JEE Main 2026 (Online) 24th January Morning Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
$$ \text { The correct stability order of the following diazonium salts is } $$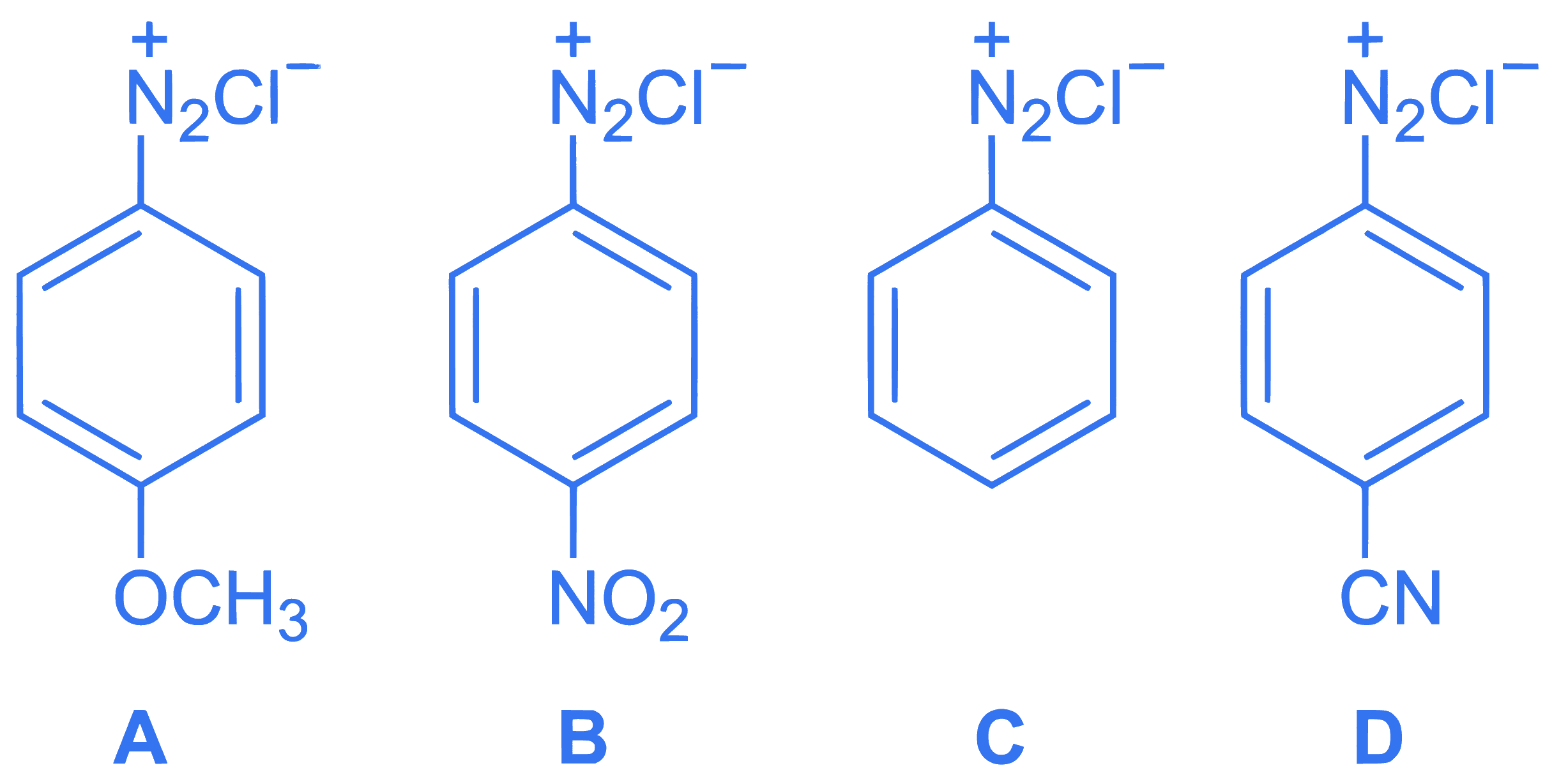

Q2.
mcq single
+4 / 1
Consider three metal chlorides $\mathrm{x}, \mathrm{y}$ and z , where x is water soluble at room temperature, y is sparingly soluble in water at room temperature and z is soluble in hot water. $\mathrm{x}, \mathrm{y}$ and z are respectively
Q3.
mcq single
+4 / 1
At $27^{\circ} \mathrm{C}$ in presence of a catalyst, activation energy of a reaction is lowered by $10 \mathrm{~kJ} \mathrm{~mol}^{-1}$. The logarithm of ratio of $\frac{\mathrm{k} \text { (catalysed) }}{\mathrm{k} \text { (uncatalysed) }}$ is….
(Consider that the frequency factor for both the reactions is same)
Q4.
mcq single
+4 / 1
$\mathrm{A} \rightarrow \mathrm{D}$ is an endothermic reaction occurring in three steps (elementary).
(i) $\mathrm{A} \rightarrow \mathrm{B} \Delta \mathrm{H}_i=+\mathrm{ve}$
(ii) $\mathrm{B} \rightarrow \mathrm{C} \Delta \mathrm{H}_{i i}=-\mathrm{ve}$
(iii) $\mathrm{C} \rightarrow \mathrm{D} \Delta \mathrm{H}_{i i i}=-\mathrm{ve}$
Which of the following graphs between potential energy ( $y$-axis) vs reaction coordinate ( $x$-axis) correctly represents the reaction profile of $A \rightarrow D$ ?
Q5.
mcq single
+4 / 1
Among the following, the CORRECT combinations are
A. $\mathrm{IF}_3 \rightarrow \mathrm{~T}$-shaped ( $\mathrm{sp}^3 \mathrm{~d}$ )
B. $\mathrm{IF}_5 \rightarrow$ Square pyramidal $\left(\mathrm{sp}^3 \mathrm{~d}^2\right)$
C. $\mathrm{IF}_7 \rightarrow$ Pentagonal bipyramidal $\left(\mathrm{sp}^3 \mathrm{~d}^3\right)$
D. $\mathrm{ClO}_4{ }^{-} \rightarrow$ Square planar ( $\mathrm{sp}^2 \mathrm{~d}$ )
Choose the correct answer from the options given below:
Q6.
mcq single
+4 / 1
Given below are statements about some molecules/ions.
Identify the CORRECT statements.
A. The dipole moment value of $\mathrm{NF}_3$ is higher than that of $\mathrm{NH}_3$.
B. The dipole moment value of $\mathrm{BeH}_2$ is zero.
C. The bond order of $\mathrm{O}_2{ }^{2-}$ and $\mathrm{F}_2$ is same.
D. The formal charge on the central oxygen atom of ozone is -1 .
E. In $\mathrm{NO}_2$, all the three atoms satisfy the octet rule, hence it is very stable.
Choose the correct answer from the options given below:
Q7.
numerical
+4 / 1
X and Y are the number of electrons involved, respectively during the oxidation of $\mathrm{I}^{-}$to $\mathrm{I}_2$ and $\mathrm{S}^{2-}$ to S by acidified $\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7$. The value of $\mathrm{X}+\mathrm{Y}$ is $\_\_\_\_$ .
Q8.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** ' $\mathrm{C}-\mathrm{Cl}$ ' bond is stronger in $\mathrm{CH}_2=\mathrm{CH}-\mathrm{Cl}$ than $\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{Cl}$
**Statement II :** The given optically active molecule, 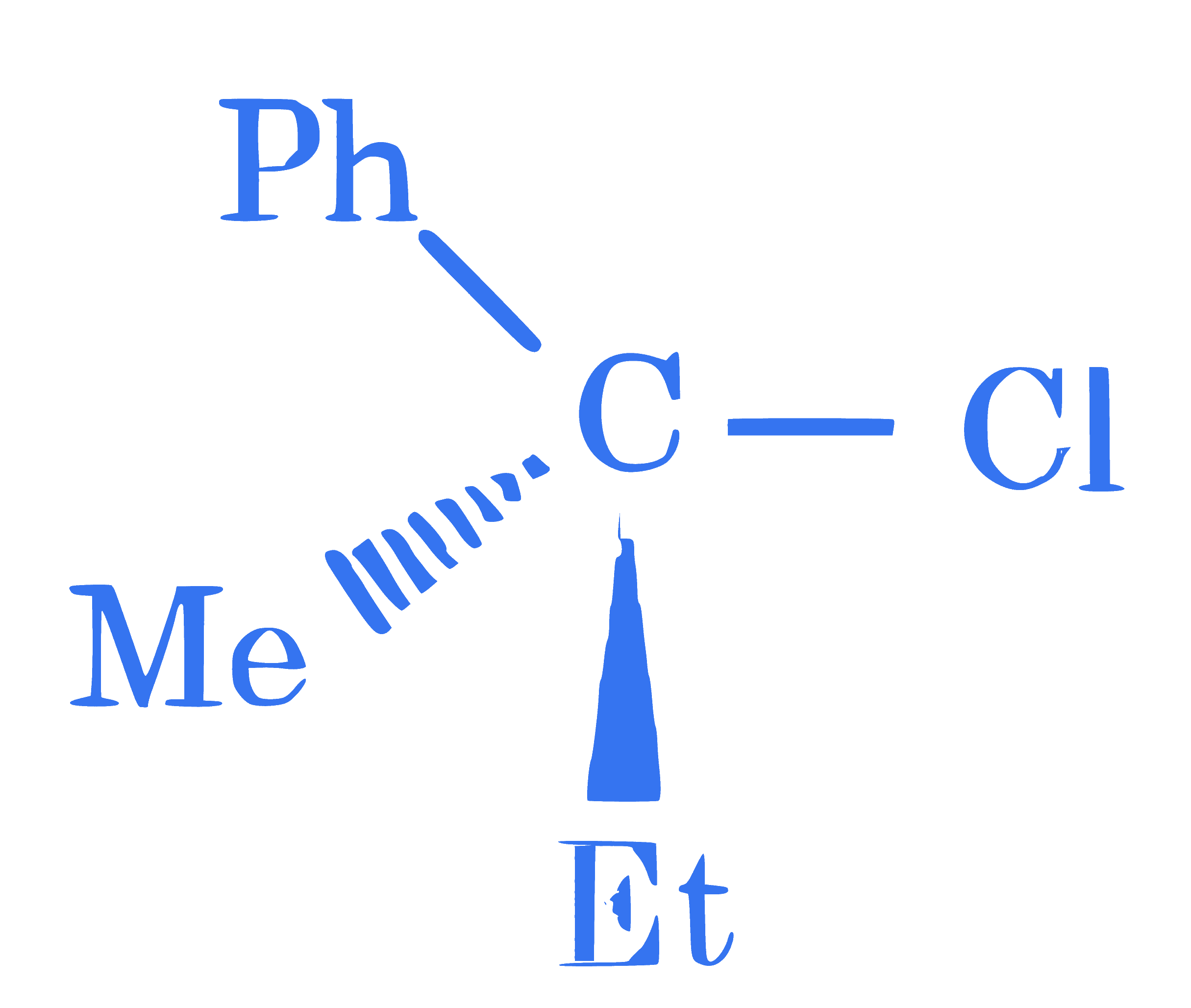 on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
 on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
Q9.
mcq single
+4 / 1
Match the List-I with List-II
List-I**Chloro derivativeList-II
ExampleA. Vinyl ChlorideI. CH$_2$ = CH - CH$_2$ClB. Benzyl ChlorideII. CH$_3$ - CH(Cl)CH$_3$C. Alkyl ChlorideIII. CH$_2$ = CHClD. Allyl ChlorideIV. 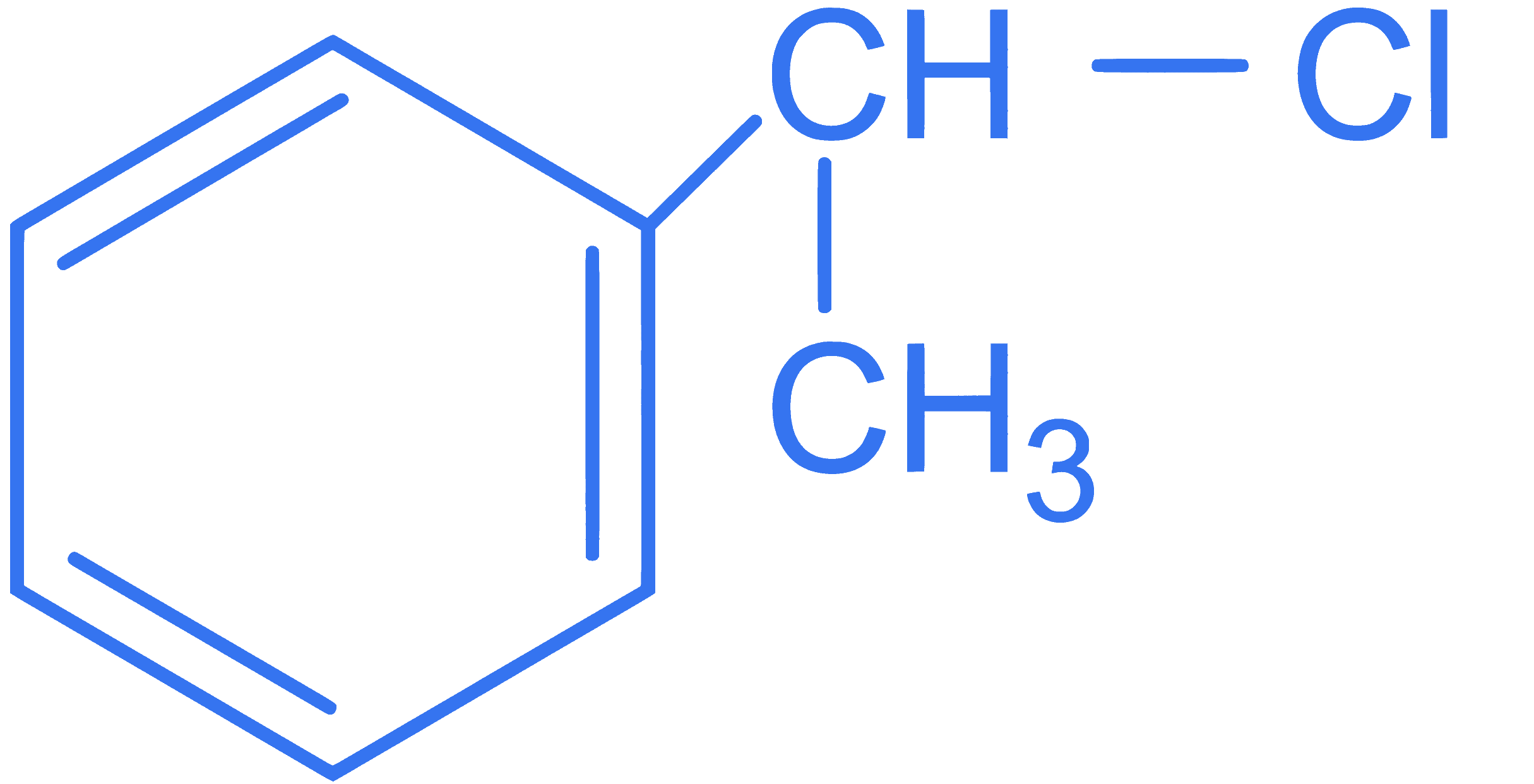 Choose the correct** answer from the options given below :
Choose the correct** answer from the options given below :
 Choose the correct** answer from the options given below :
Choose the correct** answer from the options given below :
Q10.
mcq single
+4 / 1
A solution is prepared by dissolving 0.3 g of a non-volatile non-electrolyte solute 'A' of molar mass $60 \mathrm{~g} \mathrm{~mol}^{-1}$ and 0.9 g of a non-volatile non-electrolyte solute ' B ' of molar mass $180 \mathrm{~g} \mathrm{~mol}^{-1}$ in $100 \mathrm{~mL} \mathrm{H}_2 \mathrm{O}$ at $27^{\circ} \mathrm{C}$. Osmotic pressure of the solution will be
[Given: $\mathrm{R}=0.082 \mathrm{~L} \mathrm{~atm} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ ]
Q11.
mcq single
+4 / 1
' W ' g of a non-volatile electrolyte solid solute of molar mass ' M ' $\mathrm{g} \mathrm{mol}^{-1}$ when dissolved in 100 mL water, decreases vapour pressure of water from 640 mm Hg to 600 mm Hg . If aqueous solution of the electrolyte boils at 375 K and $\mathrm{K}_{\mathrm{b}}$ for water is $0.52 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$, then the mole fraction of the electrolyte solute $\left(x_2\right)$ in the solution can be expressed as
(Given : density of water $=1 \mathrm{~g} / \mathrm{mL}$ and boiling point of water $=373 \mathrm{~K}$ )
Q12.
numerical
+4 / 1
Consider two Group IV metal ions $\mathrm{X}^{2+}$ and $\mathrm{Y}^{2+}$.
