aipmt-2015
AIPMT 2015
MEDICAL 2015 Previous Year
3 hDuration
600Total Marks
300Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 300 across 3 section(s); maximum marks: 600.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Assuming complete ionisation, same moles of which of the following compounds will require the least amount of acidified KMnO~4~ for complete oxidation?
Q2.
mcq single
+4 / 1
Gadolinium belongs to 4f series. Its atomic number is 64. Which of the following is the correct electronic configuration of gadolinium?
Q3.
mcq single
+4 / 1
In which of the following pairs, both the species are not isostructural ?
Q4.
mcq single
+4 / 1
Decreasing order of stability of O~2~, O~2~^($$-$$), O~2~^(+) and O~2~^(2$$-$$) is
Q5.
mcq single
+4 / 1
Which one of the following pairs of solution is not an acidic buffer ?
Q6.
mcq single
+4 / 1
What is the pH of the resulting solution when equal volumes of 0.1 M NaOH and 0.01 M HCl are mixed?
Q7.
mcq single
+4 / 1
The rate constant of the reaction A $$ \to $$ B is 0.6 $$ \times $$ 10^($$-$$3) mol L^($$-$$1) s^($$-$$1). If the concentration of A is 5 M, then concentration of B after 20 minutes is
Q8.
mcq single
+4 / 1
In an S~N~1 reaction on chiral centers, there is
Q9.
mcq single
+4 / 1
The number of water molecules is maximum in
Q10.
mcq single
+4 / 1
If Avogadro number N~A~, is changed from 6.022 $$ \times $$ 10^(23) mol^($$-$$1) to 6.022 $$ \times $$ 10^(20) mol^($$-$$1), this would change
Q11.
mcq single
+4 / 1
What is the mass of the precipitate formed when 50 mL of 16.9% solution of AgNO~3~ is mixed with 50 mL of 5.8% NaCl solution ? (Ag = 107.8, N = 14, O = 16, Na = 23, Cl = 35.5
Q12.
mcq single
+4 / 1
What is the mole fraction of the solute in a 1.00 m aqueous solution?
Q13.
mcq single
+4 / 1
If the equilibrium constant for
N~2(g)~ + O~2(g)~ $$\rightleftharpoons$$ 2NO~(g)~ is K, the equilibrium
constant for
$${1 \over 2}$$ N~2(g)~ + $${1 \over 2}$$O~2(g)~ $$\rightleftharpoons$$ NO~(g)~ will be
Q14.
mcq single
+4 / 1
The heat of combination of carbon to CO~2~ is $$-$$393.5 kJ/mol. The heat released upon formation of 35.2 g of CO~2~ from carbon and oxygen gas is
Q15.
mcq single
+4 / 1
Which of the statements given below is incorrect?
Q16.
mcq single
+4 / 1
The variation of the boiling points of the hydrogen halides is in the order HF > HI > HBr > HCl.
What explains the higher boiling point of hydrogen fluoride?
Q17.
mcq single
+4 / 1
The stability of +1 oxidation state among Al, Ga, In and Tl increases in the sequence
Q18.
mcq single
+4 / 1
Strong reducing behavior of H~3~PO~2~ is due to
Q19.
mcq single
+4 / 1
The formation of the oxide ion, O^(2$$-$$)~(g)~ from oxygen atom requires first an exthermic and then an endothermic step as shown below:
O~(g)~ + e^($$-$$) $$ \to $$ O^($$-$$)~(g)~; $$\Delta $$~f~H^(o) = $$-$$141 kJ mol^($$-$$1)
O^($$-$$)~(g)~ + e^($$-$$) $$ \to $$ O^(2$$-$$)~(g)~; $$\Delta $$~f~H^(o) = +780 kJ mol^($$-$$1)
Thus, process of formation of O^(2$$-$$) is isoelectronic with neon. It is due to the fact that,
Q20.
mcq single
+4 / 1
The hybridization involved in complex [Ni(CN)~4~]^($$2-$$) is (At. No. Ni = 28)
Q21.
mcq single
+4 / 1
Number of possible isomers for the complex [Co(en)~2~Cl~2~]Cl will be (en = ethylenediamine)
Q22.
mcq single
+4 / 1
The name of complex ion, [Fe(CN)~6~]^(3$$-$$) is
Q23.
mcq single
+4 / 1
The sum of coordination number and oxidation number of the metal M in the complex [M(en)~2~(C~2~O~4~)]Cl (where *en* is ethylenediamine) is
Q24.
mcq single
+4 / 1
The possible stereo-structures of CH~3~CHOHCOOH, which are optically active, are called
Q25.
mcq single
+4 / 1
Which of the following statements is not correct for a nucleophile?
Q26.
mcq single
+4 / 1
Reaction of a carbonyl compound with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is
Q27.
mcq single
+4 / 1
Which one of the following esters gets hydrolysed most easily under alkaline conditions?
Q28.
mcq single
+4 / 1
The oxidation of benzene by V~2~O~5~ in the presence of air produces
Q29.
mcq single
+4 / 1
Reaction of phenol with chloroform in presence of dilute sodium hydroxide finally introduces which one of the following functional group?
Q30.
mcq single
+4 / 1
Which of the following reaction(s) can be used for the preparation of alkyl halides?


Q31.
mcq single
+4 / 1
2,3-Dimethyl-2-butene can be prepared by heating which of the following compounds with a strong acid?
Q32.
mcq single
+4 / 1
Which of the following is not the product of dehydration of


Q33.
mcq single
+4 / 1
In the reaction with HCl, an alkene reacts in accordance with the Markovnikov's rule to give a product 1-chloro-1 methylcyclohexane. The possible alkene is
Q34.
mcq single
+4 / 1
Aqueous solution of which of the following compounds is the best conductor of electric current ?
Q35.
mcq single
+4 / 1
The number of structural isomers possible from the molecular formula C~3~H~9~N is
Q36.
mcq single
+4 / 1
The following reaction
 is known by the name
is known by the name
 is known by the name
is known by the name
Q37.
mcq single
+4 / 1
Method by which aniline cannot be prepared is
Q38.
mcq single
+4 / 1
Which is the correct order of increasing energy of the listed orbitals in the atom of titanium ? (At. no. Z = 22)
Q39.
mcq single
+4 / 1
Assuming complete ionisation, same moles of which of the following compounds will require the least amount of acidified KMnO~4~ for complete oxidation?
Q40.
mcq single
+4 / 1
Gadolinium belongs to 4f series. Its atomic number is 64. Which of the following is the correct electronic configuration of gadolinium?
Q41.
mcq single
+4 / 1
In which of the following pairs, both the species are not isostructural ?
