JEE Main 2026 (Online) 23rd January Evening Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
A student has been given a compound " $x$ " of molecular formula- $\mathrm{C}_6 \mathrm{H}_7 \mathrm{~N}$. ' $x$ ' is sparingly soluble in water. However, on addition of dilute mineral acid, ' $x$ ' becomes soluble in water. ' $x$ ' when treated with $\mathrm{CHCl}_3$ and $\mathrm{KOH}(\mathrm{alc})$, ' $y$ ' is produced. ' $y$ ' has a specific unpleasant smell. On treatment with benzenesulphonyl chloride, ' $x$ ' gives a compound ' $z$ ' which is soluble in alkali. The number of different " H " atoms present in ' z ' is:-
Q2.
mcq single
+4 / 1
$$ \text { Given below are two statements : } $$
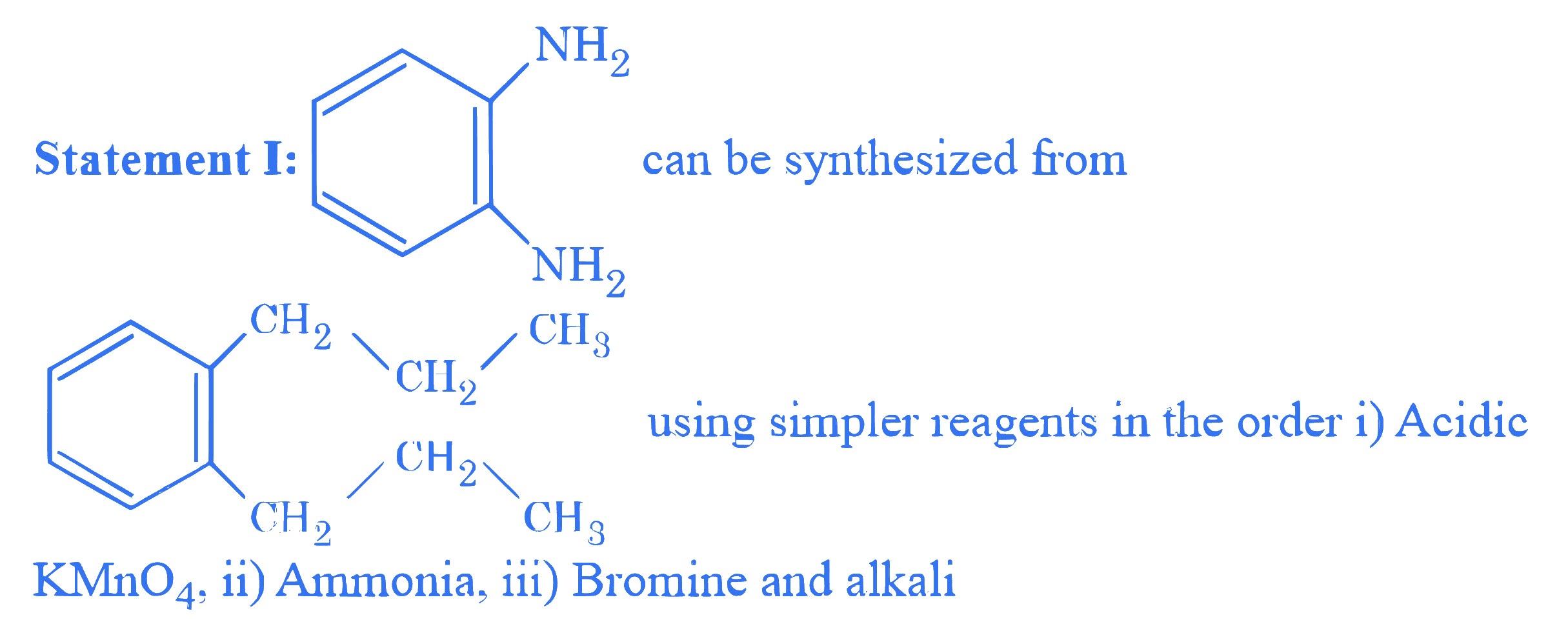
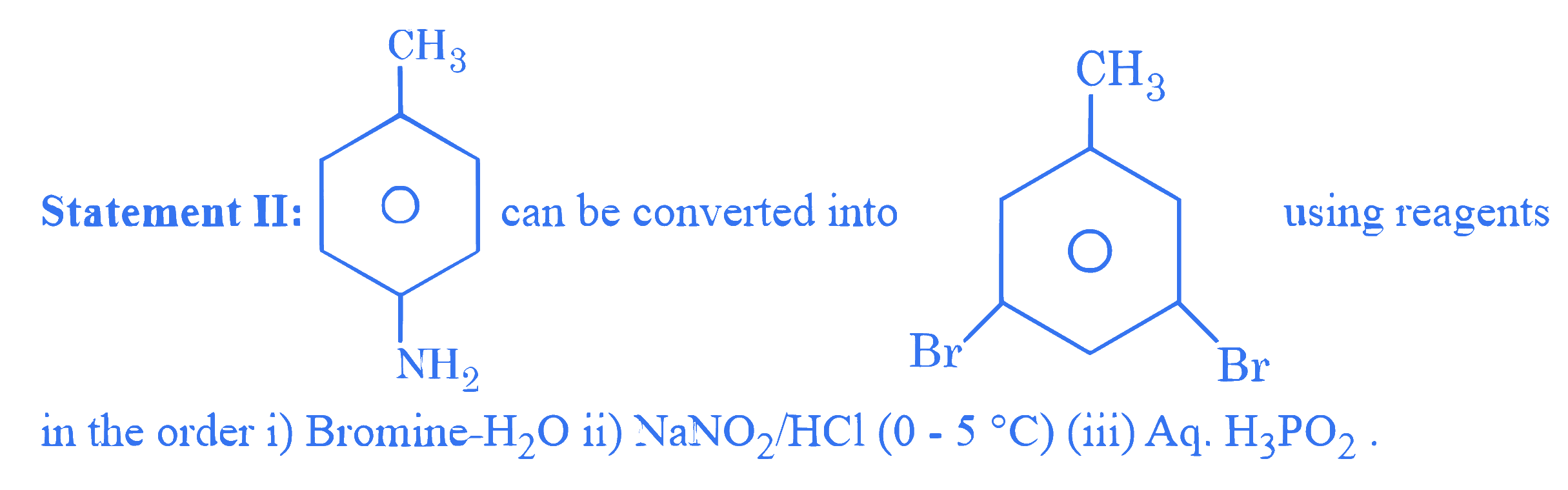 In the light of the above statements, choose the correct answer from the options given below
In the light of the above statements, choose the correct answer from the options given below

 In the light of the above statements, choose the correct answer from the options given below
In the light of the above statements, choose the correct answer from the options given below
Q3.
mcq single
+4 / 1
Observe the following reactions at $\mathrm{T}(\mathrm{K})$.
I. $\mathrm{A} \rightarrow$ products.
II. $5 \mathrm{Br}^{-}(\mathrm{aq})+\mathrm{BrO}_3{ }^{-}(\mathrm{aq})+6 \mathrm{H}^{+}(\mathrm{aq}) \rightarrow 3 \mathrm{Br}_2(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\mathrm{l})$
Both the reactions are started at 10.00 am . The rates of these reactions at 10.10 am are same. The value of $-\frac{\Delta\left[\mathrm{Br}^{-}\right]}{\Delta \mathrm{t}}$ at 10.10 am is $2 \times 10^{-4} \mathrm{~mol} \mathrm{~L}^{-1} \mathrm{~min}^{-1}$. The concentration of A at 10.10 am is $10^{-2} \mathrm{~mol} \mathrm{~L}^{-1}$. What is the first order rate constant (in $\mathrm{min}^{-1}$ ) of reaction $I$ ?
Q4.
mcq single
+4 / 1
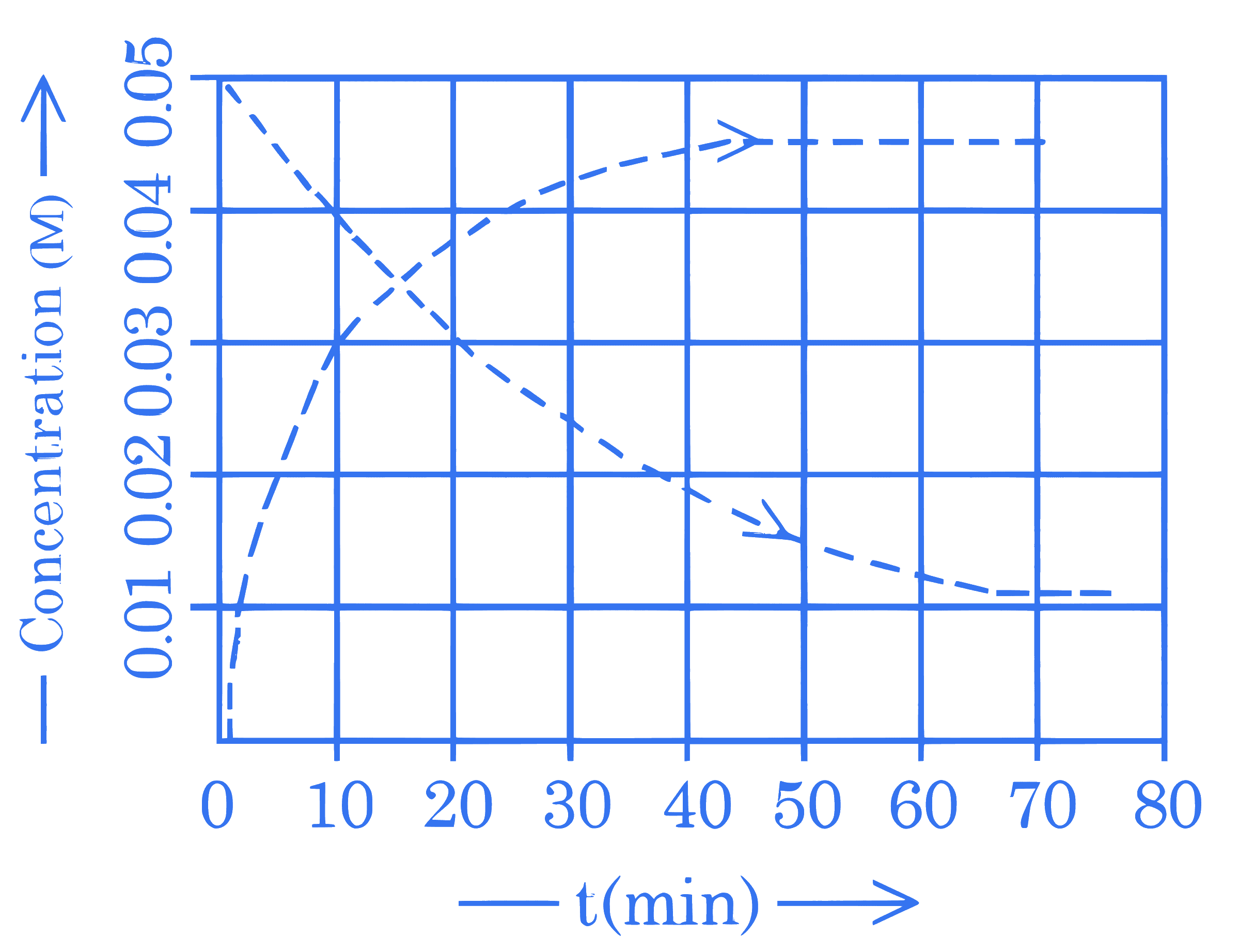 Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
 Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
Q5.
mcq single
+4 / 1
Which statements are **NOT TRUE** about $\mathrm{XeO}_2 \mathrm{~F}_2$?
A. It has a see-saw shape.
B. Xe has 5 electron pairs in its valence shell in $\mathrm{XeO}_2 \mathrm{~F}_2$.
C. The $\mathrm{O}-\mathrm{Xe}-\mathrm{O}$ bond angle is close to $180^{\circ}$.
D. The $\mathrm{F}-\mathrm{Xe}-\mathrm{F}$ bond angle is close to $180^{\circ}$.
E. Xe has 16 valence electrons in $\mathrm{XeO}_2 \mathrm{~F}_2$.
Choose the ***correct*** answer from the options given below :
Q6.
mcq single
+4 / 1
The oxidation state of chromium in the final product formed in the reaction between KI and acidified $\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7$ solution is :
Q7.
numerical
+4 / 1
200 cc of $x \times 10^{-3} \mathrm{M}$ potassium dichromate is required to oxidise 750 cc of 0.6 M Mohr's salt solution in acidic medium.
Here $x=$ $\_\_\_\_$ .
Q8.
numerical
+4 / 1
Two liquids A and B form an ideal solution. At 320 K , the vapour pressure of the solution, containing 3 mol of $A$ and 1 mol of $B$ is 500 mm Hg . At the same temperature, if 1 mol of A is further added to this solution, vapour pressure of the solution increases by 20 mm Hg . Vapour pressure (in mm Hg ) of B in pure state is $\_\_\_\_$ . (Nearest integer)
Q9.
numerical
+4 / 1
$$ \mathrm{X}_2(\mathrm{~g})+\mathrm{Y}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{Z}(\mathrm{~g}) $$
$\mathrm{X}_2(\mathrm{~g})$ and $\mathrm{Y}_2(\mathrm{~g})$ are added to a 1 L flask and it is found that the system attains the above equilibrium at $\mathrm{T}(\mathrm{K})$ with the number of moles of $\mathrm{X}_2(\mathrm{~g}), \mathrm{Y}_2(\mathrm{~g})$ and $\mathrm{Z}(\mathrm{g})$ being 3,3 and 9 mol respectively (equilibrium moles). Under this condition of equilibrium, 10 mol of $\mathrm{Z}(\mathrm{g})$ is added to the flask and the temperature is maintained at $\mathrm{T}(\mathrm{K})$. Then the number of moles of $\mathrm{Z}(\mathrm{g})$ in the flask when the new equilibrium is established is $\_\_\_\_$ . (Nearest integer)
Q10.
mcq single
+4 / 1
Elements X and Y belong to Group 15. The difference between the electronegativity values of ' X ' and phosphorus is higher than that of the difference between phosphorus and ' Y '. ' X ' & ' Y ' are respectively
Q11.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The second ionisation enthalpy of Na is larger than the corresponding ionisation enthalpy of Mg .
**Statement II :** The ionic radius of $\mathrm{O}^{2-}$ is larger than that of $\mathrm{F}^{-}$.
In the light of the above statements, choose the ***correct*** answer from the options given below :
Q12.
mcq single
+4 / 1
It is noticed that $\mathrm{Pb}^{2+}$ is more stable than $\mathrm{Pb}^{4+}$ but $\mathrm{Sn}^{2+}$ is less stable than $\mathrm{Sn}^{4+}$. Observe the following reactions.
$$ \begin{aligned} & \mathrm{PbO}_2+\mathrm{Pb} \rightarrow 2 \mathrm{PbO} ; \Delta_{\mathrm{r}} \mathrm{G}^{\circ}(1) \\ & \mathrm{SnO}_2+\mathrm{Sn} \rightarrow 2 \mathrm{SnO} ; \Delta_{\mathrm{r}} \mathrm{G}^{\circ}(2) \end{aligned} $$
Identify the correct set from the following
Q13.
mcq single
+4 / 1
Identify the CORRECT set of details from the following :
A. $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]^{3+}$ : Inner orbital complex; $\mathrm{d}^2 \mathrm{sp}^3$ hybridized
B. $\left[\mathrm{MnCl}_6\right]^{3-}$ : Outer orbital complex; $\mathrm{sp}^3 \mathrm{~d}^2$ hybridized
C. $\left[\mathrm{CoF}_6\right]^{3-}$ : Outer orbital complex; $\mathrm{d}^2 \mathrm{sp}^3$ hybridized
D. $\left[\mathrm{FeF}_6\right]^{3-}$ : Outer orbital complex; $\mathrm{sp}^3 \mathrm{~d}^2$ hybridized
E. $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}$ : Inner orbital complex; $\mathrm{sp}^3$ hybridized
Choose the correct answer from the options given below :
Q14.
numerical
+4 / 1
Total number of unpaired electrons present in the central metal atoms/ions of
$\left[\mathrm{Ni}(\mathrm{CO})_4\right],\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{PtCl}_2\left(\mathrm{NH}_3\right)_2\right],\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}$ and $\left[\mathrm{Pt}(\mathrm{CN})_4\right]^{2-}$ is $\_\_\_\_$。
Q15.
mcq single
+4 / 1
Which of the following statements are TRUE about Haloform reaction?
A. Sodium hypochlorite reacts with KI to give KOI .
B. KOI is a reducing agent.
C. $\alpha, \beta$-unsaturated methylketone 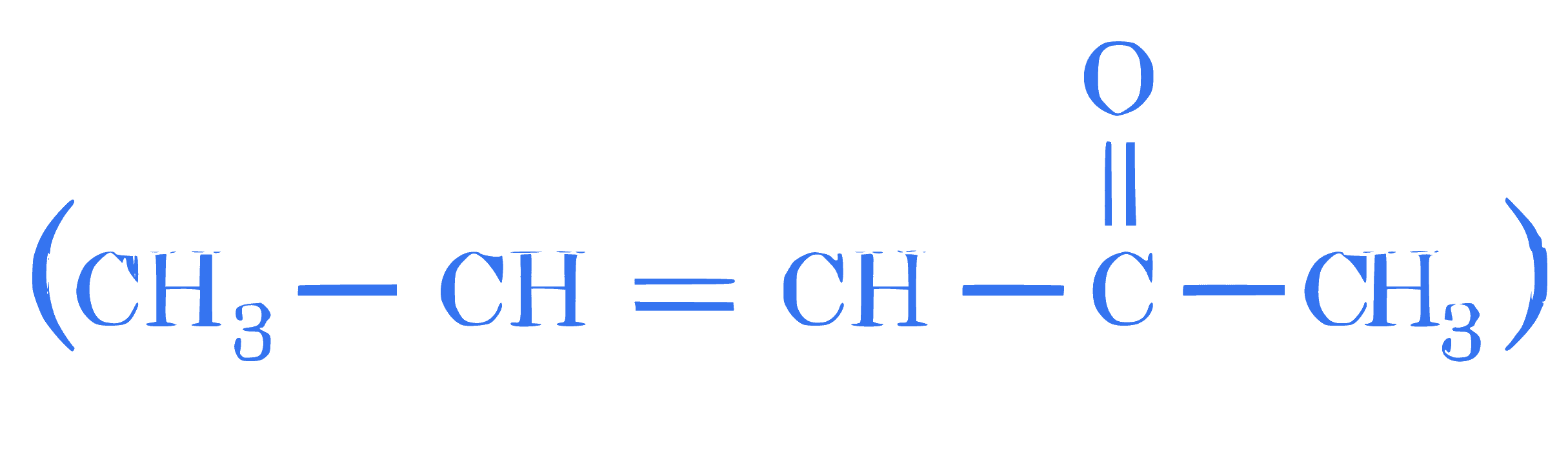 will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
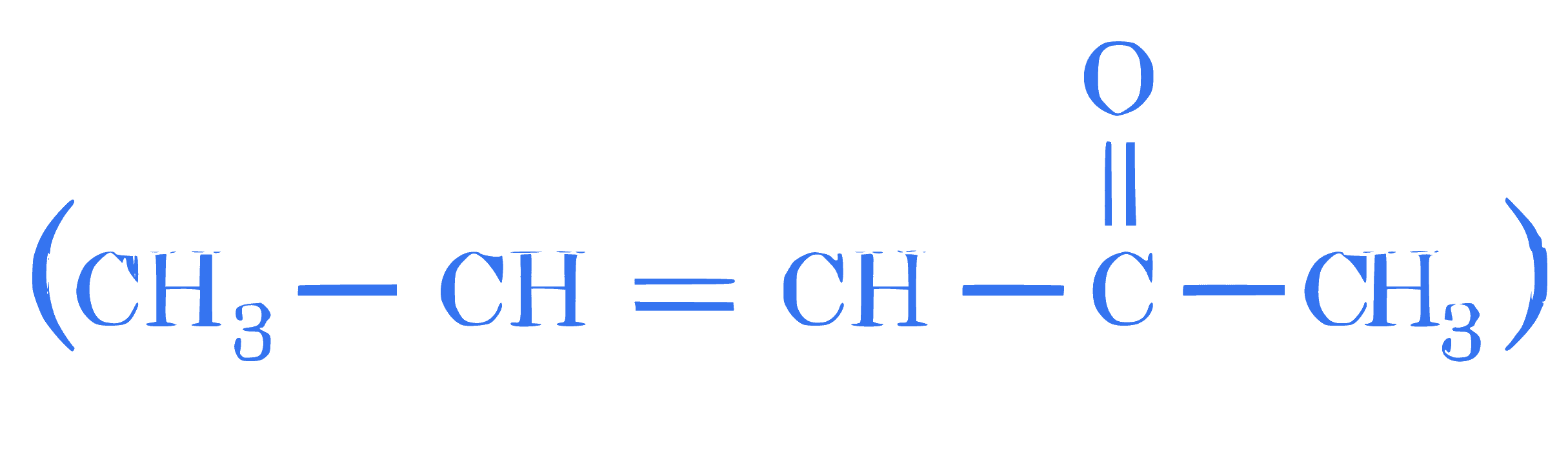 will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
Q16.
mcq single
+4 / 1
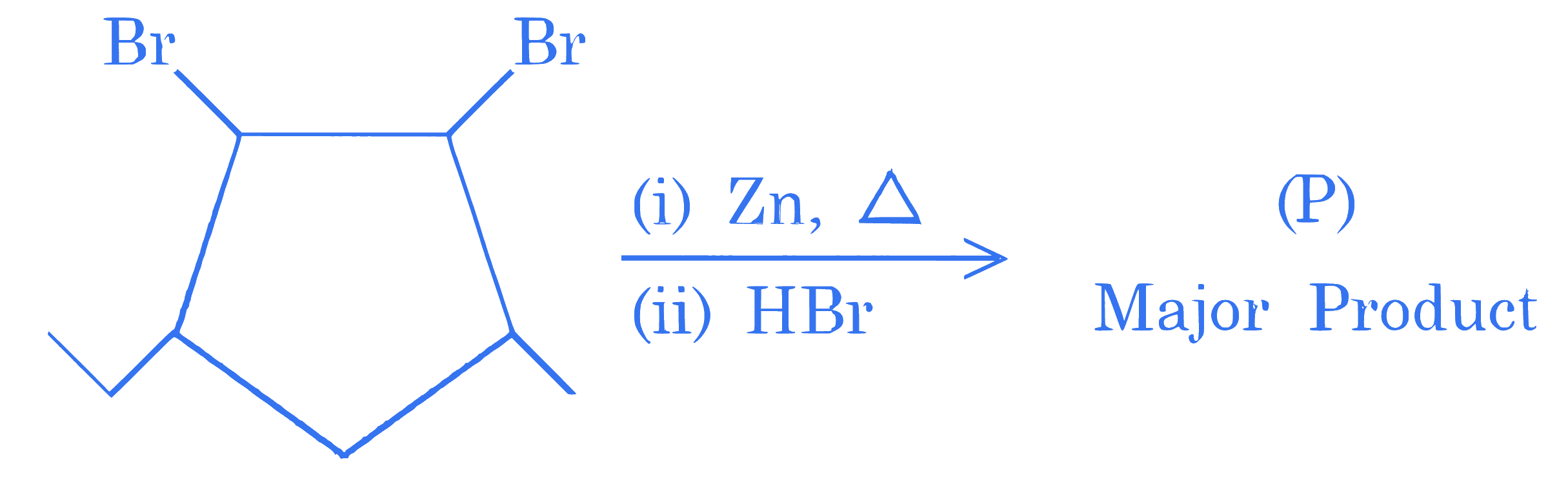 $$ \text { Identify (P) } $$
$$ \text { Identify (P) } $$
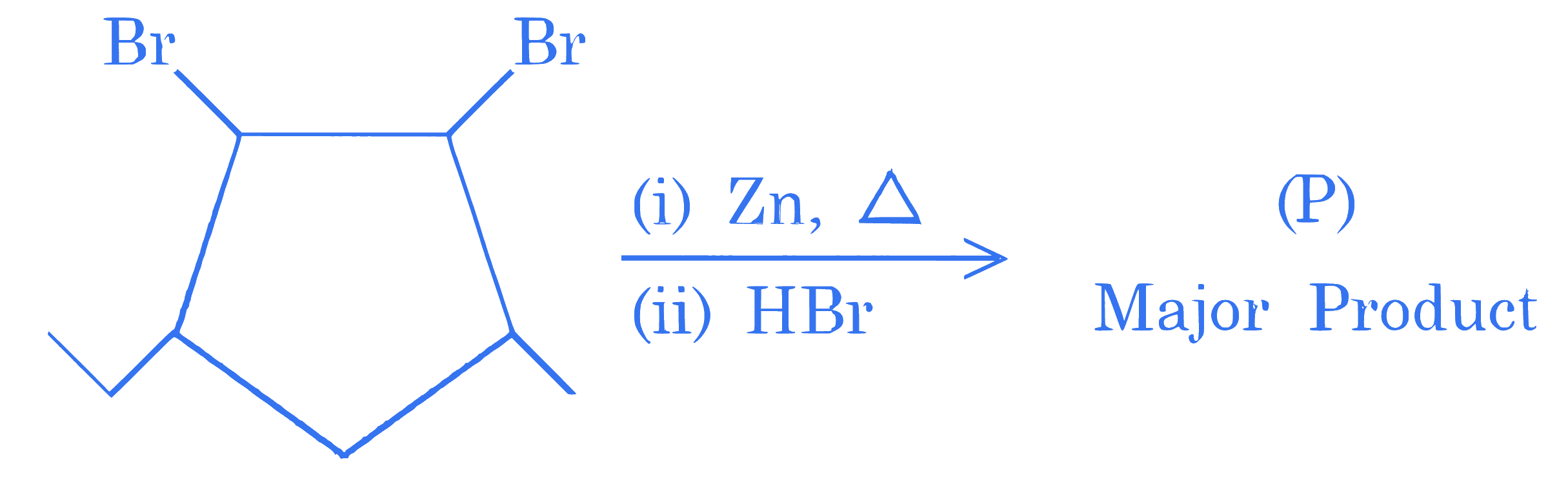 $$ \text { Identify (P) } $$
$$ \text { Identify (P) } $$
Q17.
mcq single
+4 / 1
Iodoform test can differentiate between
A. Methanol and Ethanol
B. $\mathrm{CH}_3 \mathrm{COOH}$ and $\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{COOH}$
C. Cyclohexene and cyclohexanone
D. Diethyl ether and Pentan-3-one
E. Anisole and acetone
Choose the **correct** answer from the options given below :
Q18.
mcq single
+4 / 1
In Carius method 0.2425 g of an organic compound gave 0.5253 g silver chloride. The percentage of chlorine in the organic compound is
Q19.
numerical
+4 / 1
Consider the following reaction of benzene.
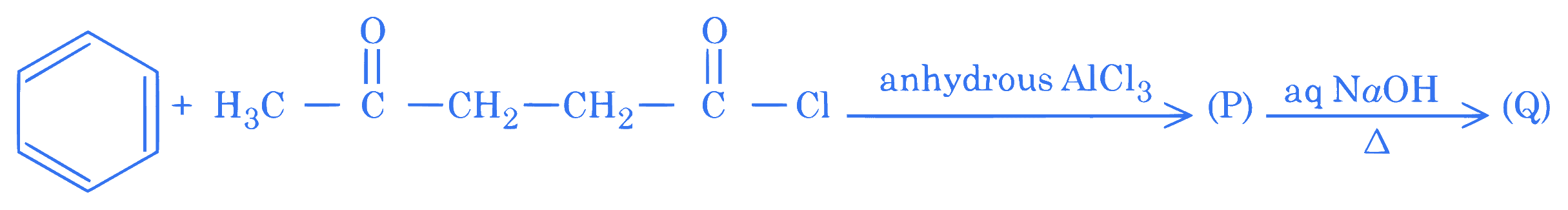 In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
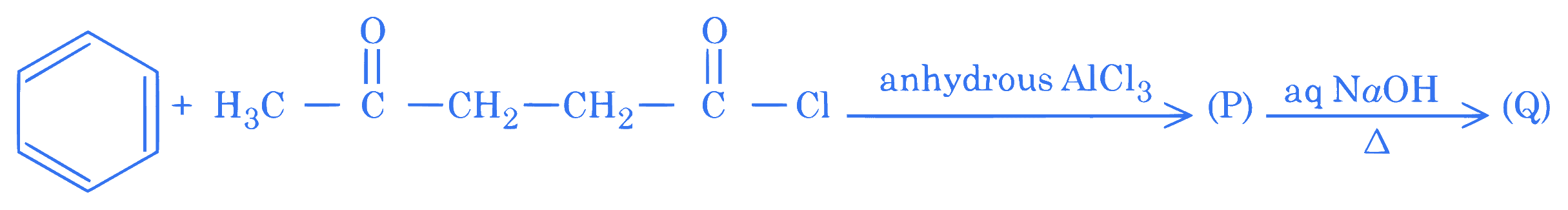 In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
Q20.
mcq single
+4 / 1
Both human DNA and RNA are chiral molecules. The chirality in DNA and RNA arises due to the presence of
Q21.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\left(\mathrm{CH}_3\right)_3 \stackrel{\oplus}{\mathrm{C}}$ is more stable than $\stackrel{\oplus}{\mathrm{C}} \mathrm{H}_3$ as nine hyperconjugation interactions are possible in $\left(\mathrm{CH}_3\right)_3 \stackrel{\oplus}{\mathrm{C}}$.
**Statement II :** $\stackrel{\oplus}{\mathrm{C}}\mathrm{H}_3$ is less stable than $\left(\mathrm{CH}_3\right)_3 \stackrel{\oplus}{\mathrm{C}}$ as only three hyperconjugation interactions are possible in $\stackrel{\oplus}{\mathrm{C}} \mathrm{H}_3$.
In the light of the above statements, choose the **correct** answer from the options given below
Q22.
mcq single
+4 / 1
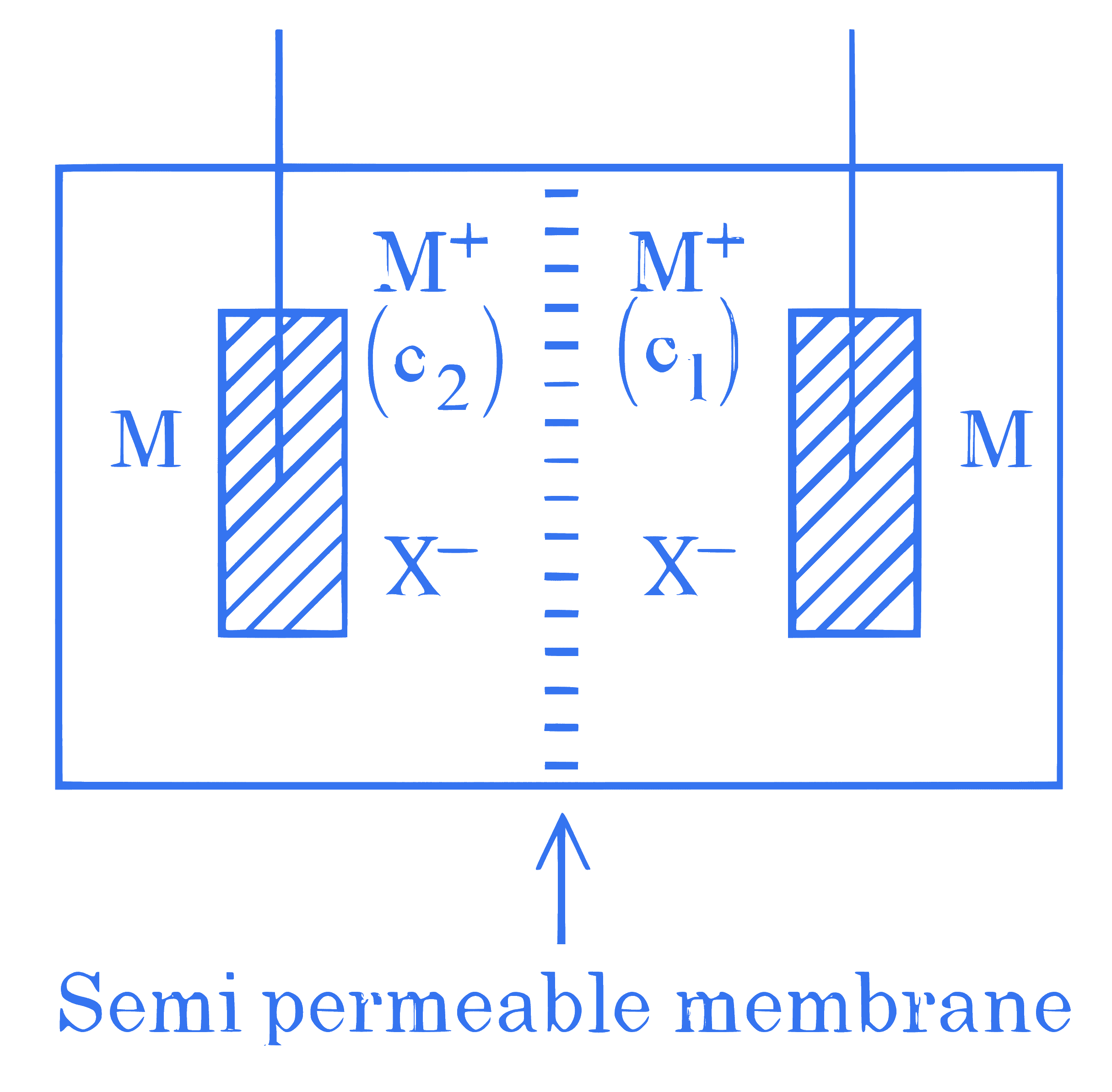 Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
 Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
Q23.
mcq single
+4 / 1
A mixed ether $(\mathrm{P})$, when heated with excess of hot concentrated hydrogen iodide produces two different alkyl iodides which when treated with aq. NaOH give compounds $(\mathrm{Q})$ and $(\mathrm{R})$. Both $(\mathrm{Q})$ and $(\mathrm{R})$ give yellow precipitate with NaOI . Identify the mixed ether $(\mathrm{P})$ :
Q24.
mcq single
+4 / 1
Identify the **INCORRECT** statements from the following :
A. Notation ${ }_{12}^{24} \mathrm{Mg}$ represents 24 protons and 12 neutrons.
B. Wavelength of a radiation of frequency $4.5 \times 10^{15} \mathrm{~s}^{-1}$ is $6.7 \times 10^{-8} \mathrm{~m}$.
C. One radiation has wavelength $=\lambda_1(900 \mathrm{~nm})$ and energy $=\mathrm{E}_1$. Other radiation has wavelength $=\lambda_2(300 \mathrm{~nm})$ and energy $=\mathrm{E}_2 \cdot \mathrm{E}_1: \mathrm{E}_2=3: 1$.
D. Number of photons of light of wavelength 2000 pm that provides 1 J of energy is $1.006 \times 10^{16}$.
Choose the correct answer from the options given below :
Q25.
mcq single
+4 / 1
The work functions of two metals $\left(\mathrm{M}_{\mathrm{A}}\right.$ and $\left.\mathrm{M}_{\mathrm{B}}\right)$ are in the $1: 2$ ratio. When these metals are exposed to photons of energy 6 eV , the kinetic energy of liberated electrons of $M_A: M_B$ is in the ratio of $2.642: 1$. The work functions (in eV ) of $M_A$ and $M_B$ are respectively.
Q26.
mcq single
+4 / 1
A student has been given a compound " $x$ " of molecular formula- $\mathrm{C}_6 \mathrm{H}_7 \mathrm{~N}$. ' $x$ ' is sparingly soluble in water. However, on addition of dilute mineral acid, ' $x$ ' becomes soluble in water. ' $x$ ' when treated with $\mathrm{CHCl}_3$ and $\mathrm{KOH}(\mathrm{alc})$, ' $y$ ' is produced. ' $y$ ' has a specific unpleasant smell. On treatment with benzenesulphonyl chloride, ' $x$ ' gives a compound ' $z$ ' which is soluble in alkali. The number of different " H " atoms present in ' z ' is:-
Q27.
mcq single
+4 / 1
$$ \text { Given below are two statements : } $$

 In the light of the above statements, choose the correct answer from the options given below
In the light of the above statements, choose the correct answer from the options given below

 In the light of the above statements, choose the correct answer from the options given below
In the light of the above statements, choose the correct answer from the options given below
Q28.
mcq single
+4 / 1
Observe the following reactions at $\mathrm{T}(\mathrm{K})$.
I. $\mathrm{A} \rightarrow$ products.
II. $5 \mathrm{Br}^{-}(\mathrm{aq})+\mathrm{BrO}_3{ }^{-}(\mathrm{aq})+6 \mathrm{H}^{+}(\mathrm{aq}) \rightarrow 3 \mathrm{Br}_2(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\mathrm{l})$
Both the reactions are started at 10.00 am . The rates of these reactions at 10.10 am are same. The value of $-\frac{\Delta\left[\mathrm{Br}^{-}\right]}{\Delta \mathrm{t}}$ at 10.10 am is $2 \times 10^{-4} \mathrm{~mol} \mathrm{~L}^{-1} \mathrm{~min}^{-1}$. The concentration of A at 10.10 am is $10^{-2} \mathrm{~mol} \mathrm{~L}^{-1}$. What is the first order rate constant (in $\mathrm{min}^{-1}$ ) of reaction $I$ ?
Q29.
mcq single
+4 / 1
 Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
 Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
Given above is the concentration vs time plot for a dissociation reaction : $\mathrm{A} \rightarrow \mathrm{nB}$.
Based on the data of the initial phase of the reaction (initial 10 min ), the value of n is $\_\_\_\_$ .
Q30.
mcq single
+4 / 1
Which statements are **NOT TRUE** about $\mathrm{XeO}_2 \mathrm{~F}_2$?
A. It has a see-saw shape.
B. Xe has 5 electron pairs in its valence shell in $\mathrm{XeO}_2 \mathrm{~F}_2$.
C. The $\mathrm{O}-\mathrm{Xe}-\mathrm{O}$ bond angle is close to $180^{\circ}$.
D. The $\mathrm{F}-\mathrm{Xe}-\mathrm{F}$ bond angle is close to $180^{\circ}$.
E. Xe has 16 valence electrons in $\mathrm{XeO}_2 \mathrm{~F}_2$.
Choose the ***correct*** answer from the options given below :
Q31.
mcq single
+4 / 1
The oxidation state of chromium in the final product formed in the reaction between KI and acidified $\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7$ solution is :
Q32.
numerical
+4 / 1
200 cc of $x \times 10^{-3} \mathrm{M}$ potassium dichromate is required to oxidise 750 cc of 0.6 M Mohr's salt solution in acidic medium.
Here $x=$ $\_\_\_\_$ .
Q33.
numerical
+4 / 1
Two liquids A and B form an ideal solution. At 320 K , the vapour pressure of the solution, containing 3 mol of $A$ and 1 mol of $B$ is 500 mm Hg . At the same temperature, if 1 mol of A is further added to this solution, vapour pressure of the solution increases by 20 mm Hg . Vapour pressure (in mm Hg ) of B in pure state is $\_\_\_\_$ . (Nearest integer)
Q34.
numerical
+4 / 1
$$ \mathrm{X}_2(\mathrm{~g})+\mathrm{Y}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{Z}(\mathrm{~g}) $$
$\mathrm{X}_2(\mathrm{~g})$ and $\mathrm{Y}_2(\mathrm{~g})$ are added to a 1 L flask and it is found that the system attains the above equilibrium at $\mathrm{T}(\mathrm{K})$ with the number of moles of $\mathrm{X}_2(\mathrm{~g}), \mathrm{Y}_2(\mathrm{~g})$ and $\mathrm{Z}(\mathrm{g})$ being 3,3 and 9 mol respectively (equilibrium moles). Under this condition of equilibrium, 10 mol of $\mathrm{Z}(\mathrm{g})$ is added to the flask and the temperature is maintained at $\mathrm{T}(\mathrm{K})$. Then the number of moles of $\mathrm{Z}(\mathrm{g})$ in the flask when the new equilibrium is established is $\_\_\_\_$ . (Nearest integer)
Q35.
mcq single
+4 / 1
Elements X and Y belong to Group 15. The difference between the electronegativity values of ' X ' and phosphorus is higher than that of the difference between phosphorus and ' Y '. ' X ' & ' Y ' are respectively
Q36.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The second ionisation enthalpy of Na is larger than the corresponding ionisation enthalpy of Mg .
**Statement II :** The ionic radius of $\mathrm{O}^{2-}$ is larger than that of $\mathrm{F}^{-}$.
In the light of the above statements, choose the ***correct*** answer from the options given below :
Q37.
mcq single
+4 / 1
It is noticed that $\mathrm{Pb}^{2+}$ is more stable than $\mathrm{Pb}^{4+}$ but $\mathrm{Sn}^{2+}$ is less stable than $\mathrm{Sn}^{4+}$. Observe the following reactions.
$$ \begin{aligned} & \mathrm{PbO}_2+\mathrm{Pb} \rightarrow 2 \mathrm{PbO} ; \Delta_{\mathrm{r}} \mathrm{G}^{\circ}(1) \\ & \mathrm{SnO}_2+\mathrm{Sn} \rightarrow 2 \mathrm{SnO} ; \Delta_{\mathrm{r}} \mathrm{G}^{\circ}(2) \end{aligned} $$
Identify the correct set from the following
Q38.
mcq single
+4 / 1
Identify the CORRECT set of details from the following :
A. $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]^{3+}$ : Inner orbital complex; $\mathrm{d}^2 \mathrm{sp}^3$ hybridized
B. $\left[\mathrm{MnCl}_6\right]^{3-}$ : Outer orbital complex; $\mathrm{sp}^3 \mathrm{~d}^2$ hybridized
C. $\left[\mathrm{CoF}_6\right]^{3-}$ : Outer orbital complex; $\mathrm{d}^2 \mathrm{sp}^3$ hybridized
D. $\left[\mathrm{FeF}_6\right]^{3-}$ : Outer orbital complex; $\mathrm{sp}^3 \mathrm{~d}^2$ hybridized
E. $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}$ : Inner orbital complex; $\mathrm{sp}^3$ hybridized
Choose the correct answer from the options given below :
Q39.
numerical
+4 / 1
Total number of unpaired electrons present in the central metal atoms/ions of
$\left[\mathrm{Ni}(\mathrm{CO})_4\right],\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{PtCl}_2\left(\mathrm{NH}_3\right)_2\right],\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}$ and $\left[\mathrm{Pt}(\mathrm{CN})_4\right]^{2-}$ is $\_\_\_\_$。
Q40.
mcq single
+4 / 1
Which of the following statements are TRUE about Haloform reaction?
A. Sodium hypochlorite reacts with KI to give KOI .
B. KOI is a reducing agent.
C. $\alpha, \beta$-unsaturated methylketone 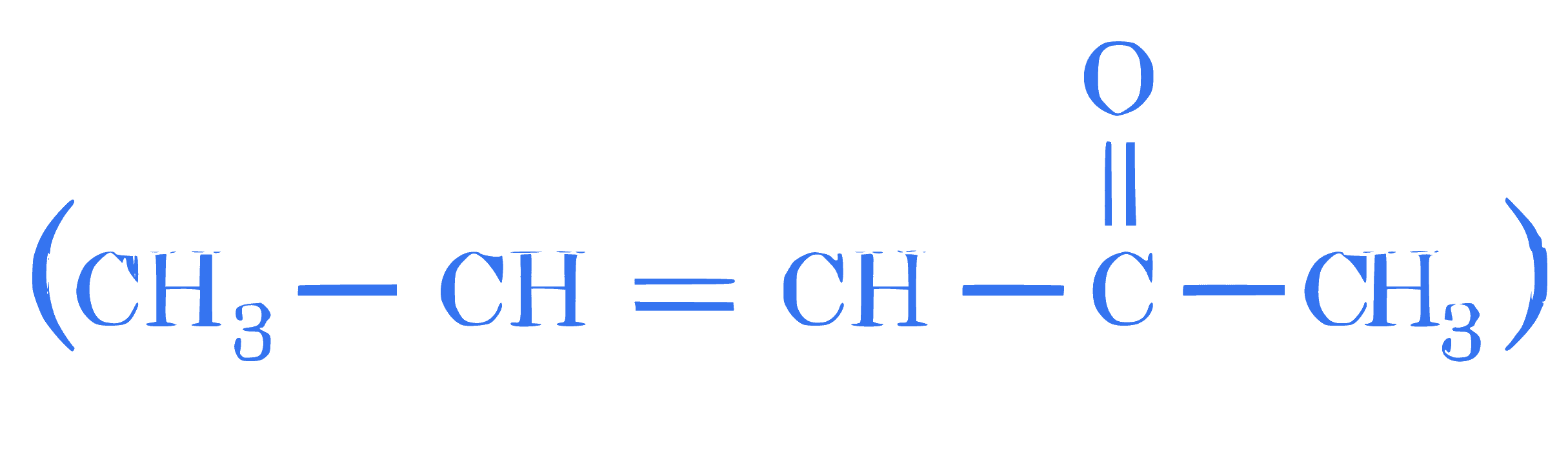 will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
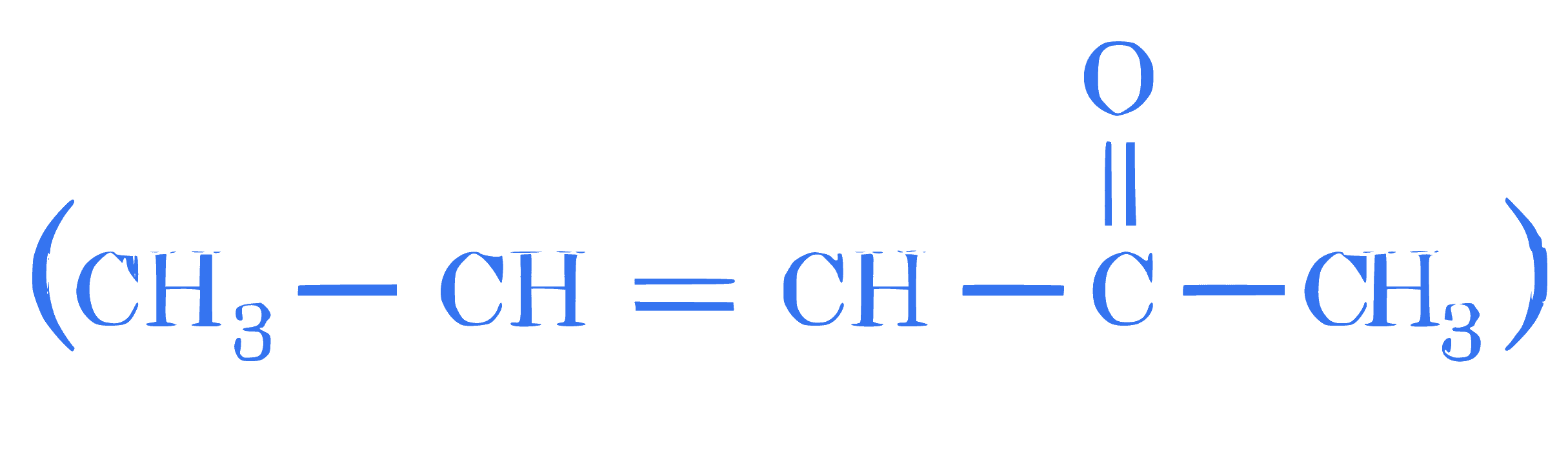 will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
will give
iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below :
Q41.
mcq single
+4 / 1
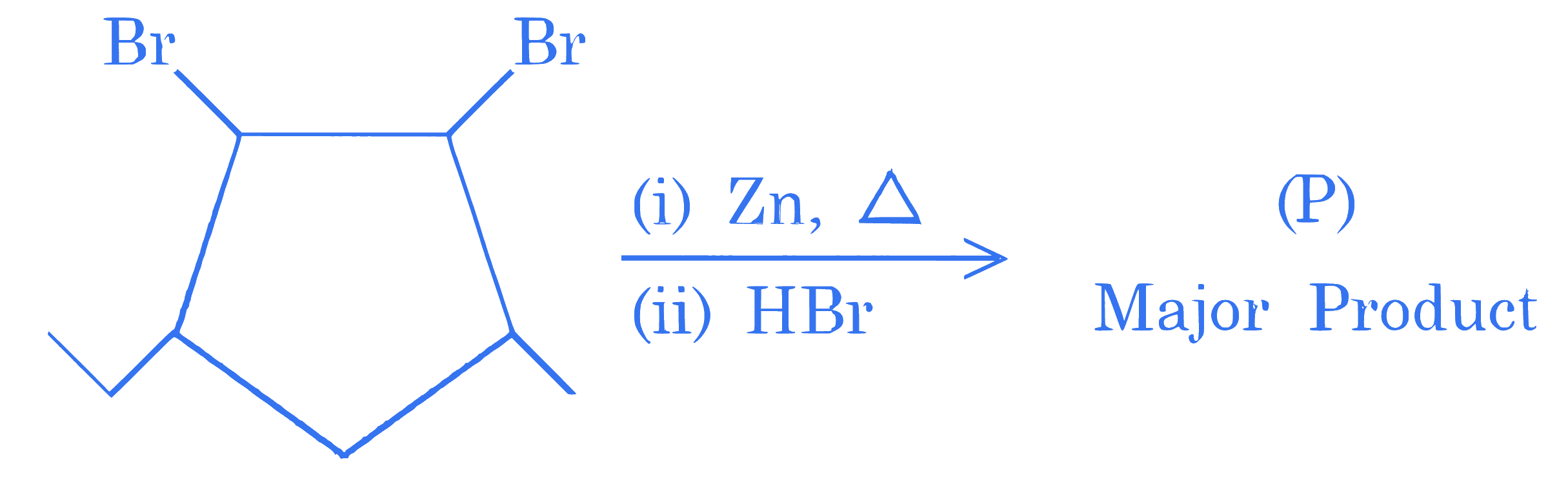 $$ \text { Identify (P) } $$
$$ \text { Identify (P) } $$
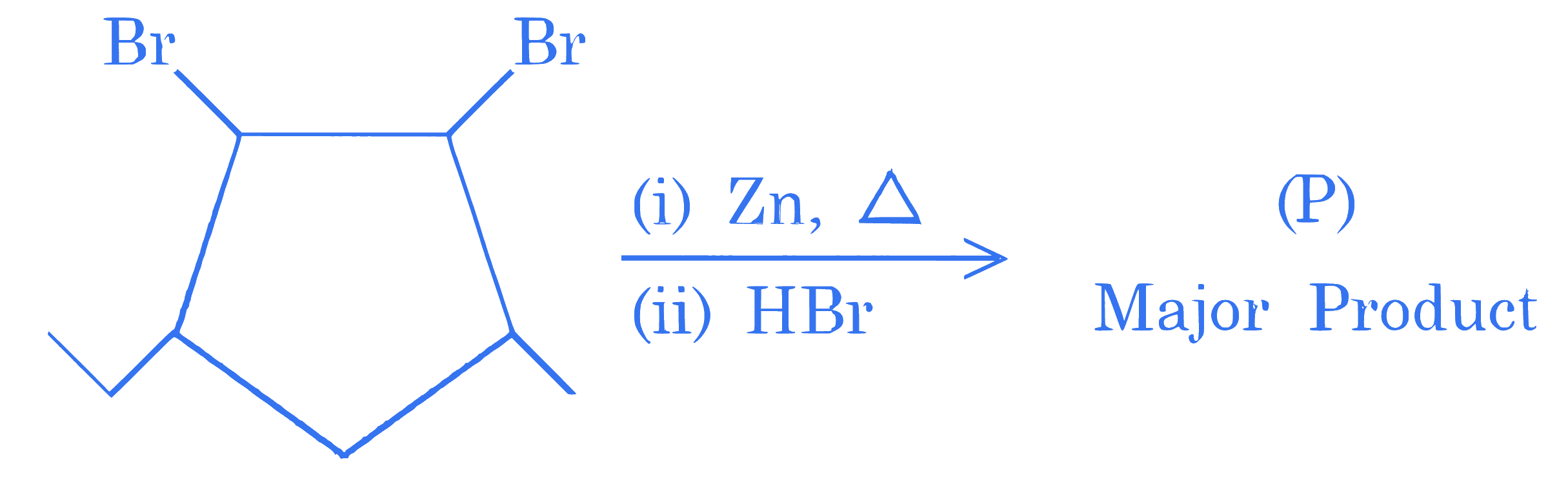 $$ \text { Identify (P) } $$
$$ \text { Identify (P) } $$
Q42.
mcq single
+4 / 1
Iodoform test can differentiate between
A. Methanol and Ethanol
B. $\mathrm{CH}_3 \mathrm{COOH}$ and $\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{COOH}$
C. Cyclohexene and cyclohexanone
D. Diethyl ether and Pentan-3-one
E. Anisole and acetone
Choose the **correct** answer from the options given below :
Q43.
mcq single
+4 / 1
In Carius method 0.2425 g of an organic compound gave 0.5253 g silver chloride. The percentage of chlorine in the organic compound is
Q44.
numerical
+4 / 1
Consider the following reaction of benzene.
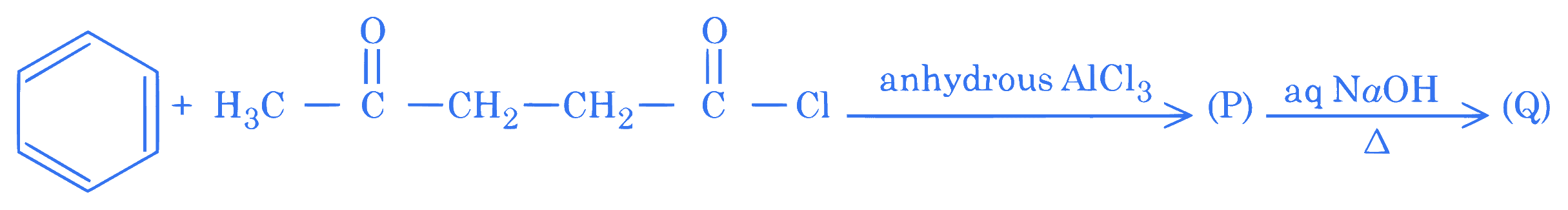 In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
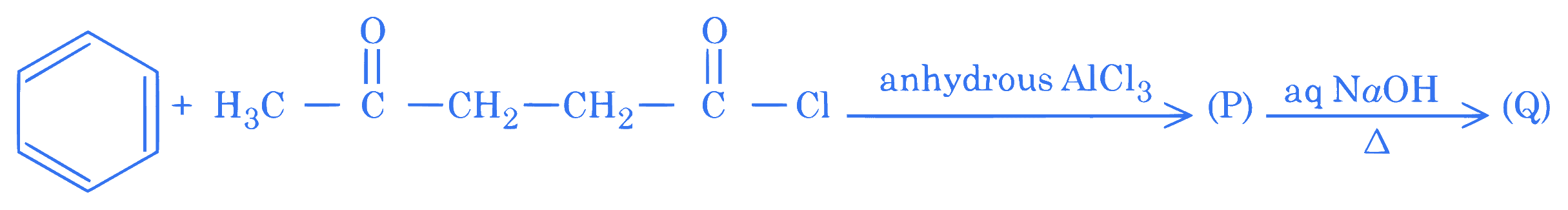 In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
In compound $(Q)$, the percentage of oxygen is $\_\_\_\_$ %. (Nearest integer)
Q45.
mcq single
+4 / 1
Both human DNA and RNA are chiral molecules. The chirality in DNA and RNA arises due to the presence of
Q46.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\left(\mathrm{CH}_3\right)_3 \stackrel{\oplus}{\mathrm{C}}$ is more stable than $\stackrel{\oplus}{\mathrm{C}} \mathrm{H}_3$ as nine hyperconjugation interactions are possible in $\left(\mathrm{CH}_3\right)_3 \stackrel{\oplus}{\mathrm{C}}$.
**Statement II :** $\stackrel{\oplus}{\mathrm{C}}\mathrm{H}_3$ is less stable than $\left(\mathrm{CH}_3\right)_3 \stackrel{\oplus}{\mathrm{C}}$ as only three hyperconjugation interactions are possible in $\stackrel{\oplus}{\mathrm{C}} \mathrm{H}_3$.
In the light of the above statements, choose the **correct** answer from the options given below
Q47.
mcq single
+4 / 1
 Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
 Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
Consider the above electrochemical cell where a metal electrode ( M ) is undergoing redox reaction by forming $\mathrm{M}^{+}\left(\mathrm{M} \rightarrow \mathrm{M}^{+}+\mathrm{e}^{-}\right)$. The cation $\mathrm{M}^{+}$is present in two different concentrations $c_1$ and $c_2$ as shown above. Which of the following statement is correct for generating a positive cell potential?
Q48.
mcq single
+4 / 1
A mixed ether $(\mathrm{P})$, when heated with excess of hot concentrated hydrogen iodide produces two different alkyl iodides which when treated with aq. NaOH give compounds $(\mathrm{Q})$ and $(\mathrm{R})$. Both $(\mathrm{Q})$ and $(\mathrm{R})$ give yellow precipitate with NaOI . Identify the mixed ether $(\mathrm{P})$ :
Q49.
mcq single
+4 / 1
Identify the **INCORRECT** statements from the following :
A. Notation ${ }_{12}^{24} \mathrm{Mg}$ represents 24 protons and 12 neutrons.
B. Wavelength of a radiation of frequency $4.5 \times 10^{15} \mathrm{~s}^{-1}$ is $6.7 \times 10^{-8} \mathrm{~m}$.
C. One radiation has wavelength $=\lambda_1(900 \mathrm{~nm})$ and energy $=\mathrm{E}_1$. Other radiation has wavelength $=\lambda_2(300 \mathrm{~nm})$ and energy $=\mathrm{E}_2 \cdot \mathrm{E}_1: \mathrm{E}_2=3: 1$.
D. Number of photons of light of wavelength 2000 pm that provides 1 J of energy is $1.006 \times 10^{16}$.
Choose the correct answer from the options given below :
Q50.
mcq single
+4 / 1
The work functions of two metals $\left(\mathrm{M}_{\mathrm{A}}\right.$ and $\left.\mathrm{M}_{\mathrm{B}}\right)$ are in the $1: 2$ ratio. When these metals are exposed to photons of energy 6 eV , the kinetic energy of liberated electrons of $M_A: M_B$ is in the ratio of $2.642: 1$. The work functions (in eV ) of $M_A$ and $M_B$ are respectively.
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
If the mean and the variance of the data
$$ \begin{array}{|c|c|c|c|c|} \hline \text { Class } & 4-8 & 8-12 & 12-16 & 16-20 \\ \hline \text { Frequency } & 3 & \lambda & 4 & 7 \\ \hline \end{array} $$
are $\mu$ and 19 respectively, then the value of $\lambda+\mu$ is :
Q2.
mcq single
+4 / 1
Let $\vec{a}, \vec{b}, \vec{c}$ be three vectors such that $\vec{a} \times \vec{b}=2(\vec{a} \times \vec{c})$. If $|\vec{a}|=1,|\vec{b}|=4,|\vec{c}|=2$, and the angle between $\vec{b}$ and $\vec{c}$ is $60^{\circ}$, then $|\vec{a} \cdot \vec{c}|$ is equal to
Q3.
mcq single
+4 / 1
Let $\vec{a}=\hat{\mathrm{i}}-2 \hat{\mathrm{j}}+3 \hat{\mathrm{k}}, \vec{b}=2 \hat{\mathrm{i}}+\hat{\mathrm{j}}-\hat{\mathrm{k}}, \vec{c}=\lambda \hat{\mathrm{i}}+\hat{\mathrm{j}}+\hat{\mathrm{k}}$ and $\vec{v}=\vec{a} \times \vec{b}$. If $\vec{v} \cdot \vec{c}=11$ and the length of the projection of $\vec{b}$ on $\vec{c}$ is $p$, then $9 p^2$ is equal to
Q4.
mcq single
+4 / 1
Bag A contains 9 white and 8 black balls, while bag B contains 6 white and 4 black balls. One ball is randomly picked up from the bag B and mixed up with the balls in the bag A . Then a ball is randomly drawn from the bag A . If the probability, that the ball drawn is white, is $\frac{\mathrm{p}}{\mathrm{q}}, \operatorname{gcd}(\mathrm{p}, \mathrm{q})=1$, then $\mathrm{p}+\mathrm{q}$ is equal to
Q5.
numerical
+4 / 1
If the image of the point $\mathrm{P}(a, 2, a)$ in the line $\frac{x}{2}=\frac{y+a}{1}=\frac{z}{1}$ is Q and the image
of Q in the line $\frac{x-2 b}{2}=\frac{y-a}{1}=\frac{z+2 b}{-5}$ is P , then $a+b$ is equal to $\_\_\_\_$ .
Q6.
mcq single
+4 / 1
Let $\sum\limits_{k=1}^n a_k=\alpha n^2+\beta n$. If $a_{10}=59$ and $a_6=7 a_1$, then $\alpha+\beta$ is equal to :
Q7.
mcq single
+4 / 1
If $f(x)=\left\{\begin{array}{cc}\frac{a|x|+x^2-2(\sin |x|)(\cos |x|)}{x} & , x \neq 0 \\ b & , x=0\end{array}\right.$
is continuous at $x=0$, then $a+b$ is equal to :
Q8.
mcq single
+4 / 1
The least value of $\left(\cos ^2 \theta-6 \sin \theta \cos \theta+3 \sin ^2 \theta+2\right)$ is
Q9.
mcq single
+4 / 1
Let $\frac{\pi}{2}<\theta<\pi$ and $\cot \theta=-\frac{1}{2 \sqrt{2}}$. Then the value of
$$ \sin \left(\frac{15 \theta}{2}\right)(\cos 8 \theta+\sin 8 \theta)+\cos \left(\frac{15 \theta}{2}\right)(\cos 8 \theta-\sin 8 \theta) $$
is equal to :
Q10.
mcq single
+4 / 1
The area of the region enclosed between the circles $x^2+y^2=4$ and $x^2+(y-2)^2=4$ is:
Q11.
mcq single
+4 / 1
The system of linear equations
$$ \begin{aligned} & x+y+z=6 \\ & 2 x+5 y+a z=36 \\ & x+2 y+3 z=b \end{aligned} $$
has :
Q12.
numerical
+4 / 1
Let $A=\left[\begin{array}{ccc}0 & 2 & -3 \\ -2 & 0 & 1 \\ 3 & -1 & 0\end{array}\right]$ and $B$ be a matrix such that $B(I-A)=I+A$. Then the sum of the diagonal elements of $\mathrm{B}^{\mathrm{T}} \mathrm{B}$ is equal to $\_\_\_\_$
Q13.
mcq single
+4 / 1
Let $\mathrm{I}(x)=\int \frac{3 d x}{(4 x+6)\left(\sqrt{4 x^2+8 x+3}\right)}$ and $\mathrm{I}(0)=\frac{\sqrt{3}}{4}+20$. If
$\mathrm{I}\left(\frac{1}{2}\right)=\frac{a \sqrt{2}}{b}+\mathrm{c}$, where $a, b, \mathrm{c} \in \mathrm{N}, \operatorname{gcd}(a, b)=1$, then $a+b+c$ is equal to :
Q14.
mcq single
+4 / 1
An equilateral triangle OAB is inscribed in the parabola $y^2=4 x$ with the vertex O at the vertex of the parabola. Then the minimum distance of the circle having $A B$ as a diameter from the origin is
Q15.
mcq single
+4 / 1
Let $\mathrm{A}(1,2)$ and $\mathrm{C}(-3,-6)$ be two diagonally opposite vertices of a rhombus, whose sides AD and BC are parallel to the line $7 x-y=14$. If $\mathrm{B}(\alpha, \beta)$ and $\mathrm{D}(\gamma, \delta)$ are the other two vertices, then $|\alpha+\beta+\gamma+\delta|$ is equal to :
Q16.
mcq single
+4 / 1
Let $\mathrm{A}=\{0,1,2, \ldots, 9\}$. Let R be a relation on A defined by $(x, y) \in \mathrm{R}$ if and only if $|x-y|$ is a multiple of 3.
Given below are two statements :
**Statement I :** $n(\mathrm{R})=36$.
**Statement II :** R is an equivalence relation.
In the light of the above statements, choose the ***correct*** answer from the options given below :
Q17.
mcq single
+4 / 1
Consider two sets $\mathrm{A}=\{x \in \mathrm{Z}:|(|x-3|-3)| \leq 1\}$ and
$\mathrm{B}=\left\{x \in \mathbb{R}-\{1,2\}: \frac{(x-2)(x-4)}{x-1} \log _e(|x-2|)=0\right\}$. Then the number of
onto functions $f: \mathrm{A} \rightarrow \mathrm{B}$ is equal to :
Q18.
mcq single
+4 / 1
If $z=\frac{\sqrt{3}}{2}+\frac{i}{2}, i=\sqrt{-1}$, then $\left(z^{201}-i\right)^8$ is equal to
Q19.
mcq single
+4 / 1
Let PQ be a chord of the hyperbola $\frac{x^2}{4}-\frac{y^2}{b^2}=1$, perpendicular to the x -axis such that OPQ is an equilateral triangle, O being the centre of the hyperbola. If the eccentricity of the hyperbola is $\sqrt{3}$, then the area of the triangle OPQ is
Q20.
numerical
+4 / 1
If the solution curve $y=f(x)$ of the differential equation
$$ \left(x^2-4\right) y^{\prime}-2 x y+2 x\left(4-x^2\right)^2=0, x>2, $$
passes through the point $(3,15)$, then the local maximum value of $f$ is $\_\_\_\_$
Q21.
mcq single
+4 / 1
If the points of intersection of the ellipses $x^2+2 y^2-6 x-12 y+23=0$ and
$4 x^2+2 y^2-20 x-12 y+35=0$ lie on a circle of radius $r$ and centre $(a, b)$, then the
value of $a b+18 r^2$ is :
Q22.
numerical
+4 / 1
Let S denote the set of 4-digit numbers $a b c d$ such that $a>b>c>d$ and P denote the set of 5 -digit numbers having product of its digits equal to 20 . Then $n(\mathrm{~S})+n(\mathrm{P})$ is equal to $\_\_\_\_$
Q23.
mcq single
+4 / 1
The number of ways, in which 16 oranges can be distributed to four children such that each child gets at least one orange, is
Q24.
mcq single
+4 / 1
The sum of all the real solutions of the equation $\log _{(x+3)}\left(6 x^2+28 x+30\right)=5-2 \log _{(6 x+10)}\left(x^2+6 x+9\right)$ is equal to :
Q25.
numerical
+4 / 1
The number of elements in the set $\mathrm{S}=\left\{x: x \in[0,100]\right.$ and $\left.\int\limits_0^x t^2 \sin (x-t) \mathrm{d} t=x^2\right\}$ is $\_\_\_\_$
Q26.
mcq single
+4 / 1
If the mean and the variance of the data
$$ \begin{array}{|c|c|c|c|c|} \hline \text { Class } & 4-8 & 8-12 & 12-16 & 16-20 \\ \hline \text { Frequency } & 3 & \lambda & 4 & 7 \\ \hline \end{array} $$
are $\mu$ and 19 respectively, then the value of $\lambda+\mu$ is :
Q27.
mcq single
+4 / 1
Let $\vec{a}, \vec{b}, \vec{c}$ be three vectors such that $\vec{a} \times \vec{b}=2(\vec{a} \times \vec{c})$. If $|\vec{a}|=1,|\vec{b}|=4,|\vec{c}|=2$, and the angle between $\vec{b}$ and $\vec{c}$ is $60^{\circ}$, then $|\vec{a} \cdot \vec{c}|$ is equal to
Q28.
mcq single
+4 / 1
Let $\vec{a}=\hat{\mathrm{i}}-2 \hat{\mathrm{j}}+3 \hat{\mathrm{k}}, \vec{b}=2 \hat{\mathrm{i}}+\hat{\mathrm{j}}-\hat{\mathrm{k}}, \vec{c}=\lambda \hat{\mathrm{i}}+\hat{\mathrm{j}}+\hat{\mathrm{k}}$ and $\vec{v}=\vec{a} \times \vec{b}$. If $\vec{v} \cdot \vec{c}=11$ and the length of the projection of $\vec{b}$ on $\vec{c}$ is $p$, then $9 p^2$ is equal to
Q29.
mcq single
+4 / 1
Bag A contains 9 white and 8 black balls, while bag B contains 6 white and 4 black balls. One ball is randomly picked up from the bag B and mixed up with the balls in the bag A . Then a ball is randomly drawn from the bag A . If the probability, that the ball drawn is white, is $\frac{\mathrm{p}}{\mathrm{q}}, \operatorname{gcd}(\mathrm{p}, \mathrm{q})=1$, then $\mathrm{p}+\mathrm{q}$ is equal to
Q30.
numerical
+4 / 1
If the image of the point $\mathrm{P}(a, 2, a)$ in the line $\frac{x}{2}=\frac{y+a}{1}=\frac{z}{1}$ is Q and the image
of Q in the line $\frac{x-2 b}{2}=\frac{y-a}{1}=\frac{z+2 b}{-5}$ is P , then $a+b$ is equal to $\_\_\_\_$ .
Q31.
mcq single
+4 / 1
Let $\sum\limits_{k=1}^n a_k=\alpha n^2+\beta n$. If $a_{10}=59$ and $a_6=7 a_1$, then $\alpha+\beta$ is equal to :
Q32.
mcq single
+4 / 1
If $f(x)=\left\{\begin{array}{cc}\frac{a|x|+x^2-2(\sin |x|)(\cos |x|)}{x} & , x \neq 0 \\ b & , x=0\end{array}\right.$
is continuous at $x=0$, then $a+b$ is equal to :
Q33.
mcq single
+4 / 1
The least value of $\left(\cos ^2 \theta-6 \sin \theta \cos \theta+3 \sin ^2 \theta+2\right)$ is
Q34.
mcq single
+4 / 1
Let $\frac{\pi}{2}<\theta<\pi$ and $\cot \theta=-\frac{1}{2 \sqrt{2}}$. Then the value of
$$ \sin \left(\frac{15 \theta}{2}\right)(\cos 8 \theta+\sin 8 \theta)+\cos \left(\frac{15 \theta}{2}\right)(\cos 8 \theta-\sin 8 \theta) $$
is equal to :
Q35.
mcq single
+4 / 1
The area of the region enclosed between the circles $x^2+y^2=4$ and $x^2+(y-2)^2=4$ is:
Q36.
mcq single
+4 / 1
The system of linear equations
$$ \begin{aligned} & x+y+z=6 \\ & 2 x+5 y+a z=36 \\ & x+2 y+3 z=b \end{aligned} $$
has :
Q37.
numerical
+4 / 1
Let $A=\left[\begin{array}{ccc}0 & 2 & -3 \\ -2 & 0 & 1 \\ 3 & -1 & 0\end{array}\right]$ and $B$ be a matrix such that $B(I-A)=I+A$. Then the sum of the diagonal elements of $\mathrm{B}^{\mathrm{T}} \mathrm{B}$ is equal to $\_\_\_\_$
Q38.
mcq single
+4 / 1
Let $\mathrm{I}(x)=\int \frac{3 d x}{(4 x+6)\left(\sqrt{4 x^2+8 x+3}\right)}$ and $\mathrm{I}(0)=\frac{\sqrt{3}}{4}+20$. If
$\mathrm{I}\left(\frac{1}{2}\right)=\frac{a \sqrt{2}}{b}+\mathrm{c}$, where $a, b, \mathrm{c} \in \mathrm{N}, \operatorname{gcd}(a, b)=1$, then $a+b+c$ is equal to :
Q39.
mcq single
+4 / 1
An equilateral triangle OAB is inscribed in the parabola $y^2=4 x$ with the vertex O at the vertex of the parabola. Then the minimum distance of the circle having $A B$ as a diameter from the origin is
Q40.
mcq single
+4 / 1
Let $\mathrm{A}(1,2)$ and $\mathrm{C}(-3,-6)$ be two diagonally opposite vertices of a rhombus, whose sides AD and BC are parallel to the line $7 x-y=14$. If $\mathrm{B}(\alpha, \beta)$ and $\mathrm{D}(\gamma, \delta)$ are the other two vertices, then $|\alpha+\beta+\gamma+\delta|$ is equal to :
Q41.
mcq single
+4 / 1
Let $\mathrm{A}=\{0,1,2, \ldots, 9\}$. Let R be a relation on A defined by $(x, y) \in \mathrm{R}$ if and only if $|x-y|$ is a multiple of 3.
Given below are two statements :
**Statement I :** $n(\mathrm{R})=36$.
**Statement II :** R is an equivalence relation.
In the light of the above statements, choose the ***correct*** answer from the options given below :
Q42.
mcq single
+4 / 1
Consider two sets $\mathrm{A}=\{x \in \mathrm{Z}:|(|x-3|-3)| \leq 1\}$ and
$\mathrm{B}=\left\{x \in \mathbb{R}-\{1,2\}: \frac{(x-2)(x-4)}{x-1} \log _e(|x-2|)=0\right\}$. Then the number of
onto functions $f: \mathrm{A} \rightarrow \mathrm{B}$ is equal to :
Q43.
mcq single
+4 / 1
If $z=\frac{\sqrt{3}}{2}+\frac{i}{2}, i=\sqrt{-1}$, then $\left(z^{201}-i\right)^8$ is equal to
Q44.
mcq single
+4 / 1
Let PQ be a chord of the hyperbola $\frac{x^2}{4}-\frac{y^2}{b^2}=1$, perpendicular to the x -axis such that OPQ is an equilateral triangle, O being the centre of the hyperbola. If the eccentricity of the hyperbola is $\sqrt{3}$, then the area of the triangle OPQ is
Q45.
numerical
+4 / 1
If the solution curve $y=f(x)$ of the differential equation
$$ \left(x^2-4\right) y^{\prime}-2 x y+2 x\left(4-x^2\right)^2=0, x>2, $$
passes through the point $(3,15)$, then the local maximum value of $f$ is $\_\_\_\_$
Q46.
mcq single
+4 / 1
If the points of intersection of the ellipses $x^2+2 y^2-6 x-12 y+23=0$ and
$4 x^2+2 y^2-20 x-12 y+35=0$ lie on a circle of radius $r$ and centre $(a, b)$, then the
value of $a b+18 r^2$ is :
Q47.
numerical
+4 / 1
Let S denote the set of 4-digit numbers $a b c d$ such that $a>b>c>d$ and P denote the set of 5 -digit numbers having product of its digits equal to 20 . Then $n(\mathrm{~S})+n(\mathrm{P})$ is equal to $\_\_\_\_$
Q48.
mcq single
+4 / 1
The number of ways, in which 16 oranges can be distributed to four children such that each child gets at least one orange, is
Q49.
mcq single
+4 / 1
The sum of all the real solutions of the equation $\log _{(x+3)}\left(6 x^2+28 x+30\right)=5-2 \log _{(6 x+10)}\left(x^2+6 x+9\right)$ is equal to :
Q50.
numerical
+4 / 1
The number of elements in the set $\mathrm{S}=\left\{x: x \in[0,100]\right.$ and $\left.\int\limits_0^x t^2 \sin (x-t) \mathrm{d} t=x^2\right\}$ is $\_\_\_\_$
Physics
Physics
Q1.
mcq single
+4 / 1
A bead $P$ sliding on a frictionless semi-circular string $(A C B)$ and it is at point $S$ at $t =0$ and at this instant the horizontal component of its velocity is $v$. Another bead $Q$ of the same mass as $P$ is ejected from point $A$ at $t=0$ along the horizontal string $A B$, with the speed $v$, friction between the beads and the respective strings may be neglected in both cases. Let $t_P$ and $t_Q$ be the respective times taken by beads $P$ and $Q$ to reach the point $B$, then the relation between $t_P$ and $t_Q$ is
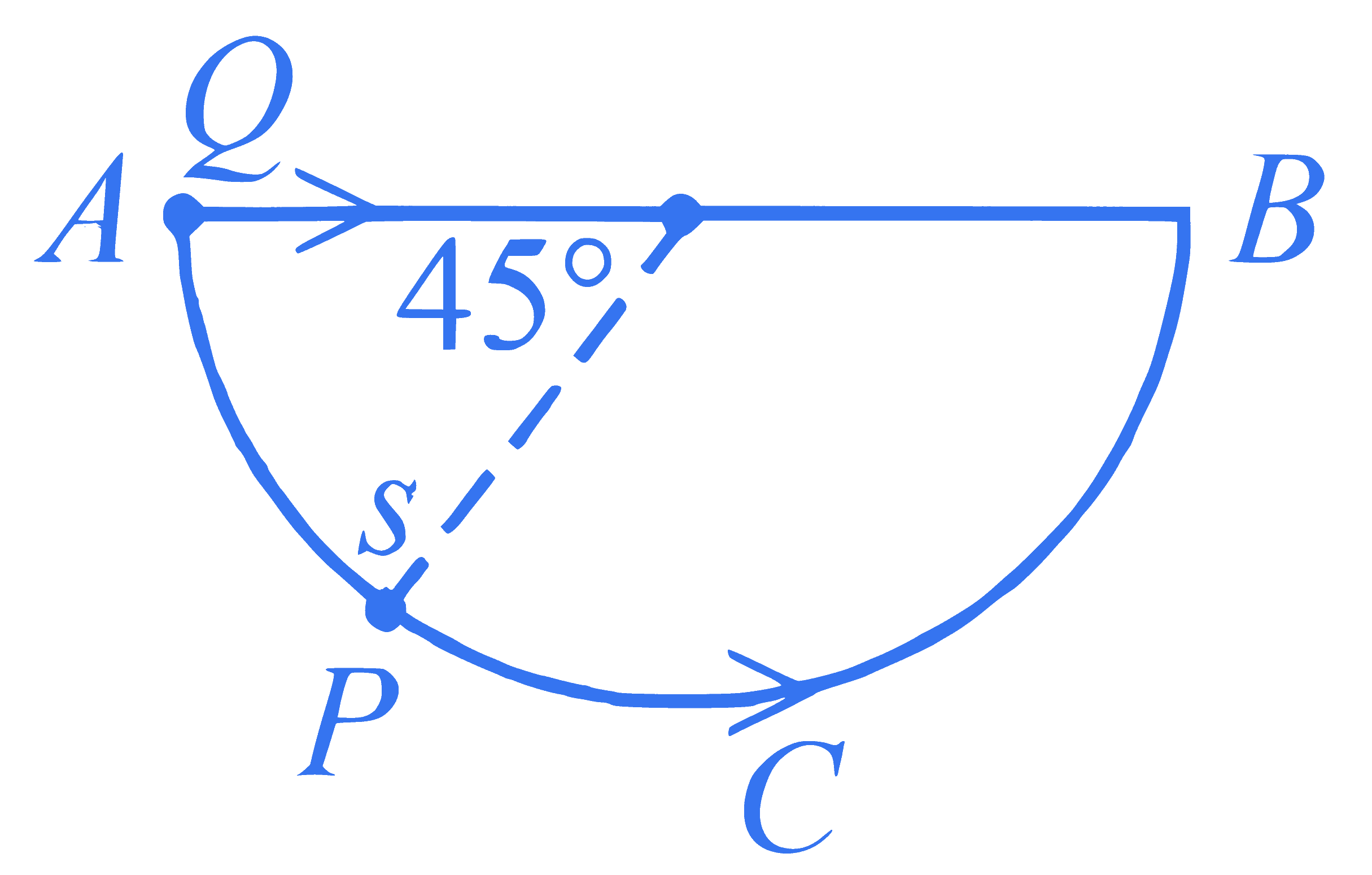

Q2.
mcq single
+4 / 1
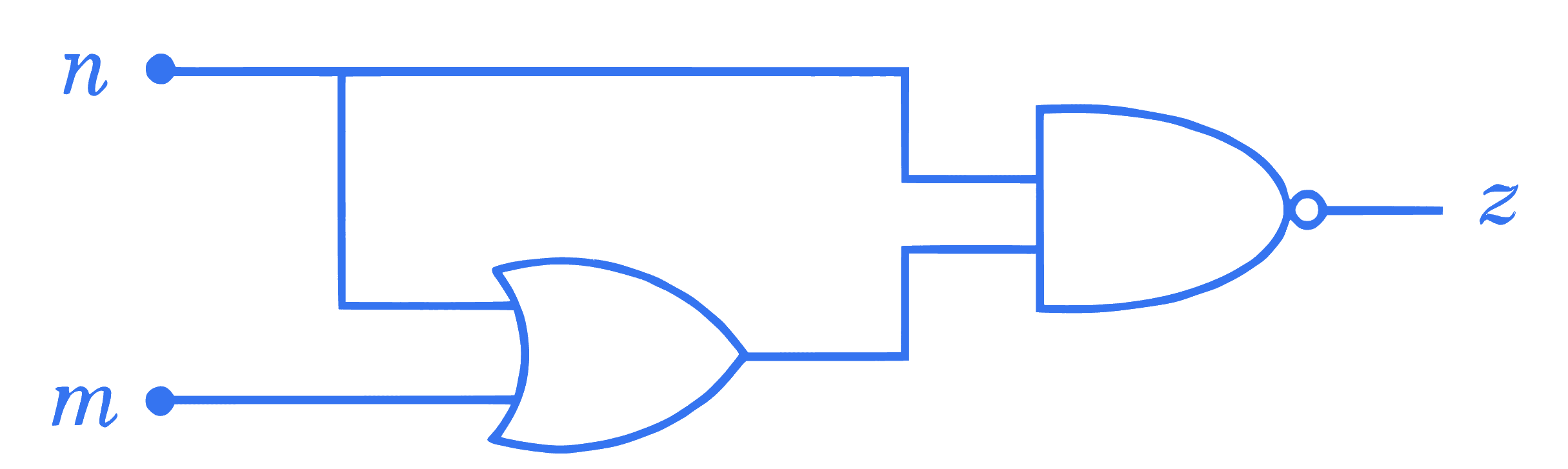
$$ \text { For the given logic gate circuit, which of the following is the correct truth table? } $$

Q3.
mcq single
+4 / 1
When an unpolarized light falls at a particular angle on a glass plate (placed in air), it is observed that the reflected beam is linearly polarized. The angle of refracted beam with respect to the normal is $\_\_\_\_$ .
$\left(\tan ^{-1}(1.52)=57.7^{\circ}\right.$, refractive indices of air and glass are 1.00 and 1.52, respectively.)
Q4.
numerical
+4 / 1
Suppose there is a uniform circular disc of mass $M \mathrm{~kg}$ and radius $r \mathrm{~m}$ shown in figure. The shaded regions are cut out from the disc. The moment of inertia of the remainder about the axis $A$ of the disc is given by $\frac{x}{256} M r^2$. The value of $x$ is $\_\_\_\_$ .


Q5.
mcq single
+4 / 1
A body of mass 14 kg initially at rest explodes and breaks into three fragments of masses in the ratio $2: 2: 3$. The two pieces of equal masses fly off perpendicular to each other with a speed of $18 \mathrm{~m} / \mathrm{s}$ each. The velocity of the heavier fragment is
$\_\_\_\_$ $\mathrm{m} / \mathrm{s}$.
Q6.
mcq single
+4 / 1
Suppose a long solenoid of 100 cm length, radius 2 cm having 500 turns per unit length, carries a current $I=10 \sin (\omega \mathrm{t}) \mathrm{A}$, where $\omega=1000 \mathrm{rad} . / \mathrm{s}$. A circular conducting loop $(B)$ of radius 1 cm coaxially slided through the solenoid at a speed $v=1 \mathrm{~cm} / \mathrm{s}$. The r.m.s. current through the loop when the coil $B$ is inserted 10 cm inside the solenoid is $${\alpha \over {\sqrt 2 }}\mu A$$. The value of $\alpha$ is $\_\_\_\_$ .
[Resistance of the loop $=10 \Omega$ ]
Q7.
mcq single
+4 / 1
A circular loop of radius 7 cm is placed in uniform magnetic field of 0.2 T directed perpendicular to plane of loop. The loop is converted into a square loop in 0.5 s . The EMF induced in the loop is $\_\_\_\_$ mV.
Q8.
mcq single
+4 / 1
The internal energy of a monoatomic gas is 3nRT. One mole of helium is kept in a cylinder having internal cross section area of $17 \mathrm{~cm}^2$ and fitted with a light movable frictionless piston. The gas is heated slowly by suppling 126 J heat. If the temperature rises by $4^{\circ} \mathrm{C}$, then the piston will move $\_\_\_\_$ cm.
(atmospheric pressure $=10^5 \mathrm{~Pa}$ )
Q9.
mcq single
+4 / 1
One mole of an ideal diatomic gas expands from volume $V$ to $2 V$ isothermally at a temperature $27^{\circ} \mathrm{C}$ and does $W$ joule of work. If the gas undergoes same magnitude of expansion adiabatically from $27^{\circ} \mathrm{C}$ doing the same amount of work $W$, then its final temperature will be (close to) $\_\_\_\_$ ${ }^{\circ} \mathrm{C}$.
$$ \left(\log _e 2=0.693\right) $$
Q10.
mcq single
+4 / 1
An air bubble of volume $2.9 \mathrm{~cm}^3$ rises from the bottom of a swimming pool of 5 m deep. At the bottom of the pool water temperature is $17^{\circ} \mathrm{C}$. The volume of the bubble when it reaches the surface, where the water temperature is $27^{\circ} \mathrm{C}$, is $\_\_\_\_$ $\mathrm{cm}^3$.
( $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$, density of water $=10^3 \mathrm{~kg} / \mathrm{m}^3$, and 1 atm pressure is $10^5 \mathrm{~Pa}$ )
Q11.
mcq single
+4 / 1
A small metallic sphere of diameter 2 mm and density $10.5 \mathrm{~g} / \mathrm{cm}^3$ is dropped in glycerine having viscosity 10 Poise and density $1.5 \mathrm{~g} / \mathrm{cm}^3$ respectively. The terminal velocity attained by the sphere is $\_\_\_\_$ $\mathrm{cm} / \mathrm{s}$.
$\left(\pi=\frac{22}{7}\right.$ and $\left.g=10 \mathrm{~m} / \mathrm{s}^2\right)$
Q12.
numerical
+4 / 1
A ball of radius $r$ and density $\rho$ dropped through a viscous liquid of density $\sigma$ and viscosity $\eta$ attains its terminal velocity at time $t$, given by $t=A \rho^a r^b \eta^c \sigma^d$, where $A$ is a constant and $a, b, c$ and $d$ are integers. The value of $\frac{b+c}{a+d}$ is $\_\_\_\_$ .
Q13.
mcq single
+4 / 1
Two shorts dipoles $(A, B), A$ having charges $\pm 2 \mu \mathrm{C}$ and length 1 cm and $B$ having charges $\pm 4 \mu \mathrm{C}$ and length 1 cm are placed with their centres 80 cm apart as shown in the figure. The electric field at a point $P$, equi-distant from the centres of both dipoles is $\_\_\_\_$ N/C.


Q14.
mcq single
+4 / 1
Two charges $7 \mu \mathrm{C}$ and $-2 \mu \mathrm{C}$ are placed at $(-9,0,0) \mathrm{cm}$ and $(9,0,0) \mathrm{cm}$ respectively in an external field $E=\frac{\mathrm{A}}{r^2} \hat{r}$, where $A=9 \times 10^5 \mathrm{~N} / \mathrm{C} . \mathrm{m}^2$. Considering the potential at infinity is 0 , the electrostatic energy of the configuration is $\_\_\_\_$ J.
Q15.
mcq single
+4 / 1
A parallel plate capacitor with plate separation 5 mm is charged by a battery. On introducing a mica sheet of 2 mm and maintaining the connections of the plates with the terminals of the battery, it is found that it draws $25 \%$ more charge from the battery. The dielectric constant of mica is $\_\_\_\_$
Q16.
numerical
+4 / 1
The size of the images of an object, formed by a thin lens are equal when the object is placed at two different positions 8 cm and 24 cm from the lens. The focal length of the lens is $\_\_\_\_$ cm.
Q17.
mcq multi
+4 / 1
A prism of angle $75^{\circ}$ and refractive index $\sqrt{3}$ is coated with thin film of refractive index 1.5 only at the back exit surface. To have total internal reflection at the back exit surface the incident angle must be $\_\_\_\_$
$\left(\sin 15^{\circ}=0.25\right.$ and $\left.\sin 25^{\circ}=0.43\right)$
Q18.
mcq single
+4 / 1
The current passing through a conducting loop in the form of equilateral triangle of side $4 \sqrt{3} \mathrm{~cm}$ is 2 A . The magnetic field at its centroid is $\alpha \times 10^{-5} \mathrm{~T}$. The value of $\alpha$ is $\_\_\_\_$ .
(Given : $\mu_{\mathrm{o}}=4 \pi \times 10^{-7}$ SI units)
Q19.
mcq single
+4 / 1
A paratrooper jumps from an aeroplane and opens a parachute after 2 s of free fall and starts deaccelerating with $3 \mathrm{~m} / \mathrm{s}^2$. At 10 m height from ground, while descending with the help of parachute, the speed of paratrooper is $5 \mathrm{~m} / \mathrm{s}$. The initial height of the airplane is $\_\_\_\_$ m.
$$ \left(\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2\right) $$
Q20.
numerical
+4 / 1
The average energy released per fission for the nucleus of ${ }_{92}^{235} \mathrm{U}$ is 190 MeV . When all the atoms of 47 g pure ${ }_{92}^{235} \mathrm{U}$ undergo fission process, the energy released is $\alpha \times 10^{23} \mathrm{MeV}$. The value of $\alpha$ is $\_\_\_\_$ .
(Avogadro Number $=6 \times 10^{23}$ per mole)
Q21.
mcq single
+4 / 1
Which of the following pair of nuclei are isobars of the element?
Q22.
numerical
+4 / 1
The velocity of sound in air is doubled when the temperature is raised from $0^{\circ} \mathrm{C}$ to $\alpha{ }^{\circ} \mathrm{C}$. The value of $\alpha$ is $\_\_\_\_$ .
Q23.
mcq single
+4 / 1
A block is sliding down on an inclined plane of slope $\theta$ and at an instant $t=0$ this block is given an upward momentum so that it starts moving up on the inclined surface with velocity $u$. The distance $(S)$ travelled by the block before its velocity become zero, is $\_\_\_\_$ .
(g = gravitational acceleration)
Q24.
mcq single
+4 / 1
To compare EMF of two cells using potentiometer the balancing lengths obtained are 200 cm and 150 cm . The least count of scale is 1 cm . The percentage error in the ratio of EMFs is $\_\_\_\_$
Q25.
mcq single
+4 / 1
The ratio of speeds of electromagnetic waves in vacuum and a medium, having dielectric constant $k=3$ and permeability of $\mu=2 \mu_0$, is ( $\mu_0=$ permeability of vacuum)
Q26.
mcq single
+4 / 1
A bead $P$ sliding on a frictionless semi-circular string $(A C B)$ and it is at point $S$ at $t =0$ and at this instant the horizontal component of its velocity is $v$. Another bead $Q$ of the same mass as $P$ is ejected from point $A$ at $t=0$ along the horizontal string $A B$, with the speed $v$, friction between the beads and the respective strings may be neglected in both cases. Let $t_P$ and $t_Q$ be the respective times taken by beads $P$ and $Q$ to reach the point $B$, then the relation between $t_P$ and $t_Q$ is


Q27.
mcq single
+4 / 1
$$ \text { For the given logic gate circuit, which of the following is the correct truth table? } $$


Q28.
mcq single
+4 / 1
When an unpolarized light falls at a particular angle on a glass plate (placed in air), it is observed that the reflected beam is linearly polarized. The angle of refracted beam with respect to the normal is $\_\_\_\_$ .
$\left(\tan ^{-1}(1.52)=57.7^{\circ}\right.$, refractive indices of air and glass are 1.00 and 1.52, respectively.)
Q29.
numerical
+4 / 1
Suppose there is a uniform circular disc of mass $M \mathrm{~kg}$ and radius $r \mathrm{~m}$ shown in figure. The shaded regions are cut out from the disc. The moment of inertia of the remainder about the axis $A$ of the disc is given by $\frac{x}{256} M r^2$. The value of $x$ is $\_\_\_\_$ .


Q30.
mcq single
+4 / 1
A body of mass 14 kg initially at rest explodes and breaks into three fragments of masses in the ratio $2: 2: 3$. The two pieces of equal masses fly off perpendicular to each other with a speed of $18 \mathrm{~m} / \mathrm{s}$ each. The velocity of the heavier fragment is
$\_\_\_\_$ $\mathrm{m} / \mathrm{s}$.
Q31.
mcq single
+4 / 1
Suppose a long solenoid of 100 cm length, radius 2 cm having 500 turns per unit length, carries a current $I=10 \sin (\omega \mathrm{t}) \mathrm{A}$, where $\omega=1000 \mathrm{rad} . / \mathrm{s}$. A circular conducting loop $(B)$ of radius 1 cm coaxially slided through the solenoid at a speed $v=1 \mathrm{~cm} / \mathrm{s}$. The r.m.s. current through the loop when the coil $B$ is inserted 10 cm inside the solenoid is $${\alpha \over {\sqrt 2 }}\mu A$$. The value of $\alpha$ is $\_\_\_\_$ .
[Resistance of the loop $=10 \Omega$ ]
Q32.
mcq single
+4 / 1
A circular loop of radius 7 cm is placed in uniform magnetic field of 0.2 T directed perpendicular to plane of loop. The loop is converted into a square loop in 0.5 s . The EMF induced in the loop is $\_\_\_\_$ mV.
Q33.
mcq single
+4 / 1
The internal energy of a monoatomic gas is 3nRT. One mole of helium is kept in a cylinder having internal cross section area of $17 \mathrm{~cm}^2$ and fitted with a light movable frictionless piston. The gas is heated slowly by suppling 126 J heat. If the temperature rises by $4^{\circ} \mathrm{C}$, then the piston will move $\_\_\_\_$ cm.
(atmospheric pressure $=10^5 \mathrm{~Pa}$ )
Q34.
mcq single
+4 / 1
One mole of an ideal diatomic gas expands from volume $V$ to $2 V$ isothermally at a temperature $27^{\circ} \mathrm{C}$ and does $W$ joule of work. If the gas undergoes same magnitude of expansion adiabatically from $27^{\circ} \mathrm{C}$ doing the same amount of work $W$, then its final temperature will be (close to) $\_\_\_\_$ ${ }^{\circ} \mathrm{C}$.
$$ \left(\log _e 2=0.693\right) $$
Q35.
mcq single
+4 / 1
An air bubble of volume $2.9 \mathrm{~cm}^3$ rises from the bottom of a swimming pool of 5 m deep. At the bottom of the pool water temperature is $17^{\circ} \mathrm{C}$. The volume of the bubble when it reaches the surface, where the water temperature is $27^{\circ} \mathrm{C}$, is $\_\_\_\_$ $\mathrm{cm}^3$.
( $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$, density of water $=10^3 \mathrm{~kg} / \mathrm{m}^3$, and 1 atm pressure is $10^5 \mathrm{~Pa}$ )
Q36.
mcq single
+4 / 1
A small metallic sphere of diameter 2 mm and density $10.5 \mathrm{~g} / \mathrm{cm}^3$ is dropped in glycerine having viscosity 10 Poise and density $1.5 \mathrm{~g} / \mathrm{cm}^3$ respectively. The terminal velocity attained by the sphere is $\_\_\_\_$ $\mathrm{cm} / \mathrm{s}$.
$\left(\pi=\frac{22}{7}\right.$ and $\left.g=10 \mathrm{~m} / \mathrm{s}^2\right)$
Q37.
numerical
+4 / 1
A ball of radius $r$ and density $\rho$ dropped through a viscous liquid of density $\sigma$ and viscosity $\eta$ attains its terminal velocity at time $t$, given by $t=A \rho^a r^b \eta^c \sigma^d$, where $A$ is a constant and $a, b, c$ and $d$ are integers. The value of $\frac{b+c}{a+d}$ is $\_\_\_\_$ .
Q38.
mcq single
+4 / 1
Two shorts dipoles $(A, B), A$ having charges $\pm 2 \mu \mathrm{C}$ and length 1 cm and $B$ having charges $\pm 4 \mu \mathrm{C}$ and length 1 cm are placed with their centres 80 cm apart as shown in the figure. The electric field at a point $P$, equi-distant from the centres of both dipoles is $\_\_\_\_$ N/C.


Q39.
mcq single
+4 / 1
Two charges $7 \mu \mathrm{C}$ and $-2 \mu \mathrm{C}$ are placed at $(-9,0,0) \mathrm{cm}$ and $(9,0,0) \mathrm{cm}$ respectively in an external field $E=\frac{\mathrm{A}}{r^2} \hat{r}$, where $A=9 \times 10^5 \mathrm{~N} / \mathrm{C} . \mathrm{m}^2$. Considering the potential at infinity is 0 , the electrostatic energy of the configuration is $\_\_\_\_$ J.
Q40.
mcq single
+4 / 1
A parallel plate capacitor with plate separation 5 mm is charged by a battery. On introducing a mica sheet of 2 mm and maintaining the connections of the plates with the terminals of the battery, it is found that it draws $25 \%$ more charge from the battery. The dielectric constant of mica is $\_\_\_\_$
Q41.
numerical
+4 / 1
The size of the images of an object, formed by a thin lens are equal when the object is placed at two different positions 8 cm and 24 cm from the lens. The focal length of the lens is $\_\_\_\_$ cm.
Q42.
mcq multi
+4 / 1
A prism of angle $75^{\circ}$ and refractive index $\sqrt{3}$ is coated with thin film of refractive index 1.5 only at the back exit surface. To have total internal reflection at the back exit surface the incident angle must be $\_\_\_\_$
$\left(\sin 15^{\circ}=0.25\right.$ and $\left.\sin 25^{\circ}=0.43\right)$
Q43.
mcq single
+4 / 1
The current passing through a conducting loop in the form of equilateral triangle of side $4 \sqrt{3} \mathrm{~cm}$ is 2 A . The magnetic field at its centroid is $\alpha \times 10^{-5} \mathrm{~T}$. The value of $\alpha$ is $\_\_\_\_$ .
(Given : $\mu_{\mathrm{o}}=4 \pi \times 10^{-7}$ SI units)
Q44.
mcq single
+4 / 1
A paratrooper jumps from an aeroplane and opens a parachute after 2 s of free fall and starts deaccelerating with $3 \mathrm{~m} / \mathrm{s}^2$. At 10 m height from ground, while descending with the help of parachute, the speed of paratrooper is $5 \mathrm{~m} / \mathrm{s}$. The initial height of the airplane is $\_\_\_\_$ m.
$$ \left(\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2\right) $$
Q45.
numerical
+4 / 1
The average energy released per fission for the nucleus of ${ }_{92}^{235} \mathrm{U}$ is 190 MeV . When all the atoms of 47 g pure ${ }_{92}^{235} \mathrm{U}$ undergo fission process, the energy released is $\alpha \times 10^{23} \mathrm{MeV}$. The value of $\alpha$ is $\_\_\_\_$ .
(Avogadro Number $=6 \times 10^{23}$ per mole)
Q46.
mcq single
+4 / 1
Which of the following pair of nuclei are isobars of the element?
Q47.
numerical
+4 / 1
The velocity of sound in air is doubled when the temperature is raised from $0^{\circ} \mathrm{C}$ to $\alpha{ }^{\circ} \mathrm{C}$. The value of $\alpha$ is $\_\_\_\_$ .
Q48.
mcq single
+4 / 1
A block is sliding down on an inclined plane of slope $\theta$ and at an instant $t=0$ this block is given an upward momentum so that it starts moving up on the inclined surface with velocity $u$. The distance $(S)$ travelled by the block before its velocity become zero, is $\_\_\_\_$ .
(g = gravitational acceleration)
Q49.
mcq single
+4 / 1
To compare EMF of two cells using potentiometer the balancing lengths obtained are 200 cm and 150 cm . The least count of scale is 1 cm . The percentage error in the ratio of EMFs is $\_\_\_\_$
Q50.
mcq single
+4 / 1
The ratio of speeds of electromagnetic waves in vacuum and a medium, having dielectric constant $k=3$ and permeability of $\mu=2 \mu_0$, is ( $\mu_0=$ permeability of vacuum)