JEE Main 2026 (Online) 2nd April Morning Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Consider the three aromatic molecules (P, Q and R) whose structures have been given below :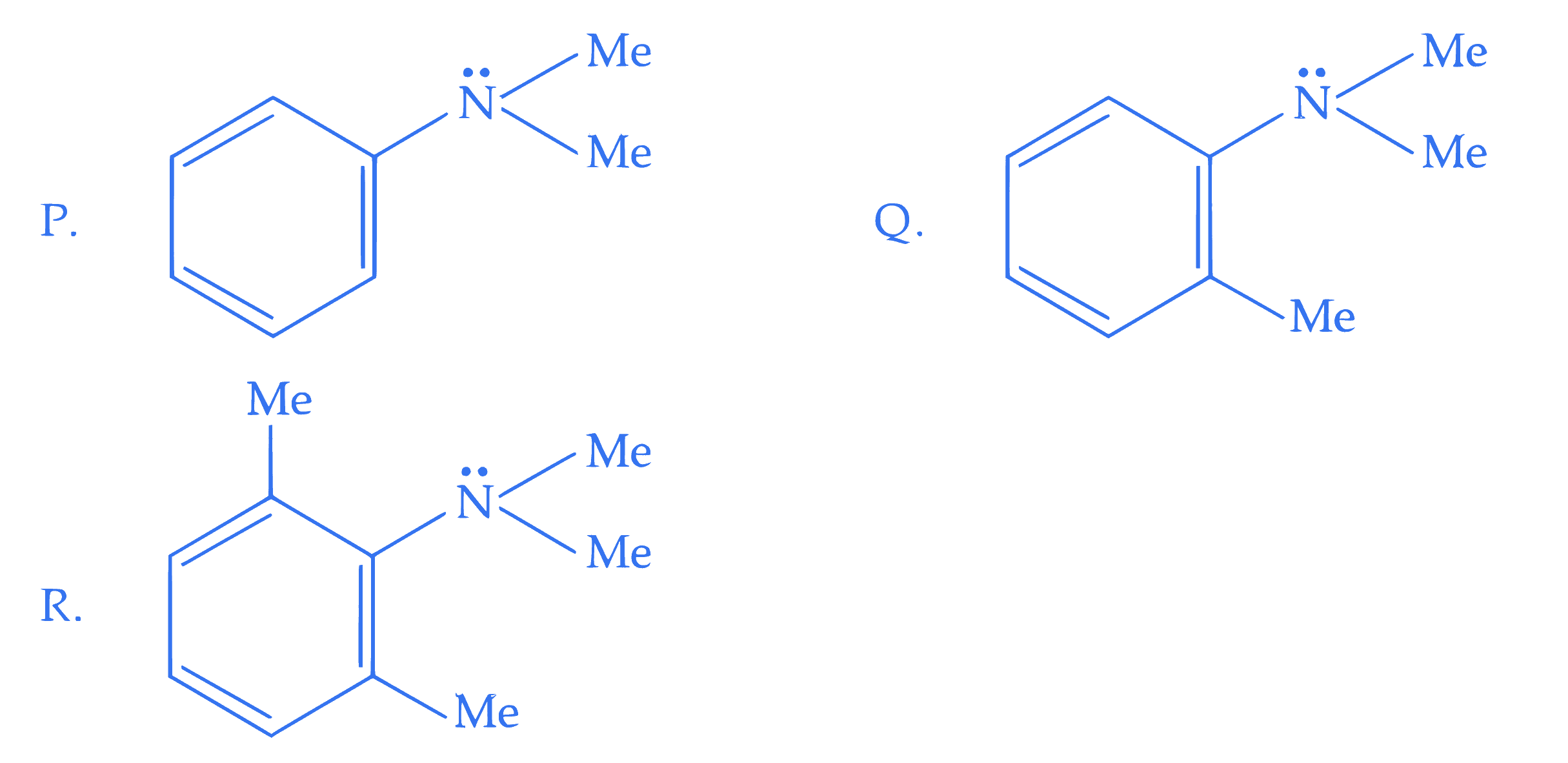 The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
 The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
Q2.
mcq single
+4 / 1
A salt with few drops of conc. HCl gives apple green colour in flame test. The group precipitate of the salt is dissolved in acetic acid and treated with K$_2$CrO$_4$ to give yellow precipitate. When the sodium carbonate extract of the salt solution is heated with conc. HNO$_3$ and ammonium molybdate, it resulted a canary yellow precipitate. The cation and anion present in the salt are respectively,
Q3.
mcq single
+4 / 1
Which of the following sets includes all the species that will change the orange colour of $\mathrm{K_2Cr_2O_7}$ in acidic medium?
Q4.
mcq single
+4 / 1
**t~100%~** is the time required for the 100% completion of the reaction while **t~1/2~** is the time required for 50% of the reaction to be completed. Which of the following option correctly represents the relation between t~100%~ and t~1/2~ for zero and first order reactions respectively?
Q5.
numerical
+4 / 1
For reaction A → P, rate constant $k = 1.5 \times 10^3 \ \mathrm{s}^{-1}$ at $27^{\circ}\mathrm{C}$
If activation energy for the above reaction is $60\ \mathrm{kJ}\ \mathrm{mol}^{-1}$, then the temperature (in $^{\circ}\mathrm{C}$) at which rate constant, $k = 4.5 \times 10^3\ \mathrm{s}^{-1}$ is ______. (Nearest integer)
Given : $\log 2 = 0.30$, $\log 3 = 0.48$, $R = 8.3\ \mathrm{J}\ \mathrm{K}^{-1}\ \mathrm{mol}^{-1}$, $\ln 10 = 2.3$
Q6.
mcq single
+4 / 1
Given below are two statements :
**Statement (I):** The correct sequence of bond lengths in the following species is :
$\ce{O2^{+}} \ce{O2^{+}} > \ce{O2^{-}} > \ce{O2^{2-}}$
In the light of the above statements, choose the **correct answer** from the options given below :
Q7.
mcq single
+4 / 1
Given below are two statements:
**Statement (I):** Oxidising power of halogens decreases in the order $F_2 > Cl_2 > Br_2 > I_2$, which is the basis of "Layer test".
**Statement (II):** "Layer test" to identify $Br_2$ and $I_2$ in aqueous solution involves the oxidation of bromide or iodide into $Br_2$ or $I_2$ respectively with $Cl_2$, which is a type of displacement redox reaction.
In the light of the above statements, choose the **correct answer** from the options given below:
Q8.
mcq single
+4 / 1
Given below are two statements :
**Statement (I) :** Benzyl chloride reacts faster in $S_N1$ mechanism than ethyl chloride.
**Statement (II) :** Ethyl carbocation intermediate is less stabilized by hyperconjugation than benzyl carbocation by resonance.
In the light of the above statements, choose the **correct answer** from the options given below :
Q9.
mcq single
+4 / 1
19.5 g of fluoro acetic acid (molar mass = 78 g mol^(−1)) is dissolved in 500 g of water at 298 K. The depression in the freezing point of water was 1^(∘)C. What is $K_a$ of fluoro acetic acid?
(For water, $K_f = 1.86$ K kg mol^(−1)). Assume molarity and molality to have same values.
Q10.
mcq single
+4 / 1
The mass of iron converted into Fe~3~O~4~ by the action of 18 g of steam is:
(Given: Molar mass of H, O and Fe are 1, 16 and 56 g mol^(−1) respectively)
Assume iron is present in excess:
Q11.
mcq single
+4 / 1
The solubility product constants of $\mathrm{Ag_2CrO_4}$ and $\mathrm{AgBr}$ are $32x$ and $4y$ respectively at 298 K.
The value of $$\left( \frac{\text{molarity of } \mathrm{Ag_2CrO_4}}{\text{molarity of } \mathrm{AgBr}} \right)$$ can be expressed as :
Q12.
mcq single
+4 / 1
Given below are two statements :
**Statement (I) :** The first ionisation enthalpy of the elements Na, Mg, Cl and Ar follows the order Na > Mg > Cl > Ar.
**Statement (II) :** Among Ca, Al, Fe and B, the third ionisation enthalpy is very high for Ca.
In the light of the above statements, choose the **correct answer** from the options given below :
Q13.
numerical
+4 / 1
At the transition temperature $T$, $A \rightleftharpoons B$ and $\Delta G^0 = 105 - 35 \log T$ where A and B are two states of substance X. The transition temperature in $^{\circ}\mathrm{C}$ when pressure is 1 atm is ________. (Nearest integer)
Q14.
mcq single
+4 / 1
Consider the following data.