Q42.
mcq single
+4 / 1
Decreasing order of stability of O~2~, O~2~^($$-$$), O~2~^(+) and O~2~^(2$$-$$) is
Q43.
mcq single
+4 / 1
Which one of the following pairs of solution is not an acidic buffer ?
Q44.
mcq single
+4 / 1
What is the pH of the resulting solution when equal volumes of 0.1 M NaOH and 0.01 M HCl are mixed?
Q45.
mcq single
+4 / 1
The rate constant of the reaction A $$ \to $$ B is 0.6 $$ \times $$ 10^($$-$$3) mol L^($$-$$1) s^($$-$$1). If the concentration of A is 5 M, then concentration of B after 20 minutes is
Q46.
mcq single
+4 / 1
In an S~N~1 reaction on chiral centers, there is
Q47.
mcq single
+4 / 1
The number of water molecules is maximum in
Q48.
mcq single
+4 / 1
If Avogadro number N~A~, is changed from 6.022 $$ \times $$ 10^(23) mol^($$-$$1) to 6.022 $$ \times $$ 10^(20) mol^($$-$$1), this would change
Q49.
mcq single
+4 / 1
What is the mass of the precipitate formed when 50 mL of 16.9% solution of AgNO~3~ is mixed with 50 mL of 5.8% NaCl solution ? (Ag = 107.8, N = 14, O = 16, Na = 23, Cl = 35.5
Q50.
mcq single
+4 / 1
What is the mole fraction of the solute in a 1.00 m aqueous solution?
Q51.
mcq single
+4 / 1
If the equilibrium constant for
N~2(g)~ + O~2(g)~ $$\rightleftharpoons$$ 2NO~(g)~ is K, the equilibrium
constant for
$${1 \over 2}$$ N~2(g)~ + $${1 \over 2}$$O~2(g)~ $$\rightleftharpoons$$ NO~(g)~ will be
Q52.
mcq single
+4 / 1
The heat of combination of carbon to CO~2~ is $$-$$393.5 kJ/mol. The heat released upon formation of 35.2 g of CO~2~ from carbon and oxygen gas is
Q53.
mcq single
+4 / 1
Which of the statements given below is incorrect?
Q54.
mcq single
+4 / 1
The variation of the boiling points of the hydrogen halides is in the order HF > HI > HBr > HCl.
What explains the higher boiling point of hydrogen fluoride?
Q55.
mcq single
+4 / 1
The stability of +1 oxidation state among Al, Ga, In and Tl increases in the sequence
Q56.
mcq single
+4 / 1
Strong reducing behavior of H~3~PO~2~ is due to
Q57.
mcq single
+4 / 1
The formation of the oxide ion, O^(2$$-$$)~(g)~ from oxygen atom requires first an exthermic and then an endothermic step as shown below:
O~(g)~ + e^($$-$$) $$ \to $$ O^($$-$$)~(g)~; $$\Delta $$~f~H^(o) = $$-$$141 kJ mol^($$-$$1)
O^($$-$$)~(g)~ + e^($$-$$) $$ \to $$ O^(2$$-$$)~(g)~; $$\Delta $$~f~H^(o) = +780 kJ mol^($$-$$1)
Thus, process of formation of O^(2$$-$$) is isoelectronic with neon. It is due to the fact that,
Q58.
mcq single
+4 / 1
The hybridization involved in complex [Ni(CN)~4~]^($$2-$$) is (At. No. Ni = 28)
Q59.
mcq single
+4 / 1
Number of possible isomers for the complex [Co(en)~2~Cl~2~]Cl will be (en = ethylenediamine)
Q60.
mcq single
+4 / 1
The name of complex ion, [Fe(CN)~6~]^(3$$-$$) is
Q61.
mcq single
+4 / 1
The sum of coordination number and oxidation number of the metal M in the complex [M(en)~2~(C~2~O~4~)]Cl (where *en* is ethylenediamine) is
Q62.
mcq single
+4 / 1
The possible stereo-structures of CH~3~CHOHCOOH, which are optically active, are called
Q63.
mcq single
+4 / 1
Which of the following statements is not correct for a nucleophile?
Q64.
mcq single
+4 / 1
Reaction of a carbonyl compound with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is
Q65.
mcq single
+4 / 1
Which one of the following esters gets hydrolysed most easily under alkaline conditions?
Q66.
mcq single
+4 / 1
The oxidation of benzene by V~2~O~5~ in the presence of air produces
Q67.
mcq single
+4 / 1
Reaction of phenol with chloroform in presence of dilute sodium hydroxide finally introduces which one of the following functional group?
Q68.
mcq single
+4 / 1
Which of the following reaction(s) can be used for the preparation of alkyl halides?
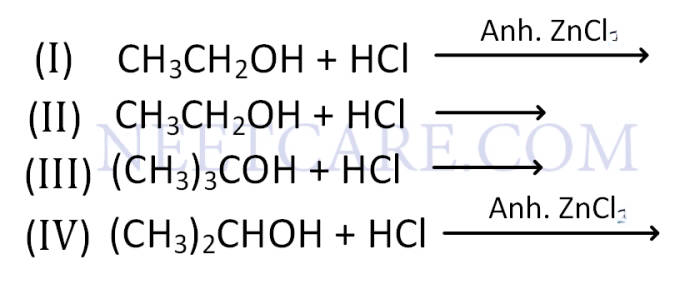

Q69.
mcq single
+4 / 1
2,3-Dimethyl-2-butene can be prepared by heating which of the following compounds with a strong acid?
Q70.
mcq single
+4 / 1
Which of the following is not the product of dehydration of


Q71.
mcq single
+4 / 1
In the reaction with HCl, an alkene reacts in accordance with the Markovnikov's rule to give a product 1-chloro-1 methylcyclohexane. The possible alkene is
Q72.
mcq single
+4 / 1
Aqueous solution of which of the following compounds is the best conductor of electric current ?
