MHT CET (PCB) 2025 9th April Evening Shift
MEDICAL 2025 Previous Year
3 hDuration
200Total Marks
394Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 394 across 3 section(s); maximum marks: 200.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+1 / 0
Which from following reagents is NOT involved in Sandmeyer reaction?
Q2.
mcq single
+1 / 0
Which among the following compounds has highest boiling point?
Q3.
mcq single
+1 / 0
Identify the element so that the last electron enters in ( $n-2$ )f sub shell.
Q4.
mcq single
+1 / 0
What is the number of unpaired electrons in $\mathrm{C}_0$ in +2 state?
Q5.
mcq single
+1 / 0
Identify the monomers used to prepare polycarbonate.
Q6.
mcq single
+1 / 0
Identify thermoplastic polymer from following.
Q7.
mcq single
+1 / 0
Identify the compound having highest value of bond angle from following.
Q8.
mcq single
+1 / 0
What type of hybridisation is found in $\mathrm{H}_2 \mathrm{Se}$ ?
Q9.
mcq single
+1 / 0
Calculate the energy associated with second orbit of $\mathrm{He}^{+}$.
Q10.
mcq single
+1 / 0
The solubility of sparingly soluble, salt $\mathrm{AX}_2$ is $1.5 \times 10^{-3} \mathrm{~mol} \mathrm{dm}^{-3}$. What is its solubility product?
Q11.
mcq single
+1 / 0
Solubility of $\mathrm{Mg}(\mathrm{OH})_2$ is ' S ' $\mathrm{mol} / \mathrm{dm}^3$ its solubility product is equal to
Q12.
mcq single
+1 / 0
Half life of a first order reaction is 3 hours. Calculate the value of $\frac{[\mathrm{A}]_0}{[\mathrm{~A}]_{\mathrm{t}}}$ after 8 hours.
Q13.
mcq single
+1 / 0
For the reaction
$$ \mathrm{N}_{2(\mathrm{~g})}+3 \mathrm{H}_{2(\mathrm{~g})} \longrightarrow 2 \mathrm{NH}_{3(\mathrm{~g})} $$
$\mathrm{N}_{2(\mathrm{~g})}$ disappears at the rate of $2 \times 10^{-4} \mathrm{~mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1}$. Find the rate of $\mathrm{NH}_3$ formation.
Q14.
mcq single
+1 / 0
Find the rate of disappearance of $B$ for the reaction $2 \mathrm{~A}+\mathrm{B} \longrightarrow 2 \mathrm{C}+\mathrm{D}$, from following.
Q15.
mcq single
+1 / 0
What is change in oxidation number of ' Mn ' during the decolourisation of acidified potassium permanganate by moist sulfur dioxide?
Q16.
mcq single
+1 / 0
What is the average oxidation number of carbon in carbon suboxide $\mathrm{C}_3 \mathrm{O}_2$ ?
Q17.
mcq single
+1 / 0
Identify the products of following reaction.
$$ \mathrm{C}_6 \mathrm{H}_5 \mathrm{COOC}_2 \mathrm{H}_5 \xrightarrow[\text { dil H}_2 \mathrm{SO}_4]{\Delta} \text { Products } $$
Q18.
mcq single
+1 / 0
Identify product ' $X$ ' obtained in the following reaction.
$$ \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{Br}+\mathrm{CH}_3 \mathrm{COOAg} \xrightarrow{\Delta} \mathrm{X}+\mathrm{AgBr} $$
Q19.
mcq single
+1 / 0
Identify the product ' X ' in following reaction.
Ethanoyl chloride $\xrightarrow{\mathrm{H}_2 \mathrm{O}} X$
Q20.
mcq single
+1 / 0
Identify product ' B ' in the following reaction.
$$ \text { Cumene } \xrightarrow[\Delta]{\mathrm{KMnO}_4, \mathrm{KOH}} \mathrm{~A} \xrightarrow{\mathrm{H}_3 \mathrm{O}^{+}} \mathrm{B} $$
Q21.
mcq single
+1 / 0
Identify positively charged sol from following.
Q22.
mcq single
+1 / 0
Identify the major product obtained in the reaction of chlorobenzene with chlorine in presence of anhydrous $\mathrm{FeCl}_3$.
Q23.
mcq single
+1 / 0
Which among the following is NOT benzylic halide?
Q24.
mcq single
+1 / 0
Which from following quantity of different gases contains maximum number of molecules?
Q25.
mcq single
+1 / 0
Calculate standard Gibbs energy for a certain reaction if $\Delta \mathrm{H}^{\circ}$ and $\Delta \mathrm{S}^{\circ}$ respectively are $219 \mathrm{~kJ} \mathrm{~mol}^{-1}$ and $-20 \mathrm{JK}^{-1}$.
Q26.
mcq single
+1 / 0
Which from following reactions performs highest + ve work at $25^{\circ} \mathrm{C}$ ?
Q27.
mcq single
+1 / 0
Calculate the change in internal energy of the system if 15 kJ of work done on the surrounding and system releases 10 kJ of heat in a particular reaction.
Q28.
mcq single
+1 / 0
Calculate cryoscopic constant of solvent if 2 g solute having molar mass $80 \mathrm{~g} \mathrm{~mol}^{-1}$ dissolved in 75 g solvent decreases its freezing point by 0.4 K .
Q29.
mcq single
+1 / 0
Calculate the osmotic pressure of 0.01 M aqueous solution of non electrolyte at 300 K .
$$ \left[\mathrm{R}=0.0821 \mathrm{~atm} \mathrm{dm}^3 \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right] $$
Q30.
mcq single
+1 / 0
Which from following units of concentrations of solutions is temperature independent?
Q31.
mcq single
+1 / 0
What is the total number of donor atoms present in Bis(ethylenediamine)dithiocyanatoplatinum (IV)?
Q32.
mcq single
+1 / 0
Identify the correct decreasing order of stability of complexes formed by divalent metal ions with same ligand.
Q33.
mcq single
+1 / 0
Which from following compounds is used as analgesics?
Q34.
mcq single
+1 / 0
Which from following compounds is called Gammexane?
Q35.
mcq single
+1 / 0
Which of the following is tertiary benzylic alcohol?
Q36.
mcq single
+1 / 0
Which of the following is a pair of dihydric phenols?
Q37.
mcq single
+1 / 0
Identify the order of reactivity of halogens towards alkanes.
Q38.
mcq single
+1 / 0
What is the total number of moles of ' C ' atoms and ' H ' atoms respectively present in n mole molecule represented by following structure?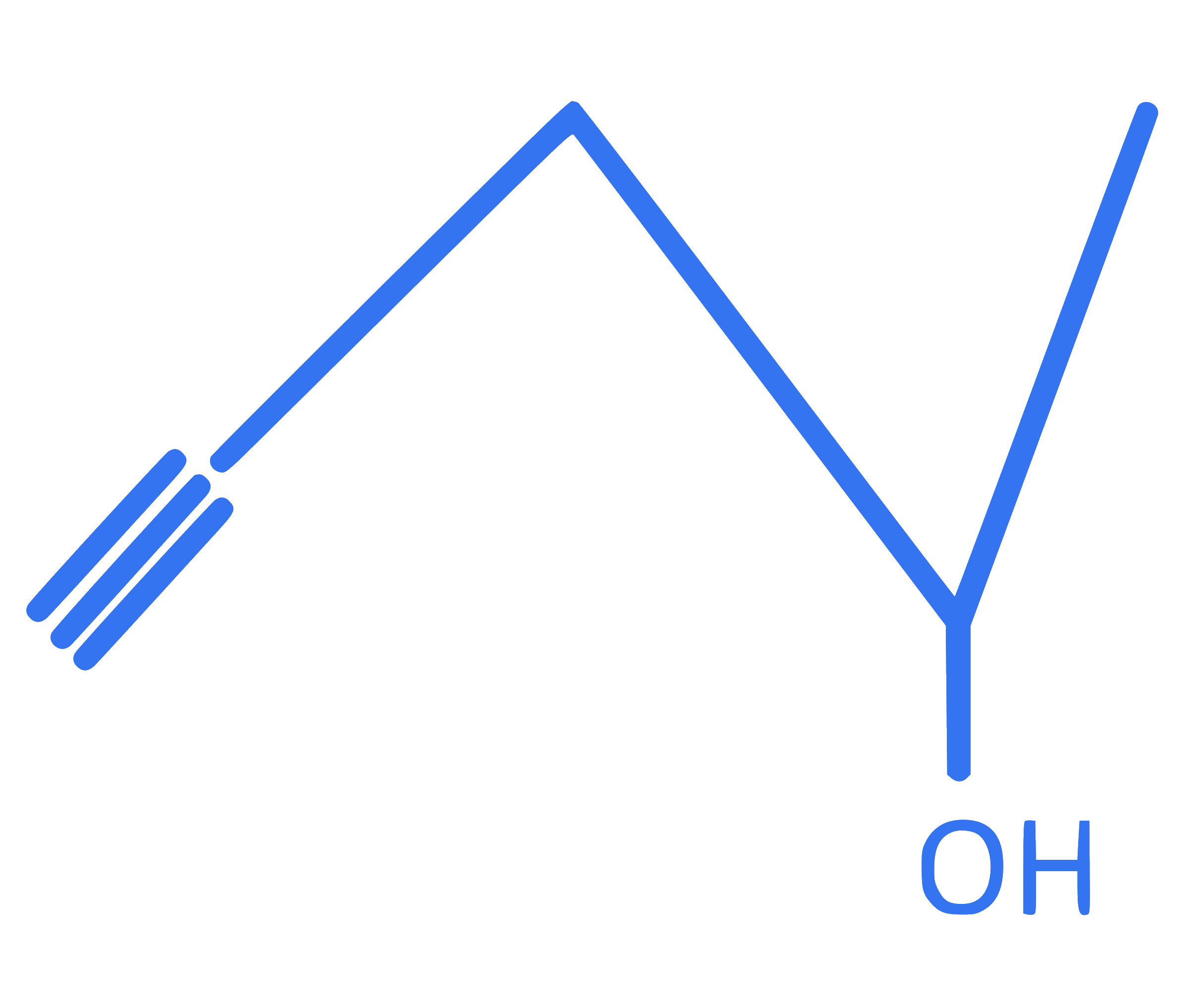