(i) $2\text{Al(s)} + 6\text{HCl(aq)} \rightarrow \text{Al}_2\text{Cl}_6\text{(aq)} + 3\text{H}_2\text{(g)} + 1200~\text{kJ/mol}$
(ii) $\text{H}_2\text{(g)} + \text{Cl}_2\text{(g)} \rightarrow 2\text{HCl(g)} + 164~\text{kJ/mol}$
(iii) $\text{HCl(g)} + \text{aq} \rightarrow \text{HCl(aq)} + 83~\text{kJ/mol}$
(iv) $\text{Al}_2\text{Cl}_6\text{(s)} + \text{aq} \rightarrow \text{Al}_2\text{Cl}_6\text{(aq)} + 663~\text{kJ/mol}$
The enthalpy of formation of anhydrous solid $\text{Al}_2\text{Cl}_6$ is :
Q15.
mcq single
+4 / 1
**Match List - I with List - II.**
List - I **Chromium (III) Complexes (en = ethylene diamine)
List - II
Δ₀ (cm⁻¹)
A
[Cr(CN)₆]³⁻
I. 15,060
B
[CrF₆]³⁻
II. 17,400
C
[Cr(H₂O)₆]³⁺
III. 22,300
D
[Cr(en)₃]³⁺
IV. 26,600
Choose the correct** answer from the options given below:
Q16.
numerical
+4 / 1
5.33 g of CrCl~3~·6H~2~O, which is a 1 : 3 electrolyte, is dissolved in water and is passed through a cation exchanger. The chloride ions in the eluted solution, on treatment with AgNO~3~ results in 8.61 g of AgCl. The ratio of moles of complex reacted and moles of AgCl formed is ________ × 10^(-2). (Nearest integer)
[Molar mass in g mol^(–1) Cr : 52, Ag : 108, Cl : 35.5, H : 1, O : 16]
Q17.
mcq single
+4 / 1
Given below are two statements :
**Statement (I)** : 1,2,3-Trihydroxypropane can be separated from water by simple distillation.
**Statement (II)** : An azeotropic mixture cannot be separated by fractional distillation.
In the light of the above statements, choose the **correct answer** from the options given below :
Q18.
mcq single
+4 / 1
Match **List - I** with **List - II**.
List - I **Mixture of Compounds
List - II
Reagent used to distinguish
A
Diethyl amine + Ethyl amine
I
Bromine water
B
Acetaldehyde + Acetone
II
CHCl₃ + KOH, Δ
C
Ethanol + Phenol
III
Neutral FeCl₃
D
Benzoic acid + Cinnamic acid
IV
Ammoniacal silver nitrate
Choose the correct** answer from the options given below :
Q19.
numerical
+4 / 1
One mole of an alkane (x) requires 8 mole oxygen for complete combustion. Sum of number of carbon and hydrogen atoms in the alkane (x) is ________.
Q20.
numerical
+4 / 1
Consider the isomers of hydrocarbon with molecular formula $C_5H_{10}$. These isomers do not decolourise $KMnO_4$ solution. These isomers are subjected to chlorination with chlorine in presence of light to give monochloro compounds. The total number of monochloro compounds (structural isomers only) formed is ________.
Q21.
mcq single
+4 / 1
**Match List - I with List - II.**
List - I**Vitamin
List - II
Name
A
Vitamin B~1~
I
Pyridoxine
B
Vitamin B~2~
II
Ascorbic acid
C
Vitamin B~6~
III
Thiamine
D
Vitamin C
IV
Riboflavin
Choose the correct** answer from the options given below:
Q22.
mcq single
+4 / 1
In IUPAC nomenclature, the **correct** order of decreasing priority of functional group is :
Q23.
mcq single
+4 / 1
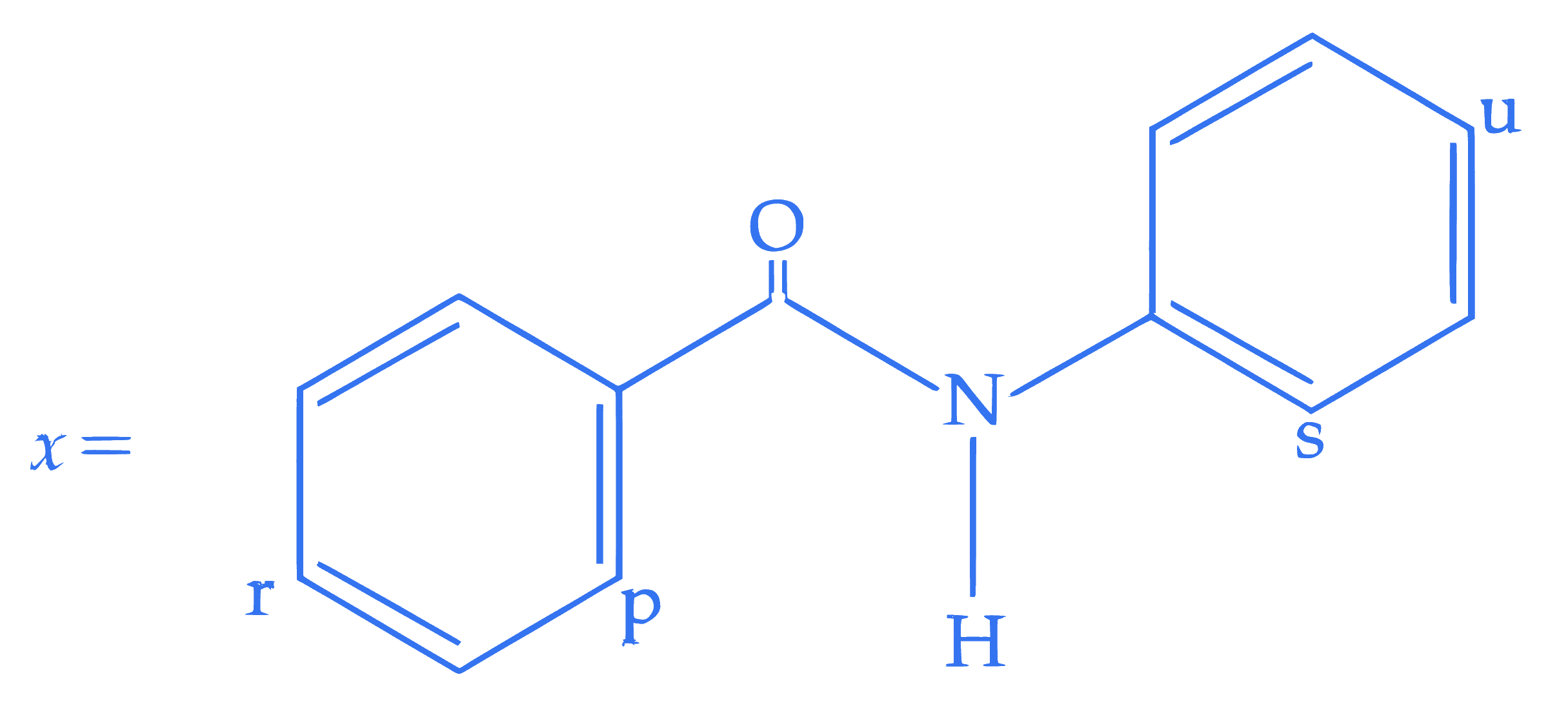
For the given molecule, "x", the preferred site for the attack of the electrophile is :

Q24.
mcq single
+4 / 1
An electrochemical cell is constructed using half cells in the direction of spontaneous change:
Fe(OH)~2~(s) + 2e^(−) → Fe(s) + 2OH^(−)(aq) E^(θ) = −0.88 V
and AgBr(s) + e^(−) → Ag(s) + Br^(−)(aq) E^(θ) = +0.07 V
Which of the following option is **correct**?
Q25.
mcq single
+4 / 1
What is the energy (in J atom^(−1)) required for the following process?
Li^(2+) (g) → Li^(3+) (g) + e^(−)
(Take the ionization energy for the H atom in the ground state as $2.18 \times 10^{−18} \text{ J atom}^{−1}$)
Q26.
mcq single
+4 / 1
Consider the three aromatic molecules (P, Q and R) whose structures have been given below : The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
 The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
The **correct** order regarding the reactivity of these compounds with $$
\mathrm{Ph}-\underset{(+)}{\mathrm{N}} \equiv \mathrm{~N} \mathrm{Cl}^{(-)}
$$ under optimum but slightly acidic medium is :
Q27.
mcq single
+4 / 1
A salt with few drops of conc. HCl gives apple green colour in flame test. The group precipitate of the salt is dissolved in acetic acid and treated with K$_2$CrO$_4$ to give yellow precipitate. When the sodium carbonate extract of the salt solution is heated with conc. HNO$_3$ and ammonium molybdate, it resulted a canary yellow precipitate. The cation and anion present in the salt are respectively,
Q28.
mcq single
+4 / 1
Which of the following sets includes all the species that will change the orange colour of $\mathrm{K_2Cr_2O_7}$ in acidic medium?
Q29.
mcq single
+4 / 1
**t~100%~** is the time required for the 100% completion of the reaction while **t~1/2~** is the time required for 50% of the reaction to be completed. Which of the following option correctly represents the relation between t~100%~ and t~1/2~ for zero and first order reactions respectively?
Q30.
numerical
+4 / 1
For reaction A → P, rate constant $k = 1.5 \times 10^3 \ \mathrm{s}^{-1}$ at $27^{\circ}\mathrm{C}$
If activation energy for the above reaction is $60\ \mathrm{kJ}\ \mathrm{mol}^{-1}$, then the temperature (in $^{\circ}\mathrm{C}$) at which rate constant, $k = 4.5 \times 10^3\ \mathrm{s}^{-1}$ is ______. (Nearest integer)
Given : $\log 2 = 0.30$, $\log 3 = 0.48$, $R = 8.3\ \mathrm{J}\ \mathrm{K}^{-1}\ \mathrm{mol}^{-1}$, $\ln 10 = 2.3$
Q31.
mcq single
+4 / 1
Given below are two statements :
**Statement (I):** The correct sequence of bond lengths in the following species is :
$\ce{O2^{+}} \ce{O2^{+}} > \ce{O2^{-}} > \ce{O2^{2-}}$
In the light of the above statements, choose the **correct answer** from the options given below :
Q32.
mcq single
+4 / 1
Given below are two statements:
**Statement (I):** Oxidising power of halogens decreases in the order $F_2 > Cl_2 > Br_2 > I_2$, which is the basis of "Layer test".
**Statement (II):** "Layer test" to identify $Br_2$ and $I_2$ in aqueous solution involves the oxidation of bromide or iodide into $Br_2$ or $I_2$ respectively with $Cl_2$, which is a type of displacement redox reaction.
In the light of the above statements, choose the **correct answer** from the options given below:
Q33.
mcq single
+4 / 1
Given below are two statements :
**Statement (I) :** Benzyl chloride reacts faster in $S_N1$ mechanism than ethyl chloride.
**Statement (II) :** Ethyl carbocation intermediate is less stabilized by hyperconjugation than benzyl carbocation by resonance.
In the light of the above statements, choose the **correct answer** from the options given below :
Q34.
mcq single
+4 / 1
19.5 g of fluoro acetic acid (molar mass = 78 g mol^(−1)) is dissolved in 500 g of water at 298 K. The depression in the freezing point of water was 1^(∘)C. What is $K_a$ of fluoro acetic acid?
(For water, $K_f = 1.86$ K kg mol^(−1)). Assume molarity and molality to have same values.
Q35.
mcq single
+4 / 1
The mass of iron converted into Fe~3~O~4~ by the action of 18 g of steam is:
(Given: Molar mass of H, O and Fe are 1, 16 and 56 g mol^(−1) respectively)
Assume iron is present in excess:
Q36.
mcq single
+4 / 1
The solubility product constants of $\mathrm{Ag_2CrO_4}$ and $\mathrm{AgBr}$ are $32x$ and $4y$ respectively at 298 K.
The value of $$\left( \frac{\text{molarity of } \mathrm{Ag_2CrO_4}}{\text{molarity of } \mathrm{AgBr}} \right)$$ can be expressed as :
Q37.
mcq single
+4 / 1
Given below are two statements :
**Statement (I) :** The first ionisation enthalpy of the elements Na, Mg, Cl and Ar follows the order Na > Mg > Cl > Ar.
**Statement (II) :** Among Ca, Al, Fe and B, the third ionisation enthalpy is very high for Ca.
In the light of the above statements, choose the **correct answer** from the options given below :
Q38.
numerical
+4 / 1
At the transition temperature $T$, $A \rightleftharpoons B$ and $\Delta G^0 = 105 - 35 \log T$ where A and B are two states of substance X. The transition temperature in $^{\circ}\mathrm{C}$ when pressure is 1 atm is ________. (Nearest integer)
Q39.
mcq single
+4 / 1
Consider the following data.
(i) $2\text{Al(s)} + 6\text{HCl(aq)} \rightarrow \text{Al}_2\text{Cl}_6\text{(aq)} + 3\text{H}_2\text{(g)} + 1200~\text{kJ/mol}$
(ii) $\text{H}_2\text{(g)} + \text{Cl}_2\text{(g)} \rightarrow 2\text{HCl(g)} + 164~\text{kJ/mol}$
(iii) $\text{HCl(g)} + \text{aq} \rightarrow \text{HCl(aq)} + 83~\text{kJ/mol}$
(iv) $\text{Al}_2\text{Cl}_6\text{(s)} + \text{aq} \rightarrow \text{Al}_2\text{Cl}_6\text{(aq)} + 663~\text{kJ/mol}$
The enthalpy of formation of anhydrous solid $\text{Al}_2\text{Cl}_6$ is :
Q40.
mcq single
+4 / 1
**Match List - I with List - II.**
List - I **Chromium (III) Complexes (en = ethylene diamine)
List - II
Δ₀ (cm⁻¹)
A
[Cr(CN)₆]³⁻
I. 15,060
B
[CrF₆]³⁻
II. 17,400
C
[Cr(H₂O)₆]³⁺
III. 22,300
D
[Cr(en)₃]³⁺
IV. 26,600
Choose the correct** answer from the options given below:
Q41.
numerical
+4 / 1
5.33 g of CrCl~3~·6H~2~O, which is a 1 : 3 electrolyte, is dissolved in water and is passed through a cation exchanger. The chloride ions in the eluted solution, on treatment with AgNO~3~ results in 8.61 g of AgCl. The ratio of moles of complex reacted and moles of AgCl formed is ________ × 10^(-2). (Nearest integer)
[Molar mass in g mol^(–1) Cr : 52, Ag : 108, Cl : 35.5, H : 1, O : 16]
Q42.
mcq single
+4 / 1
Given below are two statements :
**Statement (I)** : 1,2,3-Trihydroxypropane can be separated from water by simple distillation.
**Statement (II)** : An azeotropic mixture cannot be separated by fractional distillation.
In the light of the above statements, choose the **correct answer** from the options given below :
Q43.
mcq single
+4 / 1
Match **List - I** with **List - II**.
List - I **Mixture of Compounds
List - II
Reagent used to distinguish
A
Diethyl amine + Ethyl amine
I
Bromine water
B
Acetaldehyde + Acetone
II
CHCl₃ + KOH, Δ
C
Ethanol + Phenol
III
Neutral FeCl₃
D
Benzoic acid + Cinnamic acid
IV
Ammoniacal silver nitrate
Choose the correct** answer from the options given below :
Q44.
numerical
+4 / 1
One mole of an alkane (x) requires 8 mole oxygen for complete combustion. Sum of number of carbon and hydrogen atoms in the alkane (x) is ________.
Q45.
numerical
+4 / 1
Consider the isomers of hydrocarbon with molecular formula $C_5H_{10}$. These isomers do not decolourise $KMnO_4$ solution. These isomers are subjected to chlorination with chlorine in presence of light to give monochloro compounds. The total number of monochloro compounds (structural isomers only) formed is ________.
Q46.
mcq single
+4 / 1
**Match List - I with List - II.**
List - I**Vitamin
List - II
Name
A
Vitamin B~1~
I
Pyridoxine
B
Vitamin B~2~
II
Ascorbic acid
C
Vitamin B~6~
III
Thiamine
D
Vitamin C
IV
Riboflavin
Choose the correct** answer from the options given below:
Q47.
mcq single
+4 / 1
In IUPAC nomenclature, the **correct** order of decreasing priority of functional group is :
Q48.
mcq single
+4 / 1
For the given molecule, "x", the preferred site for the attack of the electrophile is :


Q49.
mcq single
+4 / 1
An electrochemical cell is constructed using half cells in the direction of spontaneous change:
Fe(OH)~2~(s) + 2e^(−) → Fe(s) + 2OH^(−)(aq) E^(θ) = −0.88 V
and AgBr(s) + e^(−) → Ag(s) + Br^(−)(aq) E^(θ) = +0.07 V
Which of the following option is **correct**?
Q50.
mcq single
+4 / 1
What is the energy (in J atom^(−1)) required for the following process?
Li^(2+) (g) → Li^(3+) (g) + e^(−)
(Take the ionization energy for the H atom in the ground state as $2.18 \times 10^{−18} \text{ J atom}^{−1}$)
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
If the mean of the dataClass5-1010-1515-2020-2525-3030-35Frequency2k2854k + 15is 21, then $k$ is one of the roots of the equation:
Q2.
mcq single
+4 / 1
If $\vec{a}$ and $\vec{b}$ are two vectors such that $|\vec{a}|=2$ and $|\vec{b}|=3$, then the maximum value of $3|(3 \vec{a}+2 \vec{b})|+4|(3 \vec{a}-2 \vec{b})|$ is :
Q3.
mcq single
+4 / 1
Let $\alpha, \alpha+2, \alpha \in \mathbf{Z}$, be the roots of the quadratic equation $x(x+2)+(x+1)(x+3)+(x+2)(x+4)+\ldots .+(x+\mathrm{n}-1)(x+\mathrm{n}+1)=4 \mathrm{n}$ for some $\mathrm{n} \in \mathbf{N}$. Then $\mathrm{n}+\alpha$ is equal to :
Q4.
numerical
+4 / 1
Let **a, b, c** ∈ {1, 2, 3, 4}. If the probability, that $a x^2 + 2\sqrt{2} bx + c > 0$ for all $x \in \mathbb{R}$, is $\frac{m}{n}$, $\gcd(m, n) = 1$, then $m + n$ is equal to ________.
Q5.
mcq single
+4 / 1
Let a line L passing through the point (1, 1, 1) be perpendicular to both the vectors $2\hat{i} + 2\hat{j} + \hat{k}$ and $\hat{i} + 2\hat{j} + 2\hat{k}$. If $P(a, b, c)$ is the foot of perpendicular from the origin on the line L, then the value of $34(a + b + c)$ is :
Q6.
mcq single
+4 / 1
If the point of intersection of the lines $ \frac{x+1}{3} = \frac{y+a}{5} = \frac{z+b+1}{7} $ and $ \frac{x-2}{1} = \frac{y-b}{4} = \frac{z-2a}{7} $ lies on xy-plane, then the value of $a+b$ is:
Q7.
numerical
+4 / 1
If $\sum\limits_{k=1}^{n} a_k = 6 n^3$, then $\sum\limits_{k=1}^{6} \left( \frac{a_{k+1} - a_k}{36} \right)^2$ is equal to ________.
