MHT CET (PCB) 2025 9th April Morning Shift
MEDICAL 2025 Previous Year
3 hDuration
200Total Marks
400Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 400 across 3 section(s); maximum marks: 200.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+1 / 0
Which of the following is water gas shift reaction?
Q2.
mcq single
+1 / 0
Identify product P in following reaction. Acetamide $+4[\mathrm{H}] \xrightarrow{\mathrm{Na}_2-\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}} \mathrm{P}$
Q3.
mcq single
+1 / 0
What is the number of alkyl groups attached with N -atom of amines formed when 4 n moles of alkyl halide treated with n mole alc. $\mathrm{NH}_3$ at 373 K ?
Q4.
mcq single
+1 / 0
Which from following is a transition element and also included as post actinoid element?
Q5.
mcq single
+1 / 0
Identify inner transition element from following.
Q6.
mcq single
+1 / 0
Which from following is a biodegradable polymer?
Q7.
mcq single
+1 / 0
Identify linear polymer from following.
Q8.
mcq single
+1 / 0
Which of the following compounds has highest lattice energy?
Q9.
mcq single
+1 / 0
Calculate the energy of photon in electromagnetic radiation having wavelength $400 \mathrm{~nm}\left(\mathrm{~h}=6.626 \times 10^{-34} \mathrm{Js} ; \mathrm{c}=3 \times 10^8 \mathrm{~ms}^{-1}\right)$.
Q10.
mcq single
+1 / 0
At 298 K pH of a solution is 13 . What is the number of $\mathrm{H}_3 \mathrm{O}^{+}$ions present in one mL of this solution?
Q11.
mcq single
+1 / 0
Identify the compound from following so that Ostwald's dilution law is NOT applicable to it.
Q12.
mcq single
+1 / 0
The solubility product of $\mathrm{PbI}_2$ is $2.8 \times 10^{-8}$. Calculate its solubility in $\mathrm{mol} \mathrm{dm}^{-3}$.
Q13.
mcq single
+1 / 0
Consider the reaction
$$ 3 \mathrm{I}_{(\mathrm{aq})}^{-}+\mathrm{S}_2 \mathrm{O}_{8(\mathrm{aq})}^{--} \longrightarrow \mathrm{I}_{3(\mathrm{aq})}^{-}+2 \mathrm{SO}_{4(\mathrm{aq})}^{2-} $$
rate of formation of $\mathrm{SO}_4^{2-}$ at a particular time is
$$ 2.2 \times 10^{-2} \mathrm{~mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1} $$
What is the value of $\frac{d}{d t}\left[I_3^{-}\right]$?
Q14.
mcq single
+1 / 0
The rate law for the reaction, $x \mathrm{~A}+y \mathrm{~B} \longrightarrow \mathrm{mP}+\mathrm{nQ}$, is $\mathrm{r}=\mathrm{k}[\mathrm{A}]^{\mathrm{c}} \times[\mathrm{B}]^{\mathrm{d}}$ What is the overall order of reaction?
Q15.
mcq single
+1 / 0
$60 \%$ of a first order reaction is completed in 60 minute. What is the time taken by reactant to decompose half of its original amount?
Q16.
mcq single
+1 / 0
What is the number of electrons released in conversion of $\mathrm{C}_2 \mathrm{O}_4^{2-}$ to $\mathrm{CO}_2$ ?
Q17.
mcq single
+1 / 0
Calculate the number of electrons required to reduce completely $0.1 \mathrm{~mol} \mathrm{Al}^{+3}$ ions from aqueous $\mathrm{Al}_2\left(\mathrm{SO}_4\right)_3$ solution.
Q18.
mcq single
+1 / 0
Which from following is the strongest haloacetic acid?
Q19.
mcq single
+1 / 0
Which of the following is false regarding adsorption?
Q20.
mcq single
+1 / 0
Identify zero-dimensional nanostructure from examples given below.
Q21.
mcq single
+1 / 0
Which among the following is vinylic halide?
Q22.
mcq single
+1 / 0
Which of the following compounds has highest boiling point?
Q23.
mcq single
+1 / 0
24 g carbon react with oxygen to produce 88 g carbon dioxide. Find the amount of oxygen used.
Q24.
mcq single
+1 / 0
Which from following is an example of intensive property?
Q25.
mcq single
+1 / 0
Calculate the work done in following reaction.
$$ \begin{aligned} & \mathrm{C}_2 \mathrm{H}_{4(\mathrm{~g})}+\mathrm{HCl}_{(\mathrm{g})} \longrightarrow \mathrm{C}_2 \mathrm{H}_5 \mathrm{Cl}_{(\mathrm{g})} \text { at } 27^{\circ} \mathrm{C} . \\ & \left(\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right) \end{aligned} $$
Q26.
mcq single
+1 / 0
For a certain reaction, $\Delta \mathrm{H}^{\circ}=219 \mathrm{~kJ}$ and $\Delta \mathrm{S}^{\circ}=-21 \mathrm{JK}^{-1}$. Find the value of $\Delta \mathrm{G}^{\circ}$.
Q27.
mcq single
+1 / 0
What is the molecular formula of following compound?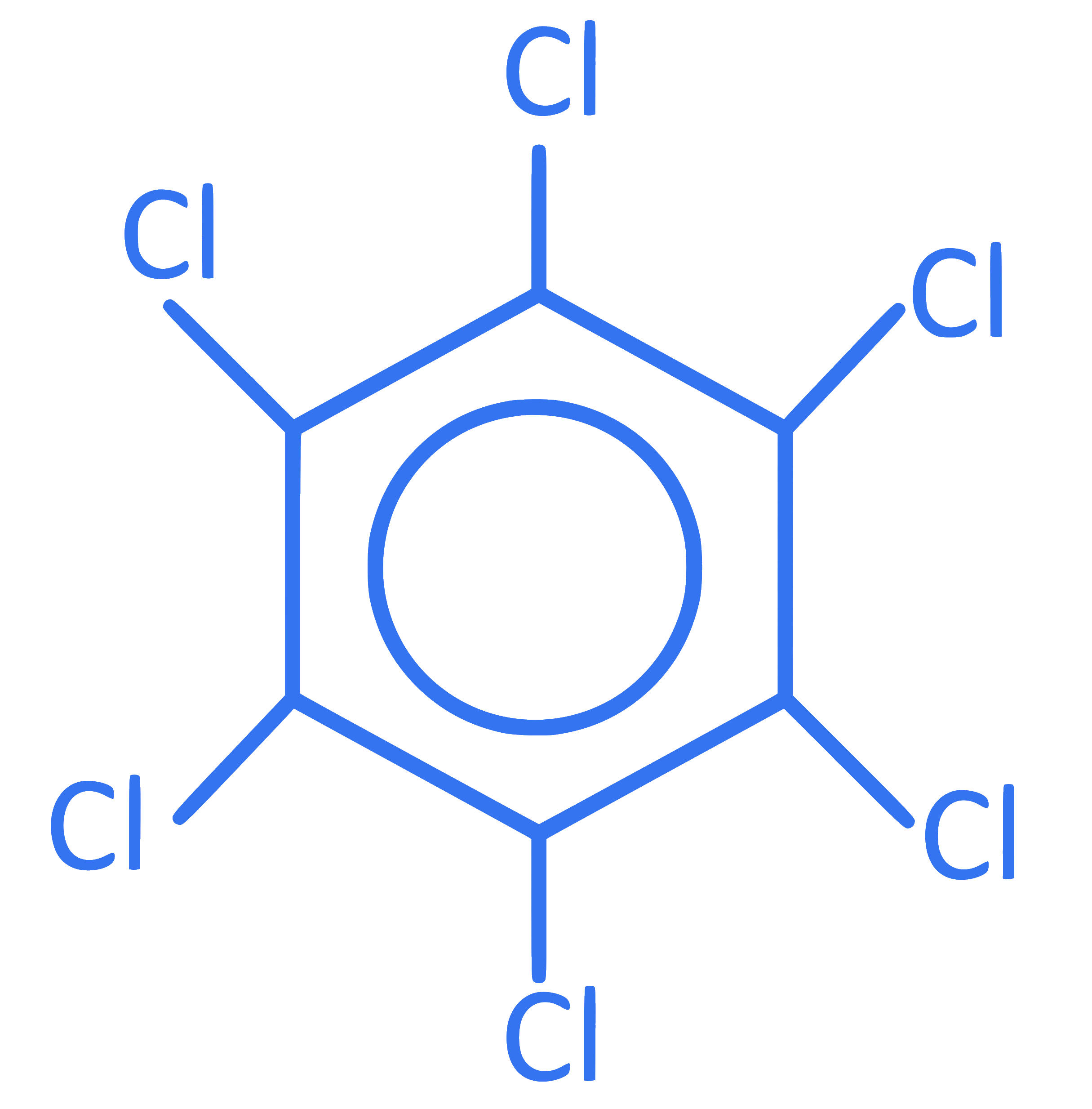

Q28.
mcq single
+1 / 0
What is the number of moles of ' C ' atoms and ' H ' atoms respectively present in n mole molecule represented as follows?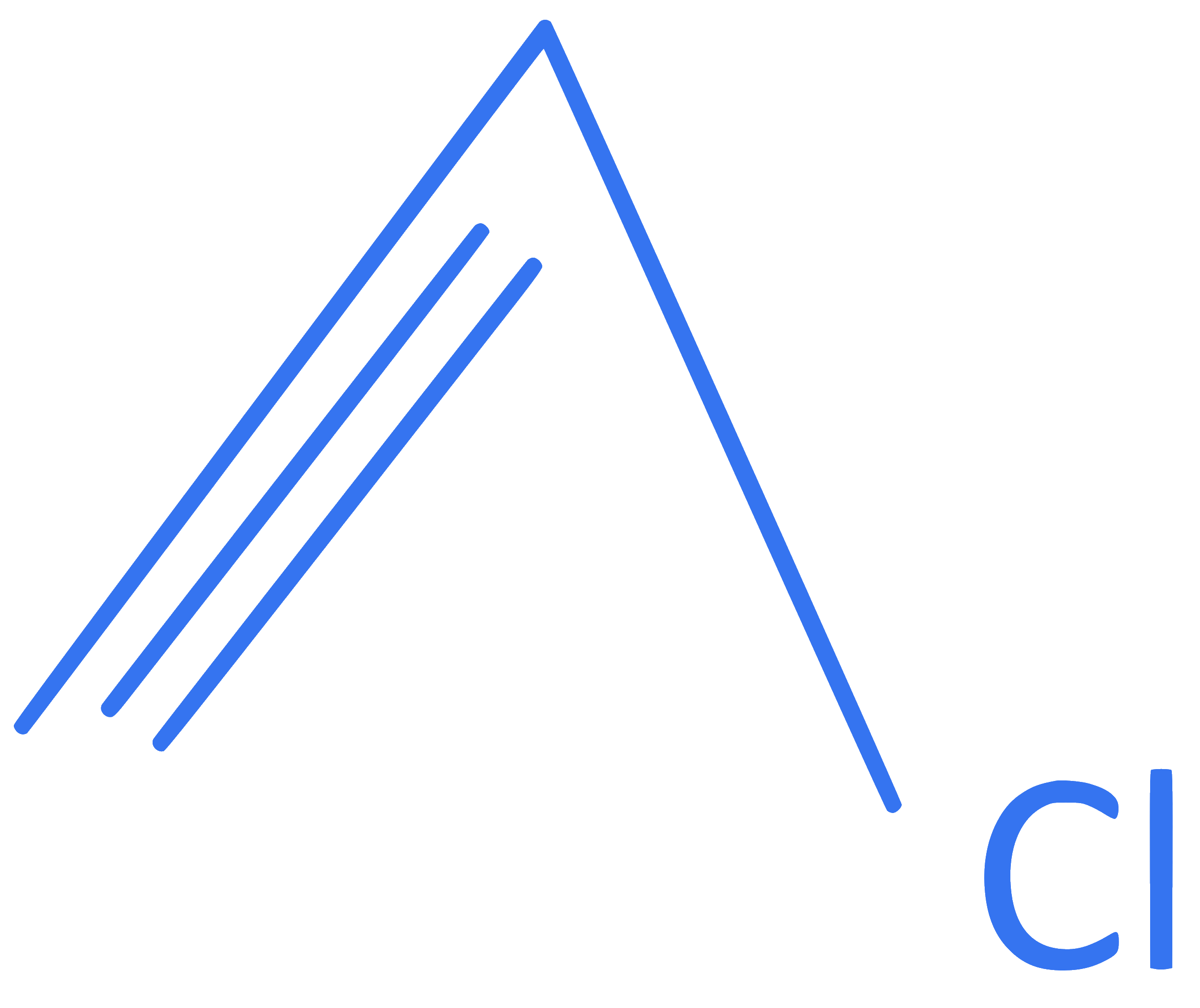