Q39.
mcq single
+1 / 0
Which from following conditions defines the critical temperature of a gas is the temperature
Q40.
mcq single
+1 / 0
Which of the following cations is involved in formation of chlorophyll molecule in plants?
Q41.
mcq single
+1 / 0
Which from following tests for glucose confirms presence of carbonyl group?
Q42.
mcq single
+1 / 0
Which from following is essential amino acid?
Q43.
mcq single
+1 / 0
What is the percent void volume in hcp lattice?
Q44.
mcq single
+1 / 0
Calculate the number of unit cells present in 0.6 g metal if the product of density and unit cell volume is $3.6 \times 10^{-22} \mathrm{~g}$.
Q45.
mcq single
+1 / 0
Find the total number of tetrahedral voids formed in 0.5 mole of a compound forming hcp structure.
Q46.
mcq single
+1 / 0
Which of the following species acts as an oxidising agent during working of hydrogenoxygen fuel cell?
Q47.
mcq single
+1 / 0
If for the cell reaction,
$$ \mathrm{A}_{(\mathrm{s})}+\mathrm{B}_{(\mathrm{aq})}^{+} \longrightarrow \mathrm{A}_{(\mathrm{aq})}^{+}+\mathrm{B}_{(\mathrm{s})} $$
if $\mathrm{E}_{\text {cell }}^{\circ}=0.0592 \mathrm{~V}$. What is equilibrium constant of reaction at 298 K ?
Q48.
mcq single
+1 / 0
What is the amount of 'Al' deposited at cathode during electrolysis of aqueous $\mathrm{AlCl}_3$ by passing 1 faraday electricity?
Q49.
mcq single
+1 / 0
Identify the product formed in the following reaction.$$
\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CHO} \xrightarrow[\text { ii) } \mathrm{H}_3 \mathrm{O}^{+}]{\text {i) } \mathrm{LiAlH}_4} \text { Product }
$$
Q50.
mcq single
+1 / 0
Which from following reagents is NOT involved in Sandmeyer reaction?
Q51.
mcq single
+1 / 0
Which among the following compounds has highest boiling point?
Q52.
mcq single
+1 / 0
Identify the element so that the last electron enters in ( $n-2$ )f sub shell.
Q53.
mcq single
+1 / 0
What is the number of unpaired electrons in $\mathrm{C}_0$ in +2 state?
Q54.
mcq single
+1 / 0
Identify the monomers used to prepare polycarbonate.
Q55.
mcq single
+1 / 0
Identify thermoplastic polymer from following.
Q56.
mcq single
+1 / 0
Identify the compound having highest value of bond angle from following.
Q57.
mcq single
+1 / 0
What type of hybridisation is found in $\mathrm{H}_2 \mathrm{Se}$ ?
Q58.
mcq single
+1 / 0
Calculate the energy associated with second orbit of $\mathrm{He}^{+}$.
Q59.
mcq single
+1 / 0
The solubility of sparingly soluble, salt $\mathrm{AX}_2$ is $1.5 \times 10^{-3} \mathrm{~mol} \mathrm{dm}^{-3}$. What is its solubility product?
Q60.
mcq single
+1 / 0
Solubility of $\mathrm{Mg}(\mathrm{OH})_2$ is ' S ' $\mathrm{mol} / \mathrm{dm}^3$ its solubility product is equal to
Q61.
mcq single
+1 / 0
Half life of a first order reaction is 3 hours. Calculate the value of $\frac{[\mathrm{A}]_0}{[\mathrm{~A}]_{\mathrm{t}}}$ after 8 hours.
Q62.
mcq single
+1 / 0
For the reaction
$$ \mathrm{N}_{2(\mathrm{~g})}+3 \mathrm{H}_{2(\mathrm{~g})} \longrightarrow 2 \mathrm{NH}_{3(\mathrm{~g})} $$
$\mathrm{N}_{2(\mathrm{~g})}$ disappears at the rate of $2 \times 10^{-4} \mathrm{~mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1}$. Find the rate of $\mathrm{NH}_3$ formation.
Q63.
mcq single
+1 / 0
Find the rate of disappearance of $B$ for the reaction $2 \mathrm{~A}+\mathrm{B} \longrightarrow 2 \mathrm{C}+\mathrm{D}$, from following.
Q64.
mcq single
+1 / 0
What is change in oxidation number of ' Mn ' during the decolourisation of acidified potassium permanganate by moist sulfur dioxide?
Q65.
mcq single
+1 / 0
What is the average oxidation number of carbon in carbon suboxide $\mathrm{C}_3 \mathrm{O}_2$ ?
Q66.
mcq single
+1 / 0
Identify the products of following reaction.
$$ \mathrm{C}_6 \mathrm{H}_5 \mathrm{COOC}_2 \mathrm{H}_5 \xrightarrow[\text { dil H}_2 \mathrm{SO}_4]{\Delta} \text { Products } $$
Q67.
mcq single
+1 / 0
Identify product ' $X$ ' obtained in the following reaction.
$$ \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{Br}+\mathrm{CH}_3 \mathrm{COOAg} \xrightarrow{\Delta} \mathrm{X}+\mathrm{AgBr} $$
Q68.
mcq single
+1 / 0
Identify the product ' X ' in following reaction.
Ethanoyl chloride $\xrightarrow{\mathrm{H}_2 \mathrm{O}} X$
Q69.
mcq single
+1 / 0
Identify product ' B ' in the following reaction.
$$ \text { Cumene } \xrightarrow[\Delta]{\mathrm{KMnO}_4, \mathrm{KOH}} \mathrm{~A} \xrightarrow{\mathrm{H}_3 \mathrm{O}^{+}} \mathrm{B} $$
Q70.
mcq single
+1 / 0
Identify positively charged sol from following.
Q71.
mcq single
+1 / 0
Identify the major product obtained in the reaction of chlorobenzene with chlorine in presence of anhydrous $\mathrm{FeCl}_3$.
Q72.
mcq single
+1 / 0
Which among the following is NOT benzylic halide?
Q73.
mcq single
+1 / 0
Which from following quantity of different gases contains maximum number of molecules?
Q74.
mcq single
+1 / 0
Calculate standard Gibbs energy for a certain reaction if $\Delta \mathrm{H}^{\circ}$ and $\Delta \mathrm{S}^{\circ}$ respectively are $219 \mathrm{~kJ} \mathrm{~mol}^{-1}$ and $-20 \mathrm{JK}^{-1}$.
Q75.
mcq single
+1 / 0
Which from following reactions performs highest + ve work at $25^{\circ} \mathrm{C}$ ?
Q76.
mcq single
+1 / 0
Calculate the change in internal energy of the system if 15 kJ of work done on the surrounding and system releases 10 kJ of heat in a particular reaction.
Q77.
mcq single
+1 / 0
Calculate cryoscopic constant of solvent if 2 g solute having molar mass $80 \mathrm{~g} \mathrm{~mol}^{-1}$ dissolved in 75 g solvent decreases its freezing point by 0.4 K .
Q78.
mcq single
+1 / 0
Calculate the osmotic pressure of 0.01 M aqueous solution of non electrolyte at 300 K .
$$ \left[\mathrm{R}=0.0821 \mathrm{~atm} \mathrm{dm}^3 \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right] $$
Q79.
mcq single
+1 / 0
Which from following units of concentrations of solutions is temperature independent?
Q80.
mcq single
+1 / 0
What is the total number of donor atoms present in Bis(ethylenediamine)dithiocyanatoplatinum (IV)?
Q81.
mcq single
+1 / 0
Identify the correct decreasing order of stability of complexes formed by divalent metal ions with same ligand.
Q82.
mcq single
+1 / 0
Which from following compounds is used as analgesics?
Q83.
mcq single
+1 / 0
Which from following compounds is called Gammexane?
Q84.
mcq single
+1 / 0
Which of the following is tertiary benzylic alcohol?
Q85.
mcq single
+1 / 0
Which of the following is a pair of dihydric phenols?
Q86.
mcq single
+1 / 0
Identify the order of reactivity of halogens towards alkanes.
Q87.
mcq single
+1 / 0
What is the total number of moles of ' C ' atoms and ' H ' atoms respectively present in n mole molecule represented by following structure?

Q88.
mcq single
+1 / 0
Which from following conditions defines the critical temperature of a gas is the temperature
Q89.
mcq single
+1 / 0
Which of the following cations is involved in formation of chlorophyll molecule in plants?
Q90.
mcq single
+1 / 0
Which from following tests for glucose confirms presence of carbonyl group?
Q91.
mcq single
+1 / 0
Which from following is essential amino acid?
Q92.
mcq single
+1 / 0
What is the percent void volume in hcp lattice?
Q93.
mcq single
+1 / 0
Calculate the number of unit cells present in 0.6 g metal if the product of density and unit cell volume is $3.6 \times 10^{-22} \mathrm{~g}$.
Q94.
mcq single
+1 / 0
Find the total number of tetrahedral voids formed in 0.5 mole of a compound forming hcp structure.
Q95.
mcq single
+1 / 0
Which of the following species acts as an oxidising agent during working of hydrogenoxygen fuel cell?
Q96.
mcq single
+1 / 0
If for the cell reaction,
$$ \mathrm{A}_{(\mathrm{s})}+\mathrm{B}_{(\mathrm{aq})}^{+} \longrightarrow \mathrm{A}_{(\mathrm{aq})}^{+}+\mathrm{B}_{(\mathrm{s})} $$
if $\mathrm{E}_{\text {cell }}^{\circ}=0.0592 \mathrm{~V}$. What is equilibrium constant of reaction at 298 K ?
Q97.
mcq single
+1 / 0
What is the amount of 'Al' deposited at cathode during electrolysis of aqueous $\mathrm{AlCl}_3$ by passing 1 faraday electricity?
Q98.
mcq single
+1 / 0
Identify the product formed in the following reaction.$$
\mathrm{CH}_3-\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CHO} \xrightarrow[\text { ii) } \mathrm{H}_3 \mathrm{O}^{+}]{\text {i) } \mathrm{LiAlH}_4} \text { Product }
$$
Physics
Physics
Q1.
mcq single
+1 / 0
At what angle should the two forces $2 \overrightarrow{\mathrm{P}}$ and $\sqrt{2} \overrightarrow{\mathrm{P}}$ act so that the resultant force is $\sqrt{10} \overrightarrow{\mathrm{P}}$ ?
Q2.
mcq single
+1 / 0
In Young's double slit experiment, waves from slits ' $\mathrm{S}_1$ ' and ' $\mathrm{S}_2$ ' have a path difference of $\frac{\lambda}{4}$ and $\frac{\lambda}{6}$ respectively at points $x$ and $y$ on the screen. The ratio of intensities at points $y$ to that at $x$ is $\left(\cos 45^{\circ}=\frac{1}{\sqrt{2}}, \cos 30^{\circ}=\sqrt{3} / 2,\right)$
Q3.
mcq single
+1 / 0
Monochromatic light of wavelength $4800 \mathop {\rm{A}}\limits^{\rm{o}}$ falls on slit of width ' $a$ '. For what value of ' $a$ ', the first maximum falls at $30^{\circ} .\left(\sin 30^{\circ}=0.5\right)$
Q4.
mcq single
+1 / 0
Two (A and B) pendulums begin to swing simultaneously. The first pendulum (A) makes five oscillations when the other (B) makes three oscillations. The ratio of the lengths of pendulum $A$ to that of $B$ is
Q5.
mcq single
+1 / 0
A particle of mass 0.02 kg executes S.H.M. about $\mathrm{x}=0$ under the influence of a force as shown in figure. The period of S.H.M. is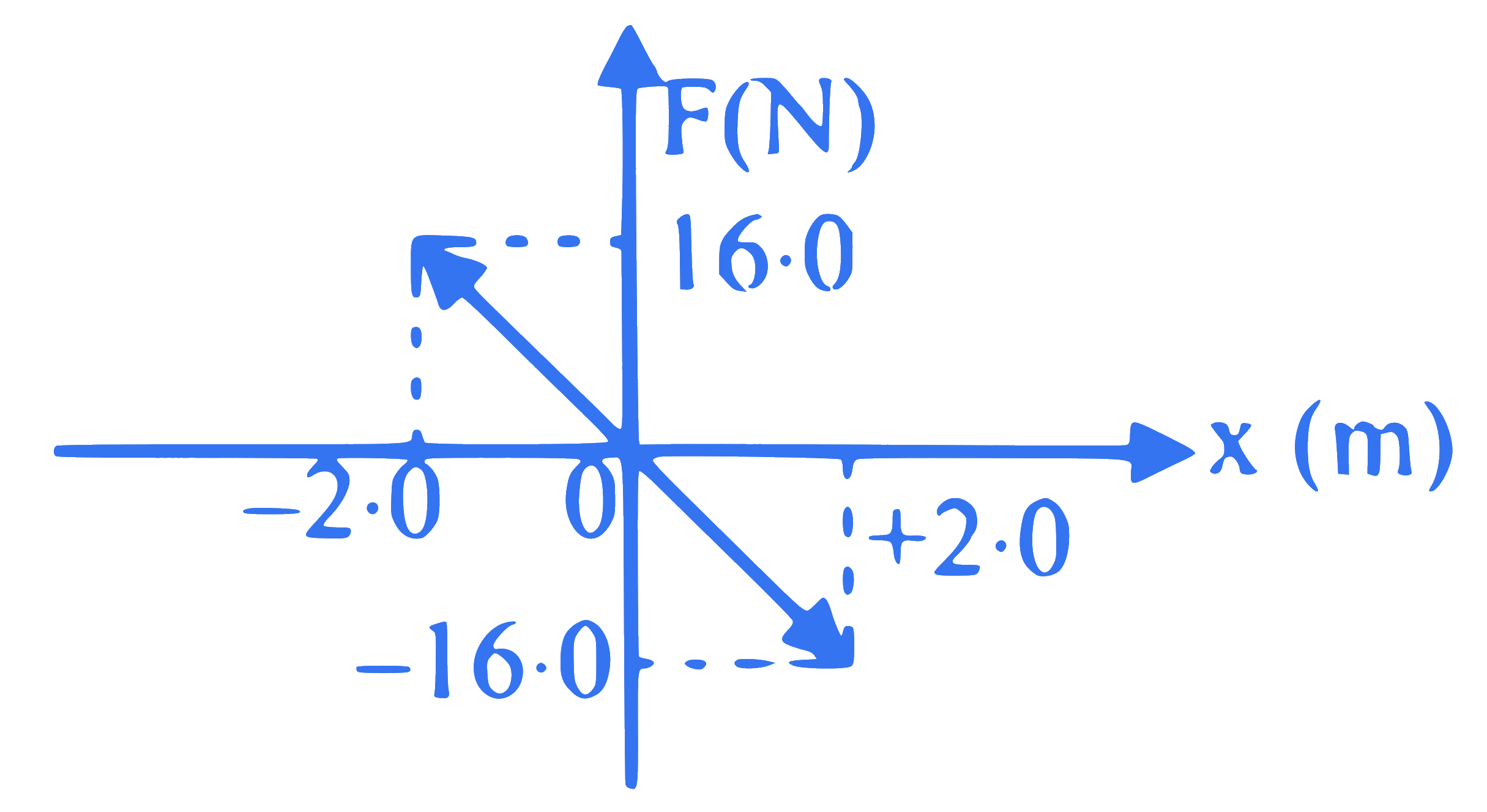

