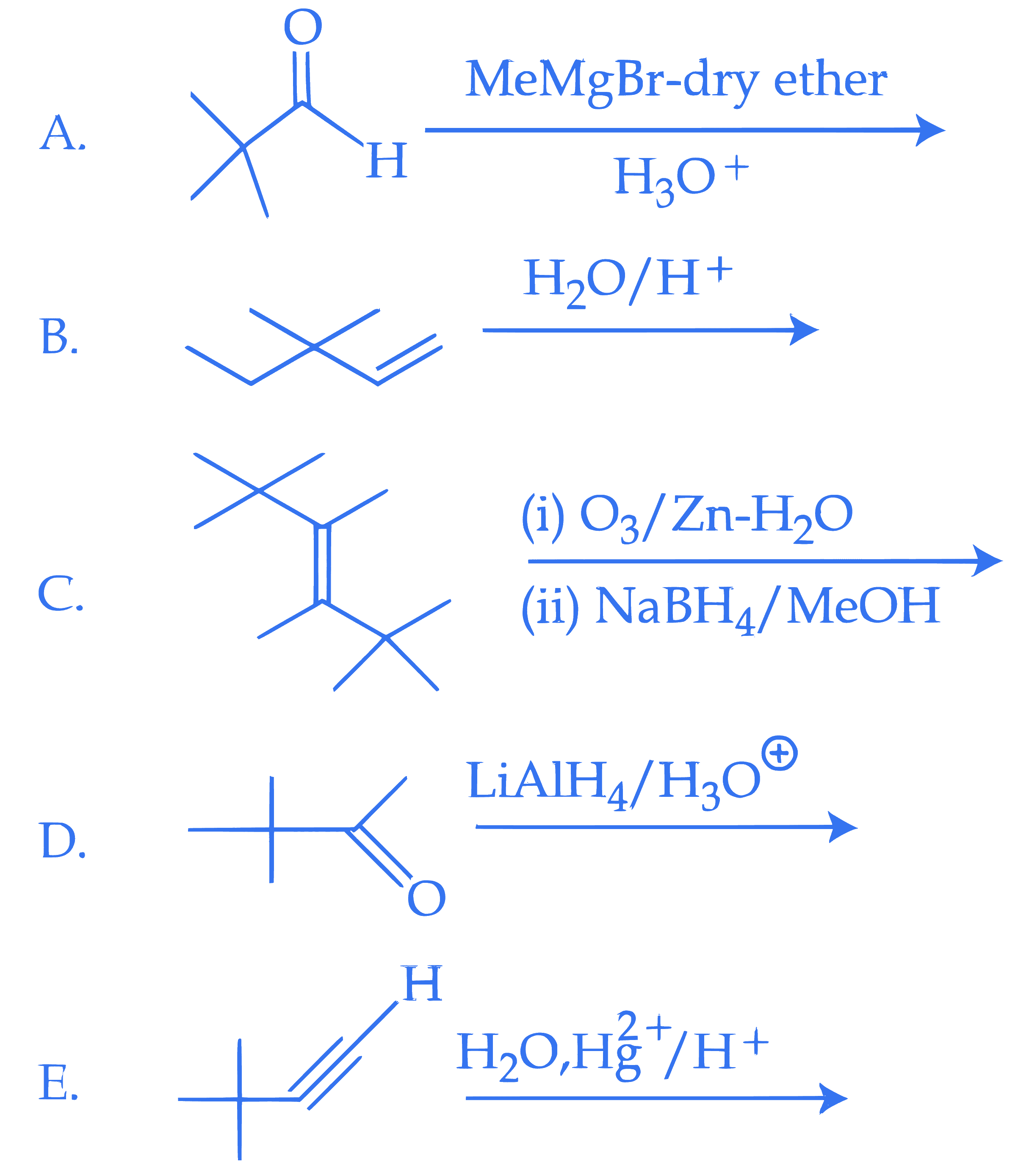 $$ \text { Choose the correct answer from the options given below : } $$
$$ \text { Choose the correct answer from the options given below : } $$
 $$ \text { Choose the correct answer from the options given below : } $$
$$ \text { Choose the correct answer from the options given below : } $$
Q25.
mcq single
+4 / 1
The energy of first (lowest) Balmer line of H atom is $x \mathrm{~J}$. The energy (in J) of second Balmer line of H atom is :
Q26.
numerical
+4 / 1
The mass of benzanilide obtained from the benzoylation reaction of 5.8 g of aniline, if yield of product is $82 \%$, is $\_\_\_\_$ g (nearest integer).
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
Q27.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The first ionization enthalpy of Cr is lower than that of Mn .
**Statement II :** The second and third ionization enthalpies of Cr are higher than those of Mn .
In the light of the above statements, choose the correct answer from the options given below :
Q28.
numerical
+4 / 1
Among the following oxides of $3 d$ elements, the number of mixed oxides are $\_\_\_\_$ .
$$ \mathrm{Ti}_2 \mathrm{O}_3, \mathrm{~V}_2 \mathrm{O}_4, \mathrm{Cr}_2 \mathrm{O}_3, \mathrm{Mn}_3 \mathrm{O}_4, \mathrm{Fe}_3 \mathrm{O}_4, \mathrm{Fe}_2 \mathrm{O}_3, \mathrm{Co}_3 \mathrm{O}_4 $$
Q29.
numerical
+4 / 1
Consider $\mathrm{A} \xrightarrow{\mathrm{k}_1} \mathrm{~B}$ and $\mathrm{C} \xrightarrow{\mathrm{k}_2} \mathrm{D}$ are two reactions. If the rate constant $\left(\mathrm{k}_1\right)$ of the $\mathrm{A} \longrightarrow \mathrm{B}$ reaction can be expressed by the following equation $\log _{10} \mathrm{k}=14.34-\frac{1.5 \times 10^4}{\mathrm{~T} / \mathrm{K}}$ and activation energy of $C \longrightarrow D$ reaction $\left(E a_2\right)$ is $\frac{1}{5}$ th of the $A \longrightarrow B$ reaction $\left(E a_1\right)$, then the value of $\left(E a_2\right)$ is
$\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (Nearest Integer)
Q30.
mcq single
+4 / 1
Correct statements regarding Arrhenius equation among the following are :
A. Factor $e^{-\mathrm{Ea} / \mathrm{RT}}$ corresponds to fraction of molecules having kinetic energy less than Ea.
B. At a given temperature, lower the Ea, faster is the reaction.
C. Increase in temperature by about $10^{\circ} \mathrm{C}$ doubles the rate of reaction.
D. Plot of $\log \mathrm{k}$ vs $\frac{1}{\mathrm{~T}}$ gives a straight line with slope $=-\frac{\mathrm{Ea}}{\mathrm{R}}$.
Choose the correct answer from the options given below :
Q31.
mcq single
+4 / 1
Among $\mathrm{H}_2 \mathrm{~S}, \mathrm{H}_2 \mathrm{O}, \mathrm{NF}_3, \mathrm{NH}_3$ and $\mathrm{CHCl}_3$, identify the molecule $(\mathrm{X})$ with lowest dipole moment value. The number of lone pairs of electrons present on the central atom of the molecule $(X)$ is :
Q32.
mcq single
+4 / 1
At $\mathrm{T}(\mathrm{K}), 100 \mathrm{~g}$ of $98 \% \mathrm{H}_2 \mathrm{SO}_4(\mathrm{w} / \mathrm{w})$ aqueous solution is mixed with 100 g of $49 \% \mathrm{H}_2 \mathrm{SO}_4(\mathrm{w} / \mathrm{w})$ aqueous solution. What is the mole fraction of $\mathrm{H}_2 \mathrm{SO}_4$ in the resultant solution?
(Given : Atomic mass $\mathrm{H}=1 \mathrm{u} ; \mathrm{S}=32 \mathrm{u} ; \mathrm{O}=16 \mathrm{u}$ ).
(Assume that temperature after mixing remains constant)
Q33.
mcq single
+4 / 1
Identify the correct statements :
A. Hydrated salts can be used as primary standard.
B. Primary standard should not undergo any reaction with air.
C. Reactions of primary standard with another substance should be instantaneous and stoichiometric.
D. Primary standard should not be soluble in water.
E. Primary standard should have low relative molar mass.
Choose the correct answer from the options given below :
Q34.
mcq single
+4 / 1
$\mathrm{A}+2 \mathrm{~B} \longrightarrow \mathrm{AB}_2$
36.0 g of 'A' (Molar mass : $60 \mathrm{~g} \mathrm{~mol}^{-1}$ ) and 56.0 g of ' B ' (Molar mass : $80 \mathrm{~g} \mathrm{~mol}^{-1}$ ) are allowed to react. Which of the following statements are correct ?
A. 'A' is the limiting reagent.
B. $77.0 \mathrm{~g}$ of $\mathrm{AB}_2$ is formed.
C. Molar mass of $\mathrm{AB}_2$ is $140 \mathrm{~g} \mathrm{~mol}^{-1}$.
D. $15.0 \mathrm{~g}$ of A is left unreacted after the completion of reaction.
Choose the correct answer from the options given below :
Q35.
mcq single
+4 / 1
Which of the following mixture gives a buffer solution with $\mathrm{pH}=9.25$ ?
