JEE Main 2026 (Online) 6th April Evening Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Consider the following organic reaction sequence. Choose the final product $(X)$ from the following (consider the major product in all intermediate reactions)
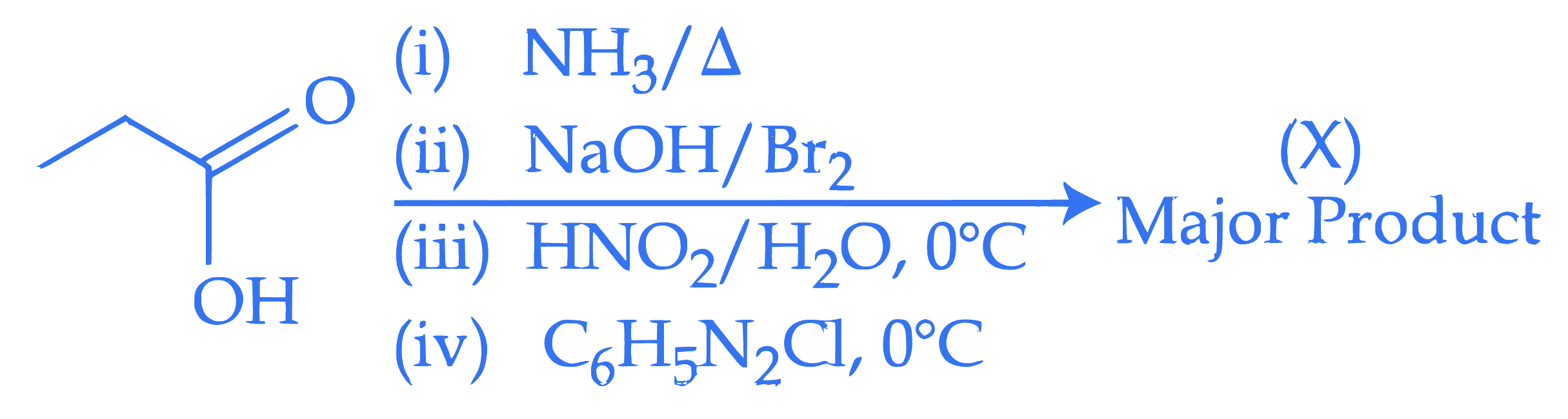
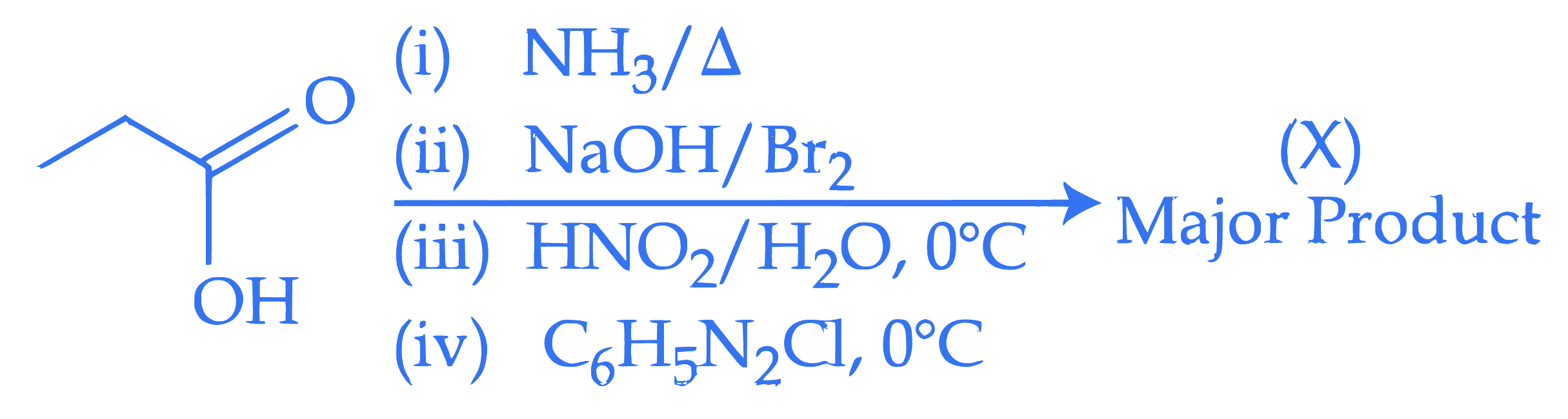
Q2.
mcq single
+4 / 1
The number of compounds from the following which can undergo reaction with $\mathrm{Br}_2 / \mathrm{KOH}$ (alcoholic) to give respective products and these respective products can also be obtained separately by Gabriel phthalimide reaction is :
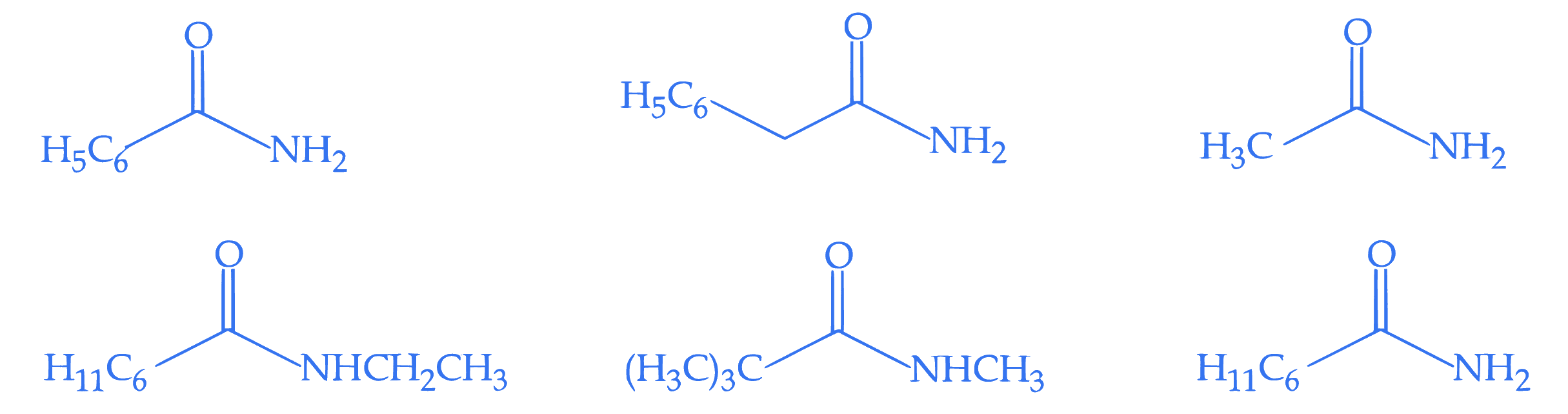

Q3.
mcq single
+4 / 1
The correct order of first $\left(\Delta_i \mathrm{H}_1\right)$ and second $\left(\Delta_i \mathrm{H}_2\right)$ ionisation enthalpy values of Cr and Mn are :
A. $\Delta_i \mathrm{H}_1: \mathrm{Cr}>\mathrm{Mn}$
B. $\Delta_i \mathrm{H}_2: \mathrm{Cr}>\mathrm{Mn}$
C. $\Delta_i \mathrm{H}_1: \mathrm{Mn}>\mathrm{Cr}$
D. $\Delta_i \mathrm{H}_2: \mathrm{Mn}>\mathrm{Cr}$
Choose the correct answer from the options given below :
Q4.
numerical
+4 / 1
Decomposition of a hydrocarbon follows the equation $\mathrm{k}=\left(5.5 \times 10^{11} \mathrm{~s}^{-1}\right) \mathrm{e}^{\frac{-28000 \mathrm{~K}}{\mathrm{~T}}}$. The activation energy of reaction is $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (Nearest Integer)
Given : $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$
Q5.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\mathrm{F}_2 \mathrm{O}<\mathrm{H}_2 \mathrm{O}<\mathrm{Cl}_2 \mathrm{O}$ is the correct trend in terms of bond angle.
**Statement II :** $\mathrm{SiF}_4, \mathrm{SnF}_4$ and $\mathrm{PbF}_4$ are ionic in nature.
In the light of the above statements, choose the correct answer from the options given below :
Q6.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of compounds among $\mathrm{SO}_2, \mathrm{SO}_3, \mathrm{SF}_4, \mathrm{SF}_6$ and $\mathrm{H}_2 \mathrm{~S}$ in which sulphur does not obey the Octet rule is 3 .
**Statement II :** Among $\left[\mathrm{H}_2 \mathrm{O}, \mathrm{ClF}_3, \mathrm{SF}_4\right],\left[\mathrm{NH}_3, \mathrm{BrF}_5, \mathrm{SF}_4\right],\left[\mathrm{BrF}_5, \mathrm{ClF}_3, \mathrm{XeF}_4\right]$ and [ $\mathrm{XeF}_4, \mathrm{ClF}_3, \mathrm{H}_2 \mathrm{O}$ ], the number of sets in which all the molecules have one lone pair of electrons on the central atom is 1 .
In the light of the above statements, choose the correct answer from the options given below :
Q7.
numerical
+4 / 1
500 mL of $0.2 \mathrm{M} \mathrm{MnO}_4^{-}$solution in basic medium when mixed with 500 mL of 1.5 M KI solution, oxidises iodide ions to liberate molecular iodine. This liberated iodine is then titrated with a standard $x \mathrm{M}$ thiosulphate solution in presence of starch till the end point. If 300 mL of thiosulphate was consumed, then the value of $x$ is $\_\_\_\_$。
Q8.
mcq single
+4 / 1
Given below are two statements :
 **Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
 **Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
Q9.
mcq single
+4 / 1
Which of the following contain the same number of atoms?
(Given : Molar mass in $\mathrm{g} \mathrm{mol}^{-1}$ of $\mathrm{H}, \mathrm{He}, \mathrm{O}$ and S are 1, 4, 16 and 32 respectively)
A. 2 g of $\mathrm{O}_2$ gas
B. 4 g of $\mathrm{SO}_2$ gas
C. 1400 mL of $\mathrm{O}_2$ at STP
D. $0.05 \mathrm{~L}$ of He at STP
E. $ 0.0625 \mathrm{~mol}$ of $\mathrm{H}_2$ gas
Choose the correct answer from the options given below :
Q10.
numerical
+4 / 1
In a closed flask at 600 K , one mole of $\mathrm{X}_2 \mathrm{Y}_4(\mathrm{~g})$ attains equilibrium as given below :
$$ \mathrm{X}_2 \mathrm{Y}_4(\mathrm{~g}) \rightleftharpoons 2 \mathrm{XY}_2(\mathrm{~g}) $$
At equilibrium, $75 \% \mathrm{X}_2 \mathrm{Y}_4(\mathrm{~g})$ was dissociated and the total pressure is 1 atm . The magnitude of $\Delta_{\mathrm{r}} \mathrm{G}^{\ominus}$ (in $\mathrm{kJ} \mathrm{mol}^{-1}$ ) at this temperature is $\_\_\_\_$ . (Nearest Integer)
(Given : $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1} ; \ln 10=2.3, \log 2=0.3, \log 3=0.48, \log 5=0.69, \log 7=0.84$ )
Q11.
mcq single
+4 / 1
Given is a concentrated solution of a weak electrolyte $A_x B_y$ of concentration ' $c$ ' and dissociation constant ' K '. The degree of dissociation is given by :
Q12.
mcq single
+4 / 1
In a period, the first ionisation enthalpy of the element at extreme left and the negative electron gain enthalpy of the extreme right element, except noble gases, are respectively.
Q13.
mcq single
+4 / 1
Match List - I with List - II.