Q6.
mcq single
+1 / 0
A particle performs S.H.M. with amplitude 'A'. The speed of the particle is $\left(\frac{1}{3}\right)^{\text {rd }}$ of the maximum speed when its displacement from the mean position is
Q7.
mcq single
+1 / 0
Two bodies A and B have their moments of inertia I and 3I respectively about their axis of rotation. their kinetic energies of rotation are also equal. The ratio of angular momenta of body A to that of body B is
Q8.
mcq single
+1 / 0
An elastic conducting material is stretched into a circular loop. It is placed with its plane perpendicular to uniform magnetic field $\mathrm{B}=0.6 \mathrm{~T}$. When loop is released, the radius starts shrinking at constant rate $3 \mathrm{~cm} / \mathrm{s}$. The induced e.m.f. in the loop at an instant when the radius of the loop is 10 cm will be (in mV )
Q9.
mcq single
+1 / 0
Two coils having self-inductances $\mathrm{L}_1=75 \mathrm{mH}$ and $L_2=48 \mathrm{mH}$ are coupled with each other. If the mutual inductance of the coils is 37.2 mH , then coefficient of coupling will be
Q10.
mcq single
+1 / 0
A bucket filled with water is revolved in a vertical circle of radius 10 m and water just not fall down. The time period of revolution will be (acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2$ )
Q11.
mcq single
+1 / 0
The angular separation between the minute hand and the hour hand of clock 40 minutes passed after $12 \mathrm{p} . \mathrm{m}$. is
Q12.
mcq single
+1 / 0
In resistance thermometer, the resistance at $0^{\circ} \mathrm{C}$ and $100^{\circ} \mathrm{C}$ are $6.74 \Omega$ and $7.74 \Omega$ respectively. The temperature corresponding to $6.53 \Omega$ resistance is
Q13.
mcq single
+1 / 0
In thermodynamic process, for free expansion, out of the following select the 'WRONG' statement.
Q14.
mcq single
+1 / 0
The heat required to increase the temperature from 373 K to 573 K at constant volume for ideal monoatomic gas of 2 moles is ( $R=$ universal gas constant)
Q15.
mcq single
+1 / 0
A Carnot engine has an efficiency of $\frac{1}{6}$. When the temperature of sink is reduced by 62 K , its efficiency is doubled. The temperature of the source is
Q16.
mcq single
+1 / 0
An ideal gas in a container of volume 500 cc is at a pressure of $2 \times 10^5 \mathrm{~N} / \mathrm{m}^2$. The average kinetic energy of each molecule is $6 \times 10^{-21} \mathrm{~J}$. The number of gas molecules in the container is
Q17.
mcq single
+1 / 0
If the r.m.s. velocity of gas is V at temperature T , then
Q18.
mcq single
+1 / 0
In photoelectric effect experiment, the stopping potential for a given metal is $\mathrm{V}_0$ (in volt) when radiation of wavelength $\lambda_0$ is used. If radiation of wavelength $5 \lambda_0$ is used with the same metal, then the stopping potential is (in volt) ( $\mathrm{h}=$ Planck's constant, $\mathrm{c}=$ velocity of light, $\mathrm{e}=$ electronic charge)
Q19.
mcq single
+1 / 0
Two identical photocathodes receive light of frequencies ' $f 1$ ' and ' $f_2$ '. The velocity of the photoelectrons of mass ' $m$ ' emitted are respectively ' $\mathrm{v}_1$ ' and ' $\mathrm{v}_2$ '. Then the correct relation is ( $\mathrm{h}=$ Planck's constant)
Q20.
mcq single
+1 / 0
If 80 J of work is required in moving an electric charge of ' 2 C ' from a point where potential is " -8 V ' to another point where potential is ' $V$ ' volt, the value of ' $V$ ' is
Q21.
mcq single
+1 / 0
If the electric flux entering and leaving an enclosed surface area are ' $\phi_1$ ' and ' $\phi_2$ ' respectively, the electric charge inside the surface will be ( $\varepsilon_0=$ permittivity of free space)
Q22.
mcq single
+1 / 0
A charge of $8 \mu \mathrm{C}$ exists at the centre of the square $A B C D$. The work done in moving a $4 \mu C$ point charge from corner A to corner B is (Along side AB )
Q23.
mcq single
+1 / 0
An object is released from a distance ' $r$ ' from the centre of the earth. The velocity of the object at the time of striking the earth will be ( $R=$ radius of earth, $r>R, g=$ acceleration due to gravity)
Q24.
mcq single
+1 / 0
An air capacitor of plate area ' $A$ ' and separation between the plates is ' $d$ ' has a capacity ' $C$ '. Two dielectric slabs are inserted between its plates in two different manners as shown in figure. The capacitance of a capacitor is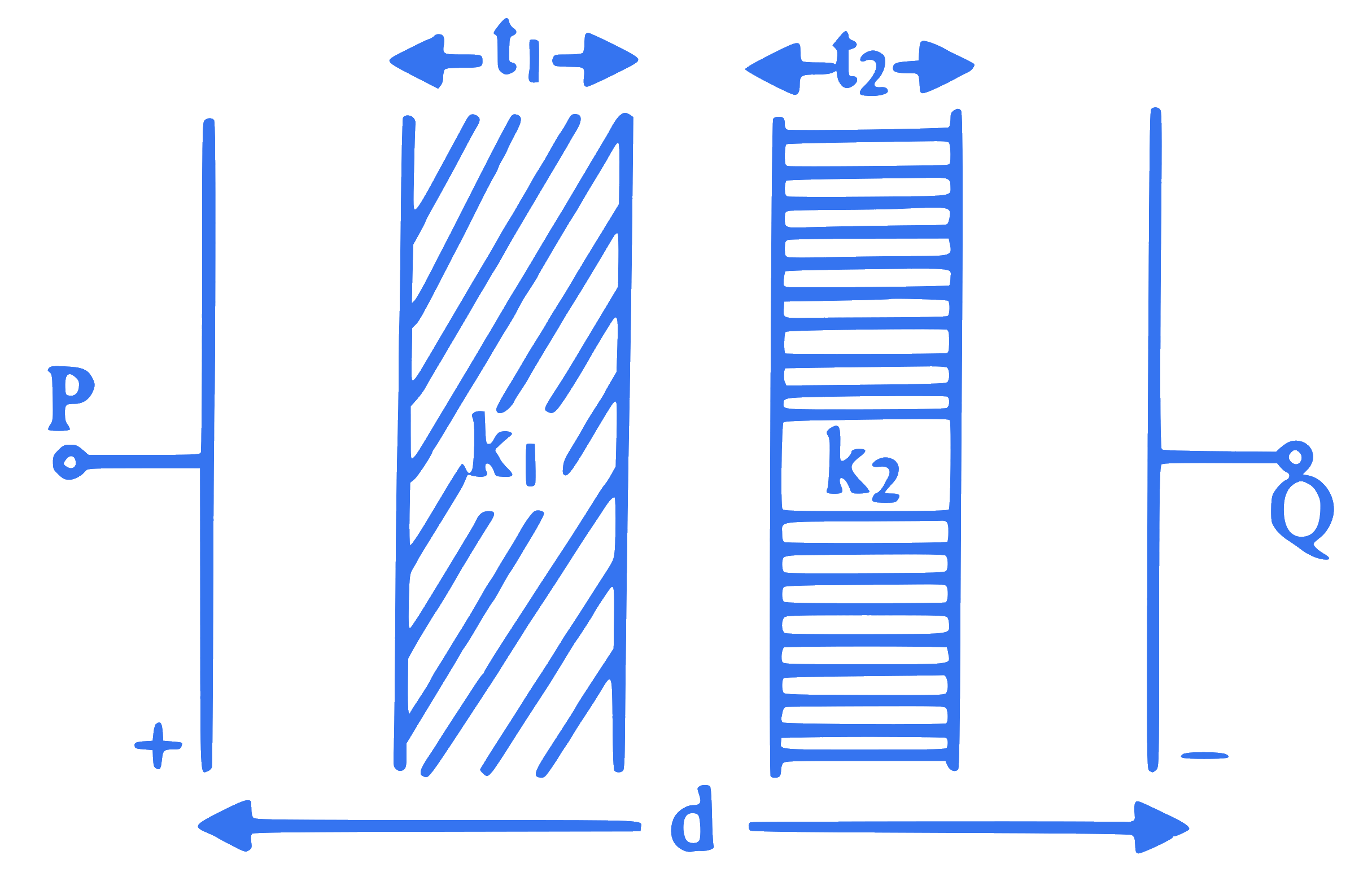

Q25.
mcq single
+1 / 0
The capacitive reactance of a capacitor connected to 50 V d.c. is
Q26.
mcq single
+1 / 0
Light propagates 2 cm distance in glass of refractive index 1.5 in certain time. In the same time same light propogates a distance of 2.25 cm in a medium. The refractive index of the medium is
Q27.
mcq single
+1 / 0
Glass has refractive index ' $n$ ' with respect to air and ' $\theta$ ' is the critical angle for a ray of light going from glass to air. If a ray of light is incident from air on the glass with angle of incidence ' $\theta$ ', corresponding angle of refraction is
Q28.
mcq single
+1 / 0
Magnetic field at the centre of a circular loop of area ' $A$ ' is ' $B$ '. The magnetic moment of the loop is ' $x B$ '. The value of ' $x$ ' is ( $\mu_0=$ permeability of vacuum or free space)
Q29.
mcq single
+1 / 0
The current $I$ is flowing through a loop $A B C D A$ as shown in the figure. The magnitude of the magnetic field at the centre ' $O$ ' is $x$ times
$$ \left(\frac{\mu_0 \mathrm{I}}{\mathrm{R}}\right)\left(\mathrm{OD}=\mathrm{R}, \mathrm{DC}=\mathrm{R}, \angle \mathrm{AOD}=90^{\circ}\right) $$
The value of ' $x$ ' is ( $\mu_0=$ permeability of free space)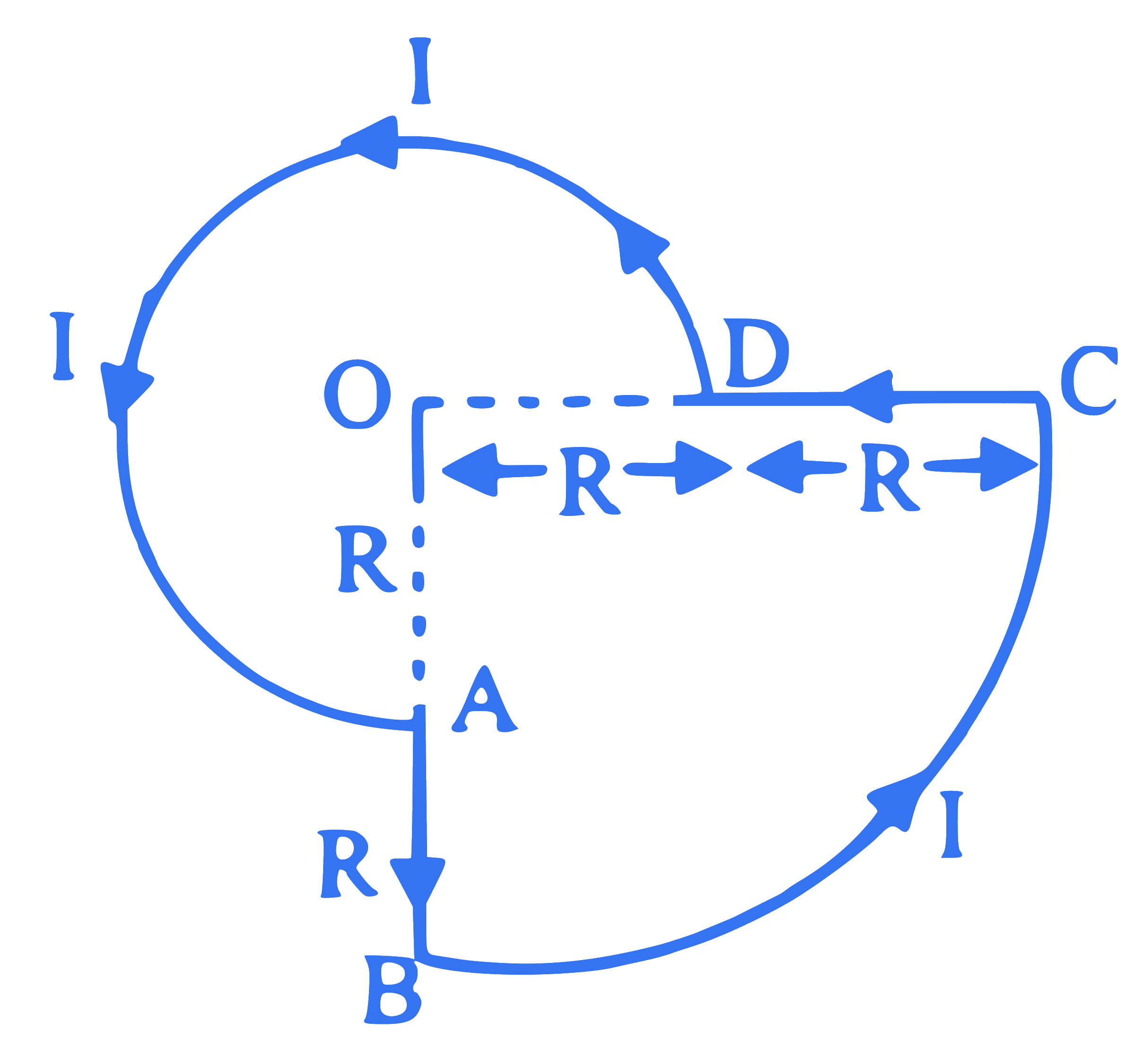