A solution containing $0.01 \mathrm{M} \mathrm{X}^{2+}$ and $0.01 \mathrm{M} \mathrm{Y}^{2+}$ is saturated with $\mathrm{H}_2 \mathrm{~S}$. The pH at which the metal sulphide YS will form as a precipitate is $\_\_\_\_$ . (Nearest integer)
(Given: $\mathrm{K}_{\mathrm{sp}}(\mathrm{XS})=1 \times 10^{-22}$ at $25^{\circ} \mathrm{C}, \mathrm{K}_{\mathrm{sp}}(\mathrm{YS})=4 \times 10^{-16}$ at $25^{\circ} \mathrm{C}$, $\left[\mathrm{H}_2 \mathrm{~S}\right]=0.1 \mathrm{M}$ in solution, $\mathrm{K}_{a 1} \times \mathrm{K}_{a 2}\left(\mathrm{H}_2 \mathrm{~S}\right)=1.0 \times 10^{-21}, \log 2=0.30$, $\log 3=0.48, \log 5=0.70)$
Q13.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\mathrm{K}>\mathrm{Mg}>\mathrm{Al}>\mathrm{B}$ is the correct order in terms of metallic character.
**Statement II :** Atomic radius is always greater than the ionic radius for any element.
In the light of the above statements, choose the correct answer from the options given below
Q14.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I Isothermal process for ideal gas system
List-II Work done (
<mi>V</mi>
<mi>f</mi>
>
<mi>V</mi>
<mi>i</mi>
<mi>V</mi>
<mi>f</mi>
>
<mi>V</mi>
<mi>i</mi>
V_(f) > V_(i) )
A.
Reversible expansion
I.
<mi mathvariant="normal">w</mi>
=
0
<mi mathvariant="normal">w</mi>
=
0
w=0
B.
Free expansion
II.
<mi mathvariant="normal">w</mi>
=
−
<mi data-mjx-auto-op="false">nR</mi>
T
ln
<msub>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mi mathvariant="normal">w</mi>
=
−
<mi data-mjx-auto-op="false">nR</mi>
T
ln
<msub>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
w=-nRT ln((V_(f))/(V_(i)))
C.
Irreversible expansion
III.
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mo data-mjx-texclass="OPEN">(</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mo data-mjx-texclass="CLOSE">)</mo>
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mrow>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
</mrow>
w=-p_(ex)(V_(f)-V_(i))
D.
Irreversible compression
IV.
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mo data-mjx-texclass="OPEN">(</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<mo data-mjx-texclass="CLOSE">)</mo>
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mrow>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
</mrow>
w=-p_(ex)(V_(i)-V_(f))
Choose the correct answer from the options given below:
Q15.
mcq single
+4 / 1
Given below are two statements:
**Statement I :** The number of paramagnetic species among $\left[\mathrm{CoF}_6\right]^{3-},\left[\mathrm{TiF}_6\right]^{3-}$, $\mathrm{V}_2 \mathrm{O}_5$ and $\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{3-}$ is 3 .
**Statement II :**
$\mathrm{K}_4\left[\mathrm{Fe}(\mathrm{CN})_6\right]<\mathrm{K}_3\left[\mathrm{Fe}(\mathrm{CN})_6\right]<\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{SO}_4 \cdot \mathrm{H}_2 \mathrm{O}<\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{Cl}_3$ is the correct order in terms of number of unpaired electron(s) present in the complexes.
In the light of the above statements, choose the correct answer from the options given below
Q16.
mcq single
+4 / 1
Consider a mixture ' X ' which is made by dissolving 0.4 mol of $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5 \mathrm{SO}_4\right] \mathrm{Br}$ and 0.4 mol of $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5 \mathrm{Br}\right] \mathrm{SO}_4$ in water to make 4 L of solution. When 2 L of mixture ' X ' is allowed to react with excess of $\mathrm{AgNO}_3$, it forms precipitate ' Y '. The rest 2 L of mixture ' X ' reacts with excess $\mathrm{BaCl}_2$ to form precipitate ' Z '. Which of the following statements is CORRECT?
Q17.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Hybridisation, shape and spin only magnetic moment of $\mathrm{K}_3\left[\mathrm{Co}\left(\mathrm{CO}_3\right)_3\right]$ is $\mathrm{sp}^3 \mathrm{~d}^2$, octahedral and 4.9 BM respectively.
**Statement II :** Geometry, hybridisation and spin only magnetic moment values $(B M)$ of the ions $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-},\left[\mathrm{MnBr}_4\right]^{2-}$ and $\left[\mathrm{CoF}_6\right]^{3-}$ respectively are square planar, tetrahedral, octahedral; $\mathrm{dsp}^2, \mathrm{sp}^3, \mathrm{sp}^3 \mathrm{~d}^2$ and $0,5.9,4.9$.
In the light of the above statements, choose the correct answer from the options given below
Q18.
mcq single
+4 / 1
$$ \text { Consider the following two reactions } \mathrm{A} \text { and } \mathrm{B} \text {. } $$
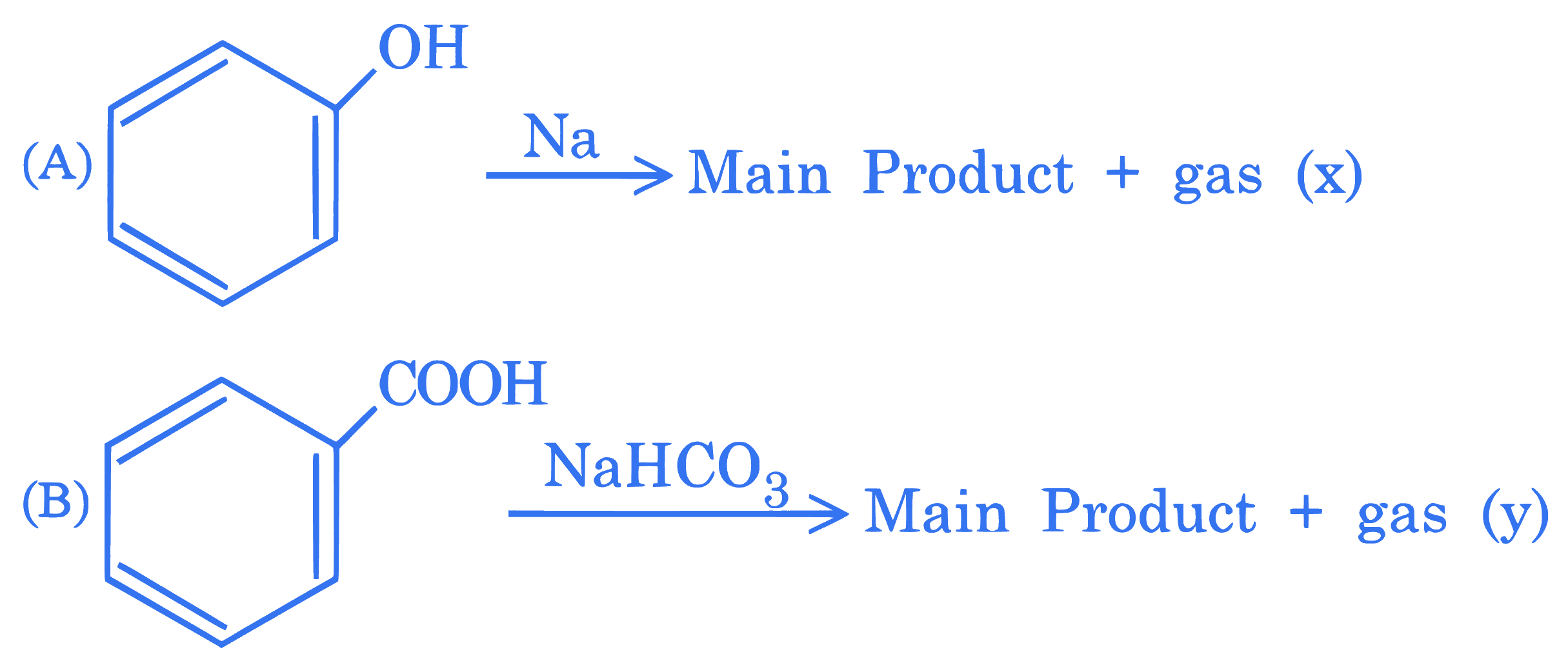 Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
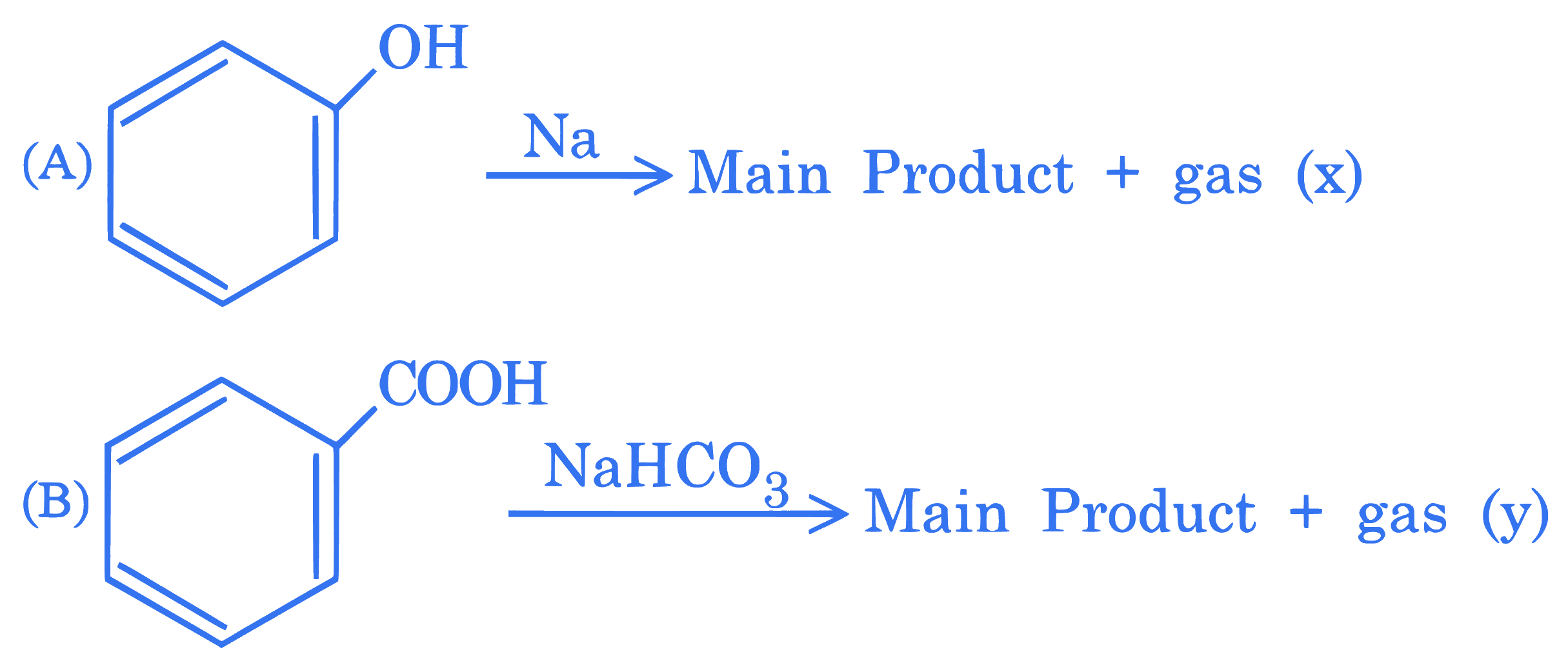 Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
Q19.
mcq single
+4 / 1
A student is given one compound among the following compounds that gives positive test with Tollen's reagent.
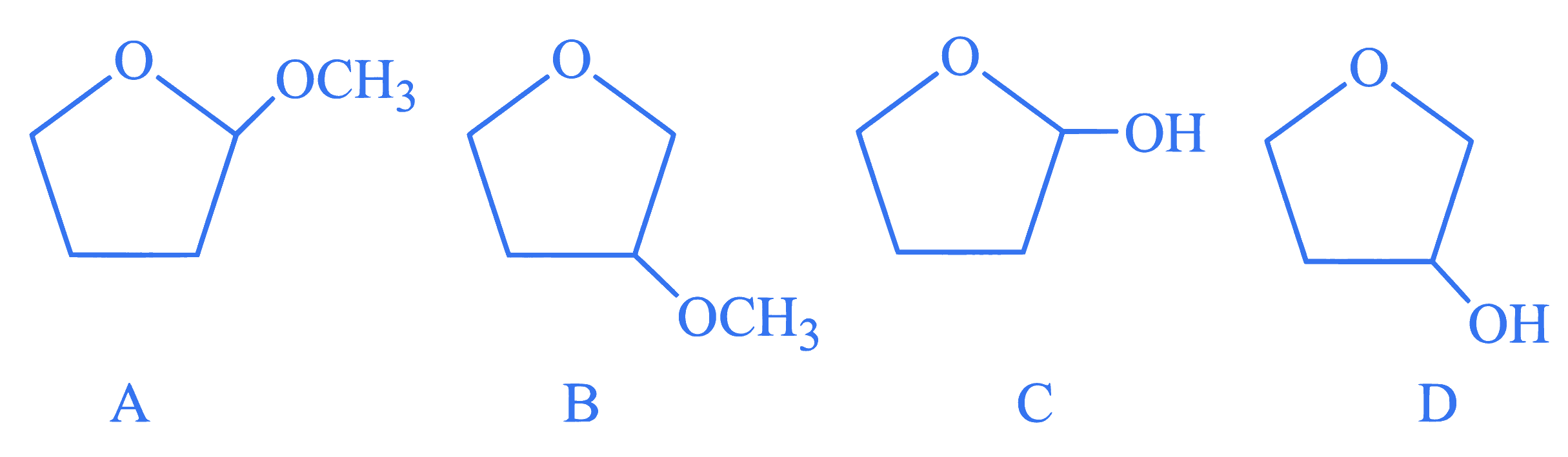 The compound is :
The compound is :
 The compound is :
The compound is :
Q20.
numerical
+4 / 1
In Dumas method for estimation of nitrogen, 0.50 g of an organic compound gave 70 mL of nitrogen collected at 300 K and 715 mm pressure. The percentage of nitrogen in the organic compound is $\_\_\_\_$ $\%$.
(Aqueous tension at 300 K is 15 mm ).
Q21.
mcq single
+4 / 1
$$ \text {Arrange the following alkenes in decreasing order of stability. } $$
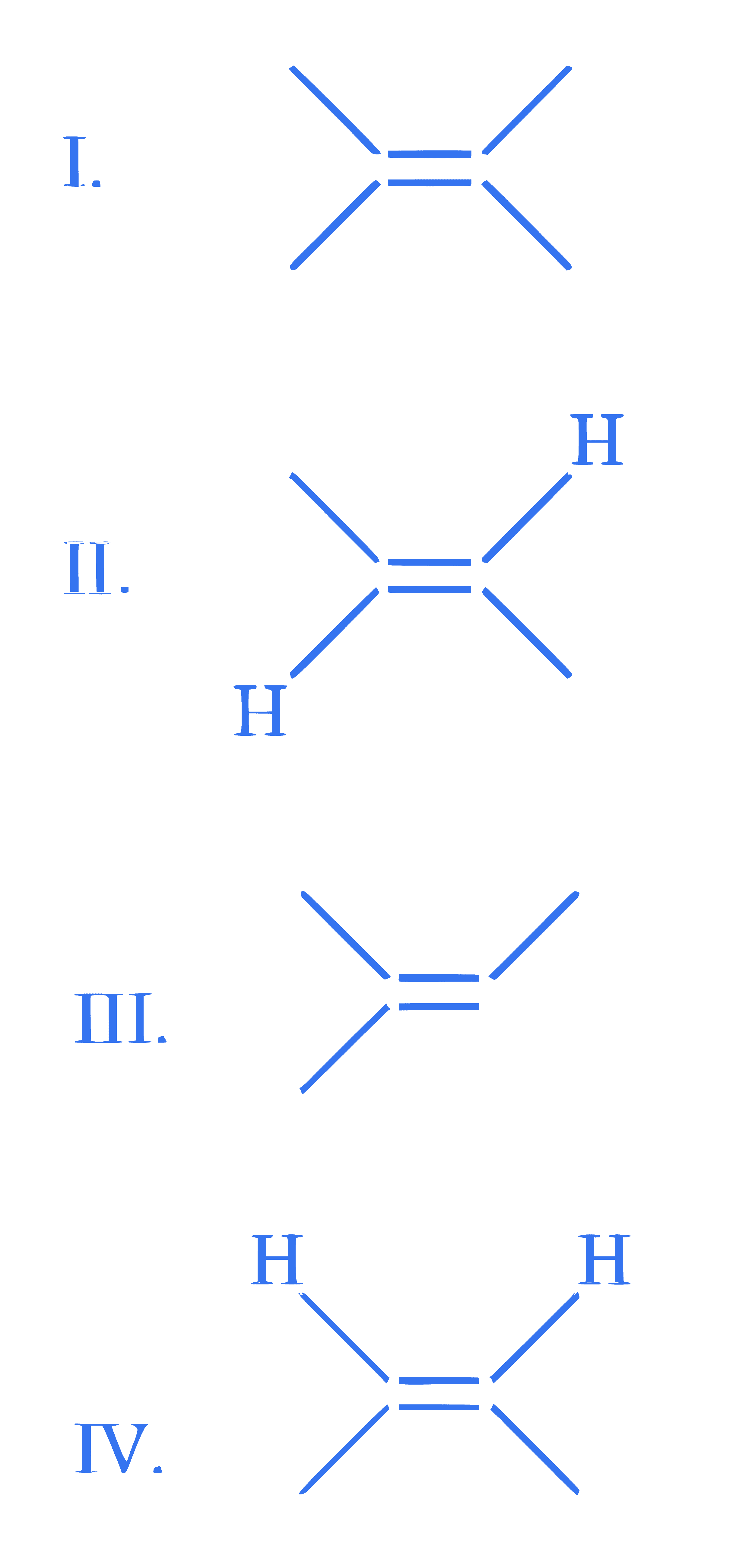 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
Q22.
mcq single
+4 / 1
$$ \text { Arrange the following carbanions in the decreasing order of stability. } $$
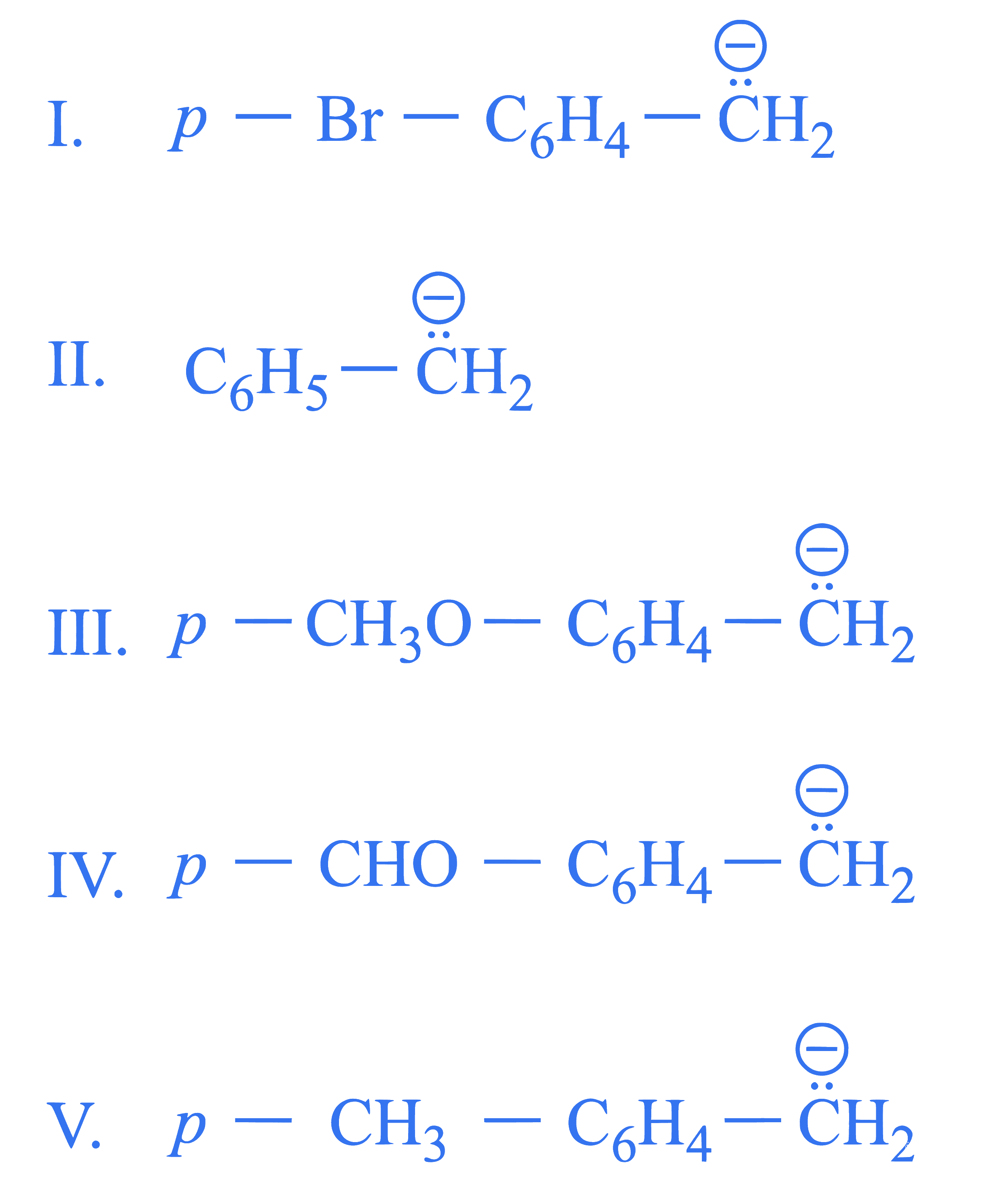 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
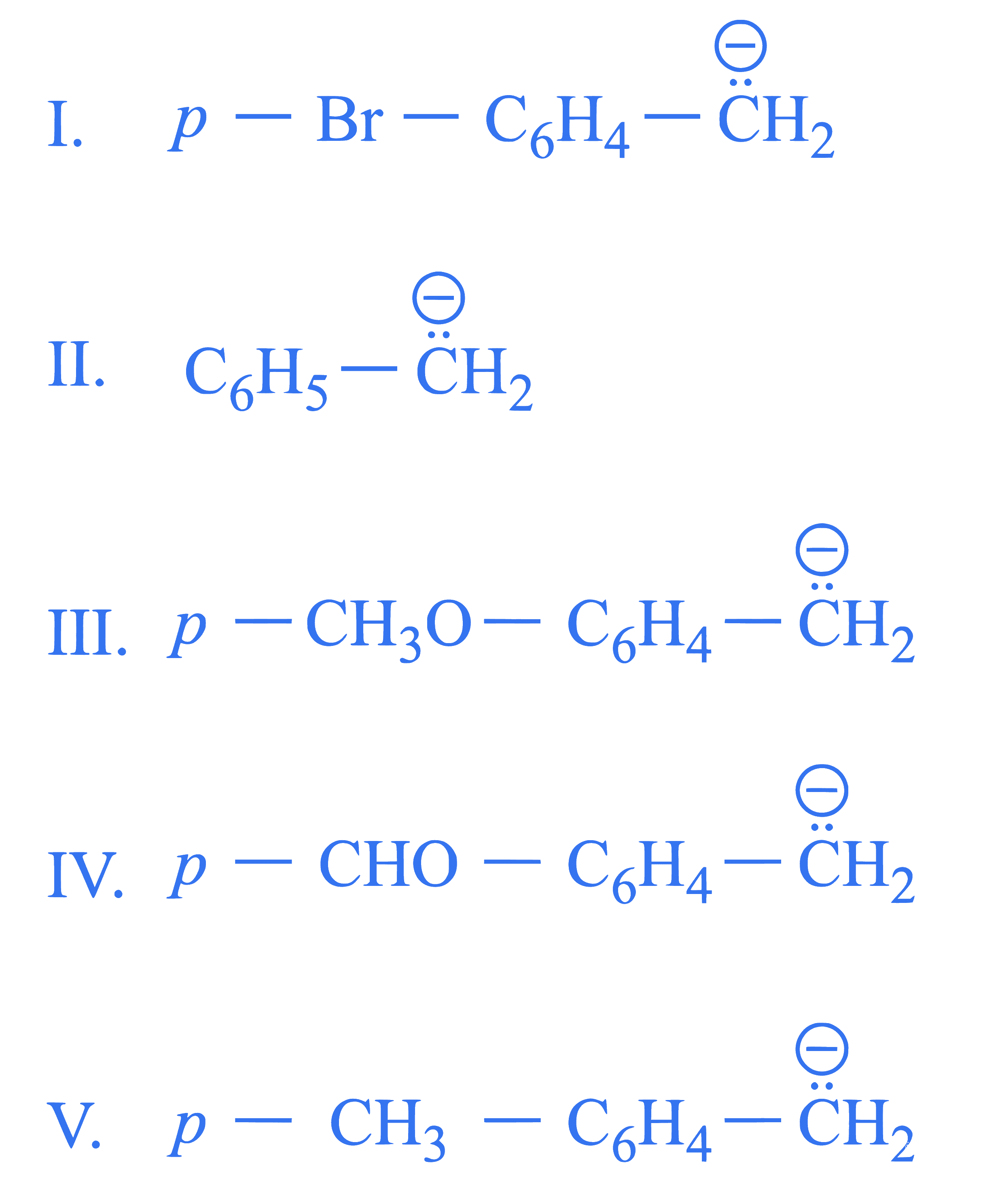 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q23.
numerical
+4 / 1
Electricity is passed through an acidic solution of $\mathrm{Cu}^{2+}$ till all the $\mathrm{Cu}^{2+}$ was exhausted, leading to the deposition of 300 mg of Cu metal. However, a current of 600 mA was continued to pass through the same solution for another 28 minutes by keeping the total volume of the solution fixed at 200 mL . The total volume of oxygen evolved at STP during the entire process is $\_\_\_\_$ mL . (Nearest integer)
[Given:
$$ \begin{aligned} & \mathrm{Cu}^{2+}(\mathrm{aq})+2 \mathrm{e}^{-} \rightarrow \mathrm{Cu}(\mathrm{~s}) \mathrm{E}_{\mathrm{red}}^{\mathrm{o}}=+0.34 \mathrm{~V} \\ & \mathrm{O}_2(\mathrm{~g})+4 \mathrm{H}^{+}+4 \mathrm{e}^{-} \rightarrow 2 \mathrm{H}_2 \mathrm{O} \mathrm{E}_{\mathrm{red}}^{\mathrm{o}}=+1.23 \mathrm{~V} \end{aligned} $$
Molar mass of $\mathrm{Cu}=63.54 \mathrm{~g} \mathrm{~mol}^{-1}$
Molar mass of $\mathrm{O}_2=32 \mathrm{~g} \mathrm{~mol}^{-1}$
Faraday Constant $=96500 \mathrm{C} \mathrm{mol}^{-1}$
Molar volume at $\mathrm{STP}=22.4 \mathrm{~L}$ ]
Q24.
mcq single
+4 / 1
A hydroxy compound $(\mathrm{X})$ with molar mass $122 \mathrm{~g} \mathrm{~mol}^{-1}$ is acetylated with acetic anhydride, using a large excess of the reagent ensuring complete acetylation of all hydroxyl groups. The product obtained has a molar mass of $290 \mathrm{~g} \mathrm{~mol}^{-1}$. The number of hydroxyl groups present in compound $(\mathrm{X})$ is:
Q25.
numerical
+4 / 1
The hydrogen spectrum consists of several spectral lines in Lyman series $\left(L_1, L_2\right.$, $\mathrm{L}_3 \ldots ; \mathrm{L}_1$ has lowest energy among Lyman series). Similarly it consists of several spectral lines in Balmer series $\left(\mathrm{B}_1, \mathrm{~B}_2, \mathrm{~B}_3 \ldots ; \mathrm{B}_1\right.$ has lowest energy among Balmer lines). The energy of $L_1$ is $x$ times the energy of $B_1$. The value of $x$ is $\_\_\_\_$ $\times 10^{-1}$
. (Nearest integer)
Q26.
mcq single
+4 / 1
$$ \text { The correct stability order of the following diazonium salts is } $$

Q27.
mcq single
+4 / 1
Consider three metal chlorides $\mathrm{x}, \mathrm{y}$ and z , where x is water soluble at room temperature, y is sparingly soluble in water at room temperature and z is soluble in hot water. $\mathrm{x}, \mathrm{y}$ and z are respectively
Q28.
mcq single
+4 / 1
At $27^{\circ} \mathrm{C}$ in presence of a catalyst, activation energy of a reaction is lowered by $10 \mathrm{~kJ} \mathrm{~mol}^{-1}$. The logarithm of ratio of $\frac{\mathrm{k} \text { (catalysed) }}{\mathrm{k} \text { (uncatalysed) }}$ is….
(Consider that the frequency factor for both the reactions is same)
Q29.
mcq single
+4 / 1
$\mathrm{A} \rightarrow \mathrm{D}$ is an endothermic reaction occurring in three steps (elementary).
(i) $\mathrm{A} \rightarrow \mathrm{B} \Delta \mathrm{H}_i=+\mathrm{ve}$
(ii) $\mathrm{B} \rightarrow \mathrm{C} \Delta \mathrm{H}_{i i}=-\mathrm{ve}$
(iii) $\mathrm{C} \rightarrow \mathrm{D} \Delta \mathrm{H}_{i i i}=-\mathrm{ve}$
Which of the following graphs between potential energy ( $y$-axis) vs reaction coordinate ( $x$-axis) correctly represents the reaction profile of $A \rightarrow D$ ?
Q30.
mcq single
+4 / 1
Among the following, the CORRECT combinations are
A. $\mathrm{IF}_3 \rightarrow \mathrm{~T}$-shaped ( $\mathrm{sp}^3 \mathrm{~d}$ )
B. $\mathrm{IF}_5 \rightarrow$ Square pyramidal $\left(\mathrm{sp}^3 \mathrm{~d}^2\right)$
C. $\mathrm{IF}_7 \rightarrow$ Pentagonal bipyramidal $\left(\mathrm{sp}^3 \mathrm{~d}^3\right)$
D. $\mathrm{ClO}_4{ }^{-} \rightarrow$ Square planar ( $\mathrm{sp}^2 \mathrm{~d}$ )
Choose the correct answer from the options given below:
Q31.
mcq single
+4 / 1
Given below are statements about some molecules/ions.
Identify the CORRECT statements.
A. The dipole moment value of $\mathrm{NF}_3$ is higher than that of $\mathrm{NH}_3$.
B. The dipole moment value of $\mathrm{BeH}_2$ is zero.
C. The bond order of $\mathrm{O}_2{ }^{2-}$ and $\mathrm{F}_2$ is same.
D. The formal charge on the central oxygen atom of ozone is -1 .
E. In $\mathrm{NO}_2$, all the three atoms satisfy the octet rule, hence it is very stable.
Choose the correct answer from the options given below:
Q32.
numerical
+4 / 1
X and Y are the number of electrons involved, respectively during the oxidation of $\mathrm{I}^{-}$to $\mathrm{I}_2$ and $\mathrm{S}^{2-}$ to S by acidified $\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7$. The value of $\mathrm{X}+\mathrm{Y}$ is $\_\_\_\_$ .
Q33.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** ' $\mathrm{C}-\mathrm{Cl}$ ' bond is stronger in $\mathrm{CH}_2=\mathrm{CH}-\mathrm{Cl}$ than $\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{Cl}$
**Statement II :** The given optically active molecule,  on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
 on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
on
hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
Q34.
mcq single
+4 / 1
Match the List-I with List-II
List-I**Chloro derivativeList-II
ExampleA. Vinyl ChlorideI. CH$_2$ = CH - CH$_2$ClB. Benzyl ChlorideII. CH$_3$ - CH(Cl)CH$_3$C. Alkyl ChlorideIII. CH$_2$ = CHClD. Allyl ChlorideIV.  Choose the correct** answer from the options given below :
Choose the correct** answer from the options given below :
 Choose the correct** answer from the options given below :
Choose the correct** answer from the options given below :
Q35.
mcq single
+4 / 1
A solution is prepared by dissolving 0.3 g of a non-volatile non-electrolyte solute 'A' of molar mass $60 \mathrm{~g} \mathrm{~mol}^{-1}$ and 0.9 g of a non-volatile non-electrolyte solute ' B ' of molar mass $180 \mathrm{~g} \mathrm{~mol}^{-1}$ in $100 \mathrm{~mL} \mathrm{H}_2 \mathrm{O}$ at $27^{\circ} \mathrm{C}$. Osmotic pressure of the solution will be
[Given: $\mathrm{R}=0.082 \mathrm{~L} \mathrm{~atm} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ ]
Q36.
mcq single
+4 / 1
' W ' g of a non-volatile electrolyte solid solute of molar mass ' M ' $\mathrm{g} \mathrm{mol}^{-1}$ when dissolved in 100 mL water, decreases vapour pressure of water from 640 mm Hg to 600 mm Hg . If aqueous solution of the electrolyte boils at 375 K and $\mathrm{K}_{\mathrm{b}}$ for water is $0.52 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$, then the mole fraction of the electrolyte solute $\left(x_2\right)$ in the solution can be expressed as
(Given : density of water $=1 \mathrm{~g} / \mathrm{mL}$ and boiling point of water $=373 \mathrm{~K}$ )
Q37.
numerical
+4 / 1
Consider two Group IV metal ions $\mathrm{X}^{2+}$ and $\mathrm{Y}^{2+}$.
A solution containing $0.01 \mathrm{M} \mathrm{X}^{2+}$ and $0.01 \mathrm{M} \mathrm{Y}^{2+}$ is saturated with $\mathrm{H}_2 \mathrm{~S}$. The pH at which the metal sulphide YS will form as a precipitate is $\_\_\_\_$ . (Nearest integer)
(Given: $\mathrm{K}_{\mathrm{sp}}(\mathrm{XS})=1 \times 10^{-22}$ at $25^{\circ} \mathrm{C}, \mathrm{K}_{\mathrm{sp}}(\mathrm{YS})=4 \times 10^{-16}$ at $25^{\circ} \mathrm{C}$, $\left[\mathrm{H}_2 \mathrm{~S}\right]=0.1 \mathrm{M}$ in solution, $\mathrm{K}_{a 1} \times \mathrm{K}_{a 2}\left(\mathrm{H}_2 \mathrm{~S}\right)=1.0 \times 10^{-21}, \log 2=0.30$, $\log 3=0.48, \log 5=0.70)$
Q38.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\mathrm{K}>\mathrm{Mg}>\mathrm{Al}>\mathrm{B}$ is the correct order in terms of metallic character.
**Statement II :** Atomic radius is always greater than the ionic radius for any element.
In the light of the above statements, choose the correct answer from the options given below
Q39.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I Isothermal process for ideal gas system
List-II Work done (
<mi>V</mi>
<mi>f</mi>
>
<mi>V</mi>
<mi>i</mi>
<mi>V</mi>
<mi>f</mi>
>
<mi>V</mi>
<mi>i</mi>
V_(f) > V_(i) )
A.
Reversible expansion
I.
<mi mathvariant="normal">w</mi>
=
0
<mi mathvariant="normal">w</mi>
=
0
w=0
B.
Free expansion
II.
<mi mathvariant="normal">w</mi>
=
−
<mi data-mjx-auto-op="false">nR</mi>
T
ln
<msub>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mi mathvariant="normal">w</mi>
=
−
<mi data-mjx-auto-op="false">nR</mi>
T
ln
<msub>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
w=-nRT ln((V_(f))/(V_(i)))
C.
Irreversible expansion
III.
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mo data-mjx-texclass="OPEN">(</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mo data-mjx-texclass="CLOSE">)</mo>
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mrow>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
</mrow>
w=-p_(ex)(V_(f)-V_(i))
D.
Irreversible compression
IV.
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mo data-mjx-texclass="OPEN">(</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
<mo data-mjx-texclass="CLOSE">)</mo>
<mi mathvariant="normal">w</mi>
=
−
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">p</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi data-mjx-auto-op="false">ex</mi>
</mrow>
</mrow>
<mrow>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">i</mi>
</mrow>
</mrow>
</msub>
<mo>−</mo>
<msub>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">V</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">f</mi>
</mrow>
</mrow>
</msub>
</mrow>
w=-p_(ex)(V_(i)-V_(f))
Choose the correct answer from the options given below:
Q40.
mcq single
+4 / 1
Given below are two statements:
**Statement I :** The number of paramagnetic species among $\left[\mathrm{CoF}_6\right]^{3-},\left[\mathrm{TiF}_6\right]^{3-}$, $\mathrm{V}_2 \mathrm{O}_5$ and $\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{3-}$ is 3 .
**Statement II :**
$\mathrm{K}_4\left[\mathrm{Fe}(\mathrm{CN})_6\right]<\mathrm{K}_3\left[\mathrm{Fe}(\mathrm{CN})_6\right]<\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{SO}_4 \cdot \mathrm{H}_2 \mathrm{O}<\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{Cl}_3$ is the correct order in terms of number of unpaired electron(s) present in the complexes.
In the light of the above statements, choose the correct answer from the options given below
Q41.
mcq single
+4 / 1
Consider a mixture ' X ' which is made by dissolving 0.4 mol of $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5 \mathrm{SO}_4\right] \mathrm{Br}$ and 0.4 mol of $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5 \mathrm{Br}\right] \mathrm{SO}_4$ in water to make 4 L of solution. When 2 L of mixture ' X ' is allowed to react with excess of $\mathrm{AgNO}_3$, it forms precipitate ' Y '. The rest 2 L of mixture ' X ' reacts with excess $\mathrm{BaCl}_2$ to form precipitate ' Z '. Which of the following statements is CORRECT?
Q42.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Hybridisation, shape and spin only magnetic moment of $\mathrm{K}_3\left[\mathrm{Co}\left(\mathrm{CO}_3\right)_3\right]$ is $\mathrm{sp}^3 \mathrm{~d}^2$, octahedral and 4.9 BM respectively.
**Statement II :** Geometry, hybridisation and spin only magnetic moment values $(B M)$ of the ions $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-},\left[\mathrm{MnBr}_4\right]^{2-}$ and $\left[\mathrm{CoF}_6\right]^{3-}$ respectively are square planar, tetrahedral, octahedral; $\mathrm{dsp}^2, \mathrm{sp}^3, \mathrm{sp}^3 \mathrm{~d}^2$ and $0,5.9,4.9$.
In the light of the above statements, choose the correct answer from the options given below
Q43.
mcq single
+4 / 1
$$ \text { Consider the following two reactions } \mathrm{A} \text { and } \mathrm{B} \text {. } $$
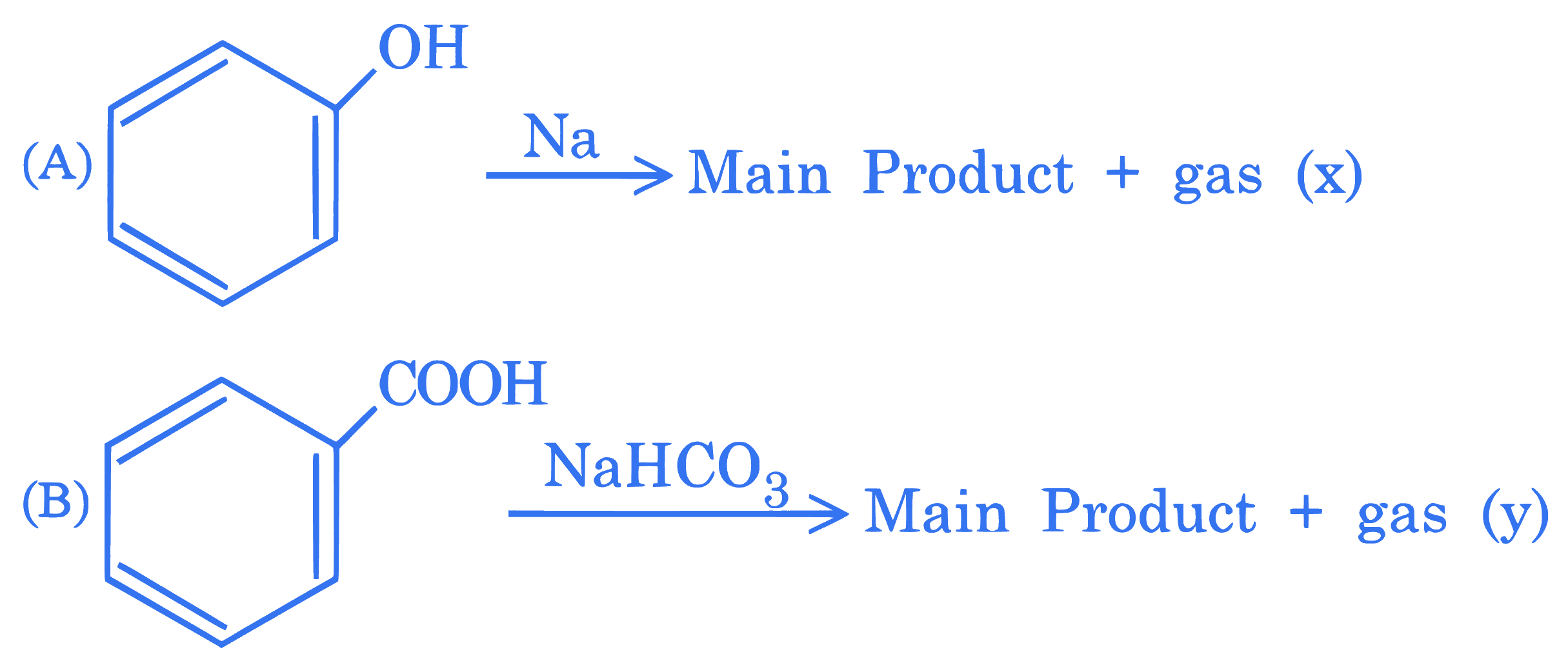 Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
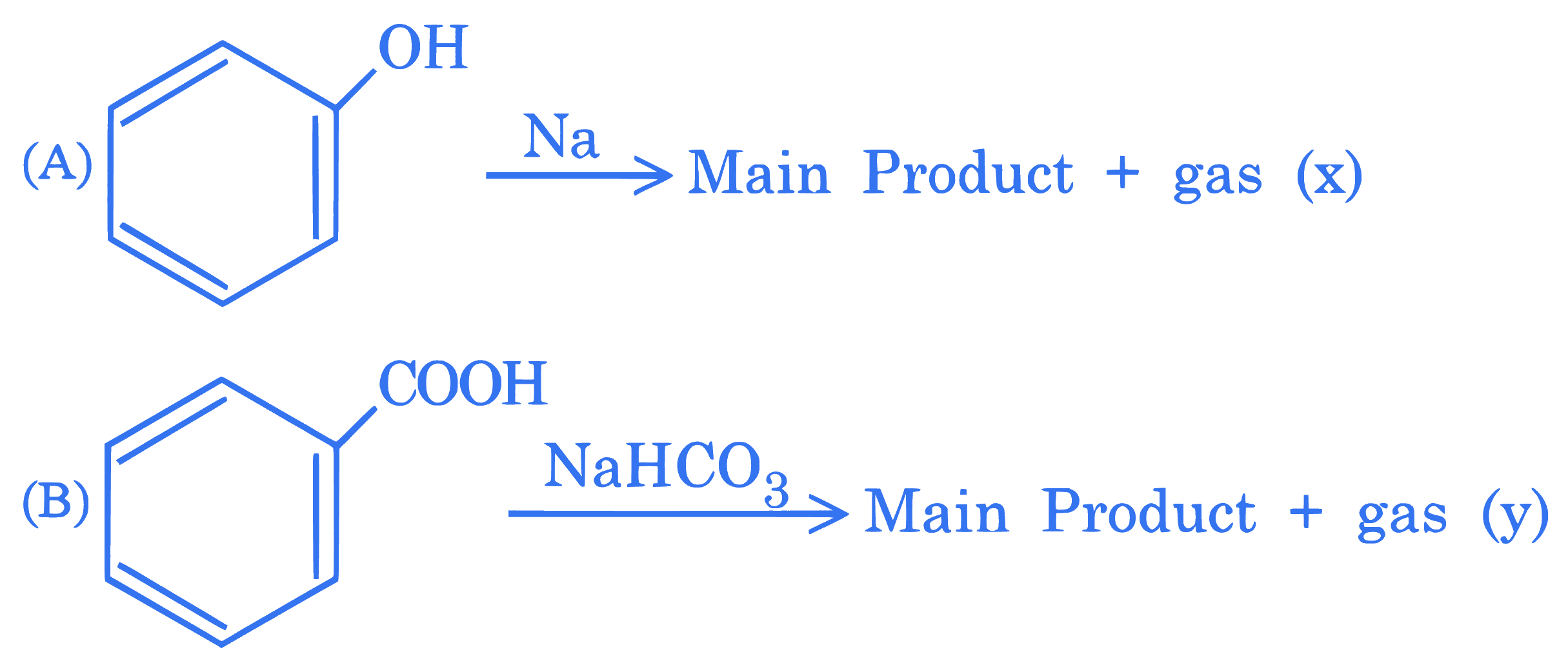 Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
Numerical value of [molar mass of $x+$ molar mass of $y$ ] is $\_\_\_\_$
Q44.
mcq single
+4 / 1
A student is given one compound among the following compounds that gives positive test with Tollen's reagent.
 The compound is :
The compound is :
 The compound is :
The compound is :
Q45.
numerical
+4 / 1
In Dumas method for estimation of nitrogen, 0.50 g of an organic compound gave 70 mL of nitrogen collected at 300 K and 715 mm pressure. The percentage of nitrogen in the organic compound is $\_\_\_\_$ $\%$.
(Aqueous tension at 300 K is 15 mm ).
Q46.
mcq single
+4 / 1
$$ \text {Arrange the following alkenes in decreasing order of stability. } $$
 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
Q47.
mcq single
+4 / 1
$$ \text { Arrange the following carbanions in the decreasing order of stability. } $$
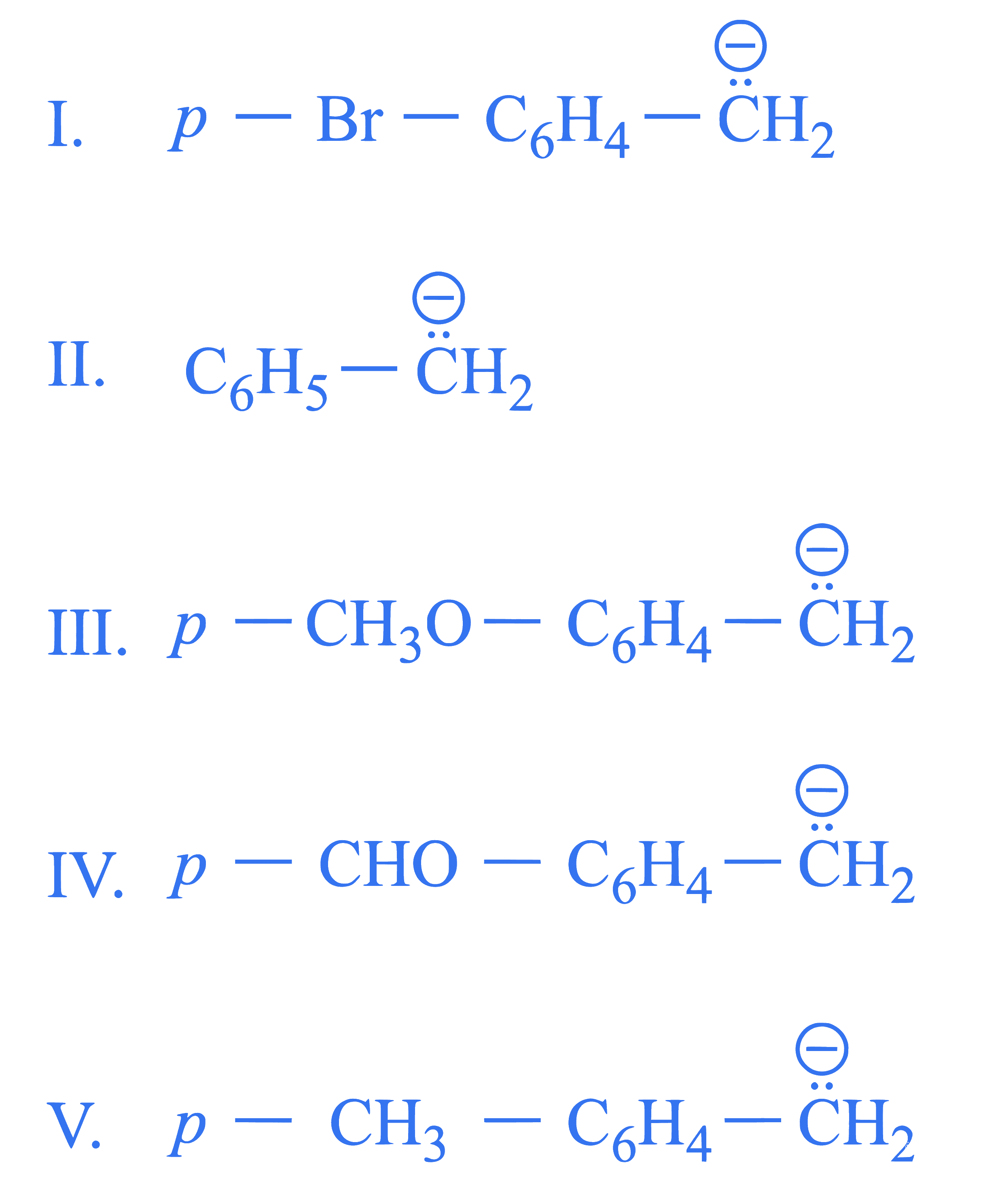 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
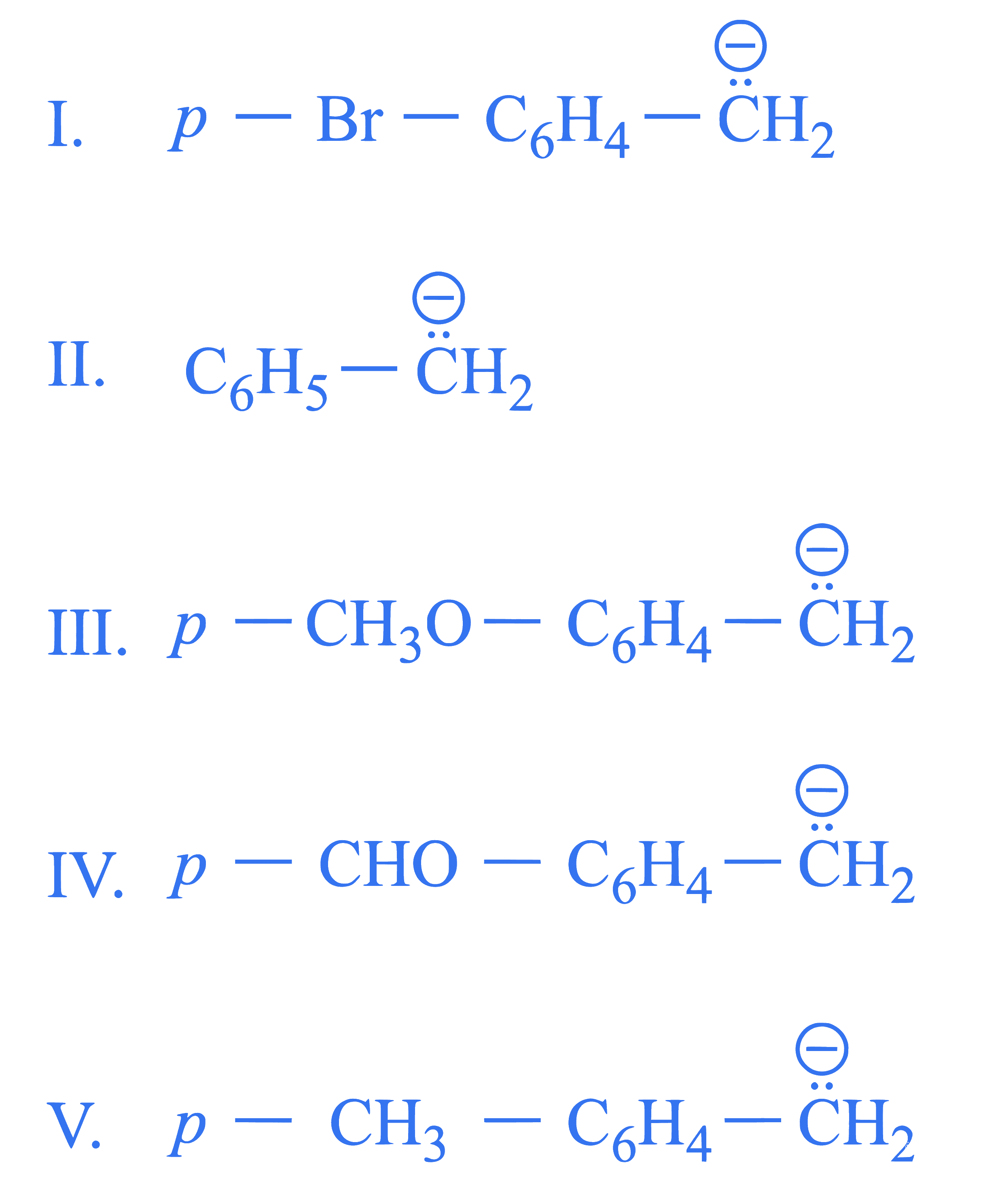 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q48.
numerical
+4 / 1
Electricity is passed through an acidic solution of $\mathrm{Cu}^{2+}$ till all the $\mathrm{Cu}^{2+}$ was exhausted, leading to the deposition of 300 mg of Cu metal. However, a current of 600 mA was continued to pass through the same solution for another 28 minutes by keeping the total volume of the solution fixed at 200 mL . The total volume of oxygen evolved at STP during the entire process is $\_\_\_\_$ mL . (Nearest integer)
[Given:
$$ \begin{aligned} & \mathrm{Cu}^{2+}(\mathrm{aq})+2 \mathrm{e}^{-} \rightarrow \mathrm{Cu}(\mathrm{~s}) \mathrm{E}_{\mathrm{red}}^{\mathrm{o}}=+0.34 \mathrm{~V} \\ & \mathrm{O}_2(\mathrm{~g})+4 \mathrm{H}^{+}+4 \mathrm{e}^{-} \rightarrow 2 \mathrm{H}_2 \mathrm{O} \mathrm{E}_{\mathrm{red}}^{\mathrm{o}}=+1.23 \mathrm{~V} \end{aligned} $$
Molar mass of $\mathrm{Cu}=63.54 \mathrm{~g} \mathrm{~mol}^{-1}$
Molar mass of $\mathrm{O}_2=32 \mathrm{~g} \mathrm{~mol}^{-1}$
Faraday Constant $=96500 \mathrm{C} \mathrm{mol}^{-1}$
Molar volume at $\mathrm{STP}=22.4 \mathrm{~L}$ ]
Q49.
mcq single
+4 / 1
A hydroxy compound $(\mathrm{X})$ with molar mass $122 \mathrm{~g} \mathrm{~mol}^{-1}$ is acetylated with acetic anhydride, using a large excess of the reagent ensuring complete acetylation of all hydroxyl groups. The product obtained has a molar mass of $290 \mathrm{~g} \mathrm{~mol}^{-1}$. The number of hydroxyl groups present in compound $(\mathrm{X})$ is:
Q50.
numerical
+4 / 1
The hydrogen spectrum consists of several spectral lines in Lyman series $\left(L_1, L_2\right.$, $\mathrm{L}_3 \ldots ; \mathrm{L}_1$ has lowest energy among Lyman series). Similarly it consists of several spectral lines in Balmer series $\left(\mathrm{B}_1, \mathrm{~B}_2, \mathrm{~B}_3 \ldots ; \mathrm{B}_1\right.$ has lowest energy among Balmer lines). The energy of $L_1$ is $x$ times the energy of $B_1$. The value of $x$ is $\_\_\_\_$ $\times 10^{-1}$
. (Nearest integer)
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
The mean and variance of a data of 10 observations are 10 and 2 , respectively. If an observations $\alpha$ in this data is replaced by $\beta$, then the mean and variance become 10.1 and 1.99 , respectively. Then $\alpha+\beta$ equals
Q2.
mcq single
+4 / 1
Let $\vec{a}=2 \hat{\mathrm{i}}+\hat{\mathrm{j}}-2 \hat{\mathrm{k}}, \vec{b}=\hat{\mathrm{i}}+\hat{\mathrm{j}}$ and $\vec{c}=\vec{a} \times \vec{b}$. Let $\vec{d}$ be a vector such that $|\vec{d}-\vec{a}|=\sqrt{11},|\vec{c} \times \vec{d}|=3$ and the angle between $\vec{c}$ and $\vec{d}$ is $\frac{\pi}{4}$. Then $\vec{a} \cdot \vec{d}$ is equal to
Q3.
mcq single
+4 / 1
Let the lines $\mathrm{L}_1: \vec{r}=\hat{\mathrm{i}}+2 \hat{\mathrm{j}}+3 \hat{\mathrm{k}}+\lambda(2 \hat{\mathrm{i}}+3 \hat{\mathrm{j}}+4 \hat{\mathrm{k}}), \lambda \in \mathbb{R}$ and $\mathrm{L}_2: \vec{r}=(4 \hat{\mathrm{i}}+\hat{\mathrm{j}})+\mu(5 \hat{\mathrm{i}}+2 \hat{\mathrm{j}}+\hat{\mathrm{k}}), \mu \in \mathbb{R}$, intersect at the point R . Let P and Q be the points lying on lines $L_1$ and $L_2$, respectively, such that $|\overrightarrow{\mathrm{PR}}|=\sqrt{29}$ and $|\overrightarrow{\mathrm{PQ}}|=\sqrt{\frac{47}{3}}$. If the point P lies in the first octant, then $27(\mathrm{QR})^2$ is equal to
Q4.
mcq single
+4 / 1
If the domain of the function
$$ f(x)=\log _{\left(10 x^2-17 x+7\right)}\left(18 x^2-11 x+1\right) $$
is $(-\infty, a) \cup(b, c) \cup(d, \infty)-\{e\}$, then
$90(a+b+c+d+e)$ equals:
Q5.
mcq single
+4 / 1
The number of the real solutions of the equation: $x|x+3|+|x-1|-2=0$ is
Q6.
mcq single
+4 / 1
From a lot containing 10 defective and 90 non-defective bulbs, 8 bulbs are selected one by one with replacement. Then the probability of getting at least 7 defective bulbs is
Q7.
numerical
+4 / 1
Let a line L passing through the point $\mathrm{P}(1,1,1)$ be perpendicular to the lines $\frac{x-4}{4}=\frac{y-1}{1}=\frac{z-1}{1}$ and $\frac{x-17}{1}=\frac{y-71}{1}=\frac{z}{0}$. Let the line L intersect the $y z-$ plane at the point Q . Another line parallel to L and passing through the point $\mathrm{S}(1,0,-1)$ intersects the $y z$-plane at the point R . Then the square of the area of the parallelogram PQRS is equal to $\_\_\_\_$ .
Q8.
mcq single
+4 / 1
Consider an A.P.: $a_1, a_2, \ldots, a_{\mathrm{n}} ; a_1>0$. If $a_2-a_1=\frac{-3}{4}, a_{\mathrm{n}}=\frac{1}{4} a_1$, and $\sum\limits_{\mathrm{i}=1}^{\mathrm{n}} a_{\mathrm{i}}=\frac{525}{2}$, then $\sum\limits_{\mathrm{i}=1}^{17} a_{\mathrm{i}}$ is equal to
Q9.
mcq single
+4 / 1
Let $729,81,9,1, \ldots$ be a sequence and $\mathrm{P}_n$ denote the product of the first $n$ terms of this sequence.
If $2 \sum\limits_{n=1}^{40}\left(\mathrm{P}_n\right)^{\frac{1}{n}}=\frac{3^\alpha-1}{3^\beta}$ and $\operatorname{gcd}(\alpha, \beta)=1$, then
$\alpha+\beta$ is equal to
Q10.
mcq single
+4 / 1
If the function $f(x)=\frac{e^x\left(e^{\tan x-x}-1\right)+\log _e(\sec x+\tan x)-x}{\tan x-x}$ is continuous at $x=0$, then the value of $f(0)$ is equal to
Q11.
mcq single
+4 / 1
Let $\alpha, \beta \in \mathbb{R}$ be such that the function $f(x)= \begin{cases}2 \alpha\left(x^2-2\right)+2 \beta x & , x<1 \\ (\alpha+3) x+(\alpha-\beta) & , x \geq 1\end{cases}$ be differentiable at all $x \in \mathbb{R}$. Then $34(\alpha+\beta)$ is equal to
Q12.
numerical
+4 / 1
Let $(2 \alpha, \alpha)$ be the largest interval in which the function $f(t)=\frac{|t+1|}{t^2}, t2$, is $\_\_\_\_$
Q13.
mcq single
+4 / 1
The value of $\frac{\sqrt{3} \operatorname{cosec} 20^{\circ}-\sec 20^{\circ}}{\cos 20^{\circ} \cos 40^{\circ} \cos 60^{\circ} \cos 80^{\circ}}$ is equal to
Q14.
mcq single
+4 / 1
If $\cot x=\frac{5}{12}$ for some $x \in\left(\pi, \frac{3 \pi}{2}\right)$, then $\sin 7 x\left(\cos \frac{13 x}{2}+\sin \frac{13 x}{2}\right)+\cos 7 x\left(\cos \frac{13 x}{2}-\sin \frac{13 x}{2}\right)$ is equal to
Q15.
mcq single
+4 / 1
Let $\mathrm{A}_1$ be the bounded area enclosed by the curves $y=x^2+2, x+y=8$ and $y$-axis that lies in the first quadrant. Let $\mathrm{A}_2$ be the bounded area enclosed by the curves $y=x^2+2, y^2=x, x=2$, and $y$-axis that lies in the first quadrant. Then $\mathrm{A}_1-\mathrm{A}_2$ is equal to
Q16.
numerical
+4 / 1
The number of $3 \times 2$ matrices A , which can be formed using the elements of the set $\{-2,-1,0,1,2\}$ such that the sum of all the diagonal elements of $\mathrm{A}^{\mathrm{T}} \mathrm{A}$ is 5 , is
$\_\_\_\_$
Q17.
mcq single
+4 / 1
Let $f(t)=\int\left(\frac{1-\sin \left(\log _e t\right)}{1-\cos \left(\log _e t\right)}\right) d t, t>1$.
If $f\left(e^{\pi / 2}\right)=-e^{\pi / 2}$ and $f\left(e^{\pi / 4}\right)=\alpha e^{\pi / 4}$, then $\alpha$ equals
Q18.
mcq single
+4 / 1
Let $\mathrm{S}=\frac{1}{25!}+\frac{1}{3!23!}+\frac{1}{5!21!}+\ldots$ up to 13 terms. If $13 \mathrm{~S}=\frac{2^k}{n!}, k \in \mathrm{~N}$, then $n+k$ is equal to
Q19.
mcq single
+4 / 1
Let $A(1,0), B(2,-1)$ and $C\left(\frac{7}{3}, \frac{4}{3}\right)$ be three points. If the equation of the bisector of the angle ABC is $\alpha x+\beta y=5$, then the value of $\alpha^2+\beta^2$ is
Q20.
mcq single
+4 / 1
Let $R$ be a relation defined on the set $\{1,2,3,4\} \times\{1,2,3,4\}$ by
$$ \mathrm{R}=\{((a, b),(c, d)): 2 a+3 b=3 c+4 d\} . $$
Then the number of elements in R is
Q21.
mcq single
+4 / 1
Let $\mathrm{S}=\left\{z \in \mathbb{C}:\left|\frac{z-6 i}{z-2 i}\right|=1\right.$ and $\left.\left|\frac{z-8+2 i}{z+2 i}\right|=\frac{3}{5}\right\}$.
Then $\sum\limits_{z \in \mathrm{~s}}|z|^2$ is equal to :
Q22.
mcq single
+4 / 1
Let a circle of radius 4 pass through the origin O , the points $\mathrm{A}(-\sqrt{3} a, 0)$ and $\mathrm{B}(0,-\sqrt{2} b)$, where $a$ and $b$ are real parameters and $a b \neq 0$. Then the locus of the centroid of $\triangle \mathrm{OAB}$ is a circle of radius
Q23.
mcq single
+4 / 1
Let each of the two ellipses $\mathrm{E}_1: \frac{x^2}{a^2}+\frac{y^2}{b^2}=1,(a>b)$ and $\mathrm{E}_2: \frac{x^2}{\mathrm{~A}^2}+\frac{y^2}{\mathrm{~B}^2}=1,(\mathrm{~A}<\mathrm{B})$ have eccentricity $\frac{4}{5}$. Let the lengths of the latus recta of $\mathrm{E}_1$ and $\mathrm{E}_2$ be $l_1$ and $l_2$, respectively, such that $2 l_1^2=9 l_2$. If the distance between the foci of $E_1$ is 8 , then the distance between the foci of $E_2$ is
Q24.
numerical
+4 / 1
The number of numbers greater than 5000 , less than 9000 and divisible by 3 , that can be formed using the digits $0,1,2,5,9$, if the repetition of the digits is allowed, is $\_\_\_\_$
Q25.
numerical
+4 / 1
Let a differentiable function $f$ satisfy the equation $\int_0^{36} f\left(\frac{t x}{36}\right) d t=4 \alpha f(x)$. If $y=f(x)$ is a standard parabola passing through the points $(2,1)$ and $(-4, \beta)$, then $\beta^\alpha$ is equal to $\_\_\_\_$ .
Q26.
mcq single
+4 / 1
The mean and variance of a data of 10 observations are 10 and 2 , respectively. If an observations $\alpha$ in this data is replaced by $\beta$, then the mean and variance become 10.1 and 1.99 , respectively. Then $\alpha+\beta$ equals
Q27.
mcq single
+4 / 1
Let $\vec{a}=2 \hat{\mathrm{i}}+\hat{\mathrm{j}}-2 \hat{\mathrm{k}}, \vec{b}=\hat{\mathrm{i}}+\hat{\mathrm{j}}$ and $\vec{c}=\vec{a} \times \vec{b}$. Let $\vec{d}$ be a vector such that $|\vec{d}-\vec{a}|=\sqrt{11},|\vec{c} \times \vec{d}|=3$ and the angle between $\vec{c}$ and $\vec{d}$ is $\frac{\pi}{4}$. Then $\vec{a} \cdot \vec{d}$ is equal to
Q28.
mcq single
+4 / 1
Let the lines $\mathrm{L}_1: \vec{r}=\hat{\mathrm{i}}+2 \hat{\mathrm{j}}+3 \hat{\mathrm{k}}+\lambda(2 \hat{\mathrm{i}}+3 \hat{\mathrm{j}}+4 \hat{\mathrm{k}}), \lambda \in \mathbb{R}$ and $\mathrm{L}_2: \vec{r}=(4 \hat{\mathrm{i}}+\hat{\mathrm{j}})+\mu(5 \hat{\mathrm{i}}+2 \hat{\mathrm{j}}+\hat{\mathrm{k}}), \mu \in \mathbb{R}$, intersect at the point R . Let P and Q be the points lying on lines $L_1$ and $L_2$, respectively, such that $|\overrightarrow{\mathrm{PR}}|=\sqrt{29}$ and $|\overrightarrow{\mathrm{PQ}}|=\sqrt{\frac{47}{3}}$. If the point P lies in the first octant, then $27(\mathrm{QR})^2$ is equal to
Q29.
mcq single
+4 / 1
If the domain of the function
$$ f(x)=\log _{\left(10 x^2-17 x+7\right)}\left(18 x^2-11 x+1\right) $$
is $(-\infty, a) \cup(b, c) \cup(d, \infty)-\{e\}$, then
$90(a+b+c+d+e)$ equals:
Q30.
mcq single
+4 / 1
The number of the real solutions of the equation: $x|x+3|+|x-1|-2=0$ is
Q31.
mcq single
+4 / 1
From a lot containing 10 defective and 90 non-defective bulbs, 8 bulbs are selected one by one with replacement. Then the probability of getting at least 7 defective bulbs is
Q32.
numerical
+4 / 1
Let a line L passing through the point $\mathrm{P}(1,1,1)$ be perpendicular to the lines $\frac{x-4}{4}=\frac{y-1}{1}=\frac{z-1}{1}$ and $\frac{x-17}{1}=\frac{y-71}{1}=\frac{z}{0}$. Let the line L intersect the $y z-$ plane at the point Q . Another line parallel to L and passing through the point $\mathrm{S}(1,0,-1)$ intersects the $y z$-plane at the point R . Then the square of the area of the parallelogram PQRS is equal to $\_\_\_\_$ .
Q33.
mcq single
+4 / 1
Consider an A.P.: $a_1, a_2, \ldots, a_{\mathrm{n}} ; a_1>0$. If $a_2-a_1=\frac{-3}{4}, a_{\mathrm{n}}=\frac{1}{4} a_1$, and $\sum\limits_{\mathrm{i}=1}^{\mathrm{n}} a_{\mathrm{i}}=\frac{525}{2}$, then $\sum\limits_{\mathrm{i}=1}^{17} a_{\mathrm{i}}$ is equal to
Q34.
mcq single
+4 / 1
Let $729,81,9,1, \ldots$ be a sequence and $\mathrm{P}_n$ denote the product of the first $n$ terms of this sequence.
If $2 \sum\limits_{n=1}^{40}\left(\mathrm{P}_n\right)^{\frac{1}{n}}=\frac{3^\alpha-1}{3^\beta}$ and $\operatorname{gcd}(\alpha, \beta)=1$, then
$\alpha+\beta$ is equal to
Q35.
mcq single
+4 / 1
If the function $f(x)=\frac{e^x\left(e^{\tan x-x}-1\right)+\log _e(\sec x+\tan x)-x}{\tan x-x}$ is continuous at $x=0$, then the value of $f(0)$ is equal to
Q36.
mcq single
+4 / 1
Let $\alpha, \beta \in \mathbb{R}$ be such that the function $f(x)= \begin{cases}2 \alpha\left(x^2-2\right)+2 \beta x & , x<1 \\ (\alpha+3) x+(\alpha-\beta) & , x \geq 1\end{cases}$ be differentiable at all $x \in \mathbb{R}$. Then $34(\alpha+\beta)$ is equal to
Q37.
numerical
+4 / 1
Let $(2 \alpha, \alpha)$ be the largest interval in which the function $f(t)=\frac{|t+1|}{t^2}, t2$, is $\_\_\_\_$
Q38.
mcq single
+4 / 1
The value of $\frac{\sqrt{3} \operatorname{cosec} 20^{\circ}-\sec 20^{\circ}}{\cos 20^{\circ} \cos 40^{\circ} \cos 60^{\circ} \cos 80^{\circ}}$ is equal to
Q39.
mcq single
+4 / 1
If $\cot x=\frac{5}{12}$ for some $x \in\left(\pi, \frac{3 \pi}{2}\right)$, then $\sin 7 x\left(\cos \frac{13 x}{2}+\sin \frac{13 x}{2}\right)+\cos 7 x\left(\cos \frac{13 x}{2}-\sin \frac{13 x}{2}\right)$ is equal to
Q40.
mcq single
+4 / 1
Let $\mathrm{A}_1$ be the bounded area enclosed by the curves $y=x^2+2, x+y=8$ and $y$-axis that lies in the first quadrant. Let $\mathrm{A}_2$ be the bounded area enclosed by the curves $y=x^2+2, y^2=x, x=2$, and $y$-axis that lies in the first quadrant. Then $\mathrm{A}_1-\mathrm{A}_2$ is equal to
Q41.
numerical
+4 / 1
The number of $3 \times 2$ matrices A , which can be formed using the elements of the set $\{-2,-1,0,1,2\}$ such that the sum of all the diagonal elements of $\mathrm{A}^{\mathrm{T}} \mathrm{A}$ is 5 , is
$\_\_\_\_$
Q42.
mcq single
+4 / 1
Let $f(t)=\int\left(\frac{1-\sin \left(\log _e t\right)}{1-\cos \left(\log _e t\right)}\right) d t, t>1$.