Given $V_1$ and $V_2$ are initial and final volumes respectively.
List - I
(Isothermal process)
List - II
(Expression)
A.
Reversible
expansion
I.
$$
\mathrm{q}=0
$$
B.
Free expansion
II.
$$
\mathrm{q}=\mathrm{nRT} \ln \frac{\mathrm{~V}_2}{\mathrm{~V}_1}
$$
C.
Irreversible
Compression
III.
$$
\mathrm{w}=-\mathrm{p}_{\mathrm{ext}}\left(\mathrm{~V}_1-\mathrm{V}_2\right)
$$
D.
Cyclic reversible
IV.
$$
\frac{\mathrm{q}_{\mathrm{rev}}}{\mathrm{~T}}=0
$$
Choose the correct answer from the options given below :
Q14.
mcq single
+4 / 1
Treatment of a gas ' $X$ ' with a freshly prepared ferrous sulphate solution gives a compound ' $Y$ ' as a brown ring. The compounds X and Y are.
Q15.
mcq single
+4 / 1
Which of the following sequences of hybridisation, geometry and magnetic nature are correct for the given coordination compounds?
A. $\left[\mathrm{NiCl}_4\right]^{2-}-\mathrm{sp}^3$, tetrahedral, paramagnetic
B. $\left[\mathrm{Ni}\left(\mathrm{NH}_3\right)_6\right]^{2+}-\mathrm{sp}^3 \mathrm{~d}^2$, octahedral, paramagnetic
C. $\left[\mathrm{Ni}(\mathrm{CO})_4\right]-\mathrm{sp}^3$, tetrahedral, paramagnetic
D. $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}-\mathrm{dsp}^2$, square planar, diamagnetic
Choose the correct answer from the options given below :
Q16.
numerical
+4 / 1
An excess of $\mathrm{AgNO}_3$ is added to 100 mL of a 0.05 M solution of tetraaquadichloridochromium (III) chloride. The number of moles of AgCl precipitated will be $\_\_\_\_$ $\times 10^{-3}$.
(Nearest integer)
Q17.
mcq single
+4 / 1
' $x$ ' is the product which is obtained by the hydrolysis of prop-1-yne in the presence of mercuric sulphate under dilute acidic medium at 333 K . ' $y$ ' is the product which is obtained by the reaction of ethane nitrile with methyl magnesium bromide in dry ether followed by hydrolysis. IUPAC name of product obtained from ' $x^{\prime}$ and ' $y$ ' in the presence of barium hydroxide followed by heating is :
Q18.
mcq single
+4 / 1
An optically active alkyl bromide $\mathrm{C}_4 \mathrm{H}_9 \mathrm{Br}$, reacts with ethanolic KOH to form major compound $[\mathrm{A}]$ which reacts with bromine to give compound $[\mathrm{B}]$. Compound $[\mathrm{B}]$ reacts with ethanolic KOH and sodamide to give compound [C]. One molecule of water adds to compound [C] on warming with mercuric sulphate and dilute sulphuric acid at 333 K to form compound [D]. The functional group in compound D will be confirmed by :
Q19.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** A mixture of $\mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}$ (sugar) and NaCl can be separated by dissolving sugar in alcohol, due to differential solubility.
**Statement II :** Rose essence from rose petals is seperated by steam distillation due to its high volatility and insolubility in $\mathrm{H}_2 \mathrm{O}$.
In the light of the above statements, choose the correct answer from the options given below :
Q20.
numerical
+4 / 1
An alkane $(Y)$ requires 8 moles of oxygen for complete combustion and on chlorination with $\mathrm{Cl}_2 / \mathrm{h} \nu,(\mathrm{Y})$ gives only one monochlorinated product $(\mathrm{Z})$. The total number of primary carbon atoms in $(Y)$ is $\_\_\_\_$ .
Q21.
mcq single
+4 / 1
Shown below is the structure of methyl acetate with three different $\alpha, \beta$ and $\gamma$ carbon - oxygen bonds.
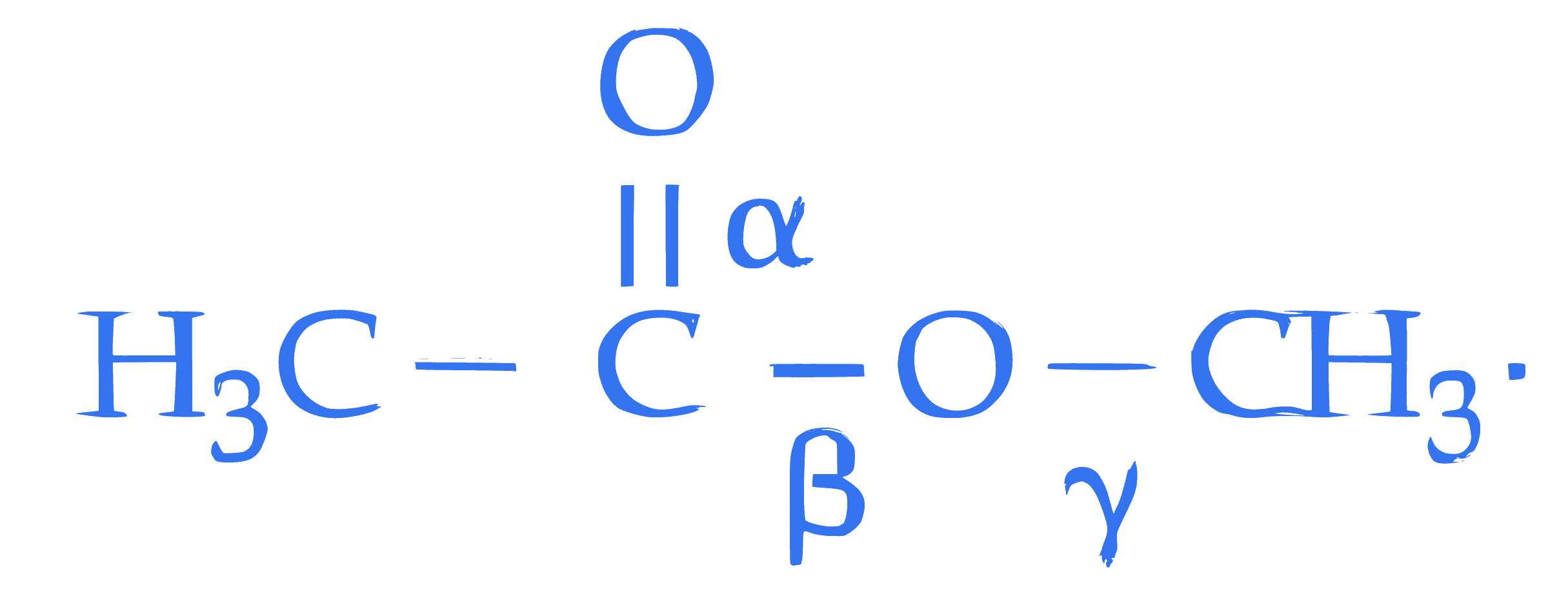 The correct order of bond lengths of these bonds is :
The correct order of bond lengths of these bonds is :
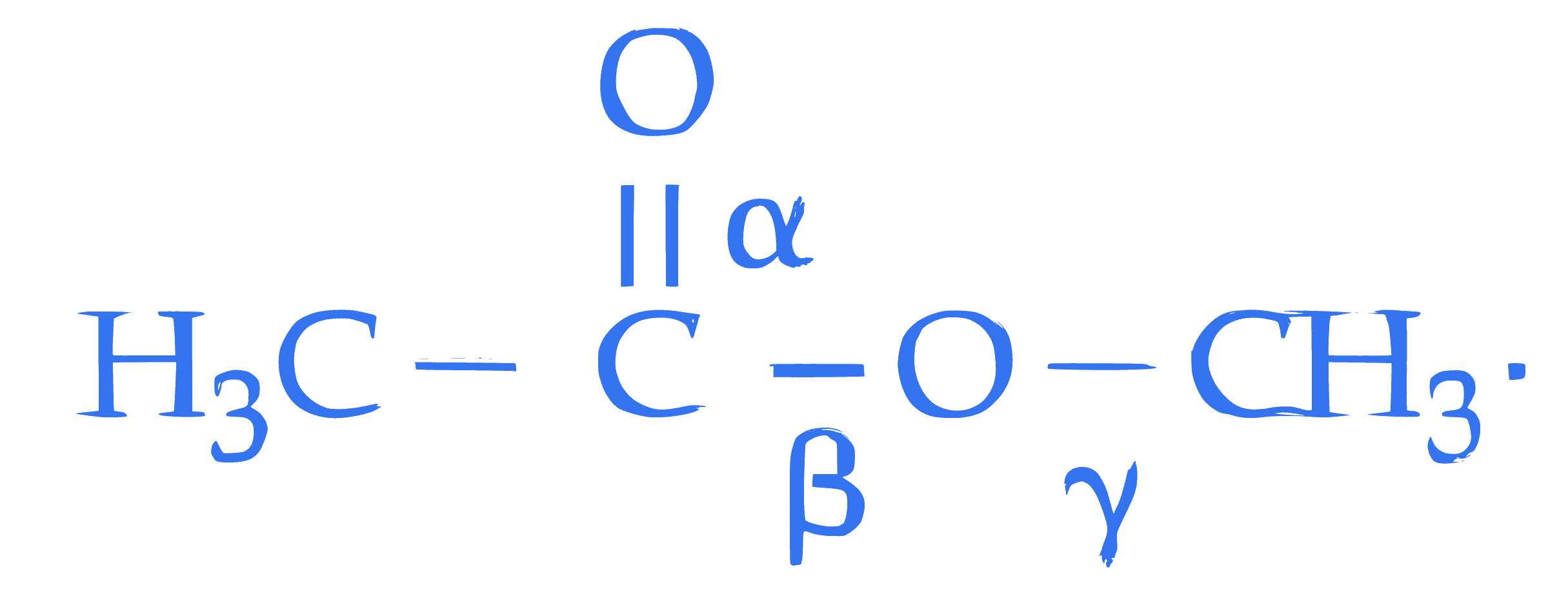 The correct order of bond lengths of these bonds is :
The correct order of bond lengths of these bonds is :
Q22.
mcq single
+4 / 1
For a general redox reaction
Anode $\quad \operatorname{Red}_1 \rightarrow \mathrm{Ox}_1^{\mathrm{n}_1^{+}}+\mathrm{n}_1 \mathrm{e}^{-}$
Cathode $\mathrm{O} x_2+\mathrm{n}_2 \mathrm{e}^{-} \rightarrow \operatorname{Red}_2^{\mathrm{n}_2-}$
Which of the following statement is incorrect?
Q23.
mcq single
+4 / 1
$$ \text { Consider the following reaction. } $$
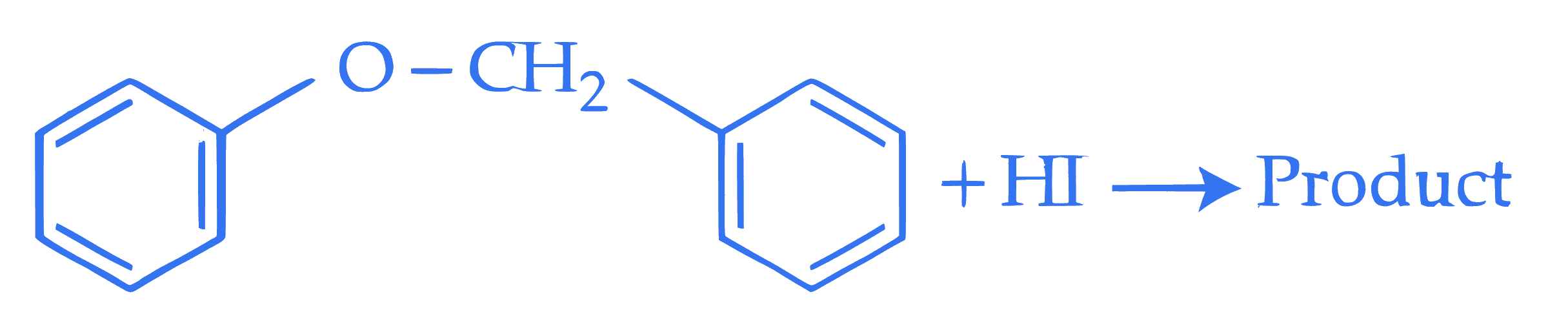 **Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
 **Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
Q24.
mcq single
+4 / 1
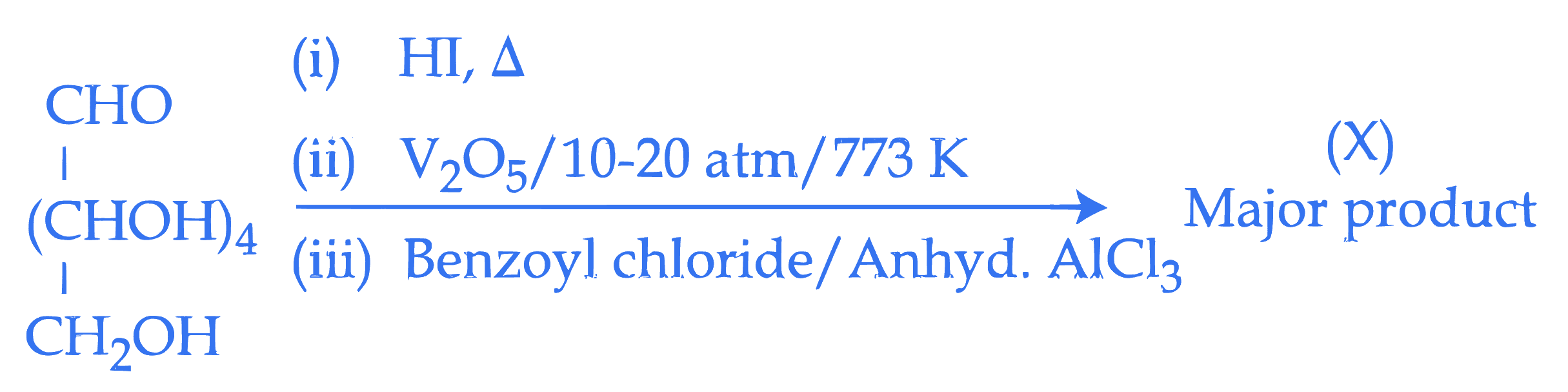
Consider the following reactions. Total number of electrons in the $\pi$ bonds and lone pair of electrons in the product $(X)$ is :

Q25.
mcq single
+4 / 1
The Bohr radius of a hydrogen like species is 70.53 pm . The species and the stationary state (n) are respectively
(Given : Hydrogen atom Bohr radius is 52.9 pm )
Q26.
mcq single
+4 / 1
Consider the following organic reaction sequence. Choose the final product $(X)$ from the following (consider the major product in all intermediate reactions)
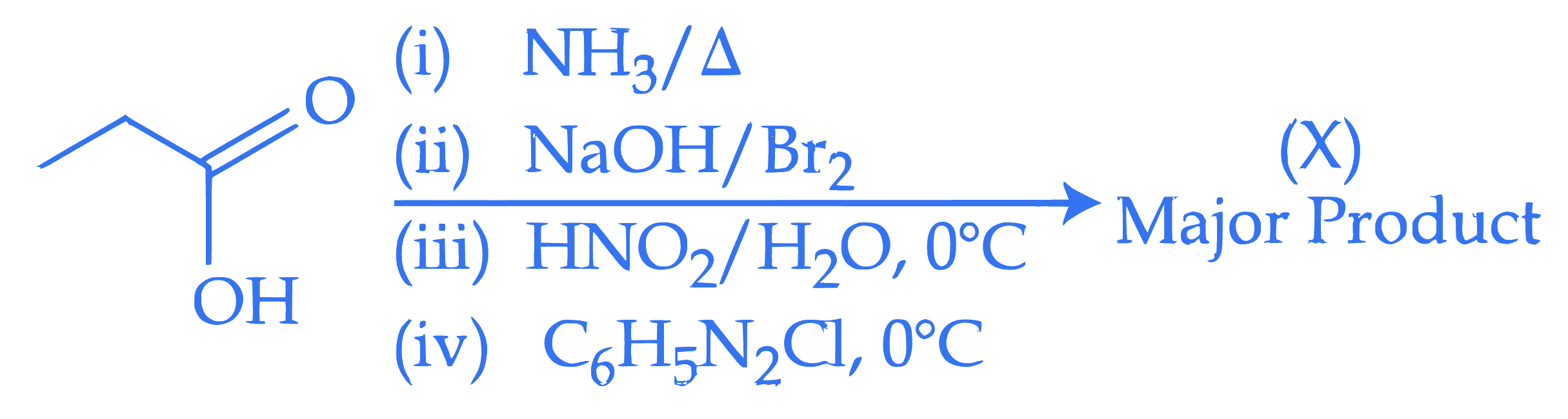
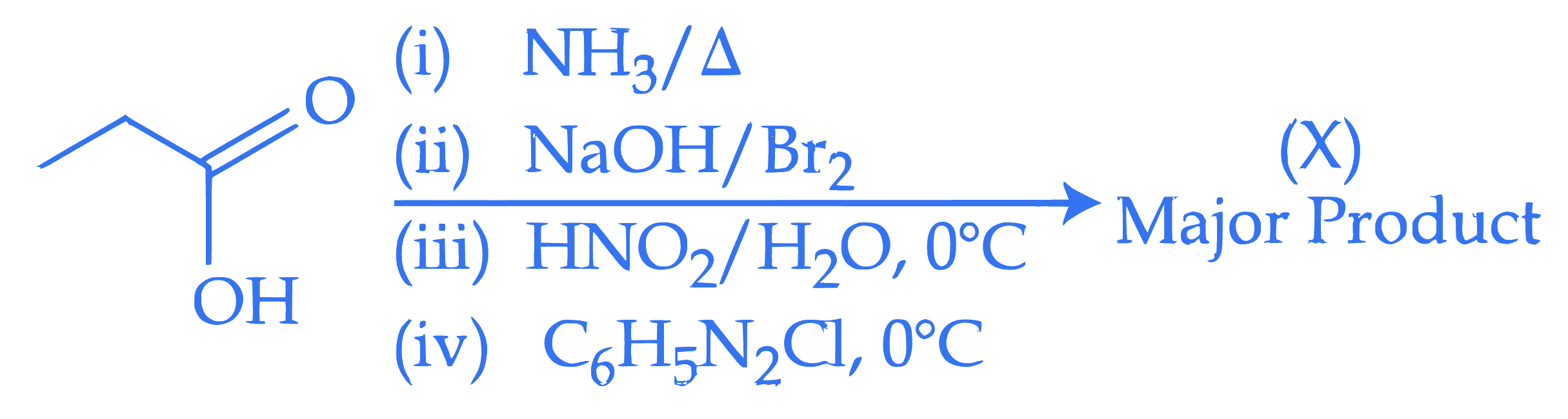
Q27.
mcq single
+4 / 1
The number of compounds from the following which can undergo reaction with $\mathrm{Br}_2 / \mathrm{KOH}$ (alcoholic) to give respective products and these respective products can also be obtained separately by Gabriel phthalimide reaction is :


Q28.
mcq single
+4 / 1
The correct order of first $\left(\Delta_i \mathrm{H}_1\right)$ and second $\left(\Delta_i \mathrm{H}_2\right)$ ionisation enthalpy values of Cr and Mn are :
A. $\Delta_i \mathrm{H}_1: \mathrm{Cr}>\mathrm{Mn}$
B. $\Delta_i \mathrm{H}_2: \mathrm{Cr}>\mathrm{Mn}$
C. $\Delta_i \mathrm{H}_1: \mathrm{Mn}>\mathrm{Cr}$
D. $\Delta_i \mathrm{H}_2: \mathrm{Mn}>\mathrm{Cr}$
Choose the correct answer from the options given below :
Q29.
numerical
+4 / 1
Decomposition of a hydrocarbon follows the equation $\mathrm{k}=\left(5.5 \times 10^{11} \mathrm{~s}^{-1}\right) \mathrm{e}^{\frac{-28000 \mathrm{~K}}{\mathrm{~T}}}$. The activation energy of reaction is $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (Nearest Integer)
Given : $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$
Q30.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** $\mathrm{F}_2 \mathrm{O}<\mathrm{H}_2 \mathrm{O}<\mathrm{Cl}_2 \mathrm{O}$ is the correct trend in terms of bond angle.
**Statement II :** $\mathrm{SiF}_4, \mathrm{SnF}_4$ and $\mathrm{PbF}_4$ are ionic in nature.
In the light of the above statements, choose the correct answer from the options given below :
Q31.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of compounds among $\mathrm{SO}_2, \mathrm{SO}_3, \mathrm{SF}_4, \mathrm{SF}_6$ and $\mathrm{H}_2 \mathrm{~S}$ in which sulphur does not obey the Octet rule is 3 .
**Statement II :** Among $\left[\mathrm{H}_2 \mathrm{O}, \mathrm{ClF}_3, \mathrm{SF}_4\right],\left[\mathrm{NH}_3, \mathrm{BrF}_5, \mathrm{SF}_4\right],\left[\mathrm{BrF}_5, \mathrm{ClF}_3, \mathrm{XeF}_4\right]$ and [ $\mathrm{XeF}_4, \mathrm{ClF}_3, \mathrm{H}_2 \mathrm{O}$ ], the number of sets in which all the molecules have one lone pair of electrons on the central atom is 1 .
In the light of the above statements, choose the correct answer from the options given below :
Q32.
numerical
+4 / 1
500 mL of $0.2 \mathrm{M} \mathrm{MnO}_4^{-}$solution in basic medium when mixed with 500 mL of 1.5 M KI solution, oxidises iodide ions to liberate molecular iodine. This liberated iodine is then titrated with a standard $x \mathrm{M}$ thiosulphate solution in presence of starch till the end point. If 300 mL of thiosulphate was consumed, then the value of $x$ is $\_\_\_\_$。
Q33.
mcq single
+4 / 1
Given below are two statements :
 **Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
 **Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** $\quad \mathrm{H}_2 \mathrm{O}$ molecules move from the chamber 1 to chamber 2 .
**Statement II :** The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass $=174 \mathrm{~g} / \mathrm{mol}$ ) in 2 L of water (at $27^{\circ} \mathrm{C}$ ) is 0.0107 bar. (Given: $\mathrm{R}=0.083 \mathrm{dm}^3$ bar $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
Q34.
mcq single
+4 / 1
Which of the following contain the same number of atoms?
(Given : Molar mass in $\mathrm{g} \mathrm{mol}^{-1}$ of $\mathrm{H}, \mathrm{He}, \mathrm{O}$ and S are 1, 4, 16 and 32 respectively)
A. 2 g of $\mathrm{O}_2$ gas
B. 4 g of $\mathrm{SO}_2$ gas
C. 1400 mL of $\mathrm{O}_2$ at STP
D. $0.05 \mathrm{~L}$ of He at STP
E. $ 0.0625 \mathrm{~mol}$ of $\mathrm{H}_2$ gas
Choose the correct answer from the options given below :
Q35.
numerical
+4 / 1
In a closed flask at 600 K , one mole of $\mathrm{X}_2 \mathrm{Y}_4(\mathrm{~g})$ attains equilibrium as given below :
$$ \mathrm{X}_2 \mathrm{Y}_4(\mathrm{~g}) \rightleftharpoons 2 \mathrm{XY}_2(\mathrm{~g}) $$
At equilibrium, $75 \% \mathrm{X}_2 \mathrm{Y}_4(\mathrm{~g})$ was dissociated and the total pressure is 1 atm . The magnitude of $\Delta_{\mathrm{r}} \mathrm{G}^{\ominus}$ (in $\mathrm{kJ} \mathrm{mol}^{-1}$ ) at this temperature is $\_\_\_\_$ . (Nearest Integer)
(Given : $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1} ; \ln 10=2.3, \log 2=0.3, \log 3=0.48, \log 5=0.69, \log 7=0.84$ )
Q36.
mcq single
+4 / 1
Given is a concentrated solution of a weak electrolyte $A_x B_y$ of concentration ' $c$ ' and dissociation constant ' K '. The degree of dissociation is given by :
Q37.
mcq single
+4 / 1
In a period, the first ionisation enthalpy of the element at extreme left and the negative electron gain enthalpy of the extreme right element, except noble gases, are respectively.
Q38.
mcq single
+4 / 1
Match List - I with List - II.
Given $V_1$ and $V_2$ are initial and final volumes respectively.
List - I
(Isothermal process)
List - II
(Expression)
A.
Reversible
expansion
I.
$$
\mathrm{q}=0
$$
B.
Free expansion
II.
$$
\mathrm{q}=\mathrm{nRT} \ln \frac{\mathrm{~V}_2}{\mathrm{~V}_1}
$$
C.
Irreversible
Compression
III.
$$
\mathrm{w}=-\mathrm{p}_{\mathrm{ext}}\left(\mathrm{~V}_1-\mathrm{V}_2\right)
$$
D.
Cyclic reversible
IV.
$$
\frac{\mathrm{q}_{\mathrm{rev}}}{\mathrm{~T}}=0
$$
Choose the correct answer from the options given below :
Q39.
mcq single
+4 / 1
Treatment of a gas ' $X$ ' with a freshly prepared ferrous sulphate solution gives a compound ' $Y$ ' as a brown ring. The compounds X and Y are.
Q40.
mcq single
+4 / 1
Which of the following sequences of hybridisation, geometry and magnetic nature are correct for the given coordination compounds?
A. $\left[\mathrm{NiCl}_4\right]^{2-}-\mathrm{sp}^3$, tetrahedral, paramagnetic
B. $\left[\mathrm{Ni}\left(\mathrm{NH}_3\right)_6\right]^{2+}-\mathrm{sp}^3 \mathrm{~d}^2$, octahedral, paramagnetic
C. $\left[\mathrm{Ni}(\mathrm{CO})_4\right]-\mathrm{sp}^3$, tetrahedral, paramagnetic
D. $\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}-\mathrm{dsp}^2$, square planar, diamagnetic
Choose the correct answer from the options given below :
Q41.
numerical
+4 / 1
An excess of $\mathrm{AgNO}_3$ is added to 100 mL of a 0.05 M solution of tetraaquadichloridochromium (III) chloride. The number of moles of AgCl precipitated will be $\_\_\_\_$ $\times 10^{-3}$.
(Nearest integer)
Q42.
mcq single
+4 / 1
' $x$ ' is the product which is obtained by the hydrolysis of prop-1-yne in the presence of mercuric sulphate under dilute acidic medium at 333 K . ' $y$ ' is the product which is obtained by the reaction of ethane nitrile with methyl magnesium bromide in dry ether followed by hydrolysis. IUPAC name of product obtained from ' $x^{\prime}$ and ' $y$ ' in the presence of barium hydroxide followed by heating is :
Q43.
mcq single
+4 / 1
An optically active alkyl bromide $\mathrm{C}_4 \mathrm{H}_9 \mathrm{Br}$, reacts with ethanolic KOH to form major compound $[\mathrm{A}]$ which reacts with bromine to give compound $[\mathrm{B}]$. Compound $[\mathrm{B}]$ reacts with ethanolic KOH and sodamide to give compound [C]. One molecule of water adds to compound [C] on warming with mercuric sulphate and dilute sulphuric acid at 333 K to form compound [D]. The functional group in compound D will be confirmed by :
Q44.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** A mixture of $\mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}$ (sugar) and NaCl can be separated by dissolving sugar in alcohol, due to differential solubility.
**Statement II :** Rose essence from rose petals is seperated by steam distillation due to its high volatility and insolubility in $\mathrm{H}_2 \mathrm{O}$.
In the light of the above statements, choose the correct answer from the options given below :
Q45.
numerical
+4 / 1
An alkane $(Y)$ requires 8 moles of oxygen for complete combustion and on chlorination with $\mathrm{Cl}_2 / \mathrm{h} \nu,(\mathrm{Y})$ gives only one monochlorinated product $(\mathrm{Z})$. The total number of primary carbon atoms in $(Y)$ is $\_\_\_\_$ .
Q46.
mcq single
+4 / 1
Shown below is the structure of methyl acetate with three different $\alpha, \beta$ and $\gamma$ carbon - oxygen bonds.
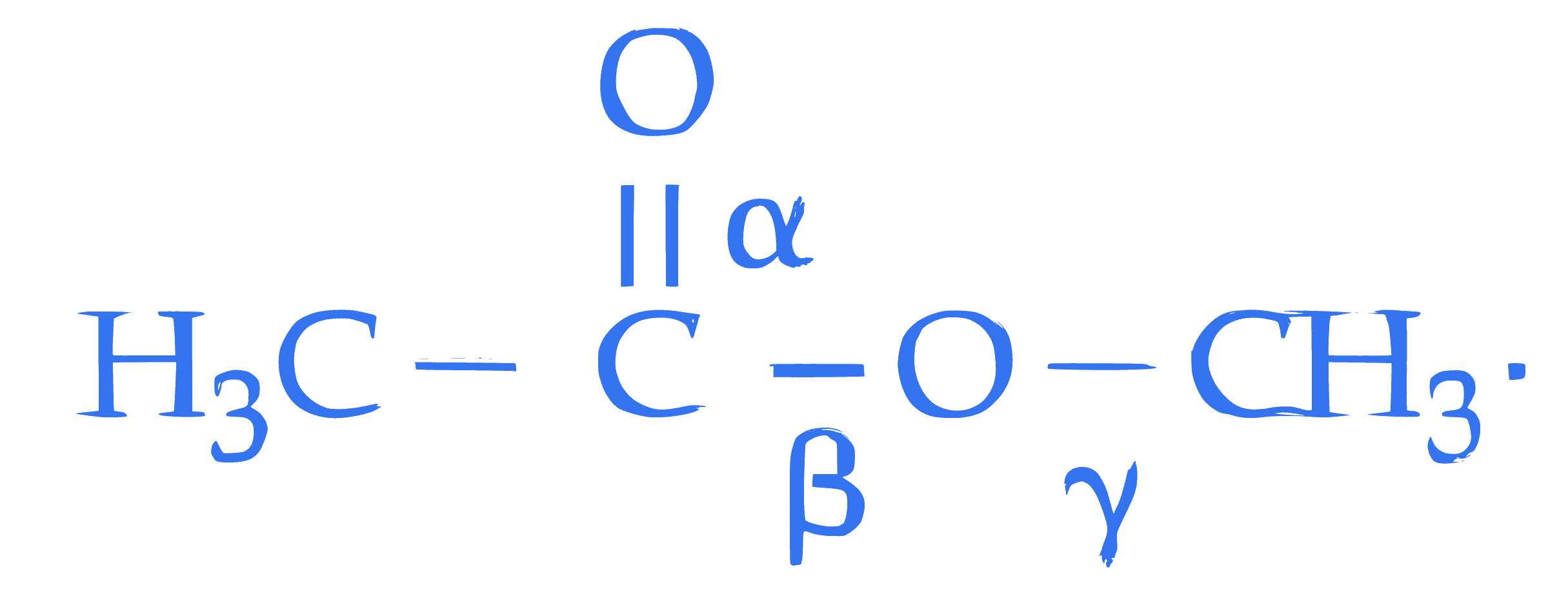 The correct order of bond lengths of these bonds is :
The correct order of bond lengths of these bonds is :
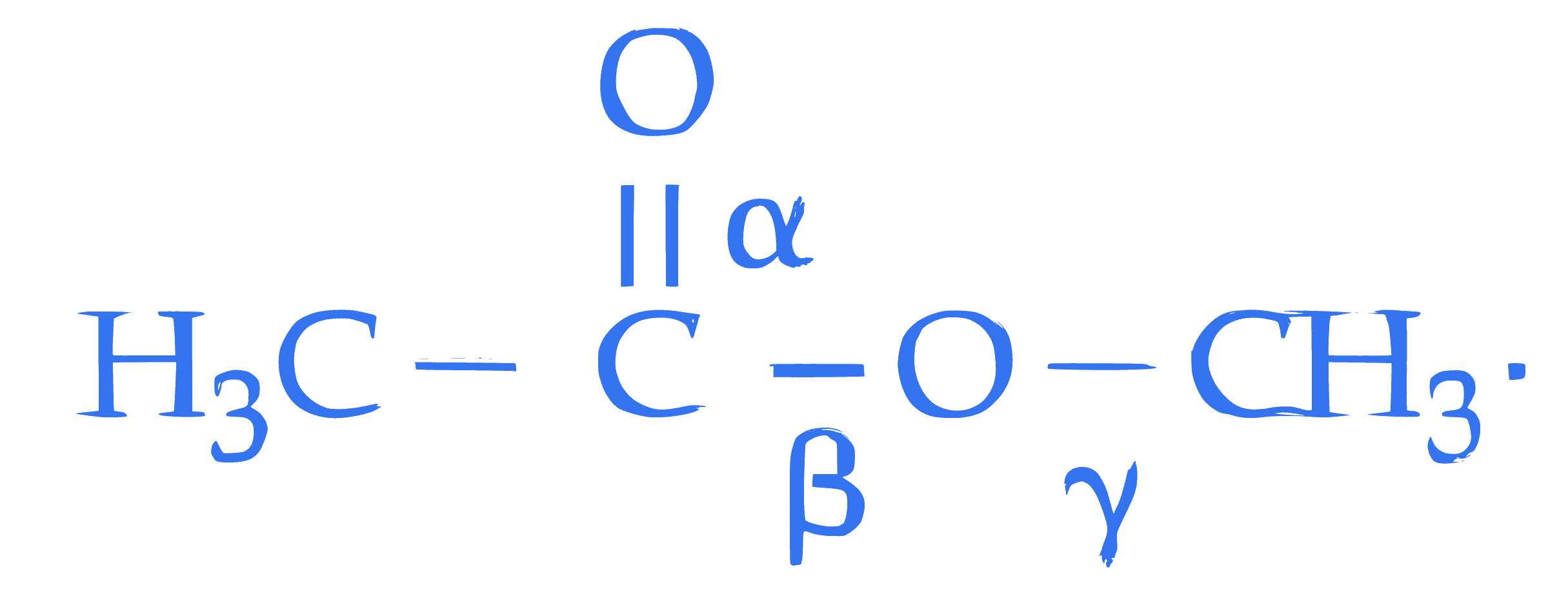 The correct order of bond lengths of these bonds is :
The correct order of bond lengths of these bonds is :
Q47.
mcq single
+4 / 1
For a general redox reaction
Anode $\quad \operatorname{Red}_1 \rightarrow \mathrm{Ox}_1^{\mathrm{n}_1^{+}}+\mathrm{n}_1 \mathrm{e}^{-}$
Cathode $\mathrm{O} x_2+\mathrm{n}_2 \mathrm{e}^{-} \rightarrow \operatorname{Red}_2^{\mathrm{n}_2-}$
Which of the following statement is incorrect?
Q48.
mcq single
+4 / 1
$$ \text { Consider the following reaction. } $$
 **Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
 **Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
**Statement I :** In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
**Statement II :** In the above reaction, the $-\mathrm{O}-\mathrm{CH}_2-$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
Q49.
mcq single
+4 / 1
Consider the following reactions. Total number of electrons in the $\pi$ bonds and lone pair of electrons in the product $(X)$ is :


Q50.
mcq single
+4 / 1
The Bohr radius of a hydrogen like species is 70.53 pm . The species and the stationary state (n) are respectively
(Given : Hydrogen atom Bohr radius is 52.9 pm )
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
Let the mean and the variance of seven observations $2,4, \alpha, 8, \beta, 12,14, \alpha<\beta$, be 8 and 16 respectively. Then the quadratic equation whose roots are $3 \alpha+2$ and $2 \beta+1$ is :
Q2.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=2 \hat{i}+3 \hat{j}+3 \hat{k}$ and $\overrightarrow{\mathrm{b}}=6 \hat{i}+3 \hat{j}+3 \hat{k}$. Then the square of the area of the triangle with adjacent sides determined by the vectors $(2 \vec{a}+3 \vec{b})$ and $(\vec{a}-\vec{b})$ is :
Q3.
mcq single
+4 / 1
Let $\mathop {\lim }\limits_{x \to 2} \frac{(\tan (x-2))\left(\mathrm{r} x^2+(\mathrm{p}-2) x-2 \mathrm{p}\right)}{(x-2)^2}=5$ for some $\mathrm{r}, \mathrm{p} \in \boldsymbol{R}$. If the set of all possible values of q , such that the roots of the equation $\mathrm{r} x^2-\mathrm{p} x+\mathrm{q}=0$ lie in $(0,2)$, be the interval $(\alpha, \beta]$, then $4(\alpha+\beta)$ equals :
Q4.
mcq single
+4 / 1
A bag contains 6 blue and 6 green balls. Pairs of balls are drawn without replacement until the bag is empty. The probability that each drawn pair consists of one blue and one green ball is :
Q5.
numerical
+4 / 1
Let the image of the point $\mathrm{P}(0,-5,0)$ in the line $\frac{x-1}{2}=\frac{y}{1}=\frac{z+1}{-2}$ be the point R and the image of the point $\mathrm{Q}\left(0, \frac{-1}{2}, 0\right)$ in the line $\frac{x-1}{-1}=\frac{y+9}{4}=\frac{z+1}{1}$ be the point S . Then the square of the area of the parallelogram PQRS is $\_\_\_\_$ .
Q6.
mcq single
+4 / 1
The shortest distance between the lines $\frac{x-4}{1}=\frac{y-3}{2}=\frac{z-2}{-3}$ and $\frac{x+2}{2}=\frac{y-6}{4}=\frac{z-5}{-5}$ is:
Q7.
mcq single
+4 / 1
Consider the quadratic equation $\left(n^2-2 n+2\right) x^2-3 x+\left(n^2-2 n+2\right)^2=0, n \in \mathbf{R}$. Let $\alpha$ be the minimum value of the product of its roots and $\beta$ be the maximum value of the sum of its roots. Then the sum of the first six terms of the G.P., whose first term is $\alpha$ and the common ratio is $\frac{\alpha}{\beta}$, is :
Q8.
mcq single
+4 / 1
The sum $1+\frac{1}{2}\left(1^2+2^2\right)+\frac{1}{3}\left(1^2+2^2+3^2\right)+\ldots$ upto 10 terms is equal to :
Q9.
numerical
+4 / 1
Let $f(x)=\left\{\begin{array}{ll}x^3+8 ; & x<0, \\ x^2-4 ; & x \geq 0,\end{array}\right.$ and $g(x)= \begin{cases}(x-8)^{1 / 3} ; & x<0, \\ (x+4)^{1 / 2} ; & x \geq 0 .\end{cases}$
Then the number of points, where the function $g \circ f$ is discontinuous, is $\_\_\_\_$ .
Q10.
mcq single
+4 / 1
The area of the region $\left\{(x, y): x^2-8 x \leq y \leq-x\right\}$ is :
Q11.
mcq single
+4 / 1
Let $\mathrm{A}=\left[\begin{array}{lll}1 & 0 & 0 \\ 3 & 1 & 0 \\ 9 & 3 & 1\end{array}\right]$ and $\mathrm{B}=\left[\mathrm{b}_{i j}\right], 1 \leq i, j \leq 3$. If $\mathrm{B}=\mathrm{A}^{99}-\mathrm{I}$, then the value of $\frac{\mathrm{b}_{31}-\mathrm{b}_{21}}{\mathrm{~b}_{32}}$ is :
Q12.
mcq single
+4 / 1
The sum of all possible values of $\theta \in[0,2 \pi]$, for which the system of equations :
$$ \begin{aligned} & x \cos 3 \theta-8 y-12 z=0 \\ & x \cos 2 \theta+3 y+3 z=0 \\ & x+y+3 z=0 \end{aligned} $$
has a non-trivial solution, is equal to :
Q13.
mcq single
+4 / 1
If $\sin \left(\tan ^{-1}(x \sqrt{2})\right)=\cot \left(\sin ^{-1} \sqrt{1-x^2}\right), x \in(0,1)$, then the value of $x$ is:
Q14.
numerical
+4 / 1
If $\left(1-x^3\right)^{10}=\sum\limits_{\mathrm{r}=0}^{10} \mathrm{a}_{\mathrm{r}} x^{\mathrm{r}}(1-x)^{30-2 \mathrm{r}}$, then $\frac{9 \mathrm{a}_9}{\mathrm{a}_{10}}$ is equal to $\_\_\_\_$ .
Q15.
numerical
+4 / 1
Let $\mathrm{R}=\left\{(x, y) \in \mathbf{N} \times \mathbf{N}: \log _{\mathrm{e}}(x+y) \leq 2\right\}$. Then the minimum number of elements, required to be added in $R$ to make it a transitive relation, is $\_\_\_\_$ .
Q16.
mcq single
+4 / 1
Let $S=\left\{z \in \mathbb{C}: z^2+\sqrt{6} i z-3=0\right\}$. Then $\sum\limits_{z \in S} z^8$ is equal to :
Q17.
mcq single
+4 / 1
The eccentricity of an ellipse $E$ with centre at the origin $O$ is $\frac{\sqrt{3}}{2}$ and its directrices are $x= \pm \frac{4 \sqrt{6}}{3}$. Let $\mathrm{H}: \frac{x^2}{\mathrm{a}^2}-\frac{y^2}{\mathrm{~b}^2}=1$ be a hyperbola whose eccentricity is equal to the length of semi-major axis of E , and whose length of latus rectum is equal to the length of minor axis of E . Then the distance between the foci of H is :
Q18.
mcq single
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be defined as $f(x)=\frac{2 x^2-3 x+2}{3 x^2+x+3}$. Then $f$ is :
Q19.
mcq single
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be such that $f(x y)=f(x) f(y)$, for all $x, y \in \mathbf{R}$ and $f(0) \neq 0$. Let $g:[1, \infty) \rightarrow \mathbf{R}$ be a differentiable function such that
$$ x^2 g(x)=\int_1^x\left(\mathrm{t}^2 f(\mathrm{t})-\operatorname{tg}(\mathrm{t})\right) d t $$
Then $g(2)$ is equal to :
Q20.
numerical
+4 / 1
Let the line $x-y=4$ intersect the circle $\mathrm{C}:(x-4)^2+(y+3)^2=9$ at the points Q and R . If $\mathrm{P}(\alpha, \beta)$ is a point on C such that $\mathrm{PQ}=\mathrm{PR}$, then $(6 \alpha+8 \beta)^2$ is equal to $\_\_\_\_$ .
Q21.
mcq single
+4 / 1
Let C be a circle having centre in the first quadrant and touching the $x$-axis at a distance of 3 units from the origin. If the circle $C$ has an intercept of length $6 \sqrt{3}$ on $y$-axis, then the length of the chord of the circle C on the line $x-y=3$ is :
Q22.
mcq single
+4 / 1
Let $x=9$ be a directrix of an ellipse E , whose centre is at the origin and eccentricity is $\frac{1}{3}$. Let $\mathrm{P}(\alpha, 0)$, $\alpha>0$, be a focus of E and AB be a chord passing through P . Then the locus of the mid point of AB is :
Q23.
mcq single
+4 / 1
A building has ground floor and 10 more floors. Nine persons enter in a lift at the ground floor. The lift goes up to the $10^{\text {th }}$ floor. The number of ways, in which any 4 persons exit at a floor and the remaining 5 persons exit at a different floor, if the lift does not stop at the first and the second floors, is equal to :
Q24.
mcq single
+4 / 1
The value of the integral $\int\limits_{-1}^1\left(\frac{x^3+|x|+1}{x^2+2|x|+1}\right) \mathrm{d} x$ is equal to:
Q25.
mcq single
+4 / 1
Let $\mathrm{A}=\left[\begin{array}{ccc}1 & 3 & -1 \\ 2 & 1 & \alpha \\ 0 & 1 & -1\end{array}\right]$ be a singular matrix. Let $f(x)=\int_0^x\left(\mathrm{t}^2+2 \mathrm{t}+3\right) \mathrm{dt}, x \in[1, \alpha]$. If M and m are respectively the maximum and the minimum values of $f$ in $[1, \alpha]$, then $3(\mathrm{M}-\mathrm{m})$ is equal to :
Q26.
mcq single
+4 / 1
Let the mean and the variance of seven observations $2,4, \alpha, 8, \beta, 12,14, \alpha<\beta$, be 8 and 16 respectively. Then the quadratic equation whose roots are $3 \alpha+2$ and $2 \beta+1$ is :
Q27.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=2 \hat{i}+3 \hat{j}+3 \hat{k}$ and $\overrightarrow{\mathrm{b}}=6 \hat{i}+3 \hat{j}+3 \hat{k}$. Then the square of the area of the triangle with adjacent sides determined by the vectors $(2 \vec{a}+3 \vec{b})$ and $(\vec{a}-\vec{b})$ is :
Q28.
mcq single
+4 / 1
Let $\mathop {\lim }\limits_{x \to 2} \frac{(\tan (x-2))\left(\mathrm{r} x^2+(\mathrm{p}-2) x-2 \mathrm{p}\right)}{(x-2)^2}=5$ for some $\mathrm{r}, \mathrm{p} \in \boldsymbol{R}$. If the set of all possible values of q , such that the roots of the equation $\mathrm{r} x^2-\mathrm{p} x+\mathrm{q}=0$ lie in $(0,2)$, be the interval $(\alpha, \beta]$, then $4(\alpha+\beta)$ equals :
Q29.
mcq single
+4 / 1
A bag contains 6 blue and 6 green balls. Pairs of balls are drawn without replacement until the bag is empty. The probability that each drawn pair consists of one blue and one green ball is :
Q30.
numerical
+4 / 1
Let the image of the point $\mathrm{P}(0,-5,0)$ in the line $\frac{x-1}{2}=\frac{y}{1}=\frac{z+1}{-2}$ be the point R and the image of the point $\mathrm{Q}\left(0, \frac{-1}{2}, 0\right)$ in the line $\frac{x-1}{-1}=\frac{y+9}{4}=\frac{z+1}{1}$ be the point S . Then the square of the area of the parallelogram PQRS is $\_\_\_\_$ .
Q31.
mcq single
+4 / 1
The shortest distance between the lines $\frac{x-4}{1}=\frac{y-3}{2}=\frac{z-2}{-3}$ and $\frac{x+2}{2}=\frac{y-6}{4}=\frac{z-5}{-5}$ is:
Q32.
mcq single
+4 / 1
Consider the quadratic equation $\left(n^2-2 n+2\right) x^2-3 x+\left(n^2-2 n+2\right)^2=0, n \in \mathbf{R}$. Let $\alpha$ be the minimum value of the product of its roots and $\beta$ be the maximum value of the sum of its roots. Then the sum of the first six terms of the G.P., whose first term is $\alpha$ and the common ratio is $\frac{\alpha}{\beta}$, is :
Q33.
mcq single
+4 / 1
The sum $1+\frac{1}{2}\left(1^2+2^2\right)+\frac{1}{3}\left(1^2+2^2+3^2\right)+\ldots$ upto 10 terms is equal to :
Q34.
numerical
+4 / 1
Let $f(x)=\left\{\begin{array}{ll}x^3+8 ; & x<0, \\ x^2-4 ; & x \geq 0,\end{array}\right.$ and $g(x)= \begin{cases}(x-8)^{1 / 3} ; & x<0, \\ (x+4)^{1 / 2} ; & x \geq 0 .\end{cases}$
Then the number of points, where the function $g \circ f$ is discontinuous, is $\_\_\_\_$ .
Q35.
mcq single
+4 / 1
The area of the region $\left\{(x, y): x^2-8 x \leq y \leq-x\right\}$ is :
Q36.
mcq single
+4 / 1
Let $\mathrm{A}=\left[\begin{array}{lll}1 & 0 & 0 \\ 3 & 1 & 0 \\ 9 & 3 & 1\end{array}\right]$ and $\mathrm{B}=\left[\mathrm{b}_{i j}\right], 1 \leq i, j \leq 3$. If $\mathrm{B}=\mathrm{A}^{99}-\mathrm{I}$, then the value of $\frac{\mathrm{b}_{31}-\mathrm{b}_{21}}{\mathrm{~b}_{32}}$ is :
Q37.
mcq single
+4 / 1
The sum of all possible values of $\theta \in[0,2 \pi]$, for which the system of equations :
$$ \begin{aligned} & x \cos 3 \theta-8 y-12 z=0 \\ & x \cos 2 \theta+3 y+3 z=0 \\ & x+y+3 z=0 \end{aligned} $$
has a non-trivial solution, is equal to :
Q38.
mcq single
+4 / 1
If $\sin \left(\tan ^{-1}(x \sqrt{2})\right)=\cot \left(\sin ^{-1} \sqrt{1-x^2}\right), x \in(0,1)$, then the value of $x$ is:
Q39.
numerical
+4 / 1
If $\left(1-x^3\right)^{10}=\sum\limits_{\mathrm{r}=0}^{10} \mathrm{a}_{\mathrm{r}} x^{\mathrm{r}}(1-x)^{30-2 \mathrm{r}}$, then $\frac{9 \mathrm{a}_9}{\mathrm{a}_{10}}$ is equal to $\_\_\_\_$ .
Q40.
numerical
+4 / 1
Let $\mathrm{R}=\left\{(x, y) \in \mathbf{N} \times \mathbf{N}: \log _{\mathrm{e}}(x+y) \leq 2\right\}$. Then the minimum number of elements, required to be added in $R$ to make it a transitive relation, is $\_\_\_\_$ .
Q41.
mcq single
+4 / 1
Let $S=\left\{z \in \mathbb{C}: z^2+\sqrt{6} i z-3=0\right\}$. Then $\sum\limits_{z \in S} z^8$ is equal to :
Q42.
mcq single
+4 / 1
The eccentricity of an ellipse $E$ with centre at the origin $O$ is $\frac{\sqrt{3}}{2}$ and its directrices are $x= \pm \frac{4 \sqrt{6}}{3}$. Let $\mathrm{H}: \frac{x^2}{\mathrm{a}^2}-\frac{y^2}{\mathrm{~b}^2}=1$ be a hyperbola whose eccentricity is equal to the length of semi-major axis of E , and whose length of latus rectum is equal to the length of minor axis of E . Then the distance between the foci of H is :
Q43.
mcq single
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be defined as $f(x)=\frac{2 x^2-3 x+2}{3 x^2+x+3}$. Then $f$ is :
Q44.
mcq single
+4 / 1
Let $f: \mathbf{R} \rightarrow \mathbf{R}$ be such that $f(x y)=f(x) f(y)$, for all $x, y \in \mathbf{R}$ and $f(0) \neq 0$. Let $g:[1, \infty) \rightarrow \mathbf{R}$ be a differentiable function such that
$$ x^2 g(x)=\int_1^x\left(\mathrm{t}^2 f(\mathrm{t})-\operatorname{tg}(\mathrm{t})\right) d t $$
Then $g(2)$ is equal to :
Q45.
numerical
+4 / 1
Let the line $x-y=4$ intersect the circle $\mathrm{C}:(x-4)^2+(y+3)^2=9$ at the points Q and R . If $\mathrm{P}(\alpha, \beta)$ is a point on C such that $\mathrm{PQ}=\mathrm{PR}$, then $(6 \alpha+8 \beta)^2$ is equal to $\_\_\_\_$ .
Q46.
mcq single
+4 / 1
Let C be a circle having centre in the first quadrant and touching the $x$-axis at a distance of 3 units from the origin. If the circle $C$ has an intercept of length $6 \sqrt{3}$ on $y$-axis, then the length of the chord of the circle C on the line $x-y=3$ is :
Q47.
mcq single
+4 / 1
Let $x=9$ be a directrix of an ellipse E , whose centre is at the origin and eccentricity is $\frac{1}{3}$. Let $\mathrm{P}(\alpha, 0)$, $\alpha>0$, be a focus of E and AB be a chord passing through P . Then the locus of the mid point of AB is :
Q48.
mcq single
+4 / 1
A building has ground floor and 10 more floors. Nine persons enter in a lift at the ground floor. The lift goes up to the $10^{\text {th }}$ floor. The number of ways, in which any 4 persons exit at a floor and the remaining 5 persons exit at a different floor, if the lift does not stop at the first and the second floors, is equal to :
Q49.
mcq single
+4 / 1
The value of the integral $\int\limits_{-1}^1\left(\frac{x^3+|x|+1}{x^2+2|x|+1}\right) \mathrm{d} x$ is equal to:
Q50.
mcq single
+4 / 1
Let $\mathrm{A}=\left[\begin{array}{ccc}1 & 3 & -1 \\ 2 & 1 & \alpha \\ 0 & 1 & -1\end{array}\right]$ be a singular matrix. Let $f(x)=\int_0^x\left(\mathrm{t}^2+2 \mathrm{t}+3\right) \mathrm{dt}, x \in[1, \alpha]$. If M and m are respectively the maximum and the minimum values of $f$ in $[1, \alpha]$, then $3(\mathrm{M}-\mathrm{m})$ is equal to :
Physics
Physics
Q1.
mcq single
+4 / 1
A body of mass 1 kg moves along a straight line with a velocity $v=2 x^2$. The work done by the body during displacement from $x=0$ to 5 m is $\_\_\_\_$ J.
Q2.
mcq single
+4 / 1
The velocity at which 6 kg mass (shown in figure) strikes the ground when it is released from a height of 6 m above the ground is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}$. Assume pulley is massless and string is light and inextensible. (Take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )

Q3.
mcq single
+4 / 1
Refer to the logic circuit given below. For two inputs ( $A=1, B=1$ ) and ( $A=0, B=1$ ), output ( Y ) will be $\_\_\_\_$ .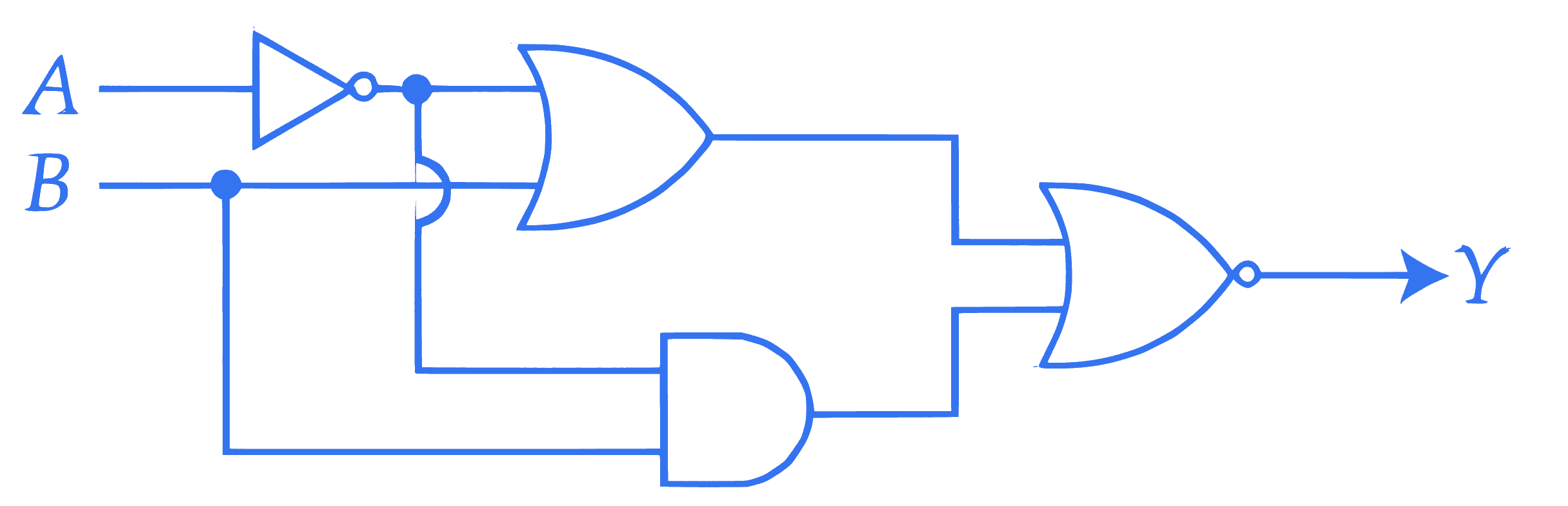

Q4.
mcq single
+4 / 1
In a Young double slit experiment, the wavelength of incident light is $6000 \mathop {\rm{A}}\limits^{\rm{o}}$, the separation between slits $S_1$ and $S_2$ is 5 cm and the distance between slits plane and screen is 50 cm , as shown in the figure below. If the resultant intensity at $P$ is equal to the intensity due to individual slits, the path difference between interfering waves is $\_\_\_\_$ Å.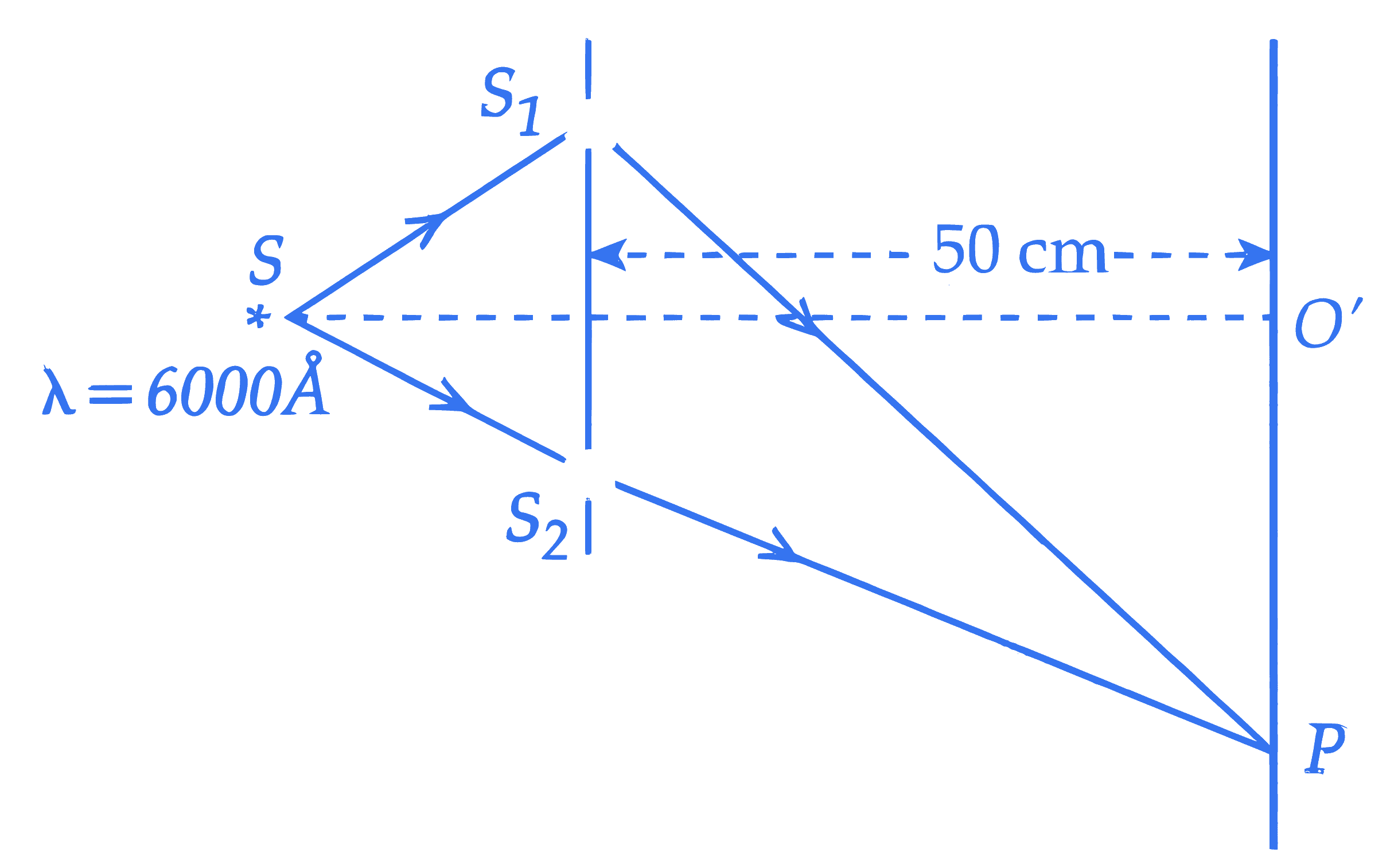

Q5.
mcq single
+4 / 1
A spring stretches by 2 mm when it is loaded with a mass of 200 g . From equilibrium position the mass is further pulled down by 2 mm and released. The frequency associated with the system and maxmimum energy in the spring are $\_\_\_\_$ Hz and $\_\_\_\_$ J, respectively.
(Take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q6.
mcq single
+4 / 1
A solid sphere $(A)$ of mass $5 m$ and a spherical shell $(B)$ of mass $m$, both having same radius, are placed on a rough surface. When a force of same magnitude is applied tangentially at the highest points of $A$ and $B$, they start rolling without slipping with an acceleration of $a_A$ and $a_B$, respectively. The ratio of $a_A$ and $a_B$ is $\_\_\_\_$ .
Q7.
mcq single
+4 / 1
Two identical bodies A and B of equal masses have initial velocities $\overrightarrow{v_1}=4 \hat{i} \mathrm{~m} / \mathrm{s}$ and $\overrightarrow{v_2}=4 \hat{j} \mathrm{~m} / \mathrm{s}$ respectively. The body A has acceleration $\overrightarrow{a_1}=6 \hat{i}+6 \hat{j} \mathrm{~m} / \mathrm{s}^2$ while the acceleration of the other body B is zero. The centre of mass of the two bodies moves in $\_\_\_\_$ path.
Q8.
mcq single
+4 / 1
A square loop of side 2 cm is placed in a time varying magnetic field with magnitude as $B=0.4 \sin (300 t)$ Tesla. The normal to the plane of loop makes an angle of $60^{\circ}$ with the field. The maximum induced emf produced in the loop is $\_\_\_\_$ mV .
Q9.
mcq single
+4 / 1
A cylinder with adiabatic walls is closed at both ends and is divided into two compartments by a frictionless adiabatic piston. Ideal gas is filled in both (left and right) the compartments at same $P, V$,
T. Heating is started from left side until pressure changes to $27 \mathrm{P} / 8$. If initial volume of each compartment was 9 litres then the final volume in right-hand side compartment is $\_\_\_\_$ litres. (for this ideal gas $\mathrm{C}_{\mathrm{P}} / \mathrm{C}_{\mathrm{V}}=1.5$ )
Q10.
mcq single
+4 / 1
If 2 mole of an ideal monoatomic gas at temperature $T$, is mixed with 6 mole of another ideal monoatomic gas at temperature $2 T$ then the temperature of mixture is:
Q11.
mcq single
+4 / 1
A cylindrical vessel of 40 cm radius is completely filled with water and its capacity is $528 \mathrm{dm}^3$ (dm : decimeter) The vessel is placed on a solid block of exactly same height as vessel. If a small hole is made at 70 cm below the top of water level, then horizontal range of water falling on the ground in the beginning is $\_\_\_\_$ cm .
Q12.
mcq single
+4 / 1
Figure represents the extension $(\Delta l)$ of a wire of length 1 meter, suspended from the ceiling of the room at one end with a load $W$ connected to the other end. If the cross-sectional area of the wire is $10^{-5} \mathrm{~m}^2$ then the Young's modulus of the wire is $\_\_\_\_$ $\mathrm{N} / \mathrm{m}^2$.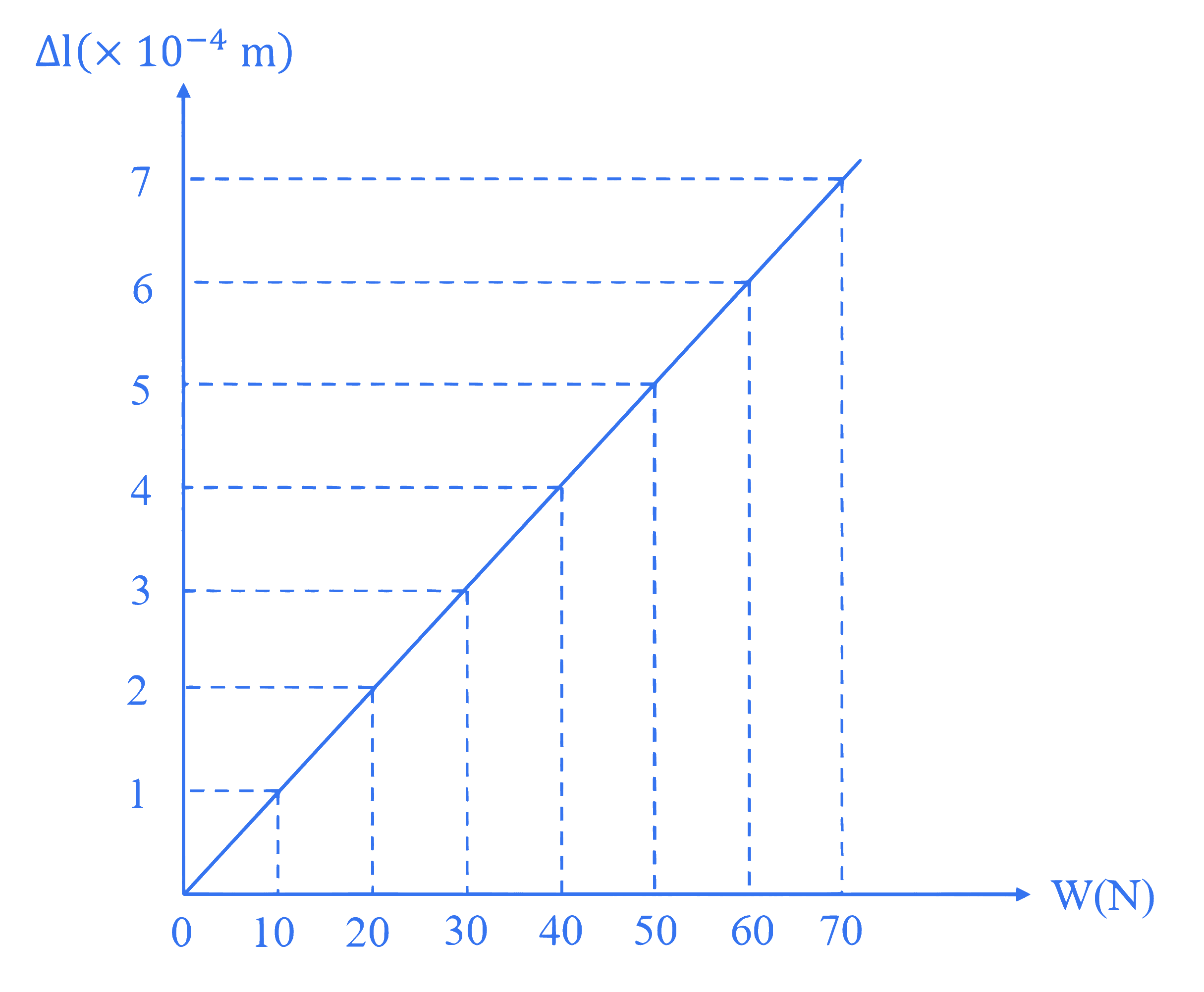

Q13.
numerical
+4 / 1
The de Broglie wavelength for an electron accelerated through the potential difference of $V_1$ volt is $\lambda_1$. When the potential difference is changed to $V_2$ volt, the associated de Broglie wavelength is increased by $50 \%$. If $\left(V_1 / V_2\right)=(9 / \alpha)$, then the value of $\alpha$ is $\_\_\_\_$。
Q14.
mcq single
+4 / 1
The electric potential as a function of $x, y$ is given by $V=5\left(x^2-y^2\right) V$. The electric field at a point $(2,3) \mathrm{m}$ is $\_\_\_\_$ $\mathrm{V} / \mathrm{m}$.
Q15.
mcq single
+4 / 1
A sphere of capacitance 100 pF is charged to a potential of 100 V . Another identical uncharged metal sphere is brought in contact with the charged sphere, then the change in the total energy stored on these spheres, when they touch is $\alpha \times 10^{-7} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
(combined capacitance of spheres is 200 pF )
Q16.
numerical
+4 / 1
A concave mirror of focal length 10 cm forms an image which is double the size of object when the object is placed at two different positions. The distance between the two positions of the object is
$\_\_\_\_$ cm.
Q17.
mcq single
+4 / 1
Angle of minimum deviation is equal to the half of the angle of prism in an equilateral prism. The refractive index of the prism is $\_\_\_\_$
Q18.
mcq single
+4 / 1
A current of 30 A each flows in opposite directions in two conducting wires, placed parallel to each other at a distance of 8 cm . The magnetic field at the mid point between the two wires is $\_\_\_\_$ $\mu \mathrm{T}$.
$$ \left(\frac{\mu_{\mathrm{o}}}{4 \pi}=10^{-7} \mathrm{~N} / \mathrm{A}^2\right) $$
Q19.
numerical
+4 / 1
A moving coil of galvanometer when shunted with $2 \Omega$ resistance gives a full scale deflection for a current of 500 mA . When a resistance of $470 \Omega$ is connected in series it gives a full scale deflection for 10 V potential applied on it. The value of resistance of galvanometer coil is $\_\_\_\_$ $\Omega$.
Q20.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
$$
\text { List - I }
$$
$$
\text { List - II }
$$
A.
Boltzmann constant
I.
$$
\left[\mathrm{M}^{-1} \mathrm{~L}^3 \mathrm{~T}^{-2}\right]
$$
B.
Stefan's constant
II.
$$
\left[\mathrm{M} \mathrm{~L}^2 \mathrm{~T}^{-1}\right]
$$
C.
Planck's constant
III.
$$
\left[\mathrm{ML}^2 \mathrm{~T}^{-2} \mathrm{~K}^{-1}\right]
$$
D.
Gravitational constant
IV.
$$
\left[\mathrm{M} \mathrm{~L}^0 \mathrm{~T}^{-3} \mathrm{~K}^{-4}\right]
$$
Choose the correct answer from the options given below :
Q21.
mcq single
+4 / 1
The percentage error in the calculated volume of a sphere, if there is $2 \%$ error in its diameter measurement, is $\_\_\_\_$ .
Q22.
mcq single
+4 / 1
The energy released if hydrogen atoms are combined to form ${ }_2^4 \mathrm{He}$ is $\_\_\_\_$ MeV .
(Take binding energies per nucleon of ${ }_1^2 \mathrm{H}$ and ${ }_2^4 \mathrm{He}$ as 1.1 MeV and 7.2 MeV , respectively)
Q23.
numerical
+4 / 1
A block takes $t$ time to slide down a plane inclined at $45^{\circ}$ to the horizontal. If the surface is made smooth (frictionless), the block takes time $\frac{t}{2}$ to slide down the plane. The coefficient of friction between the block and the inclined plane is $\left(\frac{\alpha}{100}\right)$. The value of $\alpha$ is $\_\_\_\_$ .
Q24.
numerical
+4 / 1
Two cells of emfs 1 V and 2 V and internal resistance $2 \Omega$ and $1 \Omega$, respectively connected in parallel, gave a current of 1 A through an external resistance. If the polarity of one cell is reversed, then value of current through the external resistance will be $\frac{\alpha}{5} \mathrm{~A}$. The value of $\alpha$ is $\_\_\_\_$.
Q25.
mcq single
+4 / 1
For an electromagnetic wave propagating through vacuum, $\vec{k}, \vec{E}$ and $\omega$ represent propagation vector, electric field and angular frequency, respectively. The magnetic field associated with this wave is represented by:
Q26.
mcq single
+4 / 1
A body of mass 1 kg moves along a straight line with a velocity $v=2 x^2$. The work done by the body during displacement from $x=0$ to 5 m is $\_\_\_\_$ J.
Q27.
mcq single
+4 / 1
The velocity at which 6 kg mass (shown in figure) strikes the ground when it is released from a height of 6 m above the ground is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}$. Assume pulley is massless and string is light and inextensible. (Take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )

Q28.
mcq single
+4 / 1
Refer to the logic circuit given below. For two inputs ( $A=1, B=1$ ) and ( $A=0, B=1$ ), output ( Y ) will be $\_\_\_\_$ .

Q29.
mcq single
+4 / 1
In a Young double slit experiment, the wavelength of incident light is $6000 \mathop {\rm{A}}\limits^{\rm{o}}$, the separation between slits $S_1$ and $S_2$ is 5 cm and the distance between slits plane and screen is 50 cm , as shown in the figure below. If the resultant intensity at $P$ is equal to the intensity due to individual slits, the path difference between interfering waves is $\_\_\_\_$ Å.

Q30.
mcq single
+4 / 1
A spring stretches by 2 mm when it is loaded with a mass of 200 g . From equilibrium position the mass is further pulled down by 2 mm and released. The frequency associated with the system and maxmimum energy in the spring are $\_\_\_\_$ Hz and $\_\_\_\_$ J, respectively.
(Take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q31.
mcq single
+4 / 1
A solid sphere $(A)$ of mass $5 m$ and a spherical shell $(B)$ of mass $m$, both having same radius, are placed on a rough surface. When a force of same magnitude is applied tangentially at the highest points of $A$ and $B$, they start rolling without slipping with an acceleration of $a_A$ and $a_B$, respectively. The ratio of $a_A$ and $a_B$ is $\_\_\_\_$ .
Q32.
mcq single
+4 / 1
Two identical bodies A and B of equal masses have initial velocities $\overrightarrow{v_1}=4 \hat{i} \mathrm{~m} / \mathrm{s}$ and $\overrightarrow{v_2}=4 \hat{j} \mathrm{~m} / \mathrm{s}$ respectively. The body A has acceleration $\overrightarrow{a_1}=6 \hat{i}+6 \hat{j} \mathrm{~m} / \mathrm{s}^2$ while the acceleration of the other body B is zero. The centre of mass of the two bodies moves in $\_\_\_\_$ path.
Q33.
mcq single
+4 / 1
A square loop of side 2 cm is placed in a time varying magnetic field with magnitude as $B=0.4 \sin (300 t)$ Tesla. The normal to the plane of loop makes an angle of $60^{\circ}$ with the field. The maximum induced emf produced in the loop is $\_\_\_\_$ mV .
Q34.
mcq single
+4 / 1
A cylinder with adiabatic walls is closed at both ends and is divided into two compartments by a frictionless adiabatic piston. Ideal gas is filled in both (left and right) the compartments at same $P, V$,
T. Heating is started from left side until pressure changes to $27 \mathrm{P} / 8$. If initial volume of each compartment was 9 litres then the final volume in right-hand side compartment is $\_\_\_\_$ litres. (for this ideal gas $\mathrm{C}_{\mathrm{P}} / \mathrm{C}_{\mathrm{V}}=1.5$ )
Q35.
mcq single
+4 / 1
If 2 mole of an ideal monoatomic gas at temperature $T$, is mixed with 6 mole of another ideal monoatomic gas at temperature $2 T$ then the temperature of mixture is:
Q36.
mcq single
+4 / 1
A cylindrical vessel of 40 cm radius is completely filled with water and its capacity is $528 \mathrm{dm}^3$ (dm : decimeter) The vessel is placed on a solid block of exactly same height as vessel. If a small hole is made at 70 cm below the top of water level, then horizontal range of water falling on the ground in the beginning is $\_\_\_\_$ cm .
Q37.
mcq single
+4 / 1
Figure represents the extension $(\Delta l)$ of a wire of length 1 meter, suspended from the ceiling of the room at one end with a load $W$ connected to the other end. If the cross-sectional area of the wire is $10^{-5} \mathrm{~m}^2$ then the Young's modulus of the wire is $\_\_\_\_$ $\mathrm{N} / \mathrm{m}^2$.

Q38.
numerical
+4 / 1
The de Broglie wavelength for an electron accelerated through the potential difference of $V_1$ volt is $\lambda_1$. When the potential difference is changed to $V_2$ volt, the associated de Broglie wavelength is increased by $50 \%$. If $\left(V_1 / V_2\right)=(9 / \alpha)$, then the value of $\alpha$ is $\_\_\_\_$。
Q39.
mcq single
+4 / 1
The electric potential as a function of $x, y$ is given by $V=5\left(x^2-y^2\right) V$. The electric field at a point $(2,3) \mathrm{m}$ is $\_\_\_\_$ $\mathrm{V} / \mathrm{m}$.
Q40.
mcq single
+4 / 1
A sphere of capacitance 100 pF is charged to a potential of 100 V . Another identical uncharged metal sphere is brought in contact with the charged sphere, then the change in the total energy stored on these spheres, when they touch is $\alpha \times 10^{-7} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
(combined capacitance of spheres is 200 pF )
Q41.
numerical
+4 / 1
A concave mirror of focal length 10 cm forms an image which is double the size of object when the object is placed at two different positions. The distance between the two positions of the object is
$\_\_\_\_$ cm.
Q42.
mcq single
+4 / 1
Angle of minimum deviation is equal to the half of the angle of prism in an equilateral prism. The refractive index of the prism is $\_\_\_\_$
Q43.
mcq single
+4 / 1
A current of 30 A each flows in opposite directions in two conducting wires, placed parallel to each other at a distance of 8 cm . The magnetic field at the mid point between the two wires is $\_\_\_\_$ $\mu \mathrm{T}$.
$$ \left(\frac{\mu_{\mathrm{o}}}{4 \pi}=10^{-7} \mathrm{~N} / \mathrm{A}^2\right) $$
Q44.
numerical
+4 / 1
A moving coil of galvanometer when shunted with $2 \Omega$ resistance gives a full scale deflection for a current of 500 mA . When a resistance of $470 \Omega$ is connected in series it gives a full scale deflection for 10 V potential applied on it. The value of resistance of galvanometer coil is $\_\_\_\_$ $\Omega$.
Q45.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
$$
\text { List - I }
$$
$$
\text { List - II }
$$
A.
Boltzmann constant
I.
$$
\left[\mathrm{M}^{-1} \mathrm{~L}^3 \mathrm{~T}^{-2}\right]
$$
B.
Stefan's constant
II.
$$
\left[\mathrm{M} \mathrm{~L}^2 \mathrm{~T}^{-1}\right]
$$
C.
Planck's constant
III.
$$
\left[\mathrm{ML}^2 \mathrm{~T}^{-2} \mathrm{~K}^{-1}\right]
$$
D.
Gravitational constant
IV.
$$
\left[\mathrm{M} \mathrm{~L}^0 \mathrm{~T}^{-3} \mathrm{~K}^{-4}\right]
$$
Choose the correct answer from the options given below :
Q46.
mcq single
+4 / 1
The percentage error in the calculated volume of a sphere, if there is $2 \%$ error in its diameter measurement, is $\_\_\_\_$ .
Q47.
mcq single
+4 / 1
The energy released if hydrogen atoms are combined to form ${ }_2^4 \mathrm{He}$ is $\_\_\_\_$ MeV .
(Take binding energies per nucleon of ${ }_1^2 \mathrm{H}$ and ${ }_2^4 \mathrm{He}$ as 1.1 MeV and 7.2 MeV , respectively)
Q48.
numerical
+4 / 1
A block takes $t$ time to slide down a plane inclined at $45^{\circ}$ to the horizontal. If the surface is made smooth (frictionless), the block takes time $\frac{t}{2}$ to slide down the plane. The coefficient of friction between the block and the inclined plane is $\left(\frac{\alpha}{100}\right)$. The value of $\alpha$ is $\_\_\_\_$ .
Q49.
numerical
+4 / 1
Two cells of emfs 1 V and 2 V and internal resistance $2 \Omega$ and $1 \Omega$, respectively connected in parallel, gave a current of 1 A through an external resistance. If the polarity of one cell is reversed, then value of current through the external resistance will be $\frac{\alpha}{5} \mathrm{~A}$. The value of $\alpha$ is $\_\_\_\_$.
Q50.
mcq single
+4 / 1
For an electromagnetic wave propagating through vacuum, $\vec{k}, \vec{E}$ and $\omega$ represent propagation vector, electric field and angular frequency, respectively. The magnetic field associated with this wave is represented by: