JEE Main 2026 (Online) 6th April Morning Shift
JEE 2026 Previous Year
3 hDuration
300Total Marks
150Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 150 across 3 section(s); maximum marks: 300.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
numerical
+4 / 1
$$ \text { Consider the following sequence of reactions. } $$
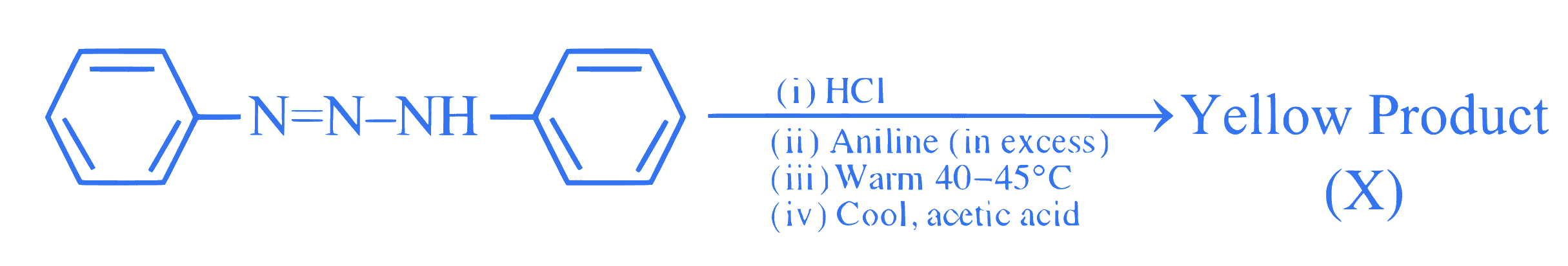 The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
 The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
Q2.
mcq single
+4 / 1
Arrange the following compounds according to increasing order of boiling points.
$$ \mathrm{n}-\mathrm{C}_4 \mathrm{H}_9 \mathrm{OH}(\mathrm{~A}), \mathrm{n}-\mathrm{C}_4 \mathrm{H}_9 \mathrm{NH}_2(\mathrm{~B}), \mathrm{n}-\mathrm{C}_4 \mathrm{H}_{10}(\mathrm{C}) \text { and } \mathrm{C}_2 \mathrm{H}_5 \mathrm{NHC}_2 \mathrm{H}_5(\mathrm{D}) . $$
Q3.
mcq single
+4 / 1
The correct statements among the following are :
A. Basic vanadium oxide is used in the manufacture of $\mathrm{H}_2 \mathrm{SO}_4$.
B. The spin-only magnetic moment value of the transition metal halide employed in Ziegler-Natta polymerization is 2.84 BM .
C. The p-block metal compound employed in Ziegler-Natta polymerization has the metal in +3 oxidation state.
D. The number of electrons present in the outer most ' d ' orbital of metal halide employed in Wacker process is 8.
Choose the correct answer from the options given below :
Q4.
numerical
+4 / 1
Sucrose hydrolyses in acidic medium into glucose and fructose by first order rate law with $t_{1 / 2}=3$ hour. The percentage of sucrose remaining after 6 hours is
$\_\_\_\_$ . (Nearest integer)
(Given $: \log 2=0.3010$ and $\log 3=0.4771$ )
Q5.
mcq single
+4 / 1
The pairs among
$\mathrm{A}=\left[\mathrm{SO}_3^{2-}, \mathrm{CO}_3^{2-}\right], \mathrm{B}=\left[\mathrm{O}_2^{2-}, \mathrm{F}_2\right], \mathrm{C}=\left[\mathrm{CN}^{-}, \mathrm{CO}\right], \mathrm{D}=\left[\mathrm{NH}_3, \mathrm{H}_3 \mathrm{O}^{+}\right]$and $\mathrm{E}=\left[\mathrm{MnO}_4^{2-}, \mathrm{CrO}_4^{2-}\right]$ that do not have similar Lewis dot structure are
Q6.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** 3-phenylpropene reacts with HBr and gives secondary alkyl bromide having a chiral carbon atom as the major product.
**Statement II :** Aryl chlorides and aryl cyanides can be prepared by Sandmeyer reaction as well as Gattermann reaction.
In the light of the above statements, choose the correct answer from the options given below
Q7.
mcq single
+4 / 1
When 0.25 moles of a non-volatile, non-ionizable solute was dissolved in 1 mole of a solvent the vapor pressure of solution was $x \%$ of vapor pressure of pure solvent. What is $x \%$ ?
Q8.
mcq single
+4 / 1
An oxide of iron contains $69.9 \%$ iron, its empirical formula, is :
(Given : Molar mass of Fe and O are 56 and $16 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.)
Q9.
mcq single
+4 / 1
One mole each of He and $\mathrm{A}(\mathrm{g})$ are taken in a 10 L closed flask and heated to 400 K to establish the following equilibrium.
$$ \mathrm{A}(\mathrm{~g}) \rightleftharpoons \mathrm{B}(\mathrm{~g}) $$
$\mathrm{K}_{\mathrm{c}}$ for this reaction at 400 K is 4.0 . The partial pressures (in atm) of He and B(g) are respectively (at equilibrium)
(Assume $\mathrm{He}, \mathrm{A}(\mathrm{g})$ and $\mathrm{B}(\mathrm{g})$ behave as ideal gases)
(Given : $\mathrm{R}=0.082 \mathrm{~L} \mathrm{~atm} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ )
Q10.
numerical
+4 / 1
First and second ionization enthalpies of lithium are $520 \mathrm{~kJ} \mathrm{~mol}^{-1}$ and $7297 \mathrm{~kJ} \mathrm{~mol}^{-1}$ respectively. Energy required to convert 3.5 mg lithium (g) into $\mathrm{Li}^{2+}(\mathrm{g})\left[\mathrm{Li}(\mathrm{g}) \rightarrow \mathrm{Li}^{2+}(\mathrm{g})\right]$ is $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (nearest integer)
[Molar mass of $\mathrm{Li}=7 \mathrm{~g} \mathrm{~mol}^{-1}$ ]
Q11.
numerical
+4 / 1
Consider the reaction $\mathrm{X} \rightleftharpoons \mathrm{Y}$ at 300 K . If $\Delta \mathrm{H}^\theta$ and K are $28.40 \mathrm{~kJ} \mathrm{~mol}^{-1}$ and $1.8 \times 10^{-7}$ at the same temperature, then the magnitude of $\Delta \mathrm{S}^\theta$ for the reaction in $\mathrm{JK}^{-1} \mathrm{~mol}^{-1}$ is $\_\_\_\_$ . (Nearest integer)
(Given : $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \ln 10=2.3, \log 3=0.48, \log 2=0.30$ )
Q12.
mcq single
+4 / 1
Arrange the following isothermal processes in order of the magnitude of the work $(\mathrm{p}-\mathrm{V})$ involved between states 1 and 2.
A. Expansion in single stage $\mathrm{w}_{\mathrm{A}}$
B. Expansion in multi stages $w_B$
C. Compression in single stage $\mathrm{w}_{\mathrm{C}}$
D. Compression in multi stages $w_D$
Choose the correct option.
Q13.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Aluminium is more electropositive than thallium as the standard electrode potential value of $\mathrm{E}^{\circ} \mathrm{Al}^{3+} / \mathrm{Al}$ is negative and $\mathrm{E}^{\circ} \mathrm{Tl}^{3+} / \mathrm{Tl}$ is positive.
**Statement II :** The sum of first three ionization enthalpies of boron is very high when compared to that of aluminium. Due to this reason boron forms covalent compounds only and aluminium forms $\mathrm{Al}^{3+}$ ion.
In the light of the above statements, choose the correct answer from the options given below
Q14.
mcq single
+4 / 1
Which of the following are true about the energy of the given d-orbitals of a tetrahedral complex?
A. $\mathrm{d}_{x y}=\mathrm{d}_{x z}>\mathrm{d}_{{x^2}-\mathrm{y}^2}$
B. $\mathrm{d}_{x y}=\mathrm{d}_{y z}>\mathrm{d}_{z^2}$
C. $\mathrm{d}_{x^2-y^2}>\mathrm{d}_{z^2}>\mathrm{d}_{x z}$
D. $\mathrm{d}_{{x^2}-y^2}=\mathrm{d}_{z^2}<\mathrm{d}_{x z}$
Choose the correct answer from the given below :
Q15.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
Electronic configuration of tetrahedral metal ion
$$
\begin{gathered}
\text { List-II } \\
\text { Crystal Field Stabilization } \\
\text { Energy }\left(\Delta_t\right)
\end{gathered}
$$
A.
$$
\mathrm{d}^2
$$
I.
-0.6
B.
$$
\mathrm{d}^4
$$
II.
-0.8
C.
$$
\mathrm{d}^6
$$
III.
-1.2
D.
$$
\mathrm{d}^8
$$
IV.
-0.4
Choose the correct answer from the options given below:
Q16.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** 2, 6-diethylcyclohexanone and 6-methyl-2-n-propylcyclohexanone are metamers.
**Statement II : **2, 2, 6, 6 - tetramethylcyclohexanone exhibits keto-enol tautomerism.
In the light of the above statements, choose the correct answer from the options given below
Q17.
mcq single
+4 / 1
$\mathrm{R}_{\mathrm{f}}$ value for 2-methylpropene in a solvent system (Ethyl acetate + ether) is 0.42 . 2-methylpropene is treated with dilute $\mathrm{H}_2 \mathrm{SO}_4$ to give major organic product $(\mathrm{X})$. $R_f$ value for $(X)$ in the same solvent system under identical condition will be:
Q18.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Methane can be prepared by decarboxylation of sodium ethanoate, Kolbe's electrolysis of sodium acetate and reaction of $\mathrm{CH}_3 \mathrm{MgBr}$ with water.
**Statement II :** Methane cannot be prepared from unsaturated hydrocarbons and by Wurtz reaction.
In the light of the above statements, choose the correct answer from the options given below
Q19.
mcq single
+4 / 1
$$ \text { Match the List-I with List-II } $$
$$
\begin{gathered}
\text { List-I } \\
\text { Amino acid }
\end{gathered}
$$
List-II
Positive reaction/Test for functional group present in side chain of amino acid
A.
Glutamine
I.
Hinsberg's test
B.
Lysine
II.
Neutral $\mathrm{FeCl}_3$ test
C.
Tyrosine
III.
Ceric ammonium nitrate test
D.
Serine
IV.
Hoffman bromamide degradation
Choose the correct answer from the options given below :
Q20.
mcq single
+4 / 1
$$ \text { Match the List-I with List-II } $$
List-I
Deficiency Disease
List-I
Vitamin
A.
Scurvy
I.
Pyridoxine
B.
Convulsions
II.