Q8.
mcq single
+4 / 1
Let A be the set of first 101 terms of an A.P., whose first term is 1 and the common difference is 5 and let B be the set of first 71 terms of an A.P., whose first term is 9 and the common difference is 7. Then the number of elements in $A \cap B$, which are divisible by 3, is:
Q9.
mcq single
+4 / 1
If $ \lim\limits_{x\to 2} \frac{\sin\left(x^3 - 5x^2 + ax + b\right)}{\left(\sqrt{x-1}-1\right) \log_e(x-1)} = m $, then $a + b + m$ is equal to:
Q10.
mcq single
+4 / 1
The number of critical points of the function $f(x) = \begin{cases} |\frac{\sin x}{x}|, & x \neq 0 \\ 1, & x = 0 \end{cases}$ in the interval $(-2\pi, 2\pi)$ is equal to:
Q11.
mcq single
+4 / 1
If $\sin\left(\frac{\pi}{18}\right) \sin\left(\frac{5\pi}{18}\right) \sin\left(\frac{7\pi}{18}\right) = K$, then the value of $\sin\left(\frac{10K\pi}{3}\right)$ is:
Q12.
mcq single
+4 / 1
Let $A = \begin{bmatrix} 1 & 2 \\ 1 & \alpha \end{bmatrix}$ and $B = \begin{bmatrix} 3 & 3 \\ \beta & 2 \end{bmatrix}$. If $A^2 - 4A + I = O$ and $B^2 - 5B - 6I = O$, then among the two statements:
(S1): $[(B-A)(B+A)]^T = \begin{bmatrix} 13 & 15 \\ 7 & 10 \end{bmatrix}$
and
(S2): $\det(\mathrm{adj}(A+B)) = -5$
Q13.
mcq single
+4 / 1
Let $\alpha, \beta \in \mathbb{R}$ be such that the system of linear equations
$ \begin{aligned} x + 2y + z &= 5 \\ 2x + y + \alpha z &= 5 \\ 8x + 4y + \beta z &= 18 \end{aligned} $
has no solution. Then $\frac{\beta}{\alpha}$ is equal to :
Q14.
mcq single
+4 / 1
Let $S = \{x \in [-\pi, \pi] : \sin x (\sin x + \cos x) = a,\ a \in \mathbb{Z} \}$. Then $n(S)$ is equal to:
Q15.
mcq single
+4 / 1
Let the mid points of the sides of a triangle ABC be $\left(\frac{5}{2}, 7\right)$, $\left(\frac{5}{2}, 3\right)$ and $(4, 5)$. If its incentre is $(h, k)$, then $3h + k$ is equal to :
Q16.
mcq single
+4 / 1
Let $x$ and $y$ be real numbers such that $50\left(\frac{2 x}{1+3 i}-\frac{y}{1-2 i}\right)=31+17 i, i=\sqrt{-1}$. Then the value of $10(x-3 y)$ is :
Q17.
numerical
+4 / 1
If the domain of the function $f(x) = \sqrt{\log_{(0.6)} (\left| \frac{2x-5}{x^2-4} \right|)}$ is $(-\infty, a] \cup \{b\} \cup [c, d) \cup (e, \infty)$, then the value of $a + b + c + d + e$ is ________.
Q18.
mcq single
+4 / 1
Let $y = y(x)$ be the solution curve of the differential equation
$(1 + \sin x)\dfrac{dy}{dx} + (y + 1)\cos x = 0,\ y(0) = 0.$ If the curve $y = y(x)$ passes through the point $\left( \alpha , \dfrac{-1}{2} \right)$,
then a value of $\alpha$ is:
Q19.
mcq single
+4 / 1
If the curve $y = f(x)$ passes through the point $(1, e)$ and satisfies the differential equation $dy = y(2 + \\log_e x) dx$, $x > 0$, then $f(e)$ is equal to :
Q20.
numerical
+4 / 1
Let a circle C have its centre in the first quadrant, intersect the coordinate axes at exactly three points and cut off equal intercepts from the coordinate axes. If the length of the chord of C on the line $x + y = 1$ is $\sqrt{14}$, then the square of the radius of C is ________.
Q21.
mcq single
+4 / 1
Let an ellipse $\frac{x^2}{a^2} + \frac{y^2}{b^2} = 1$, $a < b$, pass through the point (4, 3) and have eccentricity $\frac{\sqrt{5}}{3}$.
Then the length of its latus rectum is :
Q22.
mcq single
+4 / 1
The number of seven-digit numbers, that can be formed by using the digits 1, 2, 3, 5 and 7 such that each digit is used at least once, is:
Q23.
mcq single
+4 / 1
The number of elements in the set $S = \left\{ (r, k) : k \in \mathbb{Z} \text{ and } ^{36}C_{r+1} = \frac{6\left(^{35}C_{r}\right)}{(k^2-3)} \right\}$ is :
Q24.
numerical
+4 / 1
If $\alpha = \int\limits_{0}^{2\sqrt{3}} \log_{2}(x^{2} + 4) \, dx + \int\limits_{2}^{4} \sqrt{2x - 4} \, dx$, then $\alpha^{2}$ is equal to ________.
Q25.
mcq single
+4 / 1
Let [.] denote the greatest integer function. Then the value of $\int\limits_{0}^{3} \left( \frac{e^{x} + e^{-x}}{[x]!} \right) dx$ is:
Q26.
mcq single
+4 / 1
If the mean of the dataClass5-1010-1515-2020-2525-3030-35Frequency2k2854k + 15is 21, then $k$ is one of the roots of the equation:
Q27.
mcq single
+4 / 1
If $\vec{a}$ and $\vec{b}$ are two vectors such that $|\vec{a}|=2$ and $|\vec{b}|=3$, then the maximum value of $3|(3 \vec{a}+2 \vec{b})|+4|(3 \vec{a}-2 \vec{b})|$ is :
Q28.
mcq single
+4 / 1
Let $\alpha, \alpha+2, \alpha \in \mathbf{Z}$, be the roots of the quadratic equation $x(x+2)+(x+1)(x+3)+(x+2)(x+4)+\ldots .+(x+\mathrm{n}-1)(x+\mathrm{n}+1)=4 \mathrm{n}$ for some $\mathrm{n} \in \mathbf{N}$. Then $\mathrm{n}+\alpha$ is equal to :
Q29.
numerical
+4 / 1
Let **a, b, c** ∈ {1, 2, 3, 4}. If the probability, that $a x^2 + 2\sqrt{2} bx + c > 0$ for all $x \in \mathbb{R}$, is $\frac{m}{n}$, $\gcd(m, n) = 1$, then $m + n$ is equal to ________.
Q30.
mcq single
+4 / 1
Let a line L passing through the point (1, 1, 1) be perpendicular to both the vectors $2\hat{i} + 2\hat{j} + \hat{k}$ and $\hat{i} + 2\hat{j} + 2\hat{k}$. If $P(a, b, c)$ is the foot of perpendicular from the origin on the line L, then the value of $34(a + b + c)$ is :
Q31.
mcq single
+4 / 1
If the point of intersection of the lines $ \frac{x+1}{3} = \frac{y+a}{5} = \frac{z+b+1}{7} $ and $ \frac{x-2}{1} = \frac{y-b}{4} = \frac{z-2a}{7} $ lies on xy-plane, then the value of $a+b$ is:
Q32.
numerical
+4 / 1
If $\sum\limits_{k=1}^{n} a_k = 6 n^3$, then $\sum\limits_{k=1}^{6} \left( \frac{a_{k+1} - a_k}{36} \right)^2$ is equal to ________.
Q33.
mcq single
+4 / 1
Let A be the set of first 101 terms of an A.P., whose first term is 1 and the common difference is 5 and let B be the set of first 71 terms of an A.P., whose first term is 9 and the common difference is 7. Then the number of elements in $A \cap B$, which are divisible by 3, is:
Q34.
mcq single
+4 / 1
If $ \lim\limits_{x\to 2} \frac{\sin\left(x^3 - 5x^2 + ax + b\right)}{\left(\sqrt{x-1}-1\right) \log_e(x-1)} = m $, then $a + b + m$ is equal to:
Q35.
mcq single
+4 / 1
The number of critical points of the function $f(x) = \begin{cases} |\frac{\sin x}{x}|, & x \neq 0 \\ 1, & x = 0 \end{cases}$ in the interval $(-2\pi, 2\pi)$ is equal to:
Q36.
mcq single
+4 / 1
If $\sin\left(\frac{\pi}{18}\right) \sin\left(\frac{5\pi}{18}\right) \sin\left(\frac{7\pi}{18}\right) = K$, then the value of $\sin\left(\frac{10K\pi}{3}\right)$ is:
Q37.
mcq single
+4 / 1
Let $A = \begin{bmatrix} 1 & 2 \\ 1 & \alpha \end{bmatrix}$ and $B = \begin{bmatrix} 3 & 3 \\ \beta & 2 \end{bmatrix}$. If $A^2 - 4A + I = O$ and $B^2 - 5B - 6I = O$, then among the two statements:
(S1): $[(B-A)(B+A)]^T = \begin{bmatrix} 13 & 15 \\ 7 & 10 \end{bmatrix}$
and
(S2): $\det(\mathrm{adj}(A+B)) = -5$
Q38.
mcq single
+4 / 1
Let $\alpha, \beta \in \mathbb{R}$ be such that the system of linear equations
$ \begin{aligned} x + 2y + z &= 5 \\ 2x + y + \alpha z &= 5 \\ 8x + 4y + \beta z &= 18 \end{aligned} $
has no solution. Then $\frac{\beta}{\alpha}$ is equal to :
Q39.
mcq single
+4 / 1
Let $S = \{x \in [-\pi, \pi] : \sin x (\sin x + \cos x) = a,\ a \in \mathbb{Z} \}$. Then $n(S)$ is equal to:
Q40.
mcq single
+4 / 1
Let the mid points of the sides of a triangle ABC be $\left(\frac{5}{2}, 7\right)$, $\left(\frac{5}{2}, 3\right)$ and $(4, 5)$. If its incentre is $(h, k)$, then $3h + k$ is equal to :
Q41.
mcq single
+4 / 1
Let $x$ and $y$ be real numbers such that $50\left(\frac{2 x}{1+3 i}-\frac{y}{1-2 i}\right)=31+17 i, i=\sqrt{-1}$. Then the value of $10(x-3 y)$ is :
Q42.
numerical
+4 / 1
If the domain of the function $f(x) = \sqrt{\log_{(0.6)} (\left| \frac{2x-5}{x^2-4} \right|)}$ is $(-\infty, a] \cup \{b\} \cup [c, d) \cup (e, \infty)$, then the value of $a + b + c + d + e$ is ________.
