JEE Main 2026 (Online) 2nd April Evening Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Among $Fe^{2+}$, $Fe^{3+}$, $Cr^{2+}$ and $Zn^{2+}$, the ion that shows positive borax bead test and with highest ionisation enthalpy is :
Q2.
numerical
+4 / 1
Number of paramagnetic ions among the following d- and f-block metal ions is __________.
Mn^(2+), Cu^(2+), Zn^(2+), Yb^(2+), Sc^(3+), La^(3+), Gd^(3+), Lu^(3+), Ti^(4+), Ce^(4+)
(Atomic number of Mn = 25, Cu = 29, Zn = 30, Yb = 70, Sc = 21, La = 57, Gd = 64, Lu = 71, Ti = 22, Ce = 58)
Q3.
mcq single
+4 / 1
Consider the reaction aX → bY, for which the rate constant at 30°C is $1 \times 10^{-3}\ \text{mol}^{-1}\ \text{L}\ \text{s}^{-1}$. Which of the following statements are true?
**A.** When concentration of ‘X’ is increased to four times, the rate of reaction becomes 16 times.
**B.** The reaction is a second order reaction.
**C.** The half-life period is independent of the concentration of X.
**D.** Decomposition of N$_2$O$_5$ is an example of the above reaction.
**E.** 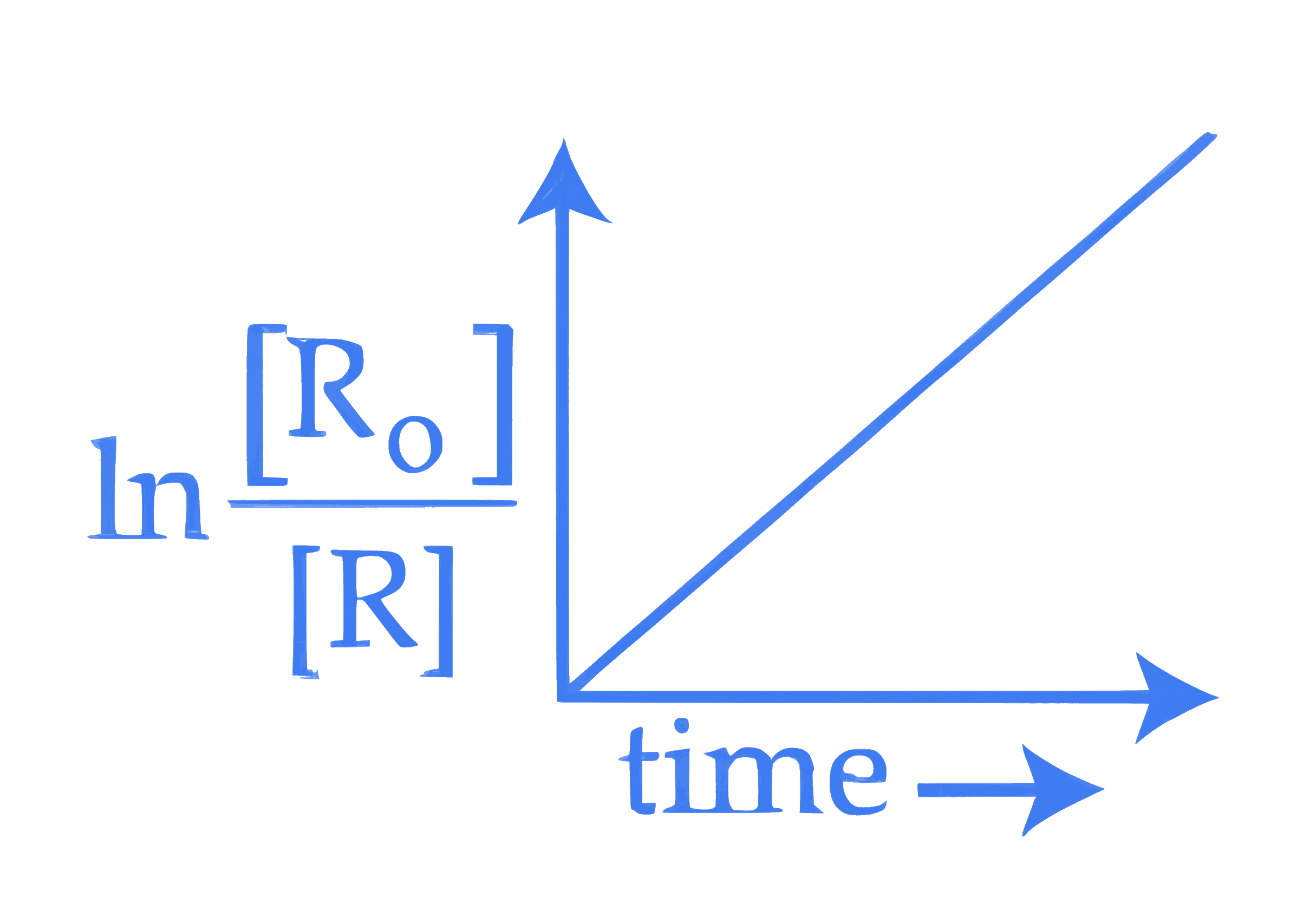 vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
 vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
Q4.
numerical
+4 / 1
Consider the following gas phase reaction being carried out in a closed vessel at $25 ^\circ$C.
$$2A(g) \longrightarrow 4B(g) + C(g)$$time
(min)total pressure of the
system
(mm Hg)30300∞600The pressure of C(g) at 30 minutes time interval would be ________ mm Hg. (nearest integer)
Q5.
mcq single
+4 / 1
**SF$_4$** is isostructural with :
**A.** BrF$_4^\ominus$
**B.** CH$_4$
**C.** IF$_4^{\oplus}$
**D.** XeF$_4$
**E.** XeO$_2$F$_2$
Choose the **correct** answer from the options given below :
Q6.
mcq single
+4 / 1
Correct statements regarding alkyl halides (R–X) among the following are :
**A.** Alcohol being less polar solvent as compared to water, alcoholic KOH favours elimination reaction with R–X.
**B.** Order of reactivity towards S~N~1 mechanism is C~6~H~5~–CH~2~–Cl > C~6~H~5~–CHCl–C~6~H~5~.
**C.** Non substituted aryl halides exhibit properties similar to alkyl halides.
**D.** Vinyl chloride is an example of haloalkene and allyl chloride is an example of haloalkyne.
**E.** R–Cl can be prepared by reacting R–OH with SOCl~2~ but Ar–Cl cannot be prepared by reacting Ar–OH with SOCl~2~.
Choose the **correct** answer from the options given below :
Q7.
mcq single
+4 / 1
Solution A is prepared by dissolving 1 g of a protein (molar mass = 50,000 g mol^(-1)) in 0.5 L of water at 300 K. Its osmotic pressure is $x$ bar. Solution B is made by dissolving 2 g of the same protein in 1 L of water at 300 K. Osmotic pressure of solution B is $y$ bar. Entire solution of A is mixed with entire solution of B at the same temperature. The osmotic pressure of resultant solution is $z$ bar. $x$, $y$ and $z$ respectively are:
(R = 0.083 L bar mol^(-1) K^(-1))
Q8.
mcq single
+4 / 1
The ratio of mass percentage (w/w) of C : H in a hydrocarbon is 12 : 1. It has two carbon atoms. The weight (in g) of CO~2~(g) formed when 3.38 g of this hydrocarbon is completely burnt in oxygen is :
(Given : Molar mass in g mol^(–1) C : 12, H : 1, O : 16)
Q9.
mcq single
+4 / 1
At 25°C, 20.0 mL of 0.2 M weak monoprotic acid HX is titrated against 0.2 M NaOH. The pH of the solution (a) at the start of the titration (when NaOH has not been added) and (b) when 10 mL of NaOH is added respectively, are :
Given :
$K_a = 5 \times 10^{-4}$
$\mathrm{p}K_a = 3.3$
$\alpha \ll 1$
Q10.
mcq single
+4 / 1
The first and second ionization constants of a weak dibasic acid H~2~A are $8.1 \times 10^{-8}$ and $1.0 \times 10^{-13}$ respectively. 0.1 mol of H~2~A was dissolved in 1 L of 0.1 M HCl solution. The concentration of HA^(-) in the resultant solution is:
Q11.
mcq single
+4 / 1
The correct set that contains all kinds (basic, acidic, amphoteric and neutral) of oxides is :
Q12.
mcq single
+4 / 1
Gas 'A' undergoes change from state 'X' to state 'Y'. In this process, the heat absorbed and work done by the gas is 10 J and 18 J respectively. Now gas is brought back to state 'X' by another process during which 6 J of heat is evolved. In the reverse process of 'Y' to 'X',
Q13.
mcq single
+4 / 1
Given below are two statements :
**Statement I:** The second ionization enthalpy of B, Al and Ga is in the order of B > Al > Ga.
**Statement II:** The correct order in terms of first ionization enthalpy is Si < Ge < Pb < Sn.
In the light of the above statements, choose the **correct** answer from the options given below :
Q14.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Among Zn, Mn, Sc and Cu, the energy required to remove the third valence electron is highest for Zn and lowest for Sc.
**Statement II :** The correct order of the following complexes in terms of CFSE is $[\text{Co(H}_2\text{O})_6]^{2+} < [\text{Co(H}_2\text{O})_6]^{3+} < [\text{Co(en)}_3]^{3+}$.
In the light of the above statements, choose the **correct** answer from the options given below :
Q15.
mcq single
+4 / 1
Which of the following complexes will show coordination isomerism?
A. $[\mathrm{Ag(NH}_3)_2][\mathrm{Ag(CN)}_2]$
B. $[\mathrm{Co(NH}_3)_6][\mathrm{Cr(CN)}_6]$
C. $[\mathrm{Co(NH}_3)_6][\mathrm{Co(CN)}_6]$
D. $[\mathrm{Fe(NH}_3)_6][\mathrm{Co(CN)}_6]$
E. $[\mathrm{Co(NH}_3)_6][\mathrm{Fe(CN)}_6]$
Choose the **correct** answer from the options given below:
Q16.
mcq single
+4 / 1
An organic compound “x” where molar ratio of C, O and H are equal, on treatment with 50% KOH under reflux followed by acidification produced “y”. The most likely structure of “y” is:
[Molar mass of ‘x’ is 58 g mol^(−1)]
Q17.
mcq single
+4 / 1
Identify compounds A and E in the following reaction sequence.
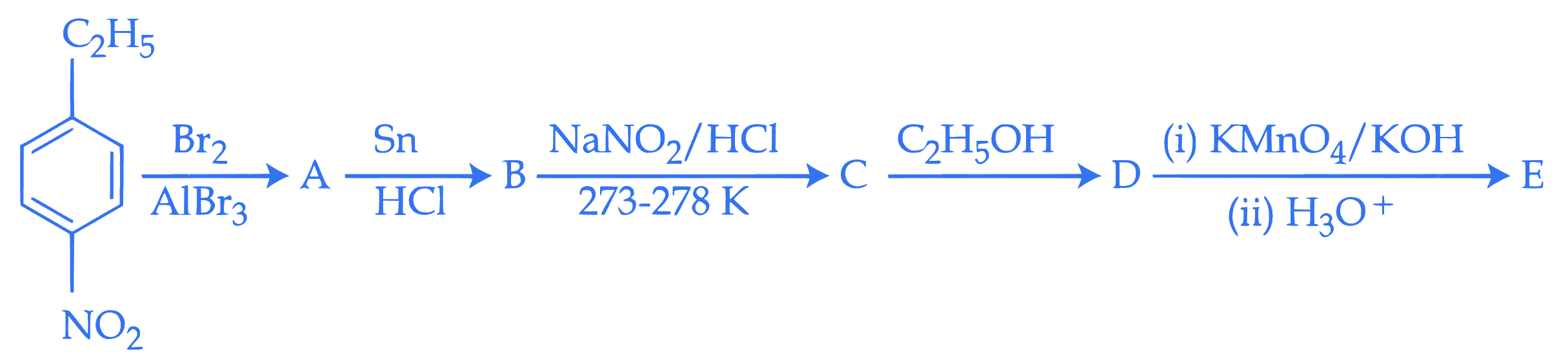

Q18.
mcq single
+4 / 1
A molecule (X) with following structure under mild acidic condition is hydrolysed to produce (Y) and (Z). Identify the correct statements about (Y) and (Z).
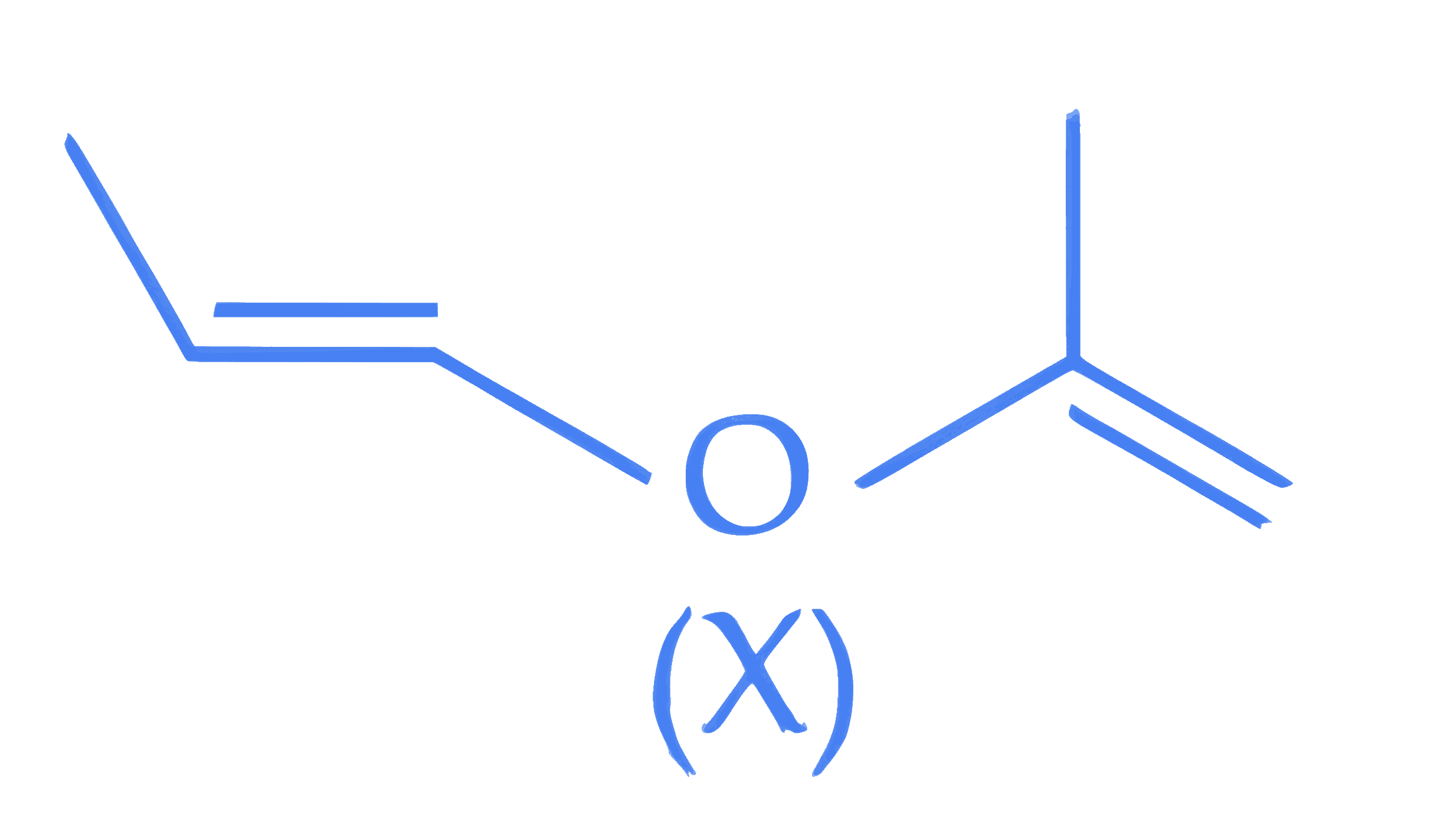 A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
 A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
Q19.
numerical
+4 / 1
Consider the following reactions sequence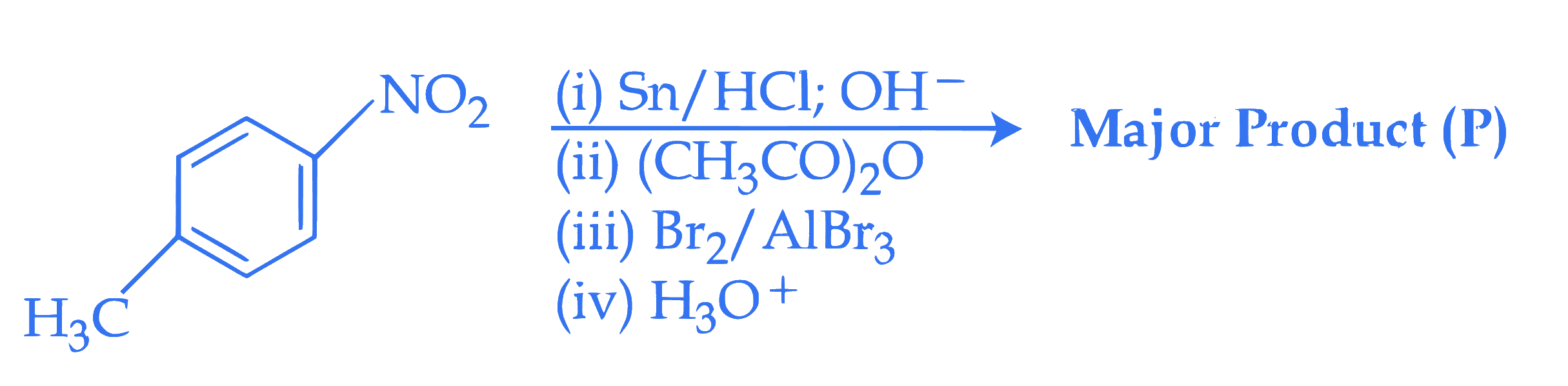 When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
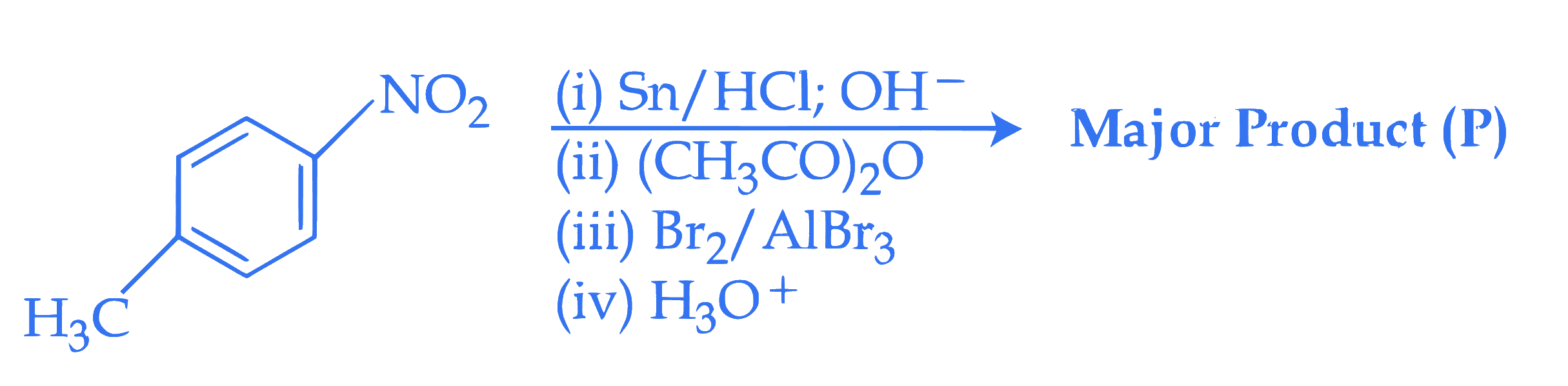 When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
Q20.
mcq single
+4 / 1
Complete combustion of $X$ g of an organic compound gave $0.25$ g of $CO_2$ and $0.12$ g of $H_2O$. If the % of carbon is $25\%$ and of hydrogen is $4.89\%$, then $X = \underline{\phantom{xxx}} \times 10^{-3}$ g. (Nearest integer)
(Molar mass of C, H and O are 12, 1 and 16 g mol$^{-1}$ respectively.)