Vitamin A
C.
Cheilosis
III.
Ascorbic Acid
D.
Xerophthalmia
IV.
Riboflavin
Choose the correct answer from the options given below :
Q21.
mcq single
+4 / 1
$$ \text { Consider the following data. } $$
$$ \begin{array}{|c|c|} \hline \text { Electrolyte } & \wedge^{\circ}_\mathbf{m}{\mathbf{(}} \mathbf{S ~ c m}^{\mathbf{2}} \mathbf{~ m o l}^{\mathbf{1}} \mathbf{)} \\ \hline \mathrm{BaCl}_2 & x_1 \\ \hline \mathrm{H}_2 \mathrm{SO}_4 & x_2 \\ \hline \mathrm{HCl} & x_3 \\ \hline \end{array} $$
$\mathrm{BaSO}_4$ is sparingly soluble in water. If the conductivity of the saturated $\mathrm{BaSO}_4$ solution is $x \mathrm{~S} \mathrm{~cm}^{-1}$ then the solubility product of $\mathrm{BaSO}_4$ can be given as (Here $\wedge_{\mathrm{m}}=\wedge^{\circ}_{\mathrm{m}}$ )
Q22.
numerical
+4 / 1
4.7 g of phenol is heated with Zn to give product X . If this reaction goes to $60 \%$ completion then the number of moles of compound X formed will be
$\_\_\_\_$ $\times 10^{-2}$.(Nearest Integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1}: \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16$ )
Q23.
mcq single
+4 / 1
Consider the following sequence of reactions
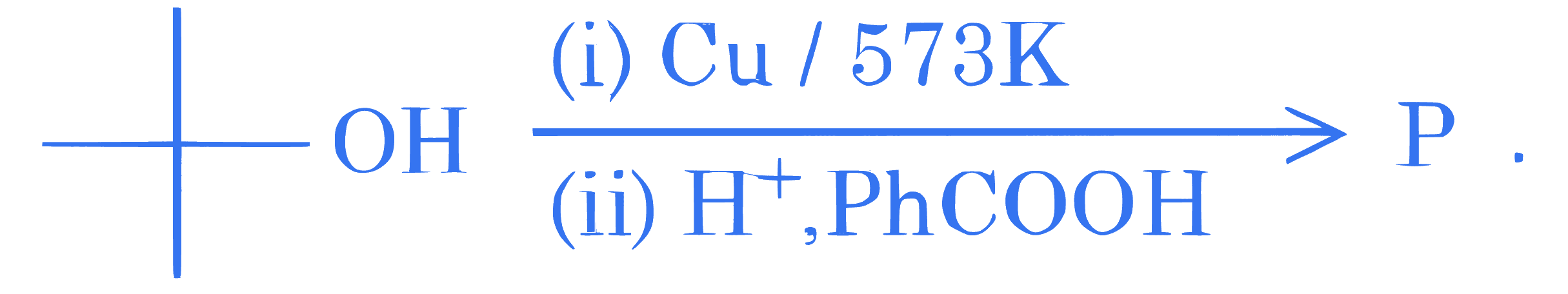 The major product P is :
The major product P is :
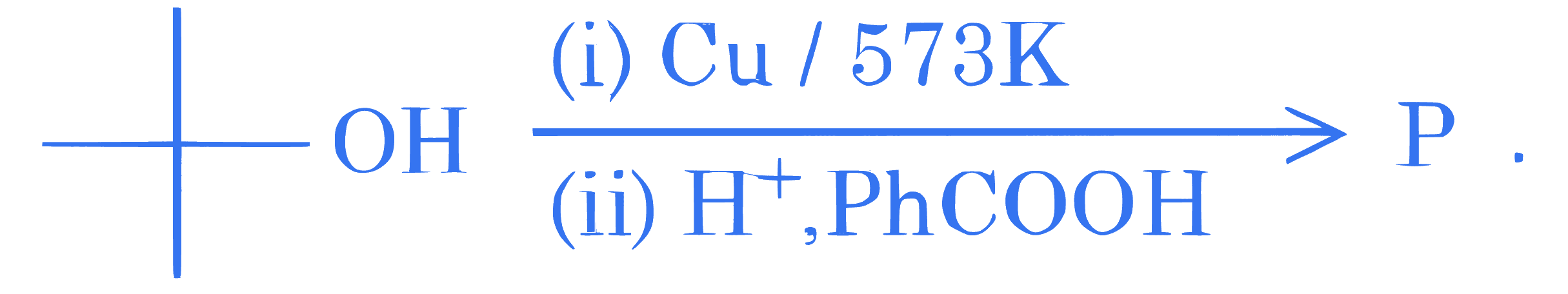 The major product P is :
The major product P is :
Q24.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
Orbital
List-II
Radial nodes and nodal plane
A.
2s
I.
1 Radial node + two nodal planes
B.
3s
II.
1 Radial node + one nodal plane
C.
3p
III.
2 Radial nodes + No nodal plane
D.
4d
IV.
1 Radial node + No nodal plane
Choose the correct answer from the options given below:
Q25.
mcq single
+4 / 1
If shortest wavelength of hydrogen atom in Lyman series is $x$, then longest wavelength in Balmer series of $\mathrm{He}^{+}$is:
Q26.
numerical
+4 / 1
$$ \text { Consider the following sequence of reactions. } $$
 The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
 The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
The percentage of nitrogen in the yellow product $(\mathrm{X})$ formed is $\_\_\_\_$ $\%$.
(Nearest Integer)
(Given Molar mass in $\mathrm{g} \mathrm{mol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{~N}: 14$ )
Q27.
mcq single
+4 / 1
Arrange the following compounds according to increasing order of boiling points.
$$ \mathrm{n}-\mathrm{C}_4 \mathrm{H}_9 \mathrm{OH}(\mathrm{~A}), \mathrm{n}-\mathrm{C}_4 \mathrm{H}_9 \mathrm{NH}_2(\mathrm{~B}), \mathrm{n}-\mathrm{C}_4 \mathrm{H}_{10}(\mathrm{C}) \text { and } \mathrm{C}_2 \mathrm{H}_5 \mathrm{NHC}_2 \mathrm{H}_5(\mathrm{D}) . $$
Q28.
mcq single
+4 / 1
The correct statements among the following are :
A. Basic vanadium oxide is used in the manufacture of $\mathrm{H}_2 \mathrm{SO}_4$.
B. The spin-only magnetic moment value of the transition metal halide employed in Ziegler-Natta polymerization is 2.84 BM .
C. The p-block metal compound employed in Ziegler-Natta polymerization has the metal in +3 oxidation state.
D. The number of electrons present in the outer most ' d ' orbital of metal halide employed in Wacker process is 8.
Choose the correct answer from the options given below :
Q29.
numerical
+4 / 1
Sucrose hydrolyses in acidic medium into glucose and fructose by first order rate law with $t_{1 / 2}=3$ hour. The percentage of sucrose remaining after 6 hours is
$\_\_\_\_$ . (Nearest integer)
(Given $: \log 2=0.3010$ and $\log 3=0.4771$ )
Q30.
mcq single
+4 / 1
The pairs among
$\mathrm{A}=\left[\mathrm{SO}_3^{2-}, \mathrm{CO}_3^{2-}\right], \mathrm{B}=\left[\mathrm{O}_2^{2-}, \mathrm{F}_2\right], \mathrm{C}=\left[\mathrm{CN}^{-}, \mathrm{CO}\right], \mathrm{D}=\left[\mathrm{NH}_3, \mathrm{H}_3 \mathrm{O}^{+}\right]$and $\mathrm{E}=\left[\mathrm{MnO}_4^{2-}, \mathrm{CrO}_4^{2-}\right]$ that do not have similar Lewis dot structure are
Q31.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** 3-phenylpropene reacts with HBr and gives secondary alkyl bromide having a chiral carbon atom as the major product.
**Statement II :** Aryl chlorides and aryl cyanides can be prepared by Sandmeyer reaction as well as Gattermann reaction.
In the light of the above statements, choose the correct answer from the options given below
Q32.
mcq single
+4 / 1
When 0.25 moles of a non-volatile, non-ionizable solute was dissolved in 1 mole of a solvent the vapor pressure of solution was $x \%$ of vapor pressure of pure solvent. What is $x \%$ ?
Q33.
mcq single
+4 / 1
An oxide of iron contains $69.9 \%$ iron, its empirical formula, is :
(Given : Molar mass of Fe and O are 56 and $16 \mathrm{~g} \mathrm{~mol}^{-1}$ respectively.)
Q34.
mcq single
+4 / 1
One mole each of He and $\mathrm{A}(\mathrm{g})$ are taken in a 10 L closed flask and heated to 400 K to establish the following equilibrium.
$$ \mathrm{A}(\mathrm{~g}) \rightleftharpoons \mathrm{B}(\mathrm{~g}) $$
$\mathrm{K}_{\mathrm{c}}$ for this reaction at 400 K is 4.0 . The partial pressures (in atm) of He and B(g) are respectively (at equilibrium)
(Assume $\mathrm{He}, \mathrm{A}(\mathrm{g})$ and $\mathrm{B}(\mathrm{g})$ behave as ideal gases)
(Given : $\mathrm{R}=0.082 \mathrm{~L} \mathrm{~atm} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ )
Q35.
numerical
+4 / 1
First and second ionization enthalpies of lithium are $520 \mathrm{~kJ} \mathrm{~mol}^{-1}$ and $7297 \mathrm{~kJ} \mathrm{~mol}^{-1}$ respectively. Energy required to convert 3.5 mg lithium (g) into $\mathrm{Li}^{2+}(\mathrm{g})\left[\mathrm{Li}(\mathrm{g}) \rightarrow \mathrm{Li}^{2+}(\mathrm{g})\right]$ is $\_\_\_\_$ $\mathrm{kJ} \mathrm{mol}^{-1}$. (nearest integer)
[Molar mass of $\mathrm{Li}=7 \mathrm{~g} \mathrm{~mol}^{-1}$ ]
Q36.
numerical
+4 / 1
Consider the reaction $\mathrm{X} \rightleftharpoons \mathrm{Y}$ at 300 K . If $\Delta \mathrm{H}^\theta$ and K are $28.40 \mathrm{~kJ} \mathrm{~mol}^{-1}$ and $1.8 \times 10^{-7}$ at the same temperature, then the magnitude of $\Delta \mathrm{S}^\theta$ for the reaction in $\mathrm{JK}^{-1} \mathrm{~mol}^{-1}$ is $\_\_\_\_$ . (Nearest integer)
(Given : $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \ln 10=2.3, \log 3=0.48, \log 2=0.30$ )
Q37.
mcq single
+4 / 1
Arrange the following isothermal processes in order of the magnitude of the work $(\mathrm{p}-\mathrm{V})$ involved between states 1 and 2.
