JEE Main 2026 (Online) 22nd January Morning Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
'A' is a neutral organic compound (M. F : $\mathrm{C}_8 \mathrm{H}_9 \mathrm{ON}$ ). On treatment with aqueous $\mathrm{Br}_2 / \mathrm{HO}^{(-)}$, ' A ' forms a compound ' B ' which is soluble in dilute acid. ' B ' on treatment with aqueous $\mathrm{NaNO}_2 / \mathrm{HCl}\left(0-5^{\circ} \mathrm{C}\right)$ produces a compound ' C ' which on treatment with $\mathrm{CuCN} / \mathrm{NaCN}$ produces ' D '. Hydrolysis of ' D ' produces ' E ' which is also obtainable from the hydrolysis of ' A '. ' E ' on treatment with acidified $\mathrm{KMnO}_4$ produces ' F '. ' F ' contains two different types of hydrogen atoms. The structure of ' A ' is
Q2.
mcq single
+4 / 1
$\mathrm{A} \rightarrow$ product (First order reaction).
Three sets of experiment were performed for a reaction under similar experimental conditions:
Run $1 \Rightarrow 100 \mathrm{~mL}$ of 10 M solution of reactant A
Run $2 \Rightarrow 200 \mathrm{~mL}$ of 10 M solution of reactant A
Run $3 \Rightarrow 100 \mathrm{~mL}$ of 10 M solution of reactant $\mathrm{A}+100 \mathrm{~mL}$ of $\mathrm{H}_2 \mathrm{O}$ added.
The correct variation of rate of reaction is
Q3.
numerical
+4 / 1
The temperature at which the rate constants of the given below two gaseous reactions become equal is $\_\_\_\_$ K. (Nearest integer)
$$ \begin{array}{ll} \mathrm{X} \longrightarrow \mathrm{Y}, & \mathrm{k}_1=10^6 e^{\frac{-30000}{\mathrm{~T}}} \\ \mathrm{P} \longrightarrow \mathrm{Q}, & \mathrm{k}_2=10^4 e^{\frac{-24000}{\mathrm{~T}}} \end{array} $$
Given : $\ln 10=2.303$
Q4.
mcq single
+4 / 1
Two p-block elements X and Y form fluorides of the type $\mathrm{EF}_3$. The fluoride compound $\mathrm{XF}_3$ is a Lewis acid and $\mathrm{YF}_3$ is a Lewis base. The hybridizations of the central atoms of $\mathrm{XF}_3$ and $\mathrm{YF}_3$ respectively are
Q5.
mcq single
+4 / 1
The formal charges on the atoms marked as (1) to (4) in the Lewis representation of $\mathrm{HNO}_3$ molecule respectively are
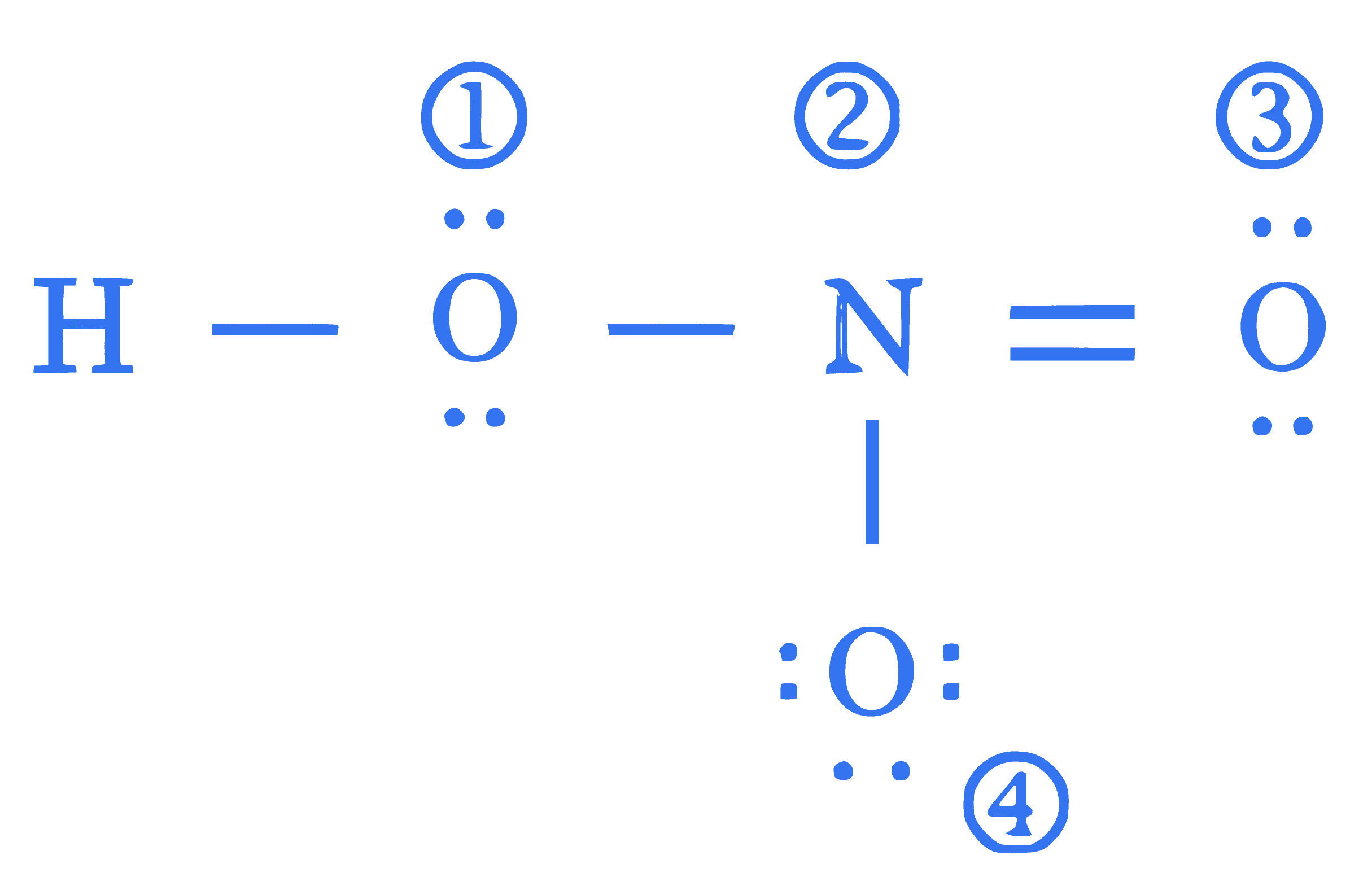
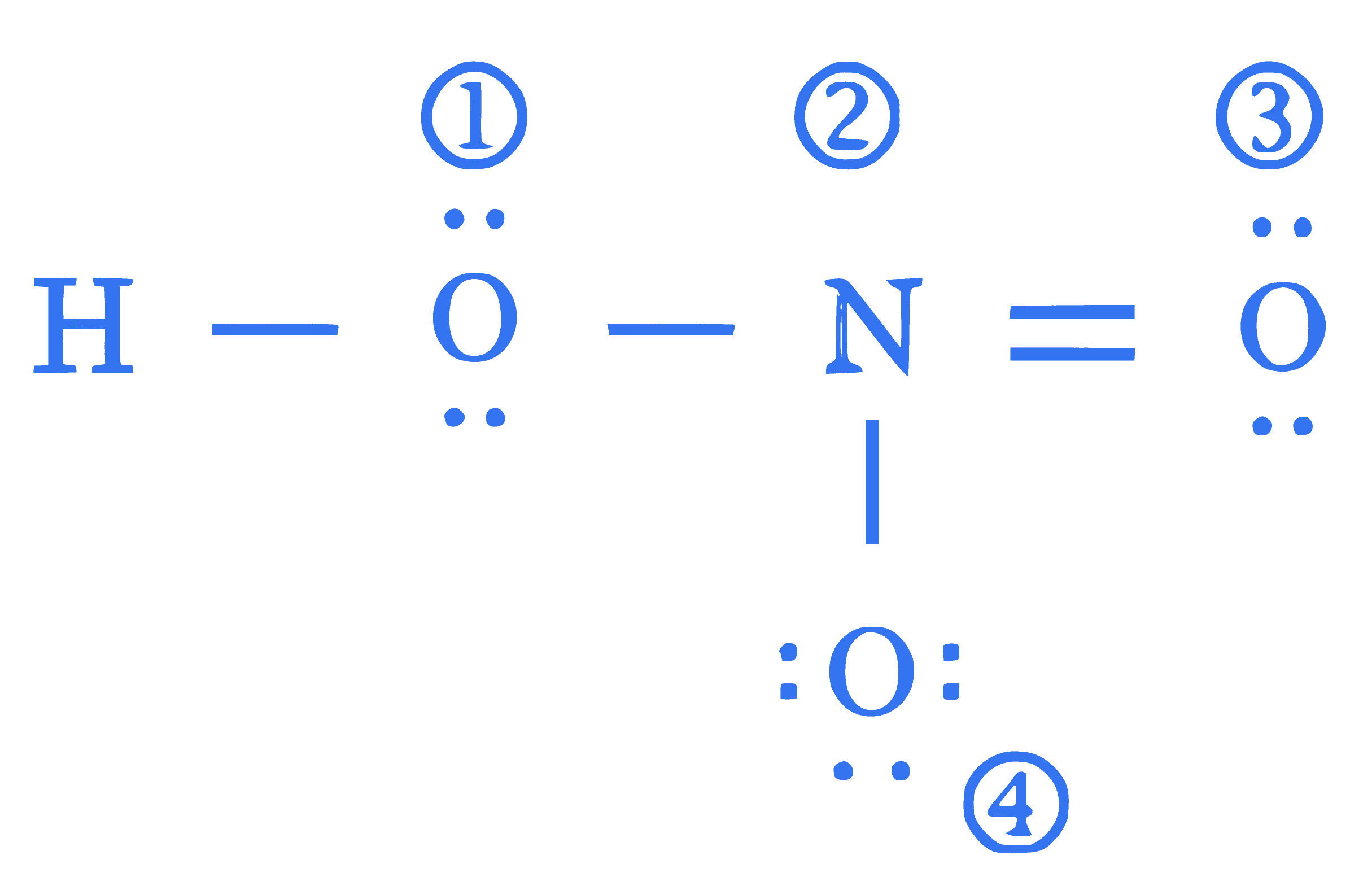
Q6.
mcq single
+4 / 1
The correct order of reactivity of $\mathrm{CH}_3 \mathrm{Br}$ in methanol with the following nucleophiles is
$\mathrm{F}^{-}, \mathrm{I}^{-}, \mathrm{C}_2 \mathrm{H}_5 \mathrm{O}^{-}$and $\mathrm{C}_6 \mathrm{H}_5 \mathrm{O}^{-}$
Q7.
mcq single
+4 / 1
The correct order of the rate of reaction of the following reactants with nucleophile by $\mathrm{S}_{\mathrm{N}} 1$ mechanism is :
(Given : Structures I and II are rigid)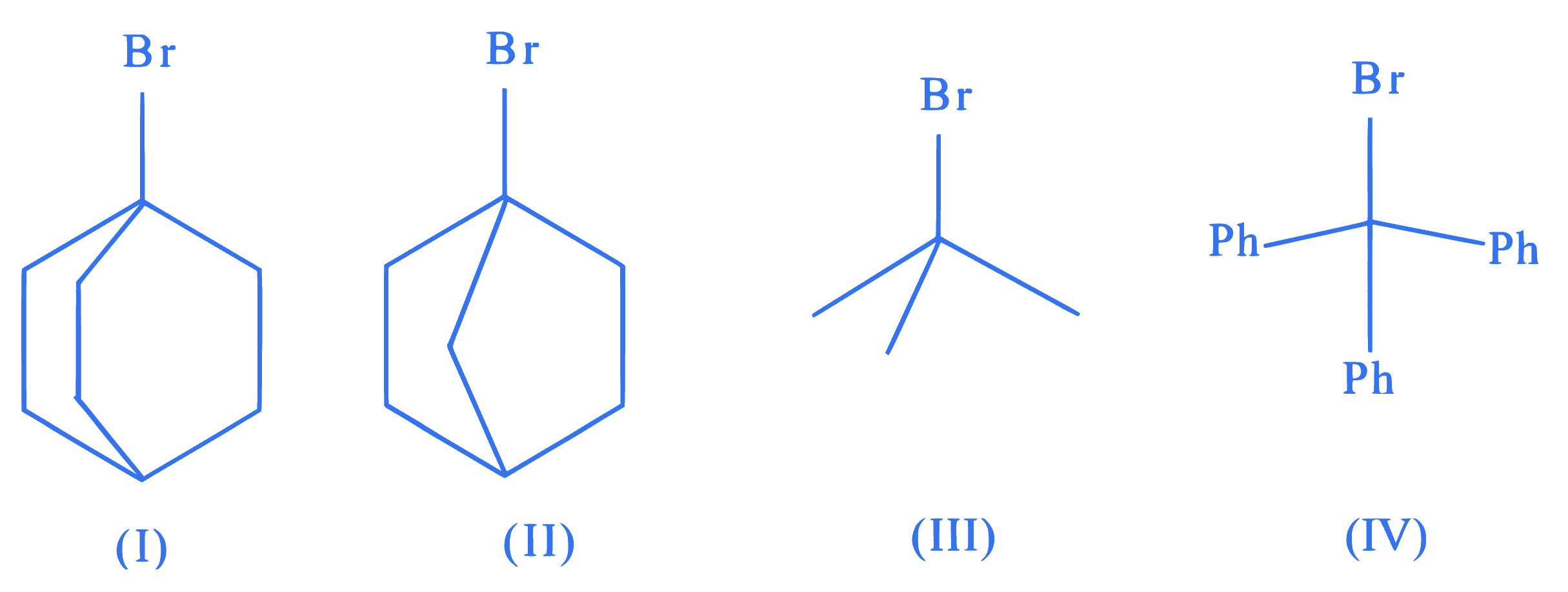