Q43.
mcq single
+4 / 1
Let $y = y(x)$ be the solution curve of the differential equation
$(1 + \sin x)\dfrac{dy}{dx} + (y + 1)\cos x = 0,\ y(0) = 0.$ If the curve $y = y(x)$ passes through the point $\left( \alpha , \dfrac{-1}{2} \right)$,
then a value of $\alpha$ is:
Q44.
mcq single
+4 / 1
If the curve $y = f(x)$ passes through the point $(1, e)$ and satisfies the differential equation $dy = y(2 + \\log_e x) dx$, $x > 0$, then $f(e)$ is equal to :
Q45.
numerical
+4 / 1
Let a circle C have its centre in the first quadrant, intersect the coordinate axes at exactly three points and cut off equal intercepts from the coordinate axes. If the length of the chord of C on the line $x + y = 1$ is $\sqrt{14}$, then the square of the radius of C is ________.
Q46.
mcq single
+4 / 1
Let an ellipse $\frac{x^2}{a^2} + \frac{y^2}{b^2} = 1$, $a < b$, pass through the point (4, 3) and have eccentricity $\frac{\sqrt{5}}{3}$.
Then the length of its latus rectum is :
Q47.
mcq single
+4 / 1
The number of seven-digit numbers, that can be formed by using the digits 1, 2, 3, 5 and 7 such that each digit is used at least once, is:
Q48.
mcq single
+4 / 1
The number of elements in the set $S = \left\{ (r, k) : k \in \mathbb{Z} \text{ and } ^{36}C_{r+1} = \frac{6\left(^{35}C_{r}\right)}{(k^2-3)} \right\}$ is :
Q49.
numerical
+4 / 1
If $\alpha = \int\limits_{0}^{2\sqrt{3}} \log_{2}(x^{2} + 4) \, dx + \int\limits_{2}^{4} \sqrt{2x - 4} \, dx$, then $\alpha^{2}$ is equal to ________.
Q50.
mcq single
+4 / 1
Let [.] denote the greatest integer function. Then the value of $\int\limits_{0}^{3} \left( \frac{e^{x} + e^{-x}}{[x]!} \right) dx$ is:
Physics
Physics
Q1.
mcq single
+4 / 1
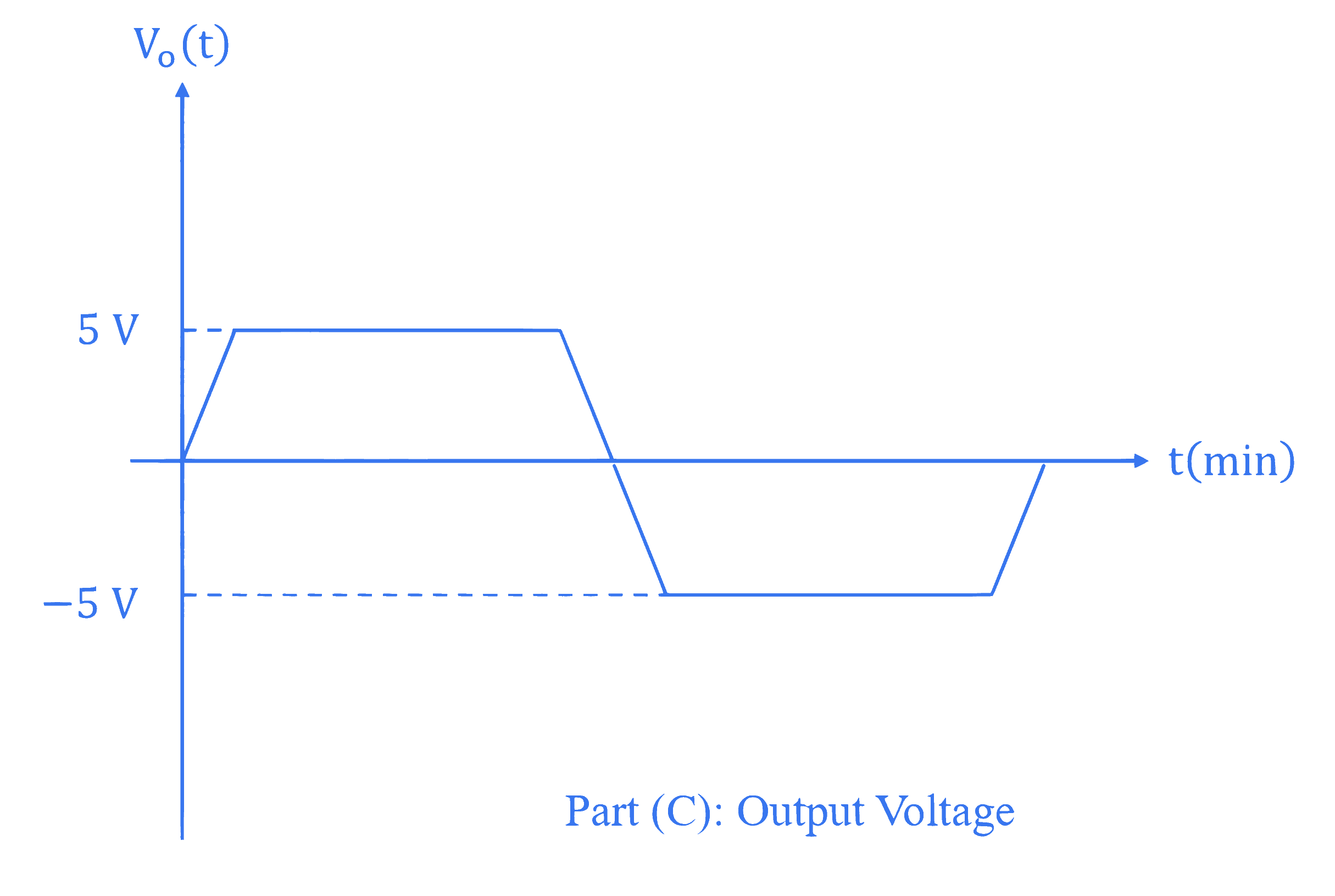
For the given circuit (shown in part (A)) the time dependent input voltage $v_{in}(t)$ and corresponding output $v_{o}(t)$ are shown in part (B) and part (C), respectively. Identify the components that are used in the circuit between points X and Y.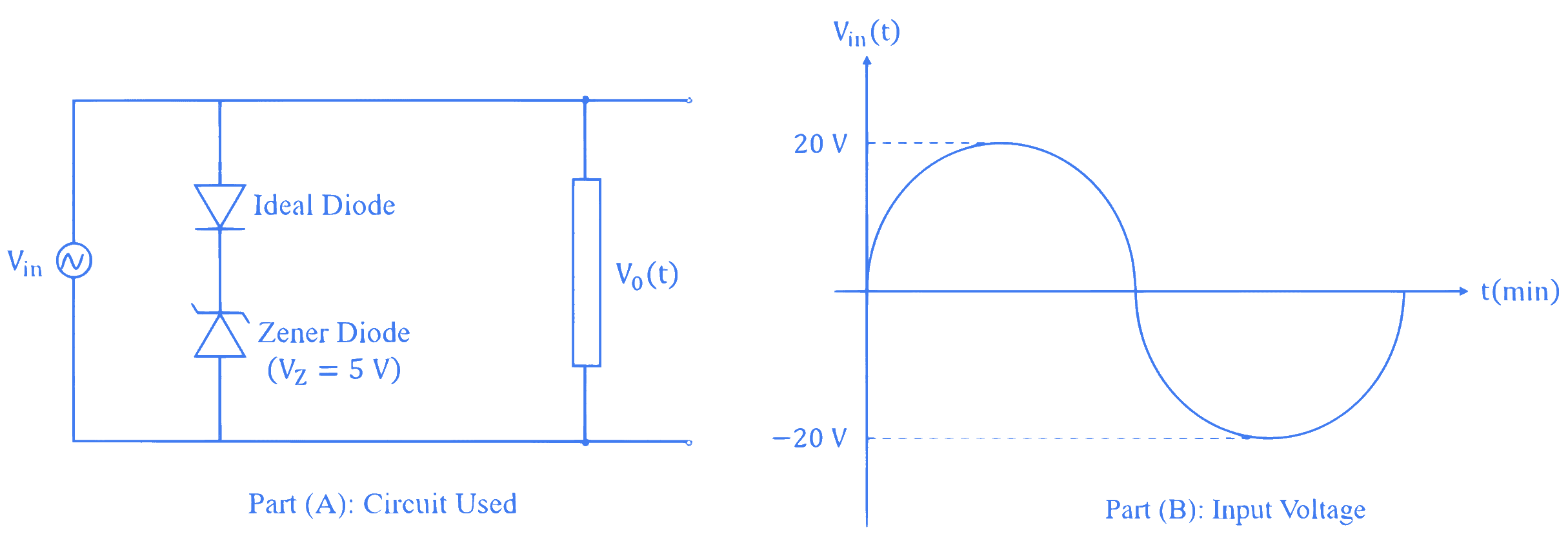


Q2.
numerical
+4 / 1
In single slit diffraction pattern, the wavelength of light used is 628 nm and slit width is 0.2 mm, the angular width of central maximum is $\alpha \times 10^{-2}$ degrees. The value of $\alpha$ is ________.