A. Expansion in single stage $\mathrm{w}_{\mathrm{A}}$
B. Expansion in multi stages $w_B$
C. Compression in single stage $\mathrm{w}_{\mathrm{C}}$
D. Compression in multi stages $w_D$
Choose the correct option.
Q38.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Aluminium is more electropositive than thallium as the standard electrode potential value of $\mathrm{E}^{\circ} \mathrm{Al}^{3+} / \mathrm{Al}$ is negative and $\mathrm{E}^{\circ} \mathrm{Tl}^{3+} / \mathrm{Tl}$ is positive.
**Statement II :** The sum of first three ionization enthalpies of boron is very high when compared to that of aluminium. Due to this reason boron forms covalent compounds only and aluminium forms $\mathrm{Al}^{3+}$ ion.
In the light of the above statements, choose the correct answer from the options given below
Q39.
mcq single
+4 / 1
Which of the following are true about the energy of the given d-orbitals of a tetrahedral complex?
A. $\mathrm{d}_{x y}=\mathrm{d}_{x z}>\mathrm{d}_{{x^2}-\mathrm{y}^2}$
B. $\mathrm{d}_{x y}=\mathrm{d}_{y z}>\mathrm{d}_{z^2}$
C. $\mathrm{d}_{x^2-y^2}>\mathrm{d}_{z^2}>\mathrm{d}_{x z}$
D. $\mathrm{d}_{{x^2}-y^2}=\mathrm{d}_{z^2}<\mathrm{d}_{x z}$
Choose the correct answer from the given below :
Q40.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
Electronic configuration of tetrahedral metal ion
$$
\begin{gathered}
\text { List-II } \\
\text { Crystal Field Stabilization } \\
\text { Energy }\left(\Delta_t\right)
\end{gathered}
$$
A.
$$
\mathrm{d}^2
$$
I.
-0.6
B.
$$
\mathrm{d}^4
$$
II.
-0.8
C.
$$
\mathrm{d}^6
$$
III.
-1.2
D.
$$
\mathrm{d}^8
$$
IV.
-0.4
Choose the correct answer from the options given below:
Q41.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** 2, 6-diethylcyclohexanone and 6-methyl-2-n-propylcyclohexanone are metamers.
**Statement II : **2, 2, 6, 6 - tetramethylcyclohexanone exhibits keto-enol tautomerism.
In the light of the above statements, choose the correct answer from the options given below
Q42.
mcq single
+4 / 1
$\mathrm{R}_{\mathrm{f}}$ value for 2-methylpropene in a solvent system (Ethyl acetate + ether) is 0.42 . 2-methylpropene is treated with dilute $\mathrm{H}_2 \mathrm{SO}_4$ to give major organic product $(\mathrm{X})$. $R_f$ value for $(X)$ in the same solvent system under identical condition will be:
Q43.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Methane can be prepared by decarboxylation of sodium ethanoate, Kolbe's electrolysis of sodium acetate and reaction of $\mathrm{CH}_3 \mathrm{MgBr}$ with water.
**Statement II :** Methane cannot be prepared from unsaturated hydrocarbons and by Wurtz reaction.
In the light of the above statements, choose the correct answer from the options given below
Q44.
mcq single
+4 / 1
$$ \text { Match the List-I with List-II } $$
$$
\begin{gathered}
\text { List-I } \\
\text { Amino acid }
\end{gathered}
$$
List-II
Positive reaction/Test for functional group present in side chain of amino acid
A.
Glutamine
I.
Hinsberg's test
B.
Lysine
II.
Neutral $\mathrm{FeCl}_3$ test
C.
Tyrosine
III.
Ceric ammonium nitrate test
D.
Serine
IV.
Hoffman bromamide degradation
Choose the correct answer from the options given below :
Q45.
mcq single
+4 / 1
$$ \text { Match the List-I with List-II } $$
List-I
Deficiency Disease
List-I
Vitamin
A.
Scurvy
I.
Pyridoxine
B.
Convulsions
II.
Vitamin A
C.
Cheilosis
III.
Ascorbic Acid
D.
Xerophthalmia
IV.
Riboflavin
Choose the correct answer from the options given below :
Q46.
mcq single
+4 / 1
$$ \text { Consider the following data. } $$
$$ \begin{array}{|c|c|} \hline \text { Electrolyte } & \wedge^{\circ}_\mathbf{m}{\mathbf{(}} \mathbf{S ~ c m}^{\mathbf{2}} \mathbf{~ m o l}^{\mathbf{1}} \mathbf{)} \\ \hline \mathrm{BaCl}_2 & x_1 \\ \hline \mathrm{H}_2 \mathrm{SO}_4 & x_2 \\ \hline \mathrm{HCl} & x_3 \\ \hline \end{array} $$
$\mathrm{BaSO}_4$ is sparingly soluble in water. If the conductivity of the saturated $\mathrm{BaSO}_4$ solution is $x \mathrm{~S} \mathrm{~cm}^{-1}$ then the solubility product of $\mathrm{BaSO}_4$ can be given as (Here $\wedge_{\mathrm{m}}=\wedge^{\circ}_{\mathrm{m}}$ )
Q47.
numerical
+4 / 1
4.7 g of phenol is heated with Zn to give product X . If this reaction goes to $60 \%$ completion then the number of moles of compound X formed will be
$\_\_\_\_$ $\times 10^{-2}$.(Nearest Integer)
(Given molar mass in $\mathrm{g} \mathrm{mol}^{-1}: \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16$ )
Q48.
mcq single
+4 / 1
Consider the following sequence of reactions
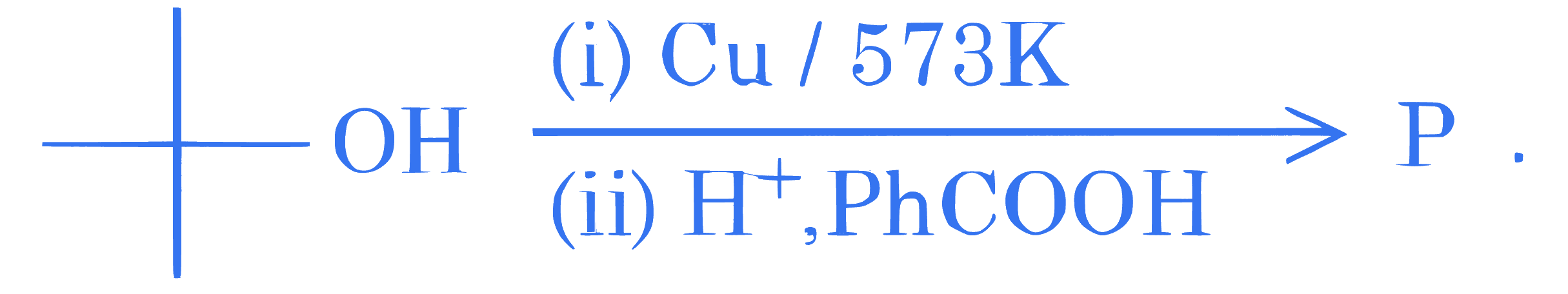 The major product P is :
The major product P is :
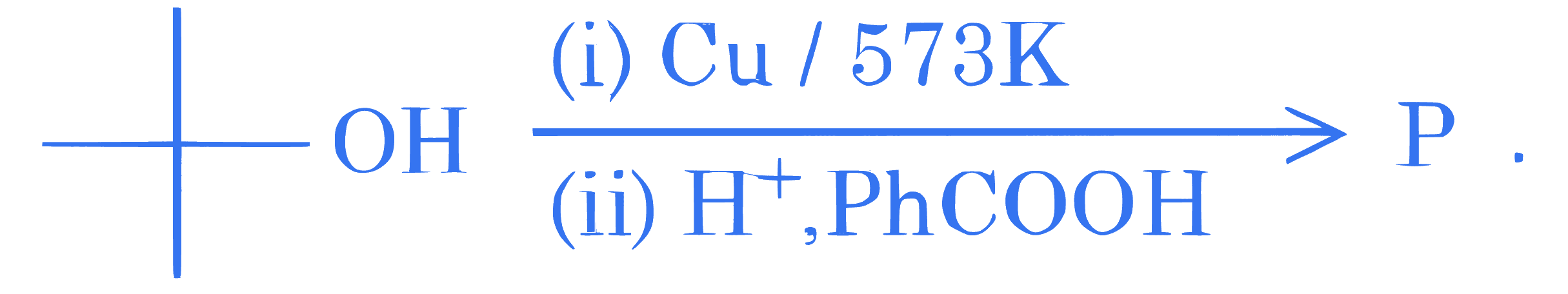 The major product P is :
The major product P is :
Q49.
mcq single
+4 / 1
$$ \text { Match the LIST-I with LIST-II } $$
List-I
Orbital
List-II
Radial nodes and nodal plane
A.
2s
I.
1 Radial node + two nodal planes
B.
3s
II.
1 Radial node + one nodal plane
C.
3p
III.
2 Radial nodes + No nodal plane
D.
4d
IV.
1 Radial node + No nodal plane
Choose the correct answer from the options given below:
Q50.
mcq single
+4 / 1
If shortest wavelength of hydrogen atom in Lyman series is $x$, then longest wavelength in Balmer series of $\mathrm{He}^{+}$is:
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
A data consists of 20 observations $x_1, x_2, \ldots, x_{20}$. If $\sum\limits_{i=1}^{20}\left(x_i+5\right)^2=2500$ and $\sum\limits_{i=1}^{20}\left(x_i-5\right)^2=100$, then the ratio of mean to standard deviation of this data is :
Q2.
numerical
+4 / 1
If $\vec{a}=\hat{i}+\hat{j}+\hat{k}, \vec{b}=\hat{j}-\hat{k}$ and $\vec{c}$ be three vectors such that $\vec{a} \times \vec{c}=\vec{b}$ and $\vec{a} \cdot \vec{c}=3$, then $\vec{c} \cdot(\vec{a}-2 \vec{b})$ is equal to $\_\_\_\_$ .
Q3.
mcq single
+4 / 1
A bag contains $(\mathrm{N}+1)$ coins -N fair coins, and one coin with 'Head' on both sides. A coin is selected at random and tossed. If the probability of getting 'Head' is $\frac{9}{16}$, then N is equal to:
Q4.
mcq single
+4 / 1
Let the image of the point $\mathrm{P}(1,6, a)$ in the line $\mathrm{L}: \frac{x}{1}=\frac{y-1}{2}=\frac{z-a+1}{b}, b>0$, be $\left(\frac{a}{3}, 0, a+c\right)$. If $\mathrm{S}(\alpha, \beta, \gamma), \alpha>0$, is the point on L such that the distance of S from the foot of perpendicular from the point P on L is $2 \sqrt{14}$, then $\alpha+\beta+\gamma$ is equal to:
Q5.
mcq single
+4 / 1
Let a line L be perpendicular to both the lines $\mathrm{L}_1: \frac{x+1}{3}=\frac{y+3}{5}=\frac{z+5}{7}$ and $\mathrm{L}_2: \frac{x-2}{1}=\frac{y-4}{4}=\frac{z-6}{7}$.