Q21.
mcq single
+4 / 1
The compound (X) on
(i) on heating in the presence of anhydrous AlCl~3~ and HCl gas gives 2,4-dimethyl pentane
(ii) aromatization gives toluene and
(iii) cyclisation gives methyl cyclohexane
The correct name of compound (X) is:
Q22.
mcq single
+4 / 1
Identify the correct pair having amino acid (A) and the hormone (B) that is iodinated derivative of the amino acid (A).
(T and Y represent one letter code for amino acids)
Q23.
mcq single
+4 / 1
Given below are two statements :
**Statement I** : In 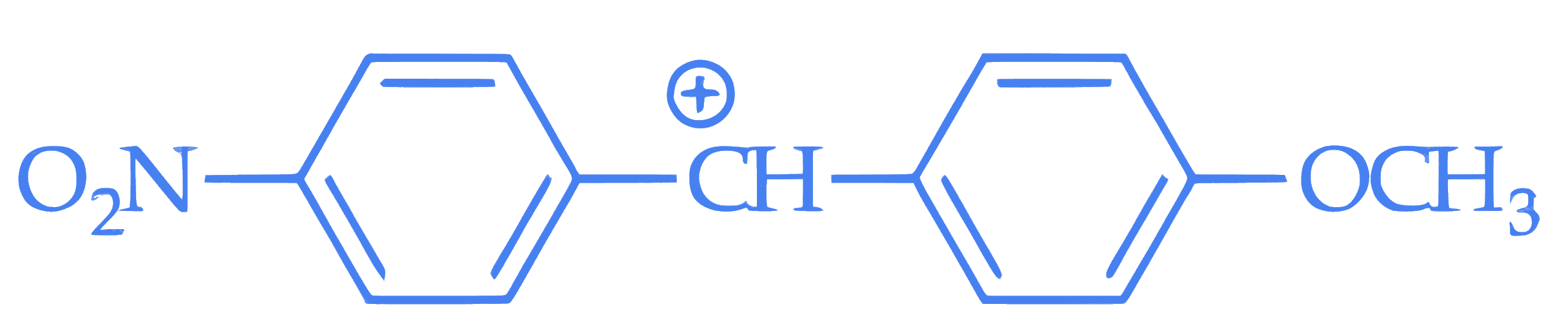 , the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In
, the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In 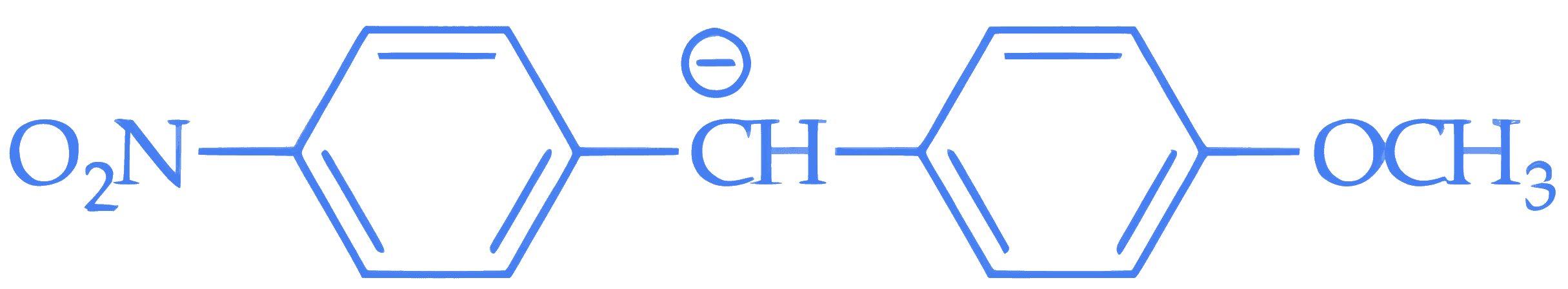 , the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
, the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
 , the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In
, the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In  , the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
, the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
Q24.
numerical
+4 / 1
Consider the following two half-cell reactions along with the standard reduction potential given :
$\mathrm{CO}_2 + 6\mathrm{H}^+ + 6e^- \longrightarrow \mathrm{CH}_3\mathrm{OH} + \mathrm{H}_2\mathrm{O}\qquad E_{\text{red}}^{\circ} = 0.02\ \mathrm{V}$
$\dfrac{1}{2} \mathrm{O}_2 + 2\mathrm{H}^+ + 2e^- \longrightarrow \mathrm{H}_2\mathrm{O}\qquad E_{\text{red}}^{\circ} = 1.23\ \mathrm{V}$
A fuel cell was set up using the above two reactions such that the cell operates under the standard condition of 1 bar pressure and 298 K temperature. The fuel cell works with 80% efficiency. If the work derived from the cell using 1 mol of $\mathrm{CH}_3\mathrm{OH}$ is used to compress an ideal gas isothermally against a constant pressure of 1 kPa, then the change in the volume of the gas,
$\Delta V = \underline{\hspace{2cm}}$ m$^3$. (nearest integer)
Given : $\mathrm{F} = 96500~\mathrm{C~mol}^{-1}$
Q25.
numerical
+4 / 1
The surface of sodium metal is irradiated with radiation of wavelength $x$ nm. The kinetic energy of ejected electrons is $2.8 \times 10^{-20}\ \text{J}$. The work function of sodium is $2.3$ eV. The value of $x$ is ________ $\times 10^2$ nm. (Nearest integer)
(Given: $h=6.6 \times 10^{-34}\ \text{J s}$ ; $1\ \text{eV} = 1.6 \times 10^{-19}\ \text{J}$ ; $c = 3.0 \times 10^8\ \text{m s}^{-1}$)
Q26.
mcq single
+4 / 1
Among $Fe^{2+}$, $Fe^{3+}$, $Cr^{2+}$ and $Zn^{2+}$, the ion that shows positive borax bead test and with highest ionisation enthalpy is :
Q27.
numerical
+4 / 1
Number of paramagnetic ions among the following d- and f-block metal ions is __________.
Mn^(2+), Cu^(2+), Zn^(2+), Yb^(2+), Sc^(3+), La^(3+), Gd^(3+), Lu^(3+), Ti^(4+), Ce^(4+)
(Atomic number of Mn = 25, Cu = 29, Zn = 30, Yb = 70, Sc = 21, La = 57, Gd = 64, Lu = 71, Ti = 22, Ce = 58)
Q28.
mcq single
+4 / 1
Consider the reaction aX → bY, for which the rate constant at 30°C is $1 \times 10^{-3}\ \text{mol}^{-1}\ \text{L}\ \text{s}^{-1}$. Which of the following statements are true?
**A.** When concentration of ‘X’ is increased to four times, the rate of reaction becomes 16 times.
**B.** The reaction is a second order reaction.
**C.** The half-life period is independent of the concentration of X.
**D.** Decomposition of N$_2$O$_5$ is an example of the above reaction.
**E.**  vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
 vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
vs time is valid for the above reaction.
Choose the **correct** answer from the options given below:
Q29.
numerical
+4 / 1
Consider the following gas phase reaction being carried out in a closed vessel at $25 ^\circ$C.
$$2A(g) \longrightarrow 4B(g) + C(g)$$time
(min)total pressure of the
system
(mm Hg)30300∞600The pressure of C(g) at 30 minutes time interval would be ________ mm Hg. (nearest integer)
Q30.
mcq single
+4 / 1
**SF$_4$** is isostructural with :
**A.** BrF$_4^\ominus$
**B.** CH$_4$
**C.** IF$_4^{\oplus}$
**D.** XeF$_4$
**E.** XeO$_2$F$_2$
Choose the **correct** answer from the options given below :
Q31.
mcq single
+4 / 1
Correct statements regarding alkyl halides (R–X) among the following are :
**A.** Alcohol being less polar solvent as compared to water, alcoholic KOH favours elimination reaction with R–X.
**B.** Order of reactivity towards S~N~1 mechanism is C~6~H~5~–CH~2~–Cl > C~6~H~5~–CHCl–C~6~H~5~.
**C.** Non substituted aryl halides exhibit properties similar to alkyl halides.
**D.** Vinyl chloride is an example of haloalkene and allyl chloride is an example of haloalkyne.
**E.** R–Cl can be prepared by reacting R–OH with SOCl~2~ but Ar–Cl cannot be prepared by reacting Ar–OH with SOCl~2~.
Choose the **correct** answer from the options given below :
Q32.
mcq single
+4 / 1
Solution A is prepared by dissolving 1 g of a protein (molar mass = 50,000 g mol^(-1)) in 0.5 L of water at 300 K. Its osmotic pressure is $x$ bar. Solution B is made by dissolving 2 g of the same protein in 1 L of water at 300 K. Osmotic pressure of solution B is $y$ bar. Entire solution of A is mixed with entire solution of B at the same temperature. The osmotic pressure of resultant solution is $z$ bar. $x$, $y$ and $z$ respectively are:
(R = 0.083 L bar mol^(-1) K^(-1))
Q33.
mcq single
+4 / 1
The ratio of mass percentage (w/w) of C : H in a hydrocarbon is 12 : 1. It has two carbon atoms. The weight (in g) of CO~2~(g) formed when 3.38 g of this hydrocarbon is completely burnt in oxygen is :
(Given : Molar mass in g mol^(–1) C : 12, H : 1, O : 16)
Q34.
mcq single
+4 / 1
At 25°C, 20.0 mL of 0.2 M weak monoprotic acid HX is titrated against 0.2 M NaOH. The pH of the solution (a) at the start of the titration (when NaOH has not been added) and (b) when 10 mL of NaOH is added respectively, are :
Given :
$K_a = 5 \times 10^{-4}$
$\mathrm{p}K_a = 3.3$
$\alpha \ll 1$
Q35.
mcq single
+4 / 1
The first and second ionization constants of a weak dibasic acid H~2~A are $8.1 \times 10^{-8}$ and $1.0 \times 10^{-13}$ respectively. 0.1 mol of H~2~A was dissolved in 1 L of 0.1 M HCl solution. The concentration of HA^(-) in the resultant solution is:
Q36.
mcq single
+4 / 1
The correct set that contains all kinds (basic, acidic, amphoteric and neutral) of oxides is :
Q37.
mcq single
+4 / 1
Gas 'A' undergoes change from state 'X' to state 'Y'. In this process, the heat absorbed and work done by the gas is 10 J and 18 J respectively. Now gas is brought back to state 'X' by another process during which 6 J of heat is evolved. In the reverse process of 'Y' to 'X',
Q38.
mcq single
+4 / 1
Given below are two statements :
**Statement I:** The second ionization enthalpy of B, Al and Ga is in the order of B > Al > Ga.
**Statement II:** The correct order in terms of first ionization enthalpy is Si < Ge < Pb < Sn.
In the light of the above statements, choose the **correct** answer from the options given below :
Q39.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Among Zn, Mn, Sc and Cu, the energy required to remove the third valence electron is highest for Zn and lowest for Sc.
**Statement II :** The correct order of the following complexes in terms of CFSE is $[\text{Co(H}_2\text{O})_6]^{2+} < [\text{Co(H}_2\text{O})_6]^{3+} < [\text{Co(en)}_3]^{3+}$.
In the light of the above statements, choose the **correct** answer from the options given below :
Q40.
mcq single
+4 / 1
Which of the following complexes will show coordination isomerism?
A. $[\mathrm{Ag(NH}_3)_2][\mathrm{Ag(CN)}_2]$
B. $[\mathrm{Co(NH}_3)_6][\mathrm{Cr(CN)}_6]$
C. $[\mathrm{Co(NH}_3)_6][\mathrm{Co(CN)}_6]$
D. $[\mathrm{Fe(NH}_3)_6][\mathrm{Co(CN)}_6]$
E. $[\mathrm{Co(NH}_3)_6][\mathrm{Fe(CN)}_6]$
Choose the **correct** answer from the options given below:
Q41.
mcq single
+4 / 1
An organic compound “x” where molar ratio of C, O and H are equal, on treatment with 50% KOH under reflux followed by acidification produced “y”. The most likely structure of “y” is:
[Molar mass of ‘x’ is 58 g mol^(−1)]
Q42.
mcq single
+4 / 1
Identify compounds A and E in the following reaction sequence.


Q43.
mcq single
+4 / 1
A molecule (X) with following structure under mild acidic condition is hydrolysed to produce (Y) and (Z). Identify the correct statements about (Y) and (Z).
 A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
 A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$_3$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the **correct** answer from the options given below :
Q44.
numerical
+4 / 1
Consider the following reactions sequence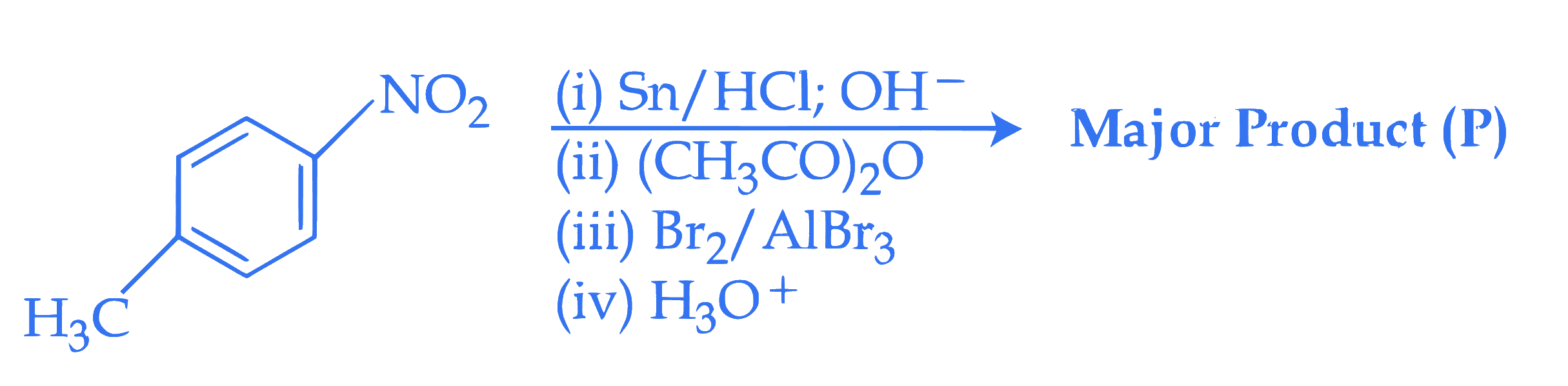 When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
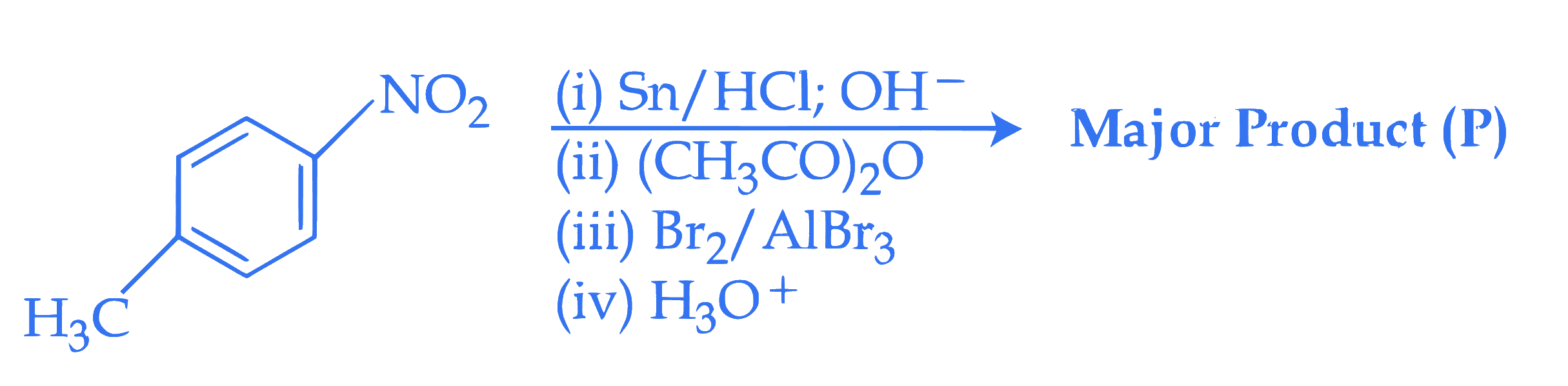 When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
When the product (P) is subjected to Carius analysis using AgNO~3~, 1.0 g of the product (P) will produce _________ g of the precipitate of AgBr. (Nearest Integer)
(Given : molar mass in g mol^(-1) C : 12, H : 1, O : 16, N : 14, Br : 80, Ag : 108)
Q45.
mcq single
+4 / 1
Complete combustion of $X$ g of an organic compound gave $0.25$ g of $CO_2$ and $0.12$ g of $H_2O$. If the % of carbon is $25\%$ and of hydrogen is $4.89\%$, then $X = \underline{\phantom{xxx}} \times 10^{-3}$ g. (Nearest integer)
(Molar mass of C, H and O are 12, 1 and 16 g mol$^{-1}$ respectively.)
Q46.
mcq single
+4 / 1
The compound (X) on
(i) on heating in the presence of anhydrous AlCl~3~ and HCl gas gives 2,4-dimethyl pentane
(ii) aromatization gives toluene and
(iii) cyclisation gives methyl cyclohexane
The correct name of compound (X) is:
Q47.
mcq single
+4 / 1
Identify the correct pair having amino acid (A) and the hormone (B) that is iodinated derivative of the amino acid (A).
(T and Y represent one letter code for amino acids)
Q48.
mcq single
+4 / 1
Given below are two statements :
**Statement I** : In  , the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In
, the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In  , the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
, the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
 , the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In
, the carbocation is stabilised by +R effect of –OCH~3~ group.
**Statement II** : In  , the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
, the carbanion is stabilised by –R effect of –NO~2~ group.
In the light of the above statements, choose the **correct** answer from the options given below:
Q49.
numerical
+4 / 1
Consider the following two half-cell reactions along with the standard reduction potential given :
$\mathrm{CO}_2 + 6\mathrm{H}^+ + 6e^- \longrightarrow \mathrm{CH}_3\mathrm{OH} + \mathrm{H}_2\mathrm{O}\qquad E_{\text{red}}^{\circ} = 0.02\ \mathrm{V}$
$\dfrac{1}{2} \mathrm{O}_2 + 2\mathrm{H}^+ + 2e^- \longrightarrow \mathrm{H}_2\mathrm{O}\qquad E_{\text{red}}^{\circ} = 1.23\ \mathrm{V}$
A fuel cell was set up using the above two reactions such that the cell operates under the standard condition of 1 bar pressure and 298 K temperature. The fuel cell works with 80% efficiency. If the work derived from the cell using 1 mol of $\mathrm{CH}_3\mathrm{OH}$ is used to compress an ideal gas isothermally against a constant pressure of 1 kPa, then the change in the volume of the gas,
$\Delta V = \underline{\hspace{2cm}}$ m$^3$. (nearest integer)
Given : $\mathrm{F} = 96500~\mathrm{C~mol}^{-1}$
Q50.
numerical
+4 / 1
The surface of sodium metal is irradiated with radiation of wavelength $x$ nm. The kinetic energy of ejected electrons is $2.8 \times 10^{-20}\ \text{J}$. The work function of sodium is $2.3$ eV. The value of $x$ is ________ $\times 10^2$ nm. (Nearest integer)
(Given: $h=6.6 \times 10^{-34}\ \text{J s}$ ; $1\ \text{eV} = 1.6 \times 10^{-19}\ \text{J}$ ; $c = 3.0 \times 10^8\ \text{m s}^{-1}$)
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
The mean and variance of **n** observations are 8 and 16, respectively. If the sum of the first (n − 1) observations is 48 and the sum of squares of the first (n − 1) observations is 496, then the value of n is :
Q2.
mcq single
+4 / 1
Two adjacent sides of a parallelogram PQRS are given by $\overrightarrow{PQ} = \hat{j} + \hat{k}$ and $\overrightarrow{PS} = \hat{i} - \hat{j}$. If the side PS is rotated about the point P by an acute angle $\alpha$ in the plane of the parallelogram so that it becomes perpendicular to the side PQ, then $\sin^2\left(\frac{5\alpha}{2}\right) - \sin^2\left(\frac{\alpha}{2}\right)$ is equal to:
Q3.
mcq single
+4 / 1
Let the vectors $\vec{a} = -\hat{i} + \hat{j} + 3\hat{k}$ and $\vec{b} = \hat{i} + 3\hat{j} + \hat{k}$. For some $\lambda, \mu \in \mathbb{R}$, let $\vec{c} = \lambda \vec{a} + \mu \vec{b}$.
If $\vec{c} \cdot (3\hat{i} - 6\hat{j} + 2\hat{k}) = 10$ and $\vec{c} \cdot (\hat{i} + \hat{j} + \hat{k}) = -2$, then $|\vec{c}|^2$ is equal to :
Q4.
mcq single
+4 / 1
Let $\alpha, \beta$ be the roots of the equation $x^2 - 3x + r = 0$, and $\frac{\alpha}{2}, 2\beta$ be the roots of the equation $x^2 + 3x + r = 0$.
If the roots of the equation $x^2 + 6x = m$ are $2\alpha + \beta + 2r$ and $\alpha - 2\beta - \frac{r}{2}$, then $m$ is equal to :
Q5.
mcq single
+4 / 1
A man throws a fair coin repeatedly. He gets 10 points for each head he throws and 5 points for each tail he throws. If the probability that he gets exactly 30 points is $\frac{m}{n}$, gcd $(m, n) = 1$, then m + n is equal to:
Q6.
mcq single
+4 / 1
Let the point A be the foot of perpendicular drawn from the point P$(a, b, 0)$ on the line
$$\frac{x-1}{2} = \frac{y-2}{1} = \frac{z-\alpha}{3}.$$
If the midpoint of the line segment PA is $$\left(0, \frac{3}{4}, -\frac{1}{4}\right),$$ then the value of $a^2 + b^2 + \alpha^2$ is equal to:
Q7.
mcq single
+4 / 1
The sum $\frac{1^3}{1} + \frac{1^3 + 2^3}{1 + 3} + \frac{1^3 + 2^3 + 3^3}{1 + 3 + 5} + \ldots$ up to 8 terms, is:
Q8.
mcq single
+4 / 1
Let $a_1, a_2, a_3, \ldots$ be an A.P. and $g_1 = a_1, g_2, g_3, \ldots$ be an increasing G.P. If $a_1 = a_2 + g_2 = 1$ and $a_3 + g_3 = 4$, then $a_{10} + g_5$ is equal to:
Q9.
numerical
+4 / 1
The number of points in the interval $[2, 4]$, at which the function $f(x) = \left[ x^2 - x - \frac{1}{2} \right]$, where $[ \cdot ]$ denotes the greatest integer function, is discontinuous, is ________.
Q10.
mcq single
+4 / 1
Let $f(x)$ be a polynomial of degree 5, and have extrema at $x = 1$ and $x = -1$. If $\lim\limits_{x \to 0} \left( \frac{f(x)}{x^3} \right) = -5$, then $f(2) - f(-2)$ is equal to:
Q11.
mcq single
+4 / 1
Let $P = \{ \theta \in [0, 4\pi] : \tan^2 \theta \neq 1 \}$ and $S = \{ a \in \mathbb{Z} : 2(\cos^8 \theta - \sin^8 \theta) \sec 2 \theta = a^2, \theta \in P \}$. Then $n(S)$ is:
Q12.
numerical
+4 / 1
If the area of the region bounded by $16x^2 - 9y^2 = 144$ and $8x - 3y = 24$ is $A$, then $3(A + 6 \log_e(3))$ is equal to ________.
Q13.
numerical
+4 / 1
Consider the matrices $A = \begin{bmatrix} 2 & -2 \\ 4 & -2 \end{bmatrix}$ and $B = \begin{bmatrix} 3 & 9 \\ 1 & 3 \end{bmatrix}$. If matrices $P$ and $Q$ are such that $PA = B$ and $AQ = B$, then the absolute value of the sum of the diagonal elements of $2(P + Q)$ is ________.
Q14.
mcq single
+4 / 1
If the system of equations
$x + 5y + 6z = 4$
$2x + 3y + 4z = 7$
$x + 6y + az = b$
has infinitely many solutions, then the point $(a, b)$ lies on the line
Q15.
mcq single
+4 / 1
Let $f(x) = \int \left( \frac{16x + 24}{x^2 + 2x - 15} \right) dx$. If $f(4) = 14 \log_e(3)$ and $f(7) = \log_e(2^\alpha \cdot 3^\beta)$, $\alpha, \beta \in \mathbb{N}$, then $\alpha + \beta$ is equal to :
Q16.
mcq single
+4 / 1
Let the parabola $y = x^2 + px + q$ passing through the point $(1, -1)$ be such that the distance between its vertex and the $x$-axis is minimum. Then the value of $p^2 + q^2$ is:
Q17.
mcq single
+4 / 1
If for $3 \leq r \leq 30$, $\left({^{30}C_{30-r}}\right) + 3\left({^{30}C_{31-r}}\right) + 3\left({^{30}C_{32-r}}\right) + \left({^{30}C_{33-r}}\right) = {^mC_r}$, then m equals :
Q18.
numerical
+4 / 1
Let $A = \{2, 3, 4, 5, 6\}$. Let $R$ be a relation on the set $A \times A$ given by $(x, y)R(z, w)$ if and only if $x$ divides $z$ and $y \leq w$. Then the number of elements in $R$ is _________.
Q19.
mcq single
+4 / 1
Let the circles $C_1:|z| = r$ and $C_2:|z - 3 - 4i| = 5$, $z \in \mathbb{C}$, be such that $C_2$ lies within $C_1$.
If $z_1$ moves on $C_1$, $z_2$ moves on $C_2$ and $\min |z_1 - z_2| = 2$, then $\max |z_1 - z_2|$ is equal to:
Q20.
mcq single
+4 / 1
Let O be the origin, and P and Q be two points on the rectangular hyperbola $xy = 12$ such that the midpoint of the line segment PQ is $\left( \frac{1}{2}, -\frac{1}{2} \right)$. Then the area of the triangle OPQ equals :
Q21.
mcq single
+4 / 1
Let $x = x(y)$ be the solution of the differential equation $2y^2 \frac{dx}{dy} - 2xy + x^2 = 0$, $y > 1$, $x(e) = e$.
Then $x(e^2)$ is equal to:
Q22.
mcq single
+4 / 1
Let a circle pass through the origin and its centre be the point of intersection of two mutually perpendicular lines $x + (k-1)y + 3 = 0$ and $2x + k^2y - 4 = 0$. If the line $x - y + 2 = 0$ intersects the circle at the points A and B, then $(AB)^2$ is equal to:
Q23.
numerical
+4 / 1
Let A be the point (3, 0) and circles with variable diameter AB touch the circle $x^2 + y^2 = 36$ internally. Let the curve C be the locus of the point B. If the eccentricity of C is $e$, then $72e^2$ is equal to ________.
Q24.
mcq single
+4 / 1
Let $p_n$ denote the total number of triangles formed by joining the vertices of an $n$-side regular polygon.
If $p_{n+1} - p_n = 66$, then the sum of all distinct prime divisors of $n$ is :
Q25.
mcq single
+4 / 1
The value of $\int\limits_{0}^{20\pi} (\sin^4 x + \cos^4 x) dx$ is equal to:
Q26.
mcq single
+4 / 1
The mean and variance of **n** observations are 8 and 16, respectively. If the sum of the first (n − 1) observations is 48 and the sum of squares of the first (n − 1) observations is 496, then the value of n is :
Q27.
mcq single
+4 / 1
Two adjacent sides of a parallelogram PQRS are given by $\overrightarrow{PQ} = \hat{j} + \hat{k}$ and $\overrightarrow{PS} = \hat{i} - \hat{j}$. If the side PS is rotated about the point P by an acute angle $\alpha$ in the plane of the parallelogram so that it becomes perpendicular to the side PQ, then $\sin^2\left(\frac{5\alpha}{2}\right) - \sin^2\left(\frac{\alpha}{2}\right)$ is equal to:
Q28.
mcq single
+4 / 1
Let the vectors $\vec{a} = -\hat{i} + \hat{j} + 3\hat{k}$ and $\vec{b} = \hat{i} + 3\hat{j} + \hat{k}$. For some $\lambda, \mu \in \mathbb{R}$, let $\vec{c} = \lambda \vec{a} + \mu \vec{b}$.
If $\vec{c} \cdot (3\hat{i} - 6\hat{j} + 2\hat{k}) = 10$ and $\vec{c} \cdot (\hat{i} + \hat{j} + \hat{k}) = -2$, then $|\vec{c}|^2$ is equal to :
Q29.
mcq single
+4 / 1
Let $\alpha, \beta$ be the roots of the equation $x^2 - 3x + r = 0$, and $\frac{\alpha}{2}, 2\beta$ be the roots of the equation $x^2 + 3x + r = 0$.
If the roots of the equation $x^2 + 6x = m$ are $2\alpha + \beta + 2r$ and $\alpha - 2\beta - \frac{r}{2}$, then $m$ is equal to :
Q30.
mcq single
+4 / 1
A man throws a fair coin repeatedly. He gets 10 points for each head he throws and 5 points for each tail he throws. If the probability that he gets exactly 30 points is $\frac{m}{n}$, gcd $(m, n) = 1$, then m + n is equal to:
Q31.
mcq single
+4 / 1
Let the point A be the foot of perpendicular drawn from the point P$(a, b, 0)$ on the line
$$\frac{x-1}{2} = \frac{y-2}{1} = \frac{z-\alpha}{3}.$$
If the midpoint of the line segment PA is $$\left(0, \frac{3}{4}, -\frac{1}{4}\right),$$ then the value of $a^2 + b^2 + \alpha^2$ is equal to:
Q32.
mcq single
+4 / 1
The sum $\frac{1^3}{1} + \frac{1^3 + 2^3}{1 + 3} + \frac{1^3 + 2^3 + 3^3}{1 + 3 + 5} + \ldots$ up to 8 terms, is:
Q33.
mcq single
+4 / 1
Let $a_1, a_2, a_3, \ldots$ be an A.P. and $g_1 = a_1, g_2, g_3, \ldots$ be an increasing G.P. If $a_1 = a_2 + g_2 = 1$ and $a_3 + g_3 = 4$, then $a_{10} + g_5$ is equal to:
Q34.
numerical
+4 / 1
The number of points in the interval $[2, 4]$, at which the function $f(x) = \left[ x^2 - x - \frac{1}{2} \right]$, where $[ \cdot ]$ denotes the greatest integer function, is discontinuous, is ________.
Q35.
mcq single
+4 / 1
Let $f(x)$ be a polynomial of degree 5, and have extrema at $x = 1$ and $x = -1$. If $\lim\limits_{x \to 0} \left( \frac{f(x)}{x^3} \right) = -5$, then $f(2) - f(-2)$ is equal to:
Q36.
mcq single
+4 / 1
Let $P = \{ \theta \in [0, 4\pi] : \tan^2 \theta \neq 1 \}$ and $S = \{ a \in \mathbb{Z} : 2(\cos^8 \theta - \sin^8 \theta) \sec 2 \theta = a^2, \theta \in P \}$. Then $n(S)$ is:
Q37.
numerical
+4 / 1
If the area of the region bounded by $16x^2 - 9y^2 = 144$ and $8x - 3y = 24$ is $A$, then $3(A + 6 \log_e(3))$ is equal to ________.
Q38.
numerical
+4 / 1
Consider the matrices $A = \begin{bmatrix} 2 & -2 \\ 4 & -2 \end{bmatrix}$ and $B = \begin{bmatrix} 3 & 9 \\ 1 & 3 \end{bmatrix}$. If matrices $P$ and $Q$ are such that $PA = B$ and $AQ = B$, then the absolute value of the sum of the diagonal elements of $2(P + Q)$ is ________.
Q39.
mcq single
+4 / 1
If the system of equations
$x + 5y + 6z = 4$
$2x + 3y + 4z = 7$
$x + 6y + az = b$
has infinitely many solutions, then the point $(a, b)$ lies on the line
Q40.
mcq single
+4 / 1
Let $f(x) = \int \left( \frac{16x + 24}{x^2 + 2x - 15} \right) dx$. If $f(4) = 14 \log_e(3)$ and $f(7) = \log_e(2^\alpha \cdot 3^\beta)$, $\alpha, \beta \in \mathbb{N}$, then $\alpha + \beta$ is equal to :
Q41.
mcq single
+4 / 1
Let the parabola $y = x^2 + px + q$ passing through the point $(1, -1)$ be such that the distance between its vertex and the $x$-axis is minimum. Then the value of $p^2 + q^2$ is:
Q42.
mcq single
+4 / 1
If for $3 \leq r \leq 30$, $\left({^{30}C_{30-r}}\right) + 3\left({^{30}C_{31-r}}\right) + 3\left({^{30}C_{32-r}}\right) + \left({^{30}C_{33-r}}\right) = {^mC_r}$, then m equals :
Q43.
numerical
+4 / 1
Let $A = \{2, 3, 4, 5, 6\}$. Let $R$ be a relation on the set $A \times A$ given by $(x, y)R(z, w)$ if and only if $x$ divides $z$ and $y \leq w$. Then the number of elements in $R$ is _________.
Q44.
mcq single
+4 / 1
Let the circles $C_1:|z| = r$ and $C_2:|z - 3 - 4i| = 5$, $z \in \mathbb{C}$, be such that $C_2$ lies within $C_1$.
If $z_1$ moves on $C_1$, $z_2$ moves on $C_2$ and $\min |z_1 - z_2| = 2$, then $\max |z_1 - z_2|$ is equal to:
Q45.
mcq single
+4 / 1
Let O be the origin, and P and Q be two points on the rectangular hyperbola $xy = 12$ such that the midpoint of the line segment PQ is $\left( \frac{1}{2}, -\frac{1}{2} \right)$. Then the area of the triangle OPQ equals :
Q46.
mcq single
+4 / 1
Let $x = x(y)$ be the solution of the differential equation $2y^2 \frac{dx}{dy} - 2xy + x^2 = 0$, $y > 1$, $x(e) = e$.
Then $x(e^2)$ is equal to:
Q47.
mcq single
+4 / 1
Let a circle pass through the origin and its centre be the point of intersection of two mutually perpendicular lines $x + (k-1)y + 3 = 0$ and $2x + k^2y - 4 = 0$. If the line $x - y + 2 = 0$ intersects the circle at the points A and B, then $(AB)^2$ is equal to:
Q48.
numerical
+4 / 1
Let A be the point (3, 0) and circles with variable diameter AB touch the circle $x^2 + y^2 = 36$ internally. Let the curve C be the locus of the point B. If the eccentricity of C is $e$, then $72e^2$ is equal to ________.
Q49.
mcq single
+4 / 1
Let $p_n$ denote the total number of triangles formed by joining the vertices of an $n$-side regular polygon.
If $p_{n+1} - p_n = 66$, then the sum of all distinct prime divisors of $n$ is :
Q50.
mcq single
+4 / 1
The value of $\int\limits_{0}^{20\pi} (\sin^4 x + \cos^4 x) dx$ is equal to:
Physics
Physics
Q1.
mcq single
+4 / 1
A spherical ball of mass 2 kg falls from a height of 10 m and is brought to rest after penetrating 10 cm into sand.
The average force exerted by sand on the ball is _________ N.
(Take $g = 10 \ \mathrm{m/s^2}$)
Q2.
mcq single
+4 / 1
If X and Y are the inputs, the given circuit works as _________.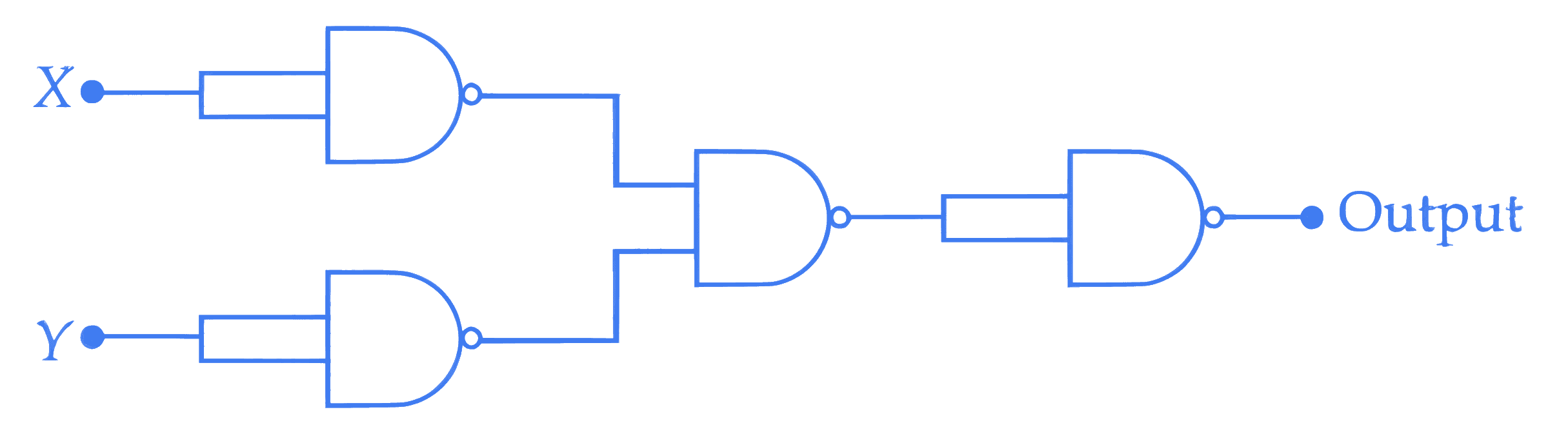

Q3.
numerical
+4 / 1
In a Young's double slit experiment, the intensity at some point on the screen is found to be $\frac{3}{4}$ times of the maximum of the interference pattern. The path difference between the interfering waves at this point is $\frac{\lambda}{x}$ where $\lambda$ is wavelength of the incident light. The value of $x$ is _______.
Q4.
mcq single
+4 / 1
The equation of motion of a particle is given by $x = a \sin(50t + \pi/3)$ cm. The particle will come to rest at time $t_1$ and it will have zero acceleration at time $t_2$. The $t_1$ and $t_2$ respectively are ________.
Q5.
