JEE Main 2026 (Online) 5th April Morning Shift
JEE 2026 Previous Year
3 hDuration
296Total Marks
148Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 148 across 3 section(s); maximum marks: 296.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
$$ \text {Identify the incorrect statements. } $$
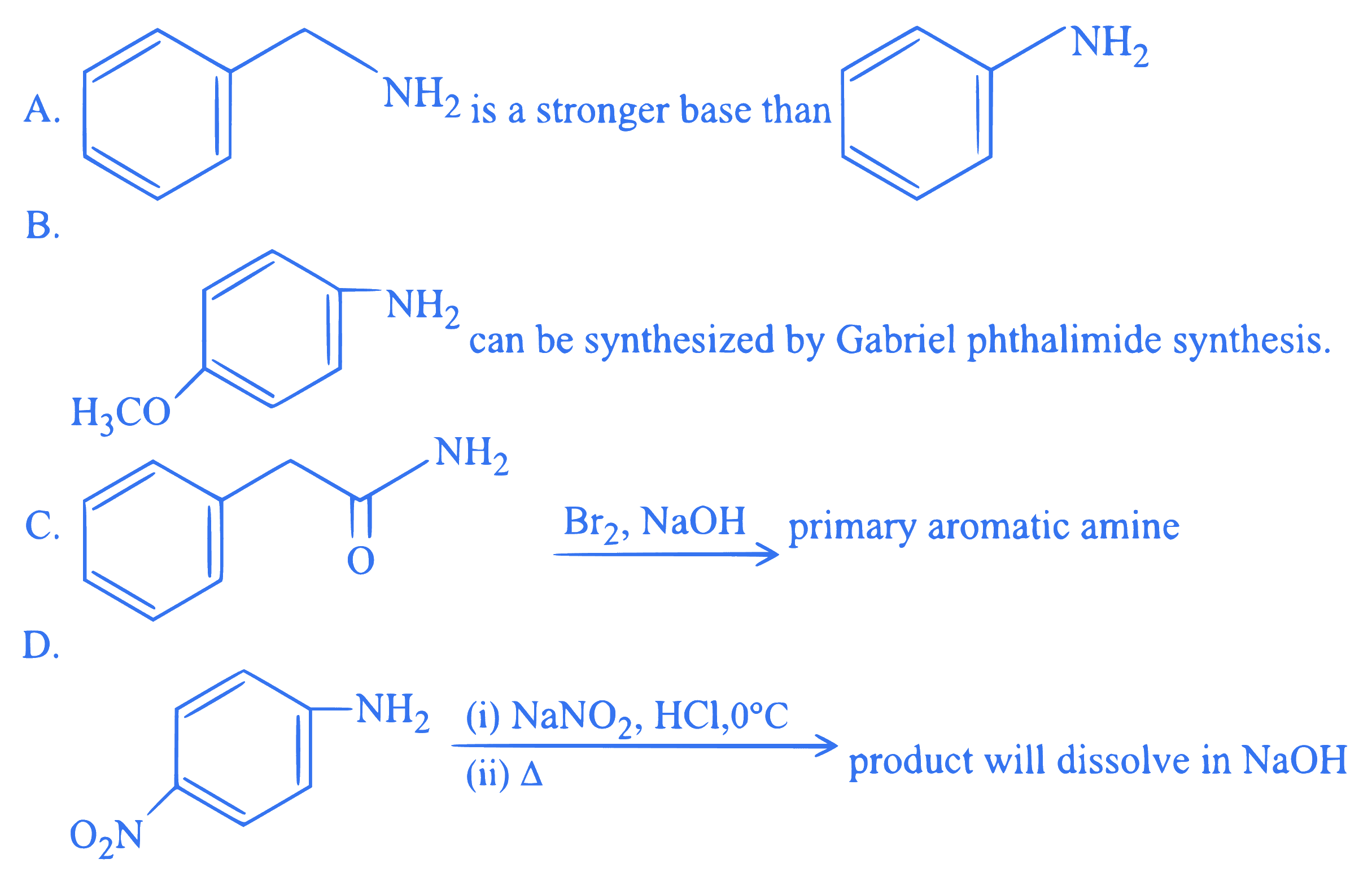 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
Q2.
mcq single
+4 / 1
$$ \text {Benzyl isocyanide can be obtained from } $$
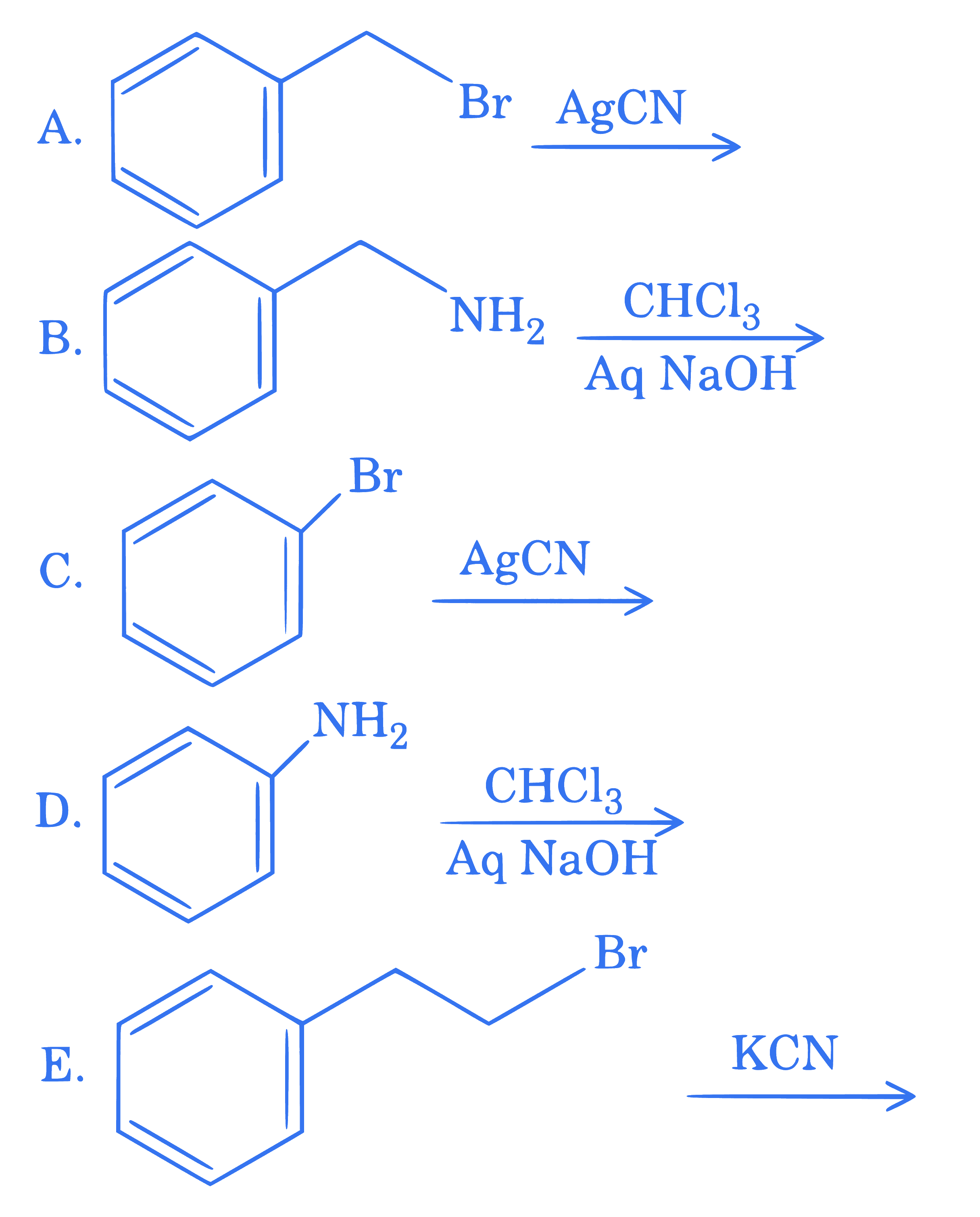 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q3.
mcq single
+4 / 1
Which of the following is NOT a physical or chemical characteristics of interstitial compounds?
Q4.
numerical
+4 / 1
If the half life of a first order reaction is 6.93 minutes then the time required for completion of $99 \%$ of the reaction will be $\_\_\_\_$ minutes.
(Given $: \log 2=0.3010$ )
Q5.
mcq single
+4 / 1
First order gas phase reaction
$$ \mathrm{A} \rightarrow \mathrm{~B}+\mathrm{C} $$
$p_t=$ initial pressure of gas $\mathrm{A}, p_t=$ total pressure of the reaction mixture at time $t$
Expression of rate constant ( $k$ ) is
Q6.
numerical
+4 / 1
Consider the following species:
$$ \mathrm{BrF}_5, \mathrm{XeF}_5^{-}, \mathrm{BF}_4^{-}, \mathrm{ICl}_4^{-}, \mathrm{XeF}_4, \mathrm{SF}_4, \mathrm{NH}_4^{+}, \mathrm{ClF}_3, \mathrm{XeF}_2, \mathrm{ICl}_2^{-} $$
Number of species having $\mathrm{sp}^3 \mathrm{~d}$ hybridized central atom is $\_\_\_\_$ .