Q8.
mcq single
+4 / 1
As compared with chlorocyclohexane, which of the following statements correctly apply to chlorobenzene?
A. The magnitude of negative charge is more on chlorine atom.
B. The $\mathrm{C}-\mathrm{Cl}$ bond has partial double bond character.
C. $\mathrm{C}-\mathrm{Cl}$ bond is less polar.
D. $\mathrm{C}-\mathrm{Cl}$ bond is longer due to repulsion between delocalised electrons of the aromatic ring and lone pairs of electrons of chlorine.
E. The $\mathrm{C}-\mathrm{Cl}$ bond is formed using $\mathrm{sp}^2$ hybridised orbital of carbon.
Choose the correct answer from the options given below :
Q9.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The Henry's law constant $\mathrm{K}_{\mathrm{H}}$ is constant with respect to variations in solution's concentration over the range for which the solution is ideally dilute.
**Statement II :** $\mathrm{K}_{\mathrm{H}}$ does not differ for the same solute in different solvents.
In the light of the above statements, choose the correct answer from the options given below
Q10.
mcq single
+4 / 1
Consider a solution of $\mathrm{CO}_2(\mathrm{~g})$ dissolved in water in a closed container.
Which one of the following plots correctly represents variation of log (partial pressure of $\mathrm{CO}_2$ in vapour phase above water) $[y$-axis $]$ with $\log$ (mole fraction of $\mathrm{CO}_2$ in water) $[x$-axis $]$ at $25^{\circ} \mathrm{C}$ ?
Q11.
mcq single
+4 / 1
In the reaction,
$$ 2 \mathrm{Al}(\mathrm{~s})+6 \mathrm{HCl}(\mathrm{aq}) \rightarrow 2 \mathrm{Al}^{3+}(\mathrm{aq})+6 \mathrm{Cl}^{-}(\mathrm{aq})+3 \mathrm{H}_2(\mathrm{~g}) $$
Q12.
numerical
+4 / 1
Dissociation of a gas $\mathrm{A}_2$ takes place according to the following chemical reaction. At equilibrium, the total pressure is 1 bar at 300 K .
$$ \mathrm{A}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{~A}(\mathrm{~g}) $$
The standard Gibbs energy of formation of the involved substances has been provided below:
$$ \begin{array}{|c|c|} \hline \text { Substance } & \Delta \mathrm{G}_{\mathrm{f}}^{\circ} / \mathrm{kJ} \mathrm{~mol}^{-1} \\ \hline \hline \mathrm{~A}_2 & -100.00 \\ \hline \mathrm{~A} & -50.832 \\ \hline \end{array} $$
The degree of dissociation of $\mathrm{A}_2(\mathrm{~g})$ is given by $\left(x \times 10^{-2}\right)^{1 / 2}$ where $x=$
$\_\_\_\_$ . (Nearest integer).
[ **Given :** $\mathrm{R}=8 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}, \log 2=0.3010, \log 3=0.48$ ]
Assume degree of dissociation is not negligible.
Q13.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I Thermodynamic Process
List-II Magnitude in kJ
A.
Work done in reversible, isothermal expansion of 2 mol of ideal gas from
2
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
2
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
2dm^(3) to
20
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
20
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
20dm^(3) at 300 K .
I.
4
B.
Work done in irreversible isothermal expansion of 1 mol ideal gas from
1
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
1
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
1m^(3) to
3
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
3
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
3m^(3) at 300 K against a constant pressure of 3 kPa .
II.
11.5
C.
Change in internal energy for adiabatic expansion of a 1 mol ideal gas with change of temperature
=
320
<mtext> </mtext>
<mi mathvariant="normal">K</mi>
=
320
<mtext></mtext>
<mi mathvariant="normal">K</mi>
=320K and
<mover>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo accent="true">―</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
</mrow>
=
<mn>3</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
<mover accentunder="false">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo>¯</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
</mrow>
=
<mn>3</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
bar(C)_(V)=(3)/(2)R.
III.
6
D.
Change in enthalpy at constant pressure of 1 mol ideal gas with change of temperature
=
337
<mtext> </mtext>
<mi mathvariant="normal">K</mi>
=
337
<mtext></mtext>
<mi mathvariant="normal">K</mi>
=337K and
<mover>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo accent="true">―</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
</mrow>
=
<mn>5</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
<mover accentunder="false">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo>¯</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
</mrow>
=
<mn>5</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
bar(C)_(p)=(5)/(2)R.
IV.
7
Q14.
mcq single
+4 / 1
A 'p'-block element $(\mathrm{E})$ and hydrogen form a binary cation $\left(\mathrm{EH}_{\mathrm{x}}\right)^{+}$, while $\mathrm{EH}_3$ on treatment with $\mathrm{K}_2 \mathrm{HgI}_4$ in alkaline medium gives a precipitate of basic mercury(II)amido-iodine. Given below are first ionisation enthalpy values $\left(\mathrm{kJ} \mathrm{mol}^{-1}\right)$ for first element each from group 13, 14, 15 and 16. Identify the correct first ionisation enthalpy value for element E.
Q15.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The halogen that makes longest bond with hydrogen in HX, has the smallest covalent radius in its group.
**Statement II :** A group 15 element's hydride $\mathrm{EH}_3$ has the lowest boiling point among corresponding hydrides of other group 15 elements. The maximum covalency of that element E is 4 .
In the light of the above statements, choose the correct answer from the options given below
Q16.
mcq single
+4 / 1
Consider the transition metal ions $\mathrm{Mn}^{3+}, \mathrm{Cr}^{3+}, \mathrm{Fe}^{3+}$ and $\mathrm{Co}^{3+}$ and all form low spin octahedral complexes. The correct decreasing order of unpaired electrons in their respective d-orbitals of the complexes is
Q17.
mcq single
+4 / 1
A first row transition metal $(\mathrm{M})$ does not liberate $\mathrm{H}_2$ gas from dilute HCl .1 mol of aqueous solution of $\mathrm{MSO}_4$ is treated with excess of aqueous KCN and then $\mathrm{H}_2 \mathrm{~S}(\mathrm{~g})$ is passed through the solution. The amount of MS (metal sulphide) formed from the above reaction is $\_\_\_\_$ mol.
Q18.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I Reagents
List-II Name of Reaction involving carbonyl compounds
A.
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
−
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
,
<mi data-mjx-auto-op="false">KOH</mi>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
−
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
,
<mi data-mjx-auto-op="false">KOH</mi>
NH_(2)-NH_(2),KOH
I.
Tollen's Test
B.
<mi data-mjx-auto-op="false">Ag</mi>
<mrow data-mjx-texclass="INNER">
<mo data-mjx-texclass="OPEN">(</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>3</mn>
</msub>
<mo data-mjx-texclass="CLOSE">)</mo>
</mrow>
<mn>2</mn>
<mi data-mjx-auto-op="false">OH</mi>
<mi data-mjx-auto-op="false">Ag</mi>
<mfenced open="(" branch_open="(" close=")" branch_close=")" separators="|">
<mrow>
<msub>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>3</mn>
</msub>
</mrow>
</mfenced>
<mn>2</mn>
<mi data-mjx-auto-op="false">OH</mi>
Ag(NH_(3))_(2)OH
II.
Clemmensen Reduction
C.
Aq.
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">CuSO</mi>
</mrow>
<mn>4</mn>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">CuSO</mi>
</mrow>
<mn>4</mn>
CuSO_(4), Sodium Potassium tartarate, KOH
III.
Wolff - Kishner Reduction
D.
<mi data-mjx-auto-op="false">Zn</mi>
−
<mi data-mjx-auto-op="false">Hg</mi>
,
<mi data-mjx-auto-op="false">HCl</mi>
<mi data-mjx-auto-op="false">Zn</mi>
−
<mi data-mjx-auto-op="false">Hg</mi>
,
<mi data-mjx-auto-op="false">HCl</mi>
Zn-Hg,HCl
IV.
Fehling's Test
Choose the correct answer from the options given below :
Q19.
numerical
+4 / 1
Sodium fusion extract of an organic compound $(\mathrm{Y})$ with $\mathrm{CHCl}_3$ and chlorine water gives violet color to the $\mathrm{CHCl}_3$ layer. 0.15 g of $(\mathrm{Y})$ gave 0.12 g of the silver halide precipitate in Carius method. Percentage of halogen in the compound $(\mathrm{Y})$ is
$\_\_\_\_$ . (Nearest integer)
(Given : molar mass $\mathrm{g} \mathrm{mol}^{-1} \mathrm{C}: 12, \mathrm{H}: 1, \mathrm{Cl}: 35.5, \mathrm{Br}: 80, \mathrm{I}: 127$ )
Q20.
numerical
+4 / 1
The cycloalkene $(\mathrm{X})$ on bromination consumes one mole of bromine per mole of $(\mathrm{X})$ and gives the product $(\mathrm{Y})$ in which $\mathrm{C}: \mathrm{Br}$ ratio is 3:1. The percentage of bromine in the product $(\mathrm{Y})$ is $\_\_\_\_$ %. (Nearest integer)
(Given : molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Q21.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Benzene is nitrated to give nitrobenzene, which on further treatment $$
\text { with } \mathrm{CH}_3 \mathrm{COCl} / \mathrm{AlCl}_3 \text { will give }
$$ **
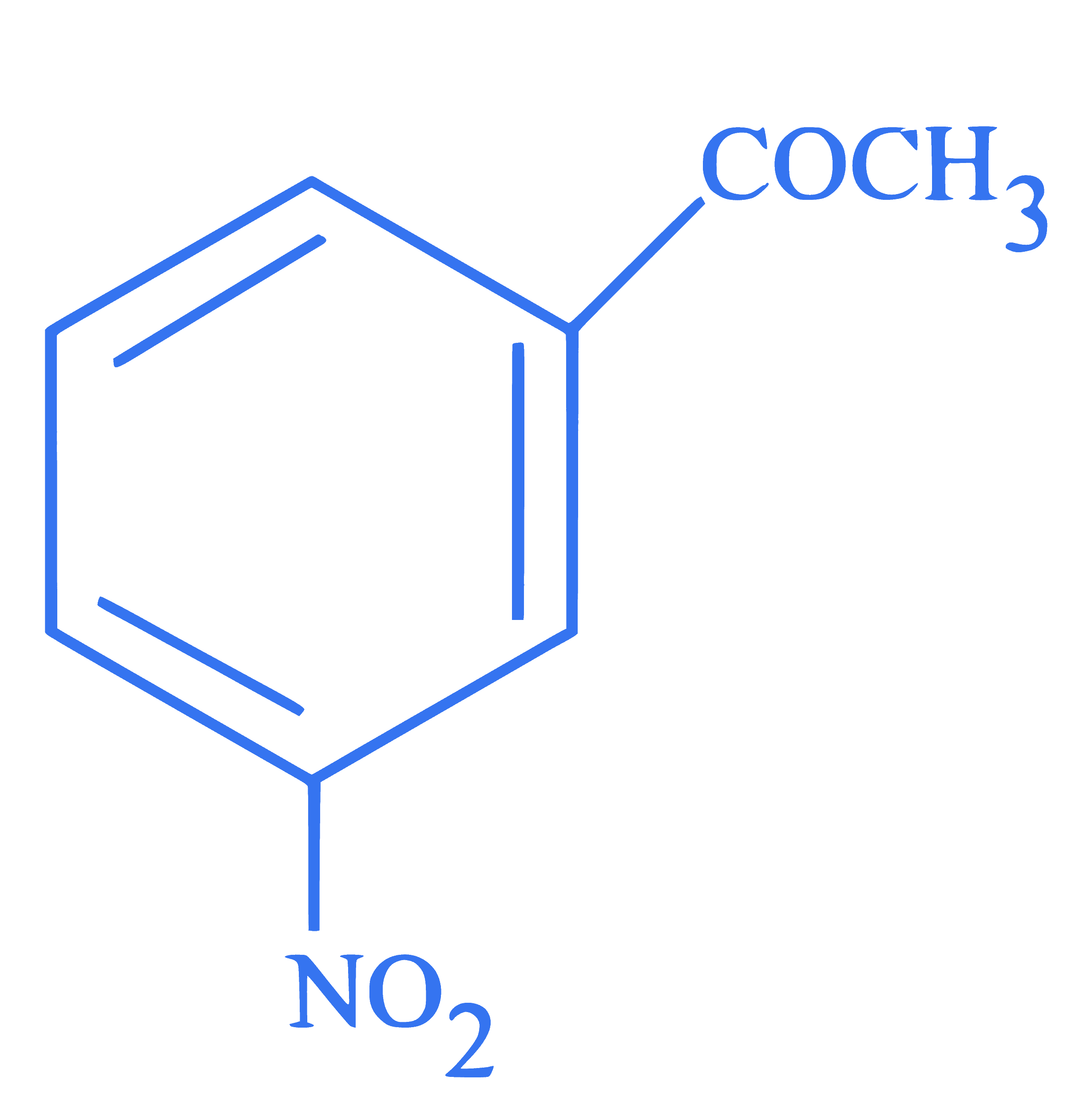 Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
 Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
Q22.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Sucrose is dextrorotatory. However, sucrose upon hydrolysis gives a solution having mixture of products. This solution shows laevorotation.
**Statement II :** Hydrolysis of sucrose gives glucose and fructose. Since the laevorotation of glucose is more than the dextrorotation of fructose, the resulting solution becomes laevorotatory.
In the light of the above statements, choose the correct answer from the options given below :
Q23.
numerical
+4 / 1
Consider the following electrochemical cell at 298 K
$\mathrm{Pt}\left|\mathrm{HSnO}_2^{-}(\mathrm{aq})\right| \mathrm{Sn}(\mathrm{OH})_6{ }^{2-}(\mathrm{aq})\left|\mathrm{OH}^{-}(\mathrm{aq})\right| \mathrm{Bi}_2 \mathrm{O}_3(\mathrm{~s}) \mid \mathrm{Bi}(\mathrm{s})$.
If the reaction quotient at a given time is $10^6$, then the cell EMF $\left(\mathrm{E}_{\text {cell }}\right)$ is
$\_\_\_\_$ $\times 10^{-1} \mathrm{~V}$ (Nearest integer).
Given the standard half-cell reduction potential as
$$ \mathrm{E}_{\mathrm{Bi}_2 \mathrm{O}_3 / \mathrm{Bi}, \mathrm{OH}^{-}}^{\circ}=-0.44 \mathrm{~V} \text { and } \mathrm{E}_{\mathrm{Sn}(\mathrm{OH})_6^{2-} / \mathrm{HSnO}_2^{-}, \mathrm{OH}^{-}}^{\circ}=-0.90 \mathrm{~V} $$
Q24.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Phenol on treatment with $\mathrm{CHCl}_3$ /aq. KOH under refluxing condition, followed by acidification produces $p$-hydroxy benzaldehyde as the major product and $o$-hydroxy benzaldehyde as the minor product.
**Statement II :** The mixture of $p$-hydroxybenzaldehyde and $o$ hydroxybenzaldehyde can be easily separated through steam distillation.
In the light of the above statements, choose the correct answer from the options given below
Q25.
mcq single
+4 / 1
The energy required by electrons, present in the first Bohr orbit of hydrogen atom to be excited to second Bohr orbit is $\_\_\_\_$ $\mathrm{J} \mathrm{mol}^{-1}$.
Given: $R_H=2.18 \times 10^{-11} \mathrm{ergs}$.
Q26.
mcq single
+4 / 1
'A' is a neutral organic compound (M. F : $\mathrm{C}_8 \mathrm{H}_9 \mathrm{ON}$ ). On treatment with aqueous $\mathrm{Br}_2 / \mathrm{HO}^{(-)}$, ' A ' forms a compound ' B ' which is soluble in dilute acid. ' B ' on treatment with aqueous $\mathrm{NaNO}_2 / \mathrm{HCl}\left(0-5^{\circ} \mathrm{C}\right)$ produces a compound ' C ' which on treatment with $\mathrm{CuCN} / \mathrm{NaCN}$ produces ' D '. Hydrolysis of ' D ' produces ' E ' which is also obtainable from the hydrolysis of ' A '. ' E ' on treatment with acidified $\mathrm{KMnO}_4$ produces ' F '. ' F ' contains two different types of hydrogen atoms. The structure of ' A ' is
Q27.
mcq single
+4 / 1
$\mathrm{A} \rightarrow$ product (First order reaction).
Three sets of experiment were performed for a reaction under similar experimental conditions:
Run $1 \Rightarrow 100 \mathrm{~mL}$ of 10 M solution of reactant A
Run $2 \Rightarrow 200 \mathrm{~mL}$ of 10 M solution of reactant A
Run $3 \Rightarrow 100 \mathrm{~mL}$ of 10 M solution of reactant $\mathrm{A}+100 \mathrm{~mL}$ of $\mathrm{H}_2 \mathrm{O}$ added.
The correct variation of rate of reaction is
Q28.
numerical
+4 / 1
The temperature at which the rate constants of the given below two gaseous reactions become equal is $\_\_\_\_$ K. (Nearest integer)
$$ \begin{array}{ll} \mathrm{X} \longrightarrow \mathrm{Y}, & \mathrm{k}_1=10^6 e^{\frac{-30000}{\mathrm{~T}}} \\ \mathrm{P} \longrightarrow \mathrm{Q}, & \mathrm{k}_2=10^4 e^{\frac{-24000}{\mathrm{~T}}} \end{array} $$
Given : $\ln 10=2.303$
Q29.
mcq single
+4 / 1
Two p-block elements X and Y form fluorides of the type $\mathrm{EF}_3$. The fluoride compound $\mathrm{XF}_3$ is a Lewis acid and $\mathrm{YF}_3$ is a Lewis base. The hybridizations of the central atoms of $\mathrm{XF}_3$ and $\mathrm{YF}_3$ respectively are
Q30.
mcq single
+4 / 1
The formal charges on the atoms marked as (1) to (4) in the Lewis representation of $\mathrm{HNO}_3$ molecule respectively are
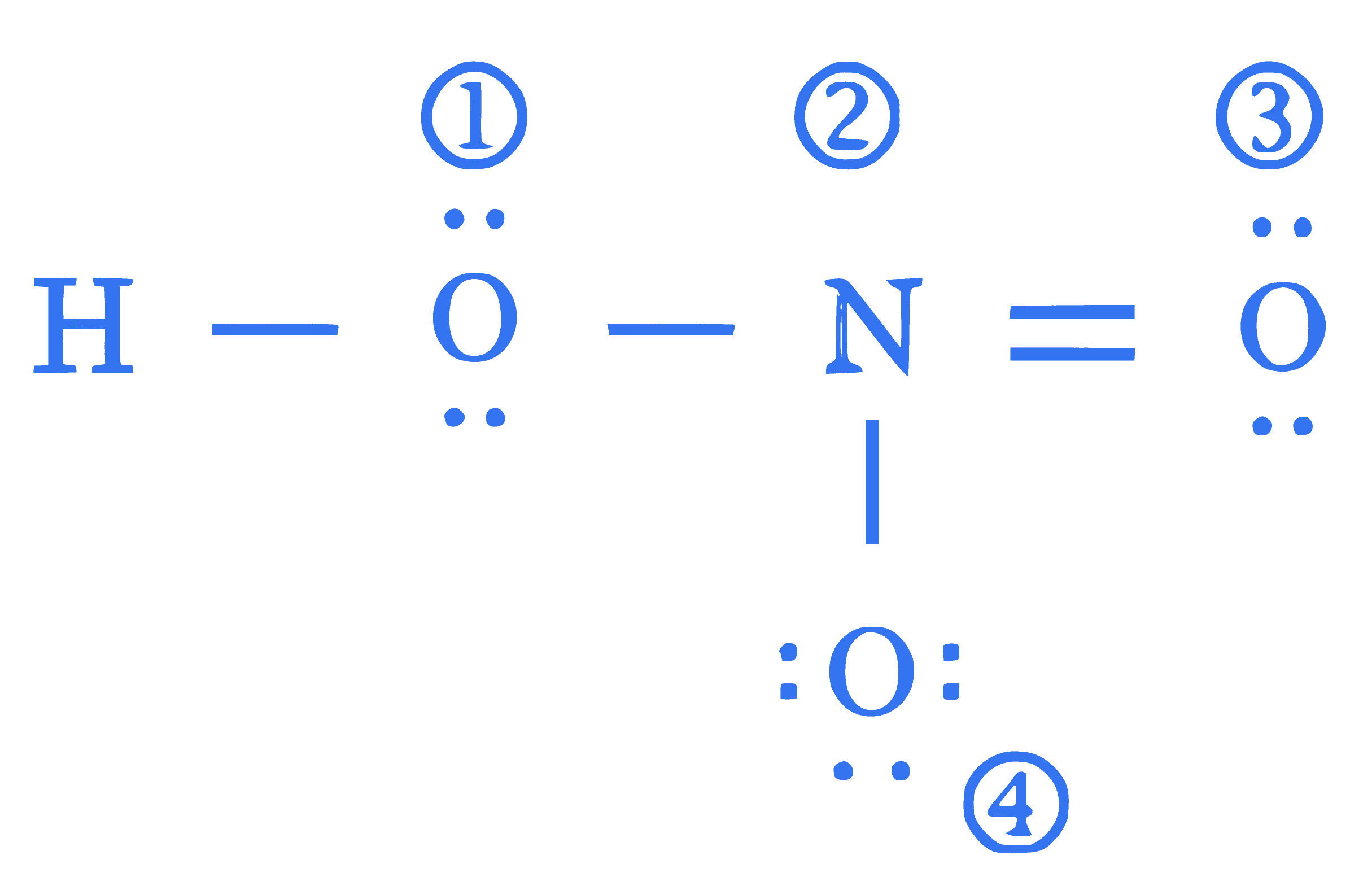
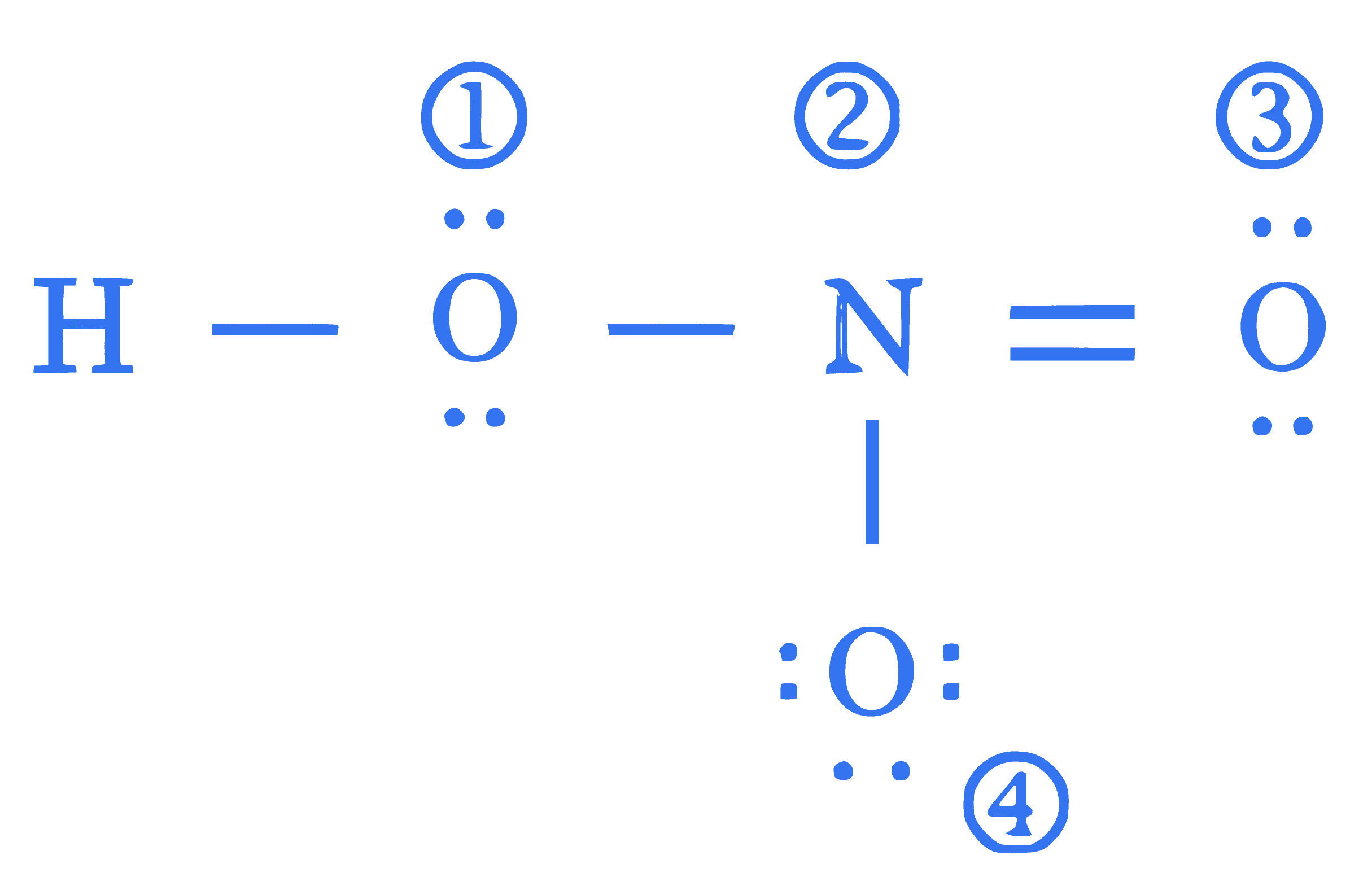
Q31.
mcq single
+4 / 1
The correct order of reactivity of $\mathrm{CH}_3 \mathrm{Br}$ in methanol with the following nucleophiles is
$\mathrm{F}^{-}, \mathrm{I}^{-}, \mathrm{C}_2 \mathrm{H}_5 \mathrm{O}^{-}$and $\mathrm{C}_6 \mathrm{H}_5 \mathrm{O}^{-}$
Q32.
mcq single
+4 / 1
The correct order of the rate of reaction of the following reactants with nucleophile by $\mathrm{S}_{\mathrm{N}} 1$ mechanism is :
(Given : Structures I and II are rigid)

