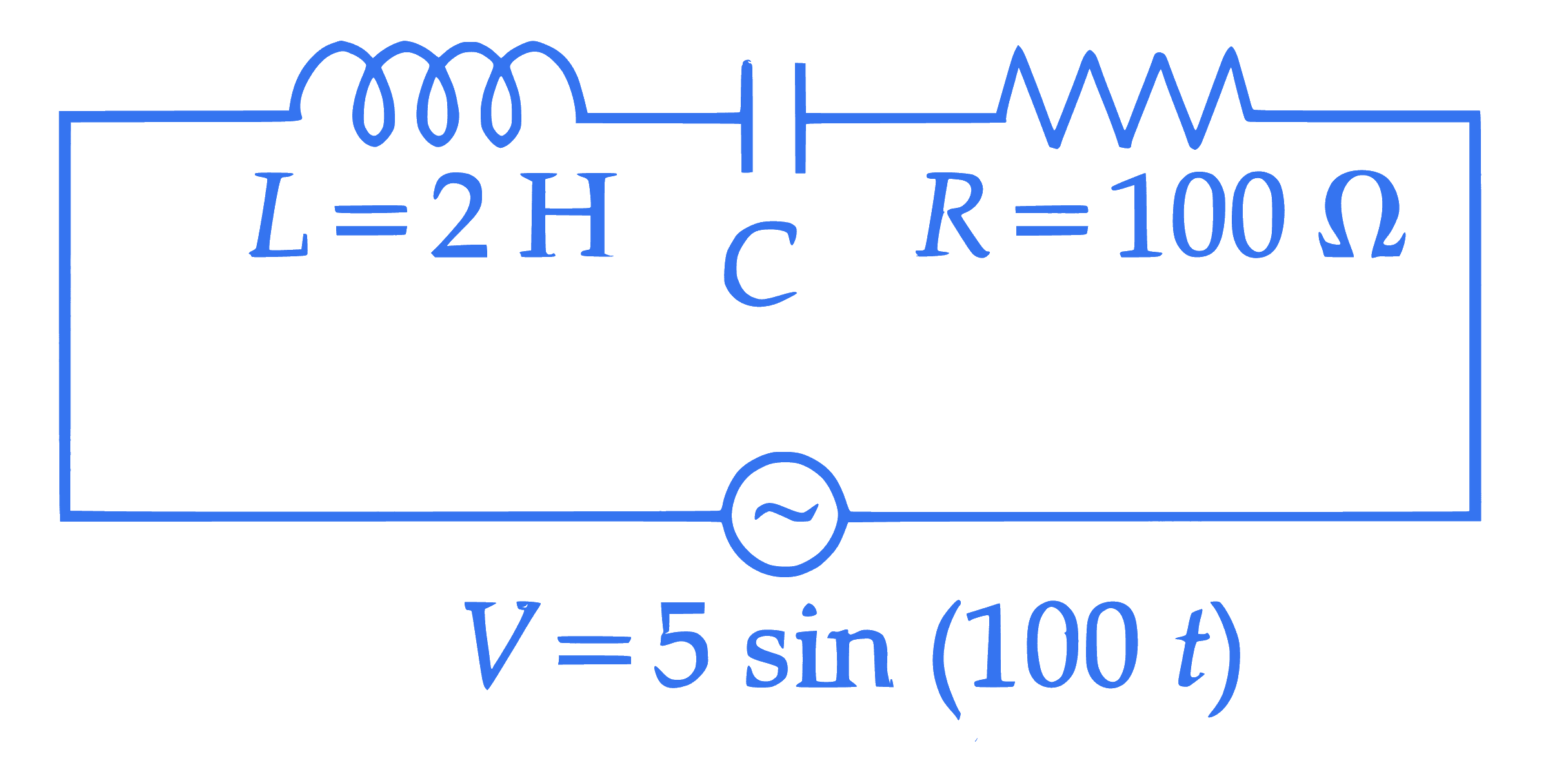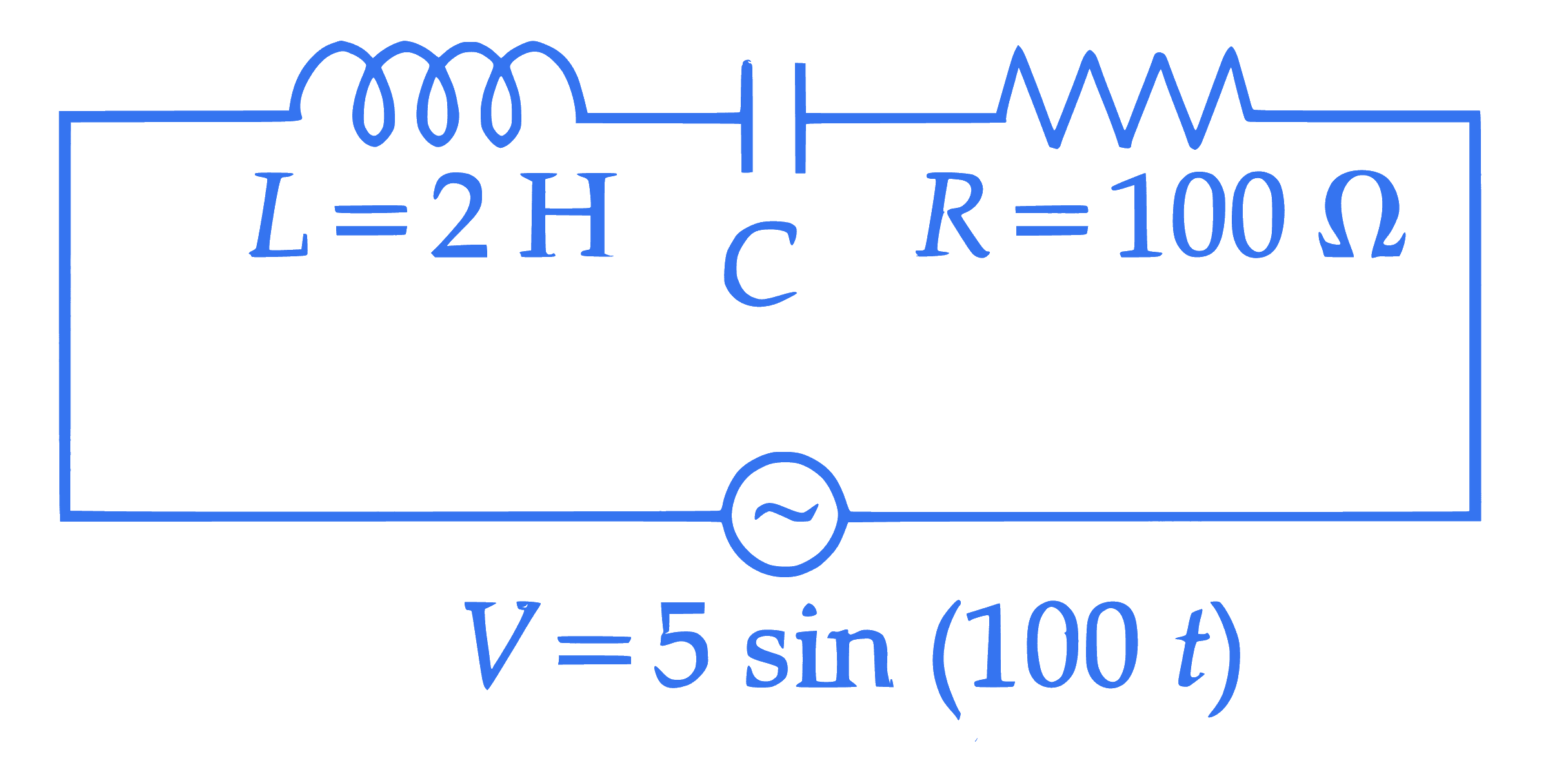JEE Main 2026 (Online) 23rd January Morning Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
148Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 148 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Consider the following sequence of reactions.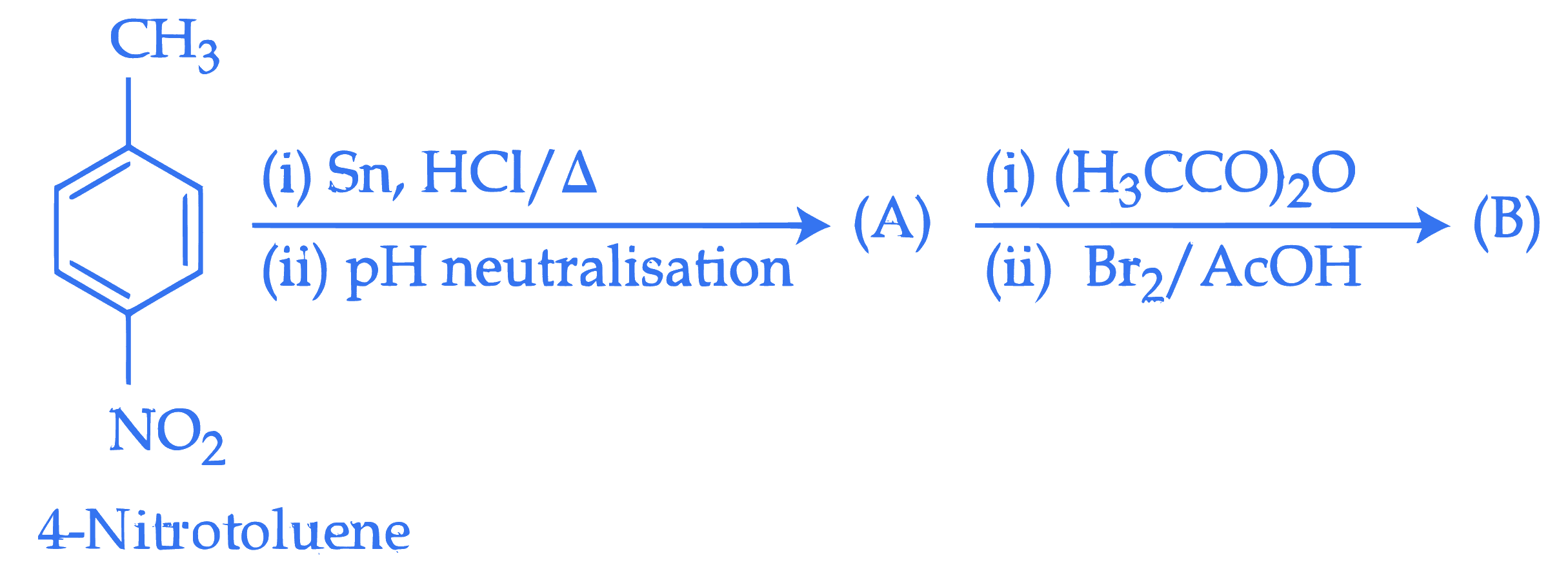 Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
 Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Q2.
numerical
+4 / 1
For the thermal decomposition of reactant $\mathrm{AB}(\mathrm{g})$, the following plot is constructed.
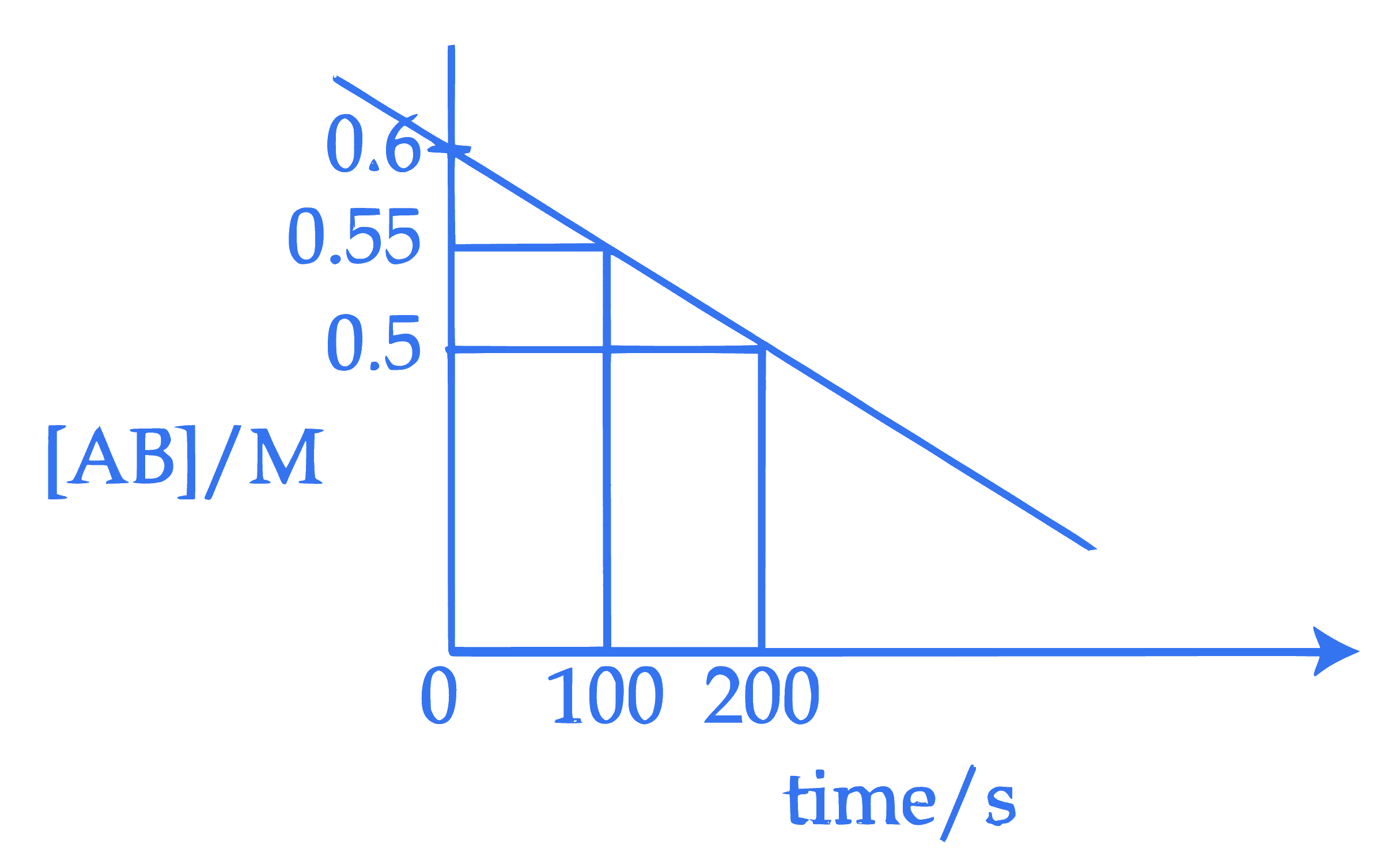 The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
 The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
Q3.
mcq single
+4 / 1
Identify the molecule $(X)$ with maximum number of lone pairs of electrons (obtained using Lewis dot structure) among $\mathrm{HNO}_3, \mathrm{H}_2 \mathrm{SO}_4, \mathrm{NF}_3$ and $\mathrm{O}_3$. Choose the correct bond angle made by the central atom of the molecule $(X)$.
Q4.
mcq single
+4 / 1
$$ \text { Consider the following compounds } $$
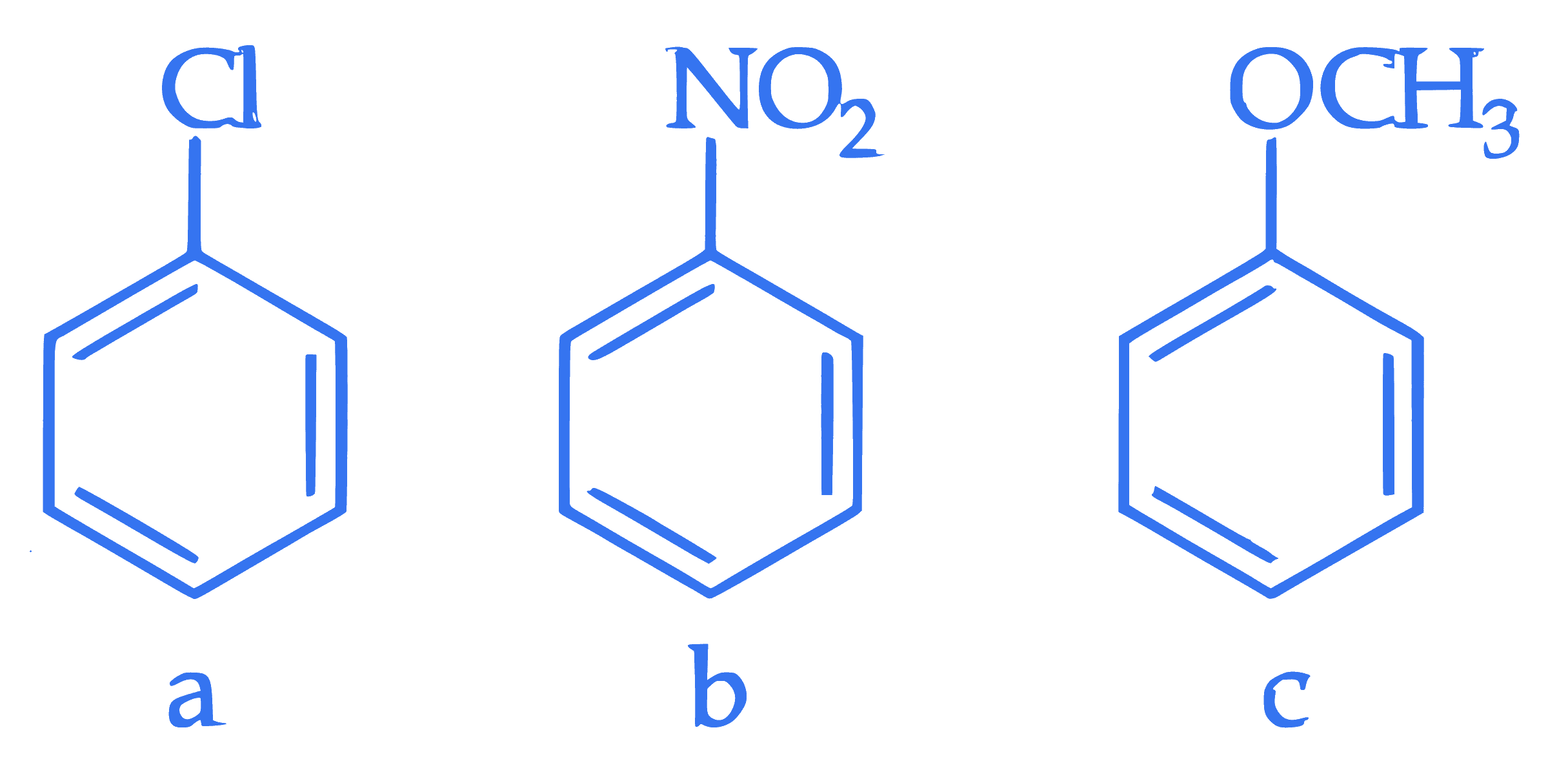 $$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
$$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
 $$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
$$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
Q5.
numerical
+4 / 1
Consider all the structural isomers with molecular formula $\mathrm{C}_5 \mathrm{H}_{11} \mathrm{Br}$ are separately treated with $\mathrm{KOH}(\mathrm{aq})$ to give respective substitution products, without any rearrangement. The number of products which can exhibit optical isomerism from these is $\_\_\_\_$。
Q6.
mcq single
+4 / 1
Which one of the following graphs accurately represents the plot of partial pressure of $\mathrm{CS}_2$ vs its mole fraction in a mixture of acetone and $\mathrm{CS}_2$ at constant temperature?
Q7.
numerical
+4 / 1
$x $ $\mathrm{mg}$ of pure HCl was used to make an aqueous solution. 25.0 mL of $0.1 \mathrm{M} \mathrm{Ba}(\mathrm{OH})_2$ solution is used when the HCl solution was titrated against it. The numerical value of $x$ is $\_\_\_\_$ $\times 10^{-1}$. (Nearest integer)
Given : Molar mass of HCl and $\mathrm{Ba}(\mathrm{OH})_2$ are 36.5 and $171.0 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.
Q8.
mcq single
+4 / 1
Consider the general reaction given below at 400 K
$$ x \mathrm{~A}(\mathrm{~g}) \rightleftharpoons y \mathrm{~B}(\mathrm{~g}) . $$
The values of $K_p$ and $K_c$ are studied under the same condition of temperature but variation in $x$ and $y$.
(i) $\mathrm{K}_{\mathrm{p}}=85.87$ and $\mathrm{K}_{\mathrm{c}}=2.586$ appropriate units
(ii) $\mathrm{K}_{\mathrm{p}}=0.862$ and $\mathrm{K}_{\mathrm{c}}=28.62$ appropriate units
The values of $x$ and $y$ in (i) and (ii) respectively are :
Q9.
numerical
+4 / 1
For the following gas phase equilibrium reaction at constant temperature,
$$ \mathrm{NH}_3(\mathrm{~g}) \rightleftharpoons 1 / 2 \mathrm{~N}_2(\mathrm{~g})+3 / 2 \mathrm{H}_2(\mathrm{~g}) $$
if the total pressure is $\sqrt{3} \mathrm{~atm}$ and the pressure equilibrium constant $\left(K_p\right)$ is 9 atm , then the degree of dissociation is given as $\left(x \times 10^{-2}\right)^{-1 / 2}$. The value of $x$ is $\_\_\_\_$ . (nearest integer)
Q10.
mcq single
+4 / 1
The correct trend in the first ionization enthalpies of the elements in the $3^{\text {rd }}$ period of periodic table is :
Q11.
mcq single
+4 / 1
A cup of water at $5^{\circ} \mathrm{C}$ (system) is placed in a microwave oven and the oven is turned on for one minute during which the water begins to boil. Which of the following option is true?
Q12.
mcq single
+4 / 1
The correct statements from the following are :
A. Ionic radii of trivalent cations of group 13 elements decreases down the group.
B. Electronegativity of group 13 elements decreases down the group.
C. Among the group 13 elements, Boron has highest first ionisation enthalpy.
D. The trichloride and triiodide of group 13 elements are covalent in nature.
Choose the correct answer from the options given below :
Q13.
numerical
+4 / 1
The crystal field splitting energy of $\left[\mathrm{Co}(\text { oxalate })_3\right]^{3-}$ complex is ' $n^{\prime}$ times that of the $\left[\mathrm{Cr}(\text { oxalate })_3\right]^{3-}$ complex. Here ' $n$ ' is $\_\_\_\_$ . (Assume $\Delta_0 \gg P$ )
Q14.
mcq single
+4 / 1
The statements that are incorrect about the nickel(II) complex of dimethylglyoxime are :
A. It is red in colour.
B. It has a high solubility in water at $\mathrm{pH}=9$.
C. The Ni ion has two unpaired d-electrons.
D. The $\mathrm{N}-\mathrm{Ni}-\mathrm{N}$ bond angle is almost close to $90^{\circ}$.
E. The complex contains four five-membered metallacycles (metal containing rings).
Choose the correct answer from the options given below :
Q15.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\left[\mathrm{CoBr}_4\right]^{2-}$ ion will absorb light of lower energy than $\left[\mathrm{CoCl}_4\right]^{2-}$ ion.
**Statement II :** In $\left[\mathrm{CoI}_4\right]^{2-}$ ion, the energy separation between the two set of d-orbitals is more than $\left[\mathrm{CoCl}_4\right]^{2-}$ ion.
In the light of the above statements, choose the correct answer from the options given below :
Q16.
mcq single
+4 / 1
From the given following (A to D) cyclic structures, those which will not react with Tollen's reagent are:
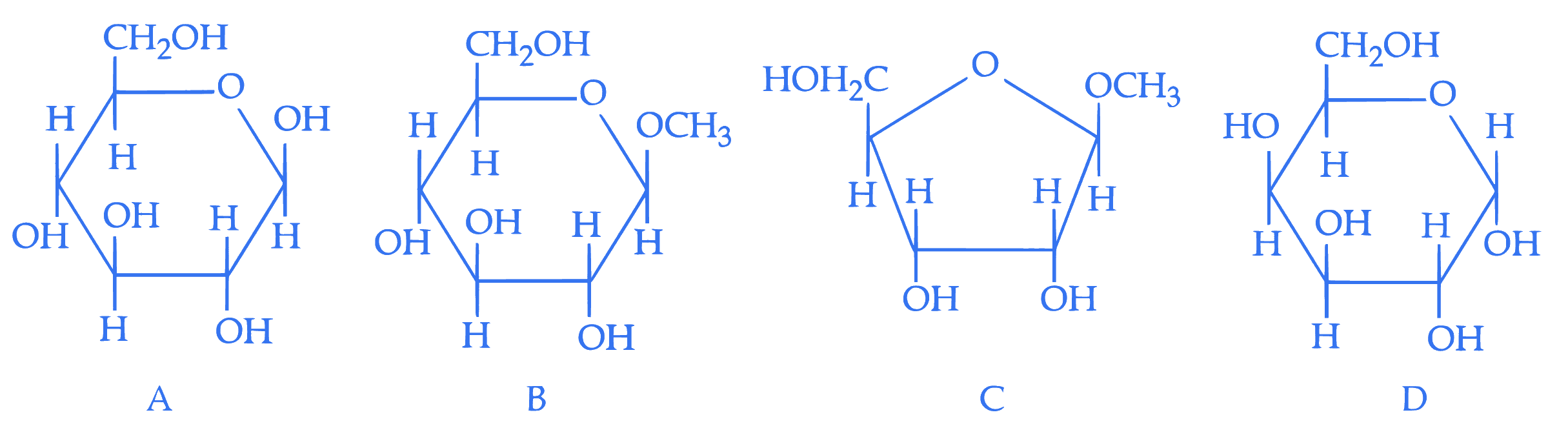