Q30.
mcq single
+1 / 0
Two solid spheres of same material but of mass ' M ' and ' 8 M ' fall simultaneously through a viscous liquid. Their terminal velocities are respectively ' V ' and ' nV '. The value ' n ' is
Q31.
mcq single
+1 / 0
When a big drop of water is formed from n small drops of water, the energy loss is 4 E , where E is the energy of the bigger drop. If $R$ and $r$ is the radius of the bigger and the smaller drop respectively then number of smaller drops $n$ is
Q32.
mcq single
+1 / 0
A small steel ball of mass ' M ', radius ' R ' and density ' $\rho$ ' falls with terminal velocity through a tube filled with glycerine of density ' $\sigma$ '. The viscous force acting on the steel ball is ( $\mathrm{g}=$ acceleration due to gravity)
Q33.
mcq single
+1 / 0
In the Bohr model of hydrogen atom, out of the following quantities, the principal quantum number is proportional to ( $\mathrm{R}, \mathrm{V}$ and E represent radius of the orbit, speed of electron and total energy of the electron respectively)
Q34.
mcq single
+1 / 0
According to Bohr's first postulate the kinetic energy $\frac{1}{2} m v^2$ of the electron in C.G.S. system is ( $\mathrm{m}=$ mass of electron, v is its velocity, r is the radius of the stationary orbit around the nucleus with charge Ze )
Q35.
mcq single
+1 / 0
A uniform metal wire has length ' $L$ ', mass ' $M$ ' and area of cross-section ' A '. It is under tension ' $T$ ' and ' $V$ ' is the speed of transverse wave along the wire. The density of the wire is
Q36.
mcq single
+1 / 0
The loudness and pitch of sound note respectively depend upon
Q37.
mcq single
+1 / 0
A transverse wave is moving along a string. The linear density of a vibrating string is $2 \times 10^{-3} \mathrm{~kg} / \mathrm{m}$. The wave equation is $y=0.02 \sin (2 x+10 t) m$. The tension in the string is
Q38.
mcq single
+1 / 0
An open organ pipe and a closed organ pipe have the frequency of their second overtone identical. The ratio of length of closed pipe to that of open pipe is (Neglect end correction)
Q39.
mcq single
+1 / 0
Out of the following properties describing diamagnetism, identify the property which is "WRONG"
Q40.
mcq single
+1 / 0
A stone is projected at an angle $60^{\circ}$ to the horizontal. The ratio of kinetic energy of the stone at point of projection to its kinetic energy at highest point of flight will be $\left(\cos 60^{\circ}=0.5\right)$
Q41.
mcq single
+1 / 0
Two bodies with kinetic energies in the ratio $3: 1$ are moving with equal linear momentum. The ratio of their masses is
Q42.
mcq single
+1 / 0
In a circuit shown, if $10 \Omega$ resistor is replaced by $20 \Omega$, then the current drawn from the battery is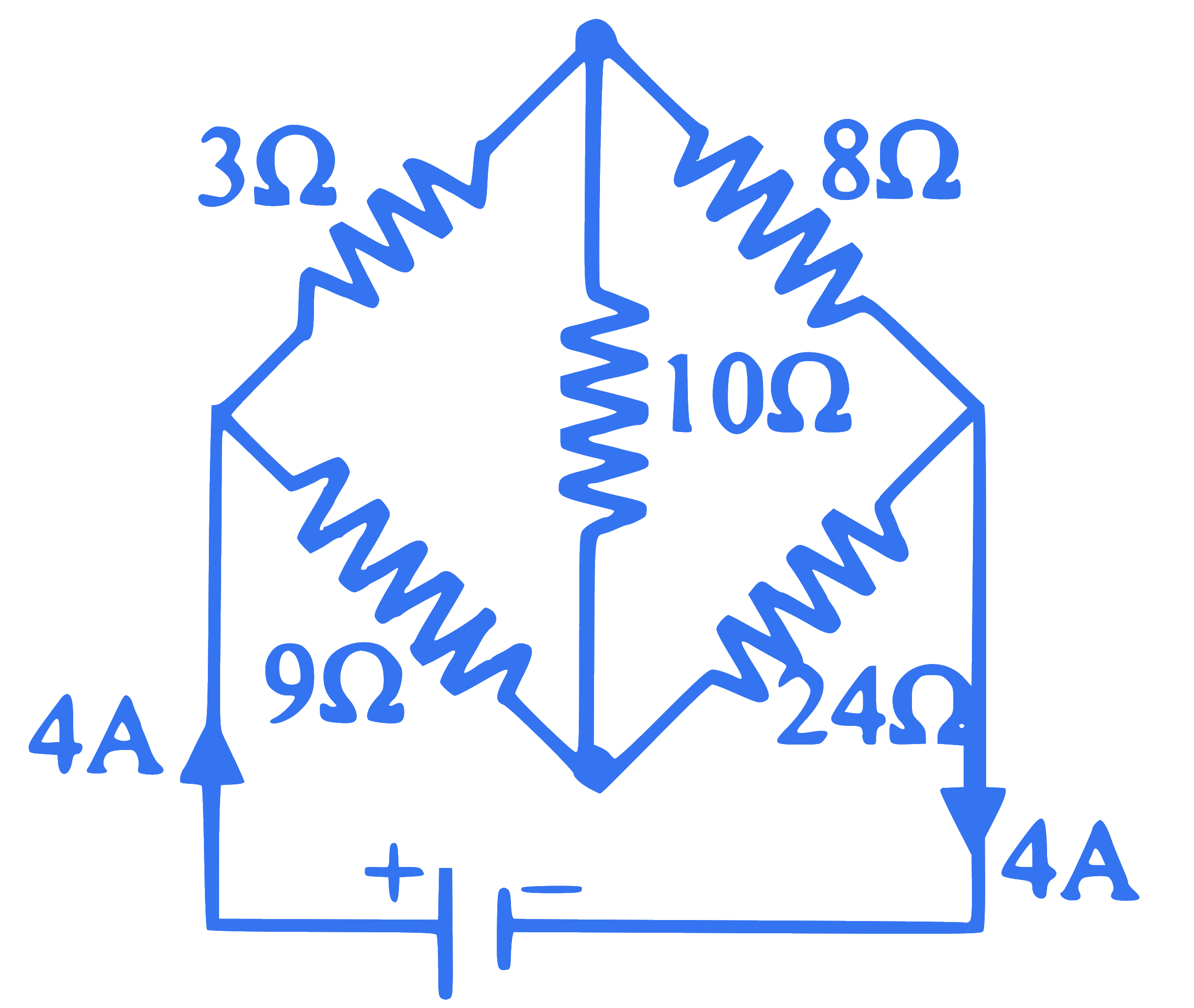

Q43.
mcq single
+1 / 0
A cell is balanced against 240 cm and 100 cm of potentiometer wire, respectively, when in open circuit and when short-circuited through a resistance of $5 \Omega$. The internal resistance of the cell is
Q44.
mcq single
+1 / 0
A physical quantity $\mathrm{P}=\frac{\mathrm{A}^3 \sqrt{\mathrm{~B}}}{\mathrm{C}^{-4} \mathrm{D}^{3 / 2}}$. Out of the quantities $\mathrm{A}, \mathrm{B}, \mathrm{C}$ and D , the quantity which brings maximum percentage error in P is
Q45.
mcq single
+1 / 0
The output Y of a given logic circuit is (For inputs $\mathrm{A}, \mathrm{B}$ and C )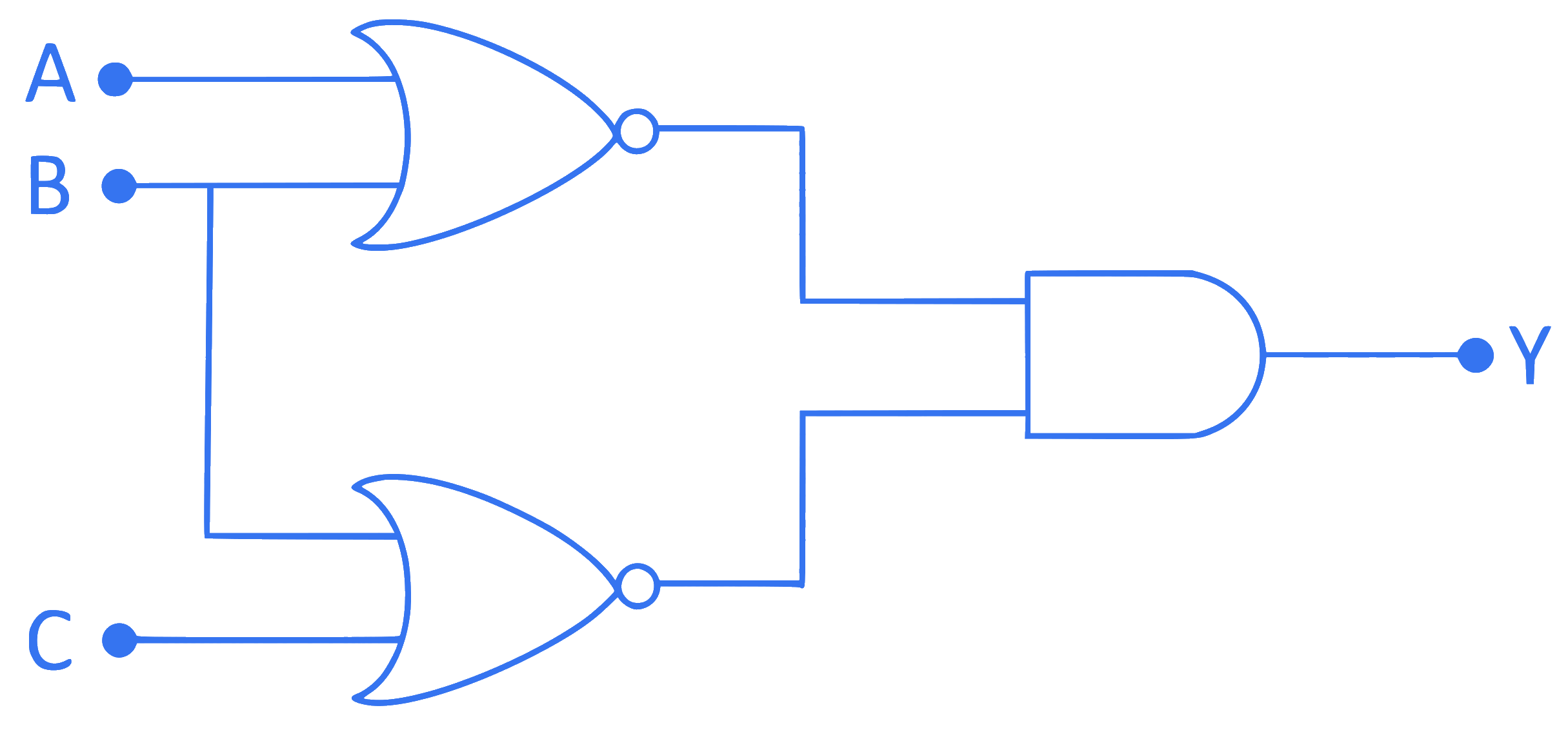