Q29.
mcq single
+1 / 0
The osmotic pressure of equimolar concentration of $\mathrm{CaCl}_2$ and urea at same temperature are 0.6 atm and 0.25 atm respectively. Calculate van't Hoff factor for $\mathrm{CaCl}_2$.
Q30.
mcq single
+1 / 0
What type of solution is the benzoic acid in benzene?
Q31.
mcq single
+1 / 0
Calculate the molal elevation constant of water if boiling point of 0.7 m solution of non volatile solute is 373.5 K .
[Boiling point of water $=373.15 \mathrm{~K}$ ]
Q32.
mcq single
+1 / 0
Which of the following hydride of chalcogens has lowest bond angle and highest bond length?
Q33.
mcq single
+1 / 0
Which of the following is the most abundant chalcogens?
Q34.
mcq single
+1 / 0
Identify the ligands present Pentaamminecarbonatocobalt(III)chloride.
Q35.
mcq single
+1 / 0
What is EAN of Mn in $\mathrm{Mn}(\mathrm{CO})_5$ ?
Q36.
mcq single
+1 / 0
Which of the following gas has high percentage by volume in air?
Q37.
mcq single
+1 / 0
Which of the following alkanes does NOT exhibit structural isomerism?
Q38.
mcq single
+1 / 0
What is IUPAC name of glyoxal?
Q39.
mcq single
+1 / 0
Which of the following is secondary benzylic alcohol?
Q40.
mcq single
+1 / 0
What is the name of t-butyl alcohol according to carbinol system?
Q41.
mcq single
+1 / 0
Which from following amino acids is synthesized in our body?
Q42.
mcq single
+1 / 0
What is the maximum number of ester linkages in a triglyceride?
Q43.
mcq single
+1 / 0
Identify the quantity of carbohydrate units formed after hydrolysis of n mole lactose.
Q44.
mcq single
+1 / 0
Which of the following statements is NOT true about vacancy defect?
Q45.
mcq single
+1 / 0
In ionic solid, anions are arranged in ccp array and cations occupy all the octahedral voids. What is the formula of ionic compound? [consider $\mathrm{A}=$ cation; $\mathrm{B}=$ anion]
Q46.
mcq single
+1 / 0
Calculate the volume occupied by a particle in simple cubic unit cell if the total volume of unit cell is $4.0 \times 10^{-23} \mathrm{~cm}^3$.
Q47.
mcq single
+1 / 0
The conductivity of 0.1 M KCl solution at $23^{\circ} \mathrm{C}$ is $0.012 \Omega^{-1} \mathrm{~cm}^{-1}$.
Resistance of a cell containing the solution at same temperature was found to be $50 \Omega$. Calculate the cell constant.
Q48.
mcq single
+1 / 0
Identify the electrolyte from following so that $\wedge_0$ is not obtained from the graphical variation of $\wedge$ against $\sqrt{\mathrm{C}}$ ?
Q49.
mcq single
+1 / 0
Which of the following on reaction with Grignards reagent followed by hydrolysis forms secondary alcohol?
Q50.
mcq single
+1 / 0
Identify an aldehyde used in margarine and food for its buttery odour.
Q51.
mcq single
+1 / 0
Which of the following is water gas shift reaction?
Q52.
mcq single
+1 / 0
Identify product P in following reaction. Acetamide $+4[\mathrm{H}] \xrightarrow{\mathrm{Na}_2-\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}} \mathrm{P}$
Q53.
mcq single
+1 / 0
What is the number of alkyl groups attached with N -atom of amines formed when 4 n moles of alkyl halide treated with n mole alc. $\mathrm{NH}_3$ at 373 K ?
Q54.
mcq single
+1 / 0
Which from following is a transition element and also included as post actinoid element?
Q55.
mcq single
+1 / 0
Identify inner transition element from following.
Q56.
mcq single
+1 / 0
Which from following is a biodegradable polymer?
Q57.
mcq single
+1 / 0
Identify linear polymer from following.
Q58.
mcq single
+1 / 0
Which of the following compounds has highest lattice energy?
Q59.
mcq single
+1 / 0
Calculate the energy of photon in electromagnetic radiation having wavelength $400 \mathrm{~nm}\left(\mathrm{~h}=6.626 \times 10^{-34} \mathrm{Js} ; \mathrm{c}=3 \times 10^8 \mathrm{~ms}^{-1}\right)$.
Q60.
mcq single
+1 / 0
At 298 K pH of a solution is 13 . What is the number of $\mathrm{H}_3 \mathrm{O}^{+}$ions present in one mL of this solution?
Q61.
mcq single
+1 / 0
Identify the compound from following so that Ostwald's dilution law is NOT applicable to it.
Q62.
mcq single
+1 / 0
The solubility product of $\mathrm{PbI}_2$ is $2.8 \times 10^{-8}$. Calculate its solubility in $\mathrm{mol} \mathrm{dm}^{-3}$.
Q63.
mcq single
+1 / 0
Consider the reaction
$$ 3 \mathrm{I}_{(\mathrm{aq})}^{-}+\mathrm{S}_2 \mathrm{O}_{8(\mathrm{aq})}^{--} \longrightarrow \mathrm{I}_{3(\mathrm{aq})}^{-}+2 \mathrm{SO}_{4(\mathrm{aq})}^{2-} $$
rate of formation of $\mathrm{SO}_4^{2-}$ at a particular time is
$$ 2.2 \times 10^{-2} \mathrm{~mol} \mathrm{dm}^{-3} \mathrm{~s}^{-1} $$
What is the value of $\frac{d}{d t}\left[I_3^{-}\right]$?
Q64.
mcq single
+1 / 0
The rate law for the reaction, $x \mathrm{~A}+y \mathrm{~B} \longrightarrow \mathrm{mP}+\mathrm{nQ}$, is $\mathrm{r}=\mathrm{k}[\mathrm{A}]^{\mathrm{c}} \times[\mathrm{B}]^{\mathrm{d}}$ What is the overall order of reaction?
Q65.
mcq single
+1 / 0
$60 \%$ of a first order reaction is completed in 60 minute. What is the time taken by reactant to decompose half of its original amount?
Q66.
mcq single
+1 / 0
What is the number of electrons released in conversion of $\mathrm{C}_2 \mathrm{O}_4^{2-}$ to $\mathrm{CO}_2$ ?
Q67.
mcq single
+1 / 0
Calculate the number of electrons required to reduce completely $0.1 \mathrm{~mol} \mathrm{Al}^{+3}$ ions from aqueous $\mathrm{Al}_2\left(\mathrm{SO}_4\right)_3$ solution.
Q68.
mcq single
+1 / 0
Which from following is the strongest haloacetic acid?
Q69.
mcq single
+1 / 0
Which of the following is false regarding adsorption?
Q70.
mcq single
+1 / 0
Identify zero-dimensional nanostructure from examples given below.
Q71.
mcq single
+1 / 0
Which among the following is vinylic halide?
Q72.
mcq single
+1 / 0
Which of the following compounds has highest boiling point?
Q73.
mcq single
+1 / 0
24 g carbon react with oxygen to produce 88 g carbon dioxide. Find the amount of oxygen used.
Q74.
mcq single
+1 / 0
Which from following is an example of intensive property?
Q75.
mcq single
+1 / 0
Calculate the work done in following reaction.
$$ \begin{aligned} & \mathrm{C}_2 \mathrm{H}_{4(\mathrm{~g})}+\mathrm{HCl}_{(\mathrm{g})} \longrightarrow \mathrm{C}_2 \mathrm{H}_5 \mathrm{Cl}_{(\mathrm{g})} \text { at } 27^{\circ} \mathrm{C} . \\ & \left(\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right) \end{aligned} $$
Q76.
mcq single
+1 / 0
For a certain reaction, $\Delta \mathrm{H}^{\circ}=219 \mathrm{~kJ}$ and $\Delta \mathrm{S}^{\circ}=-21 \mathrm{JK}^{-1}$. Find the value of $\Delta \mathrm{G}^{\circ}$.
Q77.
mcq single
+1 / 0
What is the molecular formula of following compound?

Q78.
mcq single
+1 / 0
What is the number of moles of ' C ' atoms and ' H ' atoms respectively present in n mole molecule represented as follows?

Q79.
mcq single
+1 / 0
The osmotic pressure of equimolar concentration of $\mathrm{CaCl}_2$ and urea at same temperature are 0.6 atm and 0.25 atm respectively. Calculate van't Hoff factor for $\mathrm{CaCl}_2$.
Q80.
mcq single
+1 / 0
What type of solution is the benzoic acid in benzene?
Q81.
mcq single
+1 / 0
Calculate the molal elevation constant of water if boiling point of 0.7 m solution of non volatile solute is 373.5 K .
[Boiling point of water $=373.15 \mathrm{~K}$ ]
Q82.
mcq single
+1 / 0
Which of the following hydride of chalcogens has lowest bond angle and highest bond length?
Q83.
mcq single
+1 / 0
Which of the following is the most abundant chalcogens?
Q84.
mcq single
+1 / 0
Identify the ligands present Pentaamminecarbonatocobalt(III)chloride.
Q85.
mcq single
+1 / 0
What is EAN of Mn in $\mathrm{Mn}(\mathrm{CO})_5$ ?
Q86.
mcq single
+1 / 0
Which of the following gas has high percentage by volume in air?
Q87.
mcq single
+1 / 0
Which of the following alkanes does NOT exhibit structural isomerism?
Q88.
mcq single
+1 / 0
What is IUPAC name of glyoxal?
Q89.
mcq single
+1 / 0
Which of the following is secondary benzylic alcohol?
Q90.
mcq single
+1 / 0
What is the name of t-butyl alcohol according to carbinol system?
Q91.
mcq single
+1 / 0
Which from following amino acids is synthesized in our body?
Q92.
mcq single
+1 / 0
What is the maximum number of ester linkages in a triglyceride?
Q93.
mcq single
+1 / 0
Identify the quantity of carbohydrate units formed after hydrolysis of n mole lactose.
Q94.
mcq single
+1 / 0
Which of the following statements is NOT true about vacancy defect?
Q95.
mcq single
+1 / 0
In ionic solid, anions are arranged in ccp array and cations occupy all the octahedral voids. What is the formula of ionic compound? [consider $\mathrm{A}=$ cation; $\mathrm{B}=$ anion]
Q96.
mcq single
+1 / 0
Calculate the volume occupied by a particle in simple cubic unit cell if the total volume of unit cell is $4.0 \times 10^{-23} \mathrm{~cm}^3$.
Q97.
mcq single
+1 / 0
The conductivity of 0.1 M KCl solution at $23^{\circ} \mathrm{C}$ is $0.012 \Omega^{-1} \mathrm{~cm}^{-1}$.
Resistance of a cell containing the solution at same temperature was found to be $50 \Omega$. Calculate the cell constant.
Q98.
mcq single
+1 / 0
Identify the electrolyte from following so that $\wedge_0$ is not obtained from the graphical variation of $\wedge$ against $\sqrt{\mathrm{C}}$ ?
Q99.
mcq single
+1 / 0
Which of the following on reaction with Grignards reagent followed by hydrolysis forms secondary alcohol?
Q100.
mcq single
+1 / 0
Identify an aldehyde used in margarine and food for its buttery odour.
Physics
Physics
Q1.
mcq single
+1 / 0
If $|\vec{A} \times \vec{B}|=\sqrt{3}(\vec{A} \cdot \vec{B})$ then the value of $|\vec{A}+\vec{B}|$ is $\left(\tan 60^{\circ}=\sqrt{3}, \cos 60^{\circ}=0.5\right)$
Q2.
mcq single
+1 / 0
' n ' polarising sheets are arranged such that each makes an angle $45^{\circ}$ with the proceeding sheet. An unpolarised light of intensity I is incident into this arrangement. The output intensity is I/ 64 . The value of $n$ will be $\left(\cos 45^{\circ}=1 / \sqrt{2}\right)$
Q3.
mcq single
+1 / 0
In an interference experiment, the phase difference between the waves reaching a first dark point is
Q4.
mcq single
+1 / 0
In Young's double slit experiment, the intensities at two points, for the path difference $\frac{\lambda}{4}$ and $\frac{\lambda}{3}$ are $\mathrm{I}_1$ and $\mathrm{I}_2$ respectively. If $\mathrm{I}_0$ denotes the intensity produced by each one of the individual slits then the ratio $\left(\mathrm{I}_1+\mathrm{I}_2\right): \mathrm{I}_0$ is $\left(\cos 45^{\circ}=1 / \sqrt{2}\right)\left(\cos 60^{\circ}=0.5\right)$
Q5.
mcq single
+1 / 0
The average velocity of a particle performing S.H.M. in one complete vibration is ( $\mathrm{A}=$ amplitude of S.H.M., $\omega=$ angular velocity)
Q6.
mcq single
+1 / 0
A mass ' $M$ ' is suspended from a spring of negligible mass. The spring is pulled a little and then released so that the mass executes S.H.M. of periodic time ' $T$ '. If the mass is increased by ' $m$ ', the time period becomes $4 T / 3$, then the ratio of $\frac{\mathrm{M}}{\mathrm{m}}$ is
Q7.
mcq single
+1 / 0
A particle vibrating simple harmonically has an acceleration of $16 \mathrm{~cm} / \mathrm{s}^2$, when it is at a distance of 4 cm from the mean position. Its periodic time is
Q8.
mcq single
+1 / 0
Four solid spheres each of mass ' $m$ ' and radius ' $r$ ' are arranged as shown in the figure. The moment of inertia of the system about the given axis of rotation $\mathrm{AA}^1$ is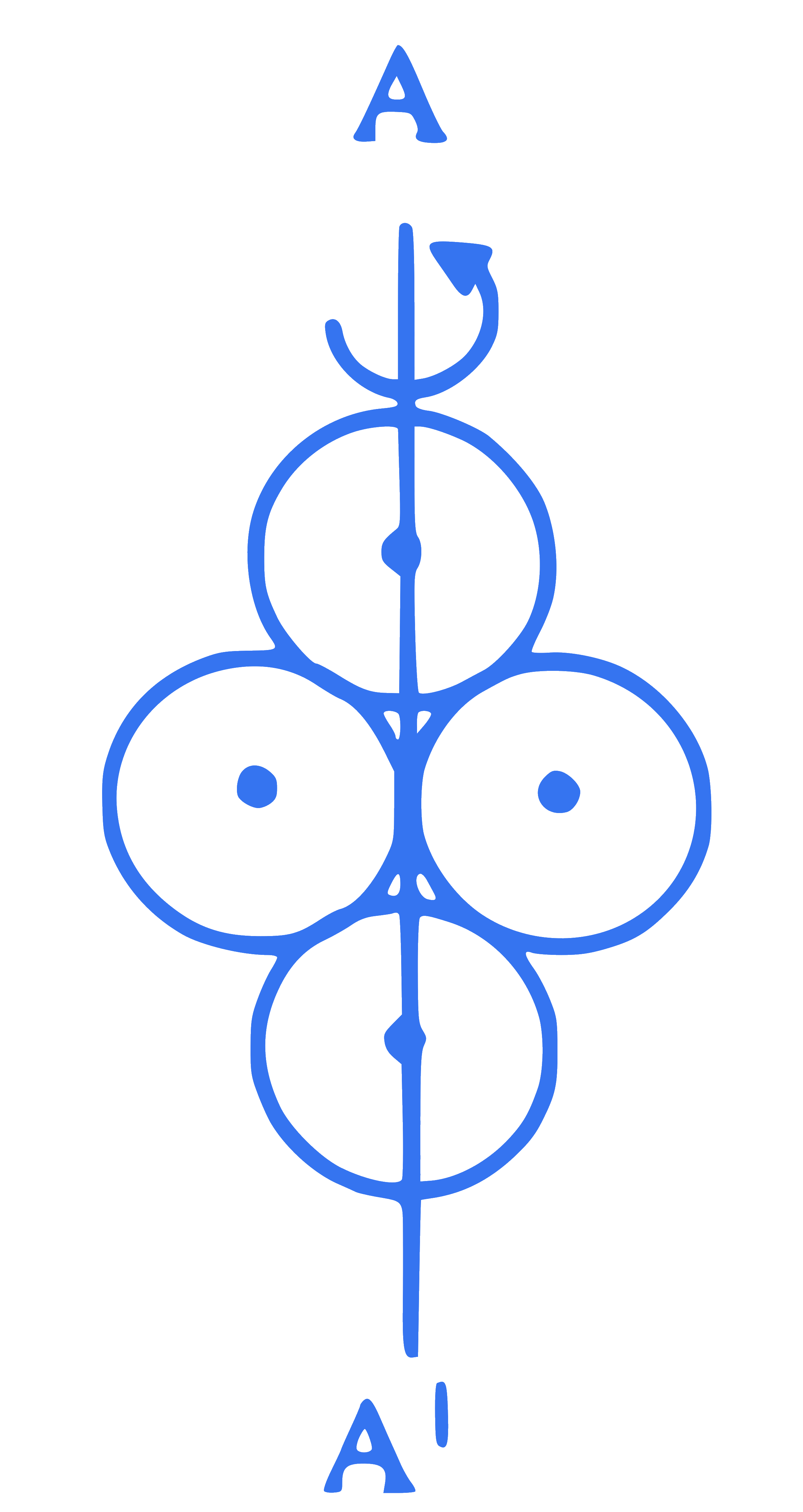

