JEE Main 2026 (Online) 5th April Evening Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Heating benzamide with bromine in an ethanolic solution of sodium hydroxide will give benzylamine.
**Statement II :** Nitration of aniline with $\mathrm{HNO}_3 / \mathrm{H}_2 \mathrm{SO}_4$ at 288 K produces $m$-nitroaniline in higher amount than $o$-nitroaniline ( pH adjusted).
In the light of the above statements, choose the correct answer from the options given below :
Q2.
mcq single
+4 / 1
A paper dipped in a dil. $\mathrm{H}_2 \mathrm{SO}_4$ solution of ' $X$ ' upon treatment with $\mathrm{SO}_2$ gas turns into green. The compound ' $X$ ' is :
Q3.
numerical
+4 / 1
For a reaction $\mathrm{A} \rightarrow \mathrm{P}$ at T K , the half life $\left(\mathrm{t}_{1 / 2}\right)$ is plotted as a function of initial concentration $[\mathrm{A}]_0$ of A as given below.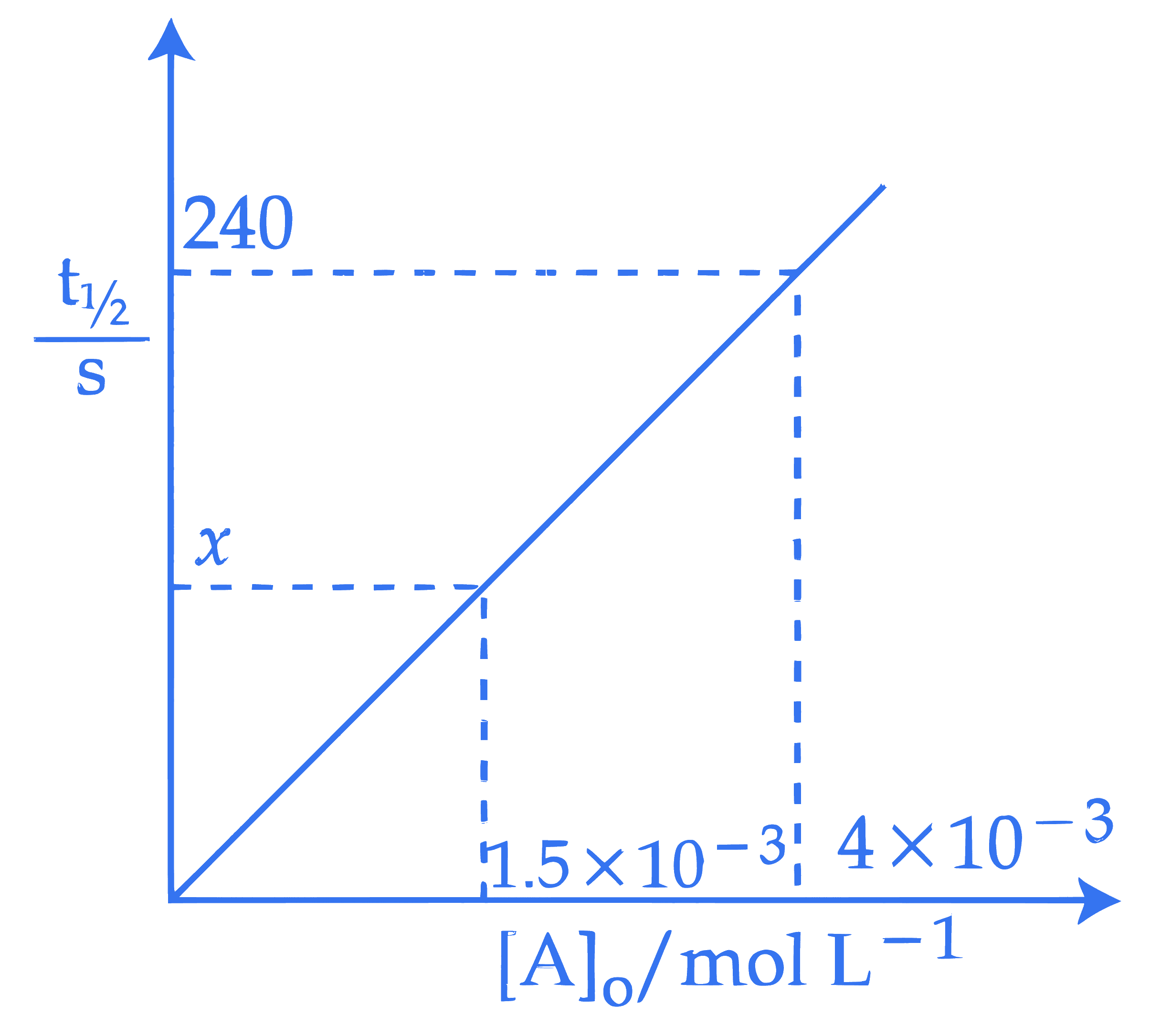 The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
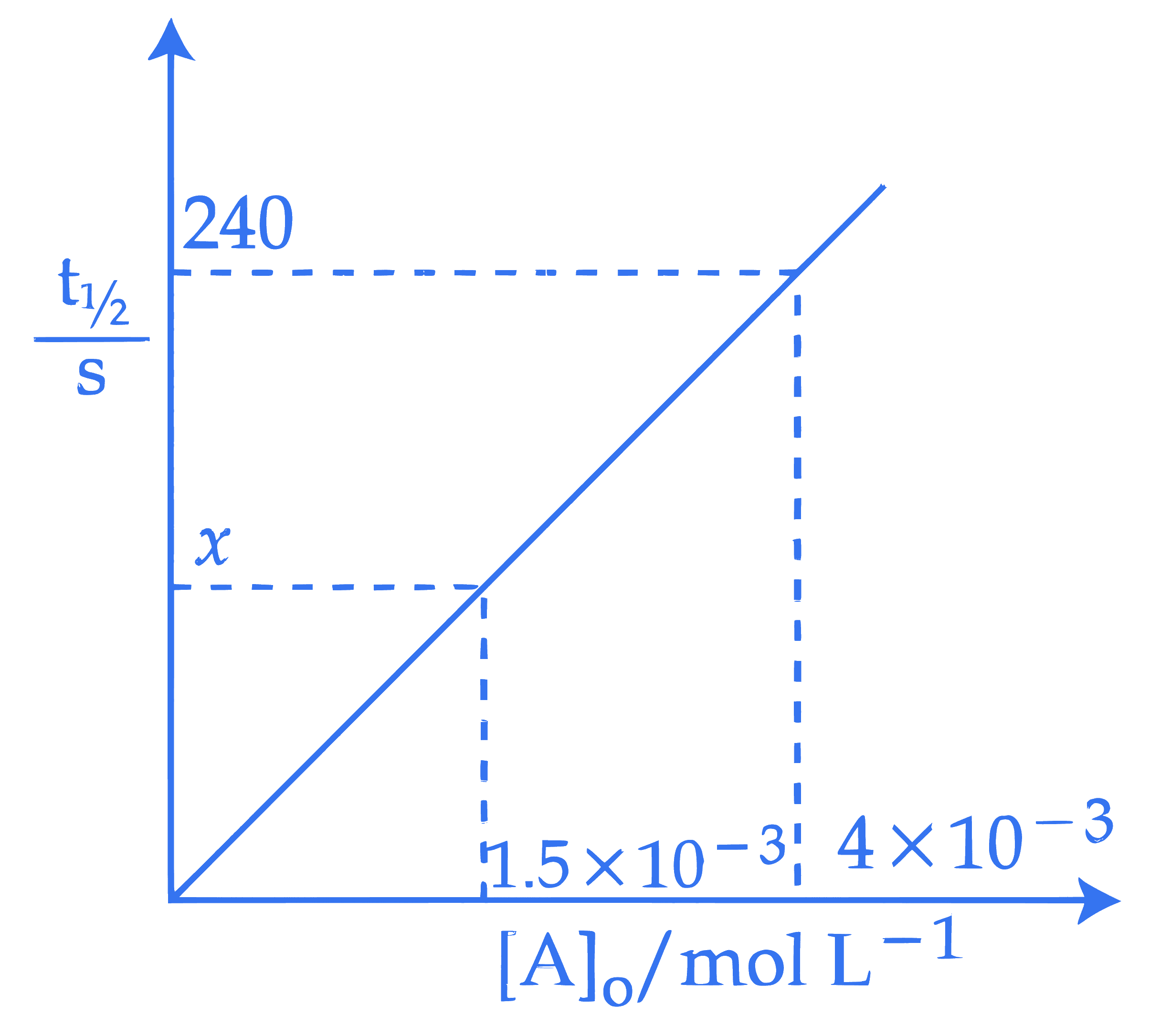 The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
Q4.
mcq single
+4 / 1
The covalent radii of atoms $A$ and $B$ are $r_A$ and $r_B$, respectively. The covalent bond length and total length of AB molecule are respectively
Q5.
numerical
+4 / 1
RMgI when treated with ice cold water liberated a gas which occupied $1.4 \mathrm{dm}^3 / \mathrm{g}$ at STP. The gas produced is further reacted with iodine in presence of $\mathrm{HIO}_3$ to give compound $(\mathrm{X})$. Compound $(\mathrm{X})$ in presence of Na and dry ether produced compound $(\mathrm{Y})$. Molar mass of compound $(\mathrm{Y})$ is $\_\_\_\_$ $\mathrm{g} \mathrm{mol}^{-1}$. (Nearest integer)
Q6.
numerical
+4 / 1
20 g hemoglobin in a 1 L aqueous solution $(\mathrm{A})$ at 300 K is separated from pure water by semi permeable membrane. At equilibrium the height of solution in a tube dipped in a solution (A) is found to be 80.0 mm higher than the tube dipped in water.
The molar mass of hemoglobin is $\_\_\_\_$ $\mathrm{kg} \mathrm{mol}^{-1}$. (Nearest integer)
(Given : $\mathrm{g}=10 \mathrm{~m} \mathrm{~s}^{-2}, \mathrm{R}=8.3 \mathrm{kPa} \mathrm{dm} \mathrm{K}^{-1} \mathrm{~mol}^{-1}$, density of solution $=1000 \mathrm{~kg} \mathrm{~m}^{-3}$ )
Q7.
mcq single
+4 / 1
What volume of hydrogen gas at STP would be liberated by action of 50 mL of $\mathrm{H}_2 \mathrm{SO}_4$ of $50 \%$ purity (density $=1.3 \mathrm{~g} \mathrm{~mL}^{-1}$ ) on 20 g of zinc?
Given : Molar mass of $\mathrm{H}, \mathrm{O}, \mathrm{S}, \mathrm{Zn}$ are 1, 16, 32, $65 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.
Q8.
mcq single
+4 / 1
The reaction $\mathrm{A}(\mathrm{g}) \rightleftharpoons \mathrm{B}(\mathrm{g})+\mathrm{C}(\mathrm{g})$ was initiated with the amount ' a ' of $\mathrm{A}(\mathrm{g})$. At equilibrium it is found that the amount of $\mathrm{A}(\mathrm{g})$ remaining is ( $\mathrm{a}-x$ ) at a total pressure of p .
The equilibrium constant Kp of the reaction can be calculated from the expression :
Q9.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of pairs among $\left[\mathrm{Al}_2 \mathrm{O}_3, \mathrm{Cr}_2 \mathrm{O}_3\right],\left[\mathrm{Cl}_2 \mathrm{O}_7, \mathrm{Mn}_2 \mathrm{O}_7\right],\left[\mathrm{Na}_2 \mathrm{O}, \mathrm{V}_2 \mathrm{O}_3\right]$ and $\left[\mathrm{CO}, \mathrm{N}_2 \mathrm{O}\right]$ that contain oxides of same nature (acidic, basic, neutral or amphoteric) is 4 .
**Statement II :** Among $\mathrm{Na}_2 \mathrm{O}, \mathrm{Al}_2 \mathrm{O}_3, \mathrm{CO}$ and $\mathrm{Cl}_2 \mathrm{O}_7$, the most basic and acidic oxides are $\mathrm{Na}_2 \mathrm{O}$ and $\mathrm{Cl}_2 \mathrm{O}_7$, respectively.
In the light of the above statements, choose the correct answer from the options given below :
Q10.
mcq single
+4 / 1
The correct order of molar heat capacities measured at 298 K and 1 bar is :
Q11.
mcq single
+4 / 1
Consider the following data for the reaction
$$ X_2(g)+Y_2(g) \rightleftharpoons 2 X Y(g) $$
at $600 \mathrm{~K}^{\circ}$. The $\Delta_{\mathrm{r}} \mathrm{G}^{\ominus}$ (in $\mathrm{kJ} \mathrm{mol}^{-1}$ ) for the reaction is :
$$ \begin{array}{|c|c|c|} \hline \text { Compound } & \Delta_f \mathrm{H}_{600 \mathrm{~K}}^{\ominus}\left(\mathrm{kJ} \mathrm{~mol}^{-1}\right) & \mathrm{S}^{\ominus}{ }_{600 \mathrm{~K}}\left(\mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}\right) \\ \hline \mathrm{XY}(\mathrm{~g}) & 42 & 200 \\ \hline \mathrm{X}_2(\mathrm{~g}) & 8 & 140 \\ \hline \mathrm{Y}_2(\mathrm{~g}) & 80 & 250 \\ \hline \end{array} $$
Q12.
mcq single
+4 / 1
Given below are two statements :
Statement I: Aluminium upon reaction with NaOH forms $\left[\mathrm{Al}(\mathrm{OH})_6\right]^{3-}$ ion.
Statement II : The geometry of $\mathrm{ICl}_4^{-}, \mathrm{ClO}_3^{-}$and $\mathrm{IBr}_2^{-}$is square planar, pyramidal and linear respectively.
In the light of the above statements, choose the correct answer from the options given below :
Q13.
mcq single
+4 / 1
Identify the correct statements from the following
A. $\left[\mathrm{Fe}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}$ is the most stable complex among $\left[\mathrm{Fe}(\mathrm{OH})_6\right]^{3-},\left[\mathrm{Fe}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}$ and $\left[\mathrm{Fe}(\mathrm{SCN})_6\right]^{3-}$
B. The stability of $\left[\mathrm{Cu}\left(\mathrm{NH}_3\right)_4\right]^{2+}$ is greater than that of $\left[\mathrm{Cu}(\mathrm{en})_2\right]^{2+}$
C. The hybridization of Fe in $\mathrm{K}_4\left[\mathrm{Fe}(\mathrm{CN})_6\right]$ is $\mathrm{d}^2 \mathrm{sp}^3$
D. $\left[\mathrm{Fe}\left(\mathrm{NO}_2\right)_3 \mathrm{Cl}_3\right]^{3-}$ exhibits linkage isomerism
E. $\quad \mathrm{NO}_2^{-}$and $\mathrm{SCN}^{-}$ligands are NOT ambidentate ligands
Choose the correct answer from the options given below :
Q14.
mcq single
+4 / 1
Given below are two statements :
Statement I: Presence of large number of unpaired electrons in transition metal atoms results in higher enthalpies of their atomisation.
Statement II: $\quad \mathrm{d}_{x y}=\mathrm{d}_{x z}=\mathrm{d}_{y z}<\mathrm{d}_{x^2-y^2}=\mathrm{d}_{z^2}$ and $\mathrm{d}_{x^2-y^2}=\mathrm{d}_{z^2}<\mathrm{d}_{x y}=\mathrm{d}_{x z}=\mathrm{d}_{y z}$ are the d-orbital splittings in $\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+}$ and $\left[\mathrm{Ni}(\mathrm{Cl})_4\right]^{2-}$ complex ions respectively.
In the light of the above statements, choose the correct answer from the options given below:
Q15.
numerical
+4 / 1
The total number of unpaired electrons present in the $d^3, d^4$ (low spin) $d^5$ (high spin), $\mathrm{d}^6$ (high spin) and $\mathrm{d}^7$ (low spin) octahedral complex systems is $\_\_\_\_$ .
Q16.
mcq single
+4 / 1
$$ \text { Complete the following reaction sequence and give the name of major product ' } \mathrm{P} \text { '. } $$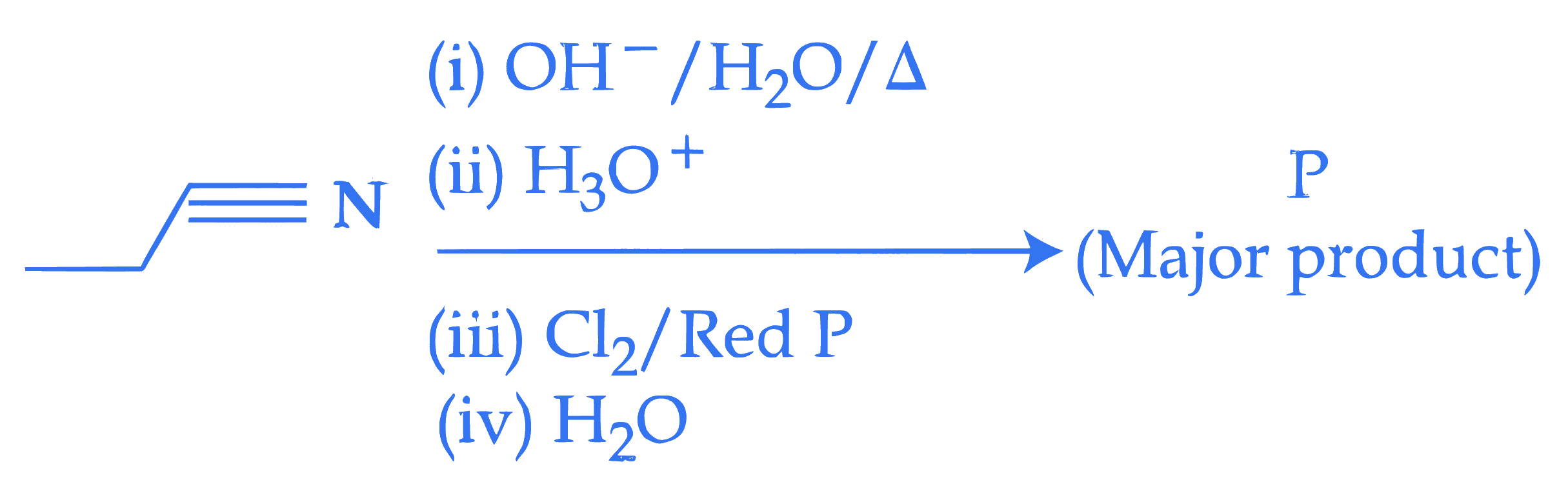
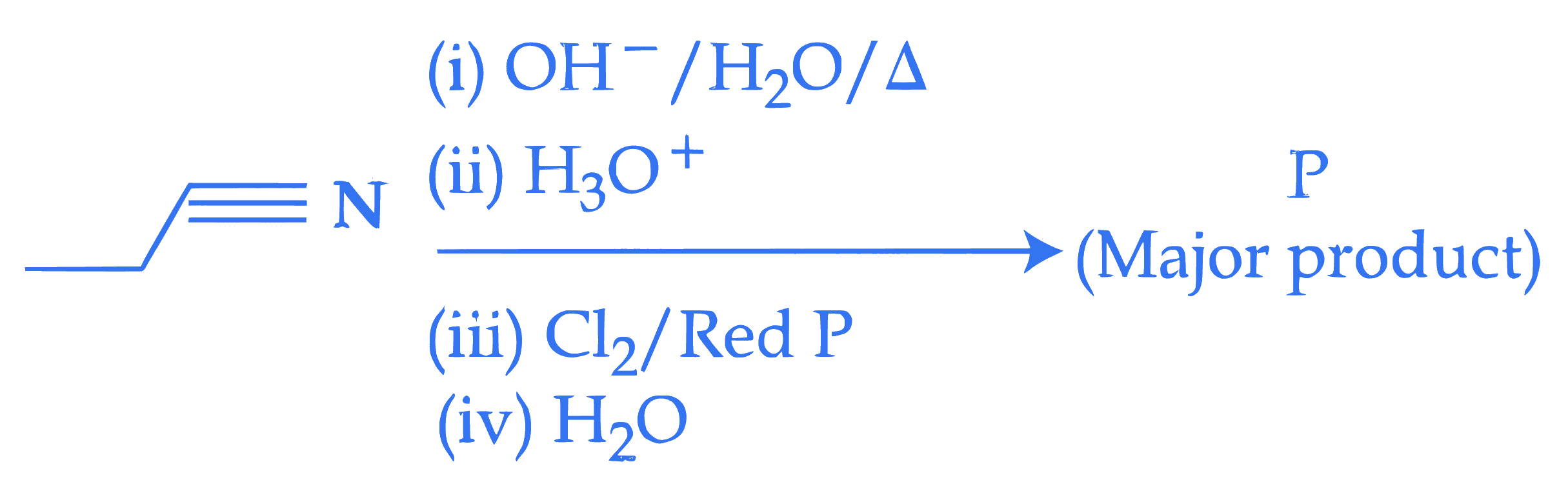
Q17.
mcq single
+4 / 1
Given below are two statements:
**Statement I**: The condensation reaction between $\mathrm{CH_3-CH=O}$ and 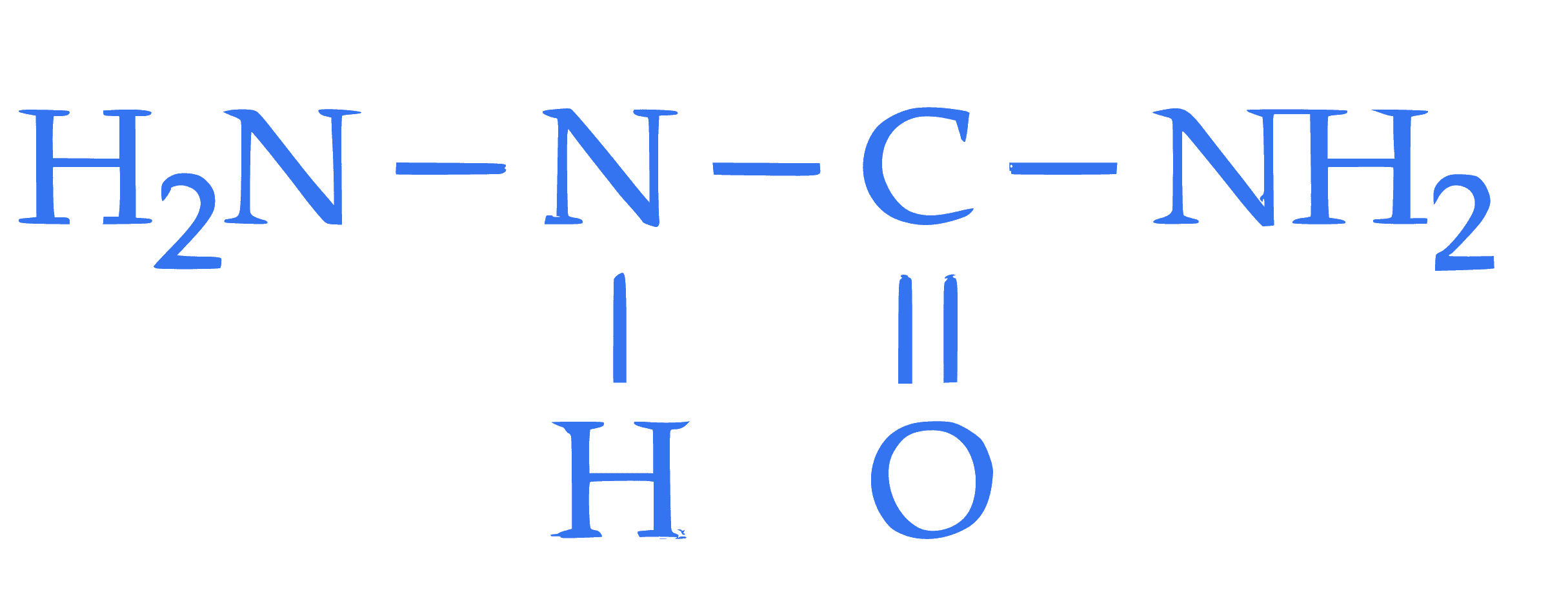 **under optimum pH will produce
**under optimum pH will produce 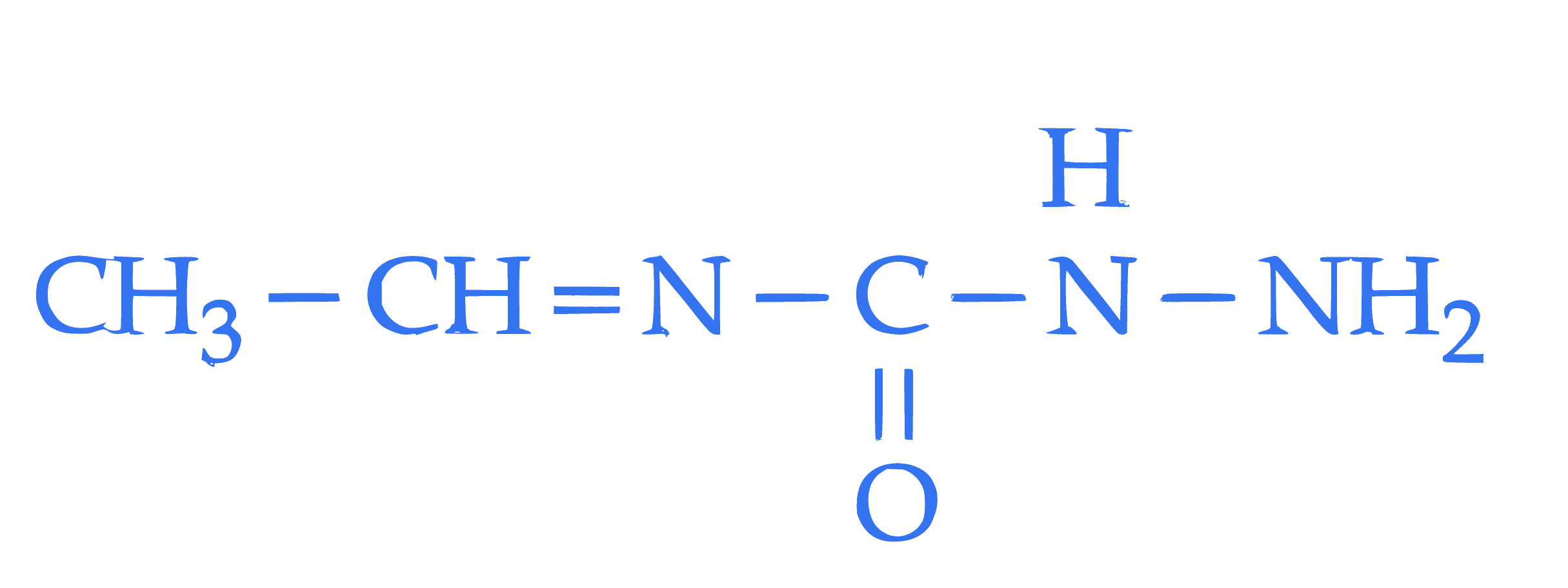 Statement II**: The molecule,
Statement II**: The molecule, 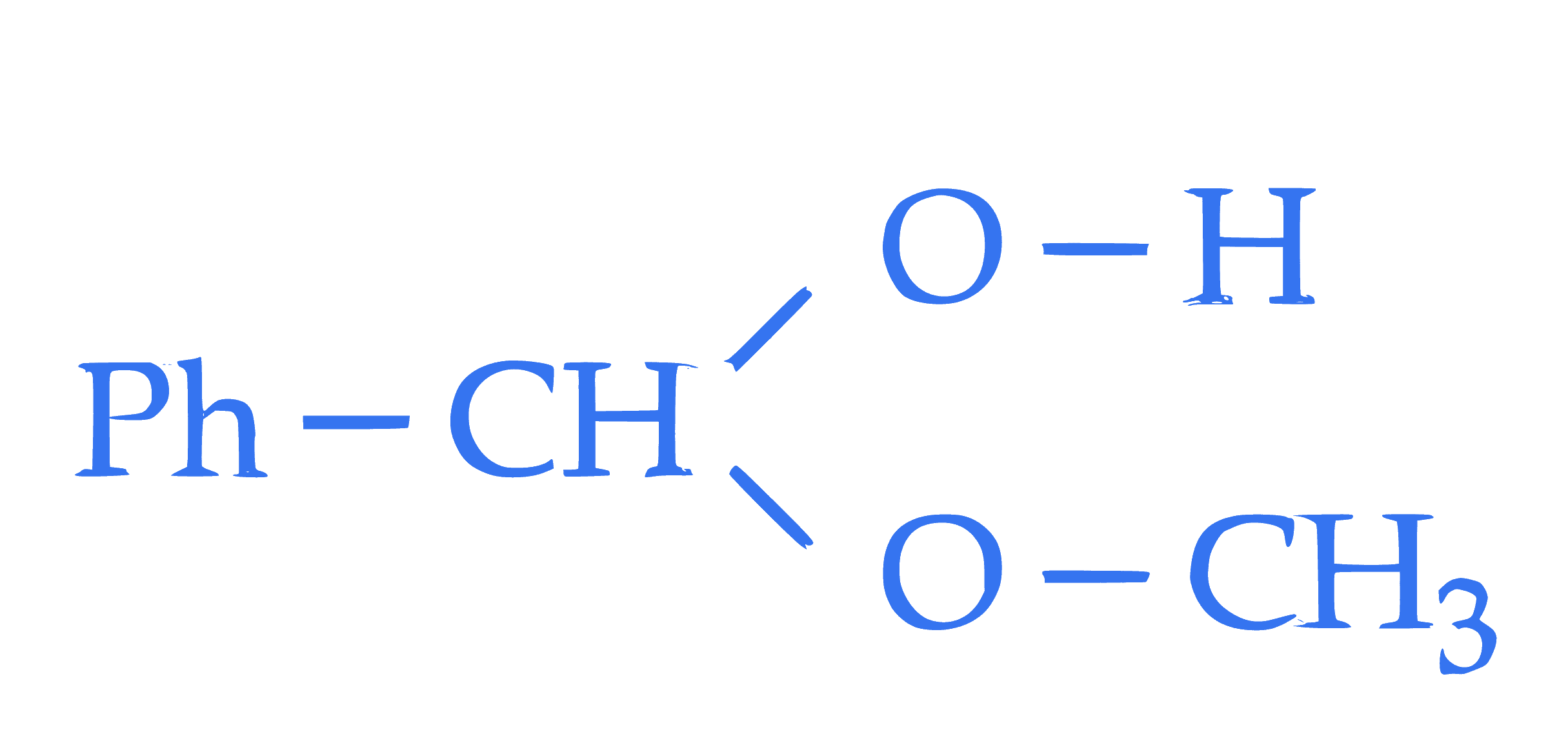 will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
 **under optimum pH will produce
**under optimum pH will produce 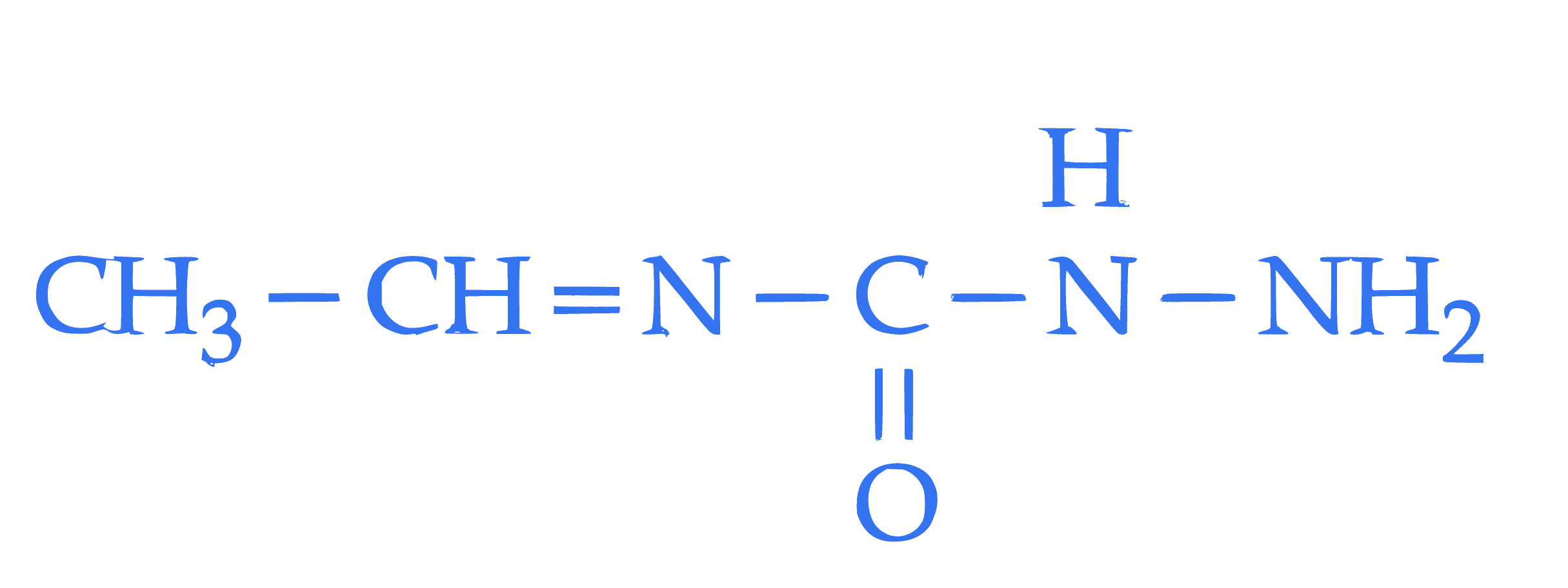 Statement II**: The molecule,
Statement II**: The molecule,  will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
Q18.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Purification technique
List - II
Used to separate
A.
Simple distillation
I.
Steam volatile compound
B.
Fractional distillation
II.
Two liquids with large difference in boiling points
C.
Steam distillation
III.
Liquid decomposing at its boiling point
D.
Distillation under reduced pressure
IV.
Two liquids with close boiling points
Choose the correct answer from the options given below :
Q19.
mcq single
+4 / 1
IUPAC name of the some alkenes are given below.
Find out the correct stability order.
A. 2-Methylbut-2-ene
B. cis-But-2-ene
C. 2,3-Dimethylbut-2-ene
D. Prop-1-ene
Choose the correct answer from the options given below :
Q20.
mcq single
+4 / 1
Identify the correct IUPAC name of hydrocarbon $(x)$ containing three primary carbon atoms and with molar mass $72 \mathrm{~g} \mathrm{~mol}^{-1}$.
Q21.
mcq single
+4 / 1
Identify the incorrect statement about tertiary structure of proteins.
Q22.
mcq single
+4 / 1
$$ \text { Given below are two statements : } $$
$$ \text { Statement I : } $$ 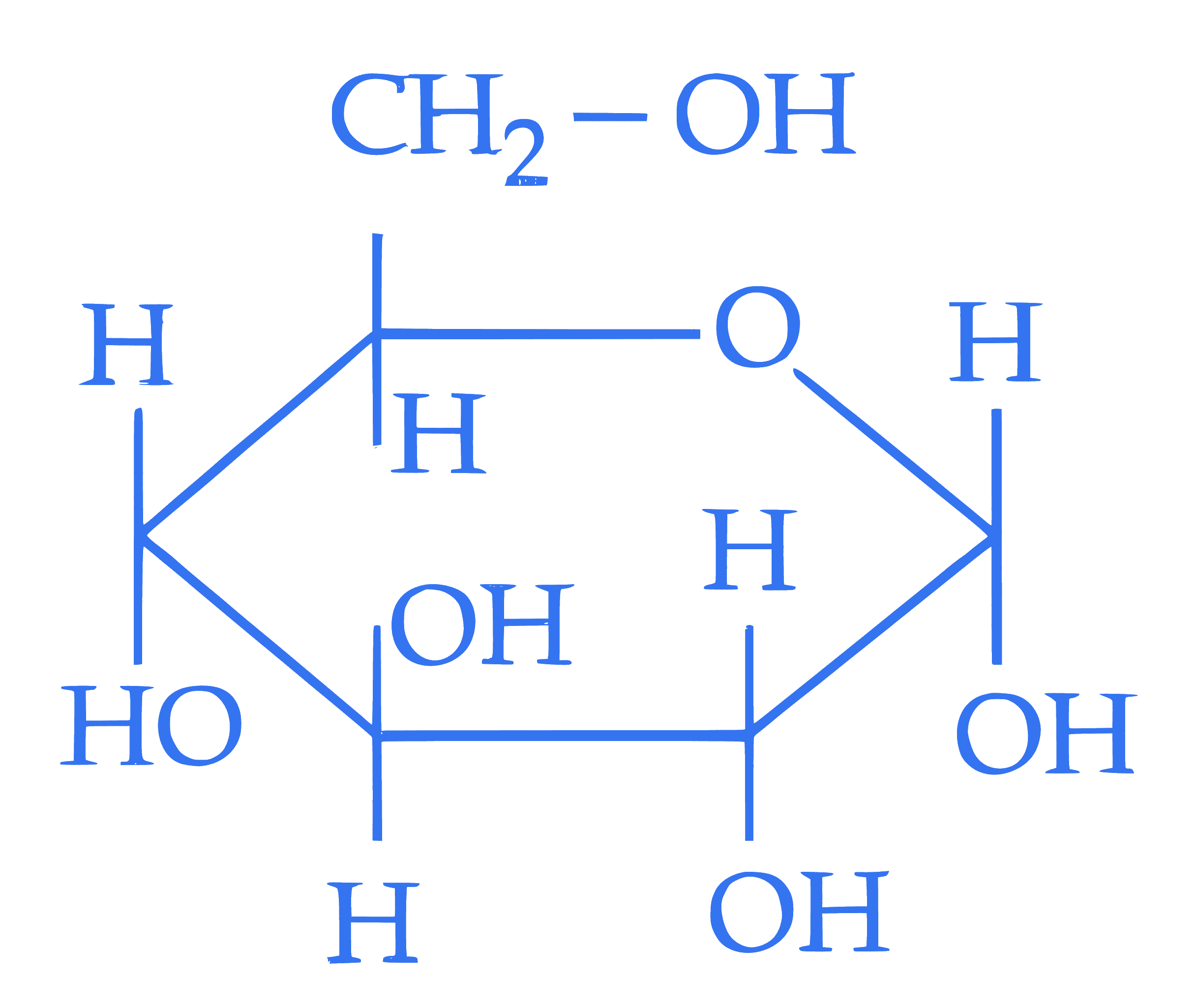 and
and 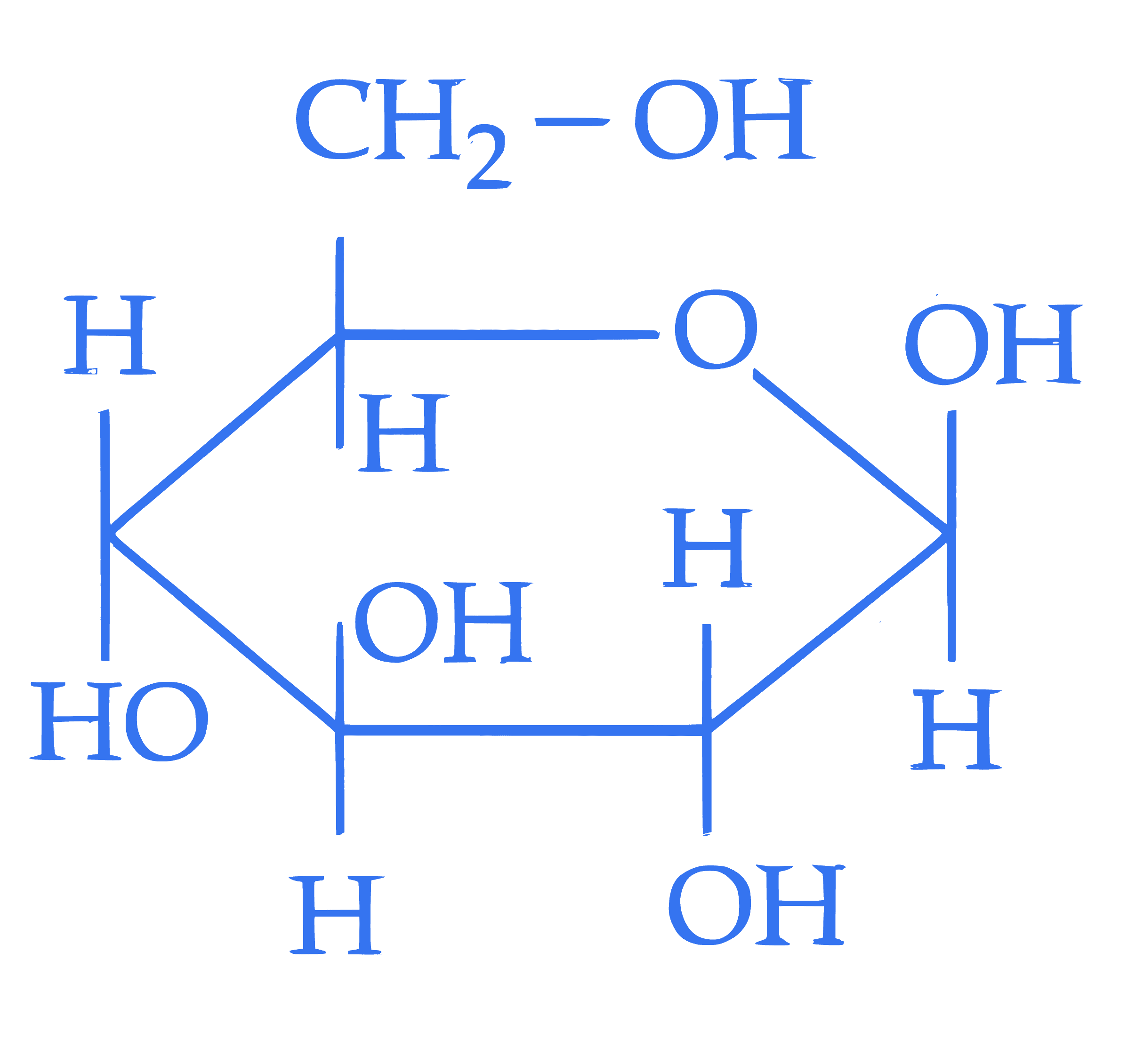 are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
 and
and  are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
Q23.
numerical
+4 / 1
At 298 K , the molar conductivity of $x \%(\mathrm{w} / \mathrm{w}) \mathrm{MX}$ solution (aqueous) is $123.5 \mathrm{~S} \mathrm{~cm}^2 \mathrm{~mol}^{-1}$. The conductance of same solution is $1.9 \times 10^{-3} \mathrm{~S}$. The value of $x$ is $\_\_\_\_$ $\times 10^{-2}$.
(Given: cell constant $=1.3 \mathrm{~cm}^{-1}$; molar mass of MX is $75 \mathrm{~g} \mathrm{~mol}^{-1}$, density of aqueous solution of MX at 298 K is $1.0 \mathrm{~g} \mathrm{~mL}^{-1}$ )
Q24.
mcq single
+4 / 1
One half cell in a voltaic cell is constructed by dipping silver rod in $\mathrm{AgNO}_3$ solution of unknown concentration, other half cell is Zn rod dipped in 1 molar solution of $\mathrm{ZnSO}_4$.
A voltage of 1.60 V is measured at 298 K for this cell. What is the concentration of $\mathrm{Ag}^{+}$ions used in terms of $\log x\left(x=\left[\mathrm{Ag}^{+}\right]\right)$?
$$ \mathrm{E}_{\mathrm{Zn}^{2+} / \mathrm{Zn}}^{\ominus}=-0.76 \mathrm{~V}, \quad \mathrm{E}_{\mathrm{Ag}^{+} / \mathrm{Ag}}^{\ominus}=+0.80 \mathrm{~V}, \frac{2.303 \mathrm{RT}}{\mathrm{~F}}=0.059 \mathrm{~V} $$
Q25.
mcq single
+4 / 1
Which of the following statement(s) is/are true ?
A. If two orbitals have the same value of ( $\mathrm{n}+l$ ), the orbital with lower value of n will have lower energy.
B. Energies of the orbitals in the same subshell increase with increase in atomic number.
C. The size of $2 \mathrm{p}_x$ orbital is less than the size of $3 \mathrm{p}_x$ orbital.
D. Among 5f, 6s, 4d, 5p and 5d orbitals, none of the orbitals have 2 radial nodes.
Choose the correct answer from the options given below :
Q26.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Heating benzamide with bromine in an ethanolic solution of sodium hydroxide will give benzylamine.
**Statement II :** Nitration of aniline with $\mathrm{HNO}_3 / \mathrm{H}_2 \mathrm{SO}_4$ at 288 K produces $m$-nitroaniline in higher amount than $o$-nitroaniline ( pH adjusted).
In the light of the above statements, choose the correct answer from the options given below :
Q27.
mcq single
+4 / 1
A paper dipped in a dil. $\mathrm{H}_2 \mathrm{SO}_4$ solution of ' $X$ ' upon treatment with $\mathrm{SO}_2$ gas turns into green. The compound ' $X$ ' is :
Q28.
numerical
+4 / 1
For a reaction $\mathrm{A} \rightarrow \mathrm{P}$ at T K , the half life $\left(\mathrm{t}_{1 / 2}\right)$ is plotted as a function of initial concentration $[\mathrm{A}]_0$ of A as given below.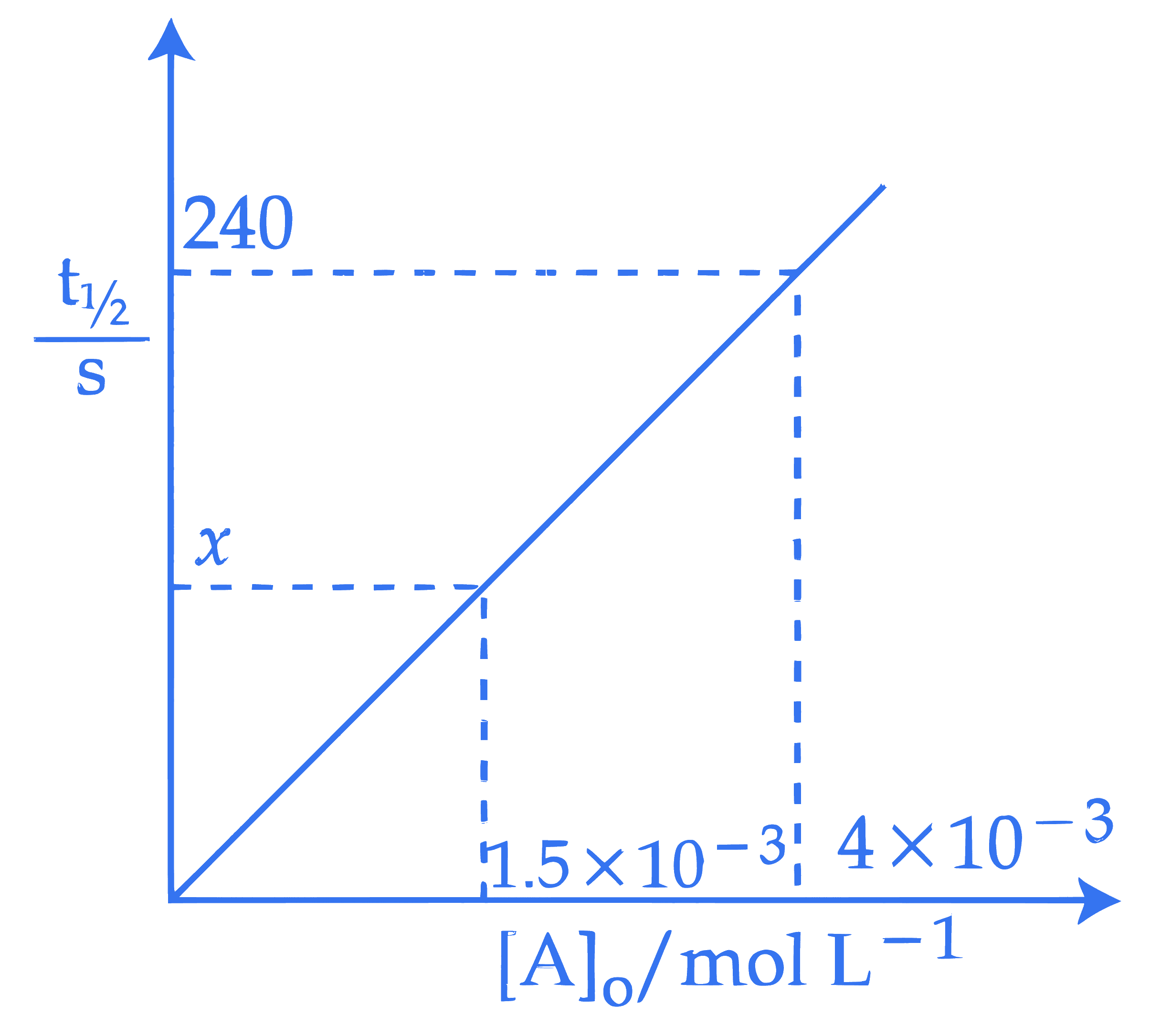 The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
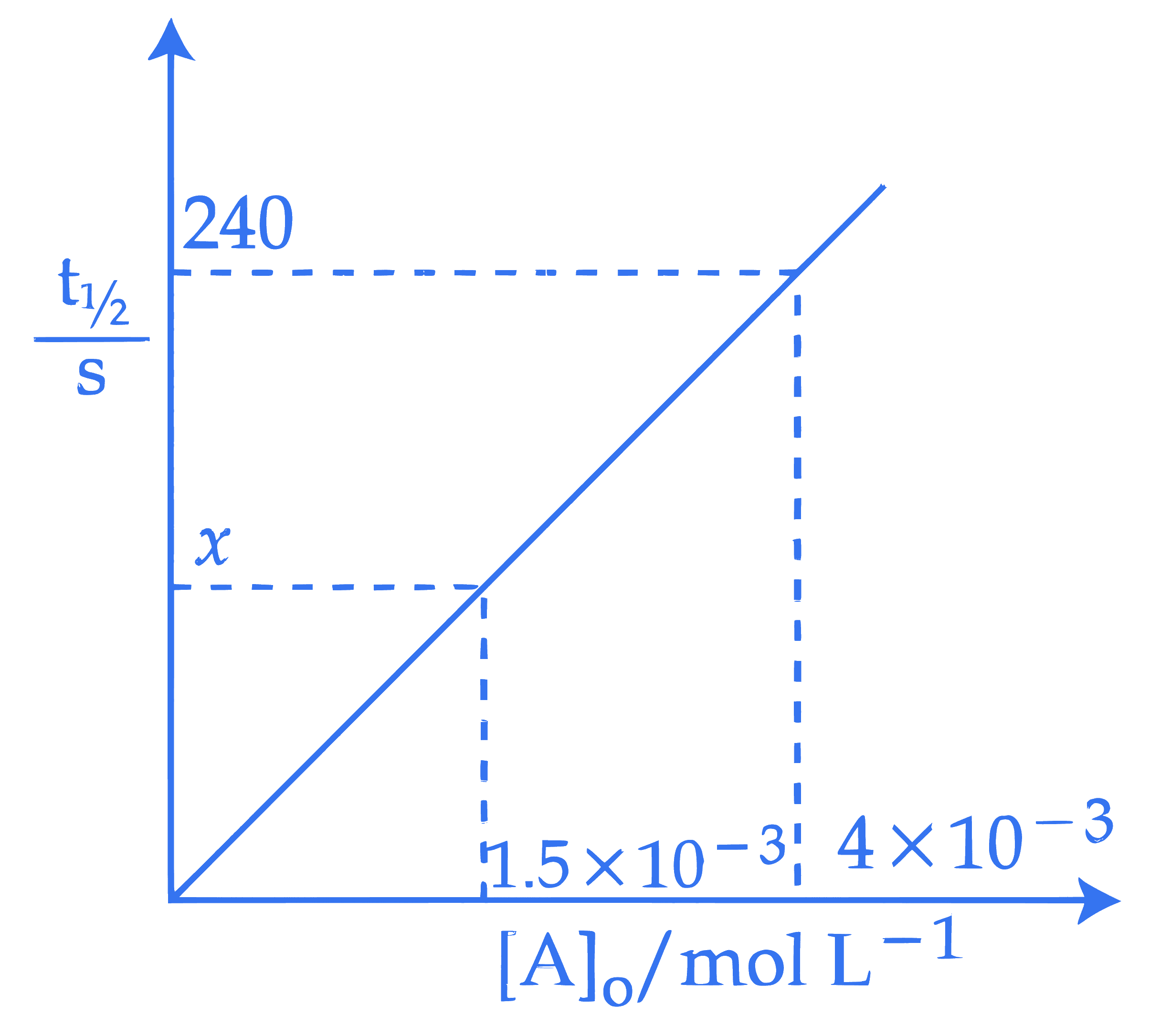 The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
The value of $x$ in the given figure is $\_\_\_\_$ s (Nearest integer)
Q29.
mcq single
+4 / 1
The covalent radii of atoms $A$ and $B$ are $r_A$ and $r_B$, respectively. The covalent bond length and total length of AB molecule are respectively
Q30.
numerical
+4 / 1
RMgI when treated with ice cold water liberated a gas which occupied $1.4 \mathrm{dm}^3 / \mathrm{g}$ at STP. The gas produced is further reacted with iodine in presence of $\mathrm{HIO}_3$ to give compound $(\mathrm{X})$. Compound $(\mathrm{X})$ in presence of Na and dry ether produced compound $(\mathrm{Y})$. Molar mass of compound $(\mathrm{Y})$ is $\_\_\_\_$ $\mathrm{g} \mathrm{mol}^{-1}$. (Nearest integer)
Q31.
numerical
+4 / 1
20 g hemoglobin in a 1 L aqueous solution $(\mathrm{A})$ at 300 K is separated from pure water by semi permeable membrane. At equilibrium the height of solution in a tube dipped in a solution (A) is found to be 80.0 mm higher than the tube dipped in water.
The molar mass of hemoglobin is $\_\_\_\_$ $\mathrm{kg} \mathrm{mol}^{-1}$. (Nearest integer)
(Given : $\mathrm{g}=10 \mathrm{~m} \mathrm{~s}^{-2}, \mathrm{R}=8.3 \mathrm{kPa} \mathrm{dm} \mathrm{K}^{-1} \mathrm{~mol}^{-1}$, density of solution $=1000 \mathrm{~kg} \mathrm{~m}^{-3}$ )
Q32.
mcq single
+4 / 1
What volume of hydrogen gas at STP would be liberated by action of 50 mL of $\mathrm{H}_2 \mathrm{SO}_4$ of $50 \%$ purity (density $=1.3 \mathrm{~g} \mathrm{~mL}^{-1}$ ) on 20 g of zinc?
Given : Molar mass of $\mathrm{H}, \mathrm{O}, \mathrm{S}, \mathrm{Zn}$ are 1, 16, 32, $65 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.
Q33.
mcq single
+4 / 1
The reaction $\mathrm{A}(\mathrm{g}) \rightleftharpoons \mathrm{B}(\mathrm{g})+\mathrm{C}(\mathrm{g})$ was initiated with the amount ' a ' of $\mathrm{A}(\mathrm{g})$. At equilibrium it is found that the amount of $\mathrm{A}(\mathrm{g})$ remaining is ( $\mathrm{a}-x$ ) at a total pressure of p .
The equilibrium constant Kp of the reaction can be calculated from the expression :
Q34.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of pairs among $\left[\mathrm{Al}_2 \mathrm{O}_3, \mathrm{Cr}_2 \mathrm{O}_3\right],\left[\mathrm{Cl}_2 \mathrm{O}_7, \mathrm{Mn}_2 \mathrm{O}_7\right],\left[\mathrm{Na}_2 \mathrm{O}, \mathrm{V}_2 \mathrm{O}_3\right]$ and $\left[\mathrm{CO}, \mathrm{N}_2 \mathrm{O}\right]$ that contain oxides of same nature (acidic, basic, neutral or amphoteric) is 4 .
**Statement II :** Among $\mathrm{Na}_2 \mathrm{O}, \mathrm{Al}_2 \mathrm{O}_3, \mathrm{CO}$ and $\mathrm{Cl}_2 \mathrm{O}_7$, the most basic and acidic oxides are $\mathrm{Na}_2 \mathrm{O}$ and $\mathrm{Cl}_2 \mathrm{O}_7$, respectively.
In the light of the above statements, choose the correct answer from the options given below :
Q35.
mcq single
+4 / 1
The correct order of molar heat capacities measured at 298 K and 1 bar is :
Q36.
mcq single
+4 / 1
Consider the following data for the reaction
$$ X_2(g)+Y_2(g) \rightleftharpoons 2 X Y(g) $$
at $600 \mathrm{~K}^{\circ}$. The $\Delta_{\mathrm{r}} \mathrm{G}^{\ominus}$ (in $\mathrm{kJ} \mathrm{mol}^{-1}$ ) for the reaction is :
$$ \begin{array}{|c|c|c|} \hline \text { Compound } & \Delta_f \mathrm{H}_{600 \mathrm{~K}}^{\ominus}\left(\mathrm{kJ} \mathrm{~mol}^{-1}\right) & \mathrm{S}^{\ominus}{ }_{600 \mathrm{~K}}\left(\mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}\right) \\ \hline \mathrm{XY}(\mathrm{~g}) & 42 & 200 \\ \hline \mathrm{X}_2(\mathrm{~g}) & 8 & 140 \\ \hline \mathrm{Y}_2(\mathrm{~g}) & 80 & 250 \\ \hline \end{array} $$
Q37.
mcq single
+4 / 1
Given below are two statements :
Statement I: Aluminium upon reaction with NaOH forms $\left[\mathrm{Al}(\mathrm{OH})_6\right]^{3-}$ ion.
Statement II : The geometry of $\mathrm{ICl}_4^{-}, \mathrm{ClO}_3^{-}$and $\mathrm{IBr}_2^{-}$is square planar, pyramidal and linear respectively.
In the light of the above statements, choose the correct answer from the options given below :
Q38.
mcq single
+4 / 1
Identify the correct statements from the following
A. $\left[\mathrm{Fe}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}$ is the most stable complex among $\left[\mathrm{Fe}(\mathrm{OH})_6\right]^{3-},\left[\mathrm{Fe}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}$ and $\left[\mathrm{Fe}(\mathrm{SCN})_6\right]^{3-}$
B. The stability of $\left[\mathrm{Cu}\left(\mathrm{NH}_3\right)_4\right]^{2+}$ is greater than that of $\left[\mathrm{Cu}(\mathrm{en})_2\right]^{2+}$
C. The hybridization of Fe in $\mathrm{K}_4\left[\mathrm{Fe}(\mathrm{CN})_6\right]$ is $\mathrm{d}^2 \mathrm{sp}^3$
D. $\left[\mathrm{Fe}\left(\mathrm{NO}_2\right)_3 \mathrm{Cl}_3\right]^{3-}$ exhibits linkage isomerism
E. $\quad \mathrm{NO}_2^{-}$and $\mathrm{SCN}^{-}$ligands are NOT ambidentate ligands
Choose the correct answer from the options given below :
Q39.
mcq single
+4 / 1
Given below are two statements :
Statement I: Presence of large number of unpaired electrons in transition metal atoms results in higher enthalpies of their atomisation.
Statement II: $\quad \mathrm{d}_{x y}=\mathrm{d}_{x z}=\mathrm{d}_{y z}<\mathrm{d}_{x^2-y^2}=\mathrm{d}_{z^2}$ and $\mathrm{d}_{x^2-y^2}=\mathrm{d}_{z^2}<\mathrm{d}_{x y}=\mathrm{d}_{x z}=\mathrm{d}_{y z}$ are the d-orbital splittings in $\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+}$ and $\left[\mathrm{Ni}(\mathrm{Cl})_4\right]^{2-}$ complex ions respectively.
In the light of the above statements, choose the correct answer from the options given below:
Q40.
numerical
+4 / 1
The total number of unpaired electrons present in the $d^3, d^4$ (low spin) $d^5$ (high spin), $\mathrm{d}^6$ (high spin) and $\mathrm{d}^7$ (low spin) octahedral complex systems is $\_\_\_\_$ .
Q41.
mcq single
+4 / 1
$$ \text { Complete the following reaction sequence and give the name of major product ' } \mathrm{P} \text { '. } $$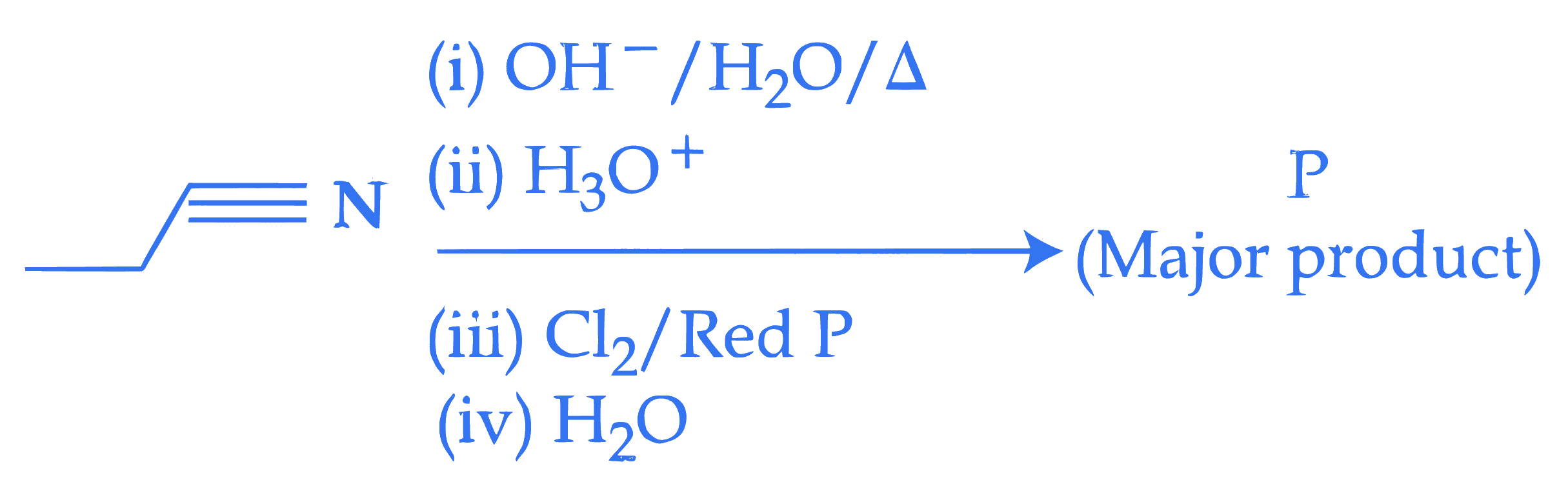
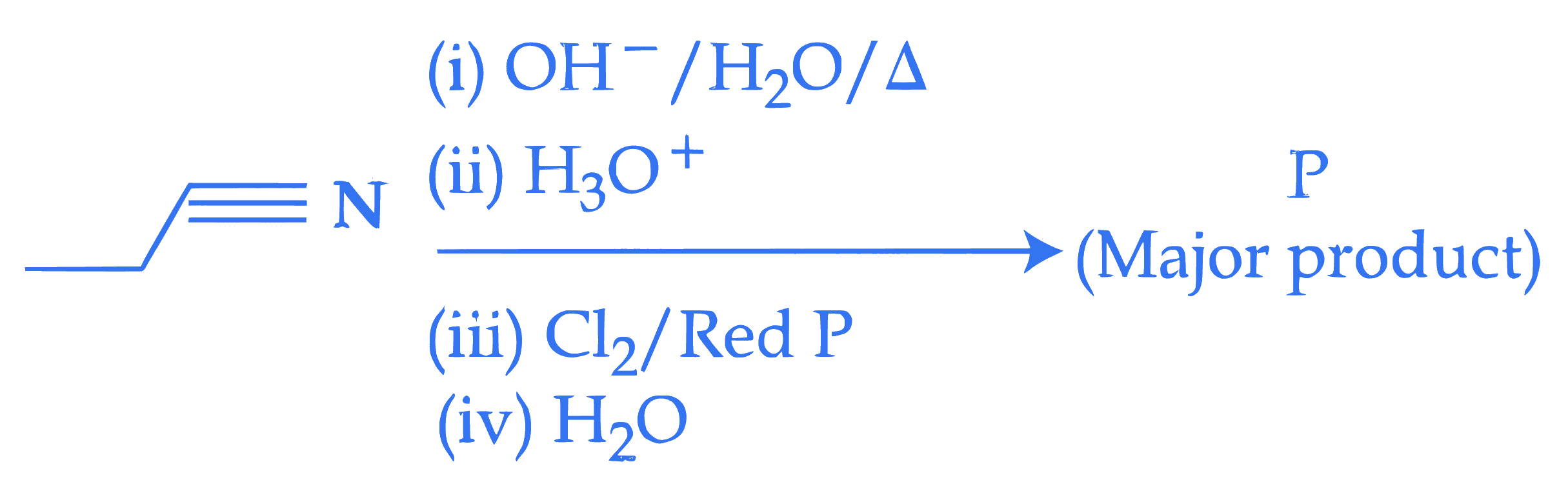
Q42.
mcq single
+4 / 1
Given below are two statements:
**Statement I**: The condensation reaction between $\mathrm{CH_3-CH=O}$ and  **under optimum pH will produce
**under optimum pH will produce 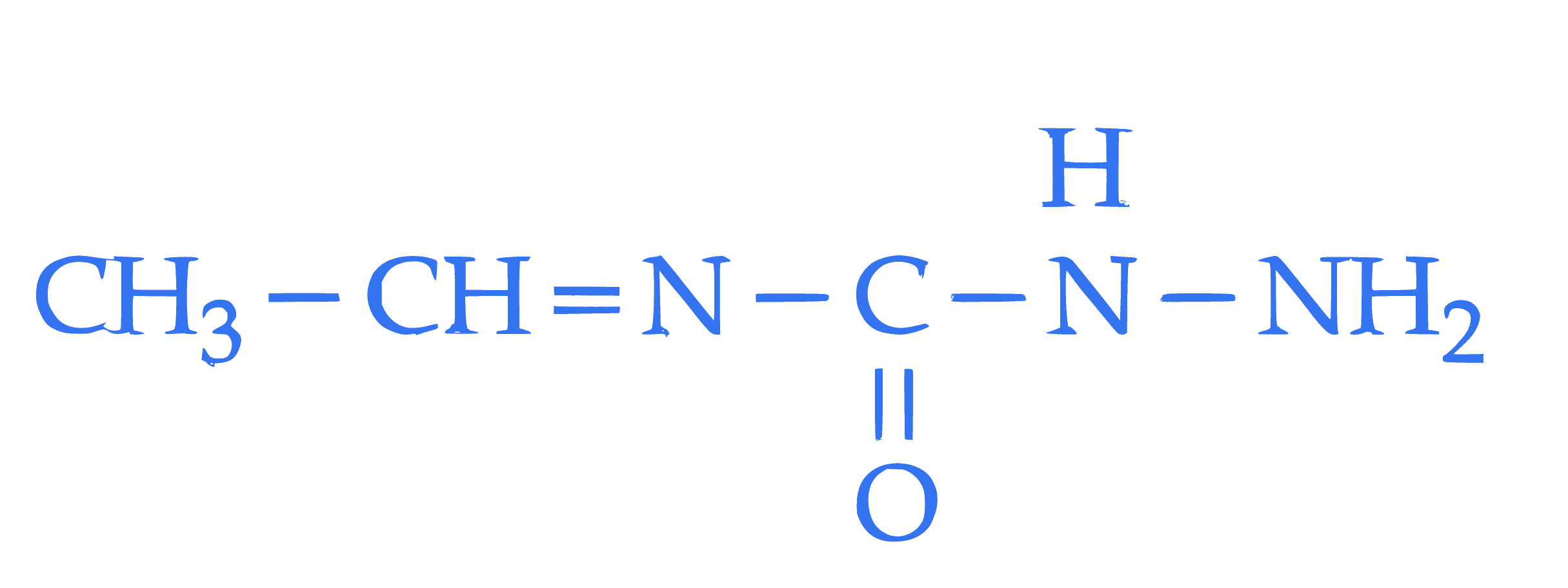 Statement II**: The molecule,
Statement II**: The molecule,  will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
 **under optimum pH will produce
**under optimum pH will produce 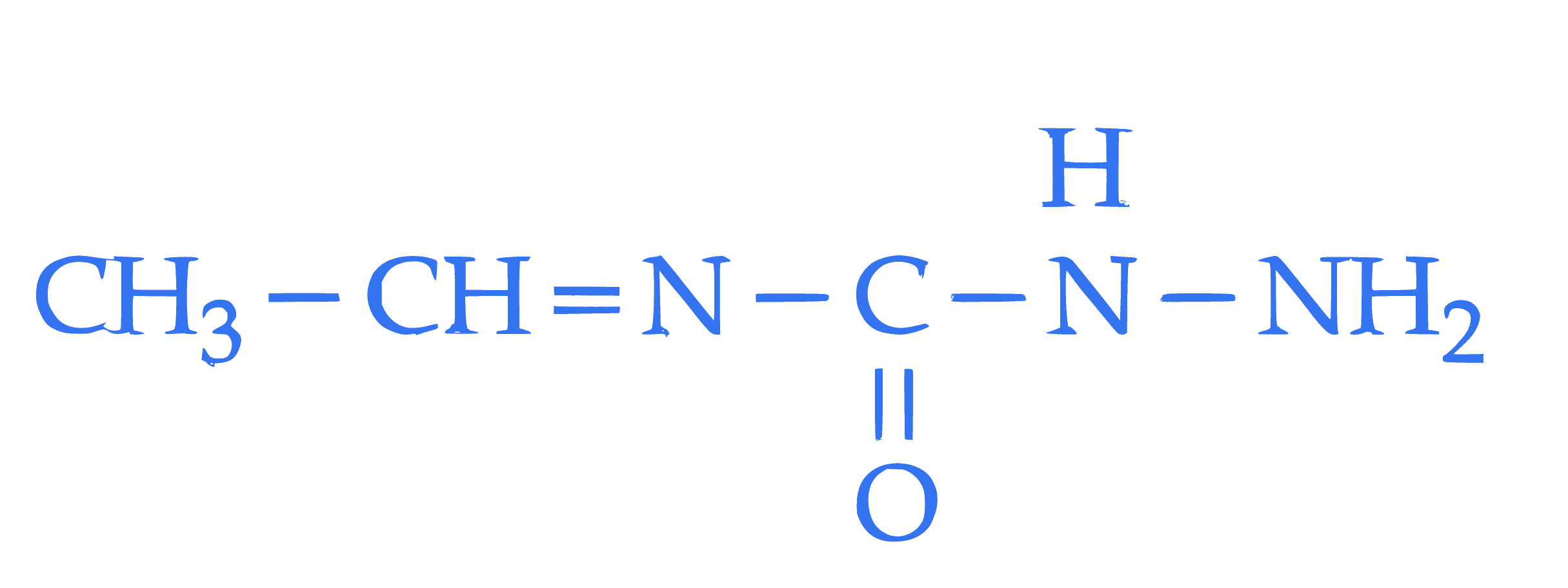 Statement II**: The molecule,
Statement II**: The molecule,  will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
will generate $\mathrm{Ph-CH=O}$ in the presence of dilute acid.
In the light of the above statements, choose the **correct** answer from the options given below:
Q43.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Purification technique
List - II
Used to separate
A.
Simple distillation
I.
Steam volatile compound
B.
Fractional distillation
II.
Two liquids with large difference in boiling points
C.
Steam distillation
III.
Liquid decomposing at its boiling point
D.
Distillation under reduced pressure
IV.
Two liquids with close boiling points
Choose the correct answer from the options given below :
Q44.
mcq single
+4 / 1
IUPAC name of the some alkenes are given below.
Find out the correct stability order.
A. 2-Methylbut-2-ene
B. cis-But-2-ene
C. 2,3-Dimethylbut-2-ene
D. Prop-1-ene
Choose the correct answer from the options given below :
Q45.
mcq single
+4 / 1
Identify the correct IUPAC name of hydrocarbon $(x)$ containing three primary carbon atoms and with molar mass $72 \mathrm{~g} \mathrm{~mol}^{-1}$.
Q46.
mcq single
+4 / 1
Identify the incorrect statement about tertiary structure of proteins.
Q47.
mcq single
+4 / 1
$$ \text { Given below are two statements : } $$
$$ \text { Statement I : } $$  and
and  are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
 and
and  are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
are two anomers of D-(+)-glucode.
Statement II : The open chain forms of D-glucose and D- fructose contain three similar chiral carbons at $\mathrm{C}_3, \mathrm{C}_4$ and $\mathrm{C}_5$.
In the light of the above statements, choose the correct answer from the options given below :
Q48.
numerical
+4 / 1
At 298 K , the molar conductivity of $x \%(\mathrm{w} / \mathrm{w}) \mathrm{MX}$ solution (aqueous) is $123.5 \mathrm{~S} \mathrm{~cm}^2 \mathrm{~mol}^{-1}$. The conductance of same solution is $1.9 \times 10^{-3} \mathrm{~S}$. The value of $x$ is $\_\_\_\_$ $\times 10^{-2}$.
(Given: cell constant $=1.3 \mathrm{~cm}^{-1}$; molar mass of MX is $75 \mathrm{~g} \mathrm{~mol}^{-1}$, density of aqueous solution of MX at 298 K is $1.0 \mathrm{~g} \mathrm{~mL}^{-1}$ )
Q49.
mcq single
+4 / 1
One half cell in a voltaic cell is constructed by dipping silver rod in $\mathrm{AgNO}_3$ solution of unknown concentration, other half cell is Zn rod dipped in 1 molar solution of $\mathrm{ZnSO}_4$.
A voltage of 1.60 V is measured at 298 K for this cell. What is the concentration of $\mathrm{Ag}^{+}$ions used in terms of $\log x\left(x=\left[\mathrm{Ag}^{+}\right]\right)$?
$$ \mathrm{E}_{\mathrm{Zn}^{2+} / \mathrm{Zn}}^{\ominus}=-0.76 \mathrm{~V}, \quad \mathrm{E}_{\mathrm{Ag}^{+} / \mathrm{Ag}}^{\ominus}=+0.80 \mathrm{~V}, \frac{2.303 \mathrm{RT}}{\mathrm{~F}}=0.059 \mathrm{~V} $$
Q50.
mcq single
+4 / 1
Which of the following statement(s) is/are true ?
A. If two orbitals have the same value of ( $\mathrm{n}+l$ ), the orbital with lower value of n will have lower energy.
B. Energies of the orbitals in the same subshell increase with increase in atomic number.
C. The size of $2 \mathrm{p}_x$ orbital is less than the size of $3 \mathrm{p}_x$ orbital.
D. Among 5f, 6s, 4d, 5p and 5d orbitals, none of the orbitals have 2 radial nodes.
Choose the correct answer from the options given below :
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
A variable $X$ takes values $0,0,2,6,12,20, \ldots, n(n-1)$ with frequencies ${ }^n C_0,{ }^n C_1,{ }^n C_2,{ }^n C_3,{ }^n C_4,{ }^n C_5, \ldots,{ }^n C_n$, respectively. If the mean of this data is 60 , then its median is :
Q2.
mcq single
+4 / 1
Let $O$ be the origin, $\overrightarrow{O P}=\vec{a}$ and $\overrightarrow{O Q}=\vec{b}$. If $R$ is the point on $\overrightarrow{O P}$ such that $\overrightarrow{O P}=5 \overrightarrow{O R}$, and $M$ is the point such that $\overrightarrow{O Q}=5 \overrightarrow{R M}$, then $\overrightarrow{P M}$ is equal to :
Q3.
mcq single
+4 / 1
The probabilities that players A and B of a team are selected for the captaincy for a tournament are 0.6 and 0.4 , respectively. If $A$ is selected the captain, the probability that the team wins the tournament is 0.8 and if B is selected the captain, the probability that the team wins the tournament is 0.7 . Then the probability, that the team wins the tournament, is :
Q4.
mcq single
+4 / 1
If the distance of the point $(a, 2,5)$ from the image of the point $(1,2,7)$ in the line $\frac{x}{1}=\frac{y-1}{1}=\frac{z-2}{2}$ is 4 , then the sum of all possible values of $a$ is equal to :
Q5.
mcq single
+4 / 1
Let a triangle PQR be such that P and Q lie on the line $\frac{x+3}{8}=\frac{y-4}{2}=\frac{z+1}{2}$ and are at a distance of 6 units from $R(1,2,3)$. If $(\alpha, \beta, \gamma)$ is the centroid of $\Delta P Q R$, then $\alpha+\beta+\gamma$ is equal to :
Q6.
mcq single
+4 / 1
Let $\alpha, \beta$ be the roots of the equation $x^2-x+\mathrm{p}=0$ and $\gamma, \delta$ be the roots the equation $x^2-4 x+\mathrm{q}=0$; $p, q \in \mathbf{Z}$. If $\alpha, \beta, \gamma, \delta$ are in G.P., then $|p+q|$ equals :
Q7.
mcq single
+4 / 1
If the sum of the first 10 terms of the series $\frac{1}{1+1^4 \times 4}+\frac{2}{1+2^4 \times 4}+\frac{3}{1+3^4 \times 4}+\frac{4}{1+4^4 \times 4}+\ldots \ldots$. is $\frac{m}{n}, \operatorname{gcd}(m, n)=1$, then $m+n$ is equal to :
Q8.
mcq single
+4 / 1
Let $\mathrm{A}_1, \mathrm{~A}_2, \mathrm{~A}_3, \ldots \ldots . ., \mathrm{A}_{39}$ be 39 arithmetic means between the numbers 59 and 159. Then the mean of $\mathrm{A}_{25}, \mathrm{~A}_{28}, \mathrm{~A}_{31}$ and $\mathrm{A}_{36}$ is equal to :
Q9.
mcq single
+4 / 1
Let $f(x)$ and $g(x)$ be twice differentiable functions satisfying $f^{\prime \prime}(x)=g^{\prime \prime}(x)$ for all $x \in \mathbf{R}, f^{\prime}(1)=2 g^{\prime}(1)=4$ and $g(2)=3 f(2)=9$. Then $f(25)-g(25)$ is equal to :
Q10.
mcq single
+4 / 1
Let $f(x)=\lim \limits_{y \rightarrow 0} \frac{(1-\cos (x y)) \tan (x y)}{y^3}$. Then the number of solutions of the equation $f(x)=\sin x$, $x \in \mathbf{R}$ is :
Q11.
numerical
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be a function such that $f(x)+3 f\left(\frac{\pi}{2}-x\right)=\sin x, x \in \mathbf{R}$. Let the maximum value of $f$ on $\mathbf{R}$ be $\alpha$. If the area of the region bounded by the curves $g(x)=x^2$ and $h(x)=\beta x^3, \beta>0$, is $\alpha^2$, then $30 \beta^3$ is equal to $\_\_\_\_$ .
Q12.
mcq single
+4 / 1
Let M be a $3 \times 3$ matrix such that $\mathrm{M}\left(\begin{array}{l}1 \\ 0 \\ 0\end{array}\right)=\left(\begin{array}{l}1 \\ 2 \\ 3\end{array}\right), \mathrm{M}\left(\begin{array}{l}0 \\ 1 \\ 0\end{array}\right)=\left(\begin{array}{l}0 \\ 1 \\ 2\end{array}\right)$ and $\mathrm{M}\left(\begin{array}{l}0 \\ 0 \\ 1\end{array}\right)=\left(\begin{array}{c}-1 \\ 1 \\ 1\end{array}\right)$. If $\mathrm{M}\left(\begin{array}{l}x \\ y \\ z\end{array}\right)=\left(\begin{array}{c}1 \\ 7 \\ 11\end{array}\right)$, then $x+y+z$ equals :
Q13.
mcq single
+4 / 1
If $f: \mathbf{N} \rightarrow \mathbf{Z}$ is defined by
$$ f(n)=\left|\begin{array}{ccc} n & -1 & -5 \\ -2 n^2 & 3(2 k+1) & 2 k+1 \\ -3 n^3 & 3 k(2 k+1) & 3 k(k+2)+1 \end{array}\right|, k \in N, $$
and $\sum\limits_{n=1}^k f(n)=98$, then $k$ is equal to :
Q14.
numerical
+4 / 1
If $S=\left\{\theta \in[-\pi, \pi]: \cos \theta \cos \frac{5 \theta}{2}=\cos 7 \theta \cos \frac{7 \theta}{2}\right\}$, then $n(S)$ is equal to $\_\_\_\_$ .
Q15.
mcq single
+4 / 1
Let the directrix of the parabola $\mathrm{P}: y^2=8 x$, cut $x$-axis at the point A . Let $\mathrm{B}(\alpha, \beta), \alpha>1$, be a point on $P$ such that the slope of $A B$ is $3 / 5$. If $B C$ is a focal chord of $P$, then six times the area of $\triangle A B C$ is :
Q16.
mcq single
+4 / 1
The coefficient of $x^2$ in the expansion of $\left(2 x^2+\frac{1}{x}\right)^{10}, x \neq 0$, is :
Q17.
numerical
+4 / 1
From the point $(-1,-1)$, two rays are sent making angles of $45^{\circ}$ with the line $x+y=0$. These rays get reflected from the mirror $x+2 y=1$. If the equations of the reflected rays are $\mathrm{a} x+\mathrm{b} y=9$ and $c x+d y=7, a, b, c, d \in \mathbf{Z}$, then the value of $a d+b c$ is $\_\_\_\_$ .
Q18.
numerical
+4 / 1
Let $\mathrm{A}=\{1,4,7\}$ and $\mathrm{B}=\{2,3,8\}$. Then the number of elements, in the relation $R=\left\{\left(\left(a_1, b_1\right),\left(a_2, b_2\right)\right) \in((A \times B) \times(A \times B)): a_1+b_2\right.$ divides $\left.a_2+b_1\right\}$ is $\_\_\_\_$ .
Q19.
mcq single
+4 / 1
Let $z_1, z_2 \in \mathbb{C}$ be the distinct solutions of the equation $z^2+4 z-(1+12 i)=0$.
Then $\left|z_1\right|^2+\left|z_2\right|^2$ is equal to :
Q20.
mcq single
+4 / 1
Let the eccentricity e of a hyperbola satisfy the equation $6 \mathrm{e}^2-11 \mathrm{e}+3=0$. If the foci of the hyperbola are $(3,5)$ and $(3,-4)$, then the length of its latus rectum is :
Q21.
mcq single
+4 / 1
Let $f:[1, \infty) \rightarrow \mathbf{R}$ be a differentiable function defined as $f(x)=\int_1^x f(\mathrm{t}) \mathrm{dt}+(1-x)\left(\log _{\mathrm{e}} x-1\right)+\mathrm{e}$.
Then the value of $f(f(1))$ is :
Q22.
numerical
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $(\tan x)^{1 / 2} \mathrm{~d} y=\left(\sec ^3 x-(\tan x)^{3 / 2} y\right) \mathrm{d} x, 0 < x <\frac{\pi}{2}, y\left(\frac{\pi}{4}\right)=\frac{6 \sqrt{2}}{5}$. If $y\left(\frac{\pi}{3}\right)=\frac{4}{5} \alpha$, then $\alpha^4$ equals
$\_\_\_\_$ .
Q23.
mcq single
+4 / 1
Let the point P be the vertex of the parabola $y=x^2-6 x+12$. If a line passing through the point P intersects the circle $x^2+y^2-2 x-4 y+3=0$ at the points R and S , then the maximum value of $(\mathrm{PR}+\mathrm{PS})^2$ is :
Q24.
mcq single
+4 / 1
A box contains 5 blue, 6 yellow and 4 red balls. The number of ways, of drawing 8 balls containing at least two balls of each colour, is :
Q25.
mcq single
+4 / 1
Let $\left(2^{1-\mathrm{a}}+2^{1+\mathrm{a}}\right), f(\mathrm{a}),\left(3^{\mathrm{a}}+3^{-\mathrm{a}}\right)$ be in A.P. and $\alpha$ be the minimum value of $f(\mathrm{a})$. Then the value of the integral $\int\limits_{\log _e(\alpha-1)}^{\log _e(\alpha)} \frac{d x}{\left(e^{2 x}-e^{-2 x}\right)}$ is :
Q26.
mcq single
+4 / 1
A variable $X$ takes values $0,0,2,6,12,20, \ldots, n(n-1)$ with frequencies ${ }^n C_0,{ }^n C_1,{ }^n C_2,{ }^n C_3,{ }^n C_4,{ }^n C_5, \ldots,{ }^n C_n$, respectively. If the mean of this data is 60 , then its median is :
Q27.
mcq single
+4 / 1
Let $O$ be the origin, $\overrightarrow{O P}=\vec{a}$ and $\overrightarrow{O Q}=\vec{b}$. If $R$ is the point on $\overrightarrow{O P}$ such that $\overrightarrow{O P}=5 \overrightarrow{O R}$, and $M$ is the point such that $\overrightarrow{O Q}=5 \overrightarrow{R M}$, then $\overrightarrow{P M}$ is equal to :
Q28.
mcq single
+4 / 1
The probabilities that players A and B of a team are selected for the captaincy for a tournament are 0.6 and 0.4 , respectively. If $A$ is selected the captain, the probability that the team wins the tournament is 0.8 and if B is selected the captain, the probability that the team wins the tournament is 0.7 . Then the probability, that the team wins the tournament, is :
Q29.
mcq single
+4 / 1
If the distance of the point $(a, 2,5)$ from the image of the point $(1,2,7)$ in the line $\frac{x}{1}=\frac{y-1}{1}=\frac{z-2}{2}$ is 4 , then the sum of all possible values of $a$ is equal to :
Q30.
mcq single
+4 / 1
Let a triangle PQR be such that P and Q lie on the line $\frac{x+3}{8}=\frac{y-4}{2}=\frac{z+1}{2}$ and are at a distance of 6 units from $R(1,2,3)$. If $(\alpha, \beta, \gamma)$ is the centroid of $\Delta P Q R$, then $\alpha+\beta+\gamma$ is equal to :
Q31.
mcq single
+4 / 1
Let $\alpha, \beta$ be the roots of the equation $x^2-x+\mathrm{p}=0$ and $\gamma, \delta$ be the roots the equation $x^2-4 x+\mathrm{q}=0$; $p, q \in \mathbf{Z}$. If $\alpha, \beta, \gamma, \delta$ are in G.P., then $|p+q|$ equals :
Q32.
mcq single
+4 / 1
If the sum of the first 10 terms of the series $\frac{1}{1+1^4 \times 4}+\frac{2}{1+2^4 \times 4}+\frac{3}{1+3^4 \times 4}+\frac{4}{1+4^4 \times 4}+\ldots \ldots$. is $\frac{m}{n}, \operatorname{gcd}(m, n)=1$, then $m+n$ is equal to :
Q33.
mcq single
+4 / 1
Let $\mathrm{A}_1, \mathrm{~A}_2, \mathrm{~A}_3, \ldots \ldots . ., \mathrm{A}_{39}$ be 39 arithmetic means between the numbers 59 and 159. Then the mean of $\mathrm{A}_{25}, \mathrm{~A}_{28}, \mathrm{~A}_{31}$ and $\mathrm{A}_{36}$ is equal to :
Q34.
mcq single
+4 / 1
Let $f(x)$ and $g(x)$ be twice differentiable functions satisfying $f^{\prime \prime}(x)=g^{\prime \prime}(x)$ for all $x \in \mathbf{R}, f^{\prime}(1)=2 g^{\prime}(1)=4$ and $g(2)=3 f(2)=9$. Then $f(25)-g(25)$ is equal to :
Q35.
mcq single
+4 / 1
Let $f(x)=\lim \limits_{y \rightarrow 0} \frac{(1-\cos (x y)) \tan (x y)}{y^3}$. Then the number of solutions of the equation $f(x)=\sin x$, $x \in \mathbf{R}$ is :
Q36.
numerical
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be a function such that $f(x)+3 f\left(\frac{\pi}{2}-x\right)=\sin x, x \in \mathbf{R}$. Let the maximum value of $f$ on $\mathbf{R}$ be $\alpha$. If the area of the region bounded by the curves $g(x)=x^2$ and $h(x)=\beta x^3, \beta>0$, is $\alpha^2$, then $30 \beta^3$ is equal to $\_\_\_\_$ .
Q37.
mcq single
+4 / 1
Let M be a $3 \times 3$ matrix such that $\mathrm{M}\left(\begin{array}{l}1 \\ 0 \\ 0\end{array}\right)=\left(\begin{array}{l}1 \\ 2 \\ 3\end{array}\right), \mathrm{M}\left(\begin{array}{l}0 \\ 1 \\ 0\end{array}\right)=\left(\begin{array}{l}0 \\ 1 \\ 2\end{array}\right)$ and $\mathrm{M}\left(\begin{array}{l}0 \\ 0 \\ 1\end{array}\right)=\left(\begin{array}{c}-1 \\ 1 \\ 1\end{array}\right)$. If $\mathrm{M}\left(\begin{array}{l}x \\ y \\ z\end{array}\right)=\left(\begin{array}{c}1 \\ 7 \\ 11\end{array}\right)$, then $x+y+z$ equals :
Q38.
mcq single
+4 / 1
If $f: \mathbf{N} \rightarrow \mathbf{Z}$ is defined by
$$ f(n)=\left|\begin{array}{ccc} n & -1 & -5 \\ -2 n^2 & 3(2 k+1) & 2 k+1 \\ -3 n^3 & 3 k(2 k+1) & 3 k(k+2)+1 \end{array}\right|, k \in N, $$
and $\sum\limits_{n=1}^k f(n)=98$, then $k$ is equal to :
Q39.
numerical
+4 / 1
If $S=\left\{\theta \in[-\pi, \pi]: \cos \theta \cos \frac{5 \theta}{2}=\cos 7 \theta \cos \frac{7 \theta}{2}\right\}$, then $n(S)$ is equal to $\_\_\_\_$ .
Q40.
mcq single
+4 / 1
Let the directrix of the parabola $\mathrm{P}: y^2=8 x$, cut $x$-axis at the point A . Let $\mathrm{B}(\alpha, \beta), \alpha>1$, be a point on $P$ such that the slope of $A B$ is $3 / 5$. If $B C$ is a focal chord of $P$, then six times the area of $\triangle A B C$ is :
Q41.
mcq single
+4 / 1
The coefficient of $x^2$ in the expansion of $\left(2 x^2+\frac{1}{x}\right)^{10}, x \neq 0$, is :
Q42.
numerical
+4 / 1
From the point $(-1,-1)$, two rays are sent making angles of $45^{\circ}$ with the line $x+y=0$. These rays get reflected from the mirror $x+2 y=1$. If the equations of the reflected rays are $\mathrm{a} x+\mathrm{b} y=9$ and $c x+d y=7, a, b, c, d \in \mathbf{Z}$, then the value of $a d+b c$ is $\_\_\_\_$ .
Q43.
numerical
+4 / 1
Let $\mathrm{A}=\{1,4,7\}$ and $\mathrm{B}=\{2,3,8\}$. Then the number of elements, in the relation $R=\left\{\left(\left(a_1, b_1\right),\left(a_2, b_2\right)\right) \in((A \times B) \times(A \times B)): a_1+b_2\right.$ divides $\left.a_2+b_1\right\}$ is $\_\_\_\_$ .
Q44.
mcq single
+4 / 1
Let $z_1, z_2 \in \mathbb{C}$ be the distinct solutions of the equation $z^2+4 z-(1+12 i)=0$.
Then $\left|z_1\right|^2+\left|z_2\right|^2$ is equal to :
Q45.
mcq single
+4 / 1
Let the eccentricity e of a hyperbola satisfy the equation $6 \mathrm{e}^2-11 \mathrm{e}+3=0$. If the foci of the hyperbola are $(3,5)$ and $(3,-4)$, then the length of its latus rectum is :
Q46.
mcq single
+4 / 1
Let $f:[1, \infty) \rightarrow \mathbf{R}$ be a differentiable function defined as $f(x)=\int_1^x f(\mathrm{t}) \mathrm{dt}+(1-x)\left(\log _{\mathrm{e}} x-1\right)+\mathrm{e}$.
Then the value of $f(f(1))$ is :
Q47.
numerical
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $(\tan x)^{1 / 2} \mathrm{~d} y=\left(\sec ^3 x-(\tan x)^{3 / 2} y\right) \mathrm{d} x, 0 < x <\frac{\pi}{2}, y\left(\frac{\pi}{4}\right)=\frac{6 \sqrt{2}}{5}$. If $y\left(\frac{\pi}{3}\right)=\frac{4}{5} \alpha$, then $\alpha^4$ equals
$\_\_\_\_$ .
Q48.
mcq single
+4 / 1
Let the point P be the vertex of the parabola $y=x^2-6 x+12$. If a line passing through the point P intersects the circle $x^2+y^2-2 x-4 y+3=0$ at the points R and S , then the maximum value of $(\mathrm{PR}+\mathrm{PS})^2$ is :
Q49.
mcq single
+4 / 1
A box contains 5 blue, 6 yellow and 4 red balls. The number of ways, of drawing 8 balls containing at least two balls of each colour, is :
Q50.
mcq single
+4 / 1
Let $\left(2^{1-\mathrm{a}}+2^{1+\mathrm{a}}\right), f(\mathrm{a}),\left(3^{\mathrm{a}}+3^{-\mathrm{a}}\right)$ be in A.P. and $\alpha$ be the minimum value of $f(\mathrm{a})$. Then the value of the integral $\int\limits_{\log _e(\alpha-1)}^{\log _e(\alpha)} \frac{d x}{\left(e^{2 x}-e^{-2 x}\right)}$ is :
Physics
Physics
Q1.
mcq single
+4 / 1
A mass of 1 kg is kept on a inclined plane with $30^{\circ}$ inclination with respect to horizontal plane and it is at rest initially. Then the whole assembly is moved up with constant velocity of $4 \mathrm{~m} / \mathrm{s}$. The work done by the frictional force in time 2 s is $\_\_\_\_$ J. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q2.
mcq single
+4 / 1
In a semiconductor $p$-n diode, the doping concentrations on $p$-side and $n$-side are $10^{15}$ atoms $/ \mathrm{cm}^3$ and $10^{18}$ atoms $/ \mathrm{cm}^3$, respectively. Which one of the following statements is true?
Q3.
mcq single
+4 / 1
The maximum intensity in a Young's double slit experiment is $I_0$. Distance between the slits $(d)$ is $5 \lambda$, where $\lambda$ is the wavelength of light used. The intensity of the fringe, exactly opposite to one of the slits on the screen, placed at $D=10 d$ is $\_\_\_\_$ .
Q4.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
$$
\text { List - I }
$$
$$
\text { List - II }
$$
A.
$$
\sin ^2 \omega t
$$
I.
Periodic with time period $T=\frac{\pi}{\omega}$ but not simple harmonic motion (SHM)
B.
$$
\sin ^3(2 \omega t)
$$
II.
Periodic with time period $T=\frac{2 \pi}{\omega}$ but Not SHM
C.
$$
\sin (\omega t)+\cos (\pi \omega t)
$$
III.
Periodic with time period $T=\frac{\pi}{\omega}$ and SHM
D.
$$
\cos \omega t+\cos 2 \omega t
$$
IV.
Non-periodic
Choose the correct answer from the options given below :
Q5.
mcq single
+4 / 1
An object of uniform density rolls up the curved path with the initial velocity $v_{\mathrm{o}}$ as shown in the figure. If the maximum height attained by an object is $\frac{7 v_0^2}{10 \mathrm{~g}}$ ( $\mathrm{g}=$ acceleration due to gravity), the object is a $\_\_\_\_$ .


Q6.
mcq single
+4 / 1
A wheel initially at rest is subjected to a uniform angular acceleration about its axis. In the first 2 s it rotates through an angle $\theta_1$ and in the next 2 s it rotates through an angle $\theta_2$. The ratio $\frac{\theta_2}{\theta_1}$ is $\_\_\_\_$ .
Q7.
mcq single
+4 / 1
A metal rod of length $L$ rotates about one end at origin with a uniform angular velocity $\omega$. The magnetic field radially falls off as $B(\mathrm{r})=B_{\mathrm{o}} \mathrm{e}^{-\lambda r} ; \lambda$ being a positive constant. The emf induced (neglecting the centripetal force on electrons in the rod) is :
Q8.
numerical
+4 / 1
A circular loop of radius 20 cm and resistance $2 \Omega$ is placed in a time varying magnetic field $\vec{B}=\left(2 t^2+2 t+3\right) T$. At $t=0$, for the plane of the loop being perpendicular to the magnetic field and, the induced current in the loop at $t=3 \mathrm{~s}$ is $\frac{\alpha}{50} \mathrm{~A}$. The value of $\alpha$ is $\_\_\_\_$ . (Take $\pi=22 / 7$ )
Q9.
numerical
+4 / 1
The heat extracted out of $x$ gram of water initially at $50^{\circ} \mathrm{C}$ to $\operatorname{cool}$ it down to $0^{\circ} \mathrm{C}$ is sufficient to evaporate $(1000-x)$ gram of water also initially at $50^{\circ} \mathrm{C}$. The value of $x$ (closest integer) is $\_\_\_\_$ .
(Take latent heat of water $2256 \mathrm{~kJ} / \mathrm{kg} . \mathrm{K}$, specific heat capacity of water $4200 \mathrm{~J} / \mathrm{kg} . \mathrm{K}$ )
Q10.
mcq single
+4 / 1
An ideal gas at pressure $P$ and temperature $T$ is expanding such that $P T^3=$ constant. The coefficient of volume expansion of the gas is $\_\_\_\_$ .
Q11.
numerical
+4 / 1
A copper wire of length 3 m is stretched by 3 mm by applying an external force. The volume of the wire is $600 \times 10^{-6} \mathrm{~m}^3$. The elastic potential energy stored in the wire in stretched condition would be
$\_\_\_\_$ J.
(Given Young modulus of copper $=1.1 \times 10^{11} \mathrm{~N} / \mathrm{m}^2$ )
Q12.
mcq single
+4 / 1
Eight mercury drops, each of radius $r$, coalesce to form a bigger drop. The surface energy released in this process is $\_\_\_\_$ - ( $S$ is the surface tension of mercury).
Q13.
mcq single
+4 / 1
An electron is travelling with a velocity $v$ in free space and when it enters a medium, its velocity is reduced by $20 \%$. The de Broglie wavelength of electron in the medium is $\alpha \lambda_0$, where $\lambda_0$ is its de Broglie wavelength in free space. The value of $\alpha$ is $\_\_\_\_$ .
Q14.
mcq single
+4 / 1
A body of mass $m$ is taken from the surface of earth to a height equal to twice the radius of earth $\left(R_e\right)$. The increase in potential energy will be $\_\_\_\_$ .
( $g$ is acceleration due to gravity at the surface of earth)
Q15.
mcq single
+4 / 1
Under steady state condition the potential difference across the capacitor in the circuit is $\_\_\_\_$ V.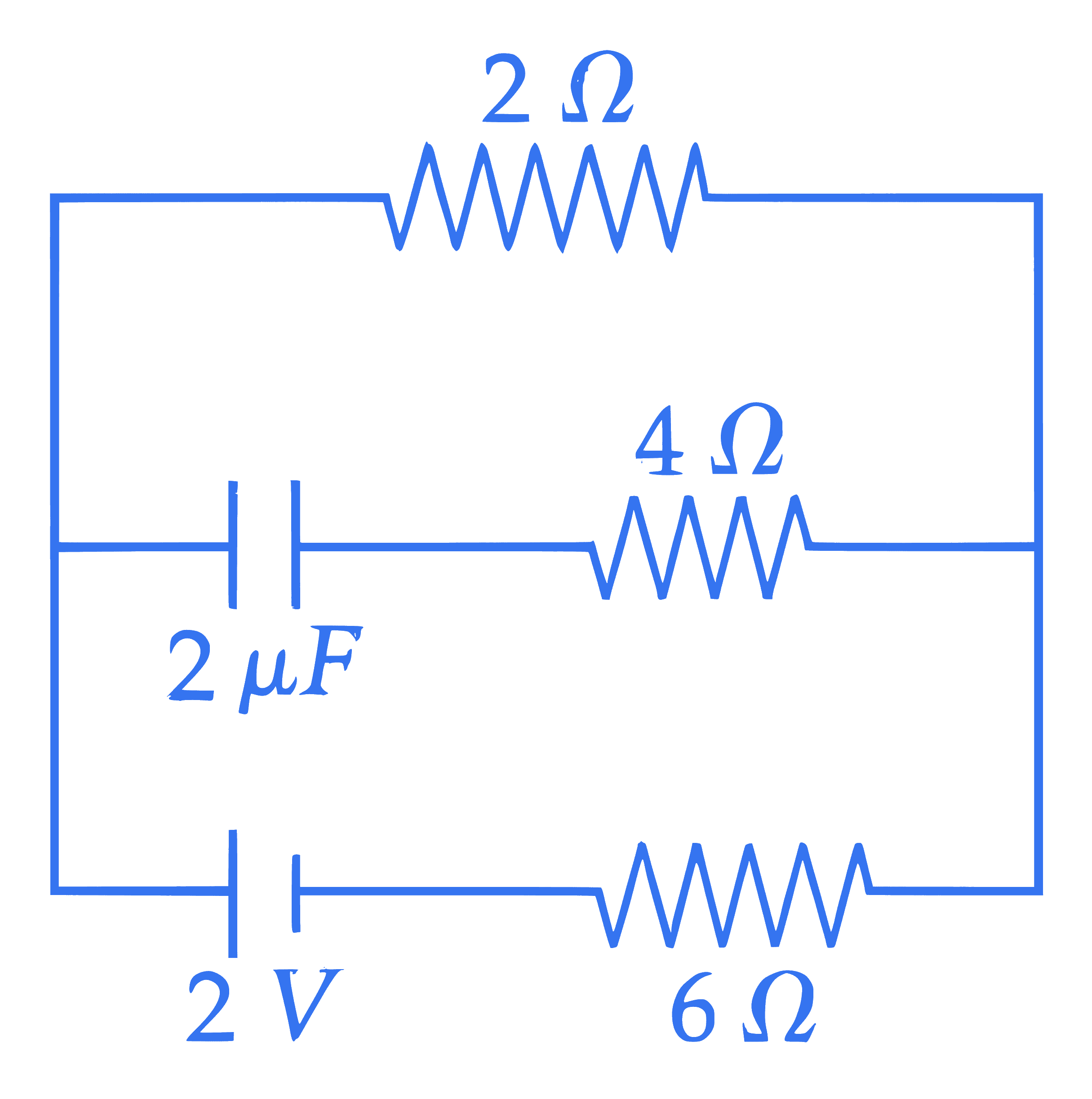

Q16.
mcq single
+4 / 1
An object $A B$ is placed 15 cm on the left of a convex lens $P$ of focal length 10 cm . Another convex lens $Q$ is now placed 15 cm right of lens $P$. If the focal length of lens $Q$ is 15 cm , the final image is $\_\_\_\_$ .
Q17.
mcq single
+4 / 1
A thin convex lens and a thin concave lens are kept in contact and are co-axial. Which of the following statements is correct for this combination of two lenses ?
Q18.
mcq single
+4 / 1
A particle of charge $q$ and mass $m$ is projected from origin with an initial velocity $\vec{v}=\left(\frac{v_0}{\sqrt{2}} \hat{x}+\frac{v_0}{\sqrt{2}} \hat{y}\right)$. There exists a uniform magnetic field $\vec{B}=B_0 \hat{z}$ and a space varying electric field $\vec{E}=E_{\mathrm{o}} \mathrm{e}^{-\lambda x} \hat{x}$ within the region $0 \leqslant x \leqslant L$. After travelling a distance such that $x$-coordinate has changed from $x=0$ to $x=L$, the change in the kinetic energy is $\_\_\_\_$ .
Q19.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
$$
\text { List - I }
$$
$$
\text { List - II }
$$
A.
Meter (L)
I.
$$
\sqrt{\frac{h c}{G}}
$$
B.
Second (S)
II.
$$
\sqrt{\frac{G h}{c^5}}
$$
C.
Kilogram (M)
III.
$$
\sqrt{\frac{K^2 L^2 c^3}{G h}}
$$
D.
Kelvin (K)
IV.
$$
\sqrt{\frac{G h}{c^3}}
$$
where h (Planck's constant), G (gravitational constant) and c (speed of light in vacuum) as fundamental units.
Choose the correct answer from the options given below :
Q20.
mcq single
+4 / 1
In an experiment to determine the resistance of a given wire using Ohm's law, the voltmeter and ammeter readings are noted as 10 V and 5 A , respectively. The least counts of voltmeter and ammeter are 500 mV and 200 mA , respectively. The estimated error in the resistance measurement is $\_\_\_\_$ $\Omega$
Q21.
mcq single
+4 / 1
The velocity $(v)$ versus time $(t)$ plot of a particle is shown in the figure, for a time interval of 40 s . The total distance travelled by the particle and the average velocity during this period are, respectively
$\_\_\_\_$.
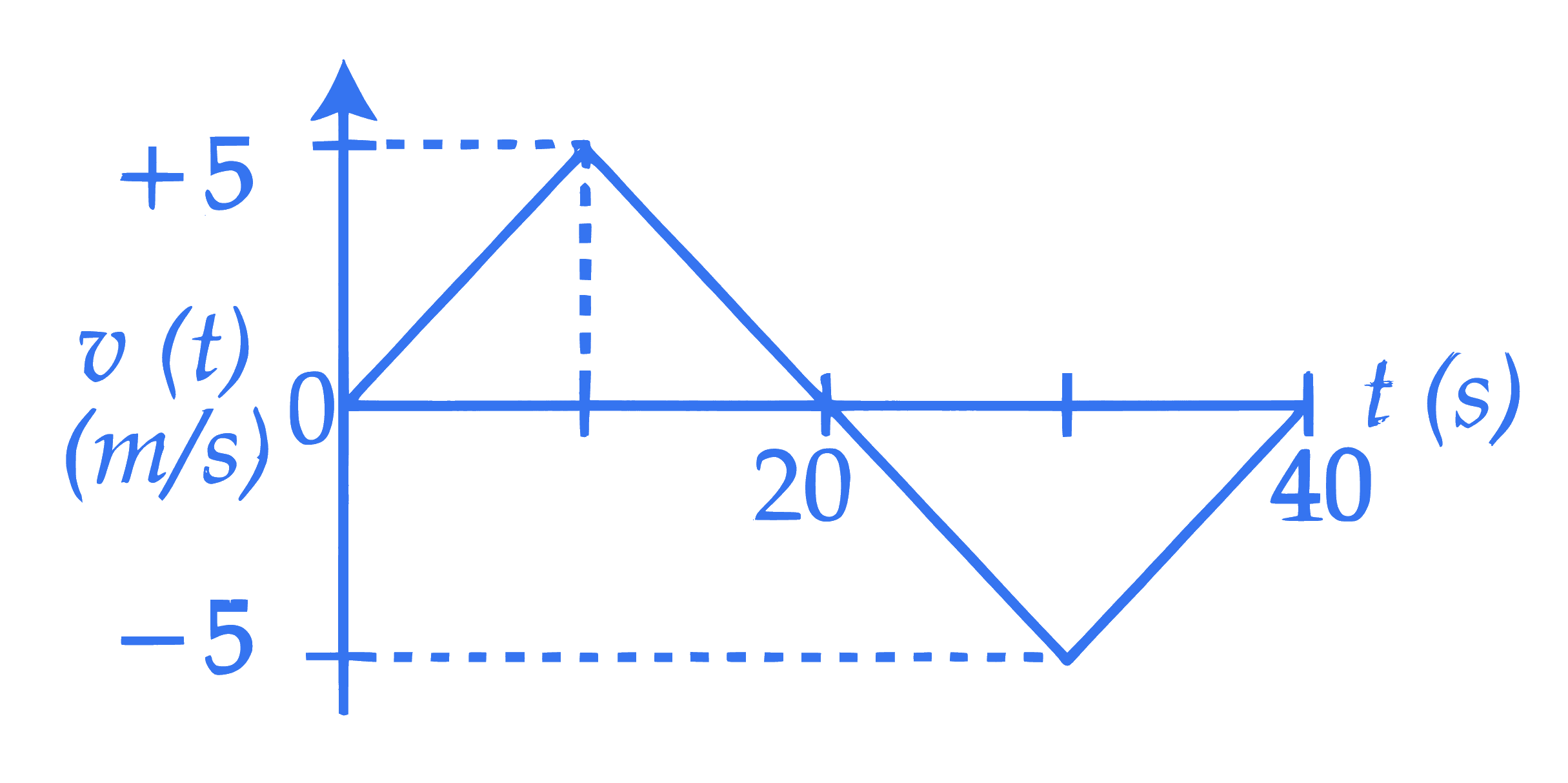

Q22.
mcq single
+4 / 1
Assuming the experimental mass of ${ }_6^{12} C$ as $12 u$, the mass defect of ${ }_6^{12} C$ atom is $\_\_\_\_$ $\mathrm{MeV} / \mathrm{c}^2$.
(Mass of proton $=1.00727 \mathrm{u}$. mass of neutron $=1.00866 \mathrm{u}, 1 \mathrm{u}=931.5 \mathrm{MeV} / \mathrm{c}^2$ and c is the speed of the light in vacuum).
Q23.
numerical
+4 / 1
When an external resistance of $5 \Omega$ is connected across terminals of a cell, a current of 0.25 A flows through it. When the $5 \Omega$ resistor is replaced by a $2 \Omega$ resistor, a current of 0.5 A flows through it. The internal resistance of the cell is $\_\_\_\_$ $\Omega$.
Q24.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
**Assertion (A) :** The electromagnetic wave exerts pressure on the surface on which they are allowed to fall.
**Reason (R) :** There is no mass associated with the electromagnetic waves.
In the light of the above statements, choose the correct answer from the options given below :
Q25.
numerical
+4 / 1
A series LCR circuit with $R=20 \Omega, L=1.6 \mathrm{H}$ and $C=40 \mu \mathrm{~F}$ is connected to a variable frequency a.c. source. The inductive reactance at resonant frequency is $\_\_\_\_$ $\Omega$.
Q26.
mcq single
+4 / 1
A mass of 1 kg is kept on a inclined plane with $30^{\circ}$ inclination with respect to horizontal plane and it is at rest initially. Then the whole assembly is moved up with constant velocity of $4 \mathrm{~m} / \mathrm{s}$. The work done by the frictional force in time 2 s is $\_\_\_\_$ J. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q27.
mcq single
+4 / 1
In a semiconductor $p$-n diode, the doping concentrations on $p$-side and $n$-side are $10^{15}$ atoms $/ \mathrm{cm}^3$ and $10^{18}$ atoms $/ \mathrm{cm}^3$, respectively. Which one of the following statements is true?
Q28.
mcq single
+4 / 1
The maximum intensity in a Young's double slit experiment is $I_0$. Distance between the slits $(d)$ is $5 \lambda$, where $\lambda$ is the wavelength of light used. The intensity of the fringe, exactly opposite to one of the slits on the screen, placed at $D=10 d$ is $\_\_\_\_$ .
Q29.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
$$
\text { List - I }
$$
$$
\text { List - II }
$$
A.
$$
\sin ^2 \omega t
$$
I.
Periodic with time period $T=\frac{\pi}{\omega}$ but not simple harmonic motion (SHM)
B.
$$
\sin ^3(2 \omega t)
$$
II.
Periodic with time period $T=\frac{2 \pi}{\omega}$ but Not SHM
C.
$$
\sin (\omega t)+\cos (\pi \omega t)
$$
III.
Periodic with time period $T=\frac{\pi}{\omega}$ and SHM
D.
$$
\cos \omega t+\cos 2 \omega t
$$
IV.
Non-periodic
Choose the correct answer from the options given below :
Q30.
mcq single
+4 / 1
An object of uniform density rolls up the curved path with the initial velocity $v_{\mathrm{o}}$ as shown in the figure. If the maximum height attained by an object is $\frac{7 v_0^2}{10 \mathrm{~g}}$ ( $\mathrm{g}=$ acceleration due to gravity), the object is a $\_\_\_\_$ .


Q31.
mcq single
+4 / 1
A wheel initially at rest is subjected to a uniform angular acceleration about its axis. In the first 2 s it rotates through an angle $\theta_1$ and in the next 2 s it rotates through an angle $\theta_2$. The ratio $\frac{\theta_2}{\theta_1}$ is $\_\_\_\_$ .
Q32.
mcq single
+4 / 1
A metal rod of length $L$ rotates about one end at origin with a uniform angular velocity $\omega$. The magnetic field radially falls off as $B(\mathrm{r})=B_{\mathrm{o}} \mathrm{e}^{-\lambda r} ; \lambda$ being a positive constant. The emf induced (neglecting the centripetal force on electrons in the rod) is :
Q33.
numerical
+4 / 1
A circular loop of radius 20 cm and resistance $2 \Omega$ is placed in a time varying magnetic field $\vec{B}=\left(2 t^2+2 t+3\right) T$. At $t=0$, for the plane of the loop being perpendicular to the magnetic field and, the induced current in the loop at $t=3 \mathrm{~s}$ is $\frac{\alpha}{50} \mathrm{~A}$. The value of $\alpha$ is $\_\_\_\_$ . (Take $\pi=22 / 7$ )
Q34.
numerical
+4 / 1
The heat extracted out of $x$ gram of water initially at $50^{\circ} \mathrm{C}$ to $\operatorname{cool}$ it down to $0^{\circ} \mathrm{C}$ is sufficient to evaporate $(1000-x)$ gram of water also initially at $50^{\circ} \mathrm{C}$. The value of $x$ (closest integer) is $\_\_\_\_$ .
(Take latent heat of water $2256 \mathrm{~kJ} / \mathrm{kg} . \mathrm{K}$, specific heat capacity of water $4200 \mathrm{~J} / \mathrm{kg} . \mathrm{K}$ )
Q35.
mcq single
+4 / 1
An ideal gas at pressure $P$ and temperature $T$ is expanding such that $P T^3=$ constant. The coefficient of volume expansion of the gas is $\_\_\_\_$ .
Q36.
numerical
+4 / 1
A copper wire of length 3 m is stretched by 3 mm by applying an external force. The volume of the wire is $600 \times 10^{-6} \mathrm{~m}^3$. The elastic potential energy stored in the wire in stretched condition would be
$\_\_\_\_$ J.
(Given Young modulus of copper $=1.1 \times 10^{11} \mathrm{~N} / \mathrm{m}^2$ )
Q37.
mcq single
+4 / 1
Eight mercury drops, each of radius $r$, coalesce to form a bigger drop. The surface energy released in this process is $\_\_\_\_$ - ( $S$ is the surface tension of mercury).
Q38.
mcq single
+4 / 1
An electron is travelling with a velocity $v$ in free space and when it enters a medium, its velocity is reduced by $20 \%$. The de Broglie wavelength of electron in the medium is $\alpha \lambda_0$, where $\lambda_0$ is its de Broglie wavelength in free space. The value of $\alpha$ is $\_\_\_\_$ .
Q39.
mcq single
+4 / 1
A body of mass $m$ is taken from the surface of earth to a height equal to twice the radius of earth $\left(R_e\right)$. The increase in potential energy will be $\_\_\_\_$ .
( $g$ is acceleration due to gravity at the surface of earth)
Q40.
mcq single
+4 / 1
Under steady state condition the potential difference across the capacitor in the circuit is $\_\_\_\_$ V.

Q41.
mcq single
+4 / 1
An object $A B$ is placed 15 cm on the left of a convex lens $P$ of focal length 10 cm . Another convex lens $Q$ is now placed 15 cm right of lens $P$. If the focal length of lens $Q$ is 15 cm , the final image is $\_\_\_\_$ .
Q42.
mcq single
+4 / 1
A thin convex lens and a thin concave lens are kept in contact and are co-axial. Which of the following statements is correct for this combination of two lenses ?
Q43.
mcq single
+4 / 1
A particle of charge $q$ and mass $m$ is projected from origin with an initial velocity $\vec{v}=\left(\frac{v_0}{\sqrt{2}} \hat{x}+\frac{v_0}{\sqrt{2}} \hat{y}\right)$. There exists a uniform magnetic field $\vec{B}=B_0 \hat{z}$ and a space varying electric field $\vec{E}=E_{\mathrm{o}} \mathrm{e}^{-\lambda x} \hat{x}$ within the region $0 \leqslant x \leqslant L$. After travelling a distance such that $x$-coordinate has changed from $x=0$ to $x=L$, the change in the kinetic energy is $\_\_\_\_$ .
Q44.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
$$
\text { List - I }
$$
$$
\text { List - II }
$$
A.
Meter (L)
I.
$$
\sqrt{\frac{h c}{G}}
$$
B.
Second (S)
II.
$$
\sqrt{\frac{G h}{c^5}}
$$
C.
Kilogram (M)
III.
$$
\sqrt{\frac{K^2 L^2 c^3}{G h}}
$$
D.
Kelvin (K)
IV.
$$
\sqrt{\frac{G h}{c^3}}
$$
where h (Planck's constant), G (gravitational constant) and c (speed of light in vacuum) as fundamental units.
Choose the correct answer from the options given below :
Q45.
mcq single
+4 / 1
In an experiment to determine the resistance of a given wire using Ohm's law, the voltmeter and ammeter readings are noted as 10 V and 5 A , respectively. The least counts of voltmeter and ammeter are 500 mV and 200 mA , respectively. The estimated error in the resistance measurement is $\_\_\_\_$ $\Omega$
Q46.
mcq single
+4 / 1
The velocity $(v)$ versus time $(t)$ plot of a particle is shown in the figure, for a time interval of 40 s . The total distance travelled by the particle and the average velocity during this period are, respectively
$\_\_\_\_$.


Q47.
mcq single
+4 / 1
Assuming the experimental mass of ${ }_6^{12} C$ as $12 u$, the mass defect of ${ }_6^{12} C$ atom is $\_\_\_\_$ $\mathrm{MeV} / \mathrm{c}^2$.
(Mass of proton $=1.00727 \mathrm{u}$. mass of neutron $=1.00866 \mathrm{u}, 1 \mathrm{u}=931.5 \mathrm{MeV} / \mathrm{c}^2$ and c is the speed of the light in vacuum).
Q48.
numerical
+4 / 1
When an external resistance of $5 \Omega$ is connected across terminals of a cell, a current of 0.25 A flows through it. When the $5 \Omega$ resistor is replaced by a $2 \Omega$ resistor, a current of 0.5 A flows through it. The internal resistance of the cell is $\_\_\_\_$ $\Omega$.
Q49.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
**Assertion (A) :** The electromagnetic wave exerts pressure on the surface on which they are allowed to fall.
**Reason (R) :** There is no mass associated with the electromagnetic waves.
In the light of the above statements, choose the correct answer from the options given below :
Q50.
numerical
+4 / 1
A series LCR circuit with $R=20 \Omega, L=1.6 \mathrm{H}$ and $C=40 \mu \mathrm{~F}$ is connected to a variable frequency a.c. source. The inductive reactance at resonant frequency is $\_\_\_\_$ $\Omega$.