numerical
+4 / 1
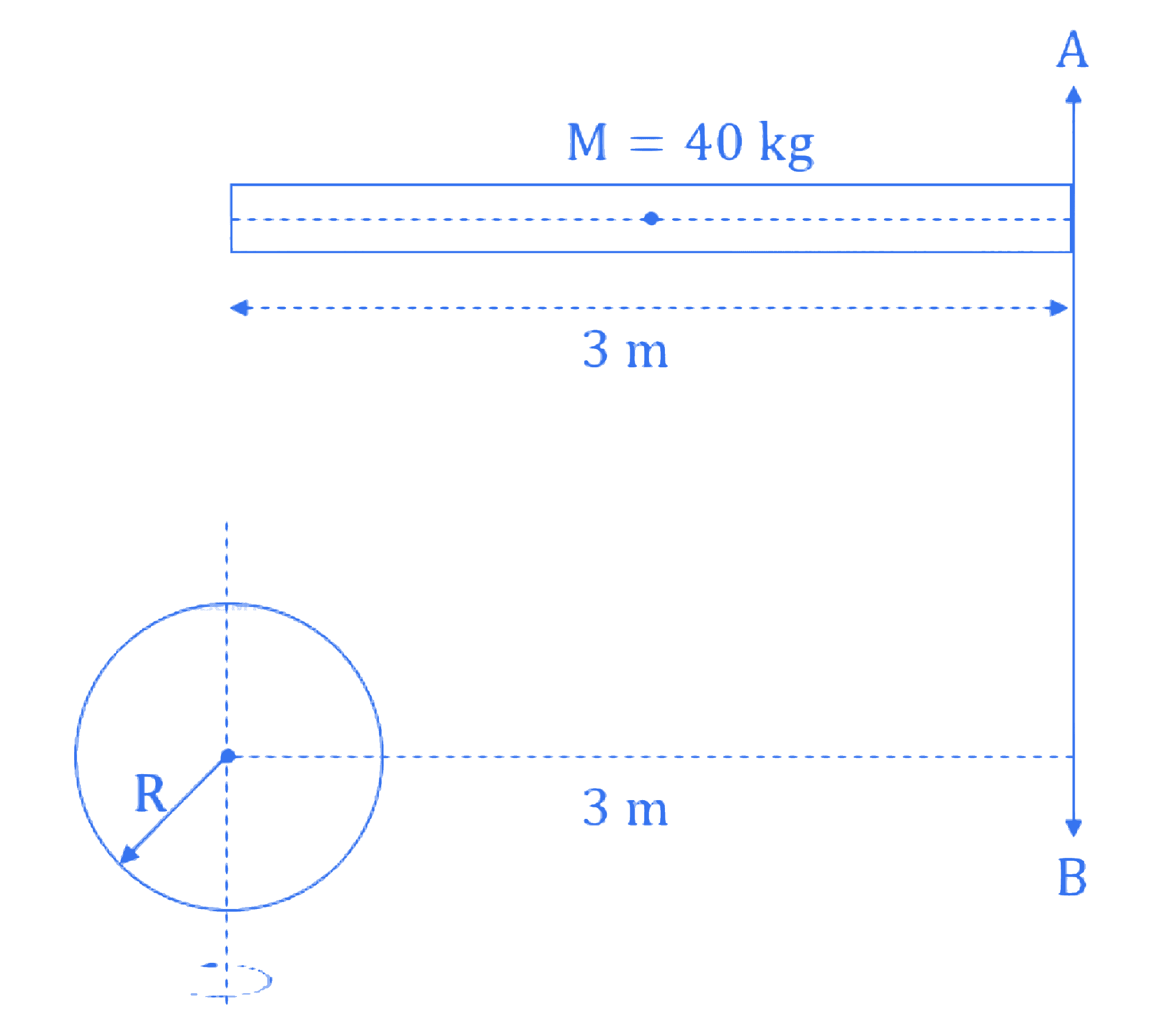
Moment of inertia about an axis *AB* for a rod of mass 40 kg and length 3 m is same as that of a solid sphere of mass of 10 kg and radius $R$ about an axis parallel to *AB* axis with separation of 3 m as shown in figure below. The value of $R$ is given as $\sqrt{\frac{\alpha}{2}}$. The value of $\alpha$ is ________.

Q6.
mcq single
+4 / 1
Two blocks of masses 2 kg and 1 kg respectively, are tied to the ends of a string which passes over a light frictionless pulley as shown in the figure below. The masses are held at rest at the same horizontal level and then released. The distance traversed by the centre of mass in 2 s is _______ m. (Take $g = 10 \; m/s^2$)

Q7.
mcq single
+4 / 1
A circular current loop of radius $R$ is placed inside square loop of side length $L$ ($L \gg R$) such that they are co-planar and their centers coincide. The permeability of free space is $\mu_0$. The mutual inductance between circular loop and square loop is ______.
Q8.
mcq single
+4 / 1
A 0.5 kg mass is in contact against the inner wall of a cylindrical drum of radius 4 m rotating about its vertical axis. The minimum rotational speed of the drum to enable the mass to remain stuck to the wall (without falling) is 5 rad/s. The coefficient of friction between the drum’s inner wall surface and mass is _________. (Take $g = 10\ \mathrm{m/s^2}$)
Q9.
mcq single
+4 / 1
A mixture of carbon dioxide and oxygen has volume 8310 cm^(3), temperature 300 K, pressure 100 kPa and mass 13.2 g. The number of moles of carbon dioxide and oxygen gases in the mixture respectively are ______.
(Assume both carbon dioxide and oxygen gases behave like ideal gases) [R = 8.31 J/mol K]
Q10.
numerical
+4 / 1
5 moles of unknown gas is heated at constant volume from 10°C to 20°C. The molar specific heat of this gas at constant pressure $c_p = 8$ cal/mol.°C and $R = 8.36$ J/mol.°C. The change in the internal energy of the gas is __________ calorie.
Q11.
mcq single
+4 / 1
The surface tension of a soap bubble is 0.03 N/m. The work done in increasing the diameter of bubble from 2 cm to 6 cm is $\alpha \pi \times 10^{-4}$ J. The value of $\alpha$ is _________. (Take $\pi = 3.14$)
Q12.
mcq single
+4 / 1
If an air bubble of diameter 2 mm rises steadily through a liquid of density 2000 kg/m^(3) at a rate of 0.5 cm/s, then the coefficient of viscosity of liquid is ______ Poise. (Take g = 10 m/s^(2))
Q13.
mcq single
+4 / 1
Two point charges $8\ \mu C$ and $-2\ \mu C$ are located at $x = 2$ cm and $x = 4$ cm, respectively on the $x$-axis. The ratio of electric flux due to these charges through two spheres of radii 3 cm and 5 cm with their centers at the origin is ________.
Q14.
mcq single
+4 / 1
Two metal plates **(A, B)** are kept horizontally with separation of **$ \left( \frac{12}{\pi} \right) $ cm**, with plate A on the top.
An atomizer jet sprays oil (density **1.5 g/cm^(3)**) droplets of radius **1 mm** horizontally. All oil droplets carry a charge **5 nC**. The potentials **V~A~** and **V~B~** are required on plates A and B respectively in order to ensure the droplets do not descend. The values of **V~A~** and **V~B~** are ______.
(Neglect the air resistance to the droplets and take **g = 10 m/s^(2)**)
Q15.
mcq single
+4 / 1
If a body of mass 1 kg falls on the earth from infinity, it attains velocity (*v*) and kinetic energy (*k*) on reaching the surface of earth. The values of *v* and *k* respectively are __________.
(Take radius of earth to be 6400 km and *g* = 9.8 m/s^(2))
Q16.
mcq single
+4 / 1
One side of an equilateral prism is painted by a transparent material of refractive index $n_2$. The refractive index of prism is $1.6$. The minimum value of $n_2$ required for total internal reflection from painted face is ________.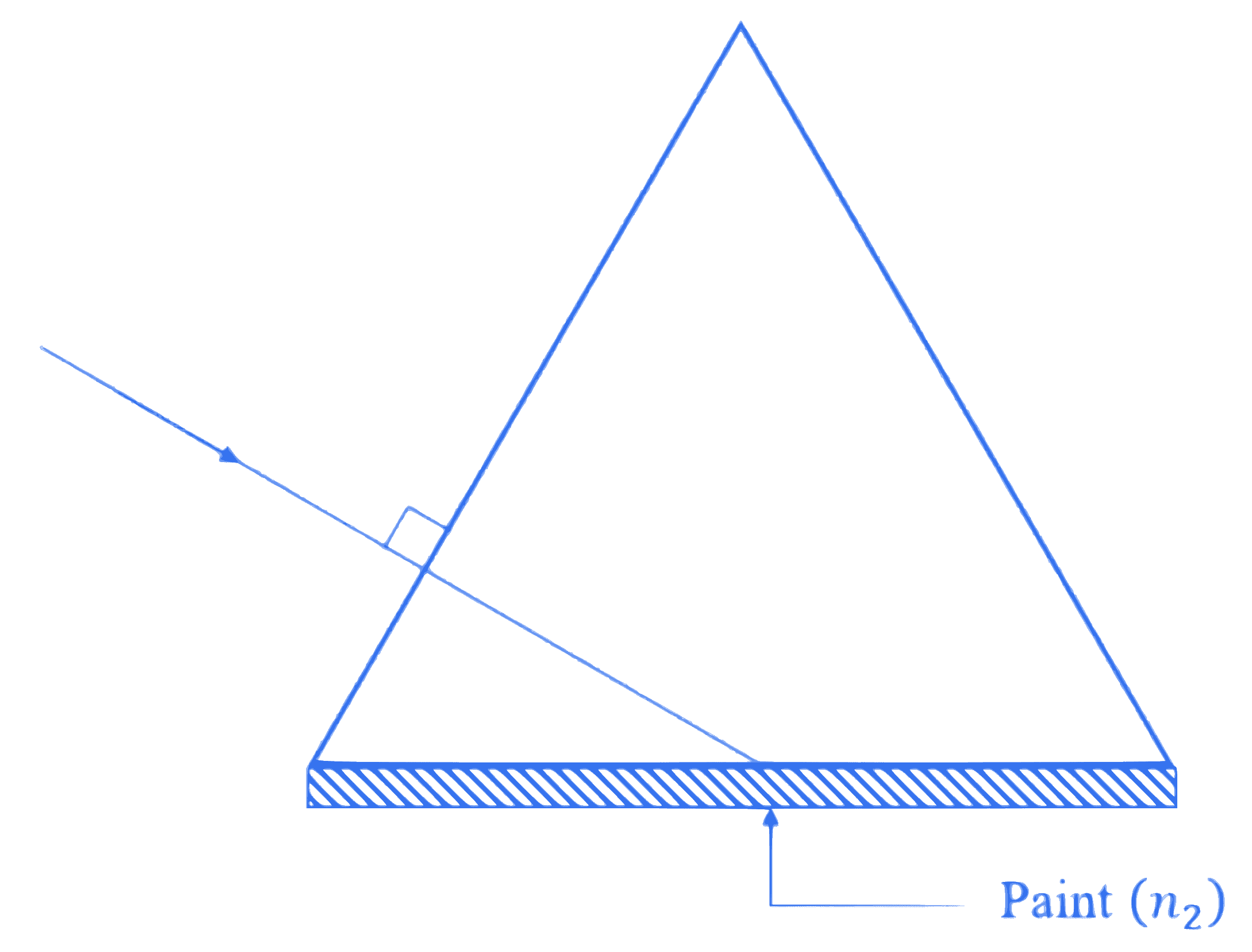

Q17.
numerical
+4 / 1
If sunlight is focused on a paper using convex lens, it starts burning the paper in shortest time when the lens is kept at 30 cm above the paper. If the radius of curvature of the lens is 60 cm then the refractive index of the lens material is $\frac{\alpha}{10}$. The value of $\alpha$ is ________.