Q33.
mcq single
+4 / 1
As compared with chlorocyclohexane, which of the following statements correctly apply to chlorobenzene?
A. The magnitude of negative charge is more on chlorine atom.
B. The $\mathrm{C}-\mathrm{Cl}$ bond has partial double bond character.
C. $\mathrm{C}-\mathrm{Cl}$ bond is less polar.
D. $\mathrm{C}-\mathrm{Cl}$ bond is longer due to repulsion between delocalised electrons of the aromatic ring and lone pairs of electrons of chlorine.
E. The $\mathrm{C}-\mathrm{Cl}$ bond is formed using $\mathrm{sp}^2$ hybridised orbital of carbon.
Choose the correct answer from the options given below :
Q34.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The Henry's law constant $\mathrm{K}_{\mathrm{H}}$ is constant with respect to variations in solution's concentration over the range for which the solution is ideally dilute.
**Statement II :** $\mathrm{K}_{\mathrm{H}}$ does not differ for the same solute in different solvents.
In the light of the above statements, choose the correct answer from the options given below
Q35.
mcq single
+4 / 1
Consider a solution of $\mathrm{CO}_2(\mathrm{~g})$ dissolved in water in a closed container.
Which one of the following plots correctly represents variation of log (partial pressure of $\mathrm{CO}_2$ in vapour phase above water) $[y$-axis $]$ with $\log$ (mole fraction of $\mathrm{CO}_2$ in water) $[x$-axis $]$ at $25^{\circ} \mathrm{C}$ ?
Q36.
mcq single
+4 / 1
In the reaction,
$$ 2 \mathrm{Al}(\mathrm{~s})+6 \mathrm{HCl}(\mathrm{aq}) \rightarrow 2 \mathrm{Al}^{3+}(\mathrm{aq})+6 \mathrm{Cl}^{-}(\mathrm{aq})+3 \mathrm{H}_2(\mathrm{~g}) $$
Q37.
numerical
+4 / 1
Dissociation of a gas $\mathrm{A}_2$ takes place according to the following chemical reaction. At equilibrium, the total pressure is 1 bar at 300 K .
$$ \mathrm{A}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{~A}(\mathrm{~g}) $$
The standard Gibbs energy of formation of the involved substances has been provided below:
$$ \begin{array}{|c|c|} \hline \text { Substance } & \Delta \mathrm{G}_{\mathrm{f}}^{\circ} / \mathrm{kJ} \mathrm{~mol}^{-1} \\ \hline \hline \mathrm{~A}_2 & -100.00 \\ \hline \mathrm{~A} & -50.832 \\ \hline \end{array} $$
The degree of dissociation of $\mathrm{A}_2(\mathrm{~g})$ is given by $\left(x \times 10^{-2}\right)^{1 / 2}$ where $x=$
$\_\_\_\_$ . (Nearest integer).
[ **Given :** $\mathrm{R}=8 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}, \log 2=0.3010, \log 3=0.48$ ]
Assume degree of dissociation is not negligible.
Q38.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I Thermodynamic Process
List-II Magnitude in kJ
A.
Work done in reversible, isothermal expansion of 2 mol of ideal gas from
2
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
2
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
2dm^(3) to
20
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
20
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">dm</mi>
</mrow>
<mn>3</mn>
20dm^(3) at 300 K .
I.
4
B.
Work done in irreversible isothermal expansion of 1 mol ideal gas from
1
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
1
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
1m^(3) to
3
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
3
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">m</mi>
</mrow>
<mn>3</mn>
3m^(3) at 300 K against a constant pressure of 3 kPa .
II.
11.5
C.
Change in internal energy for adiabatic expansion of a 1 mol ideal gas with change of temperature
=
320
<mtext> </mtext>
<mi mathvariant="normal">K</mi>
=
320
<mtext></mtext>
<mi mathvariant="normal">K</mi>
=320K and
<mover>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo accent="true">―</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
</mrow>
=
<mn>3</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
<mover accentunder="false">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo>¯</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
</mrow>
=
<mn>3</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
bar(C)_(V)=(3)/(2)R.
III.
6
D.
Change in enthalpy at constant pressure of 1 mol ideal gas with change of temperature
=
337
<mtext> </mtext>
<mi mathvariant="normal">K</mi>
=
337
<mtext></mtext>
<mi mathvariant="normal">K</mi>
=337K and
<mover>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo accent="true">―</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
</mrow>
=
<mn>5</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
<mover accentunder="false">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">C</mi>
</mrow>
<mo>¯</mo>
</mover>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
</mrow>
=
<mn>5</mn>
<mn>2</mn>
<mi mathvariant="normal">R</mi>
bar(C)_(p)=(5)/(2)R.
IV.
7
Q39.
mcq single
+4 / 1
A 'p'-block element $(\mathrm{E})$ and hydrogen form a binary cation $\left(\mathrm{EH}_{\mathrm{x}}\right)^{+}$, while $\mathrm{EH}_3$ on treatment with $\mathrm{K}_2 \mathrm{HgI}_4$ in alkaline medium gives a precipitate of basic mercury(II)amido-iodine. Given below are first ionisation enthalpy values $\left(\mathrm{kJ} \mathrm{mol}^{-1}\right)$ for first element each from group 13, 14, 15 and 16. Identify the correct first ionisation enthalpy value for element E.
Q40.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The halogen that makes longest bond with hydrogen in HX, has the smallest covalent radius in its group.
**Statement II :** A group 15 element's hydride $\mathrm{EH}_3$ has the lowest boiling point among corresponding hydrides of other group 15 elements. The maximum covalency of that element E is 4 .
In the light of the above statements, choose the correct answer from the options given below
Q41.
mcq single
+4 / 1
Consider the transition metal ions $\mathrm{Mn}^{3+}, \mathrm{Cr}^{3+}, \mathrm{Fe}^{3+}$ and $\mathrm{Co}^{3+}$ and all form low spin octahedral complexes. The correct decreasing order of unpaired electrons in their respective d-orbitals of the complexes is
Q42.
mcq single
+4 / 1
A first row transition metal $(\mathrm{M})$ does not liberate $\mathrm{H}_2$ gas from dilute HCl .1 mol of aqueous solution of $\mathrm{MSO}_4$ is treated with excess of aqueous KCN and then $\mathrm{H}_2 \mathrm{~S}(\mathrm{~g})$ is passed through the solution. The amount of MS (metal sulphide) formed from the above reaction is $\_\_\_\_$ mol.
Q43.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I Reagents
List-II Name of Reaction involving carbonyl compounds
A.
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
−
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
,
<mi data-mjx-auto-op="false">KOH</mi>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
−
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>2</mn>
,
<mi data-mjx-auto-op="false">KOH</mi>
NH_(2)-NH_(2),KOH
I.
Tollen's Test
B.
<mi data-mjx-auto-op="false">Ag</mi>
<mrow data-mjx-texclass="INNER">
<mo data-mjx-texclass="OPEN">(</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>3</mn>
</msub>
<mo data-mjx-texclass="CLOSE">)</mo>
</mrow>
<mn>2</mn>
<mi data-mjx-auto-op="false">OH</mi>
<mi data-mjx-auto-op="false">Ag</mi>
<mfenced open="(" branch_open="(" close=")" branch_close=")" separators="|">
<mrow>
<msub>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">NH</mi>
</mrow>
<mn>3</mn>
</msub>
</mrow>
</mfenced>
<mn>2</mn>
<mi data-mjx-auto-op="false">OH</mi>
Ag(NH_(3))_(2)OH
II.
Clemmensen Reduction
C.
Aq.
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">CuSO</mi>
</mrow>
<mn>4</mn>
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">CuSO</mi>
</mrow>
<mn>4</mn>
CuSO_(4), Sodium Potassium tartarate, KOH
III.
Wolff - Kishner Reduction
D.
<mi data-mjx-auto-op="false">Zn</mi>
−
<mi data-mjx-auto-op="false">Hg</mi>
,
<mi data-mjx-auto-op="false">HCl</mi>
<mi data-mjx-auto-op="false">Zn</mi>
−
<mi data-mjx-auto-op="false">Hg</mi>
,
<mi data-mjx-auto-op="false">HCl</mi>
Zn-Hg,HCl
IV.
Fehling's Test
Choose the correct answer from the options given below :
Q44.
numerical
+4 / 1
Sodium fusion extract of an organic compound $(\mathrm{Y})$ with $\mathrm{CHCl}_3$ and chlorine water gives violet color to the $\mathrm{CHCl}_3$ layer. 0.15 g of $(\mathrm{Y})$ gave 0.12 g of the silver halide precipitate in Carius method. Percentage of halogen in the compound $(\mathrm{Y})$ is
$\_\_\_\_$ . (Nearest integer)
(Given : molar mass $\mathrm{g} \mathrm{mol}^{-1} \mathrm{C}: 12, \mathrm{H}: 1, \mathrm{Cl}: 35.5, \mathrm{Br}: 80, \mathrm{I}: 127$ )
Q45.
numerical
+4 / 1
The cycloalkene $(\mathrm{X})$ on bromination consumes one mole of bromine per mole of $(\mathrm{X})$ and gives the product $(\mathrm{Y})$ in which $\mathrm{C}: \mathrm{Br}$ ratio is 3:1. The percentage of bromine in the product $(\mathrm{Y})$ is $\_\_\_\_$ %. (Nearest integer)
(Given : molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Q46.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Benzene is nitrated to give nitrobenzene, which on further treatment $$
\text { with } \mathrm{CH}_3 \mathrm{COCl} / \mathrm{AlCl}_3 \text { will give }
$$ **
 Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
 Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
Statement II :** $-\mathrm{NO}_2$ group is a $m$-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
Q47.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Sucrose is dextrorotatory. However, sucrose upon hydrolysis gives a solution having mixture of products. This solution shows laevorotation.
**Statement II :** Hydrolysis of sucrose gives glucose and fructose. Since the laevorotation of glucose is more than the dextrorotation of fructose, the resulting solution becomes laevorotatory.
In the light of the above statements, choose the correct answer from the options given below :
Q48.
numerical
+4 / 1
Consider the following electrochemical cell at 298 K
$\mathrm{Pt}\left|\mathrm{HSnO}_2^{-}(\mathrm{aq})\right| \mathrm{Sn}(\mathrm{OH})_6{ }^{2-}(\mathrm{aq})\left|\mathrm{OH}^{-}(\mathrm{aq})\right| \mathrm{Bi}_2 \mathrm{O}_3(\mathrm{~s}) \mid \mathrm{Bi}(\mathrm{s})$.
If the reaction quotient at a given time is $10^6$, then the cell EMF $\left(\mathrm{E}_{\text {cell }}\right)$ is
$\_\_\_\_$ $\times 10^{-1} \mathrm{~V}$ (Nearest integer).
Given the standard half-cell reduction potential as
$$ \mathrm{E}_{\mathrm{Bi}_2 \mathrm{O}_3 / \mathrm{Bi}, \mathrm{OH}^{-}}^{\circ}=-0.44 \mathrm{~V} \text { and } \mathrm{E}_{\mathrm{Sn}(\mathrm{OH})_6^{2-} / \mathrm{HSnO}_2^{-}, \mathrm{OH}^{-}}^{\circ}=-0.90 \mathrm{~V} $$
Q49.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Phenol on treatment with $\mathrm{CHCl}_3$ /aq. KOH under refluxing condition, followed by acidification produces $p$-hydroxy benzaldehyde as the major product and $o$-hydroxy benzaldehyde as the minor product.
**Statement II :** The mixture of $p$-hydroxybenzaldehyde and $o$ hydroxybenzaldehyde can be easily separated through steam distillation.
In the light of the above statements, choose the correct answer from the options given below
Q50.
mcq single
+4 / 1
The energy required by electrons, present in the first Bohr orbit of hydrogen atom to be excited to second Bohr orbit is $\_\_\_\_$ $\mathrm{J} \mathrm{mol}^{-1}$.
Given: $R_H=2.18 \times 10^{-11} \mathrm{ergs}$.
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{AB}}=2 \hat{i}+4 \hat{j}-5 \hat{k}$ and $\overrightarrow{\mathrm{AD}}=\hat{i}+2 \hat{j}+\lambda \hat{k}, \lambda \in \mathbb{R}$. Let the projection of the vector $\vec{v}=\hat{i}+\hat{j}+\hat{k}$ on the diagonal $\overrightarrow{\mathrm{AC}}$ of the parallelogram ABCD be of length one unit. If $\alpha, \beta$, where $\alpha>\beta$, be the roots of the equation $\lambda^2 x^2-6 \lambda x+5=0$, then $2 \alpha-\beta$ is equal to
Q2.
mcq single
+4 / 1
The number of distinct real solutions of the equation $x|x+4|+3|x+2|+10=0$ is
Q3.
mcq single
+4 / 1
Two distinct numbers $a$ and $b$ are selected at random from $1,2,3, \ldots, 50$. The probability, that their product $a b$ is divisible by 3 , is
Q4.
mcq single
+4 / 1
If a random variable $x$ has the probability distribution
$$ \begin{array}{|c|c|c|c|c|c|c|c|c|} \hline x & 0 & 1 & 2 & 3 & 4 & 5 & 6 & 7 \\ \hline \mathrm{P}(x) & 0 & 2 \mathrm{k} & \mathrm{k} & 3 \mathrm{k} & 2 \mathrm{k}^2 & 2 \mathrm{k} & \mathrm{k}^2+\mathrm{k} & 7 \mathrm{k}^2 \\ \hline \end{array} $$
$$ \text { then } \mathrm{P}(3 < x \leq 6) \text { is equal to } $$
Q5.
mcq single
+4 / 1
If the image of the point $\mathrm{P}(1,2, a)$ in the line $\frac{x-6}{3}=\frac{y-7}{2}=\frac{7-\mathrm{z}}{2}$ is $\mathrm{Q}(5, b, \mathrm{c})$, then $a^2+b^2+c^2$ is equal to
Q6.
mcq single
+4 / 1
Let $\mathrm{P}(\alpha, \beta, \gamma)$ be the point on the line $\frac{x-1}{2}=\frac{y+1}{-3}=z$ at a distance $4 \sqrt{14}$ from the point $(1,-1,0)$ and nearer to the origin. Then the shortest distance, between the lines $\frac{x-\alpha}{1}=\frac{y-\beta}{2}=\frac{z-\gamma}{3}$ and $\frac{x+5}{2}=\frac{y-10}{1}=\frac{z-3}{1}$, is equal to
Q7.
mcq single
+4 / 1
If the sum of the first four terms of an A.P. is 6 and the sum of its first six terms is 4 , then the sum of its first twelve terms is
Q8.
mcq single
+4 / 1
Let $f(x)=x^{2025}-x^{2000}, x \in[0,1]$ and the minimum value of the function $f(x)$ in the interval $[0,1]$ be $(80)^{80}(n)^{-81}$. Then $n$ is equal to
Q9.
numerical
+4 / 1
$$ \text { If } \frac{\cos ^2 48^{\circ}-\sin ^2 12^{\circ}}{\sin ^2 24^{\circ}-\sin ^2 6^{\circ}}=\frac{\alpha+\beta \sqrt{5}}{2} \text {, where } \alpha, \beta \in \mathbb{N} \text {, then } \alpha+\beta \text { is equal to ___________} $$
Q10.
mcq single
+4 / 1
Let the line $x=-1$ divide the area of the region $\left\{(x, y): 1+x^2 \leq y \leq 3-x\right\}$ in the ratio $m: n, \operatorname{gcd}(m, n)=1$. Then $m+n$ is equal to
Q11.
numerical
+4 / 1
Let A be a $3 \times 3$ matrix such that $\mathrm{A}+\mathrm{A}^{\mathrm{T}}=\mathrm{O}$. If $\mathrm{A}\left[\begin{array}{c}1 \\ -1 \\ 0\end{array}\right]=\left[\begin{array}{l}3 \\ 3 \\ 2\end{array}\right], \mathrm{A}^2\left[\begin{array}{c}1 \\ -1 \\ 0\end{array}\right]=\left[\begin{array}{c}-3 \\ 19 \\ -24\end{array}\right]$ and $\operatorname{det}(\operatorname{adj}(2 \operatorname{adj}(\mathrm{~A}+\mathrm{I})))=(2)^\alpha \cdot(3)^\beta \cdot(11)^\gamma, \alpha, \beta, \gamma$ are non-negative integers, then $\alpha+\beta+\gamma$ is equal to $\_\_\_\_$
Q12.
mcq single
+4 / 1
If $\mathrm{A}=\left[\begin{array}{ll}2 & 3 \\ 3 & 5\end{array}\right]$, then the determinant of the matrix $\left(\mathrm{A}^{2025}-3 \mathrm{~A}^{2024}+\mathrm{A}^{2023}\right)$ is
Q13.
numerical
+4 / 1
If $\int(\sin x)^{\frac{-11}{2}}(\cos x)^{\frac{-5}{2}} d x= -\frac{p_1}{q_1}(\cot x)^{\frac{9}{2}}-\frac{p_2}{q_2}(\cot x)^{\frac{5}{2}}-\frac{p_3}{q_3}(\cot x)^{\frac{1}{2}}+\frac{p_4}{q_4}(\cot x)^{\frac{-3}{2}}+\mathrm{C}$, where $p_i$ and $q_i$ are positive integers with $\operatorname{gcd}\left(p_i, q_i\right)=1$ for $i=1,2,3,4$ and C is the constant of integration, then $\frac{15 p_1 p_2 p_3 p_4}{q_1 q_2 q_3 q_4}$ is equal to $\_\_\_\_$
Q14.
mcq single
+4 / 1
If the chord joining the points $\mathrm{P}_1\left(x_1, y_1\right)$ and $\mathrm{P}_2\left(x_2, y_2\right)$ on the parabola $y^2=12 x$ subtends a right angle at the vertex of the parabola, then $x_1 x_2-y_1 y_2$ is equal to
Q15.
mcq single
+4 / 1
The number of solutions of $\tan ^{-1} 4 x+\tan ^{-1} 6 x=\frac{\pi}{6}$, where $-\frac{1}{2 \sqrt{6}} < x < \frac{1}{2 \sqrt{6}}$, is equal to :
Q16.
mcq single
+4 / 1
The coefficient of $x^{48}$ in $(1+x)+2(1+x)^2+3(1+x)^3+\ldots+100(1+x)^{100}$ is equal to
Q17.
mcq single
+4 / 1
Let the relation R on the set $\mathrm{M}=\{1,2,3, \ldots, 16\}$ be given by $\mathrm{R}=\{(x, y): 4 y=5 x-3, x, y \in \mathrm{M}\}$.
Then the minimum number of elements required to be added in R , in order to make the relation symmetric, is equal to
Q18.
numerical
+4 / 1
Let $\alpha=\frac{-1+i \sqrt{3}}{2}$ and $\beta=\frac{-1-i \sqrt{3}}{2}, i=\sqrt{-1}$. If
$$ (7-7 \alpha+9 \beta)^{20}+(9+7 \alpha-7 \beta)^{20}+(-7+9 \alpha+7 \beta)^{20}+(14+7 \alpha+7 \beta)^{20}=m^{10}, $$
then $m$ is $\_\_\_\_$
Q19.
mcq single
+4 / 1
If the line $\alpha x+2 y=1$, where $\alpha \in \mathbb{R}$, does not meet the hyperbola $x^2-9 y^2=9$, then a possible value of $\alpha$ is :
Q20.
mcq single
+4 / 1
If the domain of the function $f(x)=\sin ^{-1}\left(\frac{5-x}{3+2 x}\right)+\frac{1}{\log _e(10-x)}$ is $(-\infty, \alpha] \cup[\beta, \gamma)-\{\delta\}$, then $6(\alpha+\beta+\gamma+\delta)$ is equal to
Q21.
mcq single
+4 / 1
Let the solution curve of the differential equation $x d y-y d x=\sqrt{x^2+y^2} d x, x>0$, $y(1)=0$, be $y=y(x)$. Then $y(3)$ is equal to
Q22.
mcq single
+4 / 1
Let the set of all values of $r$, for which the circles $(x+1)^2+(y+4)^2=r^2$ and $x^2+y^2-4 x-2 y-4=0$ intersect at two distinct points be the interval $(\alpha, \beta)$. Then $\alpha \beta$ is equal to
Q23.
numerical
+4 / 1
Let ABC be a triangle. Consider four points $\mathrm{p}_1, \mathrm{p}_2, \mathrm{p}_3, \mathrm{p}_4$ on the side AB , five points $p_5, p_6, p_7, p_8, p_9$ on the side $B C$, and four points $p_{10}, p_{11}, p_{12}, p_{13}$ on the side AC . None of these points is a vertex of the triangle ABC . Then the total number of pentagons, that can be formed by taking all the vertices from the points $p_1, p_2, \ldots, p_{13}$, is $\_\_\_\_$
Q24.
mcq single
+4 / 1
Let $f:[1, \infty) \rightarrow \mathbb{R}$ be a differentiable function. If $6 \int\limits_1^x f(t) d t=3 x f(x)+x^3-4$ for all $x \geq 1$, then the value of $f(2)-f(3)$ is :
Q25.
mcq single
+4 / 1
The value of $\int\limits_{-\frac{\pi}{2}}^{\frac{\pi}{2}}\left(\frac{1}{[x]+4}\right) d x$, where $[\cdot]$ denotes the greatest integer function, is
Q26.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{AB}}=2 \hat{i}+4 \hat{j}-5 \hat{k}$ and $\overrightarrow{\mathrm{AD}}=\hat{i}+2 \hat{j}+\lambda \hat{k}, \lambda \in \mathbb{R}$. Let the projection of the vector $\vec{v}=\hat{i}+\hat{j}+\hat{k}$ on the diagonal $\overrightarrow{\mathrm{AC}}$ of the parallelogram ABCD be of length one unit. If $\alpha, \beta$, where $\alpha>\beta$, be the roots of the equation $\lambda^2 x^2-6 \lambda x+5=0$, then $2 \alpha-\beta$ is equal to
Q27.
mcq single
+4 / 1
The number of distinct real solutions of the equation $x|x+4|+3|x+2|+10=0$ is
Q28.
mcq single
+4 / 1
Two distinct numbers $a$ and $b$ are selected at random from $1,2,3, \ldots, 50$. The probability, that their product $a b$ is divisible by 3 , is
Q29.
mcq single
+4 / 1
If a random variable $x$ has the probability distribution
$$ \begin{array}{|c|c|c|c|c|c|c|c|c|} \hline x & 0 & 1 & 2 & 3 & 4 & 5 & 6 & 7 \\ \hline \mathrm{P}(x) & 0 & 2 \mathrm{k} & \mathrm{k} & 3 \mathrm{k} & 2 \mathrm{k}^2 & 2 \mathrm{k} & \mathrm{k}^2+\mathrm{k} & 7 \mathrm{k}^2 \\ \hline \end{array} $$
$$ \text { then } \mathrm{P}(3 < x \leq 6) \text { is equal to } $$
Q30.
mcq single
+4 / 1
If the image of the point $\mathrm{P}(1,2, a)$ in the line $\frac{x-6}{3}=\frac{y-7}{2}=\frac{7-\mathrm{z}}{2}$ is $\mathrm{Q}(5, b, \mathrm{c})$, then $a^2+b^2+c^2$ is equal to
Q31.
mcq single
+4 / 1
Let $\mathrm{P}(\alpha, \beta, \gamma)$ be the point on the line $\frac{x-1}{2}=\frac{y+1}{-3}=z$ at a distance $4 \sqrt{14}$ from the point $(1,-1,0)$ and nearer to the origin. Then the shortest distance, between the lines $\frac{x-\alpha}{1}=\frac{y-\beta}{2}=\frac{z-\gamma}{3}$ and $\frac{x+5}{2}=\frac{y-10}{1}=\frac{z-3}{1}$, is equal to
Q32.
mcq single
+4 / 1
If the sum of the first four terms of an A.P. is 6 and the sum of its first six terms is 4 , then the sum of its first twelve terms is
Q33.
mcq single
+4 / 1
Let $f(x)=x^{2025}-x^{2000}, x \in[0,1]$ and the minimum value of the function $f(x)$ in the interval $[0,1]$ be $(80)^{80}(n)^{-81}$. Then $n$ is equal to
Q34.
numerical
+4 / 1
$$ \text { If } \frac{\cos ^2 48^{\circ}-\sin ^2 12^{\circ}}{\sin ^2 24^{\circ}-\sin ^2 6^{\circ}}=\frac{\alpha+\beta \sqrt{5}}{2} \text {, where } \alpha, \beta \in \mathbb{N} \text {, then } \alpha+\beta \text { is equal to ___________} $$
Q35.
mcq single
+4 / 1
Let the line $x=-1$ divide the area of the region $\left\{(x, y): 1+x^2 \leq y \leq 3-x\right\}$ in the ratio $m: n, \operatorname{gcd}(m, n)=1$. Then $m+n$ is equal to
Q36.
numerical
+4 / 1
Let A be a $3 \times 3$ matrix such that $\mathrm{A}+\mathrm{A}^{\mathrm{T}}=\mathrm{O}$. If $\mathrm{A}\left[\begin{array}{c}1 \\ -1 \\ 0\end{array}\right]=\left[\begin{array}{l}3 \\ 3 \\ 2\end{array}\right], \mathrm{A}^2\left[\begin{array}{c}1 \\ -1 \\ 0\end{array}\right]=\left[\begin{array}{c}-3 \\ 19 \\ -24\end{array}\right]$ and $\operatorname{det}(\operatorname{adj}(2 \operatorname{adj}(\mathrm{~A}+\mathrm{I})))=(2)^\alpha \cdot(3)^\beta \cdot(11)^\gamma, \alpha, \beta, \gamma$ are non-negative integers, then $\alpha+\beta+\gamma$ is equal to $\_\_\_\_$
Q37.
mcq single
+4 / 1
If $\mathrm{A}=\left[\begin{array}{ll}2 & 3 \\ 3 & 5\end{array}\right]$, then the determinant of the matrix $\left(\mathrm{A}^{2025}-3 \mathrm{~A}^{2024}+\mathrm{A}^{2023}\right)$ is
Q38.
numerical
+4 / 1
If $\int(\sin x)^{\frac{-11}{2}}(\cos x)^{\frac{-5}{2}} d x= -\frac{p_1}{q_1}(\cot x)^{\frac{9}{2}}-\frac{p_2}{q_2}(\cot x)^{\frac{5}{2}}-\frac{p_3}{q_3}(\cot x)^{\frac{1}{2}}+\frac{p_4}{q_4}(\cot x)^{\frac{-3}{2}}+\mathrm{C}$, where $p_i$ and $q_i$ are positive integers with $\operatorname{gcd}\left(p_i, q_i\right)=1$ for $i=1,2,3,4$ and C is the constant of integration, then $\frac{15 p_1 p_2 p_3 p_4}{q_1 q_2 q_3 q_4}$ is equal to $\_\_\_\_$
Q39.
mcq single
+4 / 1
If the chord joining the points $\mathrm{P}_1\left(x_1, y_1\right)$ and $\mathrm{P}_2\left(x_2, y_2\right)$ on the parabola $y^2=12 x$ subtends a right angle at the vertex of the parabola, then $x_1 x_2-y_1 y_2$ is equal to
Q40.
mcq single
+4 / 1
The number of solutions of $\tan ^{-1} 4 x+\tan ^{-1} 6 x=\frac{\pi}{6}$, where $-\frac{1}{2 \sqrt{6}} < x < \frac{1}{2 \sqrt{6}}$, is equal to :
Q41.
mcq single
+4 / 1
The coefficient of $x^{48}$ in $(1+x)+2(1+x)^2+3(1+x)^3+\ldots+100(1+x)^{100}$ is equal to
Q42.
mcq single
+4 / 1
Let the relation R on the set $\mathrm{M}=\{1,2,3, \ldots, 16\}$ be given by $\mathrm{R}=\{(x, y): 4 y=5 x-3, x, y \in \mathrm{M}\}$.
Then the minimum number of elements required to be added in R , in order to make the relation symmetric, is equal to
Q43.
numerical
+4 / 1
Let $\alpha=\frac{-1+i \sqrt{3}}{2}$ and $\beta=\frac{-1-i \sqrt{3}}{2}, i=\sqrt{-1}$. If
$$ (7-7 \alpha+9 \beta)^{20}+(9+7 \alpha-7 \beta)^{20}+(-7+9 \alpha+7 \beta)^{20}+(14+7 \alpha+7 \beta)^{20}=m^{10}, $$
then $m$ is $\_\_\_\_$
Q44.
mcq single
+4 / 1
If the line $\alpha x+2 y=1$, where $\alpha \in \mathbb{R}$, does not meet the hyperbola $x^2-9 y^2=9$, then a possible value of $\alpha$ is :
Q45.
mcq single
+4 / 1
If the domain of the function $f(x)=\sin ^{-1}\left(\frac{5-x}{3+2 x}\right)+\frac{1}{\log _e(10-x)}$ is $(-\infty, \alpha] \cup[\beta, \gamma)-\{\delta\}$, then $6(\alpha+\beta+\gamma+\delta)$ is equal to
Q46.
mcq single
+4 / 1
Let the solution curve of the differential equation $x d y-y d x=\sqrt{x^2+y^2} d x, x>0$, $y(1)=0$, be $y=y(x)$. Then $y(3)$ is equal to
Q47.
mcq single
+4 / 1
Let the set of all values of $r$, for which the circles $(x+1)^2+(y+4)^2=r^2$ and $x^2+y^2-4 x-2 y-4=0$ intersect at two distinct points be the interval $(\alpha, \beta)$. Then $\alpha \beta$ is equal to
Q48.
numerical
+4 / 1
Let ABC be a triangle. Consider four points $\mathrm{p}_1, \mathrm{p}_2, \mathrm{p}_3, \mathrm{p}_4$ on the side AB , five points $p_5, p_6, p_7, p_8, p_9$ on the side $B C$, and four points $p_{10}, p_{11}, p_{12}, p_{13}$ on the side AC . None of these points is a vertex of the triangle ABC . Then the total number of pentagons, that can be formed by taking all the vertices from the points $p_1, p_2, \ldots, p_{13}$, is $\_\_\_\_$
Q49.
mcq single
+4 / 1
Let $f:[1, \infty) \rightarrow \mathbb{R}$ be a differentiable function. If $6 \int\limits_1^x f(t) d t=3 x f(x)+x^3-4$ for all $x \geq 1$, then the value of $f(2)-f(3)$ is :
Q50.
mcq single
+4 / 1
The value of $\int\limits_{-\frac{\pi}{2}}^{\frac{\pi}{2}}\left(\frac{1}{[x]+4}\right) d x$, where $[\cdot]$ denotes the greatest integer function, is
Physics
Physics
Q1.
mcq single
+4 / 1
Find the correct combination of $\mathrm{A}, \mathrm{B}, \mathrm{C}$ and D inputs which can cause the LED to glow.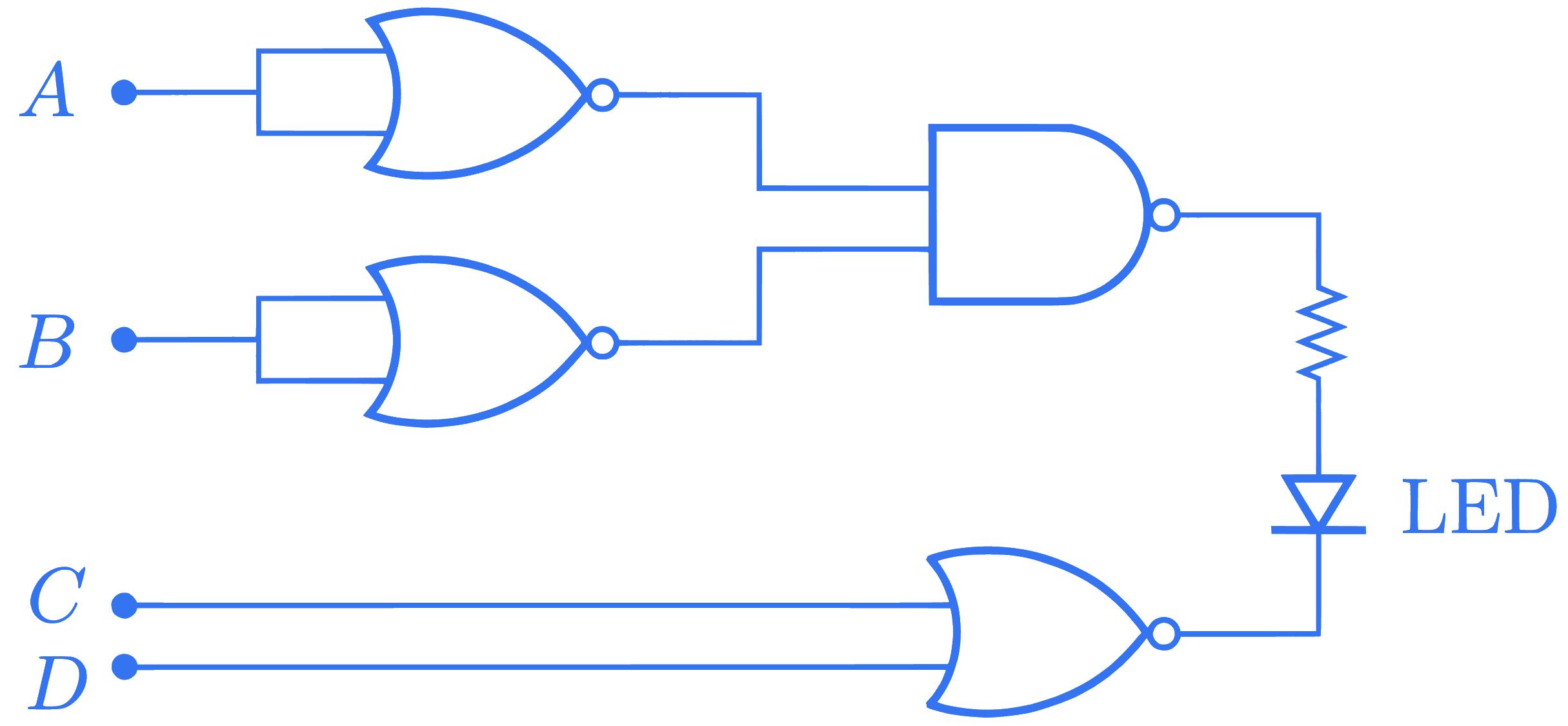

Q2.
mcq single
+4 / 1
A cylindrical tube $A B$ of length $l$, closed at both ends contains an ideal gas of 1 mol having molecular weight $M$. The tube is rotated in a horizontal plane with constant angular velocity $\omega$ about an axis perpendicular to $A B$ and passing through the edge at end $A$, as shown in the figure. If $P_A$ and $P_B$ are the pressures at $A$ and $B$ respectively, then (Consider the temperature is same at all points in the tube)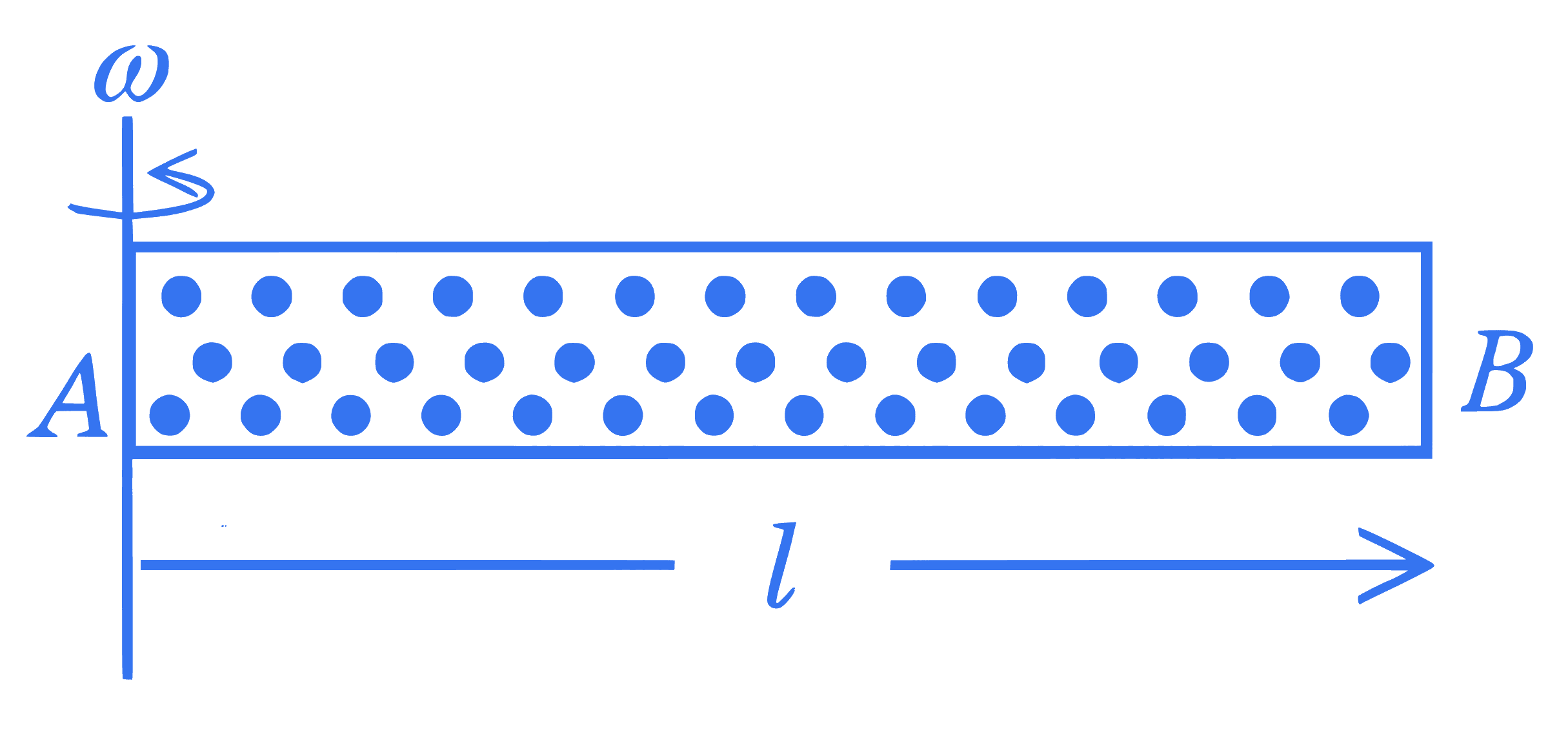