If $\theta$ is the acute angle between the lines L and $\mathrm{L}_3: \frac{x-\frac{8}{7}}{2}=\frac{y-\frac{4}{7}}{1}=\frac{z}{2}$, then $\tan \theta$ is equal to:
Q6.
mcq single
+4 / 1
The sum of the first ten terms of an A.P. is 160 and the sum of the first two terms of a G.P. is 8 . If the first term of the A.P. is equal to the common ratio of the G.P. and the first term of the G.P. is equal to common difference of the A.P., then the sum of all possible values of the first term of the G.P. is:
Q7.
numerical
+4 / 1
For the functions $f(\theta)=\alpha \tan ^2 \theta+\beta \cot ^2 \theta$, and $g(\theta)=\alpha \sin ^2 \theta+\beta \cos ^2 \theta, \alpha>\beta>0$, let $\min\limits_{0<\theta<\frac{\pi}{2}} f(\theta)=\max\limits_{0<\theta<\pi} g(\theta)$. If the first term of a G.P. is $\left(\frac{\alpha}{2 \beta}\right)$, its common ratio is $\left(\frac{2 \beta}{\alpha}\right)$ and the sum of its first 10 terms is $\frac{m}{n}, \operatorname{gcd}(m, n)=1$, then $m+n$ is equal to $\_\_\_\_$ .
Q8.
mcq single
+4 / 1
The value of $1^3-2^3+3^3-\ldots+15^3$ is:
Q9.
mcq single
+4 / 1
$$ \text { The value of } \lim\limits_{x \rightarrow 0}\left(\frac{x^2 \sin ^2 x}{x^2-\sin ^2 x}\right) \text { is : } $$
Q10.
mcq single
+4 / 1
Let $e$ be the base of natural logarithm and let $f:\{1,2,3,4\} \rightarrow\left\{1, e, e^2, e^3\right\}$ and $\mathrm{g}:\left\{1, e, e^2, e^3\right\} \rightarrow\left\{1, \frac{1}{2}, \frac{1}{3}, \frac{1}{4}\right\}$ be two bijective functions such that $f$ is strictly decreasing and $g$ is strictly increasing. If $\phi(x)=\left[f^{-1}\left\{g^{-1}\left(\frac{1}{2}\right)\right\}\right]^x$, then the area of the region $\mathrm{R}=\left\{(x, y): x^2 \leq y \leq \phi(x), 0 \leq x \leq 1\right\}$ is :
Q11.
mcq single
+4 / 1
The area of the region $\left\{(x, y): 0 \leq y \leq 6-x, y^2 \geq 4 x-3, x \geq 0\right\}$ is :
Q12.
numerical
+4 / 1
Let $A=\left[\begin{array}{ccc}-1 & 1 & -1 \\ 1 & 0 & 1 \\ 0 & 0 & 1\end{array}\right]$ satisfy
$\mathrm{A}^2+\alpha(\operatorname{adj}(\operatorname{adj}(\mathrm{A})))+\beta(\operatorname{adj}(\mathrm{A})(\operatorname{adj}(\operatorname{adj}(\mathrm{A}))))=\left[\begin{array}{ccc}2 & -2 & 2 \\ -2 & 0 & -1 \\ 0 & 0 & -1\end{array}\right]$ for some $\alpha, \beta \in \mathbb{R}$.
Then $(\alpha-\beta)^2$ is equal to $\_\_\_\_$
Q13.
mcq single
+4 / 1
Let $S=\{\theta \in(-2 \pi, 2 \pi): \cos \theta+1=\sqrt{3} \sin \theta\}$.
Then $\sum\limits_{\theta \in \mathrm{S}} \theta$ is equal to :
Q14.
mcq single
+4 / 1
Let one root of the quadratic equation in $x$ :
$$ \left(k^2-15 k+27\right) x^2+9(k-1) x+18=0 $$
be twice the other. Then the length of the latus rectum of the parabola $y^2=6 k x$ is equal to:
Q15.
mcq single
+4 / 1
Let chord PQ of length $3 \sqrt{13}$ of the parabola $y^2=12 x$ be such that the ordinates of points P and Q are in the ratio 1:2. If the chord PQ subtends an angle $\alpha$ at the focus of the parabola, then $\sin \alpha$ is equal to :
Q16.
mcq single
+4 / 1
Let $0<\alpha<1, \beta=\frac{1}{3 \alpha}$ and $\tan ^{-1}(1-\alpha)+\tan ^{-1}(1-\beta)=\frac{\pi}{4}$. Then $6(\alpha+\beta)$ is equal to:
Q17.
mcq single
+4 / 1
If the coefficients of the middle terms in the binomial expansions of $(1+\alpha x)^{26}$ and $(1-\alpha x)^{28}, \alpha \neq 0$, are equal, then the value of $\alpha$ is:
Q18.
mcq single
+4 / 1
Let the set of all values of $k \in \mathbb{R}$ such that the equation $z(\bar{z}+2+i)+k(2+3 i)=0, z \in \mathrm{C}$, has at least one solution, be the interval $[\alpha, \beta]$. Then $9(\alpha+\beta)$ is equal to:
Q19.
mcq single
+4 / 1
Let $e_1$ and $e_2$ be two distinct roots of the equation $x^2-a x+2=0$. Let the sets $\left\{a \in \mathbb{R}: e_1\right.$ and $e_2$ are the eccentricities of hyperbolas $\}=(\alpha, \beta)$, and $\left\{a \in \mathbb{R}: e_1\right.$ and $e_2$ are the eccentricities of an ellipse and a hyperbola, respectively $\}=(\gamma, \infty)$.
Then $\alpha^2+\beta^2+\gamma^2$ is equal to:
Q20.
mcq single
+4 / 1
If the eccentricity $e$ of the hyperbola $\frac{x^2}{a^2}-\frac{y^2}{b^2}=1$, passing through $(6,4 \sqrt{3})$, satisfies $15\left(e^2+1\right)=34 e$, then the length of the latus rectum of the hyperbola $\frac{x^2}{b^2}-\frac{y^2}{2\left(a^2+1\right)}=1$ is:
Q21.
mcq single
+4 / 1
Let [ • ] denote the greatest integer function. If the domain of the function $f(x)=\sin ^{-1}\left(\frac{x+[x]}{3}\right)$ is $[\alpha, \beta)$, then $\alpha^2+\beta^2$ is equal to:
Q22.
numerical
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $\left(x^2-x \sqrt{x^2-1}\right) d y+\left(y\left(x-\sqrt{x^2-1}\right)-x\right) d x=0, x \geq 1$. If $y(1)=1$, then the greatest integer less than $y(\sqrt{5})$ is $\_\_\_\_$ .
Q23.
numerical
+4 / 1
Let the centre of the circle $x^2+y^2+2 \mathrm{~g} x+2 f y+25=0$ be in the first quadrant and lie on the line $2 x-y=4$. Let the area of an equilateral triangle inscribed in the circle be $27 \sqrt{3}$. Then the square of the length of the chord of the circle on the line $x=1$ is $\_\_\_\_$ .
Q24.
mcq single
+4 / 1
The number of 4-letter words, with or without meaning, each consisting of two vowels and two consonants that can be formed from the letters of the word INCONSEQUENTIAL, without repeating any letter, is:
Q25.
mcq single
+4 / 1
The value of the integral $\int\limits_{-\frac{\pi}{4}}^{\frac{\pi}{4}}\left(\frac{32 \cos ^4 x}{1+e^{\sin x}}\right) d x$ is :
Q26.
mcq single
+4 / 1
A data consists of 20 observations $x_1, x_2, \ldots, x_{20}$. If $\sum\limits_{i=1}^{20}\left(x_i+5\right)^2=2500$ and $\sum\limits_{i=1}^{20}\left(x_i-5\right)^2=100$, then the ratio of mean to standard deviation of this data is :
Q27.
numerical
+4 / 1
If $\vec{a}=\hat{i}+\hat{j}+\hat{k}, \vec{b}=\hat{j}-\hat{k}$ and $\vec{c}$ be three vectors such that $\vec{a} \times \vec{c}=\vec{b}$ and $\vec{a} \cdot \vec{c}=3$, then $\vec{c} \cdot(\vec{a}-2 \vec{b})$ is equal to $\_\_\_\_$ .
Q28.
mcq single
+4 / 1
A bag contains $(\mathrm{N}+1)$ coins -N fair coins, and one coin with 'Head' on both sides. A coin is selected at random and tossed. If the probability of getting 'Head' is $\frac{9}{16}$, then N is equal to:
Q29.
mcq single
+4 / 1
Let the image of the point $\mathrm{P}(1,6, a)$ in the line $\mathrm{L}: \frac{x}{1}=\frac{y-1}{2}=\frac{z-a+1}{b}, b>0$, be $\left(\frac{a}{3}, 0, a+c\right)$. If $\mathrm{S}(\alpha, \beta, \gamma), \alpha>0$, is the point on L such that the distance of S from the foot of perpendicular from the point P on L is $2 \sqrt{14}$, then $\alpha+\beta+\gamma$ is equal to:
Q30.
mcq single
+4 / 1
Let a line L be perpendicular to both the lines $\mathrm{L}_1: \frac{x+1}{3}=\frac{y+3}{5}=\frac{z+5}{7}$ and $\mathrm{L}_2: \frac{x-2}{1}=\frac{y-4}{4}=\frac{z-6}{7}$.
If $\theta$ is the acute angle between the lines L and $\mathrm{L}_3: \frac{x-\frac{8}{7}}{2}=\frac{y-\frac{4}{7}}{1}=\frac{z}{2}$, then $\tan \theta$ is equal to:
Q31.
mcq single
+4 / 1
The sum of the first ten terms of an A.P. is 160 and the sum of the first two terms of a G.P. is 8 . If the first term of the A.P. is equal to the common ratio of the G.P. and the first term of the G.P. is equal to common difference of the A.P., then the sum of all possible values of the first term of the G.P. is:
Q32.
numerical
+4 / 1
For the functions $f(\theta)=\alpha \tan ^2 \theta+\beta \cot ^2 \theta$, and $g(\theta)=\alpha \sin ^2 \theta+\beta \cos ^2 \theta, \alpha>\beta>0$, let $\min\limits_{0<\theta<\frac{\pi}{2}} f(\theta)=\max\limits_{0<\theta<\pi} g(\theta)$. If the first term of a G.P. is $\left(\frac{\alpha}{2 \beta}\right)$, its common ratio is $\left(\frac{2 \beta}{\alpha}\right)$ and the sum of its first 10 terms is $\frac{m}{n}, \operatorname{gcd}(m, n)=1$, then $m+n$ is equal to $\_\_\_\_$ .
Q33.
mcq single
+4 / 1
The value of $1^3-2^3+3^3-\ldots+15^3$ is:
Q34.
mcq single
+4 / 1
$$ \text { The value of } \lim\limits_{x \rightarrow 0}\left(\frac{x^2 \sin ^2 x}{x^2-\sin ^2 x}\right) \text { is : } $$
Q35.
mcq single
+4 / 1
Let $e$ be the base of natural logarithm and let $f:\{1,2,3,4\} \rightarrow\left\{1, e, e^2, e^3\right\}$ and $\mathrm{g}:\left\{1, e, e^2, e^3\right\} \rightarrow\left\{1, \frac{1}{2}, \frac{1}{3}, \frac{1}{4}\right\}$ be two bijective functions such that $f$ is strictly decreasing and $g$ is strictly increasing. If $\phi(x)=\left[f^{-1}\left\{g^{-1}\left(\frac{1}{2}\right)\right\}\right]^x$, then the area of the region $\mathrm{R}=\left\{(x, y): x^2 \leq y \leq \phi(x), 0 \leq x \leq 1\right\}$ is :
Q36.
mcq single
+4 / 1
The area of the region $\left\{(x, y): 0 \leq y \leq 6-x, y^2 \geq 4 x-3, x \geq 0\right\}$ is :
Q37.
numerical
+4 / 1
Let $A=\left[\begin{array}{ccc}-1 & 1 & -1 \\ 1 & 0 & 1 \\ 0 & 0 & 1\end{array}\right]$ satisfy
$\mathrm{A}^2+\alpha(\operatorname{adj}(\operatorname{adj}(\mathrm{A})))+\beta(\operatorname{adj}(\mathrm{A})(\operatorname{adj}(\operatorname{adj}(\mathrm{A}))))=\left[\begin{array}{ccc}2 & -2 & 2 \\ -2 & 0 & -1 \\ 0 & 0 & -1\end{array}\right]$ for some $\alpha, \beta \in \mathbb{R}$.
Then $(\alpha-\beta)^2$ is equal to $\_\_\_\_$
Q38.
mcq single
+4 / 1
Let $S=\{\theta \in(-2 \pi, 2 \pi): \cos \theta+1=\sqrt{3} \sin \theta\}$.
Then $\sum\limits_{\theta \in \mathrm{S}} \theta$ is equal to :
Q39.
mcq single
+4 / 1
Let one root of the quadratic equation in $x$ :
$$ \left(k^2-15 k+27\right) x^2+9(k-1) x+18=0 $$
be twice the other. Then the length of the latus rectum of the parabola $y^2=6 k x$ is equal to:
Q40.
mcq single
+4 / 1
Let chord PQ of length $3 \sqrt{13}$ of the parabola $y^2=12 x$ be such that the ordinates of points P and Q are in the ratio 1:2. If the chord PQ subtends an angle $\alpha$ at the focus of the parabola, then $\sin \alpha$ is equal to :
Q41.
mcq single
+4 / 1
Let $0<\alpha<1, \beta=\frac{1}{3 \alpha}$ and $\tan ^{-1}(1-\alpha)+\tan ^{-1}(1-\beta)=\frac{\pi}{4}$. Then $6(\alpha+\beta)$ is equal to:
Q42.
mcq single
+4 / 1
If the coefficients of the middle terms in the binomial expansions of $(1+\alpha x)^{26}$ and $(1-\alpha x)^{28}, \alpha \neq 0$, are equal, then the value of $\alpha$ is:
Q43.
mcq single
+4 / 1
Let the set of all values of $k \in \mathbb{R}$ such that the equation $z(\bar{z}+2+i)+k(2+3 i)=0, z \in \mathrm{C}$, has at least one solution, be the interval $[\alpha, \beta]$. Then $9(\alpha+\beta)$ is equal to:
Q44.
mcq single
+4 / 1
Let $e_1$ and $e_2$ be two distinct roots of the equation $x^2-a x+2=0$. Let the sets $\left\{a \in \mathbb{R}: e_1\right.$ and $e_2$ are the eccentricities of hyperbolas $\}=(\alpha, \beta)$, and $\left\{a \in \mathbb{R}: e_1\right.$ and $e_2$ are the eccentricities of an ellipse and a hyperbola, respectively $\}=(\gamma, \infty)$.
Then $\alpha^2+\beta^2+\gamma^2$ is equal to:
Q45.
mcq single
+4 / 1
If the eccentricity $e$ of the hyperbola $\frac{x^2}{a^2}-\frac{y^2}{b^2}=1$, passing through $(6,4 \sqrt{3})$, satisfies $15\left(e^2+1\right)=34 e$, then the length of the latus rectum of the hyperbola $\frac{x^2}{b^2}-\frac{y^2}{2\left(a^2+1\right)}=1$ is:
Q46.
mcq single
+4 / 1
Let [ • ] denote the greatest integer function. If the domain of the function $f(x)=\sin ^{-1}\left(\frac{x+[x]}{3}\right)$ is $[\alpha, \beta)$, then $\alpha^2+\beta^2$ is equal to:
Q47.
numerical
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $\left(x^2-x \sqrt{x^2-1}\right) d y+\left(y\left(x-\sqrt{x^2-1}\right)-x\right) d x=0, x \geq 1$. If $y(1)=1$, then the greatest integer less than $y(\sqrt{5})$ is $\_\_\_\_$ .
Q48.
numerical
+4 / 1
Let the centre of the circle $x^2+y^2+2 \mathrm{~g} x+2 f y+25=0$ be in the first quadrant and lie on the line $2 x-y=4$. Let the area of an equilateral triangle inscribed in the circle be $27 \sqrt{3}$. Then the square of the length of the chord of the circle on the line $x=1$ is $\_\_\_\_$ .
Q49.
mcq single
+4 / 1
The number of 4-letter words, with or without meaning, each consisting of two vowels and two consonants that can be formed from the letters of the word INCONSEQUENTIAL, without repeating any letter, is:
Q50.
mcq single
+4 / 1
The value of the integral $\int\limits_{-\frac{\pi}{4}}^{\frac{\pi}{4}}\left(\frac{32 \cos ^4 x}{1+e^{\sin x}}\right) d x$ is :
Physics
Physics
Q1.
mcq single
+4 / 1
The rain drop of mass 1 g , starts with zero velocity from a height of 1 km . It hits the ground with a speed of $5 \mathrm{~m} / \mathrm{s}$. The work done by the unknown resistive force is $\_\_\_\_$ J.
(take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q2.
mcq single
+4 / 1
The maximum rated power of the LED is 2 mW and it is used in the circuit with input voltage of 5 V as shown in the figure below. The current through resistance $R_S$ is 0.5 mA .
The minimum value of the resistance of $R_S$, to ensure that the LED is not damaged is $\_\_\_\_$ $\mathrm{k} \Omega$.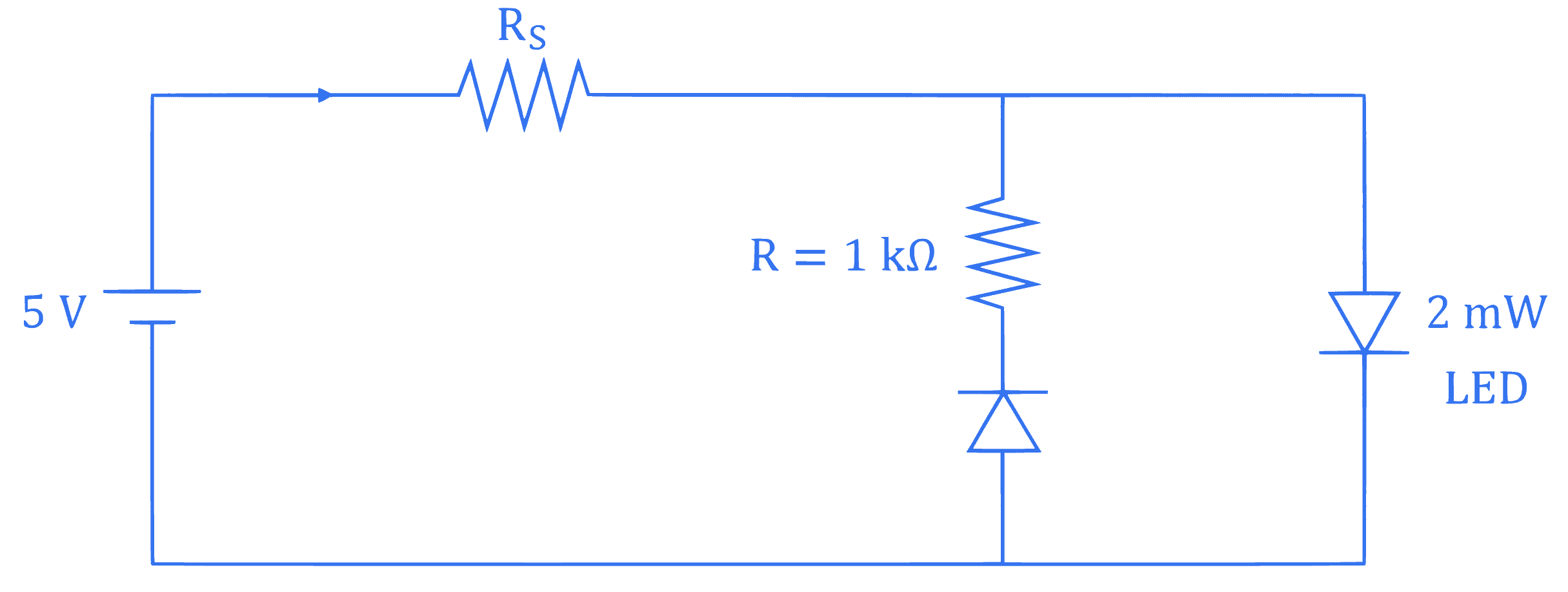

Q3.
numerical
+4 / 1
An unpolarized light of intensity $I_0$ passes through polarizer and then through a certain optically active solution and finally it goes to analyser. If the angle between analyser and polariser is $0^{\circ}$ and intensity of light emerged from analyser is $\frac{3}{8} I_0$, the angle of rotation of the light by the solution with respect to analyser is $\_\_\_\_$ degrees.
Q4.
mcq single
+4 / 1
In interference experiment the path difference between two interfering waves at a point $A$ on the screen is $\lambda / 3$, where $\lambda$ is the wavelength of these waves, and at another point $B$ the path difference is $\lambda / 6$. The ratio of intensities at points $A$ and $B$ is $\_\_\_\_$ .
Q5.
mcq single
+4 / 1
A particle is executing simple harmonic motion. Its amplitude is $A$ and time period is 5 sec . The time required by it to move from $x=A$ to $x=\frac{A}{\sqrt{2}}$ is $\_\_\_\_$ sec.
Q6.
mcq single
+4 / 1
A solid sphere of radius 4 cm and mass 5 kg is rotating (rotation axis is passing through the centre of the sphere) with an angular velocity of 1200 rpm . It is brought to rest in 10 s by applying a constant torque. The torque applied and the number of rotations it made before it comes to rest are $\_\_\_\_$ and $\_\_\_\_$ respectively.
Q7.
mcq single
+4 / 1
The position of center of mass of three masses $2 \mathrm{~kg}, 3 \mathrm{~kg}$ and 15 kg placed with respect to mid point $(p)$ of normal bisector, as shown in the figure is $\_\_\_\_$ .

Q8.
mcq single
+4 / 1
An inductor of inductance 10 mH having resistance of $100 \Omega$ is connected to battery of E.M.F. 1.0 V through a switch as shown in the figure below. After switch is closed, the ratio of instantaneous voltages across the inductor when the current passing through it is 2 mA and 4 mA is $\_\_\_\_$ .
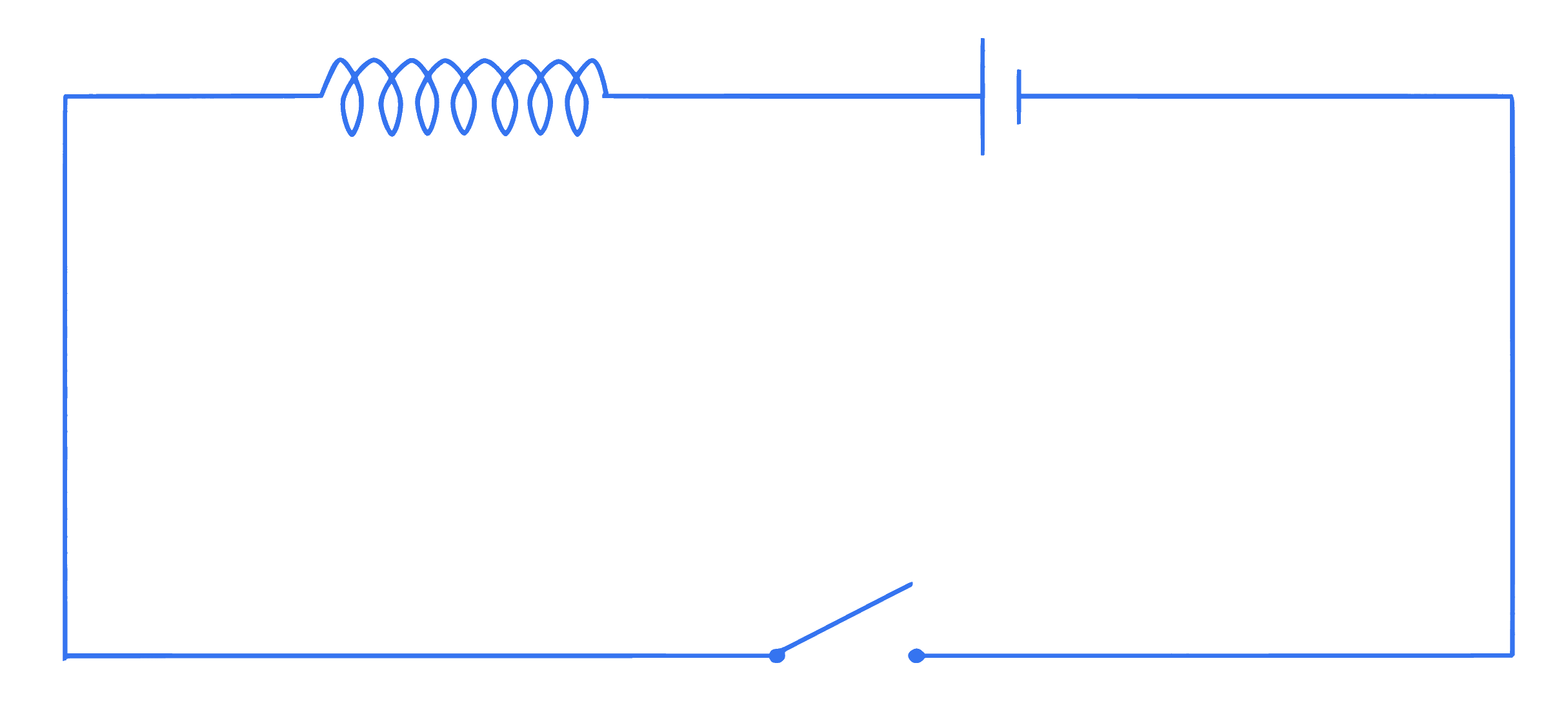

Q9.
mcq single
+4 / 1
A smooth inclined plane ends in a vertical circular loop, as shown in the figure. A small body is released from height $h$ as shown. If the body exerts a force of three times its weight on the plane at the highest point of circle then the height $h=\alpha R$. The value of $\alpha$ is $\_\_\_\_$
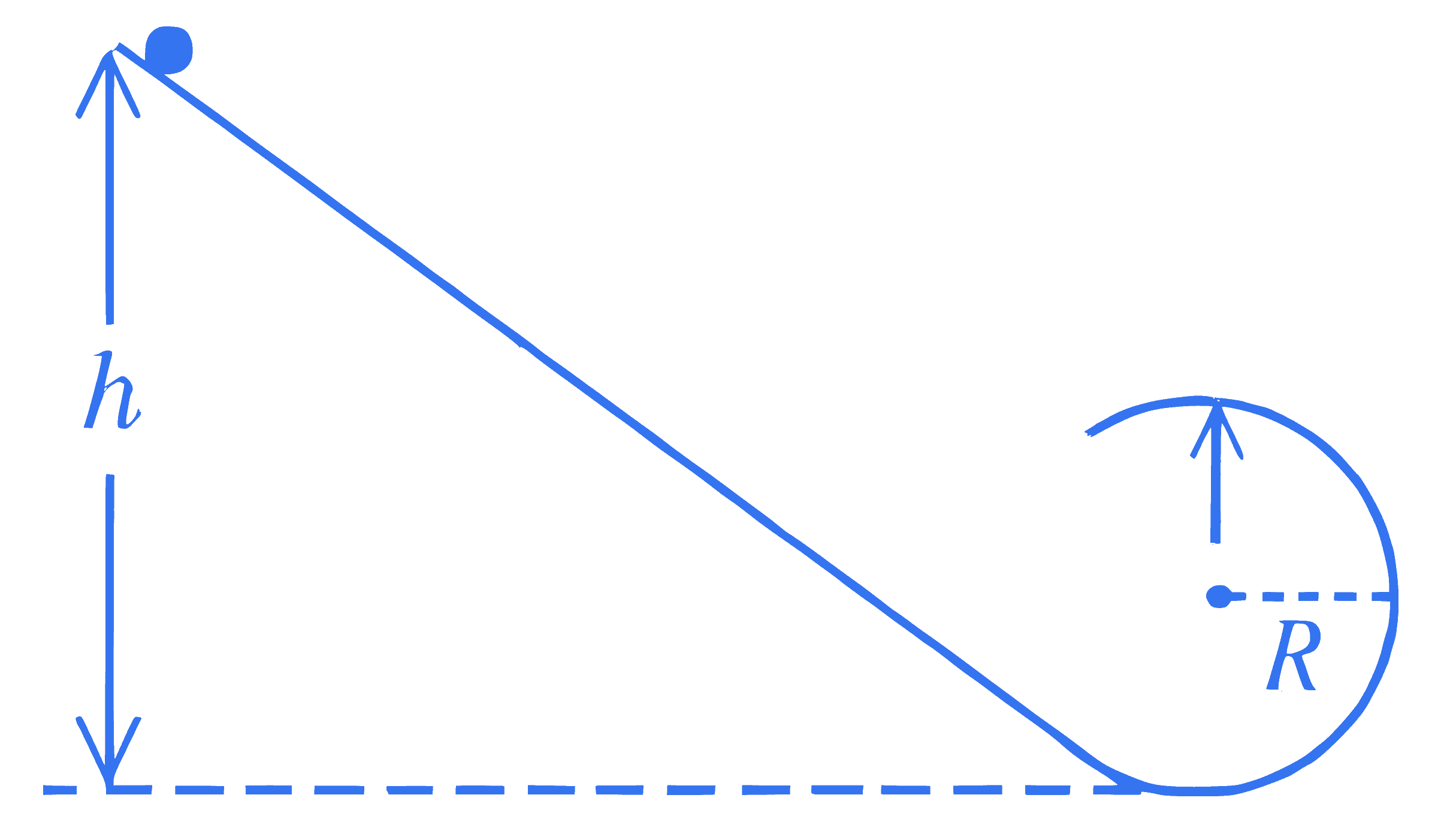

Q10.
mcq single
+4 / 1
Two closed vessels of same volume are joined through a narrow tube and both vessels are filled with air of pressure 90 kPa and temperature 400 K . Keeping the temperature of one vessel constant at 400 K the second vessel temperature is raised to 500 K . The final pressure in the vessels is $\_\_\_\_$ kPa .
Q11.
mcq single
+4 / 1
A lift of mass 1600 kg is supported by thick iron wire. If the maximum stress which the wire can withstand is $4 \times 10^8 \mathrm{~N} / \mathrm{m}^2$ and its radius is 4 mm , then maximum acceleration the lift can take is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}^2$.
(take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ and $\pi=3.14$ )
Q12.
mcq single
+4 / 1
The two wires $A$ and $B$ of equal cross-section but of different materials are joined together. The ratio of Young's modulus of wire $A$ and wire $B$ is 20/11. When the joined wire is kept under certain tension the elongations in the wires $A$ and $B$ are equal. If the length of wire $A$ is 2.2 m , then the length of wire $B$ is
$\_\_\_\_$ m.
Q13.
numerical
+4 / 1
A certain gas is isothermally compressed to $\left(\frac{1}{3}\right)^{\mathrm{rd}}$ of its initial volume $\left(V_{\mathrm{o}}=3\right.$ litre) by applying required pressure. If the bulk modulus of the gas is $3 \times 10^5 \mathrm{~N} / \mathrm{m}^2$, the magnitude of work done on the gas is $\_\_\_\_$ J.
Q14.
mcq single
+4 / 1
A thin half ring of radius 35 cm is uniformly charged with a total charge of $Q$ coulomb. If the magnitude of the electric field at centre of the half ring is $100 \mathrm{~V} / \mathrm{m}$, then the value of $Q$ is $\_\_\_\_$ nC .
$$ \left(\epsilon_0=8.85 \times 10^{-12} \mathrm{C}^2 / \mathrm{Nm}^2 \text { and } \pi=3.14\right) $$
Q15.
numerical
+4 / 1
A three coulomb charge moves from the point $(0,-2,-5)$ to the point $(5,1,2)$ in an electric field expressed as $\vec{E}=2 x \hat{\mathrm{i}}+3 \mathrm{y}^2 \hat{\mathrm{j}}+4 \hat{\mathrm{k}} \mathrm{N} / \mathrm{C}$. The work done in moving the charge is $\_\_\_\_$ J.
Q16.
mcq single
+4 / 1
A spherical interface lens of radius $R$ separates two media of refractive indices 1 and 1.4 respectively as shown in the figure below. A point source is placed at a distance of $4 R$ in front of spherical interface. The magnitude of the magnification of point source image is $\_\_\_\_$ .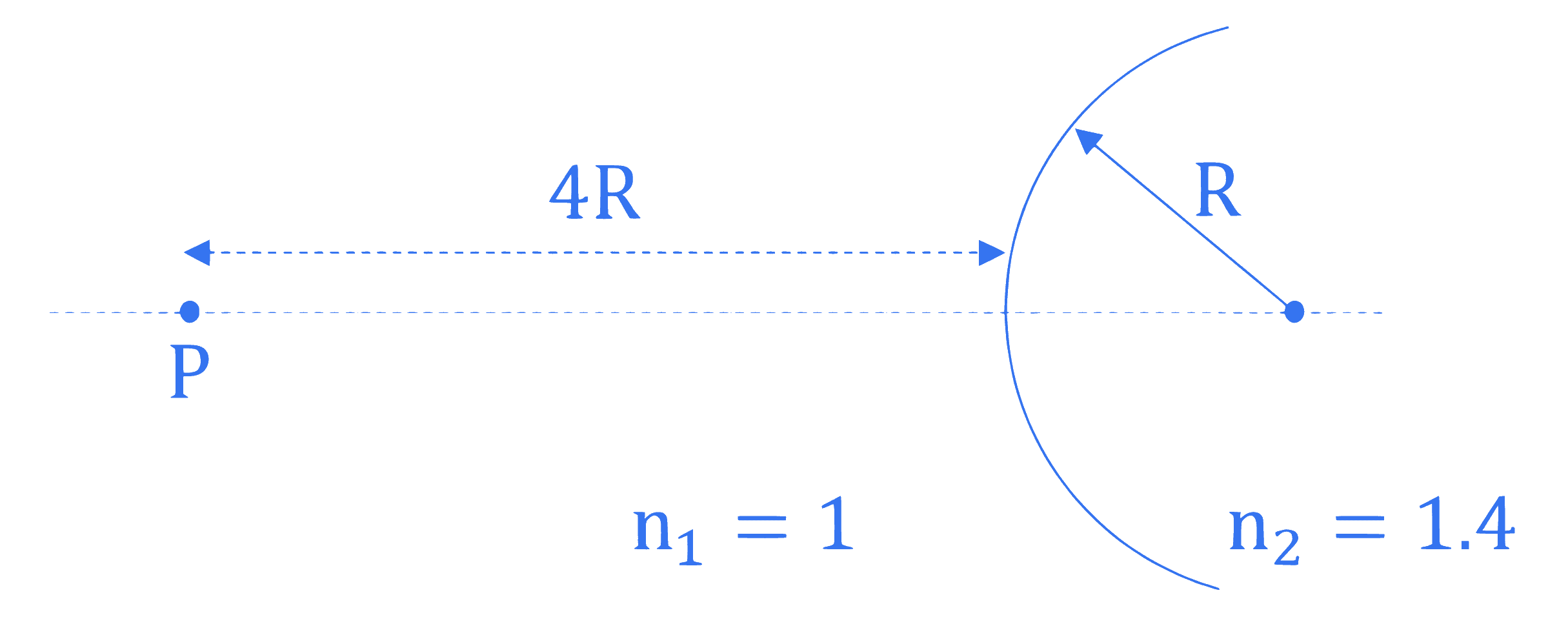

Q17.
mcq single
+4 / 1
A small cube of side 1 mm is placed at the centre of a circular loop of radius 10 cm carrying a current of 2 A . The magnetic energy stored inside the cube is $\alpha \times 10^{-14} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
$$ \left(\mu_{\mathrm{o}}=4 \pi \times 10^{-7} \mathrm{Tm} / \mathrm{A}, \pi=3.14\right) $$
Q18.
mcq single
+4 / 1
The density $\rho$ of a uniform cylinder is determined by measuring its mass $m$, length $l$ and diameter $d$. The measured values of $m, l$ and $d$ are $97.42 \pm 0.02 \mathrm{~g}$, $8.35 \pm 0.05 \mathrm{~mm}$ and $20.20 \pm 0.02 \mathrm{~mm}$, respectively. Calculated percentage fractional error in $\rho$ is $\_\_\_\_$ .
Q19.
mcq single
+4 / 1
The potential energy of a particle changes with distance $x$ from a fixed origin as $V=\frac{A \sqrt{x}}{x+B}$, where $A$ and $B$ are constant with appropriate dimensions. The dimensions of $A B$ are $\_\_\_\_$
Q20.
numerical
+4 / 1
The energy released when $\frac{7}{17.13} \mathrm{~kg}$ of ${ }_3^7 \mathrm{Li}$ is converted into ${ }_2^4 \mathrm{He}$ by proton bombardment is $\alpha \times 10^{32} \mathrm{eV}$. The value of $\alpha$ is $\_\_\_\_$ . (Nearest integer) (Mass of ${ }_3^7 \mathrm{Li}=7.0183 \mathrm{u}$, mass of ${ }_2^4 \mathrm{He}=4.004 \mathrm{u}$, mass of proton $=1.008 \mathrm{u}$ and $1 \mathrm{u}=931 \mathrm{MeV} / \mathrm{c}^2$ and Avogadro number $=6.0 \times 10^{23}$ )
Q21.
mcq single
+4 / 1
The ratio of momentum of the photons of the $1^{\text {st }}$ and $2^{\text {nd }}$ line of Balmer series of Hydrogen atoms is $\alpha / \beta$. The possible values of $\alpha$ and $\beta$ are:-
Q22.
mcq single
+4 / 1
Two blocks ( $P$ and $Q$ ) with respectively masses 2 kg and 1.5 kg are joined by a massless thread. These blocks are mounted on a frictionless pully which is fixed on the edge of a cube $(S)$, as shown in the figure below. Block $P$ is positioned on the top surface which has no friction and block $Q$ is in contact with side-surface, having coefficient friction $\mu$. The cube ( $S$ ) moves towards the right with acceleration of $\frac{g}{2}$, where $g$ is gravitational acceleration. During this movement the block $P$ and $Q$ remain stationary. The value of $\mu$ is $\_\_\_\_$ (take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )


Q23.
numerical
+4 / 1
Refer to the circuit diagram given below. The heat generated across the $6 \Omega$ resistance in 100 second is $\frac{\alpha}{100} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ . (Nearest integer)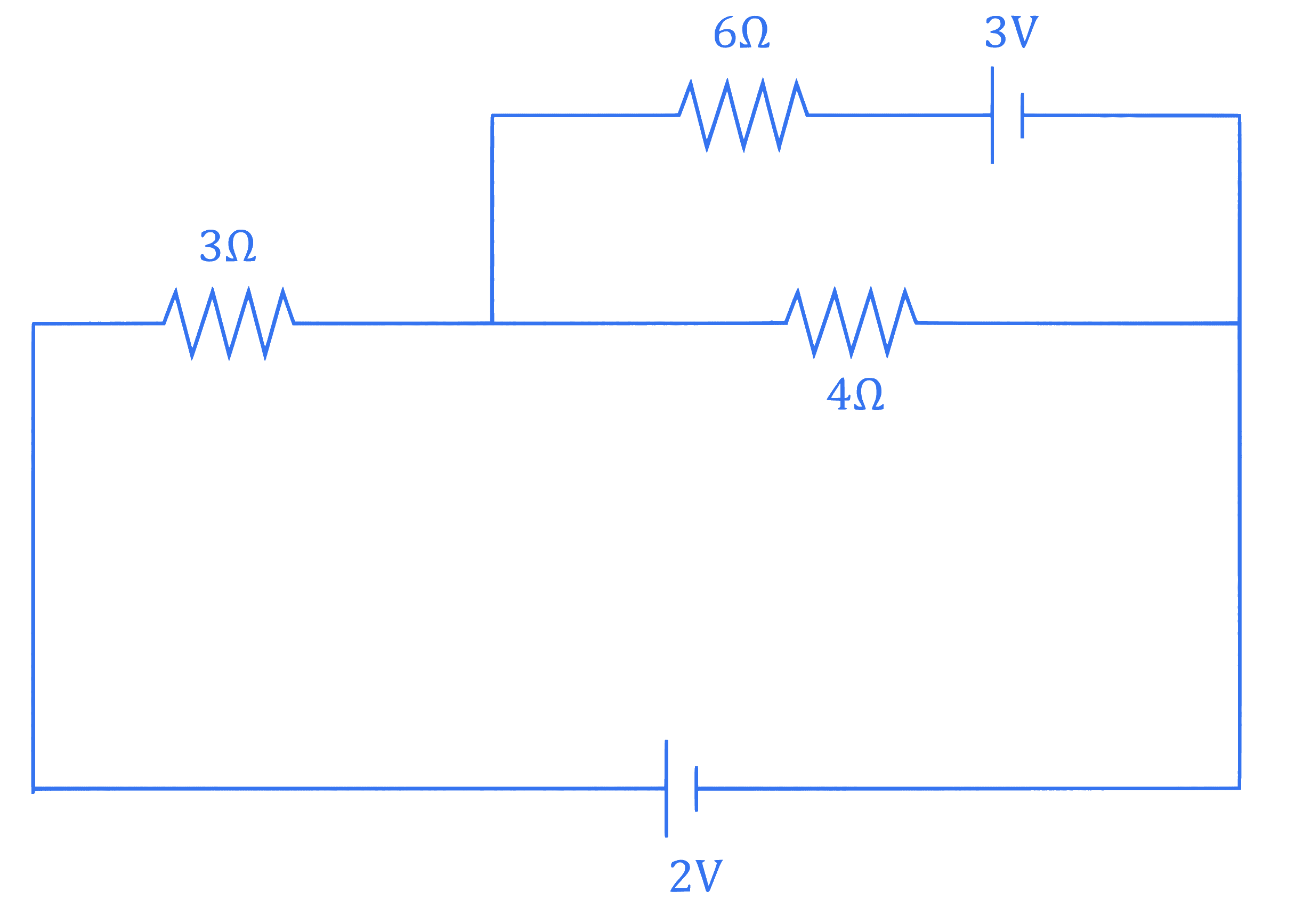

Q24.
mcq single
+4 / 1
A point light source emits E.M. waves in free space. A detector, placed at a distance of $L \mathrm{~m}$, measures the intensity as $I_{\mathrm{o}}$. The detector is now shifted to another location on the same spherical surface ensuring the angle between original location and new location as $45^{\circ}$. The measured intensity at new location will be $\_\_\_\_$ .
Q25.
mcq single
+4 / 1
A LCR series circuit driven with $E_{r m s}=90 \mathrm{~V}$ at frequency $f_{\mathrm{d}}=30 \mathrm{~Hz}$ has resistance $R=80 \Omega$, an inductance with inductive reactance $X_L=20.0 \Omega$ and capacitance with capacitive reactance $X_C=80.0 \Omega$. The power factor of the circuit is $\_\_\_\_$ .
Q26.
mcq single
+4 / 1
The rain drop of mass 1 g , starts with zero velocity from a height of 1 km . It hits the ground with a speed of $5 \mathrm{~m} / \mathrm{s}$. The work done by the unknown resistive force is $\_\_\_\_$ J.
(take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q27.
mcq single
+4 / 1
The maximum rated power of the LED is 2 mW and it is used in the circuit with input voltage of 5 V as shown in the figure below. The current through resistance $R_S$ is 0.5 mA .
The minimum value of the resistance of $R_S$, to ensure that the LED is not damaged is $\_\_\_\_$ $\mathrm{k} \Omega$.

Q28.
numerical
+4 / 1
An unpolarized light of intensity $I_0$ passes through polarizer and then through a certain optically active solution and finally it goes to analyser. If the angle between analyser and polariser is $0^{\circ}$ and intensity of light emerged from analyser is $\frac{3}{8} I_0$, the angle of rotation of the light by the solution with respect to analyser is $\_\_\_\_$ degrees.
Q29.
mcq single
+4 / 1
In interference experiment the path difference between two interfering waves at a point $A$ on the screen is $\lambda / 3$, where $\lambda$ is the wavelength of these waves, and at another point $B$ the path difference is $\lambda / 6$. The ratio of intensities at points $A$ and $B$ is $\_\_\_\_$ .
Q30.
mcq single
+4 / 1
A particle is executing simple harmonic motion. Its amplitude is $A$ and time period is 5 sec . The time required by it to move from $x=A$ to $x=\frac{A}{\sqrt{2}}$ is $\_\_\_\_$ sec.
Q31.
mcq single
+4 / 1
A solid sphere of radius 4 cm and mass 5 kg is rotating (rotation axis is passing through the centre of the sphere) with an angular velocity of 1200 rpm . It is brought to rest in 10 s by applying a constant torque. The torque applied and the number of rotations it made before it comes to rest are $\_\_\_\_$ and $\_\_\_\_$ respectively.
Q32.
mcq single
+4 / 1
The position of center of mass of three masses $2 \mathrm{~kg}, 3 \mathrm{~kg}$ and 15 kg placed with respect to mid point $(p)$ of normal bisector, as shown in the figure is $\_\_\_\_$ .

Q33.
mcq single
+4 / 1
An inductor of inductance 10 mH having resistance of $100 \Omega$ is connected to battery of E.M.F. 1.0 V through a switch as shown in the figure below. After switch is closed, the ratio of instantaneous voltages across the inductor when the current passing through it is 2 mA and 4 mA is $\_\_\_\_$ .


Q34.
mcq single
+4 / 1
A smooth inclined plane ends in a vertical circular loop, as shown in the figure. A small body is released from height $h$ as shown. If the body exerts a force of three times its weight on the plane at the highest point of circle then the height $h=\alpha R$. The value of $\alpha$ is $\_\_\_\_$


Q35.
mcq single
+4 / 1
Two closed vessels of same volume are joined through a narrow tube and both vessels are filled with air of pressure 90 kPa and temperature 400 K . Keeping the temperature of one vessel constant at 400 K the second vessel temperature is raised to 500 K . The final pressure in the vessels is $\_\_\_\_$ kPa .
Q36.
mcq single
+4 / 1
A lift of mass 1600 kg is supported by thick iron wire. If the maximum stress which the wire can withstand is $4 \times 10^8 \mathrm{~N} / \mathrm{m}^2$ and its radius is 4 mm , then maximum acceleration the lift can take is $\_\_\_\_$ $\mathrm{m} / \mathrm{s}^2$.
(take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ and $\pi=3.14$ )
Q37.
mcq single
+4 / 1
The two wires $A$ and $B$ of equal cross-section but of different materials are joined together. The ratio of Young's modulus of wire $A$ and wire $B$ is 20/11. When the joined wire is kept under certain tension the elongations in the wires $A$ and $B$ are equal. If the length of wire $A$ is 2.2 m , then the length of wire $B$ is
$\_\_\_\_$ m.
Q38.
numerical
+4 / 1
A certain gas is isothermally compressed to $\left(\frac{1}{3}\right)^{\mathrm{rd}}$ of its initial volume $\left(V_{\mathrm{o}}=3\right.$ litre) by applying required pressure. If the bulk modulus of the gas is $3 \times 10^5 \mathrm{~N} / \mathrm{m}^2$, the magnitude of work done on the gas is $\_\_\_\_$ J.
Q39.
mcq single
+4 / 1
A thin half ring of radius 35 cm is uniformly charged with a total charge of $Q$ coulomb. If the magnitude of the electric field at centre of the half ring is $100 \mathrm{~V} / \mathrm{m}$, then the value of $Q$ is $\_\_\_\_$ nC .
$$ \left(\epsilon_0=8.85 \times 10^{-12} \mathrm{C}^2 / \mathrm{Nm}^2 \text { and } \pi=3.14\right) $$
Q40.
numerical
+4 / 1
A three coulomb charge moves from the point $(0,-2,-5)$ to the point $(5,1,2)$ in an electric field expressed as $\vec{E}=2 x \hat{\mathrm{i}}+3 \mathrm{y}^2 \hat{\mathrm{j}}+4 \hat{\mathrm{k}} \mathrm{N} / \mathrm{C}$. The work done in moving the charge is $\_\_\_\_$ J.
Q41.
mcq single
+4 / 1
A spherical interface lens of radius $R$ separates two media of refractive indices 1 and 1.4 respectively as shown in the figure below. A point source is placed at a distance of $4 R$ in front of spherical interface. The magnitude of the magnification of point source image is $\_\_\_\_$ .

Q42.
mcq single
+4 / 1
A small cube of side 1 mm is placed at the centre of a circular loop of radius 10 cm carrying a current of 2 A . The magnetic energy stored inside the cube is $\alpha \times 10^{-14} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ .
$$ \left(\mu_{\mathrm{o}}=4 \pi \times 10^{-7} \mathrm{Tm} / \mathrm{A}, \pi=3.14\right) $$
Q43.
mcq single
+4 / 1
The density $\rho$ of a uniform cylinder is determined by measuring its mass $m$, length $l$ and diameter $d$. The measured values of $m, l$ and $d$ are $97.42 \pm 0.02 \mathrm{~g}$, $8.35 \pm 0.05 \mathrm{~mm}$ and $20.20 \pm 0.02 \mathrm{~mm}$, respectively. Calculated percentage fractional error in $\rho$ is $\_\_\_\_$ .
Q44.
mcq single
+4 / 1
The potential energy of a particle changes with distance $x$ from a fixed origin as $V=\frac{A \sqrt{x}}{x+B}$, where $A$ and $B$ are constant with appropriate dimensions. The dimensions of $A B$ are $\_\_\_\_$
Q45.
numerical
+4 / 1
The energy released when $\frac{7}{17.13} \mathrm{~kg}$ of ${ }_3^7 \mathrm{Li}$ is converted into ${ }_2^4 \mathrm{He}$ by proton bombardment is $\alpha \times 10^{32} \mathrm{eV}$. The value of $\alpha$ is $\_\_\_\_$ . (Nearest integer) (Mass of ${ }_3^7 \mathrm{Li}=7.0183 \mathrm{u}$, mass of ${ }_2^4 \mathrm{He}=4.004 \mathrm{u}$, mass of proton $=1.008 \mathrm{u}$ and $1 \mathrm{u}=931 \mathrm{MeV} / \mathrm{c}^2$ and Avogadro number $=6.0 \times 10^{23}$ )
Q46.
mcq single
+4 / 1
The ratio of momentum of the photons of the $1^{\text {st }}$ and $2^{\text {nd }}$ line of Balmer series of Hydrogen atoms is $\alpha / \beta$. The possible values of $\alpha$ and $\beta$ are:-
Q47.
mcq single
+4 / 1
Two blocks ( $P$ and $Q$ ) with respectively masses 2 kg and 1.5 kg are joined by a massless thread. These blocks are mounted on a frictionless pully which is fixed on the edge of a cube $(S)$, as shown in the figure below. Block $P$ is positioned on the top surface which has no friction and block $Q$ is in contact with side-surface, having coefficient friction $\mu$. The cube ( $S$ ) moves towards the right with acceleration of $\frac{g}{2}$, where $g$ is gravitational acceleration. During this movement the block $P$ and $Q$ remain stationary. The value of $\mu$ is $\_\_\_\_$ (take $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )


Q48.
numerical
+4 / 1
Refer to the circuit diagram given below. The heat generated across the $6 \Omega$ resistance in 100 second is $\frac{\alpha}{100} \mathrm{~J}$. The value of $\alpha$ is $\_\_\_\_$ . (Nearest integer)

Q49.
mcq single
+4 / 1
A point light source emits E.M. waves in free space. A detector, placed at a distance of $L \mathrm{~m}$, measures the intensity as $I_{\mathrm{o}}$. The detector is now shifted to another location on the same spherical surface ensuring the angle between original location and new location as $45^{\circ}$. The measured intensity at new location will be $\_\_\_\_$ .
Q50.
mcq single
+4 / 1
A LCR series circuit driven with $E_{r m s}=90 \mathrm{~V}$ at frequency $f_{\mathrm{d}}=30 \mathrm{~Hz}$ has resistance $R=80 \Omega$, an inductance with inductive reactance $X_L=20.0 \Omega$ and capacitance with capacitive reactance $X_C=80.0 \Omega$. The power factor of the circuit is $\_\_\_\_$ .