Q18.
mcq single
+4 / 1
A particle having charge $10^{-9}$ C moving in $x$-$y$ plane in fields of $0.4 \hat{j}$ N/C and $4 \times 10^{-3} \hat{k}$ T experiences a force of $(4 \hat{i} + 2 \hat{j}) \times 10^{-10}$ N. The velocity of the particle at that instant is _________ m/s.
Q19.
mcq single
+4 / 1
Dimensions of universal gravitational constant ($G$) in terms of Planck's constant ($h$), distance ($L$), mass ($M$) and time ($T$) are _______.
Q20.
mcq single
+4 / 1
In a screw gauge the zero of main scale reference line coincides with the fifth division of the circular scale when two studs are in contact. There are 100 divisions in circular scale and pitch of screw gauge is 0.1 mm. When diameter of a sphere is measured, the reading of main scale is 5 mm and 50^(th) division of circular scale coincides with the reference line of main scale. The diameter of sphere is ______ mm.
Q21.
mcq single
+4 / 1
The binding energy per nucleon of $^{209}_{83}Bi$ is _______ MeV.
[Take $m(^{209}_{83}Bi) = 208.980388\ \text{u}$, $m_p = 1.007825\ \text{u}$, $m_n = 1.008665\ \text{u}$, $1\ \text{u} = 931\ \text{MeV}/c^2$]
Q22.
numerical
+4 / 1
Using Bohr’s model, calculate the ratio of the magnetic fields generated due to the motion of the electrons in the 2^(nd) and 4^(th) orbits of hydrogen atom ________.
Q23.
mcq single
+4 / 1
Two resistors of 200 $\Omega$ and 400 $\Omega$ are connected in series with a battery of 100 V. A bulb rated at 200 V, 100 W is connected across the 400 $\Omega$ resistance. The potential drop across the bulb is ________ V.
Q24.
mcq single
+4 / 1
An electromagnetic wave travels in free space along the *x*-direction. At a particular point in space and time, $\vec{B} = 2 \times 10^{-7} \hat{j}$ T is associated with this wave. The value of corresponding electric field $\vec{E}$ at this point is _______ V/m.
Q25.
mcq single
+4 / 1
The figure given below shows an *LCR* series circuit with two switches S~1~ and S~2~. When switch S~1~ is closed keeping S~2~ open, the phase difference (φ) between the current and source voltage is 30° and phase difference is 60° when S~2~ is closed keeping S~1~ open. The value of (3L~1~ − L~2~) is ______ H.

Q26.
mcq single
+4 / 1
A spherical ball of mass 2 kg falls from a height of 10 m and is brought to rest after penetrating 10 cm into sand.
The average force exerted by sand on the ball is _________ N.
(Take $g = 10 \ \mathrm{m/s^2}$)
Q27.
mcq single
+4 / 1
If X and Y are the inputs, the given circuit works as _________.

Q28.
numerical
+4 / 1
In a Young's double slit experiment, the intensity at some point on the screen is found to be $\frac{3}{4}$ times of the maximum of the interference pattern. The path difference between the interfering waves at this point is $\frac{\lambda}{x}$ where $\lambda$ is wavelength of the incident light. The value of $x$ is _______.
Q29.
mcq single
+4 / 1
The equation of motion of a particle is given by $x = a \sin(50t + \pi/3)$ cm. The particle will come to rest at time $t_1$ and it will have zero acceleration at time $t_2$. The $t_1$ and $t_2$ respectively are ________.
Q30.
numerical
+4 / 1
Moment of inertia about an axis *AB* for a rod of mass 40 kg and length 3 m is same as that of a solid sphere of mass of 10 kg and radius $R$ about an axis parallel to *AB* axis with separation of 3 m as shown in figure below. The value of $R$ is given as $\sqrt{\frac{\alpha}{2}}$. The value of $\alpha$ is ________.


Q31.
mcq single
+4 / 1
Two blocks of masses 2 kg and 1 kg respectively, are tied to the ends of a string which passes over a light frictionless pulley as shown in the figure below. The masses are held at rest at the same horizontal level and then released. The distance traversed by the centre of mass in 2 s is _______ m. (Take $g = 10 \; m/s^2$)

Q32.
mcq single
+4 / 1
A circular current loop of radius $R$ is placed inside square loop of side length $L$ ($L \gg R$) such that they are co-planar and their centers coincide. The permeability of free space is $\mu_0$. The mutual inductance between circular loop and square loop is ______.
Q33.
mcq single
+4 / 1
A 0.5 kg mass is in contact against the inner wall of a cylindrical drum of radius 4 m rotating about its vertical axis. The minimum rotational speed of the drum to enable the mass to remain stuck to the wall (without falling) is 5 rad/s. The coefficient of friction between the drum’s inner wall surface and mass is _________. (Take $g = 10\ \mathrm{m/s^2}$)
Q34.
mcq single
+4 / 1
A mixture of carbon dioxide and oxygen has volume 8310 cm^(3), temperature 300 K, pressure 100 kPa and mass 13.2 g. The number of moles of carbon dioxide and oxygen gases in the mixture respectively are ______.
(Assume both carbon dioxide and oxygen gases behave like ideal gases) [R = 8.31 J/mol K]
Q35.
numerical
+4 / 1
5 moles of unknown gas is heated at constant volume from 10°C to 20°C. The molar specific heat of this gas at constant pressure $c_p = 8$ cal/mol.°C and $R = 8.36$ J/mol.°C. The change in the internal energy of the gas is __________ calorie.
Q36.
mcq single
+4 / 1
The surface tension of a soap bubble is 0.03 N/m. The work done in increasing the diameter of bubble from 2 cm to 6 cm is $\alpha \pi \times 10^{-4}$ J. The value of $\alpha$ is _________. (Take $\pi = 3.14$)
Q37.
mcq single
+4 / 1
If an air bubble of diameter 2 mm rises steadily through a liquid of density 2000 kg/m^(3) at a rate of 0.5 cm/s, then the coefficient of viscosity of liquid is ______ Poise. (Take g = 10 m/s^(2))
Q38.
mcq single
+4 / 1
Two point charges $8\ \mu C$ and $-2\ \mu C$ are located at $x = 2$ cm and $x = 4$ cm, respectively on the $x$-axis. The ratio of electric flux due to these charges through two spheres of radii 3 cm and 5 cm with their centers at the origin is ________.
Q39.
mcq single
+4 / 1
Two metal plates **(A, B)** are kept horizontally with separation of **$ \left( \frac{12}{\pi} \right) $ cm**, with plate A on the top.
An atomizer jet sprays oil (density **1.5 g/cm^(3)**) droplets of radius **1 mm** horizontally. All oil droplets carry a charge **5 nC**. The potentials **V~A~** and **V~B~** are required on plates A and B respectively in order to ensure the droplets do not descend. The values of **V~A~** and **V~B~** are ______.
(Neglect the air resistance to the droplets and take **g = 10 m/s^(2)**)
Q40.
mcq single
+4 / 1
If a body of mass 1 kg falls on the earth from infinity, it attains velocity (*v*) and kinetic energy (*k*) on reaching the surface of earth. The values of *v* and *k* respectively are __________.
(Take radius of earth to be 6400 km and *g* = 9.8 m/s^(2))
Q41.
mcq single
+4 / 1
One side of an equilateral prism is painted by a transparent material of refractive index $n_2$. The refractive index of prism is $1.6$. The minimum value of $n_2$ required for total internal reflection from painted face is ________.

Q42.
numerical
+4 / 1
If sunlight is focused on a paper using convex lens, it starts burning the paper in shortest time when the lens is kept at 30 cm above the paper. If the radius of curvature of the lens is 60 cm then the refractive index of the lens material is $\frac{\alpha}{10}$. The value of $\alpha$ is ________.
Q43.
mcq single
+4 / 1
A particle having charge $10^{-9}$ C moving in $x$-$y$ plane in fields of $0.4 \hat{j}$ N/C and $4 \times 10^{-3} \hat{k}$ T experiences a force of $(4 \hat{i} + 2 \hat{j}) \times 10^{-10}$ N. The velocity of the particle at that instant is _________ m/s.
Q44.
mcq single
+4 / 1
Dimensions of universal gravitational constant ($G$) in terms of Planck's constant ($h$), distance ($L$), mass ($M$) and time ($T$) are _______.
Q45.
mcq single
+4 / 1
In a screw gauge the zero of main scale reference line coincides with the fifth division of the circular scale when two studs are in contact. There are 100 divisions in circular scale and pitch of screw gauge is 0.1 mm. When diameter of a sphere is measured, the reading of main scale is 5 mm and 50^(th) division of circular scale coincides with the reference line of main scale. The diameter of sphere is ______ mm.
Q46.
mcq single
+4 / 1
The binding energy per nucleon of $^{209}_{83}Bi$ is _______ MeV.
[Take $m(^{209}_{83}Bi) = 208.980388\ \text{u}$, $m_p = 1.007825\ \text{u}$, $m_n = 1.008665\ \text{u}$, $1\ \text{u} = 931\ \text{MeV}/c^2$]
Q47.
numerical
+4 / 1
Using Bohr’s model, calculate the ratio of the magnetic fields generated due to the motion of the electrons in the 2^(nd) and 4^(th) orbits of hydrogen atom ________.
Q48.
mcq single
+4 / 1
Two resistors of 200 $\Omega$ and 400 $\Omega$ are connected in series with a battery of 100 V. A bulb rated at 200 V, 100 W is connected across the 400 $\Omega$ resistance. The potential drop across the bulb is ________ V.
Q49.
mcq single
+4 / 1
An electromagnetic wave travels in free space along the *x*-direction. At a particular point in space and time, $\vec{B} = 2 \times 10^{-7} \hat{j}$ T is associated with this wave. The value of corresponding electric field $\vec{E}$ at this point is _______ V/m.
Q50.
mcq single
+4 / 1
The figure given below shows an *LCR* series circuit with two switches S~1~ and S~2~. When switch S~1~ is closed keeping S~2~ open, the phase difference (φ) between the current and source voltage is 30° and phase difference is 60° when S~2~ is closed keeping S~1~ open. The value of (3L~1~ − L~2~) is ______ H.