Q9.
mcq single
+1 / 0
Four thin rods of same mass M and same length L form a square as shown in figure. Moment of inertia of this system about an axis passing through point O and perpendicular to its plane is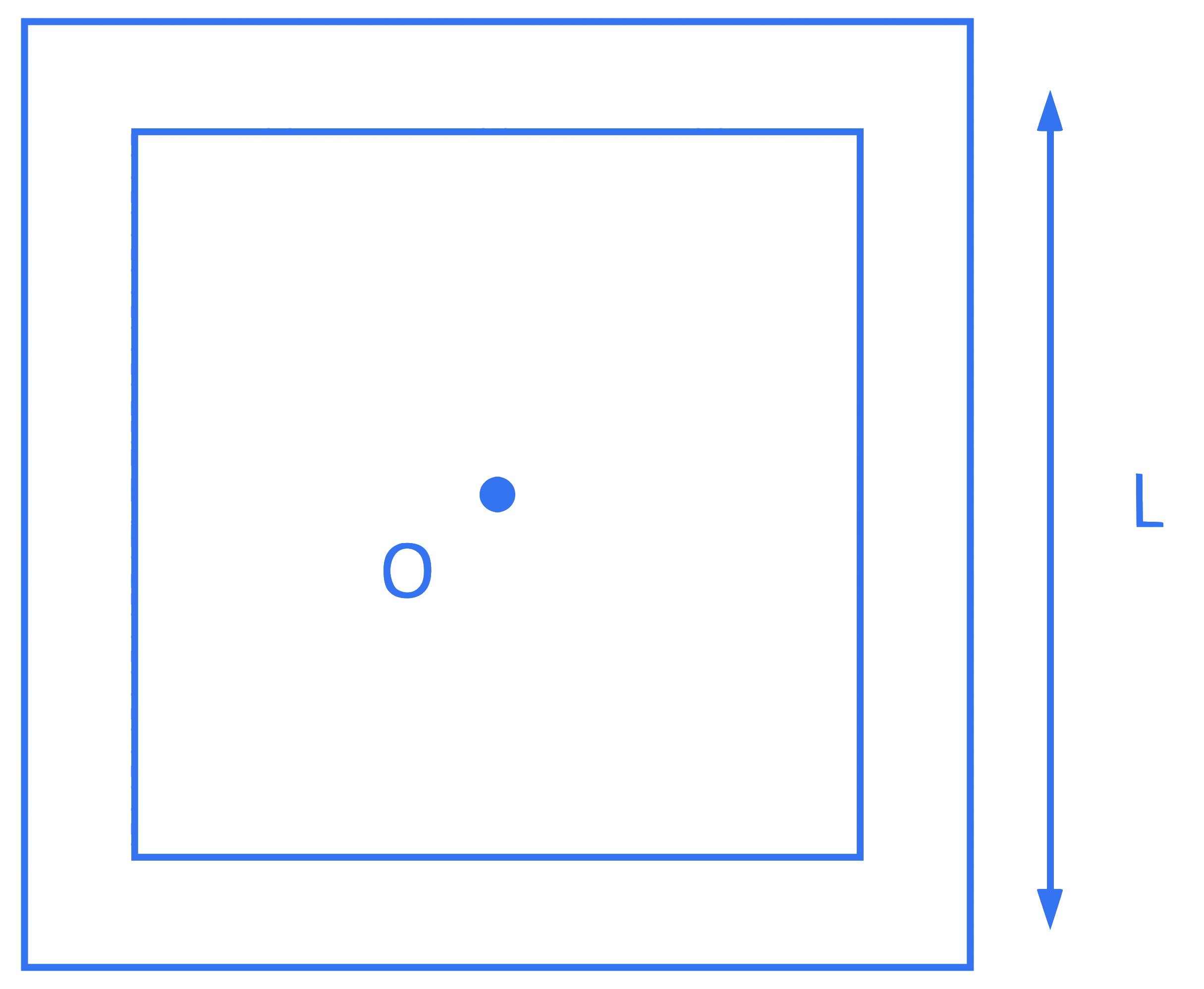

Q10.
mcq single
+1 / 0
Two inductors of 88 mH each are joined in parallel. The current passing through this combination is 4 A . The energy stored in this combination of inductors in joule is
Q11.
mcq single
+1 / 0
If current ' i ' is passing through the solenoid of diameter ' $d$ ' having number of turns per unit length ' $n$ ', then the inductance per unit length near the middle of a long solenoid is directly proportional to
Q12.
mcq single
+1 / 0
A bicycle wheel of diameter ' $D$ ' has ' $N$ ' number of spokes. Wheel is rotating at the rate of ' $x$ ' revolutions per minute, perpendicular to the horizontal component of earth's magnetic field ' $\mathrm{B}_{\mathrm{H}}$ '. The e.m.f. induced between the rim and the centre of the wheel will be
Q13.
mcq single
+1 / 0
A string of length $L$ is fixed at one end and carries a mass m at the other end. The string makes $\frac{3}{\pi}$ revolutions per second around the vertical axis through the fixed end. The tension in the string is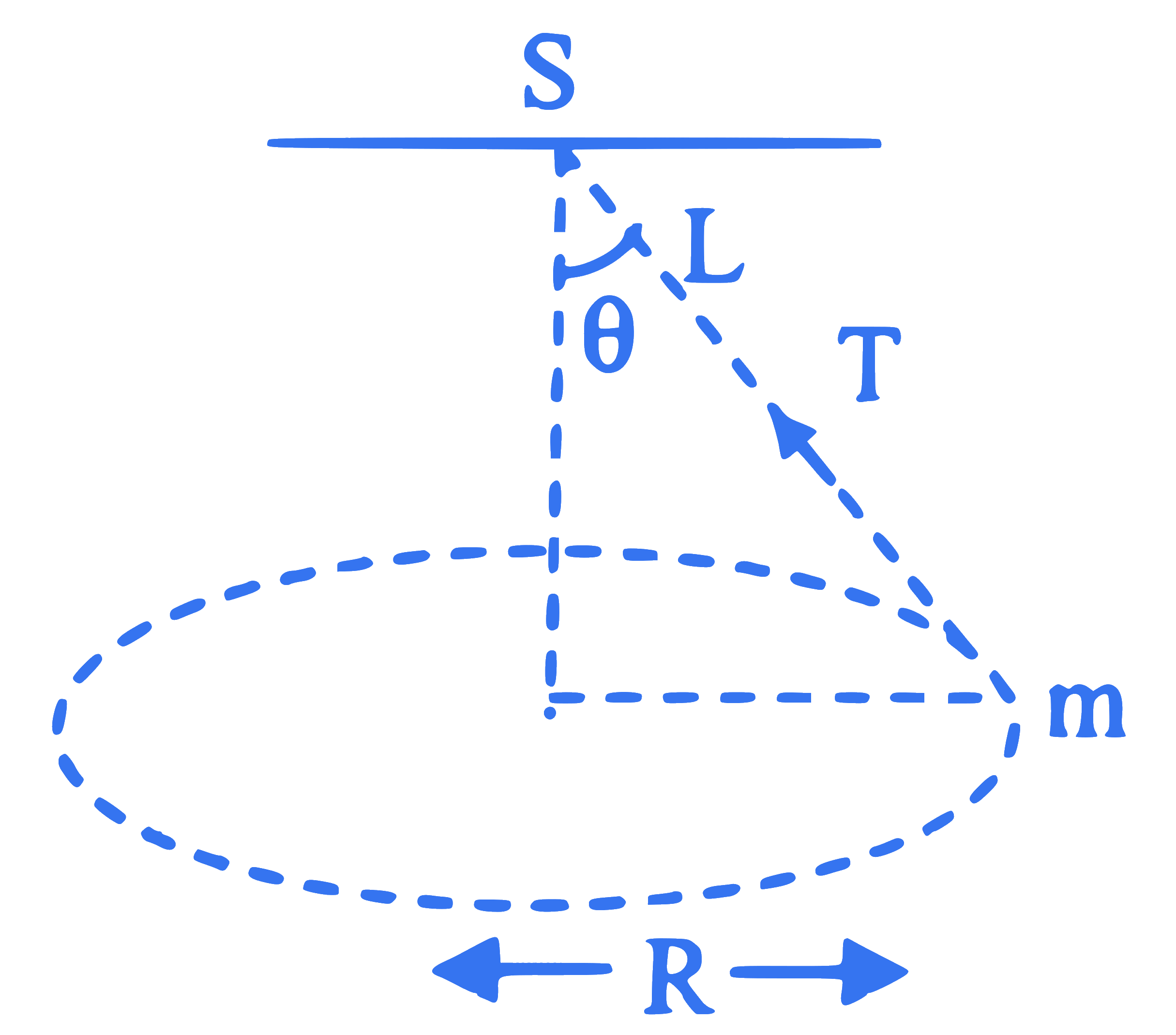

Q14.
mcq single
+1 / 0
Two spherical black bodies of radius $\mathrm{R}_1$ and $\mathrm{R}_2$ with surface temperatures $\mathrm{T}_1$ and $\mathrm{T}_2$ respectively. If $T_1=2 T_2$ and they radiate the same power then relation between $R_1$ and $R_2$ is
Q15.
mcq single
+1 / 0
In an adiabatic change, the pressure and temperature of a diatomic gas are related as $\mathrm{P} \propto \mathrm{T}^{\mathrm{c}}$, where c is a constant. The value of c is (consider gas molecules are rigid)
Q16.
mcq single
+1 / 0
A system is compressed adiabatically, then its temperature
Q17.
mcq single
+1 / 0
When the r.m.s. velocity of a gas is denoted by ' V ' and temperature of the gas by T , which of the following relations is TRUE?
Q18.
mcq single
+1 / 0
A gas at $800^{\circ} \mathrm{C}$ is cooled until both its pressure and volume are halved. The final temperature of the gas will be
Q19.
mcq single
+1 / 0
The average kinetic energy of a molecule of the gas is
Q20.
mcq single
+1 / 0
A photon and an electron have equal energy ' $E$ '. The ratio of wavelength of photon to wavelength of electron is proportional to
Q21.
mcq single
+1 / 0
The energy of a photon is equal to the kinetic energy of proton. If $\lambda_1$ is the de-Broglie wavelength of a proton, $\lambda_2$ is the wavelength associated with the photon and if E is the energy of photon then $\lambda_2: \lambda_1$ is
Q22.
mcq single
+1 / 0
The charges are arranged at the four corners of square $A B C D$ of side ' $d$ ' as shown in figure. The work required to put this arrangement together is given by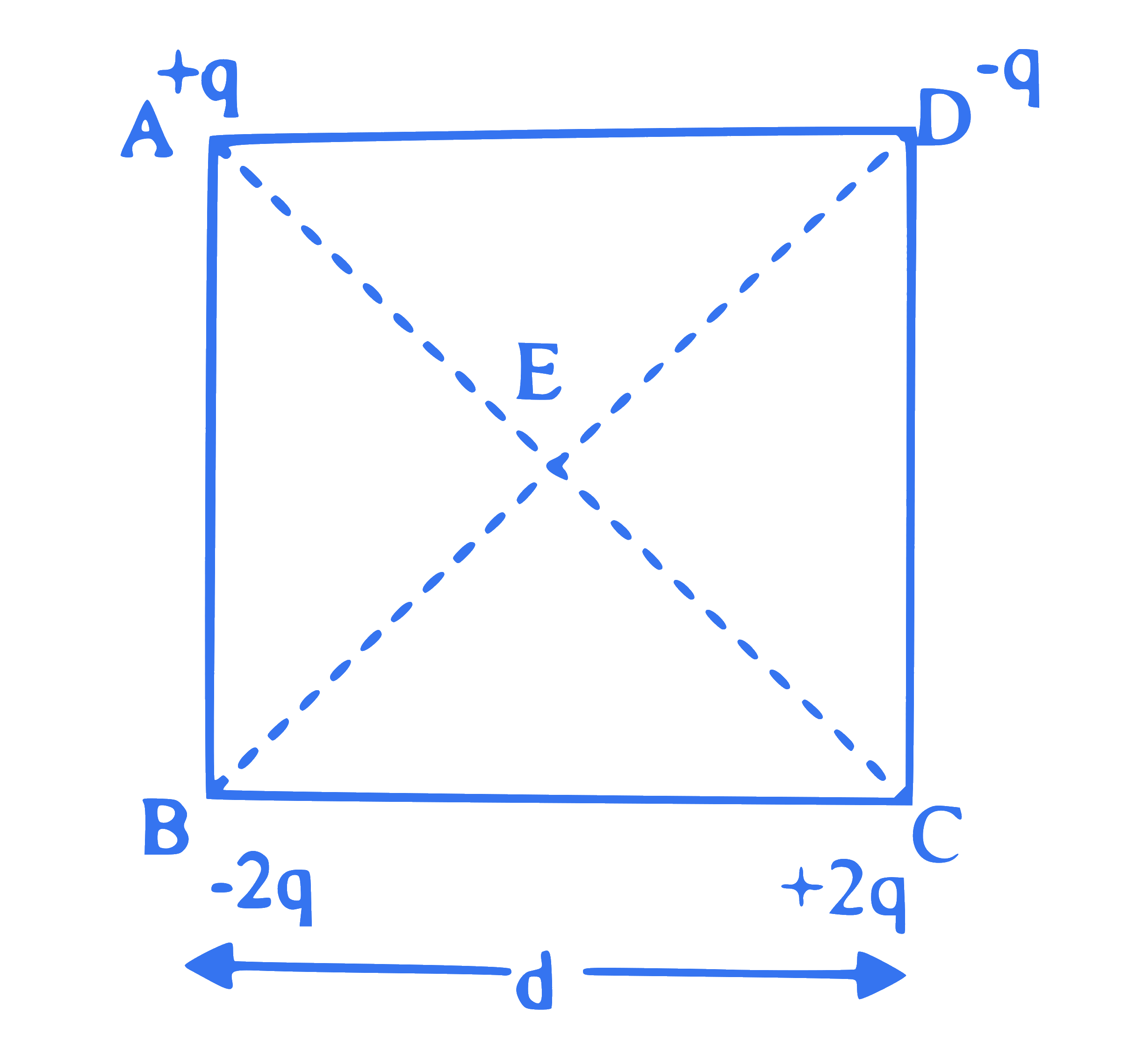

Q23.
mcq single
+1 / 0
A simple pendulum has mass 2 gram and charge $2 \mu \mathrm{C}$. In a uniform horizontal electric field of intensity $1000 \mathrm{~V} / \mathrm{m}$, pendulum is at rest. At equilibrium, the angle made by the pendulum with the vertical is ( $\mathrm{g}=$ acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2$ )
Q24.
mcq single
+1 / 0
The value of acceleration due to gravity (g) becomes $\left(\frac{g}{3}\right)$ at height ' $h$ ' above the earth's surface. If ' R ' is the radius of earth, the height h will be equal to
Q25.
mcq single
+1 / 0
When three capacitors of capacitances ' C ', ' 2 C '? and ' 3 C ' are connected in series. The potential difference applied across the combination is ' $V$ '. The energy stored in the circuit is
Q26.
mcq single
+1 / 0
A lens of power +5 D is put in contact with a lens of power -3 D . The combination will behave like a
Q27.
mcq single
+1 / 0
In a projectile motion shown in figure, which one of the following statements is NOT CORRECT?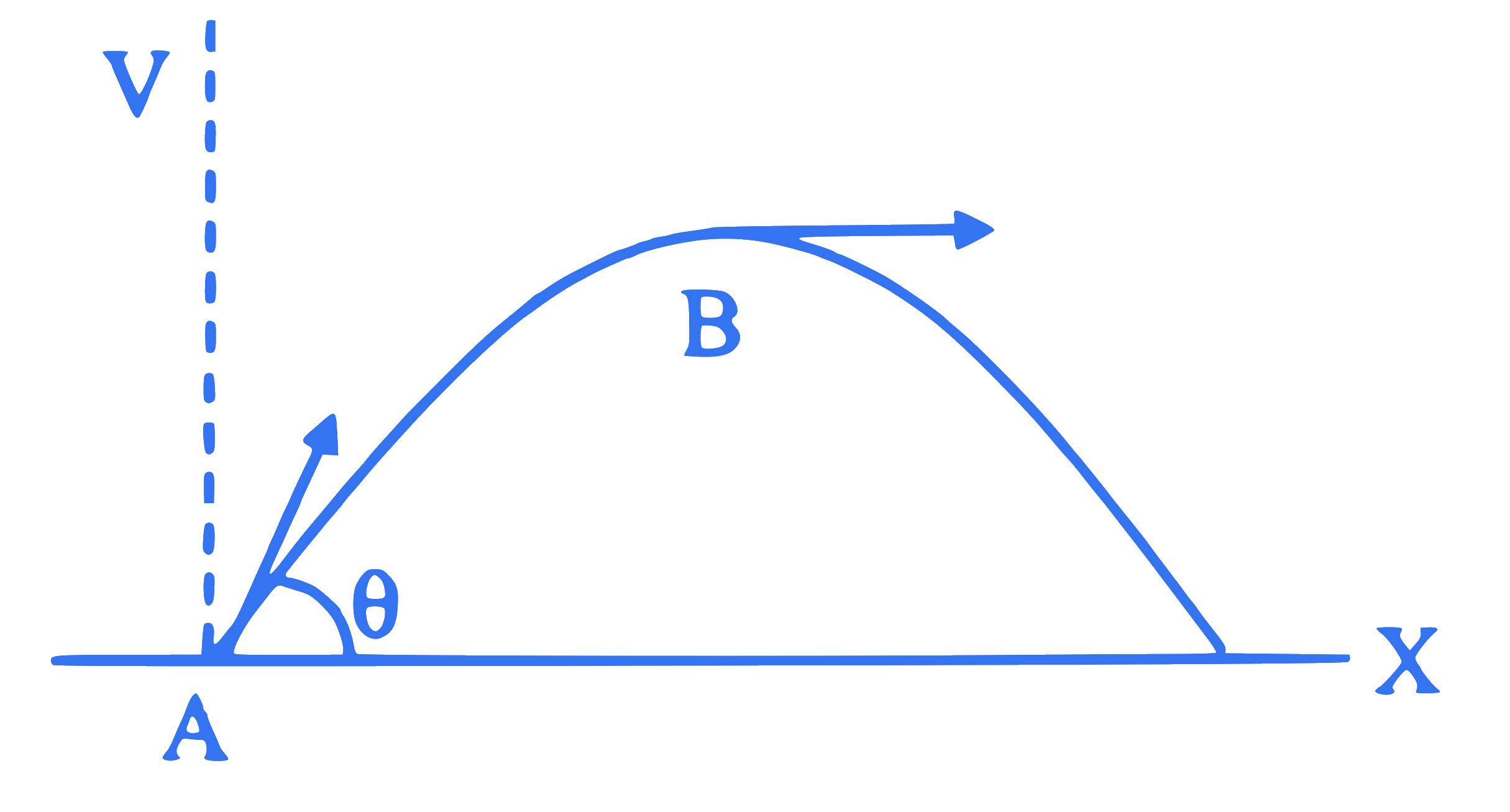

Q28.
mcq single
+1 / 0
The magnetic field at the centre of a current carrying circular coil of area ' $A$ ' is ' $B$ '. The magnetic moment of the coil is $x$ times $\left(2 B / \mu_0\right)$. The value of $x$ is ( $\mu_0=$ permeability of free space)
Q29.
mcq single
+1 / 0
A straight wire of mass ' M ' and length 2 m is placed in a magnetic field of 2 T which is acting perpendicular to the length of the wire. When a current of 1 A flows through the wire, the wire experiences an upthrust and leviates in a magnetic field. The mass ' $M$ ' of the wire is (acceleration due to gravity $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q30.
mcq single
+1 / 0
A spherical liquid drop of radius R is divided into 64 equal droplets. If surface tension is $T$ then the work done in this process will be
Q31.
mcq single
+1 / 0
A glass capillary of radius 0.35 mm is inclined at $60^{\circ}$ with the vertical in water. The length of water column in the capillary tube is (surface tension of water $=7 \times 10^{-2} \mathrm{Nm}^{-1}$, acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2, \cos 0^{\circ}=1$, $\cos 60^{\circ}=0.5$, density of water $=1 \mathrm{gram} / \mathrm{cm}^3$ )
Q32.
mcq single
+1 / 0
If the shape of the liquid surface is curved, then the
Q33.
mcq single
+1 / 0
The series limit for the frequency of Balmer series is ' $\mathrm{v}_{\mathrm{B}}$ ', then the series limit frequency of Paschen series ' $v_p$ ' is
Q34.
mcq single
+1 / 0
In the spectrum of hydrogen atom the ratio of the longest wavelength in the Balmer series to the longest wavelength in the Lyman series is
Q35.
mcq single
+1 / 0
A passenger is sitting in a train which is moving fast. The engine blows a whistle of frequency ' n '. If the apparent frequency of sound heard by the passenger is $n^1$ then
Q36.
mcq single
+1 / 0
A wave is travelling in the negative x direction having displacement 3 cm along Y direction, wavelength $2 \pi \mathrm{~m}$ and frequency $\left(\frac{1}{2 \pi}\right) \mathrm{Hz}$ is represented as ( $t=$ time)
Q37.
mcq single
+1 / 0
Two points on a travelling wave having frequency 500 Hz and velocity $300 \mathrm{~m} / \mathrm{s}$ are $30^{\circ}$ out of phase, then the minimum distance between the two points is
Q38.
mcq single
+1 / 0
A simple harmonic progressive wave is given by $y=A \sin 2 \pi\left(n t-\frac{x}{\lambda}\right)$. If the wave velocity is equal to $=\frac{1}{3}$ (maximum particle velocity) then the wavelength $\lambda$ is given by
Q39.
mcq single
+1 / 0
Force is applied to a body of mass 2 kg at rest on a frictionless horizontal surface as shown in the force against time $(\mathrm{F}-\mathrm{t})$ graph. The speed of the body after 1 s is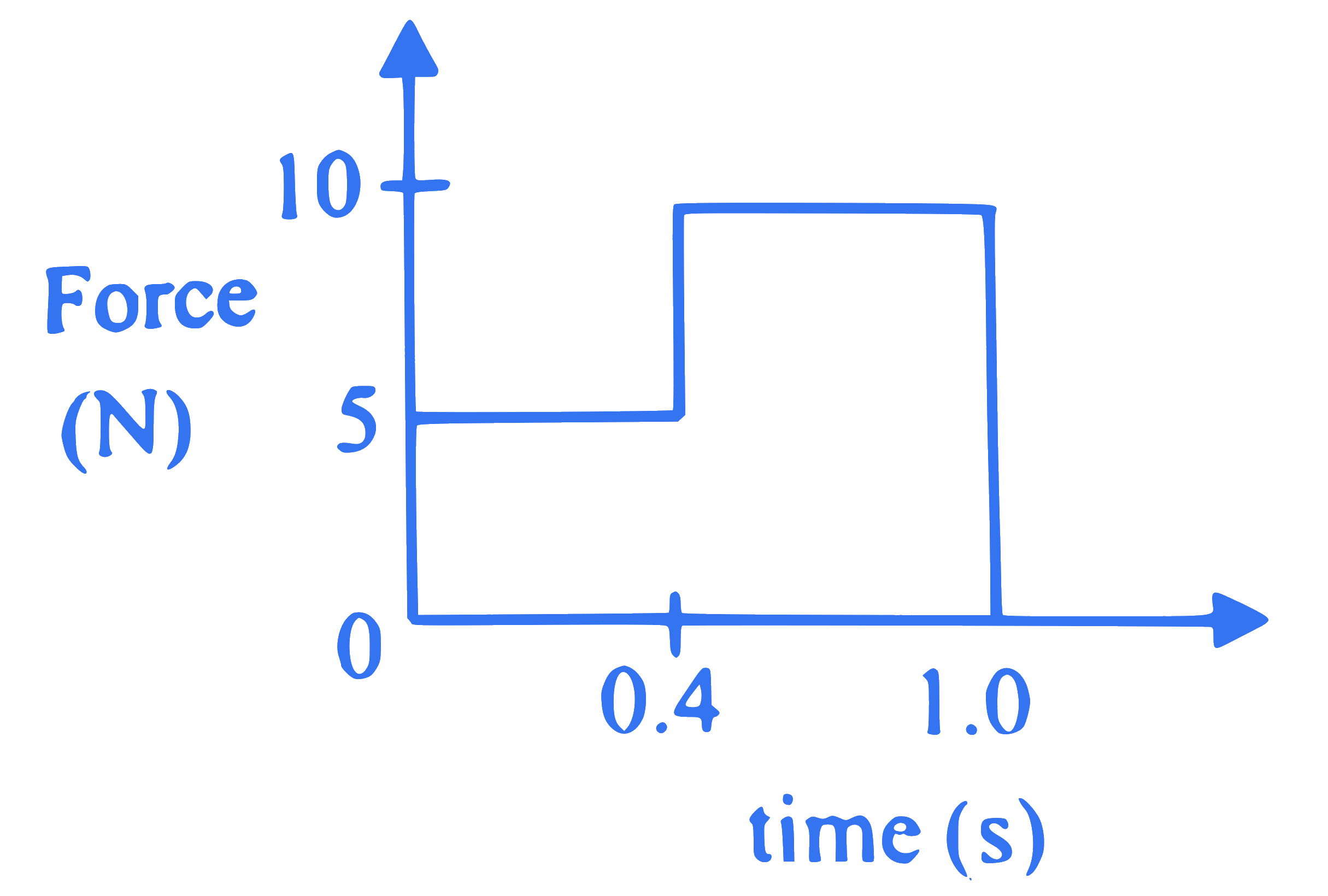

Q40.
mcq single
+1 / 0
A solenoid has core of a material with relative permeability 301 and its winding carries a current of 2 A . The number of turns of the solenoid is 600 per metre. The magnetization of the material is
Q41.
mcq single
+1 / 0
The range of voltmeter of resistance $300 \Omega$ is 5 V . The resistance required to be connected to convert it into an ammeter of range 5 A is nearly
Q42.
mcq single
+1 / 0
In a given circuit, AB is a uniform potentiometer wire. If the value of resistance ' $R$ ' is slightly increased, the balance point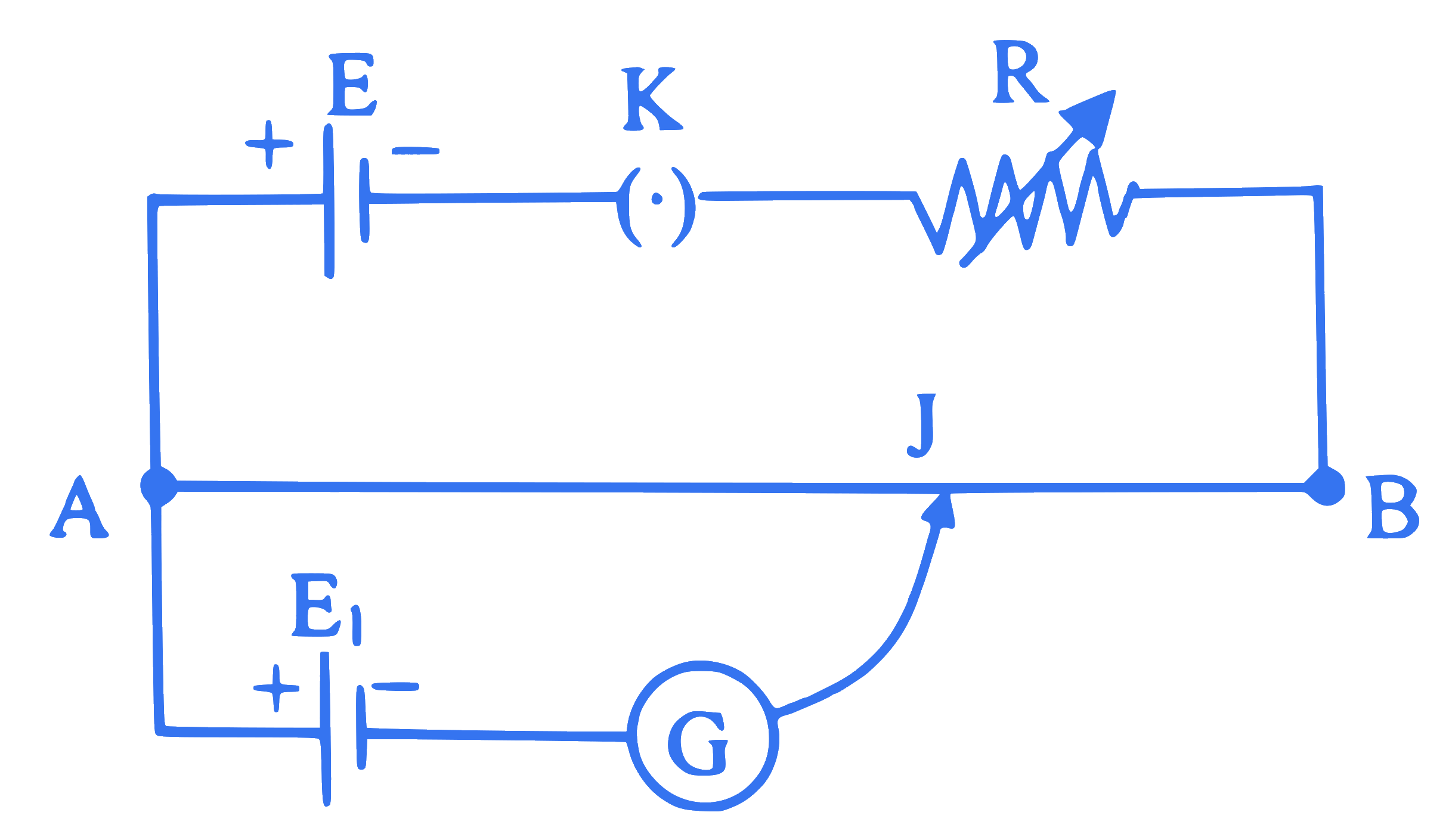

Q43.
mcq single
+1 / 0
A rigid body weighs 25 gram and has a measured volume of $5 \mathrm{~cm}^3$. The possible error in the measurement of mass and volume are 0.01 gram and $0.1 \mathrm{~cm}^3$. The maximum error in the density will be
Q44.
mcq single
+1 / 0
In common emitter configuration, $\beta, \mathrm{R}_{\mathrm{L}}$ and r are the a.c. current gain, load resistance and the input resistance of a transistor respectively. The voltage and power gain respectively are
Q45.
mcq single
+1 / 0
When forward bias is applied to a p-n junction, then the potential barrier and the width of the depletion region respectively.
Q46.
mcq single
+1 / 0
The order of (a) transformer (b) diode rectifier (c) filter (d) voltage regulator in a block diagram of simple rectifier circuit is as
(a) b, d, c, a
(b) a, c, d, b
(c) a, b, c, d
(d) d, c, a, b
Q47.
mcq single
+1 / 0
An alternating voltage $\mathrm{V}=100 \sqrt{2} \sin (50 \mathrm{t})$ is connected to $2 \mu \mathrm{~F}$ capacitor through a.c. ammeter. The reading of the ammeter is
Q48.
mcq single
+1 / 0
A capacitor of capacitance ' C ' is connected across a.c. source of voltage V as $\mathrm{V}=\mathrm{V}_0 \sin \omega \mathrm{t}$. The displacement current between the plates of the capacitor would be
Q49.
mcq single
+1 / 0
An inductor of 0.5 mH , capacitor of $20 \mu \mathrm{~F}$ and resistance of $20 \Omega$ are connected in series with a 220 V a.c. source. If the current is in phase with e.m.f., the maximum current in the circuit is $\sqrt{x} A$. The value of $x$ is
Q50.
mcq single
+1 / 0
In an a.c. circuit $I=50 \sin 100 \pi t$. The time required for the current to achieve its peak value will be
Q51.
mcq single
+1 / 0
If $|\vec{A} \times \vec{B}|=\sqrt{3}(\vec{A} \cdot \vec{B})$ then the value of $|\vec{A}+\vec{B}|$ is $\left(\tan 60^{\circ}=\sqrt{3}, \cos 60^{\circ}=0.5\right)$
Q52.
mcq single
+1 / 0
' n ' polarising sheets are arranged such that each makes an angle $45^{\circ}$ with the proceeding sheet. An unpolarised light of intensity I is incident into this arrangement. The output intensity is I/ 64 . The value of $n$ will be $\left(\cos 45^{\circ}=1 / \sqrt{2}\right)$
Q53.
mcq single
+1 / 0
In an interference experiment, the phase difference between the waves reaching a first dark point is
Q54.
mcq single
+1 / 0
In Young's double slit experiment, the intensities at two points, for the path difference $\frac{\lambda}{4}$ and $\frac{\lambda}{3}$ are $\mathrm{I}_1$ and $\mathrm{I}_2$ respectively. If $\mathrm{I}_0$ denotes the intensity produced by each one of the individual slits then the ratio $\left(\mathrm{I}_1+\mathrm{I}_2\right): \mathrm{I}_0$ is $\left(\cos 45^{\circ}=1 / \sqrt{2}\right)\left(\cos 60^{\circ}=0.5\right)$
Q55.
mcq single
+1 / 0
The average velocity of a particle performing S.H.M. in one complete vibration is ( $\mathrm{A}=$ amplitude of S.H.M., $\omega=$ angular velocity)
Q56.
mcq single
+1 / 0
A mass ' $M$ ' is suspended from a spring of negligible mass. The spring is pulled a little and then released so that the mass executes S.H.M. of periodic time ' $T$ '. If the mass is increased by ' $m$ ', the time period becomes $4 T / 3$, then the ratio of $\frac{\mathrm{M}}{\mathrm{m}}$ is
Q57.
mcq single
+1 / 0
A particle vibrating simple harmonically has an acceleration of $16 \mathrm{~cm} / \mathrm{s}^2$, when it is at a distance of 4 cm from the mean position. Its periodic time is
Q58.
mcq single
+1 / 0
Four solid spheres each of mass ' $m$ ' and radius ' $r$ ' are arranged as shown in the figure. The moment of inertia of the system about the given axis of rotation $\mathrm{AA}^1$ is

Q59.
mcq single
+1 / 0
Four thin rods of same mass M and same length L form a square as shown in figure. Moment of inertia of this system about an axis passing through point O and perpendicular to its plane is

Q60.
mcq single
+1 / 0
Two inductors of 88 mH each are joined in parallel. The current passing through this combination is 4 A . The energy stored in this combination of inductors in joule is
Q61.
mcq single
+1 / 0
If current ' i ' is passing through the solenoid of diameter ' $d$ ' having number of turns per unit length ' $n$ ', then the inductance per unit length near the middle of a long solenoid is directly proportional to
Q62.
mcq single
+1 / 0
A bicycle wheel of diameter ' $D$ ' has ' $N$ ' number of spokes. Wheel is rotating at the rate of ' $x$ ' revolutions per minute, perpendicular to the horizontal component of earth's magnetic field ' $\mathrm{B}_{\mathrm{H}}$ '. The e.m.f. induced between the rim and the centre of the wheel will be
Q63.
mcq single
+1 / 0
A string of length $L$ is fixed at one end and carries a mass m at the other end. The string makes $\frac{3}{\pi}$ revolutions per second around the vertical axis through the fixed end. The tension in the string is

Q64.
mcq single
+1 / 0
Two spherical black bodies of radius $\mathrm{R}_1$ and $\mathrm{R}_2$ with surface temperatures $\mathrm{T}_1$ and $\mathrm{T}_2$ respectively. If $T_1=2 T_2$ and they radiate the same power then relation between $R_1$ and $R_2$ is
Q65.
mcq single
+1 / 0
In an adiabatic change, the pressure and temperature of a diatomic gas are related as $\mathrm{P} \propto \mathrm{T}^{\mathrm{c}}$, where c is a constant. The value of c is (consider gas molecules are rigid)
Q66.
mcq single
+1 / 0
A system is compressed adiabatically, then its temperature
Q67.
mcq single
+1 / 0
When the r.m.s. velocity of a gas is denoted by ' V ' and temperature of the gas by T , which of the following relations is TRUE?
Q68.
mcq single
+1 / 0
A gas at $800^{\circ} \mathrm{C}$ is cooled until both its pressure and volume are halved. The final temperature of the gas will be
Q69.
mcq single
+1 / 0
The average kinetic energy of a molecule of the gas is
Q70.
mcq single
+1 / 0
A photon and an electron have equal energy ' $E$ '. The ratio of wavelength of photon to wavelength of electron is proportional to
Q71.
mcq single
+1 / 0
The energy of a photon is equal to the kinetic energy of proton. If $\lambda_1$ is the de-Broglie wavelength of a proton, $\lambda_2$ is the wavelength associated with the photon and if E is the energy of photon then $\lambda_2: \lambda_1$ is
Q72.
mcq single
+1 / 0
The charges are arranged at the four corners of square $A B C D$ of side ' $d$ ' as shown in figure. The work required to put this arrangement together is given by

Q73.
mcq single
+1 / 0
A simple pendulum has mass 2 gram and charge $2 \mu \mathrm{C}$. In a uniform horizontal electric field of intensity $1000 \mathrm{~V} / \mathrm{m}$, pendulum is at rest. At equilibrium, the angle made by the pendulum with the vertical is ( $\mathrm{g}=$ acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2$ )
Q74.
mcq single
+1 / 0
The value of acceleration due to gravity (g) becomes $\left(\frac{g}{3}\right)$ at height ' $h$ ' above the earth's surface. If ' R ' is the radius of earth, the height h will be equal to
Q75.
mcq single
+1 / 0
When three capacitors of capacitances ' C ', ' 2 C '? and ' 3 C ' are connected in series. The potential difference applied across the combination is ' $V$ '. The energy stored in the circuit is
Q76.
mcq single
+1 / 0
A lens of power +5 D is put in contact with a lens of power -3 D . The combination will behave like a
Q77.
mcq single
+1 / 0
In a projectile motion shown in figure, which one of the following statements is NOT CORRECT?

Q78.
mcq single
+1 / 0
The magnetic field at the centre of a current carrying circular coil of area ' $A$ ' is ' $B$ '. The magnetic moment of the coil is $x$ times $\left(2 B / \mu_0\right)$. The value of $x$ is ( $\mu_0=$ permeability of free space)
Q79.
mcq single
+1 / 0
A straight wire of mass ' M ' and length 2 m is placed in a magnetic field of 2 T which is acting perpendicular to the length of the wire. When a current of 1 A flows through the wire, the wire experiences an upthrust and leviates in a magnetic field. The mass ' $M$ ' of the wire is (acceleration due to gravity $\mathrm{g}=10 \mathrm{~m} / \mathrm{s}^2$ )
Q80.
mcq single
+1 / 0
A spherical liquid drop of radius R is divided into 64 equal droplets. If surface tension is $T$ then the work done in this process will be
Q81.
mcq single
+1 / 0
A glass capillary of radius 0.35 mm is inclined at $60^{\circ}$ with the vertical in water. The length of water column in the capillary tube is (surface tension of water $=7 \times 10^{-2} \mathrm{Nm}^{-1}$, acceleration due to gravity $=10 \mathrm{~m} / \mathrm{s}^2, \cos 0^{\circ}=1$, $\cos 60^{\circ}=0.5$, density of water $=1 \mathrm{gram} / \mathrm{cm}^3$ )
Q82.
mcq single
+1 / 0
If the shape of the liquid surface is curved, then the
Q83.
mcq single
+1 / 0
The series limit for the frequency of Balmer series is ' $\mathrm{v}_{\mathrm{B}}$ ', then the series limit frequency of Paschen series ' $v_p$ ' is
Q84.
mcq single
+1 / 0
In the spectrum of hydrogen atom the ratio of the longest wavelength in the Balmer series to the longest wavelength in the Lyman series is
Q85.
mcq single
+1 / 0
A passenger is sitting in a train which is moving fast. The engine blows a whistle of frequency ' n '. If the apparent frequency of sound heard by the passenger is $n^1$ then
Q86.
mcq single
+1 / 0
A wave is travelling in the negative x direction having displacement 3 cm along Y direction, wavelength $2 \pi \mathrm{~m}$ and frequency $\left(\frac{1}{2 \pi}\right) \mathrm{Hz}$ is represented as ( $t=$ time)
Q87.
mcq single
+1 / 0
Two points on a travelling wave having frequency 500 Hz and velocity $300 \mathrm{~m} / \mathrm{s}$ are $30^{\circ}$ out of phase, then the minimum distance between the two points is
Q88.
mcq single
+1 / 0
A simple harmonic progressive wave is given by $y=A \sin 2 \pi\left(n t-\frac{x}{\lambda}\right)$. If the wave velocity is equal to $=\frac{1}{3}$ (maximum particle velocity) then the wavelength $\lambda$ is given by
Q89.
mcq single
+1 / 0
Force is applied to a body of mass 2 kg at rest on a frictionless horizontal surface as shown in the force against time $(\mathrm{F}-\mathrm{t})$ graph. The speed of the body after 1 s is

Q90.
mcq single
+1 / 0
A solenoid has core of a material with relative permeability 301 and its winding carries a current of 2 A . The number of turns of the solenoid is 600 per metre. The magnetization of the material is
Q91.
mcq single
+1 / 0
The range of voltmeter of resistance $300 \Omega$ is 5 V . The resistance required to be connected to convert it into an ammeter of range 5 A is nearly
Q92.
mcq single
+1 / 0
In a given circuit, AB is a uniform potentiometer wire. If the value of resistance ' $R$ ' is slightly increased, the balance point

Q93.
mcq single
+1 / 0
A rigid body weighs 25 gram and has a measured volume of $5 \mathrm{~cm}^3$. The possible error in the measurement of mass and volume are 0.01 gram and $0.1 \mathrm{~cm}^3$. The maximum error in the density will be
Q94.
mcq single
+1 / 0
In common emitter configuration, $\beta, \mathrm{R}_{\mathrm{L}}$ and r are the a.c. current gain, load resistance and the input resistance of a transistor respectively. The voltage and power gain respectively are
Q95.
mcq single
+1 / 0
When forward bias is applied to a p-n junction, then the potential barrier and the width of the depletion region respectively.
Q96.
mcq single
+1 / 0
The order of (a) transformer (b) diode rectifier (c) filter (d) voltage regulator in a block diagram of simple rectifier circuit is as
(a) b, d, c, a
(b) a, c, d, b
(c) a, b, c, d
(d) d, c, a, b
Q97.
mcq single
+1 / 0
An alternating voltage $\mathrm{V}=100 \sqrt{2} \sin (50 \mathrm{t})$ is connected to $2 \mu \mathrm{~F}$ capacitor through a.c. ammeter. The reading of the ammeter is
Q98.
mcq single
+1 / 0
A capacitor of capacitance ' C ' is connected across a.c. source of voltage V as $\mathrm{V}=\mathrm{V}_0 \sin \omega \mathrm{t}$. The displacement current between the plates of the capacitor would be
Q99.
mcq single
+1 / 0
An inductor of 0.5 mH , capacitor of $20 \mu \mathrm{~F}$ and resistance of $20 \Omega$ are connected in series with a 220 V a.c. source. If the current is in phase with e.m.f., the maximum current in the circuit is $\sqrt{x} A$. The value of $x$ is
Q100.
mcq single
+1 / 0
In an a.c. circuit $I=50 \sin 100 \pi t$. The time required for the current to achieve its peak value will be
Biology
Biology
Q1.
mcq single
+1 / 0
The famous wine of Goa, traditionally called 'fenny', is made by fermenting the fleshy pedicels of __________ fruits.
Q2.
mcq single
+1 / 0
Spores of which one of the microbial biocontrol agents are used to get rid of butterfly?
Q3.
mcq single
+1 / 0
Which one of the following is NOT a cyanobacterial biofertilizer?
Q4.
mcq single
+1 / 0
Following changes occur during the successive seral stages of ecological succession EXCEPT _______
Q5.
mcq single
+1 / 0
Following are the limitations of ecological pyramids EXCEPT __________ .
Q6.
mcq single
+1 / 0
Given below are two statements.
Statement I - The long term storage places like sediments and oceanic water are termed as 'sinks'.
Statement II - Human activities like fossil fuel burning and deforestation have affected the global carbon cycle.
In the light of above statements, select the correct option given below:
Q7.
mcq single
+1 / 0
The amount of ________ determines the pattern of ecological succession in the given area.
Q8.
mcq single
+1 / 0
Following table shows ABO blood group in man. Observe carefully and identify 'P', 'Q' and ' R '.

Q9.
mcq single
+1 / 0
In a dihybrid cross, $9: 3: 3: 1$ ratio, denotes that ___________.
Q10.
mcq single
+1 / 0
Which of the following statements are correct regarding chromosomal basis of sex determination?
i. The mechanism of sex determination in insect is XX - XO type.
ii. In honey-bee, haplo-diploid type of sex determination is observed.
iii. Human males have homomorphic sex chromosomes.
iv. Human females have heteromorphic sex chromosomes.
v. Chromosomal mechanism of sex determination in birds is ZW-ZZ type.
Q11.
mcq single
+1 / 0
Sexually reproduced offsprings tend to differ from their parents. This is known as __________
Q12.
mcq single
+1 / 0
If you cross two heterozygous plants, how many of the all possible $F_1$ offsprings will also be heterozygous?
Q13.
mcq single
+1 / 0
Pongidae includes $\qquad$
Q14.
mcq single
+1 / 0
Due to formation of oxyhaemoglobin, the pH of blood decreases due to release of $\mathrm{H}^{+}$ion and the $\mathrm{H}_2 \mathrm{CO}_3$ dissociates into $\mathrm{H}_2 \mathrm{O}$ and $\mathrm{CO}_2$. This is called __________ .
Q15.
mcq single
+1 / 0
How does the Hb act as buffer?
Q16.
mcq single
+1 / 0
Match the Column I and II
Column (I)
Column (II)
i.
$\mathrm{CO}_2$ dissolved in plasma
a.
23 %
ii.
$\mathrm{CO}_2$ transported as bicarbonate ions
b.
7 %
iii.
$\mathrm{CO}_2$ transported by RBC as carbaminohaemoglobin
c.
70 %
iv.
$\mathrm{O}_2$ transportation by RBCs as oxyhaemoglobin
d.
97 %
Choose the correct option:
Q17.
mcq single
+1 / 0
Select the INCORRECT statements.
i. The rate of formation of $\mathrm{H}_2 \mathrm{CO}_3$ is slower in RBCs and faster in plasma.
ii. Dissociation of $\mathrm{H}_2 \mathrm{CO}_3$ into $\mathrm{H}^{+}$and $\mathrm{HCO}_3^{-}$ takes place due to carbonic anhydrase.
iii. The carbonic anhydrase is present in leucocytes,
iv. The prosthetic group of carbonic anhydrase is Mn .
Select the correct option.
Q18.
mcq single
+1 / 0
Which one of the following genetically engineered microorganisms, used for clearing oil spills in the sea, was given first biopatent?
Q19.
mcq single
+1 / 0
Which one of the following is NOT a traditional knowledge passed down through generations?
Q20.
mcq single
+1 / 0
Resting potential of an axon is ___________ .
Q21.
mcq single
+1 / 0
Which of the following statements is INCORRECT regarding macula lutea?
Q22.
mcq single
+1 / 0
Select the correct statements regarding sympathetic nervous system.
i. It consists of 31 pairs of sympathetic ganglia.
ii. It is also called cranio- sacral outflow.
iii. It controls body activities like fight and flight
iv. Pre-ganglionic fibres are long and post response. ganglionic fibres are short.
v. Adrenaline is produced towards terminal end of post-ganglionic fibres.
Q23.
mcq single
+1 / 0
Disorder caused due to accumulation of amyloid proteins in CNS is ___________
Q24.
mcq single
+1 / 0
About _________ percentage of starch gets converted to maltose in mouth.
Q25.
mcq single
+1 / 0
Select the mismatched pair
Q26.
mcq single
+1 / 0
Small intestine opens into large intestine at the junction of _________
Q27.
mcq single
+1 / 0
Identify P and Q regions in the diagram of histology of alimentary canal and identify the type of tissue.

Q28.
mcq single
+1 / 0
Which one of the following combination of bacterial strains used by Griffith, when injected to mice will NOT kill them?
Q29.
mcq single
+1 / 0
Identify the mRNA transcribed from the DNA having following nucleotide sequence.

Q30.
mcq single
+1 / 0
Glycine is coded by GGA in prokaryotic cell, it will code for ________ in eukaryotic cell.
Q31.
mcq single
+1 / 0
The core DNA in the nucleosome is _________ base pairs long
Q32.
mcq single
+1 / 0
Which type of mutation causes sickle cell anaemia?
Q33.
mcq single
+1 / 0
If the sequence of gene loci on a chromosome are A-B-C-D-E-O-T. Which of the following gene pair will have least probability of being inherited together?
Q34.
mcq single
+1 / 0
Any foreign substance invading body and capable of stimulating an immune response is _________ .
Q35.
mcq single
+1 / 0
Subcutaneous Filariasis is caused by __________
Q36.
mcq single
+1 / 0
Symptoms like disappearance of subcutaneous fat, prominent ribs, dry skin and atrophy of digestive glands are seen in one year old infant. Identify the disease this child is suffering from
Q37.
mcq single
+1 / 0
Excessive dosage of ____________ causes extreme happiness, irritability and paranoia
Q38.
mcq single
+1 / 0
Given below are two statements about absorption of water in plants.
Statement I - Rate of water absorption is maximum at soil temperature between 40 to $60^{\circ} \mathrm{C}$.
Statement II - High concentration of solutes in soil water increases the rate of absorption of water.
In the light of above statements, select the correct option given below:
Q39.
mcq single
+1 / 0
Stomatal transpiration takes place during day time in the following plants EXCEPT __________
Q40.
mcq single
+1 / 0
Water constitutes about ______________to ____________ $\%$ of plant cells and tissues.
Q41.
mcq single
+1 / 0
Match the concepts given in Column I with their scientist who proposed them given in Column II.
Column (I)
Column (II)
i.
Osmotic absorption
a.
Bohem
ii.
Non-osmotic absorption
b.
Dixon and Jolly
iii.
Capillarity theory
c.
Atkins and Priestley
iv.
Cohesion- tension theory
d.
Kramer and Thiman
Choose the correct option:
Q42.
mcq single
+1 / 0
The following forms of water EXCEPT _________ water is NOT available to plants for absorption.
Q43.
mcq single
+1 / 0
The quantity of CO and hydrocarbons in the exhaust of a motor vehicle can be reduced by installing ____________ in it.
i. a scrubber
ii. an electrostatic precipitator
iii. a catalytic converter
Select the correct option:
Q44.
mcq single
+1 / 0
Given below are two statements.
Statement I - Thickness of ozone is more above the poles than at the equator.
Statement II - The 'ozone holes' are generally found in Antarctic region.
In the light of above statements, select the correct option given below:
Q45.
mcq single
+1 / 0
Match the Column I with Column II with reference to biomagnification of DDT.
Column (I)
(Organism)
Column (II)
(DDT content)
i.
Small fishes
a.
0.04 ppm
ii.
Fish eating Birds of Prey
b.
0.5 ppm
iii.
Large fishes
c.
25 ppm
iv.
Zooplankton
d.
2.ppm
Choose the correct option:
Q46.
mcq single
+1 / 0
In the above logistic growth curve of population, identify ' $A$ ', ' $B$ ' and ' $C$ '.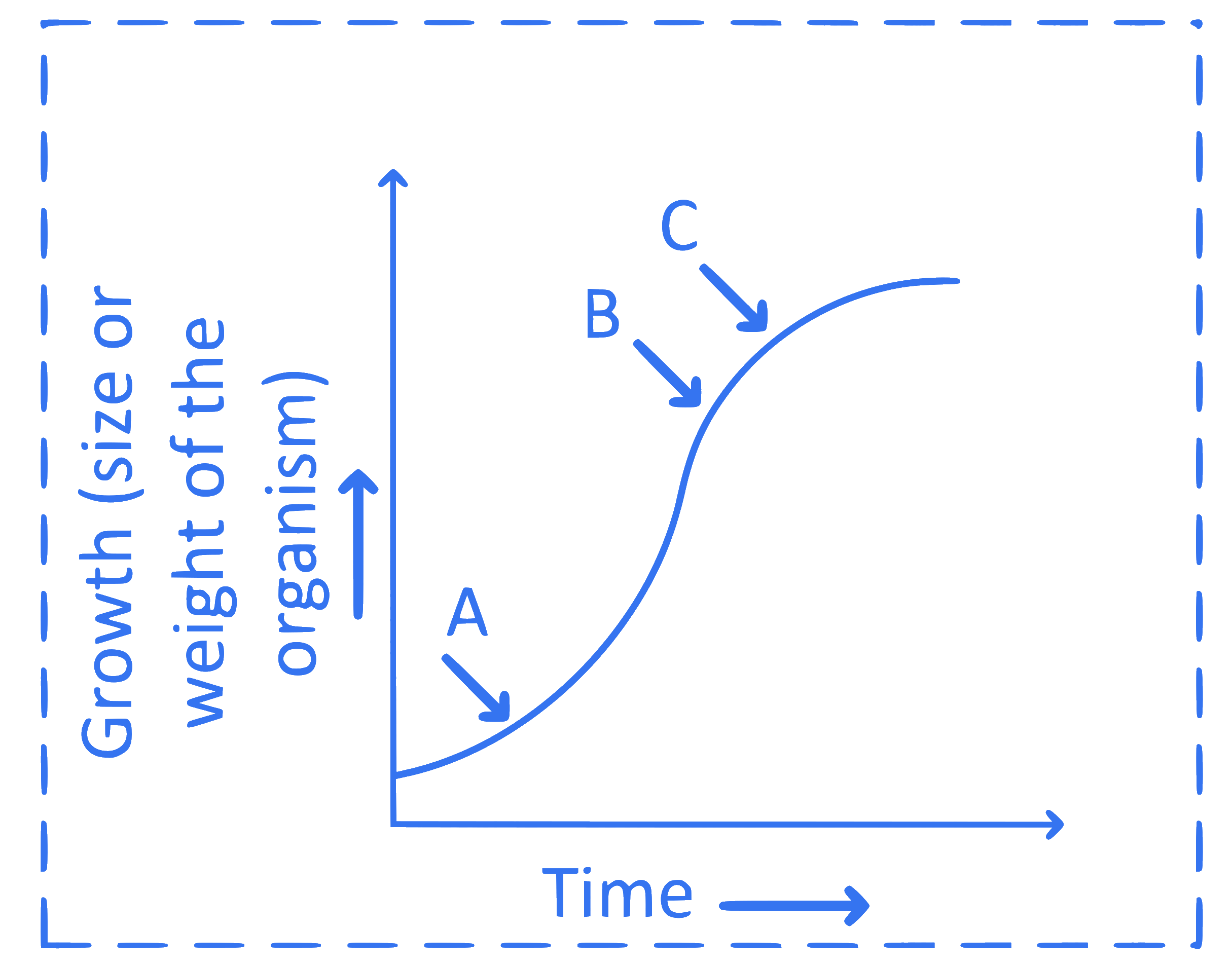

Q47.
mcq single
+1 / 0
Which one of the following is INCORRECT regarding 'resource partitioning'?
Q48.
mcq single
+1 / 0
Interaction between flamingos visiting Mumbai west coast and resident fishes is of _________ type.
Q49.
mcq single
+1 / 0
Match the Column I with Column II.
Column (I)
(Parasite)
\begin{aligned}
& \text { Column (II) } \\
& \text { (Host) }
\end{aligned}
i.
Ticks
a.
Marine fish
ii.
Copepods
b.
Dog
iii.
Cuscuta
c.
Indian crow
iv.
Asian Koel
d.
Hedge plants
Choose the correct option:
Q50.
mcq single
+1 / 0
The smallest WBC is __________.
Q51.
mcq single
+1 / 0
If 10 ml of the blood sample of a healthy person, containing added anticoagulant, is subjected to centrifugation, then approximately how much will be the amount of plasma?
Q52.
mcq single
+1 / 0
Given below are two statements.
Statement I - Wall of the left ventricle is more muscular and about three times thicker than that of the right ventricle.
Statement II - Columnae carneae are present in the wall of atria as well as ventricles.
In the light of above statements, select the correct option given below:
Q53.
mcq single
+1 / 0
Which of the following is NOT the function of erythrocytes?
Q54.
mcq single
+1 / 0
Which of the following covering of testis originates from peritoneum?
Q55.
mcq single
+1 / 0
Which of the following is NOT an acrosomal reaction?
Q56.
mcq single
+1 / 0
The blastocyst stage of development is reached by the end of ___________ day.
Q57.
mcq single
+1 / 0
Given below are two statements.
Statement I - In the follicular phase of menstrual cycle, the thickness of endometrium reaches $3-5 \mathrm{~mm}$.
Statement II - FSH stimulates 6-12 primary follicles to proceed in development to form secondary follicles.
In the light of above statements, Select the correct option given below:
Q58.
mcq single
+1 / 0
With respect to spermatogenesis, match Column I with Column II and select the correct option.
Column I
Column II
i.
Phase of growth
a.
Spermatid
ii.
Spermiogenesis
b.
Primary Spermatocyte
iii.
Production of spermatogonia
c.
Meiosis
iv.
Phase
maturation of
d.
Mitosis
Q59.
mcq single
+1 / 0
Given below is a list of hormones. How many of the following are hypothalamic hormones?
i. Thyroid stimulating Hormone.
ii. Follicle stimulating hormone.
iii. Oxytocin
iv. Collip's hormone
v. Prolactostatin
vi. Gonadotropin Releasing hormone
vii. Adrenocorticotropin releasing hormone.
Select the correct option:
Q60.
mcq single
+1 / 0
Which one of the following neurohypophyseal hormones brings about increase in BP?
Q61.
mcq single
+1 / 0
Which one of the following hormones is a modified amino acid?
Q62.
mcq single
+1 / 0
Gland which was considered vestigial but later found to produce melatonin is _________
Q63.
mcq single
+1 / 0
Given below are two statements.
Statement I - The pituitary gland is situated in sella turcica of palate bone.
Statement II - The pituitary gland is attached to the hypothalamus by a hypophyseal stalk.
In the light of above statements, select the correct option given below:
Q64.
mcq single
+1 / 0
The purposeful manipulation in the heredity of crops and the production of new superior varieties of existing crop plants is called ___________
Q65.
mcq single
+1 / 0
Match a type of artificial method of vegetative reproduction given in Column I with examples given in Column II.
Column (I)
Column (II)
i.
Root cutting
a.
Apple
ii.
Bud grafting
b.
Sansevieria
iii.
Stem cutting
c.
Blackberry
iv.
Leaf cutting
d.
Rose
Choose the correct option:
Q66.
mcq single
+1 / 0
The current loss of biodiversity which is progressing at an alarming rate is estimated to be ________ to __________ times faster than prehuman times.
Q67.
mcq single
+1 / 0
Which one of the following represents qualitative growth in plants?
Q68.
mcq single
+1 / 0
If dark period is interrupted by a flash of red light of 660 nm , then SDP will not flower. But if it is immediately followed by _________ then SDP will flower.
Q69.
mcq single
+1 / 0
The figure shows photoperiodism. Identify its correct explanation from the following.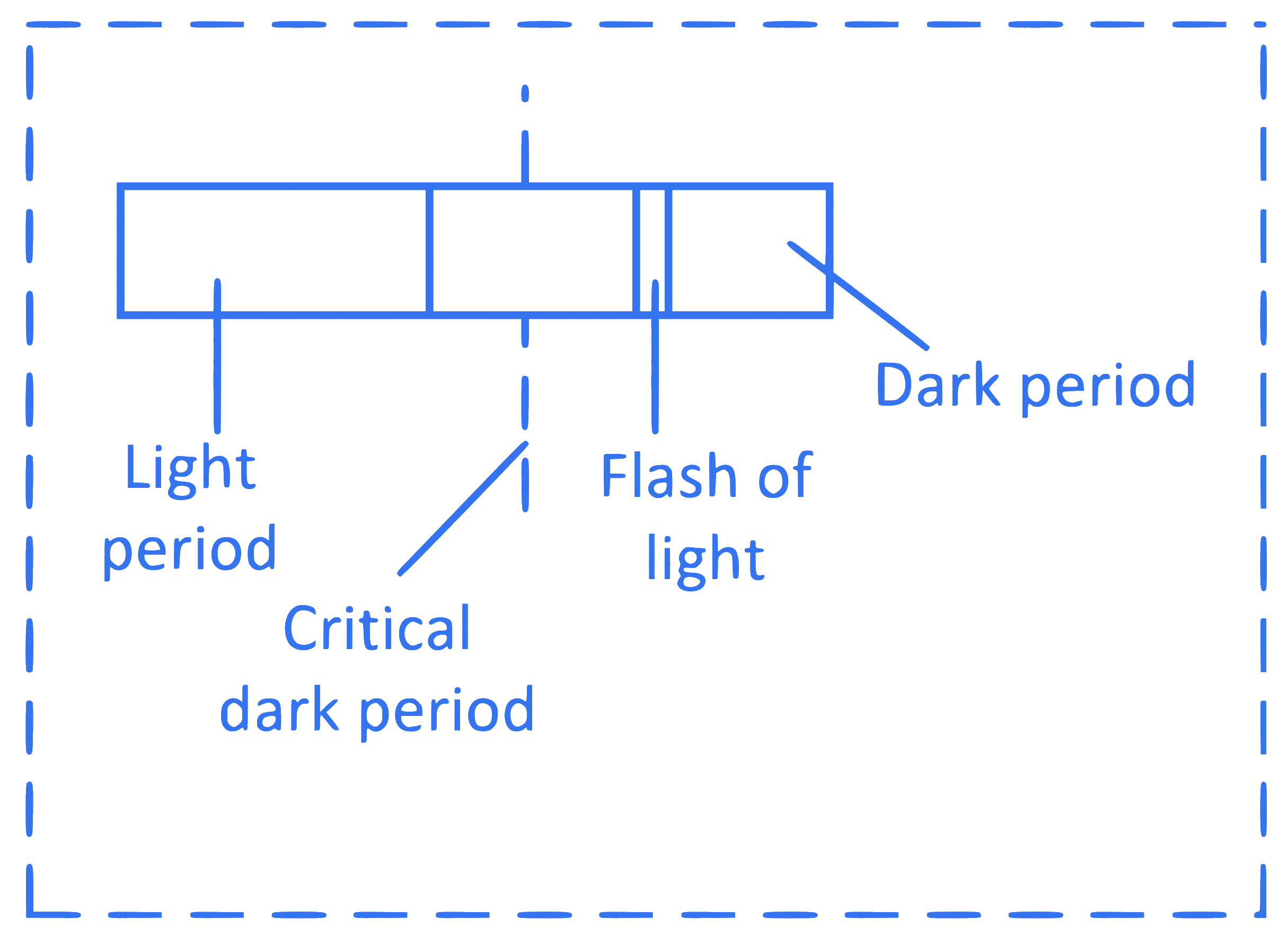

Q70.
mcq single
+1 / 0
Which one of the following plant hormones does NOT prevent abscission in plants?
Q71.
mcq single
+1 / 0
Which one of the following enters mitochondrial matrix after glycolysis?
Q72.
mcq single
+1 / 0
How many times dehydrogenation occurs during Krebs cycle?
Q73.
mcq single
+1 / 0
In Glycolysis, which one of the following conversion shows ATP formation?
Q74.
mcq single
+1 / 0
Total number of ATP molecules formed by substrate level phosphorylation in complete oxidation of one glucose molecule are ___________.
Q75.
mcq single
+1 / 0
Given below are two statements.
Statement I - Glycolysis occurs in cytoplasm of the cell.
Statement II - Glycolysis is a common process in aerobic as well as anaerobic respiration.
In the light of above statements, select the correct option given below:
Q76.
mcq single
+1 / 0
Darwin's finches are good example of ____________
Q77.
mcq single
+1 / 0
Match concepts of origin of life in Column I with their examples given in Column II.
Column (I)
Column (II)
i.
Geographical
a.
Moths of Great Britain in 1845
ii.
Convergent evolution
b.
Bones of forelimbs in man and whale
iii.
Divergent evolution
c.
Wings of butterfly and bird
iv.
Natural selection
d.
Darwin finches
Choose the correct option:
Q78.
mcq single
+1 / 0
Given below are two statements.
Statement I - Directional selection favours intermediate forms and eliminates both phenotypic extremes.
Statement II - In directional selection, more individuals acquire value other than mean character value.
In the light of above statements, select the correct option given below:
Q79.
mcq single
+1 / 0
Arrange the following events of chemical evolution in proper sequence.
i. formation of nucleic acid
ii. formation of eobionts
iii. formation of protocell
iv. formation of complex organic compounds
Q80.
mcq single
+1 / 0
Which of the following pairs of organisms are ureotelic?
Q81.
mcq single
+1 / 0
Which of the following is correct regarding cystine stones?
i. These are formed in response to bacterial infection.
ii. It is a genetic disorder.
iii. These grow quickly and become quite large.
iv. Occurs in people who consume high protein diet.
v. People who suffer excrete too much of certain amino acid.
Q82.
mcq single
+1 / 0
Which of the following is INCORRECT in case of severe haemorrhage?
Q83.
mcq single
+1 / 0
In oliguria condition, urine output is less than ___________.
Q84.
mcq single
+1 / 0
Match the Columns I, II and III based an excretion.
Column I
(Part of Nephron)
Column II
(Type of cells)
Column III
(Function)
a.
Bownman's capsule
(i)
Squamous epithelium
p.
passage of filterate
b.
Neck of nephron
(ii)
Cuboidal epithelium
q.
selective reabsorption
c.
Proximal convoluted tubule
(iii)
Ciliated epithelium
r.
filtration of blood
Q85.
mcq single
+1 / 0
Primary urine contains all of the following, EXCEPT
Q86.
mcq single
+1 / 0
Given below are two statements.
Statement I - Defective development of foetus is examined by amniocentesis.
Statement II - Defective foetus can be terminated after 20 weeks of pregnancy.
In the light of above statements, select the correct option given below:
Q87.
mcq single
+1 / 0
Which of the following pairs are haploid structures?
Q88.
mcq single
+1 / 0
Growth of the pollen tube in ovule is guided by the secretions of
Q89.
mcq single
+1 / 0
When a biotic agent is bat, the pollination is called
Q90.
mcq single
+1 / 0
Germ pores of pollen grains are useful for
Q91.
mcq single
+1 / 0
Identify the labels ' X ', ${ }^{\prime} \mathrm{Y}^{\prime}$ and ' Z ' in following figure.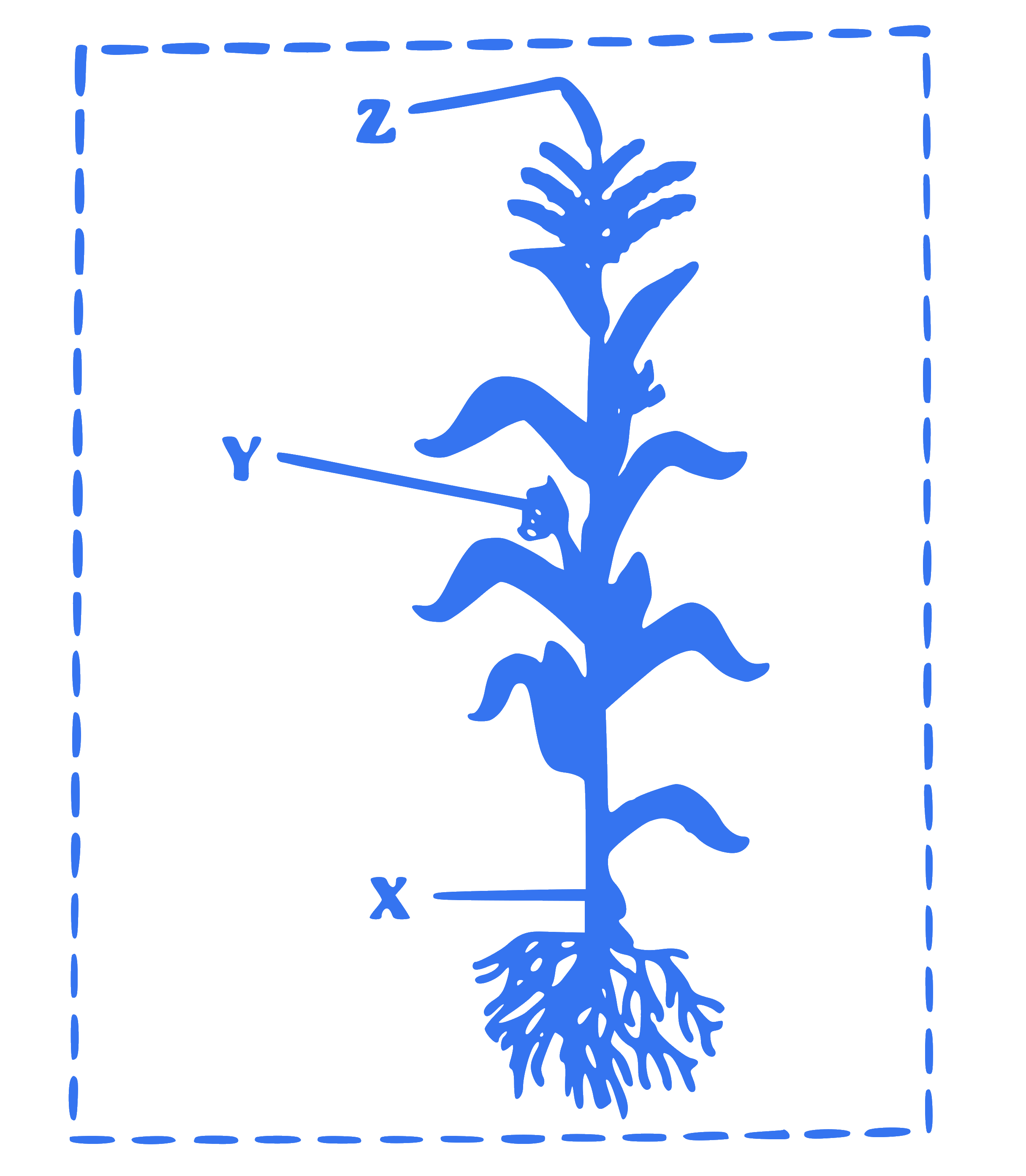

Q92.
mcq single
+1 / 0
A graph shows effect of substrate concentration on enzyme activity. In this graph ' Y ' axis represents.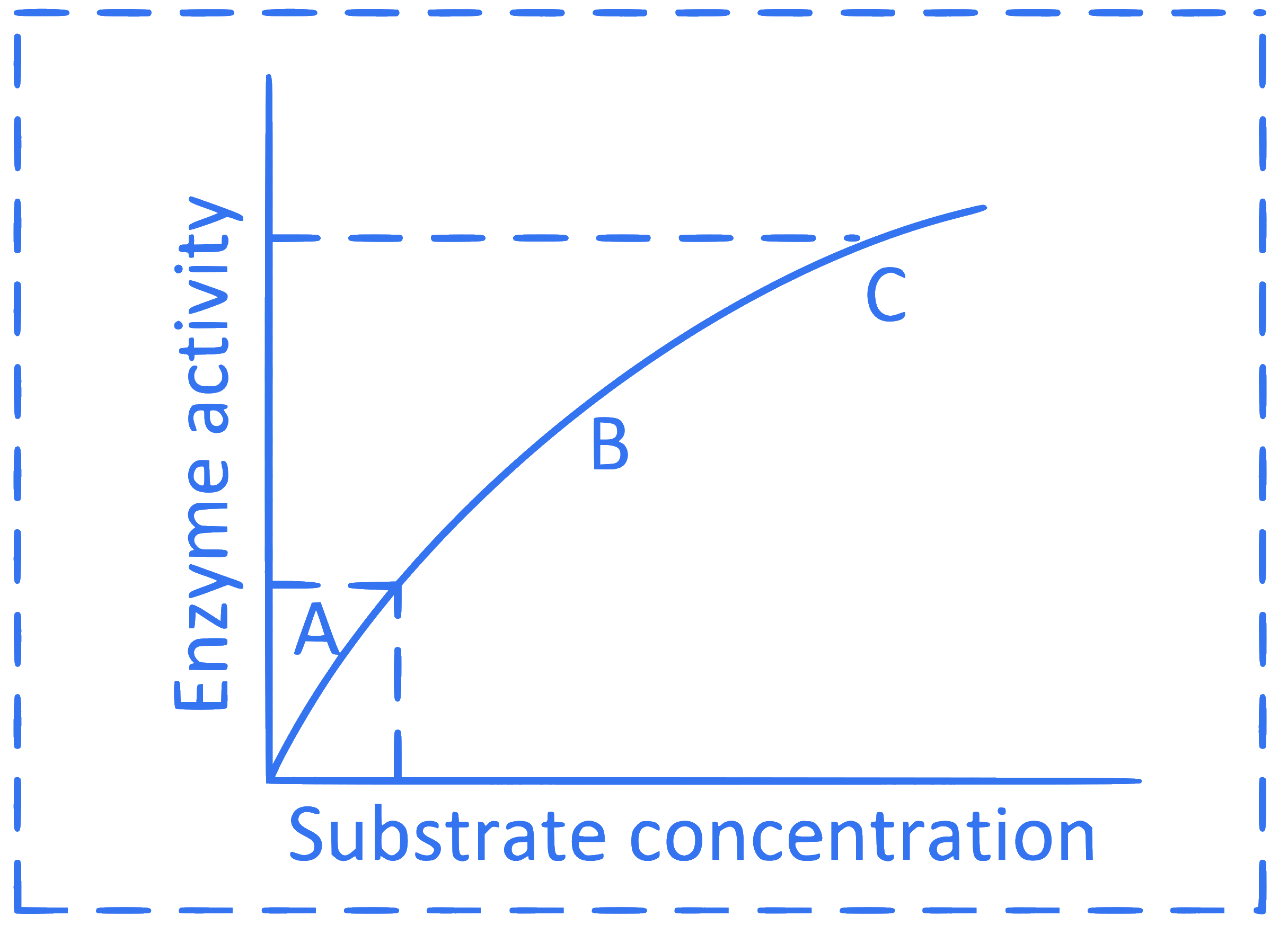

Q93.
mcq single
+1 / 0
Which of the following organic acid is used in medicine for solubility of $\mathrm{Ca}^{++}$?
Q94.
mcq single
+1 / 0
A molecule of triglyceride is composed of
Q95.
mcq single
+1 / 0
Cerebrosides found in white matter of brain and myelin sheath contains
Q96.
mcq single
+1 / 0
Which one of the following is NOT a prime element?
Q97.
mcq single
+1 / 0
What is the time duration required for completion of one PCR Cycle?
Q98.
mcq single
+1 / 0
Given below are two statements.
Statement I - The recognition sites of RENs are 4-8 nucleotides bps long and are characterized by a particular type of internal symmetry.
Statement II - Different restriction enzymes found in different organisms recognise different nucleotide sequences and cut the DNA at different sites.
In the light of above statements, select the correct option given below:
Q99.
mcq single
+1 / 0
In genetic engineering, the foreign DNA is also called ___________ .
Q100.
mcq single
+1 / 0
Minerals are absorbed by plants in the ionic form through _________ region of roots.
Q101.
mcq single
+1 / 0
The famous wine of Goa, traditionally called 'fenny', is made by fermenting the fleshy pedicels of __________ fruits.
Q102.
mcq single
+1 / 0
Spores of which one of the microbial biocontrol agents are used to get rid of butterfly?
Q103.
mcq single
+1 / 0
Which one of the following is NOT a cyanobacterial biofertilizer?
Q104.
mcq single
+1 / 0
Following changes occur during the successive seral stages of ecological succession EXCEPT _______
Q105.
mcq single
+1 / 0
Following are the limitations of ecological pyramids EXCEPT __________ .
Q106.
mcq single
+1 / 0
Given below are two statements.
Statement I - The long term storage places like sediments and oceanic water are termed as 'sinks'.
Statement II - Human activities like fossil fuel burning and deforestation have affected the global carbon cycle.
In the light of above statements, select the correct option given below:
Q107.
mcq single
+1 / 0
The amount of ________ determines the pattern of ecological succession in the given area.
Q108.
mcq single
+1 / 0
Following table shows ABO blood group in man. Observe carefully and identify 'P', 'Q' and ' R '.

Q109.
mcq single
+1 / 0
In a dihybrid cross, $9: 3: 3: 1$ ratio, denotes that ___________.
Q110.
mcq single
+1 / 0
Which of the following statements are correct regarding chromosomal basis of sex determination?
i. The mechanism of sex determination in insect is XX - XO type.
ii. In honey-bee, haplo-diploid type of sex determination is observed.
iii. Human males have homomorphic sex chromosomes.
iv. Human females have heteromorphic sex chromosomes.
v. Chromosomal mechanism of sex determination in birds is ZW-ZZ type.
Q111.
mcq single
+1 / 0
Sexually reproduced offsprings tend to differ from their parents. This is known as __________
Q112.
mcq single
+1 / 0
If you cross two heterozygous plants, how many of the all possible $F_1$ offsprings will also be heterozygous?
Q113.
mcq single
+1 / 0
Pongidae includes $\qquad$
Q114.
mcq single
+1 / 0
Due to formation of oxyhaemoglobin, the pH of blood decreases due to release of $\mathrm{H}^{+}$ion and the $\mathrm{H}_2 \mathrm{CO}_3$ dissociates into $\mathrm{H}_2 \mathrm{O}$ and $\mathrm{CO}_2$. This is called __________ .
Q115.
mcq single
+1 / 0
How does the Hb act as buffer?
Q116.
mcq single
+1 / 0
Match the Column I and II
Column (I)
Column (II)
i.
$\mathrm{CO}_2$ dissolved in plasma
a.
23 %
ii.
$\mathrm{CO}_2$ transported as bicarbonate ions
b.
7 %
iii.
$\mathrm{CO}_2$ transported by RBC as carbaminohaemoglobin
c.
70 %
iv.
$\mathrm{O}_2$ transportation by RBCs as oxyhaemoglobin
d.
97 %
Choose the correct option:
Q117.
mcq single
+1 / 0
Select the INCORRECT statements.
i. The rate of formation of $\mathrm{H}_2 \mathrm{CO}_3$ is slower in RBCs and faster in plasma.
ii. Dissociation of $\mathrm{H}_2 \mathrm{CO}_3$ into $\mathrm{H}^{+}$and $\mathrm{HCO}_3^{-}$ takes place due to carbonic anhydrase.
iii. The carbonic anhydrase is present in leucocytes,
iv. The prosthetic group of carbonic anhydrase is Mn .
Select the correct option.
Q118.
mcq single
+1 / 0
Which one of the following genetically engineered microorganisms, used for clearing oil spills in the sea, was given first biopatent?
Q119.
mcq single
+1 / 0
Which one of the following is NOT a traditional knowledge passed down through generations?
Q120.
mcq single
+1 / 0
Resting potential of an axon is ___________ .
Q121.
mcq single
+1 / 0
Which of the following statements is INCORRECT regarding macula lutea?
Q122.
mcq single
+1 / 0
Select the correct statements regarding sympathetic nervous system.
i. It consists of 31 pairs of sympathetic ganglia.
ii. It is also called cranio- sacral outflow.
iii. It controls body activities like fight and flight
iv. Pre-ganglionic fibres are long and post response. ganglionic fibres are short.
v. Adrenaline is produced towards terminal end of post-ganglionic fibres.
Q123.
mcq single
+1 / 0
Disorder caused due to accumulation of amyloid proteins in CNS is ___________
Q124.
mcq single
+1 / 0
About _________ percentage of starch gets converted to maltose in mouth.
Q125.
mcq single
+1 / 0
Select the mismatched pair
Q126.
mcq single
+1 / 0
Small intestine opens into large intestine at the junction of _________
Q127.
mcq single
+1 / 0
Identify P and Q regions in the diagram of histology of alimentary canal and identify the type of tissue.

Q128.
mcq single
+1 / 0
Which one of the following combination of bacterial strains used by Griffith, when injected to mice will NOT kill them?
Q129.
mcq single
+1 / 0
Identify the mRNA transcribed from the DNA having following nucleotide sequence.

Q130.
mcq single
+1 / 0
Glycine is coded by GGA in prokaryotic cell, it will code for ________ in eukaryotic cell.
Q131.
mcq single
+1 / 0
The core DNA in the nucleosome is _________ base pairs long
Q132.
mcq single
+1 / 0
Which type of mutation causes sickle cell anaemia?
Q133.
mcq single
+1 / 0
If the sequence of gene loci on a chromosome are A-B-C-D-E-O-T. Which of the following gene pair will have least probability of being inherited together?
Q134.
mcq single
+1 / 0
Any foreign substance invading body and capable of stimulating an immune response is _________ .
Q135.
mcq single
+1 / 0
Subcutaneous Filariasis is caused by __________
Q136.
mcq single
+1 / 0
Symptoms like disappearance of subcutaneous fat, prominent ribs, dry skin and atrophy of digestive glands are seen in one year old infant. Identify the disease this child is suffering from
Q137.
mcq single
+1 / 0
Excessive dosage of ____________ causes extreme happiness, irritability and paranoia
Q138.
mcq single
+1 / 0
Given below are two statements about absorption of water in plants.
Statement I - Rate of water absorption is maximum at soil temperature between 40 to $60^{\circ} \mathrm{C}$.
Statement II - High concentration of solutes in soil water increases the rate of absorption of water.
In the light of above statements, select the correct option given below:
Q139.
mcq single
+1 / 0
Stomatal transpiration takes place during day time in the following plants EXCEPT __________
Q140.
mcq single
+1 / 0
Water constitutes about ______________to ____________ $\%$ of plant cells and tissues.
Q141.
mcq single
+1 / 0
Match the concepts given in Column I with their scientist who proposed them given in Column II.
Column (I)
Column (II)
i.
Osmotic absorption
a.
Bohem
ii.
Non-osmotic absorption
b.
Dixon and Jolly
iii.
Capillarity theory
c.
Atkins and Priestley
iv.
Cohesion- tension theory
d.
Kramer and Thiman
Choose the correct option:
Q142.
mcq single
+1 / 0
The following forms of water EXCEPT _________ water is NOT available to plants for absorption.
Q143.
mcq single
+1 / 0
The quantity of CO and hydrocarbons in the exhaust of a motor vehicle can be reduced by installing ____________ in it.
i. a scrubber
ii. an electrostatic precipitator
iii. a catalytic converter
Select the correct option:
Q144.
mcq single
+1 / 0
Given below are two statements.
Statement I - Thickness of ozone is more above the poles than at the equator.
Statement II - The 'ozone holes' are generally found in Antarctic region.
In the light of above statements, select the correct option given below:
Q145.
mcq single
+1 / 0
Match the Column I with Column II with reference to biomagnification of DDT.
Column (I)
(Organism)
Column (II)
(DDT content)
i.
Small fishes
a.
0.04 ppm
ii.
Fish eating Birds of Prey
b.
0.5 ppm
iii.
Large fishes
c.
25 ppm
iv.
Zooplankton
d.
2.ppm
Choose the correct option:
Q146.
mcq single
+1 / 0
In the above logistic growth curve of population, identify ' $A$ ', ' $B$ ' and ' $C$ '.

Q147.
mcq single
+1 / 0
Which one of the following is INCORRECT regarding 'resource partitioning'?
Q148.
mcq single
+1 / 0
Interaction between flamingos visiting Mumbai west coast and resident fishes is of _________ type.
Q149.
mcq single
+1 / 0
Match the Column I with Column II.
Column (I)
(Parasite)
\begin{aligned}
& \text { Column (II) } \\
& \text { (Host) }
\end{aligned}
i.
Ticks
a.
Marine fish
ii.
Copepods
b.
Dog
iii.
Cuscuta
c.
Indian crow
iv.
Asian Koel
d.
Hedge plants
Choose the correct option:
Q150.
mcq single
+1 / 0
The smallest WBC is __________.
Q151.
mcq single
+1 / 0
If 10 ml of the blood sample of a healthy person, containing added anticoagulant, is subjected to centrifugation, then approximately how much will be the amount of plasma?
Q152.
mcq single
+1 / 0
Given below are two statements.
Statement I - Wall of the left ventricle is more muscular and about three times thicker than that of the right ventricle.
Statement II - Columnae carneae are present in the wall of atria as well as ventricles.
In the light of above statements, select the correct option given below:
Q153.
mcq single
+1 / 0
Which of the following is NOT the function of erythrocytes?
Q154.
mcq single
+1 / 0
Which of the following covering of testis originates from peritoneum?
Q155.
mcq single
+1 / 0
Which of the following is NOT an acrosomal reaction?
Q156.
mcq single
+1 / 0
The blastocyst stage of development is reached by the end of ___________ day.
Q157.
mcq single
+1 / 0
Given below are two statements.
Statement I - In the follicular phase of menstrual cycle, the thickness of endometrium reaches $3-5 \mathrm{~mm}$.
Statement II - FSH stimulates 6-12 primary follicles to proceed in development to form secondary follicles.
In the light of above statements, Select the correct option given below:
Q158.
mcq single
+1 / 0
With respect to spermatogenesis, match Column I with Column II and select the correct option.
Column I
Column II
i.
Phase of growth
a.
Spermatid
ii.
Spermiogenesis
b.
Primary Spermatocyte
iii.
Production of spermatogonia
c.
Meiosis
iv.
Phase
maturation of
d.
Mitosis
Q159.
mcq single
+1 / 0
Given below is a list of hormones. How many of the following are hypothalamic hormones?
i. Thyroid stimulating Hormone.
ii. Follicle stimulating hormone.
iii. Oxytocin
iv. Collip's hormone
v. Prolactostatin
vi. Gonadotropin Releasing hormone
vii. Adrenocorticotropin releasing hormone.
Select the correct option:
Q160.
mcq single
+1 / 0
Which one of the following neurohypophyseal hormones brings about increase in BP?
Q161.
mcq single
+1 / 0
Which one of the following hormones is a modified amino acid?
Q162.
mcq single
+1 / 0
Gland which was considered vestigial but later found to produce melatonin is _________
Q163.
mcq single
+1 / 0
Given below are two statements.
Statement I - The pituitary gland is situated in sella turcica of palate bone.
Statement II - The pituitary gland is attached to the hypothalamus by a hypophyseal stalk.
In the light of above statements, select the correct option given below:
Q164.
mcq single
+1 / 0
The purposeful manipulation in the heredity of crops and the production of new superior varieties of existing crop plants is called ___________
Q165.
mcq single
+1 / 0
Match a type of artificial method of vegetative reproduction given in Column I with examples given in Column II.
Column (I)
Column (II)
i.
Root cutting
a.
Apple
ii.
Bud grafting
b.
Sansevieria
iii.
Stem cutting
c.
Blackberry
iv.
Leaf cutting
d.
Rose
Choose the correct option:
Q166.
mcq single
+1 / 0
The current loss of biodiversity which is progressing at an alarming rate is estimated to be ________ to __________ times faster than prehuman times.
Q167.
mcq single
+1 / 0
Which one of the following represents qualitative growth in plants?
Q168.
mcq single
+1 / 0
If dark period is interrupted by a flash of red light of 660 nm , then SDP will not flower. But if it is immediately followed by _________ then SDP will flower.
Q169.
mcq single
+1 / 0
The figure shows photoperiodism. Identify its correct explanation from the following.

Q170.
mcq single
+1 / 0
Which one of the following plant hormones does NOT prevent abscission in plants?
Q171.
mcq single
+1 / 0
Which one of the following enters mitochondrial matrix after glycolysis?
Q172.
mcq single
+1 / 0
How many times dehydrogenation occurs during Krebs cycle?
Q173.
mcq single
+1 / 0
In Glycolysis, which one of the following conversion shows ATP formation?
Q174.
mcq single
+1 / 0
Total number of ATP molecules formed by substrate level phosphorylation in complete oxidation of one glucose molecule are ___________.
Q175.
mcq single
+1 / 0
Given below are two statements.
Statement I - Glycolysis occurs in cytoplasm of the cell.
Statement II - Glycolysis is a common process in aerobic as well as anaerobic respiration.
In the light of above statements, select the correct option given below:
Q176.
mcq single
+1 / 0
Darwin's finches are good example of ____________
Q177.
mcq single
+1 / 0
Match concepts of origin of life in Column I with their examples given in Column II.
Column (I)
Column (II)
i.
Geographical
a.
Moths of Great Britain in 1845
ii.
Convergent evolution
b.
Bones of forelimbs in man and whale
iii.
Divergent evolution
c.
Wings of butterfly and bird
iv.
Natural selection
d.
Darwin finches
Choose the correct option:
Q178.
mcq single
+1 / 0
Given below are two statements.
Statement I - Directional selection favours intermediate forms and eliminates both phenotypic extremes.
Statement II - In directional selection, more individuals acquire value other than mean character value.
In the light of above statements, select the correct option given below:
Q179.
mcq single
+1 / 0
Arrange the following events of chemical evolution in proper sequence.
i. formation of nucleic acid
ii. formation of eobionts
iii. formation of protocell
iv. formation of complex organic compounds
Q180.
mcq single
+1 / 0
Which of the following pairs of organisms are ureotelic?
Q181.
mcq single
+1 / 0
Which of the following is correct regarding cystine stones?
i. These are formed in response to bacterial infection.
ii. It is a genetic disorder.
iii. These grow quickly and become quite large.
iv. Occurs in people who consume high protein diet.
v. People who suffer excrete too much of certain amino acid.
Q182.
mcq single
+1 / 0
Which of the following is INCORRECT in case of severe haemorrhage?
Q183.
mcq single
+1 / 0
In oliguria condition, urine output is less than ___________.
Q184.
mcq single
+1 / 0
Match the Columns I, II and III based an excretion.
Column I
(Part of Nephron)
Column II
(Type of cells)
Column III
(Function)
a.
Bownman's capsule
(i)
Squamous epithelium
p.
passage of filterate
b.
Neck of nephron
(ii)
Cuboidal epithelium
q.
selective reabsorption
c.
Proximal convoluted tubule
(iii)
Ciliated epithelium
r.
filtration of blood
Q185.
mcq single
+1 / 0
Primary urine contains all of the following, EXCEPT
Q186.
mcq single
+1 / 0
Given below are two statements.
Statement I - Defective development of foetus is examined by amniocentesis.
Statement II - Defective foetus can be terminated after 20 weeks of pregnancy.
In the light of above statements, select the correct option given below:
Q187.
mcq single
+1 / 0
Which of the following pairs are haploid structures?
Q188.
mcq single
+1 / 0
Growth of the pollen tube in ovule is guided by the secretions of
Q189.
mcq single
+1 / 0
When a biotic agent is bat, the pollination is called
Q190.
mcq single
+1 / 0
Germ pores of pollen grains are useful for
Q191.
mcq single
+1 / 0
Identify the labels ' X ', ${ }^{\prime} \mathrm{Y}^{\prime}$ and ' Z ' in following figure.

Q192.
mcq single
+1 / 0
A graph shows effect of substrate concentration on enzyme activity. In this graph ' Y ' axis represents.

Q193.
mcq single
+1 / 0
Which of the following organic acid is used in medicine for solubility of $\mathrm{Ca}^{++}$?
Q194.
mcq single
+1 / 0
A molecule of triglyceride is composed of
Q195.
mcq single
+1 / 0
Cerebrosides found in white matter of brain and myelin sheath contains
Q196.
mcq single
+1 / 0
Which one of the following is NOT a prime element?
Q197.
mcq single
+1 / 0
What is the time duration required for completion of one PCR Cycle?
Q198.
mcq single
+1 / 0
Given below are two statements.
Statement I - The recognition sites of RENs are 4-8 nucleotides bps long and are characterized by a particular type of internal symmetry.
Statement II - Different restriction enzymes found in different organisms recognise different nucleotide sequences and cut the DNA at different sites.
In the light of above statements, select the correct option given below:
Q199.
mcq single
+1 / 0
In genetic engineering, the foreign DNA is also called ___________ .
Q200.
mcq single
+1 / 0
Minerals are absorbed by plants in the ionic form through _________ region of roots.