Q73.
mcq single
+4 / 1
The number of structural isomers possible from the molecular formula C~3~H~9~N is
Q74.
mcq single
+4 / 1
The following reaction
 is known by the name
is known by the name
 is known by the name
is known by the name
Q75.
mcq single
+4 / 1
Method by which aniline cannot be prepared is
Q76.
mcq single
+4 / 1
Which is the correct order of increasing energy of the listed orbitals in the atom of titanium ? (At. no. Z = 22)
Physics
Physics
Q1.
mcq single
+4 / 1
At the first minimum adjacent to the central maximum of a single-slit diffraction pattern, the phase difference between the Huygen's wavelet from the edge of the slit and the wavelet from the midpoint of the slit is
Q2.
mcq single
+4 / 1
Two slits in Young's experiment have widths in the ratio 1 : 25. The ratio of intensity at the maxima and minima in the interference pattern, $${{{I_{max}}} \over {{I_{min}}}}$$ is
Q3.
mcq single
+4 / 1
An automobile moves on a road with a speed of 54 km h^($$-$$1). The radius of its wheels is 0.45 m and the moment of inertia of the wheel about its axis of rotation is 3 kg m^(2). If the vehicle is brought to rest in 15 s, the magnitude of average torque transmitted by its brakes to the wheel is
Q4.
mcq single
+4 / 1
Point masses m~1~ and m~2~ are placed at the opposite ends of a rigid rod of length L, and negligible mass. The rod is to be set rotating about an axis perpendicular to it. The position of point P on this rod through which the axis should pass so that the work required to set the rod rotating with angular velocity $$\omega $$~0~ is minimum, is given by


Q5.
mcq single
+4 / 1
A force $$\overrightarrow F = \alpha \widehat i + 3\widehat j + 6\widehat k$$ is acting at a point $$\overrightarrow r = 2\widehat i - 6\widehat j - 12\widehat k$$. The value of $$\alpha $$ for which angular momentum about origin is conserved is
Q6.
mcq single
+4 / 1
An electron moves on a straight line path XY as shown. The abcd is a coil adjacent to the path of electron. What will be the direction of current, if any, induced in the coil ?


Q7.
mcq single
+4 / 1
If dimensions of critical velocity $$\upsilon $$~c~ of a liquid flowing through a tube are expressed as $$\left[ {{\eta ^x}{\rho ^y}{r^z}} \right]$$ where $$\eta ,\rho $$ and r are the coefficient of viscosity of liquid, density of liquid and radius of the tube respectively, then the values of x, y and z are given by
Q8.
mcq single
+4 / 1
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas ?
Q9.
mcq single
+4 / 1
Two vessels separately contain two ideal gases A and B at the same temperature, the pressure of A being twice that of B. Under such conditions, the density of A is found to be 1.5 times the density of B. The ratio of molecular weight of A and B is
Q10.
mcq single
+4 / 1
The coefficient of performance of a refrigerator is 5. If the temperature inside freezer is $$-$$20^(o)C, the temperature of the surroundings to which it rejects heat is
Q11.
mcq single
+4 / 1
The value of coefficient of volume expansion of glycerin is 5 $$ \times $$ 10^($$-$$4) K^($$-$$1). The fractional change in the density of glycerin for a rise of 40^(o)C in its temperature, is
Q12.
mcq single
+4 / 1
Water rises to a height h in capillary tube. If the length of capillary tube above the surface of water is made less than h, then
Q13.
mcq single
+4 / 1
The cylindrical tube of a spray pump has radius R, one end of which has n fine holes, each of radius r. If the speed of the liquid in the tube is V, the speed of the ejection of the liquid through the holes is
Q14.
mcq single
+4 / 1
The Young's modulus of steel is twice that of brass. Two wires of same length and of same area of cross section, one of steel and another of brass are suspended from the same roof. If we want the lower ends of the wires to be at the same level, then the weights added to the steel and brass wires must be in the ratio of
Q15.
mcq single
+4 / 1
If potential (in volts) in a region is expressed as V(x, y, z) = 6xy $$-$$ y + 2yz, the electric field (in N/C) at point (1, 1, 0) is
Q16.
mcq single
+4 / 1
A remote-sensing satellite of earth revolves in a circular orbit at a height of 0.25 $$ \times $$ 10^(6) m above the surface of earth. If earth's radius is 6.38 $$ \times $$ 10^(6) m and g = 9.8 ms^($$-$$2), then the orbital speed of the satellite is
Q17.
mcq single
+4 / 1
A satellite S is moving in an elliptical orbit around the earth. The mass of the satellite is very small compared to the mass of the earth. Then,
Q18.
mcq single
+4 / 1
A parallel plate air capacitor has capacity C, distance of separation between plates is d and potential difference $$V$$ is applied between the plates. Force of attraction between the plates of the parallel plate air capacitor is
Q19.
mcq single
+4 / 1
A beam of light consisting of red, green and blue colours is incifent on a right angled prism. The refractive index of the material of the prism for the above red, green and blue wavelengths are 1.39, 1.44 and 1.47 respectively.
 The prism will
The prism will
 The prism will
The prism will
Q20.
mcq single
+4 / 1
In an astronomical telescope in normal adjustment a straight black line of length L is drawn on inside part of objective lens. The eye-piece forms a real image of this line. The length of this image is $$I$$. The magnification of the telescope is
Q21.
mcq single
+4 / 1
A particle is executing a simple harmonic motion. Its maximum acceleration is $$\alpha $$ and maximum velocity is $$\beta $$. Them, its time period of vibration will be
Q22.
mcq single
+4 / 1
In the given figure, a diode D is connected to an external resistance R = 100 $$\Omega $$ and an e.m.f. of 3.5 V. If the barrier potential developed across the diode is 0.5 V, the current in the citcuit will be


Q23.
mcq single
+4 / 1
A proton and an alpha particle both enter a region of uniform magnetic field B, moving at right angles to the field B. If the radius of circular orbits for both the particles is equal and the kinetic energy acquired by proton is 1 MeV, the energy acquired by the alpha particle will be
Q24.
mcq single
+4 / 1
A photoelectric surface is illuminated successively by monochromatic light of wavelength $$\lambda $$ and If the maximum kinetic energy of the emitted photoelectrons in the second case is 3 times that in the first case, the work function of the surface of the material is
(h = Planck's constant, C = speed of light)
Q25.
mcq single
+4 / 1
Light of wavelength 500 nm is incifent on a metal with work function 2.28 eV. The de Broglie wavelength of the emitted electron is
Q26.
mcq single
+4 / 1
In the spectrum of hydrogen, the ratio of the longest wavelength in the Lyman series to the longest wavelength in the Balmer series is
Q27.
mcq single
+4 / 1
A nucleus of uranium decays at rest into nuclei of thorium and helium. Then
Q28.
mcq single
+4 / 1
If vectors $$\overrightarrow A = \cos \omega t\widehat i + \sin \omega t\widehat j$$ and $$\overrightarrow B = \cos {{\omega t} \over 2}\widehat i + \sin {{\omega t} \over 2}\widehat j$$ are functions of time, then the value of t at which they are orthogonal to each other is
Q29.
mcq single
+4 / 1
The positions vector of a particle $$\overrightarrow R $$ as a function of time is given by $$\overrightarrow R $$ = 4sin(2$$\pi $$t)$$\widehat i$$ + 4cos(2$$\pi $$t)$$\widehat j$$. Where R is in meters, t is in seconds and $$\widehat i$$ and $$\widehat j$$ denote unit vectors along x-and y-directions, respectively. Which one of the following statements is wrong for the motion of particle?
