MHT CET (PCB) 2024 22th April Evening Shift
MEDICAL 2024 Previous Year
3 hDuration
200Total Marks
400Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 400 across 3 section(s); maximum marks: 200.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+1 / 0
Identify the product ' B ' in the following sequence of reactions.
Ethanenitrile $\xrightarrow{\mathrm{SnCl}_2, \mathrm{HCl}} \mathrm{A} \xrightarrow{\mathrm{H}_3 \mathrm{O}^{+}} \mathrm{B}+\mathrm{NH}_4 \mathrm{Cl}$
Q2.
mcq single
+1 / 0
Which of the following has the highest basic strength?
Q3.
mcq single
+1 / 0
Identify false statement about zinc.
Q4.
mcq single
+1 / 0
Which from following metal ions in their respective oxidation states forms colourless compounds?
Q5.
mcq single
+1 / 0
Which from following polymers is obtained by addition polymerisation method?
Q6.
mcq single
+1 / 0
Which from the following statements about nylon 2-nylon 6 is NOT correct?
Q7.
mcq single
+1 / 0
Identify a molecule having octahedral geometry as per VSEPR theory.
Q8.
mcq single
+1 / 0
Which of the following is NOT an example of diamagnetic substance?
Q9.
mcq single
+1 / 0
What is the radius of first orbit of monopositive helium ion?
Q10.
mcq multi
+1 / 0
What is the representation of element ( X ) having mass number 40 containing 22 neutrons.
Q11.
mcq single
+1 / 0
Which from the following buffers is used for precipitation of cations of IIIA group in qualitative analysis?
Q12.
mcq single
+1 / 0
The dissociation constant of weak monoacidic base is $1.8 \times 10^{-5}$. Calculate the degree of dissociation in 0.02 M solution.
Q13.
mcq single
+1 / 0
Calculate the pH of buffer solution containing 0.12 M weak acid and 0.48 M of its salt with strong base if $\mathrm{pK}_{\mathrm{a}}$ is 3.82 .
Q14.
mcq single
+1 / 0
What is rate constant of a first order reaction having half life 138.6 minute?
Q15.
mcq single
+1 / 0
What is the order of a reaction having unit of the rate constant $\mathrm{mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1}$ ?
Q16.
mcq single
+1 / 0
For the reaction,
$$ 3 \mathrm{I}_{(\mathrm{aq})}^{-}+\mathrm{S}_2 \mathrm{O}_{8(\mathrm{aq})}^{2-} \longrightarrow 2 \mathrm{SO}_{4(\mathrm{aq})}^{2-}+\mathrm{I}_{3(\mathrm{aq})}^{-} $$
rate of formation of $\mathrm{SO}_{4(\text { aq })}^{2-}$ is $0.044 \mathrm{~mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1}$.
What is the rate of consumption of $\mathrm{S}_2 \mathrm{O}_{8(\mathrm{aq})}^{2-}$ ?
Q17.
mcq single
+1 / 0
Identify oxidant in following reaction.
$$ 3 \mathrm{H}_3 \mathrm{AsO}_{3(\mathrm{aq})}+\mathrm{BrO}_{3(\mathrm{aq})}^{-} \longrightarrow \mathrm{Br}_{(\mathrm{aq})}^{-}+3 \mathrm{H}_3 \mathrm{AsO}_{4(\mathrm{aq})} $$
Q18.
mcq single
+1 / 0
Calculate the number of moles of electrons required to convert $1.1 \mathrm{~mol} \mathrm{Cr}_2 \mathrm{O}_7^{2-}$ to $\mathrm{Cr}^{3+}$ in acidic medium.
Q19.
mcq single
+1 / 0
Which of the following statements is NOT true about lyophobic colloids?
Q20.
mcq single
+1 / 0
Alkyl chlorides on heating with metal fluorides form alkyl fluorides. This reaction is known as
Q21.
mcq single
+1 / 0
Which of the following compounds has highest boiling point?
Q22.
mcq single
+1 / 0
Identify the correct increasing order of boiling point of following compounds.
Q23.
mcq single
+1 / 0
What is the percentage by mass of oxygen in water, if percentage by mass of hydrogen is $11.1 \%$ ?
Q24.
mcq single
+1 / 0
If four different elements $\mathrm{A}, \mathrm{B}, \mathrm{C}$ and D having outer electronic configuration as,
$$ \mathrm{A}=3 \mathrm{~s}^2 3 \mathrm{p}^4, \mathrm{~B}=3 \mathrm{~s}^2 3 \mathrm{p}^5, \mathrm{C}=4 \mathrm{~s}^2 4 \mathrm{p}^4, \mathrm{D}=4 \mathrm{~s}^2 4 \mathrm{p}^5 $$
identify the element with lowest ionization enthalpy $\left(\Delta_1 \mathrm{H}_1\right)$
Q25.
mcq single
+1 / 0
Identify the element having highest value of first ionization enthalpy.
Q26.
mcq single
+1 / 0
Which of the following symbols represents heat of reaction at constant pressure?
Q27.
mcq single
+1 / 0
One mole of a perfect gas expands isothermally and reversibly from $10 \mathrm{dm}^3$ to $20 \mathrm{dm}^3$ at 300 K . Find $\Delta \mathrm{U}, \mathrm{q}$ and work done respectively in the process. $\left(\mathrm{R}=8.3 \times 10^{-3} \mathrm{~kJ} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right)$
Q28.
mcq single
+1 / 0
Calculate heat of formation of $\mathrm{SO}_2$ from following equations.
$$ \begin{aligned} & \mathrm{S}+\frac{3}{2} \mathrm{O}_2 \longrightarrow \mathrm{SO}_3, \Delta \mathrm{H}=-2 \mathrm{xkJ} \\ & \mathrm{SO}_2+\frac{1}{2} \mathrm{O}_2 \longrightarrow \mathrm{SO}_3, \Delta \mathrm{H}=-\mathrm{ykJ} \end{aligned} $$
Q29.
mcq single
+1 / 0
Calculate $\Delta \mathrm{T}_{\mathrm{b}}$ of 0.02 m solution if molal elevation constant for solvent is $0.52 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$.
Q30.
mcq single
+1 / 0
Calculate the mass of solute dissolved in $1 \mathrm{dm}^3$ water has osmotic pressure 3.0 atm at 300 K [Molar mass of solute $=41 \mathrm{~g} \mathrm{~mol}^{-1}$, $\mathrm{R}=0.082 \mathrm{dm}^3 \mathrm{~atm} \mathrm{~K} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ ]
Q31.
mcq single
+1 / 0
What is the expected value of $\Delta T_f$ for 0.2 m aqueous $\mathrm{CaCl}_2$ solution if $\Delta \mathrm{T}_{\mathrm{f}}$ for 0.2 m sucrose solution is $x \mathrm{~K}$ ?
Q32.
mcq single
+1 / 0
Identify a ligand having two donor atoms and uses pairs of electrons on both donor atoms.
Q33.
mcq single
+1 / 0
Identify anionic complex from following.
Q34.
mcq single
+1 / 0
Which of the following solvents is safe to use and considered as green solvent?
Q35.
mcq single
+1 / 0
Identify a pair of compounds that exhibits functional group isomerism from following.
Q36.
mcq single
+1 / 0
How many isomers of $\mathrm{C}_4 \mathrm{H}_{11} \mathrm{~N}$ are tertiary amines?
Q37.
mcq single
+1 / 0
$$ \text { What is IUPAC name of following compound? } $$

Q38.
mcq single
+1 / 0
Identify IUPAC name of the following compound.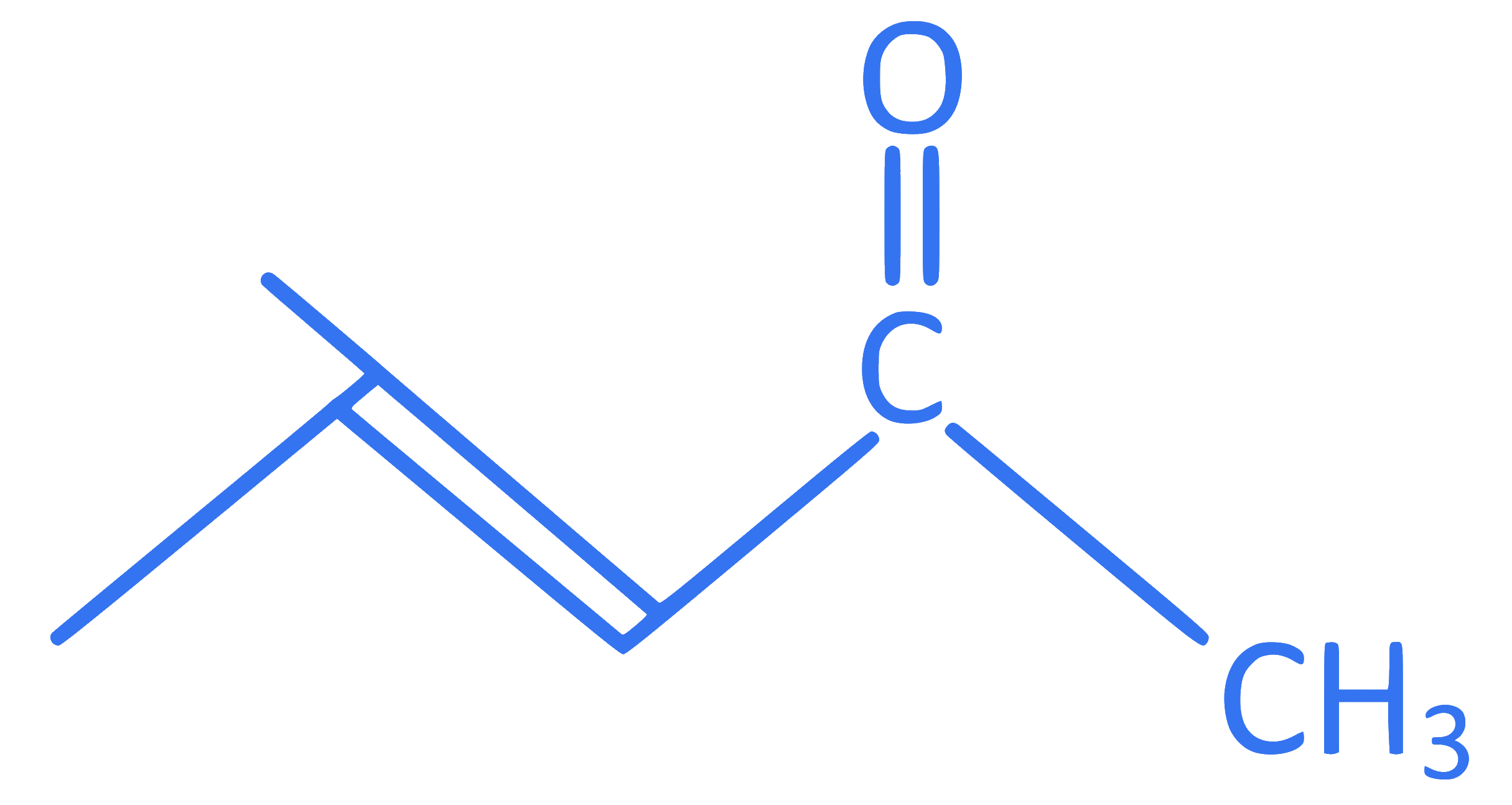

Q39.
mcq single
+1 / 0
Which alkali metal is used for heat transfer in nuclear power station?
Q40.
mcq single
+1 / 0
Which of the following is a tertiary allylic alcohol?
Q41.
mcq single
+1 / 0
Identify the major product formed when 2-chlorobutane is boiled with concentrated alcoholic KOH .
Q42.
mcq single
+1 / 0
Find increase in temperature for a gas when first its pressure and then volume both are doubled at 400 K .
Q43.
mcq single
+1 / 0
Which from following amino acids contains sulfur in it's side chain?
Q44.
mcq single
+1 / 0
Which carbon atoms of glucose in open chain structure numbered from $\mathrm{C}-1$ to $\mathrm{C}-6$ are NOT chiral?
Q45.
mcq single
+1 / 0
Calculate the number of atoms in 0.4 gram of metal if it forms fcc structure.
$$ \left[\rho \times \mathrm{a}^3=1.2 \times 10^{-22} \mathrm{~g}\right] $$
Q46.
mcq single
+1 / 0
Calculate the volume of bcc unit cell if the radius of an atom in it is 216.5 pm .
Q47.
mcq single
+1 / 0
The molar conductivity of 0.02 M HCl is $408.4 \Omega^{-1} \mathrm{~cm}^2 \mathrm{~mol}^{-1}$ at $25^{\circ} \mathrm{C}$. Calculate it's conductivity.
Q48.
mcq single
+1 / 0
Which of the following substances is NOT required in the construction of $\mathrm{H}_2-\mathrm{O}_2$ fuel cell?
Q49.
mcq single
+1 / 0
Identify functional group from following that forms pink colour when schiffs reagent is added in it.
Q50.
mcq single
+1 / 0
Identify the product formed in the following reaction.
$$ \mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CHO} \xrightarrow[\text { ii) } \mathrm{H}_3 \mathrm{O}^{+}]{\text {i) } \mathrm{LiAlH}_4} \text { product } $$
Q51.
mcq single
+1 / 0
Identify the product ' B ' in the following sequence of reactions.
Ethanenitrile $\xrightarrow{\mathrm{SnCl}_2, \mathrm{HCl}} \mathrm{A} \xrightarrow{\mathrm{H}_3 \mathrm{O}^{+}} \mathrm{B}+\mathrm{NH}_4 \mathrm{Cl}$
Q52.
mcq single
+1 / 0
Which of the following has the highest basic strength?
Q53.
mcq single
+1 / 0
Identify false statement about zinc.
Q54.
mcq single
+1 / 0
Which from following metal ions in their respective oxidation states forms colourless compounds?
Q55.
mcq single
+1 / 0
Which from following polymers is obtained by addition polymerisation method?
Q56.
mcq single
+1 / 0
Which from the following statements about nylon 2-nylon 6 is NOT correct?
Q57.
mcq single
+1 / 0
Identify a molecule having octahedral geometry as per VSEPR theory.
Q58.
mcq single
+1 / 0
Which of the following is NOT an example of diamagnetic substance?
Q59.
mcq single
+1 / 0
What is the radius of first orbit of monopositive helium ion?
Q60.
mcq multi
+1 / 0
What is the representation of element ( X ) having mass number 40 containing 22 neutrons.
Q61.
mcq single
+1 / 0
Which from the following buffers is used for precipitation of cations of IIIA group in qualitative analysis?
Q62.
mcq single
+1 / 0
The dissociation constant of weak monoacidic base is $1.8 \times 10^{-5}$. Calculate the degree of dissociation in 0.02 M solution.
Q63.
mcq single
+1 / 0
Calculate the pH of buffer solution containing 0.12 M weak acid and 0.48 M of its salt with strong base if $\mathrm{pK}_{\mathrm{a}}$ is 3.82 .
Q64.
mcq single
+1 / 0
What is rate constant of a first order reaction having half life 138.6 minute?
Q65.
mcq single
+1 / 0
What is the order of a reaction having unit of the rate constant $\mathrm{mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1}$ ?
Q66.
mcq single
+1 / 0
For the reaction,
$$ 3 \mathrm{I}_{(\mathrm{aq})}^{-}+\mathrm{S}_2 \mathrm{O}_{8(\mathrm{aq})}^{2-} \longrightarrow 2 \mathrm{SO}_{4(\mathrm{aq})}^{2-}+\mathrm{I}_{3(\mathrm{aq})}^{-} $$
rate of formation of $\mathrm{SO}_{4(\text { aq })}^{2-}$ is $0.044 \mathrm{~mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1}$.
What is the rate of consumption of $\mathrm{S}_2 \mathrm{O}_{8(\mathrm{aq})}^{2-}$ ?
Q67.
mcq single
+1 / 0
Identify oxidant in following reaction.
$$ 3 \mathrm{H}_3 \mathrm{AsO}_{3(\mathrm{aq})}+\mathrm{BrO}_{3(\mathrm{aq})}^{-} \longrightarrow \mathrm{Br}_{(\mathrm{aq})}^{-}+3 \mathrm{H}_3 \mathrm{AsO}_{4(\mathrm{aq})} $$
Q68.
mcq single
+1 / 0
Calculate the number of moles of electrons required to convert $1.1 \mathrm{~mol} \mathrm{Cr}_2 \mathrm{O}_7^{2-}$ to $\mathrm{Cr}^{3+}$ in acidic medium.
Q69.
mcq single
+1 / 0
Which of the following statements is NOT true about lyophobic colloids?
Q70.
mcq single
+1 / 0
Alkyl chlorides on heating with metal fluorides form alkyl fluorides. This reaction is known as
Q71.
mcq single
+1 / 0
Which of the following compounds has highest boiling point?
Q72.
mcq single
+1 / 0
Identify the correct increasing order of boiling point of following compounds.
Q73.
mcq single
+1 / 0
What is the percentage by mass of oxygen in water, if percentage by mass of hydrogen is $11.1 \%$ ?
Q74.
mcq single
+1 / 0
If four different elements $\mathrm{A}, \mathrm{B}, \mathrm{C}$ and D having outer electronic configuration as,
$$ \mathrm{A}=3 \mathrm{~s}^2 3 \mathrm{p}^4, \mathrm{~B}=3 \mathrm{~s}^2 3 \mathrm{p}^5, \mathrm{C}=4 \mathrm{~s}^2 4 \mathrm{p}^4, \mathrm{D}=4 \mathrm{~s}^2 4 \mathrm{p}^5 $$
identify the element with lowest ionization enthalpy $\left(\Delta_1 \mathrm{H}_1\right)$
Q75.
mcq single
+1 / 0
Identify the element having highest value of first ionization enthalpy.
Q76.
mcq single
+1 / 0
Which of the following symbols represents heat of reaction at constant pressure?
Q77.
mcq single
+1 / 0
One mole of a perfect gas expands isothermally and reversibly from $10 \mathrm{dm}^3$ to $20 \mathrm{dm}^3$ at 300 K . Find $\Delta \mathrm{U}, \mathrm{q}$ and work done respectively in the process. $\left(\mathrm{R}=8.3 \times 10^{-3} \mathrm{~kJ} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right)$
Q78.
mcq single
+1 / 0
Calculate heat of formation of $\mathrm{SO}_2$ from following equations.
$$ \begin{aligned} & \mathrm{S}+\frac{3}{2} \mathrm{O}_2 \longrightarrow \mathrm{SO}_3, \Delta \mathrm{H}=-2 \mathrm{xkJ} \\ & \mathrm{SO}_2+\frac{1}{2} \mathrm{O}_2 \longrightarrow \mathrm{SO}_3, \Delta \mathrm{H}=-\mathrm{ykJ} \end{aligned} $$
Q79.
mcq single
+1 / 0
Calculate $\Delta \mathrm{T}_{\mathrm{b}}$ of 0.02 m solution if molal elevation constant for solvent is $0.52 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$.
Q80.
mcq single
+1 / 0
Calculate the mass of solute dissolved in $1 \mathrm{dm}^3$ water has osmotic pressure 3.0 atm at 300 K [Molar mass of solute $=41 \mathrm{~g} \mathrm{~mol}^{-1}$, $\mathrm{R}=0.082 \mathrm{dm}^3 \mathrm{~atm} \mathrm{~K} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ ]
Q81.
mcq single
+1 / 0
What is the expected value of $\Delta T_f$ for 0.2 m aqueous $\mathrm{CaCl}_2$ solution if $\Delta \mathrm{T}_{\mathrm{f}}$ for 0.2 m sucrose solution is $x \mathrm{~K}$ ?
Q82.
mcq single
+1 / 0
Identify a ligand having two donor atoms and uses pairs of electrons on both donor atoms.
Q83.
mcq single
+1 / 0
Identify anionic complex from following.
Q84.
mcq single
+1 / 0
Which of the following solvents is safe to use and considered as green solvent?
Q85.
mcq single
+1 / 0
Identify a pair of compounds that exhibits functional group isomerism from following.
Q86.
mcq single
+1 / 0
How many isomers of $\mathrm{C}_4 \mathrm{H}_{11} \mathrm{~N}$ are tertiary amines?
Q87.
mcq single
+1 / 0
$$ \text { What is IUPAC name of following compound? } $$

Q88.
mcq single
+1 / 0
Identify IUPAC name of the following compound.

Q89.
mcq single
+1 / 0
Which alkali metal is used for heat transfer in nuclear power station?
Q90.
mcq single
+1 / 0
Which of the following is a tertiary allylic alcohol?
Q91.
mcq single
+1 / 0
Identify the major product formed when 2-chlorobutane is boiled with concentrated alcoholic KOH .
Q92.
mcq single
+1 / 0
Find increase in temperature for a gas when first its pressure and then volume both are doubled at 400 K .
Q93.
mcq single
+1 / 0
Which from following amino acids contains sulfur in it's side chain?
Q94.
mcq single
+1 / 0
Which carbon atoms of glucose in open chain structure numbered from $\mathrm{C}-1$ to $\mathrm{C}-6$ are NOT chiral?
Q95.
mcq single
+1 / 0
Calculate the number of atoms in 0.4 gram of metal if it forms fcc structure.
$$ \left[\rho \times \mathrm{a}^3=1.2 \times 10^{-22} \mathrm{~g}\right] $$
Q96.
mcq single
+1 / 0
Calculate the volume of bcc unit cell if the radius of an atom in it is 216.5 pm .
Q97.
mcq single
+1 / 0
The molar conductivity of 0.02 M HCl is $408.4 \Omega^{-1} \mathrm{~cm}^2 \mathrm{~mol}^{-1}$ at $25^{\circ} \mathrm{C}$. Calculate it's conductivity.
Q98.
mcq single
+1 / 0
Which of the following substances is NOT required in the construction of $\mathrm{H}_2-\mathrm{O}_2$ fuel cell?
Q99.
mcq single
+1 / 0
Identify functional group from following that forms pink colour when schiffs reagent is added in it.
Q100.
mcq single
+1 / 0
Identify the product formed in the following reaction.
$$ \mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CHO} \xrightarrow[\text { ii) } \mathrm{H}_3 \mathrm{O}^{+}]{\text {i) } \mathrm{LiAlH}_4} \text { product } $$
Physics
Physics
Q1.
mcq single
+1 / 0
A spring has length ' $L$ ' and force constant ' $K$ '. It is cut into two springs of length ' $\mathrm{L}_1$ ' and ' $\mathrm{L}_2$ ' such that $\mathrm{L}_1=\mathrm{nL}_2$ ( n is an integer). The force constant of the spring of length ' $\mathrm{L}_2$ ' is
Q2.
mcq single
+1 / 0
In Young's double slit experiment, fringe width is 1.4 mm with light of wavelength $6000 $$\mathop {\rm{A}}\limits^{\rm{o}} $. If the light of wavelength $5400 $$\mathop {\rm{A}}\limits^{\rm{o}} $ is used, with no other change in the experimental set up. The change in fringe width is
Q3.
mcq single
+1 / 0
In a single slit diffraction experiment, slit of width ' a ' and incident light of wavelength $5600 $$\mathop {\rm{A}}\limits^{\rm{o}} $, the first minimum is observed at angle $30^{\circ}$. The first secondary maximum is observed at angle $\left(\operatorname{Sin} 30^{\circ}=0.5\right)$
Q4.
mcq single
+1 / 0
A particle starts from mean position and performs S.H.M. with period 6 second. At what time its kinetic energy is $50 \%$ of total energy?
$$ \left(\cos 45^{\circ}=\frac{1}{\sqrt{2}}\right) $$
Q5.
mcq single
+1 / 0
Two bodies A and B have their moments of inertia ' I ' and ' 2 I ' respectively about their axis of rotation. If their kinetic energy of rotation are equal then angular momentum of body A to that of body $B$ will be in the ratio
Q6.
mcq single
+1 / 0
A solid sphere and a ring have equal mass and equal radius of gyration. If sphere is rotating about its diameter and ring about an axis passing through centre and perpendicular to its plane, then the ratio of radius of sphere to that of ring is $\sqrt{\frac{x}{2}}$ then the value of ' $x$ ' is
Q7.
mcq single
+1 / 0
The centre of mass of a system of particles does NOT depend on
Q8.
mcq single
+1 / 0
A coil of area $12 \mathrm{~cm}^2$ has 250 turns. Magnetic field of $0.2 \mathrm{~Wb} / \mathrm{m}^2$ is perpendicular to the plane of the coil. The field is reduced to $0.1 \mathrm{~Wb} / \mathrm{m}^2$ in 0.1 second. The magnitude of induced e.m.f. in the coil is
Q9.
mcq single
+1 / 0
The flux linked with the coil at any instant ' t ' is given by $\phi=12 t^2-60 t+275$. The magnitude of induced e.m.f. at $\mathrm{t}=3 \mathrm{~second}$ is
Q10.
mcq single
+1 / 0
Which one of the following statements is false for a particle moving in a circle with constant angular speed?
Q11.
mcq single
+1 / 0
Pressure of the gas remaining same, the temperature at which r. m. s. speed of the gas molecules is double its value at $27^{\circ} \mathrm{C}$ is
Q12.
mcq single
+1 / 0
An ideal gas is heated from $27^{\circ} \mathrm{C}$ to $627^{\circ} \mathrm{C}$ at constant pressure. If initial volume of gas is $4 \mathrm{~m}^3$, then the final volume of the gas will be
Q13.
mcq single
+1 / 0
Rate of flow of heat through a cylindrical rod is $\mathrm{H}_1$. The temperature of the ends of the rod are ' $\mathrm{T}_1$ ' and ' $\mathrm{T}_2$ '. If all the dimensions of the rod become double and the temperature difference remains the same, the rate of flow of heat becomes ' $\mathrm{H}_2$ '. Then $\mathrm{H}_2=$
Q14.
mcq single
+1 / 0
For which of the following combinations of working temperatures, the efficiency of Carnot's engine is maximum?
Q15.
mcq single
+1 / 0
A blackbody at $1227^{\circ} \mathrm{C}$ emits radiation with maximum intensity at a wavelength of $5600 $$\mathop {\rm{A}}\limits^{\rm{o}} $. If the temperature of the body is increased by $1000^{\circ} \mathrm{C}$, the maximum intensity will be at wavelength
Q16.
mcq single
+1 / 0
An ideal gas of mass ' $M$ ' is in the state ' $A$ ' goes to another state B via three different processes. If $Q_1, Q_2$ and $Q_3$ denote the heat absorbed by the gas along the paths 1,2 and 3 respectively, then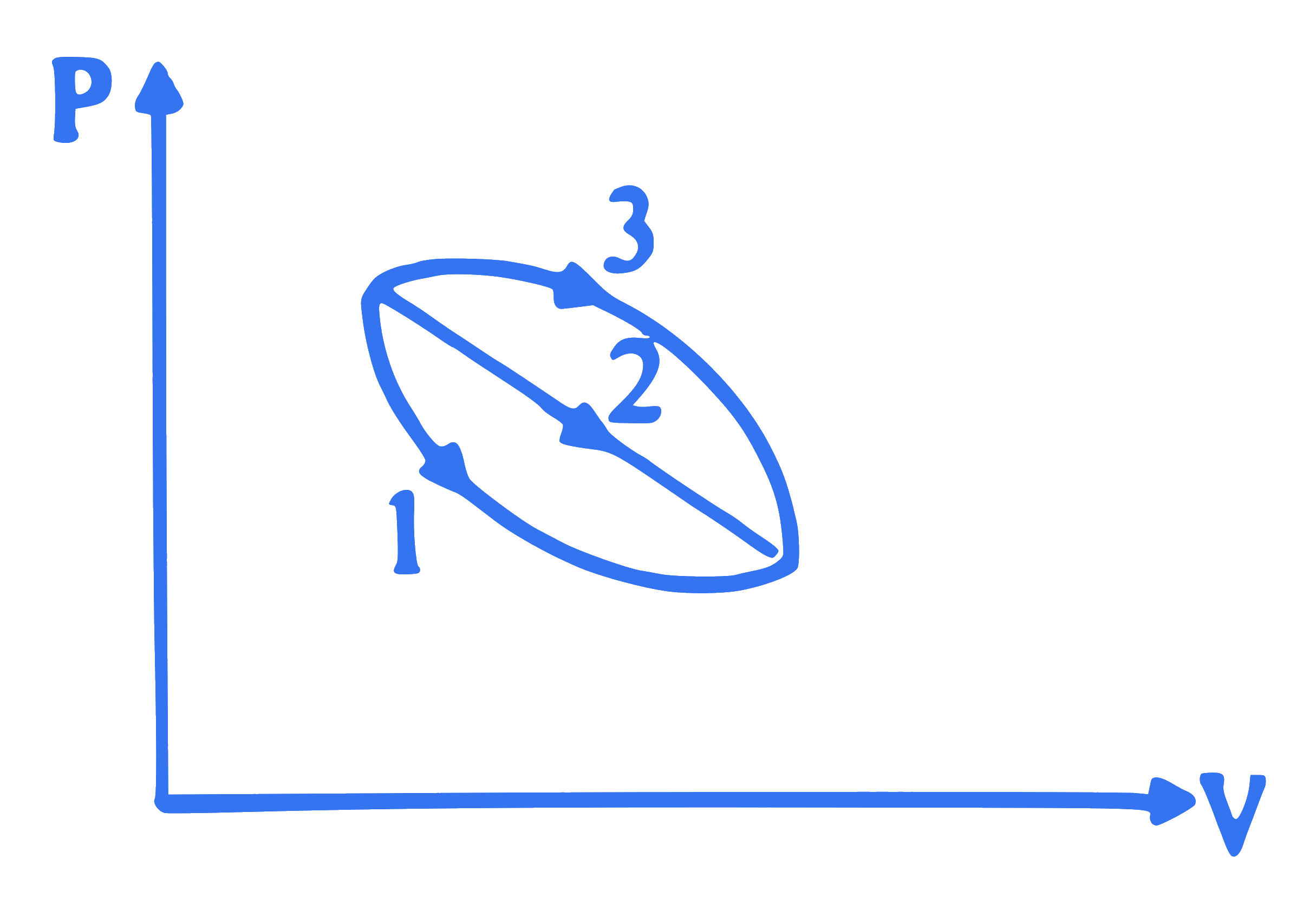

Q17.
mcq single
+1 / 0
In the Davisson Germer experiment, the velocity of electrons emitted from the electron gun can be increased by
Q18.
mcq single
+1 / 0
The work function of a photosensitive metallic surface is $h v_0$. If photons of energy (2.5) $h v_0$ fall on this surface, the electrons come out with maximum velocity ' v '. When the photon energy is increased to $7 \mathrm{~h} v_0$, the maximum velocity of photoelectrons will be
Q19.
mcq single
+1 / 0
The potentials at points A and B are $\mathrm{V}_{\mathrm{A}}$ and $\mathrm{V}_{\mathrm{B}}$ respectively for the charges $+q$ and $-q$ placed at distances ' $x$ ' each from points $A$ and $B$ as shown in figure. The distance between points $A$ and B is ' y '. The net potential $\left(\mathrm{V}_{\mathrm{A}}-\mathrm{V}_{\mathrm{B}}\right)$ is proportional to
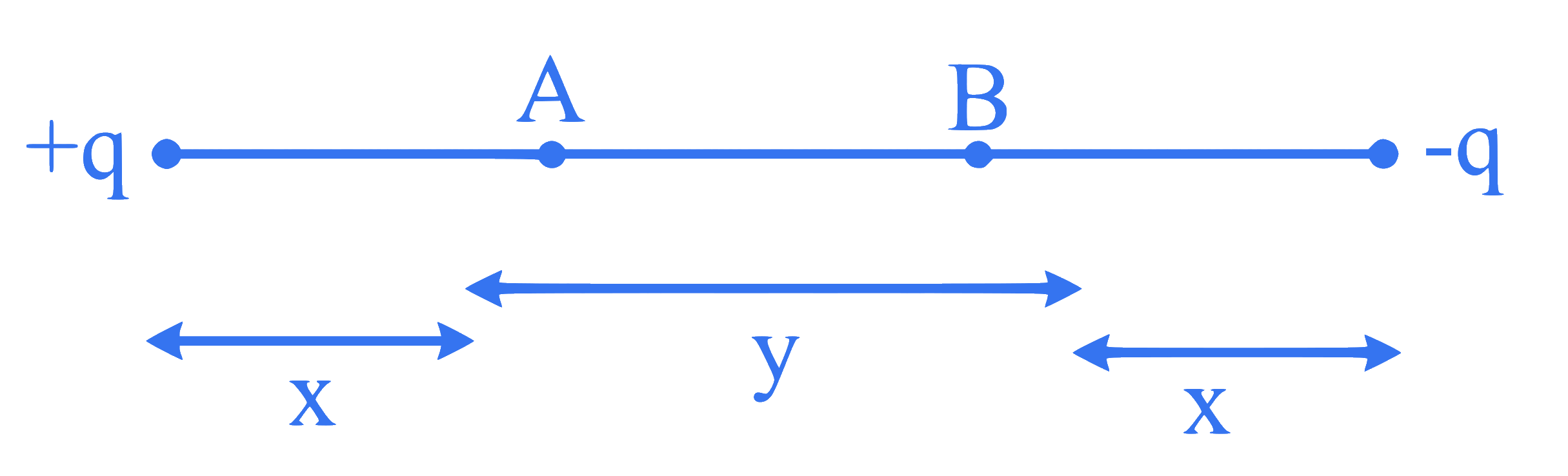

