JEE Main 2026 (Online) 8th April Evening Shift
JEE 2026 Previous Year
3 hDuration
296Total Marks
148Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 148 across 3 section(s); maximum marks: 296.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Which statements are True?
A. In Hoffmann bromamide degradation, 4 moles of NaOH and 2 moles of $\mathrm{Br}_2$ are consumed per mole of an amide
B. Hoffmann bromamide reaction is not given by alkyl amides.
C. Primary amines can be synthesized by Hoffmann bromamide degradation.
D. Secondary amide on reaction with $\mathrm{Br}_2$ and NaOH will give secondary amine.
E. The by-products of Hoffmann degradation are $\mathrm{Na}_2 \mathrm{CO}_3, \mathrm{NaBr}$ and $\mathrm{H}_2 \mathrm{O}$.
Choose the correct answer from the options given below :
Q2.
mcq single
+4 / 1
Which of the following amino acid will give violet coloured complex with neutral ferric chloride solution?
Q3.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of pairs among $\left[\mathrm{Ti}^{4+}, \mathrm{V}^{2+}\right],\left[\mathrm{V}^{2+}, \mathrm{Mn}^{2+}\right],\left[\mathrm{Mn}^{2+}, \mathrm{Fe}^{3+}\right]$ and $\left[\mathrm{V}^{2+}, \mathrm{Cr}^{2+}\right]$ in which both ions are coloured is 3 .
**Statement II :** The number of pairs among $\left[\mathrm{La}^{3+}, \mathrm{Yb}^{2+}\right],\left[\mathrm{Lu}^{3+}, \mathrm{Ce}^{4+}\right]$ and $\left[\mathrm{Ac}^{3+}, \mathrm{Lr}^{3+}\right]$ ions in which both are diamagnetic is 3 .
In the light of the above statements, choose the correct from the options given below :
Q4.
mcq single
+4 / 1
Given below are two statements for catalytic properties of transition metals.
**Statement I :** First row transition metals which act as catalyst utilise their 3d electrons only for formation of bonds between reactant molecules and atoms on the surface of catalyst.
**Statement II :** There is increase in the concentration of reactants on the surface of catalyst which strengthens the bonds in reacting molecules.
In the light of the above statements, choose the correct answer from the options given below :
Q5.
mcq single
+4 / 1
Given below are two statements :
$$ \mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1} \text { and } 1 \mathrm{cal}=4.2 \mathrm{~J} $$
**Statement I :** When $\mathrm{Ea}=12.6 \mathrm{kcal} / \mathrm{mol}$, the room temperature rate constant is doubled by a $10^{\circ} \mathrm{C}$ increase in temperature ( 298 K to 308 K )
**Statement II :** For a first order reactions $\mathrm{A} \rightarrow \mathrm{B}$,
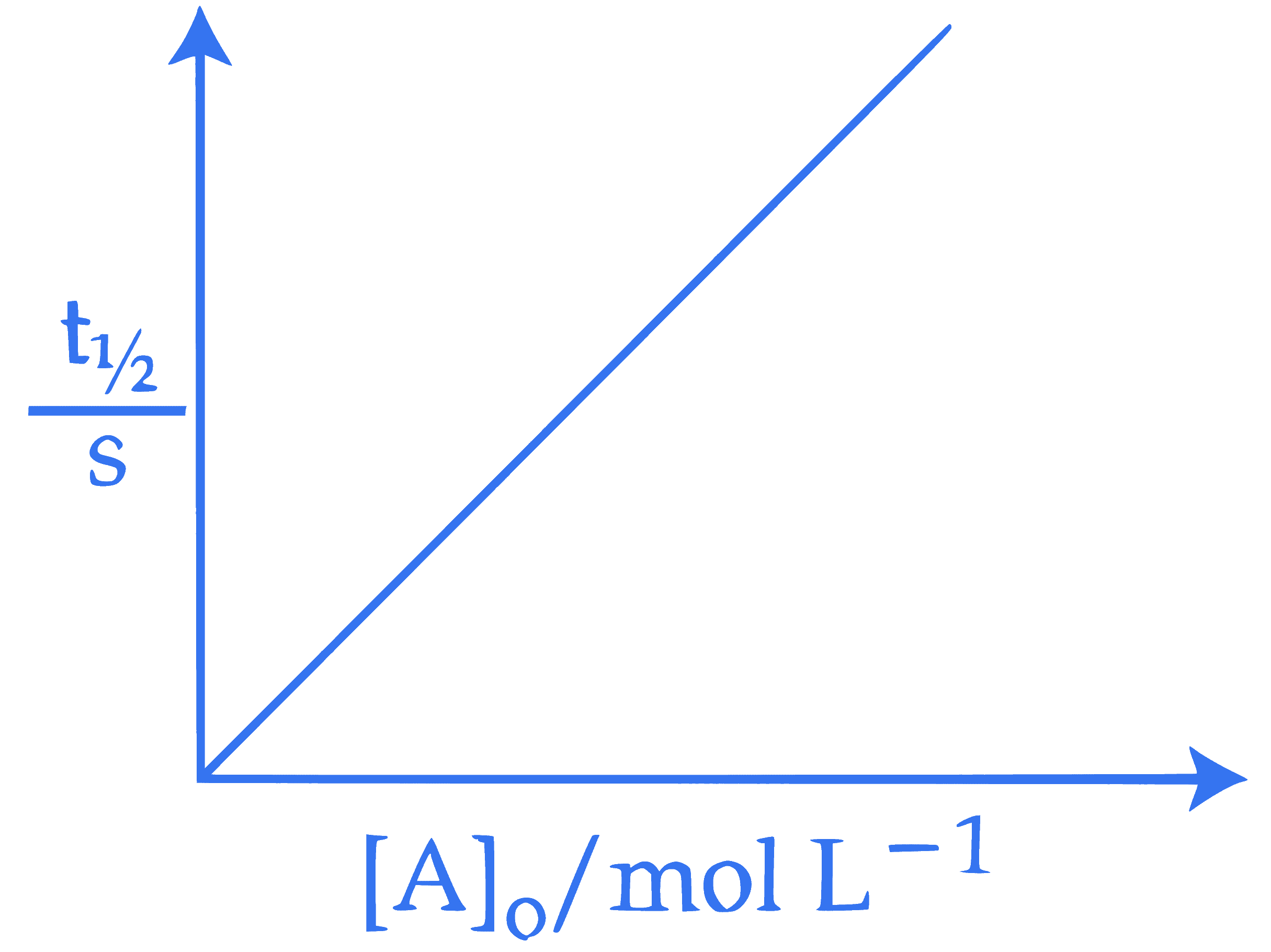 Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
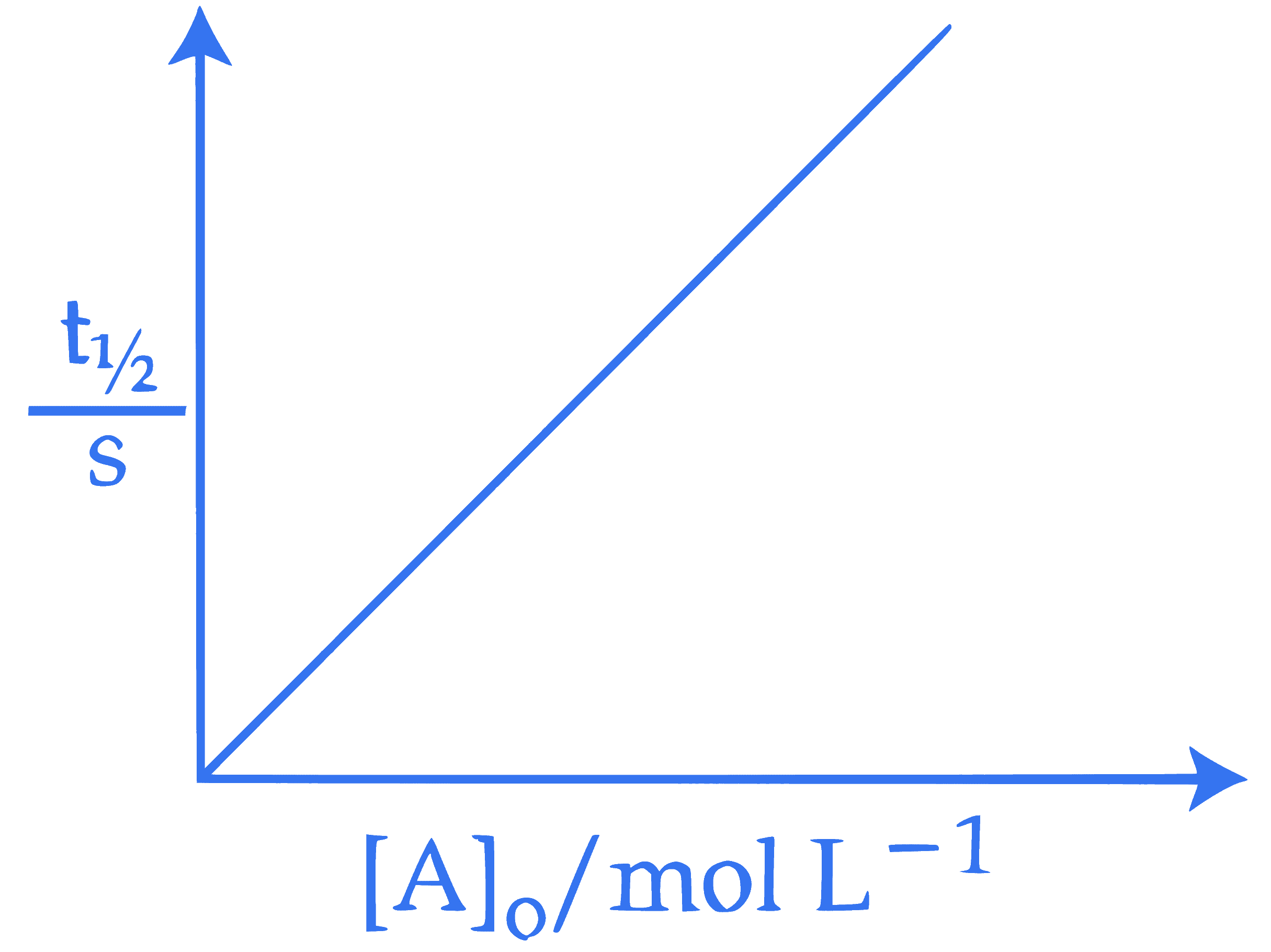 Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
Q6.
mcq single
+4 / 1
Bromine trifluoride autoionizes to form $\mathrm{BrF}_2^{\oplus}$ and $\mathrm{BrF}_4^{\ominus}$. The shapes of the cation and anion are respectively $\_\_\_\_$ and $\_\_\_\_$ .
Q7.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Due to increase in van der Waals forces, the order of boiling points is $\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{I}>\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{I}>\mathrm{CH}_3 \mathrm{I}$.
$$ \text { Statement II : } $$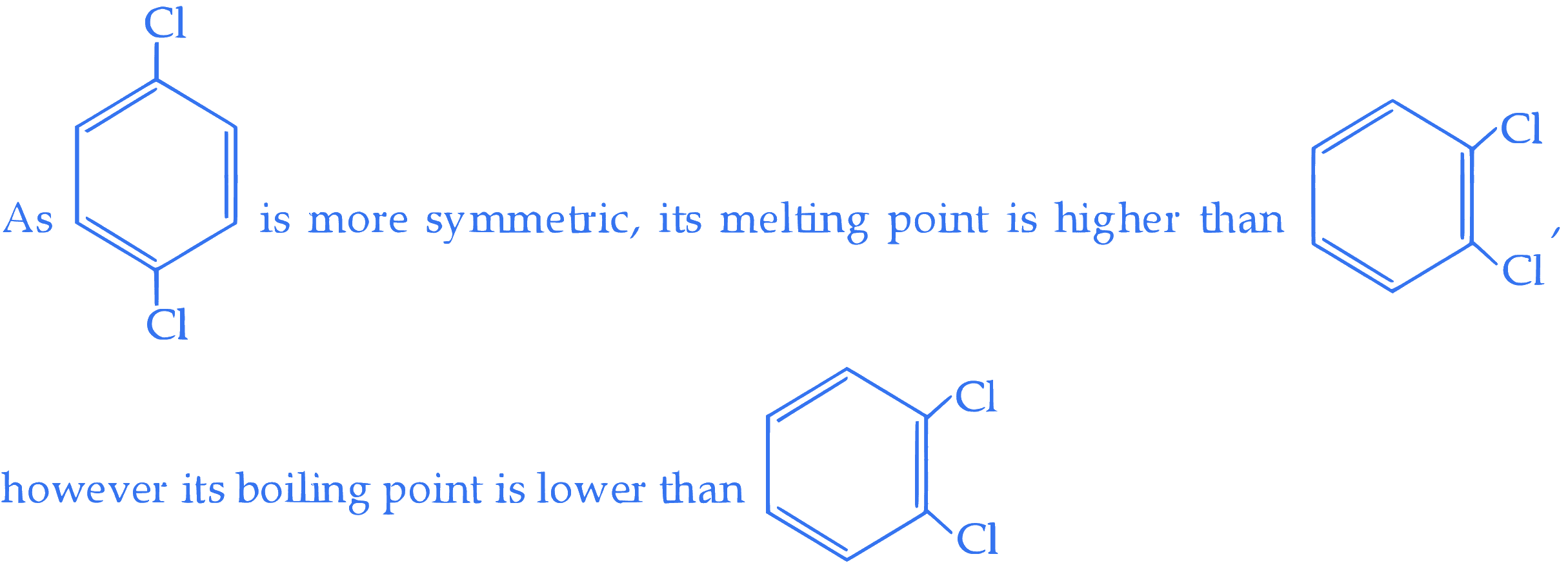 In the light of the above statements, choose the correct answer from the options given below :
In the light of the above statements, choose the correct answer from the options given below :
 In the light of the above statements, choose the correct answer from the options given below :
In the light of the above statements, choose the correct answer from the options given below :
Q8.
mcq single
+4 / 1
Given below are two statements :
Given : Molar mass of $\mathrm{C}, \mathrm{H}, \mathrm{O}, \mathrm{Cl}$ are $12,1,16$ and $35.5 \mathrm{~g} \mathrm{~mol}^{-1}$, respectively
**Statement I :** In $30 \%(\mathrm{w} / \mathrm{w})$ solution of methanol in $\mathrm{CCl}_4($ at T K$)$, the mole fraction of $\mathrm{CCl}_4$ is equal to 0.33 .
**Statement II :** Mixture of methanol and $\mathrm{CCl}_4$ shows positive deviation from Raoult's law. In the light of the above statements, choose the correct answer from the options given below :
Q9.
mcq single
+4 / 1
Which of the following statements are not correct?
A. For water, magnitude of $\mathrm{K}_{\mathrm{b}}$ is more than the magnitude of $\mathrm{K}_{\mathrm{f}}$.
B. The elevation in boiling point of water when a non-volatile solute is added to it is larger in magnitude than its depression in freezing point.
C. Osmotic pressure measurement is preferred over any other colligative property to determine molar mass of proteins and polymers.
$$ \text { D. The dimerised form of benzoic acid in benzene is } $$ 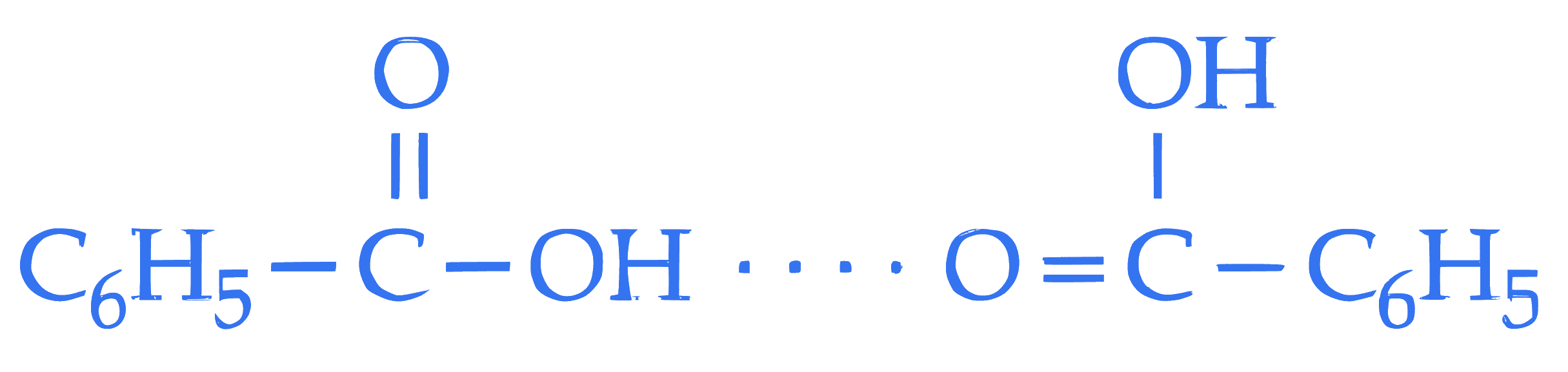 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q10.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Mass of substance
List - II
Number of atoms
A.
1.8 mg water
I.
$$
2 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
B.
9.8 mg sulphuric acid
II.
$$
1.5 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
C.
1.8 mg carbon
III.
$$
3 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
D.
5.85 mg salt $(\mathrm{NaCl})$
IV.
$$
7 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
$$ \text { Choose the correct answer from the options given below : } $$
Q11.
numerical
+4 / 1
Solid carbon, CaO and $\mathrm{CaCO}_3$ are mixed and allowed to attain equilibrium at T K .
$$ \begin{array}{ll} \mathrm{CaCO}_3(\mathrm{~s}) \rightleftharpoons \mathrm{CaO}(\mathrm{~s})+\mathrm{CO}_2(\mathrm{~g}) & \mathrm{Kp}_1=0.08 \mathrm{~atm} \\ \mathrm{C}(\mathrm{~s})+\mathrm{CO}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{CO}(\mathrm{~g}) & \mathrm{Kp}_2=2 \mathrm{~atm} \end{array} $$
The partial pressure of CO is __ $\times 10^{-1} \mathrm{~atm}$
Q12.
mcq single
+4 / 1
Consider the following reactions in which all the reactants and products are present in gaseous state
$$ \begin{aligned} & 2 x y \rightleftharpoons x_2+y_2 \,\,\mathrm{~K}_1=2.5 \times 10^5 \\ & x y+\frac{1}{2} z_2 \rightleftharpoons x y z \,\,\mathrm{~K}_2=5 \times 10^{-3} \end{aligned} $$
The value of $\mathrm{K}_3$ for the equilibrium $\frac{1}{2} x_2+\frac{1}{2} y_2+\frac{1}{2} z_2 \rightleftharpoons x y z$ is :
Q13.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Electronic configuration of neutral atom (where $n=2$ )
List - II
$1^{\text {st }}$ Ionization Energy ( $\mathbf{k J ~ m o l}^{-\mathbf{1}}$ )
A.
$$
\mathrm{ns}^2
$$
I.
2080
B.
$$
\mathrm{ns}^2 \mathrm{np}^1
$$
II.
899
C.
$$
n s^2 n p^3
$$
III.
800
D.
$$
n s^2 n p^6
$$
IV.
1402
Choose the correct answer from the options given below :
Q14.
numerical
+4 / 1
Consider the reaction
$$ 2 \mathrm{H}_2 \mathrm{~S}(\mathrm{~g})+3 \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(\mathrm{l})+2 \mathrm{SO}_2(\mathrm{~g}) $$
The magnitude of enthalpy change for the reaction in $\mathrm{kJ} \mathrm{mol}^{-1}$ is $\_\_\_\_$ . (Nearest integer)
$$ \begin{aligned} Given:\,\,& \Delta_{\mathrm{f}} \mathrm{H}^{\ominus}\left(\mathrm{H}_2 \mathrm{~S}\right)=-20.1 \mathrm{~kJ} \mathrm{~mol}^{-1} \\ & \Delta_{\mathrm{f}} \mathrm{H}^{\ominus}\left(\mathrm{H}_2 \mathrm{O}\right)=-286.0 \mathrm{~kJ} \mathrm{~mol}^{-1} \\ & \Delta_{\mathrm{f}} \mathrm{H}^{\ominus}\left(\mathrm{SO}_2\right)=-297.0 \mathrm{~kJ} \mathrm{~mol}^{-1} \end{aligned} $$
Q15.
mcq single
+4 / 1
Find the correct statements related to group 15 hydrides.
A. Reducing nature increases from $\mathrm{NH}_3$ to $\mathrm{BiH}_3$
B. Tendency to donate lone pair of electrons decreases from $\mathrm{NH}_3$ to $\mathrm{BiH}_3$
C. The stability of hydrides decreases from $\mathrm{NH}_3$ to $\mathrm{BiH}_3$
D. HEH bond angle decreases from $\mathrm{NH}_3$ to $\mathrm{SbH}_3(\mathrm{E}=$ Elements of group 15)
Choose the correct answer from the options given below :
Q16.
numerical
+4 / 1
Number of paramagnetic complexes among the following is $\_\_\_\_$ .
$$ \begin{aligned} & {\left[\mathrm{MnBr}_4\right]^{2-},\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CO})_4\right],\left[\mathrm{CoF}_6\right]^{3-},\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{4-},\left[\mathrm{Mn}(\mathrm{CN})_6\right]^{3-},\left[\mathrm{Ti}(\mathrm{CN})_6\right]^{3-},} \\ & {\left[\mathrm{Cu}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+},\left[\mathrm{Co}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}} \end{aligned} $$
Q17.
numerical
+4 / 1
' $x$ ' is the product which is obtained from benzene by reacting it with carbon monoxide and hydrogen chloride in the presence of cuprous chloride. ' $y$ ' is the major product obtained from the benzene by reacting it with ethanoyl chloride in the presence of anhydrous $\mathrm{AlCl}_3$. Product (major) obtained by heating $x$ and $y$ in the presence of alkali is $z$. Total number of $\pi$ (pi) electrons in $z$ is $\_\_\_\_$ .
Q18.
mcq single
+4 / 1
$$ \text { Consider the following reaction. } $$
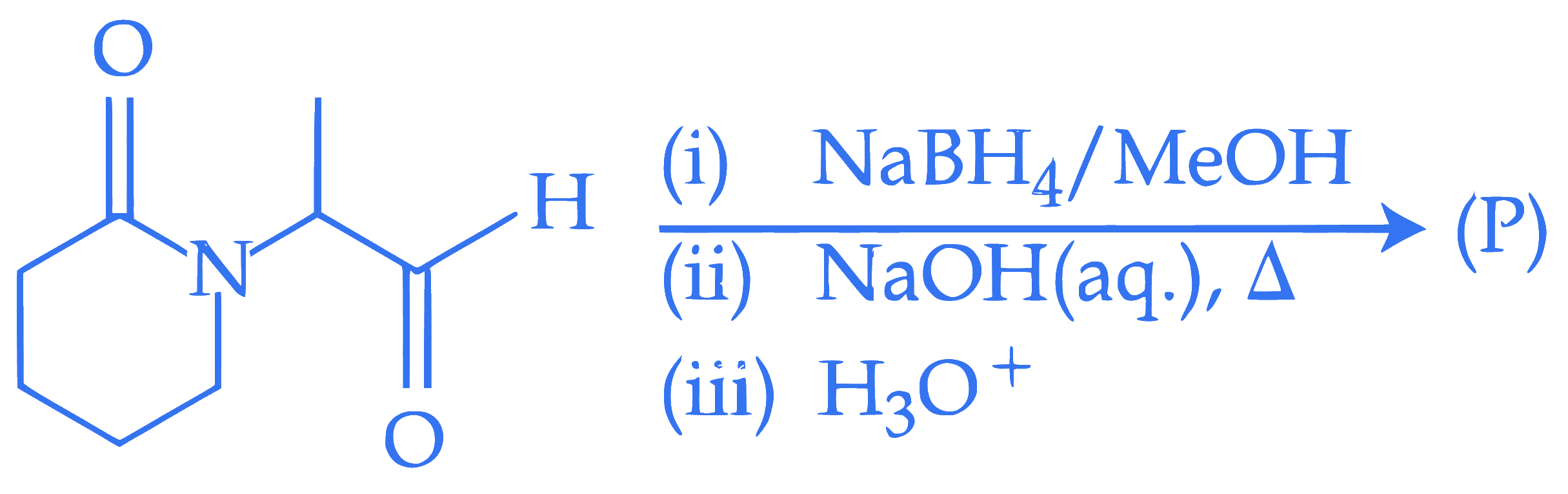 $$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
$$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
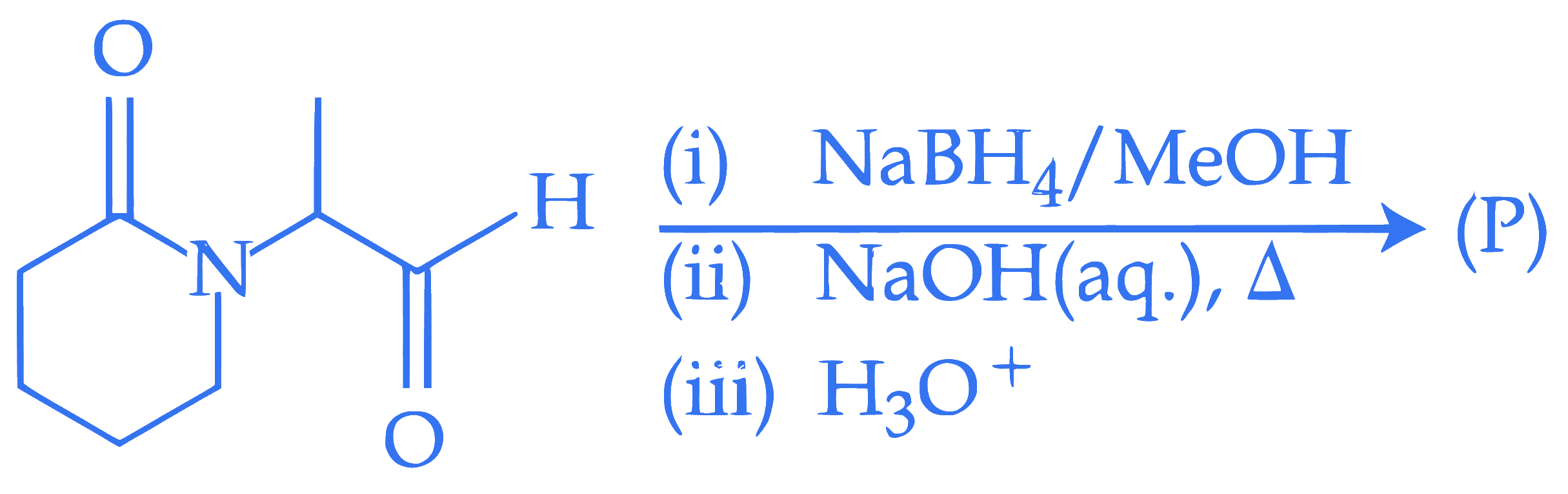 $$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
$$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
Q19.
mcq single
+4 / 1
n-Butane on monochlorination under photochemical condition gives an optically active compound " P ". " P " on further chlorination gives dichloro compounds.
The number of dichloro compounds obtained (ignore stereoisomers) is :
Q20.
mcq single
+4 / 1
$$ \text { The major product of which of the following reaction is not obtained by rearrangement reaction? } $$
Q21.
mcq single
+4 / 1
The incorrect statement from the following with respect to carbohydrates is :
Q22.
mcq single
+4 / 1
$$ \text { The total number of aromatic compounds/species from the following is } $$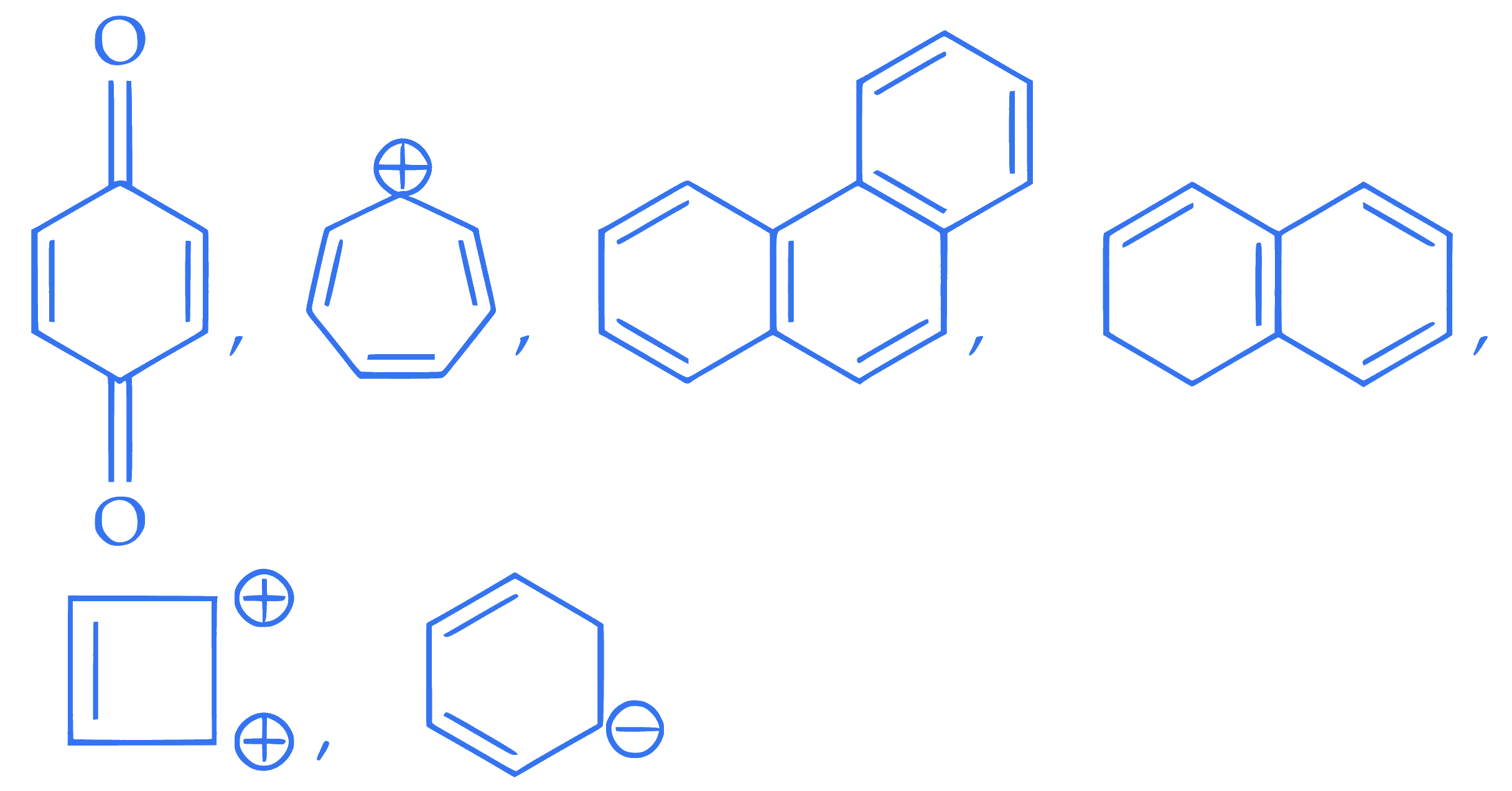

Q23.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Vapours of the liquid with higher boiling point condense before vapours of the liquid with lower boiling points in fractional distillation.
**Statement II :** The vapours rising up in the fractionating column become richer in high boiling component of the mixture.
In the light of the above statements, choose the correct answer from the options given below :
Q24.
mcq single
+4 / 1
Given at 298 K :
$$ \begin{aligned} & \mathrm{E}_{\mathrm{Fe}^{2+} / \mathrm{Fe}}^{\ominus}=\mathrm{X} \text { Volt } \\ & \mathrm{E}_{\mathrm{Fe}^{3+} / \mathrm{Fe}}^{\ominus}=\mathrm{Y} \text { Volt } \end{aligned} $$
The $\mathrm{E}_{\mathrm{Fe}^{3+} / \mathrm{Fe}^{2+}}^{\ominus}$ in Volt at 298 K is given by :
Q25.
numerical
+4 / 1
Consider two radiations of wavelengths
- $\lambda_1=2000\mathop {\rm{A}}\limits^{\rm{o}}$
- $\lambda_2=6000 \mathop {\rm{A}}\limits^{\rm{o}}$
The ratio of the energies of these two radiations $\left(\frac{E_1}{E_2}\right)$ is $\_\_\_\_$ (Nearest integer).
Q26.
mcq single
+4 / 1
Which statements are True?
A. In Hoffmann bromamide degradation, 4 moles of NaOH and 2 moles of $\mathrm{Br}_2$ are consumed per mole of an amide
B. Hoffmann bromamide reaction is not given by alkyl amides.
C. Primary amines can be synthesized by Hoffmann bromamide degradation.
D. Secondary amide on reaction with $\mathrm{Br}_2$ and NaOH will give secondary amine.
E. The by-products of Hoffmann degradation are $\mathrm{Na}_2 \mathrm{CO}_3, \mathrm{NaBr}$ and $\mathrm{H}_2 \mathrm{O}$.
Choose the correct answer from the options given below :
Q27.
mcq single
+4 / 1
Which of the following amino acid will give violet coloured complex with neutral ferric chloride solution?
Q28.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** The number of pairs among $\left[\mathrm{Ti}^{4+}, \mathrm{V}^{2+}\right],\left[\mathrm{V}^{2+}, \mathrm{Mn}^{2+}\right],\left[\mathrm{Mn}^{2+}, \mathrm{Fe}^{3+}\right]$ and $\left[\mathrm{V}^{2+}, \mathrm{Cr}^{2+}\right]$ in which both ions are coloured is 3 .
**Statement II :** The number of pairs among $\left[\mathrm{La}^{3+}, \mathrm{Yb}^{2+}\right],\left[\mathrm{Lu}^{3+}, \mathrm{Ce}^{4+}\right]$ and $\left[\mathrm{Ac}^{3+}, \mathrm{Lr}^{3+}\right]$ ions in which both are diamagnetic is 3 .
In the light of the above statements, choose the correct from the options given below :
Q29.
mcq single
+4 / 1
Given below are two statements for catalytic properties of transition metals.
**Statement I :** First row transition metals which act as catalyst utilise their 3d electrons only for formation of bonds between reactant molecules and atoms on the surface of catalyst.
**Statement II :** There is increase in the concentration of reactants on the surface of catalyst which strengthens the bonds in reacting molecules.
In the light of the above statements, choose the correct answer from the options given below :
Q30.
mcq single
+4 / 1
Given below are two statements :
$$ \mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1} \text { and } 1 \mathrm{cal}=4.2 \mathrm{~J} $$
**Statement I :** When $\mathrm{Ea}=12.6 \mathrm{kcal} / \mathrm{mol}$, the room temperature rate constant is doubled by a $10^{\circ} \mathrm{C}$ increase in temperature ( 298 K to 308 K )
**Statement II :** For a first order reactions $\mathrm{A} \rightarrow \mathrm{B}$,
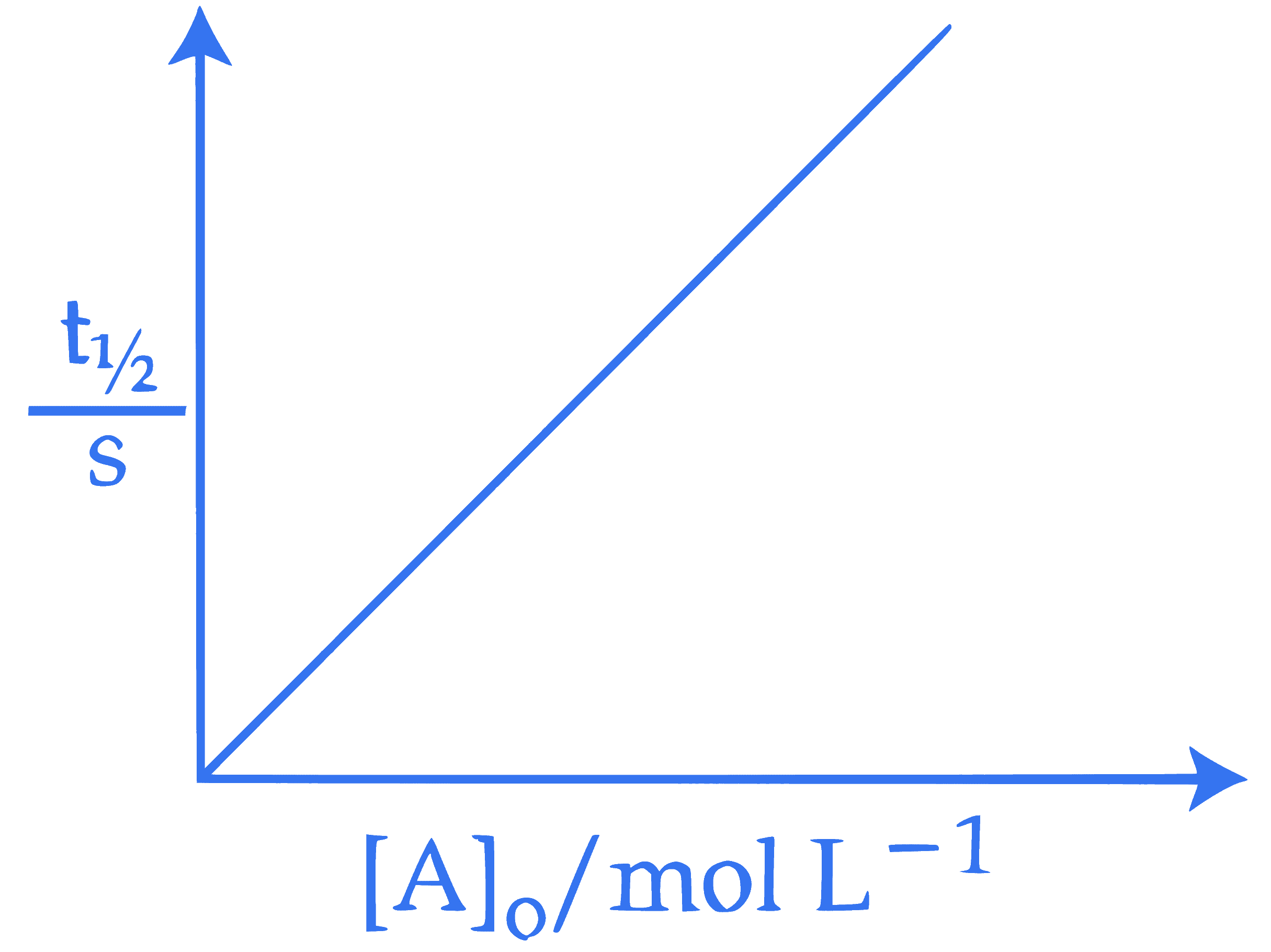 Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
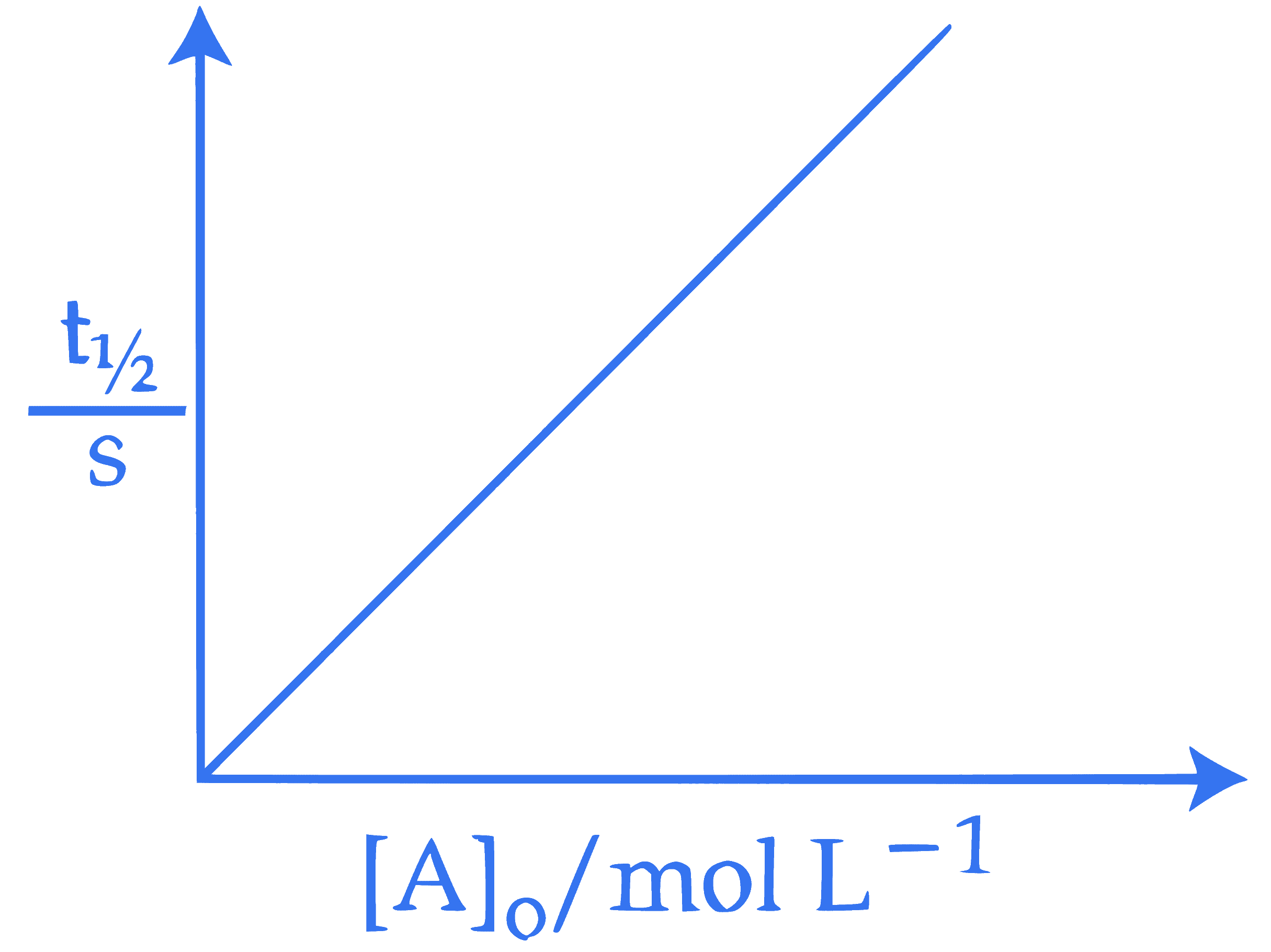 Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
Here $[A]_0$ is the initial concentration of $A$ and $t_{1 / 2}$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
Q31.
mcq single
+4 / 1
Bromine trifluoride autoionizes to form $\mathrm{BrF}_2^{\oplus}$ and $\mathrm{BrF}_4^{\ominus}$. The shapes of the cation and anion are respectively $\_\_\_\_$ and $\_\_\_\_$ .
Q32.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Due to increase in van der Waals forces, the order of boiling points is $\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{I}>\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{I}>\mathrm{CH}_3 \mathrm{I}$.
$$ \text { Statement II : } $$ In the light of the above statements, choose the correct answer from the options given below :
In the light of the above statements, choose the correct answer from the options given below :
 In the light of the above statements, choose the correct answer from the options given below :
In the light of the above statements, choose the correct answer from the options given below :
Q33.
mcq single
+4 / 1
Given below are two statements :
Given : Molar mass of $\mathrm{C}, \mathrm{H}, \mathrm{O}, \mathrm{Cl}$ are $12,1,16$ and $35.5 \mathrm{~g} \mathrm{~mol}^{-1}$, respectively
**Statement I :** In $30 \%(\mathrm{w} / \mathrm{w})$ solution of methanol in $\mathrm{CCl}_4($ at T K$)$, the mole fraction of $\mathrm{CCl}_4$ is equal to 0.33 .
**Statement II :** Mixture of methanol and $\mathrm{CCl}_4$ shows positive deviation from Raoult's law. In the light of the above statements, choose the correct answer from the options given below :
Q34.
mcq single
+4 / 1
Which of the following statements are not correct?
A. For water, magnitude of $\mathrm{K}_{\mathrm{b}}$ is more than the magnitude of $\mathrm{K}_{\mathrm{f}}$.
B. The elevation in boiling point of water when a non-volatile solute is added to it is larger in magnitude than its depression in freezing point.
C. Osmotic pressure measurement is preferred over any other colligative property to determine molar mass of proteins and polymers.
$$ \text { D. The dimerised form of benzoic acid in benzene is } $$  Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q35.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Mass of substance
List - II
Number of atoms
A.
1.8 mg water
I.
$$
2 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
B.
9.8 mg sulphuric acid
II.
$$
1.5 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
C.
1.8 mg carbon
III.
$$
3 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
D.
5.85 mg salt $(\mathrm{NaCl})$
IV.
$$
7 \times 10^{-4} \times \mathrm{N}_{\mathrm{A}}
$$
$$ \text { Choose the correct answer from the options given below : } $$
Q36.
numerical
+4 / 1
Solid carbon, CaO and $\mathrm{CaCO}_3$ are mixed and allowed to attain equilibrium at T K .
$$ \begin{array}{ll} \mathrm{CaCO}_3(\mathrm{~s}) \rightleftharpoons \mathrm{CaO}(\mathrm{~s})+\mathrm{CO}_2(\mathrm{~g}) & \mathrm{Kp}_1=0.08 \mathrm{~atm} \\ \mathrm{C}(\mathrm{~s})+\mathrm{CO}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{CO}(\mathrm{~g}) & \mathrm{Kp}_2=2 \mathrm{~atm} \end{array} $$
The partial pressure of CO is __ $\times 10^{-1} \mathrm{~atm}$
Q37.
mcq single
+4 / 1
Consider the following reactions in which all the reactants and products are present in gaseous state
$$ \begin{aligned} & 2 x y \rightleftharpoons x_2+y_2 \,\,\mathrm{~K}_1=2.5 \times 10^5 \\ & x y+\frac{1}{2} z_2 \rightleftharpoons x y z \,\,\mathrm{~K}_2=5 \times 10^{-3} \end{aligned} $$
The value of $\mathrm{K}_3$ for the equilibrium $\frac{1}{2} x_2+\frac{1}{2} y_2+\frac{1}{2} z_2 \rightleftharpoons x y z$ is :
Q38.
mcq single
+4 / 1
$$ \text { Match List - I with List - II. } $$
List - I
Electronic configuration of neutral atom (where $n=2$ )
List - II
$1^{\text {st }}$ Ionization Energy ( $\mathbf{k J ~ m o l}^{-\mathbf{1}}$ )
A.
$$
\mathrm{ns}^2
$$
I.
2080
B.
$$
\mathrm{ns}^2 \mathrm{np}^1
$$
II.
899
C.
$$
n s^2 n p^3
$$
III.
800
D.
$$
n s^2 n p^6
$$
IV.
1402
Choose the correct answer from the options given below :
Q39.
numerical
+4 / 1
Consider the reaction
$$ 2 \mathrm{H}_2 \mathrm{~S}(\mathrm{~g})+3 \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(\mathrm{l})+2 \mathrm{SO}_2(\mathrm{~g}) $$
The magnitude of enthalpy change for the reaction in $\mathrm{kJ} \mathrm{mol}^{-1}$ is $\_\_\_\_$ . (Nearest integer)
$$ \begin{aligned} Given:\,\,& \Delta_{\mathrm{f}} \mathrm{H}^{\ominus}\left(\mathrm{H}_2 \mathrm{~S}\right)=-20.1 \mathrm{~kJ} \mathrm{~mol}^{-1} \\ & \Delta_{\mathrm{f}} \mathrm{H}^{\ominus}\left(\mathrm{H}_2 \mathrm{O}\right)=-286.0 \mathrm{~kJ} \mathrm{~mol}^{-1} \\ & \Delta_{\mathrm{f}} \mathrm{H}^{\ominus}\left(\mathrm{SO}_2\right)=-297.0 \mathrm{~kJ} \mathrm{~mol}^{-1} \end{aligned} $$
Q40.
mcq single
+4 / 1
Find the correct statements related to group 15 hydrides.
A. Reducing nature increases from $\mathrm{NH}_3$ to $\mathrm{BiH}_3$
B. Tendency to donate lone pair of electrons decreases from $\mathrm{NH}_3$ to $\mathrm{BiH}_3$
C. The stability of hydrides decreases from $\mathrm{NH}_3$ to $\mathrm{BiH}_3$
D. HEH bond angle decreases from $\mathrm{NH}_3$ to $\mathrm{SbH}_3(\mathrm{E}=$ Elements of group 15)
Choose the correct answer from the options given below :
Q41.
numerical
+4 / 1
Number of paramagnetic complexes among the following is $\_\_\_\_$ .
$$ \begin{aligned} & {\left[\mathrm{MnBr}_4\right]^{2-},\left[\mathrm{NiCl}_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-},\left[\mathrm{Ni}(\mathrm{CO})_4\right],\left[\mathrm{CoF}_6\right]^{3-},\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{4-},\left[\mathrm{Mn}(\mathrm{CN})_6\right]^{3-},\left[\mathrm{Ti}(\mathrm{CN})_6\right]^{3-},} \\ & {\left[\mathrm{Cu}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+},\left[\mathrm{Co}\left(\mathrm{C}_2 \mathrm{O}_4\right)_3\right]^{3-}} \end{aligned} $$
Q42.
numerical
+4 / 1
' $x$ ' is the product which is obtained from benzene by reacting it with carbon monoxide and hydrogen chloride in the presence of cuprous chloride. ' $y$ ' is the major product obtained from the benzene by reacting it with ethanoyl chloride in the presence of anhydrous $\mathrm{AlCl}_3$. Product (major) obtained by heating $x$ and $y$ in the presence of alkali is $z$. Total number of $\pi$ (pi) electrons in $z$ is $\_\_\_\_$ .
Q43.
mcq single
+4 / 1
$$ \text { Consider the following reaction. } $$
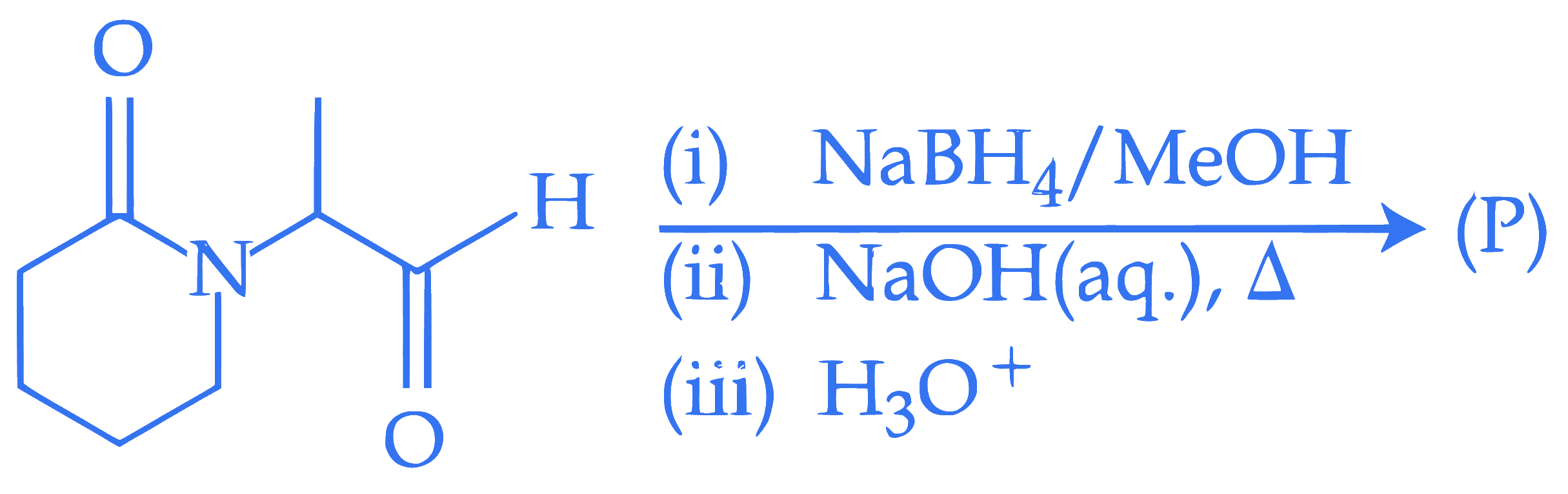 $$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
$$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
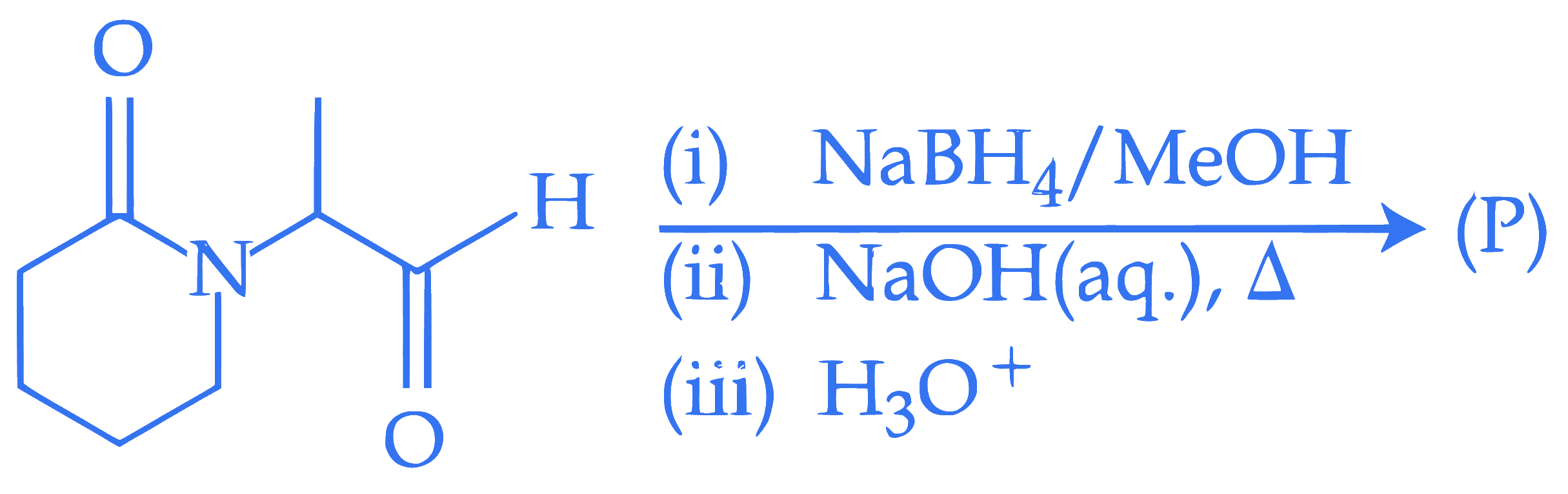 $$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
$$ \text { The major product }(\mathrm{P}) \text { formed is : } $$
Q44.
mcq single
+4 / 1
n-Butane on monochlorination under photochemical condition gives an optically active compound " P ". " P " on further chlorination gives dichloro compounds.
The number of dichloro compounds obtained (ignore stereoisomers) is :
Q45.
mcq single
+4 / 1
$$ \text { The major product of which of the following reaction is not obtained by rearrangement reaction? } $$
Q46.
mcq single
+4 / 1
The incorrect statement from the following with respect to carbohydrates is :
Q47.
mcq single
+4 / 1
$$ \text { The total number of aromatic compounds/species from the following is } $$

Q48.
mcq single
+4 / 1
Given below are two statements :
**Statement I :** Vapours of the liquid with higher boiling point condense before vapours of the liquid with lower boiling points in fractional distillation.
**Statement II :** The vapours rising up in the fractionating column become richer in high boiling component of the mixture.
In the light of the above statements, choose the correct answer from the options given below :
Q49.
mcq single
+4 / 1
Given at 298 K :
$$ \begin{aligned} & \mathrm{E}_{\mathrm{Fe}^{2+} / \mathrm{Fe}}^{\ominus}=\mathrm{X} \text { Volt } \\ & \mathrm{E}_{\mathrm{Fe}^{3+} / \mathrm{Fe}}^{\ominus}=\mathrm{Y} \text { Volt } \end{aligned} $$
The $\mathrm{E}_{\mathrm{Fe}^{3+} / \mathrm{Fe}^{2+}}^{\ominus}$ in Volt at 298 K is given by :
Q50.
numerical
+4 / 1
Consider two radiations of wavelengths
- $\lambda_1=2000\mathop {\rm{A}}\limits^{\rm{o}}$
- $\lambda_2=6000 \mathop {\rm{A}}\limits^{\rm{o}}$
The ratio of the energies of these two radiations $\left(\frac{E_1}{E_2}\right)$ is $\_\_\_\_$ (Nearest integer).
Mathematics
Mathematics
Q1.
mcq single
+4 / 1
A set of four observations has mean 1 and variance 13. Another set of six observations has mean 2 and variance 1 . Then, the variance of all these 10 observations is equal to :
Q2.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=4 \hat{i}-\hat{j}+3 \hat{k}, \overrightarrow{\mathrm{~b}}=10 \hat{i}+2 \hat{j}-\hat{k}$ and a vector $\overrightarrow{\mathrm{c}}$ be such that $2(\overrightarrow{\mathrm{a}} \times \overrightarrow{\mathrm{b}})+3(\overrightarrow{\mathrm{~b}} \times \overrightarrow{\mathrm{c}})=\overrightarrow{0}$.
If $\overrightarrow{\mathrm{a}} \cdot \overrightarrow{\mathrm{c}}=15$, then $\overrightarrow{\mathrm{c}} \cdot(\hat{i}+\hat{j}-3 \hat{k})$ is equal to :
Q3.
mcq single
+4 / 1
A candidate has to go to the examination centre to appear in an examination. The candidate uses only one means of transportation for the entire distance out of bus, scooter and car. The probabilities of the candidate going by bus, scooter and car, respectively, are $\frac{2}{5}, \frac{1}{5}$ and $\frac{2}{5}$. The probabilities that the candidate reaches late at the examination centre are $\frac{1}{5}, \frac{1}{3}$ and $\frac{1}{4}$ if the candidate uses bus, scooter and car, respectively. Given that the candidate reached late at the examination centre, the probability that the candidate travelled by bus is :
Q4.
mcq single
+4 / 1
Let the foot of perpendicular from the point $(\lambda, 2,3)$ on the line $\frac{x-4}{1}=\frac{y-9}{2}=\frac{z-5}{1}$ be the point ( $1, \mu, 2$ ). Then the distance between the lines $\frac{x-1}{2}=\frac{y-2}{3}=\frac{z+4}{6}$ and $\frac{x-\lambda}{2}=\frac{y-\mu}{3}=\frac{z+5}{6}$ is equal to :
Q5.
numerical
+4 / 1
Let a line $L_1$ pass through the origin and be perpendicular to the lines
$\mathrm{L}_2: \overrightarrow{\mathrm{r}}=(3+\mathrm{t}) \hat{i}+(2 \mathrm{t}-1) \hat{j}+(2 \mathrm{t}+4) \hat{k}$ and
$\mathrm{L}_3: \overrightarrow{\mathrm{r}}=(3+2 \mathrm{~s}) \hat{i}+(3+2 \mathrm{~s}) \hat{j}+(2+\mathrm{s}) \hat{k}, \mathrm{t}, \mathrm{s} \in \mathbf{R}$.
If $(a, b, c), a \in \mathbf{Z}$, is the point on $\mathrm{L}_3$ at a distance of $\sqrt{17}$ from the point of intersection of $\mathrm{L}_1$ and $\mathrm{L}_2$, then $(\mathrm{a}+\mathrm{b}+\mathrm{c})^2$ is equal to $\_\_\_\_$ .
Q6.
mcq single
+4 / 1
Let $\alpha=3+4+8+9+13+14+\ldots$ upto 40 terms. If $(\tan \beta)^{\frac{\alpha}{1020}}$ is a root of the equation $x^2+x-2=0, \beta \in\left(0, \frac{\pi}{2}\right)$, then $\sin ^2 \beta+3 \cos ^2 \beta$ is equal to :
Q7.
mcq single
+4 / 1
For the function $f(x)=\mathrm{e}^{\sin |x|}-|x|, x \in \mathbf{R}$, consider the following statements :
**Statement I :** $ f$ is differentiable for all $x \in \mathbf{R}$.
**Statement II :** $ f$ is increasing in $\left(-\pi,-\frac{\pi}{2}\right)$.
In the light of the above statements, choose the correct answer from the options given below :
Q8.
mcq single
+4 / 1
Let $f:(1, \infty) \rightarrow \mathbf{R}$ be a function defined as $f(x)=\frac{x-1}{x+1}$. Let $f^{i+1}(x)=f\left(f^i(x)\right), i=1,2, \ldots, 25$, where $f^1(x)=f(x)$. If $g(x)+f^{26}(x)=0, x \in(1, \infty)$, then the area of the region bounded by the curves $y=g(x), 2 y=2 x-3, y=0$ and $x=4$ is :
Q9.
mcq single
+4 / 1
Let $A=\left[\begin{array}{lll}\alpha & 1 & 2 \\ 2 & 3 & 0 \\ 0 & 4 & 5\end{array}\right]$ and $B=\left[\begin{array}{ccc}1 & 0 & 0 \\ 0 & -5 \alpha & 0 \\ 0 & 4 \alpha & -2 \alpha\end{array}\right]+\operatorname{adj}(A)$. If $\operatorname{det}(B)=66$, then $\operatorname{det}(\operatorname{adj}(A))$ equals :
Q10.
mcq single
+4 / 1
If the system of linear equations :
$$ \begin{aligned} & x+y+z=6 \\ & x+2 y+5 z=10 \\ & 2 x+3 y+\lambda z=\mu \end{aligned} $$
has infinitely many solutions, then the value of $\lambda+\mu$ equals:
Q11.
mcq single
+4 / 1
Let O be the vertex of the parabola $y^2=4 x$ and its chords OP and OQ are perpendicular to each other. If the locus of the mid-point of the line segment PQ is a conic C , then the length of its latus rectum is :
Q12.
mcq single
+4 / 1
Let $\alpha=3 \sin ^{-1}\left(\frac{6}{11}\right)$ and $\beta=3 \cos ^{-1}\left(\frac{4}{9}\right)$, where inverse trigonometric functions take only the principal values.
Given below are two statements :
**Statement I :** $\quad \cos (\alpha+\beta)>0$.
**Statement II :** $\quad \cos (\alpha)<0$.
In the light of the above statements, choose the correct answer from the options given below :
Q13.
mcq single
+4 / 1
If $26\left(\frac{2^3}{3}\left({ }^{12} \mathrm{C}_2\right)+\frac{2^5}{5}\left({ }^{12} \mathrm{C}_4\right)+\frac{2^7}{7}\left({ }^{12} \mathrm{C}_6\right)+\cdots+\frac{2^{13}}{13}\left({ }^{12} \mathrm{C}_{12}\right)\right)=3^{13}-\alpha$, then $\alpha$ is equal to :
Q14.
mcq single
+4 / 1
If a straight line drawn through the point of intersection of the lines $4 x+3 y-1=0$ and $3 x+4 y-1=0$, meets the co-ordinate axes at the points P and Q , then the locus of the mid point of PQ is :
Q15.
mcq single
+4 / 1
Consider the relation $R$ on the set $\{-2,-1,0,1,2\}$ defined by $(a, b) \in R$ if and only if $1+a b>0$. Then, among the statements :
I. The number of elements in R is 17
II. R is an equivalence relation
Q16.
mcq single
+4 / 1
The number of values of $z \in \mathbb{C}$, satisfying the equations $|z-(4+8 i)|=\sqrt{10}$ and $|z-(3+5 i)|+|z-(5+11 i)|=4 \sqrt{5}$, is $:$
Q17.
numerical
+4 / 1
Let $f$ be a polynomial function such that $\log _2(f(x))=\left(\log _2\left(2+\frac{2}{3}+\frac{2}{9}+\ldots \ldots \infty\right)\right) \cdot \log _3\left(1+\frac{f(x)}{f(1 / x)}\right), x>0$ and $f(6)=37$. Then $\sum\limits_{\mathrm{n}=1}^{10} f(\mathrm{n})$ is equal to $\_\_\_\_$ .
Q18.
mcq single
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $x \sqrt{1-x^2} d y+\left(y \sqrt{1-x^2}-x \cos ^{-1} x\right) d x=0, x \in(0,1), \lim _{x \rightarrow 1^{-}} y(x)=1$. Then $y\left(\frac{1}{2}\right)$ equals :
Q19.
numerical
+4 / 1
Consider the circle C : $x^2+y^2-6 x-8 y-11=0$. Let a variable chord AB of the circle C subtend a right angle at the origin. If the locus of the foot of the perpendicular drawn from the origin on the chord AB is the circle $x^2+y^2-\alpha x-\beta y-\gamma=0$, then $\alpha+\beta+2 \gamma$ is equal to $\_\_\_\_$ .
Q20.
mcq single
+4 / 1
Let $\frac{x^2}{f\left(a^2+7 a+3\right)}+\frac{y^2}{f(3 a+15)}=1$ represent an ellipse with major axis along $y$-axis, where $f$ is a strictly decreasing positive function on $\mathbf{R}$. If the set of all possible values of $a$ is $\mathbf{R}-[\alpha, \beta]$, then $\alpha^2+\beta^2$ is equal to :
Q21.
mcq single
+4 / 1
A person has three different bags and four different books. The number of ways, in which he can put these books in the bags so that no bag is empty, is :
Q22.
numerical
+4 / 1
The sum of squares of all the real solutions of the equation
$\log _{(x+1)}\left(2 x^2+5 x+3\right)=4-\log _{(2 x+3)}\left(x^2+2 x+1\right)$ is equal to $\_\_\_\_$ .
Q23.
numerical
+4 / 1
If $\int\limits_{\pi / 6}^{\pi / 4}\left(\cot \left(x-\frac{\pi}{3}\right) \cot \left(x+\frac{\pi}{3}\right)+1\right) d x=\alpha \log _{\mathrm{e}}(\sqrt{3}-1)$, then $9 \alpha^2$ is equal to $\_\_\_\_$ .
Q24.
mcq single
+4 / 1
Let $f(x)=\left\{\begin{array}{cc}\frac{1}{3}, & x \leq \pi / 2 \\ \frac{\mathrm{~b}(1-\sin x)}{(\pi-2 x)^2}, & x>\pi / 2\end{array}\right.$. If $f$ is continuous at $x=\pi / 2$, then the value of $\int\limits_0^{3 \mathrm{~b}-6}\left|x^2+2 x-3\right| \mathrm{d} x$ is :
Q25.
mcq single
+4 / 1
The value of the integral $\int\limits_0^2 \frac{\sqrt{x\left(x^2+x+1\right)}}{(\sqrt{x+1})\left(\sqrt{x^4+x^2+1}\right)} \mathrm{d} x$ is equal to:
Q26.
mcq single
+4 / 1
A set of four observations has mean 1 and variance 13. Another set of six observations has mean 2 and variance 1 . Then, the variance of all these 10 observations is equal to :
Q27.
mcq single
+4 / 1
Let $\overrightarrow{\mathrm{a}}=4 \hat{i}-\hat{j}+3 \hat{k}, \overrightarrow{\mathrm{~b}}=10 \hat{i}+2 \hat{j}-\hat{k}$ and a vector $\overrightarrow{\mathrm{c}}$ be such that $2(\overrightarrow{\mathrm{a}} \times \overrightarrow{\mathrm{b}})+3(\overrightarrow{\mathrm{~b}} \times \overrightarrow{\mathrm{c}})=\overrightarrow{0}$.
If $\overrightarrow{\mathrm{a}} \cdot \overrightarrow{\mathrm{c}}=15$, then $\overrightarrow{\mathrm{c}} \cdot(\hat{i}+\hat{j}-3 \hat{k})$ is equal to :
Q28.
mcq single
+4 / 1
A candidate has to go to the examination centre to appear in an examination. The candidate uses only one means of transportation for the entire distance out of bus, scooter and car. The probabilities of the candidate going by bus, scooter and car, respectively, are $\frac{2}{5}, \frac{1}{5}$ and $\frac{2}{5}$. The probabilities that the candidate reaches late at the examination centre are $\frac{1}{5}, \frac{1}{3}$ and $\frac{1}{4}$ if the candidate uses bus, scooter and car, respectively. Given that the candidate reached late at the examination centre, the probability that the candidate travelled by bus is :
Q29.
mcq single
+4 / 1
Let the foot of perpendicular from the point $(\lambda, 2,3)$ on the line $\frac{x-4}{1}=\frac{y-9}{2}=\frac{z-5}{1}$ be the point ( $1, \mu, 2$ ). Then the distance between the lines $\frac{x-1}{2}=\frac{y-2}{3}=\frac{z+4}{6}$ and $\frac{x-\lambda}{2}=\frac{y-\mu}{3}=\frac{z+5}{6}$ is equal to :
Q30.
numerical
+4 / 1
Let a line $L_1$ pass through the origin and be perpendicular to the lines
$\mathrm{L}_2: \overrightarrow{\mathrm{r}}=(3+\mathrm{t}) \hat{i}+(2 \mathrm{t}-1) \hat{j}+(2 \mathrm{t}+4) \hat{k}$ and
$\mathrm{L}_3: \overrightarrow{\mathrm{r}}=(3+2 \mathrm{~s}) \hat{i}+(3+2 \mathrm{~s}) \hat{j}+(2+\mathrm{s}) \hat{k}, \mathrm{t}, \mathrm{s} \in \mathbf{R}$.
If $(a, b, c), a \in \mathbf{Z}$, is the point on $\mathrm{L}_3$ at a distance of $\sqrt{17}$ from the point of intersection of $\mathrm{L}_1$ and $\mathrm{L}_2$, then $(\mathrm{a}+\mathrm{b}+\mathrm{c})^2$ is equal to $\_\_\_\_$ .
Q31.
mcq single
+4 / 1
Let $\alpha=3+4+8+9+13+14+\ldots$ upto 40 terms. If $(\tan \beta)^{\frac{\alpha}{1020}}$ is a root of the equation $x^2+x-2=0, \beta \in\left(0, \frac{\pi}{2}\right)$, then $\sin ^2 \beta+3 \cos ^2 \beta$ is equal to :
Q32.
mcq single
+4 / 1
For the function $f(x)=\mathrm{e}^{\sin |x|}-|x|, x \in \mathbf{R}$, consider the following statements :
**Statement I :** $ f$ is differentiable for all $x \in \mathbf{R}$.
**Statement II :** $ f$ is increasing in $\left(-\pi,-\frac{\pi}{2}\right)$.
In the light of the above statements, choose the correct answer from the options given below :
Q33.
mcq single
+4 / 1
Let $f:(1, \infty) \rightarrow \mathbf{R}$ be a function defined as $f(x)=\frac{x-1}{x+1}$. Let $f^{i+1}(x)=f\left(f^i(x)\right), i=1,2, \ldots, 25$, where $f^1(x)=f(x)$. If $g(x)+f^{26}(x)=0, x \in(1, \infty)$, then the area of the region bounded by the curves $y=g(x), 2 y=2 x-3, y=0$ and $x=4$ is :
Q34.
mcq single
+4 / 1
Let $A=\left[\begin{array}{lll}\alpha & 1 & 2 \\ 2 & 3 & 0 \\ 0 & 4 & 5\end{array}\right]$ and $B=\left[\begin{array}{ccc}1 & 0 & 0 \\ 0 & -5 \alpha & 0 \\ 0 & 4 \alpha & -2 \alpha\end{array}\right]+\operatorname{adj}(A)$. If $\operatorname{det}(B)=66$, then $\operatorname{det}(\operatorname{adj}(A))$ equals :
Q35.
mcq single
+4 / 1
If the system of linear equations :
$$ \begin{aligned} & x+y+z=6 \\ & x+2 y+5 z=10 \\ & 2 x+3 y+\lambda z=\mu \end{aligned} $$
has infinitely many solutions, then the value of $\lambda+\mu$ equals:
Q36.
mcq single
+4 / 1
Let O be the vertex of the parabola $y^2=4 x$ and its chords OP and OQ are perpendicular to each other. If the locus of the mid-point of the line segment PQ is a conic C , then the length of its latus rectum is :
Q37.
mcq single
+4 / 1
Let $\alpha=3 \sin ^{-1}\left(\frac{6}{11}\right)$ and $\beta=3 \cos ^{-1}\left(\frac{4}{9}\right)$, where inverse trigonometric functions take only the principal values.
Given below are two statements :
**Statement I :** $\quad \cos (\alpha+\beta)>0$.
**Statement II :** $\quad \cos (\alpha)<0$.
In the light of the above statements, choose the correct answer from the options given below :
Q38.
mcq single
+4 / 1
If $26\left(\frac{2^3}{3}\left({ }^{12} \mathrm{C}_2\right)+\frac{2^5}{5}\left({ }^{12} \mathrm{C}_4\right)+\frac{2^7}{7}\left({ }^{12} \mathrm{C}_6\right)+\cdots+\frac{2^{13}}{13}\left({ }^{12} \mathrm{C}_{12}\right)\right)=3^{13}-\alpha$, then $\alpha$ is equal to :
Q39.
mcq single
+4 / 1
If a straight line drawn through the point of intersection of the lines $4 x+3 y-1=0$ and $3 x+4 y-1=0$, meets the co-ordinate axes at the points P and Q , then the locus of the mid point of PQ is :
Q40.
mcq single
+4 / 1
Consider the relation $R$ on the set $\{-2,-1,0,1,2\}$ defined by $(a, b) \in R$ if and only if $1+a b>0$. Then, among the statements :
I. The number of elements in R is 17
II. R is an equivalence relation
Q41.
mcq single
+4 / 1
The number of values of $z \in \mathbb{C}$, satisfying the equations $|z-(4+8 i)|=\sqrt{10}$ and $|z-(3+5 i)|+|z-(5+11 i)|=4 \sqrt{5}$, is $:$
Q42.
numerical
+4 / 1
Let $f$ be a polynomial function such that $\log _2(f(x))=\left(\log _2\left(2+\frac{2}{3}+\frac{2}{9}+\ldots \ldots \infty\right)\right) \cdot \log _3\left(1+\frac{f(x)}{f(1 / x)}\right), x>0$ and $f(6)=37$. Then $\sum\limits_{\mathrm{n}=1}^{10} f(\mathrm{n})$ is equal to $\_\_\_\_$ .
Q43.
mcq single
+4 / 1
Let $y=y(x)$ be the solution of the differential equation $x \sqrt{1-x^2} d y+\left(y \sqrt{1-x^2}-x \cos ^{-1} x\right) d x=0, x \in(0,1), \lim _{x \rightarrow 1^{-}} y(x)=1$. Then $y\left(\frac{1}{2}\right)$ equals :
Q44.
numerical
+4 / 1
Consider the circle C : $x^2+y^2-6 x-8 y-11=0$. Let a variable chord AB of the circle C subtend a right angle at the origin. If the locus of the foot of the perpendicular drawn from the origin on the chord AB is the circle $x^2+y^2-\alpha x-\beta y-\gamma=0$, then $\alpha+\beta+2 \gamma$ is equal to $\_\_\_\_$ .
Q45.
mcq single
+4 / 1
Let $\frac{x^2}{f\left(a^2+7 a+3\right)}+\frac{y^2}{f(3 a+15)}=1$ represent an ellipse with major axis along $y$-axis, where $f$ is a strictly decreasing positive function on $\mathbf{R}$. If the set of all possible values of $a$ is $\mathbf{R}-[\alpha, \beta]$, then $\alpha^2+\beta^2$ is equal to :
Q46.
mcq single
+4 / 1
A person has three different bags and four different books. The number of ways, in which he can put these books in the bags so that no bag is empty, is :
Q47.
numerical
+4 / 1
The sum of squares of all the real solutions of the equation
$\log _{(x+1)}\left(2 x^2+5 x+3\right)=4-\log _{(2 x+3)}\left(x^2+2 x+1\right)$ is equal to $\_\_\_\_$ .
Q48.
numerical
+4 / 1
If $\int\limits_{\pi / 6}^{\pi / 4}\left(\cot \left(x-\frac{\pi}{3}\right) \cot \left(x+\frac{\pi}{3}\right)+1\right) d x=\alpha \log _{\mathrm{e}}(\sqrt{3}-1)$, then $9 \alpha^2$ is equal to $\_\_\_\_$ .
Q49.
mcq single
+4 / 1
Let $f(x)=\left\{\begin{array}{cc}\frac{1}{3}, & x \leq \pi / 2 \\ \frac{\mathrm{~b}(1-\sin x)}{(\pi-2 x)^2}, & x>\pi / 2\end{array}\right.$. If $f$ is continuous at $x=\pi / 2$, then the value of $\int\limits_0^{3 \mathrm{~b}-6}\left|x^2+2 x-3\right| \mathrm{d} x$ is :
Q50.
mcq single
+4 / 1
The value of the integral $\int\limits_0^2 \frac{\sqrt{x\left(x^2+x+1\right)}}{(\sqrt{x+1})\left(\sqrt{x^4+x^2+1}\right)} \mathrm{d} x$ is equal to:
Physics
Physics
Q1.
mcq single
+4 / 1
$$ \text { The output } Y \text { for the given inputs } A \text { and } B \text { to the circuit is : } $$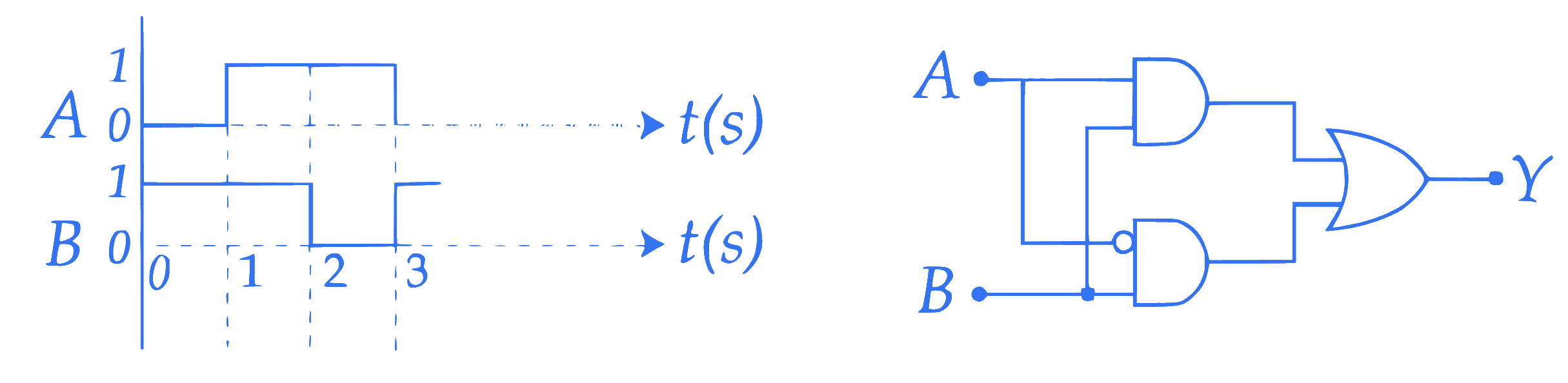
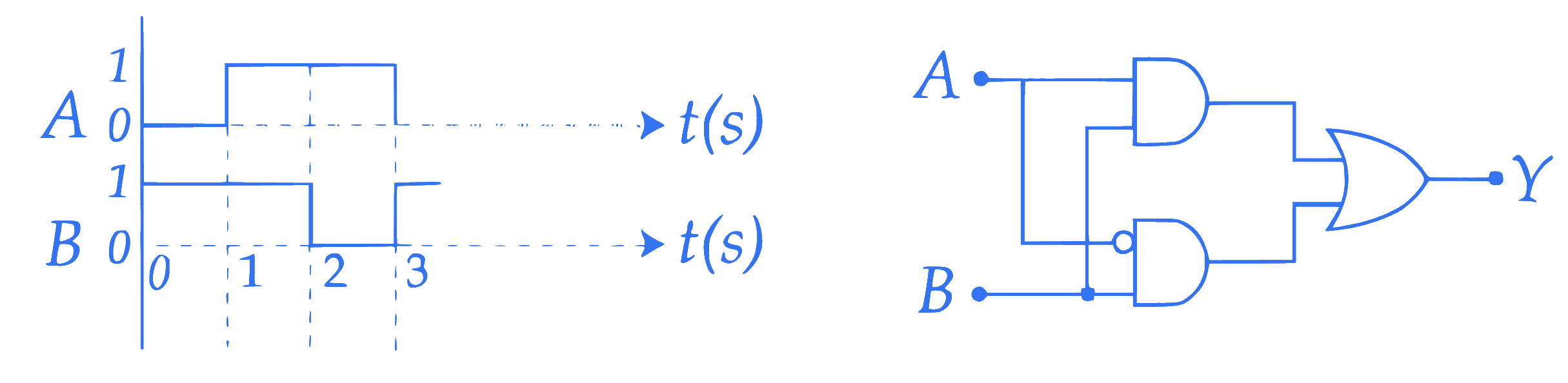
Q2.
mcq single
+4 / 1
The frequency of oscillation of a mass $m$ suspended by a spring is $v_1$. If the length of the spring is cut to half, the same mass oscillates with frequency $v_2$. The value of $v_2 / v_1$ is $\_\_\_\_$ .
Q3.
mcq single
+4 / 1
A solid cylinder having radius $R$ and length $L$ is slipping on a rough horizontal plane. At time $t=0$ the cylinder has a translational velocity $v_{\mathrm{o}}=49 \mathrm{~m} / \mathrm{s}$, perpendicular to its axis and a rotational velocity $v_{\mathrm{o}} / 4 R$ about the centre. The time taken by the cylinder to start rolling is $\_\_\_\_$ seconds. (coefficient of kinetic friction $\mu_K=0.25$ and $g=9.8 \mathrm{~m} / \mathrm{s}^2$ )
Q4.
numerical
+4 / 1
Two masses of 3.4 kg and 2.5 kg are accelerated from an initial speed of $5 \mathrm{~m} / \mathrm{s}$ and $12 \mathrm{~m} / \mathrm{s}$, respectively. The distances traversed by the masses in the $5^{\text {th }}$ second are 104 m and 129 m , respectively. The ratio of their momenta after 10 s is $\frac{x}{8}$. The value of $x$ is $\_\_\_\_$。
Q5.
mcq single
+4 / 1
A 30 cm long solenoid has 10 turns per cm and area of $5 \mathrm{~cm}^2$. The current through the solenoid coil varies from 2 A to 4 A in 3.14 s . The e.m.f. induced in the coil is $\alpha \times 10^{-5} \mathrm{~V}$. The value $\alpha$ is $\_\_\_\_$ .
Q6.
mcq single
+4 / 1
A car moving with a speed of $54 \mathrm{~km} / \mathrm{h}$ takes a turn of radius 20 m . A simple pendulum is suspended from the ceiling of the car. Determine the angle made by the string of the pendulum with the vertical during the turning. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q7.
mcq single
+4 / 1
Initial pressure and volume of a monoatomic ideal gas are $P$ and $V$. The change in internal energy of this gas in adiabatic expansion to volume $V_{\text {final }}=27 \mathrm{~V}$ is $\_\_\_\_$ J.
Q8.
mcq single
+4 / 1
One mole of diatomic gas having rotational modes only is kept in a cylinder with a piston system. The cross-section area of the cylinder is $4 \mathrm{~cm}^2$. The gas is heated slowly to raise the temperature by $1.2^{\circ} \mathrm{C}$ during which the piston moves by 25 mm . The amount of heat supplied to the gas is $\_\_\_\_$ J.
(Atmospheric pressure $=100 \mathrm{kPa}, R=8.3 \mathrm{~J} / \mathrm{mol} . \mathrm{K}$ ) (Neglect mass of the piston)
Q9.
mcq single
+4 / 1
A spherical liquid drop of radius $R$ acquires the terminal velocity $v_1$ when falls through a gas of viscosity $\eta$. Now the drop is broken into 64 identical droplets and each droplet acquires terminal velocity $v_2$ falling through the same gas. The ratio of terminal velocities $v_1 / v_2$ is $\_\_\_\_$ .
Q10.
mcq single
+4 / 1
A liquid of density $600 \mathrm{~kg} / \mathrm{m}^3$ flowing steadily in a tube of varying cross-section. The cross-section at a point $A$ is $1.0 \mathrm{~cm}^2$ and that at $B$ is $20 \mathrm{~mm}^2$. Both the points $A$ and $B$ are in same horizontal plane, the speed of the liquid at $A$ is $10 \mathrm{~cm} / \mathrm{s}$. The difference in pressures at $A$ and $B$ points is $\_\_\_\_$ Pa.
Q11.
mcq single
+4 / 1
A monochromatic source of light operating at 15 kW emits $2.5 \times 10^{22}$ photons $/ \mathrm{s}$. The region of an electromagnetic spectrum to which the emitted electromagnetic radiation belongs to $\_\_\_\_$。
(Take $h=6.6 \times 10^{-34} \mathrm{~J} . \mathrm{s}$ and $c=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ).
Q12.
mcq single
+4 / 1
$K_1$ and $K_2$ be the maximum kinetic energies of photoelectrons emitted from a surface of a given material for the light of wavelength $\lambda_1$ and $\lambda_2$, respectively. If $\lambda_1=2 \lambda_2$ then the work function of material is given by :
Q13.
mcq single
+4 / 1
Two point charges $\mathrm{q}_1=3 \mu C$ and $\mathrm{q}_2=-4 \mu C$ are placed at points $(2 \hat{i}+3 \hat{j}+3 \hat{k})$ and $(\hat{i}+\hat{j}+\hat{k})$ respectively. Force on charge $\mathrm{q}_2$ is $\_\_\_\_$ N. (Take $\frac{1}{4 \pi \epsilon_0}=9 \times 10^9$ SI Units)
Q14.
numerical
+4 / 1
The stored charge in the capacitor in steady state of the following circuit is $\_\_\_\_$ $\mu \mathrm{C}$.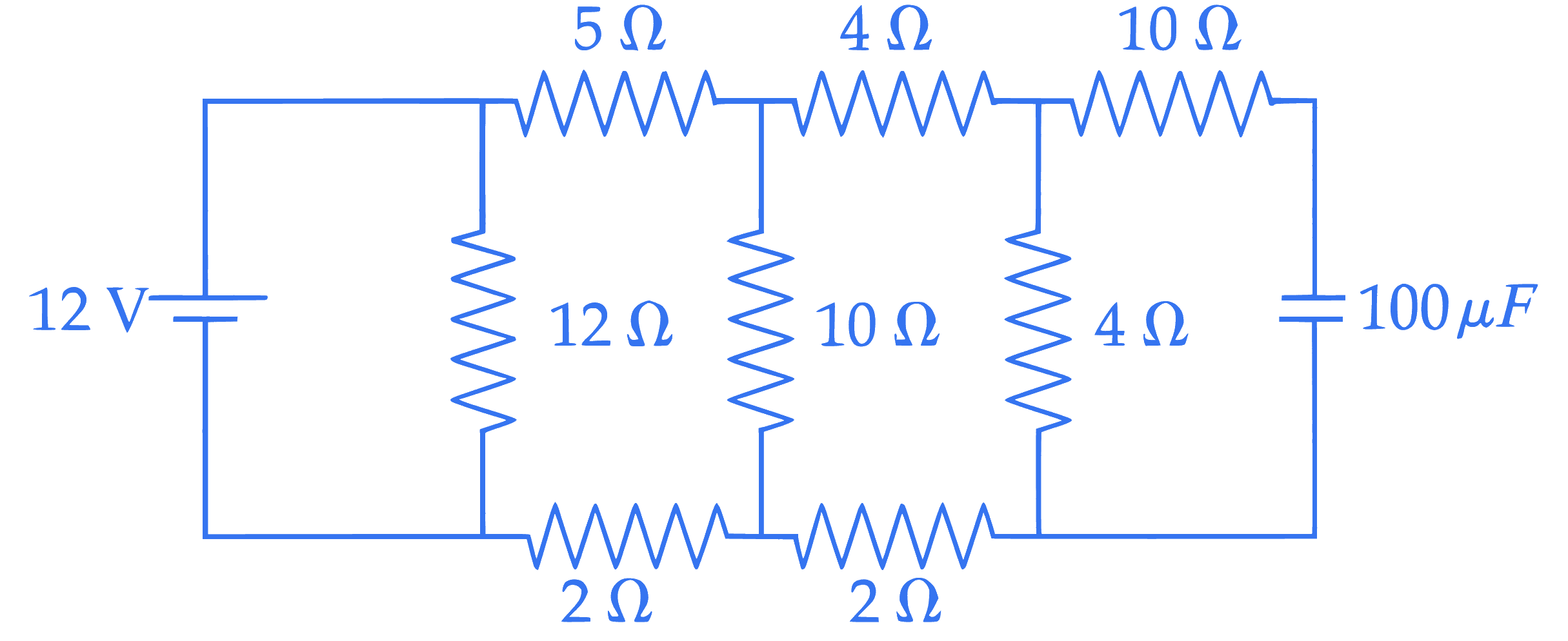

Q15.
numerical
+4 / 1
A parallel plate capacitor is having separation between plates 0.885 mm . It has a capacitance of $1 \mu \mathrm{~F}$ when the space between the plates is filled with an insulating material of resistivity $1 \times 10^{13} \Omega \mathrm{~m}$ and resistance $17.7 \times 10^{14} \Omega$. Relative permittivity of the insulating material is $\alpha \times 10^7$. The value of $\alpha$ is $\_\_\_\_$ .
(Take permittivity of free space $=8.85 \times 10^{-12} \mathrm{~F} / \mathrm{m}$ )
Q16.
mcq single
+4 / 1
A thin biconvex lens is prepared from the glass $(\mu=1.5)$ both curved surfaces of which have equal radii of 20 cm each. Left side surface of the lens is silvered from outside to make it reflecting. To have the position of image and object at the same place, the object should be placed, from the lens at a distance of $\_\_\_\_$ cm.
Q17.
mcq single
+4 / 1
Light ray incident along a vector $\overrightarrow{A O}(\overrightarrow{A O}=2 \hat{i}-3 \hat{j})$ emerges out along vector $\overrightarrow{O B}(\overrightarrow{O B}=C \hat{i}-4 \hat{j})$ as shown in the figure below. The value of $C$ is $\_\_\_\_$ .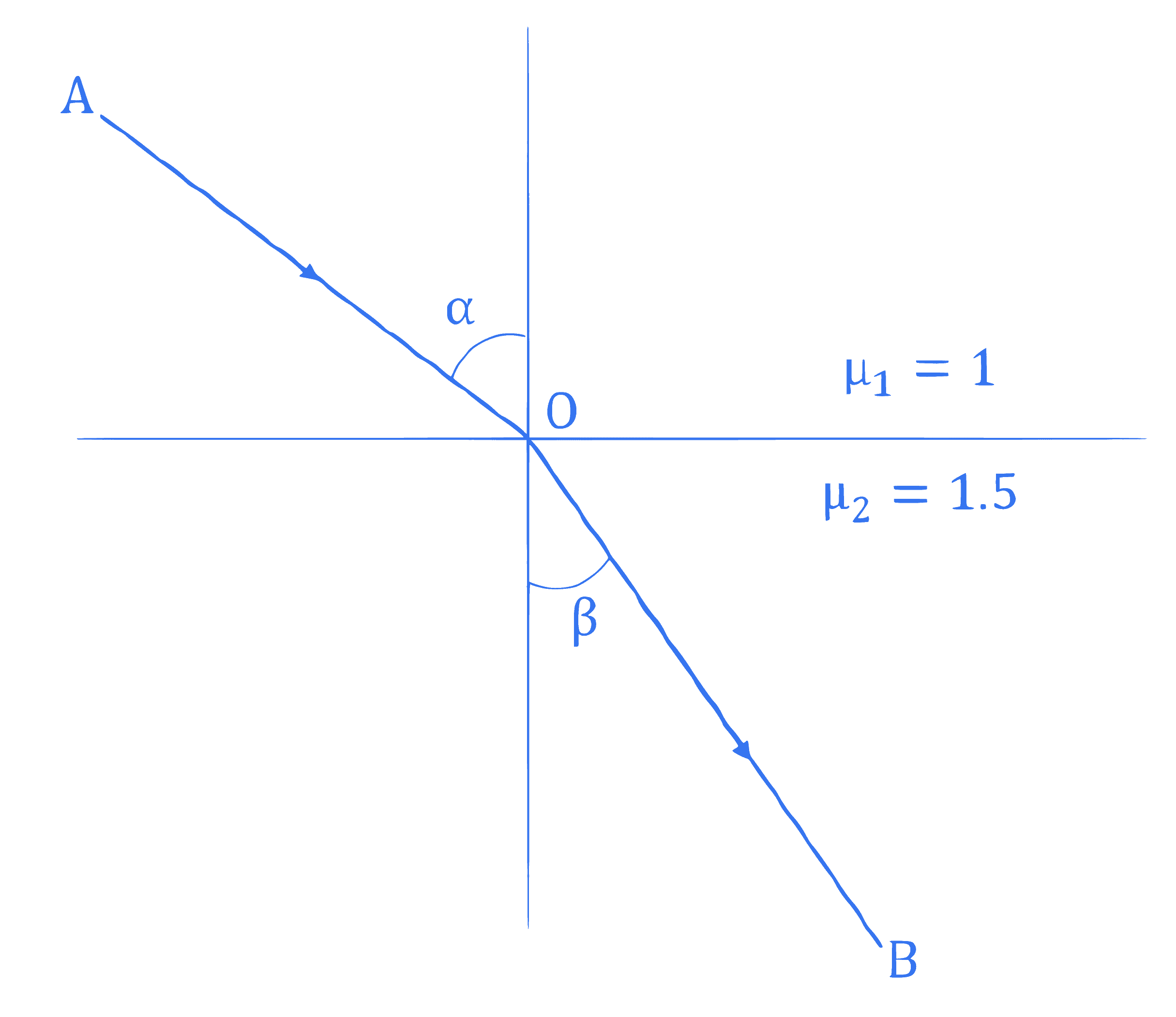

Q18.
numerical
+4 / 1
A 5 mg particle carrying a charge of $5 \pi \times 10^{-6} \mathrm{C}$ is moving with velocity of $(3 \hat{i}+2 \hat{k}) \times 10^{-2} \mathrm{~m} / \mathrm{s}$ in a region having magnetic field $\vec{B}=0.1 \hat{k} \mathrm{~Wb} / \mathrm{m}^2$. It moves a distance of $\alpha$ meter along $\hat{k}$ when it completes 5 revolutions. The value of $\alpha$ is $\_\_\_\_$。
Q19.
mcq single
+4 / 1
A current carrying circular loop of radius 2 cm with unit normal $\hat{n}=\frac{\hat{k}+\hat{i}}{\sqrt{2}}$ is placed in a magnetic field, $\vec{B}=B_o(3 \hat{i}+2 \hat{k})$. If $B_o=4 \times 10^{-3} \mathrm{~T}$ and current $I=100 \sqrt{2} \mathrm{~A}$, the torque experienced by the loop is $\_\_\_\_$ Wb.A. ( $\pi=3.14$ )
Q20.
mcq single
+4 / 1
A new unit ( $\alpha$ ) of length is chosen such that it is equal to the speed of light in vacuum. What is the distance between Venus and Earth in terms of $\alpha$ units if light takes 6 min. 40 s to cover this distance?
Q21.
mcq single
+4 / 1
Consider the equation $H=\frac{x^p \epsilon^q E^r}{t^s}$
Where $H=$ magnetic field; $E=$ electric field, $\epsilon=$ permittivity, $x=$ distance, $t=$ time The values of $p, q, r$ and $s$ respectively are :
Q22.
mcq single
+4 / 1
A gas balloon is going up with a constant velocity of $10 \mathrm{~m} / \mathrm{s}$. When this balloon reached a height of 75 m , a stone is dropped from it and balloon keeps moving up with the same velocity. The height of the balloon when the stone hits the ground is $\_\_\_\_$ m. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q23.
mcq single
+4 / 1
Two radioactive substances A and B of mass numbers 200 and 212 respectively, shows spontaneous $\alpha$-decay with same $Q$ value of 1 MeV . The ratio of energies of $\alpha$-rays produced by A and B is $\_\_\_\_$ .
Q24.
mcq single
+4 / 1
Two identical bodies, projected with the same speed at two different angles cover the same horizontal range $R$. If the time of flight of these bodies are 5 s and 10 s , respectively, then the value of $R$ is
$\_\_\_\_$ m. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q25.
mcq single
+4 / 1
$$ \text { The output } Y \text { for the given inputs } A \text { and } B \text { to the circuit is : } $$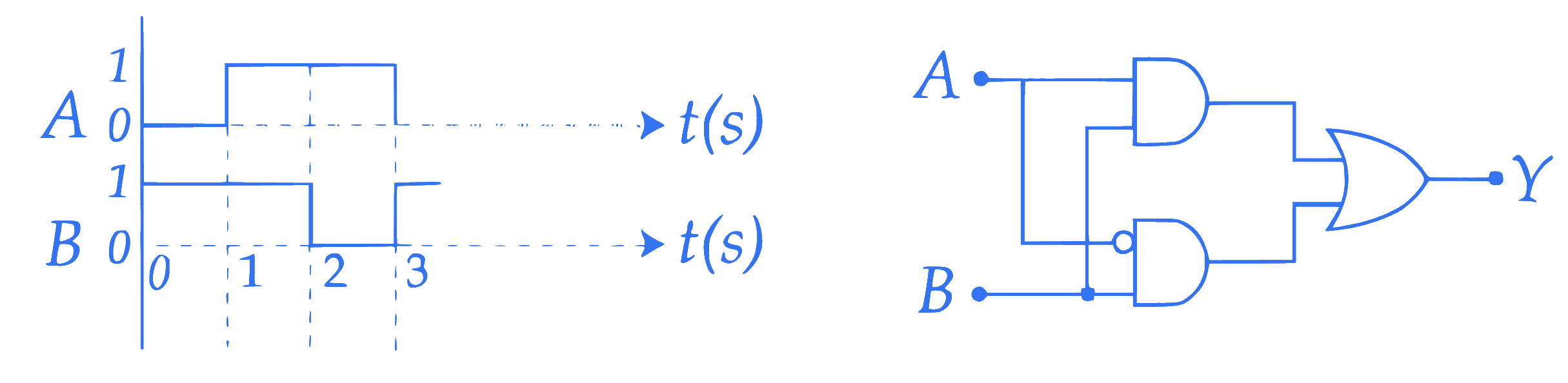
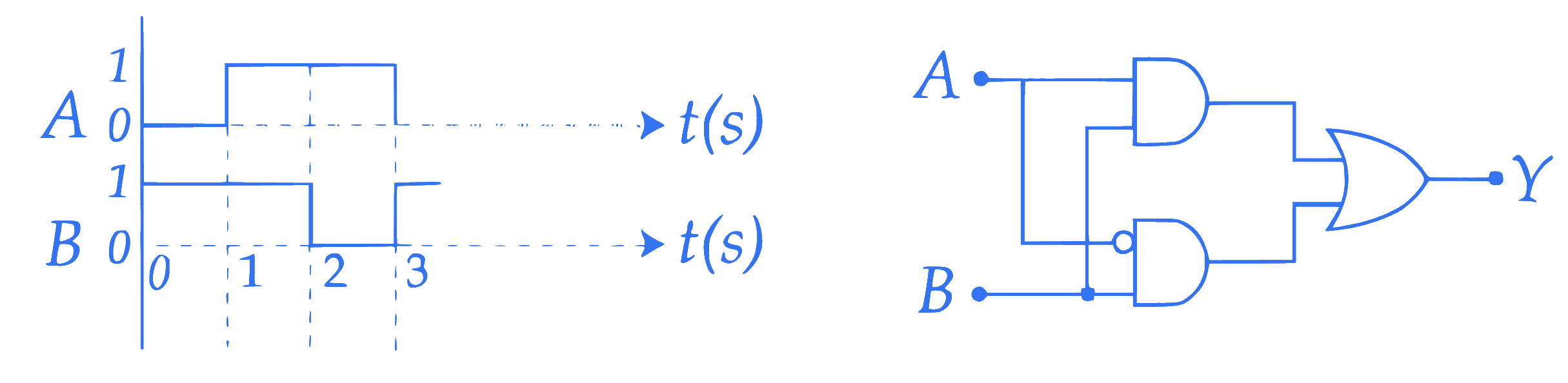
Q26.
mcq single
+4 / 1
The frequency of oscillation of a mass $m$ suspended by a spring is $v_1$. If the length of the spring is cut to half, the same mass oscillates with frequency $v_2$. The value of $v_2 / v_1$ is $\_\_\_\_$ .
Q27.
mcq single
+4 / 1
A solid cylinder having radius $R$ and length $L$ is slipping on a rough horizontal plane. At time $t=0$ the cylinder has a translational velocity $v_{\mathrm{o}}=49 \mathrm{~m} / \mathrm{s}$, perpendicular to its axis and a rotational velocity $v_{\mathrm{o}} / 4 R$ about the centre. The time taken by the cylinder to start rolling is $\_\_\_\_$ seconds. (coefficient of kinetic friction $\mu_K=0.25$ and $g=9.8 \mathrm{~m} / \mathrm{s}^2$ )
Q28.
numerical
+4 / 1
Two masses of 3.4 kg and 2.5 kg are accelerated from an initial speed of $5 \mathrm{~m} / \mathrm{s}$ and $12 \mathrm{~m} / \mathrm{s}$, respectively. The distances traversed by the masses in the $5^{\text {th }}$ second are 104 m and 129 m , respectively. The ratio of their momenta after 10 s is $\frac{x}{8}$. The value of $x$ is $\_\_\_\_$。
Q29.
mcq single
+4 / 1
A 30 cm long solenoid has 10 turns per cm and area of $5 \mathrm{~cm}^2$. The current through the solenoid coil varies from 2 A to 4 A in 3.14 s . The e.m.f. induced in the coil is $\alpha \times 10^{-5} \mathrm{~V}$. The value $\alpha$ is $\_\_\_\_$ .
Q30.
mcq single
+4 / 1
A car moving with a speed of $54 \mathrm{~km} / \mathrm{h}$ takes a turn of radius 20 m . A simple pendulum is suspended from the ceiling of the car. Determine the angle made by the string of the pendulum with the vertical during the turning. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q31.
mcq single
+4 / 1
Initial pressure and volume of a monoatomic ideal gas are $P$ and $V$. The change in internal energy of this gas in adiabatic expansion to volume $V_{\text {final }}=27 \mathrm{~V}$ is $\_\_\_\_$ J.
Q32.
mcq single
+4 / 1
One mole of diatomic gas having rotational modes only is kept in a cylinder with a piston system. The cross-section area of the cylinder is $4 \mathrm{~cm}^2$. The gas is heated slowly to raise the temperature by $1.2^{\circ} \mathrm{C}$ during which the piston moves by 25 mm . The amount of heat supplied to the gas is $\_\_\_\_$ J.
(Atmospheric pressure $=100 \mathrm{kPa}, R=8.3 \mathrm{~J} / \mathrm{mol} . \mathrm{K}$ ) (Neglect mass of the piston)
Q33.
mcq single
+4 / 1
A spherical liquid drop of radius $R$ acquires the terminal velocity $v_1$ when falls through a gas of viscosity $\eta$. Now the drop is broken into 64 identical droplets and each droplet acquires terminal velocity $v_2$ falling through the same gas. The ratio of terminal velocities $v_1 / v_2$ is $\_\_\_\_$ .
Q34.
mcq single
+4 / 1
A liquid of density $600 \mathrm{~kg} / \mathrm{m}^3$ flowing steadily in a tube of varying cross-section. The cross-section at a point $A$ is $1.0 \mathrm{~cm}^2$ and that at $B$ is $20 \mathrm{~mm}^2$. Both the points $A$ and $B$ are in same horizontal plane, the speed of the liquid at $A$ is $10 \mathrm{~cm} / \mathrm{s}$. The difference in pressures at $A$ and $B$ points is $\_\_\_\_$ Pa.
Q35.
mcq single
+4 / 1
A monochromatic source of light operating at 15 kW emits $2.5 \times 10^{22}$ photons $/ \mathrm{s}$. The region of an electromagnetic spectrum to which the emitted electromagnetic radiation belongs to $\_\_\_\_$。
(Take $h=6.6 \times 10^{-34} \mathrm{~J} . \mathrm{s}$ and $c=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ).
Q36.
mcq single
+4 / 1
$K_1$ and $K_2$ be the maximum kinetic energies of photoelectrons emitted from a surface of a given material for the light of wavelength $\lambda_1$ and $\lambda_2$, respectively. If $\lambda_1=2 \lambda_2$ then the work function of material is given by :
Q37.
mcq single
+4 / 1
Two point charges $\mathrm{q}_1=3 \mu C$ and $\mathrm{q}_2=-4 \mu C$ are placed at points $(2 \hat{i}+3 \hat{j}+3 \hat{k})$ and $(\hat{i}+\hat{j}+\hat{k})$ respectively. Force on charge $\mathrm{q}_2$ is $\_\_\_\_$ N. (Take $\frac{1}{4 \pi \epsilon_0}=9 \times 10^9$ SI Units)
Q38.
numerical
+4 / 1
The stored charge in the capacitor in steady state of the following circuit is $\_\_\_\_$ $\mu \mathrm{C}$.

Q39.
numerical
+4 / 1
A parallel plate capacitor is having separation between plates 0.885 mm . It has a capacitance of $1 \mu \mathrm{~F}$ when the space between the plates is filled with an insulating material of resistivity $1 \times 10^{13} \Omega \mathrm{~m}$ and resistance $17.7 \times 10^{14} \Omega$. Relative permittivity of the insulating material is $\alpha \times 10^7$. The value of $\alpha$ is $\_\_\_\_$ .
(Take permittivity of free space $=8.85 \times 10^{-12} \mathrm{~F} / \mathrm{m}$ )
Q40.
mcq single
+4 / 1
A thin biconvex lens is prepared from the glass $(\mu=1.5)$ both curved surfaces of which have equal radii of 20 cm each. Left side surface of the lens is silvered from outside to make it reflecting. To have the position of image and object at the same place, the object should be placed, from the lens at a distance of $\_\_\_\_$ cm.
Q41.
mcq single
+4 / 1
Light ray incident along a vector $\overrightarrow{A O}(\overrightarrow{A O}=2 \hat{i}-3 \hat{j})$ emerges out along vector $\overrightarrow{O B}(\overrightarrow{O B}=C \hat{i}-4 \hat{j})$ as shown in the figure below. The value of $C$ is $\_\_\_\_$ .

Q42.
numerical
+4 / 1
A 5 mg particle carrying a charge of $5 \pi \times 10^{-6} \mathrm{C}$ is moving with velocity of $(3 \hat{i}+2 \hat{k}) \times 10^{-2} \mathrm{~m} / \mathrm{s}$ in a region having magnetic field $\vec{B}=0.1 \hat{k} \mathrm{~Wb} / \mathrm{m}^2$. It moves a distance of $\alpha$ meter along $\hat{k}$ when it completes 5 revolutions. The value of $\alpha$ is $\_\_\_\_$。
Q43.
mcq single
+4 / 1
A current carrying circular loop of radius 2 cm with unit normal $\hat{n}=\frac{\hat{k}+\hat{i}}{\sqrt{2}}$ is placed in a magnetic field, $\vec{B}=B_o(3 \hat{i}+2 \hat{k})$. If $B_o=4 \times 10^{-3} \mathrm{~T}$ and current $I=100 \sqrt{2} \mathrm{~A}$, the torque experienced by the loop is $\_\_\_\_$ Wb.A. ( $\pi=3.14$ )
Q44.
mcq single
+4 / 1
A new unit ( $\alpha$ ) of length is chosen such that it is equal to the speed of light in vacuum. What is the distance between Venus and Earth in terms of $\alpha$ units if light takes 6 min. 40 s to cover this distance?
Q45.
mcq single
+4 / 1
Consider the equation $H=\frac{x^p \epsilon^q E^r}{t^s}$
Where $H=$ magnetic field; $E=$ electric field, $\epsilon=$ permittivity, $x=$ distance, $t=$ time The values of $p, q, r$ and $s$ respectively are :
Q46.
mcq single
+4 / 1
A gas balloon is going up with a constant velocity of $10 \mathrm{~m} / \mathrm{s}$. When this balloon reached a height of 75 m , a stone is dropped from it and balloon keeps moving up with the same velocity. The height of the balloon when the stone hits the ground is $\_\_\_\_$ m. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q47.
mcq single
+4 / 1
Two radioactive substances A and B of mass numbers 200 and 212 respectively, shows spontaneous $\alpha$-decay with same $Q$ value of 1 MeV . The ratio of energies of $\alpha$-rays produced by A and B is $\_\_\_\_$ .
Q48.
mcq single
+4 / 1
Two identical bodies, projected with the same speed at two different angles cover the same horizontal range $R$. If the time of flight of these bodies are 5 s and 10 s , respectively, then the value of $R$ is
$\_\_\_\_$ m. (Take $g=10 \mathrm{~m} / \mathrm{s}^2$ )