JEE Main 2026 (Online) 4th April Morning Shift
JEE 2026 Previous Year
3 hDuration
296Total Marks
148Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 148 across 3 section(s); maximum marks: 296.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Product C of the following reaction sequence will be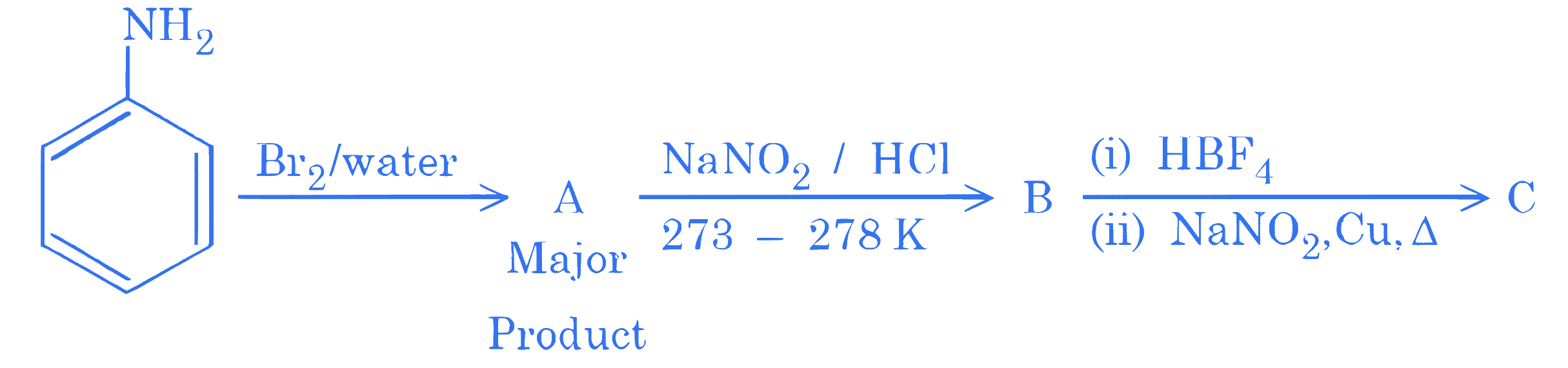

Q2.
mcq single
+4 / 1
The correct statements among the following are,
A. $\mathrm{Mo}(\mathrm{VI})$ and $\mathrm{W}(\mathrm{VI})$ are less stable than $\mathrm{Cr}(\mathrm{VI})$.
B. $\mathrm{Ce}^{4+}$ and $\mathrm{Tb}^{4+}$ are oxidant while $\mathrm{Eu}^{2+}$ and $\mathrm{Yb}^{2+}$ are reductant.
C. Cm and Am have seven unpaired electrons.
D. Actinoid contraction is greater from element to element than lanthanoid contraction.
Choose the correct answer from the options given below :
Q3.
mcq single
+4 / 1
Correct statements from the following are
A. Potassium dichromate is an oxidising agent and it oxidises $\mathrm{FeSO}_4$ to $\mathrm{Fe}_2\left(\mathrm{SO}_4\right)_3$ in acidic medium.
B. Sodium dichromate can be used as primary standard in volumetric estimation.
C. $\mathrm{CrO}_4^{2-}$ and $\mathrm{Cr}_2 \mathrm{O}_7^{2-}$ are interconvertible in aqueous solution by varying the pH of the solution.
D. $\mathrm{Cr}-\mathrm{O}-\mathrm{Cr}$ bond angle in $\mathrm{Cr}_2 \mathrm{O}_7^{2-}$ is $126^{\circ}$.
Choose the correct answer from the options given below :
Q4.
mcq single
+4 / 1
Consider the first order reaction $\mathrm{R} \rightarrow \mathrm{P}$.
The fraction of molecules decomposed in the given first order reaction can be expressed as
Q5.
numerical
+4 / 1
According to Lewis theory, the total number of $\sigma$ bond-pairs and lone pair of electrons around the central atom of $\mathrm{XeO}_6{ }^{4-}$ ion is $\_\_\_\_$ .
Q6.
mcq single
+4 / 1
In order to oxidise a mixture of 1 mole each of $\mathrm{FeC}_2 \mathrm{O}_4, \mathrm{Fe}_2\left(\mathrm{C}_2 \mathrm{O}_4\right)_3, \mathrm{FeSO}_4$ and $\mathrm{Fe}_2\left(\mathrm{SO}_4\right)_3$ in acidic medium, the number of moles of $\mathrm{KMnO}_4$ required is
Q7.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
$$
\begin{gathered}
\text { List-I } \\
\text { Name of reaction }
\end{gathered}
$$
$$
\begin{gathered}
\text { List-II } \\
\text { Reagent or catalyst used }
\end{gathered}
$$
A.
Finkelstein reaction
I.
$\mathrm{SbF}_3$
B.
Swarts reaction
II.
Na, dry ether
C.
Sandmeyer's reaction
III.
NaI
D.
Fittig reaction
IV.
$\mathrm{Cu}_2 \mathrm{Cl}_2$
Choose the correct answer from the options given below :
Q8.
numerical
+4 / 1
A non-volatile, non-electrolyte solid solute when dissolved in 40 g of a solvent, the vapour pressure of the solvent decreased from 760 mm Hg to 750 mm Hg . If the same solution boils at 320 K , then the number of moles of the solvent present in the solution is $\_\_\_\_$ . (Nearest integer)
[Given: boiling point of the pure solvent $=319.5 \mathrm{~K}$,
$\mathrm{K}_{\mathrm{b}}$ of the solvent $=0.3 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$ ]
Q9.
mcq single
+4 / 1
Number of moles and number of molecules in 1.4187 L of $\mathrm{SO}_2$ at STP respectively are
Q10.
mcq single
+4 / 1
At $\mathrm{T}(\mathrm{K})$, the equilibrium constant of
$\mathrm{A}_2(g)+\mathrm{B}_2(g) \rightleftharpoons \mathrm{C}(g)$ is $2.7 \times 10^{-5}$.
What is the equilibrium constant for
$\frac{1}{3} \mathrm{~A}_2(\mathrm{~g})+\frac{1}{3} \mathrm{~B}_2(\mathrm{~g}) \rightleftharpoons \frac{1}{3} \mathrm{C}(\mathrm{g})$ at the same temperature?
Q11.
numerical
+4 / 1
The pH of a solution obtained by mixing 5 mL of $0.1 \mathrm{M} \mathrm{NH}_4 \mathrm{OH}$ solution with 250 mL of $0.1 \mathrm{M} \mathrm{NH}_4 \mathrm{Cl}$ solution is $\_\_\_\_$ $\times 10^{-2}$.(Nearest integer) Given: $\mathrm{pK}_{\mathrm{b}}\left(\mathrm{NH}_4 \mathrm{OH}\right)=4.74$
$$ \begin{aligned} & \log 2=0.30 \\ & \log 3=0.48 \\ & \log 5=0.70 \end{aligned} $$
Q12.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** For an ideal gas, heat capacity at constant volume is always greater than the heat capacity at constant pressure.
**Statement II :** In a constant volume process, no work is produced and all the heat withdrawn goes into the chaotic motion and is reflected by a temperature increase of the ideal gas.
In the light of the above statements, choose the correct answer from the options given below
Q13.
mcq single
+4 / 1
A monoatomic anion (A ) has 45 neutrons and 36 electrons. Atomic mass, group in the periodic table and physical state at room temperature of the element (A) respectively are
Q14.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The covalency of oxygen is generally two but it can exceed upto four. The oxidation state of oxygen in $\mathrm{SO}_2$ is -2 and in $\mathrm{OF}_2$ it is +2 .
**Statement II :** The anomalous behaviour of oxygen when compared to the other elements of group 16 is due to its small size and high electronegativity.
In the light of the above statements, choose the correct answer from the options given below
Q15.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
Complex ion
List-II
Calculated spin only magnetic moment (BM)
A.
$$
\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
I.
3.87
B.
$$
\left[\mathrm{Co}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
II.
5.92
C.
$$
\left[\mathrm{Cu}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
III.
4.90
D.
$$
\left[\mathrm{Mn}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
IV.
1.73
Choose the correct answer from the options given below :
Q16.
numerical
+4 / 1
$$ \text { Consider the following sequence of reactions to give the major product }(\mathrm{X}) $$
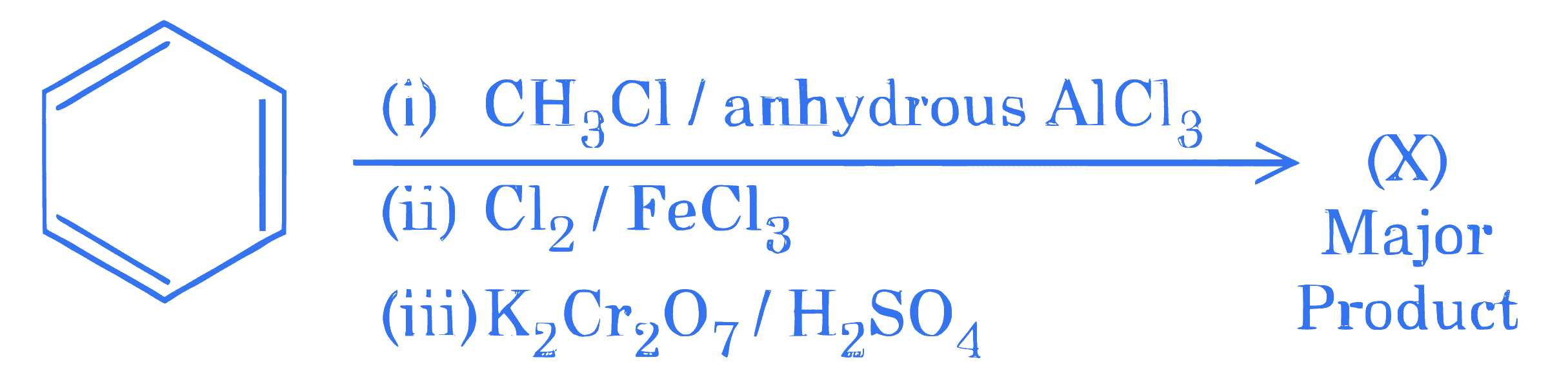 P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
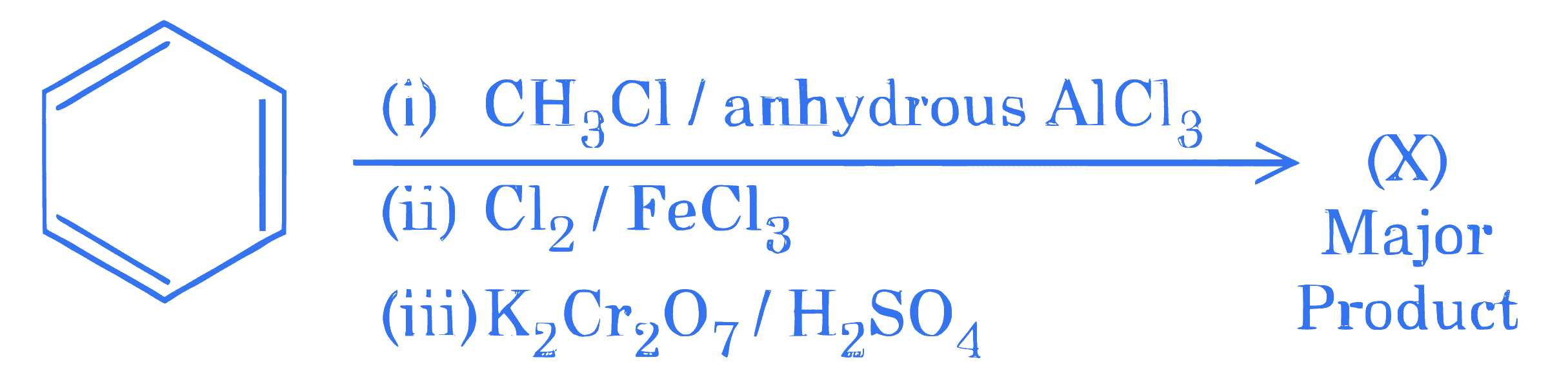 P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
Q17.
numerical
+4 / 1
2.0 g of a bromo hydrocarbon $(\mathrm{X})$ was subjected to Carius analysis, gave 3.36 g of AgBr . The percentage of carbon in the compound $(\mathrm{X})$ is $26.7 \%$. Total number of carbon atoms in the empirical formula for compound $(\mathrm{X})$ is $\_\_\_\_$ .
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{Br}: 80, \mathrm{Ag}: 108$ )
Q18.
mcq single
+4 / 1
Amongst the following, the total number of compounds soluble in aqueous NaOH at room temperature is :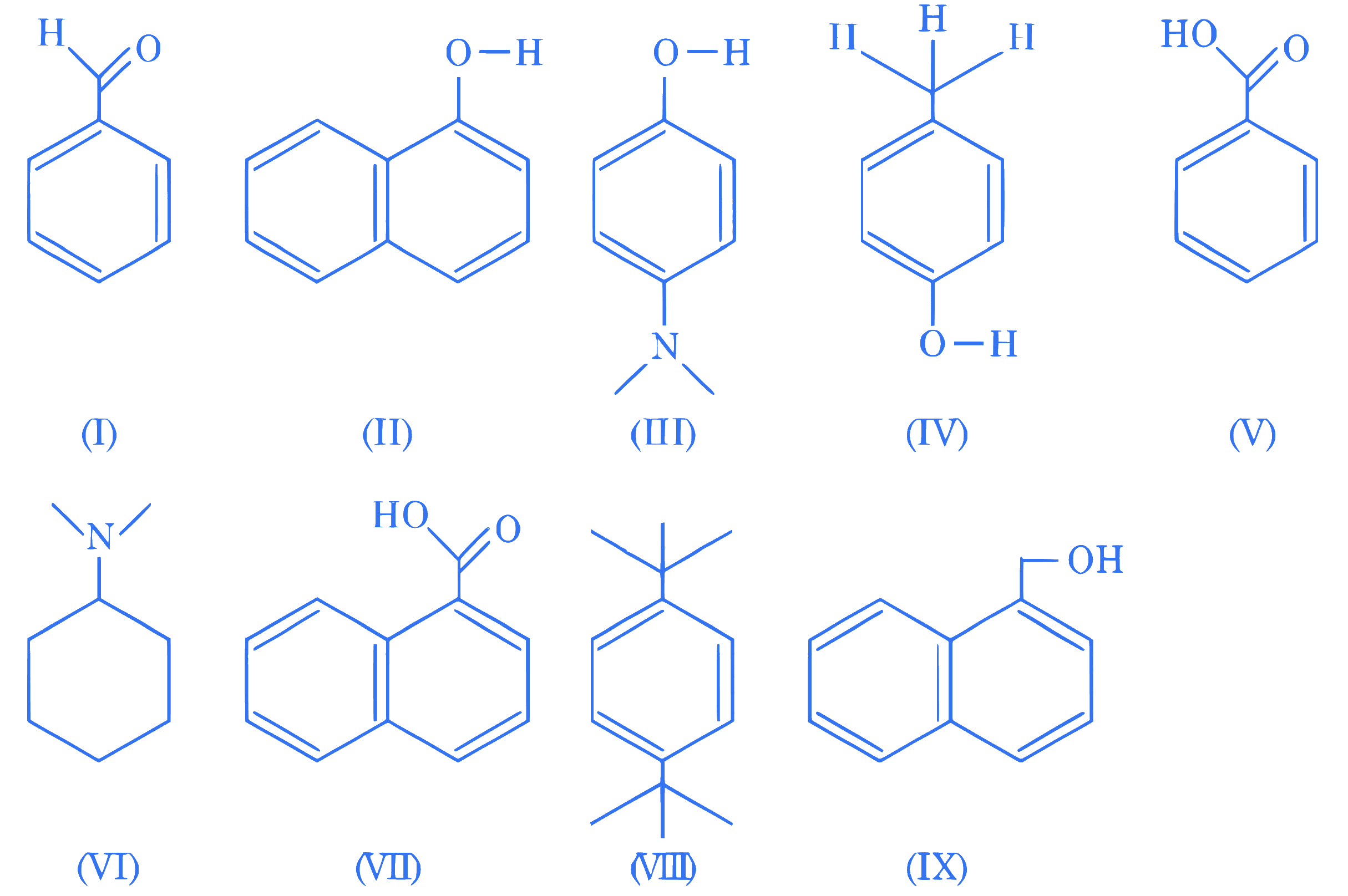

Q19.
mcq single
+4 / 1
$$ \text { An alkene }(\mathrm{X}) \text { on ozonolysis followed by reduction gives following products. } $$
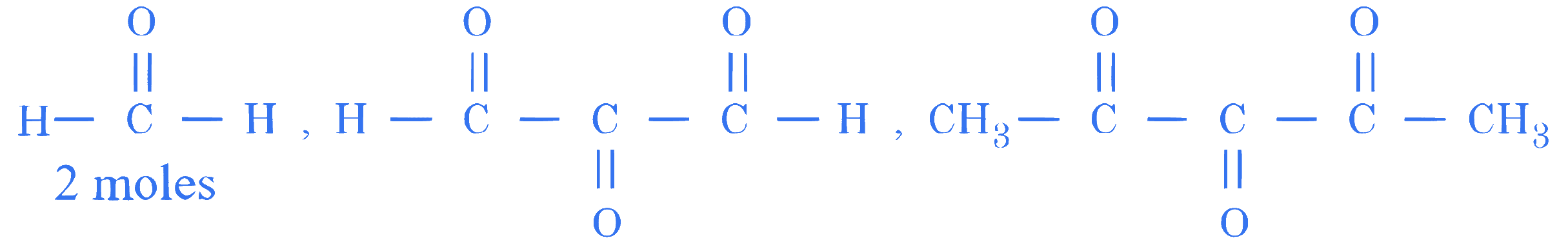 $$ \text { The alkene }(\mathrm{X}) \text { is : } $$
$$ \text { The alkene }(\mathrm{X}) \text { is : } $$
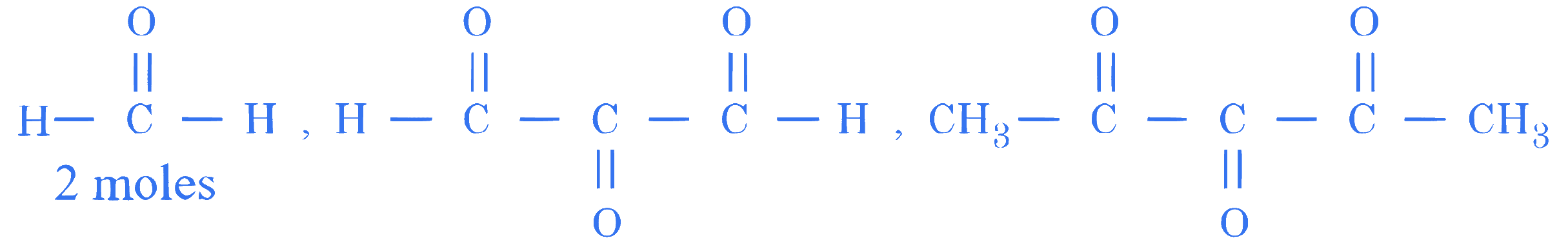 $$ \text { The alkene }(\mathrm{X}) \text { is : } $$
$$ \text { The alkene }(\mathrm{X}) \text { is : } $$
Q20.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The structure of Maltose is given below:
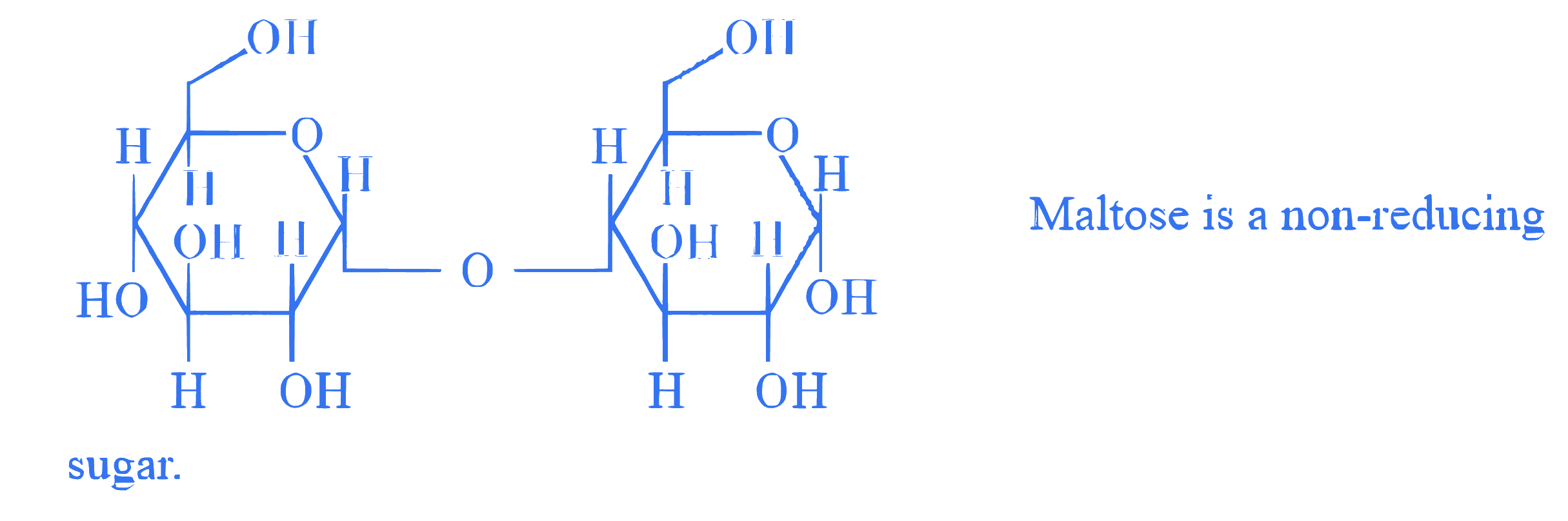 **Statement II :** The structure of Lactose is given below :
**Statement II :** The structure of Lactose is given below :
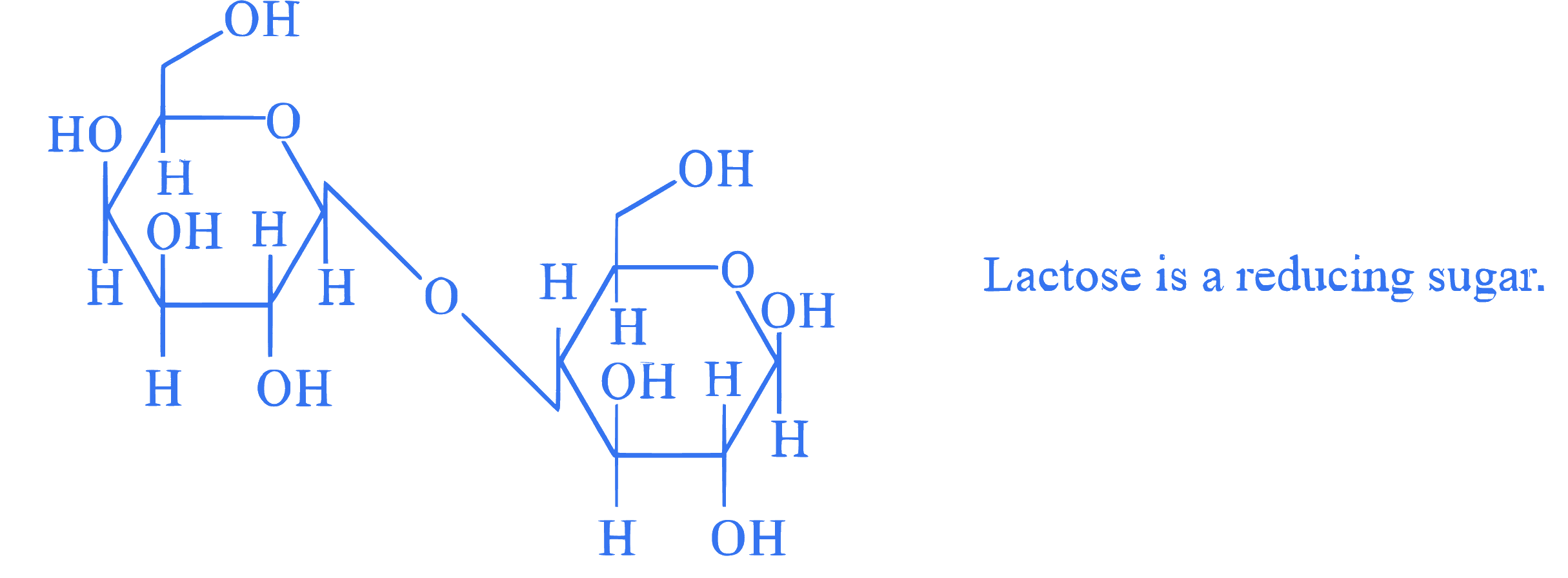 In the light of the above statements, choose the **correct** answer from the options given below :
In the light of the above statements, choose the **correct** answer from the options given below :
 **Statement II :** The structure of Lactose is given below :
**Statement II :** The structure of Lactose is given below :
 In the light of the above statements, choose the **correct** answer from the options given below :
In the light of the above statements, choose the **correct** answer from the options given below :
Q21.
mcq single
+4 / 1
Match the List-I with List-II
List-I**Name of amino acidList-II
One letter symbol/typeA. ArginineI. D/Non-essentialB. Aspartic acidII. R/EssentialC. LysineIII. E/Non-essentialD. Glutamic acidIV. K/Essential
Choose the correct** answer from the options given below :
Q22.
mcq single
+4 / 1
Increasing order of electron withdrawing power of following functional groups is :
a. -CN
b. -COOH
c. $-\mathrm{NO}_2$
d. -I
Q23.
mcq single
+4 / 1
$$ \text { Identify the colour of compound ' } \mathrm{X} \text { ' in the sequence of the reaction. } $$
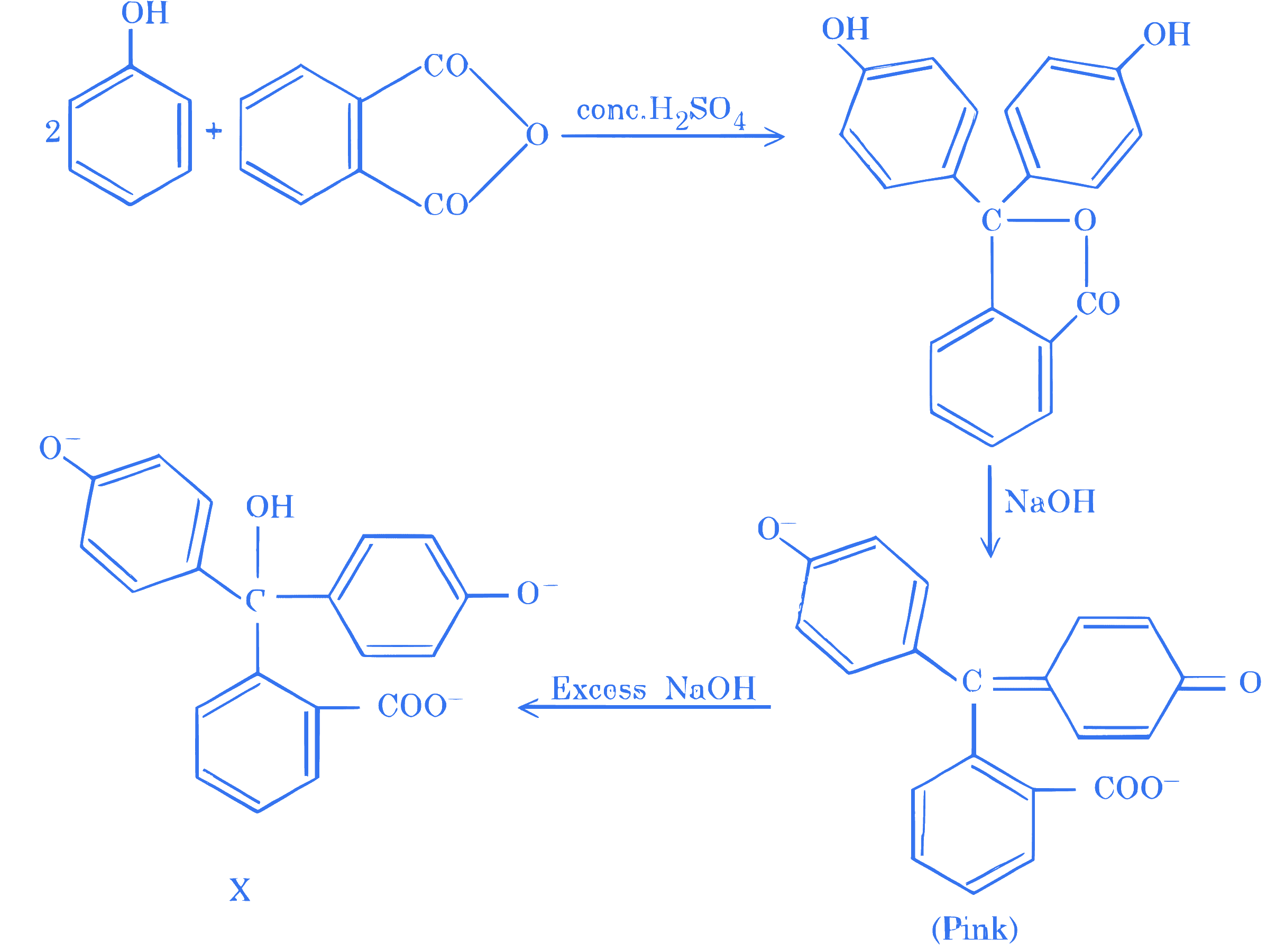

Q24.
mcq single
+4 / 1
What is the ratio of wave number of first line (lowest energy line) of Balmer series of H atomic spectrum to first line of its Brackett series?
Q25.
mcq single
+4 / 1
Which of the following is correct set of 4 quantum numbers of $19^{\text {th }}$ electron in Chromium (Atomic number $=24$ ) in accordance with Aufbau principle?
Q26.
mcq single
+4 / 1
Product C of the following reaction sequence will be

Q27.
mcq single
+4 / 1
The correct statements among the following are,
A. $\mathrm{Mo}(\mathrm{VI})$ and $\mathrm{W}(\mathrm{VI})$ are less stable than $\mathrm{Cr}(\mathrm{VI})$.
B. $\mathrm{Ce}^{4+}$ and $\mathrm{Tb}^{4+}$ are oxidant while $\mathrm{Eu}^{2+}$ and $\mathrm{Yb}^{2+}$ are reductant.
C. Cm and Am have seven unpaired electrons.
D. Actinoid contraction is greater from element to element than lanthanoid contraction.
Choose the correct answer from the options given below :
Q28.
mcq single
+4 / 1
Correct statements from the following are
A. Potassium dichromate is an oxidising agent and it oxidises $\mathrm{FeSO}_4$ to $\mathrm{Fe}_2\left(\mathrm{SO}_4\right)_3$ in acidic medium.
B. Sodium dichromate can be used as primary standard in volumetric estimation.
C. $\mathrm{CrO}_4^{2-}$ and $\mathrm{Cr}_2 \mathrm{O}_7^{2-}$ are interconvertible in aqueous solution by varying the pH of the solution.
D. $\mathrm{Cr}-\mathrm{O}-\mathrm{Cr}$ bond angle in $\mathrm{Cr}_2 \mathrm{O}_7^{2-}$ is $126^{\circ}$.
Choose the correct answer from the options given below :
Q29.
mcq single
+4 / 1
Consider the first order reaction $\mathrm{R} \rightarrow \mathrm{P}$.
The fraction of molecules decomposed in the given first order reaction can be expressed as
Q30.
numerical
+4 / 1
According to Lewis theory, the total number of $\sigma$ bond-pairs and lone pair of electrons around the central atom of $\mathrm{XeO}_6{ }^{4-}$ ion is $\_\_\_\_$ .
Q31.
mcq single
+4 / 1
In order to oxidise a mixture of 1 mole each of $\mathrm{FeC}_2 \mathrm{O}_4, \mathrm{Fe}_2\left(\mathrm{C}_2 \mathrm{O}_4\right)_3, \mathrm{FeSO}_4$ and $\mathrm{Fe}_2\left(\mathrm{SO}_4\right)_3$ in acidic medium, the number of moles of $\mathrm{KMnO}_4$ required is
Q32.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
$$
\begin{gathered}
\text { List-I } \\
\text { Name of reaction }
\end{gathered}
$$
$$
\begin{gathered}
\text { List-II } \\
\text { Reagent or catalyst used }
\end{gathered}
$$
A.
Finkelstein reaction
I.
$\mathrm{SbF}_3$
B.
Swarts reaction
II.
Na, dry ether
C.
Sandmeyer's reaction
III.
NaI
D.
Fittig reaction
IV.
$\mathrm{Cu}_2 \mathrm{Cl}_2$
Choose the correct answer from the options given below :
Q33.
numerical
+4 / 1
A non-volatile, non-electrolyte solid solute when dissolved in 40 g of a solvent, the vapour pressure of the solvent decreased from 760 mm Hg to 750 mm Hg . If the same solution boils at 320 K , then the number of moles of the solvent present in the solution is $\_\_\_\_$ . (Nearest integer)
[Given: boiling point of the pure solvent $=319.5 \mathrm{~K}$,
$\mathrm{K}_{\mathrm{b}}$ of the solvent $=0.3 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$ ]
Q34.
mcq single
+4 / 1
Number of moles and number of molecules in 1.4187 L of $\mathrm{SO}_2$ at STP respectively are
Q35.
mcq single
+4 / 1
At $\mathrm{T}(\mathrm{K})$, the equilibrium constant of
$\mathrm{A}_2(g)+\mathrm{B}_2(g) \rightleftharpoons \mathrm{C}(g)$ is $2.7 \times 10^{-5}$.
What is the equilibrium constant for
$\frac{1}{3} \mathrm{~A}_2(\mathrm{~g})+\frac{1}{3} \mathrm{~B}_2(\mathrm{~g}) \rightleftharpoons \frac{1}{3} \mathrm{C}(\mathrm{g})$ at the same temperature?
Q36.
numerical
+4 / 1
The pH of a solution obtained by mixing 5 mL of $0.1 \mathrm{M} \mathrm{NH}_4 \mathrm{OH}$ solution with 250 mL of $0.1 \mathrm{M} \mathrm{NH}_4 \mathrm{Cl}$ solution is $\_\_\_\_$ $\times 10^{-2}$.(Nearest integer) Given: $\mathrm{pK}_{\mathrm{b}}\left(\mathrm{NH}_4 \mathrm{OH}\right)=4.74$
$$ \begin{aligned} & \log 2=0.30 \\ & \log 3=0.48 \\ & \log 5=0.70 \end{aligned} $$
Q37.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** For an ideal gas, heat capacity at constant volume is always greater than the heat capacity at constant pressure.
**Statement II :** In a constant volume process, no work is produced and all the heat withdrawn goes into the chaotic motion and is reflected by a temperature increase of the ideal gas.
In the light of the above statements, choose the correct answer from the options given below
Q38.
mcq single
+4 / 1
A monoatomic anion (A ) has 45 neutrons and 36 electrons. Atomic mass, group in the periodic table and physical state at room temperature of the element (A) respectively are
Q39.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The covalency of oxygen is generally two but it can exceed upto four. The oxidation state of oxygen in $\mathrm{SO}_2$ is -2 and in $\mathrm{OF}_2$ it is +2 .
**Statement II :** The anomalous behaviour of oxygen when compared to the other elements of group 16 is due to its small size and high electronegativity.
In the light of the above statements, choose the correct answer from the options given below
Q40.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
Complex ion
List-II
Calculated spin only magnetic moment (BM)
A.
$$
\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
I.
3.87
B.
$$
\left[\mathrm{Co}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
II.
5.92
C.
$$
\left[\mathrm{Cu}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
III.
4.90
D.
$$
\left[\mathrm{Mn}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}
$$
IV.
1.73
Choose the correct answer from the options given below :
Q41.
numerical
+4 / 1
$$ \text { Consider the following sequence of reactions to give the major product }(\mathrm{X}) $$
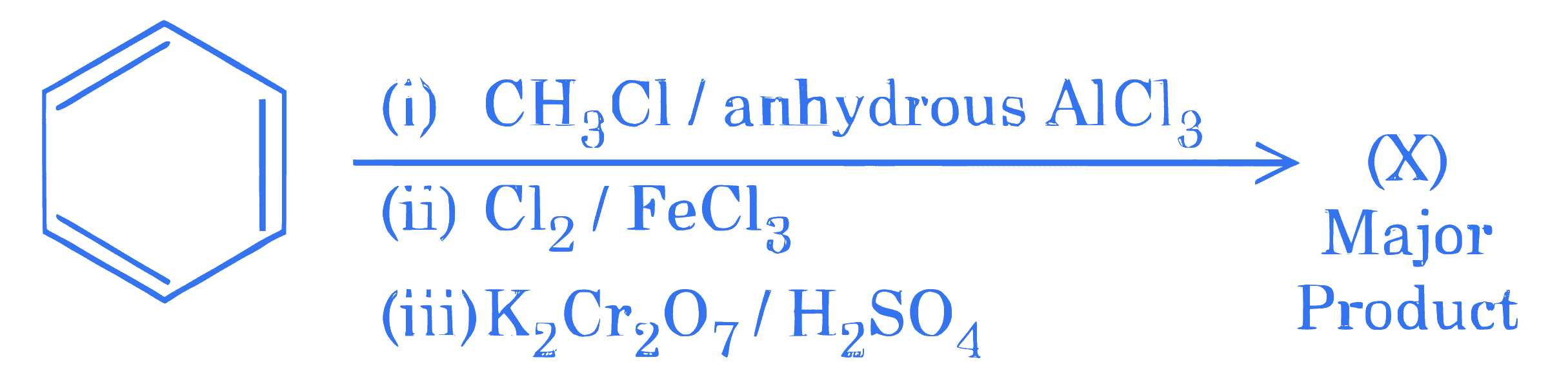 P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
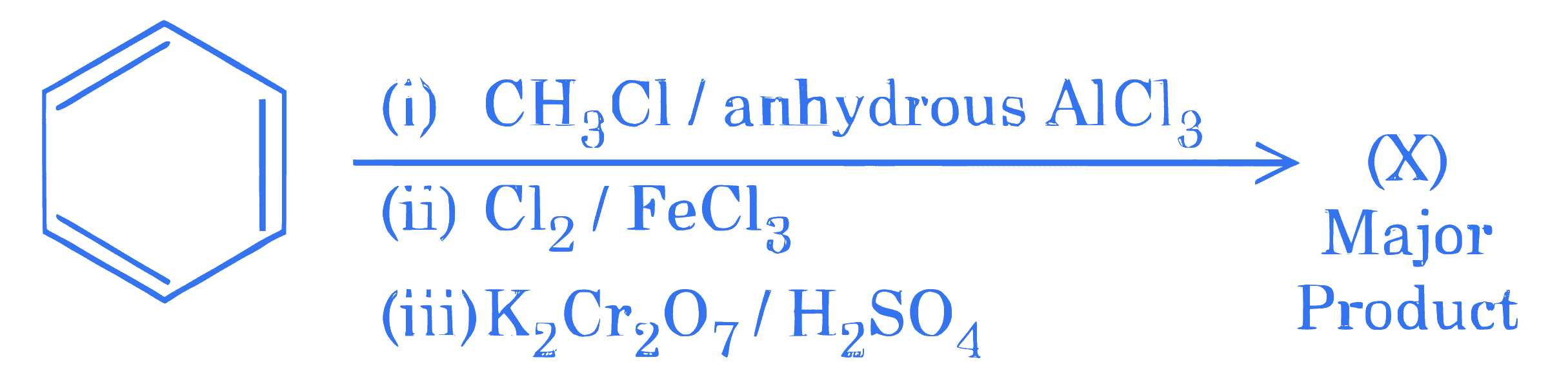 P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
P g of the major product $(\mathrm{X})$ formed is reacted with $\mathrm{NaHCO}_3$ solution to liberate a gas which occupied $11.2 \mathrm{dm}^3$ at STP.
$$ \mathrm{P}=\text {______________ g. } $$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Cl}: 35.5$ )
Q42.
numerical
+4 / 1
2.0 g of a bromo hydrocarbon $(\mathrm{X})$ was subjected to Carius analysis, gave 3.36 g of AgBr . The percentage of carbon in the compound $(\mathrm{X})$ is $26.7 \%$. Total number of carbon atoms in the empirical formula for compound $(\mathrm{X})$ is $\_\_\_\_$ .
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{Br}: 80, \mathrm{Ag}: 108$ )
Q43.
mcq single
+4 / 1
Amongst the following, the total number of compounds soluble in aqueous NaOH at room temperature is :

Q44.
mcq single
+4 / 1
$$ \text { An alkene }(\mathrm{X}) \text { on ozonolysis followed by reduction gives following products. } $$
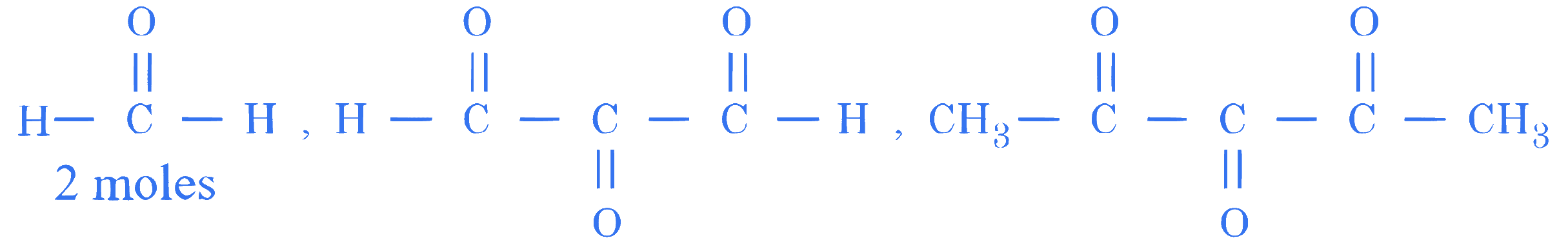 $$ \text { The alkene }(\mathrm{X}) \text { is : } $$
$$ \text { The alkene }(\mathrm{X}) \text { is : } $$
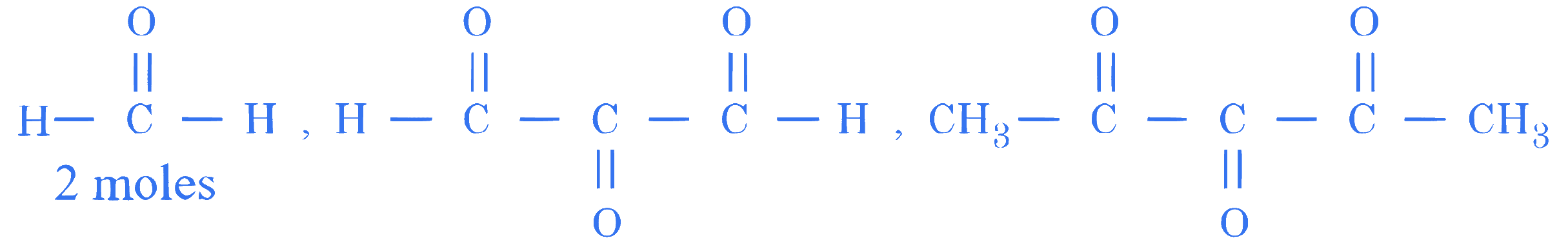 $$ \text { The alkene }(\mathrm{X}) \text { is : } $$
$$ \text { The alkene }(\mathrm{X}) \text { is : } $$
Q45.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The structure of Maltose is given below:
 **Statement II :** The structure of Lactose is given below :
**Statement II :** The structure of Lactose is given below :
 In the light of the above statements, choose the **correct** answer from the options given below :
In the light of the above statements, choose the **correct** answer from the options given below :
 **Statement II :** The structure of Lactose is given below :
**Statement II :** The structure of Lactose is given below :
 In the light of the above statements, choose the **correct** answer from the options given below :
In the light of the above statements, choose the **correct** answer from the options given below :
Q46.
mcq single
+4 / 1
Match the List-I with List-II
List-I**Name of amino acidList-II
One letter symbol/typeA. ArginineI. D/Non-essentialB. Aspartic acidII. R/EssentialC. LysineIII. E/Non-essentialD. Glutamic acidIV. K/Essential
Choose the correct** answer from the options given below :
Q47.
mcq single
+4 / 1
Increasing order of electron withdrawing power of following functional groups is :
a. -CN
b. -COOH
c. $-\mathrm{NO}_2$
d. -I
Q48.
mcq single
+4 / 1
$$ \text { Identify the colour of compound ' } \mathrm{X} \text { ' in the sequence of the reaction. } $$


Q49.
mcq single
+4 / 1
What is the ratio of wave number of first line (lowest energy line) of Balmer series of H atomic spectrum to first line of its Brackett series?
Q50.
mcq single
+4 / 1
Which of the following is correct set of 4 quantum numbers of $19^{\text {th }}$ electron in Chromium (Atomic number $=24$ ) in accordance with Aufbau principle?
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
Suppose that the mean and median of the non-negative numbers $21,8,17, a, 51,103, b, 13,67,(a>b)$, are 40 and 21 , respectively. If the mean deviation about the median is 26 , then $2 a$ is equal to :
Q2.
mcq single
+4 / 1
If the set of all solutions of $\left|x^2+x-9\right|=|x|+\left|x^2-9\right|$ is $[\alpha, \beta] \cup[\gamma, \infty)$, then ( $\alpha^2+\beta^2+\gamma^2$ ) is equal to:
Q3.
numerical
+4 / 1
A coin is tossed 8 times. If the probability that exactly 4 heads appear in the first six tosses and exactly 3 heads appear in the last five tosses is $p$, then $96 p$ is equal to $\_\_\_\_$ .
Q4.
mcq single
+4 / 1
The square of the distance of the point $(-2,-8,6)$ from the line $\frac{x-1}{1}=\frac{y-1}{2}=\frac{z}{-1}$ along the line $\frac{x+5}{1}=\frac{y+5}{-1}=\frac{z}{2}$ is equal to:
Q5.
mcq single
+4 / 1
A line with direction ratios $1,-1,2$ intersects the lines $\frac{x}{2}=\frac{y}{3}=\frac{z+1}{3}$ and $\frac{x+1}{-1}=\frac{y-2}{1}=\frac{z}{4}$ at the points P and Q , respectively. If the length of the line segment PQ is $\alpha$, then $225 \alpha^2$ is equal to:
Q6.
mcq single
+4 / 1
The first term of an A.P. of 30 non-negative terms is $\frac{10}{3}$. If the sum of this A.P. is the cube of its last term, then its common difference is:
Q7.
numerical
+4 / 1
The number of points, at which the function $f(x)=\max \left\{6 x, 2+3 x^2\right\}+|x-1| \cos \left|x^2-\frac{1}{4}\right|, x \in(-\pi, \pi)$, is not differentiable, is
$\_\_\_\_$ .
Q8.
numerical
+4 / 1
Let $\overrightarrow{a_k}=\left(\tan \theta_k\right) \hat{i}+\hat{j}$ and $\overrightarrow{b_k}=\hat{i}-\left(\cot \theta_k\right) \hat{j}$, where $\theta_k=\frac{2^{k-1} \pi}{2^n+1}$, for some $n \in \mathbb{N}, n>5$. Then the value of $\frac{\sum\limits_{k=1}^n\left|\overrightarrow{a_k}\right|^2}{\sum\limits_{k=1}^n\left|\overrightarrow{b_k}\right|^2}$ is
Q9.
numerical
+4 / 1
If $\mathrm{A}=\frac{\sin 3^{\circ}}{\cos 9^{\circ}}+\frac{\sin 9^{\circ}}{\cos 27^{\circ}}+\frac{\sin 27^{\circ}}{\cos 81^{\circ}}$ and $\mathrm{B}=\tan 81^{\circ}-\tan 3^{\circ}$, then $\frac{\mathrm{B}}{\mathrm{A}}$ is equal to
$\_\_\_\_$ .
Q10.
mcq single
+4 / 1
The area of the region $\{(x, y): y \leq \pi-|x|, y \leq|x \sin x|, y \geq 0\}$ is:
Q11.
mcq single
+4 / 1
Let $A=\left[\begin{array}{ccc}1 & 1 & 2 \\ -2 & 0 & 1 \\ 1 & 3 & 5\end{array}\right]$. Then the sum of all elements of the matrix $\operatorname{adj}\left(\operatorname{adj}\left(2(\operatorname{adj} \mathrm{~A})^{-1}\right)\right)$ is equal to:
Q12.
mcq single
+4 / 1
Let $\mathrm{S}=\left\{\mathrm{A}=\left[\begin{array}{ll}a & b \\ c & d\end{array}\right]: a, b, c, d \in\{0,1,2,3,4\}\right.$ and $\left.\mathrm{A}^2-4 \mathrm{~A}+3 \mathrm{I}=0\right\}$ be a set of $2 \times 2$ matrices. Then the number of matrices in S , for which the sum of the diagonal elements is equal to 4 , is :
Q13.
mcq single
+4 / 1
Let the smallest value of $k \in \mathbb{N}$, for which the coefficient of $x^3$ in $(1+x)^3+(1+x)^4+(1+x)^5+\ldots+(1+x)^{99}+(1+k x)^{100}, x \neq 0$, is $\left(43 n+\frac{101}{4}\right)\left({ }^{100} \mathrm{C}_3\right)$ for some $n \in \mathrm{~N}$, be $p$. Then the value of $p+n$ is :
Q14.
mcq single
+4 / 1
Let the vertex A of a triangle ABC be $(1,2)$, and the mid-point of the side AB be $(5,-1)$. If the centroid of this triangle is $(3,4)$ and its circumcenter is $(\alpha, \beta)$, then $21(\alpha+\beta)$ is equal to :
Q15.
mcq single
+4 / 1
Let the line $\mathrm{L}_1: x+3=0$ intersect the lines $\mathrm{L}_2: x-y=0$ and $\mathrm{L}_3: 3 x+y=0$ at the points A and B , respectively. Let the bisector of the obtuse angle between the lines $L_2$ and $L_3$ intersect the line $L_1$ at the point $C$. Then $B C^2: A C^2$ is equal to:
Q16.
mcq single
+4 / 1
Let $z$ be a complex number such that $|z+2|=|z-2|$ and arg $\left(\frac{z+3}{z-i}\right)=\frac{\pi}{4}$. Then $|z|^2$ is equal to:
Q17.
mcq single
+4 / 1
The number of functions $f:\{1,2,3,4\} \rightarrow\{a, b, c\}$, which are not onto, is :
Q18.
mcq single
+4 / 1
Let [•] denote the greatest integer function. If the domain of the function
$f(x)=\cos ^{-1}\left(\frac{4 x+2[x]}{3}\right)$ is $[\alpha, \beta]$, then $12(\alpha+\beta)$ is equal to :
Q19.
mcq single
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $\frac{d y}{d x}=\left(1+x+x^2\right)\left(1-y+y^2\right), y(0)=\frac{1}{2}$. Then $(2 y(1)-1)$ is equal to
Q20.
mcq single
+4 / 1
Suppose that two chords, drawn from the point $(1,2)$ on the circle $x^2+y^2+x-3 y=0$ are bisected by the $y$-axis. If the other ends of these chords are R and S , and the mid point of the line segment RS is $(\alpha, \beta)$, then $6(\alpha+\beta)$ is equal to :
Q21.
numerical
+4 / 1
Consider the parabola $\mathrm{P}: y^2=4 k x$ and the ellipse $\mathrm{E}: \frac{x^2}{a^2}+\frac{y^2}{b^2}=1$. Let the line segment joining the points of intersection of P and E , be their latus rectums. If the eccentricity of E is $e$, then $e^2+2 \sqrt{2}$ is equal to $\_\_\_\_$ .
Q22.
mcq single
+4 / 1
If $y=\tan ^{-1}\left(\frac{3 \cos x-4 \sin x}{4 \cos x+3 \sin x}\right)+2 \tan ^{-1}\left(\frac{x}{1+\sqrt{1-x^2}}\right)$, then $\frac{d y}{d x}$ at $x=\frac{\sqrt{3}}{2}$ is equal to :
Q23.
mcq single
+4 / 1
The number of ways, of forming a queue of 4 boys and 3 girls such that all the girls are not together, is:
Q24.
mcq single
+4 / 1
Let $f$ be a real polynomial of degree $n$ such that $f(x)=f^{\prime}(x) f^{\prime \prime}(x)$, for all $x \in \mathbb{R}$. If $f(0)=0$, then $36\left(f^{\prime}(2)+f^{\prime \prime}(2)+\int_0^2 f(x) d x\right)$ is equal to:
Q25.
mcq single
+4 / 1
Let $\int\limits_{-2}^2(|\sin x|+[x \sin x]) d x=2(3-\cos 2)+\beta$, where [ ⋅ ] is the greatest integer function. Then $\beta \sin \left(\frac{\beta}{2}\right)$ equals:
Q26.
mcq single
+4 / 1
Suppose that the mean and median of the non-negative numbers $21,8,17, a, 51,103, b, 13,67,(a>b)$, are 40 and 21 , respectively. If the mean deviation about the median is 26 , then $2 a$ is equal to :
Q27.
mcq single
+4 / 1
If the set of all solutions of $\left|x^2+x-9\right|=|x|+\left|x^2-9\right|$ is $[\alpha, \beta] \cup[\gamma, \infty)$, then ( $\alpha^2+\beta^2+\gamma^2$ ) is equal to:
Q28.
numerical
+4 / 1
A coin is tossed 8 times. If the probability that exactly 4 heads appear in the first six tosses and exactly 3 heads appear in the last five tosses is $p$, then $96 p$ is equal to $\_\_\_\_$ .
Q29.
mcq single
+4 / 1
The square of the distance of the point $(-2,-8,6)$ from the line $\frac{x-1}{1}=\frac{y-1}{2}=\frac{z}{-1}$ along the line $\frac{x+5}{1}=\frac{y+5}{-1}=\frac{z}{2}$ is equal to:
Q30.
mcq single
+4 / 1
A line with direction ratios $1,-1,2$ intersects the lines $\frac{x}{2}=\frac{y}{3}=\frac{z+1}{3}$ and $\frac{x+1}{-1}=\frac{y-2}{1}=\frac{z}{4}$ at the points P and Q , respectively. If the length of the line segment PQ is $\alpha$, then $225 \alpha^2$ is equal to:
Q31.
mcq single
+4 / 1
The first term of an A.P. of 30 non-negative terms is $\frac{10}{3}$. If the sum of this A.P. is the cube of its last term, then its common difference is:
Q32.
numerical
+4 / 1
The number of points, at which the function $f(x)=\max \left\{6 x, 2+3 x^2\right\}+|x-1| \cos \left|x^2-\frac{1}{4}\right|, x \in(-\pi, \pi)$, is not differentiable, is
$\_\_\_\_$ .
Q33.
numerical
+4 / 1
Let $\overrightarrow{a_k}=\left(\tan \theta_k\right) \hat{i}+\hat{j}$ and $\overrightarrow{b_k}=\hat{i}-\left(\cot \theta_k\right) \hat{j}$, where $\theta_k=\frac{2^{k-1} \pi}{2^n+1}$, for some $n \in \mathbb{N}, n>5$. Then the value of $\frac{\sum\limits_{k=1}^n\left|\overrightarrow{a_k}\right|^2}{\sum\limits_{k=1}^n\left|\overrightarrow{b_k}\right|^2}$ is
Q34.
numerical
+4 / 1
If $\mathrm{A}=\frac{\sin 3^{\circ}}{\cos 9^{\circ}}+\frac{\sin 9^{\circ}}{\cos 27^{\circ}}+\frac{\sin 27^{\circ}}{\cos 81^{\circ}}$ and $\mathrm{B}=\tan 81^{\circ}-\tan 3^{\circ}$, then $\frac{\mathrm{B}}{\mathrm{A}}$ is equal to
$\_\_\_\_$ .
Q35.
mcq single
+4 / 1
The area of the region $\{(x, y): y \leq \pi-|x|, y \leq|x \sin x|, y \geq 0\}$ is:
Q36.
mcq single
+4 / 1
Let $A=\left[\begin{array}{ccc}1 & 1 & 2 \\ -2 & 0 & 1 \\ 1 & 3 & 5\end{array}\right]$. Then the sum of all elements of the matrix $\operatorname{adj}\left(\operatorname{adj}\left(2(\operatorname{adj} \mathrm{~A})^{-1}\right)\right)$ is equal to:
Q37.
mcq single
+4 / 1
Let $\mathrm{S}=\left\{\mathrm{A}=\left[\begin{array}{ll}a & b \\ c & d\end{array}\right]: a, b, c, d \in\{0,1,2,3,4\}\right.$ and $\left.\mathrm{A}^2-4 \mathrm{~A}+3 \mathrm{I}=0\right\}$ be a set of $2 \times 2$ matrices. Then the number of matrices in S , for which the sum of the diagonal elements is equal to 4 , is :
Q38.
mcq single
+4 / 1
Let the smallest value of $k \in \mathbb{N}$, for which the coefficient of $x^3$ in $(1+x)^3+(1+x)^4+(1+x)^5+\ldots+(1+x)^{99}+(1+k x)^{100}, x \neq 0$, is $\left(43 n+\frac{101}{4}\right)\left({ }^{100} \mathrm{C}_3\right)$ for some $n \in \mathrm{~N}$, be $p$. Then the value of $p+n$ is :
Q39.
mcq single
+4 / 1
Let the vertex A of a triangle ABC be $(1,2)$, and the mid-point of the side AB be $(5,-1)$. If the centroid of this triangle is $(3,4)$ and its circumcenter is $(\alpha, \beta)$, then $21(\alpha+\beta)$ is equal to :
Q40.
mcq single
+4 / 1
Let the line $\mathrm{L}_1: x+3=0$ intersect the lines $\mathrm{L}_2: x-y=0$ and $\mathrm{L}_3: 3 x+y=0$ at the points A and B , respectively. Let the bisector of the obtuse angle between the lines $L_2$ and $L_3$ intersect the line $L_1$ at the point $C$. Then $B C^2: A C^2$ is equal to:
Q41.
mcq single
+4 / 1
Let $z$ be a complex number such that $|z+2|=|z-2|$ and arg $\left(\frac{z+3}{z-i}\right)=\frac{\pi}{4}$. Then $|z|^2$ is equal to:
Q42.
mcq single
+4 / 1
The number of functions $f:\{1,2,3,4\} \rightarrow\{a, b, c\}$, which are not onto, is :
Q43.
mcq single
+4 / 1
Let [•] denote the greatest integer function. If the domain of the function
$f(x)=\cos ^{-1}\left(\frac{4 x+2[x]}{3}\right)$ is $[\alpha, \beta]$, then $12(\alpha+\beta)$ is equal to :
Q44.
mcq single
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $\frac{d y}{d x}=\left(1+x+x^2\right)\left(1-y+y^2\right), y(0)=\frac{1}{2}$. Then $(2 y(1)-1)$ is equal to
Q45.
mcq single
+4 / 1
Suppose that two chords, drawn from the point $(1,2)$ on the circle $x^2+y^2+x-3 y=0$ are bisected by the $y$-axis. If the other ends of these chords are R and S , and the mid point of the line segment RS is $(\alpha, \beta)$, then $6(\alpha+\beta)$ is equal to :
Q46.
numerical
+4 / 1
Consider the parabola $\mathrm{P}: y^2=4 k x$ and the ellipse $\mathrm{E}: \frac{x^2}{a^2}+\frac{y^2}{b^2}=1$. Let the line segment joining the points of intersection of P and E , be their latus rectums. If the eccentricity of E is $e$, then $e^2+2 \sqrt{2}$ is equal to $\_\_\_\_$ .
Q47.
mcq single
+4 / 1
If $y=\tan ^{-1}\left(\frac{3 \cos x-4 \sin x}{4 \cos x+3 \sin x}\right)+2 \tan ^{-1}\left(\frac{x}{1+\sqrt{1-x^2}}\right)$, then $\frac{d y}{d x}$ at $x=\frac{\sqrt{3}}{2}$ is equal to :
Q48.
mcq single
+4 / 1
The number of ways, of forming a queue of 4 boys and 3 girls such that all the girls are not together, is:
Q49.
mcq single
+4 / 1
Let $f$ be a real polynomial of degree $n$ such that $f(x)=f^{\prime}(x) f^{\prime \prime}(x)$, for all $x \in \mathbb{R}$. If $f(0)=0$, then $36\left(f^{\prime}(2)+f^{\prime \prime}(2)+\int_0^2 f(x) d x\right)$ is equal to:
Q50.
mcq single
+4 / 1
Let $\int\limits_{-2}^2(|\sin x|+[x \sin x]) d x=2(3-\cos 2)+\beta$, where [ ⋅ ] is the greatest integer function. Then $\beta \sin \left(\frac{\beta}{2}\right)$ equals:
Physics
Physics
Q1.
numerical
+4 / 1
A 1 kg block subjected to two simultaneous forces $(2 \hat{\mathrm{i}}+3 \hat{\mathrm{j}}+4 \hat{\mathrm{k}}) \mathrm{N}$ and $(3 \hat{\mathrm{i}}-\hat{\mathrm{j}}-2 \hat{\mathrm{k}}) \mathrm{N}$ is moved a distance of 25 m along $(3 \hat{\mathrm{i}}-4 \hat{\mathrm{j}})$ direction. The work done in this process is $\_\_\_\_$ J.
Q2.
numerical
+4 / 1
A body of mass 2 kg begins to move under the influence of time dependent force $\overrightarrow{\mathrm{F}}=\left(2 \mathrm{t} \hat{\mathrm{i}}+6 \mathrm{t}^2 \hat{\mathrm{j}}\right) \mathrm{N}$, where $\hat{\mathrm{i}}$ and $\hat{\mathrm{j}}$ are unit vectors along $x$ and y -axis respectively. The power produced by the force at $t=2 \mathrm{~s}$ is $\_\_\_\_$ W.
Q3.
mcq single
+4 / 1
Two 4 bits binary numbers, $A=1101$ and $B=1010$ are given in the inputs of a logic circuit shown in figure below. The output $(Y)$ will be :
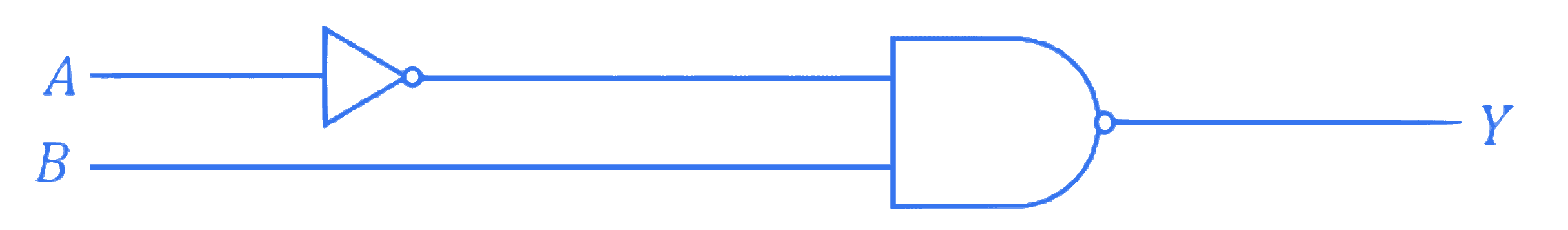

Q4.
mcq single
+4 / 1
Consider a circuit consisting of a capacitor $(20 \mu \mathrm{~F})$, resistor $(100 \Omega)$ and two identical diodes as shown in figure. The resistance of diode under forward biasing condition is $10 \Omega$. The time constant of the circuit is $\alpha \times 10^{-3} \mathrm{~s}$. The value of $\alpha$ is $\_\_\_\_$
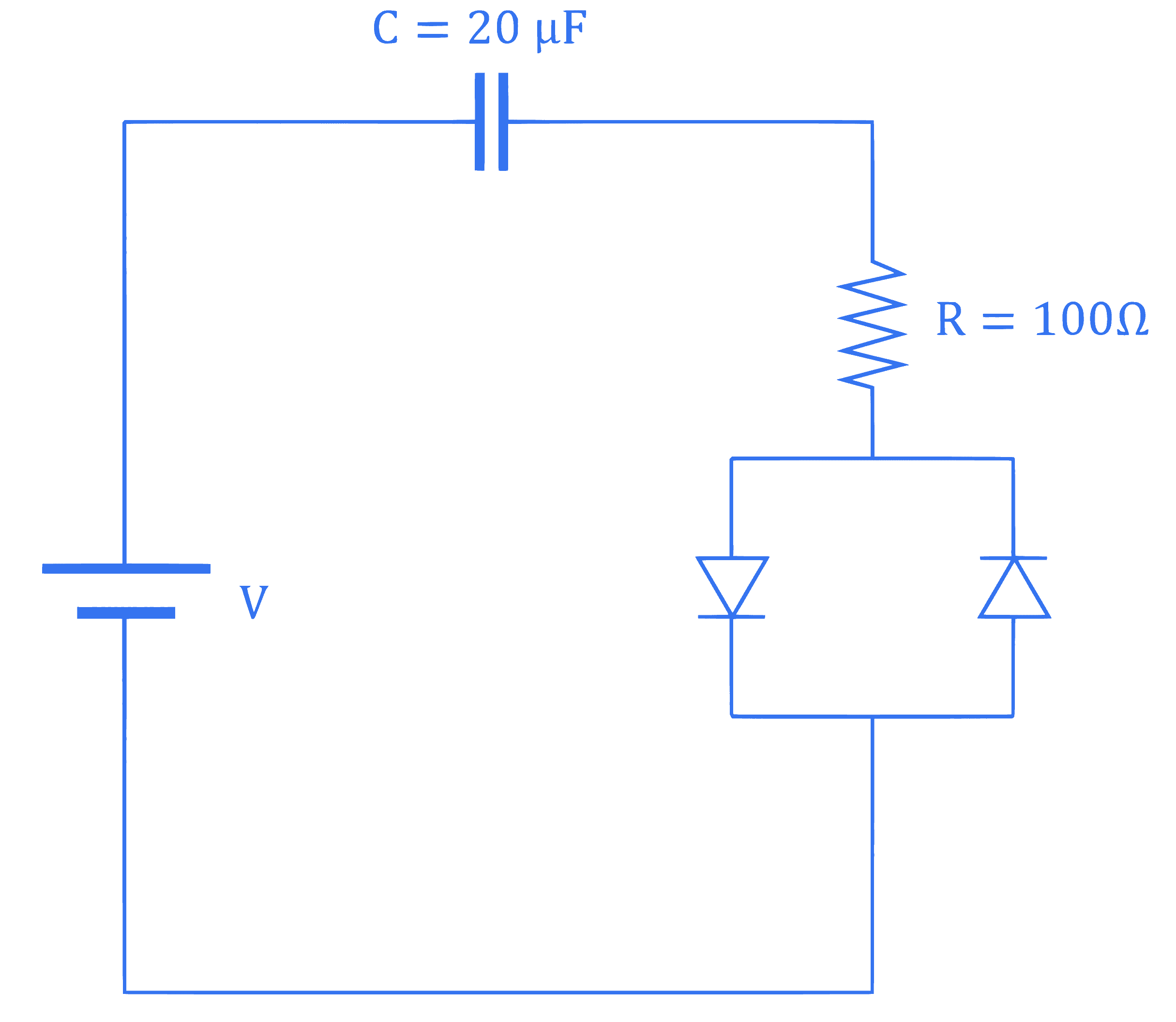

Q5.
mcq single
+4 / 1
A slit of width $a$ is illuminated by light of wavelength $\lambda$. The linear separation between $1^{\text {st }}$ and $3^{\text {rd }}$ minima in the diffraction pattern produced on a screen placed at a distance $D$ from the slit system is $\_\_\_\_$ .
Q6.
mcq single
+4 / 1
An unpolarized light is incident on the plane interface of air-dielectric medium shown in figure. If the incident angle is equal to Brewster angle, identify the expression representing reflected wave.


Q7.
numerical
+4 / 1
The velocity of a particle executing simple harmonic motion along $x$-axis is described as $v^2=50-x^2$, where $x$ represents displacement. If the time period of motion is $\frac{x}{7} \mathrm{~s}$, the value of $x$ is $\_\_\_\_$ .
Q8.
mcq single
+4 / 1
A solid sphere of mass $M$ and radius $R$ is divided into two unequal parts. The smaller part having mass $M / 8$ is converted into a sphere of radius $r$ and the larger part is converted into a circular disc of thickness $t$ and radius $2 R$. If $I_1$ is moment of inertia of a sphere having radius $r$ about an axis through its centre and $I_2$ is the moment of inertia of a disc about its diameter, the ratio of their moment of inertia $I_2 / I_1=$ $\_\_\_\_$
Q9.
mcq single
+4 / 1
An ideal gas undergoes a process maintaining relation between pressure $(P)$ and $\operatorname{volume}(V)$ as $P=P_{\mathrm{o}}\left(1+\left(\frac{V_{\mathrm{o}}}{V}\right)^2\right)^{-1}$, where $P_{\mathrm{o}}$ and $V_{\mathrm{o}}$ are constants. If two samples $A$ and $B$ (two moles each) with initial volumes $V_{\mathrm{o}}$ and $3 V_{\mathrm{o}}$ respectively undergo above mentioned process and attain same pressure, then the difference at the temperatures of these samples, $T_B-T_A$ is $\_\_\_\_$ .
( $R=$ gas constant)
Q10.
mcq single
+4 / 1
One gas of $n_1$ mole of molecules at temperature $T_1$, volume $V_1$, and pressure $P_1$, and another gas of $n_2$ mole of molecules at temperature $T_2$, volume $V_2$, and pressure $P_2$, are mixed resulting in pressure $P$ and volume $V$ of the mixture. The temperature of the mixture is $\_\_\_\_$ .
Q11.
mcq single
+4 / 1
The increase in the pressure required to decrease the volume ( $\Delta V$ ) of water is $6.3 \times 10^7 \mathrm{~N} / \mathrm{m}^2$. The percentage decrease in the volume is $\_\_\_\_$ .
(Bulk modulus of water $=2.1 \times 10^9 \mathrm{~N} / \mathrm{m}^2$.)
Q12.
numerical
+4 / 1
The surface tension of a soap solution is $3.5 \times 10^{-2} \mathrm{~N} / \mathrm{m}$. The work required to increase the radius of a soap bubble from 1 cm to 2 cm is $\alpha \times 10^{-6} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
$$ (\pi=22 / 7) $$
Q13.
mcq single
+4 / 1
A string $A$ of length 0.314 m and Young's modulus $2 \times 10^{10} \mathrm{~N} / \mathrm{m}^2$ is connected to another string $B$ of length and Young's modulus both twice of those of $A$. This series combination of strings is then suspended from a rigid support and its free end is fixed to a load of mass 0.8 kg . The net change in length of the combination is $\_\_\_\_$ mm.
(radius of both the strings is 0.2 mm and acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2$ ) (Mass of both strings is to be neglected as compared to the mass of load)
Q14.
mcq single
+4 / 1
The graph shows variation of stopping potential $V_{\mathrm{o}}$ with the frequency $v$ of the incident radiation for three photosensitive metals $X_1, X_2$ and $X_3$. Which metal will give out electrons with greater kinetic energy, for the same wavelength of incident radiation?
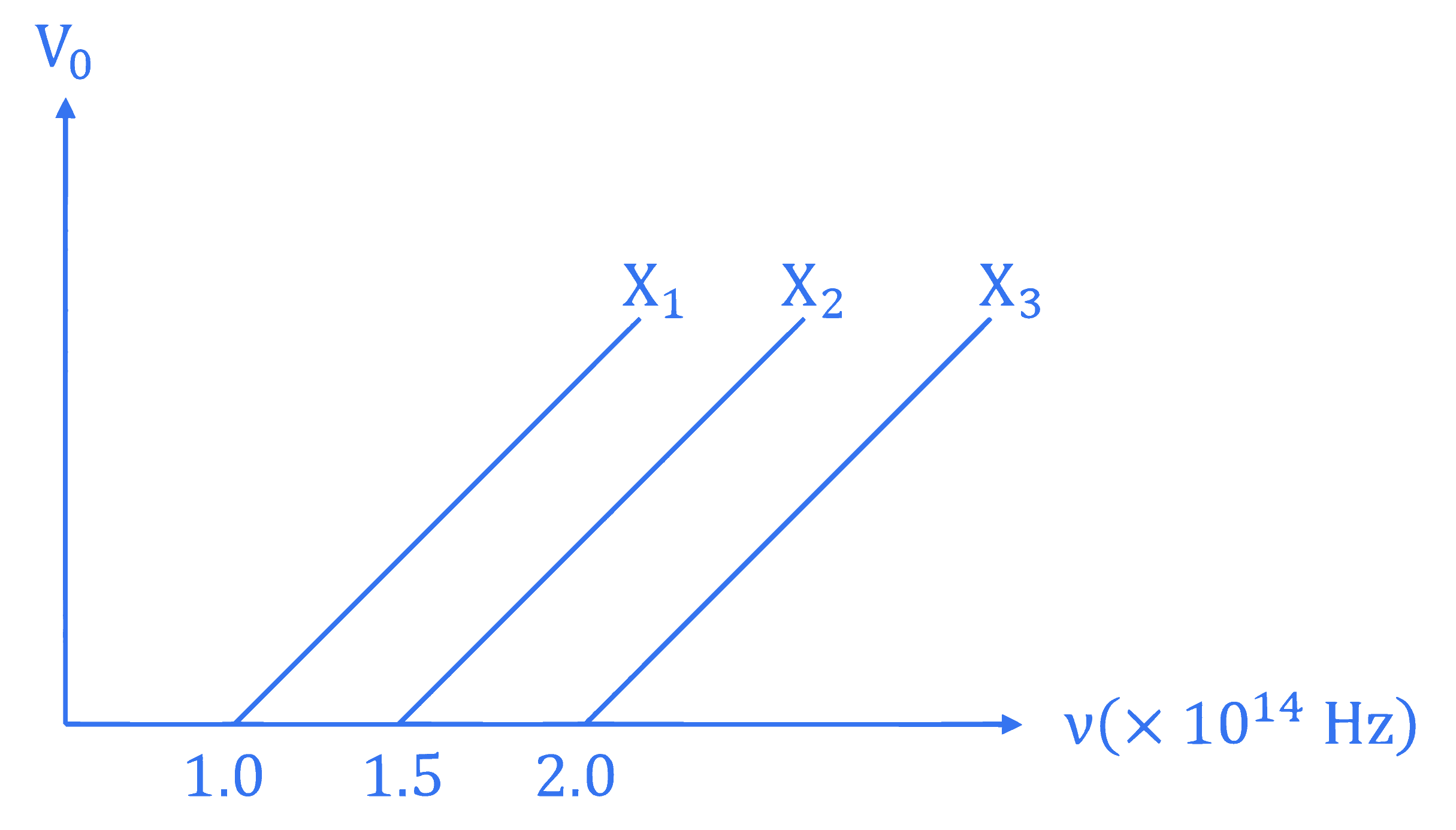

Q15.
mcq single
+4 / 1
A parallel plate air capacitor is connected to a battery. The plates are pulled apart at uniform speed $v$. If $x$ is the separation between the plates at any instant, then the time rate of change of electrostatic energy of the capacitor is proportional to $x^\alpha$, where $\alpha$ is $\_\_\_\_$ .
Q16.
mcq single
+4 / 1
A rod of length 10 cm lies along the principle axis of a concave mirror of focal length 10 cm as shown in figure. The length of the image is $\_\_\_\_$ cm.
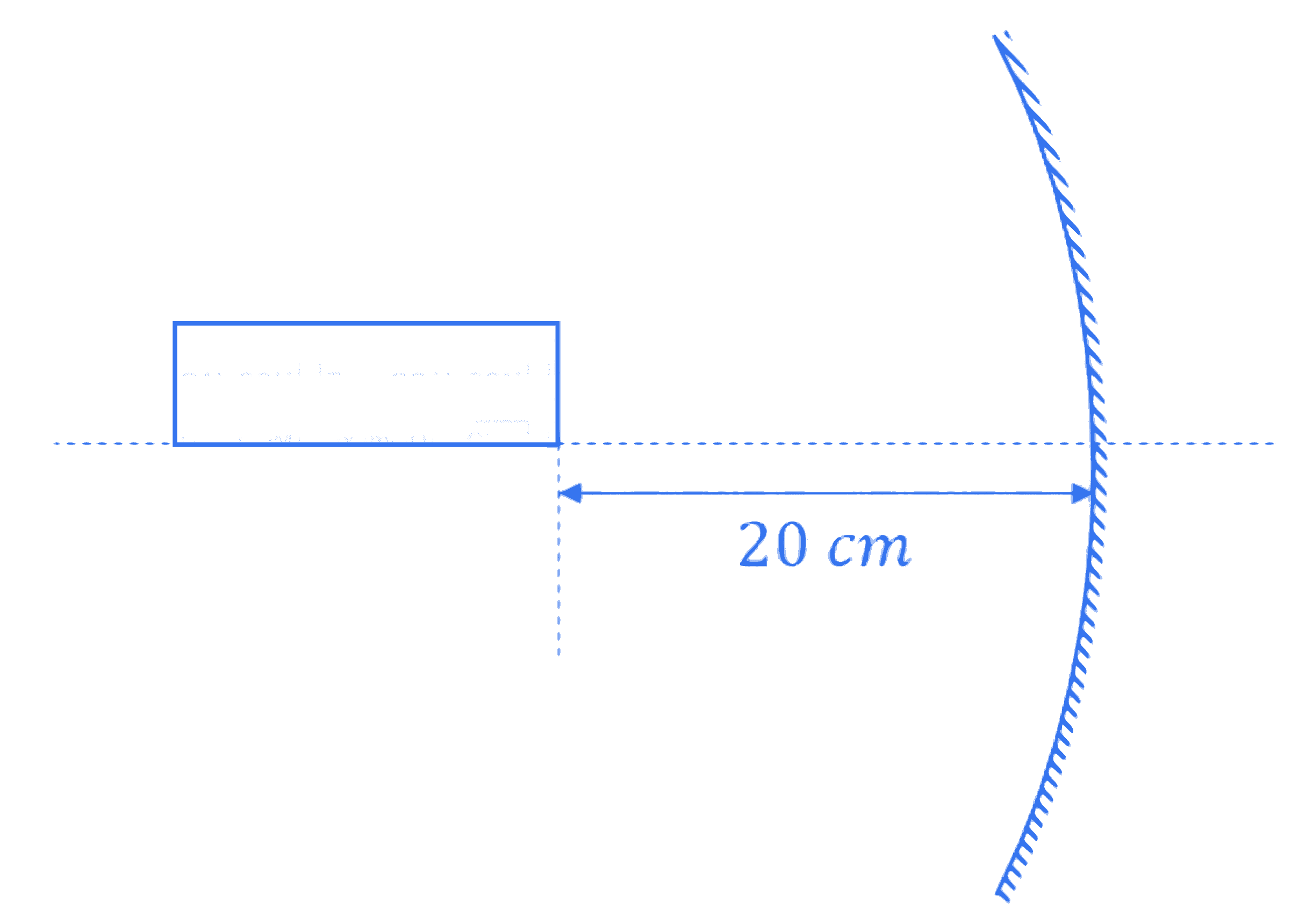

Q17.
mcq single
+4 / 1
An insulated wire is wound so that it forms a flat coil with $N=200$ turns. The radius of the innermost turn is $r_1=3 \mathrm{~cm}$, and of the outermost turn $r_2=6 \mathrm{~cm}$. If 20 mA current flows in it then the magnetic moment will be $\alpha \times 10^{-2} \mathrm{~A} . \mathrm{m}^2$. The value of $\alpha$ is $\_\_\_\_$ .
Q18.
mcq single
+4 / 1
In a screw gauge when the circular scale is given five complete rotations it moves linearly by 2.5 mm . If the circular scale has 100 divisions, the least count of screw gauge is $\_\_\_\_$ mm.
Q19.
mcq single
+4 / 1
Two nuclei of mass number 3 combine with another nucleus of mass number 4 to yield a nucleus of mass number 10. If the binding energy per nucleon for the mass numbers 3,4 and 10 are $5.6 \mathrm{MeV}, 7.4 \mathrm{MeV}$ and 6.1 MeV , respectively, then in the process, $\Delta \mathrm{Mc}^2=$ $\_\_\_\_$ MeV .
Q20.
mcq single
+4 / 1
The two projectiles are projected with the same initial velocities at the $15^{\circ}$ and $30^{\circ}$ with respect to the horizontal. The ratio of their ranges is $1: x$. The value of $x$ is
Q21.
mcq single
+4 / 1
The time taken by a block of mass $m$ to slide down from the highest point to the lowest point on a rough inclined plane is $50 \%$ more compared to the time taken by the same block on identical inclined smooth plane. Both inclined planes are at $45^{\circ}$ with the horizontal. The coefficient of kinetic friction between the rough inclined surface and block is $\_\_\_\_$
Q22.
mcq single
+4 / 1
The voltage and the current between $A$ and $B$ points shown in the circuit are $\_\_\_\_$ .
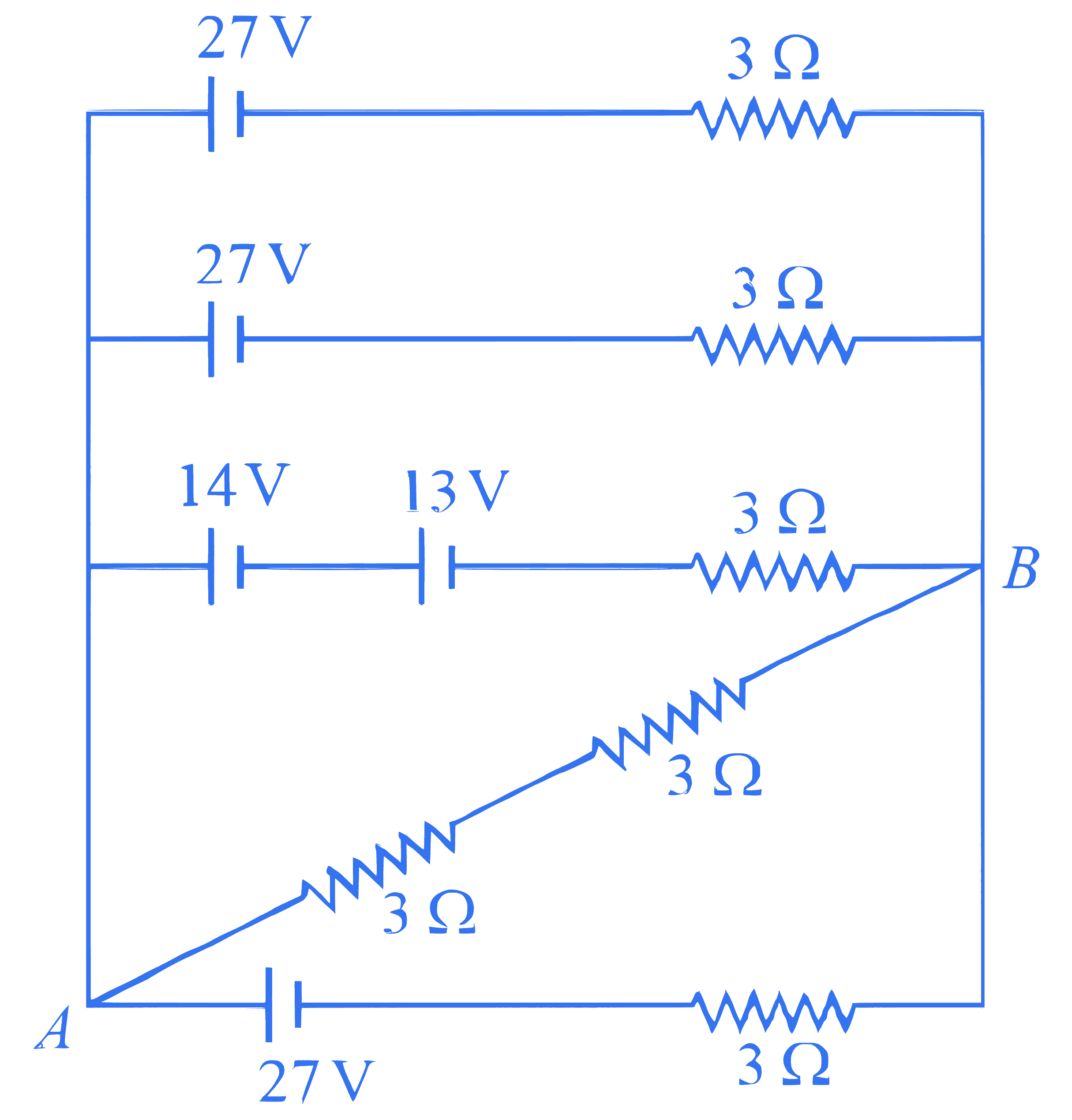

Q23.
mcq single
+4 / 1
A voltmeter with internal resistance of $x \Omega$ can be used to measure upto 20 V . In order to increase its measuring range to 30 V , the required modification is to $\_\_\_\_$ .
Q24.
numerical
+4 / 1
An inductor of 10 mH , capacitor of $0.1 \mu \mathrm{~F}$ and a resistor of $100 \Omega$ are connected in series across an $a . c$ power supply $220 \mathrm{~V}, 70 \mathrm{~Hz}$. The power factor of the given circuit is 0.5 . The difference in the inductive reactance and capacitance reactance is $\sqrt{3} \alpha \Omega$. The value of $\alpha$ is $\_\_\_\_$ .
Q25.
numerical
+4 / 1
A 1 kg block subjected to two simultaneous forces $(2 \hat{\mathrm{i}}+3 \hat{\mathrm{j}}+4 \hat{\mathrm{k}}) \mathrm{N}$ and $(3 \hat{\mathrm{i}}-\hat{\mathrm{j}}-2 \hat{\mathrm{k}}) \mathrm{N}$ is moved a distance of 25 m along $(3 \hat{\mathrm{i}}-4 \hat{\mathrm{j}})$ direction. The work done in this process is $\_\_\_\_$ J.
Q26.
numerical
+4 / 1
A body of mass 2 kg begins to move under the influence of time dependent force $\overrightarrow{\mathrm{F}}=\left(2 \mathrm{t} \hat{\mathrm{i}}+6 \mathrm{t}^2 \hat{\mathrm{j}}\right) \mathrm{N}$, where $\hat{\mathrm{i}}$ and $\hat{\mathrm{j}}$ are unit vectors along $x$ and y -axis respectively. The power produced by the force at $t=2 \mathrm{~s}$ is $\_\_\_\_$ W.
Q27.
mcq single
+4 / 1
Two 4 bits binary numbers, $A=1101$ and $B=1010$ are given in the inputs of a logic circuit shown in figure below. The output $(Y)$ will be :


Q28.
mcq single
+4 / 1
Consider a circuit consisting of a capacitor $(20 \mu \mathrm{~F})$, resistor $(100 \Omega)$ and two identical diodes as shown in figure. The resistance of diode under forward biasing condition is $10 \Omega$. The time constant of the circuit is $\alpha \times 10^{-3} \mathrm{~s}$. The value of $\alpha$ is $\_\_\_\_$


Q29.
mcq single
+4 / 1
A slit of width $a$ is illuminated by light of wavelength $\lambda$. The linear separation between $1^{\text {st }}$ and $3^{\text {rd }}$ minima in the diffraction pattern produced on a screen placed at a distance $D$ from the slit system is $\_\_\_\_$ .
Q30.
mcq single
+4 / 1
An unpolarized light is incident on the plane interface of air-dielectric medium shown in figure. If the incident angle is equal to Brewster angle, identify the expression representing reflected wave.


Q31.
numerical
+4 / 1
The velocity of a particle executing simple harmonic motion along $x$-axis is described as $v^2=50-x^2$, where $x$ represents displacement. If the time period of motion is $\frac{x}{7} \mathrm{~s}$, the value of $x$ is $\_\_\_\_$ .
Q32.
mcq single
+4 / 1
A solid sphere of mass $M$ and radius $R$ is divided into two unequal parts. The smaller part having mass $M / 8$ is converted into a sphere of radius $r$ and the larger part is converted into a circular disc of thickness $t$ and radius $2 R$. If $I_1$ is moment of inertia of a sphere having radius $r$ about an axis through its centre and $I_2$ is the moment of inertia of a disc about its diameter, the ratio of their moment of inertia $I_2 / I_1=$ $\_\_\_\_$
Q33.
mcq single
+4 / 1
An ideal gas undergoes a process maintaining relation between pressure $(P)$ and $\operatorname{volume}(V)$ as $P=P_{\mathrm{o}}\left(1+\left(\frac{V_{\mathrm{o}}}{V}\right)^2\right)^{-1}$, where $P_{\mathrm{o}}$ and $V_{\mathrm{o}}$ are constants. If two samples $A$ and $B$ (two moles each) with initial volumes $V_{\mathrm{o}}$ and $3 V_{\mathrm{o}}$ respectively undergo above mentioned process and attain same pressure, then the difference at the temperatures of these samples, $T_B-T_A$ is $\_\_\_\_$ .
( $R=$ gas constant)
Q34.
mcq single
+4 / 1
One gas of $n_1$ mole of molecules at temperature $T_1$, volume $V_1$, and pressure $P_1$, and another gas of $n_2$ mole of molecules at temperature $T_2$, volume $V_2$, and pressure $P_2$, are mixed resulting in pressure $P$ and volume $V$ of the mixture. The temperature of the mixture is $\_\_\_\_$ .
Q35.
mcq single
+4 / 1
The increase in the pressure required to decrease the volume ( $\Delta V$ ) of water is $6.3 \times 10^7 \mathrm{~N} / \mathrm{m}^2$. The percentage decrease in the volume is $\_\_\_\_$ .
(Bulk modulus of water $=2.1 \times 10^9 \mathrm{~N} / \mathrm{m}^2$.)
Q36.
numerical
+4 / 1
The surface tension of a soap solution is $3.5 \times 10^{-2} \mathrm{~N} / \mathrm{m}$. The work required to increase the radius of a soap bubble from 1 cm to 2 cm is $\alpha \times 10^{-6} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
$$ (\pi=22 / 7) $$
Q37.
mcq single
+4 / 1
A string $A$ of length 0.314 m and Young's modulus $2 \times 10^{10} \mathrm{~N} / \mathrm{m}^2$ is connected to another string $B$ of length and Young's modulus both twice of those of $A$. This series combination of strings is then suspended from a rigid support and its free end is fixed to a load of mass 0.8 kg . The net change in length of the combination is $\_\_\_\_$ mm.
(radius of both the strings is 0.2 mm and acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2$ ) (Mass of both strings is to be neglected as compared to the mass of load)
Q38.
mcq single
+4 / 1
The graph shows variation of stopping potential $V_{\mathrm{o}}$ with the frequency $v$ of the incident radiation for three photosensitive metals $X_1, X_2$ and $X_3$. Which metal will give out electrons with greater kinetic energy, for the same wavelength of incident radiation?


Q39.
mcq single
+4 / 1
A parallel plate air capacitor is connected to a battery. The plates are pulled apart at uniform speed $v$. If $x$ is the separation between the plates at any instant, then the time rate of change of electrostatic energy of the capacitor is proportional to $x^\alpha$, where $\alpha$ is $\_\_\_\_$ .
Q40.
mcq single
+4 / 1
A rod of length 10 cm lies along the principle axis of a concave mirror of focal length 10 cm as shown in figure. The length of the image is $\_\_\_\_$ cm.


Q41.
mcq single
+4 / 1
An insulated wire is wound so that it forms a flat coil with $N=200$ turns. The radius of the innermost turn is $r_1=3 \mathrm{~cm}$, and of the outermost turn $r_2=6 \mathrm{~cm}$. If 20 mA current flows in it then the magnetic moment will be $\alpha \times 10^{-2} \mathrm{~A} . \mathrm{m}^2$. The value of $\alpha$ is $\_\_\_\_$ .
Q42.
mcq single
+4 / 1
In a screw gauge when the circular scale is given five complete rotations it moves linearly by 2.5 mm . If the circular scale has 100 divisions, the least count of screw gauge is $\_\_\_\_$ mm.
Q43.
mcq single
+4 / 1
Two nuclei of mass number 3 combine with another nucleus of mass number 4 to yield a nucleus of mass number 10. If the binding energy per nucleon for the mass numbers 3,4 and 10 are $5.6 \mathrm{MeV}, 7.4 \mathrm{MeV}$ and 6.1 MeV , respectively, then in the process, $\Delta \mathrm{Mc}^2=$ $\_\_\_\_$ MeV .
Q44.
mcq single
+4 / 1
The two projectiles are projected with the same initial velocities at the $15^{\circ}$ and $30^{\circ}$ with respect to the horizontal. The ratio of their ranges is $1: x$. The value of $x$ is
Q45.
mcq single
+4 / 1
The time taken by a block of mass $m$ to slide down from the highest point to the lowest point on a rough inclined plane is $50 \%$ more compared to the time taken by the same block on identical inclined smooth plane. Both inclined planes are at $45^{\circ}$ with the horizontal. The coefficient of kinetic friction between the rough inclined surface and block is $\_\_\_\_$
Q46.
mcq single
+4 / 1
The voltage and the current between $A$ and $B$ points shown in the circuit are $\_\_\_\_$ .


Q47.
mcq single
+4 / 1
A voltmeter with internal resistance of $x \Omega$ can be used to measure upto 20 V . In order to increase its measuring range to 30 V , the required modification is to $\_\_\_\_$ .
Q48.
numerical
+4 / 1
An inductor of 10 mH , capacitor of $0.1 \mu \mathrm{~F}$ and a resistor of $100 \Omega$ are connected in series across an $a . c$ power supply $220 \mathrm{~V}, 70 \mathrm{~Hz}$. The power factor of the given circuit is 0.5 . The difference in the inductive reactance and capacitance reactance is $\sqrt{3} \alpha \Omega$. The value of $\alpha$ is $\_\_\_\_$ .