Given : $\mathrm{pK}_{\mathrm{b}}\left(\mathrm{NH}_4 \mathrm{OH}\right)=4.75$
Q36.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\mathrm{C}\mathrm{Se}>\mathrm{Te}>\mathrm{Po}>\mathrm{O}$ is the correct order in terms of the magnitude of electron gain enthalpy values.
In the light of the above statements, choose the correct answer from the options given below :
Q37.
numerical
+4 / 1
If the enthalpy of sublimation of Li is $155 \mathrm{~kJ} \mathrm{~mol}^{-1}$, enthalpy of dissociation of $\mathrm{F}_2$ is $150 \mathrm{~kJ} \mathrm{~mol}^{-1}$, ionization enthalpy of Li is $520 \mathrm{~kJ} \mathrm{~mol}^{-1}$, electron gain enthalpy of F is $-313 \mathrm{~kJ} \mathrm{~mol}^{-1}$, standard enthalpy of formation of LiF is $-594 \mathrm{~kJ} \mathrm{~mol}^{-1}$. The magnitude of lattice enthalpy of LiF is $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (Nearest Integer)
Q38.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Elements ' $X$ ' and ' $Y$ ' are the most and least electronegative elements, respectively among $\mathrm{N}, \mathrm{As}, \mathrm{Sb}$ and P . The nature of the oxides $\mathrm{X}_2 \mathrm{O}_3$ and $\mathrm{Y}_2 \mathrm{O}_3$ is acidic and amphoteric, respectively.
**Statement II :** $\mathrm{BCl}_3$ is covalent in nature and gets hydrolysed in water. It produces $\left[\mathrm{B}(\mathrm{OH})_4\right]^{-}$ and $\left[\mathrm{B}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+}$ in aqueous medium.
In the light of the above statements, choose the correct answer from the options given below :
Q39.
mcq single
+4 / 1
$\left[\mathrm{Ni}\left(\mathrm{PPh}_3\right)_2 \mathrm{Cl}_2\right]$ is a paramagnetic complex. Identify the INCORRECT statements about this complex.
A. The complex exhibits geometrical isomerism.
B. The complex is white in colour.
C. The calculated spin-only magnetic moment of the complex is 2.84 BM .
D. The calculated CFSE (Crystal Field Stabilization Energy) of Ni in this complex is $-0.8 \Delta_{\mathrm{o}}$.
E. The geometrical arrangement of ligands in this complex is similar to that in $\mathrm{Ni}(\mathrm{CO})_4$.
Choose the correct answer from the options given below :
Q40.
mcq single
+4 / 1
The compound $\mathrm{A}, \mathrm{C}_8 \mathrm{H}_8 \mathrm{O}_2$ reacts with acetophenone to form a single product via cross-Aldol condensation. The compound A on reaction with conc. NaOH forms a substituted benzyl alcohol as one of the two products. The compound A is :
Q41.
mcq single
+4 / 1
When 1 g of compound $(\mathrm{X})$ is subjected to Kjeldahl's method for estimation of nitrogen, 15 mL 1 M $\mathrm{H}_2 \mathrm{SO}_4$ was neutralized by ammonia evolved. The percentage of nitrogen in compound $(\mathrm{X})$ is :
Q42.
mcq single
+4 / 1
The dibromo compound $[\mathrm{P}]$ (molecular formula: $\mathrm{C}_9 \mathrm{H}_{10} \mathrm{Br}_2$ ) when heated with excess sodamide followed by treatment with dilute HCl gives [Q]. On warming [Q] with mercuric sulphate and dilute sulphuric acid yield $[R]$ which gives positive Iodoform test but negative Tollen's test. The compound $[\mathrm{P}]$ is :
Q43.
mcq single
+4 / 1
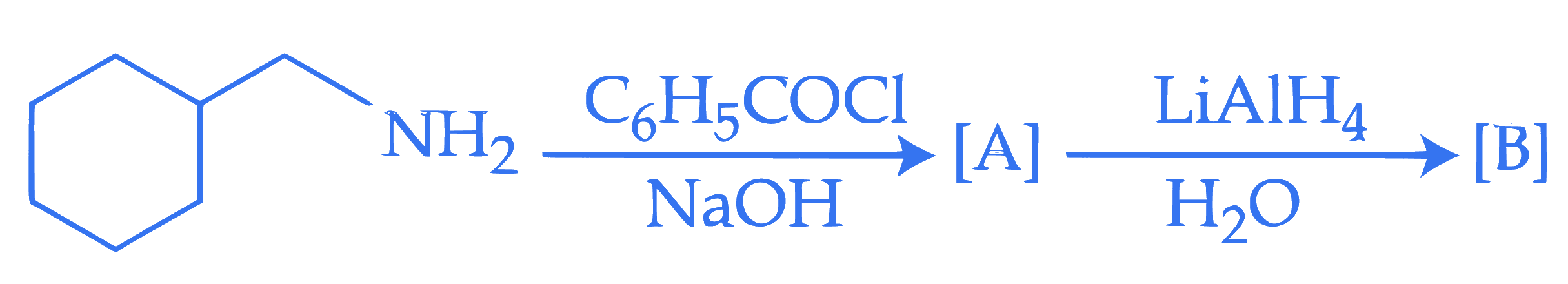 $$ \text { The final product }[\mathrm{B}] \text { is : } $$
$$ \text { The final product }[\mathrm{B}] \text { is : } $$
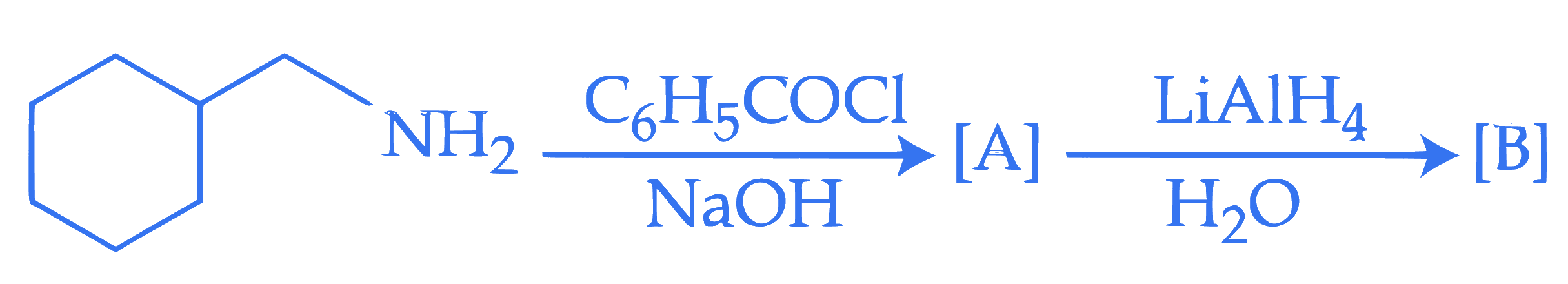 $$ \text { The final product }[\mathrm{B}] \text { is : } $$
$$ \text { The final product }[\mathrm{B}] \text { is : } $$
Q44.
mcq single
+4 / 1
Consider the following reaction :
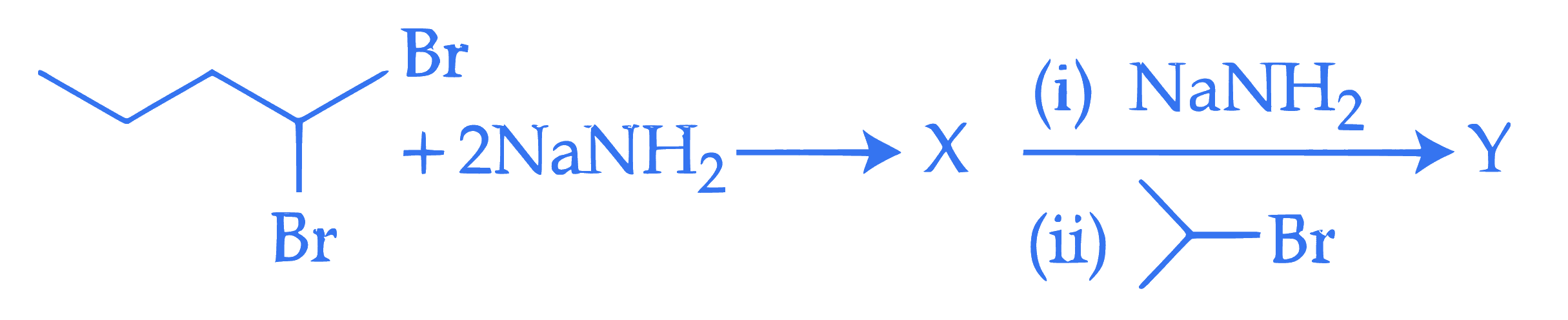 The product Y formed is :
The product Y formed is :
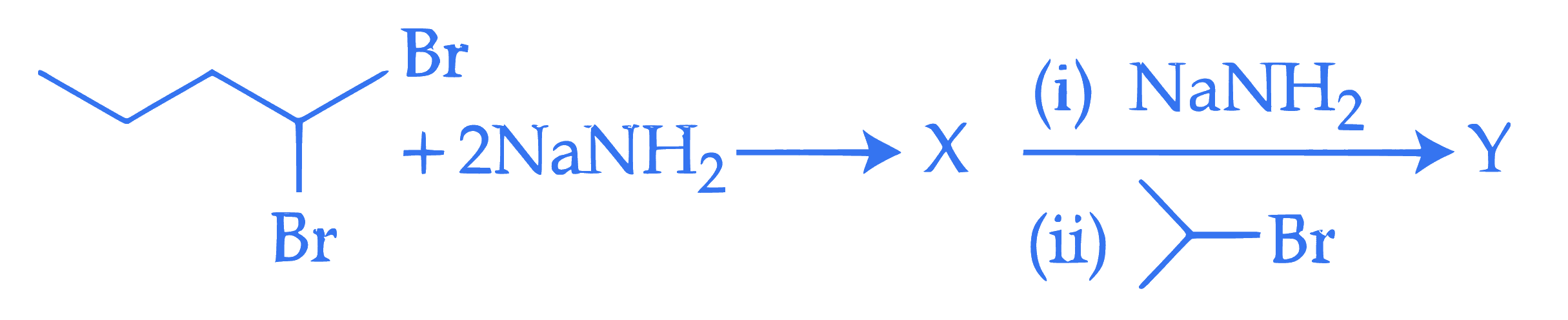 The product Y formed is :
The product Y formed is :
Q45.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Reaction of Glucose with
List - II
Product formed
A.
