MHT CET (PCB) 2024 22th April Morning Shift
MEDICAL 2024 Previous Year
3 hDuration
200Total Marks
400Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 400 across 3 section(s); maximum marks: 200.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+1 / 0
Which of the following isomeric amines has the highest boiling point?
Q2.
mcq single
+1 / 0
How many moles of iodomethane are consumed in the following conversion?
$$ \mathrm{CH}_3 \mathrm{NH}_2 \xrightarrow[\Delta]{\mathrm{CH}_3 \mathrm{l}}\left(\mathrm{CH}_3\right)_4 \mathrm{~N}^{+} \mathrm{I}^{-} $$
Q3.
mcq single
+1 / 0
Which element from following does NOT exhibit magnetic moment in +1 state?
Q4.
mcq single
+1 / 0
Identify false statement about transition elements.
Q5.
mcq single
+1 / 0
Which from following is a semisynthetic polymer?
Q6.
mcq single
+1 / 0
Which among the following is a monomer of natural rubber?
Q7.
mcq single
+1 / 0
What is the formal charge present on oxygen atom (numbered 1) in Lewis structure of $\mathrm{CO}_2$ ?

Q8.
mcq single
+1 / 0
What are the possible values of magnetic quantum number for $p$ orbital?
Q9.
mcq single
+1 / 0
What is the frequency of red light having wave length 750 nm ?
Q10.
mcq single
+1 / 0
Which from the following equations represents the relation between solubility ( $\mathrm{mol} \mathrm{L}^{-1}$ ) and solubility product for the salt $\mathrm{B}_2 \mathrm{~A}$ ?
Q11.
mcq single
+1 / 0
Calculate the pOH of buffer solution formed from 0.3 M weak base and 0.45 M of its salt with strong acid $\left[\mathrm{pK}_{\mathrm{b}}=4.7447\right]$
Q12.
mcq single
+1 / 0
Calculate degree of dissociation of a weak monobasic acid in 0.01 M solution if dissociation constant is $1.6 \times 10^{-5}$.
Q13.
mcq single
+1 / 0
Calculate activation energy for a reaction if it's rate doubles when temperature is raised from $20^{\circ} \mathrm{C}$ to $35^{\circ} \mathrm{C}\left(\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right)$
Q14.
mcq single
+1 / 0
If half life of a first order reaction is 10 minutes, find the time required to decrease concentration of reactant from 0.08 M to 0.02 M .
Q15.
mcq single
+1 / 0
Which of the following equation exhibits integrated rate law equation for first order reaction?
Q16.
mcq single
+1 / 0
Which from following elements is a decay product of thorium?
Q17.
mcq single
+1 / 0
Which of the following changes exhibit that nitrogen undergoes oxidation?
Q18.
mcq single
+1 / 0
Identify the product obtained when alkyl alkanoate is hydrolysed with dilute HCl .
Q19.
mcq single
+1 / 0
Identify the reagent ' R ' necessary to bring the following conversion.
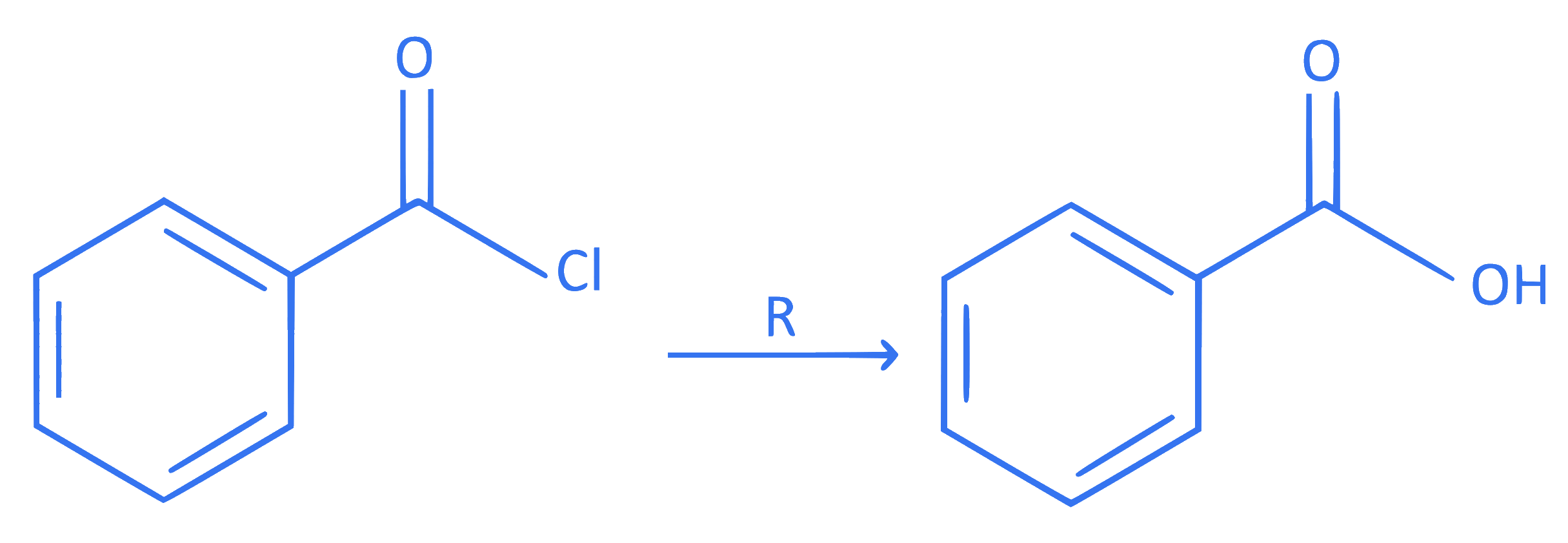

Q20.
mcq single
+1 / 0
$$ \text { Identify ' } A \text { ' in the following reaction. } $$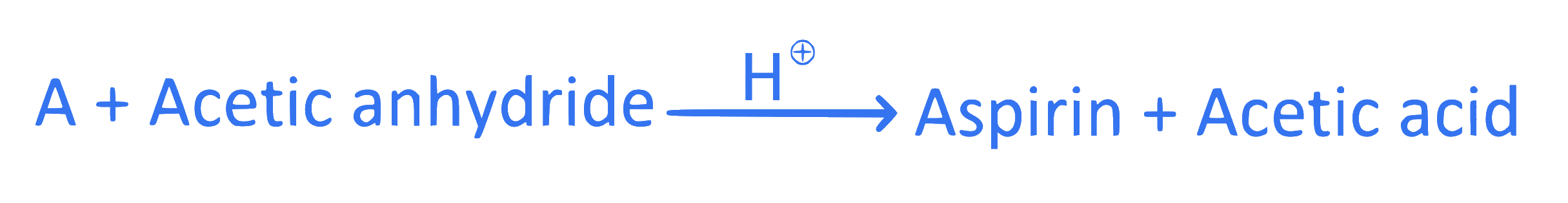

Q21.
mcq single
+1 / 0
Which among the following is the strongest acid?
Q22.
mcq single
+1 / 0
Which from following statements is NOT true about physisorption?
Q23.
mcq single
+1 / 0
Which of the following acid strength is used to get good yield of alkyl iodide from alcohol and sodium iodide in phosphoric acid?
Q24.
mcq single
+1 / 0
$$ \text { Which among the following is haloarene? } $$
Q25.
mcq single
+1 / 0
What is the ratio of mass of nitrogen that combines with 16 parts by weight of oxygen in $\mathrm{N}_2 \mathrm{O}, \mathrm{NO}, \mathrm{NO}_2$ ?
Q26.
mcq single
+1 / 0
If $\mathrm{A}, \mathrm{B}, \mathrm{C}, \mathrm{D}$ are four different elements with outer electronic configuration as $\mathrm{A}=4 \mathrm{~s}^2 4 \mathrm{p}^4, \mathrm{~B}=4 \mathrm{~s}^2 4 \mathrm{p}^5, \mathrm{C}=5 \mathrm{~s}^2 5 \mathrm{p}^4, \mathrm{D}=5 \mathrm{~s}^2 5 \mathrm{p}^5$.
Find the element having highest ionization enthalpy $\left(\Delta_1 \mathrm{H}_1\right)$
Q27.
mcq single
+1 / 0
What is the change in internal energy for $2 \mathrm{CO}_{(\mathrm{g})}+\mathrm{O}_{2(\mathrm{~g})} \rightarrow 2 \mathrm{CO}_{2(\mathrm{~g})}$ at $25^{\circ} \mathrm{C}$ ?
( $\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \Delta \mathrm{H}=-560 \mathrm{~kJ}$ )
Q28.
mcq single
+1 / 0
Which from following terms is explained by first law of thermodynamics?
Q29.
mcq single
+1 / 0
The entropy of vaporisation of benzene is $85 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$. When 117 g of benzene vaporises at its boiling point, what is entropy change of surrounding if process is at equilibrium?
Q30.
mcq single
+1 / 0
Which from following groups exhibits +I effect?
Q31.
mcq single
+1 / 0
Calculate the Henry's law constant at $25^{\circ} \mathrm{C}$ if solubility of gas in liquid is $2.1 \times 10^{-2} \mathrm{~mol} \mathrm{dm}^{-3}$ at 0.18 bar.
Q32.
mcq single
+1 / 0
If osmotic pressure of 0.2 M aqueous glucose solution is 5 atm at 300 K . Calculate the concentration of glucose solution having osmotic pressure 2.5 atm at same temperature.
Q33.
mcq single
+1 / 0
Which from following is an example of solution of solid as solute and liquid as solvent?
Q34.
mcq single
+1 / 0
Which from following complexes is having ambidentate ligand in it?
Q35.
mcq single
+1 / 0
Identify the coordination number of aluminium in potassium trioxalatoaluminate(III).
Q36.
mcq single
+1 / 0
Which from following chemical activities does NOT exhibit good atom economy according to the principles of green chemistry?
Q37.
mcq single
+1 / 0
What is the name of isopropyl alcohol according to carbinol system?
Q38.
mcq single
+1 / 0
What is the IUPAC name of following compound?
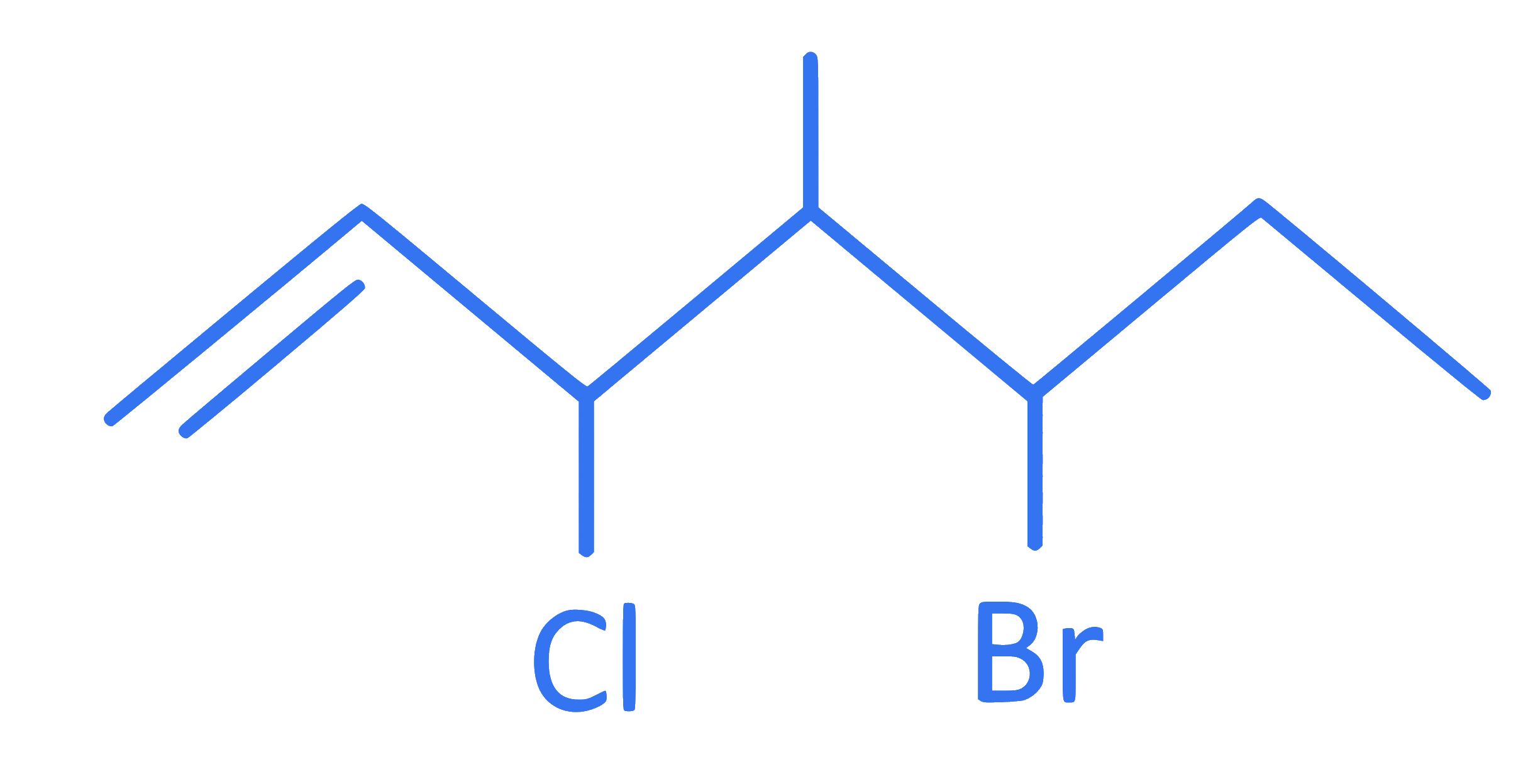