Q3.
mcq single
+4 / 1
A solid sphere of mass 5 kg and radius 10 cm is kept in contact with another solid sphere of mass 10 kg and radius 20 cm . The moment of inertia of this pair of spheres about the tangent passing through the point of contact is $\_\_\_\_$ $\mathrm{kg} \cdot \mathrm{m}^2$.
Q4.
numerical
+4 / 1
A circular disc has radius $R_1$ and thickness $T_1$. Another circular disc made of the same material has radius $R_2$ and thickness $T_2$. If the moment of inertia of both discs are same and $\frac{R_1}{R_2}=2$ then $\frac{T_1}{T_2}=\frac{1}{\alpha}$. The value of $\alpha$ is $\_\_\_\_$ .
Q5.
numerical
+4 / 1
Inductance of a coil with $10^4$ turns is 10 mH and it is connected to a dc source of 10 V with internal resistance of $10 \Omega$. The energy density in the inductor when the current reaches $\left(\frac{1}{e}\right)$ of its maximum value is $\alpha \pi \times \frac{1}{e^2} \mathrm{~J} / \mathrm{m}^3$. The value of $\alpha$ is
$\_\_\_\_$ .
$$ \left(\mu_0=4 \pi \times 10^{-7} \mathrm{Tm} / \mathrm{A}\right) . $$
Q6.
mcq single
+4 / 1
Three identical coils $C_1, C_2$ and $C_3$ are closely placed such that they share a common axis. $C_2$ is exactly midway. $C_1$ carries current $I$ in anti-clockwise direction while $C_3$ carries current $I$ in clockwise direction. An induced current flows through $C_2$ will be in clockwise direction when
Q7.
mcq single
+4 / 1
$X P Q Y$ is a vertical smooth long loop having a total resistance $R$ where $P X$ is parallel to $Q Y$ and separation between them is $l$. A constant magnetic field $B$ perpendicular to the plane of the loop exists in the entire space. A rod $C D$ of length $L(L>l)$ and mass $m$ is made to slide down from rest under the gravity as shown in figure. The terminal speed acquired by the rod is $\_\_\_\_$ $\mathrm{m} / \mathrm{s} .(\mathrm{g}=$ acceleration due to gravity)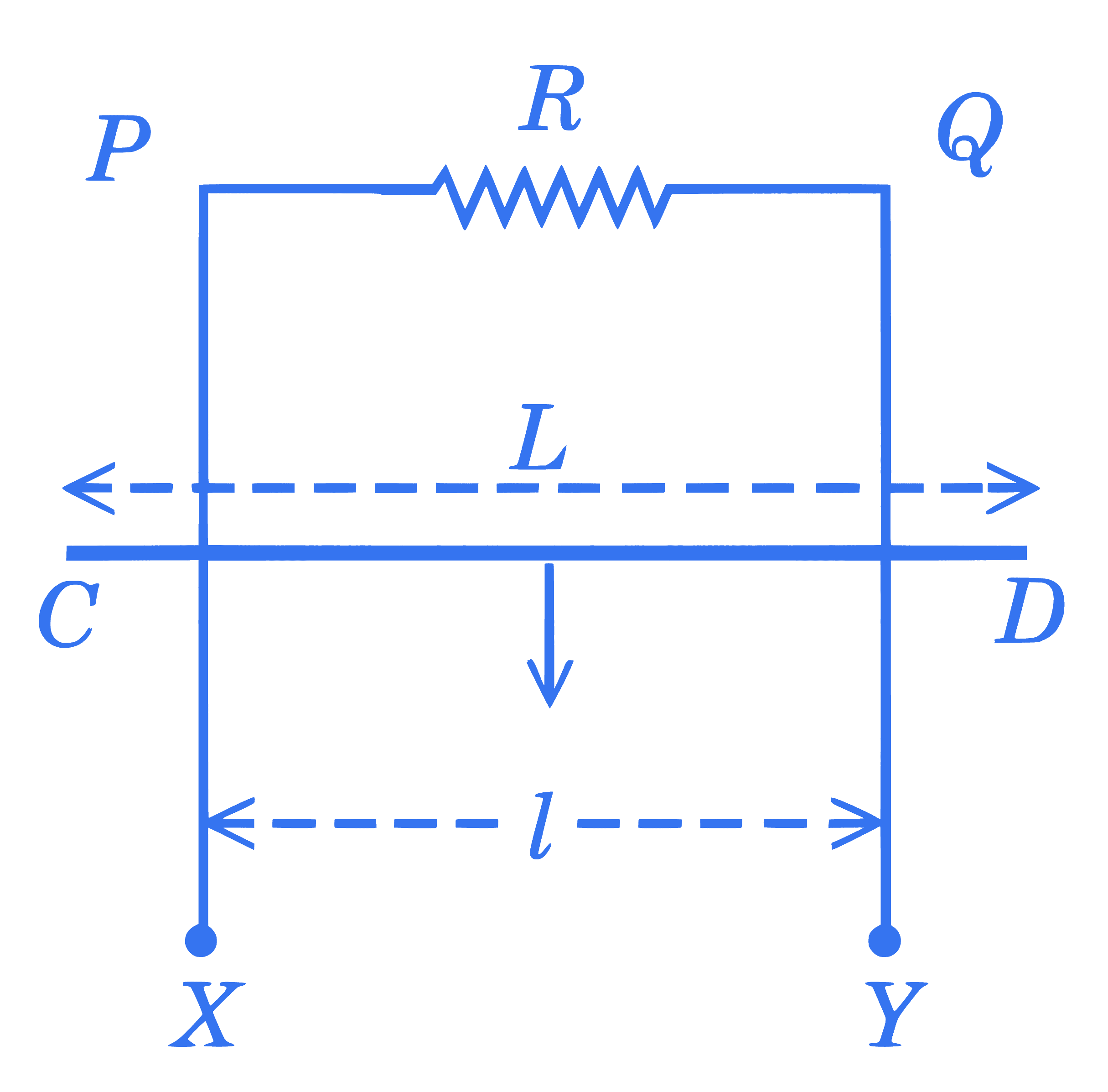

Q8.
mcq single
+4 / 1
The volume of an ideal gas increases 8 times and temperature becomes $(1 / 4)^{\text {th }}$ of initial temperature during a reversible change. If there is no exchange of heat in this process $(\Delta \mathrm{Q}=0)$ then identify the gas from the following options (Assuming the gases given in the options are ideal gases) :
Q9.
mcq single
+4 / 1
Rods $x$ and $y$ of equal dimensions but of different materials are joined as shown in figure. Temperatures of end points $A$ and $F$ are maintained at $100^{\circ} \mathrm{C}$ and $40^{\circ} \mathrm{C}$ respectively. Given the thermal conductivity of $\operatorname{rod} x$ is three times of that of $\operatorname{rod} y$, the temperature at junction points $B$ and $E$ are (close to):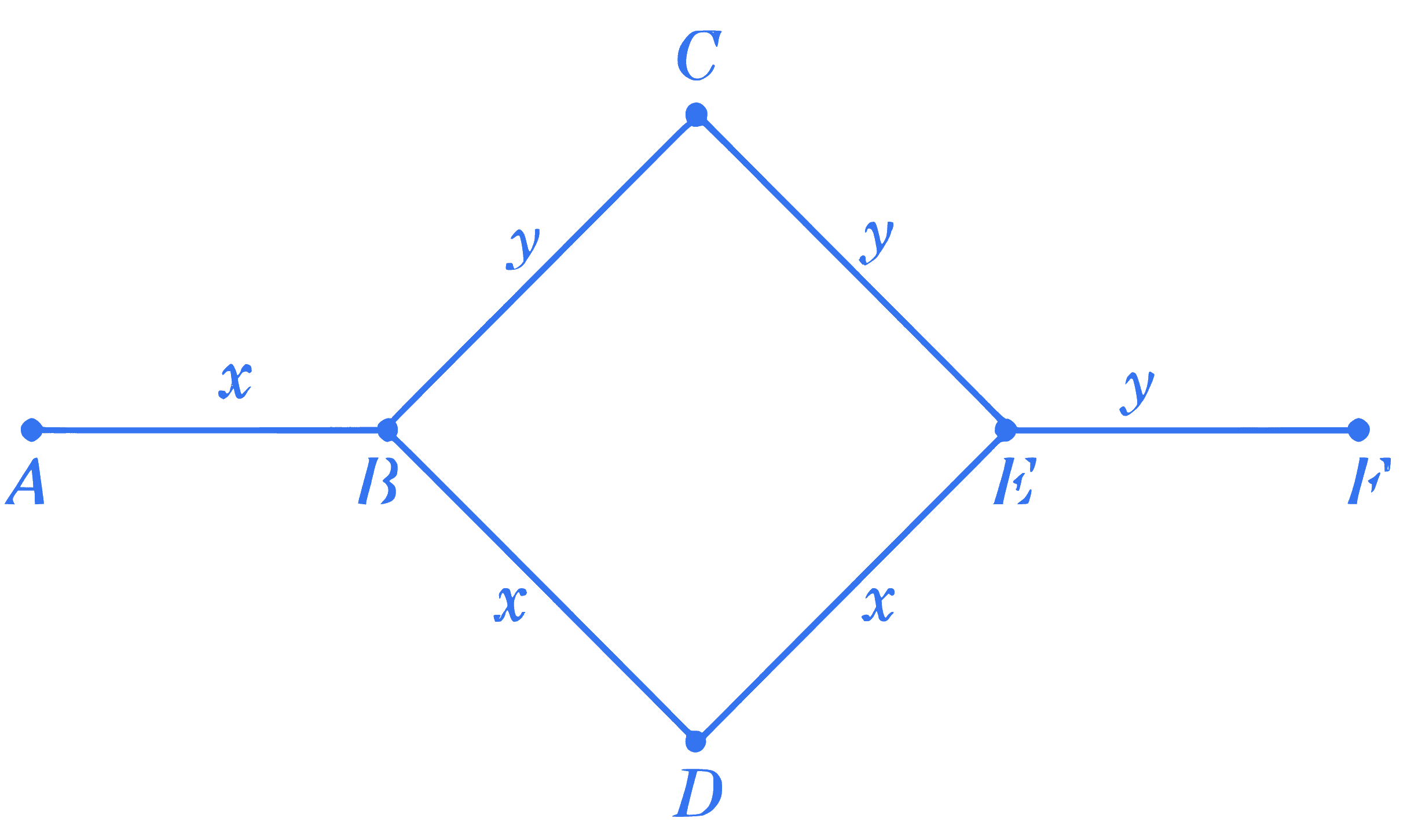

Q10.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Pressure of a fluid is exerted only on a solid surface in contact as the fluid-pressure does not exist everywhere in a still fluid.
**Statement II :** Excess potential energy of the molecules on the surface of a liquid, when compared to interior, results in surface tension.
In the light of the above statements, choose the correct answer from the options given below
Q11.
mcq single
+4 / 1
Electric field in a region is given by $\vec{E}=A x \hat{i}+B y \hat{j}$, where $A=10 \mathrm{~V} / \mathrm{m}^2$ and $B=5 \mathrm{~V} / \mathrm{m}^2$. If the electric potential at a point $(10,20)$ is 500 V , then the electric potential at origin is $\_\_\_\_$ V.
Q12.
mcq single
+4 / 1
A simple pendulum has a bob with mass $m$ and charge $q$. The pendulum string has negligible mass. When a uniform and horizontal electric field $\vec{E}$ is applied, the tension in the string changes. The final tension in the string, when pendulum attains an equilibrium position is $\_\_\_\_$ .
Q13.
mcq single
+4 / 1
Six point charges are kept $60^{\circ}$ apart from each other on the circumference of a circle of radius $R$ as shown in figure. The net electric field at the center of the circle is $\_\_\_\_$ .
( $\epsilon_0$ is permittivity of free space)
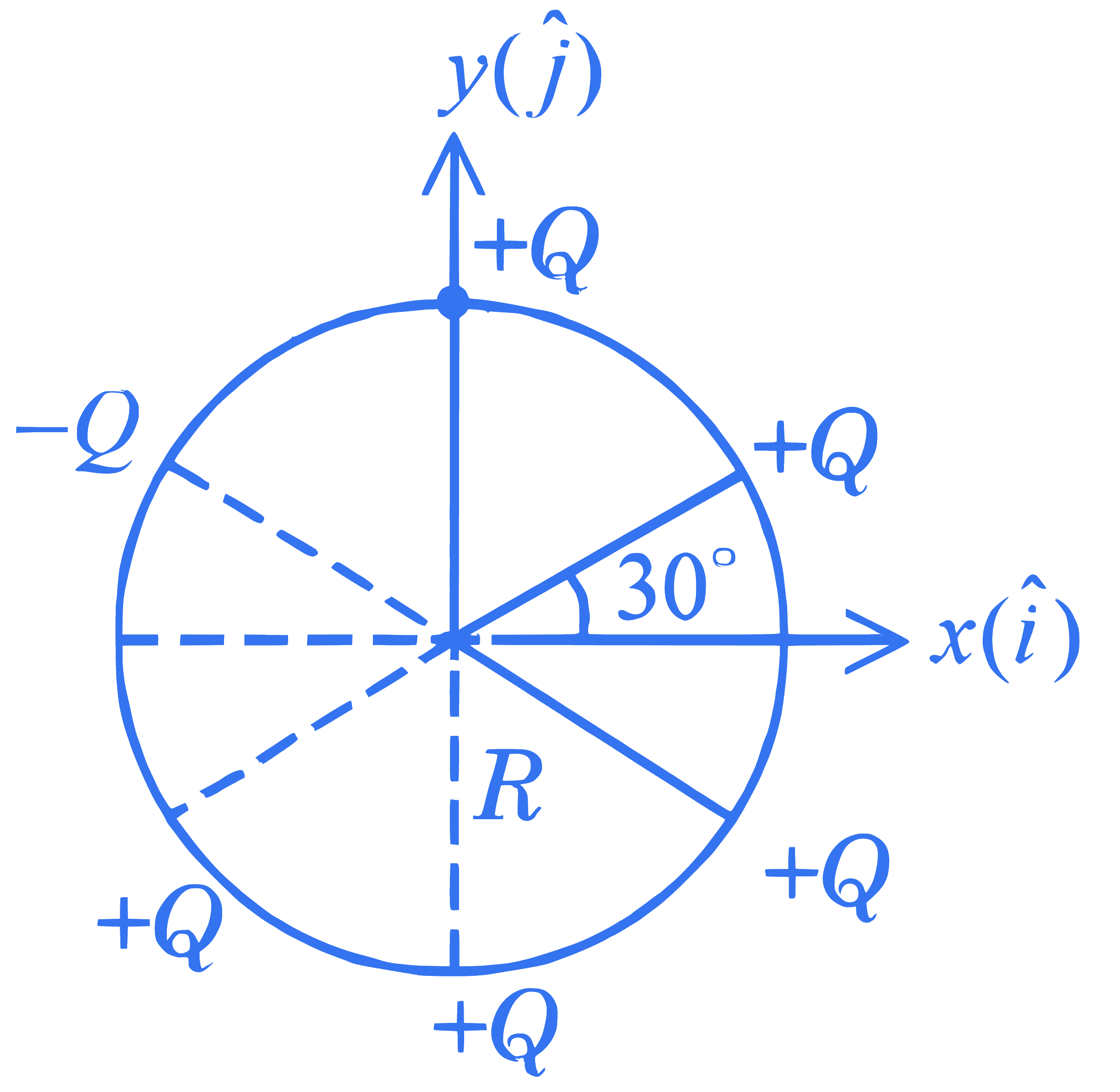

Q14.
mcq single
+4 / 1
Net gravitational force at the center of a square is found to be $F_1$ when four particles having mass $M, 2 M, 3 M$ and $4 M$ are placed at the four corners of the square as shown in figure and it is $F_2$ when the positions of $3 M$ and $4 M$ are interchanged. The ratio $\frac{F_1}{F_2}$ is $\frac{\alpha}{\sqrt{5}}$. The value of $\alpha$ is $\_\_\_\_$ .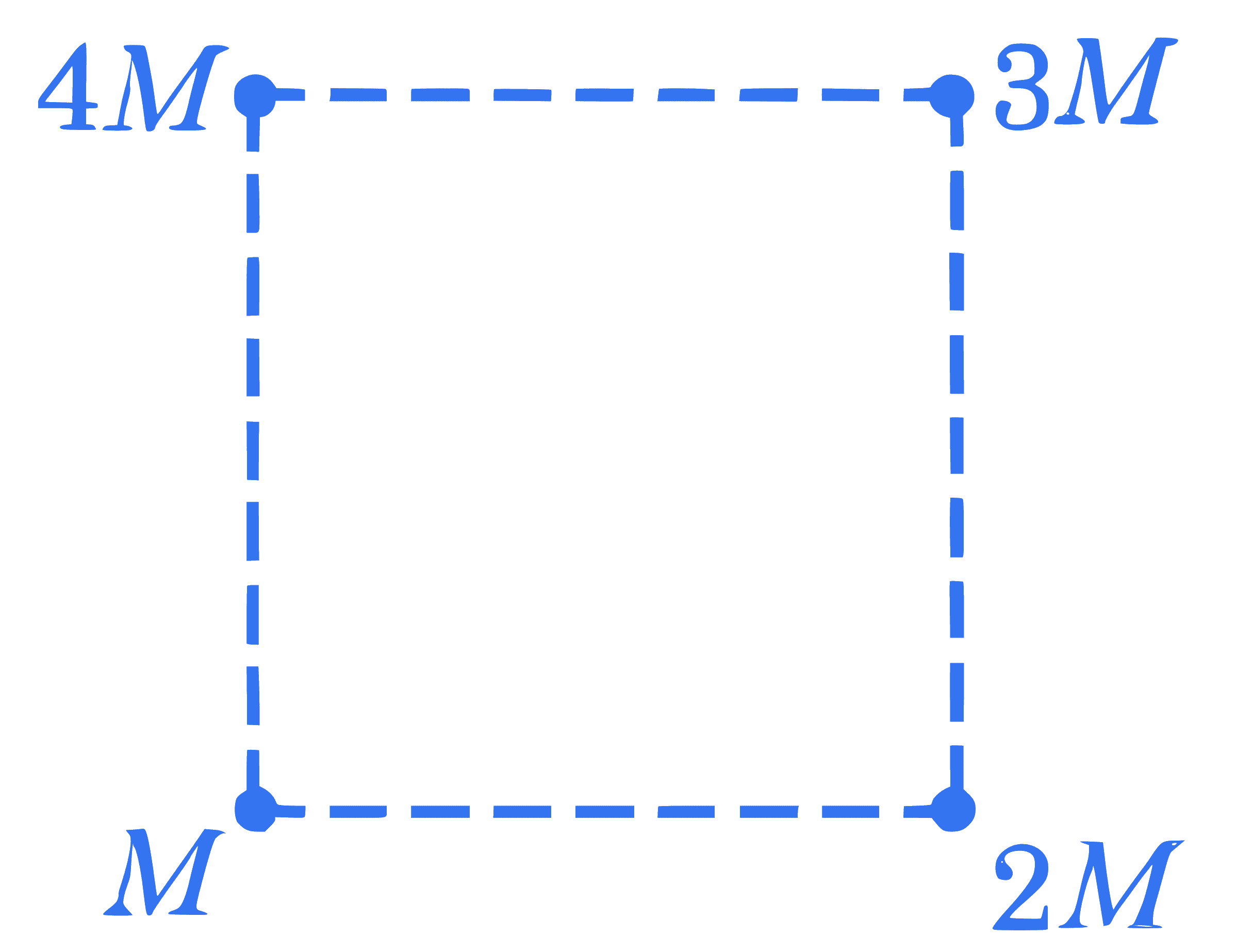

Q15.
mcq single
+4 / 1
The escape velocity from a spherical planet $A$ is $10 \mathrm{~km} / \mathrm{s}$. The escape velocity from another planet $B$ whose density and radius are $10 \%$ of those of planet $A$, is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}$.
Q16.
mcq single
+4 / 1
Consider an equilateral prism (refractive index $\sqrt{2}$ ). A ray of light is incident on its one surface at a certain angle $i$. If the emergent ray is found to graze along the other surface then the angle of refraction at the incident surface is close to $\_\_\_\_$
Q17.
numerical
+4 / 1
A parallel beam of light travelling in air (refractive index 1.0) is incident on a convex spherical glass surface of radius of curvature 50 cm . Refractive index of glass is 1.5 . The rays converge to a point at a distance $x \mathrm{~cm}$ from the centre of the curvature of the spherical surface. The value of $x$ is $\_\_\_\_$ cm .
Q18.
mcq single
+4 / 1
A thin convex lens of focal length 5 cm and a thin concave lens of focal length 4 cm are combined together (without any gap) and this combination has magnification $m_1$ when an object is placed 10 cm before the convex lens. Keeping the positions of convex lens and object undisturbed a gap of 1 cm is introduced between the lenses by moving the concave lens away, which lead to a change in magnification of total lens system to $m_2$. The value of $\left|\frac{m_1}{m_2}\right|$ is $\_\_\_\_$ .
Q19.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
List-II
A.
Spring constant
I.
M
L
2
T
−
2
K
−
1
M
L
2
T
−
2
K
−
1
ML^(2)T^(-2)K^(-1)
B.
Thermal conductivity
II.
ML
0
T
−
2
ML
0
T
−
2
ML^(0)T^(-2)
C.
Boltzmann constant
III.
M
L
2
T
−
3
A
−
2
M
L
2
T
−
3
A
−
2
ML^(2)T^(-3)A^(-2)
D.
Inductive reactance
IV.
M
L
T
−
3
K
−
1
M
L
T
−
3
K
−
1
MLT^(-3)K^(-1)
Choose the correct answer from the options given below:
Q20.
mcq single
+4 / 1
$7.9 \mathrm{MeV} \alpha$-particle scatters from a target material of atomic number 79 . From the given data the estimated diameter of nuclei of the target material is (approximately) $\_\_\_\_$ m.
$$ \left[\frac{1}{4 \pi \epsilon_{\mathrm{o}}}=9 \times 10^9 \mathrm{Nm}^2 / \mathrm{C}^2 \text { and electron charge }=1.6 \times 10^{-19} \mathrm{C}\right] $$
Q21.
mcq single
+4 / 1
The minimum frequency of photon required to break a particle of mass 15.348 amu into $4 \alpha$ particles is $\_\_\_\_$ kHz .
[mass of He nucleus = $4.002 \mathrm{amu}, 1 \mathrm{amu}=1.66 \times 10^{-27} \mathrm{~kg}, \mathrm{~h}=6.6 \times 10^{-34} \mathrm{~J} . \mathrm{s}$ and $\mathrm{c}=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ]
Q22.
mcq single
+4 / 1
A projectile is thrown upward at an angle $60^{\circ}$ with the horizontal. The speed of the projectile is $20 \mathrm{~m} / \mathrm{s}$ when its direction of motion is $45^{\circ}$ with the horizontal. The initial speed of the projectile is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}$.
Q23.
numerical
+4 / 1
Two loudspeakers $\left(L_1\right.$ and $\left.L_2\right)$ are placed with a separation of 10 m , as shown in figure. Both speakers are fed with an audio input signal of same frequency with constant volume. A voice recorder, initially at point $A$, at equidistance to both loud speakers, is moved by 25 m along the line $A B$ while monitoring the audio signal. The measured signal was found to undergo 10 cycles of minima and maxima during the movement. The frequency of the input signal is $\_\_\_\_$ Hz (Speed of sound in air is $324 \mathrm{~m} / \mathrm{s}$ and $\sqrt{5}=2.23$ )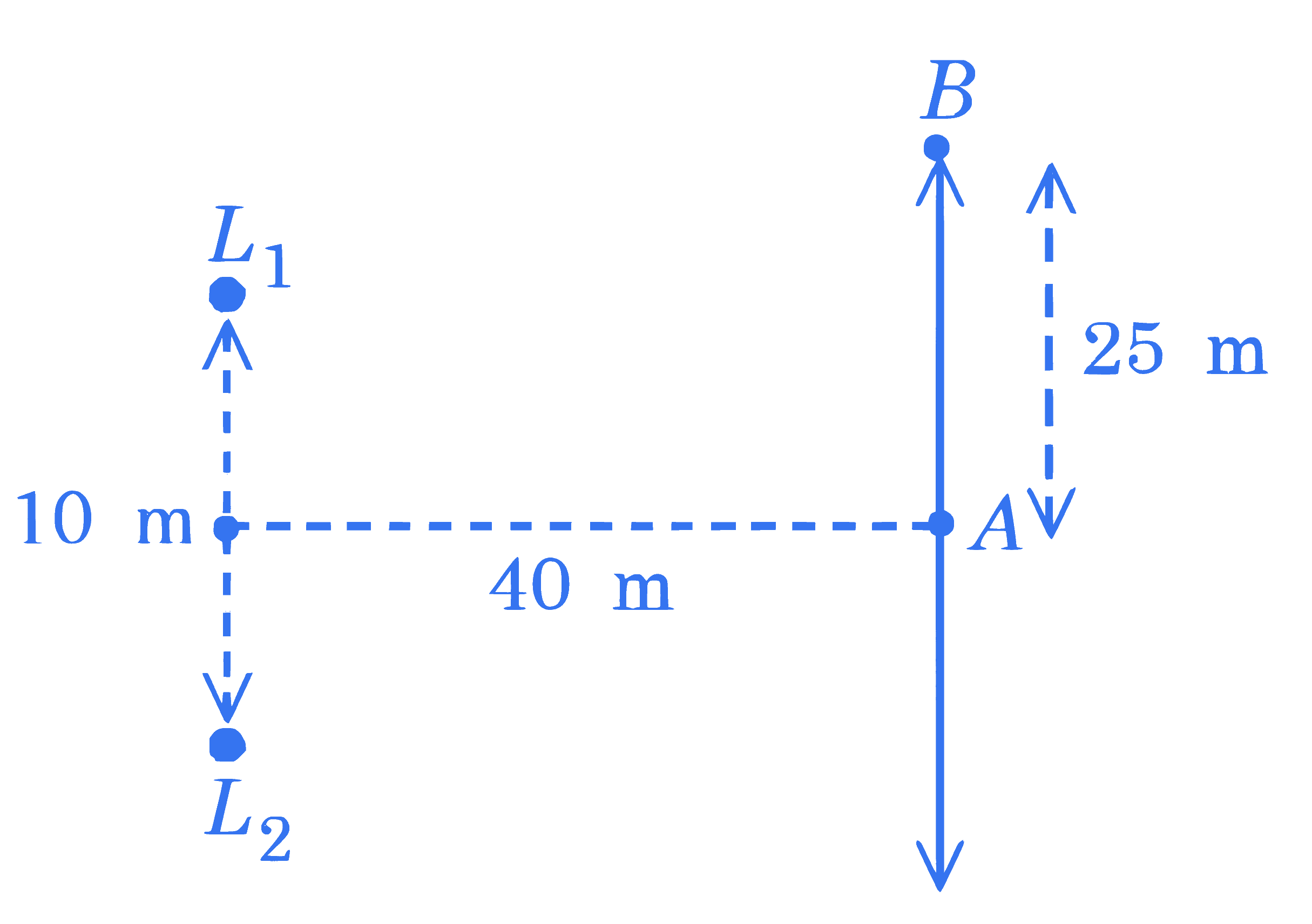

Q24.
mcq single
+4 / 1
A meter bridge with two resistances $R_1$ and $R_2$ as shown in figure was balanced (null point) at 40 cm from the point $P$. The null point changed to 50 cm from the point $P$, when $16 \Omega$ resistance is connected in parallel to $R_2$. The values of resistances $R_1$ and $R_2$ are $\_\_\_\_$ .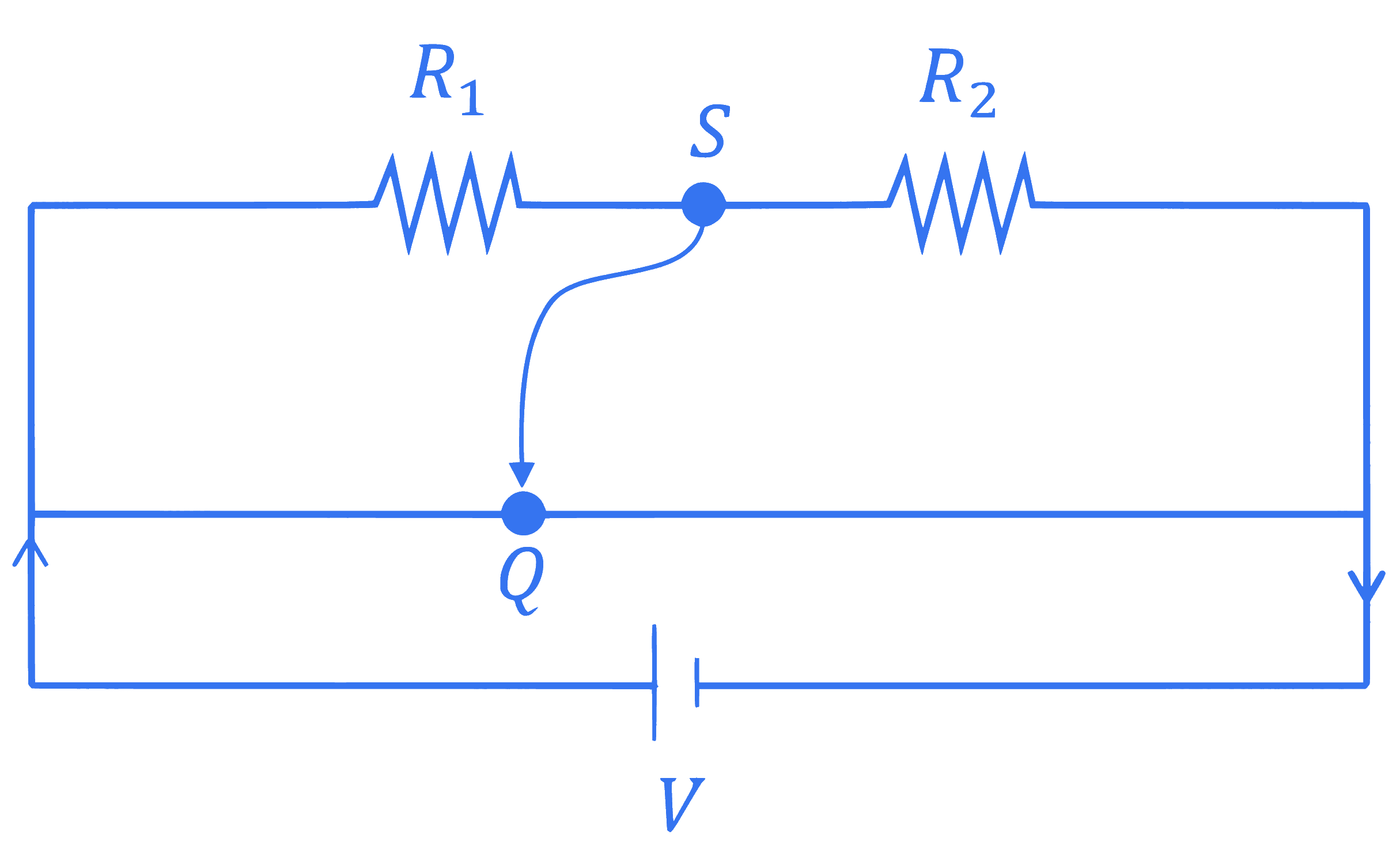

Q25.
numerical
+4 / 1
The electric field of a plane electromagnetic wave, travelling in an unknown nonmagnetic medium is given by,
$$ E_{\mathrm{y}}=20 \sin \left(3 \times 10^6 x-4.5 \times 10^{14} \mathrm{t}\right) \mathrm{V} / \mathrm{m} $$
(where $x, \mathrm{t}$ and other values have S.I. units). The dielectric constant of the medium is $\_\_\_\_$
(speed of light in free space is $3 \times 10^8 \mathrm{~m} / \mathrm{s}$ )
Q26.
mcq single
+4 / 1
Find the correct combination of $\mathrm{A}, \mathrm{B}, \mathrm{C}$ and D inputs which can cause the LED to glow.

Q27.
mcq single
+4 / 1
A cylindrical tube $A B$ of length $l$, closed at both ends contains an ideal gas of 1 mol having molecular weight $M$. The tube is rotated in a horizontal plane with constant angular velocity $\omega$ about an axis perpendicular to $A B$ and passing through the edge at end $A$, as shown in the figure. If $P_A$ and $P_B$ are the pressures at $A$ and $B$ respectively, then (Consider the temperature is same at all points in the tube)

Q28.
mcq single
+4 / 1
A solid sphere of mass 5 kg and radius 10 cm is kept in contact with another solid sphere of mass 10 kg and radius 20 cm . The moment of inertia of this pair of spheres about the tangent passing through the point of contact is $\_\_\_\_$ $\mathrm{kg} \cdot \mathrm{m}^2$.
Q29.
numerical
+4 / 1
A circular disc has radius $R_1$ and thickness $T_1$. Another circular disc made of the same material has radius $R_2$ and thickness $T_2$. If the moment of inertia of both discs are same and $\frac{R_1}{R_2}=2$ then $\frac{T_1}{T_2}=\frac{1}{\alpha}$. The value of $\alpha$ is $\_\_\_\_$ .
Q30.
numerical
+4 / 1
Inductance of a coil with $10^4$ turns is 10 mH and it is connected to a dc source of 10 V with internal resistance of $10 \Omega$. The energy density in the inductor when the current reaches $\left(\frac{1}{e}\right)$ of its maximum value is $\alpha \pi \times \frac{1}{e^2} \mathrm{~J} / \mathrm{m}^3$. The value of $\alpha$ is
$\_\_\_\_$ .
$$ \left(\mu_0=4 \pi \times 10^{-7} \mathrm{Tm} / \mathrm{A}\right) . $$
Q31.
mcq single
+4 / 1
Three identical coils $C_1, C_2$ and $C_3$ are closely placed such that they share a common axis. $C_2$ is exactly midway. $C_1$ carries current $I$ in anti-clockwise direction while $C_3$ carries current $I$ in clockwise direction. An induced current flows through $C_2$ will be in clockwise direction when
Q32.
mcq single
+4 / 1
$X P Q Y$ is a vertical smooth long loop having a total resistance $R$ where $P X$ is parallel to $Q Y$ and separation between them is $l$. A constant magnetic field $B$ perpendicular to the plane of the loop exists in the entire space. A rod $C D$ of length $L(L>l)$ and mass $m$ is made to slide down from rest under the gravity as shown in figure. The terminal speed acquired by the rod is $\_\_\_\_$ $\mathrm{m} / \mathrm{s} .(\mathrm{g}=$ acceleration due to gravity)

Q33.
mcq single
+4 / 1
The volume of an ideal gas increases 8 times and temperature becomes $(1 / 4)^{\text {th }}$ of initial temperature during a reversible change. If there is no exchange of heat in this process $(\Delta \mathrm{Q}=0)$ then identify the gas from the following options (Assuming the gases given in the options are ideal gases) :
Q34.
mcq single
+4 / 1
Rods $x$ and $y$ of equal dimensions but of different materials are joined as shown in figure. Temperatures of end points $A$ and $F$ are maintained at $100^{\circ} \mathrm{C}$ and $40^{\circ} \mathrm{C}$ respectively. Given the thermal conductivity of $\operatorname{rod} x$ is three times of that of $\operatorname{rod} y$, the temperature at junction points $B$ and $E$ are (close to):

Q35.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Pressure of a fluid is exerted only on a solid surface in contact as the fluid-pressure does not exist everywhere in a still fluid.
**Statement II :** Excess potential energy of the molecules on the surface of a liquid, when compared to interior, results in surface tension.
In the light of the above statements, choose the correct answer from the options given below
Q36.
mcq single
+4 / 1
Electric field in a region is given by $\vec{E}=A x \hat{i}+B y \hat{j}$, where $A=10 \mathrm{~V} / \mathrm{m}^2$ and $B=5 \mathrm{~V} / \mathrm{m}^2$. If the electric potential at a point $(10,20)$ is 500 V , then the electric potential at origin is $\_\_\_\_$ V.
Q37.
mcq single
+4 / 1
A simple pendulum has a bob with mass $m$ and charge $q$. The pendulum string has negligible mass. When a uniform and horizontal electric field $\vec{E}$ is applied, the tension in the string changes. The final tension in the string, when pendulum attains an equilibrium position is $\_\_\_\_$ .
Q38.
mcq single
+4 / 1
Six point charges are kept $60^{\circ}$ apart from each other on the circumference of a circle of radius $R$ as shown in figure. The net electric field at the center of the circle is $\_\_\_\_$ .
( $\epsilon_0$ is permittivity of free space)


Q39.
mcq single
+4 / 1
Net gravitational force at the center of a square is found to be $F_1$ when four particles having mass $M, 2 M, 3 M$ and $4 M$ are placed at the four corners of the square as shown in figure and it is $F_2$ when the positions of $3 M$ and $4 M$ are interchanged. The ratio $\frac{F_1}{F_2}$ is $\frac{\alpha}{\sqrt{5}}$. The value of $\alpha$ is $\_\_\_\_$ .

Q40.
mcq single
+4 / 1
The escape velocity from a spherical planet $A$ is $10 \mathrm{~km} / \mathrm{s}$. The escape velocity from another planet $B$ whose density and radius are $10 \%$ of those of planet $A$, is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}$.
Q41.
mcq single
+4 / 1
Consider an equilateral prism (refractive index $\sqrt{2}$ ). A ray of light is incident on its one surface at a certain angle $i$. If the emergent ray is found to graze along the other surface then the angle of refraction at the incident surface is close to $\_\_\_\_$
Q42.
numerical
+4 / 1
A parallel beam of light travelling in air (refractive index 1.0) is incident on a convex spherical glass surface of radius of curvature 50 cm . Refractive index of glass is 1.5 . The rays converge to a point at a distance $x \mathrm{~cm}$ from the centre of the curvature of the spherical surface. The value of $x$ is $\_\_\_\_$ cm .
Q43.
mcq single
+4 / 1
A thin convex lens of focal length 5 cm and a thin concave lens of focal length 4 cm are combined together (without any gap) and this combination has magnification $m_1$ when an object is placed 10 cm before the convex lens. Keeping the positions of convex lens and object undisturbed a gap of 1 cm is introduced between the lenses by moving the concave lens away, which lead to a change in magnification of total lens system to $m_2$. The value of $\left|\frac{m_1}{m_2}\right|$ is $\_\_\_\_$ .
Q44.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
List-II
A.
Spring constant
I.
M
L
2
T
−
2
K
−
1
M
L
2
T
−
2
K
−
1
ML^(2)T^(-2)K^(-1)
B.
Thermal conductivity
II.
ML
0
T
−
2
ML
0
T
−
2
ML^(0)T^(-2)
C.
Boltzmann constant
III.
M
L
2
T
−
3
A
−
2
M
L
2
T
−
3
A
−
2
ML^(2)T^(-3)A^(-2)
D.
Inductive reactance
IV.
M
L
T
−
3
K
−
1
M
L
T
−
3
K
−
1
MLT^(-3)K^(-1)
Choose the correct answer from the options given below:
Q45.
mcq single
+4 / 1
$7.9 \mathrm{MeV} \alpha$-particle scatters from a target material of atomic number 79 . From the given data the estimated diameter of nuclei of the target material is (approximately) $\_\_\_\_$ m.
$$ \left[\frac{1}{4 \pi \epsilon_{\mathrm{o}}}=9 \times 10^9 \mathrm{Nm}^2 / \mathrm{C}^2 \text { and electron charge }=1.6 \times 10^{-19} \mathrm{C}\right] $$
Q46.
mcq single
+4 / 1
The minimum frequency of photon required to break a particle of mass 15.348 amu into $4 \alpha$ particles is $\_\_\_\_$ kHz .
[mass of He nucleus = $4.002 \mathrm{amu}, 1 \mathrm{amu}=1.66 \times 10^{-27} \mathrm{~kg}, \mathrm{~h}=6.6 \times 10^{-34} \mathrm{~J} . \mathrm{s}$ and $\mathrm{c}=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ]
Q47.
mcq single
+4 / 1
A projectile is thrown upward at an angle $60^{\circ}$ with the horizontal. The speed of the projectile is $20 \mathrm{~m} / \mathrm{s}$ when its direction of motion is $45^{\circ}$ with the horizontal. The initial speed of the projectile is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}$.
Q48.
numerical
+4 / 1
Two loudspeakers $\left(L_1\right.$ and $\left.L_2\right)$ are placed with a separation of 10 m , as shown in figure. Both speakers are fed with an audio input signal of same frequency with constant volume. A voice recorder, initially at point $A$, at equidistance to both loud speakers, is moved by 25 m along the line $A B$ while monitoring the audio signal. The measured signal was found to undergo 10 cycles of minima and maxima during the movement. The frequency of the input signal is $\_\_\_\_$ Hz (Speed of sound in air is $324 \mathrm{~m} / \mathrm{s}$ and $\sqrt{5}=2.23$ )

Q49.
mcq single
+4 / 1
A meter bridge with two resistances $R_1$ and $R_2$ as shown in figure was balanced (null point) at 40 cm from the point $P$. The null point changed to 50 cm from the point $P$, when $16 \Omega$ resistance is connected in parallel to $R_2$. The values of resistances $R_1$ and $R_2$ are $\_\_\_\_$ .

Q50.
numerical
+4 / 1
The electric field of a plane electromagnetic wave, travelling in an unknown nonmagnetic medium is given by,
$$ E_{\mathrm{y}}=20 \sin \left(3 \times 10^6 x-4.5 \times 10^{14} \mathrm{t}\right) \mathrm{V} / \mathrm{m} $$
(where $x, \mathrm{t}$ and other values have S.I. units). The dielectric constant of the medium is $\_\_\_\_$
(speed of light in free space is $3 \times 10^8 \mathrm{~m} / \mathrm{s}$ )