Hydroxylamine
I.
Gluconic acid
B.
$\mathrm{Br}_2$ water
II.
Glucose pentacetate
C.
Excess acetic anhydride
III.
Saccharic acid
D.
Concentrated $\mathrm{HNO}_3$
IV.
Glucoxime
Choose the correct answer from the options given below :
Q46.
mcq single
+4 / 1
$$ \text { The IUPAC name of the following compound is : } $$


Q47.
numerical
+4 / 1
Consider the following electrochemical cell :
$$ \mathrm{Pt}\left|\mathrm{O}_2(\mathrm{~g})(1 \mathrm{bar})\right| \mathrm{HCl}(\mathrm{aq}) \| \mathrm{M}^{2+}(\mathrm{aq}, 1.0 \mathrm{M}) \mid \mathrm{M}(\mathrm{~s}) $$
The pH above which, oxygen gas would start to evolve at anode is $\_\_\_\_$ (nearest integer).
$$ \left.\left[\begin{array}{ll} \text { Given : } & \mathrm{E}_{\mathrm{M}^{2+} / \mathrm{M}}^{\mathrm{o}}=0.994 \mathrm{~V} \\ & \mathrm{E}_{\mathrm{O}_2 / \mathrm{H}_2 \mathrm{O}}^{\mathrm{o}}=1.23 \mathrm{~V} \end{array}\right\} \text { standard reduction potential }
\\ \text {and} \frac{\mathrm{RT}}{\mathrm{F}}(2.303)=0.059 \mathrm{~V} \text {at the given condition}\right] $$
Q48.
mcq single
+4 / 1
Consider the following reduction processes :
$$ \begin{aligned} & \mathrm{Al}^{3+}+3 \mathrm{e}^{-} \longrightarrow \mathrm{Al}(\mathrm{~s}), \mathrm{E}^0=-1.66 \mathrm{~V} \\ & \mathrm{Fe}^{3+}+\mathrm{e}^{-} \longrightarrow \mathrm{Fe}^{2+}, \mathrm{E}^0=+0.77 \mathrm{~V} \\ & \mathrm{Co}^{3+}+\mathrm{e}^{-} \longrightarrow \mathrm{Co}^{2+}, \mathrm{E}^0=+1.81 \mathrm{~V} \\ & \mathrm{Cr}^{3+}+3 \mathrm{e}^{-} \longrightarrow \mathrm{Cr}(\mathrm{~s}), \mathrm{E}^0=-0.74 \mathrm{~V} \end{aligned} $$
The tendency to act as reducing agent decreases in the order :
Q49.
mcq single
+4 / 1
$$ \text { 3, 3-Dimethyl-2-butanol cannot be prepared by : } $$
 $$ \text { Choose the correct answer from the options given below : } $$
$$ \text { Choose the correct answer from the options given below : } $$
 $$ \text { Choose the correct answer from the options given below : } $$
$$ \text { Choose the correct answer from the options given below : } $$
Q50.
mcq single
+4 / 1
The energy of first (lowest) Balmer line of H atom is $x \mathrm{~J}$. The energy (in J) of second Balmer line of H atom is :
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
If the mean deviation about the median of the numbers $\mathrm{k}, 2 \mathrm{k}, 3 \mathrm{k}, \ldots ., 1000 \mathrm{k}$ is 500 , then $\mathrm{k}^2$ is equal to :
Q2.
numerical
+4 / 1
Let a vector $\overrightarrow{\mathrm{a}}=\sqrt{2} \hat{i}-\hat{j}+\lambda \hat{k}, \lambda>0$, make an obtuse angle with the vector $\overrightarrow{\mathrm{b}}=-\lambda^2 \hat{i}+4 \sqrt{2} \hat{j}+4 \sqrt{2} \hat{k}$ and an angle $\theta, \frac{\pi}{6}<\theta<\frac{\pi}{2}$, with the positive $z$-axis. If the set of all possible values of $\lambda$ is $(\alpha, \beta)-\{\gamma\}$, then $\alpha+\beta+\gamma$ is equal to $\_\_\_\_$ .
Q3.
mcq single
+4 / 1
Let $\vec{a}=2 \hat{i}-\hat{j}+\hat{k}$ and $\vec{b}=\lambda \hat{j}+2 \hat{k}, \lambda \in \boldsymbol{Z}$ be two vectors. Let $\vec{c}=\vec{a} \times \vec{b}$ and $\vec{d}$ be a vector of magnitude 2 in $y z$-plane. If $|\vec{c}|=\sqrt{53}$, then the maximum possible value of $(\vec{c} \cdot \vec{d})^2$ is equal to :
Q4.
mcq single
+4 / 1
Let $\alpha, \beta$ be the roots of the quadratic equation $12 x^2-20 x+3 \lambda=0, \lambda \in \mathbf{Z}$. If $\frac{1}{2} \leqslant|\beta-\alpha| \leqslant \frac{3}{2}$, then the sum of all possible values of $\lambda$ is :
Q5.
mcq single
+4 / 1
Let L be the line $\frac{x+1}{2}=\frac{y+1}{3}=\frac{z+3}{6}$ and let S be the set of all points $(\mathrm{a}, \mathrm{b}, \mathrm{c})$ on L , whose distance from the line $\frac{x+1}{2}=\frac{y+1}{3}=\frac{z-9}{0}$ along the line $L$ is 7 . Then $\sum\limits_{(a, b, c) \in S}(a+b+c)$ is equal to :
Q6.
numerical
+4 / 1
Suppose $\mathrm{a}, \mathrm{b}, \mathrm{c}$ are in A.P. and $\mathrm{a}^2, 2 \mathrm{~b}^2, \mathrm{c}^2$ are in G.P. If $\mathrm{a}<\mathrm{b}<\mathrm{c}$ and $\mathrm{a}+\mathrm{b}+\mathrm{c}=1$, then $9\left(\mathrm{a}^2+\mathrm{b}^2+\mathrm{c}^2\right)$ is equal to $\_\_\_\_$ .
Q7.
mcq single
+4 / 1
If $\lim\limits_{x \rightarrow 0} \frac{\mathrm{e}^{(\mathrm{a}-1) x}+2 \cos \mathrm{~b} x+(\mathrm{c}-2) \mathrm{e}^{-x}}{x \cos x-\log _{\mathrm{e}}(1+x)}=2$, then $\mathrm{a}^2+\mathrm{b}^2+\mathrm{c}^2$ is equal to :
Q8.
mcq single
+4 / 1
Let $[\cdot]$ denote the greatest integer function, and let $f(x)=\min \left\{\sqrt{2} x, x^2\right\}$.
Let $\mathrm{S}=\left\{x \in(-2,2)\right.$ : the function $\mathrm{g}(x)=|x|\left[x^2\right]$ is discontinuous at $\left.x\right\}$.
Then $\sum\limits_{x \in \mathrm{~S}} f(x)$ equals
Q9.
numerical
+4 / 1
Let $\cos (\alpha+\beta)=-\frac{1}{10}$ and $\sin (\alpha-\beta)=\frac{3}{8}$, where $0<\alpha<\frac{\pi}{3}$ and $0<\beta<\frac{\pi}{4}$. If $\tan 2 \alpha=\frac{3(1-r \sqrt{5})}{\sqrt{11}(s+\sqrt{5})}, r, s \in N$, then $r+s$ is equal to $\_\_\_\_$ .