Q39.
mcq single
+1 / 0
Identify the use of calcium carbonate from following.
Q40.
mcq single
+1 / 0
Which of the following is primary benzylic alcohol?
Q41.
mcq single
+1 / 0
Identify the product ' $C$ ' formed in the following series of reactions.
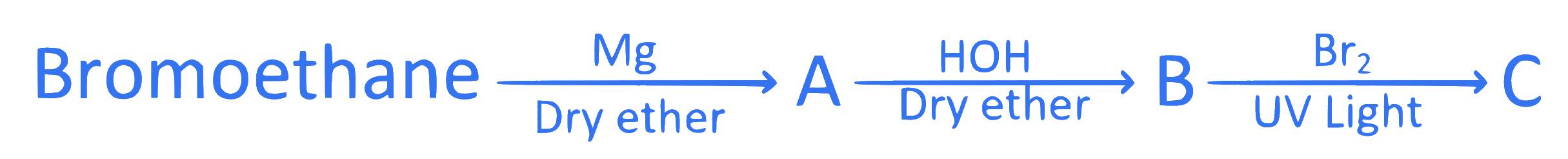
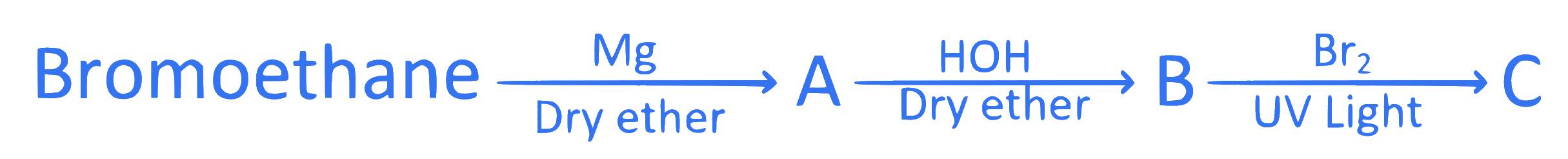
Q42.
mcq single
+1 / 0
Calculate the final pressure required to reduce the volume of a gas to one third, if initial pressure is $1.6 \times 10^5 \mathrm{Nm}^{-2}$.
Q43.
mcq single
+1 / 0
What is the number of chiral carbon atoms and number of formyl groups respectively present in ribose?
Q44.
mcq single
+1 / 0
Which amino acid from following contains highest number of N atoms in it?
Q45.
mcq single
+1 / 0
What is the minimum number of spheres required to develop an octahedral void?
Q46.
mcq single
+1 / 0
Calculate the molar mass of an element having density $19.2 \mathrm{~g} \mathrm{~cm}^{-3}$ if it forms fcc structure $\left[\mathrm{a}^3 \times \mathrm{N}_{\mathrm{A}}=40 \mathrm{~cm}^3 \mathrm{~mol}^{-1}\right]$
Q47.
mcq single
+1 / 0
Calculate the volume occupied by particle in simple cubic unit cell if volume of unit cell is $5.5 \times 10^{-22} \mathrm{~cm}^3$.
Q48.
mcq single
+1 / 0
Which of the following is selected as cathode for a galvanic cell set up with nickel anode?
Q49.
mcq single
+1 / 0
The molar conductivity of $0.02 \mathrm{~mol} \mathrm{dm} \mathrm{dm}^{-3}$ solution of sodium hydroxide is $230.5 \Omega^{-1} \mathrm{~cm}^2 \mathrm{~mol}^{-1}$. What is it's conductivity?
Q50.
mcq single
+1 / 0
What is the quantity of electricity required to liberate $112 \mathrm{~cm}^3$ of hydrogen gas at STP from acidified water?
Q51.
mcq single
+1 / 0
Which of the following isomeric amines has the highest boiling point?
Q52.
mcq single
+1 / 0
How many moles of iodomethane are consumed in the following conversion?
$$ \mathrm{CH}_3 \mathrm{NH}_2 \xrightarrow[\Delta]{\mathrm{CH}_3 \mathrm{l}}\left(\mathrm{CH}_3\right)_4 \mathrm{~N}^{+} \mathrm{I}^{-} $$
Q53.
mcq single
+1 / 0
Which element from following does NOT exhibit magnetic moment in +1 state?
Q54.
mcq single
+1 / 0
Identify false statement about transition elements.
Q55.
mcq single
+1 / 0
Which from following is a semisynthetic polymer?
Q56.
mcq single
+1 / 0
Which among the following is a monomer of natural rubber?
Q57.
mcq single
+1 / 0
What is the formal charge present on oxygen atom (numbered 1) in Lewis structure of $\mathrm{CO}_2$ ?

Q58.
mcq single
+1 / 0
What are the possible values of magnetic quantum number for $p$ orbital?
Q59.
mcq single
+1 / 0
What is the frequency of red light having wave length 750 nm ?
Q60.
mcq single
+1 / 0
Which from the following equations represents the relation between solubility ( $\mathrm{mol} \mathrm{L}^{-1}$ ) and solubility product for the salt $\mathrm{B}_2 \mathrm{~A}$ ?
Q61.
mcq single
+1 / 0
Calculate the pOH of buffer solution formed from 0.3 M weak base and 0.45 M of its salt with strong acid $\left[\mathrm{pK}_{\mathrm{b}}=4.7447\right]$
Q62.
mcq single
+1 / 0
Calculate degree of dissociation of a weak monobasic acid in 0.01 M solution if dissociation constant is $1.6 \times 10^{-5}$.
Q63.
mcq single
+1 / 0
Calculate activation energy for a reaction if it's rate doubles when temperature is raised from $20^{\circ} \mathrm{C}$ to $35^{\circ} \mathrm{C}\left(\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right)$
Q64.
mcq single
+1 / 0
If half life of a first order reaction is 10 minutes, find the time required to decrease concentration of reactant from 0.08 M to 0.02 M .
Q65.
mcq single
+1 / 0
Which of the following equation exhibits integrated rate law equation for first order reaction?
Q66.
mcq single
+1 / 0
Which from following elements is a decay product of thorium?
Q67.
mcq single
+1 / 0
Which of the following changes exhibit that nitrogen undergoes oxidation?
Q68.
mcq single
+1 / 0
Identify the product obtained when alkyl alkanoate is hydrolysed with dilute HCl .
Q69.
mcq single
+1 / 0
Identify the reagent ' R ' necessary to bring the following conversion.


Q70.
mcq single
+1 / 0
$$ \text { Identify ' } A \text { ' in the following reaction. } $$

Q71.
mcq single
+1 / 0
Which among the following is the strongest acid?
Q72.
mcq single
+1 / 0
Which from following statements is NOT true about physisorption?
Q73.
mcq single
+1 / 0
Which of the following acid strength is used to get good yield of alkyl iodide from alcohol and sodium iodide in phosphoric acid?
Q74.
mcq single
+1 / 0
$$ \text { Which among the following is haloarene? } $$
Q75.
mcq single
+1 / 0
What is the ratio of mass of nitrogen that combines with 16 parts by weight of oxygen in $\mathrm{N}_2 \mathrm{O}, \mathrm{NO}, \mathrm{NO}_2$ ?
Q76.
mcq single
+1 / 0
If $\mathrm{A}, \mathrm{B}, \mathrm{C}, \mathrm{D}$ are four different elements with outer electronic configuration as $\mathrm{A}=4 \mathrm{~s}^2 4 \mathrm{p}^4, \mathrm{~B}=4 \mathrm{~s}^2 4 \mathrm{p}^5, \mathrm{C}=5 \mathrm{~s}^2 5 \mathrm{p}^4, \mathrm{D}=5 \mathrm{~s}^2 5 \mathrm{p}^5$.
Find the element having highest ionization enthalpy $\left(\Delta_1 \mathrm{H}_1\right)$
Q77.
mcq single
+1 / 0
What is the change in internal energy for $2 \mathrm{CO}_{(\mathrm{g})}+\mathrm{O}_{2(\mathrm{~g})} \rightarrow 2 \mathrm{CO}_{2(\mathrm{~g})}$ at $25^{\circ} \mathrm{C}$ ?
( $\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \Delta \mathrm{H}=-560 \mathrm{~kJ}$ )
Q78.
mcq single
+1 / 0
Which from following terms is explained by first law of thermodynamics?
Q79.
mcq single
+1 / 0
The entropy of vaporisation of benzene is $85 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$. When 117 g of benzene vaporises at its boiling point, what is entropy change of surrounding if process is at equilibrium?
Q80.
mcq single
+1 / 0
Which from following groups exhibits +I effect?
Q81.
mcq single
+1 / 0
Calculate the Henry's law constant at $25^{\circ} \mathrm{C}$ if solubility of gas in liquid is $2.1 \times 10^{-2} \mathrm{~mol} \mathrm{dm}^{-3}$ at 0.18 bar.
Q82.
mcq single
+1 / 0
If osmotic pressure of 0.2 M aqueous glucose solution is 5 atm at 300 K . Calculate the concentration of glucose solution having osmotic pressure 2.5 atm at same temperature.
Q83.
mcq single
+1 / 0
Which from following is an example of solution of solid as solute and liquid as solvent?
Q84.
mcq single
+1 / 0
Which from following complexes is having ambidentate ligand in it?
Q85.
mcq single
+1 / 0
Identify the coordination number of aluminium in potassium trioxalatoaluminate(III).
Q86.
mcq single
+1 / 0
Which from following chemical activities does NOT exhibit good atom economy according to the principles of green chemistry?
Q87.
mcq single
+1 / 0
What is the name of isopropyl alcohol according to carbinol system?
Q88.
mcq single
+1 / 0
What is the IUPAC name of following compound?


Q89.
mcq single
+1 / 0
Identify the use of calcium carbonate from following.
Q90.
mcq single
+1 / 0
Which of the following is primary benzylic alcohol?
Q91.
mcq single
+1 / 0
Identify the product ' $C$ ' formed in the following series of reactions.
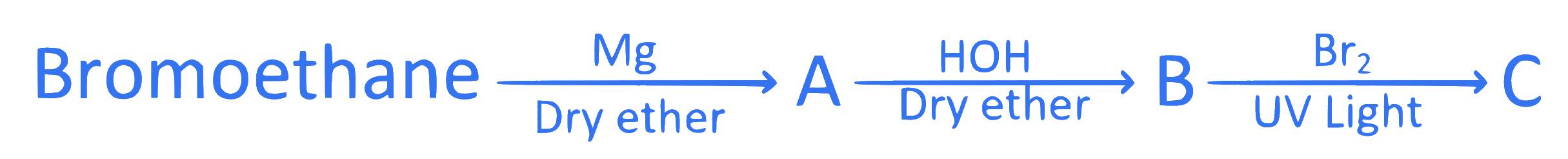
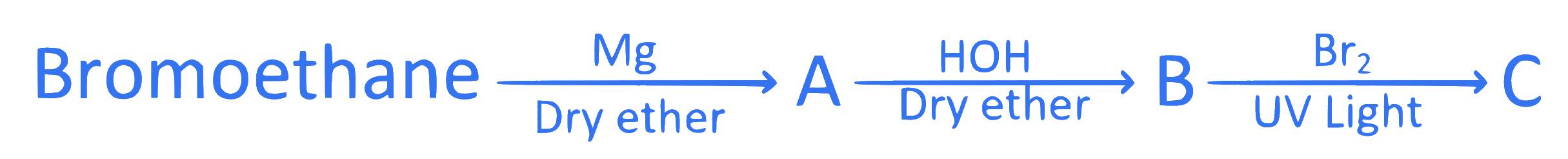
Q92.
mcq single
+1 / 0
Calculate the final pressure required to reduce the volume of a gas to one third, if initial pressure is $1.6 \times 10^5 \mathrm{Nm}^{-2}$.
Q93.
mcq single
+1 / 0
What is the number of chiral carbon atoms and number of formyl groups respectively present in ribose?
Q94.
mcq single
+1 / 0
Which amino acid from following contains highest number of N atoms in it?
Q95.
mcq single
+1 / 0
What is the minimum number of spheres required to develop an octahedral void?
Q96.
mcq single
+1 / 0
Calculate the molar mass of an element having density $19.2 \mathrm{~g} \mathrm{~cm}^{-3}$ if it forms fcc structure $\left[\mathrm{a}^3 \times \mathrm{N}_{\mathrm{A}}=40 \mathrm{~cm}^3 \mathrm{~mol}^{-1}\right]$
Q97.
mcq single
+1 / 0
Calculate the volume occupied by particle in simple cubic unit cell if volume of unit cell is $5.5 \times 10^{-22} \mathrm{~cm}^3$.
Q98.
mcq single
+1 / 0
Which of the following is selected as cathode for a galvanic cell set up with nickel anode?
Q99.
mcq single
+1 / 0
The molar conductivity of $0.02 \mathrm{~mol} \mathrm{dm} \mathrm{dm}^{-3}$ solution of sodium hydroxide is $230.5 \Omega^{-1} \mathrm{~cm}^2 \mathrm{~mol}^{-1}$. What is it's conductivity?
Q100.
mcq single
+1 / 0
What is the quantity of electricity required to liberate $112 \mathrm{~cm}^3$ of hydrogen gas at STP from acidified water?
Physics
Physics
Q1.
mcq single
+1 / 0
In a biprism experiment, fifth dark fringe is obtained at a point. A thin transparent film of refractive index ' $\mu$ ' is placed in one of the interfering paths. Now $7^{\text {th }}$ bright fringe is obtained at the same point. If ' $\lambda$ ' is the wavelength of light used, the thickness of film is equal to
Q2.
mcq single
+1 / 0
In biprism experiment the maximum intensity is ' $\mathrm{I}_0$ '. If the path difference between the two interfering waves is ' $\lambda / 4$ ' then intensity at the point on the screen is
$$ \left[\sin 45^{\circ}=\cos 45^{\circ}=\frac{1}{\sqrt{2}}\right] $$
Q3.
mcq single
+1 / 0
A pendulum is performing simple harmonic motion. The acceleration of the bob is $20 \mathrm{~cm} \mathrm{~s}^{-2}$ at a distance of 5 cm from mean position. The time period of oscillation is
Q4.
mcq single
+1 / 0
A particle of mass ' m ' is executing S.H.M. about the origin on $x$-axis with frequency $\sqrt{\frac{\mathrm{ka}}{\pi \mathrm{m}}}$, where ' $k$ ' is a constant and ' $a$ ' is the amplitude of S.H.M. If ' x ' is a displacement of a particle, at time ' $t$ ', potential energy of the particle will be
Q5.
mcq single
+1 / 0
Two particles execute S.H.M. of same amplitude and frequency along the same straight line path. They pass each other when going in opposite directions, each time their displacement is half the amplitude. The phase difference between them is $\left(\sin 30^{\circ}=0 \cdot 5\right)$
Q6.
mcq single
+1 / 0
Four point masses, each of mass ' $m$ ' are arranged in $X-Y$ plane as shown in the figure. The moment of inertia of this system about $X$-axis is
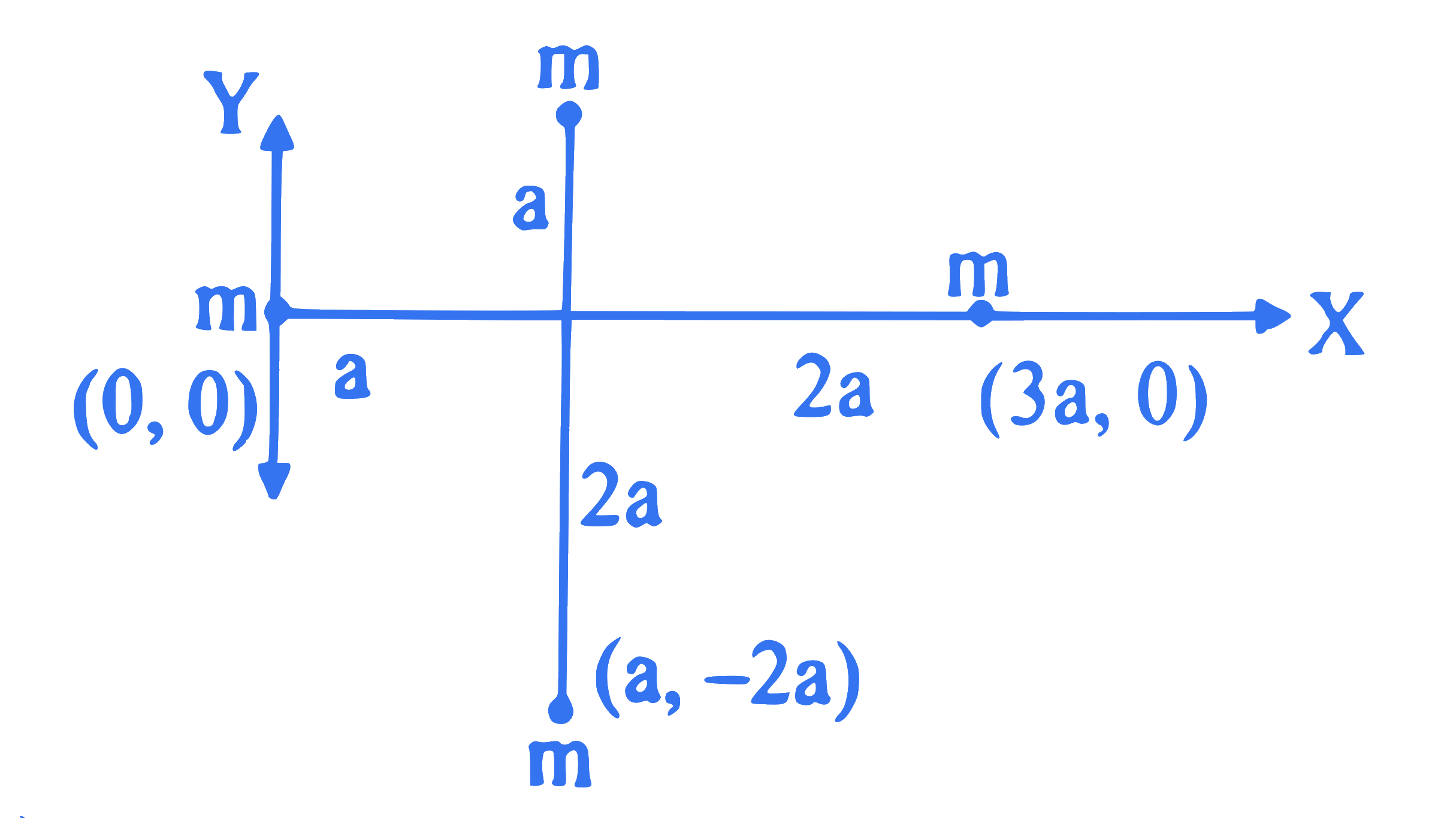

Q7.
mcq single
+1 / 0
A thin rod of length ' 4 L ' and mass ' 4 m ' is bent at the points as shown in the figure. The moment of inertia of the rod about an axis passing through point ' $O$ ' and perpendicular to plane of the paper is
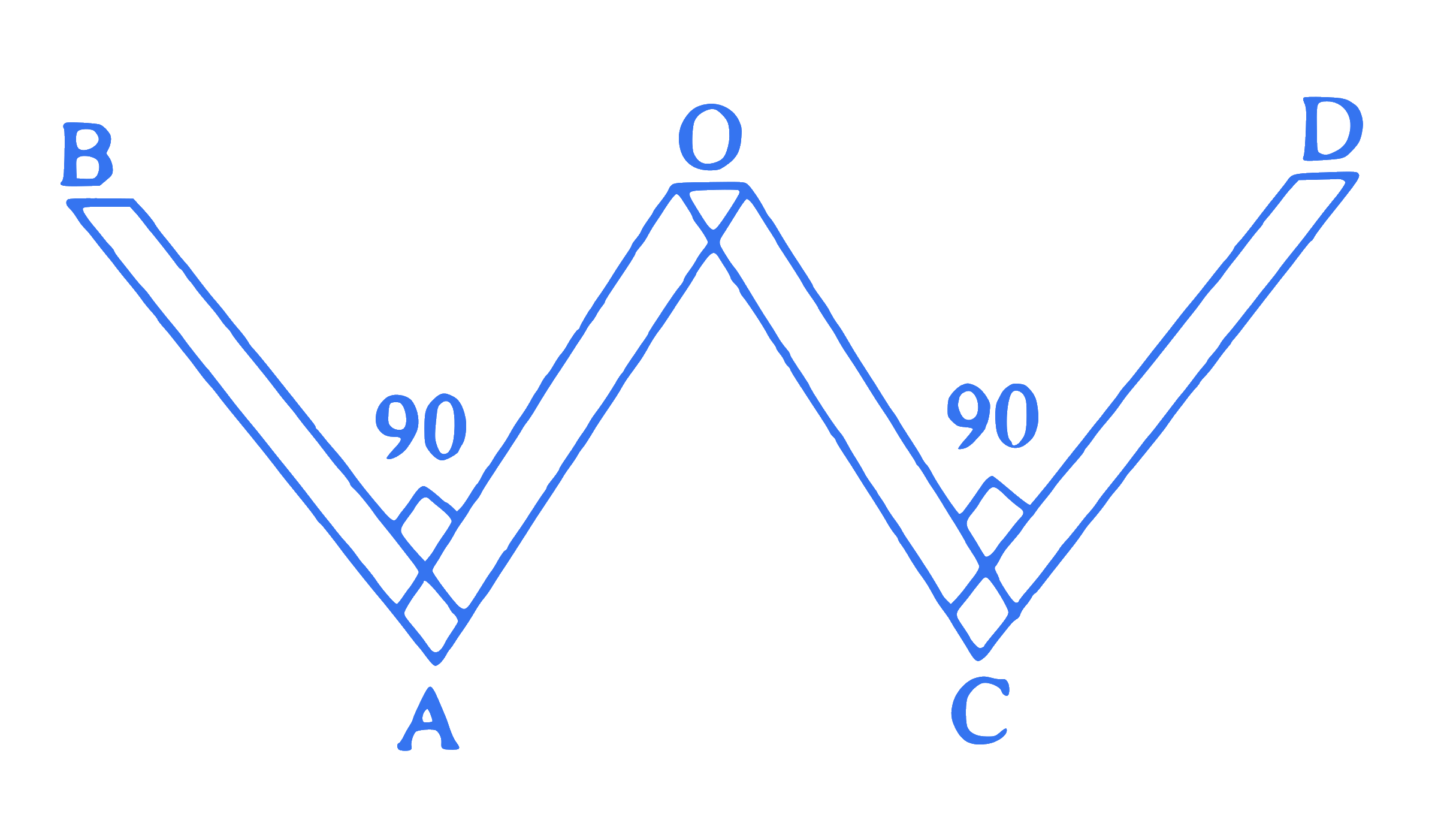

Q8.
mcq single
+1 / 0
Four masses of $1 \mathrm{~kg}, 2 \mathrm{~kg}, 3 \mathrm{~kg}$ and 4 kg are kept at co-ordinates $(0,0) \mathrm{m},(0,1) \mathrm{m}$ and $(1,0) \mathrm{m}$ respectively. Using the co-ordinates of centre of mass its position vector is
Q9.
mcq single
+1 / 0
Figure shows a rectangular frame situated in a constant magnetic field. A wire BC of length 1 m is moved out with velocity $4 \mathrm{~m} / \mathrm{s}$. Magnetic field strength is 0.15 T . Force acting on the wire $B C$ is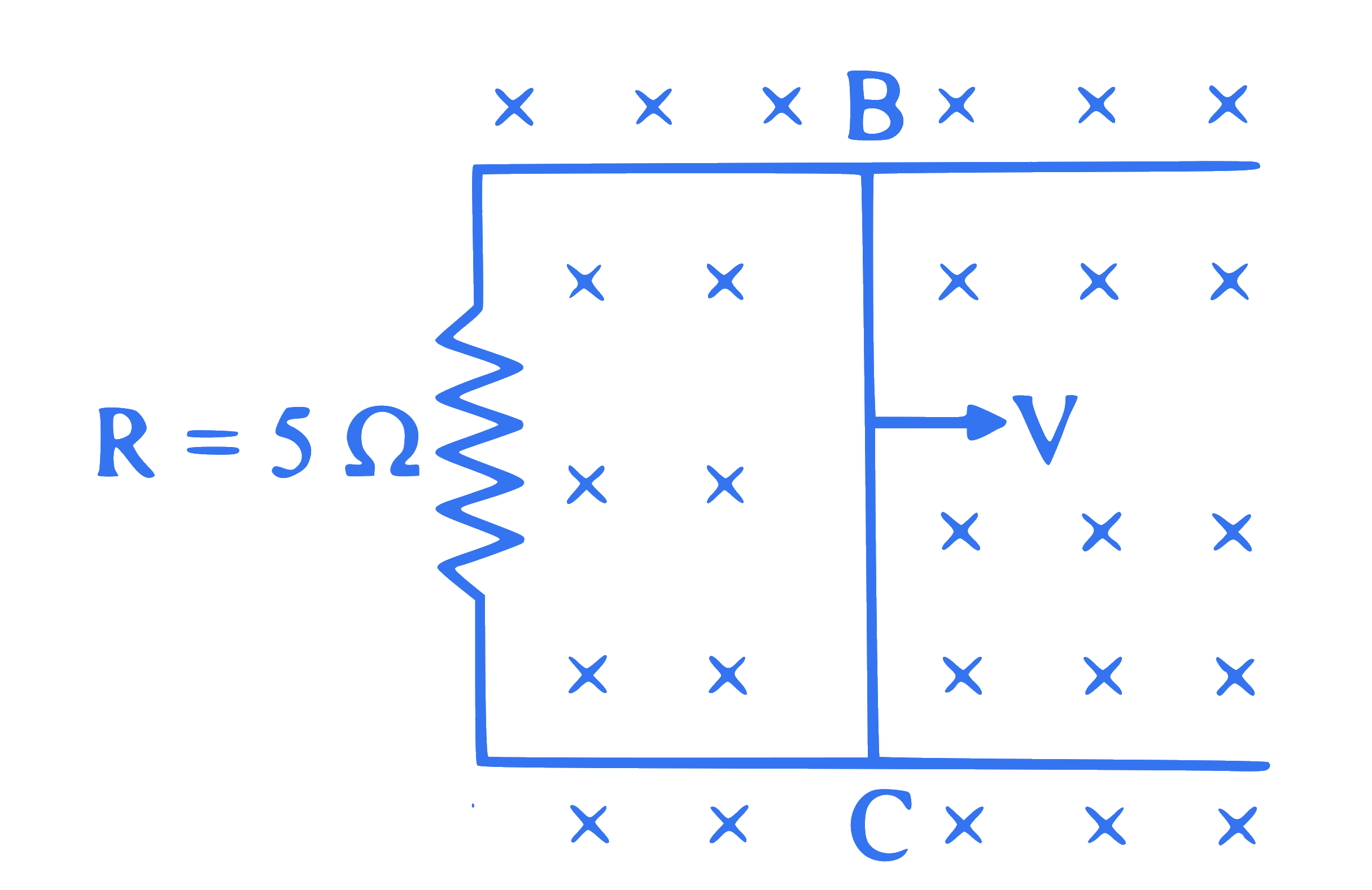
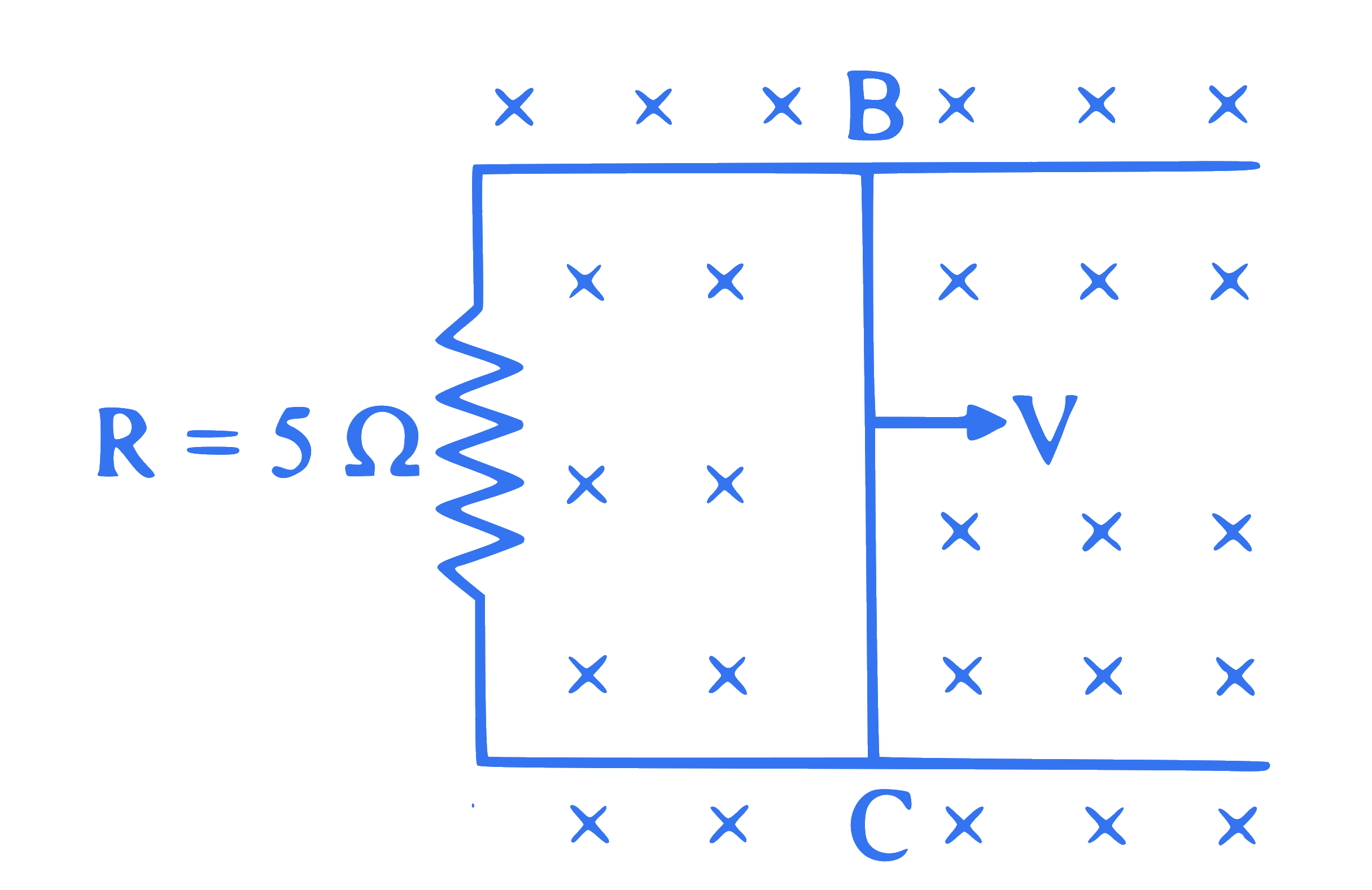
Q10.
mcq single
+1 / 0
Two different coils have self - inductance $\mathrm{L}_1=9 \mathrm{mH}$ and $\mathrm{L}_2=3 \mathrm{mH}$. The current in first coil is increased at a constant rate. The current in the second coil is also increased at the same constant rate. At certain instant of time, the power given to the two coils is same. At that time, there was current and induced voltage in the two coils. At the same instant, the ratio of the energy stored in the first coil to that in second coil is
Q11.
mcq single
+1 / 0
In case of well of death which is a vertical cylindrical wall of radius ' $r$ ' inside which vehicle is driven in horizontal circles. If ' $m$ ' is mass of vehicle, ' $V$ ' is the velocity and ' $\mu_s$ ' is the coefficient of static friction between the wheels of vehicle and walls then correct relation is [ $\mathrm{g}=$ acceleration due to gravity]
Q12.
mcq single
+1 / 0
A spherical body of radius ' $r$ ' radiates power ' $P$ ' at $T$ kelvin. If the radius $s$ halved and the temperature doubled the power radiated in the same time ' t ' will be
Q13.
mcq single
+1 / 0
The volume of a block of metal at $30^{\circ} \mathrm{C}$ changes by $0.12 \%$ when its temperature is increased to $70^{\circ} \mathrm{C}$. The coefficient of linear expansion of the metal is
Q14.
mcq single
+1 / 0
In thermodynamic process, which of the following statements is not true?
Q15.
mcq single
+1 / 0
A fixed mass of gas at constant pressure occupies a volume ' $V$ '. The gas undergoes a rise in temperature so that the r.m.s. velocity of the molecule is doubled. The new volume will be
Q16.
mcq single
+1 / 0
A monoatomic ideal gas, initially at temperature ' $\mathrm{T}_1$ '' is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature ' $\mathrm{T}_2$ ' by releasing the piston suddenly. $L_1$ and $L_2$ are the lengths of the gas columns before and after the expansion respectively. Then $\frac{T_1}{T_2}$ is
Q17.
mcq single
+1 / 0
Two stars 'A' and 'B' radiate maximum energy at $5200 \mathop {\rm{A}}\limits^ \circ$ and $6500 \mathop {\rm{A}}\limits^ \circ$ respectively. Then the ratio of absolute temperatures of stars ' A ' and ' $B$ ' is
$$\mathop {\rm{A}}\limits^ \circ $$
Q18.
mcq single
+1 / 0
A photon and an electron have an equal energy ' E '. The ratio of wavelength ' $\lambda_{\mathrm{p}}$ ' of photon to that of electron ' $\lambda_{\mathrm{e}}$ ' is proportional to
Q19.
mcq single
+1 / 0
A metal surface is illuminated by light of two different wavelengths 207 nm and 414 nm . The maximum speeds of photoelectrons corresponding to these wavelengths are $\mathrm{u}_1$ and $\dot{u}_2$ respectively with $u_1: u_2=2: 1$. The work function of the metal is $(\mathrm{hc}=1242 \mathrm{eV} \mathrm{nm})$
Q20.
mcq single
+1 / 0
In the diagram, the total electric flux through the closed surface ' S ' is
[Given $\mathrm{q}=$ charge
$\varepsilon_0=$ permittivity of free space]
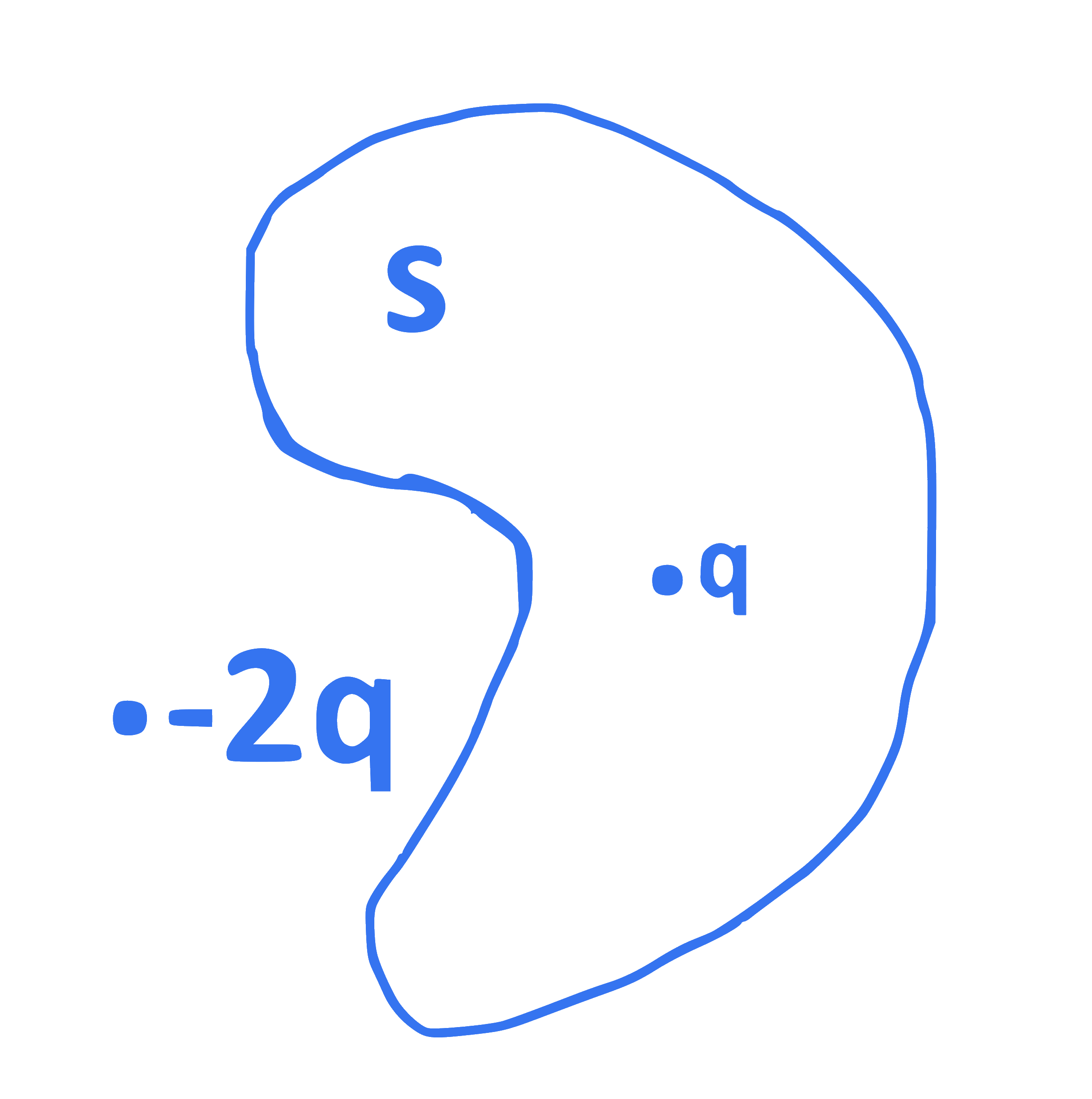
Q21.
mcq single
+1 / 0
A hollow charged metal sphere has a radius ' $r$ '. If the potential difference between its surface and a point at a distance ' 3 r ' from the centre is ' $v$ ', then the electric field intensity at a distance ' $3 r$ ' is
Q22.
mcq single
+1 / 0
A satellite is revolving round the earth with orbital speed ' $\mathrm{V}_0$ '. If it stops suddenly, the speed with which it will strike the surface of the earth would be ( $\mathrm{V}_{\mathrm{e}}=$ escape velocity of a particle on earth's surface)
Q23.
mcq single
+1 / 0
The height at which the acceleration due to gravity becomes $\frac{g}{4}$ in terms of $R$ is $[R=$ the radius of the earth $]$
Q24.
mcq single
+1 / 0
A parallel combination of two capacitors of capacities ' C ' and ' $\frac{\mathrm{C}}{3}$, respectively is connected across a battery of 12 volt. When both capacitors are fully charged, the charge and energy stored in them is $Q_1, Q_2$ and $E_1, E_2$ respectively. Then the ratio of $\left(E_1-E_2\right)$ to $\left(Q_1-Q_2\right)$ is
Q25.
mcq single
+1 / 0
In between the plates of parallel plate capacitor of plate separation 'd' a dielectric plate of thickness ' $t$ ' is inserted. The capacitance becomes one-third of the original capacity without dielectric. The dielectric constant of the plate is
Q26.
mcq single
+1 / 0
A transparent sphere of refractive index ' $\mu$ ' and radius of curvature ' $R$ ' is kept in air. A point object is placed at a distance ' d ' from the surface of the sphere so that the real image is formed at the same distance ' $d$ ' from exactly opposite side of the sphere. The distance ' $d$ ' is
Q27.
mcq single
+1 / 0
For a given medium, the speed of light and the polarising angle are ' $v$ ' and ' $i_p$ ' respectively, then ( $\mathrm{c}=$ speed of light in vaccum )
Q28.
mcq single
+1 / 0
When a ray of light is refracted from one medium to another, then the wavelength changes from $6000 \mathop {\rm{A}}\limits^ \circ$ to $4000 \mathop {\rm{A}}\limits^ \circ$. The critical angle for the interface will be
Q29.
mcq single
+1 / 0
A stone is thrown upward with a speed 'u' from the top of a tower reaches the ground with velocity ' $3 u$ '. The height of the tower is ( $g=$ acceleration due to gravity)
Q30.
mcq single
+1 / 0
A charged particle is subjected to acceleration in a cyclotron which consists of two dees ' $\mathrm{D}_1$ ' and ' $D_2$ '. The charged particle undergoes increase in its speed.
Q31.
mcq single
+1 / 0
The magnetic field at the centre of a current carrying circular coil of area ' $A$ ' is ' $B$ '. The magnetic moment of the coil is ( $\mu_0=$. permeability of free space)
Q32.
mcq single
+1 / 0
Water is flowing through a horizontal pipe in a streamline flow. At the narrowest part of the pipe
Q33.
mcq single
+1 / 0
A light metal disc of radius ' $r$ ' floats on water surface and bends the surface downwards along the perimeter making an angle ' $\theta$ ' with the vertical edge of the disc. If the weight of water displaced by the disc is ' W ', the weight of the metal disc is [ $\mathrm{T}=$ surface tension of water]
Q34.
mcq single
+1 / 0
Work done to get ' $n$ ' spherical drops of equal size from a single spherical drop of water, is proportional to
Q35.
mcq single
+1 / 0
If an electron in hydrogen atom jumps from $3^{\text {rd }}$ orbit to $2^{\text {nd }}$ orbit it emits a photon of wavelength ' $\lambda$ '. When it emits a photon from $4^{\text {th }}$ orbit to $3^{\text {rd }}$ orbit then the corresponding wavelength of emitted photon will be
Q36.
mcq single
+1 / 0
The angular momentum of electron in hydrogen atom in first orbit is ' $L$ '. The change in angular momentum if electron is in second orbit of hydrogen atom is
Q37.
mcq single
+1 / 0
Two uniform strings ' $A$ ' and ' $B$ ' made of steel are made to vibrate under same tension. If the first overtone of ' $A$ ' is equal to second overtone of ' B ' and radius of ' A ' is twice that of ' B '. Then the ratio of length of string ' $A$ ' to that of ' $B$ ' is
Q38.
mcq single
+1 / 0
The pitch of whistle of an engine appears to drop by $30 \%$ of the original value when it passes a stationary observer. If speed of sound in air is $350 \mathrm{~m} / \mathrm{s}$, then the speed of engine in $\mathrm{m} / \mathrm{s}$ is
Q39.
mcq single
+1 / 0
A pipe closed at one end vibrating in fifth overtone is in unison with open pipe vibrating in its fifth overtone. The ratio of $l_{\mathrm{c}}: l_{\mathrm{o}}$ is $\left[l_{\mathrm{c}}=\right.$ vibrating length of closed pipe, $l_0=$ vibrating length of open pipe]
Q40.
mcq single
+1 / 0
The equation of simple harmonic progressive wave is given by $y=A \sin (100 \pi t-4 x)$. The distance between two particles having a phase difference of $\left(\frac{\pi}{4}\right)^c$ is
Q41.
mcq single
+1 / 0
A solenoid is connected to a battery so that a steady current flows through it, If an iron core is inserted into the solenoid, then the current in the coil
Q42.
mcq single
+1 / 0
A solenoid having 400 turns/metre has a core of a material with relative permeability 300 . If a current of 0.5 A is passed through it, then the magnetisation of the core material is nearly
Q43.
mcq single
+1 / 0
Which one of the following is the correct equation for the electric circuit shown in the figure?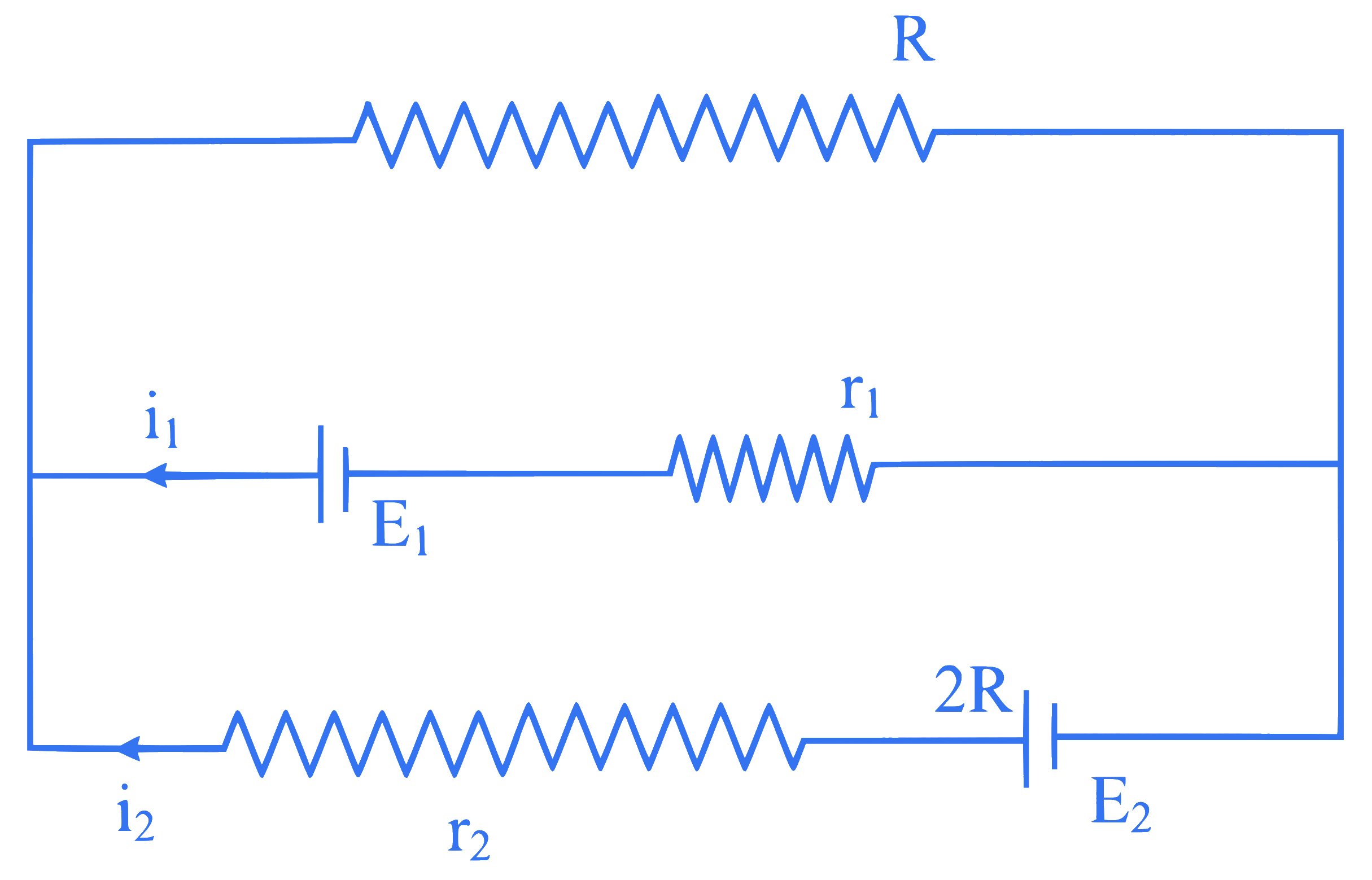

Q44.
mcq single
+1 / 0
A galvanometer of resistance ' $G$ ' can be converted into a voltmeter of range ( $0-\mathrm{V}$ ) volt by connecting a resistance ' $R$ ' in series with it. The resistance ' $R$ required to change its range from $\left(0-\frac{\mathrm{V}}{4}\right)$ volt will be
Q45.
mcq single
+1 / 0
For a common emitter transistor configuration the ratio of $\frac{\mathrm{I}_{\mathrm{C}}}{\mathrm{I}_{\mathrm{E}}}=0.96$, then the current gain in this configuration is
Q46.
mcq single
+1 / 0
The Boolean expression for $\mathrm{X}-\mathrm{OR}$ gate $C=(A \oplus B)$ is equivalent to
Q47.
mcq single
+1 / 0
Diode and resistance are connected as shown in figure. Out of the following statements which one is TRUE?
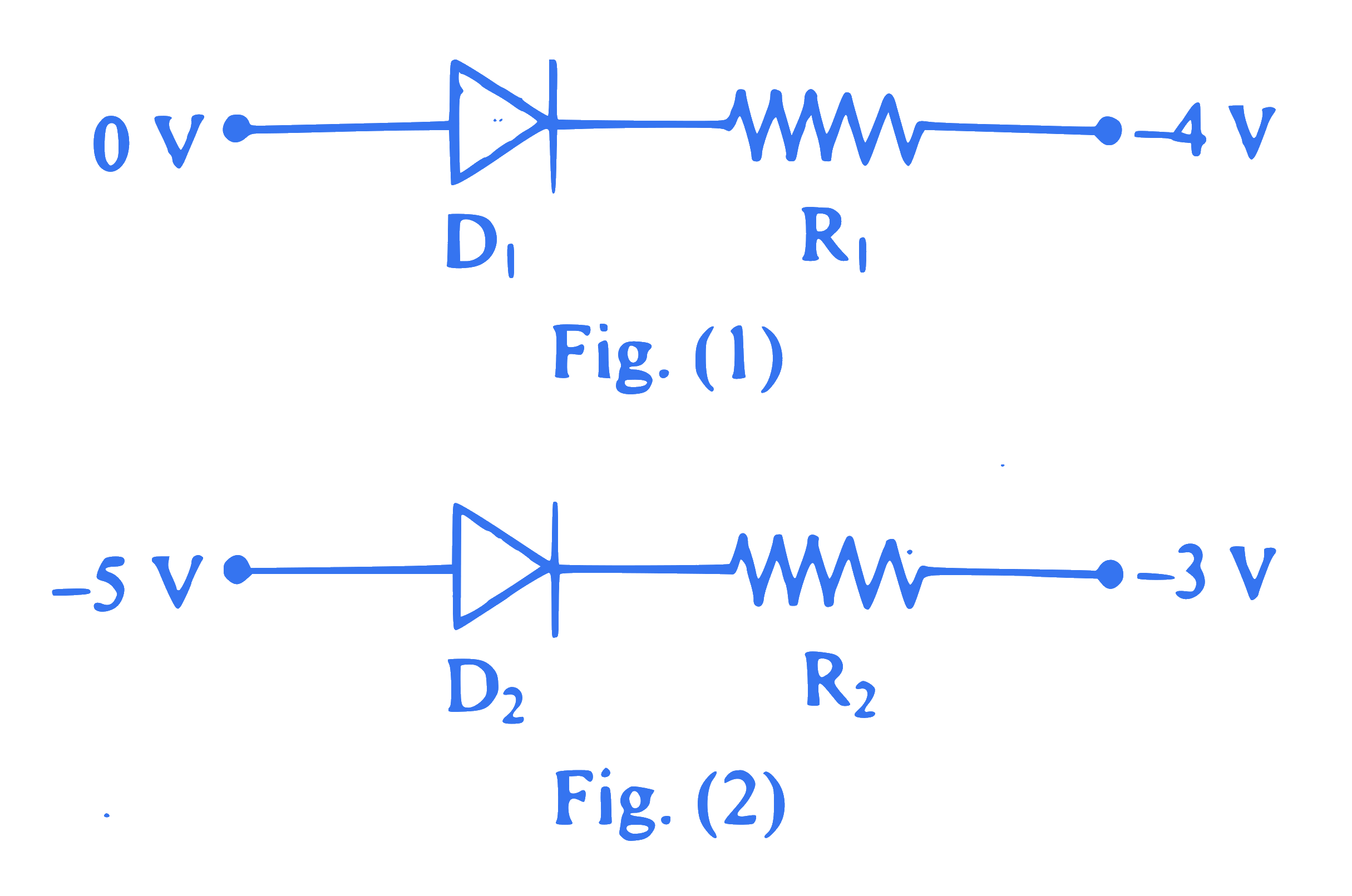

Q48.
mcq single
+1 / 0
In an oscillating LC circuit, the maximum charge on the capacitor is ' $Q$ '. When the energy is stored equally between the electric and magnetic fields, the instantaneous charge on the capacitor ' $q$ ' is
Q49.
mcq single
+1 / 0
The instantaneous value of current in an a.c. circuit is $I=2 \sin \left[100 \pi t+\frac{\pi}{3}\right]$ A. The current will be maximum for the first time at $\left(\sin 90^{\circ}=1\right)$
Q50.
mcq single
+1 / 0
The resistance of a coil for d.c. is $5 \Omega$. In a.c., the resistance will
Q51.
mcq single
+1 / 0
In a biprism experiment, fifth dark fringe is obtained at a point. A thin transparent film of refractive index ' $\mu$ ' is placed in one of the interfering paths. Now $7^{\text {th }}$ bright fringe is obtained at the same point. If ' $\lambda$ ' is the wavelength of light used, the thickness of film is equal to
Q52.
mcq single
+1 / 0
In biprism experiment the maximum intensity is ' $\mathrm{I}_0$ '. If the path difference between the two interfering waves is ' $\lambda / 4$ ' then intensity at the point on the screen is
$$ \left[\sin 45^{\circ}=\cos 45^{\circ}=\frac{1}{\sqrt{2}}\right] $$
Q53.
mcq single
+1 / 0
A pendulum is performing simple harmonic motion. The acceleration of the bob is $20 \mathrm{~cm} \mathrm{~s}^{-2}$ at a distance of 5 cm from mean position. The time period of oscillation is
Q54.
mcq single
+1 / 0
A particle of mass ' m ' is executing S.H.M. about the origin on $x$-axis with frequency $\sqrt{\frac{\mathrm{ka}}{\pi \mathrm{m}}}$, where ' $k$ ' is a constant and ' $a$ ' is the amplitude of S.H.M. If ' x ' is a displacement of a particle, at time ' $t$ ', potential energy of the particle will be
Q55.
mcq single
+1 / 0
Two particles execute S.H.M. of same amplitude and frequency along the same straight line path. They pass each other when going in opposite directions, each time their displacement is half the amplitude. The phase difference between them is $\left(\sin 30^{\circ}=0 \cdot 5\right)$
Q56.
mcq single
+1 / 0
Four point masses, each of mass ' $m$ ' are arranged in $X-Y$ plane as shown in the figure. The moment of inertia of this system about $X$-axis is


Q57.
mcq single
+1 / 0
A thin rod of length ' 4 L ' and mass ' 4 m ' is bent at the points as shown in the figure. The moment of inertia of the rod about an axis passing through point ' $O$ ' and perpendicular to plane of the paper is


Q58.
mcq single
+1 / 0
Four masses of $1 \mathrm{~kg}, 2 \mathrm{~kg}, 3 \mathrm{~kg}$ and 4 kg are kept at co-ordinates $(0,0) \mathrm{m},(0,1) \mathrm{m}$ and $(1,0) \mathrm{m}$ respectively. Using the co-ordinates of centre of mass its position vector is
Q59.
mcq single
+1 / 0
Figure shows a rectangular frame situated in a constant magnetic field. A wire BC of length 1 m is moved out with velocity $4 \mathrm{~m} / \mathrm{s}$. Magnetic field strength is 0.15 T . Force acting on the wire $B C$ is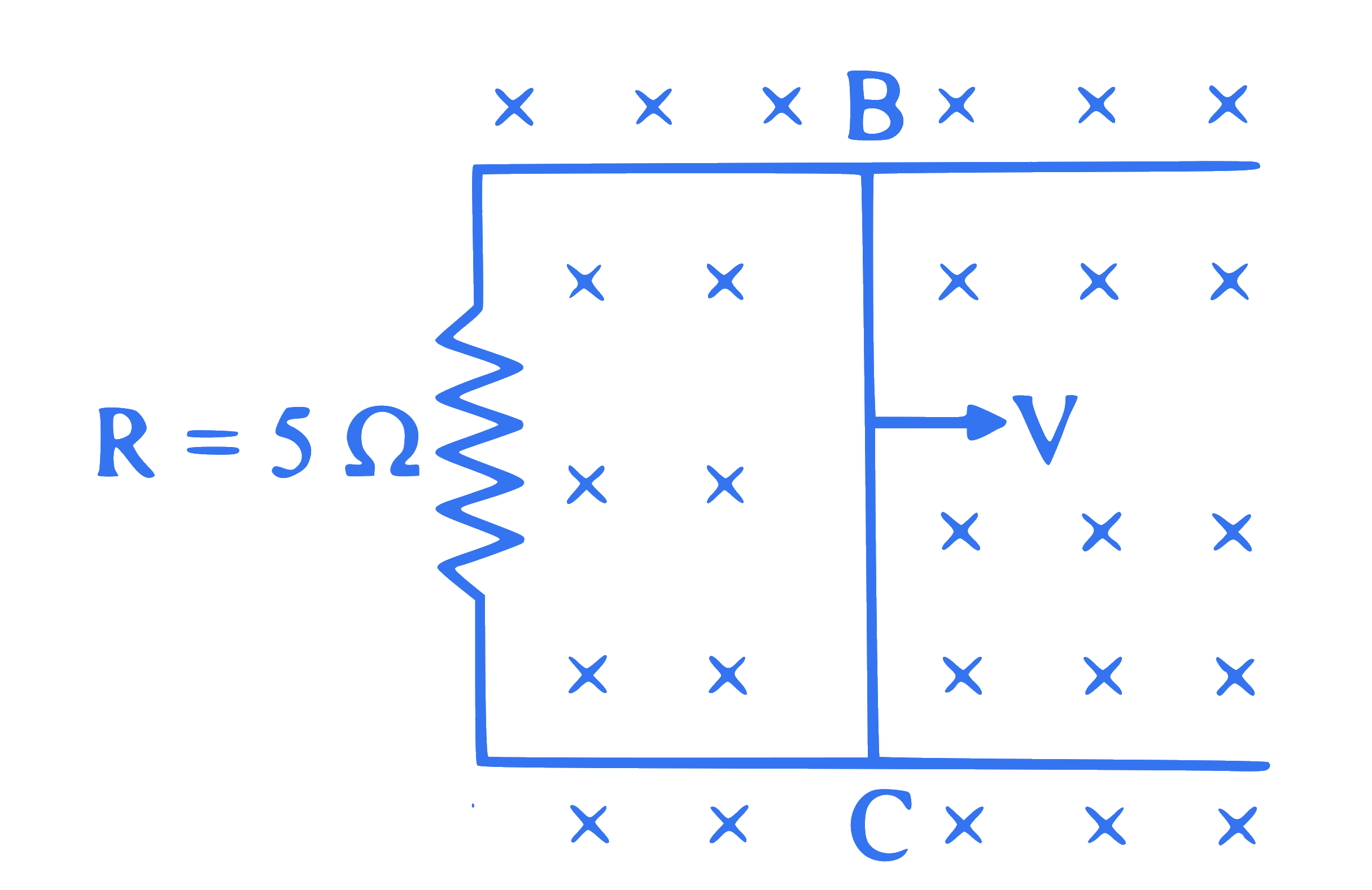
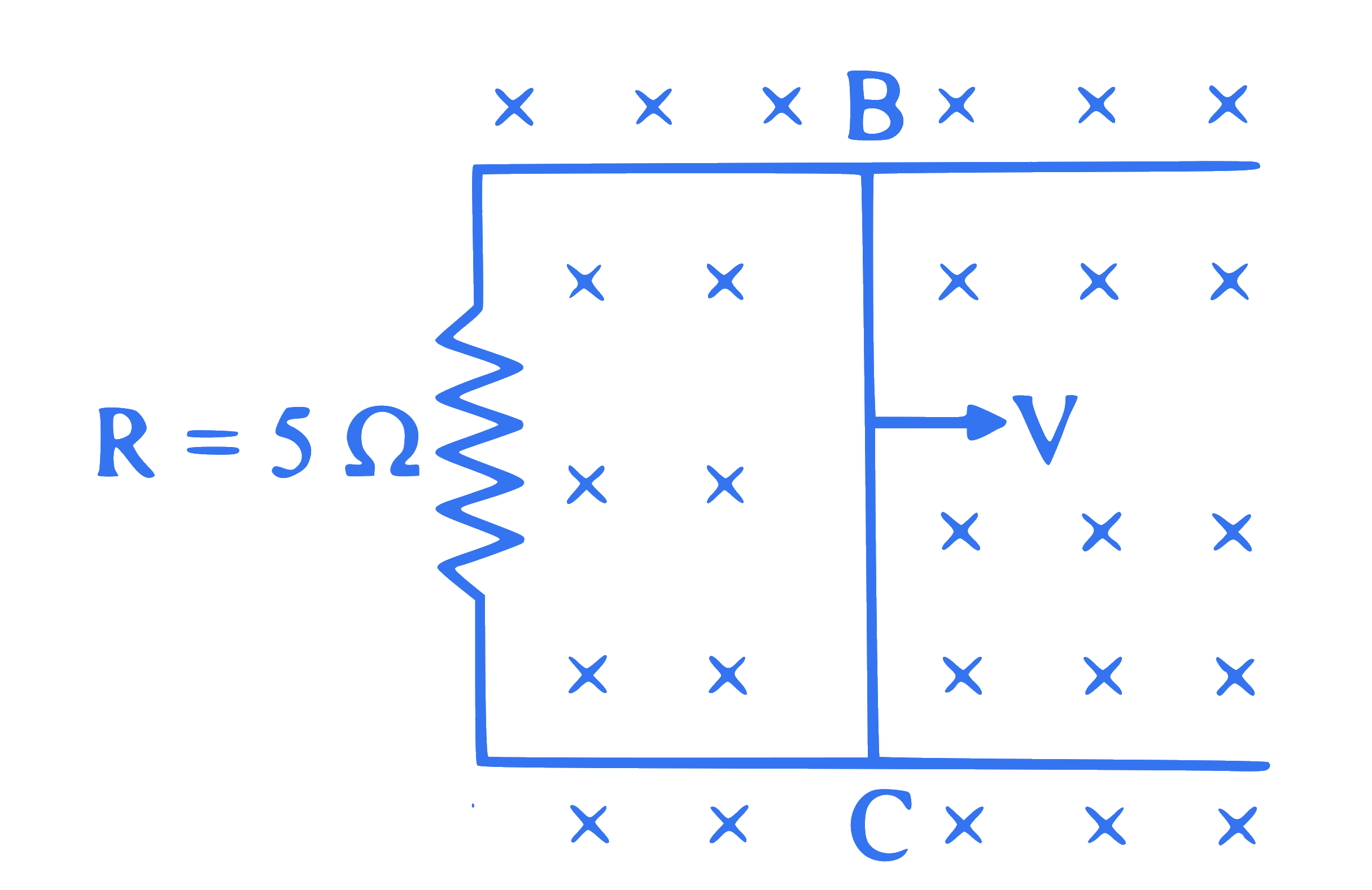
Q60.
mcq single
+1 / 0
Two different coils have self - inductance $\mathrm{L}_1=9 \mathrm{mH}$ and $\mathrm{L}_2=3 \mathrm{mH}$. The current in first coil is increased at a constant rate. The current in the second coil is also increased at the same constant rate. At certain instant of time, the power given to the two coils is same. At that time, there was current and induced voltage in the two coils. At the same instant, the ratio of the energy stored in the first coil to that in second coil is
Q61.
mcq single
+1 / 0
In case of well of death which is a vertical cylindrical wall of radius ' $r$ ' inside which vehicle is driven in horizontal circles. If ' $m$ ' is mass of vehicle, ' $V$ ' is the velocity and ' $\mu_s$ ' is the coefficient of static friction between the wheels of vehicle and walls then correct relation is [ $\mathrm{g}=$ acceleration due to gravity]
Q62.
mcq single
+1 / 0
A spherical body of radius ' $r$ ' radiates power ' $P$ ' at $T$ kelvin. If the radius $s$ halved and the temperature doubled the power radiated in the same time ' t ' will be
Q63.
mcq single
+1 / 0
The volume of a block of metal at $30^{\circ} \mathrm{C}$ changes by $0.12 \%$ when its temperature is increased to $70^{\circ} \mathrm{C}$. The coefficient of linear expansion of the metal is
Q64.
mcq single
+1 / 0
In thermodynamic process, which of the following statements is not true?
Q65.
mcq single
+1 / 0
A fixed mass of gas at constant pressure occupies a volume ' $V$ '. The gas undergoes a rise in temperature so that the r.m.s. velocity of the molecule is doubled. The new volume will be
Q66.
mcq single
+1 / 0
A monoatomic ideal gas, initially at temperature ' $\mathrm{T}_1$ '' is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature ' $\mathrm{T}_2$ ' by releasing the piston suddenly. $L_1$ and $L_2$ are the lengths of the gas columns before and after the expansion respectively. Then $\frac{T_1}{T_2}$ is
Q67.
mcq single
+1 / 0
Two stars 'A' and 'B' radiate maximum energy at $5200 \mathop {\rm{A}}\limits^ \circ$ and $6500 \mathop {\rm{A}}\limits^ \circ$ respectively. Then the ratio of absolute temperatures of stars ' A ' and ' $B$ ' is
$$\mathop {\rm{A}}\limits^ \circ $$
Q68.
mcq single
+1 / 0
A photon and an electron have an equal energy ' E '. The ratio of wavelength ' $\lambda_{\mathrm{p}}$ ' of photon to that of electron ' $\lambda_{\mathrm{e}}$ ' is proportional to
Q69.
mcq single
+1 / 0
A metal surface is illuminated by light of two different wavelengths 207 nm and 414 nm . The maximum speeds of photoelectrons corresponding to these wavelengths are $\mathrm{u}_1$ and $\dot{u}_2$ respectively with $u_1: u_2=2: 1$. The work function of the metal is $(\mathrm{hc}=1242 \mathrm{eV} \mathrm{nm})$
Q70.
mcq single
+1 / 0
In the diagram, the total electric flux through the closed surface ' S ' is
[Given $\mathrm{q}=$ charge
$\varepsilon_0=$ permittivity of free space]
Q71.
mcq single
+1 / 0
A hollow charged metal sphere has a radius ' $r$ '. If the potential difference between its surface and a point at a distance ' 3 r ' from the centre is ' $v$ ', then the electric field intensity at a distance ' $3 r$ ' is
Q72.
mcq single
+1 / 0
A satellite is revolving round the earth with orbital speed ' $\mathrm{V}_0$ '. If it stops suddenly, the speed with which it will strike the surface of the earth would be ( $\mathrm{V}_{\mathrm{e}}=$ escape velocity of a particle on earth's surface)
Q73.
mcq single
+1 / 0
The height at which the acceleration due to gravity becomes $\frac{g}{4}$ in terms of $R$ is $[R=$ the radius of the earth $]$
Q74.
mcq single
+1 / 0
A parallel combination of two capacitors of capacities ' C ' and ' $\frac{\mathrm{C}}{3}$, respectively is connected across a battery of 12 volt. When both capacitors are fully charged, the charge and energy stored in them is $Q_1, Q_2$ and $E_1, E_2$ respectively. Then the ratio of $\left(E_1-E_2\right)$ to $\left(Q_1-Q_2\right)$ is
Q75.
mcq single
+1 / 0
In between the plates of parallel plate capacitor of plate separation 'd' a dielectric plate of thickness ' $t$ ' is inserted. The capacitance becomes one-third of the original capacity without dielectric. The dielectric constant of the plate is
Q76.
mcq single
+1 / 0
A transparent sphere of refractive index ' $\mu$ ' and radius of curvature ' $R$ ' is kept in air. A point object is placed at a distance ' d ' from the surface of the sphere so that the real image is formed at the same distance ' $d$ ' from exactly opposite side of the sphere. The distance ' $d$ ' is
Q77.
mcq single
+1 / 0
For a given medium, the speed of light and the polarising angle are ' $v$ ' and ' $i_p$ ' respectively, then ( $\mathrm{c}=$ speed of light in vaccum )
Q78.
mcq single
+1 / 0
When a ray of light is refracted from one medium to another, then the wavelength changes from $6000 \mathop {\rm{A}}\limits^ \circ$ to $4000 \mathop {\rm{A}}\limits^ \circ$. The critical angle for the interface will be
Q79.
mcq single
+1 / 0
A stone is thrown upward with a speed 'u' from the top of a tower reaches the ground with velocity ' $3 u$ '. The height of the tower is ( $g=$ acceleration due to gravity)
Q80.
mcq single
+1 / 0
A charged particle is subjected to acceleration in a cyclotron which consists of two dees ' $\mathrm{D}_1$ ' and ' $D_2$ '. The charged particle undergoes increase in its speed.
Q81.
mcq single
+1 / 0
The magnetic field at the centre of a current carrying circular coil of area ' $A$ ' is ' $B$ '. The magnetic moment of the coil is ( $\mu_0=$. permeability of free space)
Q82.
mcq single
+1 / 0
Water is flowing through a horizontal pipe in a streamline flow. At the narrowest part of the pipe
Q83.
mcq single
+1 / 0
A light metal disc of radius ' $r$ ' floats on water surface and bends the surface downwards along the perimeter making an angle ' $\theta$ ' with the vertical edge of the disc. If the weight of water displaced by the disc is ' W ', the weight of the metal disc is [ $\mathrm{T}=$ surface tension of water]
Q84.
mcq single
+1 / 0
Work done to get ' $n$ ' spherical drops of equal size from a single spherical drop of water, is proportional to
Q85.
mcq single
+1 / 0
If an electron in hydrogen atom jumps from $3^{\text {rd }}$ orbit to $2^{\text {nd }}$ orbit it emits a photon of wavelength ' $\lambda$ '. When it emits a photon from $4^{\text {th }}$ orbit to $3^{\text {rd }}$ orbit then the corresponding wavelength of emitted photon will be
Q86.
mcq single
+1 / 0
The angular momentum of electron in hydrogen atom in first orbit is ' $L$ '. The change in angular momentum if electron is in second orbit of hydrogen atom is
Q87.
mcq single
+1 / 0
Two uniform strings ' $A$ ' and ' $B$ ' made of steel are made to vibrate under same tension. If the first overtone of ' $A$ ' is equal to second overtone of ' B ' and radius of ' A ' is twice that of ' B '. Then the ratio of length of string ' $A$ ' to that of ' $B$ ' is
Q88.
mcq single
+1 / 0
The pitch of whistle of an engine appears to drop by $30 \%$ of the original value when it passes a stationary observer. If speed of sound in air is $350 \mathrm{~m} / \mathrm{s}$, then the speed of engine in $\mathrm{m} / \mathrm{s}$ is
Q89.
mcq single
+1 / 0
A pipe closed at one end vibrating in fifth overtone is in unison with open pipe vibrating in its fifth overtone. The ratio of $l_{\mathrm{c}}: l_{\mathrm{o}}$ is $\left[l_{\mathrm{c}}=\right.$ vibrating length of closed pipe, $l_0=$ vibrating length of open pipe]
Q90.
mcq single
+1 / 0
The equation of simple harmonic progressive wave is given by $y=A \sin (100 \pi t-4 x)$. The distance between two particles having a phase difference of $\left(\frac{\pi}{4}\right)^c$ is
Q91.
mcq single
+1 / 0
A solenoid is connected to a battery so that a steady current flows through it, If an iron core is inserted into the solenoid, then the current in the coil
Q92.
mcq single
+1 / 0
A solenoid having 400 turns/metre has a core of a material with relative permeability 300 . If a current of 0.5 A is passed through it, then the magnetisation of the core material is nearly
Q93.
mcq single
+1 / 0
Which one of the following is the correct equation for the electric circuit shown in the figure?

Q94.
mcq single
+1 / 0
A galvanometer of resistance ' $G$ ' can be converted into a voltmeter of range ( $0-\mathrm{V}$ ) volt by connecting a resistance ' $R$ ' in series with it. The resistance ' $R$ required to change its range from $\left(0-\frac{\mathrm{V}}{4}\right)$ volt will be
Q95.
mcq single
+1 / 0
For a common emitter transistor configuration the ratio of $\frac{\mathrm{I}_{\mathrm{C}}}{\mathrm{I}_{\mathrm{E}}}=0.96$, then the current gain in this configuration is
Q96.
mcq single
+1 / 0
The Boolean expression for $\mathrm{X}-\mathrm{OR}$ gate $C=(A \oplus B)$ is equivalent to
Q97.
mcq single
+1 / 0
Diode and resistance are connected as shown in figure. Out of the following statements which one is TRUE?


Q98.
mcq single
+1 / 0
In an oscillating LC circuit, the maximum charge on the capacitor is ' $Q$ '. When the energy is stored equally between the electric and magnetic fields, the instantaneous charge on the capacitor ' $q$ ' is
Q99.
mcq single
+1 / 0
The instantaneous value of current in an a.c. circuit is $I=2 \sin \left[100 \pi t+\frac{\pi}{3}\right]$ A. The current will be maximum for the first time at $\left(\sin 90^{\circ}=1\right)$
Q100.
mcq single
+1 / 0
The resistance of a coil for d.c. is $5 \Omega$. In a.c., the resistance will
Biology
Biology
Q1.
mcq single
+1 / 0
Following are viral diseases in poultry, EXCEPT ________
Q2.
mcq single
+1 / 0
Given below are two statements:
Statement I - Toddy is made by fermenting fleshy pedicels of cashews nuts.
Statement II - Fenny is made by fermenting the sugar sap extracted from palm plants and coconut palm.
In light of above statements, select the correct answer from the option given below.
Q3.
mcq single
+1 / 0
How many of the following are the symbiotic nitrogen fixing micro-organisms?
Rhizobium, Anabaena, Frankia, Azotobacter, Nostoc, Clostridium, Beijerinckia, Klebsiella
Q4.
mcq single
+1 / 0
Given below are two statements.
Statement I - An ecosystem is a self regulatory and self sustaining structural and functional unit of nature.
Statement II - Entire biosphere can be considered as one global ecosystem.
In light of above statements, choose the most appropriate answer from the options given below.
Q5.
mcq single
+1 / 0
Primary productivity in an ecosystem refers to the rate of generation of _________ .
Q6.
mcq single
+1 / 0
Which of the following characteristics are seen in a stable community?
i. It is strong enough to withstand environmental disturbance and recovers quickly.
ii. It is resistant to invasive species.
iii. It exhibits constant change in biomass production over a period of time.
iv. It has more species diversity.
Q7.
mcq single
+1 / 0
Which of the following is INCORRECT about Primary and Secondary ecological successions?
Q8.
mcq single
+1 / 0
Given below are two statements.
Statement I - Biome constitute, a large regional terrestrial unit delimited by a specific climate zone having major vegetation zone and associated fauna.
Statement II - Biome is the fourth level of ecological hierarchy.
In light of above statements, select the correct answer from the option given below.
Q9.
mcq single
+1 / 0
Which of the following are the initial and final steps during the process of ecological succession?
Q10.
mcq single
+1 / 0
Monohybrid crosses are useful in demonstrating which of the following laws of inheritance suggested by Mendel?
Q11.
mcq single
+1 / 0
Which of the following statements are correct?
i. $X$ chromosome has large amount of euchromatin.
ii. heterochromatin is genetically inert.
iii. Both X and Y chromosomes are homologous.
iv. Crossing over does not take place in sex chromosomes in female.
Choose the correct option.
Q12.
mcq single
+1 / 0
Match the disorder in column I with their nature in column II.
Column - I
Column - II
i.
Thalassemia
a.
X monosomy
ii.
Turner's syndrome
b.
Extra X chromosomes in male
iii
Klinefelter's syndrome
c.
$$
21^{\text {st }} \text { Trisomy }
$$
iv.
Down's syndrome
d.
Mendelian disorder
Q13.
mcq single
+1 / 0
Which one of the following is an EXCEPTION to the generalization made by Mendel on the basis of his experiments on garden pea plant?
Q14.
mcq single
+1 / 0
Given below are two statements:
Statement I - Back cross is the crossing of $F_1$ hybrid with one of the two parents from which they were derived.
Statement II - Test cross involves the crossing of $F_1$ - hybrid with its homozygous recessive parent.
In light of above statements, choose the correct answer from the option given below.
Q15.
mcq single
+1 / 0
Given below are two statements.
Statement I - Nervous tissue is without lymphatic vessels.
Statement II - Nervous tissue is endodermal in origin.
In light of above statements, choose the most appropriate correct answer from the option given below.
Q16.
mcq single
+1 / 0
Given below are two statements:
Statement I - The muscularis of intestine can be differentiated into longitudinal and oblique muscles only.
Statement II - The muscularis of stomach is made up of outer- longitudinal, middle oblique and inner circular muscles.
In light of above statements, choose the most appropriate answer from the option given below.
Q17.
mcq single
+1 / 0
Complete the following analogy.
Parietal wall of Bowman's capsule : squamous epithelium : : PCT : ___________ epithelium.
Q18.
mcq single
+1 / 0
Complete the following analogy with respect to classification of mammals.
$$ \text { Marsupials : (i) :: monotremes : (ii) } $$
Q19.
mcq single
+1 / 0
Respiratory centres that control the rate and depth of breathing are located in __________
Q20.
mcq single
+1 / 0
Haemoglobin has maximum affinity for
Q21.
mcq single
+1 / 0
Select INCORRECT statements with respects transport of $\mathrm{CO}_2$ by RBCs and plasma.
i. $23 \%$ of $\mathrm{CO}_2$ released from tissue cells diffuse into plasma first and then into RBCs.
ii. Within RBCs carbonic acid immediately dissociates into $\mathrm{H}^{+}$and $\mathrm{HCO}_3^{-}$.
iii. In RBCs $\mathrm{CO}_2$ combines with $\mathrm{H}_2 \mathrm{O}$ to form carbonic acid.
iv. $\mathrm{H}^{+}$ions move out of RBCs and combine with $\mathrm{Na}^{+}$to form $\mathrm{NaHCO}_3$.
v. $7 \%$ of $\mathrm{CO}_2$ is transported in dissolved form as carbonic acid in plasma.
In the light of above statements choose the correct answer from option given below.
Q22.
mcq single
+1 / 0
The _______ gene from Bacillus thuringiensis produces a protein that forms crystalline inclusions in bacterial spores.
Q23.
mcq single
+1 / 0
Arrange the following in sequence during the transmission of nerve impulse at chemical synapse.
i. Neurotransmitter binds with receptors of post synaptic cell.
ii. Calcium channels open and the calcium diffuses inward from ECF.
iii. Impulse travels along the axon of presynaptic neuron.
iv. Release of neurotransmitter by exocytosis.
v. Synaptic vesicles fuse with cell membrane of axon terminal of presynaptic neuron.
Q24.
mcq single
+1 / 0
Lateral sulcus separates ________ .
Q25.
mcq single
+1 / 0
Match column I with column II and select the correct option.
Column I
Column II
i.
Frontal lobe
a.
Gustato receptors
ii.
Parietal lobe
b.
Olfactory receptors
iii.
Temporal lobe
c.
Sense of vision
iv.
Occipital lobe
d.
Broca's area
Q26.
mcq single
+1 / 0
CSF is secreted by
Q27.
mcq single
+1 / 0
Complete the following reaction by replacing (Y) with proper word.
$$ \text { Emulsified fats } \xrightarrow{\text { Lipases }} \text { Fatty acids }+(\mathrm{Y}) $$
Q28.
mcq single
+1 / 0
Chemical digestion of which one of the following starts first in human alimentary canal?
Q29.
mcq single
+1 / 0
Given below are two statements.
Statement I - Root hair is composed of two layers.
Statement II - Outer layer of root hair cell wall is composed of cellulose and inner layer is made up of pectin.
In light of above statements, choose the most appropriate answer from the option given below.
Q30.
mcq single
+1 / 0
Two different amino acids are never encoded by the same codon, this character of genetic code is called ____________.
Q31.
mcq single
+1 / 0
All the 64 codons in the dictionary of genetic code were deciphered by __________
Q32.
mcq single
+1 / 0
The size of mRNA is generally related to $\qquad$
Q33.
mcq single
+1 / 0
In the processing of hnRNA in eukaryotic cell, the primary transcripts are processed in the following sequence.
Q34.
mcq single
+1 / 0
Given below are two statements:
Statement I - In prokaryotes, there is only replicon however in eukaryotes, these are several replicons in tandem.
Statement II - Two separated strands in a replicon of DNA are prevented from re-joining by helicase enzyme.
In light of above statements, choose the correct answer from the option given below.
Q35.
mcq single
+1 / 0
In which of the following, bilirubin is excreted in urine?
Q36.
mcq single
+1 / 0
Which one of the following diseases is caused by a fungus?
Q37.
mcq single
+1 / 0
In an antibody, disulphide bonds are present___________
i. between two heavy chains
ii. between two light chains
iii. between the constant region of light chain and the constant region of a heavy chain
iv. between antigen binding sites of both the heavy and light chains
v. between the variable region of light chain and that of heavy chain
Select the correct answer from the options given below
Q38.
mcq single
+1 / 0
Cells of malignant tumor spread from one organ to other via blood or lymph and cause new tumor. This property is called ___________ .
Q39.
mcq single
+1 / 0
Angina pectoris is severe pain and heaviness in chest that is mainly caused due to
Q40.
mcq single
+1 / 0
Atherosclerosis can be treated with recombinant protein called __________
Q41.
mcq single
+1 / 0
With reference to adolescence, what is mhGAP?
Q42.
mcq single
+1 / 0
The water which percolates deep in the soil is ___________
Q43.
mcq single
+1 / 0
Given below are two statements:
Statement I - During night, guard cells become flaccid.
Statement II - During day time, guard cells become turgid due to endosmosis.
In light of above statements, choose the most appropriate answer from the option given below.
Q44.
mcq single
+1 / 0
Capillarity theory was proposed by __________
Q45.
mcq single
+1 / 0
Accessory cells of stomata are reservoirs of_________.
Q46.
mcq single
+1 / 0
The term 'Niche' denotes the _________ role played by an organism in its environment.
Q47.
mcq single
+1 / 0
Dispersal of pollens is an example of
Q48.
mcq single
+1 / 0
Given below are two statements.
Statement I - Calotropis growing in abandoned fields is never consumed by cattle and goats.
Statement II - Calotropis produces highly poisonous cardiac glycosides.
In light of above statements, select the most appropriate answer from the option given below.
Q49.
mcq single
+1 / 0
The highest level of ecological hierarchy in ecological organization is
Q50.
mcq single
+1 / 0
Match the valves of human heart in column I with the respective opening they guard in column II and select the correct option.
Column - I
Column - II
i.
Eustachian valve
a.
Opening
of pulmonary aorta
ii.
Thebesian valve
b.
Left atrioventricular aperture
iii
Mitral valve
c.
opening of inferior vena cava
iv.
Semilunar valve
d.
opening of coronary sinus
Q51.
mcq single
+1 / 0
Mature erythrocytes are enucleated in following animals EXCEPT
i. Rat
ii. Monkey
iii. Llama
iv. Camel
v. Elephant Choose the correct answer from options given below.
Q52.
mcq single
+1 / 0
Which one of the following cells secrete heparin, histamine and serotonin?
Q53.
mcq single
+1 / 0
Blood plasma transports oxygen in dissolved state, percentage of which is ________ .
Q54.
mcq single
+1 / 0
In eukaryotic organisms, replication of DNA occurs in _________
Q55.
mcq single
+1 / 0
Hormone causing vigorous contraction of myometrium to initiate parturition is ___________ .
Q56.
mcq single
+1 / 0
The fully developed foetus gives signals for uterine contractions by secreting __________ hormones.
Q57.
mcq single
+1 / 0
Complete the analogy with respect to structure of sperm
Nebenkern : ' X ' : : Acrosome is formed from : ' Y '
Q58.
mcq single
+1 / 0
During oogenesis the meiotic division is arrested at________ before fertilization.
Q59.
mcq single
+1 / 0
Median vertical depression of external genitalia in females, enclosing the urethral and vaginal opening is _________
Q60.
mcq single
+1 / 0
Complete the analogy with respect to reproductive system.
Male : Penis : : Female : __________ .
Q61.
mcq single
+1 / 0
Blood osmolarity decreases with the secretion of
i. ADH
ii. aldosterone
iii. angiotensin II
iv. ANP
Q62.
mcq single
+1 / 0
Identify label ' X ' from the given diagram and name the cells, which regulate concentration of calcium and phosphorous in the blood and select the correct option given below.
Q63.
mcq single
+1 / 0
Islets of Langerhans are _________ .
Q64.
mcq single
+1 / 0
Which one of the following pair of hormones is NOT antagonistic?
Q65.
mcq single
+1 / 0
Hypersecretion of glucocorticoid leads to___________
Q66.
mcq single
+1 / 0
Identify the INCORRECT statement.
Q67.
mcq single
+1 / 0
Match the scientist in column I with their contribution in column II and choose the correct option.
Column - I
Column - II
i.
Alexander Van Humboldt
a.
Estimated 7 million species round the globe
ii.
Rober ay
b.
Rivet popper hypothesis
iii
David TIllman
c.
Species Area Relationship
iv.
Paul Ehrlich
d.
Productivity stability hypothesis
Q68.
mcq single
+1 / 0
Damage to biodiversity takes place due to natural reasons which includes the following EXCEPT
Q69.
mcq single
+1 / 0
Phenotypically tall plants can be obtained from genetically dwarf maize plants by application of_________
Q70.
mcq single
+1 / 0
Which plant hormone is called antitranspirant?
Q71.
mcq single
+1 / 0
While studying flowering behaviour in __________ and ___________ plant, Garner and Allard discovered photoperiodism.
Q72.
mcq single
+1 / 0
The number of ATP molecules generated in aerobic respiration after oxidation of two molecules of pyruvic acid is.
Q73.
mcq single
+1 / 0
Given below are two statements:
Statement I - Reactions involved in Krebs cycle are anabolic and catabolic.
Statement II - During oxidation of acetyl Co-A, stepwise oxidation of acetyl part of acetyl Co-A occurs.
In light of above statements, choose the most appropriate answer from the option given below.
Q74.
mcq single
+1 / 0
The only 5C intermediate formed during reactions of TCA cycle is __________
Q75.
mcq single
+1 / 0
The number of reduced coenzymes $\mathrm{NADH}+\mathrm{H}^{+}$ formed during complete oxidation of one molecule of glucose.
Q76.
mcq single
+1 / 0
Homo sapiens are most closely related to________
Q77.
mcq single
+1 / 0
DDT resistant mosquitoes and different beak sizes of African seed cracker finches show
_________ type of selection respectively.
Q78.
mcq single
+1 / 0
Which one of the following is the most primitive ancestor of Homo sapiens?
Q79.
mcq single
+1 / 0
An organ with sphincters at its origin is
Q80.
mcq single
+1 / 0
Tubular fluid tends to become acidic in the_________ of nephron.
Q81.
mcq single
+1 / 0
Given below are two statements.
Statement I - Defecation is an involuntary process that takes place through anal opening guarded by sphincter muscles.
Statement II - Distension, of rectum stimulates pressure sensitive receptors that initiate a neural reflex for egestion.
In light of above statements, select the correct answer from the option given below.
Q82.
mcq single
+1 / 0
People who consume high protein diet can develop __________ stones.
Q83.
mcq single
+1 / 0
All are events of pollen-pistil interaction EXCEPT
Q84.
mcq single
+1 / 0
In sea grass, pollens are long, ribbon like and without exine exhibiting __________ pollination.
Q85.
mcq single
+1 / 0
In a male gametophyte, ploidy of tube cell and a male gamete is __________
Q86.
mcq single
+1 / 0
In an angiospermic embryo, upper swollen suspensor cell towards the micropyle, functions as_________.
Q87.
mcq single
+1 / 0
Bird pollinated flowers are usually __________
Q88.
mcq single
+1 / 0
Just before fertilization, the angiosperm's embryo sac contains_________ and ___________nuclei.
Q89.
mcq single
+1 / 0
Just before fertilization, the angiosperm's embryo sac contains ________ and ________ nuclei.
Q90.
mcq single
+1 / 0
Given below are two statements.
Statement I - Oxaloacetate is useful as a precursor for synthesis of aspartic acid.
Statement II - $\alpha$-ketoglutarate is useful for synthesis of glutamic acid.
In light of above statements, select the correct answer from the option given below.
Q91.
mcq single
+1 / 0
The number of amino groups in the amino acids present in amides is __________
Q92.
mcq single
+1 / 0
Match column I with column II and select the correct option
Column - I
Column - II
i.
Carrot and spinach
a.
High protein content
ii.
Bitter gourd
b.
More Vit A and minerals
iii
Maize
c.
More Vit C
iv.
Wheat Atlas-66
c.
Twice the amount of lysine and tryptophan
Q93.
mcq single
+1 / 0
In large proteins, peptide chains are much looped, twisted and folded back on themselves. The folded structure is due to the formation of________bonds
Q94.
mcq single
+1 / 0
Enzymes are needed only in small quantities to catalyse reactions because
Q95.
mcq single
+1 / 0
Secondary metabolites like glucosinolates are produced by cabbage to
Q96.
mcq single
+1 / 0
Agarose gel electrophoresis is used to separate DNA fragments. The DNA fragments separate due to __________ .
Q97.
mcq single
+1 / 0
When the phage DNA enters the host bacterium, the host cell protects itself from the viral DNA attacks with the help of _________ enzyme.
Q98.
mcq single
+1 / 0
The restriction enzymes used as molecular scissors are type of ____________
Q99.
mcq single
+1 / 0
Match elements given in Column I with their deficiency symptoms shown by plants given in Column II
Column - I
Column - II
i.
Eustachian valve
a.
Brown heart disease
ii.
Boron
b.
Malformed leaves
iii
Zinc
c.
Poor growth of the plant
iv.
Copper
c.
Die-back of shoots
Q100.
mcq single
+1 / 0
Which of the following bacteria do not solubilize the insoluble rock phosphate?
Q101.
mcq single
+1 / 0
Following are viral diseases in poultry, EXCEPT ________
Q102.
mcq single
+1 / 0
Given below are two statements:
Statement I - Toddy is made by fermenting fleshy pedicels of cashews nuts.
Statement II - Fenny is made by fermenting the sugar sap extracted from palm plants and coconut palm.
In light of above statements, select the correct answer from the option given below.
Q103.
mcq single
+1 / 0
How many of the following are the symbiotic nitrogen fixing micro-organisms?
Rhizobium, Anabaena, Frankia, Azotobacter, Nostoc, Clostridium, Beijerinckia, Klebsiella
Q104.
mcq single
+1 / 0
Given below are two statements.
Statement I - An ecosystem is a self regulatory and self sustaining structural and functional unit of nature.
Statement II - Entire biosphere can be considered as one global ecosystem.
In light of above statements, choose the most appropriate answer from the options given below.
Q105.
mcq single
+1 / 0
Primary productivity in an ecosystem refers to the rate of generation of _________ .
Q106.
mcq single
+1 / 0
Which of the following characteristics are seen in a stable community?
i. It is strong enough to withstand environmental disturbance and recovers quickly.
ii. It is resistant to invasive species.
iii. It exhibits constant change in biomass production over a period of time.
iv. It has more species diversity.
Q107.
mcq single
+1 / 0
Which of the following is INCORRECT about Primary and Secondary ecological successions?
Q108.
mcq single
+1 / 0
Given below are two statements.
Statement I - Biome constitute, a large regional terrestrial unit delimited by a specific climate zone having major vegetation zone and associated fauna.
Statement II - Biome is the fourth level of ecological hierarchy.
In light of above statements, select the correct answer from the option given below.
Q109.
mcq single
+1 / 0
Which of the following are the initial and final steps during the process of ecological succession?
Q110.
mcq single
+1 / 0
Monohybrid crosses are useful in demonstrating which of the following laws of inheritance suggested by Mendel?
Q111.
mcq single
+1 / 0
Which of the following statements are correct?
i. $X$ chromosome has large amount of euchromatin.
ii. heterochromatin is genetically inert.
iii. Both X and Y chromosomes are homologous.
iv. Crossing over does not take place in sex chromosomes in female.
Choose the correct option.
Q112.
mcq single
+1 / 0
Match the disorder in column I with their nature in column II.
Column - I
Column - II
i.
Thalassemia
a.
X monosomy
ii.
Turner's syndrome
b.
Extra X chromosomes in male
iii
Klinefelter's syndrome
c.
$$
21^{\text {st }} \text { Trisomy }
$$
iv.
Down's syndrome
d.
Mendelian disorder
Q113.
mcq single
+1 / 0
Which one of the following is an EXCEPTION to the generalization made by Mendel on the basis of his experiments on garden pea plant?
Q114.
mcq single
+1 / 0
Given below are two statements:
Statement I - Back cross is the crossing of $F_1$ hybrid with one of the two parents from which they were derived.
Statement II - Test cross involves the crossing of $F_1$ - hybrid with its homozygous recessive parent.
In light of above statements, choose the correct answer from the option given below.
Q115.
mcq single
+1 / 0
Given below are two statements.
Statement I - Nervous tissue is without lymphatic vessels.
Statement II - Nervous tissue is endodermal in origin.
In light of above statements, choose the most appropriate correct answer from the option given below.
Q116.
mcq single
+1 / 0
Given below are two statements:
Statement I - The muscularis of intestine can be differentiated into longitudinal and oblique muscles only.
Statement II - The muscularis of stomach is made up of outer- longitudinal, middle oblique and inner circular muscles.
In light of above statements, choose the most appropriate answer from the option given below.
Q117.
mcq single
+1 / 0
Complete the following analogy.
Parietal wall of Bowman's capsule : squamous epithelium : : PCT : ___________ epithelium.
Q118.
mcq single
+1 / 0
Complete the following analogy with respect to classification of mammals.
$$ \text { Marsupials : (i) :: monotremes : (ii) } $$
Q119.
mcq single
+1 / 0
Respiratory centres that control the rate and depth of breathing are located in __________
Q120.
mcq single
+1 / 0
Haemoglobin has maximum affinity for
Q121.
mcq single
+1 / 0
Select INCORRECT statements with respects transport of $\mathrm{CO}_2$ by RBCs and plasma.
i. $23 \%$ of $\mathrm{CO}_2$ released from tissue cells diffuse into plasma first and then into RBCs.
ii. Within RBCs carbonic acid immediately dissociates into $\mathrm{H}^{+}$and $\mathrm{HCO}_3^{-}$.
iii. In RBCs $\mathrm{CO}_2$ combines with $\mathrm{H}_2 \mathrm{O}$ to form carbonic acid.
iv. $\mathrm{H}^{+}$ions move out of RBCs and combine with $\mathrm{Na}^{+}$to form $\mathrm{NaHCO}_3$.
v. $7 \%$ of $\mathrm{CO}_2$ is transported in dissolved form as carbonic acid in plasma.
In the light of above statements choose the correct answer from option given below.
Q122.
mcq single
+1 / 0
The _______ gene from Bacillus thuringiensis produces a protein that forms crystalline inclusions in bacterial spores.
Q123.
mcq single
+1 / 0
Arrange the following in sequence during the transmission of nerve impulse at chemical synapse.
i. Neurotransmitter binds with receptors of post synaptic cell.
ii. Calcium channels open and the calcium diffuses inward from ECF.
iii. Impulse travels along the axon of presynaptic neuron.
iv. Release of neurotransmitter by exocytosis.
v. Synaptic vesicles fuse with cell membrane of axon terminal of presynaptic neuron.
Q124.
mcq single
+1 / 0
Lateral sulcus separates ________ .
Q125.
mcq single
+1 / 0
Match column I with column II and select the correct option.
Column I
Column II
i.
Frontal lobe
a.
Gustato receptors
ii.
Parietal lobe
b.
Olfactory receptors
iii.
Temporal lobe
c.
Sense of vision
iv.
Occipital lobe
d.
Broca's area
Q126.
mcq single
+1 / 0
CSF is secreted by
Q127.
mcq single
+1 / 0
Complete the following reaction by replacing (Y) with proper word.
$$ \text { Emulsified fats } \xrightarrow{\text { Lipases }} \text { Fatty acids }+(\mathrm{Y}) $$
Q128.
mcq single
+1 / 0
Chemical digestion of which one of the following starts first in human alimentary canal?
Q129.
mcq single
+1 / 0
Given below are two statements.
Statement I - Root hair is composed of two layers.
Statement II - Outer layer of root hair cell wall is composed of cellulose and inner layer is made up of pectin.
In light of above statements, choose the most appropriate answer from the option given below.
Q130.
mcq single
+1 / 0
Two different amino acids are never encoded by the same codon, this character of genetic code is called ____________.
Q131.
mcq single
+1 / 0
All the 64 codons in the dictionary of genetic code were deciphered by __________
Q132.
mcq single
+1 / 0
The size of mRNA is generally related to $\qquad$
Q133.
mcq single
+1 / 0
In the processing of hnRNA in eukaryotic cell, the primary transcripts are processed in the following sequence.
Q134.
mcq single
+1 / 0
Given below are two statements:
Statement I - In prokaryotes, there is only replicon however in eukaryotes, these are several replicons in tandem.
Statement II - Two separated strands in a replicon of DNA are prevented from re-joining by helicase enzyme.
In light of above statements, choose the correct answer from the option given below.
Q135.
mcq single
+1 / 0
In which of the following, bilirubin is excreted in urine?
Q136.
mcq single
+1 / 0
Which one of the following diseases is caused by a fungus?
Q137.
mcq single
+1 / 0
In an antibody, disulphide bonds are present___________
i. between two heavy chains
ii. between two light chains
iii. between the constant region of light chain and the constant region of a heavy chain
iv. between antigen binding sites of both the heavy and light chains
v. between the variable region of light chain and that of heavy chain
Select the correct answer from the options given below
Q138.
mcq single
+1 / 0
Cells of malignant tumor spread from one organ to other via blood or lymph and cause new tumor. This property is called ___________ .
Q139.
mcq single
+1 / 0
Angina pectoris is severe pain and heaviness in chest that is mainly caused due to
Q140.
mcq single
+1 / 0
Atherosclerosis can be treated with recombinant protein called __________
Q141.
mcq single
+1 / 0
With reference to adolescence, what is mhGAP?
Q142.
mcq single
+1 / 0
The water which percolates deep in the soil is ___________
Q143.
mcq single
+1 / 0
Given below are two statements:
Statement I - During night, guard cells become flaccid.
Statement II - During day time, guard cells become turgid due to endosmosis.
In light of above statements, choose the most appropriate answer from the option given below.
Q144.
mcq single
+1 / 0
Capillarity theory was proposed by __________
Q145.
mcq single
+1 / 0
Accessory cells of stomata are reservoirs of_________.
Q146.
mcq single
+1 / 0
The term 'Niche' denotes the _________ role played by an organism in its environment.
Q147.
mcq single
+1 / 0
Dispersal of pollens is an example of
Q148.
mcq single
+1 / 0
Given below are two statements.
Statement I - Calotropis growing in abandoned fields is never consumed by cattle and goats.
Statement II - Calotropis produces highly poisonous cardiac glycosides.
In light of above statements, select the most appropriate answer from the option given below.
Q149.
mcq single
+1 / 0
The highest level of ecological hierarchy in ecological organization is
Q150.
mcq single
+1 / 0
Match the valves of human heart in column I with the respective opening they guard in column II and select the correct option.
Column - I
Column - II
i.
Eustachian valve
a.
Opening
of pulmonary aorta
ii.
Thebesian valve
b.
Left atrioventricular aperture
iii
Mitral valve
c.
opening of inferior vena cava
iv.
Semilunar valve
d.
opening of coronary sinus
Q151.
mcq single
+1 / 0
Mature erythrocytes are enucleated in following animals EXCEPT
i. Rat
ii. Monkey
iii. Llama
iv. Camel
v. Elephant Choose the correct answer from options given below.
Q152.
mcq single
+1 / 0
Which one of the following cells secrete heparin, histamine and serotonin?
Q153.
mcq single
+1 / 0
Blood plasma transports oxygen in dissolved state, percentage of which is ________ .
Q154.
mcq single
+1 / 0
In eukaryotic organisms, replication of DNA occurs in _________
Q155.
mcq single
+1 / 0
Hormone causing vigorous contraction of myometrium to initiate parturition is ___________ .
Q156.
mcq single
+1 / 0
The fully developed foetus gives signals for uterine contractions by secreting __________ hormones.
Q157.
mcq single
+1 / 0
Complete the analogy with respect to structure of sperm
Nebenkern : ' X ' : : Acrosome is formed from : ' Y '
Q158.
mcq single
+1 / 0
During oogenesis the meiotic division is arrested at________ before fertilization.
Q159.
mcq single
+1 / 0
Median vertical depression of external genitalia in females, enclosing the urethral and vaginal opening is _________
Q160.
mcq single
+1 / 0
Complete the analogy with respect to reproductive system.
Male : Penis : : Female : __________ .
Q161.
mcq single
+1 / 0
Blood osmolarity decreases with the secretion of
i. ADH
ii. aldosterone
iii. angiotensin II
iv. ANP
Q162.
mcq single
+1 / 0
Identify label ' X ' from the given diagram and name the cells, which regulate concentration of calcium and phosphorous in the blood and select the correct option given below.
Q163.
mcq single
+1 / 0
Islets of Langerhans are _________ .
Q164.
mcq single
+1 / 0
Which one of the following pair of hormones is NOT antagonistic?
Q165.
mcq single
+1 / 0
Hypersecretion of glucocorticoid leads to___________
Q166.
mcq single
+1 / 0
Identify the INCORRECT statement.
Q167.
mcq single
+1 / 0
Match the scientist in column I with their contribution in column II and choose the correct option.
Column - I
Column - II
i.
Alexander Van Humboldt
a.
Estimated 7 million species round the globe
ii.
Rober ay
b.
Rivet popper hypothesis
iii
David TIllman
c.
Species Area Relationship
iv.
Paul Ehrlich
d.
Productivity stability hypothesis
Q168.
mcq single
+1 / 0
Damage to biodiversity takes place due to natural reasons which includes the following EXCEPT
Q169.
mcq single
+1 / 0
Phenotypically tall plants can be obtained from genetically dwarf maize plants by application of_________
Q170.
mcq single
+1 / 0
Which plant hormone is called antitranspirant?
Q171.
mcq single
+1 / 0
While studying flowering behaviour in __________ and ___________ plant, Garner and Allard discovered photoperiodism.
Q172.
mcq single
+1 / 0
The number of ATP molecules generated in aerobic respiration after oxidation of two molecules of pyruvic acid is.
Q173.
mcq single
+1 / 0
Given below are two statements:
Statement I - Reactions involved in Krebs cycle are anabolic and catabolic.
Statement II - During oxidation of acetyl Co-A, stepwise oxidation of acetyl part of acetyl Co-A occurs.
In light of above statements, choose the most appropriate answer from the option given below.
Q174.
mcq single
+1 / 0
The only 5C intermediate formed during reactions of TCA cycle is __________
Q175.
mcq single
+1 / 0
The number of reduced coenzymes $\mathrm{NADH}+\mathrm{H}^{+}$ formed during complete oxidation of one molecule of glucose.
Q176.
mcq single
+1 / 0
Homo sapiens are most closely related to________
Q177.
mcq single
+1 / 0
DDT resistant mosquitoes and different beak sizes of African seed cracker finches show
_________ type of selection respectively.
Q178.
mcq single
+1 / 0
Which one of the following is the most primitive ancestor of Homo sapiens?
Q179.
mcq single
+1 / 0
An organ with sphincters at its origin is
Q180.
mcq single
+1 / 0
Tubular fluid tends to become acidic in the_________ of nephron.
Q181.
mcq single
+1 / 0
Given below are two statements.
Statement I - Defecation is an involuntary process that takes place through anal opening guarded by sphincter muscles.
Statement II - Distension, of rectum stimulates pressure sensitive receptors that initiate a neural reflex for egestion.
In light of above statements, select the correct answer from the option given below.
Q182.
mcq single
+1 / 0
People who consume high protein diet can develop __________ stones.
Q183.
mcq single
+1 / 0
All are events of pollen-pistil interaction EXCEPT
Q184.
mcq single
+1 / 0
In sea grass, pollens are long, ribbon like and without exine exhibiting __________ pollination.
Q185.
mcq single
+1 / 0
In a male gametophyte, ploidy of tube cell and a male gamete is __________
Q186.
mcq single
+1 / 0
In an angiospermic embryo, upper swollen suspensor cell towards the micropyle, functions as_________.
Q187.
mcq single
+1 / 0
Bird pollinated flowers are usually __________
Q188.
mcq single
+1 / 0
Just before fertilization, the angiosperm's embryo sac contains_________ and ___________nuclei.
Q189.
mcq single
+1 / 0
Just before fertilization, the angiosperm's embryo sac contains ________ and ________ nuclei.
Q190.
mcq single
+1 / 0
Given below are two statements.
Statement I - Oxaloacetate is useful as a precursor for synthesis of aspartic acid.
Statement II - $\alpha$-ketoglutarate is useful for synthesis of glutamic acid.
In light of above statements, select the correct answer from the option given below.
Q191.
mcq single
+1 / 0
The number of amino groups in the amino acids present in amides is __________
Q192.
mcq single
+1 / 0
Match column I with column II and select the correct option
Column - I
Column - II
i.
Carrot and spinach
a.
High protein content
ii.
Bitter gourd
b.
More Vit A and minerals
iii
Maize
c.
More Vit C
iv.
Wheat Atlas-66
c.
Twice the amount of lysine and tryptophan
Q193.
mcq single
+1 / 0
In large proteins, peptide chains are much looped, twisted and folded back on themselves. The folded structure is due to the formation of________bonds
Q194.
mcq single
+1 / 0
Enzymes are needed only in small quantities to catalyse reactions because
Q195.
mcq single
+1 / 0
Secondary metabolites like glucosinolates are produced by cabbage to
Q196.
mcq single
+1 / 0
Agarose gel electrophoresis is used to separate DNA fragments. The DNA fragments separate due to __________ .
Q197.
mcq single
+1 / 0
When the phage DNA enters the host bacterium, the host cell protects itself from the viral DNA attacks with the help of _________ enzyme.
Q198.
mcq single
+1 / 0
The restriction enzymes used as molecular scissors are type of ____________
Q199.
mcq single
+1 / 0
Match elements given in Column I with their deficiency symptoms shown by plants given in Column II
Column - I
Column - II
i.
Eustachian valve
a.
Brown heart disease
ii.
Boron
b.
Malformed leaves
iii
Zinc
c.
Poor growth of the plant
iv.
Copper
c.
Die-back of shoots
Q200.
mcq single
+1 / 0
Which of the following bacteria do not solubilize the insoluble rock phosphate?