Q17.
mcq single
+4 / 1
' $x$ ' is the product which is obtained from propanenitrile and stannous chloride in the presence of hydrochloric acid followed by hydrolysis. ' $y$ ' is the product which is obtained from the but-2-ene by the ozonolysis followed by hydrolysis. From the following, which product is not obtained when one mole of ' $x$ ' and one mole of ' $y$ ' react with each other in the presence of alkali followed by heating?
Q18.
mcq single
+4 / 1
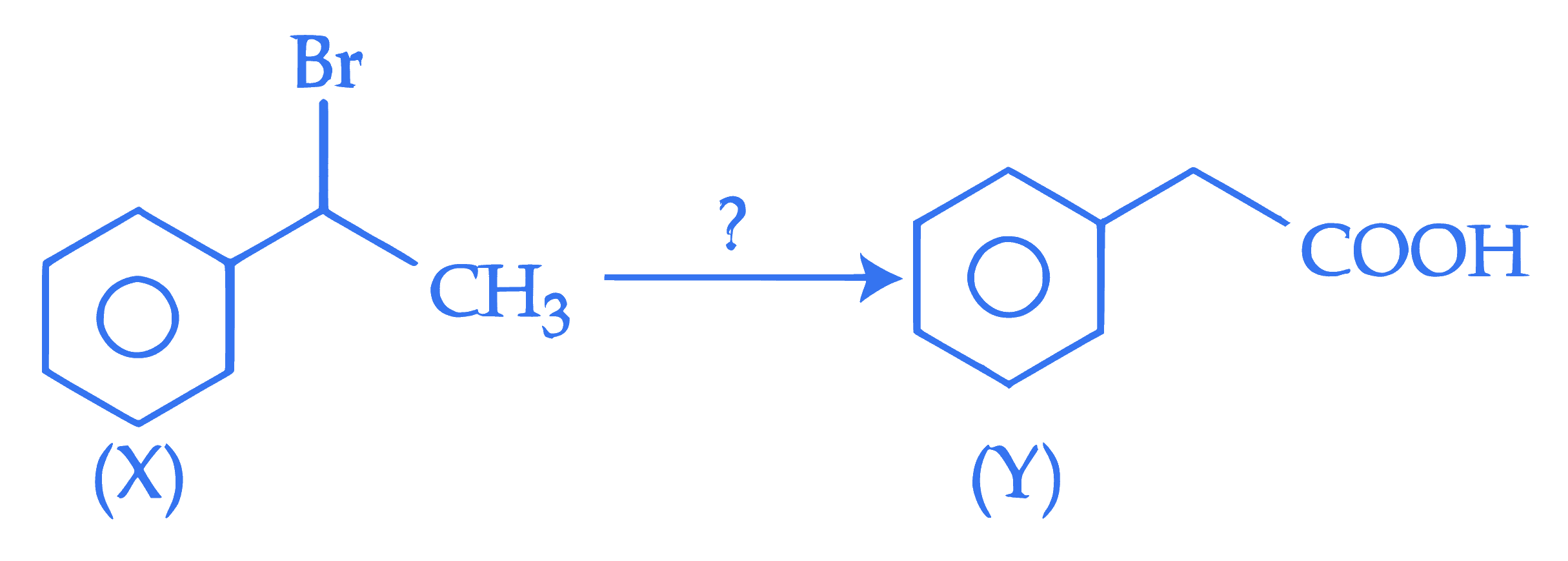 The correct sequence of reagents for the above conversion of X to Y is :
The correct sequence of reagents for the above conversion of X to Y is :
 The correct sequence of reagents for the above conversion of X to Y is :
The correct sequence of reagents for the above conversion of X to Y is :
Q19.
mcq single
+4 / 1
$$ \text { Compound ' } \mathrm{P} \text { ' undergoes the following sequence of reactions : } $$
 $$ ' \mathrm{P}^{\prime} \text { is : } $$
$$ ' \mathrm{P}^{\prime} \text { is : } $$
 $$ ' \mathrm{P}^{\prime} \text { is : } $$
$$ ' \mathrm{P}^{\prime} \text { is : } $$
Q20.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Functional group (detection)
List - II
Change observed during detection
A.
Unsaturation (Baeyer's test)
I.
Red colour appears
B.
Alcoholic group (Ceric ammonium nitrate test)
II.
Silver mirror appears
C.
Aldehyde group (Tollen's reagent)
III.
Violet colour appears
D.
Phenolic group ( $\mathrm{FeCl}_3$ test)
IV.
Discharge of pink colour
Q21.
mcq single
+4 / 1
But-2-yne and hydrogen (one mole each) are separately treated with (i) $\mathrm{Pd} / \mathrm{C}$ and (ii) $\mathrm{Na} /$ liq. $\mathrm{NH}_3$ to give the products X and Y respectively.
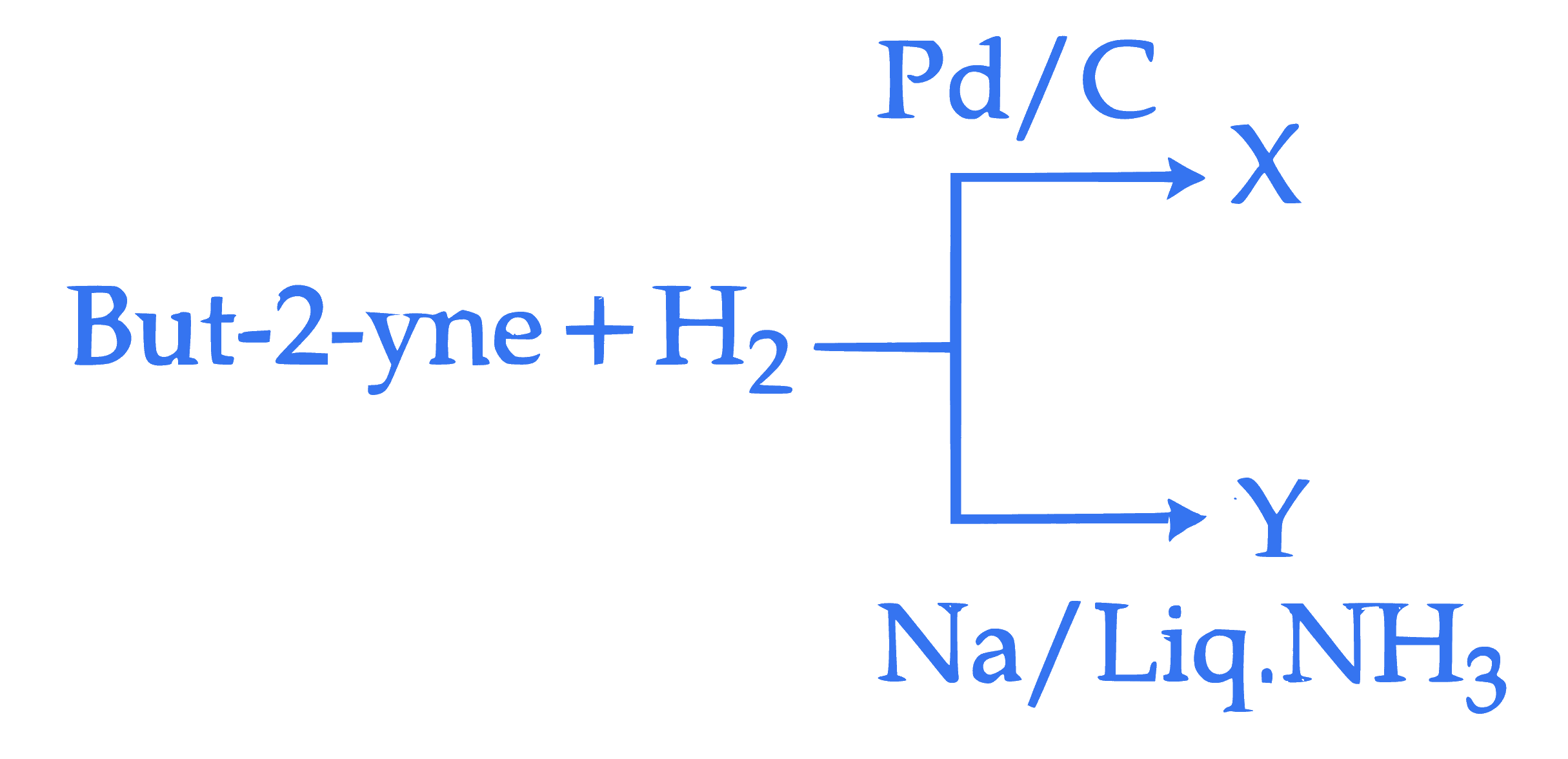 Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
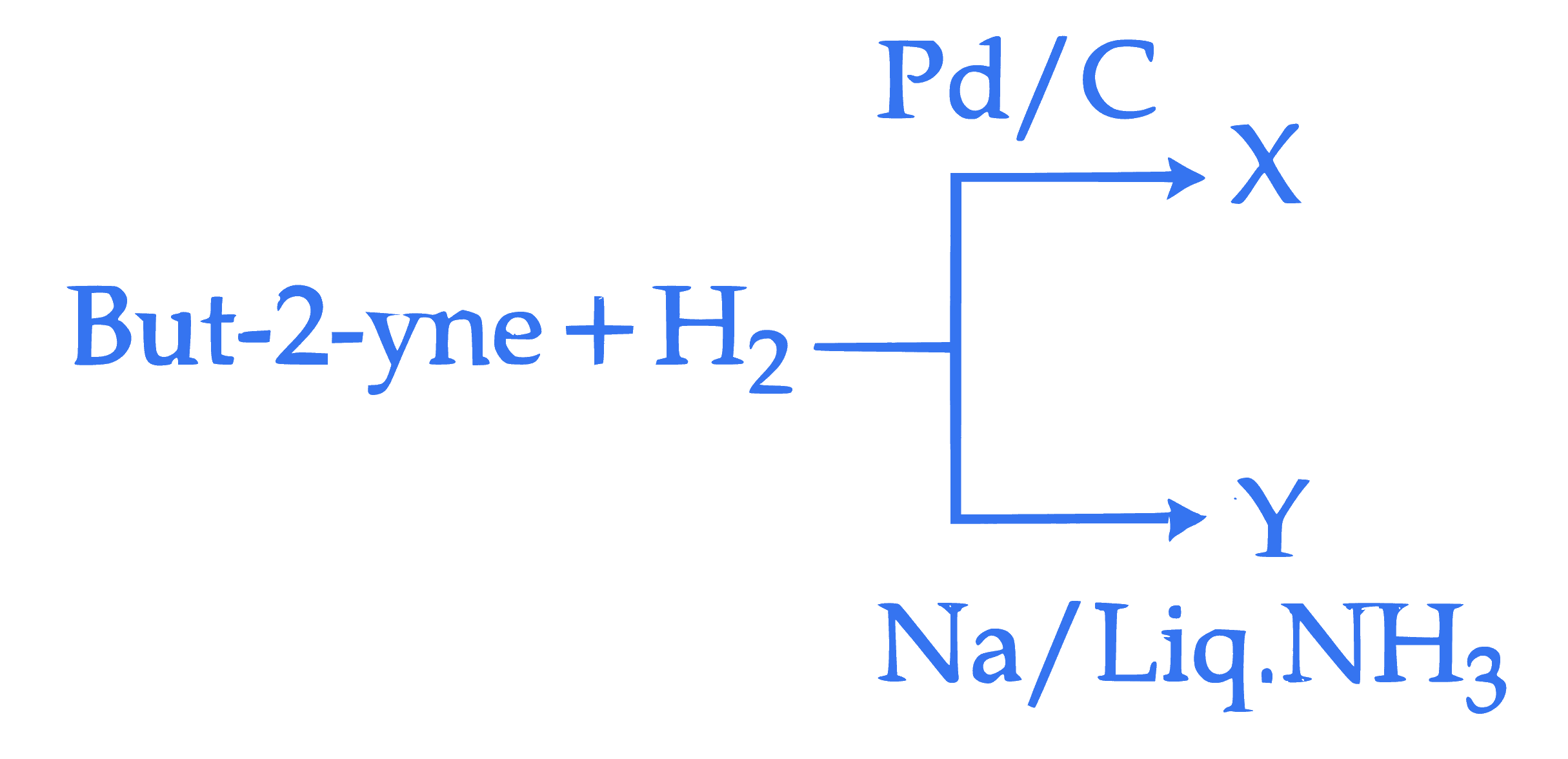 Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
Q22.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Sublimation is used for the separation and purification of compounds with low melting point.
**Statement II :** The boiling point of a liquid increases as the external pressure is reduced. In the light of the above statements,
choose the correct answer from the options given below :
Q23.
mcq single
+4 / 1
In the given electrochemical cell, $\mathrm{Ag}(\mathrm{s})|\mathrm{AgCl}(\mathrm{s})| \mathrm{FeCl}_2(\mathrm{aq}), \mathrm{FeCl}_3(\mathrm{aq}) \mid \mathrm{Pt}(\mathrm{s})$ at 298 K , the cell potential ( $\mathrm{E}_{\text {cell }}$ ) will increase when :
A. Concentration of $\mathrm{Fe}^{2+}$ is increased.
B. Concentration of $\mathrm{Fe}^{3+}$ is decreased.
C. Concentration of $\mathrm{Fe}^{2+}$ is decreased.
D. Concentration of $\mathrm{Fe}^{3+}$ is increased.
E. Concentration of $\mathrm{Cl}^{-}$is increased.
Choose the correct answer from the options given below :
Q24.
mcq single
+4 / 1
Given,
(A) $\mathrm{n}=5, \mathrm{~m}_1=-1$
(B) $\mathrm{n}=3, \mathrm{l}=2, \mathrm{~m}_1=-1, \mathrm{~m}_{\mathrm{s}}=+\frac{1}{2}$
The maximum number of electron(s) in an atom that can have the quantum numbers as given in (A) and (B) respectively are :
Q25.
mcq single
+4 / 1
Which of the following statements regarding the energy of the stationary state is true in the following one - electron systems ?
Q26.
mcq single
+4 / 1
Consider the following sequence of reactions. Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
 Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce
$\_\_\_\_$ mg of $B$.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16, \mathrm{Br}: 80$ )
Q27.
numerical
+4 / 1
For the thermal decomposition of reactant $\mathrm{AB}(\mathrm{g})$, the following plot is constructed.
 The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
 The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
The half life of the reaction is ' $x^{\prime} \,\mathrm{min}$.
$x=$ $\_\_\_\_$ min. (Nearest integer)
Q28.
mcq single
+4 / 1
Identify the molecule $(X)$ with maximum number of lone pairs of electrons (obtained using Lewis dot structure) among $\mathrm{HNO}_3, \mathrm{H}_2 \mathrm{SO}_4, \mathrm{NF}_3$ and $\mathrm{O}_3$. Choose the correct bond angle made by the central atom of the molecule $(X)$.
Q29.
mcq single
+4 / 1
$$ \text { Consider the following compounds } $$
 $$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
$$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
 $$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
$$ \text { Arrange these compounds in the increasing order of reactivity with nitrating mixture. } $$
Q30.
numerical
+4 / 1
Consider all the structural isomers with molecular formula $\mathrm{C}_5 \mathrm{H}_{11} \mathrm{Br}$ are separately treated with $\mathrm{KOH}(\mathrm{aq})$ to give respective substitution products, without any rearrangement. The number of products which can exhibit optical isomerism from these is $\_\_\_\_$。
Q31.
mcq single
+4 / 1
Which one of the following graphs accurately represents the plot of partial pressure of $\mathrm{CS}_2$ vs its mole fraction in a mixture of acetone and $\mathrm{CS}_2$ at constant temperature?
Q32.
numerical
+4 / 1
$x $ $\mathrm{mg}$ of pure HCl was used to make an aqueous solution. 25.0 mL of $0.1 \mathrm{M} \mathrm{Ba}(\mathrm{OH})_2$ solution is used when the HCl solution was titrated against it. The numerical value of $x$ is $\_\_\_\_$ $\times 10^{-1}$. (Nearest integer)
Given : Molar mass of HCl and $\mathrm{Ba}(\mathrm{OH})_2$ are 36.5 and $171.0 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.
Q33.
mcq single
+4 / 1
Consider the general reaction given below at 400 K
$$ x \mathrm{~A}(\mathrm{~g}) \rightleftharpoons y \mathrm{~B}(\mathrm{~g}) . $$
The values of $K_p$ and $K_c$ are studied under the same condition of temperature but variation in $x$ and $y$.
(i) $\mathrm{K}_{\mathrm{p}}=85.87$ and $\mathrm{K}_{\mathrm{c}}=2.586$ appropriate units
(ii) $\mathrm{K}_{\mathrm{p}}=0.862$ and $\mathrm{K}_{\mathrm{c}}=28.62$ appropriate units
The values of $x$ and $y$ in (i) and (ii) respectively are :
Q34.
numerical
+4 / 1
For the following gas phase equilibrium reaction at constant temperature,
$$ \mathrm{NH}_3(\mathrm{~g}) \rightleftharpoons 1 / 2 \mathrm{~N}_2(\mathrm{~g})+3 / 2 \mathrm{H}_2(\mathrm{~g}) $$
if the total pressure is $\sqrt{3} \mathrm{~atm}$ and the pressure equilibrium constant $\left(K_p\right)$ is 9 atm , then the degree of dissociation is given as $\left(x \times 10^{-2}\right)^{-1 / 2}$. The value of $x$ is $\_\_\_\_$ . (nearest integer)
Q35.
mcq single
+4 / 1
The correct trend in the first ionization enthalpies of the elements in the $3^{\text {rd }}$ period of periodic table is :
Q36.
mcq single
+4 / 1
A cup of water at $5^{\circ} \mathrm{C}$ (system) is placed in a microwave oven and the oven is turned on for one minute during which the water begins to boil. Which of the following option is true?
Q37.
mcq single
+4 / 1
The correct statements from the following are :
A. Ionic radii of trivalent cations of group 13 elements decreases down the group.
B. Electronegativity of group 13 elements decreases down the group.
C. Among the group 13 elements, Boron has highest first ionisation enthalpy.
D. The trichloride and triiodide of group 13 elements are covalent in nature.
Choose the correct answer from the options given below :
Q38.
numerical
+4 / 1
The crystal field splitting energy of $\left[\mathrm{Co}(\text { oxalate })_3\right]^{3-}$ complex is ' $n^{\prime}$ times that of the $\left[\mathrm{Cr}(\text { oxalate })_3\right]^{3-}$ complex. Here ' $n$ ' is $\_\_\_\_$ . (Assume $\Delta_0 \gg P$ )
Q39.
mcq single
+4 / 1
The statements that are incorrect about the nickel(II) complex of dimethylglyoxime are :
A. It is red in colour.
B. It has a high solubility in water at $\mathrm{pH}=9$.
C. The Ni ion has two unpaired d-electrons.
D. The $\mathrm{N}-\mathrm{Ni}-\mathrm{N}$ bond angle is almost close to $90^{\circ}$.
E. The complex contains four five-membered metallacycles (metal containing rings).
Choose the correct answer from the options given below :
Q40.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\left[\mathrm{CoBr}_4\right]^{2-}$ ion will absorb light of lower energy than $\left[\mathrm{CoCl}_4\right]^{2-}$ ion.
**Statement II :** In $\left[\mathrm{CoI}_4\right]^{2-}$ ion, the energy separation between the two set of d-orbitals is more than $\left[\mathrm{CoCl}_4\right]^{2-}$ ion.
In the light of the above statements, choose the correct answer from the options given below :
Q41.
mcq single
+4 / 1
From the given following (A to D) cyclic structures, those which will not react with Tollen's reagent are:


Q42.
mcq single
+4 / 1
' $x$ ' is the product which is obtained from propanenitrile and stannous chloride in the presence of hydrochloric acid followed by hydrolysis. ' $y$ ' is the product which is obtained from the but-2-ene by the ozonolysis followed by hydrolysis. From the following, which product is not obtained when one mole of ' $x$ ' and one mole of ' $y$ ' react with each other in the presence of alkali followed by heating?
Q43.
mcq single
+4 / 1
 The correct sequence of reagents for the above conversion of X to Y is :
The correct sequence of reagents for the above conversion of X to Y is :
 The correct sequence of reagents for the above conversion of X to Y is :
The correct sequence of reagents for the above conversion of X to Y is :
Q44.
mcq single
+4 / 1
$$ \text { Compound ' } \mathrm{P} \text { ' undergoes the following sequence of reactions : } $$
 $$ ' \mathrm{P}^{\prime} \text { is : } $$
$$ ' \mathrm{P}^{\prime} \text { is : } $$
 $$ ' \mathrm{P}^{\prime} \text { is : } $$
$$ ' \mathrm{P}^{\prime} \text { is : } $$
Q45.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Functional group (detection)
List - II
Change observed during detection
A.
Unsaturation (Baeyer's test)
I.
Red colour appears
B.
Alcoholic group (Ceric ammonium nitrate test)
II.
Silver mirror appears
C.
Aldehyde group (Tollen's reagent)
III.
Violet colour appears
D.
Phenolic group ( $\mathrm{FeCl}_3$ test)
IV.
Discharge of pink colour
Q46.
mcq single
+4 / 1
But-2-yne and hydrogen (one mole each) are separately treated with (i) $\mathrm{Pd} / \mathrm{C}$ and (ii) $\mathrm{Na} /$ liq. $\mathrm{NH}_3$ to give the products X and Y respectively.
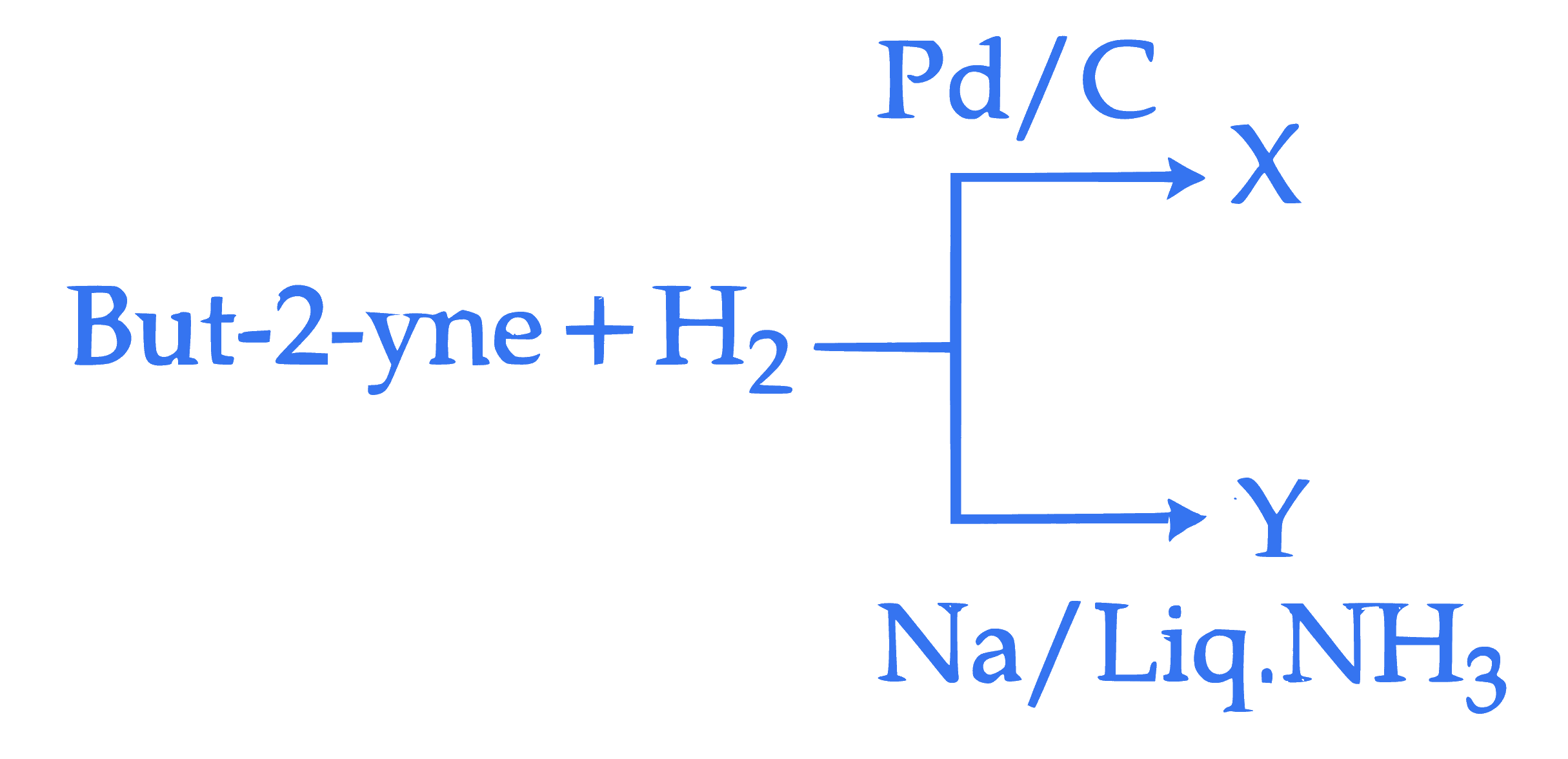 Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
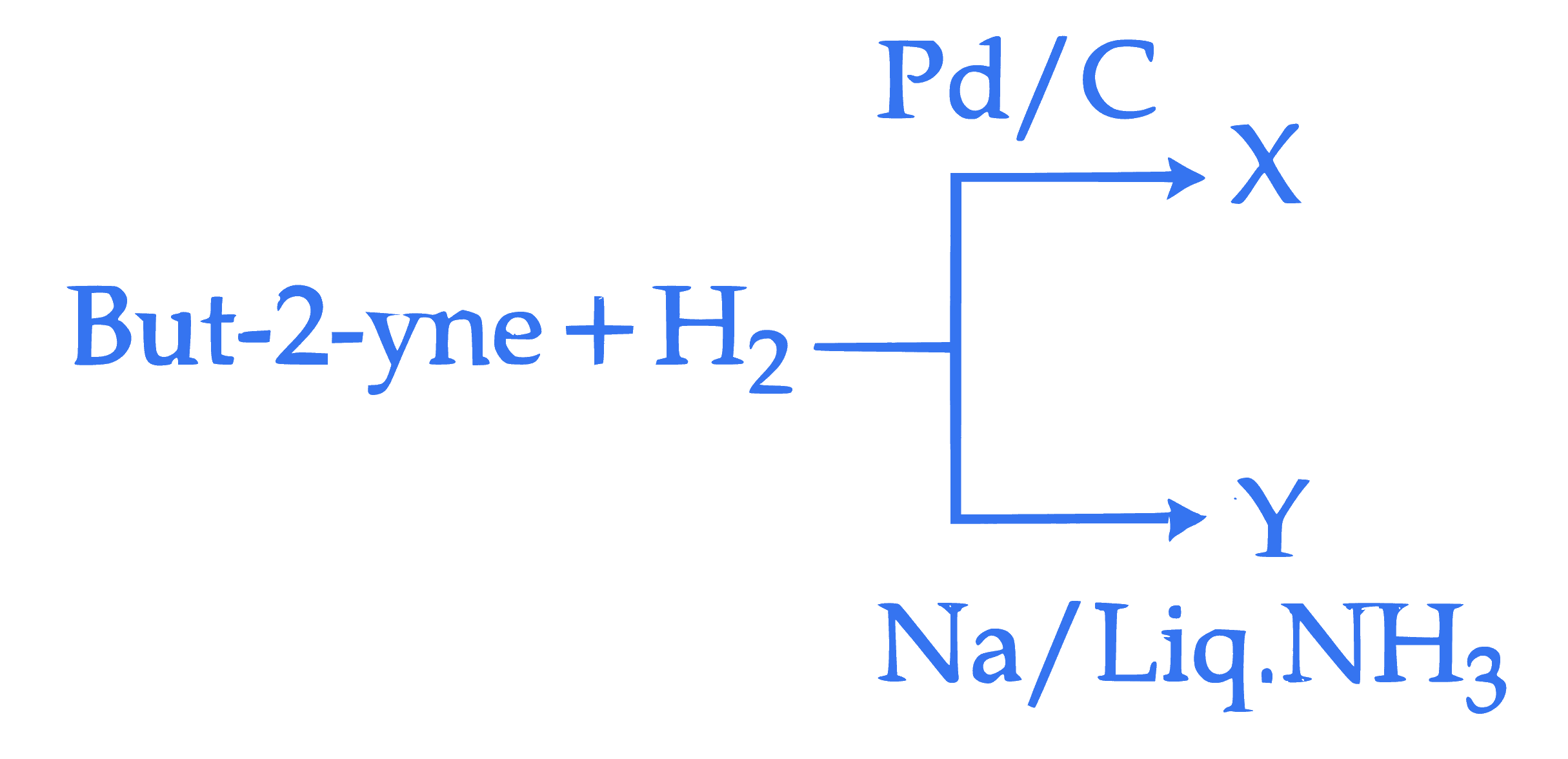 Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of $X$ is zero.
C. Boiling point of X is higher than Y .
D. X and Y react with $\mathrm{O}_3 / \mathrm{Zn}+\mathrm{H}_2 \mathrm{O}$ to give different products.
Choose the correct answer from the options given below :
Q47.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Sublimation is used for the separation and purification of compounds with low melting point.
**Statement II :** The boiling point of a liquid increases as the external pressure is reduced. In the light of the above statements,
choose the correct answer from the options given below :
Q48.
mcq single
+4 / 1
In the given electrochemical cell, $\mathrm{Ag}(\mathrm{s})|\mathrm{AgCl}(\mathrm{s})| \mathrm{FeCl}_2(\mathrm{aq}), \mathrm{FeCl}_3(\mathrm{aq}) \mid \mathrm{Pt}(\mathrm{s})$ at 298 K , the cell potential ( $\mathrm{E}_{\text {cell }}$ ) will increase when :
A. Concentration of $\mathrm{Fe}^{2+}$ is increased.
B. Concentration of $\mathrm{Fe}^{3+}$ is decreased.
C. Concentration of $\mathrm{Fe}^{2+}$ is decreased.
D. Concentration of $\mathrm{Fe}^{3+}$ is increased.
E. Concentration of $\mathrm{Cl}^{-}$is increased.
Choose the correct answer from the options given below :
Q49.
mcq single
+4 / 1
Given,
(A) $\mathrm{n}=5, \mathrm{~m}_1=-1$
(B) $\mathrm{n}=3, \mathrm{l}=2, \mathrm{~m}_1=-1, \mathrm{~m}_{\mathrm{s}}=+\frac{1}{2}$
The maximum number of electron(s) in an atom that can have the quantum numbers as given in (A) and (B) respectively are :
Q50.
mcq single
+4 / 1
Which of the following statements regarding the energy of the stationary state is true in the following one - electron systems ?
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
Let the mean and variance of 8 numbers $-10,-7,-1, x, y, 9,2,16$ be $\frac{7}{2}$ and $\frac{293}{4}$, respectively.
Then the mean of 4 numbers $x, y, x+y+1,|x-y|$ is :
Q2.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=-\hat{i}+\hat{j}+2 \hat{k}, \overrightarrow{\mathrm{~b}}=\hat{i}-\hat{j}-3 \hat{k}, \overrightarrow{\mathrm{c}}=\overrightarrow{\mathrm{a}} \times \overrightarrow{\mathrm{b}}$ and $\overrightarrow{\mathrm{d}}=\overrightarrow{\mathrm{c}} \times \overrightarrow{\mathrm{a}}$. Then $(\vec{a}-\vec{b}) \cdot \vec{d}$ is equal to :
Q3.
mcq single
+4 / 1
A building construction work can be completed by two masons A and B together in 22.5 days. Mason A alone can complete the construction work in 24 days less than mason B alone. Then mason A alone will complete the construction work in :
Q4.
mcq single
+4 / 1
If $\alpha$ and $\beta(\alpha<\beta)$ are the roots of the equation $(-2+\sqrt{3})(|\sqrt{x}-3|)+(x-6 \sqrt{x})+(9-2 \sqrt{3})=0, x \geqslant 0$, then $\sqrt{\frac{\beta}{\alpha}}+\sqrt{\alpha \beta}$ is equal to :
Q5.
numerical
+4 / 1
From the first 100 natural numbers, two numbers first $a$ and then $b$ are selected randomly without replacement. If the probability that $\mathrm{a}-\mathrm{b} \geqslant 10$ is $\frac{\mathrm{m}}{\mathrm{n}}, \operatorname{gcd}(\mathrm{m}, \mathrm{n})=1$, then $\mathrm{m}+\mathrm{n}$ is equal to
$\_\_\_\_$ .
Q6.
mcq single
+4 / 1
The vertices B and C of a triangle ABC lie on the line $\frac{x}{1}=\frac{1-y}{-2}=\frac{\mathrm{z}-2}{3}$. The coordinates of A and $B$ are $(1,6,3)$ and $(4,9, \alpha)$ respectively and $C$ is at a distance of 10 units from $B$. The area (in sq. units) of $\triangle A B C$ is :
Q7.
mcq single
+4 / 1
Let the direction cosines of two lines satisfy the equations : $4 l+m-n=0$ and $2 m n+10 n l+3 l m=0$.
Then the cosine of the acute angle between these lines is :
Q8.
mcq single
+4 / 1
Let $f(x)= \begin{cases}\frac{\mathrm{a} x^2+2 \mathrm{a} x+3}{4 x^2+4 x-3} & , x \neq-\frac{3}{2}, \frac{1}{2} \\ \mathrm{~b} & , x=-\frac{3}{2}, \frac{1}{2}\end{cases}$ be continuous at $x=-\frac{3}{2}$. If $f \circ f(x)=\frac{7}{5}$, then $x$ is equal to:
Q9.
mcq single
+4 / 1
Let $\alpha$ and $\beta$ respectively be the maximum and the minimum values of the function $f(\theta)=4\left(\sin ^4\left(\frac{7 \pi}{2}-\theta\right)+\sin ^4(11 \pi+\theta)\right)-2\left(\sin ^6\left(\frac{3 \pi}{2}-\theta\right)+\sin ^6(9 \pi-\theta)\right), \theta \in \mathbf{R}$.
Then $\alpha+2 \beta$ is equal to :
Q10.
numerical
+4 / 1
Let the area of the region bounded by the curve $y=\max \{\sin x, \cos x\}$, lines $x=0, x=\frac{3 \pi}{2}$, and the $x$-axis be A . Then, $\mathrm{A}+\mathrm{A}^2$ is equal to $\_\_\_\_$。
Q11.
mcq single
+4 / 1
Among the statements :
I: If $\left|\begin{array}{ccc}1 & \cos \alpha & \cos \beta \\ \cos \alpha & 1 & \cos \gamma \\ \cos \beta & \cos \gamma & 1\end{array}\right|=\left|\begin{array}{ccc}0 & \cos \alpha & \cos \beta \\ \cos \alpha & 0 & \cos \gamma \\ \cos \beta & \cos \gamma & 0\end{array}\right|$, then $\cos ^2 \alpha+\cos ^2 \beta+\cos ^2 \gamma=\frac{3}{2}$, and
II: If $\left|\begin{array}{ccc}x^2+x & x+1 & x-2 \\ 2 x^2+3 x-1 & 3 x & 3 x-3 \\ x^2+2 x+3 & 2 x-1 & 2 x-1\end{array}\right|=\mathrm{p} x+\mathrm{q}$, then $\mathrm{p}^2=196 \mathrm{q}^2$,
Q12.
numerical
+4 / 1
Let $|\mathrm{A}|=6$, where A is a $3 \times 3$ matrix. If $\left|\operatorname{adj}\left(3\operatorname{adj}\left(\mathrm{A}^2 \cdot \operatorname{adj}(2 \mathrm{~A})\right)\right)\right|=2^{\mathrm{m}} \cdot 3^{\mathrm{n}}, \mathrm{m}, \mathrm{n} \in \mathbf{N}$, then $\mathrm{m}+\mathrm{n}$ is equal to
$\_\_\_\_$ .
Q13.
mcq single
+4 / 1
Number of solutions of $\sqrt{3} \cos 2 \theta+8 \cos \theta+3 \sqrt{3}=0, \theta \in[-3 \pi, 2 \pi]$ is :
Q14.
mcq single
+4 / 1
Let $f(x)=\int \frac{\left(2-x^2\right) \cdot \mathrm{e}^x}{(\sqrt{1+x})(1-x)^{3 / 2}} \mathrm{~d} x$. If $f(0)=0$, then $f\left(\frac{1}{2}\right)$ is equal to:
Q15.
mcq single
+4 / 1
The value of $\frac{{ }^{100} \mathrm{C}_{50}}{51}+\frac{{ }^{100} \mathrm{C}_{51}}{52}+\ldots .+\frac{{ }^{100} \mathrm{C}_{100}}{101}$ is:
Q16.
mcq single
+4 / 1
The sum of all possible values of $\mathbf{n} \in \mathbf{N}$, so that the coefficients of $x, x^2$ and $x^3$ in the expansion of $\left(1+x^2\right)^2(1+x)^{\mathrm{n}}$, are in arithmetic progression is :
Q17.
mcq single
+4 / 1
A rectangle is formed by the lines $x=0, y=0, x=3$ and $y=4$. Let the line L be perpendicular to $3 x+y+6=0$ and divide the area of the rectangle into two equal parts. Then the distance of the point $\left(\frac{1}{2},-5\right)$ from the line $L$ is equal to :
Q18.
mcq single
+4 / 1
Let $\mathrm{A}=\{-2,-1,0,1,2,3,4\}$. Let R be a relation on A defined by $x \mathrm{R} y$ if and only if $2 x+y \leqslant 2$. Let $l$ be the number of elements in R . Let m and n be the minimum number of elements required to be added in R to make it reflexive and symmetric relations respectively. Then $\mathrm{l}+\mathrm{m}+\mathrm{n}$ is equal to :
Q19.
mcq single
+4 / 1
Let $\mathrm{S}=\{z: 3 \leqslant|2 z-3(1+\mathrm{i})| \leqslant 7\}$ be a set of complex numbers.
Then $\operatorname{Min}_{z \in S}\left|\left(z+\frac{1}{2}(5+3 i)\right)\right|$ is equal to :
Q20.
mcq single
+4 / 1
Let the domain of the function $f(x)=\log _3 \log _5 \log _7\left(9 x-x^2-13\right)$ be the interval $(\mathrm{m}, \mathrm{n})$. Let the hyperbola $\frac{x^2}{\mathrm{a}^2}-\frac{y^2}{\mathrm{~b}^2}=1$ have eccentricity $\frac{\mathrm{n}}{3}$ and the length of the latus rectum $\frac{8 \mathrm{~m}}{3}$. Then $\mathrm{b}^2-\mathrm{a}^2$ is equal to :
Q21.
mcq single
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $x^4 \mathrm{~d} y+\left(4 x^3 y+2 \sin x\right) \mathrm{d} x=0, x>0, y\left(\frac{\pi}{2}\right)=0$.
Then $\pi^4 y\left(\frac{\pi}{3}\right)$ is equal to :
Q22.
numerical
+4 / 1
Let $f$ be a twice differentiable non-negative function such that $(f(x))^2=25+\int_0^x\left((f(\mathrm{t}))^2+\left(f^{\prime}(\mathrm{t})\right)^2\right) \mathrm{dt}$. Then the mean of $f\left(\log _{\mathrm{e}}(1)\right), f\left(\log _{\mathrm{e}}(2)\right), \ldots . ., f\left(\log _{\mathrm{e}}(625)\right)$ is equal to $\_\_\_\_$ .
Q23.
mcq single
+4 / 1
Let the line $y-x=1$ intersect the ellipse $\frac{x^2}{2}+\frac{y^2}{1}=1$ at the points A and B . Then the angle made by the line segment AB at the center of the ellipse is :
Q24.
numerical
+4 / 1
The number of 4 -letter words, with or without meaning, which can be formed using the letters PQRPQRSTUVP, is $\_\_\_\_$ .
Q25.
mcq single
+4 / 1
The value of the integral $\int_{\frac{\pi}{24}}^{\frac{5 \pi}{24}} \frac{\mathrm{~d} x}{1+\sqrt[3]{\tan 2 x}}$ is :
Q26.
mcq single
+4 / 1
Let the mean and variance of 8 numbers $-10,-7,-1, x, y, 9,2,16$ be $\frac{7}{2}$ and $\frac{293}{4}$, respectively.
Then the mean of 4 numbers $x, y, x+y+1,|x-y|$ is :
Q27.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=-\hat{i}+\hat{j}+2 \hat{k}, \overrightarrow{\mathrm{~b}}=\hat{i}-\hat{j}-3 \hat{k}, \overrightarrow{\mathrm{c}}=\overrightarrow{\mathrm{a}} \times \overrightarrow{\mathrm{b}}$ and $\overrightarrow{\mathrm{d}}=\overrightarrow{\mathrm{c}} \times \overrightarrow{\mathrm{a}}$. Then $(\vec{a}-\vec{b}) \cdot \vec{d}$ is equal to :
Q28.
mcq single
+4 / 1
A building construction work can be completed by two masons A and B together in 22.5 days. Mason A alone can complete the construction work in 24 days less than mason B alone. Then mason A alone will complete the construction work in :
Q29.
mcq single
+4 / 1
If $\alpha$ and $\beta(\alpha<\beta)$ are the roots of the equation $(-2+\sqrt{3})(|\sqrt{x}-3|)+(x-6 \sqrt{x})+(9-2 \sqrt{3})=0, x \geqslant 0$, then $\sqrt{\frac{\beta}{\alpha}}+\sqrt{\alpha \beta}$ is equal to :
Q30.
numerical
+4 / 1
From the first 100 natural numbers, two numbers first $a$ and then $b$ are selected randomly without replacement. If the probability that $\mathrm{a}-\mathrm{b} \geqslant 10$ is $\frac{\mathrm{m}}{\mathrm{n}}, \operatorname{gcd}(\mathrm{m}, \mathrm{n})=1$, then $\mathrm{m}+\mathrm{n}$ is equal to
$\_\_\_\_$ .
Q31.
mcq single
+4 / 1
The vertices B and C of a triangle ABC lie on the line $\frac{x}{1}=\frac{1-y}{-2}=\frac{\mathrm{z}-2}{3}$. The coordinates of A and $B$ are $(1,6,3)$ and $(4,9, \alpha)$ respectively and $C$ is at a distance of 10 units from $B$. The area (in sq. units) of $\triangle A B C$ is :
Q32.
mcq single
+4 / 1
Let the direction cosines of two lines satisfy the equations : $4 l+m-n=0$ and $2 m n+10 n l+3 l m=0$.
Then the cosine of the acute angle between these lines is :
Q33.
mcq single
+4 / 1
Let $f(x)= \begin{cases}\frac{\mathrm{a} x^2+2 \mathrm{a} x+3}{4 x^2+4 x-3} & , x \neq-\frac{3}{2}, \frac{1}{2} \\ \mathrm{~b} & , x=-\frac{3}{2}, \frac{1}{2}\end{cases}$ be continuous at $x=-\frac{3}{2}$. If $f \circ f(x)=\frac{7}{5}$, then $x$ is equal to:
Q34.
mcq single
+4 / 1
Let $\alpha$ and $\beta$ respectively be the maximum and the minimum values of the function $f(\theta)=4\left(\sin ^4\left(\frac{7 \pi}{2}-\theta\right)+\sin ^4(11 \pi+\theta)\right)-2\left(\sin ^6\left(\frac{3 \pi}{2}-\theta\right)+\sin ^6(9 \pi-\theta)\right), \theta \in \mathbf{R}$.
Then $\alpha+2 \beta$ is equal to :
Q35.
numerical
+4 / 1
Let the area of the region bounded by the curve $y=\max \{\sin x, \cos x\}$, lines $x=0, x=\frac{3 \pi}{2}$, and the $x$-axis be A . Then, $\mathrm{A}+\mathrm{A}^2$ is equal to $\_\_\_\_$。
Q36.
mcq single
+4 / 1
Among the statements :
I: If $\left|\begin{array}{ccc}1 & \cos \alpha & \cos \beta \\ \cos \alpha & 1 & \cos \gamma \\ \cos \beta & \cos \gamma & 1\end{array}\right|=\left|\begin{array}{ccc}0 & \cos \alpha & \cos \beta \\ \cos \alpha & 0 & \cos \gamma \\ \cos \beta & \cos \gamma & 0\end{array}\right|$, then $\cos ^2 \alpha+\cos ^2 \beta+\cos ^2 \gamma=\frac{3}{2}$, and
II: If $\left|\begin{array}{ccc}x^2+x & x+1 & x-2 \\ 2 x^2+3 x-1 & 3 x & 3 x-3 \\ x^2+2 x+3 & 2 x-1 & 2 x-1\end{array}\right|=\mathrm{p} x+\mathrm{q}$, then $\mathrm{p}^2=196 \mathrm{q}^2$,
Q37.
numerical
+4 / 1
Let $|\mathrm{A}|=6$, where A is a $3 \times 3$ matrix. If $\left|\operatorname{adj}\left(3\operatorname{adj}\left(\mathrm{A}^2 \cdot \operatorname{adj}(2 \mathrm{~A})\right)\right)\right|=2^{\mathrm{m}} \cdot 3^{\mathrm{n}}, \mathrm{m}, \mathrm{n} \in \mathbf{N}$, then $\mathrm{m}+\mathrm{n}$ is equal to
$\_\_\_\_$ .
Q38.
mcq single
+4 / 1
Number of solutions of $\sqrt{3} \cos 2 \theta+8 \cos \theta+3 \sqrt{3}=0, \theta \in[-3 \pi, 2 \pi]$ is :
Q39.
mcq single
+4 / 1
Let $f(x)=\int \frac{\left(2-x^2\right) \cdot \mathrm{e}^x}{(\sqrt{1+x})(1-x)^{3 / 2}} \mathrm{~d} x$. If $f(0)=0$, then $f\left(\frac{1}{2}\right)$ is equal to:
Q40.
mcq single
+4 / 1
The value of $\frac{{ }^{100} \mathrm{C}_{50}}{51}+\frac{{ }^{100} \mathrm{C}_{51}}{52}+\ldots .+\frac{{ }^{100} \mathrm{C}_{100}}{101}$ is:
Q41.
mcq single
+4 / 1
The sum of all possible values of $\mathbf{n} \in \mathbf{N}$, so that the coefficients of $x, x^2$ and $x^3$ in the expansion of $\left(1+x^2\right)^2(1+x)^{\mathrm{n}}$, are in arithmetic progression is :
Q42.
mcq single
+4 / 1
A rectangle is formed by the lines $x=0, y=0, x=3$ and $y=4$. Let the line L be perpendicular to $3 x+y+6=0$ and divide the area of the rectangle into two equal parts. Then the distance of the point $\left(\frac{1}{2},-5\right)$ from the line $L$ is equal to :
Q43.
mcq single
+4 / 1
Let $\mathrm{A}=\{-2,-1,0,1,2,3,4\}$. Let R be a relation on A defined by $x \mathrm{R} y$ if and only if $2 x+y \leqslant 2$. Let $l$ be the number of elements in R . Let m and n be the minimum number of elements required to be added in R to make it reflexive and symmetric relations respectively. Then $\mathrm{l}+\mathrm{m}+\mathrm{n}$ is equal to :
Q44.
mcq single
+4 / 1
Let $\mathrm{S}=\{z: 3 \leqslant|2 z-3(1+\mathrm{i})| \leqslant 7\}$ be a set of complex numbers.
Then $\operatorname{Min}_{z \in S}\left|\left(z+\frac{1}{2}(5+3 i)\right)\right|$ is equal to :
Q45.
mcq single
+4 / 1
Let the domain of the function $f(x)=\log _3 \log _5 \log _7\left(9 x-x^2-13\right)$ be the interval $(\mathrm{m}, \mathrm{n})$. Let the hyperbola $\frac{x^2}{\mathrm{a}^2}-\frac{y^2}{\mathrm{~b}^2}=1$ have eccentricity $\frac{\mathrm{n}}{3}$ and the length of the latus rectum $\frac{8 \mathrm{~m}}{3}$. Then $\mathrm{b}^2-\mathrm{a}^2$ is equal to :
Q46.
mcq single
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $x^4 \mathrm{~d} y+\left(4 x^3 y+2 \sin x\right) \mathrm{d} x=0, x>0, y\left(\frac{\pi}{2}\right)=0$.
Then $\pi^4 y\left(\frac{\pi}{3}\right)$ is equal to :
Q47.
numerical
+4 / 1
Let $f$ be a twice differentiable non-negative function such that $(f(x))^2=25+\int_0^x\left((f(\mathrm{t}))^2+\left(f^{\prime}(\mathrm{t})\right)^2\right) \mathrm{dt}$. Then the mean of $f\left(\log _{\mathrm{e}}(1)\right), f\left(\log _{\mathrm{e}}(2)\right), \ldots . ., f\left(\log _{\mathrm{e}}(625)\right)$ is equal to $\_\_\_\_$ .
Q48.
mcq single
+4 / 1
Let the line $y-x=1$ intersect the ellipse $\frac{x^2}{2}+\frac{y^2}{1}=1$ at the points A and B . Then the angle made by the line segment AB at the center of the ellipse is :
Q49.
numerical
+4 / 1
The number of 4 -letter words, with or without meaning, which can be formed using the letters PQRPQRSTUVP, is $\_\_\_\_$ .
Q50.
mcq single
+4 / 1
The value of the integral $\int_{\frac{\pi}{24}}^{\frac{5 \pi}{24}} \frac{\mathrm{~d} x}{1+\sqrt[3]{\tan 2 x}}$ is :
Physics
Physics
Q1.
mcq single
+4 / 1
An object is projected with kinetic energy $K$ from a point $A$ at an angle $60^{\circ}$ with the horizontal. The ratio of the difference in kinetic energies at points $B$ and $C$ to that at point $A$ (see figure), in the absence of air friction is :
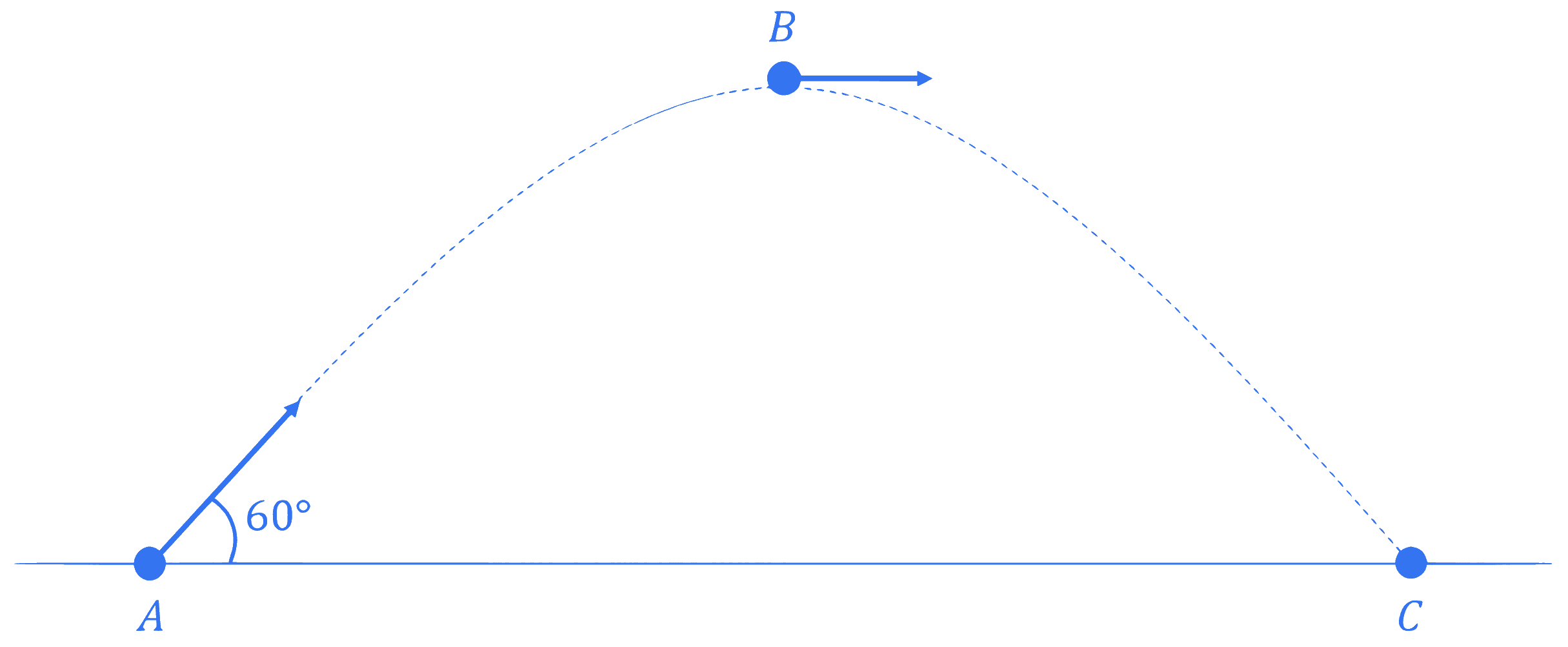