Q10.
mcq single
+4 / 1
The area of the region $\mathrm{A}=\left\{(x, y): 4 x^2+y^2 \leqslant 8\right.$ and $\left.y^2 \leqslant 4 x\right\}$ is:
Q11.
mcq single
+4 / 1
Let n be the number obtained on rolling a fair die. If the probability that the system
$$ \begin{aligned} & x-\mathrm{n} y+z=6 \\ & x+(\mathrm{n}-2) y+(\mathrm{n}+1) z=8 \\ & \quad(\mathrm{n}-1) y+z=1 \end{aligned} $$
has a unique solution is $\frac{k}{6}$, then the sum of $k$ and all possible values of $n$ is :
Q12.
mcq single
+4 / 1
If $X=\left[\begin{array}{l}x \\ y \\ z\end{array}\right]$ is a solution of the system of equations $A X=B$, where $\operatorname{adj} A=\left[\begin{array}{ccc}4 & 2 & 2 \\ -5 & 0 & 5 \\ 1 & -2 & 3\end{array}\right]$ and $\mathrm{B}=\left[\begin{array}{l}4 \\ 0 \\ 2\end{array}\right]$, then $|x+y+z|$ is equal to :
Q13.
mcq single
+4 / 1
Let the locus of the mid-point of the chord through the origin $O$ of the parabola $y^2=4 x$ be the curve S . Let P be any point on S . Then the locus of the point, which internally divides OP in the ratio 3 : 1, is :
Q14.
mcq single
+4 / 1
Let $\mathrm{C}_{\mathrm{r}}$ denote the coefficient of $x^{\mathrm{r}}$ in the binomial expansion of $(1+x)^{\mathrm{n}}, \mathrm{n} \in \mathrm{N}, 0 \leq \mathrm{r} \leq \mathrm{n}$. If
$P_n=C_0-C_1+\frac{2^2}{3} C_2-\frac{2^3}{4} C_3+\ldots . .+\frac{(-2)^n}{n+1} C_n$, then the value of $\sum\limits_{n=1}^{25} \frac{1}{P_{2 n}}$ equals.
Q15.
mcq single
+4 / 1
Among the statements
$(S 1)$ : If $A(5,-1)$ and $B(-2,3)$ are two vertices of a triangle, whose orthocentre is $(0,0)$, then its third vertex is $(-4,-7)$
and
(S2) : If positive numbers $2 a, b, c$ are three consecutive terms of an A.P., then the lines $a x+b y+c=0$ are concurrent at $(2,-2)$,
Q16.
numerical
+4 / 1
Let S be the set of the first 11 natural numbers. Then the number of elements in $A=\{B \subseteq S: n(B) \geqslant 2$ and the product of all elements of $B$ is even $\}$ is $\_\_\_\_$ .
Q17.
mcq single
+4 / 1
The number of elements in the relation $\mathrm{R}=\left\{(x, y): 4 x^2+y^2<52, x, y \in \mathbf{Z}\right\}$ is
Q18.
mcq single
+4 / 1
Let $\mathrm{S}=\left\{z \in \mathbb{C}: 4 z^2+\bar{z}=0\right\}$. Then $\sum\limits_{z \in \mathrm{~S}}|z|^2$ is equal to:
Q19.
mcq single
+4 / 1
Let $\mathrm{P}(10,2 \sqrt{15})$ be a point on the hyperbola $\frac{x^2}{\mathrm{a}^2}-\frac{y^2}{\mathrm{~b}^2}=1$, whose foci are S and $\mathrm{S}^{\prime}$. If the length of its latus rectum is 8 , then the square of the area of $\Delta \mathrm{PSS}^{\prime}$ is equal to :
Q20.
mcq single
+4 / 1
Let $f(x)=[x]^2-[x+3]-3, x \in \mathbf{R}$, where [.] is the greatest integer funtion. Then
Q21.
mcq single
+4 / 1
Let the domain of the function $f(x)=\log _3 \log _5\left(7-\log _2\left(x^2-10 x+85\right)\right)+\sin ^{-1}\left(\left|\frac{3 x-7}{17-x}\right|\right)$ be $(\alpha, \beta]$. Then $\alpha+\beta$ is equal to :
Q22.
mcq single
+4 / 1
Let $f$ and $g$ be functions satisfying $f(x+y)=f(x) f(y), f(1)=7$ and $g(x+y)=g(x y), g(1)=1$, for all $x, y \in \mathbf{N}$. If $\sum\limits_{x=1}^{\mathrm{n}}\left(\frac{f(x)}{\mathrm{g}(x)}\right)=19607$, then n is equal to :
Q23.
mcq single
+4 / 1
If $y=y(x)$ satisfies the differential equation $16(\sqrt{x+9 \sqrt{x}})(4+\sqrt{9+\sqrt{x}}) \cos y \mathrm{~d} y=(1+2 \sin y) \mathrm{d} x, x>0$ and $y(256)=\frac{\pi}{2}, y(49)=\alpha$, then $2 \sin \alpha$ is equal to :
Q24.
mcq single
+4 / 1
Let S and $\mathrm{S}^{\prime}$ be the foci of the ellipse $\frac{x^2}{25}+\frac{y^2}{9}=1$ and $\mathrm{P}(\alpha, \beta)$ be a point on the ellipse in the first quadrant. If $(\mathrm{SP})^2+\left(\mathrm{S}^{\prime} \mathrm{P}\right)^2-\mathrm{SP} \cdot \mathrm{S}^{\prime} \mathrm{P}=37$, then $\alpha^2+\beta^2$ is equal to :
Q25.
numerical
+4 / 1
Let [.] be the greatest integer function. If $\alpha=\int\limits_0^{64}\left(x^{1 / 3}-\left[x^{1 / 3}\right]\right) \mathrm{d} x$, then $\frac{1}{\pi} \int\limits_0^{\alpha \pi}\left(\frac{\sin ^2 \theta}{\sin ^6 \theta+\cos ^6 \theta}\right) \mathrm{d} \theta$ is equal to $\_\_\_\_$ .
Q26.
mcq single
+4 / 1
If the mean deviation about the median of the numbers $\mathrm{k}, 2 \mathrm{k}, 3 \mathrm{k}, \ldots ., 1000 \mathrm{k}$ is 500 , then $\mathrm{k}^2$ is equal to :
Q27.
numerical
+4 / 1
Let a vector $\overrightarrow{\mathrm{a}}=\sqrt{2} \hat{i}-\hat{j}+\lambda \hat{k}, \lambda>0$, make an obtuse angle with the vector $\overrightarrow{\mathrm{b}}=-\lambda^2 \hat{i}+4 \sqrt{2} \hat{j}+4 \sqrt{2} \hat{k}$ and an angle $\theta, \frac{\pi}{6}<\theta<\frac{\pi}{2}$, with the positive $z$-axis. If the set of all possible values of $\lambda$ is $(\alpha, \beta)-\{\gamma\}$, then $\alpha+\beta+\gamma$ is equal to $\_\_\_\_$ .
Q28.
mcq single
+4 / 1
Let $\vec{a}=2 \hat{i}-\hat{j}+\hat{k}$ and $\vec{b}=\lambda \hat{j}+2 \hat{k}, \lambda \in \boldsymbol{Z}$ be two vectors. Let $\vec{c}=\vec{a} \times \vec{b}$ and $\vec{d}$ be a vector of magnitude 2 in $y z$-plane. If $|\vec{c}|=\sqrt{53}$, then the maximum possible value of $(\vec{c} \cdot \vec{d})^2$ is equal to :
Q29.
mcq single
+4 / 1
Let $\alpha, \beta$ be the roots of the quadratic equation $12 x^2-20 x+3 \lambda=0, \lambda \in \mathbf{Z}$. If $\frac{1}{2} \leqslant|\beta-\alpha| \leqslant \frac{3}{2}$, then the sum of all possible values of $\lambda$ is :
Q30.
mcq single
+4 / 1
Let L be the line $\frac{x+1}{2}=\frac{y+1}{3}=\frac{z+3}{6}$ and let S be the set of all points $(\mathrm{a}, \mathrm{b}, \mathrm{c})$ on L , whose distance from the line $\frac{x+1}{2}=\frac{y+1}{3}=\frac{z-9}{0}$ along the line $L$ is 7 . Then $\sum\limits_{(a, b, c) \in S}(a+b+c)$ is equal to :
Q31.
numerical
+4 / 1
Suppose $\mathrm{a}, \mathrm{b}, \mathrm{c}$ are in A.P. and $\mathrm{a}^2, 2 \mathrm{~b}^2, \mathrm{c}^2$ are in G.P. If $\mathrm{a}<\mathrm{b}<\mathrm{c}$ and $\mathrm{a}+\mathrm{b}+\mathrm{c}=1$, then $9\left(\mathrm{a}^2+\mathrm{b}^2+\mathrm{c}^2\right)$ is equal to $\_\_\_\_$ .
Q32.
mcq single
+4 / 1
If $\lim\limits_{x \rightarrow 0} \frac{\mathrm{e}^{(\mathrm{a}-1) x}+2 \cos \mathrm{~b} x+(\mathrm{c}-2) \mathrm{e}^{-x}}{x \cos x-\log _{\mathrm{e}}(1+x)}=2$, then $\mathrm{a}^2+\mathrm{b}^2+\mathrm{c}^2$ is equal to :
Q33.
mcq single
+4 / 1
Let $[\cdot]$ denote the greatest integer function, and let $f(x)=\min \left\{\sqrt{2} x, x^2\right\}$.
Let $\mathrm{S}=\left\{x \in(-2,2)\right.$ : the function $\mathrm{g}(x)=|x|\left[x^2\right]$ is discontinuous at $\left.x\right\}$.