If $f\left(e^{\pi / 2}\right)=-e^{\pi / 2}$ and $f\left(e^{\pi / 4}\right)=\alpha e^{\pi / 4}$, then $\alpha$ equals
Q43.
mcq single
+4 / 1
Let $\mathrm{S}=\frac{1}{25!}+\frac{1}{3!23!}+\frac{1}{5!21!}+\ldots$ up to 13 terms. If $13 \mathrm{~S}=\frac{2^k}{n!}, k \in \mathrm{~N}$, then $n+k$ is equal to
Q44.
mcq single
+4 / 1
Let $A(1,0), B(2,-1)$ and $C\left(\frac{7}{3}, \frac{4}{3}\right)$ be three points. If the equation of the bisector of the angle ABC is $\alpha x+\beta y=5$, then the value of $\alpha^2+\beta^2$ is
Q45.
mcq single
+4 / 1
Let $R$ be a relation defined on the set $\{1,2,3,4\} \times\{1,2,3,4\}$ by
$$ \mathrm{R}=\{((a, b),(c, d)): 2 a+3 b=3 c+4 d\} . $$
Then the number of elements in R is
Q46.
mcq single
+4 / 1
Let $\mathrm{S}=\left\{z \in \mathbb{C}:\left|\frac{z-6 i}{z-2 i}\right|=1\right.$ and $\left.\left|\frac{z-8+2 i}{z+2 i}\right|=\frac{3}{5}\right\}$.
Then $\sum\limits_{z \in \mathrm{~s}}|z|^2$ is equal to :
Q47.
mcq single
+4 / 1
Let a circle of radius 4 pass through the origin O , the points $\mathrm{A}(-\sqrt{3} a, 0)$ and $\mathrm{B}(0,-\sqrt{2} b)$, where $a$ and $b$ are real parameters and $a b \neq 0$. Then the locus of the centroid of $\triangle \mathrm{OAB}$ is a circle of radius
Q48.
mcq single
+4 / 1
Let each of the two ellipses $\mathrm{E}_1: \frac{x^2}{a^2}+\frac{y^2}{b^2}=1,(a>b)$ and $\mathrm{E}_2: \frac{x^2}{\mathrm{~A}^2}+\frac{y^2}{\mathrm{~B}^2}=1,(\mathrm{~A}<\mathrm{B})$ have eccentricity $\frac{4}{5}$. Let the lengths of the latus recta of $\mathrm{E}_1$ and $\mathrm{E}_2$ be $l_1$ and $l_2$, respectively, such that $2 l_1^2=9 l_2$. If the distance between the foci of $E_1$ is 8 , then the distance between the foci of $E_2$ is
Q49.
numerical
+4 / 1
The number of numbers greater than 5000 , less than 9000 and divisible by 3 , that can be formed using the digits $0,1,2,5,9$, if the repetition of the digits is allowed, is $\_\_\_\_$
Q50.
numerical
+4 / 1
Let a differentiable function $f$ satisfy the equation $\int_0^{36} f\left(\frac{t x}{36}\right) d t=4 \alpha f(x)$. If $y=f(x)$ is a standard parabola passing through the points $(2,1)$ and $(-4, \beta)$, then $\beta^\alpha$ is equal to $\_\_\_\_$ .
Physics
Physics
Q1.
numerical
+4 / 1
A voltage regulating circuit consisting of Zener diode, having break-down voltage of 10 V and maximum power dissipation of 0.4 W , is operated at 15 V . The approximate value of protective resistance in this circuit is $\_\_\_\_$ $\Omega$.
Q2.
mcq single
+4 / 1
A spring of force constant $15 \mathrm{~N} / \mathrm{m}$ is cut into two pieces. If the ratio of their length is $1: 3$, then the force constant of smaller piece is $\_\_\_\_$ $\mathrm{N} / \mathrm{m}$.
Q3.
mcq single
+4 / 1
A cylindrical block of mass $M$ and area of cross section $A$ is floating in a liquid of density $\rho$ and with its axis vertical. When depressed a little and released the block starts oscillating. The period of oscillation is $\_\_\_\_$
Q4.
mcq single
+4 / 1
Two masses 400 g and 350 g are suspended from the ends of a light string passing over a heavy pulley of radius 2 cm . When released from rest the heavier mass is observed to fall 81 cm in 9 s . The rotational inertia of the pulley is $\_\_\_\_$ $\mathrm{kg} \cdot \mathrm{m}^2$. $\left(\mathrm{g}=9.8 \mathrm{~m} / \mathrm{s}^2\right)$
Q5.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
List-II
A.
Magnetic induction
I.
<mi mathvariant="normal">M</mi>
<mi mathvariant="normal">L</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mi mathvariant="normal">L</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
MLT^(-2)A^(-2)
B.
Magnetic flux
II.
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
ML^(2)T^(-2)A^(-2)
C.
Magnetic permeability
III.
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>0</mn>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>0</mn>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
ML^(0)T^(-2)A^(-1)
D.
Self inductance
IV.
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
ML^(2)T^(-2)A^(-1)
Choose the correct answer from the options given below:
Q6.
mcq single
+4 / 1
Density of water at $4^{\circ} \mathrm{C}$ and $20^{\circ} \mathrm{C}$ are $1000 \mathrm{~kg} / \mathrm{m}^3$ and $998 \mathrm{~kg} / \mathrm{m}^3$ respectively. The increase in internal energy of 4 kg of water when it is heated from $4^{\circ} \mathrm{C}$ to $20^{\circ} \mathrm{C}$ is $\_\_\_\_$ J.
(specific heat capacity of water $=4.2 \mathrm{~J} / \mathrm{kg}$. and 1 atmospheric pressure $=10^5 \mathrm{~Pa}$ )
Q7.
numerical
+4 / 1
A gas of certain mass filled in a closed cylinder at a pressure of 3.23 kPa has temperature $50^{\circ} \mathrm{C}$. The gas is now heated to double its temperature. The modified pressure is $\_\_\_\_$ Pa .
Q8.
numerical
+4 / 1
A short bar magnet placed with its axis at $30^{\circ}$ with an external field of 800 Gauss, experiences a torque of $0.016 \mathrm{~N} . \mathrm{m}$. The work done in moving it from most stable to most unstable position is $\alpha \times 10^{-3} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
Q9.
numerical
+4 / 1
Sixty four rain drops of radius 1 mm each falling down with a terminal velocity of $10 \mathrm{~cm} / \mathrm{s}$ coalesce to form a bigger drop. The terminal velocity of bigger drop is
$\_\_\_\_$ $\mathrm{cm} / \mathrm{s}$.
Q10.
mcq single
+4 / 1
A brass wire of length 2 m and radius 1 mm at $27^{\circ} \mathrm{C}$ is held taut between two rigid supports. Initially it was cooled to a temperature of $-43^{\circ} \mathrm{C}$ creating a tension $T$ in the wire. The temperature to which the wire has to be cooled in order to increase the tension in it to $1.4 T$, is $\_\_\_\_$ ${ }^{\circ} \mathrm{C}$.
Q11.
mcq single
+4 / 1
The electrostatic potential in a charged spherical region of radius $r$ varies as $V=a r^3+b$, where $a$ and $b$ are constants. The total charge in the sphere of unit radius is $\alpha \times \pi a \in_0$. The value of $\alpha$ is $\_\_\_\_$ .
(permittivity of vacuum is $\epsilon_0$ )
Q12.
mcq single
+4 / 1
Three charges $+2 q,+3 q$ and $-4 q$ are situated at $(0,-3 a),(2 a, 0)$ and $(-2 a, 0)$ respectively in the $x y$ plane. The resultant dipole moment about origin is $\_\_\_\_$ .
Q13.
mcq single
+4 / 1
There are three co-centric conducting spherical shells $A, B$ and $C$ of radii $a, b$ and $c$ respectively ( $c>b>a$ ) and they are charged with charge $q_1, q_2$ and $q_3$ respectively. The potentials of the spheres $A, B$ and $C$ respectively, are :
Q14.
mcq single
+4 / 1
Three masses $200 \mathrm{~kg}, 300 \mathrm{~kg}$ and 400 kg are placed at the vertices of an equilateral triangle with sides 20 m . They are rearranged on the vertices of a bigger triangle of side 25 m and with the same centre. The work done in this process $\_\_\_\_$ J. (Gravitational constant $\mathrm{G}=6.7 \times 10^{-11} \mathrm{~N} \mathrm{~m}^2 / \mathrm{kg}^2$ )
Q15.
mcq single
+4 / 1
An unpolarised light is incident at an interface of two dielectric media having refractive indices of 2 (incident medium) and $2 \sqrt{3}$ (medium) respectively. To satisfy the condition that reflected and refracted rays are perpendicular to each other, the angle of incidence is $\_\_\_\_$
Q16.
mcq single
+4 / 1
In a microscope of tube length 10 cm two convex lenses are arranged with focal length of 2 cm and 5 cm . Total magnification obtained with this system for normal adjustment is $(5)^k$. The value of $k$ is $\_\_\_\_$ .
Q17.
mcq single
+4 / 1
The exit surface of a prism with refractive index $n$ is coated with a material having refractive index $\frac{n}{2}$. When this prism is set for minimum angle of deviation, it exactly meets the condition of critical angle. The prism angle is $\_\_\_\_$ .
Q18.
mcq single
+4 / 1
Two electrons are moving in orbits of two hydrogen like atoms with speeds $3 \times 10^5 \mathrm{~m} / \mathrm{s}$ and $2.5 \times 10^5 \mathrm{~m} / \mathrm{s}$ respectively. If the radii of these orbits are nearly same then the possible order of energy states are $\_\_\_\_$ respectively.
Q19.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** For all elements, greater the mass of the nucleus, greater is the binding energy per nucleon.
**Statement II :** For all elements, nuclei with less binding energy per nucleon transforms to nuclei with greater binding energy per nucleon.
In the light of the above statements, choose the correct answer from the options given below
Q20.
mcq single
+4 / 1
A boy throws a ball into air at $45^{\circ}$ from the horizontal to land it on a roof of a building of height $H$. If the ball attains maximum height in 2 s and lands on the building in 3 s after launch, then value of $H$ is $\_\_\_\_$ m.
$$ \left(\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2\right) $$
Q21.
numerical
+4 / 1
In the given figure the blocks $A, B$ and $C$ weigh $4 \mathrm{~kg}, 6 \mathrm{~kg}$ and 8 kg respectively. The co-efficient of sliding friction between any two surfaces is 0.5 . The force $\vec{F}$ required to slide the block $C$ with constant speed is $\_\_\_\_$ N . (Use $g=10 \mathrm{~m} / \mathrm{s}^2$ )

Q22.
mcq single
+4 / 1
Two resistors $2 \Omega$ and $3 \Omega$ are connected in the gaps of bridge as shown in figure. The null point is obtained with the contact of jockey at some point on wire $X Y$. When an unknown resistor is connected in parallel with $3 \Omega$ resistor, the null point is shifted by 22.5 cm toward $Y$. The resistance of unknown resistor is $\_\_\_\_$ $\Omega$.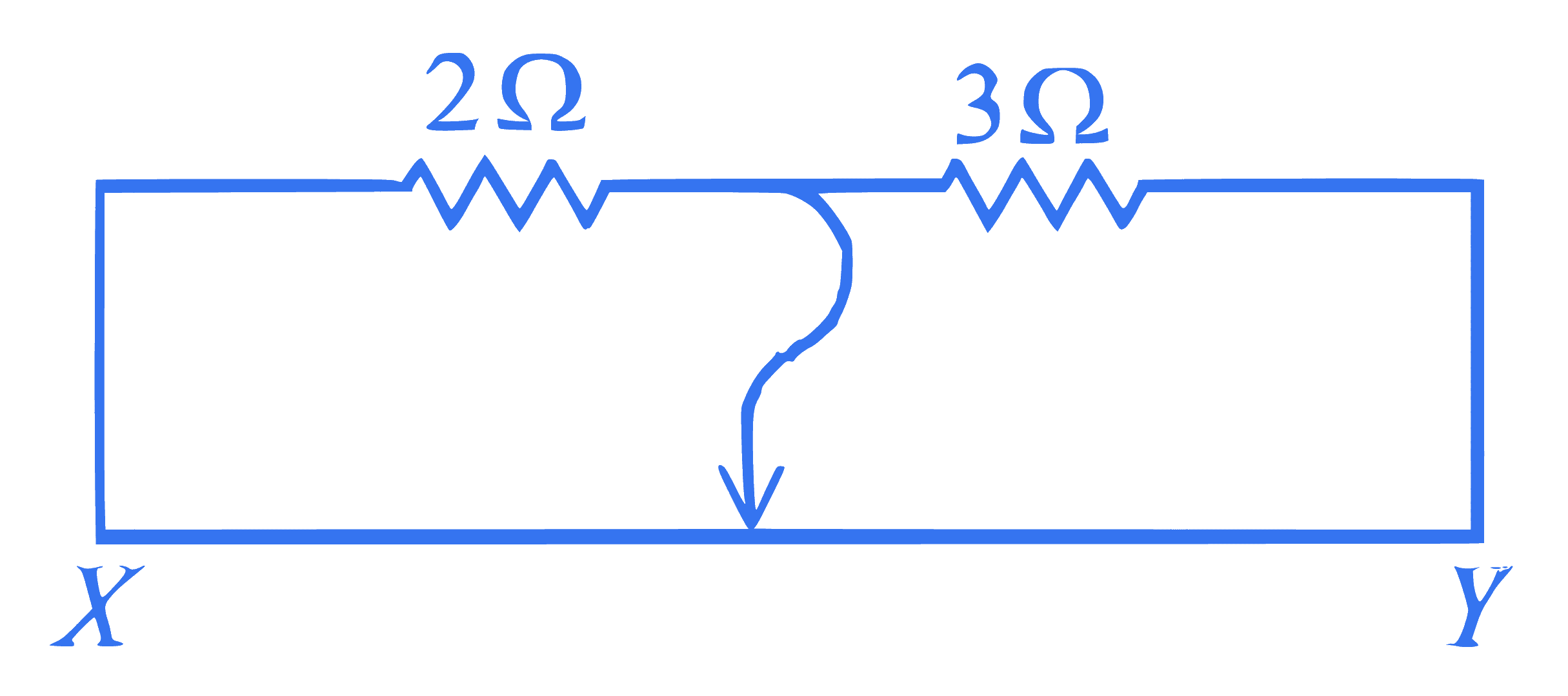

Q23.
mcq single
+4 / 1
Two resistors of $100 \Omega$ each are connected in series with a 9 V battery. A voltmeter of $400 \Omega$ resistance is connected to measure the voltage drop across one of the resistors. The voltmeter reading is $\_\_\_\_$ V.