Q20.
mcq single
+1 / 0
Three charges $Q,-2 q$ and $-2 q$ are placed at the vertices of an isosceles right-angled triangle as shown in figure. The net electrostatic potential energy is zero if $Q$ is equal to
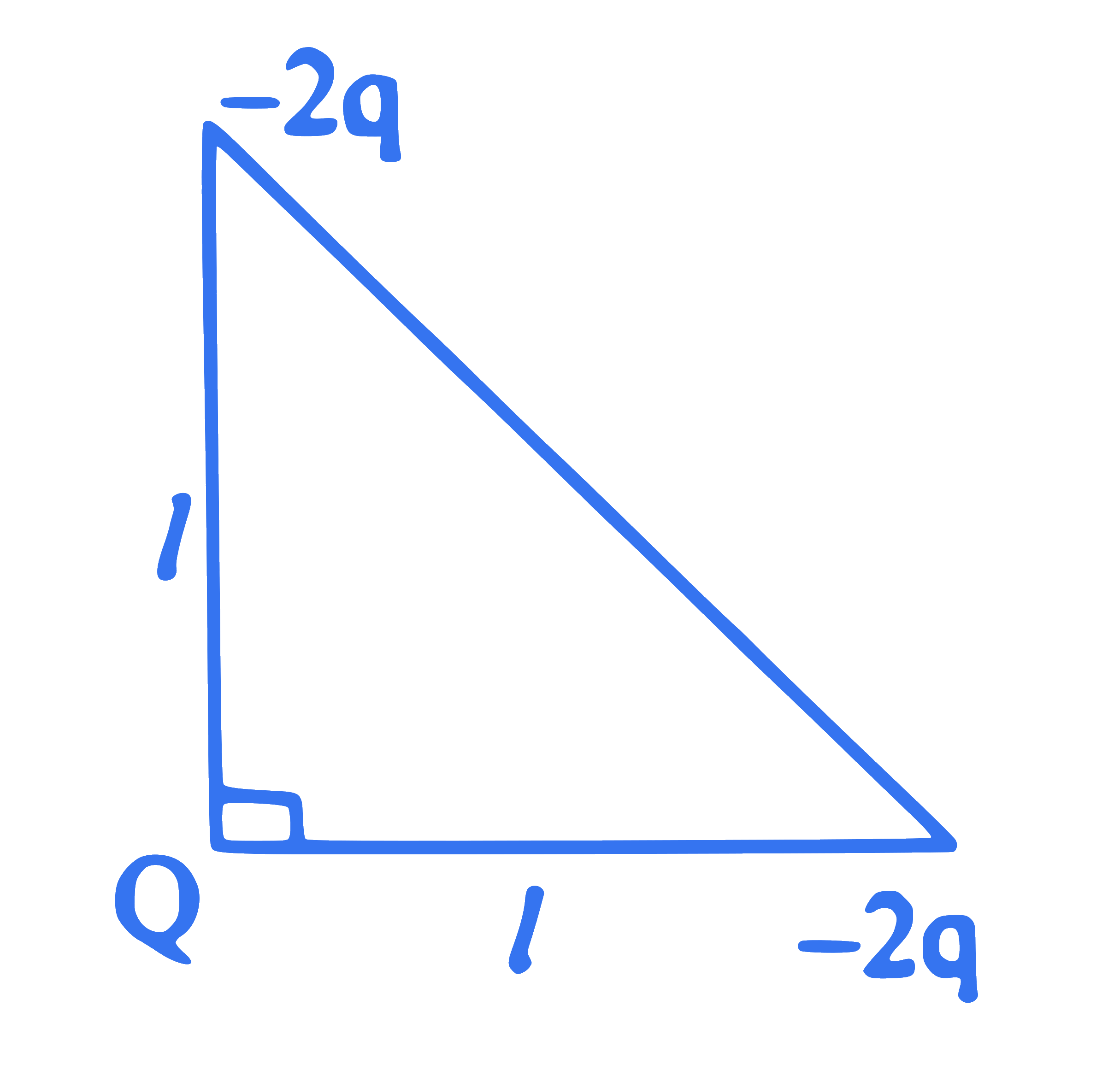

Q21.
mcq single
+1 / 0
The wrong statement out of the following statements is
Q22.
mcq single
+1 / 0
A large insulated sphere of radius ' $r$ ', charged with ' $Q$ ' units of electricity, is placed in contact with a small insulated uncharged sphere of radius ' R ' and is then separated. The charge on the smaller sphere will now be
Q23.
mcq single
+1 / 0
The period of revolution of planet A around the sun is 8 times that of $B$. The distance of $A$ from the sun is how many times greater than that of $B$ from the sun?
Q24.
mcq single
+1 / 0
A satellite of mass ' $m$ ' is orbiting the earth of radius ' $R$ ' at height ' $h$ ' from the surface of earth. The total energy of the satellite is ( $\mathrm{g}=$ acceleration due to gravity at the earth's surface)
Q25.
mcq single
+1 / 0
A plane wavefront of width ' $x$ ' is incident on an air-water interface and the corresponding refracted wavefront has a width ' y ' as shown in figure. The refractive index of air with respect to water in terms of distances ' $w$ ' and ' $z$ ' is $(\mathrm{AD}=\mathrm{w}, \mathrm{CB}=\mathrm{z})$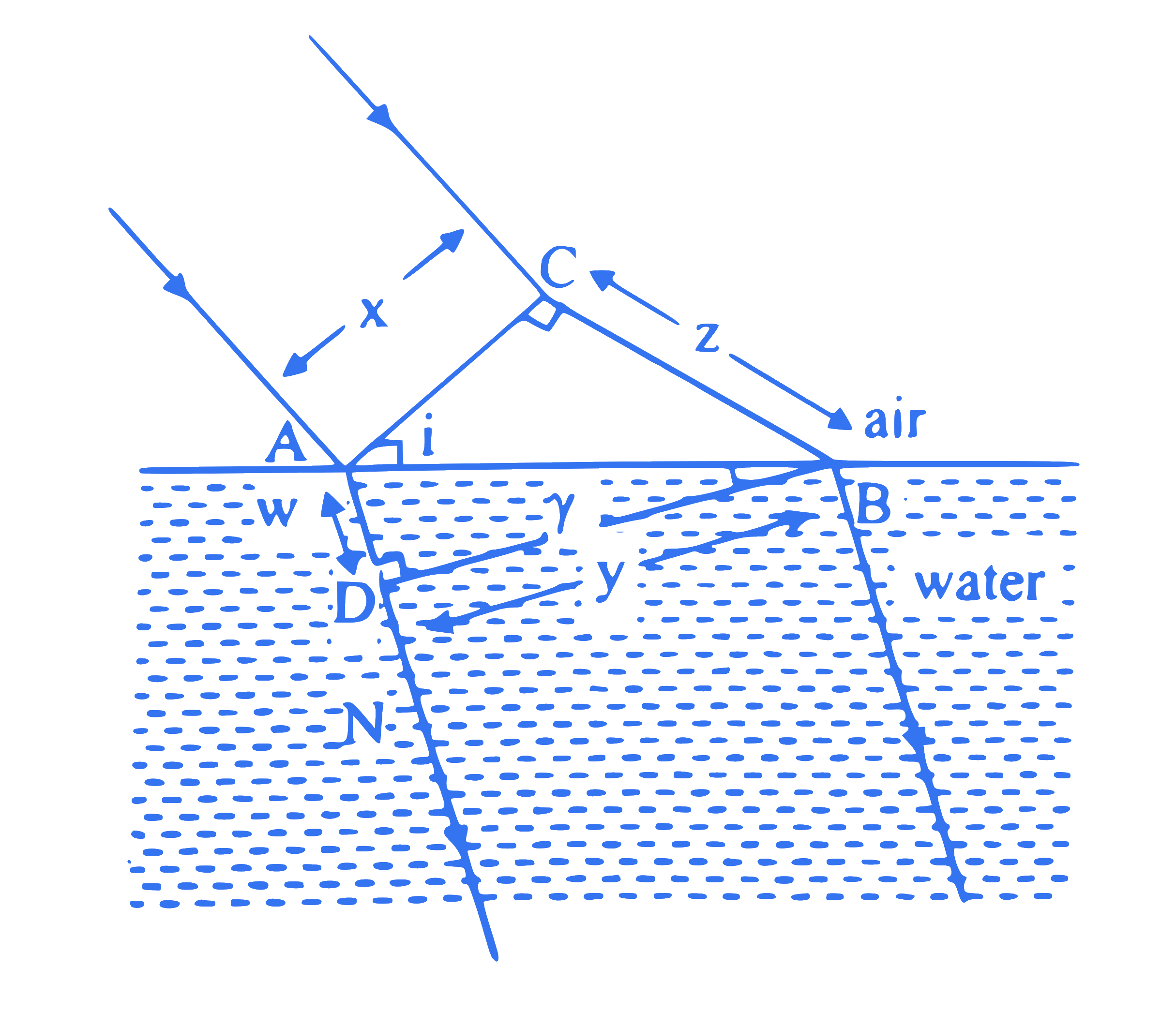

Q26.
mcq single
+1 / 0
The intermediate image formed by an objective lens of a compound microscope is
Q27.
mcq single
+1 / 0
A thin concavo-convex lens with convex face receiving incident rays has radii of curvatures 12 cm and 24 cm respectively. If refractive index of material of lens is 1.5 , then the focal length of the lens is
Q28.
mcq single
+1 / 0
Two objects A and B are separated horizontally by distance ' a '. Object B moves in a direction perpendicular to distance ' $a$ ' with velocity ' $V_1$ '. Simultaneously object A covers shortest distance with velocity ' $V$ ' and meets object $B$ in time ' $t$ '. The time ' $t$ ' is given by
Q29.
mcq single
+1 / 0
The magnitude of magnetic induction at a point on the axis at a large distance ' $r$ ' from the centre of a circular coil of ' $n$ ' turns and area ' $A$ ' carrying current ' I ' is ( $\mu_0=$ permeabilty of free space)
Q30.
mcq single
+1 / 0
To manufacture a solenoid of length ' $l$ ' and inductance ' $L$ ', the length of the thin wire required is (cross - sectional diameter of a solenoid is considerably less than length, $\mu_0=$ permeability of free space)
Q31.
mcq single
+1 / 0
A circular coil carrying current ' $I$ ' has radius ' $R$ ' and magnetic field at the centre is ' $B$ '. At what distance from the centre along the axis of the same coil, the magnetic field will be $\frac{\mathrm{B}}{8}$ ?
Q32.
mcq single
+1 / 0
A soap bubble of radius $R$ is surrounded by another soap bubble of radius $2 R$ as shown in figure. The excess pressure inside the smaller soap bubble will be ( $T=$ surface tension of soap solution)

Q33.
mcq single
+1 / 0
Water rises up to height ' $x$ ' in a capillary tube immersed vertically in water. When the whole arrangement is taken to a depth 'd' in a mine, the water level rises height ' $Y$ '. If ' $R$ ' is the radius of earth then the ratio $(\mathrm{Y} / \mathrm{x})$ is
Q34.
mcq single
+1 / 0
At two points on a horizontal tube of varying cross-section the radii are 1 cm and $\mathbf{0 . 4 ~ c m}$, velocities of fluid are $\mathrm{V}_1, \mathrm{~V}_2$ and pressure difference ( $P_1-P_2$ ) between these points is 4.9 cm of water. The value of $\sqrt{V_2^2-V_1^2}$ is ( $\mathrm{g}=$ acceleration due to gravity $\mathrm{g}=980 \mathrm{~cm} / \mathrm{s}^2$ )
Q35.
mcq single
+1 / 0
If $R_1$ and $R_2$ are the radii of the atomic nuclei of mass numbers 27 and 125 respectively, then the ratio $R_2: R_1$ is
Q36.
mcq single
+1 / 0
Radioactive materials A and B have decay constants ' $9 \lambda$ ' and ' $\lambda$ ' respectively. Initially they have same number of nuclei. The ratio of number of nuclei of material ' $A$ ' to that of ' $B$ ' will be $\left(\frac{1}{\mathrm{e}}\right)$ after time ' $t$ '. So ' $t$ ' is equal to
Q37.
mcq single
+1 / 0
A simple harmonic progressive wave is given by equation $y=\operatorname{asin} 2 \pi\left(n t-\frac{x}{\lambda}\right)$. If the wave velocity is equal to $\frac{1}{4} \times$ (maximum particle velocity), then the wavelength ' $\lambda$ ' is (Given $\rightarrow \mathrm{a}=$ amplitude, $\mathrm{n}=$ frequency, $\mathrm{t}=$ time, $\mathrm{y}=$ displacement, $\mathrm{x}=$ distance )
Q38.
mcq single
+1 / 0
The third overtone of a closed pipe of length ' $\mathrm{L}_{\mathrm{c}}$ ' has the same frequency as the third overtone of an open pipe of length ' $L_0$ '. The ratio ' $\mathrm{L}_{\mathrm{c}}$ ': ' $\mathrm{L}_0$ ' is equal to (Neglecting end correction)
Q39.
mcq single
+1 / 0
Two sounding sources send waves at certain temperature in air of wavelength 60 cm and 60.6 cm respectively. The frequency of sources differ by 5 Hz . The velocity of sound in air at same temperature is
Q40.
mcq single
+1 / 0
Three tuning forks $\mathrm{A}, \mathrm{B}$ and C have respective frequencies $\mathrm{n}_1, \mathrm{n}_2$ and $\mathrm{n}_3$ related as $\mathrm{n}_1=1.03 \mathrm{n}_2$ and $n_3=0.99 n_2$. When $A$ and $C$ are sounded together 4 beats are heard per second. The frequencies of fork B and C are respectively
Q41.
mcq single
+1 / 0
A magnet having a magnetic dipole moment ' M is placed in two magnetic fields ' $\mathrm{B}_1$ ' and ' $\mathrm{B}_2$ ' respectively. If it is displaced slightly from the equilibrium position, it oscillates 60 times in 20 second in field ' $\mathrm{B}_1$ ' and 60 times in 30 second in field ' $B_2$ '. The ratio of field ' $B_1$ ' to that of ' $\mathrm{B}_2{ }^{\prime}$ is
Q42.
mcq single
+1 / 0
Rods ' $A$ ', ' $B$ ' and ' $C$ ' are made of a paramagnetic, a ferromagnetic and a diamagnetic substance respectively. A magnet is brought close to them, it will
Q43.
mcq single
+1 / 0
A galvanometer of resistance ' $G$ ' is converted into an ammeter of resistance $\frac{\mathrm{G}}{40}$, by connecting a shunt ' $S$ ' to it. The part of main current passing through the galvanometer is
Q44.
mcq single
+1 / 0
$$ \text { In the following figure, the current } \mathrm{I} \text { is equal to } $$
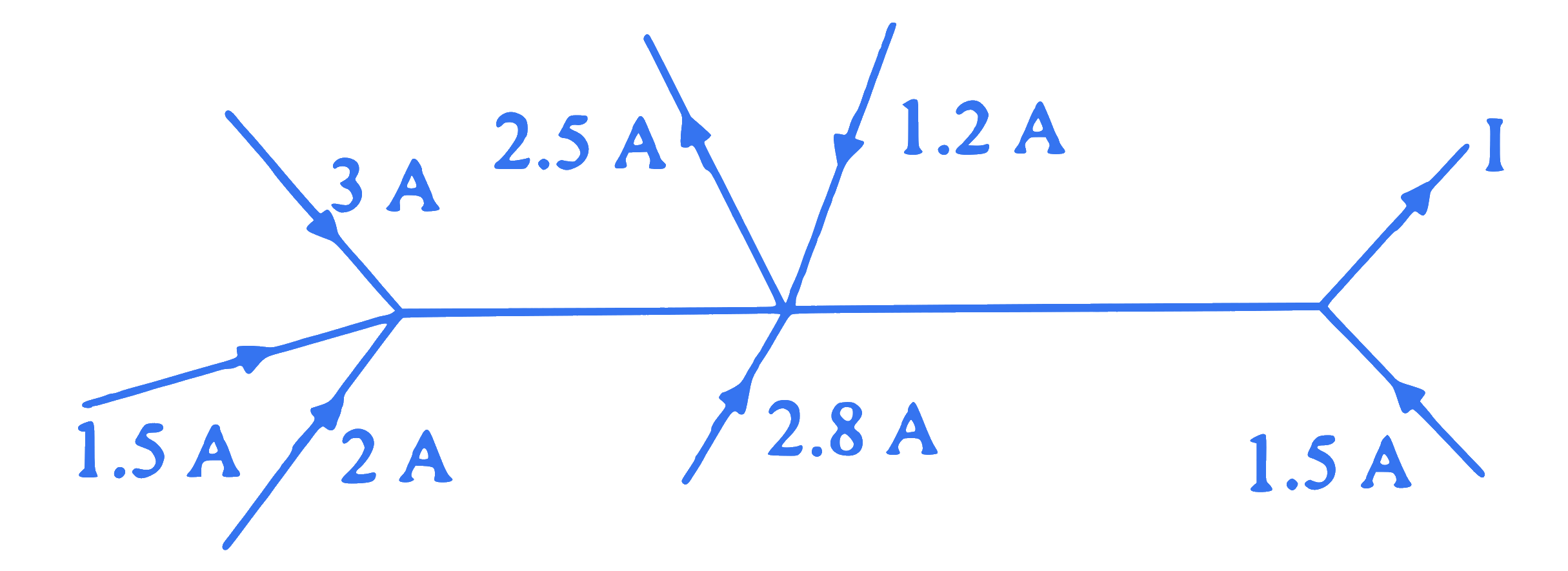
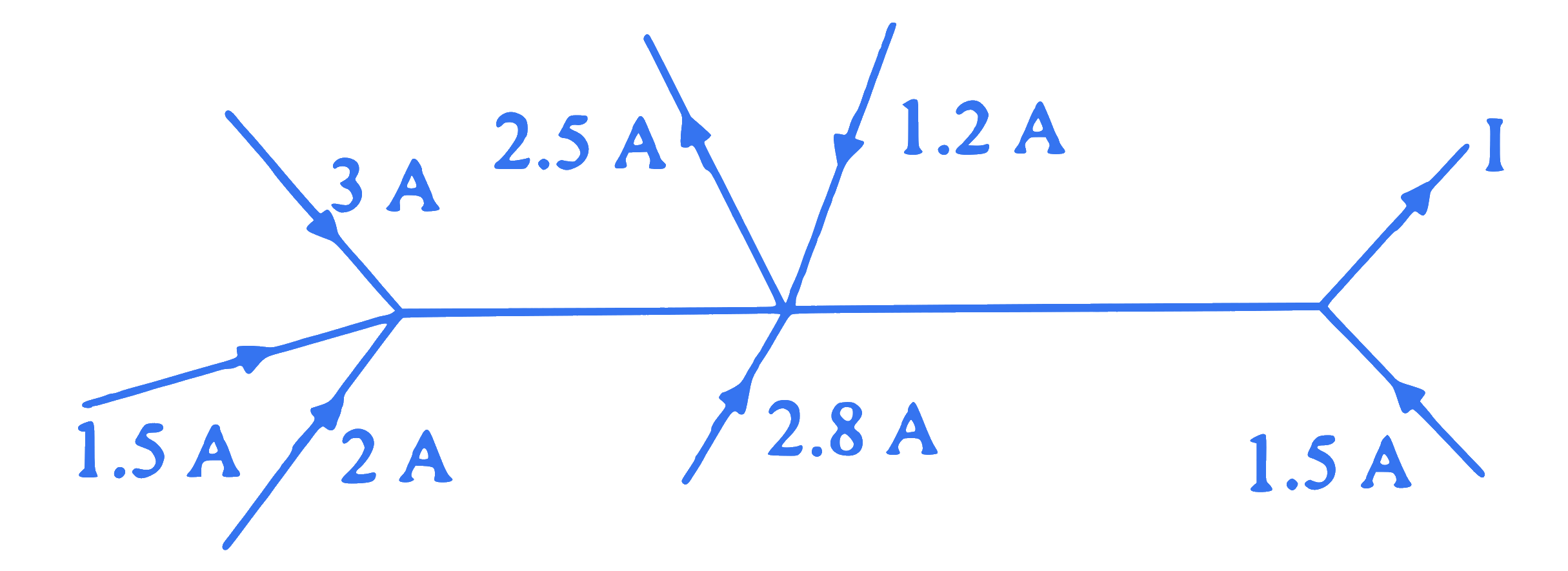
Q45.
mcq single
+1 / 0
By increasing the temperature, the electrical conductivity of a conductor and a semiconductor,
Q46.
mcq single
+1 / 0
To get an output $\mathrm{Y}=1$ in the given logic circuit, the correct choice for the input is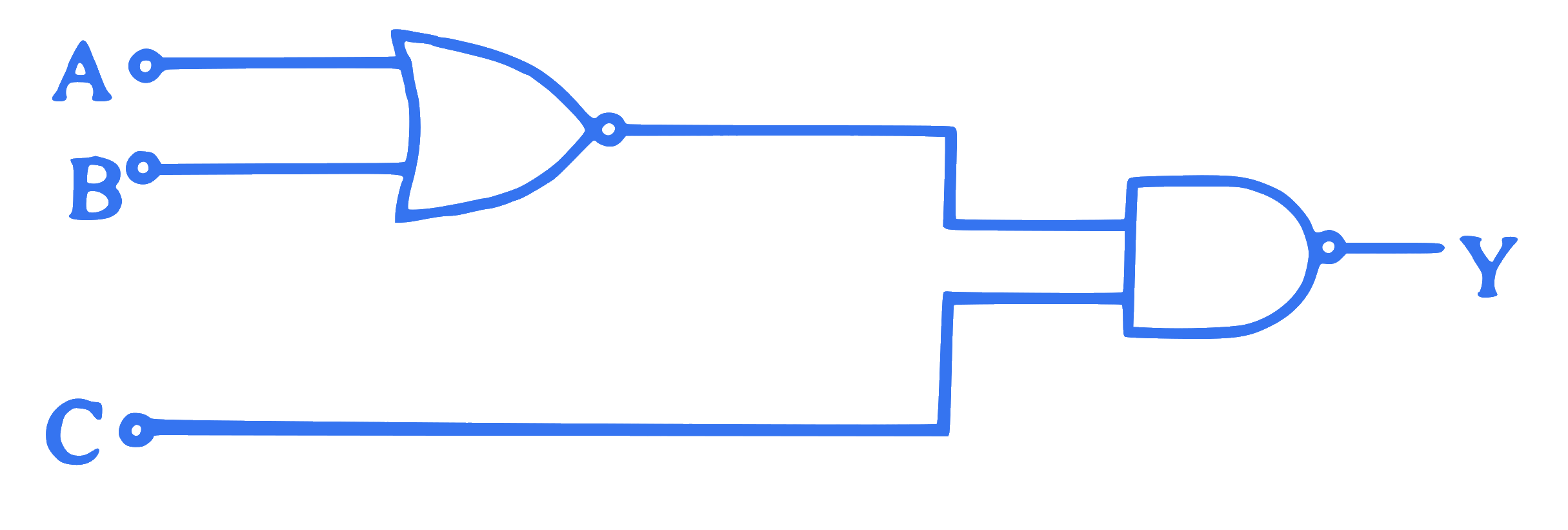

Q47.
mcq single
+1 / 0
For a transistor, the current amplification factor ' $\alpha$ ' is 0.8 . The transistor is then connected in common emitter configuration. The change in the collector current when the base current changes by 6 mA is
Q48.
mcq single
+1 / 0
A resistor of $400 \Omega$, an inductance of 0.4 H are in series with an a.c. source of e.m.f. $\mathrm{E}=200 \sqrt{2} \sin (1000 \mathrm{t})$. The power factor of the circuit is
Q49.
mcq single
+1 / 0
In the series LCR circuit shown in figure, the impedance is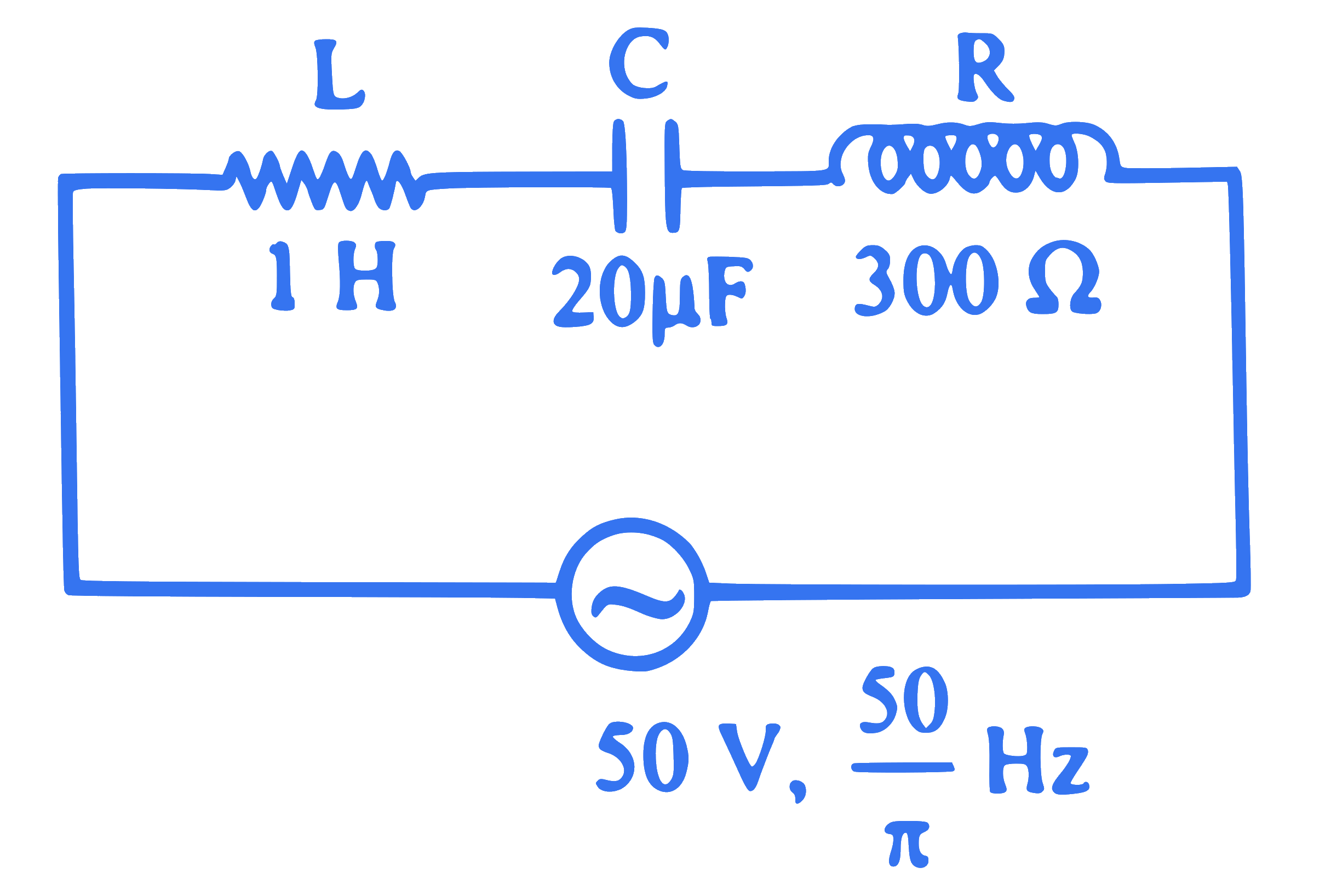

Q50.
mcq single
+1 / 0
An alternating current is given by $\mathrm{I}=100 \sin (50 \pi t)$. How many times will the current become zero in one second?
Q51.
mcq single
+1 / 0
A spring has length ' $L$ ' and force constant ' $K$ '. It is cut into two springs of length ' $\mathrm{L}_1$ ' and ' $\mathrm{L}_2$ ' such that $\mathrm{L}_1=\mathrm{nL}_2$ ( n is an integer). The force constant of the spring of length ' $\mathrm{L}_2$ ' is
Q52.
mcq single
+1 / 0
In Young's double slit experiment, fringe width is 1.4 mm with light of wavelength $6000 $$\mathop {\rm{A}}\limits^{\rm{o}} $. If the light of wavelength $5400 $$\mathop {\rm{A}}\limits^{\rm{o}} $ is used, with no other change in the experimental set up. The change in fringe width is
Q53.
mcq single
+1 / 0
In a single slit diffraction experiment, slit of width ' a ' and incident light of wavelength $5600 $$\mathop {\rm{A}}\limits^{\rm{o}} $, the first minimum is observed at angle $30^{\circ}$. The first secondary maximum is observed at angle $\left(\operatorname{Sin} 30^{\circ}=0.5\right)$
Q54.
mcq single
+1 / 0
A particle starts from mean position and performs S.H.M. with period 6 second. At what time its kinetic energy is $50 \%$ of total energy?
$$ \left(\cos 45^{\circ}=\frac{1}{\sqrt{2}}\right) $$
Q55.
mcq single
+1 / 0
Two bodies A and B have their moments of inertia ' I ' and ' 2 I ' respectively about their axis of rotation. If their kinetic energy of rotation are equal then angular momentum of body A to that of body $B$ will be in the ratio
Q56.
mcq single
+1 / 0
A solid sphere and a ring have equal mass and equal radius of gyration. If sphere is rotating about its diameter and ring about an axis passing through centre and perpendicular to its plane, then the ratio of radius of sphere to that of ring is $\sqrt{\frac{x}{2}}$ then the value of ' $x$ ' is
Q57.
mcq single
+1 / 0
The centre of mass of a system of particles does NOT depend on
Q58.
mcq single
+1 / 0
A coil of area $12 \mathrm{~cm}^2$ has 250 turns. Magnetic field of $0.2 \mathrm{~Wb} / \mathrm{m}^2$ is perpendicular to the plane of the coil. The field is reduced to $0.1 \mathrm{~Wb} / \mathrm{m}^2$ in 0.1 second. The magnitude of induced e.m.f. in the coil is
Q59.
mcq single
+1 / 0
The flux linked with the coil at any instant ' t ' is given by $\phi=12 t^2-60 t+275$. The magnitude of induced e.m.f. at $\mathrm{t}=3 \mathrm{~second}$ is
Q60.
mcq single
+1 / 0
Which one of the following statements is false for a particle moving in a circle with constant angular speed?
Q61.
mcq single
+1 / 0
Pressure of the gas remaining same, the temperature at which r. m. s. speed of the gas molecules is double its value at $27^{\circ} \mathrm{C}$ is
Q62.
mcq single
+1 / 0
An ideal gas is heated from $27^{\circ} \mathrm{C}$ to $627^{\circ} \mathrm{C}$ at constant pressure. If initial volume of gas is $4 \mathrm{~m}^3$, then the final volume of the gas will be
Q63.
mcq single
+1 / 0
Rate of flow of heat through a cylindrical rod is $\mathrm{H}_1$. The temperature of the ends of the rod are ' $\mathrm{T}_1$ ' and ' $\mathrm{T}_2$ '. If all the dimensions of the rod become double and the temperature difference remains the same, the rate of flow of heat becomes ' $\mathrm{H}_2$ '. Then $\mathrm{H}_2=$
Q64.
mcq single
+1 / 0
For which of the following combinations of working temperatures, the efficiency of Carnot's engine is maximum?
Q65.
mcq single
+1 / 0
A blackbody at $1227^{\circ} \mathrm{C}$ emits radiation with maximum intensity at a wavelength of $5600 $$\mathop {\rm{A}}\limits^{\rm{o}} $. If the temperature of the body is increased by $1000^{\circ} \mathrm{C}$, the maximum intensity will be at wavelength
Q66.
mcq single
+1 / 0
An ideal gas of mass ' $M$ ' is in the state ' $A$ ' goes to another state B via three different processes. If $Q_1, Q_2$ and $Q_3$ denote the heat absorbed by the gas along the paths 1,2 and 3 respectively, then

Q67.
mcq single
+1 / 0
In the Davisson Germer experiment, the velocity of electrons emitted from the electron gun can be increased by
Q68.
mcq single
+1 / 0
The work function of a photosensitive metallic surface is $h v_0$. If photons of energy (2.5) $h v_0$ fall on this surface, the electrons come out with maximum velocity ' v '. When the photon energy is increased to $7 \mathrm{~h} v_0$, the maximum velocity of photoelectrons will be
Q69.
mcq single
+1 / 0
The potentials at points A and B are $\mathrm{V}_{\mathrm{A}}$ and $\mathrm{V}_{\mathrm{B}}$ respectively for the charges $+q$ and $-q$ placed at distances ' $x$ ' each from points $A$ and $B$ as shown in figure. The distance between points $A$ and B is ' y '. The net potential $\left(\mathrm{V}_{\mathrm{A}}-\mathrm{V}_{\mathrm{B}}\right)$ is proportional to


Q70.
mcq single
+1 / 0
Three charges $Q,-2 q$ and $-2 q$ are placed at the vertices of an isosceles right-angled triangle as shown in figure. The net electrostatic potential energy is zero if $Q$ is equal to


Q71.
mcq single
+1 / 0
The wrong statement out of the following statements is
Q72.
mcq single
+1 / 0
A large insulated sphere of radius ' $r$ ', charged with ' $Q$ ' units of electricity, is placed in contact with a small insulated uncharged sphere of radius ' R ' and is then separated. The charge on the smaller sphere will now be
Q73.
mcq single
+1 / 0
The period of revolution of planet A around the sun is 8 times that of $B$. The distance of $A$ from the sun is how many times greater than that of $B$ from the sun?
Q74.
mcq single
+1 / 0
A satellite of mass ' $m$ ' is orbiting the earth of radius ' $R$ ' at height ' $h$ ' from the surface of earth. The total energy of the satellite is ( $\mathrm{g}=$ acceleration due to gravity at the earth's surface)
Q75.
mcq single
+1 / 0
A plane wavefront of width ' $x$ ' is incident on an air-water interface and the corresponding refracted wavefront has a width ' y ' as shown in figure. The refractive index of air with respect to water in terms of distances ' $w$ ' and ' $z$ ' is $(\mathrm{AD}=\mathrm{w}, \mathrm{CB}=\mathrm{z})$

Q76.
mcq single
+1 / 0
The intermediate image formed by an objective lens of a compound microscope is
Q77.
mcq single
+1 / 0
A thin concavo-convex lens with convex face receiving incident rays has radii of curvatures 12 cm and 24 cm respectively. If refractive index of material of lens is 1.5 , then the focal length of the lens is
Q78.
mcq single
+1 / 0
Two objects A and B are separated horizontally by distance ' a '. Object B moves in a direction perpendicular to distance ' $a$ ' with velocity ' $V_1$ '. Simultaneously object A covers shortest distance with velocity ' $V$ ' and meets object $B$ in time ' $t$ '. The time ' $t$ ' is given by
Q79.
mcq single
+1 / 0
The magnitude of magnetic induction at a point on the axis at a large distance ' $r$ ' from the centre of a circular coil of ' $n$ ' turns and area ' $A$ ' carrying current ' I ' is ( $\mu_0=$ permeabilty of free space)
Q80.
mcq single
+1 / 0
To manufacture a solenoid of length ' $l$ ' and inductance ' $L$ ', the length of the thin wire required is (cross - sectional diameter of a solenoid is considerably less than length, $\mu_0=$ permeability of free space)
Q81.
mcq single
+1 / 0
A circular coil carrying current ' $I$ ' has radius ' $R$ ' and magnetic field at the centre is ' $B$ '. At what distance from the centre along the axis of the same coil, the magnetic field will be $\frac{\mathrm{B}}{8}$ ?
Q82.
mcq single
+1 / 0
A soap bubble of radius $R$ is surrounded by another soap bubble of radius $2 R$ as shown in figure. The excess pressure inside the smaller soap bubble will be ( $T=$ surface tension of soap solution)

Q83.
mcq single
+1 / 0
Water rises up to height ' $x$ ' in a capillary tube immersed vertically in water. When the whole arrangement is taken to a depth 'd' in a mine, the water level rises height ' $Y$ '. If ' $R$ ' is the radius of earth then the ratio $(\mathrm{Y} / \mathrm{x})$ is
Q84.
mcq single
+1 / 0
At two points on a horizontal tube of varying cross-section the radii are 1 cm and $\mathbf{0 . 4 ~ c m}$, velocities of fluid are $\mathrm{V}_1, \mathrm{~V}_2$ and pressure difference ( $P_1-P_2$ ) between these points is 4.9 cm of water. The value of $\sqrt{V_2^2-V_1^2}$ is ( $\mathrm{g}=$ acceleration due to gravity $\mathrm{g}=980 \mathrm{~cm} / \mathrm{s}^2$ )
Q85.
mcq single
+1 / 0
If $R_1$ and $R_2$ are the radii of the atomic nuclei of mass numbers 27 and 125 respectively, then the ratio $R_2: R_1$ is
Q86.
mcq single
+1 / 0
Radioactive materials A and B have decay constants ' $9 \lambda$ ' and ' $\lambda$ ' respectively. Initially they have same number of nuclei. The ratio of number of nuclei of material ' $A$ ' to that of ' $B$ ' will be $\left(\frac{1}{\mathrm{e}}\right)$ after time ' $t$ '. So ' $t$ ' is equal to
Q87.
mcq single
+1 / 0
A simple harmonic progressive wave is given by equation $y=\operatorname{asin} 2 \pi\left(n t-\frac{x}{\lambda}\right)$. If the wave velocity is equal to $\frac{1}{4} \times$ (maximum particle velocity), then the wavelength ' $\lambda$ ' is (Given $\rightarrow \mathrm{a}=$ amplitude, $\mathrm{n}=$ frequency, $\mathrm{t}=$ time, $\mathrm{y}=$ displacement, $\mathrm{x}=$ distance )
Q88.
mcq single
+1 / 0
The third overtone of a closed pipe of length ' $\mathrm{L}_{\mathrm{c}}$ ' has the same frequency as the third overtone of an open pipe of length ' $L_0$ '. The ratio ' $\mathrm{L}_{\mathrm{c}}$ ': ' $\mathrm{L}_0$ ' is equal to (Neglecting end correction)
Q89.
mcq single
+1 / 0
Two sounding sources send waves at certain temperature in air of wavelength 60 cm and 60.6 cm respectively. The frequency of sources differ by 5 Hz . The velocity of sound in air at same temperature is
Q90.
mcq single
+1 / 0
Three tuning forks $\mathrm{A}, \mathrm{B}$ and C have respective frequencies $\mathrm{n}_1, \mathrm{n}_2$ and $\mathrm{n}_3$ related as $\mathrm{n}_1=1.03 \mathrm{n}_2$ and $n_3=0.99 n_2$. When $A$ and $C$ are sounded together 4 beats are heard per second. The frequencies of fork B and C are respectively
Q91.
mcq single
+1 / 0
A magnet having a magnetic dipole moment ' M is placed in two magnetic fields ' $\mathrm{B}_1$ ' and ' $\mathrm{B}_2$ ' respectively. If it is displaced slightly from the equilibrium position, it oscillates 60 times in 20 second in field ' $\mathrm{B}_1$ ' and 60 times in 30 second in field ' $B_2$ '. The ratio of field ' $B_1$ ' to that of ' $\mathrm{B}_2{ }^{\prime}$ is
Q92.
mcq single
+1 / 0
Rods ' $A$ ', ' $B$ ' and ' $C$ ' are made of a paramagnetic, a ferromagnetic and a diamagnetic substance respectively. A magnet is brought close to them, it will
Q93.
mcq single
+1 / 0
A galvanometer of resistance ' $G$ ' is converted into an ammeter of resistance $\frac{\mathrm{G}}{40}$, by connecting a shunt ' $S$ ' to it. The part of main current passing through the galvanometer is
Q94.
mcq single
+1 / 0
$$ \text { In the following figure, the current } \mathrm{I} \text { is equal to } $$
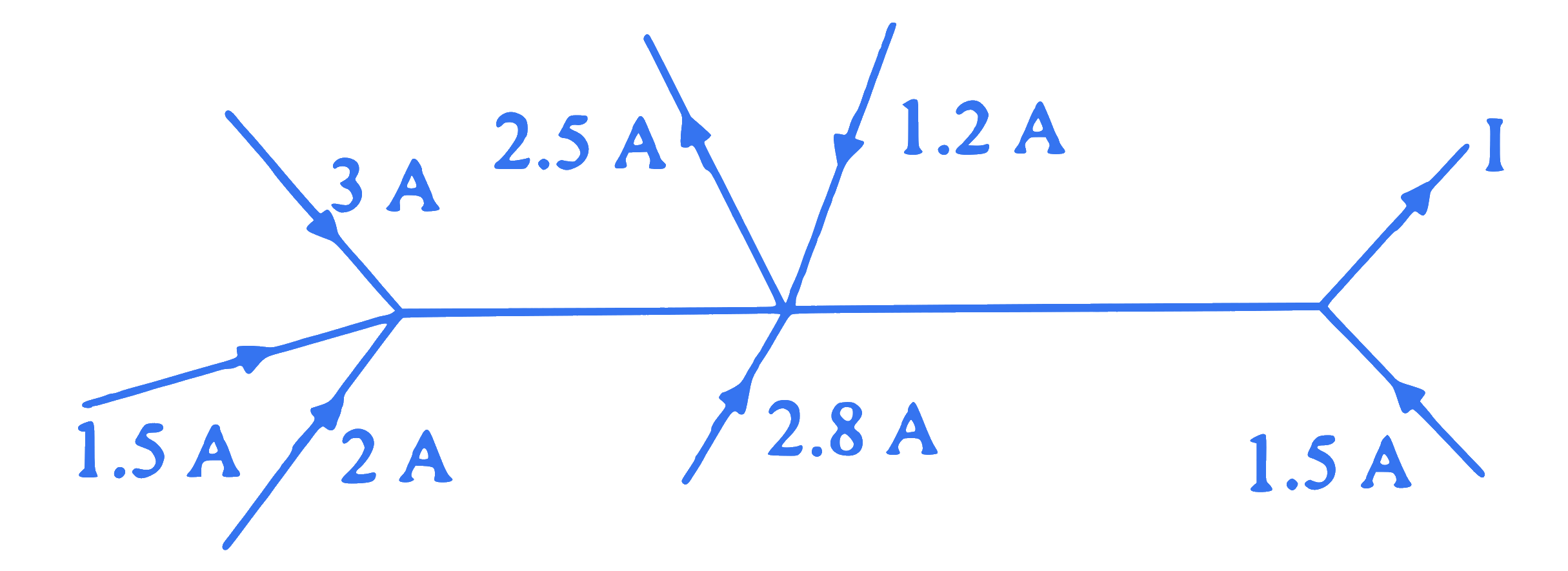
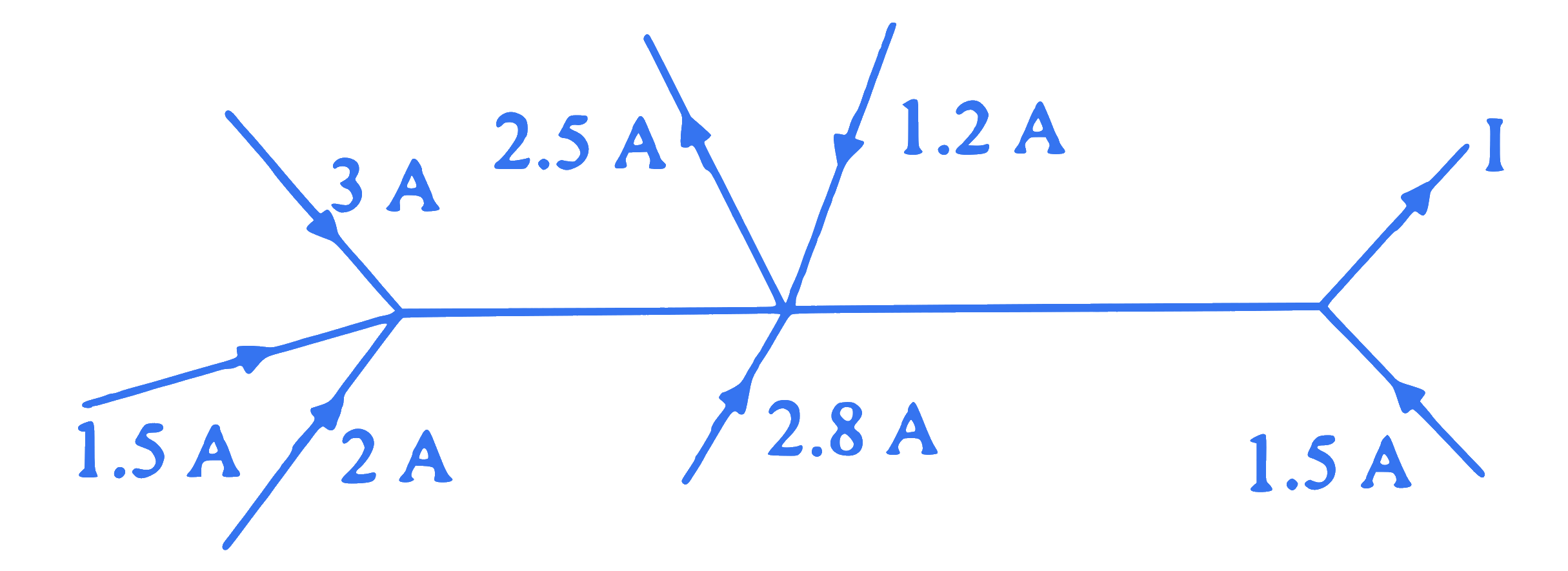
Q95.
mcq single
+1 / 0
By increasing the temperature, the electrical conductivity of a conductor and a semiconductor,
Q96.
mcq single
+1 / 0
To get an output $\mathrm{Y}=1$ in the given logic circuit, the correct choice for the input is

Q97.
mcq single
+1 / 0
For a transistor, the current amplification factor ' $\alpha$ ' is 0.8 . The transistor is then connected in common emitter configuration. The change in the collector current when the base current changes by 6 mA is
Q98.
mcq single
+1 / 0
A resistor of $400 \Omega$, an inductance of 0.4 H are in series with an a.c. source of e.m.f. $\mathrm{E}=200 \sqrt{2} \sin (1000 \mathrm{t})$. The power factor of the circuit is
Q99.
mcq single
+1 / 0
In the series LCR circuit shown in figure, the impedance is

Q100.
mcq single
+1 / 0
An alternating current is given by $\mathrm{I}=100 \sin (50 \pi t)$. How many times will the current become zero in one second?
Biology
Biology
Q1.
mcq single
+1 / 0
Which of the following microbes is used in the production of yoghurt?
Q2.
mcq single
+1 / 0
Protozoans like Nosema locustae is used for controlling the target pests like ________
Q3.
mcq single
+1 / 0
All of the raw materials that are extracted from an ecosystem are called ________ services.
Q4.
mcq single
+1 / 0
Phosphorus cycle does not pass through __________ .
Q5.
mcq single
+1 / 0
The pyramid of biomass is inverted in________ ecosystem.
Q6.
mcq single
+1 / 0
Observe the given figure of Morgan's experiment for yellow body and white eyed female Drosophila crossed with brown body and red eyed male Drosophila. What will be the percentage of parental type in $F_2$ generation?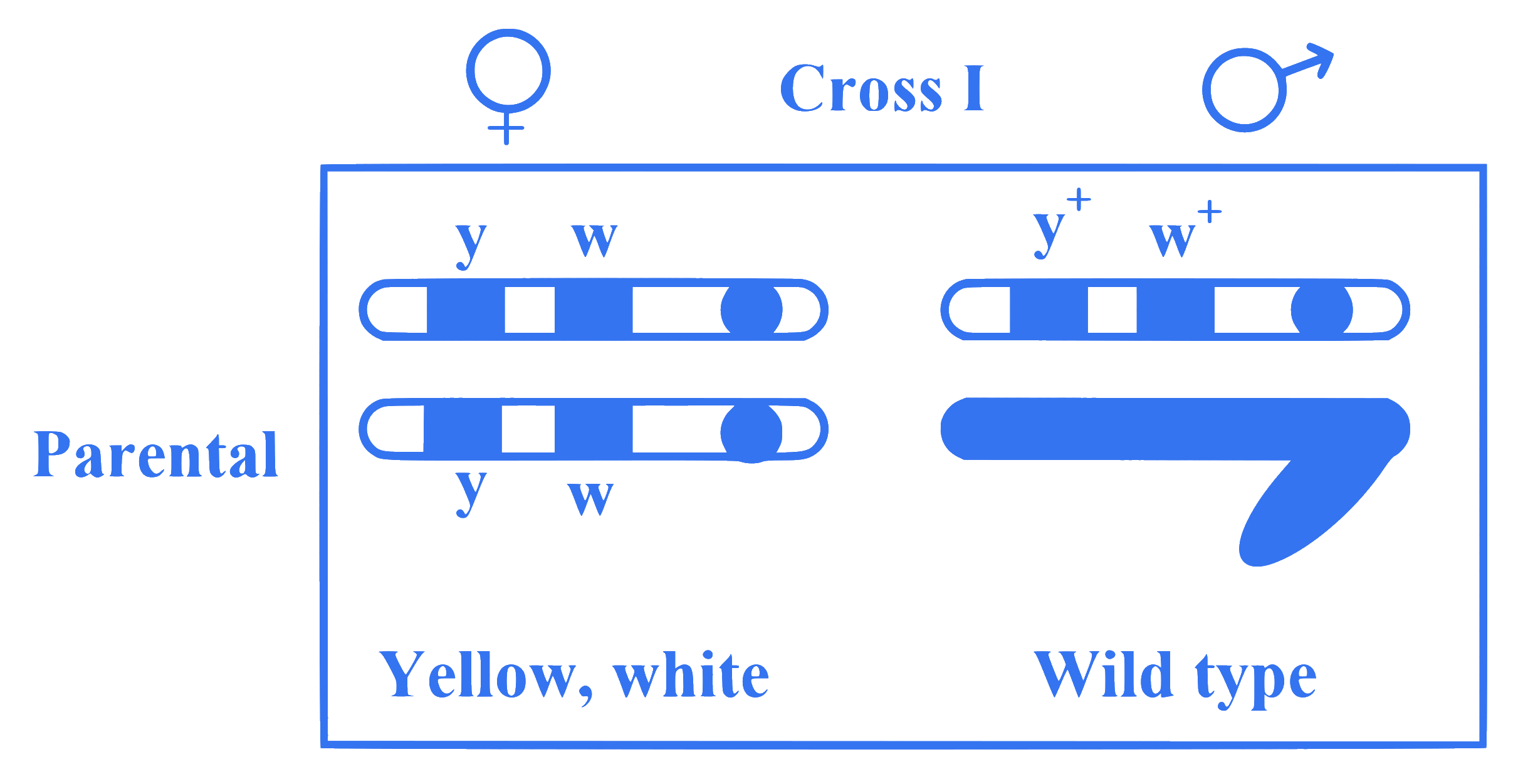
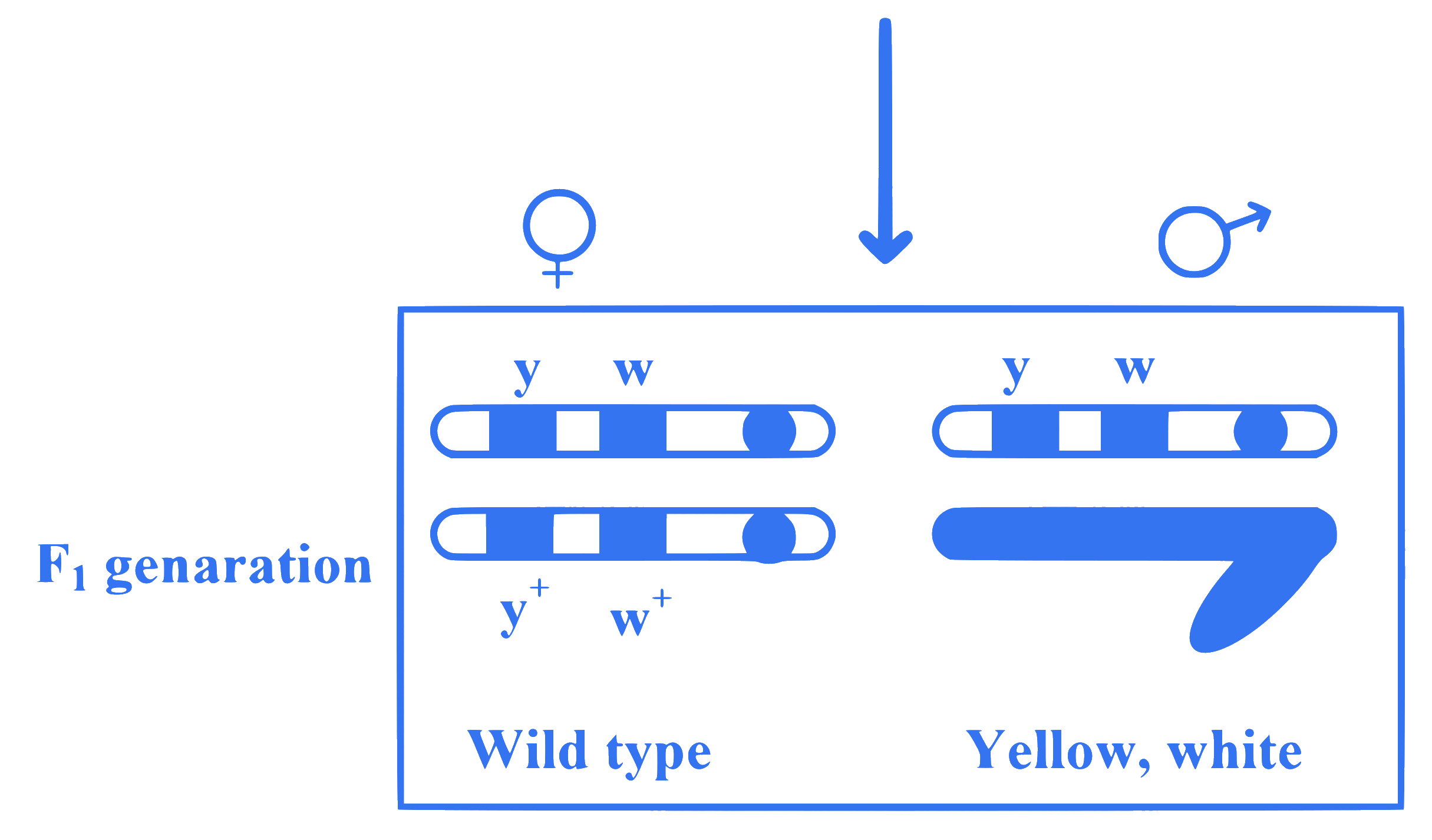
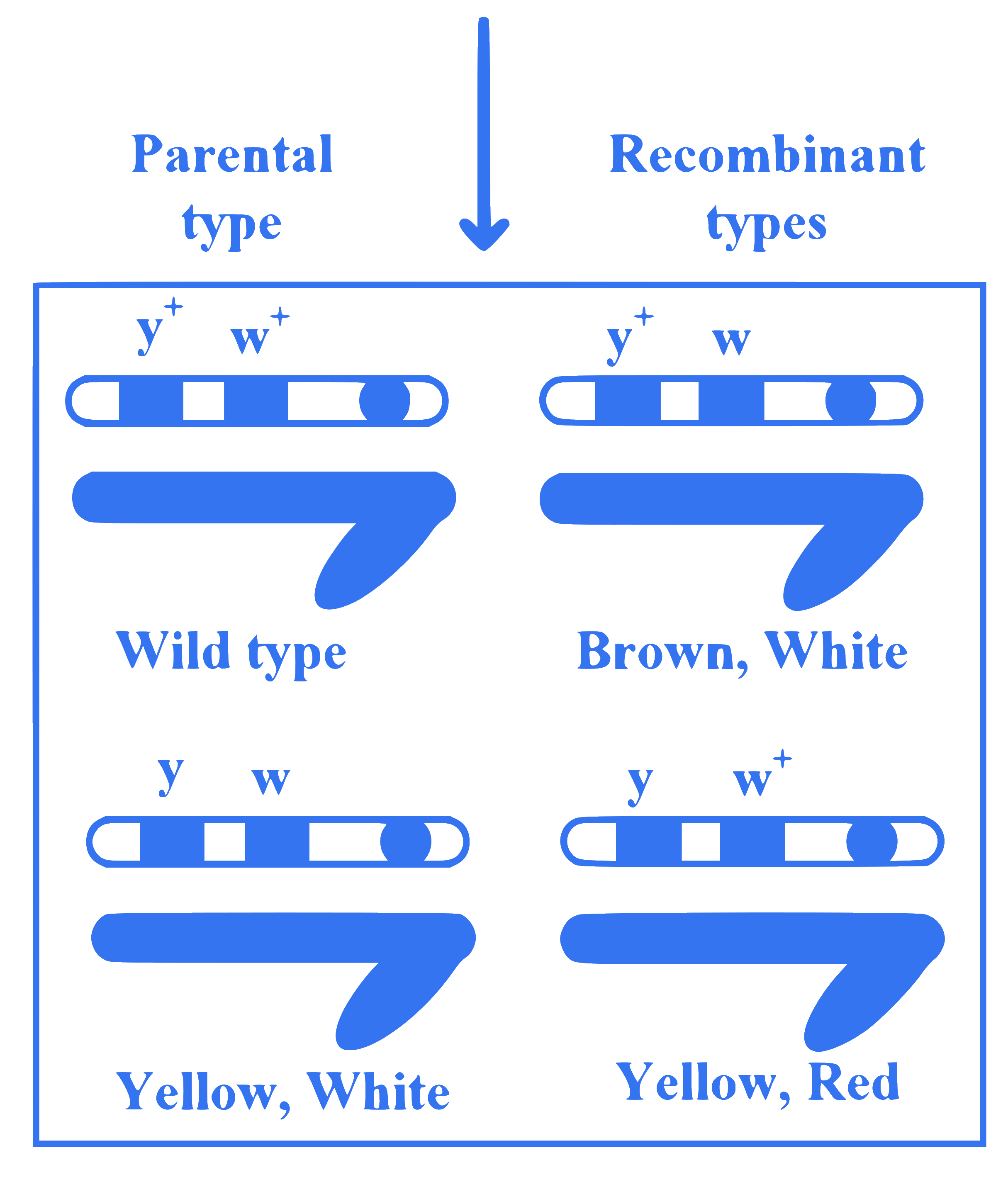
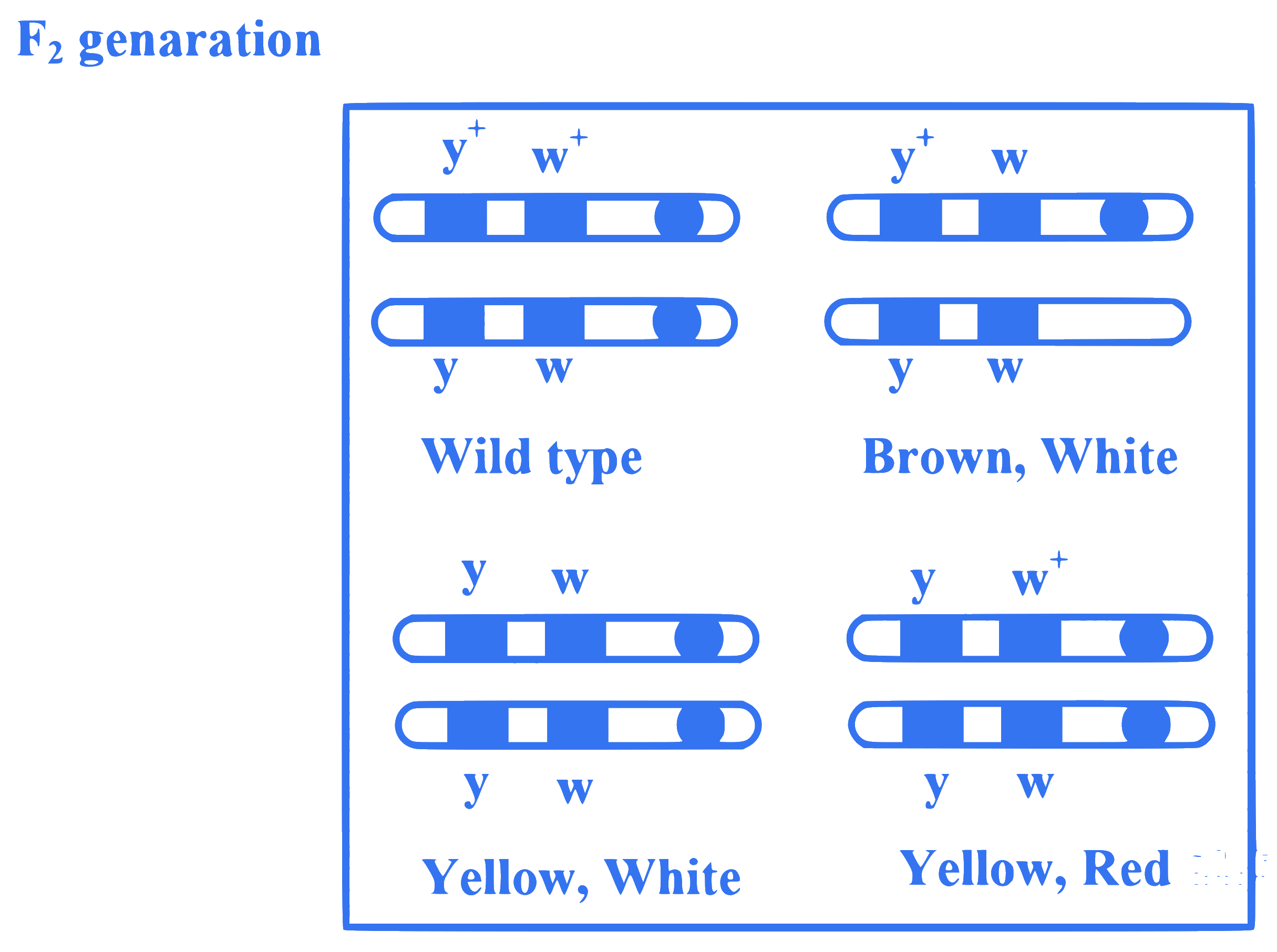




Q7.
mcq single
+1 / 0
Chromosomal basis of inheritance suggested by Sutton and Boveri is based on ________
Q8.
mcq single
+1 / 0
Choose the correct statement(s) with respect to Down's syndrome
i. Down's syndrome results from non-disjunction of $21^{\text {st }}$ chromosome during gamete formation.
ii. Down's syndrome is a sex chromosomal disorder observed in immature mothers who are below the age of 18 .
iii. These patients have mild mental retardation, poor skeletal development, flat hands, stubby fingers and palms are broader.
iv. The have 44 A with XO type of karyotype.
v. The risk of giving birth to a Down Syndrome child is more in mothers who are over 45 years of age.
Q9.
mcq single
+1 / 0
Non-disjunction of X-chromosomes during meiosis in the formation of ovum leads to _________ syndrome.
Q10.
mcq single
+1 / 0
Test cross is used to confirm $\qquad$ .
Q11.
mcq single
+1 / 0
A type of chromosomal aberration, in which there is no loss or gain of gene complement of the chromosome is $\qquad$ .
Q12.
mcq single
+1 / 0
Structural and functional unit of liver is ________.
Q13.
mcq single
+1 / 0
$$ \text { Match the following characteristics of tooth. } $$
Column I
Column II
i.
Enamel
a.
Made up of calcified connective tissue.
ii.
Dentine
b.
Contains blood vessels and nerves
iii.
Pulp cavity
c.
Covered by cementum
iv.
Root
d.
$$
\text { Hardest substance }
$$
Choose the correct option from below.
Q14.
mcq single
+1 / 0
Parenchymal circulation is found in _________
Q15.
mcq single
+1 / 0
Which one of the following statements is INCORRECT?
Q16.
mcq single
+1 / 0
$$ \text { Match the Column I with Column II } $$
Column I
(Animals)
Column II
(Respiratory organs)
i.
Sponges
a.
External gills
ii.
Insects
b.
Plasma membrane
iii.
Limulus
c.
Tracheae
iv.
Amphibian tadpoles
d.
Book gills
Select the correct option
Q17.
mcq single
+1 / 0
Which of the following Cyanobacteria is found associated with both the lichens and the plants like Cycas and Azolla?
Q18.
mcq single
+1 / 0
The partial pressure of carbon dioxide $\left(\mathrm{ppCO}_2\right)$ of blood entering the pulmonary capillaries is_______.
Q19.
mcq single
+1 / 0
Given below are two statements.
Statement I - GMO crops could potentially have negative effects on human health and natural biodiversity.
Statement II - Insertion of a gene from Brazil nut to increase the production of methionine in soyabean has caused allergic reaction to those who have nut allergies.
In the light of above statements, choose the correct option given below:
Q20.
mcq single
+1 / 0
Cranial nerve $\underline{\mathrm{i}}$ is also called pneumogastric, $\underline{\mathrm{i}} \underline{\mathrm{i}}$ is also called vestibulocochlear, and iii is also called Dentist's Nerve.
Q21.
mcq single
+1 / 0
In a polarised state of nerve membrane, sodiumpotassium exchange pump actively restores the ions on their appropriate side because ________ against concentration electrochemical gradient.
Q22.
mcq single
+1 / 0
Consumption of which drug increases the concentration of dopamine in the brain?
Q23.
mcq single
+1 / 0
With reference to role of internal ear in equilibrium receptors for dynamic balance of body lie in i while the receptors for static or linear balance of body lie in ii
Q24.
mcq single
+1 / 0
Match the parts of human eye in Column I with their respective characteristics in Column II.
Column I
Column II
i.
Fovea Centralis
a.
Thick, Muscular ring like structure at junction of choroid and iris.
ii.
Conjunctiva
b.
Area of retina from which the optic nerve and blood vessels leave the eyeball.
iii.
Blind spot
c.
Exposed, transparent part of sclera and cornea.
iv.
Ciliary body
d.
Place of formation of sharpest vision.
Choose the correct option from below.
Q25.
mcq single
+1 / 0
Action of salivary amylase stops when it mixes with gastric juice in stomach because _________ .
Q26.
mcq single
+1 / 0
The process in which an organism consumes and utilises food substances is _________ .
Q27.
mcq single
+1 / 0
Which of the following pair of organs do NOT produce digestive enzymes?
Q28.
mcq single
+1 / 0
Select the INCORRECT statement.
Q29.
mcq single
+1 / 0
Select the INCORRECT statement.
Q30.
mcq single
+1 / 0
Which of the following is the role of SSBP's during DNA replication?
Q31.
mcq single
+1 / 0
The number of nucleotides in tRNA is _________ .
Q32.
mcq single
+1 / 0
Match the process in Column I with the action in Column II.
Column I
Column II
i.
Tailing
a.
Removal of introns
ii.
Capping
b.
Polyadenylation
iii.
Translocation
c.
Addition of $\mathrm{mG}_{\mathrm{ppp}}$ at $5^{\prime}$ end of hnRNA
iv.
Splicing
d.
Movement of ribosome on mRNA.
Choose the correct match from the options given below.
Q33.
mcq single
+1 / 0
Which bonds hold together the two complementary polynucleotide chains of DNA?
Q34.
mcq single
+1 / 0
During allergic conditions which of the following increase in number?
Q35.
mcq single
+1 / 0
The larvae of Wuchereria bancrofti undergo _________moultings before they become adults and settle in the lymphatic system of human.
Q36.
mcq single
+1 / 0
Interferons are class of cytokines to stimulate other cells against ________ infections.
Q37.
mcq single
+1 / 0
Which of the following is caused by neither bacteria nor viruses?
Q38.
mcq single
+1 / 0
Which one of the following is the correct description of Streptococcus pneumoniae causing pneumonia?
Q39.
mcq single
+1 / 0
Female Aedes mosquito acts as a vector in the transmission of ________
Q40.
mcq single
+1 / 0
Water is the best medium for the transport of dissolved minerals and food molecules because it has ______ .
Q41.
mcq single
+1 / 0
The additional apoplastic route of water that allows direct access to xylem and phloem is along the margins of __________ .
Q42.
mcq single
+1 / 0
The correct sequence of water absorption by root hair cell is ________
Q43.
mcq single
+1 / 0
Given below are two statements about asexual reproduction.
Statement I - In asexual reproduction, the ploidy of progeny is identical to the parent.
Statement II - The progeny is also referred to as clone.
In the light of above statements, choose the correct option given below:
Q44.
mcq single
+1 / 0
Leaf cutting is successful in __________ .
Q45.
mcq single
+1 / 0
Given below are two statements.
Statement I-Budding is most common method of asexual reproduction in unicellular yeast.
Statement II - In yeast, budding occurs during unfavorable condition.
In the light of above statements, choose the most appropriate answer from the option given below:
Q46.
mcq single
+1 / 0
Gene pool is __________ .
Q47.
mcq single
+1 / 0
Which of the following pair of organisms are euryhaline in nature?
Q48.
mcq single
+1 / 0
What is the horizontal distribution of different species occupying levels on land or in water called?
Q49.
mcq single
+1 / 0
Match types of niche in Column I with its explanation in Column II.
Column I
(Type of niche)
Column II
(Explanation)
i.
Habitat
a.
absence of all competitors
ii.
Hypervolume
b.
physical space occupied by the organism
iii.
Fundamental
c.
presence of competition for resources
iv.
Realized
d.
considers biotic and abiotic environmental factors
Select the correct option
Q50.
mcq single
+1 / 0
Which one of the following is INCORRECT with respect to Verhulst - Pearl Logisitc Growth.
Q51.
mcq single
+1 / 0
Given below are two statements regarding, competitive interaction.
Statement I - Competition is the type of interaction where totally unrelated species may compete for the same resource.
Statement II - Competition occurs only when resources are limited.
In the light of above statements, choose the correct option given below:
Q52.
mcq single
+1 / 0
Which of the following in INCORRECT about conformers?
Q53.
mcq single
+1 / 0
Given below are two statements.
Statement I - The heart sound 'Lub' is produced due to simultaneous closure of semilunar valves. Statement II - The heart sound 'Dub' is produced due to simultaneous closure of cuspid valves.
In the light of above statements, choose the correct option given below:
Q54.
mcq single
+1 / 0
The QRS complex in a normal ECG represents _________.
Q55.
mcq single
+1 / 0
The erythropoietic organs of a foetus are ________and _________.
Q56.
mcq single
+1 / 0
Which of the following is NOT a feature of capacitation process of sperm?
Q57.
mcq single
+1 / 0
In human female, the number of primordial follicles in the ovary at the time of birth is more than _______ million.
Q58.
mcq single
+1 / 0
Complete the following analogy about human sperm.
Nebenkern : mitochondria :: Acrosome : _______
Q59.
mcq single
+1 / 0
Which of the following hormones is secreted by theca interna covering the Graafian follicle?
Q60.
mcq single
+1 / 0
Match hormones given in Column I with their source gland in Column II.
Column I
Column II
i.
Oxytocin
a.
Adrenal medulla
ii.
Melatonin
b.
Thyroid gland
iii.
Catecholamines
c.
Pineal gland
iv.
Calcitonin
d.
Hypothalamus
Choose the correct option from below.
Q61.
mcq single
+1 / 0
Match the hormones given in Column I with their respective source organs of secretion, given in Column II.
Column I
(Hormone)
Column II
(Source of secretion)
i.
hCG
a.
Adenohypophysis of pituitary gland.
ii.
CCK
b.
Placenta.
iii.
ICSH
c.
Pars intermedia of hypophysis gland.
iv.
MSH
d.
Gastro - intestinal mucosa.
Choose the correct option from below.
Q62.
mcq single
+1 / 0
Given below are two statements.
Statement I - Mode of action of catecholamines, peptide and polypeptide hormones is through cell membrane receptors.
Statement II - These hormones are non-steroid, water soluble and lipid insoluble hormones.
In the light of above statements, choose the correct option from below:
Q63.
mcq single
+1 / 0
Demineralization of bone resulting in softening, bending and fracture of bones, common in women who have reached menopause is called i and is caused due to ii.
Q64.
mcq single
+1 / 0
The purposeful manipulation in the heredity of crops and the production of new superior varieties of existing crop plants is called _________
Q65.
mcq single
+1 / 0
Match the Column I containing mutant varieties with Column II containing respective crop plants.
Column I
Column II
i.
Jagannath
a.
Cotton
ii.
NP- 836
b.
Cabbage
iii.
Indore - 2
c.
Rice
iv.
Regina - II
d.
Wheat
Choose the correct option from below.
Q66.
mcq single
+1 / 0
The water hyacinth (Eichhornia crassipes) is a native of _________
Q67.
mcq single
+1 / 0
Which one of the following is the main objective of proposing Biological diversity Act 2002?
Q68.
mcq single
+1 / 0
Identify the sacred groves found in India from the following list.
i. Khasi and Jaintia Hills in Meghalaya.
ii. Western Ghat regions of Maharashtra.
iii. Aravalli Hills of Rajasthan.
iv. Chanda and Sarguja areas of Madhya Pradesh.
v. Ladakh region of Himalayas.
Choose the correct option from given below
Q69.
mcq single
+1 / 0
Which one of the following is also called 'Terror of Bengal' ?
Q70.
mcq single
+1 / 0
Which one of the following occurs maximum during exponential growth phase in plants?
Q71.
mcq single
+1 / 0
In which one of the following conditions tobacco plants do NOT flower?
Q72.
mcq single
+1 / 0
According to proton transport theory of stomatal mechanism, protons are formed in the guard cells when _________ .
Q73.
mcq single
+1 / 0
Given below are two statements.
Statement I - Gibberellins promote flowering in long day plants.
Statement II - ABA inhibits flowering in long day plants but stimulates flowering in short day plants. In the light of above statements, choose the correct option given below:
Q74.
mcq single
+1 / 0
How many $\mathrm{CO}_2$ molecules are released after oxidation of one acetyl CoA in Krebs cycle?
Q75.
mcq single
+1 / 0
How many phosphorylation reactions occur for formation of fructose 1,6 biphosphate during the preparatory phase of glycolysis?
Q76.
mcq single
+1 / 0
The final electron acceptor in ETS of cellular respiration is
Q77.
mcq single
+1 / 0
Which of the following reaction occurs during conversion of $\alpha$-ketoglutarate $\rightarrow$ Succinyl Co-A?
Q78.
mcq single
+1 / 0
Given below are two statements.
Statement I - Skeletal muscles usually derive energy by anaerobic respiration.
Statement II - After vigorous exercise lactic acid accumulates in muscle fibres leading to muscle fatigue.
In the light of above statements, choose the most appropriate answer from the options given below:
Q79.
mcq single
+1 / 0
Select the correct set of evolutionary characters of man.
Q80.
mcq single
+1 / 0
Which one of the following occurs due to changes in the environment and genetic variations of the species?
Q81.
mcq single
+1 / 0
Select the INCORRECT statement regarding RNA world hypothesis.
Q82.
mcq single
+1 / 0
Choose the INCORRECT pair.
Q83.
mcq single
+1 / 0
Juxta glomerular apparatus in kidney releases Renin, when a person is __________
Q84.
mcq single
+1 / 0
Which of the following are true about sharks?
i. Their body fluids is isosmotic to external environment.
ii. They are osmoconformers.
iii. They control their internal environment independent of external environment.
iv. They are osmoregulators.
Q85.
mcq single
+1 / 0
Most energy saving mode of excretion is_________.
Q86.
mcq single
+1 / 0
Kidneys are NOT associated with secretion of
Q87.
mcq single
+1 / 0
Which contraceptive device attracts macrophages in the uterine cavity for phagocytosis of sperms?
Q88.
mcq single
+1 / 0
How many nuclei migrate from each pole to the centre of developing female gametophyte to form secondary nucleus?
Q89.
mcq single
+1 / 0
Choose the correct sequence of events in pollenpistil interaction from the options given below:
i. Entry of pollen tube into synergid.
ii. Development of pollen tube.
iii. Deposition of pollen grains on the stigma.
iv. Release of contents from pollen tube.
Q90.
mcq single
+1 / 0
Match the types of endosperms given in Column I with examples given in Column II
Column I
Column II
i.
Cellular
a.
Coconut
ii.
Nuclear
b.
Asphodelus
iii.
Helobial
c.
Petunia
Q91.
mcq single
+1 / 0
To achieve pollination, attractions and reward are required in
Q92.
mcq single
+1 / 0
Most important fuel of living cells is _________ .
Q93.
mcq single
+1 / 0
Basic proteins consist of more basic amino acids and these amino acids exist as a cation at the physiological pH of 7.4.
Which among the following are basic amino acids?
Q94.
mcq single
+1 / 0
Which one of the following is considered as molecular scissors in modern biotechnology?
Q95.
mcq single
+1 / 0
During PCR cycle, the temperature range in the step of annealing is________
Q96.
mcq single
+1 / 0
Match the Column I containing restriction enzymes with Column II containing recognition sequences.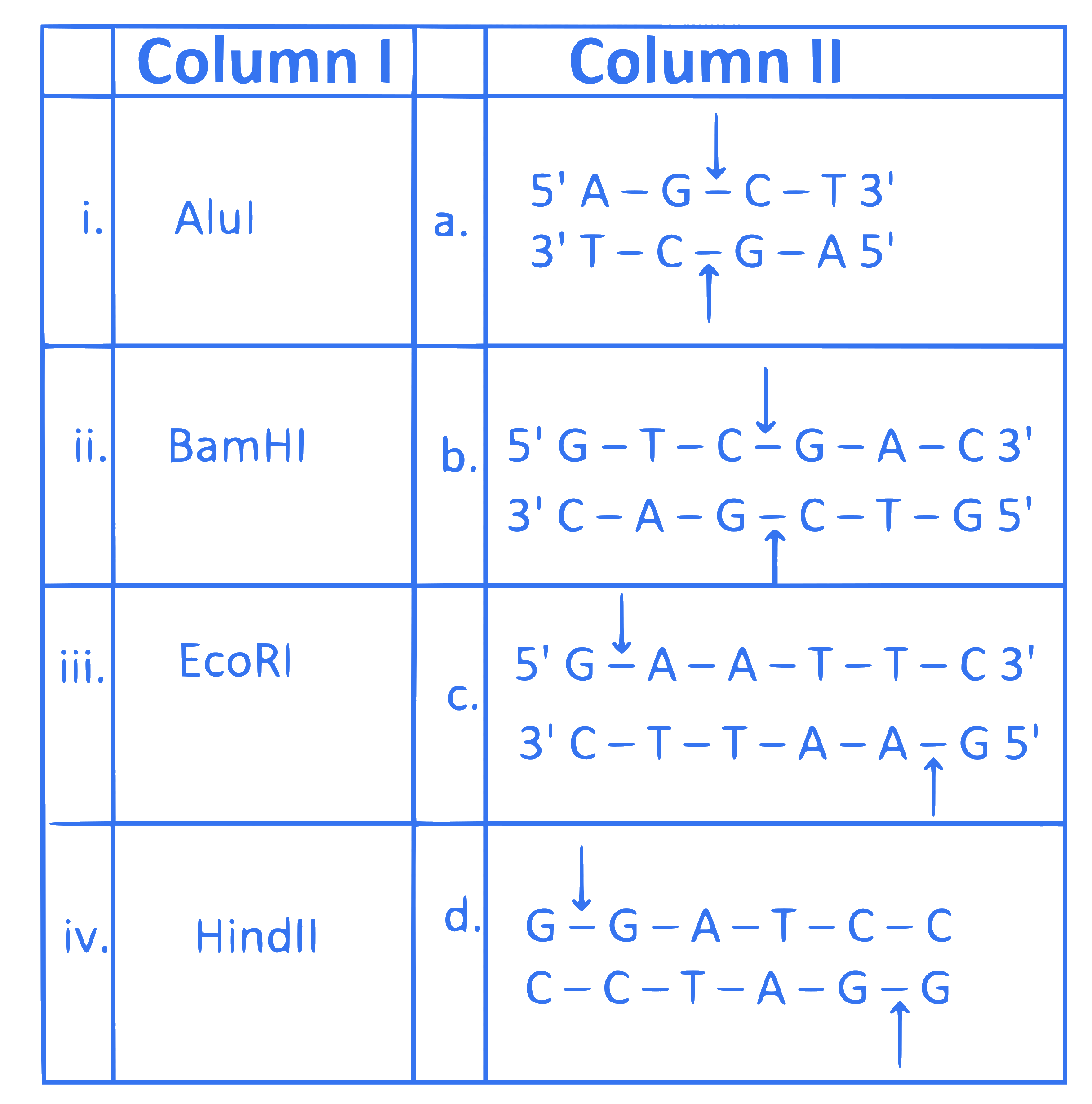

Q97.
mcq single
+1 / 0
The manipulation of genetic material towards a desired end using "in vitro" process is called _________.
Q98.
mcq single
+1 / 0
Nearly $80 \%$ of nitrogen found in human tissues originate from ________ nitrogen fixation.
Q99.
mcq single
+1 / 0
Which one of the following minerals is NOT mobilized in plants?
Q100.
mcq single
+1 / 0
Match the mineral deficiency disorders given in Column I with their symptoms given in Column II.
Column I
Column II
i.
Chlorosis
a.
Localised death of tissues in leaves
ii.
Necrosis
b.
Premature fall of flowers, fruits and leaves
iii.
Motlling
c.
Yellowing of leaves due to absence of chlorophyll
iv.
Abscission
d.
Appearance of green and non- green patches on leaves
Choose the correct option from below.
Q101.
mcq single
+1 / 0
Which of the following microbes is used in the production of yoghurt?
Q102.
mcq single
+1 / 0
Protozoans like Nosema locustae is used for controlling the target pests like ________
Q103.
mcq single
+1 / 0
All of the raw materials that are extracted from an ecosystem are called ________ services.
Q104.
mcq single
+1 / 0
Phosphorus cycle does not pass through __________ .
Q105.
mcq single
+1 / 0
The pyramid of biomass is inverted in________ ecosystem.
Q106.
mcq single
+1 / 0
Observe the given figure of Morgan's experiment for yellow body and white eyed female Drosophila crossed with brown body and red eyed male Drosophila. What will be the percentage of parental type in $F_2$ generation?







Q107.
mcq single
+1 / 0
Chromosomal basis of inheritance suggested by Sutton and Boveri is based on ________
Q108.
mcq single
+1 / 0
Choose the correct statement(s) with respect to Down's syndrome
i. Down's syndrome results from non-disjunction of $21^{\text {st }}$ chromosome during gamete formation.
ii. Down's syndrome is a sex chromosomal disorder observed in immature mothers who are below the age of 18 .
iii. These patients have mild mental retardation, poor skeletal development, flat hands, stubby fingers and palms are broader.
iv. The have 44 A with XO type of karyotype.
v. The risk of giving birth to a Down Syndrome child is more in mothers who are over 45 years of age.
Q109.
mcq single
+1 / 0
Non-disjunction of X-chromosomes during meiosis in the formation of ovum leads to _________ syndrome.
Q110.
mcq single
+1 / 0
Test cross is used to confirm $\qquad$ .
Q111.
mcq single
+1 / 0
A type of chromosomal aberration, in which there is no loss or gain of gene complement of the chromosome is $\qquad$ .
Q112.
mcq single
+1 / 0
Structural and functional unit of liver is ________.
Q113.
mcq single
+1 / 0
$$ \text { Match the following characteristics of tooth. } $$
Column I
Column II
i.
Enamel
a.
Made up of calcified connective tissue.
ii.
Dentine
b.
Contains blood vessels and nerves
iii.
Pulp cavity
c.
Covered by cementum
iv.
Root
d.
$$
\text { Hardest substance }
$$
Choose the correct option from below.
Q114.
mcq single
+1 / 0
Parenchymal circulation is found in _________
Q115.
mcq single
+1 / 0
Which one of the following statements is INCORRECT?
Q116.
mcq single
+1 / 0
$$ \text { Match the Column I with Column II } $$
Column I
(Animals)
Column II
(Respiratory organs)
i.
Sponges
a.
External gills
ii.
Insects
b.
Plasma membrane
iii.
Limulus
c.
Tracheae
iv.
Amphibian tadpoles
d.
Book gills
Select the correct option
Q117.
mcq single
+1 / 0
Which of the following Cyanobacteria is found associated with both the lichens and the plants like Cycas and Azolla?
Q118.
mcq single
+1 / 0
The partial pressure of carbon dioxide $\left(\mathrm{ppCO}_2\right)$ of blood entering the pulmonary capillaries is_______.
Q119.
mcq single
+1 / 0
Given below are two statements.
Statement I - GMO crops could potentially have negative effects on human health and natural biodiversity.
Statement II - Insertion of a gene from Brazil nut to increase the production of methionine in soyabean has caused allergic reaction to those who have nut allergies.
In the light of above statements, choose the correct option given below:
Q120.
mcq single
+1 / 0
Cranial nerve $\underline{\mathrm{i}}$ is also called pneumogastric, $\underline{\mathrm{i}} \underline{\mathrm{i}}$ is also called vestibulocochlear, and iii is also called Dentist's Nerve.
Q121.
mcq single
+1 / 0
In a polarised state of nerve membrane, sodiumpotassium exchange pump actively restores the ions on their appropriate side because ________ against concentration electrochemical gradient.
Q122.
mcq single
+1 / 0
Consumption of which drug increases the concentration of dopamine in the brain?
Q123.
mcq single
+1 / 0
With reference to role of internal ear in equilibrium receptors for dynamic balance of body lie in i while the receptors for static or linear balance of body lie in ii
Q124.
mcq single
+1 / 0
Match the parts of human eye in Column I with their respective characteristics in Column II.
Column I
Column II
i.
Fovea Centralis
a.
Thick, Muscular ring like structure at junction of choroid and iris.
ii.
Conjunctiva
b.
Area of retina from which the optic nerve and blood vessels leave the eyeball.
iii.
Blind spot
c.
Exposed, transparent part of sclera and cornea.
iv.
Ciliary body
d.
Place of formation of sharpest vision.
Choose the correct option from below.
Q125.
mcq single
+1 / 0
Action of salivary amylase stops when it mixes with gastric juice in stomach because _________ .
Q126.
mcq single
+1 / 0
The process in which an organism consumes and utilises food substances is _________ .
Q127.
mcq single
+1 / 0
Which of the following pair of organs do NOT produce digestive enzymes?
Q128.
mcq single
+1 / 0
Select the INCORRECT statement.
Q129.
mcq single
+1 / 0
Select the INCORRECT statement.
Q130.
mcq single
+1 / 0
Which of the following is the role of SSBP's during DNA replication?
Q131.
mcq single
+1 / 0
The number of nucleotides in tRNA is _________ .
Q132.
mcq single
+1 / 0
Match the process in Column I with the action in Column II.
Column I
Column II
i.
Tailing
a.
Removal of introns
ii.
Capping
b.
Polyadenylation
iii.
Translocation
c.
Addition of $\mathrm{mG}_{\mathrm{ppp}}$ at $5^{\prime}$ end of hnRNA
iv.
Splicing
d.
Movement of ribosome on mRNA.
Choose the correct match from the options given below.
Q133.
mcq single
+1 / 0
Which bonds hold together the two complementary polynucleotide chains of DNA?
Q134.
mcq single
+1 / 0
During allergic conditions which of the following increase in number?
Q135.
mcq single
+1 / 0
The larvae of Wuchereria bancrofti undergo _________moultings before they become adults and settle in the lymphatic system of human.
Q136.
mcq single
+1 / 0
Interferons are class of cytokines to stimulate other cells against ________ infections.
Q137.
mcq single
+1 / 0
Which of the following is caused by neither bacteria nor viruses?
Q138.
mcq single
+1 / 0
Which one of the following is the correct description of Streptococcus pneumoniae causing pneumonia?
Q139.
mcq single
+1 / 0
Female Aedes mosquito acts as a vector in the transmission of ________
Q140.
mcq single
+1 / 0
Water is the best medium for the transport of dissolved minerals and food molecules because it has ______ .
Q141.
mcq single
+1 / 0
The additional apoplastic route of water that allows direct access to xylem and phloem is along the margins of __________ .
Q142.
mcq single
+1 / 0
The correct sequence of water absorption by root hair cell is ________
Q143.
mcq single
+1 / 0
Given below are two statements about asexual reproduction.
Statement I - In asexual reproduction, the ploidy of progeny is identical to the parent.
Statement II - The progeny is also referred to as clone.
In the light of above statements, choose the correct option given below:
Q144.
mcq single
+1 / 0
Leaf cutting is successful in __________ .
Q145.
mcq single
+1 / 0
Given below are two statements.
Statement I-Budding is most common method of asexual reproduction in unicellular yeast.
Statement II - In yeast, budding occurs during unfavorable condition.
In the light of above statements, choose the most appropriate answer from the option given below:
Q146.
mcq single
+1 / 0
Gene pool is __________ .
Q147.
mcq single
+1 / 0
Which of the following pair of organisms are euryhaline in nature?
Q148.
mcq single
+1 / 0
What is the horizontal distribution of different species occupying levels on land or in water called?
Q149.
mcq single
+1 / 0
Match types of niche in Column I with its explanation in Column II.
Column I
(Type of niche)
Column II
(Explanation)
i.
Habitat
a.
absence of all competitors
ii.
Hypervolume
b.
physical space occupied by the organism
iii.
Fundamental
c.
presence of competition for resources
iv.
Realized
d.
considers biotic and abiotic environmental factors
Select the correct option
Q150.
mcq single
+1 / 0
Which one of the following is INCORRECT with respect to Verhulst - Pearl Logisitc Growth.
Q151.
mcq single
+1 / 0
Given below are two statements regarding, competitive interaction.
Statement I - Competition is the type of interaction where totally unrelated species may compete for the same resource.
Statement II - Competition occurs only when resources are limited.
In the light of above statements, choose the correct option given below:
Q152.
mcq single
+1 / 0
Which of the following in INCORRECT about conformers?
Q153.
mcq single
+1 / 0
Given below are two statements.
Statement I - The heart sound 'Lub' is produced due to simultaneous closure of semilunar valves. Statement II - The heart sound 'Dub' is produced due to simultaneous closure of cuspid valves.
In the light of above statements, choose the correct option given below:
Q154.
mcq single
+1 / 0
The QRS complex in a normal ECG represents _________.
Q155.
mcq single
+1 / 0
The erythropoietic organs of a foetus are ________and _________.
Q156.
mcq single
+1 / 0
Which of the following is NOT a feature of capacitation process of sperm?
Q157.
mcq single
+1 / 0
In human female, the number of primordial follicles in the ovary at the time of birth is more than _______ million.
Q158.
mcq single
+1 / 0
Complete the following analogy about human sperm.
Nebenkern : mitochondria :: Acrosome : _______
Q159.
mcq single
+1 / 0
Which of the following hormones is secreted by theca interna covering the Graafian follicle?
Q160.
mcq single
+1 / 0
Match hormones given in Column I with their source gland in Column II.
Column I
Column II
i.
Oxytocin
a.
Adrenal medulla
ii.
Melatonin
b.
Thyroid gland
iii.
Catecholamines
c.
Pineal gland
iv.
Calcitonin
d.
Hypothalamus
Choose the correct option from below.
Q161.
mcq single
+1 / 0
Match the hormones given in Column I with their respective source organs of secretion, given in Column II.
Column I
(Hormone)
Column II
(Source of secretion)
i.
hCG
a.
Adenohypophysis of pituitary gland.
ii.
CCK
b.
Placenta.
iii.
ICSH
c.
Pars intermedia of hypophysis gland.
iv.
MSH
d.
Gastro - intestinal mucosa.
Choose the correct option from below.
Q162.
mcq single
+1 / 0
Given below are two statements.
Statement I - Mode of action of catecholamines, peptide and polypeptide hormones is through cell membrane receptors.
Statement II - These hormones are non-steroid, water soluble and lipid insoluble hormones.
In the light of above statements, choose the correct option from below:
Q163.
mcq single
+1 / 0
Demineralization of bone resulting in softening, bending and fracture of bones, common in women who have reached menopause is called i and is caused due to ii.
Q164.
mcq single
+1 / 0
The purposeful manipulation in the heredity of crops and the production of new superior varieties of existing crop plants is called _________
Q165.
mcq single
+1 / 0
Match the Column I containing mutant varieties with Column II containing respective crop plants.
Column I
Column II
i.
Jagannath
a.
Cotton
ii.
NP- 836
b.
Cabbage
iii.
Indore - 2
c.
Rice
iv.
Regina - II
d.
Wheat
Choose the correct option from below.
Q166.
mcq single
+1 / 0
The water hyacinth (Eichhornia crassipes) is a native of _________
Q167.
mcq single
+1 / 0
Which one of the following is the main objective of proposing Biological diversity Act 2002?
Q168.
mcq single
+1 / 0
Identify the sacred groves found in India from the following list.
i. Khasi and Jaintia Hills in Meghalaya.
ii. Western Ghat regions of Maharashtra.
iii. Aravalli Hills of Rajasthan.
iv. Chanda and Sarguja areas of Madhya Pradesh.
v. Ladakh region of Himalayas.
Choose the correct option from given below
Q169.
mcq single
+1 / 0
Which one of the following is also called 'Terror of Bengal' ?
Q170.
mcq single
+1 / 0
Which one of the following occurs maximum during exponential growth phase in plants?
Q171.
mcq single
+1 / 0
In which one of the following conditions tobacco plants do NOT flower?
Q172.
mcq single
+1 / 0
According to proton transport theory of stomatal mechanism, protons are formed in the guard cells when _________ .
Q173.
mcq single
+1 / 0
Given below are two statements.
Statement I - Gibberellins promote flowering in long day plants.
Statement II - ABA inhibits flowering in long day plants but stimulates flowering in short day plants. In the light of above statements, choose the correct option given below:
Q174.
mcq single
+1 / 0
How many $\mathrm{CO}_2$ molecules are released after oxidation of one acetyl CoA in Krebs cycle?
Q175.
mcq single
+1 / 0
How many phosphorylation reactions occur for formation of fructose 1,6 biphosphate during the preparatory phase of glycolysis?
Q176.
mcq single
+1 / 0
The final electron acceptor in ETS of cellular respiration is
Q177.
mcq single
+1 / 0
Which of the following reaction occurs during conversion of $\alpha$-ketoglutarate $\rightarrow$ Succinyl Co-A?
Q178.
mcq single
+1 / 0
Given below are two statements.
Statement I - Skeletal muscles usually derive energy by anaerobic respiration.
Statement II - After vigorous exercise lactic acid accumulates in muscle fibres leading to muscle fatigue.
In the light of above statements, choose the most appropriate answer from the options given below:
Q179.
mcq single
+1 / 0
Select the correct set of evolutionary characters of man.
Q180.
mcq single
+1 / 0
Which one of the following occurs due to changes in the environment and genetic variations of the species?
Q181.
mcq single
+1 / 0
Select the INCORRECT statement regarding RNA world hypothesis.
Q182.
mcq single
+1 / 0
Choose the INCORRECT pair.
Q183.
mcq single
+1 / 0
Juxta glomerular apparatus in kidney releases Renin, when a person is __________
Q184.
mcq single
+1 / 0
Which of the following are true about sharks?
i. Their body fluids is isosmotic to external environment.
ii. They are osmoconformers.
iii. They control their internal environment independent of external environment.
iv. They are osmoregulators.
Q185.
mcq single
+1 / 0
Most energy saving mode of excretion is_________.
Q186.
mcq single
+1 / 0
Kidneys are NOT associated with secretion of
Q187.
mcq single
+1 / 0
Which contraceptive device attracts macrophages in the uterine cavity for phagocytosis of sperms?
Q188.
mcq single
+1 / 0
How many nuclei migrate from each pole to the centre of developing female gametophyte to form secondary nucleus?
Q189.
mcq single
+1 / 0
Choose the correct sequence of events in pollenpistil interaction from the options given below:
i. Entry of pollen tube into synergid.
ii. Development of pollen tube.
iii. Deposition of pollen grains on the stigma.
iv. Release of contents from pollen tube.
Q190.
mcq single
+1 / 0
Match the types of endosperms given in Column I with examples given in Column II
Column I
Column II
i.
Cellular
a.
Coconut
ii.
Nuclear
b.
Asphodelus
iii.
Helobial
c.
Petunia
Q191.
mcq single
+1 / 0
To achieve pollination, attractions and reward are required in
Q192.
mcq single
+1 / 0
Most important fuel of living cells is _________ .
Q193.
mcq single
+1 / 0
Basic proteins consist of more basic amino acids and these amino acids exist as a cation at the physiological pH of 7.4.
Which among the following are basic amino acids?
Q194.
mcq single
+1 / 0
Which one of the following is considered as molecular scissors in modern biotechnology?
Q195.
mcq single
+1 / 0
During PCR cycle, the temperature range in the step of annealing is________
Q196.
mcq single
+1 / 0
Match the Column I containing restriction enzymes with Column II containing recognition sequences.

Q197.
mcq single
+1 / 0
The manipulation of genetic material towards a desired end using "in vitro" process is called _________.
Q198.
mcq single
+1 / 0
Nearly $80 \%$ of nitrogen found in human tissues originate from ________ nitrogen fixation.
Q199.
mcq single
+1 / 0
Which one of the following minerals is NOT mobilized in plants?
Q200.
mcq single
+1 / 0
Match the mineral deficiency disorders given in Column I with their symptoms given in Column II.
Column I
Column II
i.
Chlorosis
a.
Localised death of tissues in leaves
ii.
Necrosis
b.
Premature fall of flowers, fruits and leaves
iii.
Motlling
c.
Yellowing of leaves due to absence of chlorophyll
iv.
Abscission
d.
Appearance of green and non- green patches on leaves
Choose the correct option from below.