Then $\sum\limits_{x \in \mathrm{~S}} f(x)$ equals
Q34.
numerical
+4 / 1
Let $\cos (\alpha+\beta)=-\frac{1}{10}$ and $\sin (\alpha-\beta)=\frac{3}{8}$, where $0<\alpha<\frac{\pi}{3}$ and $0<\beta<\frac{\pi}{4}$. If $\tan 2 \alpha=\frac{3(1-r \sqrt{5})}{\sqrt{11}(s+\sqrt{5})}, r, s \in N$, then $r+s$ is equal to $\_\_\_\_$ .
Q35.
mcq single
+4 / 1
The area of the region $\mathrm{A}=\left\{(x, y): 4 x^2+y^2 \leqslant 8\right.$ and $\left.y^2 \leqslant 4 x\right\}$ is:
Q36.
mcq single
+4 / 1
Let n be the number obtained on rolling a fair die. If the probability that the system
$$ \begin{aligned} & x-\mathrm{n} y+z=6 \\ & x+(\mathrm{n}-2) y+(\mathrm{n}+1) z=8 \\ & \quad(\mathrm{n}-1) y+z=1 \end{aligned} $$
has a unique solution is $\frac{k}{6}$, then the sum of $k$ and all possible values of $n$ is :
Q37.
mcq single
+4 / 1
If $X=\left[\begin{array}{l}x \\ y \\ z\end{array}\right]$ is a solution of the system of equations $A X=B$, where $\operatorname{adj} A=\left[\begin{array}{ccc}4 & 2 & 2 \\ -5 & 0 & 5 \\ 1 & -2 & 3\end{array}\right]$ and $\mathrm{B}=\left[\begin{array}{l}4 \\ 0 \\ 2\end{array}\right]$, then $|x+y+z|$ is equal to :
Q38.
mcq single
+4 / 1
Let the locus of the mid-point of the chord through the origin $O$ of the parabola $y^2=4 x$ be the curve S . Let P be any point on S . Then the locus of the point, which internally divides OP in the ratio 3 : 1, is :
Q39.
mcq single
+4 / 1
Let $\mathrm{C}_{\mathrm{r}}$ denote the coefficient of $x^{\mathrm{r}}$ in the binomial expansion of $(1+x)^{\mathrm{n}}, \mathrm{n} \in \mathrm{N}, 0 \leq \mathrm{r} \leq \mathrm{n}$. If
$P_n=C_0-C_1+\frac{2^2}{3} C_2-\frac{2^3}{4} C_3+\ldots . .+\frac{(-2)^n}{n+1} C_n$, then the value of $\sum\limits_{n=1}^{25} \frac{1}{P_{2 n}}$ equals.
Q40.
mcq single
+4 / 1
Among the statements
$(S 1)$ : If $A(5,-1)$ and $B(-2,3)$ are two vertices of a triangle, whose orthocentre is $(0,0)$, then its third vertex is $(-4,-7)$
and
(S2) : If positive numbers $2 a, b, c$ are three consecutive terms of an A.P., then the lines $a x+b y+c=0$ are concurrent at $(2,-2)$,
Q41.
numerical
+4 / 1
Let S be the set of the first 11 natural numbers. Then the number of elements in $A=\{B \subseteq S: n(B) \geqslant 2$ and the product of all elements of $B$ is even $\}$ is $\_\_\_\_$ .
Q42.
mcq single
+4 / 1
The number of elements in the relation $\mathrm{R}=\left\{(x, y): 4 x^2+y^2<52, x, y \in \mathbf{Z}\right\}$ is
Q43.
mcq single
+4 / 1
Let $\mathrm{S}=\left\{z \in \mathbb{C}: 4 z^2+\bar{z}=0\right\}$. Then $\sum\limits_{z \in \mathrm{~S}}|z|^2$ is equal to:
Q44.
mcq single
+4 / 1
Let $\mathrm{P}(10,2 \sqrt{15})$ be a point on the hyperbola $\frac{x^2}{\mathrm{a}^2}-\frac{y^2}{\mathrm{~b}^2}=1$, whose foci are S and $\mathrm{S}^{\prime}$. If the length of its latus rectum is 8 , then the square of the area of $\Delta \mathrm{PSS}^{\prime}$ is equal to :
Q45.
mcq single
+4 / 1
Let $f(x)=[x]^2-[x+3]-3, x \in \mathbf{R}$, where [.] is the greatest integer funtion. Then
Q46.
mcq single
+4 / 1
Let the domain of the function $f(x)=\log _3 \log _5\left(7-\log _2\left(x^2-10 x+85\right)\right)+\sin ^{-1}\left(\left|\frac{3 x-7}{17-x}\right|\right)$ be $(\alpha, \beta]$. Then $\alpha+\beta$ is equal to :
Q47.
mcq single
+4 / 1
Let $f$ and $g$ be functions satisfying $f(x+y)=f(x) f(y), f(1)=7$ and $g(x+y)=g(x y), g(1)=1$, for all $x, y \in \mathbf{N}$. If $\sum\limits_{x=1}^{\mathrm{n}}\left(\frac{f(x)}{\mathrm{g}(x)}\right)=19607$, then n is equal to :
Q48.
mcq single
+4 / 1
If $y=y(x)$ satisfies the differential equation $16(\sqrt{x+9 \sqrt{x}})(4+\sqrt{9+\sqrt{x}}) \cos y \mathrm{~d} y=(1+2 \sin y) \mathrm{d} x, x>0$ and $y(256)=\frac{\pi}{2}, y(49)=\alpha$, then $2 \sin \alpha$ is equal to :
Q49.
mcq single
+4 / 1
Let S and $\mathrm{S}^{\prime}$ be the foci of the ellipse $\frac{x^2}{25}+\frac{y^2}{9}=1$ and $\mathrm{P}(\alpha, \beta)$ be a point on the ellipse in the first quadrant. If $(\mathrm{SP})^2+\left(\mathrm{S}^{\prime} \mathrm{P}\right)^2-\mathrm{SP} \cdot \mathrm{S}^{\prime} \mathrm{P}=37$, then $\alpha^2+\beta^2$ is equal to :
Q50.
numerical
+4 / 1
Let [.] be the greatest integer function. If $\alpha=\int\limits_0^{64}\left(x^{1 / 3}-\left[x^{1 / 3}\right]\right) \mathrm{d} x$, then $\frac{1}{\pi} \int\limits_0^{\alpha \pi}\left(\frac{\sin ^2 \theta}{\sin ^6 \theta+\cos ^6 \theta}\right) \mathrm{d} \theta$ is equal to $\_\_\_\_$ .
Physics
Physics
Q1.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** An object moves from position $r_1$ to position $r_2$ under a conservative force field $\vec{F}$. The work done by the force is $W=-\int\limits_{r_1}^{r_2} \vec{F} \cdot \overrightarrow{d r}$.
**Statement II :** Any object moving from one location to another location can follow infinite number of paths. Therefore, the amount of work done by the object changes with the path it follows for a conservative force.
In the light of the above statements, choose the correct answer from the options given below :
Q2.
mcq single
+4 / 1
The correct truth table for the given input data of the following logic gate is :
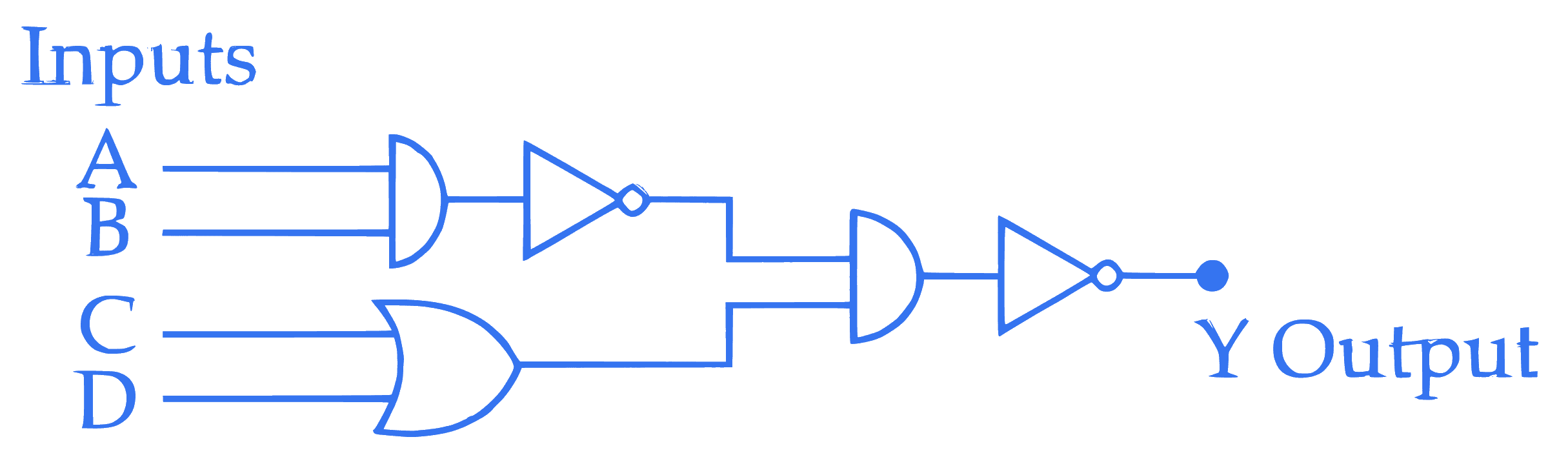

Q3.
mcq single
+4 / 1
Which of the following are true for a single slit diffraction?