Q24.
mcq single
+4 / 1
\text { Match the LIST-I with LIST-II }
List-I
List-II
A.
Radio-wave
I.
is produced by Magnetron valve
B.
Micro-wave
II.
due to change in the vibrational modes of atoms
C.
Infrared-wave
III.
due to inner shell electrons moving from higher energy level to lower energy level
D.
X-ray
IV.
due to rapid acceleration of electrons
Choose the correct answer from the options given below:
Q25.
mcq single
+4 / 1
For the series LCR circuit connected with $220 \mathrm{~V}, 50 \mathrm{~Hz}$ a.c source as shown in the figure, the power factor is $\frac{\alpha}{10}$. The value of $\alpha$ is $\_\_\_\_$ .
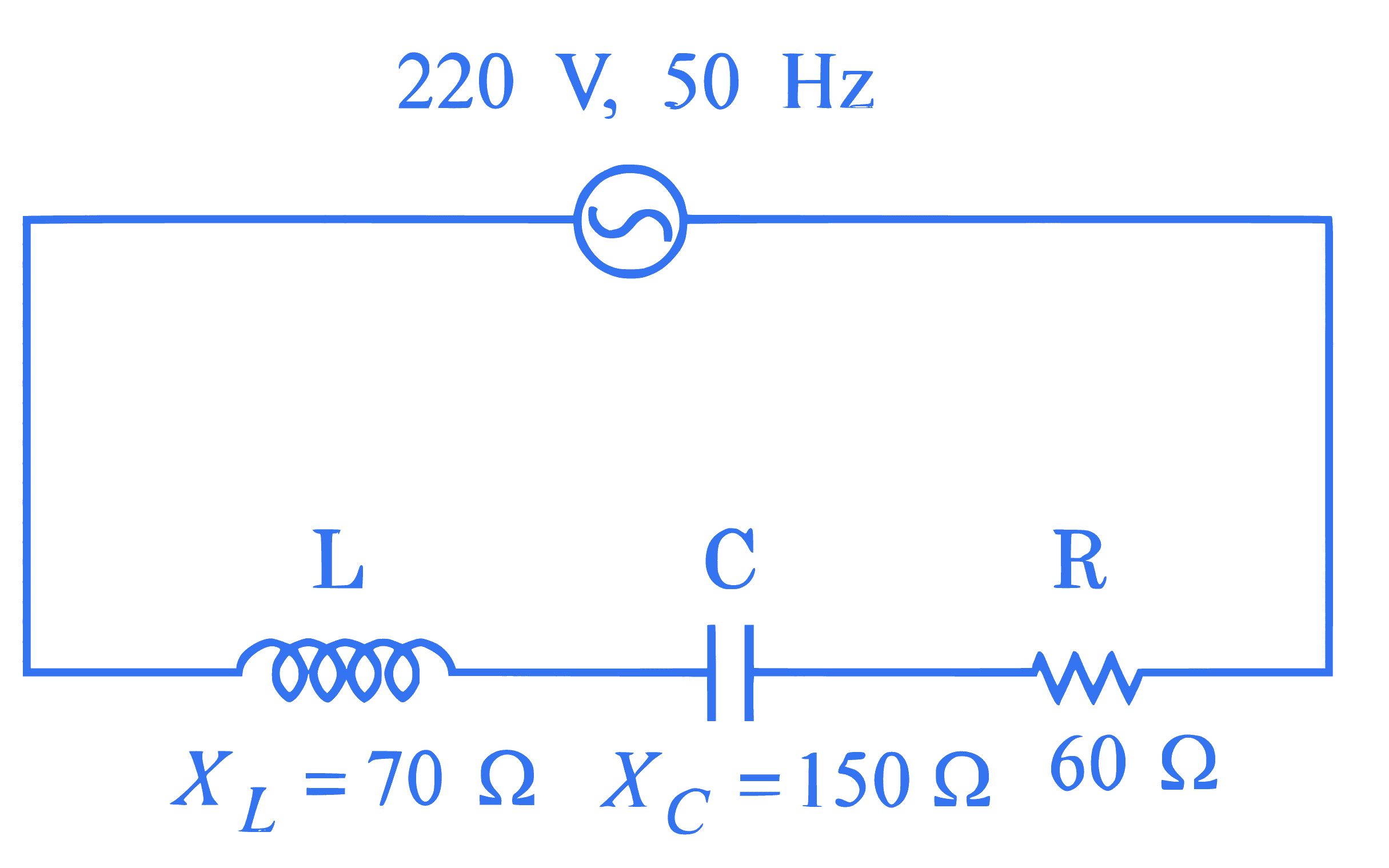

Q26.
numerical
+4 / 1
A voltage regulating circuit consisting of Zener diode, having break-down voltage of 10 V and maximum power dissipation of 0.4 W , is operated at 15 V . The approximate value of protective resistance in this circuit is $\_\_\_\_$ $\Omega$.
Q27.
mcq single
+4 / 1
A spring of force constant $15 \mathrm{~N} / \mathrm{m}$ is cut into two pieces. If the ratio of their length is $1: 3$, then the force constant of smaller piece is $\_\_\_\_$ $\mathrm{N} / \mathrm{m}$.
Q28.
mcq single
+4 / 1
A cylindrical block of mass $M$ and area of cross section $A$ is floating in a liquid of density $\rho$ and with its axis vertical. When depressed a little and released the block starts oscillating. The period of oscillation is $\_\_\_\_$
Q29.
mcq single
+4 / 1
Two masses 400 g and 350 g are suspended from the ends of a light string passing over a heavy pulley of radius 2 cm . When released from rest the heavier mass is observed to fall 81 cm in 9 s . The rotational inertia of the pulley is $\_\_\_\_$ $\mathrm{kg} \cdot \mathrm{m}^2$. $\left(\mathrm{g}=9.8 \mathrm{~m} / \mathrm{s}^2\right)$
Q30.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
List-II
A.
Magnetic induction
I.
<mi mathvariant="normal">M</mi>
<mi mathvariant="normal">L</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mi mathvariant="normal">L</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
MLT^(-2)A^(-2)
B.
Magnetic flux
II.
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
ML^(2)T^(-2)A^(-2)
C.
Magnetic permeability
III.
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>0</mn>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>0</mn>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
ML^(0)T^(-2)A^(-1)
D.
Self inductance
IV.
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext> </mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
<mi mathvariant="normal">M</mi>
<mrow data-mjx-texclass="ORD">
<mi mathvariant="normal">L</mi>
</mrow>
<mn>2</mn>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">T</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>2</mn>
</mrow>
<mrow data-mjx-texclass="ORD">
<mtext></mtext>
<mi mathvariant="normal">A</mi>
</mrow>
<mrow data-mjx-texclass="ORD">
<mo>−</mo>
<mn>1</mn>
</mrow>
ML^(2)T^(-2)A^(-1)
Choose the correct answer from the options given below:
Q31.
mcq single
+4 / 1
Density of water at $4^{\circ} \mathrm{C}$ and $20^{\circ} \mathrm{C}$ are $1000 \mathrm{~kg} / \mathrm{m}^3$ and $998 \mathrm{~kg} / \mathrm{m}^3$ respectively. The increase in internal energy of 4 kg of water when it is heated from $4^{\circ} \mathrm{C}$ to $20^{\circ} \mathrm{C}$ is $\_\_\_\_$ J.
(specific heat capacity of water $=4.2 \mathrm{~J} / \mathrm{kg}$. and 1 atmospheric pressure $=10^5 \mathrm{~Pa}$ )
Q32.
numerical
+4 / 1
A gas of certain mass filled in a closed cylinder at a pressure of 3.23 kPa has temperature $50^{\circ} \mathrm{C}$. The gas is now heated to double its temperature. The modified pressure is $\_\_\_\_$ Pa .
Q33.
numerical
+4 / 1
A short bar magnet placed with its axis at $30^{\circ}$ with an external field of 800 Gauss, experiences a torque of $0.016 \mathrm{~N} . \mathrm{m}$. The work done in moving it from most stable to most unstable position is $\alpha \times 10^{-3} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
Q34.
numerical
+4 / 1
Sixty four rain drops of radius 1 mm each falling down with a terminal velocity of $10 \mathrm{~cm} / \mathrm{s}$ coalesce to form a bigger drop. The terminal velocity of bigger drop is
$\_\_\_\_$ $\mathrm{cm} / \mathrm{s}$.
Q35.
mcq single
+4 / 1
A brass wire of length 2 m and radius 1 mm at $27^{\circ} \mathrm{C}$ is held taut between two rigid supports. Initially it was cooled to a temperature of $-43^{\circ} \mathrm{C}$ creating a tension $T$ in the wire. The temperature to which the wire has to be cooled in order to increase the tension in it to $1.4 T$, is $\_\_\_\_$ ${ }^{\circ} \mathrm{C}$.
Q36.
mcq single
+4 / 1
The electrostatic potential in a charged spherical region of radius $r$ varies as $V=a r^3+b$, where $a$ and $b$ are constants. The total charge in the sphere of unit radius is $\alpha \times \pi a \in_0$. The value of $\alpha$ is $\_\_\_\_$ .
(permittivity of vacuum is $\epsilon_0$ )
Q37.
mcq single
+4 / 1
Three charges $+2 q,+3 q$ and $-4 q$ are situated at $(0,-3 a),(2 a, 0)$ and $(-2 a, 0)$ respectively in the $x y$ plane. The resultant dipole moment about origin is $\_\_\_\_$ .
Q38.
mcq single
+4 / 1
There are three co-centric conducting spherical shells $A, B$ and $C$ of radii $a, b$ and $c$ respectively ( $c>b>a$ ) and they are charged with charge $q_1, q_2$ and $q_3$ respectively. The potentials of the spheres $A, B$ and $C$ respectively, are :
Q39.
mcq single
+4 / 1
Three masses $200 \mathrm{~kg}, 300 \mathrm{~kg}$ and 400 kg are placed at the vertices of an equilateral triangle with sides 20 m . They are rearranged on the vertices of a bigger triangle of side 25 m and with the same centre. The work done in this process $\_\_\_\_$ J. (Gravitational constant $\mathrm{G}=6.7 \times 10^{-11} \mathrm{~N} \mathrm{~m}^2 / \mathrm{kg}^2$ )
Q40.
mcq single
+4 / 1
An unpolarised light is incident at an interface of two dielectric media having refractive indices of 2 (incident medium) and $2 \sqrt{3}$ (medium) respectively. To satisfy the condition that reflected and refracted rays are perpendicular to each other, the angle of incidence is $\_\_\_\_$
Q41.
mcq single
+4 / 1
In a microscope of tube length 10 cm two convex lenses are arranged with focal length of 2 cm and 5 cm . Total magnification obtained with this system for normal adjustment is $(5)^k$. The value of $k$ is $\_\_\_\_$ .
Q42.
mcq single
+4 / 1
The exit surface of a prism with refractive index $n$ is coated with a material having refractive index $\frac{n}{2}$. When this prism is set for minimum angle of deviation, it exactly meets the condition of critical angle. The prism angle is $\_\_\_\_$ .
Q43.
mcq single
+4 / 1
Two electrons are moving in orbits of two hydrogen like atoms with speeds $3 \times 10^5 \mathrm{~m} / \mathrm{s}$ and $2.5 \times 10^5 \mathrm{~m} / \mathrm{s}$ respectively. If the radii of these orbits are nearly same then the possible order of energy states are $\_\_\_\_$ respectively.
Q44.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** For all elements, greater the mass of the nucleus, greater is the binding energy per nucleon.
**Statement II :** For all elements, nuclei with less binding energy per nucleon transforms to nuclei with greater binding energy per nucleon.
In the light of the above statements, choose the correct answer from the options given below
Q45.
mcq single
+4 / 1
A boy throws a ball into air at $45^{\circ}$ from the horizontal to land it on a roof of a building of height $H$. If the ball attains maximum height in 2 s and lands on the building in 3 s after launch, then value of $H$ is $\_\_\_\_$ m.
$$ \left(\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2\right) $$
Q46.
numerical
+4 / 1
In the given figure the blocks $A, B$ and $C$ weigh $4 \mathrm{~kg}, 6 \mathrm{~kg}$ and 8 kg respectively. The co-efficient of sliding friction between any two surfaces is 0.5 . The force $\vec{F}$ required to slide the block $C$ with constant speed is $\_\_\_\_$ N . (Use $g=10 \mathrm{~m} / \mathrm{s}^2$ )

Q47.
mcq single
+4 / 1
Two resistors $2 \Omega$ and $3 \Omega$ are connected in the gaps of bridge as shown in figure. The null point is obtained with the contact of jockey at some point on wire $X Y$. When an unknown resistor is connected in parallel with $3 \Omega$ resistor, the null point is shifted by 22.5 cm toward $Y$. The resistance of unknown resistor is $\_\_\_\_$ $\Omega$.

Q48.
mcq single
+4 / 1
Two resistors of $100 \Omega$ each are connected in series with a 9 V battery. A voltmeter of $400 \Omega$ resistance is connected to measure the voltage drop across one of the resistors. The voltmeter reading is $\_\_\_\_$ V.
Q49.
mcq single
+4 / 1
\text { Match the LIST-I with LIST-II }
List-I
List-II
A.
Radio-wave
I.
is produced by Magnetron valve
B.
Micro-wave
II.
due to change in the vibrational modes of atoms
C.
Infrared-wave
III.
due to inner shell electrons moving from higher energy level to lower energy level
D.
X-ray
IV.
due to rapid acceleration of electrons
Choose the correct answer from the options given below:
Q50.
mcq single
+4 / 1
For the series LCR circuit connected with $220 \mathrm{~V}, 50 \mathrm{~Hz}$ a.c source as shown in the figure, the power factor is $\frac{\alpha}{10}$. The value of $\alpha$ is $\_\_\_\_$ .