Q2.
mcq single
+4 / 1
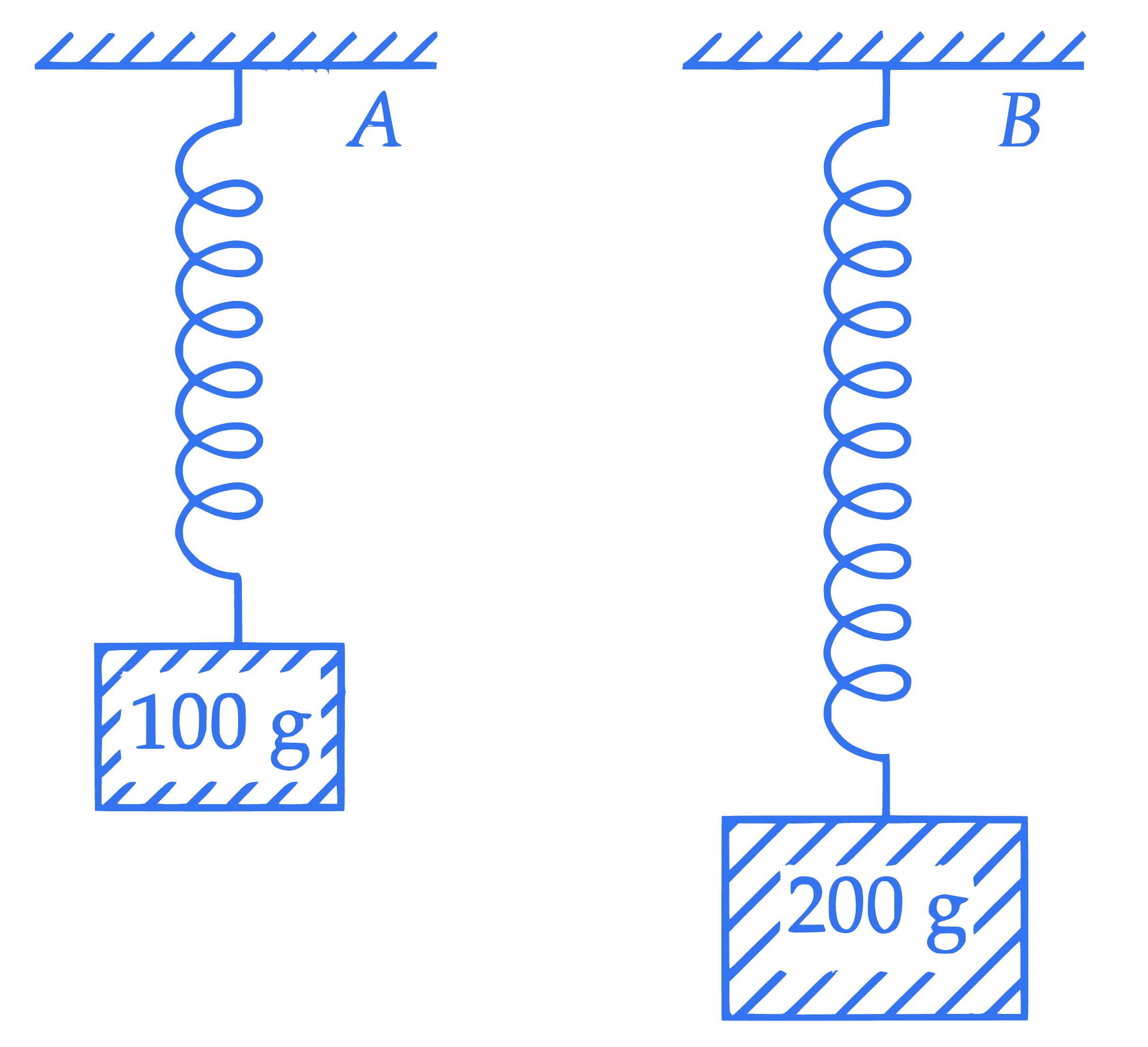
Two blocks with masses 100 g and 200 g are attached to the ends of springs $A$ and $B$ as shown in figure. The energy stored in $A$ is $E$. The energy stored in $B$, when spring constants $k_A, k_B$ of $A$ and $B$, respectively satisfy the relation $4 k_A=3 k_B$, is :

Q3.
numerical
+4 / 1
In two separate Young's double-slit experimental set-ups and two monochromatic light sources of different wavelengths are used to get fringes of equal width. The ratios of the slits separations and that of the wavelengths of light used are $ 2 : 1 $ and $1: 2$ respectively. The corresponding ratio of the distances between the slits and the respective screens ( $D_1 / D_2$ ) is $\_\_\_\_$。
Q4.
mcq single
+4 / 1
A simple pendulum of string length 30 cm performs 20 oscillations in 10 s . The length of the string required for the pendulum to perform 40 oscillations in the same time duration is
$\_\_\_\_$ cm . [Assume that the mass of the pendulum remains same.]
Q5.
mcq single
+4 / 1
Two small balls with masses $m$ and 2 m are attached to both ends of a rigid rod of length $d$ and negligible mass. If angular momentum of this system is $L$ about an axis (A) passing through its centre of mass and perpendicular to the rod then angular velocity of the system about $A$ is :
Q6.
mcq single
+4 / 1
The moment of inertia of a square loop made of four uniform solid cylinders, each having radius $R$ and length $L(\mathrm{R}<\mathrm{L})$ about an axis passing through the mid points of opposite sides, is (Take the mass of the entire loop as $M$ ) :
Q7.
mcq single
+4 / 1
In a perfectly inelastic collision, two spheres made of the same material with masses 15 kg and 25 kg , moving in opposite directions with speeds of $10 \mathrm{~m} / \mathrm{s}$ and $30 \mathrm{~m} / \mathrm{s}$, respectively, strike each other and stick together. The rise in temperature (in ${ }^{\circ} \mathrm{C}$ ), if all the heat produced during the collision is retained by these spheres, is :
(specific heat of sphere material $31 \mathrm{cal} / \mathrm{kg} .{ }^{\circ} \mathrm{C}$ and $1 \mathrm{cal}=4.2 \mathrm{~J}$ )
Q8.
mcq single
+4 / 1
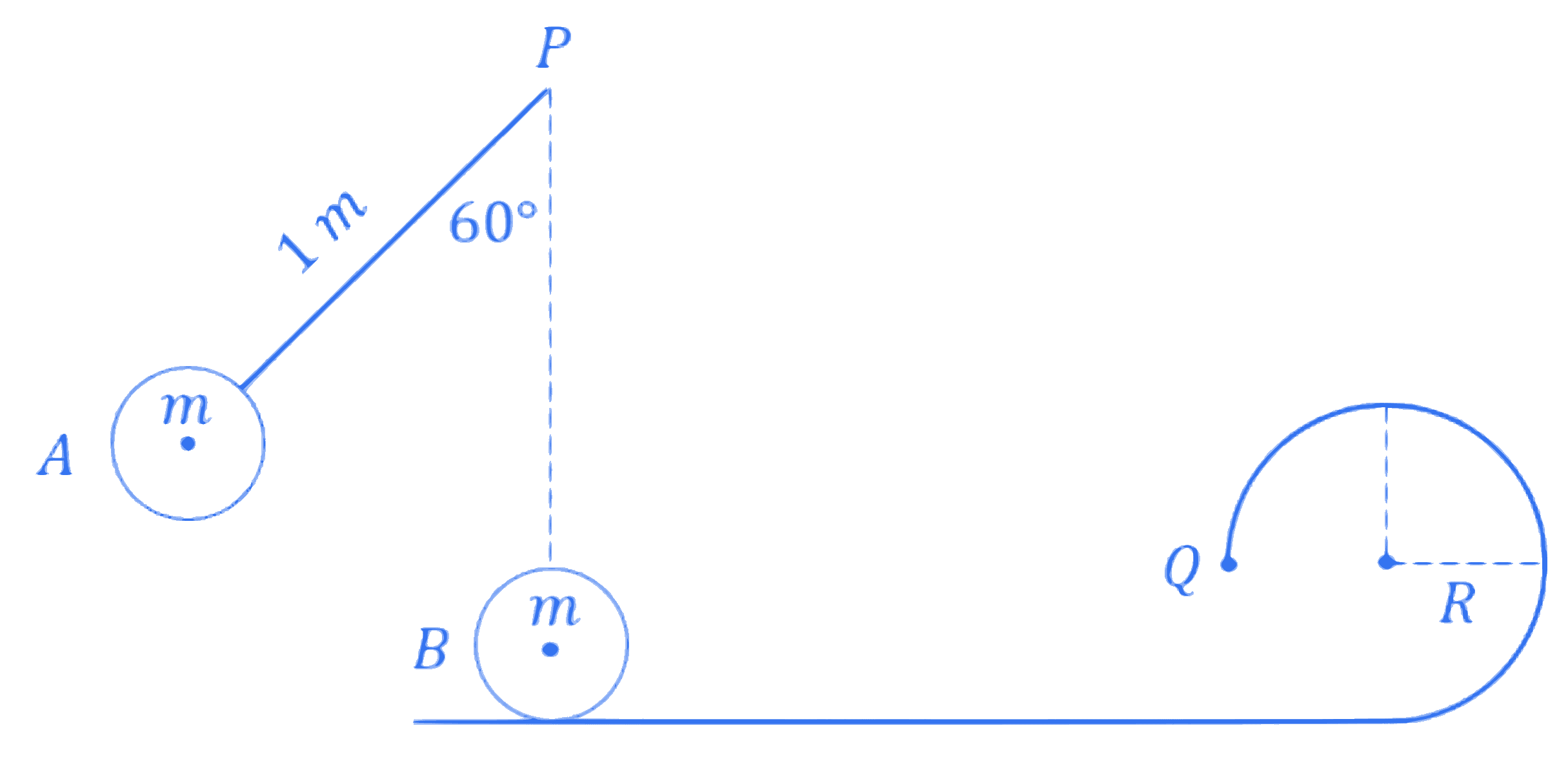
A small bob $A$ of mass $m$ is attached to a massless rigid rod of length 1 m pivoted at point $P$ and kept at an angle of $60^{\circ}$ with vertical as shown in figure. At distance of 1 m below point $P$, an identical bob $B$ is kept at rest on a smooth horizontal surface that extends to a circular track of radius $R$ as shown in figure. If bob $B$ just manages to complete the circular path of radius $R$ upto a point $Q$ after being hit elastically by $\operatorname{bob} A$, then radius $R$ is $\_\_\_\_$ m.

Q9.
numerical
+4 / 1
A simple pendulum made of mass 10 g and a metallic wire of length 10 cm is suspended vertically in a uniform magnetic field of 2 T . The magnetic field direction is perpendicular to the plane of oscillations of the pendulum. If the pendulum is released from an angle of $60^{\circ}$ with vertical, then maximum induced EMF between the point of suspension and point of oscillation is
$\_\_\_\_$ mV . (Take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q10.
mcq single
+4 / 1
A 20 m long uniform copper wire held horizontally is allowed to fall under the gravity $\left(g=10 \mathrm{~m} / \mathrm{s}^2\right)$ through a uniform horizontal magnetic field of 0.5 Gauss perpendicular to the length of the wire. The induced EMF across the wire when it travells a vertical distance of 200 m is $\_\_\_\_$ mV .
Q11.
mcq single
+4 / 1
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Consider a ferromagnetic material :
**Assertion (A) :** The individual atoms in a ferromagnetic material possess a magnetic dipole moment and interact with one another in such a way that they spontaneously align themselves forming domains.
**Reason (R) :** At high enough temperature, the domain structure of ferromagnetic material disintegrates. Thus, magnetization will disappear at high enough temperature known as Curie temperature.
In the light of the above statements, choose the correct answer from the options given below :
Q12.
mcq single
+4 / 1
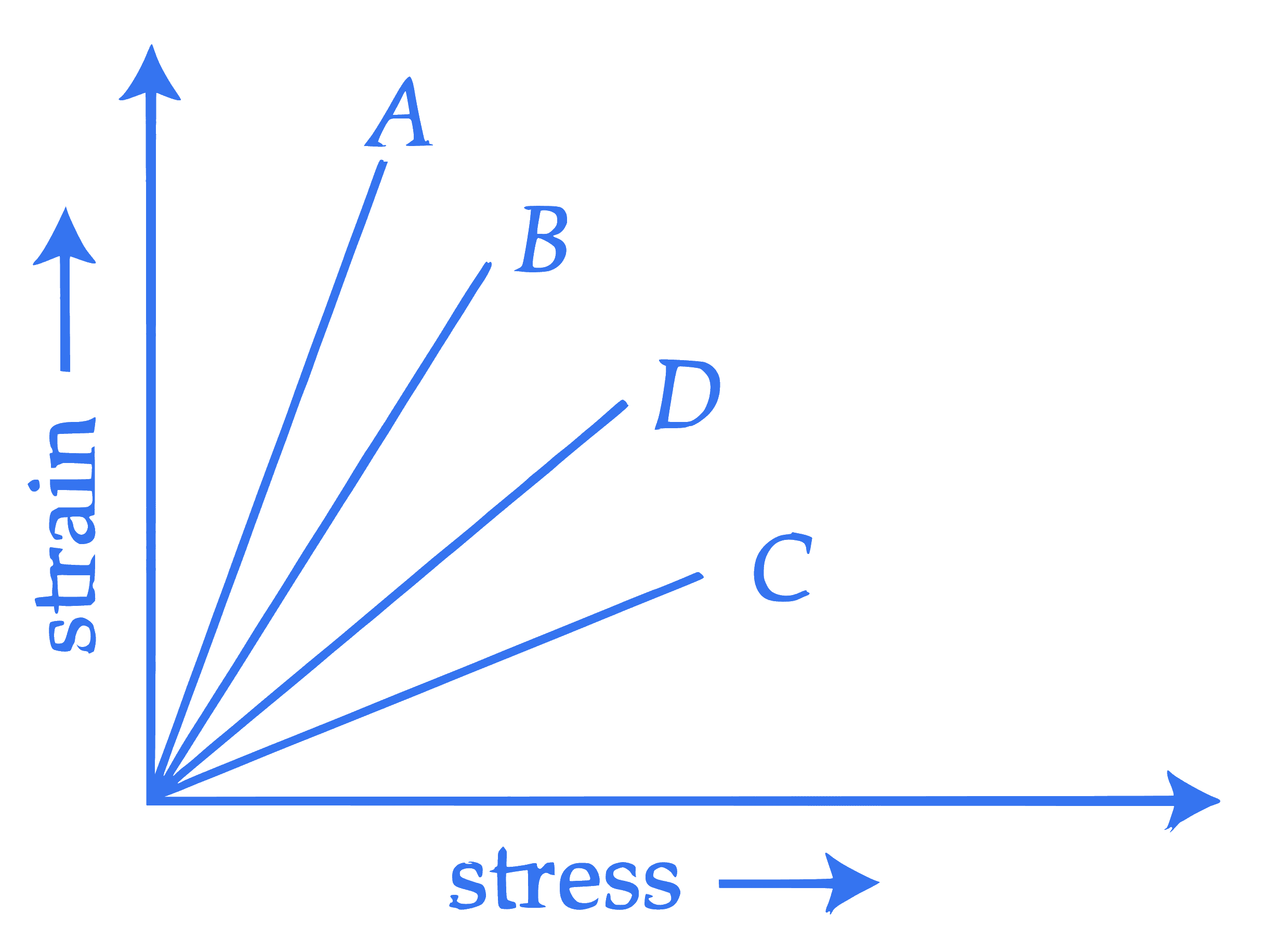
The strain-stress plot for materials $A, B, C$ and $D$ is shown in the figure. Which material has the largest Young's modulus ?

Q13.
mcq single
+4 / 1
The de Broglie wavelength of an oxygen molecule at $27^{\circ} \mathrm{C}$ is $x \times 10^{-12} \mathrm{~m}$. The value of $x$ is (take Planck's constant $=6.63 \times 10^{-34} \mathrm{~J} . \mathrm{s}$, Boltzmann constant $=1.38 \times 10^{-23} \mathrm{~J} / \mathrm{K}$, mass of oxygen molecule $=5.31 \times 10^{-26} \mathrm{~kg}$ )
Q14.
mcq single
+4 / 1
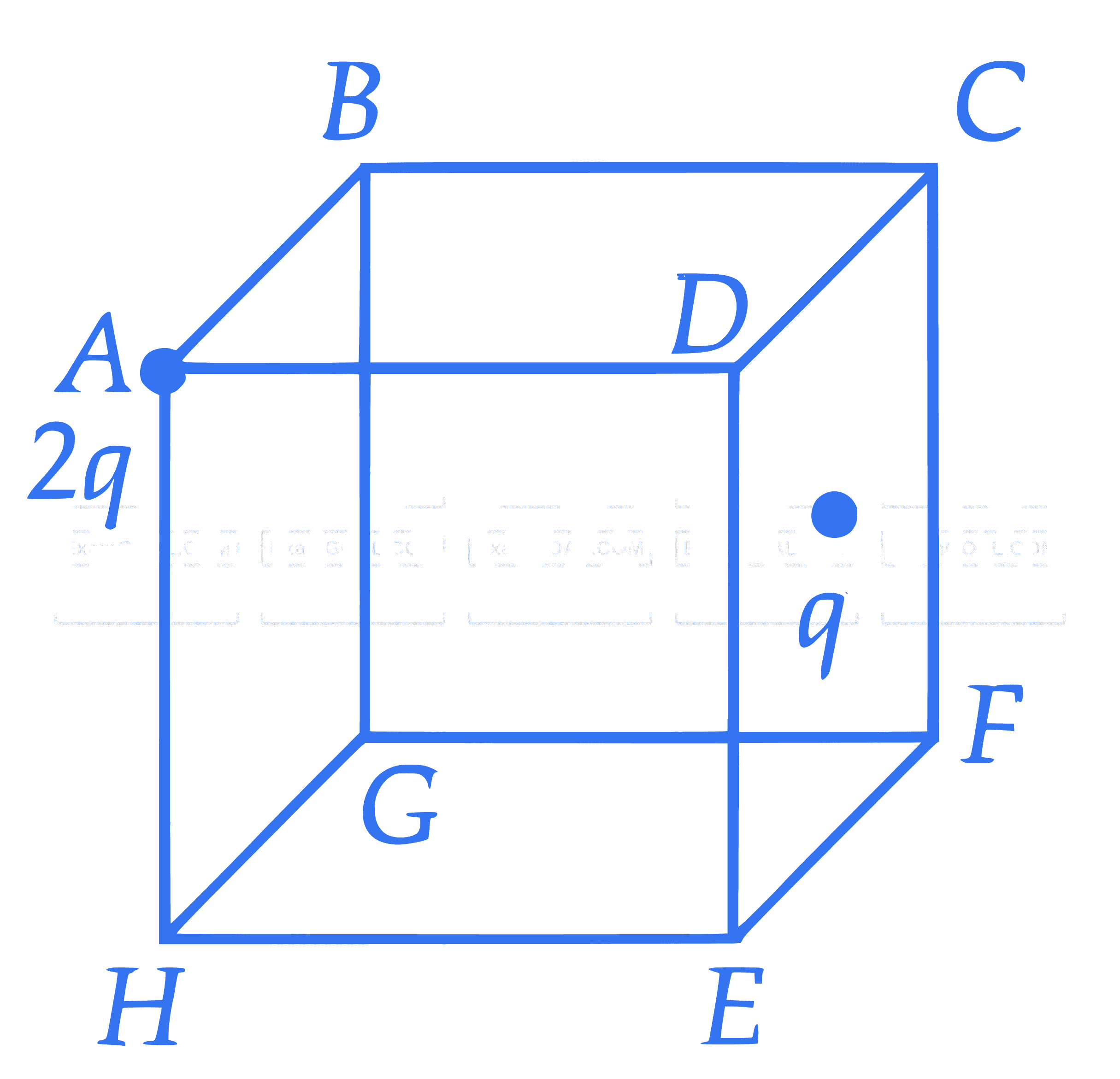
Two point charges $2 q$ and $q$ are placed at vertex $A$ and centre of face $C D E F$ of the cube as shown in figure. The electric flux passing through the cube is:

Q15.
numerical
+4 / 1
The space between the plates of a parallel plate capacitor of capacitance C (without any dielectric) is now filled with three dielectric slabs of dielectric constants $K_1=2, K_2=3$ and $K_3=5$ (as shown in figure). If new capacitance is $\frac{n}{3} C$ then the value of $n$ is $\_\_\_\_$ .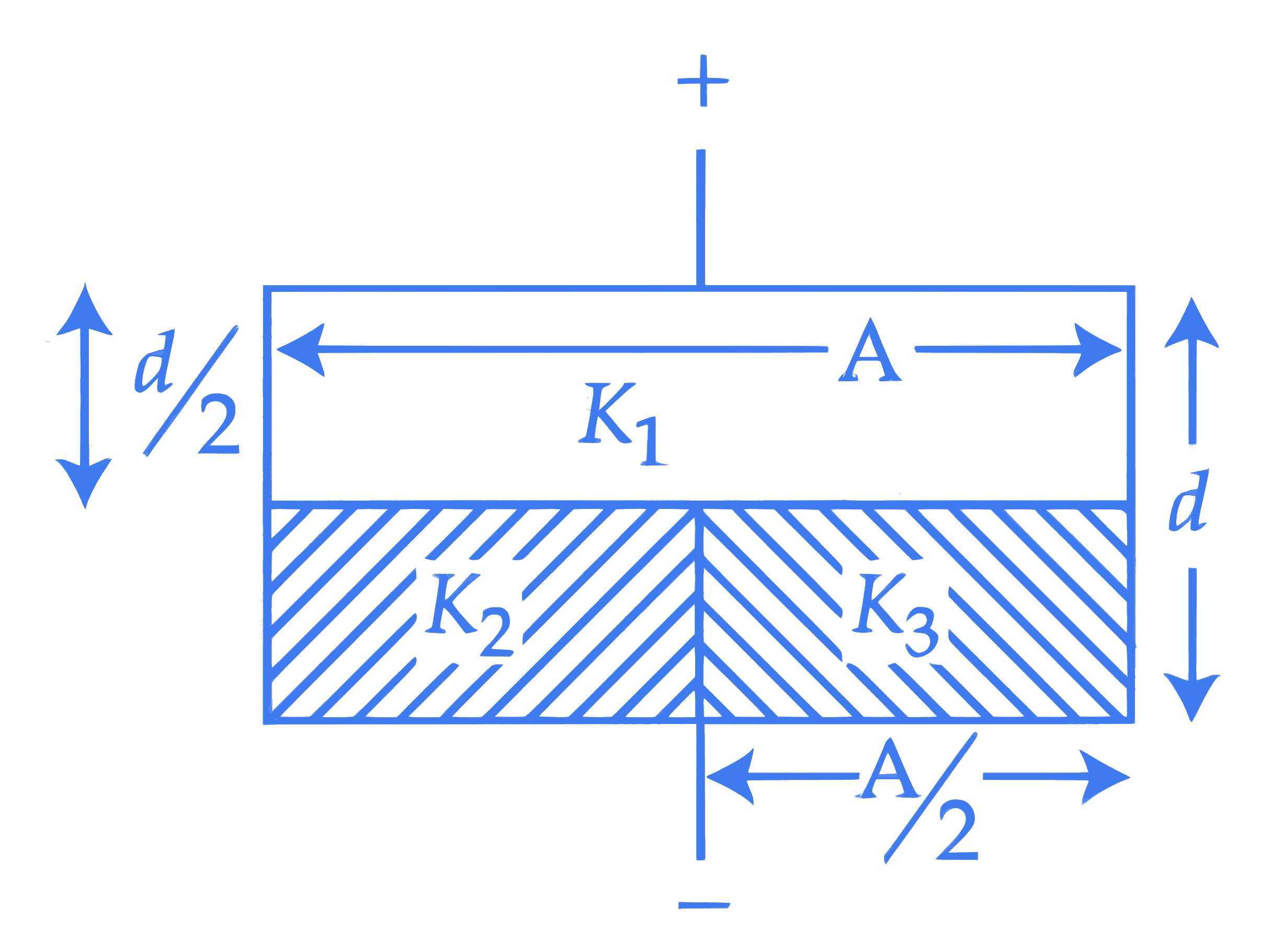

Q16.
mcq single
+4 / 1
Consider light travelling from a medium $A$ to medium $B$ separated by a plane interface. If the light undergoes total internal reflection during its travel from medium $A$ to $B$ and the speed of light in media $A$ and $B$ are $2.4 \times 10^8 \mathrm{~m} / \mathrm{s}$ and $2.7 \times 10^8 \mathrm{~m} / \mathrm{s}$, respectively, then the value of critical angle is :
Q17.
mcq single
+4 / 1
A thin prism with angle $5^{\circ}$ of refractive index 1.72 is combined with another prism of refractive index 1.9 to produce dispersion without deviation. The angle of second prism is $\_\_\_\_$ .
Q18.
mcq single
+4 / 1
Four persons measure the length of a rod as $20.00 \mathrm{~cm}, 19.75 \mathrm{~cm}, 17.01 \mathrm{~cm}$ and 18.25 cm . The relative error in the measurement of average length of the rod is :
Q19.
mcq single
+4 / 1
In a screw gauge, the zero of the circular scale lies 3 divisions above the horizontal pitch line when their metallic studs are brought in contact. Using this instrument thickness of a sheet is measured. If pitch scale reading is 1 mm and the circular scale reading is 51 then the correct thickness of the sheet is $\_\_\_\_$ mm.
[Assume least count is 0.01 mm ]
Q20.
mcq single
+4 / 1
In hydrogen atom spectrum, ( $R \rightarrow$ Rydberg's constant)
A. the maximum wavelength of the radiation of Lyman series is $\frac{4}{3 R}$
B. the Balmer series lies in the visible region of the spectrum
C. the minimum wavelength of the radiation of Paschen series is $\frac{9}{R}$
D. the minimum wavelength of Lyman series is $\frac{5}{4 R}$
Choose the correct answer from the options given below :
Q21.
mcq single
+4 / 1
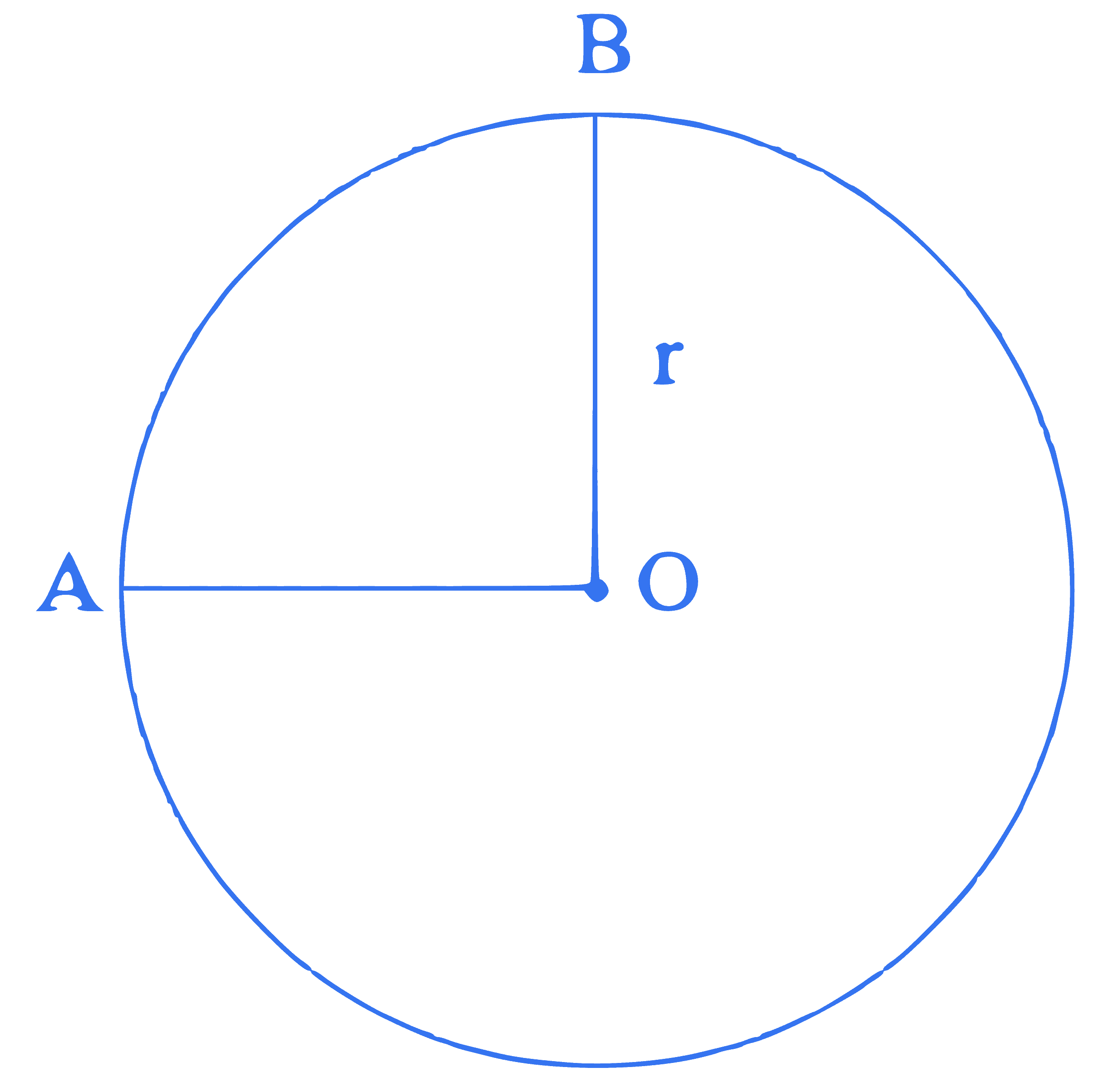
A wire of uniform resistance $\lambda \Omega / \mathrm{m}$ is bent into a circle of radius $r$ and another piece of wire with length $2 r$ is connected between points $A$ and $B(\mathrm{AOB})$ as shown in figure. The equivalent resistance between points $A$ and $B$ is $\_\_\_\_$ $\Omega$.

Q22.
numerical
+4 / 1
The equation of the electric field of an electromagnetic wave propagating through free space is given by : $E=\sqrt{377} \sin \left(6.27 \times 10^3 t-2.09 \times 10^{-5} x\right) \mathrm{N} / \mathrm{C}$
The average power of the electromagnetic wave is $\left(\frac{1}{\alpha}\right) \mathrm{W} / \mathrm{m}^2$. The value of $\alpha$ is
$$ \left(\text { Take } \sqrt{\frac{\mu_0}{\varepsilon_o}}=377 \text { in SI units }\right) $$
Q23.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Relation
List - II
Law
A.
$$
\oint \vec{E} \cdot \overrightarrow{d l}=-\frac{d}{d t} \oint \vec{B} \cdot \overrightarrow{d a}
$$
I.
Ampere's circuital law
B.
$$
\oint \vec{B} \cdot \overrightarrow{d l}=\mu_0\left(I+\epsilon_0 \frac{d \phi_E}{d t}\right)
$$
II.
Faraday's laws of electromagnetic induction
C.
$$
\oint \vec{E} \cdot \overrightarrow{d a}=\frac{1}{\epsilon_0} \int_{\mathrm{v}} \rho \mathrm{dv}
$$
III.
Ampere - Maxwell law
D.
$$
\oint \vec{B} \cdot \overrightarrow{d l}=\mu_0 I
$$
IV.
Gauss's law of electrostatics
Choose the correct answer from the options given below :
Q24.
numerical
+4 / 1
Using a variable frequency a.c. voltage source the maximum current measured in the given LCR circuit is 50 mA for $V=5 \sin (100 t)$ The values of $L$ and $R$ are shown in the figure. The capacitance of the capacitor ( C ) used is $\_\_\_\_$ $\mu \mathrm{F}$.
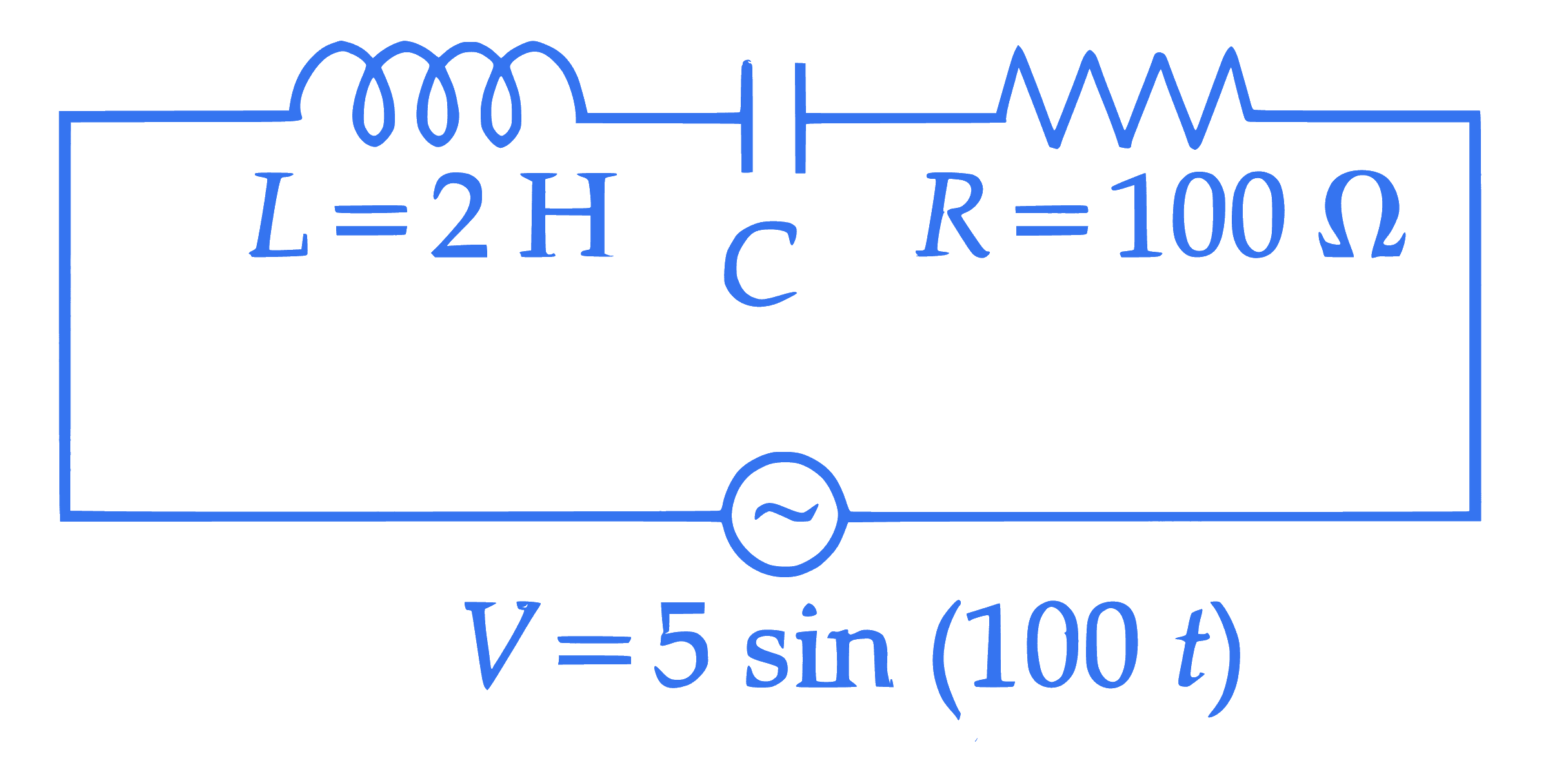
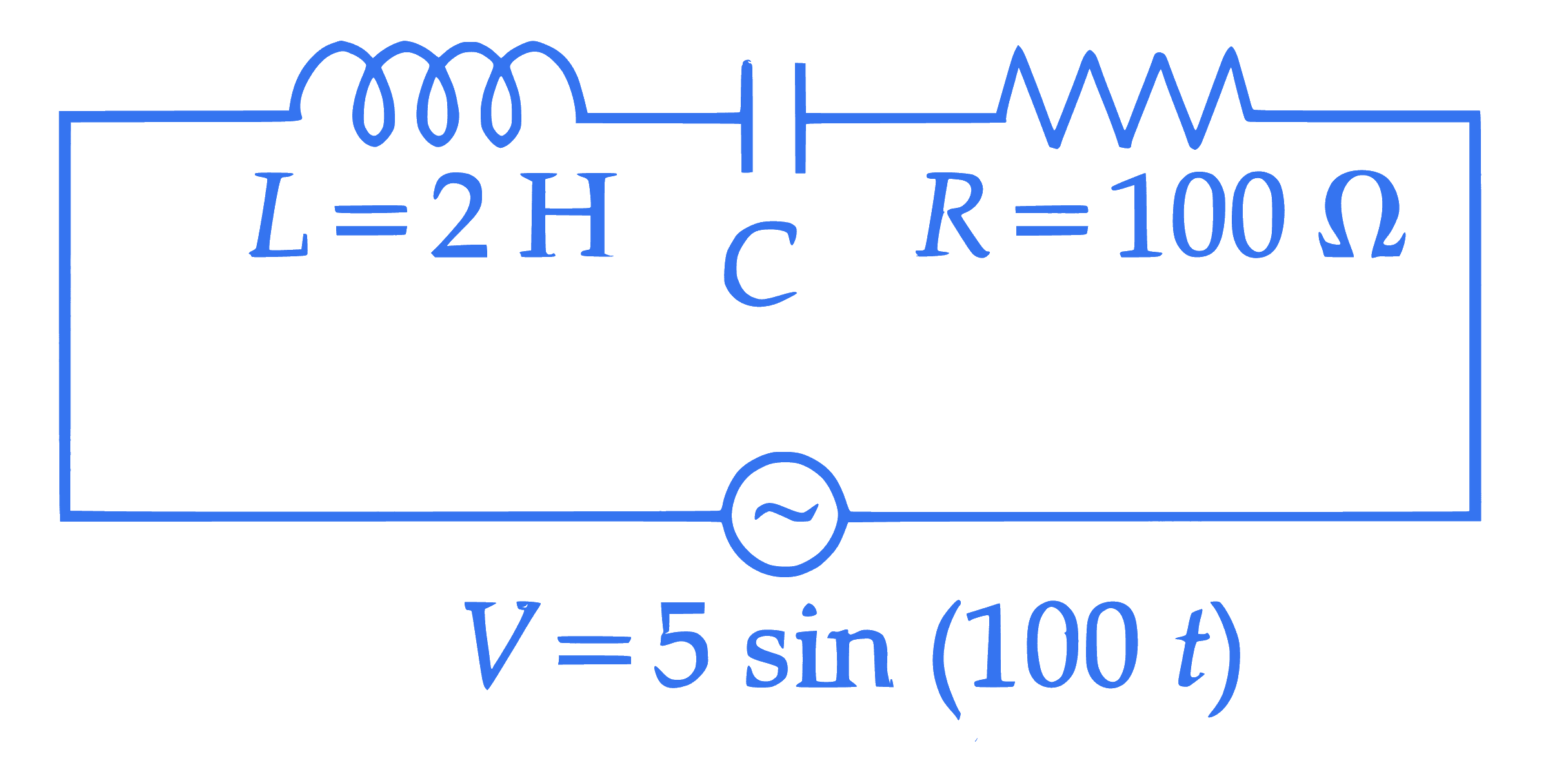
Q25.
mcq single
+4 / 1
An object is projected with kinetic energy $K$ from a point $A$ at an angle $60^{\circ}$ with the horizontal. The ratio of the difference in kinetic energies at points $B$ and $C$ to that at point $A$ (see figure), in the absence of air friction is :


Q26.
mcq single
+4 / 1
Two blocks with masses 100 g and 200 g are attached to the ends of springs $A$ and $B$ as shown in figure. The energy stored in $A$ is $E$. The energy stored in $B$, when spring constants $k_A, k_B$ of $A$ and $B$, respectively satisfy the relation $4 k_A=3 k_B$, is :


Q27.
numerical
+4 / 1
In two separate Young's double-slit experimental set-ups and two monochromatic light sources of different wavelengths are used to get fringes of equal width. The ratios of the slits separations and that of the wavelengths of light used are $ 2 : 1 $ and $1: 2$ respectively. The corresponding ratio of the distances between the slits and the respective screens ( $D_1 / D_2$ ) is $\_\_\_\_$。
Q28.
mcq single
+4 / 1
A simple pendulum of string length 30 cm performs 20 oscillations in 10 s . The length of the string required for the pendulum to perform 40 oscillations in the same time duration is
$\_\_\_\_$ cm . [Assume that the mass of the pendulum remains same.]
Q29.
mcq single
+4 / 1
Two small balls with masses $m$ and 2 m are attached to both ends of a rigid rod of length $d$ and negligible mass. If angular momentum of this system is $L$ about an axis (A) passing through its centre of mass and perpendicular to the rod then angular velocity of the system about $A$ is :
Q30.
mcq single
+4 / 1
The moment of inertia of a square loop made of four uniform solid cylinders, each having radius $R$ and length $L(\mathrm{R}<\mathrm{L})$ about an axis passing through the mid points of opposite sides, is (Take the mass of the entire loop as $M$ ) :
Q31.
mcq single
+4 / 1
In a perfectly inelastic collision, two spheres made of the same material with masses 15 kg and 25 kg , moving in opposite directions with speeds of $10 \mathrm{~m} / \mathrm{s}$ and $30 \mathrm{~m} / \mathrm{s}$, respectively, strike each other and stick together. The rise in temperature (in ${ }^{\circ} \mathrm{C}$ ), if all the heat produced during the collision is retained by these spheres, is :
(specific heat of sphere material $31 \mathrm{cal} / \mathrm{kg} .{ }^{\circ} \mathrm{C}$ and $1 \mathrm{cal}=4.2 \mathrm{~J}$ )
Q32.
mcq single
+4 / 1
A small bob $A$ of mass $m$ is attached to a massless rigid rod of length 1 m pivoted at point $P$ and kept at an angle of $60^{\circ}$ with vertical as shown in figure. At distance of 1 m below point $P$, an identical bob $B$ is kept at rest on a smooth horizontal surface that extends to a circular track of radius $R$ as shown in figure. If bob $B$ just manages to complete the circular path of radius $R$ upto a point $Q$ after being hit elastically by $\operatorname{bob} A$, then radius $R$ is $\_\_\_\_$ m.


Q33.
numerical
+4 / 1
A simple pendulum made of mass 10 g and a metallic wire of length 10 cm is suspended vertically in a uniform magnetic field of 2 T . The magnetic field direction is perpendicular to the plane of oscillations of the pendulum. If the pendulum is released from an angle of $60^{\circ}$ with vertical, then maximum induced EMF between the point of suspension and point of oscillation is
$\_\_\_\_$ mV . (Take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q34.
mcq single
+4 / 1
A 20 m long uniform copper wire held horizontally is allowed to fall under the gravity $\left(g=10 \mathrm{~m} / \mathrm{s}^2\right)$ through a uniform horizontal magnetic field of 0.5 Gauss perpendicular to the length of the wire. The induced EMF across the wire when it travells a vertical distance of 200 m is $\_\_\_\_$ mV .
Q35.
mcq single
+4 / 1
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Consider a ferromagnetic material :
**Assertion (A) :** The individual atoms in a ferromagnetic material possess a magnetic dipole moment and interact with one another in such a way that they spontaneously align themselves forming domains.
**Reason (R) :** At high enough temperature, the domain structure of ferromagnetic material disintegrates. Thus, magnetization will disappear at high enough temperature known as Curie temperature.
In the light of the above statements, choose the correct answer from the options given below :
Q36.
mcq single
+4 / 1
The strain-stress plot for materials $A, B, C$ and $D$ is shown in the figure. Which material has the largest Young's modulus ?


Q37.
mcq single
+4 / 1
The de Broglie wavelength of an oxygen molecule at $27^{\circ} \mathrm{C}$ is $x \times 10^{-12} \mathrm{~m}$. The value of $x$ is (take Planck's constant $=6.63 \times 10^{-34} \mathrm{~J} . \mathrm{s}$, Boltzmann constant $=1.38 \times 10^{-23} \mathrm{~J} / \mathrm{K}$, mass of oxygen molecule $=5.31 \times 10^{-26} \mathrm{~kg}$ )
Q38.
mcq single
+4 / 1
Two point charges $2 q$ and $q$ are placed at vertex $A$ and centre of face $C D E F$ of the cube as shown in figure. The electric flux passing through the cube is:


Q39.
numerical
+4 / 1
The space between the plates of a parallel plate capacitor of capacitance C (without any dielectric) is now filled with three dielectric slabs of dielectric constants $K_1=2, K_2=3$ and $K_3=5$ (as shown in figure). If new capacitance is $\frac{n}{3} C$ then the value of $n$ is $\_\_\_\_$ .

Q40.
mcq single
+4 / 1
Consider light travelling from a medium $A$ to medium $B$ separated by a plane interface. If the light undergoes total internal reflection during its travel from medium $A$ to $B$ and the speed of light in media $A$ and $B$ are $2.4 \times 10^8 \mathrm{~m} / \mathrm{s}$ and $2.7 \times 10^8 \mathrm{~m} / \mathrm{s}$, respectively, then the value of critical angle is :
Q41.
mcq single
+4 / 1
A thin prism with angle $5^{\circ}$ of refractive index 1.72 is combined with another prism of refractive index 1.9 to produce dispersion without deviation. The angle of second prism is $\_\_\_\_$ .
Q42.
mcq single
+4 / 1
Four persons measure the length of a rod as $20.00 \mathrm{~cm}, 19.75 \mathrm{~cm}, 17.01 \mathrm{~cm}$ and 18.25 cm . The relative error in the measurement of average length of the rod is :
Q43.
mcq single
+4 / 1
In a screw gauge, the zero of the circular scale lies 3 divisions above the horizontal pitch line when their metallic studs are brought in contact. Using this instrument thickness of a sheet is measured. If pitch scale reading is 1 mm and the circular scale reading is 51 then the correct thickness of the sheet is $\_\_\_\_$ mm.
[Assume least count is 0.01 mm ]
Q44.
mcq single
+4 / 1
In hydrogen atom spectrum, ( $R \rightarrow$ Rydberg's constant)
A. the maximum wavelength of the radiation of Lyman series is $\frac{4}{3 R}$
B. the Balmer series lies in the visible region of the spectrum
C. the minimum wavelength of the radiation of Paschen series is $\frac{9}{R}$
D. the minimum wavelength of Lyman series is $\frac{5}{4 R}$
Choose the correct answer from the options given below :
Q45.
mcq single
+4 / 1
A wire of uniform resistance $\lambda \Omega / \mathrm{m}$ is bent into a circle of radius $r$ and another piece of wire with length $2 r$ is connected between points $A$ and $B(\mathrm{AOB})$ as shown in figure. The equivalent resistance between points $A$ and $B$ is $\_\_\_\_$ $\Omega$.


Q46.
numerical
+4 / 1
The equation of the electric field of an electromagnetic wave propagating through free space is given by : $E=\sqrt{377} \sin \left(6.27 \times 10^3 t-2.09 \times 10^{-5} x\right) \mathrm{N} / \mathrm{C}$
The average power of the electromagnetic wave is $\left(\frac{1}{\alpha}\right) \mathrm{W} / \mathrm{m}^2$. The value of $\alpha$ is
$$ \left(\text { Take } \sqrt{\frac{\mu_0}{\varepsilon_o}}=377 \text { in SI units }\right) $$
Q47.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Relation
List - II
Law
A.
$$
\oint \vec{E} \cdot \overrightarrow{d l}=-\frac{d}{d t} \oint \vec{B} \cdot \overrightarrow{d a}
$$
I.
Ampere's circuital law
B.
$$
\oint \vec{B} \cdot \overrightarrow{d l}=\mu_0\left(I+\epsilon_0 \frac{d \phi_E}{d t}\right)
$$
II.
Faraday's laws of electromagnetic induction
C.
$$
\oint \vec{E} \cdot \overrightarrow{d a}=\frac{1}{\epsilon_0} \int_{\mathrm{v}} \rho \mathrm{dv}
$$
III.
Ampere - Maxwell law
D.
$$
\oint \vec{B} \cdot \overrightarrow{d l}=\mu_0 I
$$
IV.
Gauss's law of electrostatics
Choose the correct answer from the options given below :
Q48.
numerical
+4 / 1
Using a variable frequency a.c. voltage source the maximum current measured in the given LCR circuit is 50 mA for $V=5 \sin (100 t)$ The values of $L$ and $R$ are shown in the figure. The capacitance of the capacitor ( C ) used is $\_\_\_\_$ $\mu \mathrm{F}$.