A. Width of central maxima increases with increase in wavelength keeping slit width constant.
B. Width of central maxima increases with decrease in wavelength keeping slit width constant.
C. Width of central maxima increases with decrease in slit width at constant wavelength.
D. Width of central maxima increases with increase in slit width at constant wavelength.
E. Brightness of central maxima increases for decrease in wavelength at constant slit width.
Q4.
mcq single
+4 / 1
The wavelength of light, while it is passing through water is 540 nm . The refractive index of water is $4 / 3$. The wavelength of the same light when it is passing through a transparent medium having refractive index of $3 / 2$ is $\_\_\_\_$ nm.
Q5.
mcq single
+4 / 1
Using a simple pendulum experiment $g$ is determind by measuring its time period $T$. Which of the following plots represent the correct relation between the pendulum length $L$ and time period $T$ ?
Q6.
mcq single
+4 / 1
A uniform bar of length 12 cm and mass 20 m lies on a smooth horizontal table. Two point masses $m$ and $2 m$ are moving in opposite directions with same speed of $v$ and in the same plane as the bar, as shown in figure. These masses strike the bar simultaneously and get stuck to it. After collision the entire system is rotating with angular frequency $\omega$. The ratio of $v$ and $\omega$ is :
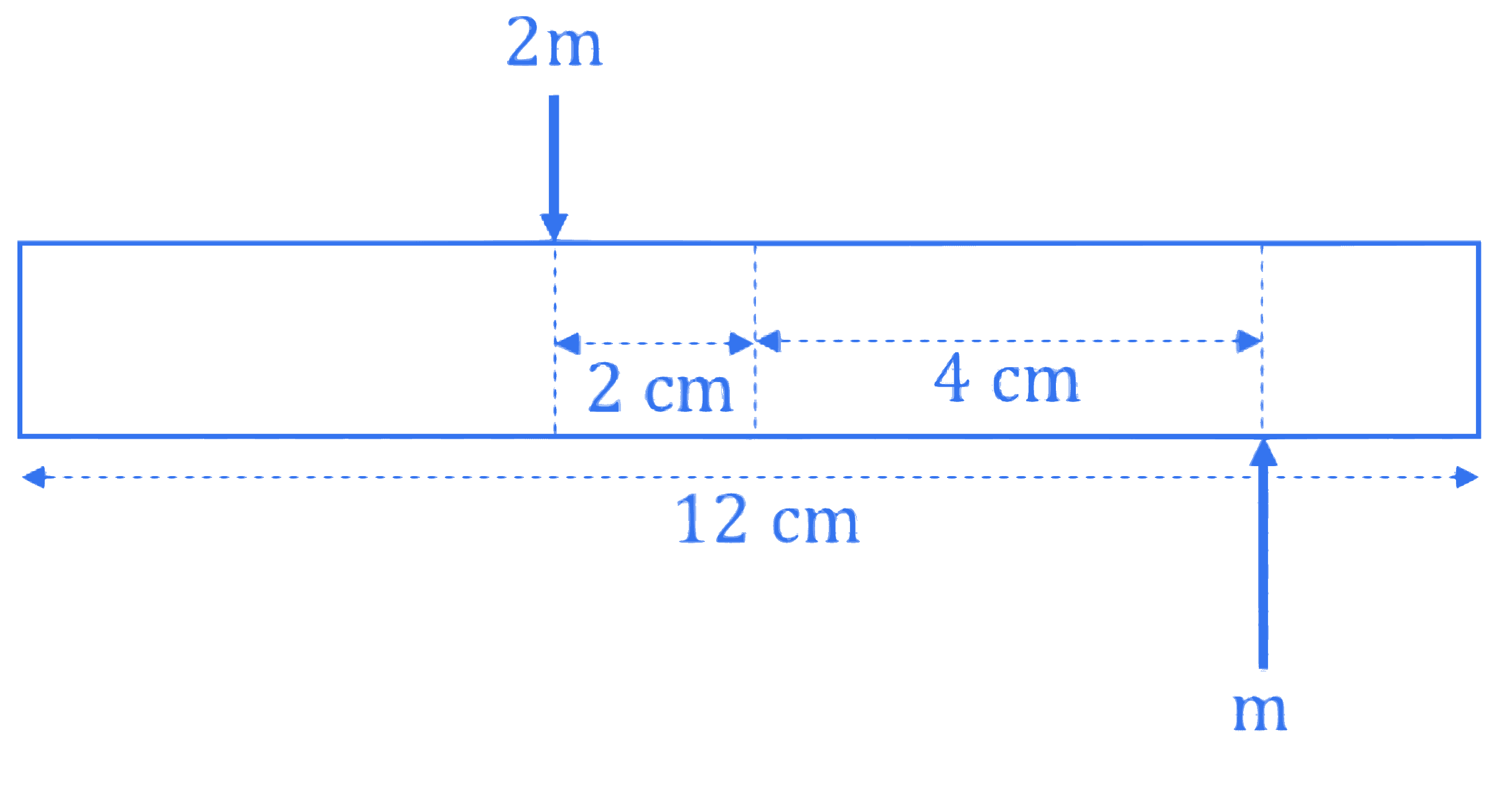

Q7.
numerical
+4 / 1
Two masses $m$ and 2 m are connected by a light string going over a pulley (disc) of mass 30 m with radius $r=0.1 \mathrm{~m}$. The pulley is mounted in a vertical plane and it is free to rotate about its axis. The 2 m mass is released from rest and its speed when it has descended through a height of 3.6 m is
$\_\_\_\_$ $\mathrm{m} / \mathrm{s}$. (Assume string does not slip and $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q8.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** For a mechanical system of many particles total kinetic energy is the sum of kinetic energies of all the particles.
**Statement II :** The total kinetic energy can be the sum of kinetic energy of the center of mass w.r.t to the origin and the kinetic energy of all the particles w.r.t. the center of mass as the reference.
In the light of the above statements, choose the correct answer from the options given below :
Q9.
numerical
+4 / 1
A conducting circular loop is rotated about its diameter at a constant angular speed of $100 \mathrm{rad} / \mathrm{s}$ in a magnetic field of 0.5 T perpendicular to the axis of rotation. When the loop is rotated by $30^{\circ}$ from the horizontal position, the induced EMF is 15.4 mV . The radius of the loop is $\_\_\_\_$ mm.
$$ \left(\text { Take } \pi=\frac{22}{7}\right) $$
Q10.
mcq single
+4 / 1
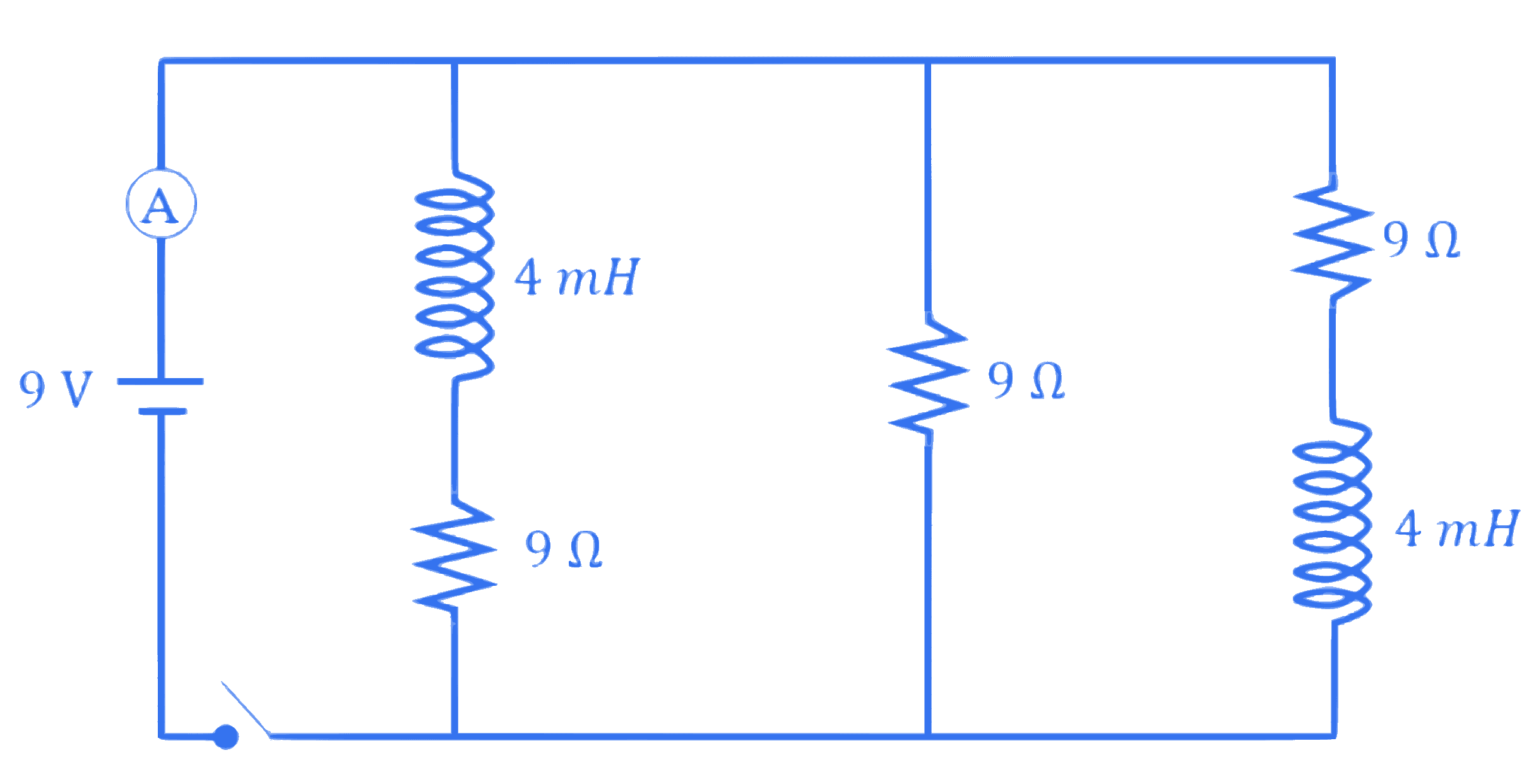
Figure shows the circuit that contains three resistances ( $9 \Omega$ each) and two inductors ( 4 mH each). The reading of ammeter at the moment switch $K$ is turned ON , is $\_\_\_\_$ A.