Q3.
mcq single
+4 / 1
The position of an object having mass 0.1 kg as a function of time $t$ is given as
$\vec{r} = \left( 10 t^2 \hat{i} + 5 t^3 \hat{j} \right)$ m. At $t = 1$ s, which of the following statements are **correct**?
**A.** The linear momentum $\vec{p} = \left( 2 \hat{i} + 1.5 \hat{j} \right)$ kg·m/s.
**B.** The force acting on the object $\vec{F} = \left( 2 \hat{i} + 3 \hat{j} \right)$ N.
**C.** The angular momentum of the object about its origin $\vec{L} = 15 \hat{k}$ J·s.
**D.** The torque acting on the object about its origin $\vec{\tau} = 20 \hat{k}$ N·m.
Choose the **correct** answer from the options given below:
Q4.
mcq single
+4 / 1
When a coil is placed in a time dependent magnetic field the power dissipated in it is $P$. The number of turns, area of the coil and radius of the coil wire are $N$, $A$ and $r$ respectively. For a second coil number of turns, area of the coil and radius of the coil wire are $2N$, $2A$ and $3r$ respectively. When the first coil is replaced with second coil the power dissipated in it is $\sqrt{2} \,\alpha P$. The value of $\alpha$ is ______.
Q5.
mcq single
+4 / 1
A particle is rotating in a circular path and at any instant its motion can be described as
$\theta = \frac{5t^4}{40} - \frac{t^3}{3}$.
The angular acceleration of the particle after 10 seconds is _________ rad/s^(2).
Q6.
mcq single
+4 / 1
Heat is supplied to a diatomic gas at constant pressure. Then the ratio of $\Delta Q : \Delta U : \Delta W$ is ______.
Q7.
numerical
+4 / 1
A vessel contains 0.15 m^(3) of a gas at pressure 8 bar and temperature 140 °C with $c_p = 3R$ and $c_v = 2R$. It is expanded adiabatically till pressure falls to 1 bar. The work done during this process is _________ kJ. (R is gas constant)
Q8.
numerical
+4 / 1
A tub is filled with water and a wooden cube $10\ \text{cm} \times 10\ \text{cm} \times 10\ \text{cm}$ is placed in the water. The wooden cube is found to float on the water with a part of it submerged in water. When a metal coin is placed on the wooden cube, the submerged part is increased by $3.87$ cm. The mass of the metal coin is ________ gram.
(Take water density as $1\ \text{g/cm}^3$ and density of wood as $0.4\ \text{g/cm}^3$)
Q9.
numerical
+4 / 1
A uniform wire of length $l$ of weight $w$ is suspended from the roof with a weight of $W$ at the other end. The stress in the wire at $\frac{l}{3}$ distance from the top is $\left( \frac{W}{A} + \frac{2}{\gamma} \frac{w}{A} \right)$, where $A$ is the cross sectional area of the wire. The value of $\gamma$ is ________.
Q10.
mcq single
+4 / 1
A liquid drop of diameter 2 mm breaks into 512 droplets. The change in surface energy is $\alpha \times 10^{-6}$ J. The value of $\alpha$ is _______. (Take surface tension of liquid = 0.08 N/m)
Q11.
mcq single
+4 / 1
For a certain metal, when monochromatic light of wavelength $\lambda$ is incident, the stopping potential for photoelectrons is $3V_0$. When the same metal is illuminated by light of wavelength $2\lambda$, then the stopping potential becomes $V_0$. The threshold wavelength for photoelectric emission for the given metal is $\alpha \lambda$. The value of $\alpha$ is ______.
Q12.
mcq single
+4 / 1
Two short electric dipoles **A** and **B** having dipole moment $p_1$ and $p_2$ respectively are placed with their axis mutually perpendicular as shown in the figure. The resultant electric field at a point $x$ is making an angle of $60^{\circ}$ with the line joining points $O$ and $x$. The ratio of the dipole moments $p_2/p_1$ is _________.


Q13.
mcq single
+4 / 1
Two charged conducting spheres $S_1$ and $S_2$ of radii 8 cm and 18 cm are connected to each other by a wire. After equilibrium is established, the ratio of electric fields on $S_1$ and $S_2$ spheres are $E_{S1}$ and $E_{S2}$ respectively. The value of $\dfrac{E_{S1}}{E_{S2}}$ is ________.
Q14.
mcq single
+4 / 1
A planet ($P_1$) is moving around the star of mass $2M$ in the orbit of radius $R$. Another planet ($P_2$) is moving around another star of mass $4M$ in a orbit of radius $2R$. Ratio of time periods of revolution of $P_2$ and $P_1$ is ________.
Q15.
mcq single
+4 / 1
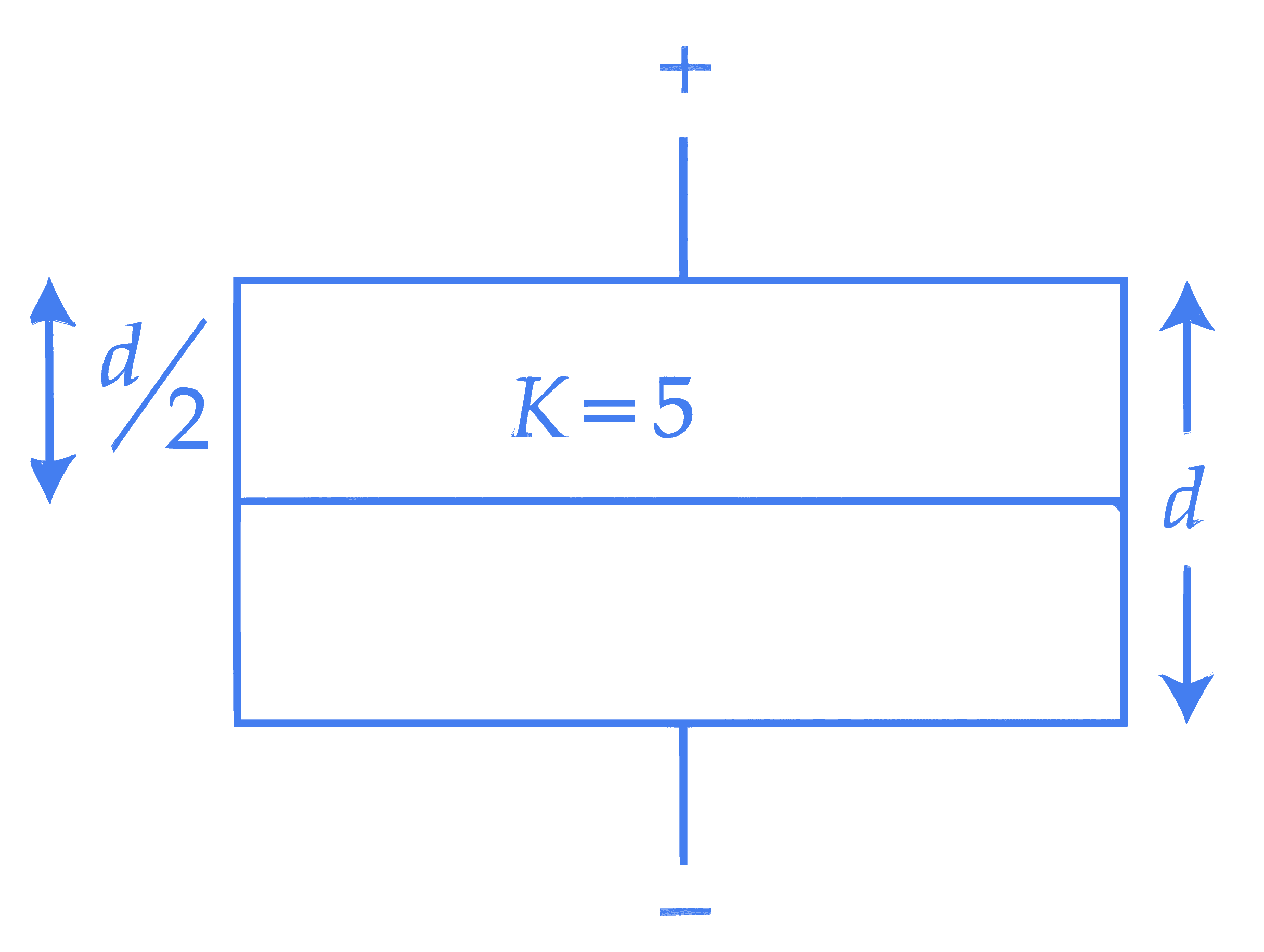
A parallel plate air capacitor has a capacitance $C$. When it is half filled as shown in figure with a dielectric constant $K = 5$, the percentage increase in the capacitance is ________.

Q16.
mcq single
+4 / 1
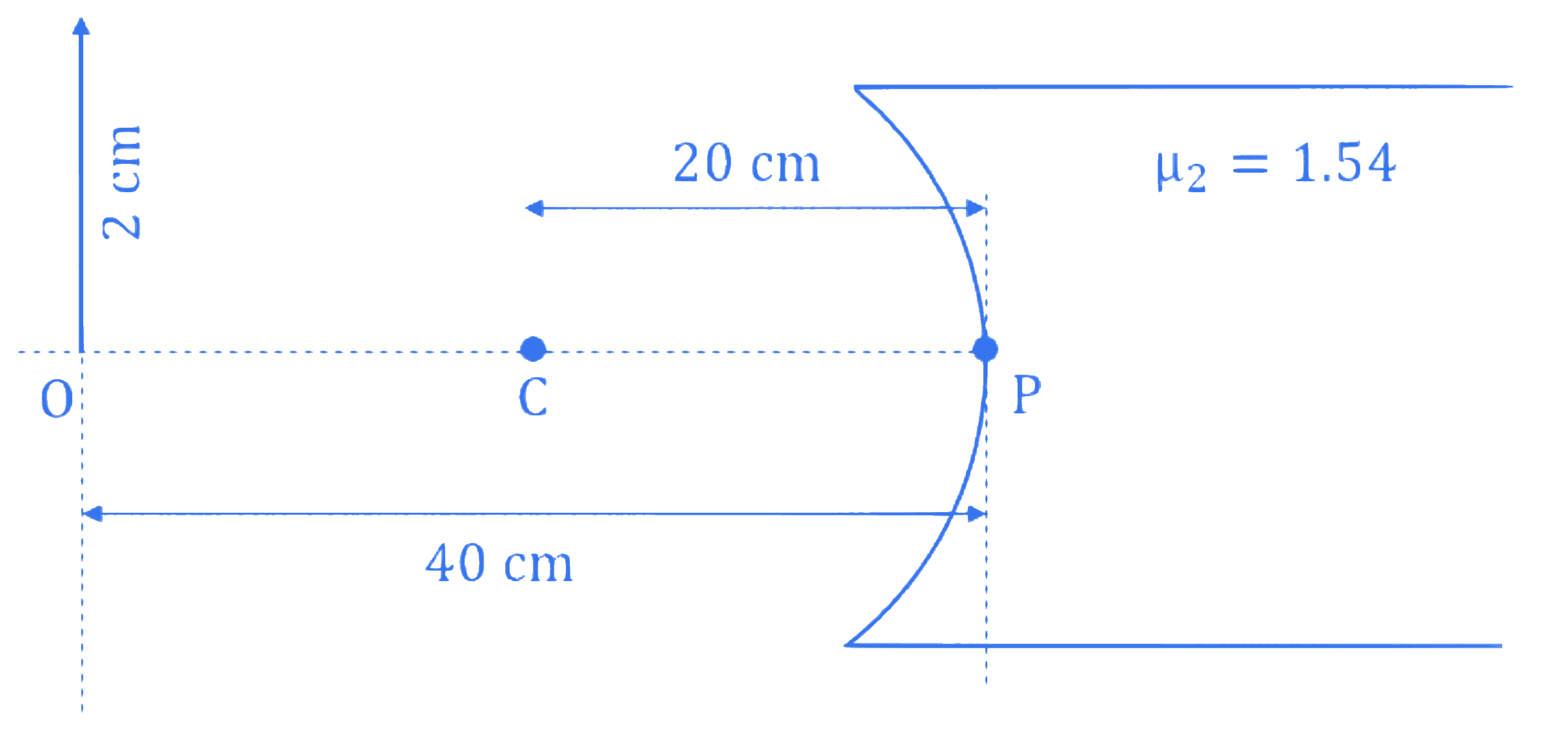
Refer the figure given below. $\mu_1$ and $\mu_2$ are refractive indices of air and lens material. The height of image will be _______ cm.

Q17.
mcq single
+4 / 1
For a thin symmetric prism made of glass (refractive index 1.5), the ratio of incident angle and minimum deviation will be _______.
Q18.
numerical
+4 / 1
1 μC charge moving with velocity $\vec{v} = (\hat{i} - 2\hat{j} + 3\hat{k})$ m/s in the region of magnetic field $\vec{B} = (2\hat{i} + 3\hat{j} - 5\hat{k})$ T. The magnitude of force acting on it is $\sqrt{\alpha} \times 10^{-6}$ N. The value of $\alpha$ is _______.
Q19.
mcq single
+4 / 1
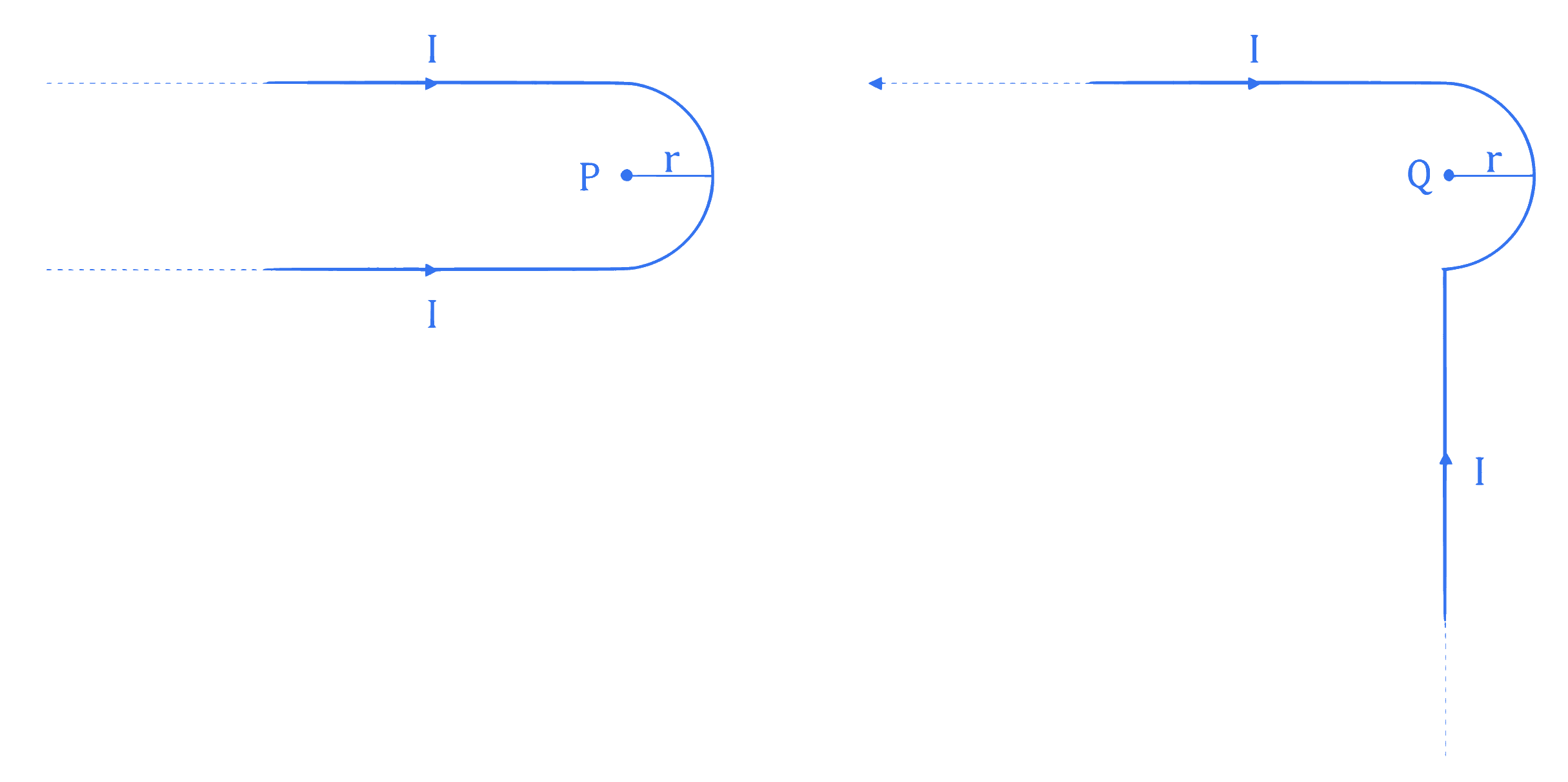
Two identical long current carrying wires are bent into the shapes shown in the following figures. If the magnitude of magnetic fields at the centres P and Q of a semicircular arc are $B_1$ and $B_2$ respectively, then the ratio $\frac{B_1}{B_2}$ is ______.

Q20.
mcq single
+4 / 1
The dimensional formula of $\frac{1}{2} \epsilon_0 E^2$ ($\epsilon_0$ = permittivity of vacuum and $E$ = electric field) is $M^a L^b T^c$.
The value of $2a - b + c =$ ________.
Q21.
mcq single
+4 / 1
The diameter of a wire measured by a screw gauge of least count 0.001 cm is 0.08 cm. The length measured by a scale of least count 0.1 cm is 150 cm. When a weight of 100 N is applied to the wire, the extension in length is 0.5 cm, measured by a micrometer of least count 0.001 cm. The error in the measured Young’s modulus is $\alpha \times 10^9 \ \mathrm{N/m}^2$. The value of $\alpha$ is ________.
(Ignore the contribution of the load to Young’s modulus error calculation)
Q22.
mcq single
+4 / 1
Angular momentum of an electron in a hydrogen atom is $\frac{3h}{\pi}$, then the energy of the electron is _____ eV.
Q23.
mcq single
+4 / 1
The velocity of a particle is given as $\vec{v} = -x \hat{i} + 2y \hat{j} - z \hat{k}$ m/s. The magnitude of acceleration at point (1, 2, 4) is ________ m/s^(2).
Q24.
mcq single
+4 / 1
The equation of a plane progressive wave is given by $y = 5 \cos \pi \left( 200t - \frac{x}{150} \right)$ where $x$ and $y$ are in cm and $t$ is in second. The velocity of the wave is ________ m/s.
Q25.
mcq single
+4 / 1
An electromagnetic wave travelling in **x-direction** is described by field equation
$E_y = 300 \sin \omega \left( t - \frac{x}{c} \right)$.
If the electron is restricted to move in **y-direction** only with speed of $1.5 \times 10^6$ m/s then ratio of maximum electric and magnetic forces acting on the electron is ______.
Q26.
mcq single
+4 / 1
For the given circuit (shown in part (A)) the time dependent input voltage $v_{in}(t)$ and corresponding output $v_{o}(t)$ are shown in part (B) and part (C), respectively. Identify the components that are used in the circuit between points X and Y.



Q27.
numerical
+4 / 1
In single slit diffraction pattern, the wavelength of light used is 628 nm and slit width is 0.2 mm, the angular width of central maximum is $\alpha \times 10^{-2}$ degrees. The value of $\alpha$ is ________.
Q28.
mcq single
+4 / 1
The position of an object having mass 0.1 kg as a function of time $t$ is given as
$\vec{r} = \left( 10 t^2 \hat{i} + 5 t^3 \hat{j} \right)$ m. At $t = 1$ s, which of the following statements are **correct**?
**A.** The linear momentum $\vec{p} = \left( 2 \hat{i} + 1.5 \hat{j} \right)$ kg·m/s.
**B.** The force acting on the object $\vec{F} = \left( 2 \hat{i} + 3 \hat{j} \right)$ N.
**C.** The angular momentum of the object about its origin $\vec{L} = 15 \hat{k}$ J·s.
**D.** The torque acting on the object about its origin $\vec{\tau} = 20 \hat{k}$ N·m.
Choose the **correct** answer from the options given below:
Q29.
mcq single
+4 / 1
When a coil is placed in a time dependent magnetic field the power dissipated in it is $P$. The number of turns, area of the coil and radius of the coil wire are $N$, $A$ and $r$ respectively. For a second coil number of turns, area of the coil and radius of the coil wire are $2N$, $2A$ and $3r$ respectively. When the first coil is replaced with second coil the power dissipated in it is $\sqrt{2} \,\alpha P$. The value of $\alpha$ is ______.
Q30.
mcq single
+4 / 1
A particle is rotating in a circular path and at any instant its motion can be described as
$\theta = \frac{5t^4}{40} - \frac{t^3}{3}$.
The angular acceleration of the particle after 10 seconds is _________ rad/s^(2).
Q31.
mcq single
+4 / 1
Heat is supplied to a diatomic gas at constant pressure. Then the ratio of $\Delta Q : \Delta U : \Delta W$ is ______.
Q32.
numerical
+4 / 1
A vessel contains 0.15 m^(3) of a gas at pressure 8 bar and temperature 140 °C with $c_p = 3R$ and $c_v = 2R$. It is expanded adiabatically till pressure falls to 1 bar. The work done during this process is _________ kJ. (R is gas constant)
Q33.
numerical
+4 / 1
A tub is filled with water and a wooden cube $10\ \text{cm} \times 10\ \text{cm} \times 10\ \text{cm}$ is placed in the water. The wooden cube is found to float on the water with a part of it submerged in water. When a metal coin is placed on the wooden cube, the submerged part is increased by $3.87$ cm. The mass of the metal coin is ________ gram.
(Take water density as $1\ \text{g/cm}^3$ and density of wood as $0.4\ \text{g/cm}^3$)
Q34.
numerical
+4 / 1
A uniform wire of length $l$ of weight $w$ is suspended from the roof with a weight of $W$ at the other end. The stress in the wire at $\frac{l}{3}$ distance from the top is $\left( \frac{W}{A} + \frac{2}{\gamma} \frac{w}{A} \right)$, where $A$ is the cross sectional area of the wire. The value of $\gamma$ is ________.
Q35.
mcq single
+4 / 1
A liquid drop of diameter 2 mm breaks into 512 droplets. The change in surface energy is $\alpha \times 10^{-6}$ J. The value of $\alpha$ is _______. (Take surface tension of liquid = 0.08 N/m)
Q36.
mcq single
+4 / 1
For a certain metal, when monochromatic light of wavelength $\lambda$ is incident, the stopping potential for photoelectrons is $3V_0$. When the same metal is illuminated by light of wavelength $2\lambda$, then the stopping potential becomes $V_0$. The threshold wavelength for photoelectric emission for the given metal is $\alpha \lambda$. The value of $\alpha$ is ______.
Q37.
mcq single
+4 / 1
Two short electric dipoles **A** and **B** having dipole moment $p_1$ and $p_2$ respectively are placed with their axis mutually perpendicular as shown in the figure. The resultant electric field at a point $x$ is making an angle of $60^{\circ}$ with the line joining points $O$ and $x$. The ratio of the dipole moments $p_2/p_1$ is _________.


Q38.
mcq single
+4 / 1
Two charged conducting spheres $S_1$ and $S_2$ of radii 8 cm and 18 cm are connected to each other by a wire. After equilibrium is established, the ratio of electric fields on $S_1$ and $S_2$ spheres are $E_{S1}$ and $E_{S2}$ respectively. The value of $\dfrac{E_{S1}}{E_{S2}}$ is ________.
Q39.
mcq single
+4 / 1
A planet ($P_1$) is moving around the star of mass $2M$ in the orbit of radius $R$. Another planet ($P_2$) is moving around another star of mass $4M$ in a orbit of radius $2R$. Ratio of time periods of revolution of $P_2$ and $P_1$ is ________.
Q40.
mcq single
+4 / 1
A parallel plate air capacitor has a capacitance $C$. When it is half filled as shown in figure with a dielectric constant $K = 5$, the percentage increase in the capacitance is ________.


Q41.
mcq single
+4 / 1
Refer the figure given below. $\mu_1$ and $\mu_2$ are refractive indices of air and lens material. The height of image will be _______ cm.


Q42.
mcq single
+4 / 1
For a thin symmetric prism made of glass (refractive index 1.5), the ratio of incident angle and minimum deviation will be _______.
Q43.
numerical
+4 / 1
1 μC charge moving with velocity $\vec{v} = (\hat{i} - 2\hat{j} + 3\hat{k})$ m/s in the region of magnetic field $\vec{B} = (2\hat{i} + 3\hat{j} - 5\hat{k})$ T. The magnitude of force acting on it is $\sqrt{\alpha} \times 10^{-6}$ N. The value of $\alpha$ is _______.
Q44.
mcq single
+4 / 1
Two identical long current carrying wires are bent into the shapes shown in the following figures. If the magnitude of magnetic fields at the centres P and Q of a semicircular arc are $B_1$ and $B_2$ respectively, then the ratio $\frac{B_1}{B_2}$ is ______.


Q45.
mcq single
+4 / 1
The dimensional formula of $\frac{1}{2} \epsilon_0 E^2$ ($\epsilon_0$ = permittivity of vacuum and $E$ = electric field) is $M^a L^b T^c$.
The value of $2a - b + c =$ ________.
Q46.
mcq single
+4 / 1
The diameter of a wire measured by a screw gauge of least count 0.001 cm is 0.08 cm. The length measured by a scale of least count 0.1 cm is 150 cm. When a weight of 100 N is applied to the wire, the extension in length is 0.5 cm, measured by a micrometer of least count 0.001 cm. The error in the measured Young’s modulus is $\alpha \times 10^9 \ \mathrm{N/m}^2$. The value of $\alpha$ is ________.
(Ignore the contribution of the load to Young’s modulus error calculation)
Q47.
mcq single
+4 / 1
Angular momentum of an electron in a hydrogen atom is $\frac{3h}{\pi}$, then the energy of the electron is _____ eV.
Q48.
mcq single
+4 / 1
The velocity of a particle is given as $\vec{v} = -x \hat{i} + 2y \hat{j} - z \hat{k}$ m/s. The magnitude of acceleration at point (1, 2, 4) is ________ m/s^(2).
Q49.
mcq single
+4 / 1
The equation of a plane progressive wave is given by $y = 5 \cos \pi \left( 200t - \frac{x}{150} \right)$ where $x$ and $y$ are in cm and $t$ is in second. The velocity of the wave is ________ m/s.
Q50.
mcq single
+4 / 1
An electromagnetic wave travelling in **x-direction** is described by field equation
$E_y = 300 \sin \omega \left( t - \frac{x}{c} \right)$.
If the electron is restricted to move in **y-direction** only with speed of $1.5 \times 10^6$ m/s then ratio of maximum electric and magnetic forces acting on the electron is ______.