Q30.
mcq single
+4 / 1
4.0 g of a gas occupies 22.4 litres at NTP. The specific heat capacity of the gas at constant volume is 5.0 J K^($$-$$1) mol^($$-$$1). If the speed of sound in this gas at NTP is 952 m s^($$-$$1), then the heat capacity at constant pressure is
(Take gas constant R $$=$$ 8.3 J K^($$-$$1) mol^($$-$$1))
Q31.
mcq single
+4 / 1
A string is stretched between fixed points separated by 75.0 cm. It is observed to have resonant frequencies of 420 Hz and 315 Hz. There are no other resonant frequencies between these two. The lowest resonant frequency for this string is
Q32.
mcq single
+4 / 1
The fundamental frequency of a closed organ pipe of length 20 cm is equal to the second overtone of an organ pipe open at both the ends. The length of organ pipe open at both the ends is
Q33.
mcq single
+4 / 1
A plank with a box on it at one end is gradually raised about the other end. As the angle of inclination with the horizontal reaches 30^(o), the box starts to slip and slides 4.0 m down the plank in 4.0 s.
 The coefficients of static and kinetic friction between the box and the plank will be, respectively
The coefficients of static and kinetic friction between the box and the plank will be, respectively
 The coefficients of static and kinetic friction between the box and the plank will be, respectively
The coefficients of static and kinetic friction between the box and the plank will be, respectively
Q34.
mcq single
+4 / 1
Two stones of masses m and 2m are whirled in horizontal circles, the heavier one in a radius $${r \over 2}$$ and the lighter one in radius r. The tangential speed of lighter stone is n times that of the value of heavier stone when they exprience same centripetal forces. The value of n is
Q35.
mcq single
+4 / 1
A rectangular coil of length 0.12 m and width 0.1 m having 50 turns of wire is suspended vertically in a uniform magnetic field of strength 0.2 weber/m^(2). The coil carries a current of 2 A. If the plane of the coil is inclined at an angle of 30^(o) with the direction of the field, the torque required to keep the coil in stable equilibrium will be
Q36.
mcq single
+4 / 1
Two particles A and B, move with constant velocities $$\overrightarrow {{v_1}} $$ and $$\overrightarrow {{v_2}} $$. At the initial moment their position vectors are $$\overrightarrow {{r_1}} $$ and $$\overrightarrow {{r_2}} $$ respectively. The condition for particles A and B for their collision is
Q37.
mcq single
+4 / 1
On a frictionless surface, a block of mass M moving at speed v collides elastically with another block of same mass M which is initially at rest. After collision the first block moves at an angle $$\theta $$ to its initial direction and has a speed $${v \over 3}.$$ The second block's speed after the collision is
Q38.
mcq single
+4 / 1
The heart of a man pumps 5 litres of blood through the arteries per minute at a pressure of 150 mm of mercury. If the density of mercury be 13.6 $$ \times $$ 10^(3) kg/m^(3) and g = 10 m/s^(2) then the power (in watt) is
Q39.
mcq single
+4 / 1
A ball is thrown vertically downwards from a height of 20 m with an initial velocity v~0~. It collides with the ground, losses 50 percent of its energy in collision and rebounds to the same height. The initial velocity v~0~ is
(Take g = 10 m s^($$-$$2))
Q40.
mcq single
+4 / 1
Two metal wires of identical dimensions are connected in series. If $$\sigma $$~1~ and $$\sigma $$~2~ are the conductivity of the combination is
Q41.
mcq single
+4 / 1
A potentiometer wire of length L and a resistance r are connected in series with a battery of e.m.f. E~0~ and a resistance r~1~. An unknown e.m.f. E is balanced at a length $$l$$ of the potentiometer wire. The e.m.f. E will be given by
Q42.
mcq single
+4 / 1
A circuit contains an ammeter, a battery of 30 V and a resistance 40.8 ohm all connected in series. If the ammeter has a coil of resistance 480 ohm and a shunt of 20 ohm, the reading in the ammeter will be
Q43.
mcq single
+4 / 1
The energy of the em waves is of the order of 15 keV. To which part of the spectrum does it belong ?
Q44.
mcq single
+4 / 1
A series R-C circuit is connected to an alternating voltage source. Consider two situations :
(a) When capacitor is air filled.
(b) When capacitor is mica filled.
Current through resistor is $$I$$ and voltage across capacitor is $$V$$ then
Q45.
mcq single
+4 / 1
At the first minimum adjacent to the central maximum of a single-slit diffraction pattern, the phase difference between the Huygen's wavelet from the edge of the slit and the wavelet from the midpoint of the slit is
Q46.
mcq single
+4 / 1
Two slits in Young's experiment have widths in the ratio 1 : 25. The ratio of intensity at the maxima and minima in the interference pattern, $${{{I_{max}}} \over {{I_{min}}}}$$ is
Q47.
mcq single
+4 / 1
An automobile moves on a road with a speed of 54 km h^($$-$$1). The radius of its wheels is 0.45 m and the moment of inertia of the wheel about its axis of rotation is 3 kg m^(2). If the vehicle is brought to rest in 15 s, the magnitude of average torque transmitted by its brakes to the wheel is
Q48.
mcq single
+4 / 1
Point masses m~1~ and m~2~ are placed at the opposite ends of a rigid rod of length L, and negligible mass. The rod is to be set rotating about an axis perpendicular to it. The position of point P on this rod through which the axis should pass so that the work required to set the rod rotating with angular velocity $$\omega $$~0~ is minimum, is given by


Q49.
mcq single
+4 / 1
A force $$\overrightarrow F = \alpha \widehat i + 3\widehat j + 6\widehat k$$ is acting at a point $$\overrightarrow r = 2\widehat i - 6\widehat j - 12\widehat k$$. The value of $$\alpha $$ for which angular momentum about origin is conserved is
Q50.
mcq single
+4 / 1
An electron moves on a straight line path XY as shown. The abcd is a coil adjacent to the path of electron. What will be the direction of current, if any, induced in the coil ?
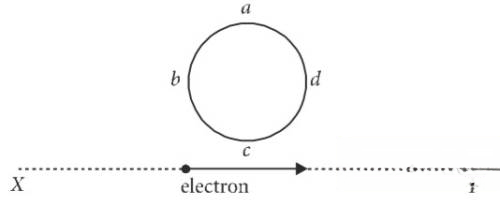

Q51.
mcq single
+4 / 1
If dimensions of critical velocity $$\upsilon $$~c~ of a liquid flowing through a tube are expressed as $$\left[ {{\eta ^x}{\rho ^y}{r^z}} \right]$$ where $$\eta ,\rho $$ and r are the coefficient of viscosity of liquid, density of liquid and radius of the tube respectively, then the values of x, y and z are given by
Q52.
mcq single
+4 / 1
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas ?
Q53.
mcq single
+4 / 1
Two vessels separately contain two ideal gases A and B at the same temperature, the pressure of A being twice that of B. Under such conditions, the density of A is found to be 1.5 times the density of B. The ratio of molecular weight of A and B is
Q54.
mcq single
+4 / 1
The coefficient of performance of a refrigerator is 5. If the temperature inside freezer is $$-$$20^(o)C, the temperature of the surroundings to which it rejects heat is
Q55.
mcq single
+4 / 1
The value of coefficient of volume expansion of glycerin is 5 $$ \times $$ 10^($$-$$4) K^($$-$$1). The fractional change in the density of glycerin for a rise of 40^(o)C in its temperature, is
Q56.
mcq single
+4 / 1
Water rises to a height h in capillary tube. If the length of capillary tube above the surface of water is made less than h, then
Q57.
mcq single
+4 / 1
The cylindrical tube of a spray pump has radius R, one end of which has n fine holes, each of radius r. If the speed of the liquid in the tube is V, the speed of the ejection of the liquid through the holes is
Q58.
mcq single
+4 / 1
The Young's modulus of steel is twice that of brass. Two wires of same length and of same area of cross section, one of steel and another of brass are suspended from the same roof. If we want the lower ends of the wires to be at the same level, then the weights added to the steel and brass wires must be in the ratio of
Q59.
mcq single
+4 / 1
If potential (in volts) in a region is expressed as V(x, y, z) = 6xy $$-$$ y + 2yz, the electric field (in N/C) at point (1, 1, 0) is
Q60.
mcq single
+4 / 1
A remote-sensing satellite of earth revolves in a circular orbit at a height of 0.25 $$ \times $$ 10^(6) m above the surface of earth. If earth's radius is 6.38 $$ \times $$ 10^(6) m and g = 9.8 ms^($$-$$2), then the orbital speed of the satellite is
Q61.
mcq single
+4 / 1
A satellite S is moving in an elliptical orbit around the earth. The mass of the satellite is very small compared to the mass of the earth. Then,
Q62.
mcq single
+4 / 1
A parallel plate air capacitor has capacity C, distance of separation between plates is d and potential difference $$V$$ is applied between the plates. Force of attraction between the plates of the parallel plate air capacitor is
Q63.
mcq single
+4 / 1
A beam of light consisting of red, green and blue colours is incifent on a right angled prism. The refractive index of the material of the prism for the above red, green and blue wavelengths are 1.39, 1.44 and 1.47 respectively.
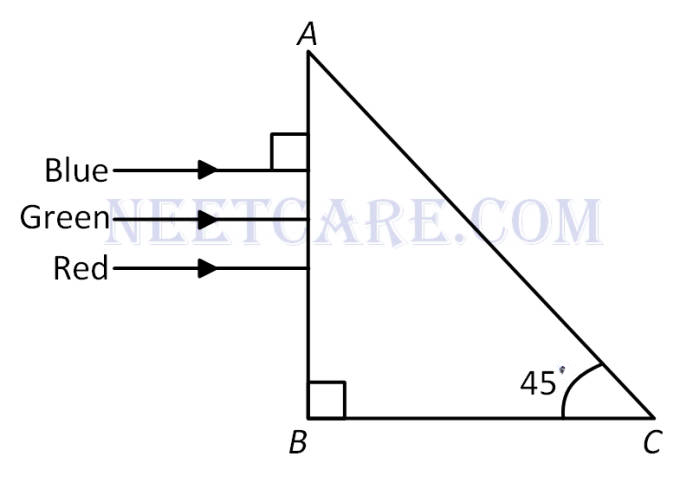 The prism will
The prism will
 The prism will
The prism will
Q64.
mcq single
+4 / 1
In an astronomical telescope in normal adjustment a straight black line of length L is drawn on inside part of objective lens. The eye-piece forms a real image of this line. The length of this image is $$I$$. The magnification of the telescope is
Q65.
mcq single
+4 / 1
A particle is executing a simple harmonic motion. Its maximum acceleration is $$\alpha $$ and maximum velocity is $$\beta $$. Them, its time period of vibration will be
Q66.
mcq single
+4 / 1
In the given figure, a diode D is connected to an external resistance R = 100 $$\Omega $$ and an e.m.f. of 3.5 V. If the barrier potential developed across the diode is 0.5 V, the current in the citcuit will be


Q67.
mcq single
+4 / 1
A proton and an alpha particle both enter a region of uniform magnetic field B, moving at right angles to the field B. If the radius of circular orbits for both the particles is equal and the kinetic energy acquired by proton is 1 MeV, the energy acquired by the alpha particle will be
Q68.
mcq single
+4 / 1
A photoelectric surface is illuminated successively by monochromatic light of wavelength $$\lambda $$ and If the maximum kinetic energy of the emitted photoelectrons in the second case is 3 times that in the first case, the work function of the surface of the material is
(h = Planck's constant, C = speed of light)
Q69.
mcq single
+4 / 1
Light of wavelength 500 nm is incifent on a metal with work function 2.28 eV. The de Broglie wavelength of the emitted electron is
Q70.
mcq single
+4 / 1
In the spectrum of hydrogen, the ratio of the longest wavelength in the Lyman series to the longest wavelength in the Balmer series is
Q71.
mcq single
+4 / 1
A nucleus of uranium decays at rest into nuclei of thorium and helium. Then
Q72.
mcq single
+4 / 1
If vectors $$\overrightarrow A = \cos \omega t\widehat i + \sin \omega t\widehat j$$ and $$\overrightarrow B = \cos {{\omega t} \over 2}\widehat i + \sin {{\omega t} \over 2}\widehat j$$ are functions of time, then the value of t at which they are orthogonal to each other is
Q73.
mcq single
+4 / 1
The positions vector of a particle $$\overrightarrow R $$ as a function of time is given by $$\overrightarrow R $$ = 4sin(2$$\pi $$t)$$\widehat i$$ + 4cos(2$$\pi $$t)$$\widehat j$$. Where R is in meters, t is in seconds and $$\widehat i$$ and $$\widehat j$$ denote unit vectors along x-and y-directions, respectively. Which one of the following statements is wrong for the motion of particle?
Q74.
mcq single
+4 / 1
4.0 g of a gas occupies 22.4 litres at NTP. The specific heat capacity of the gas at constant volume is 5.0 J K^($$-$$1) mol^($$-$$1). If the speed of sound in this gas at NTP is 952 m s^($$-$$1), then the heat capacity at constant pressure is
(Take gas constant R $$=$$ 8.3 J K^($$-$$1) mol^($$-$$1))
Q75.
mcq single
+4 / 1
A string is stretched between fixed points separated by 75.0 cm. It is observed to have resonant frequencies of 420 Hz and 315 Hz. There are no other resonant frequencies between these two. The lowest resonant frequency for this string is
Q76.
mcq single
+4 / 1
The fundamental frequency of a closed organ pipe of length 20 cm is equal to the second overtone of an organ pipe open at both the ends. The length of organ pipe open at both the ends is
Q77.
mcq single
+4 / 1
A plank with a box on it at one end is gradually raised about the other end. As the angle of inclination with the horizontal reaches 30^(o), the box starts to slip and slides 4.0 m down the plank in 4.0 s.
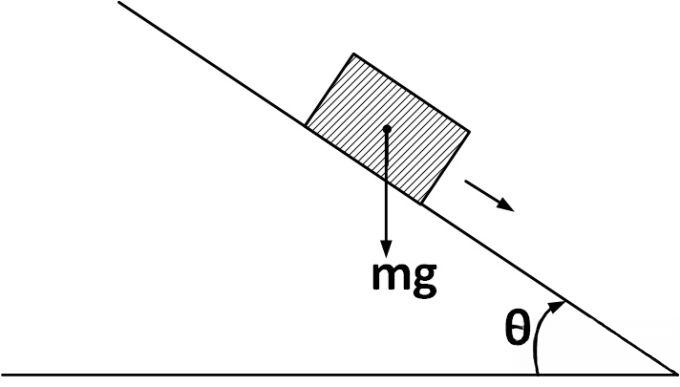 The coefficients of static and kinetic friction between the box and the plank will be, respectively
The coefficients of static and kinetic friction between the box and the plank will be, respectively
 The coefficients of static and kinetic friction between the box and the plank will be, respectively
The coefficients of static and kinetic friction between the box and the plank will be, respectively
Q78.
mcq single
+4 / 1
Two stones of masses m and 2m are whirled in horizontal circles, the heavier one in a radius $${r \over 2}$$ and the lighter one in radius r. The tangential speed of lighter stone is n times that of the value of heavier stone when they exprience same centripetal forces. The value of n is
Q79.
mcq single
+4 / 1
A rectangular coil of length 0.12 m and width 0.1 m having 50 turns of wire is suspended vertically in a uniform magnetic field of strength 0.2 weber/m^(2). The coil carries a current of 2 A. If the plane of the coil is inclined at an angle of 30^(o) with the direction of the field, the torque required to keep the coil in stable equilibrium will be
Q80.
mcq single
+4 / 1
Two particles A and B, move with constant velocities $$\overrightarrow {{v_1}} $$ and $$\overrightarrow {{v_2}} $$. At the initial moment their position vectors are $$\overrightarrow {{r_1}} $$ and $$\overrightarrow {{r_2}} $$ respectively. The condition for particles A and B for their collision is
Q81.
mcq single
+4 / 1
On a frictionless surface, a block of mass M moving at speed v collides elastically with another block of same mass M which is initially at rest. After collision the first block moves at an angle $$\theta $$ to its initial direction and has a speed $${v \over 3}.$$ The second block's speed after the collision is
Q82.
mcq single
+4 / 1
The heart of a man pumps 5 litres of blood through the arteries per minute at a pressure of 150 mm of mercury. If the density of mercury be 13.6 $$ \times $$ 10^(3) kg/m^(3) and g = 10 m/s^(2) then the power (in watt) is
Q83.
mcq single
+4 / 1
A ball is thrown vertically downwards from a height of 20 m with an initial velocity v~0~. It collides with the ground, losses 50 percent of its energy in collision and rebounds to the same height. The initial velocity v~0~ is
(Take g = 10 m s^($$-$$2))
Q84.
mcq single
+4 / 1
Two metal wires of identical dimensions are connected in series. If $$\sigma $$~1~ and $$\sigma $$~2~ are the conductivity of the combination is
Q85.
mcq single
+4 / 1
A potentiometer wire of length L and a resistance r are connected in series with a battery of e.m.f. E~0~ and a resistance r~1~. An unknown e.m.f. E is balanced at a length $$l$$ of the potentiometer wire. The e.m.f. E will be given by
Q86.
mcq single
+4 / 1
A circuit contains an ammeter, a battery of 30 V and a resistance 40.8 ohm all connected in series. If the ammeter has a coil of resistance 480 ohm and a shunt of 20 ohm, the reading in the ammeter will be
Q87.
mcq single
+4 / 1
The energy of the em waves is of the order of 15 keV. To which part of the spectrum does it belong ?
Q88.
mcq single
+4 / 1
A series R-C circuit is connected to an alternating voltage source. Consider two situations :
(a) When capacitor is air filled.
(b) When capacitor is mica filled.
Current through resistor is $$I$$ and voltage across capacitor is $$V$$ then
Biology
Biology
Q1.
mcq single
+4 / 1
Match the following list of microbes and
their importance -
Column I
Column II
(a) Sacharomyces cerevisiae
(i) Production of immunosuppressive agent
(b) Monascus purpureus
(ii) Ripening of Swiss cheese
(c) Trichoderma polysporum
(iii) Commercial production of ethanol
(d) Propionibacterium sharmanii
(iv) Production of blood cholesterol lowering agents
Q2.
mcq single
+4 / 1
During ecological succession :
Q3.
mcq single
+4 / 1
In which of the following both pairs have
correct combination?
Q4.
mcq single
+4 / 1
Most animals that live in deep oceanic
waters are -
Q5.
mcq single
+4 / 1
The function of the gap junction is to -
Q6.
mcq single
+4 / 1
The body cell in cockroach discharge their
nitrogenous waste in the haemolymph
mainly in the form of -
Q7.
mcq single
+4 / 1
Chromatophores take part in
Q8.
mcq single
+4 / 1
In photosynthesis, the light-independent reactions take place at
Q9.
mcq single
+4 / 1
Metagenesis refers to
Q10.
mcq single
+4 / 1
A jawless fish, which lays eggs in fresh water and whose ammocoetes larvae after metamorphosis return to the ocean is
Q11.
mcq single
+4 / 1
Body having meshwork of cells, internal cavities lined with food filtering flagellated cells and indirect development are the characteristics of Phylum
Q12.
mcq single
+4 / 1
Which one is a wrong statement?
Q13.
mcq single
+4 / 1
Name the pulmonary disease in which alveolar surface area involved in gas exchage is drastically reduced due to damage in the alveolar walls.
Q14.
mcq single
+4 / 1
The introduction of t-DNA into plants
involves -
Q15.
mcq single
+4 / 1
Golden rice is a genetically modified crop
plant where the incorporated gene is meant
for biosynthesis of -
Q16.
mcq single
+4 / 1
In mammalian eye, the 'fovea' is the center of the visual field, where
Q17.
mcq single
+4 / 1
Destruction of the anterior horn cells of the spinal cord would result in loss of
Q18.
mcq single
+4 / 1
Read the different components from (1) to
(4) in the list given below and tell the
correct order of the components with
reference to their arrangement from outer
side to inner side in a woody dicot stem :
(1) Secondary cortex
(2) Wood
(3) Secondary phoem
(4) Phellem
The correct order is :
Q19.
mcq single
+4 / 1
Which of the following structures is not
found in a prokaryotic cell?
Q20.
mcq single
+4 / 1
Cellular organelles with membranes are -
Q21.
mcq single
+4 / 1
Match the columns and identify the correct
option.
Column I
Column II
(a) Thylakoids
(i) Disc-shaped sacs in Golgi
apparatus
(b) Cristae
(ii) Condensed structure of DNA
(c) Cisternae
(iii) Flat membranous sacs in
stroma
(d) Chromatin
(iv) Infoldings in mitochondria
Q22.
mcq single
+4 / 1
Which of the following are not membranebound ?
Q23.
mcq single
+4 / 1
Balbiani rings are sites of :
Q24.
mcq single
+4 / 1
Which one of the following is not
applicable to RNA?
Q25.
mcq single
+4 / 1
Satellite DNA is important because it :
Q26.
mcq single
+4 / 1
Identify the correct order of organisation of
genetic material from largest to smallest :
Q27.
mcq single
+4 / 1
If you suspect major deficiency of
antibodies in a person, to which of the
following would you look for confirmatory
evidence ?
Q28.
mcq single
+4 / 1
Which of the following diseases is caused
by a protozoan?
Q29.
mcq single
+4 / 1
Which of the following immunoglobulins
does constitute the largest percentage in
human milk?
Q30.
mcq single
+4 / 1
Grafted kidney may be rejected in a patient
due to -
Q31.
mcq single
+4 / 1
An association of individuals of different
species living in the same habitat and
having functional interactions is -
Q32.
mcq single
+4 / 1
In which of the following interactions both
partners are adversely affected?
Q33.
mcq single
+4 / 1
Doctors use stethoscope to hear the sounds produced during each cardiac cycle. The second sound is heard when
Q34.
mcq single
+4 / 1
Which one of the following animals has two separate circulatory pathways?
Q35.
mcq single
+4 / 1
Arrange the following events of meiosis in correct sequence
(i) Crossing over
(ii) Synapsis
(iii) Terminalisation of chiasmata
(iv) Disappearance of nucleolus
Q36.
mcq single
+4 / 1
In human females, meiosis-II is not completed until
Q37.
mcq single
+4 / 1
Which of the following layers in an antral follicle is acellular?
Q38.
mcq single
+4 / 1
Ectopic pregnancies are referred to as
Q39.
mcq single
+4 / 1
Which of the following events is not associated with ovulation in human life?
Q40.
mcq single
+4 / 1
Which one of the following hormones is not involved in sugar metabolism?
Q41.
mcq single
+4 / 1
Which one of the following hormones though synthesised elsewhere, is stored and released by the master gland?
Q42.
mcq single
+4 / 1
The species confined to a particular region
and not found elsewhere is termed as -
Q43.
mcq single
+4 / 1
Auxin can be bioassayed by
Q44.
mcq single
+4 / 1
Which of the following joints would allow no movements?
Q45.
mcq single
+4 / 1
Which of the following is not a function of the skeletal system?
Q46.
mcq single
+4 / 1
Choose the wrong statement.
Q47.
mcq single
+4 / 1
Cell wall is absent in
Q48.
mcq single
+4 / 1
The structures that help some bacteria to attach to rocks and/or host tissues are
Q49.
mcq single
+4 / 1
Select the wrong statement.
Q50.
mcq single
+4 / 1
The imperfect fungi which are decomposers
of litter and help in mineral cycling belong to
Q51.
mcq single
+4 / 1
In which group of organisms the cell walls form two thin overlapping shells which fit together?
Q52.
mcq single
+4 / 1
Pick up the wrong statement.
Q53.
mcq single
+4 / 1
Human urine is usually acidic because
Q54.
mcq single
+4 / 1
A childless couple can be assisted to have a
child through a technique called GIFT. The
full form of this technique is :
Q55.
mcq single
+4 / 1
Filiform apparatus is characteristic feature of
Q56.
mcq single
+4 / 1
Male gametophyte in angiosperms produces
Q57.
mcq single
+4 / 1
Flowers are unisexual in
Q58.
mcq single
+4 / 1
Coconut water from a tender coconut is
Q59.
mcq single
+4 / 1
In angiosperms, microsporogenesis and mega-sporogenesis
Q60.
mcq single
+4 / 1
Which one of the following fruits is parthenocarpic?
Q61.
mcq single
+4 / 1
The wheat grain has an embryo with one large, shield shaped cotyledon known as
Q62.
mcq single
+4 / 1
Among China rose, mustard, brinjal, potato, guava, cucumber, onion and tulip, how many plants have superior ovary?
Q63.
mcq single
+4 / 1
Axile placentation is present in
Q64.
mcq single
+4 / 1
Roots play insignificant role in absorption of water in
Q65.
mcq single
+4 / 1
Which of the following biomolecules does have a phosphodiester bond?
Q66.
mcq single
+4 / 1
The chitinous exoskeleton of arthropods is formed by the polymerisation of
Q67.
mcq single
+4 / 1
The cutting of DNA at specific locations
became possible with the discovery of -
Q68.
mcq single
+4 / 1
The DNA molecule to which the gene of
interest is integrated for cloning is called -
Q69.
mcq single
+4 / 1
Match the following list of microbes and
their importance -
Column I
Column II
(a) Sacharomyces cerevisiae
(i) Production of immunosuppressive agent
(b) Monascus purpureus
(ii) Ripening of Swiss cheese
(c) Trichoderma polysporum
(iii) Commercial production of ethanol
(d) Propionibacterium sharmanii
(iv) Production of blood cholesterol lowering agents
Q70.
mcq single
+4 / 1
During ecological succession :
Q71.
mcq single
+4 / 1
In which of the following both pairs have
correct combination?
Q72.
mcq single
+4 / 1
Most animals that live in deep oceanic
waters are -
Q73.
mcq single
+4 / 1
The function of the gap junction is to -
Q74.
mcq single
+4 / 1
The body cell in cockroach discharge their
nitrogenous waste in the haemolymph
mainly in the form of -
Q75.
mcq single
+4 / 1
Chromatophores take part in
Q76.
mcq single
+4 / 1
In photosynthesis, the light-independent reactions take place at
Q77.
mcq single
+4 / 1
Metagenesis refers to
Q78.
mcq single
+4 / 1
A jawless fish, which lays eggs in fresh water and whose ammocoetes larvae after metamorphosis return to the ocean is
Q79.
mcq single
+4 / 1
Body having meshwork of cells, internal cavities lined with food filtering flagellated cells and indirect development are the characteristics of Phylum
Q80.
mcq single
+4 / 1
Which one is a wrong statement?
Q81.
mcq single
+4 / 1
Name the pulmonary disease in which alveolar surface area involved in gas exchage is drastically reduced due to damage in the alveolar walls.
Q82.
mcq single
+4 / 1
The introduction of t-DNA into plants
involves -
Q83.
mcq single
+4 / 1
Golden rice is a genetically modified crop
plant where the incorporated gene is meant
for biosynthesis of -
Q84.
mcq single
+4 / 1
In mammalian eye, the 'fovea' is the center of the visual field, where
Q85.
mcq single
+4 / 1
Destruction of the anterior horn cells of the spinal cord would result in loss of
Q86.
mcq single
+4 / 1
Read the different components from (1) to
(4) in the list given below and tell the
correct order of the components with
reference to their arrangement from outer
side to inner side in a woody dicot stem :
(1) Secondary cortex
(2) Wood
(3) Secondary phoem
(4) Phellem
The correct order is :
Q87.
mcq single
+4 / 1
Which of the following structures is not
found in a prokaryotic cell?
Q88.
mcq single
+4 / 1
Cellular organelles with membranes are -
Q89.
mcq single
+4 / 1
Match the columns and identify the correct
option.
Column I
Column II
(a) Thylakoids
(i) Disc-shaped sacs in Golgi
apparatus
(b) Cristae
(ii) Condensed structure of DNA
(c) Cisternae
(iii) Flat membranous sacs in
stroma
(d) Chromatin
(iv) Infoldings in mitochondria
Q90.
mcq single
+4 / 1
Which of the following are not membranebound ?
Q91.
mcq single
+4 / 1
Balbiani rings are sites of :
Q92.
mcq single
+4 / 1
Which one of the following is not
applicable to RNA?
Q93.
mcq single
+4 / 1
Satellite DNA is important because it :
Q94.
mcq single
+4 / 1
Identify the correct order of organisation of
genetic material from largest to smallest :
Q95.
mcq single
+4 / 1
If you suspect major deficiency of
antibodies in a person, to which of the
following would you look for confirmatory
evidence ?
Q96.
mcq single
+4 / 1
Which of the following diseases is caused
by a protozoan?
Q97.
mcq single
+4 / 1
Which of the following immunoglobulins
does constitute the largest percentage in
human milk?
Q98.
mcq single
+4 / 1
Grafted kidney may be rejected in a patient
due to -
Q99.
mcq single
+4 / 1
An association of individuals of different
species living in the same habitat and
having functional interactions is -
Q100.
mcq single
+4 / 1
In which of the following interactions both
partners are adversely affected?
Q101.
mcq single
+4 / 1
Doctors use stethoscope to hear the sounds produced during each cardiac cycle. The second sound is heard when
Q102.
mcq single
+4 / 1
Which one of the following animals has two separate circulatory pathways?
Q103.
mcq single
+4 / 1
Arrange the following events of meiosis in correct sequence
(i) Crossing over
(ii) Synapsis
(iii) Terminalisation of chiasmata
(iv) Disappearance of nucleolus
Q104.
mcq single
+4 / 1
In human females, meiosis-II is not completed until
Q105.
mcq single
+4 / 1
Which of the following layers in an antral follicle is acellular?
Q106.
mcq single
+4 / 1
Ectopic pregnancies are referred to as
Q107.
mcq single
+4 / 1
Which of the following events is not associated with ovulation in human life?
Q108.
mcq single
+4 / 1
Which one of the following hormones is not involved in sugar metabolism?
Q109.
mcq single
+4 / 1
Which one of the following hormones though synthesised elsewhere, is stored and released by the master gland?
Q110.
mcq single
+4 / 1
The species confined to a particular region
and not found elsewhere is termed as -
Q111.
mcq single
+4 / 1
Auxin can be bioassayed by
Q112.
mcq single
+4 / 1
Which of the following joints would allow no movements?
Q113.
mcq single
+4 / 1
Which of the following is not a function of the skeletal system?
Q114.
mcq single
+4 / 1
Choose the wrong statement.
Q115.
mcq single
+4 / 1
Cell wall is absent in
Q116.
mcq single
+4 / 1
The structures that help some bacteria to attach to rocks and/or host tissues are
Q117.
mcq single
+4 / 1
Select the wrong statement.
Q118.
mcq single
+4 / 1
The imperfect fungi which are decomposers
of litter and help in mineral cycling belong to
Q119.
mcq single
+4 / 1
In which group of organisms the cell walls form two thin overlapping shells which fit together?
Q120.
mcq single
+4 / 1
Pick up the wrong statement.
Q121.
mcq single
+4 / 1
Human urine is usually acidic because
Q122.
mcq single
+4 / 1
A childless couple can be assisted to have a
child through a technique called GIFT. The
full form of this technique is :
Q123.
mcq single
+4 / 1
Filiform apparatus is characteristic feature of
Q124.
mcq single
+4 / 1
Male gametophyte in angiosperms produces
Q125.
mcq single
+4 / 1
Flowers are unisexual in
Q126.
mcq single
+4 / 1
Coconut water from a tender coconut is
Q127.
mcq single
+4 / 1
In angiosperms, microsporogenesis and mega-sporogenesis
Q128.
mcq single
+4 / 1
Which one of the following fruits is parthenocarpic?
Q129.
mcq single
+4 / 1
The wheat grain has an embryo with one large, shield shaped cotyledon known as
Q130.
mcq single
+4 / 1
Among China rose, mustard, brinjal, potato, guava, cucumber, onion and tulip, how many plants have superior ovary?
Q131.
mcq single
+4 / 1
Axile placentation is present in
Q132.
mcq single
+4 / 1
Roots play insignificant role in absorption of water in
Q133.
mcq single
+4 / 1
Which of the following biomolecules does have a phosphodiester bond?
Q134.
mcq single
+4 / 1
The chitinous exoskeleton of arthropods is formed by the polymerisation of
Q135.
mcq single
+4 / 1
The cutting of DNA at specific locations
became possible with the discovery of -
Q136.
mcq single
+4 / 1
The DNA molecule to which the gene of
interest is integrated for cloning is called -