Q11.
numerical
+4 / 1
An insulated cylinder of volume $60 \mathrm{~cm}^3$ is filled with a gas at $27^{\circ} \mathrm{C}$ and 2 atmospheric pressure. Then the gas is compressed making the final volume as $20 \mathrm{~cm}^3$ while allowing the temperature to rise to $77^{\circ} \mathrm{C}$. The final pressure is $\_\_\_\_$ atmospheric pressure.
Q12.
mcq single
+4 / 1
Consider two boxes containing ideal gases $A$ and $B$ such that their temperatures, pressures and number densities are same. The molecular size of $A$ is half of that of $B$ and mass of molecule $A$ is four times that of $B$. If the collision frequency in gas $B$ is $32 \times 10^{18} / \mathrm{s}$ then collision frequency in gas $A$ is $\_\_\_\_$ /s.
Q13.
mcq single
+4 / 1
When a part of a straight capillary tube is placed vertically in a liquid, the liquid raises upto certain height $h$. If the inner radius of the capillary tube, density of the liquid and surface tension of the liquid decrease by $1 \%$ each, then the height of the liquid in the tube will change by $\_\_\_\_$ $\%$.
Q14.
mcq single
+4 / 1
Light is incident on a metallic plate having work function $110 \times 10^{-20} \mathrm{~J}$. If the produced photoelectrons have zero kinetic energy then the angular frequency of the incident light is $\_\_\_\_$ rad/s. $\left(\mathrm{h}=6.63 \times 10^{-34} \mathrm{~J} . \mathrm{s}\right)$.
Q15.
mcq single
+4 / 1
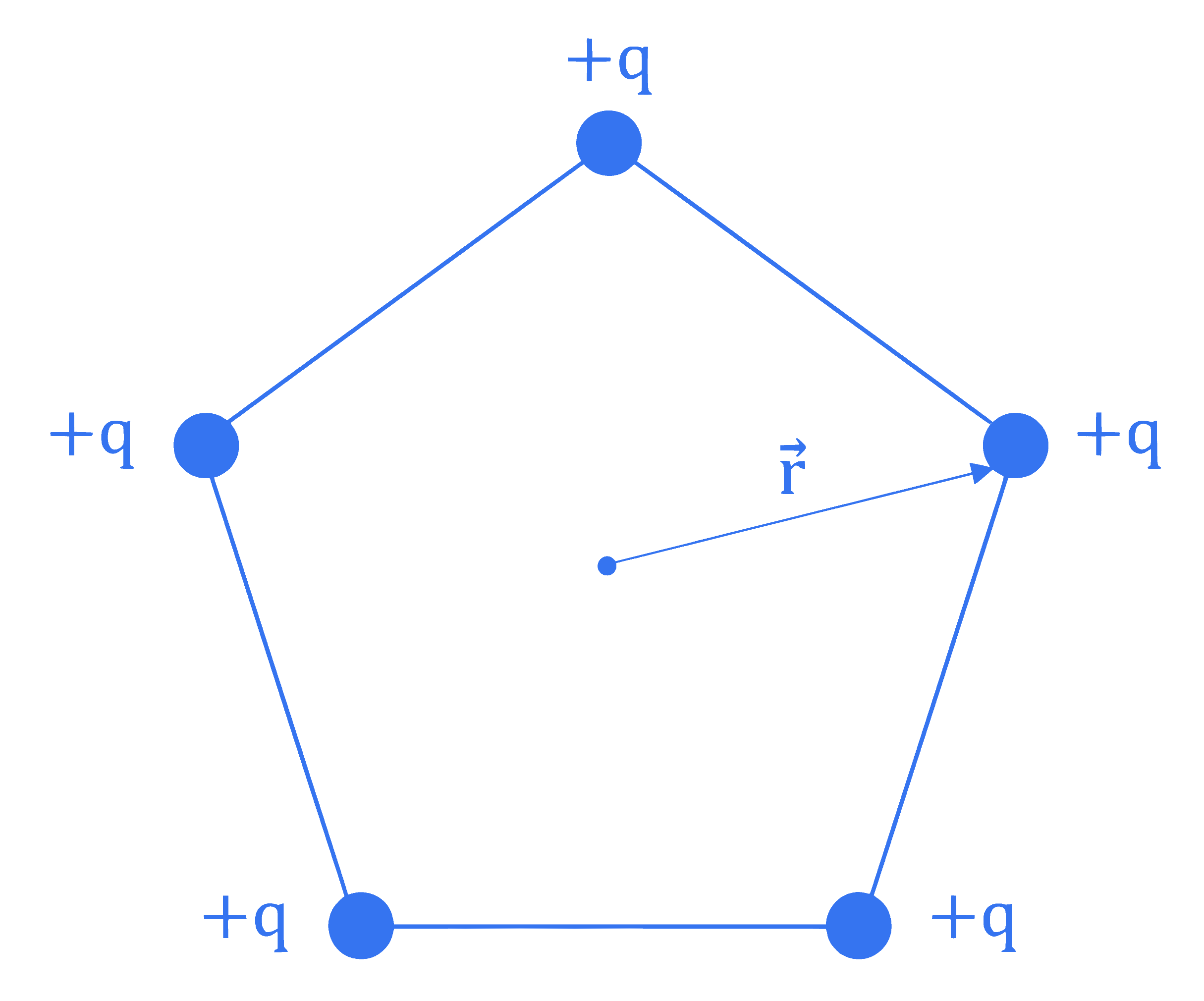
Five positive charges each having charge $q$ are placed at the vertices of a pentagon as shown in the figure. The electric potential $(V)$ and the electric field $(\vec{E})$ at the center $O$ of the pentagon due to these five positive charges are :

Q16.
mcq single
+4 / 1
Three small identical bubbles of water having same charge on each coalesce to form a bigger bubble. Then the ratio of the potentials on one initial bubble and that on the resultant bigger bubble is :
Q17.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** A satellite is moving around earth in the orbit very close to the earth surface. The time period of revolution of satellite depends upon the density of earth.
**Statement II :** The time period of revolution of the satellite is $T=2 \pi \sqrt{\frac{R_e}{g}}$ (for satellite very close to the earth surface), where $R_{\mathrm{e}}$ radius of earth and $g$ acceleration due to gravity. In the light of the above statements, choose the correct answer from the options given below :
Q18.
numerical
+4 / 1
A capacitor $P$ with capacitance $10 \times 10^{-6} \mathrm{~F}$ is fully charged with a potential difference of 6.0 V and disconnected from the battery. The charged capacitor $P$ is connected across another capacitor $Q$ with capacitance $20 \times 10^{-6} \mathrm{~F}$. The charge on capacitor $Q$ when equilibrium is established will be $\alpha \times 10^{-5} C$ (assume capacitor $Q$ does not have any charge initially), the value of $\alpha$ is $\_\_\_\_$ .
Q19.
mcq single
+4 / 1
In parallax method for the determination of focal length of a concave mirror, the object should always be placed:
Q20.
mcq single
+4 / 1
If $\epsilon, E$ and $t$ represent the free space permittivity, electric field and time respectively, then the unit of $\frac{\epsilon E}{t}$ will be :
Q21.
mcq single
+4 / 1
The smallest wavelength of Lyman series is 91 nm . The difference between the largest wavelengths of Paschen and Balmer series is nearly $\_\_\_\_$ nm.
Q22.
mcq single
+4 / 1
In an open organ pipe $\nu_3$ and $\nu_6$ are $3^{\text {rd }}$ and $6^{\text {th }}$ harmonic frequencies, respectively. If $\nu_6-\nu_3=2200 \mathrm{~Hz}$ then length of the pipe is $\_\_\_\_$ mm .
(Take velocity of sound in air is $330 \mathrm{~m} / \mathrm{s}$.)