Q46.
mcq single
+1 / 0
The main difference in the working of a step-up transformer and CE amplifier is
Q47.
mcq single
+1 / 0
For a p-type semiconductor, the relation between the number density ' $\mathrm{n}_{\mathrm{h}}$ ' and ' $\mathrm{n}_{\mathrm{e}}$ ' is
Q48.
mcq single
+1 / 0
The frequency of oscillation of LCR series circuit is ' $F$ '. The value of capacitance is tripled, then new frequency becomes ' xF '. The value of ' $x$ ' is
Q49.
mcq single
+1 / 0
Out of the following graphs, the correct graphical relation for LC parallel resonant circuit at resonance is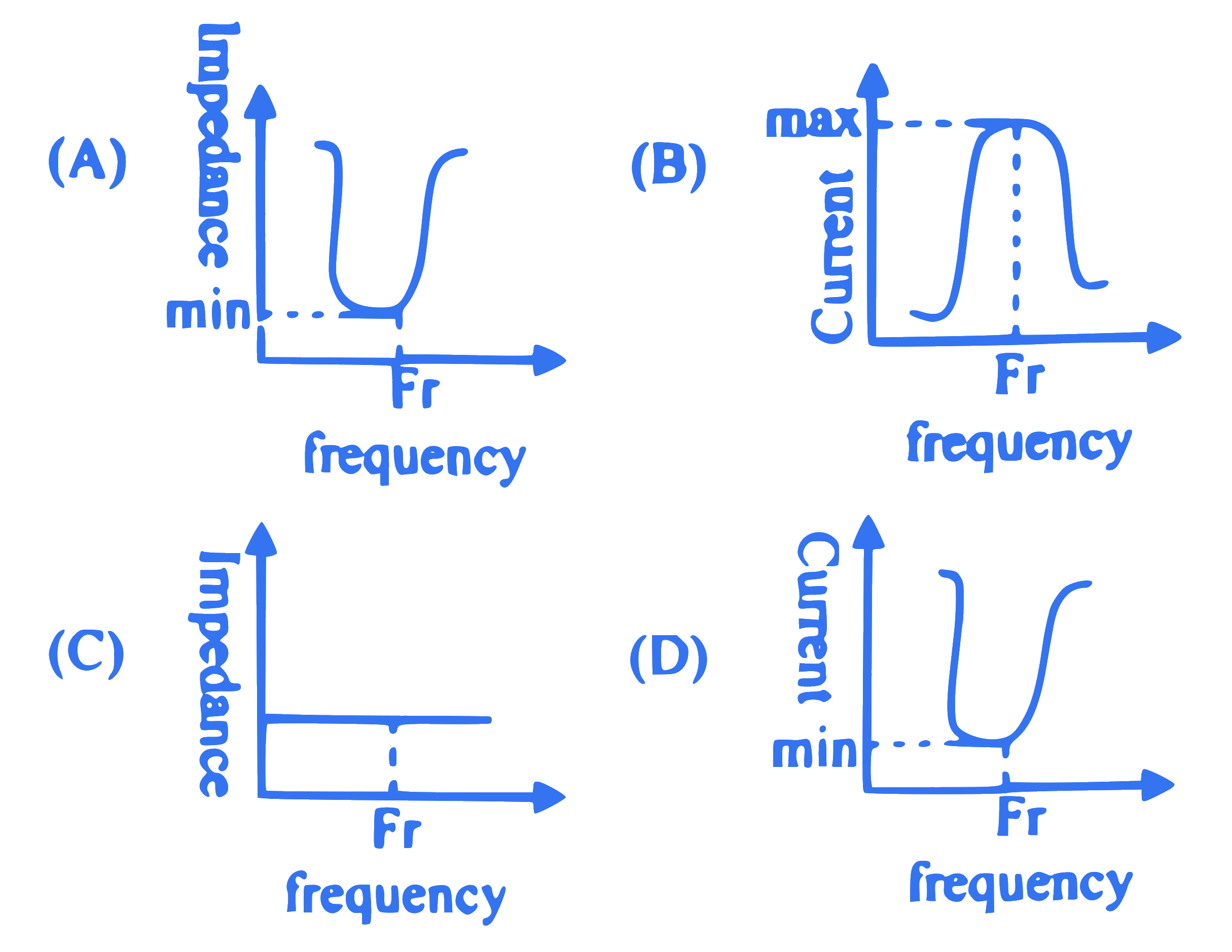

Q50.
mcq single
+1 / 0
At what angle should the two forces $2 \overrightarrow{\mathrm{P}}$ and $\sqrt{2} \overrightarrow{\mathrm{P}}$ act so that the resultant force is $\sqrt{10} \overrightarrow{\mathrm{P}}$ ?
Q51.
mcq single
+1 / 0
In Young's double slit experiment, waves from slits ' $\mathrm{S}_1$ ' and ' $\mathrm{S}_2$ ' have a path difference of $\frac{\lambda}{4}$ and $\frac{\lambda}{6}$ respectively at points $x$ and $y$ on the screen. The ratio of intensities at points $y$ to that at $x$ is $\left(\cos 45^{\circ}=\frac{1}{\sqrt{2}}, \cos 30^{\circ}=\sqrt{3} / 2,\right)$
Q52.
mcq single
+1 / 0
Monochromatic light of wavelength $4800 \mathop {\rm{A}}\limits^{\rm{o}}$ falls on slit of width ' $a$ '. For what value of ' $a$ ', the first maximum falls at $30^{\circ} .\left(\sin 30^{\circ}=0.5\right)$
Q53.
mcq single
+1 / 0
Two (A and B) pendulums begin to swing simultaneously. The first pendulum (A) makes five oscillations when the other (B) makes three oscillations. The ratio of the lengths of pendulum $A$ to that of $B$ is
Q54.
mcq single
+1 / 0
A particle of mass 0.02 kg executes S.H.M. about $\mathrm{x}=0$ under the influence of a force as shown in figure. The period of S.H.M. is

Q55.
mcq single
+1 / 0
A particle performs S.H.M. with amplitude 'A'. The speed of the particle is $\left(\frac{1}{3}\right)^{\text {rd }}$ of the maximum speed when its displacement from the mean position is
Q56.
mcq single
+1 / 0
Two bodies A and B have their moments of inertia I and 3I respectively about their axis of rotation. their kinetic energies of rotation are also equal. The ratio of angular momenta of body A to that of body B is
Q57.
mcq single
+1 / 0
An elastic conducting material is stretched into a circular loop. It is placed with its plane perpendicular to uniform magnetic field $\mathrm{B}=0.6 \mathrm{~T}$. When loop is released, the radius starts shrinking at constant rate $3 \mathrm{~cm} / \mathrm{s}$. The induced e.m.f. in the loop at an instant when the radius of the loop is 10 cm will be (in mV )
Q58.
mcq single
+1 / 0
Two coils having self-inductances $\mathrm{L}_1=75 \mathrm{mH}$ and $L_2=48 \mathrm{mH}$ are coupled with each other. If the mutual inductance of the coils is 37.2 mH , then coefficient of coupling will be
Q59.
mcq single
+1 / 0
A bucket filled with water is revolved in a vertical circle of radius 10 m and water just not fall down. The time period of revolution will be (acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2$ )
Q60.
mcq single
+1 / 0
The angular separation between the minute hand and the hour hand of clock 40 minutes passed after $12 \mathrm{p} . \mathrm{m}$. is
Q61.
mcq single
+1 / 0
In resistance thermometer, the resistance at $0^{\circ} \mathrm{C}$ and $100^{\circ} \mathrm{C}$ are $6.74 \Omega$ and $7.74 \Omega$ respectively. The temperature corresponding to $6.53 \Omega$ resistance is
Q62.
mcq single
+1 / 0
In thermodynamic process, for free expansion, out of the following select the 'WRONG' statement.
Q63.
mcq single
+1 / 0
The heat required to increase the temperature from 373 K to 573 K at constant volume for ideal monoatomic gas of 2 moles is ( $R=$ universal gas constant)
Q64.
mcq single
+1 / 0
A Carnot engine has an efficiency of $\frac{1}{6}$. When the temperature of sink is reduced by 62 K , its efficiency is doubled. The temperature of the source is
Q65.
mcq single
+1 / 0
An ideal gas in a container of volume 500 cc is at a pressure of $2 \times 10^5 \mathrm{~N} / \mathrm{m}^2$. The average kinetic energy of each molecule is $6 \times 10^{-21} \mathrm{~J}$. The number of gas molecules in the container is
Q66.
mcq single
+1 / 0
If the r.m.s. velocity of gas is V at temperature T , then
Q67.
mcq single
+1 / 0
In photoelectric effect experiment, the stopping potential for a given metal is $\mathrm{V}_0$ (in volt) when radiation of wavelength $\lambda_0$ is used. If radiation of wavelength $5 \lambda_0$ is used with the same metal, then the stopping potential is (in volt) ( $\mathrm{h}=$ Planck's constant, $\mathrm{c}=$ velocity of light, $\mathrm{e}=$ electronic charge)
Q68.
mcq single
+1 / 0
Two identical photocathodes receive light of frequencies ' $f 1$ ' and ' $f_2$ '. The velocity of the photoelectrons of mass ' $m$ ' emitted are respectively ' $\mathrm{v}_1$ ' and ' $\mathrm{v}_2$ '. Then the correct relation is ( $\mathrm{h}=$ Planck's constant)
Q69.
mcq single
+1 / 0
If 80 J of work is required in moving an electric charge of ' 2 C ' from a point where potential is " -8 V ' to another point where potential is ' $V$ ' volt, the value of ' $V$ ' is
Q70.
mcq single
+1 / 0
If the electric flux entering and leaving an enclosed surface area are ' $\phi_1$ ' and ' $\phi_2$ ' respectively, the electric charge inside the surface will be ( $\varepsilon_0=$ permittivity of free space)
Q71.
mcq single
+1 / 0
A charge of $8 \mu \mathrm{C}$ exists at the centre of the square $A B C D$. The work done in moving a $4 \mu C$ point charge from corner A to corner B is (Along side AB )
Q72.
mcq single
+1 / 0
An object is released from a distance ' $r$ ' from the centre of the earth. The velocity of the object at the time of striking the earth will be ( $R=$ radius of earth, $r>R, g=$ acceleration due to gravity)
Q73.
mcq single
+1 / 0
An air capacitor of plate area ' $A$ ' and separation between the plates is ' $d$ ' has a capacity ' $C$ '. Two dielectric slabs are inserted between its plates in two different manners as shown in figure. The capacitance of a capacitor is

Q74.
mcq single
+1 / 0
The capacitive reactance of a capacitor connected to 50 V d.c. is
Q75.
mcq single
+1 / 0
Light propagates 2 cm distance in glass of refractive index 1.5 in certain time. In the same time same light propogates a distance of 2.25 cm in a medium. The refractive index of the medium is
Q76.
mcq single
+1 / 0
Glass has refractive index ' $n$ ' with respect to air and ' $\theta$ ' is the critical angle for a ray of light going from glass to air. If a ray of light is incident from air on the glass with angle of incidence ' $\theta$ ', corresponding angle of refraction is
Q77.
mcq single
+1 / 0
Magnetic field at the centre of a circular loop of area ' $A$ ' is ' $B$ '. The magnetic moment of the loop is ' $x B$ '. The value of ' $x$ ' is ( $\mu_0=$ permeability of vacuum or free space)
Q78.
mcq single
+1 / 0
The current $I$ is flowing through a loop $A B C D A$ as shown in the figure. The magnitude of the magnetic field at the centre ' $O$ ' is $x$ times
$$ \left(\frac{\mu_0 \mathrm{I}}{\mathrm{R}}\right)\left(\mathrm{OD}=\mathrm{R}, \mathrm{DC}=\mathrm{R}, \angle \mathrm{AOD}=90^{\circ}\right) $$
The value of ' $x$ ' is ( $\mu_0=$ permeability of free space)

Q79.
mcq single
+1 / 0
Two solid spheres of same material but of mass ' M ' and ' 8 M ' fall simultaneously through a viscous liquid. Their terminal velocities are respectively ' V ' and ' nV '. The value ' n ' is
Q80.
mcq single
+1 / 0
When a big drop of water is formed from n small drops of water, the energy loss is 4 E , where E is the energy of the bigger drop. If $R$ and $r$ is the radius of the bigger and the smaller drop respectively then number of smaller drops $n$ is
Q81.
mcq single
+1 / 0
A small steel ball of mass ' M ', radius ' R ' and density ' $\rho$ ' falls with terminal velocity through a tube filled with glycerine of density ' $\sigma$ '. The viscous force acting on the steel ball is ( $\mathrm{g}=$ acceleration due to gravity)
Q82.
mcq single
+1 / 0
In the Bohr model of hydrogen atom, out of the following quantities, the principal quantum number is proportional to ( $\mathrm{R}, \mathrm{V}$ and E represent radius of the orbit, speed of electron and total energy of the electron respectively)
Q83.
mcq single
+1 / 0
According to Bohr's first postulate the kinetic energy $\frac{1}{2} m v^2$ of the electron in C.G.S. system is ( $\mathrm{m}=$ mass of electron, v is its velocity, r is the radius of the stationary orbit around the nucleus with charge Ze )
Q84.
mcq single
+1 / 0
A uniform metal wire has length ' $L$ ', mass ' $M$ ' and area of cross-section ' A '. It is under tension ' $T$ ' and ' $V$ ' is the speed of transverse wave along the wire. The density of the wire is
Q85.
mcq single
+1 / 0
The loudness and pitch of sound note respectively depend upon
Q86.
mcq single
+1 / 0
A transverse wave is moving along a string. The linear density of a vibrating string is $2 \times 10^{-3} \mathrm{~kg} / \mathrm{m}$. The wave equation is $y=0.02 \sin (2 x+10 t) m$. The tension in the string is
Q87.
mcq single
+1 / 0
An open organ pipe and a closed organ pipe have the frequency of their second overtone identical. The ratio of length of closed pipe to that of open pipe is (Neglect end correction)
Q88.
mcq single
+1 / 0
Out of the following properties describing diamagnetism, identify the property which is "WRONG"
Q89.
mcq single
+1 / 0
A stone is projected at an angle $60^{\circ}$ to the horizontal. The ratio of kinetic energy of the stone at point of projection to its kinetic energy at highest point of flight will be $\left(\cos 60^{\circ}=0.5\right)$
Q90.
mcq single
+1 / 0
Two bodies with kinetic energies in the ratio $3: 1$ are moving with equal linear momentum. The ratio of their masses is
Q91.
mcq single
+1 / 0
In a circuit shown, if $10 \Omega$ resistor is replaced by $20 \Omega$, then the current drawn from the battery is

Q92.
mcq single
+1 / 0
A cell is balanced against 240 cm and 100 cm of potentiometer wire, respectively, when in open circuit and when short-circuited through a resistance of $5 \Omega$. The internal resistance of the cell is
Q93.
mcq single
+1 / 0
A physical quantity $\mathrm{P}=\frac{\mathrm{A}^3 \sqrt{\mathrm{~B}}}{\mathrm{C}^{-4} \mathrm{D}^{3 / 2}}$. Out of the quantities $\mathrm{A}, \mathrm{B}, \mathrm{C}$ and D , the quantity which brings maximum percentage error in P is
Q94.
mcq single
+1 / 0
The output Y of a given logic circuit is (For inputs $\mathrm{A}, \mathrm{B}$ and C )

Q95.
mcq single
+1 / 0
The main difference in the working of a step-up transformer and CE amplifier is
Q96.
mcq single
+1 / 0
For a p-type semiconductor, the relation between the number density ' $\mathrm{n}_{\mathrm{h}}$ ' and ' $\mathrm{n}_{\mathrm{e}}$ ' is
Q97.
mcq single
+1 / 0
The frequency of oscillation of LCR series circuit is ' $F$ '. The value of capacitance is tripled, then new frequency becomes ' xF '. The value of ' $x$ ' is
Q98.
mcq single
+1 / 0
Out of the following graphs, the correct graphical relation for LC parallel resonant circuit at resonance is

Biology
Biology
Q1.
mcq single
+1 / 0
Eremothecium ashbyi is used to obtain __________ .
Q2.
mcq single
+1 / 0
Match Column I containing names of enzymes with Column II containing their microbial source.
Column I
Column II
i.
Invertase
a.
Sclerotinialibertine
ii.
Pectinase
b.
Trichoderma koningii
iii.
Lipase
c.
Saccharomyces cerevisiae
iv.
Cellulase
d.
Candida lipolytica
Choose the correct option:
Q3.
mcq single
+1 / 0
Which one of the following is a non-symbiotic biofertilizer?
Q4.
mcq single
+1 / 0
Which one of the ecosystem service is necessary for crop production and fruit formation?
Q5.
mcq single
+1 / 0
Detritus food chain begins with ___________ .
Q6.
mcq single
+1 / 0
Following components of the ecosystem are all linked to function as an 'ecosystem unit' EXCEPT ________
Q7.
mcq single
+1 / 0
The terminal stage of xerarch succession is represented by _________ .
Q8.
mcq single
+1 / 0
Red (RR) flowered plant of Mirabilis is crossed with white (rr) flowered plant. All the offsprings in $\mathrm{F}_1$ generation are pink ( Rr ). This is an indication that the ' R ' gene is __________
Q9.
mcq single
+1 / 0
Which one of the following is NOT a sex linked disorder?
Q10.
mcq single
+1 / 0
Which one of the following is an important symptom of haemophilia?
Q11.
mcq single
+1 / 0
What is the probability of producing a colourblind son if a colourblind male marries a normal visioned female?
Q12.
mcq single
+1 / 0
Non-disjunction of X - chromosome during meiosis leads to __________
Q13.
mcq single
+1 / 0
A large population is showing random mating and not undergoing any evolutionary change. If frequency of allele A is 0.6 , then find out the percentage of homozygous recessive and heterozygous organisms respectively?
Q14.
mcq single
+1 / 0
The palate separates _________ .
Q15.
mcq single
+1 / 0
Given below are two statements.
Statement I -The right lung is larger and divided into two lobes.
Statement II -The left lung is smaller and divided into three lobes.
In the light of above statements, select the correct option given below:
Q16.
mcq single
+1 / 0
Select INCORRECT statement about the Hering-Breuer reflex.
Q17.
mcq single
+1 / 0
Elimination of defective gene and insertion of healthy gene in a genome is achieved by ________
Q18.
mcq single
+1 / 0
Which one of the following is NOT an example of unconditioned reflex?
Q19.
mcq single
+1 / 0
Match parts of human ear in Column I with their structure / characteristic in Column II.
Column I
Column II
i.
Organ of Corti
a.
Dynamic balance is maintained
ii.
Semi-circular Canal
b.
Sensory epithelium over basilar membrane of cochlea
iii.
Tympanic membrane
c.
Connects middle ear to the pharynx
iv.
Eustachian tube
d.
Transmits sound waves to middle ear
Choose the correct option:
Q20.
mcq single
+1 / 0
Smallest branch of mixed spinal nerve is $\qquad$
Q21.
mcq single
+1 / 0
Match the properties of nerve fibres in Column I with their explanation in Column II
Column I
Column II
i.
Irritability
a.
Halting of nerve impulse transmission due to exhaustion of neurotransmitter.
ii.
Synaptic fatigue
b.
Ability to perceive stimulus and enter into a state of activity.
iii.
Conductivity
c.
Many subliminal stimuli are given in quick succession.
iv.
Summation effect
d.
Ability to transmit the excitation.
Choose the correct option:
Q22.
mcq single
+1 / 0
Select the INCORRECT statement regarding spinal cord from the following.
Q23.
mcq single
+1 / 0
Action of salivary amylase stops when food gets mixed with gastric juice due to __________
Q24.
mcq single
+1 / 0
Glisson's capsule is _________
Q25.
mcq single
+1 / 0
Match the types of glands in Column I with their location in Column II
Column I
Column II
i.
Parotid gland
a.
Below the lower jaw
ii.
Submandibular gland
b.
In front of the ear
iii.
Sublingual gland
c.
Submucosa of small intestine
iv.
Brunner's gland
d.
Below the tongue
Choose the correct option
Q26.
mcq single
+1 / 0
Select the INCORRECT statement regarding physiology of digestion.
Q27.
mcq single
+1 / 0
The enzyme enterokinase helps in conversion of _________.
Q28.
mcq single
+1 / 0
Exons on hnRNA codes for $\qquad$
Q29.
mcq single
+1 / 0
In an operon, when repressor protein attaches to the operator then __________ .
Q30.
mcq single
+1 / 0
The very first step of initiation of translation is
Q31.
mcq single
+1 / 0
Identify the correct sequence of steps in Hershey and Chase's experiment.
Q32.
mcq single
+1 / 0
Given below are two statements.
Statement I - In Meselson and Stahl's experiment the samples were separated by using CsCl equilibrium gradients.
Statement II - ${ }^{15} \mathrm{~N}$ is a heavy isotope and can be separated based on density gradient.
In the light of above statements, select the correct option given below:
Q33.
mcq single
+1 / 0
In which one of the following, treatment includes use of protective mask and gear to minimize inflammation?
Q34.
mcq single
+1 / 0
Which among the following disease is called as modern pandemic?
Q35.
mcq single
+1 / 0
Reason for introducing vaccine into the body is ___________.
Q36.
mcq single
+1 / 0
Transmission of malaria is through __________
Q37.
mcq single
+1 / 0
Given below are two statements about sap in xylem.
Statement I - Under normal conditions the sap in the xylem cell shows negative hydrostatic pressure.
Statement II - It also shows very low osmotic pressure.
In the light of above statements, Select the correct option given below:
Q38.
mcq single
+1 / 0
Given below are two statements about absorption of water in plants.
Statement I - Water passes into the interface of cell wall and plasma membrane by diffusion.
Statement II -Water at the interface of cell wall and plasma membrane enters into the cytoplasm of the cell by osmosis.
In the light of above statements, select the correct option given below:
Q39.
mcq single
+1 / 0
An additional apoplastic route that allows direct access to xylem in plants is along the secondary roots because they __________
Q40.
mcq single
+1 / 0
For facilitated diffusion in plant________ is essential.
Q41.
mcq single
+1 / 0
When roots are deprived of __________ they show a sudden drop in active absorption.
Q42.
mcq single
+1 / 0
Cuticular transpiration contributes to __________ $\%$ of total transpiration in plants.
Q43.
mcq single
+1 / 0
Given below are two statements.
Statement I - There are different modes of asexual reproduction in plants.
Statement II - Sexual reproduction in plants, involves two major events.
In the light of above statements, select the correct option given below:
Q44.
mcq single
+1 / 0
Noise can cause the following EXCEPT __________.
Q45.
mcq single
+1 / 0
'Algal bloom' mainly includes _________ .
Q46.
mcq single
+1 / 0
The purpose of biological treatment of sewage is to __________ .
Q47.
mcq single
+1 / 0
Which of the following depicts Logistic growth curve of population?
Q48.
mcq single
+1 / 0
Select the correct sequential level with decreasing complexity of ecological organisation.
Q49.
mcq single
+1 / 0
Which one of the following does not show suspend type of adaptation?
Q50.
mcq single
+1 / 0
Given below are two statements.
Statement I - Osmoconformers are animals, that are iso-osmotic to the surroundings.
Statement II - Marine organisms mostly are osmoconformers because their body fluids and external environment are iso-osmotic in nature. In the light of above statements, select the correct option given below:
Q51.
mcq single
+1 / 0
Number of deaths with no competition and abundance of resources such as food and water is ________.
Q52.
mcq single
+1 / 0
Match the Column I with Column II regarding cardiac cycle.
Column I
Column II
i.
Atrial diastole (AD)
a.
0.5 sec
ii.
Atrial systole (AS)
b.
0.7 sec
iii.
Ventricular systole (VS)
c.
0.1 sec
iv.
Ventricular diastole (VD)
d.
0.3 sec
Choose the correct option:
Q53.
mcq single
+1 / 0
By using methylene blue, which of the following leucocytes can be stained and identified?
Q54.
mcq single
+1 / 0
Given below are two statements with reference to measurement of blood pressure.
Statement I - The appearance of Korotkoff sounds for the first time due to release of external pressure indicates diastolic pressure. Statement II - The disappearance of Korotkoff sounds for the first time due to release of external pressure indicates systolic pressure.
In the light of above statements, select the correct option given below:
Q55.
mcq single
+1 / 0
If Rh antigens are introduced into the body of $\mathrm{Rh}^{-\mathrm{ve}}$ person for second time, then it leads to _________
Q56.
mcq single
+1 / 0
How many electrodes are generally positioned on chest during recording of ECG?
Q57.
mcq single
+1 / 0
Given below are two statements.
Statement I - In testis, vasa efferentia connect the rete testis with head of epididymis.
Statement II - The vas deferens arises from the corpus or body of epididymis.
In the light of above statements, select the correct option given below:
Q58.
mcq single
+1 / 0
Fluid filled antrum is present between secondary oocyte and _________
Q59.
mcq single
+1 / 0
Select INCORRECT statement regarding cells of Sertoli.
Q60.
mcq single
+1 / 0
Select the INCORRECT statement regarding the prostate gland.
Q61.
mcq single
+1 / 0
Tremor of fingers, increased irritability, tachycardia, weight loss, etc are the symptoms of _________ .
Q62.
mcq single
+1 / 0
Match the pair.
Column I
(Hormone)
Column II
(Function)
i.
Erythropoietin
a.
absorption of dietary calcium
ii.
Calcitriol
b.
water retention during severe dehydration
iii.
Renin
c.
production of RBC
iv.
Antidiuretic hormone
d.
increases blood pressure
Choose the correct option:
Q63.
mcq single
+1 / 0
Match the Column I with Column II regarding adrenal gland.
Column I
Column II
i.
Zona glomerulosa
a.
Nor- epinephrine
ii.
Zona fasciculata
b.
Aldosterone
iii.
Zona Reticularis
c.
Cortisol
iv.
Adrenal medulla
d.
Androgens and Estradiols
Choose the correct option:
Q64.
mcq single
+1 / 0
Which one of the following is a salt- retaining hormone?
Q65.
mcq single
+1 / 0
Which of the following cells are present in maximum (highest) number in the Islet of Langerhans?
Q66.
mcq single
+1 / 0
Following are modern techniques used to protect endangered species EXCEPT _________
Q67.
mcq single
+1 / 0
Match the items given in Column I with their number found in India given in Column II
Column I
Column II
i.
National Parks
a.
14
ii.
Biosphere reserves
b.
90
iii.
Varieties of mango
c.
448
iv.
Wildlife Sanctuaries
d.
1000
Choose the correct option
Q68.
mcq single
+1 / 0
While describing the dynamics of growth in plants, Relative Growth Rate (RGR) is determined by the following formula.
Q69.
mcq single
+1 / 0
Which one of the following phytohormones causes epinasty?
Q70.
mcq single
+1 / 0
Match the types of plants in Column I with the time of the year when they flower given in Column II.
Column I
Column II
i.
Short day plants
a.
throughout the year
ii.
Long day plants
b.
in winter
iii.
Day neutral Plants
c.
in summer
Choose the correct option:
Q71.
mcq single
+1 / 0
Which one of the following is the activator for enzyme enolase in glycolysis?
Q72.
mcq single
+1 / 0
Given below are two statements.
Statement I - F 1 part of oxysome catalyse the synthesis of ATP from ADP
Statement II - Protons are channeled from outer chamber of mitochondria into inner chamber by $\mathrm{F}_0$ part of oxysome.
In the light of above statements, select the correct option given below:
Q73.
mcq single
+1 / 0
During aerobic respiration, out of 38 ATP molecules produced from one glucose, how many ATP molecules are formed through ETS?
Q74.
mcq single
+1 / 0
Which one of the following is a correct statement?
Q75.
mcq single
+1 / 0
Which one of the following occurs during glycolysis?
Q76.
mcq single
+1 / 0
Connecting link between ape and man is ____________ in the course of human evolution.
Q77.
mcq single
+1 / 0
Select the correct pair regarding origin of life
Q78.
mcq single
+1 / 0
Example of geographical isolation is __________
Q79.
mcq single
+1 / 0
According to Darwinism, __________ leads to evolution.
Q80.
mcq single
+1 / 0
People who don't drink enough water and consume high protein diet, may suffer from
__________ kidney stones.
Q81.
mcq single
+1 / 0
Role of Angiotensin converting enzyme is ___________.
Q82.
mcq single
+1 / 0
The Krebs-Henseleit cycle of urea formation is a sequence of biochemical reactions that takes place in___________
Q83.
mcq single
+1 / 0
Renal medulla contains __________ .
i. Malpighian bodies and convoluted tubules
ii. blood vessels and glomerulus
iii. Loop of Henle
iv. Collecting ducts
Q84.
mcq single
+1 / 0
Legal framework for MTP act 2017 aims for following EXCEPT_________
Q85.
mcq single
+1 / 0
When there is blockage in the fallopian tube, zygote is transferred to fallopian tube for further development. This is __________ type of ART.
Q86.
mcq single
+1 / 0
Given below are two statements.
Statement I - Microspore is covered by two layered covering called sporoderm.
Statement II - Sporopollenin is not present in the region of germ pores in microspore.
In the light of above statements, Select the correct option given below:
Q87.
mcq single
+1 / 0
In the given diagram of developing monocot embryo, identify the labels 'i', 'ii' and 'iii'.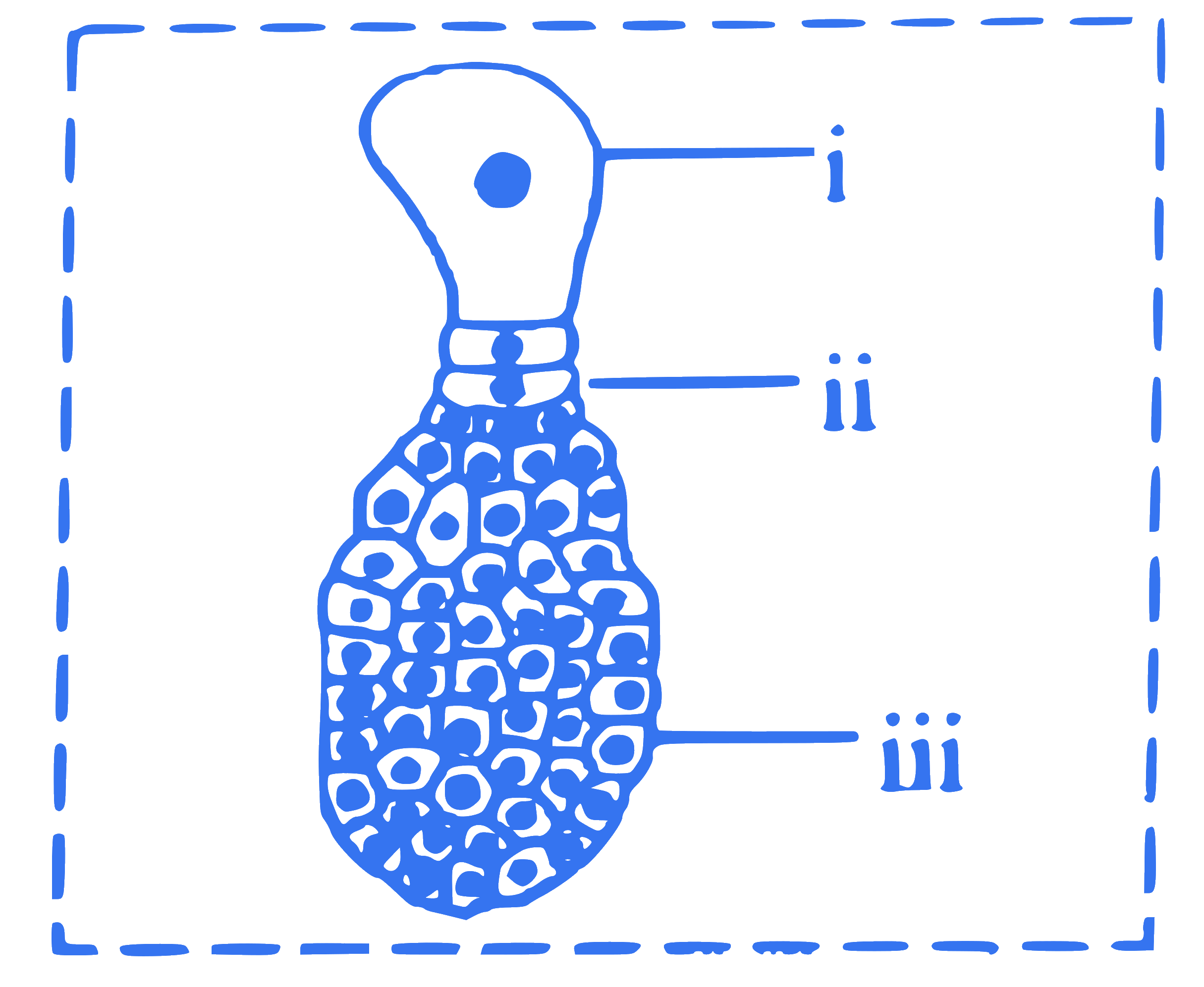

Q88.
mcq single
+1 / 0
Match the parts of anatropous ovule given in Column I with their ploidy given in Column II.
Column I
Column II
i.
Antipodals
a.
2n
ii.
Nucellus.
b.
3n
iii.
PEN
c.
n
Choose the correct option given below
Q89.
mcq single
+1 / 0
Which of the following plant shows 'Lever mechanism' of pollination?
Q90.
mcq single
+1 / 0
How many meiotic and mitotic divisions respectively are required to form male gametophyte from pollen mother cell?
Q91.
mcq single
+1 / 0
Given below are two statements.
Statement I - All monosaccharides are reducing sugars.
Statement II - Polysaccharides are too big to escape from the cell.
In the light of above statements, select the correct option given below:
Q92.
mcq single
+1 / 0
Which secondary metabolites are added to wines to improve astringency?
Q93.
mcq single
+1 / 0
Number of pyrimidine bases found in nucleic acids is / are
Q94.
mcq single
+1 / 0
Types of Carbohydrates are given in Column I while their examples are given in Column II.
Column I
Column II
i.
Monosaccharide
a.
Malt Sugar
ii.
Heteropolysaccharide
b.
Glycogen
iii.
Disaccharide
c.
Hyaluronic acid
iv.
Homopolysaccharide
d.
Glyceraldehyde
Choose the correct option:
Q95.
mcq single
+1 / 0
Which one of the following is NOT a technique used for direct transfer of foreign DNA into the protoplast of host cell without using vector?
Q96.
mcq single
+1 / 0
$$ \text { Match the restriction enzyme in Column I with their recognition sequences in Column II. } $$
 Choose the correct option:
Choose the correct option:
 Choose the correct option:
Choose the correct option:
Q97.
mcq single
+1 / 0
Which of the following technique is used for invitro amplification of gene?
Q98.
mcq single
+1 / 0
The transfer of rDNA into a bacterial cell for the purpose of its cloning, is assisted by ________ ions.
Q99.
mcq single
+1 / 0
In tissue culture technique, to maintain aseptic conditions the nutrient medium is autoclaved at constant pressure of _________ $\mathrm{lb} / \mathrm{sq}$ inch for continuous __________ minutes.
Q100.
mcq single
+1 / 0
Eremothecium ashbyi is used to obtain __________ .
Q101.
mcq single
+1 / 0
Match Column I containing names of enzymes with Column II containing their microbial source.
Column I
Column II
i.
Invertase
a.
Sclerotinialibertine
ii.
Pectinase
b.
Trichoderma koningii
iii.
Lipase
c.
Saccharomyces cerevisiae
iv.
Cellulase
d.
Candida lipolytica
Choose the correct option:
Q102.
mcq single
+1 / 0
Which one of the following is a non-symbiotic biofertilizer?
Q103.
mcq single
+1 / 0
Which one of the ecosystem service is necessary for crop production and fruit formation?
Q104.
mcq single
+1 / 0
Detritus food chain begins with ___________ .
Q105.
mcq single
+1 / 0
Following components of the ecosystem are all linked to function as an 'ecosystem unit' EXCEPT ________
Q106.
mcq single
+1 / 0
The terminal stage of xerarch succession is represented by _________ .
Q107.
mcq single
+1 / 0
Red (RR) flowered plant of Mirabilis is crossed with white (rr) flowered plant. All the offsprings in $\mathrm{F}_1$ generation are pink ( Rr ). This is an indication that the ' R ' gene is __________
Q108.
mcq single
+1 / 0
Which one of the following is NOT a sex linked disorder?
Q109.
mcq single
+1 / 0
Which one of the following is an important symptom of haemophilia?
Q110.
mcq single
+1 / 0
What is the probability of producing a colourblind son if a colourblind male marries a normal visioned female?
Q111.
mcq single
+1 / 0
Non-disjunction of X - chromosome during meiosis leads to __________
Q112.
mcq single
+1 / 0
A large population is showing random mating and not undergoing any evolutionary change. If frequency of allele A is 0.6 , then find out the percentage of homozygous recessive and heterozygous organisms respectively?
Q113.
mcq single
+1 / 0
The palate separates _________ .
Q114.
mcq single
+1 / 0
Given below are two statements.
Statement I -The right lung is larger and divided into two lobes.
Statement II -The left lung is smaller and divided into three lobes.
In the light of above statements, select the correct option given below:
Q115.
mcq single
+1 / 0
Select INCORRECT statement about the Hering-Breuer reflex.
Q116.
mcq single
+1 / 0
Elimination of defective gene and insertion of healthy gene in a genome is achieved by ________
Q117.
mcq single
+1 / 0
Which one of the following is NOT an example of unconditioned reflex?
Q118.
mcq single
+1 / 0
Match parts of human ear in Column I with their structure / characteristic in Column II.
Column I
Column II
i.
Organ of Corti
a.
Dynamic balance is maintained
ii.
Semi-circular Canal
b.
Sensory epithelium over basilar membrane of cochlea
iii.
Tympanic membrane
c.
Connects middle ear to the pharynx
iv.
Eustachian tube
d.
Transmits sound waves to middle ear
Choose the correct option:
Q119.
mcq single
+1 / 0
Smallest branch of mixed spinal nerve is $\qquad$
Q120.
mcq single
+1 / 0
Match the properties of nerve fibres in Column I with their explanation in Column II
Column I
Column II
i.
Irritability
a.
Halting of nerve impulse transmission due to exhaustion of neurotransmitter.
ii.
Synaptic fatigue
b.
Ability to perceive stimulus and enter into a state of activity.
iii.
Conductivity
c.
Many subliminal stimuli are given in quick succession.
iv.
Summation effect
d.
Ability to transmit the excitation.
Choose the correct option:
Q121.
mcq single
+1 / 0
Select the INCORRECT statement regarding spinal cord from the following.
Q122.
mcq single
+1 / 0
Action of salivary amylase stops when food gets mixed with gastric juice due to __________
Q123.
mcq single
+1 / 0
Glisson's capsule is _________
Q124.
mcq single
+1 / 0
Match the types of glands in Column I with their location in Column II
Column I
Column II
i.
Parotid gland
a.
Below the lower jaw
ii.
Submandibular gland
b.
In front of the ear
iii.
Sublingual gland
c.
Submucosa of small intestine
iv.
Brunner's gland
d.
Below the tongue
Choose the correct option
Q125.
mcq single
+1 / 0
Select the INCORRECT statement regarding physiology of digestion.
Q126.
mcq single
+1 / 0
The enzyme enterokinase helps in conversion of _________.
Q127.
mcq single
+1 / 0
Exons on hnRNA codes for $\qquad$
Q128.
mcq single
+1 / 0
In an operon, when repressor protein attaches to the operator then __________ .
Q129.
mcq single
+1 / 0
The very first step of initiation of translation is
Q130.
mcq single
+1 / 0
Identify the correct sequence of steps in Hershey and Chase's experiment.
Q131.
mcq single
+1 / 0
Given below are two statements.
Statement I - In Meselson and Stahl's experiment the samples were separated by using CsCl equilibrium gradients.
Statement II - ${ }^{15} \mathrm{~N}$ is a heavy isotope and can be separated based on density gradient.
In the light of above statements, select the correct option given below:
Q132.
mcq single
+1 / 0
In which one of the following, treatment includes use of protective mask and gear to minimize inflammation?
Q133.
mcq single
+1 / 0
Which among the following disease is called as modern pandemic?
Q134.
mcq single
+1 / 0
Reason for introducing vaccine into the body is ___________.
Q135.
mcq single
+1 / 0
Transmission of malaria is through __________
Q136.
mcq single
+1 / 0
Given below are two statements about sap in xylem.
Statement I - Under normal conditions the sap in the xylem cell shows negative hydrostatic pressure.
Statement II - It also shows very low osmotic pressure.
In the light of above statements, Select the correct option given below:
Q137.
mcq single
+1 / 0
Given below are two statements about absorption of water in plants.
Statement I - Water passes into the interface of cell wall and plasma membrane by diffusion.
Statement II -Water at the interface of cell wall and plasma membrane enters into the cytoplasm of the cell by osmosis.
In the light of above statements, select the correct option given below:
Q138.
mcq single
+1 / 0
An additional apoplastic route that allows direct access to xylem in plants is along the secondary roots because they __________
Q139.
mcq single
+1 / 0
For facilitated diffusion in plant________ is essential.
Q140.
mcq single
+1 / 0
When roots are deprived of __________ they show a sudden drop in active absorption.
Q141.
mcq single
+1 / 0
Cuticular transpiration contributes to __________ $\%$ of total transpiration in plants.
Q142.
mcq single
+1 / 0
Given below are two statements.
Statement I - There are different modes of asexual reproduction in plants.
Statement II - Sexual reproduction in plants, involves two major events.
In the light of above statements, select the correct option given below:
Q143.
mcq single
+1 / 0
Noise can cause the following EXCEPT __________.
Q144.
mcq single
+1 / 0
'Algal bloom' mainly includes _________ .
Q145.
mcq single
+1 / 0
The purpose of biological treatment of sewage is to __________ .
Q146.
mcq single
+1 / 0
Which of the following depicts Logistic growth curve of population?
Q147.
mcq single
+1 / 0
Select the correct sequential level with decreasing complexity of ecological organisation.
Q148.
mcq single
+1 / 0
Which one of the following does not show suspend type of adaptation?
Q149.
mcq single
+1 / 0
Given below are two statements.
Statement I - Osmoconformers are animals, that are iso-osmotic to the surroundings.
Statement II - Marine organisms mostly are osmoconformers because their body fluids and external environment are iso-osmotic in nature. In the light of above statements, select the correct option given below:
Q150.
mcq single
+1 / 0
Number of deaths with no competition and abundance of resources such as food and water is ________.
Q151.
mcq single
+1 / 0
Match the Column I with Column II regarding cardiac cycle.
Column I
Column II
i.
Atrial diastole (AD)
a.
0.5 sec
ii.
Atrial systole (AS)
b.
0.7 sec
iii.
Ventricular systole (VS)
c.
0.1 sec
iv.
Ventricular diastole (VD)
d.
0.3 sec
Choose the correct option:
Q152.
mcq single
+1 / 0
By using methylene blue, which of the following leucocytes can be stained and identified?
Q153.
mcq single
+1 / 0
Given below are two statements with reference to measurement of blood pressure.
Statement I - The appearance of Korotkoff sounds for the first time due to release of external pressure indicates diastolic pressure. Statement II - The disappearance of Korotkoff sounds for the first time due to release of external pressure indicates systolic pressure.
In the light of above statements, select the correct option given below:
Q154.
mcq single
+1 / 0
If Rh antigens are introduced into the body of $\mathrm{Rh}^{-\mathrm{ve}}$ person for second time, then it leads to _________
Q155.
mcq single
+1 / 0
How many electrodes are generally positioned on chest during recording of ECG?
Q156.
mcq single
+1 / 0
Given below are two statements.
Statement I - In testis, vasa efferentia connect the rete testis with head of epididymis.
Statement II - The vas deferens arises from the corpus or body of epididymis.
In the light of above statements, select the correct option given below:
Q157.
mcq single
+1 / 0
Fluid filled antrum is present between secondary oocyte and _________
Q158.
mcq single
+1 / 0
Select INCORRECT statement regarding cells of Sertoli.
Q159.
mcq single
+1 / 0
Select the INCORRECT statement regarding the prostate gland.
Q160.
mcq single
+1 / 0
Tremor of fingers, increased irritability, tachycardia, weight loss, etc are the symptoms of _________ .
Q161.
mcq single
+1 / 0
Match the pair.
Column I
(Hormone)
Column II
(Function)
i.
Erythropoietin
a.
absorption of dietary calcium
ii.
Calcitriol
b.
water retention during severe dehydration
iii.
Renin
c.
production of RBC
iv.
Antidiuretic hormone
d.
increases blood pressure
Choose the correct option:
Q162.
mcq single
+1 / 0
Match the Column I with Column II regarding adrenal gland.
Column I
Column II
i.
Zona glomerulosa
a.
Nor- epinephrine
ii.
Zona fasciculata
b.
Aldosterone
iii.
Zona Reticularis
c.
Cortisol
iv.
Adrenal medulla
d.
Androgens and Estradiols
Choose the correct option:
Q163.
mcq single
+1 / 0
Which one of the following is a salt- retaining hormone?
Q164.
mcq single
+1 / 0
Which of the following cells are present in maximum (highest) number in the Islet of Langerhans?
Q165.
mcq single
+1 / 0
Following are modern techniques used to protect endangered species EXCEPT _________
Q166.
mcq single
+1 / 0
Match the items given in Column I with their number found in India given in Column II
Column I
Column II
i.
National Parks
a.
14
ii.
Biosphere reserves
b.
90
iii.
Varieties of mango
c.
448
iv.
Wildlife Sanctuaries
d.
1000
Choose the correct option
Q167.
mcq single
+1 / 0
While describing the dynamics of growth in plants, Relative Growth Rate (RGR) is determined by the following formula.
Q168.
mcq single
+1 / 0
Which one of the following phytohormones causes epinasty?
Q169.
mcq single
+1 / 0
Match the types of plants in Column I with the time of the year when they flower given in Column II.
Column I
Column II
i.
Short day plants
a.
throughout the year
ii.
Long day plants
b.
in winter
iii.
Day neutral Plants
c.
in summer
Choose the correct option:
Q170.
mcq single
+1 / 0
Which one of the following is the activator for enzyme enolase in glycolysis?
Q171.
mcq single
+1 / 0
Given below are two statements.
Statement I - F 1 part of oxysome catalyse the synthesis of ATP from ADP
Statement II - Protons are channeled from outer chamber of mitochondria into inner chamber by $\mathrm{F}_0$ part of oxysome.
In the light of above statements, select the correct option given below:
Q172.
mcq single
+1 / 0
During aerobic respiration, out of 38 ATP molecules produced from one glucose, how many ATP molecules are formed through ETS?
Q173.
mcq single
+1 / 0
Which one of the following is a correct statement?
Q174.
mcq single
+1 / 0
Which one of the following occurs during glycolysis?
Q175.
mcq single
+1 / 0
Connecting link between ape and man is ____________ in the course of human evolution.
Q176.
mcq single
+1 / 0
Select the correct pair regarding origin of life
Q177.
mcq single
+1 / 0
Example of geographical isolation is __________
Q178.
mcq single
+1 / 0
According to Darwinism, __________ leads to evolution.
Q179.
mcq single
+1 / 0
People who don't drink enough water and consume high protein diet, may suffer from
__________ kidney stones.
Q180.
mcq single
+1 / 0
Role of Angiotensin converting enzyme is ___________.
Q181.
mcq single
+1 / 0
The Krebs-Henseleit cycle of urea formation is a sequence of biochemical reactions that takes place in___________
Q182.
mcq single
+1 / 0
Renal medulla contains __________ .
i. Malpighian bodies and convoluted tubules
ii. blood vessels and glomerulus
iii. Loop of Henle
iv. Collecting ducts
Q183.
mcq single
+1 / 0
Legal framework for MTP act 2017 aims for following EXCEPT_________
Q184.
mcq single
+1 / 0
When there is blockage in the fallopian tube, zygote is transferred to fallopian tube for further development. This is __________ type of ART.
Q185.
mcq single
+1 / 0
Given below are two statements.
Statement I - Microspore is covered by two layered covering called sporoderm.
Statement II - Sporopollenin is not present in the region of germ pores in microspore.
In the light of above statements, Select the correct option given below:
Q186.
mcq single
+1 / 0
In the given diagram of developing monocot embryo, identify the labels 'i', 'ii' and 'iii'.

Q187.
mcq single
+1 / 0
Match the parts of anatropous ovule given in Column I with their ploidy given in Column II.
Column I
Column II
i.
Antipodals
a.
2n
ii.
Nucellus.
b.
3n
iii.
PEN
c.
n
Choose the correct option given below
Q188.
mcq single
+1 / 0
Which of the following plant shows 'Lever mechanism' of pollination?
Q189.
mcq single
+1 / 0
How many meiotic and mitotic divisions respectively are required to form male gametophyte from pollen mother cell?
Q190.
mcq single
+1 / 0
Given below are two statements.
Statement I - All monosaccharides are reducing sugars.
Statement II - Polysaccharides are too big to escape from the cell.
In the light of above statements, select the correct option given below:
Q191.
mcq single
+1 / 0
Which secondary metabolites are added to wines to improve astringency?
Q192.
mcq single
+1 / 0
Number of pyrimidine bases found in nucleic acids is / are
Q193.
mcq single
+1 / 0
Types of Carbohydrates are given in Column I while their examples are given in Column II.
Column I
Column II
i.
Monosaccharide
a.
Malt Sugar
ii.
Heteropolysaccharide
b.
Glycogen
iii.
Disaccharide
c.
Hyaluronic acid
iv.
Homopolysaccharide
d.
Glyceraldehyde
Choose the correct option:
Q194.
mcq single
+1 / 0
Which one of the following is NOT a technique used for direct transfer of foreign DNA into the protoplast of host cell without using vector?
Q195.
mcq single
+1 / 0
$$ \text { Match the restriction enzyme in Column I with their recognition sequences in Column II. } $$
 Choose the correct option:
Choose the correct option:
 Choose the correct option:
Choose the correct option:
Q196.
mcq single
+1 / 0
Which of the following technique is used for invitro amplification of gene?
Q197.
mcq single
+1 / 0
The transfer of rDNA into a bacterial cell for the purpose of its cloning, is assisted by ________ ions.
Q198.
mcq single
+1 / 0
In tissue culture technique, to maintain aseptic conditions the nutrient medium is autoclaved at constant pressure of _________ $\mathrm{lb} / \mathrm{sq}$ inch for continuous __________ minutes.