Q7.
mcq single
+4 / 1
What is the mole fraction of water in $10 \%$ by weight $(\mathrm{w} / \mathrm{w})$ of aqueous urea solution?
[Given: Molar mass of $\mathrm{H}, \mathrm{O}, \mathrm{C}$ and N are $1,16,12$ and $14 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.]
Q8.
mcq single
+4 / 1
How many grams of residue is obtained by heating 2.76 g of silver carbonate?
(Given : Molar mass of $\mathrm{C}, \mathrm{O}$ and Ag are 12, 16 and $108 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively)
Q9.
numerical
+4 / 1
The values of pressure equilibrium constant recorded at different temperatures for the following equilibrium reaction have been given below $\mathrm{A}(\mathrm{g}) \rightleftharpoons \mathrm{B}(\mathrm{g})+\mathrm{C}(\mathrm{g})$
$$ \begin{array}{|c|c|} \hline \frac{1}{\mathrm{~T}}\left(\mathrm{~K}^{-1}\right) & \log _{10} \mathrm{~K}_{\mathrm{p}} \\ \hline 0.05 & 3.5 \\ \hline 0.06 & 2.5 \\ \hline 0.07 & 1.5 \\ \hline \end{array} $$
The magnitude of $\frac{\Delta \mathrm{H}^{\circ}}{\mathrm{R}}$ calculated from the above data is $\_\_\_\_$ . (Nearest integer)
Q10.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Sodium dichromate and potassium dichromate are classified as primary standards in titrimetric analysis.
**Statement II :** Phenolphthalein is a weak base, therefore it dissociates in acidic medium.
In the light of the above statements, choose the correct answer from the options given below
Q11.
mcq single
+4 / 1
Arrange the following resultant mixtures in increasing order of their pH values
A. $10 \mathrm{~mL} 0.2 \mathrm{M} \mathrm{Ca}(\mathrm{OH})_2+25 \mathrm{~mL} 0.1 \mathrm{M} \mathrm{HCl}$
B. $10 \mathrm{~mL} 0.01 \mathrm{M} \mathrm{H}_2 \mathrm{SO}_4+10 \mathrm{~mL} 0.01 \mathrm{M} \mathrm{Ca}(\mathrm{OH})_2$
C. $10 \mathrm{~mL} 0.1 \mathrm{M} \mathrm{H}_2 \mathrm{SO}_4+10 \mathrm{~mL} 0.1 \mathrm{M} \mathrm{KOH}$
Choose the correct answer from the options given below :
Q12.
mcq single
+4 / 1
$\mathrm{M}_3 \mathrm{~A}_2$ is a sparingly soluble salt of molar mass $y \mathrm{~g} \mathrm{~mol}^{-1}$ and solubility $x \mathrm{~g} \mathrm{~L}^{-1}$. The ratio of the molar concentration of the anion $\left(\mathrm{A}^{3-}\right)$ to the solubility product of the salt is
Q13.
mcq single
+4 / 1
Given below are two statements:
Statement I: The correct order of electronegativity of fluorine, oxygen and nitrogen is $\mathrm{F}>\mathrm{O}>\mathrm{N}$.
Statement II: The oxidation state of oxygen in $\mathrm{OF}_2$ is +2 and in $\mathrm{Na}_2 \mathrm{O}$ is -2 .
In the light of the above statements, choose the correct answer from the options given below
Q14.
mcq single
+4 / 1
Correct statements from the following are:
A. Nitrogen in oxidation states from +1 to +4 disproportionates in acid medium.
B. Nitrogen has the ability to form $\mathrm{d} \pi$ - $\mathrm{p} \pi$ multiple bonds with itself and other elements with small size and high electronegativity.
C. N-N single bond is stronger than P-P single bond.
D. Nitrogen has highest density in its group due to small size.
E. The maximum covalency of nitrogen is four since it has only four valence orbitals for bonding.
Choose the correct answer from the options given below:
Q15.
mcq single
+4 / 1
The correct statements about metal carbonyls are
A. The metal-carbon bonds in metal carbonyls possess both $\sigma$ and $\pi$ character.
B. Due to synergic bonding interactions between metal and CO ligand, the metal-carbon bond becomes weak.
C. The metal-carbon $\sigma$ bond is formed by the donation of lone pair of electrons on the carbonyl carbon into a vacant orbital of metal.
D. The metal-carbon $\pi$ bond is formed by the donation of electrons from filled d-orbital of metal into vacant $\pi^*$ orbital of CO .
Choose the correct answer from the options given below :
Q16.
mcq single
+4 / 1
Given below are two statements:
Statement I: Each electron in $\mathrm{e}_{\mathrm{g}}$ orbitals destabilizes the orbitals by $+0.6 \Delta_{\mathrm{o}}$ and each electron in the $t_{2 g}$ orbitals stabilizes the orbitals by $-0.4 \Delta_0$ in an octahedral field on the basis of crystal field theory.
Statement II: All the d - orbitals of the transition metals have the same energy in their free atomic state but when a complex is formed the ligands destroy the degeneracy of these orbitals on the basis of crystal field theory.
In the light of the above statements, choose the correct answer from the options given below
Q17.
numerical
+4 / 1
In an estimation of sulphur by Carius method 0.2 g of the substance gave 0.6 g of $\mathrm{BaSO}_4$. The percentage of sulphur in the substance is $\_\_\_\_$%.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{~S}: 32, \mathrm{BaSO}_4: 231$ )
Q18.
mcq single
+4 / 1
For the following Friedel Craft's alkylation reaction, which of the statements are correct?
 A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
 A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
Q19.
mcq single
+4 / 1
Identify the correct statements.
A. Glucose exists in two anomeric forms.
B. Anomers of glucose differ in configuration at $\mathrm{C}-1$ in cyclic hemiacetal structure.
C. Melting point of $\alpha$ - anomer of glucose is greater than $\beta$ - anomer.
D. Specific rotation of $\alpha$ - anomer is $+19^{\circ}$ while for $\beta$ - anomer is $+112^{\circ}$
E. $\alpha$ and $\beta$ - anomers of glucose are prepared by crystallization of saturated glucose solution at 303 K and 371 K respectively.
Choose the correct answer from the options given below :
Q20.
mcq single
+4 / 1
Given below are two statements:
Statement I: On the basis of inductive effect, the order of stability of alkyl carbanions is $\mathrm{CH}_3{ }^{-}>\mathrm{CH}_3-\mathrm{CH}_2{ }^{-}>\left(\mathrm{CH}_3\right)_2 \mathrm{CH}^{-}>\left(\mathrm{CH}_3\right)_3 \mathrm{C}^{-}$.
Statement II: Allyl and benzyl carbanions are more stabilised by inductive effect and not by resonance effect.
In the light of the above statements, choose the correct answer from the options given below
Q21.
numerical
+4 / 1
One mole of phenol is treated with dilute $\mathrm{HNO}_3$ at 298 K to give a mixture of products. The mixture is separated by steam distillation. The steam volatile compound $(\mathrm{X})$ is separated. The increase in percentage of oxygen in $(\mathrm{X})$ with respect to phenol is $\_\_\_\_$ $\times 10^{-1} \%$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
Q22.
mcq single
+4 / 1
Consider compounds $\mathrm{A}, \mathrm{B}$ and C with following structural formulae
$$ \begin{aligned} & \mathrm{A}=\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{OH} \\ & \mathrm{~B}=\mathrm{CH}_2=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}_3 \\ & \mathrm{C}=\mathrm{HO}-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}(\mathrm{OH})-\mathrm{CH}_3 \end{aligned} $$
For the conversion of B from A , reagent (D) required is $\_\_\_\_$ and structural formula of product $(\mathrm{E})$ obtained when C undergoes same reaction using excess reagent (D) is $\_\_\_\_$ .
Q23.
mcq single
+4 / 1
Arrange the following atomic orbitals of multi electron atoms in order of increasing energy.
A. $n=3, l=2, m=+1$
B. $n=4, l=0, m=0$
C. $n=6, l=1, m=0$
D. $n=5, l=1, m=+1$
E. $n=2, l=1, m=+1$
Choose the correct answer from the options given below:
Q24.
mcq single
+4 / 1
Identify the correct statements from the following :
A. Heisenberg uncertainty principle is applicable to electrons.
B. The size of $2 p_x$ orbital is less than the size of $3 p_x$ orbital.
C. The energy of 2 s orbital of H atom is equal to the energy of 2 s orbital of Li .
D. The electronic configuration of Cr is $[\mathrm{Ar}] 3 \mathrm{~d}^5 4 \mathrm{~s}^1$
Choose the correct answer from the options given below:
Q25.
mcq single
+4 / 1
$$ \text {Identify the incorrect statements. } $$
 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
 Choose the correct answer from the options given below:
Choose the correct answer from the options given below:
Q26.
mcq single
+4 / 1
$$ \text {Benzyl isocyanide can be obtained from } $$
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q27.
mcq single
+4 / 1
Which of the following is NOT a physical or chemical characteristics of interstitial compounds?
Q28.
numerical
+4 / 1
If the half life of a first order reaction is 6.93 minutes then the time required for completion of $99 \%$ of the reaction will be $\_\_\_\_$ minutes.
(Given $: \log 2=0.3010$ )
Q29.
mcq single
+4 / 1
First order gas phase reaction
$$ \mathrm{A} \rightarrow \mathrm{~B}+\mathrm{C} $$
$p_t=$ initial pressure of gas $\mathrm{A}, p_t=$ total pressure of the reaction mixture at time $t$
Expression of rate constant ( $k$ ) is
Q30.
numerical
+4 / 1
Consider the following species:
$$ \mathrm{BrF}_5, \mathrm{XeF}_5^{-}, \mathrm{BF}_4^{-}, \mathrm{ICl}_4^{-}, \mathrm{XeF}_4, \mathrm{SF}_4, \mathrm{NH}_4^{+}, \mathrm{ClF}_3, \mathrm{XeF}_2, \mathrm{ICl}_2^{-} $$
Number of species having $\mathrm{sp}^3 \mathrm{~d}$ hybridized central atom is $\_\_\_\_$ .
Q31.
mcq single
+4 / 1
What is the mole fraction of water in $10 \%$ by weight $(\mathrm{w} / \mathrm{w})$ of aqueous urea solution?
[Given: Molar mass of $\mathrm{H}, \mathrm{O}, \mathrm{C}$ and N are $1,16,12$ and $14 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.]
Q32.
mcq single
+4 / 1
How many grams of residue is obtained by heating 2.76 g of silver carbonate?
(Given : Molar mass of $\mathrm{C}, \mathrm{O}$ and Ag are 12, 16 and $108 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively)
Q33.
numerical
+4 / 1
The values of pressure equilibrium constant recorded at different temperatures for the following equilibrium reaction have been given below $\mathrm{A}(\mathrm{g}) \rightleftharpoons \mathrm{B}(\mathrm{g})+\mathrm{C}(\mathrm{g})$
$$ \begin{array}{|c|c|} \hline \frac{1}{\mathrm{~T}}\left(\mathrm{~K}^{-1}\right) & \log _{10} \mathrm{~K}_{\mathrm{p}} \\ \hline 0.05 & 3.5 \\ \hline 0.06 & 2.5 \\ \hline 0.07 & 1.5 \\ \hline \end{array} $$
The magnitude of $\frac{\Delta \mathrm{H}^{\circ}}{\mathrm{R}}$ calculated from the above data is $\_\_\_\_$ . (Nearest integer)
Q34.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Sodium dichromate and potassium dichromate are classified as primary standards in titrimetric analysis.
**Statement II :** Phenolphthalein is a weak base, therefore it dissociates in acidic medium.
In the light of the above statements, choose the correct answer from the options given below
Q35.
mcq single
+4 / 1
Arrange the following resultant mixtures in increasing order of their pH values
A. $10 \mathrm{~mL} 0.2 \mathrm{M} \mathrm{Ca}(\mathrm{OH})_2+25 \mathrm{~mL} 0.1 \mathrm{M} \mathrm{HCl}$
B. $10 \mathrm{~mL} 0.01 \mathrm{M} \mathrm{H}_2 \mathrm{SO}_4+10 \mathrm{~mL} 0.01 \mathrm{M} \mathrm{Ca}(\mathrm{OH})_2$
C. $10 \mathrm{~mL} 0.1 \mathrm{M} \mathrm{H}_2 \mathrm{SO}_4+10 \mathrm{~mL} 0.1 \mathrm{M} \mathrm{KOH}$
Choose the correct answer from the options given below :
Q36.
mcq single
+4 / 1
$\mathrm{M}_3 \mathrm{~A}_2$ is a sparingly soluble salt of molar mass $y \mathrm{~g} \mathrm{~mol}^{-1}$ and solubility $x \mathrm{~g} \mathrm{~L}^{-1}$. The ratio of the molar concentration of the anion $\left(\mathrm{A}^{3-}\right)$ to the solubility product of the salt is
Q37.
mcq single
+4 / 1
Given below are two statements:
Statement I: The correct order of electronegativity of fluorine, oxygen and nitrogen is $\mathrm{F}>\mathrm{O}>\mathrm{N}$.
Statement II: The oxidation state of oxygen in $\mathrm{OF}_2$ is +2 and in $\mathrm{Na}_2 \mathrm{O}$ is -2 .
In the light of the above statements, choose the correct answer from the options given below
Q38.
mcq single
+4 / 1
Correct statements from the following are:
A. Nitrogen in oxidation states from +1 to +4 disproportionates in acid medium.
B. Nitrogen has the ability to form $\mathrm{d} \pi$ - $\mathrm{p} \pi$ multiple bonds with itself and other elements with small size and high electronegativity.
C. N-N single bond is stronger than P-P single bond.
D. Nitrogen has highest density in its group due to small size.
E. The maximum covalency of nitrogen is four since it has only four valence orbitals for bonding.
Choose the correct answer from the options given below:
Q39.
mcq single
+4 / 1
The correct statements about metal carbonyls are
A. The metal-carbon bonds in metal carbonyls possess both $\sigma$ and $\pi$ character.
B. Due to synergic bonding interactions between metal and CO ligand, the metal-carbon bond becomes weak.
C. The metal-carbon $\sigma$ bond is formed by the donation of lone pair of electrons on the carbonyl carbon into a vacant orbital of metal.
D. The metal-carbon $\pi$ bond is formed by the donation of electrons from filled d-orbital of metal into vacant $\pi^*$ orbital of CO .
Choose the correct answer from the options given below :
Q40.
mcq single
+4 / 1
Given below are two statements:
Statement I: Each electron in $\mathrm{e}_{\mathrm{g}}$ orbitals destabilizes the orbitals by $+0.6 \Delta_{\mathrm{o}}$ and each electron in the $t_{2 g}$ orbitals stabilizes the orbitals by $-0.4 \Delta_0$ in an octahedral field on the basis of crystal field theory.
Statement II: All the d - orbitals of the transition metals have the same energy in their free atomic state but when a complex is formed the ligands destroy the degeneracy of these orbitals on the basis of crystal field theory.
In the light of the above statements, choose the correct answer from the options given below
Q41.
numerical
+4 / 1
In an estimation of sulphur by Carius method 0.2 g of the substance gave 0.6 g of $\mathrm{BaSO}_4$. The percentage of sulphur in the substance is $\_\_\_\_$%.
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{~S}: 32, \mathrm{BaSO}_4: 231$ )
Q42.
mcq single
+4 / 1
For the following Friedel Craft's alkylation reaction, which of the statements are correct?
 A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
 A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below :
Q43.
mcq single
+4 / 1
Identify the correct statements.
A. Glucose exists in two anomeric forms.
B. Anomers of glucose differ in configuration at $\mathrm{C}-1$ in cyclic hemiacetal structure.
C. Melting point of $\alpha$ - anomer of glucose is greater than $\beta$ - anomer.
D. Specific rotation of $\alpha$ - anomer is $+19^{\circ}$ while for $\beta$ - anomer is $+112^{\circ}$
E. $\alpha$ and $\beta$ - anomers of glucose are prepared by crystallization of saturated glucose solution at 303 K and 371 K respectively.
Choose the correct answer from the options given below :
Q44.
mcq single
+4 / 1
Given below are two statements:
Statement I: On the basis of inductive effect, the order of stability of alkyl carbanions is $\mathrm{CH}_3{ }^{-}>\mathrm{CH}_3-\mathrm{CH}_2{ }^{-}>\left(\mathrm{CH}_3\right)_2 \mathrm{CH}^{-}>\left(\mathrm{CH}_3\right)_3 \mathrm{C}^{-}$.
Statement II: Allyl and benzyl carbanions are more stabilised by inductive effect and not by resonance effect.
In the light of the above statements, choose the correct answer from the options given below
Q45.
numerical
+4 / 1
One mole of phenol is treated with dilute $\mathrm{HNO}_3$ at 298 K to give a mixture of products. The mixture is separated by steam distillation. The steam volatile compound $(\mathrm{X})$ is separated. The increase in percentage of oxygen in $(\mathrm{X})$ with respect to phenol is $\_\_\_\_$ $\times 10^{-1} \%$
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14, \mathrm{O}: 16$ )
Q46.
mcq single
+4 / 1
Consider compounds $\mathrm{A}, \mathrm{B}$ and C with following structural formulae
$$ \begin{aligned} & \mathrm{A}=\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{OH} \\ & \mathrm{~B}=\mathrm{CH}_2=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}_3 \\ & \mathrm{C}=\mathrm{HO}-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}(\mathrm{OH})-\mathrm{CH}_3 \end{aligned} $$
For the conversion of B from A , reagent (D) required is $\_\_\_\_$ and structural formula of product $(\mathrm{E})$ obtained when C undergoes same reaction using excess reagent (D) is $\_\_\_\_$ .
Q47.
mcq single
+4 / 1
Arrange the following atomic orbitals of multi electron atoms in order of increasing energy.
A. $n=3, l=2, m=+1$
B. $n=4, l=0, m=0$
C. $n=6, l=1, m=0$
D. $n=5, l=1, m=+1$
E. $n=2, l=1, m=+1$
Choose the correct answer from the options given below:
Q48.
mcq single
+4 / 1
Identify the correct statements from the following :
A. Heisenberg uncertainty principle is applicable to electrons.
B. The size of $2 p_x$ orbital is less than the size of $3 p_x$ orbital.
C. The energy of 2 s orbital of H atom is equal to the energy of 2 s orbital of Li .
D. The electronic configuration of Cr is $[\mathrm{Ar}] 3 \mathrm{~d}^5 4 \mathrm{~s}^1$
Choose the correct answer from the options given below:
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
The mean deviation about the mean for the data
$$ \begin{array}{|c|c|c|c|c|c|c|} \hline x_i & 5 & 7 & 9 & 10 & 12 & 15 \\ \hline f_i & 8 & 6 & 2 & 2 & 2 & 6 \\ \hline \end{array} $$
$$ \text { is equal to: } $$
Q2.
mcq single
+4 / 1
Let $\vec{a}=\sqrt{7} \hat{i}+\hat{j}-\hat{k}$ and $\vec{b}=\hat{j}+2 \hat{k}$. If $\vec{r}$ is a vector such that $\vec{r} \times \vec{a}+\vec{a} \times \vec{b}=\overrightarrow{0}$ and $\vec{r} \cdot \vec{a}=0$, then $|3 \vec{r}|^2$ is equal to:
Q3.
mcq single
+4 / 1
A letter is known to have arrived by post either from KANPUR or from ANANTPUR. On the envelope just two consecutive letters AN are visible. The probability, that the letter came from ANANTPUR, is:
Q4.
mcq single
+4 / 1
The square of the distance of the point $\mathrm{P}(5,6,7)$ from the line $\frac{x-2}{2}=\frac{y-5}{3}=\frac{z-2}{4}$ is equal to:
Q5.
mcq single
+4 / 1
$\vec{r}=(\hat{i}+\hat{j}-\hat{k})+\lambda(a \hat{i}-\hat{j}), a \neq 0$ and $\vec{r}=(4 \hat{i}-\hat{k})+\mu(2 \hat{i}+a \hat{k})$ from the origin is :
Q6.
mcq single
+4 / 1
Let the sum of the first $n$ terms of an A.P. be $3 n^2+5 n$. Then the sum of squares of the first 10 terms of the A.P. is:
Q7.
mcq single
+4 / 1
$\sum_{n=1}^{10}\left(\frac{528}{n(n+1)(n+2)}\right)$ is equal to:
Q8.
mcq single
+4 / 1
The product of all possible values of $\alpha$, for which
$\lim \limits_{x \rightarrow 0}\left(\frac{1-\cos (\alpha x) \cos ((\alpha+1) x) \cos ((\alpha+2) x)}{\sin ^2((\alpha+1) x)}\right)=2$, is :
Q9.
mcq single
+4 / 1
Let $f: \mathbb{R} \rightarrow \mathbb{R}$ be a differentiable function such that $f\left(\frac{x+y}{3}\right)=\frac{f(x)+f(y)}{3}$ for all $x, y \in \mathbb{R}$, and $f^{\prime}(0)=3$. Then the minimum value of the function $g(x)=3+e^x f(x)$, is:
Q10.
mcq single
+4 / 1
Let $\tan A, \tan B$, where $A, B \in\left(-\frac{\pi}{2}, \frac{\pi}{2}\right)$, be the roots of the quadratic equation $x^2-2 x-5=0$. Then $20 \sin ^2\left(\frac{A+B}{2}\right)$ is equal to:
Q11.
mcq single
+4 / 1
The area of the region $\mathrm{R}=\left\{(x, y): x y \leq 27,1 \leq y \leq x^2\right\}$ is equal to :
Q12.
mcq single
+4 / 1
Consider the system of linear equations in $x, y, z$ :
$$ \begin{aligned} & x+2 y+t z=0 \\ & 6 x+y+5 t z=0 \\ & 3 x+t^2 y+f(t) z=0 \end{aligned} $$
where $f: \mathbb{R} \rightarrow \mathbb{R}$ is a differentiable function. If this system has infinitely many solutions for all $t \in \mathbb{R}$, then $f$
Q13.
mcq single
+4 / 1
Let A be a $3 \times 3$ matrix such that
$$ \mathrm{A}^{\mathrm{T}}\left[\begin{array}{l} 1 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 5 \\ 2 \\ 2 \end{array}\right], \mathrm{A}^{\mathrm{T}}\left[\begin{array}{l} 0 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 3 \\ 1 \\ 1 \end{array}\right], \mathrm{A}\left[\begin{array}{l} 1 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 3 \\ 4 \\ 4 \end{array}\right] \text { and } \mathrm{A}\left[\begin{array}{l} 0 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 1 \\ 3 \\ 1 \end{array}\right] . $$
If $\operatorname{det}(A)=1$, then $\operatorname{det}\left(\operatorname{adj}\left(A^2+A\right)\right)$ is equal to:
Q14.
mcq single
+4 / 1
The sum of all the integral values of $p$ such that the equation $3 \sin ^2 x+12 \cos x-3=p, x \in \mathbb{R}$, has at least one solution, is:
Q15.
numerical
+4 / 1
If $\frac{\pi}{4}+\sum\limits_{p=1}^{11} \tan ^{-1}\left(\frac{2^{p-1}}{1+2^{2 p-1}}\right)=\alpha$, then $\tan \alpha$ is equal to $\_\_\_\_$ .
Q16.
numerical
+4 / 1
If the sum of the coefficients of $x^7$ and $x^{14}$ in the expansion of $\left(\frac{1}{x^3}-x^4\right)^n, x \neq 0$, is zero, then the value of $n$ is $\_\_\_\_$ .
Q17.
mcq single
+4 / 1
In an equilateral triangle $P Q R$, let the vertex $P$ be at $(3,5)$ and the side $Q R$ be along the line $x+y=4$. If the orthocentre of the triangle PQR is $(\alpha, \beta)$, then $9(\alpha+\beta)$ is equal to:
Q18.
mcq single
+4 / 1
Let $a, b \in \mathbb{C}$. Let $\alpha, \beta$ be the roots of the equation $x^2+a x+b=0$. If $\beta-\alpha=\sqrt{11}$ and $\beta^2-\alpha^2=3 i \sqrt{11}$, then $\left(\beta^3-\alpha^3\right)^2$ is equal to:
Q19.
numerical
+4 / 1
Let $\mathrm{A}=\{1,2,3,4,5,6\}$. The number of one-one functions $f: \mathrm{A} \rightarrow \mathrm{A}$ such that $f(1) \geq 3, f(3) \leq 4$ and $f(2)+f(3)=5$, is $\_\_\_\_$ .
Q20.
numerical
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $x \sin \left(\frac{y}{x}\right) d y=\left(y \sin \left(\frac{y}{x}\right)-x\right) d x, y(1)=\frac{\pi}{2}$ and let $\alpha=\cos \left(\frac{y\left(e^{12}\right)}{e^{12}}\right)$. Then the number of integral value of $p$, for which the equation $x^2+y^2-2 p x+2 p y+\alpha+2=0$ represents a circle of radius $r \leq 6$, is $\_\_\_\_$ .
Q21.
mcq single
+4 / 1
Let P be a moving point on the circle $x^2+y^2-6 x-8 y+21=0$. Then, the maximum distance of P from the vertex of the parabola $x^2+6 x+y+13=0$ is equal to:
Q22.
mcq single
+4 / 1
Let a focus of the ellipse $\mathrm{E}: \frac{x^2}{a^2}+\frac{y^2}{b^2}=1$ be $\mathrm{S}(4,0)$ and its eccentricity be $\frac{4}{5}$. If the point $\mathrm{P}(3, \alpha)$ lies on E and O is the origin, then the area of $\triangle \mathrm{POS}$ is equal to:
Q23.
numerical
+4 / 1
Two players A and B play a series of games of badminton. The player, who wins 5 games first, wins the series. Assuming that no game ends in a draw, the number of ways, in which player A wins the series is $\_\_\_\_$ .
Q24.
mcq single
+4 / 1
The value of the integral $\int\limits_{\frac{\pi}{6}}^{\frac{\pi}{3}}\left(\frac{4-\operatorname{cosec}^2 x}{\cos ^4 x}\right) d x$ is :
Q25.
mcq single
+4 / 1
The value of the integral $\int\limits_0^{\infty} \frac{\log _e(x)}{x^2+4} d x$ is:
Q26.
mcq single
+4 / 1
The mean deviation about the mean for the data
$$ \begin{array}{|c|c|c|c|c|c|c|} \hline x_i & 5 & 7 & 9 & 10 & 12 & 15 \\ \hline f_i & 8 & 6 & 2 & 2 & 2 & 6 \\ \hline \end{array} $$
$$ \text { is equal to: } $$
Q27.
mcq single
+4 / 1
Let $\vec{a}=\sqrt{7} \hat{i}+\hat{j}-\hat{k}$ and $\vec{b}=\hat{j}+2 \hat{k}$. If $\vec{r}$ is a vector such that $\vec{r} \times \vec{a}+\vec{a} \times \vec{b}=\overrightarrow{0}$ and $\vec{r} \cdot \vec{a}=0$, then $|3 \vec{r}|^2$ is equal to:
Q28.
mcq single
+4 / 1
A letter is known to have arrived by post either from KANPUR or from ANANTPUR. On the envelope just two consecutive letters AN are visible. The probability, that the letter came from ANANTPUR, is:
Q29.
mcq single
+4 / 1
The square of the distance of the point $\mathrm{P}(5,6,7)$ from the line $\frac{x-2}{2}=\frac{y-5}{3}=\frac{z-2}{4}$ is equal to:
Q30.
mcq single
+4 / 1
$\vec{r}=(\hat{i}+\hat{j}-\hat{k})+\lambda(a \hat{i}-\hat{j}), a \neq 0$ and $\vec{r}=(4 \hat{i}-\hat{k})+\mu(2 \hat{i}+a \hat{k})$ from the origin is :
Q31.
mcq single
+4 / 1
Let the sum of the first $n$ terms of an A.P. be $3 n^2+5 n$. Then the sum of squares of the first 10 terms of the A.P. is:
Q32.
mcq single
+4 / 1
$\sum_{n=1}^{10}\left(\frac{528}{n(n+1)(n+2)}\right)$ is equal to:
Q33.
mcq single
+4 / 1
The product of all possible values of $\alpha$, for which
$\lim \limits_{x \rightarrow 0}\left(\frac{1-\cos (\alpha x) \cos ((\alpha+1) x) \cos ((\alpha+2) x)}{\sin ^2((\alpha+1) x)}\right)=2$, is :
Q34.
mcq single
+4 / 1
Let $f: \mathbb{R} \rightarrow \mathbb{R}$ be a differentiable function such that $f\left(\frac{x+y}{3}\right)=\frac{f(x)+f(y)}{3}$ for all $x, y \in \mathbb{R}$, and $f^{\prime}(0)=3$. Then the minimum value of the function $g(x)=3+e^x f(x)$, is:
Q35.
mcq single
+4 / 1
Let $\tan A, \tan B$, where $A, B \in\left(-\frac{\pi}{2}, \frac{\pi}{2}\right)$, be the roots of the quadratic equation $x^2-2 x-5=0$. Then $20 \sin ^2\left(\frac{A+B}{2}\right)$ is equal to:
Q36.
mcq single
+4 / 1
The area of the region $\mathrm{R}=\left\{(x, y): x y \leq 27,1 \leq y \leq x^2\right\}$ is equal to :
Q37.
mcq single
+4 / 1
Consider the system of linear equations in $x, y, z$ :
$$ \begin{aligned} & x+2 y+t z=0 \\ & 6 x+y+5 t z=0 \\ & 3 x+t^2 y+f(t) z=0 \end{aligned} $$
where $f: \mathbb{R} \rightarrow \mathbb{R}$ is a differentiable function. If this system has infinitely many solutions for all $t \in \mathbb{R}$, then $f$
Q38.
mcq single
+4 / 1
Let A be a $3 \times 3$ matrix such that
$$ \mathrm{A}^{\mathrm{T}}\left[\begin{array}{l} 1 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 5 \\ 2 \\ 2 \end{array}\right], \mathrm{A}^{\mathrm{T}}\left[\begin{array}{l} 0 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 3 \\ 1 \\ 1 \end{array}\right], \mathrm{A}\left[\begin{array}{l} 1 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 3 \\ 4 \\ 4 \end{array}\right] \text { and } \mathrm{A}\left[\begin{array}{l} 0 \\ 0 \\ 1 \end{array}\right]=\left[\begin{array}{l} 1 \\ 3 \\ 1 \end{array}\right] . $$
If $\operatorname{det}(A)=1$, then $\operatorname{det}\left(\operatorname{adj}\left(A^2+A\right)\right)$ is equal to:
Q39.
mcq single
+4 / 1
The sum of all the integral values of $p$ such that the equation $3 \sin ^2 x+12 \cos x-3=p, x \in \mathbb{R}$, has at least one solution, is:
Q40.
numerical
+4 / 1
If $\frac{\pi}{4}+\sum\limits_{p=1}^{11} \tan ^{-1}\left(\frac{2^{p-1}}{1+2^{2 p-1}}\right)=\alpha$, then $\tan \alpha$ is equal to $\_\_\_\_$ .
Q41.
numerical
+4 / 1
If the sum of the coefficients of $x^7$ and $x^{14}$ in the expansion of $\left(\frac{1}{x^3}-x^4\right)^n, x \neq 0$, is zero, then the value of $n$ is $\_\_\_\_$ .
Q42.
mcq single
+4 / 1
In an equilateral triangle $P Q R$, let the vertex $P$ be at $(3,5)$ and the side $Q R$ be along the line $x+y=4$. If the orthocentre of the triangle PQR is $(\alpha, \beta)$, then $9(\alpha+\beta)$ is equal to:
Q43.
mcq single
+4 / 1
Let $a, b \in \mathbb{C}$. Let $\alpha, \beta$ be the roots of the equation $x^2+a x+b=0$. If $\beta-\alpha=\sqrt{11}$ and $\beta^2-\alpha^2=3 i \sqrt{11}$, then $\left(\beta^3-\alpha^3\right)^2$ is equal to:
Q44.
numerical
+4 / 1
Let $\mathrm{A}=\{1,2,3,4,5,6\}$. The number of one-one functions $f: \mathrm{A} \rightarrow \mathrm{A}$ such that $f(1) \geq 3, f(3) \leq 4$ and $f(2)+f(3)=5$, is $\_\_\_\_$ .
Q45.
numerical
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $x \sin \left(\frac{y}{x}\right) d y=\left(y \sin \left(\frac{y}{x}\right)-x\right) d x, y(1)=\frac{\pi}{2}$ and let $\alpha=\cos \left(\frac{y\left(e^{12}\right)}{e^{12}}\right)$. Then the number of integral value of $p$, for which the equation $x^2+y^2-2 p x+2 p y+\alpha+2=0$ represents a circle of radius $r \leq 6$, is $\_\_\_\_$ .
Q46.
mcq single
+4 / 1
Let P be a moving point on the circle $x^2+y^2-6 x-8 y+21=0$. Then, the maximum distance of P from the vertex of the parabola $x^2+6 x+y+13=0$ is equal to:
Q47.
mcq single
+4 / 1
Let a focus of the ellipse $\mathrm{E}: \frac{x^2}{a^2}+\frac{y^2}{b^2}=1$ be $\mathrm{S}(4,0)$ and its eccentricity be $\frac{4}{5}$. If the point $\mathrm{P}(3, \alpha)$ lies on E and O is the origin, then the area of $\triangle \mathrm{POS}$ is equal to:
Q48.
numerical
+4 / 1
Two players A and B play a series of games of badminton. The player, who wins 5 games first, wins the series. Assuming that no game ends in a draw, the number of ways, in which player A wins the series is $\_\_\_\_$ .
Q49.
mcq single
+4 / 1
The value of the integral $\int\limits_{\frac{\pi}{6}}^{\frac{\pi}{3}}\left(\frac{4-\operatorname{cosec}^2 x}{\cos ^4 x}\right) d x$ is :
Q50.
mcq single
+4 / 1
The value of the integral $\int\limits_0^{\infty} \frac{\log _e(x)}{x^2+4} d x$ is:
Physics
Physics
Q1.
mcq single
+4 / 1
For the given logic circuit, which of the following inputs combination will make both LED-1 and LED-2 to glow?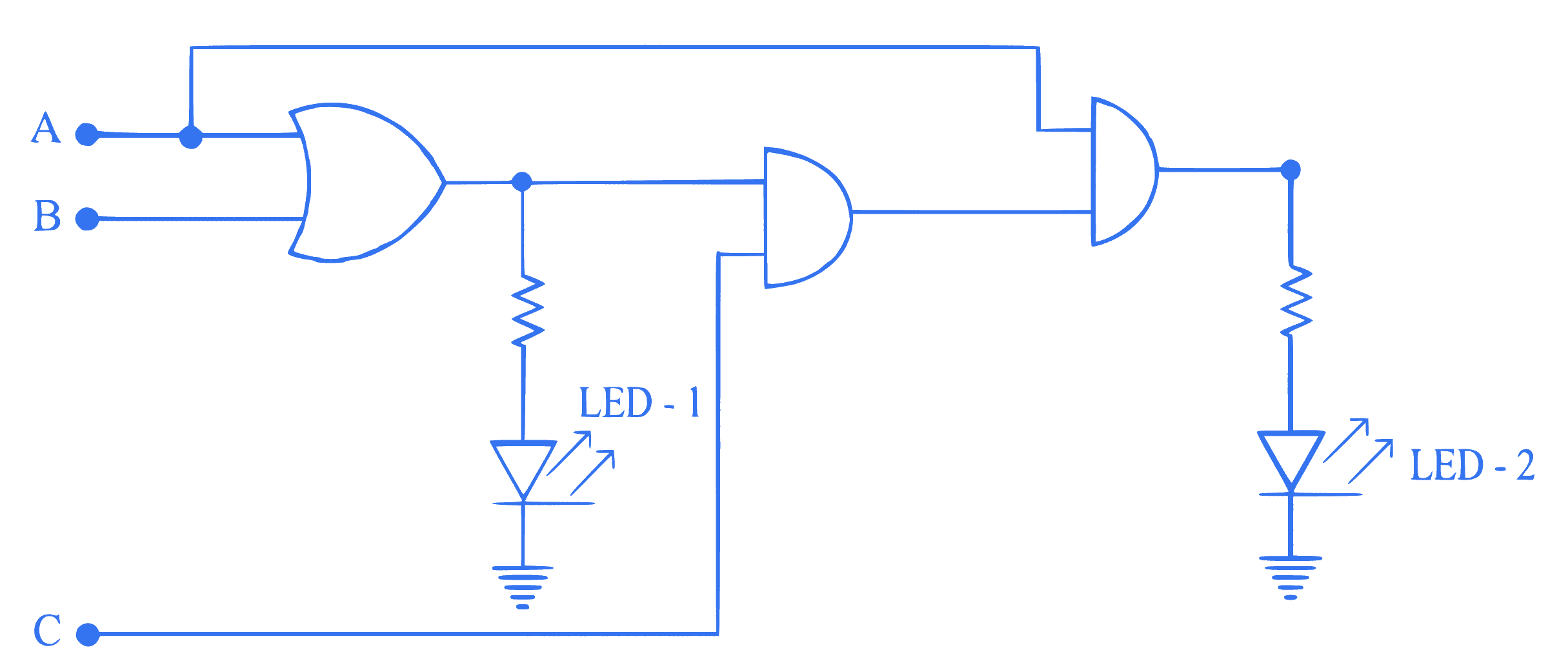

Q2.
mcq single
+4 / 1
In Young's double slit experiment, the fringe width of the interference pattern produced on the screen is $2.4 \mu \mathrm{~m}$. If the experiment is carried out in another medium having refractive index 1.2 , the fringe width will be $\_\_\_\_$ $\mu \mathrm{m}$.
Q3.
numerical
+4 / 1
In the given circuit below inductance values of $L_1, L_2$ and $L_3$ are same. The magnetic energy stored in the entire circuit is $\left(U_t\right)$ and that stored in the $\mathrm{L}_2$ inductor is $\left(U_l\right)$. $U_t / U_l$ is $\_\_\_\_$ .
(Ignore the mutual inductance if any)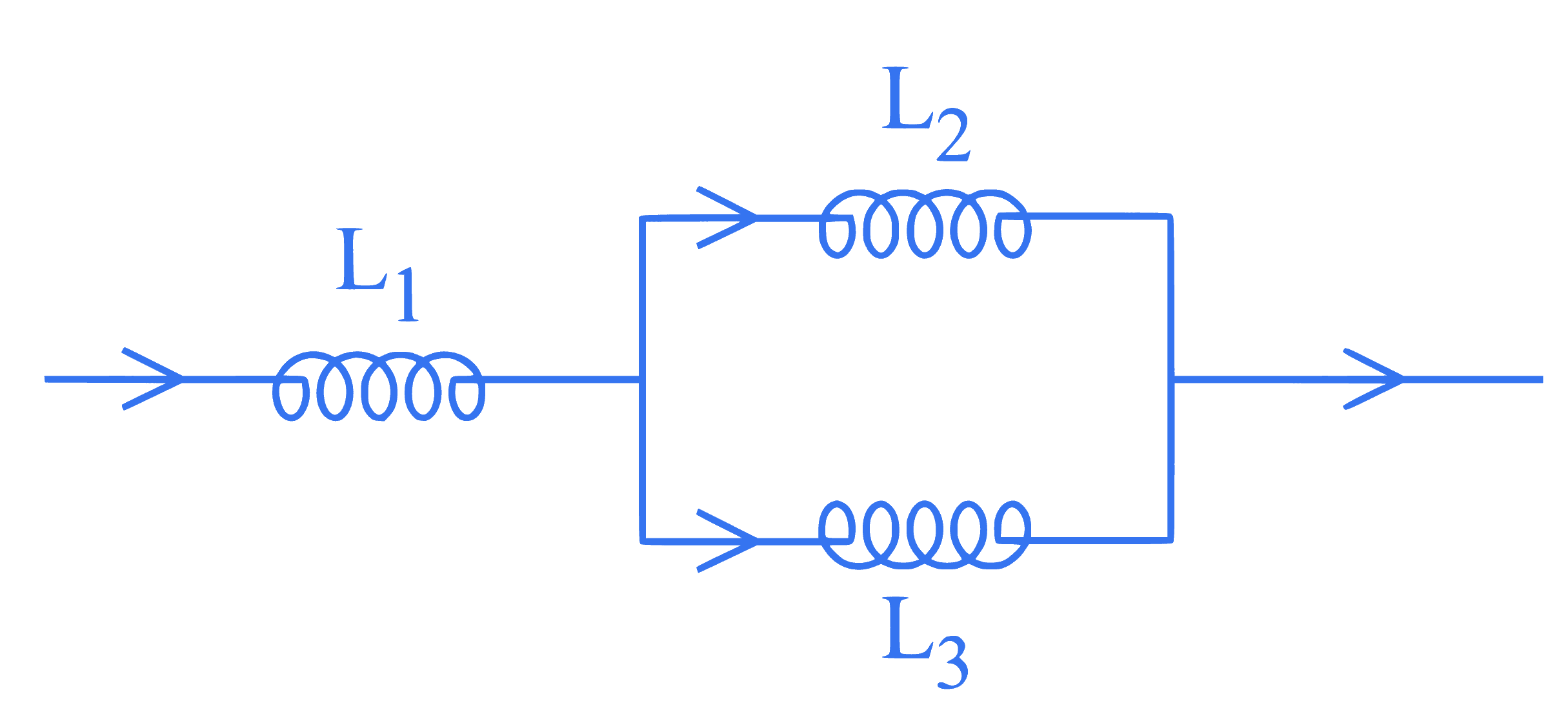

Q4.
mcq single
+4 / 1
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason $\mathbf{R}$
**Statement I :** Change in internal energy of a system containing $n$ mole of ideal gas can be written as $\Delta \mathrm{U}=n \mathrm{C}_v\left(T_{\mathrm{f}}-T_i\right)=\frac{n R}{\gamma-1}\left(T_{\mathrm{f}}-T_i\right)$, where $\gamma=\frac{C_p}{C_v}, T_i=$ initial temperature, $T_{\mathrm{f}}=$ final temperature.
**Statement II :** Relation between degree of freedom $f$ and $\gamma\left(=C_p / C_v\right)$ is $\left(\gamma=1+\frac{2}{f}\right)$
Choose the correct answer from the options given below
Q5.
mcq single
+4 / 1
Consider the following statements:
A. Zeroth law of thermodynamics gives concept of temperature
B. First law of thermodynamics gives concept of internal energy
C. In isothermal expansion of ideal gas, $\Delta Q \neq \Delta W$
D. Product of intensive and extensive variables is extensive
E. The ratio of any extensive variable to mass will be an extensive variable
Choose the correct combination of statements from the options given below:
Q6.
mcq single
+4 / 1
The Young's modulus of steel wire of radius $r$ and length $L$ is $Y$.
If the radius $r$ and length $L$ of the wire are doubled then the value of $Y$
Q7.
numerical
+4 / 1
A cube has side length 5 cm and modulus of rigidity $10^5 \mathrm{~N} / \mathrm{m}^2$. The displacement produced by a force of 10 N in the upper face of cube is $\_\_\_\_$ mm.
Q8.
mcq single
+4 / 1
Two wires as shown in the figure below, made of steel and have breaking stress of $12 \times 10^8 \mathrm{~N} / \mathrm{m}^2$. Area of cross-section of upper wire is $0.008 \mathrm{~cm}^2$ and of lower wire is $0.004 \mathrm{~cm}^2$. The maximum mass that can be added to pan without breaking any wire is $\_\_\_\_$ kg.


Q9.
mcq single
+4 / 1
An electron of mass $m$ is moving in an electric field $\vec{E}=-2 E_{\mathrm{o}} \hat{i}\left(E_{\mathrm{o}}=\right.$ constant $\left.>0\right)$, with an initial velocity $\vec{V}=v_{\mathrm{o}} \hat{i} \left(v_{\mathrm{o}}=\right.$ constant $\left.>0\right)$. If $\lambda_{\mathrm{o}}=\frac{h}{4 m v_{\mathrm{o}}}$, its de Broglie wavelength at time $t$ is
$\_\_\_\_$ .
( $e=$ charge of electron)
Q10.
mcq single
+4 / 1
Light source having wavelength 331 nm is used to generate photo-electrons whose stopping potential is 0.2 V . The work function of the used metal in the experiment is $\alpha \times 10^{-19} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
$$ \left(\mathrm{h}=6.62 \times 10^{-34} \mathrm{~J} \mathrm{~s}, \mathrm{e}=1.6 \times 10^{-19} \mathrm{C} \text { and } \mathrm{c}=3 \times 10^8 \mathrm{~m} / \mathrm{s}\right) $$
Q11.
mcq single
+4 / 1
When one moves from a point 16 km below the earth's surface to a point 16 km above the earth's surface. The change in g is approximately $\alpha \%$. The value of $\alpha$ is $\_\_\_\_$ .
(Take radius of the earth $=6400 \mathrm{~km}$.)
Q12.
mcq single
+4 / 1
A compound microscope is designed with two symmetric biconvex lenses. The objective lens is cut vertically, creating two identical plano-convex lenses. One of them is used in place of original objective lens. To retain same magnification keeping the object distance unchanged, the tube length has to be
Q13.
mcq single
+4 / 1
A ray of light passing through an equilateral prism is having velocity $2.12 \times 10^8 \mathrm{~m} / \mathrm{s}$ in the prism material, then the minimum angle of deviation is
$\_\_\_\_$ degrees.
Q14.
numerical
+4 / 1
The charged particle moving in a uniform magnetic field of $(3 \hat{i}+2 \hat{j}) \mathrm{T}$ has an acceleration $\left(4 \hat{i}-\frac{x}{2} \hat{j}\right) \mathrm{m} / \mathrm{s}^2$. The value of $x$ is
Q15.
mcq single
+4 / 1
$L, C$ and $R$ represents physical quantities inductance, capacitance and resistance respectively. The dimensional formula $\mathrm{ML}^2 \mathrm{~T}^{-4} \mathrm{~A}^{-2}$ corresponds to $\_\_\_\_$ .
Q16.
mcq single
+4 / 1
In a Vernier calipers, when both jaws touch each other, zero of the Vernier scale is shifted to the right of zero of the main scale and $7^{\text {th }}$ Vernier division coincides with a main scale reading. If the value of 1 main scale division is 1 mm and there are 10 Vernier scale divisions, then the Vernier caliper has
Q17.
numerical
+4 / 1
From 18 m height above the ground a ball is dropped from rest . The height above the ground at which the magnitude of velocity equal to the magnitude of acceleration (in the same set of units) due to gravity is $\_\_\_\_$ m.
(Take $g=10 \mathrm{~m} / \mathrm{s}^2$ and neglect the air resistance)
Q18.
mcq single
+4 / 1
In the hydrogen atom, the electron makes a transition from the higher orbit (i) to a lower orbit $(f)$. The ratio of the radius of the orbits in given by $r_i: r_f=16: 4$. The wavelength of photon emitted due to this transition is $\_\_\_\_$ nm.
(Given Rydberg constant $=1.0973 \times 10^7 / \mathrm{m}$ )
Q19.
numerical
+4 / 1
A transverse wave on a string is described by $y=3 \sin (36 t+0.018 x+\pi / 4)$. where $x, y$ are in cm and $t$ in seconds. The least distance between the two successive crests in the wave is $\_\_\_\_$ cm . (Nearest integer)
$$ (\pi=3.14) $$
Q20.
mcq single
+4 / 1
Three masses $m_1=4 \mathrm{~kg}, m_2=4 \mathrm{~kg}$ and $m_3=6 \mathrm{~kg}$ are suspended from a fixed smooth frictionless pully as shown in the figure below. The value of $T_1 / T_2$ is
$\_\_\_\_$
(take $g=10 \mathrm{~m} / \mathrm{s}^2$ )

Q21.
mcq single
+4 / 1
A wedge $Y$ with mass of 10 kg and all frictionless surfaces and the inclined surface making $37^{\circ}$ with horizontal. A block $X$ with mass 2 kg is placed at the highest point of the wedge as shown in figure is at rest. At $t=0$ wedge ( $Y$ ) is pulled toward right with constant force $(f)$ of 24 N . Taking the block $X$ at rest at $t=0$, the time taken by it to slide down 8.8 m on the slope, while $Y$ is on the move, is $\_\_\_\_$ s.
$\left(\right.$ take $\tan \left(37^{\circ}\right)=3 / 4$ and $\left.\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2\right)$

Q22.
mcq single
+4 / 1
Refer to the figure given below, current between terminals $A$ and $B$ is
$\_\_\_\_$ A.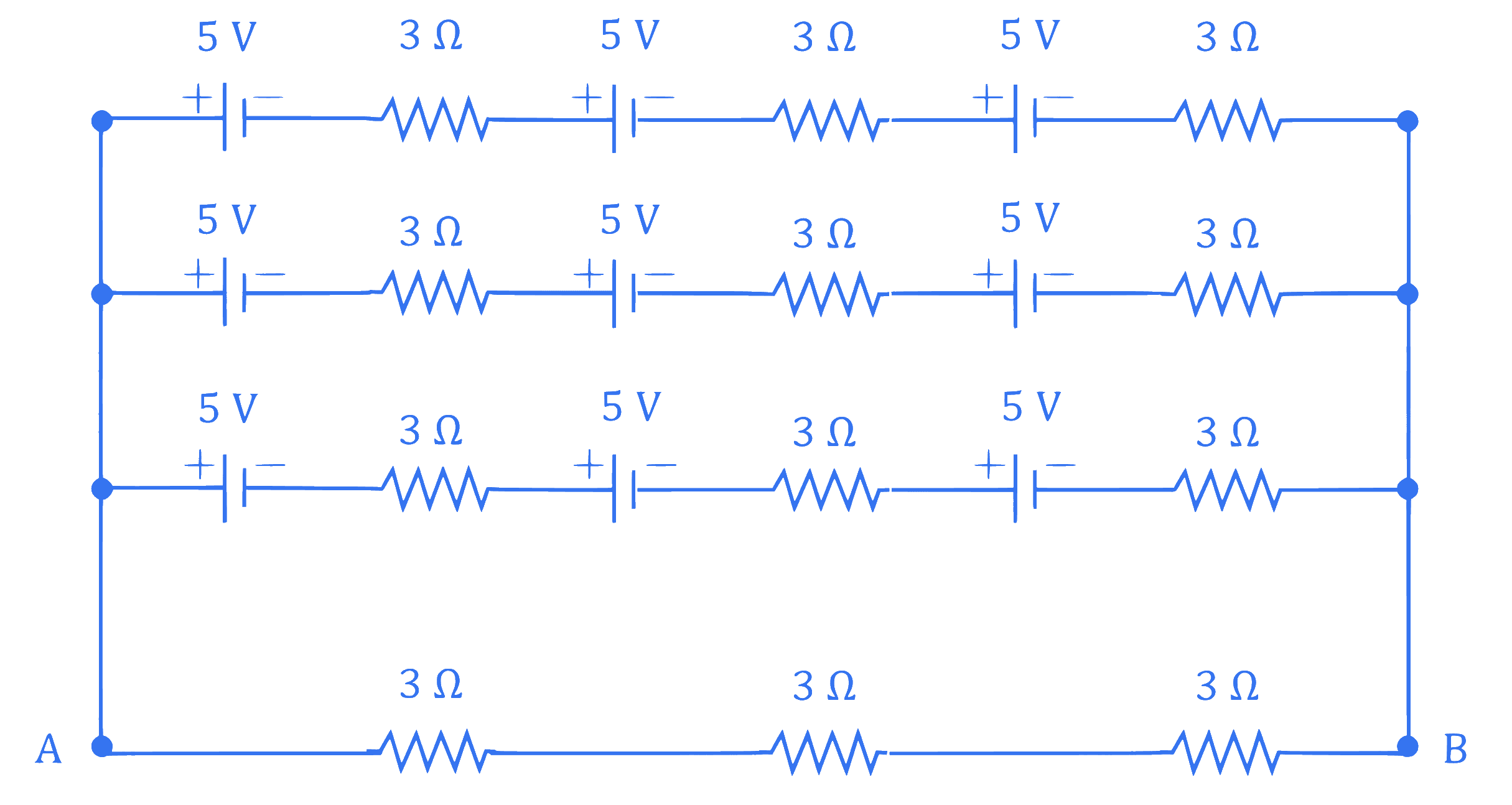

Q23.
mcq single
+4 / 1
Refer to the figure given below. The values of $I_1, I_2$ and $I_3$ are $\_\_\_\_$ .


Q24.
mcq single
+4 / 1
A displacement current of 4.0 A can be set up in the space between two parallel plates of $6 \mu \mathrm{~F}$ capacitor. The rate of change of potential difference across the plates of the capacitor is nearly $\alpha \times 10^6 \mathrm{~V} / \mathrm{s}$. The value of $\alpha$ is $\_\_\_\_$ .
Q25.
mcq single
+4 / 1
An a.c. source of angular frequency $\omega$ is connected across a resistor $R$ and a capacitor $C$ in series. The current is observed as $I$. Now the frequency of the source is changed to $\omega / 4$, (keeping the voltage unchanged) the current is found to be $I / 3$. The ratio of resistance to reactance at frequency $\omega$ is
Q26.
mcq single
+4 / 1
For the given logic circuit, which of the following inputs combination will make both LED-1 and LED-2 to glow?

Q27.
mcq single
+4 / 1
In Young's double slit experiment, the fringe width of the interference pattern produced on the screen is $2.4 \mu \mathrm{~m}$. If the experiment is carried out in another medium having refractive index 1.2 , the fringe width will be $\_\_\_\_$ $\mu \mathrm{m}$.
Q28.
numerical
+4 / 1
In the given circuit below inductance values of $L_1, L_2$ and $L_3$ are same. The magnetic energy stored in the entire circuit is $\left(U_t\right)$ and that stored in the $\mathrm{L}_2$ inductor is $\left(U_l\right)$. $U_t / U_l$ is $\_\_\_\_$ .
(Ignore the mutual inductance if any)

Q29.
mcq single
+4 / 1
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason $\mathbf{R}$
**Statement I :** Change in internal energy of a system containing $n$ mole of ideal gas can be written as $\Delta \mathrm{U}=n \mathrm{C}_v\left(T_{\mathrm{f}}-T_i\right)=\frac{n R}{\gamma-1}\left(T_{\mathrm{f}}-T_i\right)$, where $\gamma=\frac{C_p}{C_v}, T_i=$ initial temperature, $T_{\mathrm{f}}=$ final temperature.
**Statement II :** Relation between degree of freedom $f$ and $\gamma\left(=C_p / C_v\right)$ is $\left(\gamma=1+\frac{2}{f}\right)$
Choose the correct answer from the options given below
Q30.
mcq single
+4 / 1
Consider the following statements:
A. Zeroth law of thermodynamics gives concept of temperature
B. First law of thermodynamics gives concept of internal energy
C. In isothermal expansion of ideal gas, $\Delta Q \neq \Delta W$
D. Product of intensive and extensive variables is extensive
E. The ratio of any extensive variable to mass will be an extensive variable
Choose the correct combination of statements from the options given below:
Q31.
mcq single
+4 / 1
The Young's modulus of steel wire of radius $r$ and length $L$ is $Y$.
If the radius $r$ and length $L$ of the wire are doubled then the value of $Y$
Q32.
numerical
+4 / 1
A cube has side length 5 cm and modulus of rigidity $10^5 \mathrm{~N} / \mathrm{m}^2$. The displacement produced by a force of 10 N in the upper face of cube is $\_\_\_\_$ mm.
Q33.
mcq single
+4 / 1
Two wires as shown in the figure below, made of steel and have breaking stress of $12 \times 10^8 \mathrm{~N} / \mathrm{m}^2$. Area of cross-section of upper wire is $0.008 \mathrm{~cm}^2$ and of lower wire is $0.004 \mathrm{~cm}^2$. The maximum mass that can be added to pan without breaking any wire is $\_\_\_\_$ kg.


Q34.
mcq single
+4 / 1
An electron of mass $m$ is moving in an electric field $\vec{E}=-2 E_{\mathrm{o}} \hat{i}\left(E_{\mathrm{o}}=\right.$ constant $\left.>0\right)$, with an initial velocity $\vec{V}=v_{\mathrm{o}} \hat{i} \left(v_{\mathrm{o}}=\right.$ constant $\left.>0\right)$. If $\lambda_{\mathrm{o}}=\frac{h}{4 m v_{\mathrm{o}}}$, its de Broglie wavelength at time $t$ is
$\_\_\_\_$ .
( $e=$ charge of electron)
Q35.
mcq single
+4 / 1
Light source having wavelength 331 nm is used to generate photo-electrons whose stopping potential is 0.2 V . The work function of the used metal in the experiment is $\alpha \times 10^{-19} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
$$ \left(\mathrm{h}=6.62 \times 10^{-34} \mathrm{~J} \mathrm{~s}, \mathrm{e}=1.6 \times 10^{-19} \mathrm{C} \text { and } \mathrm{c}=3 \times 10^8 \mathrm{~m} / \mathrm{s}\right) $$
Q36.
mcq single
+4 / 1
When one moves from a point 16 km below the earth's surface to a point 16 km above the earth's surface. The change in g is approximately $\alpha \%$. The value of $\alpha$ is $\_\_\_\_$ .
(Take radius of the earth $=6400 \mathrm{~km}$.)
Q37.
mcq single
+4 / 1
A compound microscope is designed with two symmetric biconvex lenses. The objective lens is cut vertically, creating two identical plano-convex lenses. One of them is used in place of original objective lens. To retain same magnification keeping the object distance unchanged, the tube length has to be
Q38.
mcq single
+4 / 1
A ray of light passing through an equilateral prism is having velocity $2.12 \times 10^8 \mathrm{~m} / \mathrm{s}$ in the prism material, then the minimum angle of deviation is
$\_\_\_\_$ degrees.
Q39.
numerical
+4 / 1
The charged particle moving in a uniform magnetic field of $(3 \hat{i}+2 \hat{j}) \mathrm{T}$ has an acceleration $\left(4 \hat{i}-\frac{x}{2} \hat{j}\right) \mathrm{m} / \mathrm{s}^2$. The value of $x$ is
Q40.
mcq single
+4 / 1
$L, C$ and $R$ represents physical quantities inductance, capacitance and resistance respectively. The dimensional formula $\mathrm{ML}^2 \mathrm{~T}^{-4} \mathrm{~A}^{-2}$ corresponds to $\_\_\_\_$ .
Q41.
mcq single
+4 / 1
In a Vernier calipers, when both jaws touch each other, zero of the Vernier scale is shifted to the right of zero of the main scale and $7^{\text {th }}$ Vernier division coincides with a main scale reading. If the value of 1 main scale division is 1 mm and there are 10 Vernier scale divisions, then the Vernier caliper has
Q42.
numerical
+4 / 1
From 18 m height above the ground a ball is dropped from rest . The height above the ground at which the magnitude of velocity equal to the magnitude of acceleration (in the same set of units) due to gravity is $\_\_\_\_$ m.
(Take $g=10 \mathrm{~m} / \mathrm{s}^2$ and neglect the air resistance)
Q43.
mcq single
+4 / 1
In the hydrogen atom, the electron makes a transition from the higher orbit (i) to a lower orbit $(f)$. The ratio of the radius of the orbits in given by $r_i: r_f=16: 4$. The wavelength of photon emitted due to this transition is $\_\_\_\_$ nm.
(Given Rydberg constant $=1.0973 \times 10^7 / \mathrm{m}$ )
Q44.
numerical
+4 / 1
A transverse wave on a string is described by $y=3 \sin (36 t+0.018 x+\pi / 4)$. where $x, y$ are in cm and $t$ in seconds. The least distance between the two successive crests in the wave is $\_\_\_\_$ cm . (Nearest integer)
$$ (\pi=3.14) $$
Q45.
mcq single
+4 / 1
Three masses $m_1=4 \mathrm{~kg}, m_2=4 \mathrm{~kg}$ and $m_3=6 \mathrm{~kg}$ are suspended from a fixed smooth frictionless pully as shown in the figure below. The value of $T_1 / T_2$ is
$\_\_\_\_$
(take $g=10 \mathrm{~m} / \mathrm{s}^2$ )

Q46.
mcq single
+4 / 1
A wedge $Y$ with mass of 10 kg and all frictionless surfaces and the inclined surface making $37^{\circ}$ with horizontal. A block $X$ with mass 2 kg is placed at the highest point of the wedge as shown in figure is at rest. At $t=0$ wedge ( $Y$ ) is pulled toward right with constant force $(f)$ of 24 N . Taking the block $X$ at rest at $t=0$, the time taken by it to slide down 8.8 m on the slope, while $Y$ is on the move, is $\_\_\_\_$ s.
$\left(\right.$ take $\tan \left(37^{\circ}\right)=3 / 4$ and $\left.\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2\right)$

Q47.
mcq single
+4 / 1
Refer to the figure given below, current between terminals $A$ and $B$ is
$\_\_\_\_$ A.

Q48.
mcq single
+4 / 1
Refer to the figure given below. The values of $I_1, I_2$ and $I_3$ are $\_\_\_\_$ .


Q49.
mcq single
+4 / 1
A displacement current of 4.0 A can be set up in the space between two parallel plates of $6 \mu \mathrm{~F}$ capacitor. The rate of change of potential difference across the plates of the capacitor is nearly $\alpha \times 10^6 \mathrm{~V} / \mathrm{s}$. The value of $\alpha$ is $\_\_\_\_$ .
Q50.
mcq single
+4 / 1
An a.c. source of angular frequency $\omega$ is connected across a resistor $R$ and a capacitor $C$ in series. The current is observed as $I$. Now the frequency of the source is changed to $\omega / 4$, (keeping the voltage unchanged) the current is found to be $I / 3$. The ratio of resistance to reactance at frequency $\omega$ is