Q23.
numerical
+4 / 1
A cylindrical conductor of length 2 m and area of cross-section $0.2 \mathrm{~mm}^2$ carries an electric current of 1.6 A when its ends are connected to a 2 V battery. Mobility of electrons in the conductor is $\alpha \times 10^{-3} \mathrm{~m}^2 / \mathrm{V} . \mathrm{s}$. The value of $\alpha$ is :
(electron concentration $=5 \times 10^{28} / \mathrm{m}^3$ and electron charge $=1.6 \times 10^{-19} \mathrm{C}$ )
Q24.
mcq single
+4 / 1
An electric power line having total resistance of $2 \Omega$, delivers 1 kW of power at 250 V . The percentage efficiency of transmission line is $\_\_\_\_$ .
Q25.
mcq single
+4 / 1
A laser beam has intensity of $4.0 \times 10^{14} \mathrm{~W} / \mathrm{m}^2$. The amplitude of magnetic field associated with beam is $\_\_\_\_$ T.
(Take $\epsilon_{\mathrm{o}}=8.85 \times 10^{-12} \mathrm{C}^2 / \mathrm{Nm}^2$ and $\mathrm{c}=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ )
Q26.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** An object moves from position $r_1$ to position $r_2$ under a conservative force field $\vec{F}$. The work done by the force is $W=-\int\limits_{r_1}^{r_2} \vec{F} \cdot \overrightarrow{d r}$.
**Statement II :** Any object moving from one location to another location can follow infinite number of paths. Therefore, the amount of work done by the object changes with the path it follows for a conservative force.
In the light of the above statements, choose the correct answer from the options given below :
Q27.
mcq single
+4 / 1
The correct truth table for the given input data of the following logic gate is :


Q28.
mcq single
+4 / 1
Which of the following are true for a single slit diffraction?
A. Width of central maxima increases with increase in wavelength keeping slit width constant.
B. Width of central maxima increases with decrease in wavelength keeping slit width constant.
C. Width of central maxima increases with decrease in slit width at constant wavelength.
D. Width of central maxima increases with increase in slit width at constant wavelength.
E. Brightness of central maxima increases for decrease in wavelength at constant slit width.
Q29.
mcq single
+4 / 1
The wavelength of light, while it is passing through water is 540 nm . The refractive index of water is $4 / 3$. The wavelength of the same light when it is passing through a transparent medium having refractive index of $3 / 2$ is $\_\_\_\_$ nm.
Q30.
mcq single
+4 / 1
Using a simple pendulum experiment $g$ is determind by measuring its time period $T$. Which of the following plots represent the correct relation between the pendulum length $L$ and time period $T$ ?
Q31.
mcq single
+4 / 1
A uniform bar of length 12 cm and mass 20 m lies on a smooth horizontal table. Two point masses $m$ and $2 m$ are moving in opposite directions with same speed of $v$ and in the same plane as the bar, as shown in figure. These masses strike the bar simultaneously and get stuck to it. After collision the entire system is rotating with angular frequency $\omega$. The ratio of $v$ and $\omega$ is :


Q32.
numerical
+4 / 1
Two masses $m$ and 2 m are connected by a light string going over a pulley (disc) of mass 30 m with radius $r=0.1 \mathrm{~m}$. The pulley is mounted in a vertical plane and it is free to rotate about its axis. The 2 m mass is released from rest and its speed when it has descended through a height of 3.6 m is
$\_\_\_\_$ $\mathrm{m} / \mathrm{s}$. (Assume string does not slip and $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q33.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** For a mechanical system of many particles total kinetic energy is the sum of kinetic energies of all the particles.
**Statement II :** The total kinetic energy can be the sum of kinetic energy of the center of mass w.r.t to the origin and the kinetic energy of all the particles w.r.t. the center of mass as the reference.
In the light of the above statements, choose the correct answer from the options given below :
Q34.
numerical
+4 / 1
A conducting circular loop is rotated about its diameter at a constant angular speed of $100 \mathrm{rad} / \mathrm{s}$ in a magnetic field of 0.5 T perpendicular to the axis of rotation. When the loop is rotated by $30^{\circ}$ from the horizontal position, the induced EMF is 15.4 mV . The radius of the loop is $\_\_\_\_$ mm.
$$ \left(\text { Take } \pi=\frac{22}{7}\right) $$
Q35.
mcq single
+4 / 1
Figure shows the circuit that contains three resistances ( $9 \Omega$ each) and two inductors ( 4 mH each). The reading of ammeter at the moment switch $K$ is turned ON , is $\_\_\_\_$ A.


Q36.
numerical
+4 / 1
An insulated cylinder of volume $60 \mathrm{~cm}^3$ is filled with a gas at $27^{\circ} \mathrm{C}$ and 2 atmospheric pressure. Then the gas is compressed making the final volume as $20 \mathrm{~cm}^3$ while allowing the temperature to rise to $77^{\circ} \mathrm{C}$. The final pressure is $\_\_\_\_$ atmospheric pressure.
Q37.
mcq single
+4 / 1
Consider two boxes containing ideal gases $A$ and $B$ such that their temperatures, pressures and number densities are same. The molecular size of $A$ is half of that of $B$ and mass of molecule $A$ is four times that of $B$. If the collision frequency in gas $B$ is $32 \times 10^{18} / \mathrm{s}$ then collision frequency in gas $A$ is $\_\_\_\_$ /s.
Q38.
mcq single
+4 / 1
When a part of a straight capillary tube is placed vertically in a liquid, the liquid raises upto certain height $h$. If the inner radius of the capillary tube, density of the liquid and surface tension of the liquid decrease by $1 \%$ each, then the height of the liquid in the tube will change by $\_\_\_\_$ $\%$.
Q39.
mcq single
+4 / 1
Light is incident on a metallic plate having work function $110 \times 10^{-20} \mathrm{~J}$. If the produced photoelectrons have zero kinetic energy then the angular frequency of the incident light is $\_\_\_\_$ rad/s. $\left(\mathrm{h}=6.63 \times 10^{-34} \mathrm{~J} . \mathrm{s}\right)$.
Q40.
mcq single
+4 / 1
Five positive charges each having charge $q$ are placed at the vertices of a pentagon as shown in the figure. The electric potential $(V)$ and the electric field $(\vec{E})$ at the center $O$ of the pentagon due to these five positive charges are :


Q41.
mcq single
+4 / 1
Three small identical bubbles of water having same charge on each coalesce to form a bigger bubble. Then the ratio of the potentials on one initial bubble and that on the resultant bigger bubble is :
Q42.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** A satellite is moving around earth in the orbit very close to the earth surface. The time period of revolution of satellite depends upon the density of earth.
**Statement II :** The time period of revolution of the satellite is $T=2 \pi \sqrt{\frac{R_e}{g}}$ (for satellite very close to the earth surface), where $R_{\mathrm{e}}$ radius of earth and $g$ acceleration due to gravity. In the light of the above statements, choose the correct answer from the options given below :
Q43.
numerical
+4 / 1
A capacitor $P$ with capacitance $10 \times 10^{-6} \mathrm{~F}$ is fully charged with a potential difference of 6.0 V and disconnected from the battery. The charged capacitor $P$ is connected across another capacitor $Q$ with capacitance $20 \times 10^{-6} \mathrm{~F}$. The charge on capacitor $Q$ when equilibrium is established will be $\alpha \times 10^{-5} C$ (assume capacitor $Q$ does not have any charge initially), the value of $\alpha$ is $\_\_\_\_$ .
Q44.
mcq single
+4 / 1
In parallax method for the determination of focal length of a concave mirror, the object should always be placed:
Q45.
mcq single
+4 / 1
If $\epsilon, E$ and $t$ represent the free space permittivity, electric field and time respectively, then the unit of $\frac{\epsilon E}{t}$ will be :
Q46.
mcq single
+4 / 1
The smallest wavelength of Lyman series is 91 nm . The difference between the largest wavelengths of Paschen and Balmer series is nearly $\_\_\_\_$ nm.
Q47.
mcq single
+4 / 1
In an open organ pipe $\nu_3$ and $\nu_6$ are $3^{\text {rd }}$ and $6^{\text {th }}$ harmonic frequencies, respectively. If $\nu_6-\nu_3=2200 \mathrm{~Hz}$ then length of the pipe is $\_\_\_\_$ mm .
(Take velocity of sound in air is $330 \mathrm{~m} / \mathrm{s}$.)
Q48.
numerical
+4 / 1
A cylindrical conductor of length 2 m and area of cross-section $0.2 \mathrm{~mm}^2$ carries an electric current of 1.6 A when its ends are connected to a 2 V battery. Mobility of electrons in the conductor is $\alpha \times 10^{-3} \mathrm{~m}^2 / \mathrm{V} . \mathrm{s}$. The value of $\alpha$ is :
(electron concentration $=5 \times 10^{28} / \mathrm{m}^3$ and electron charge $=1.6 \times 10^{-19} \mathrm{C}$ )
Q49.
mcq single
+4 / 1
An electric power line having total resistance of $2 \Omega$, delivers 1 kW of power at 250 V . The percentage efficiency of transmission line is $\_\_\_\_$ .
Q50.
mcq single
+4 / 1
A laser beam has intensity of $4.0 \times 10^{14} \mathrm{~W} / \mathrm{m}^2$. The amplitude of magnetic field associated with beam is $\_\_\_\_$ T.
(Take $\epsilon_{\mathrm{o}}=8.85 \times 10^{-12} \mathrm{C}^2 / \mathrm{Nm}^2$ and $\mathrm{c}=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ )