NEET 2016 Phase 2
MEDICAL 2016 Previous Year
3 hDuration
644Total Marks
322Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 322 across 3 section(s); maximum marks: 644.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Which one of the following statements related to lanthanoids is incorrect?
Q2.
mcq single
+4 / 1
Among the following, which one is a wrong statement ?
Q3.
mcq single
+4 / 1
The hybridizations of atomic orbitals of nitrogen in NO$$_2^ + $$, NO$$_3^ - $$ and NH$$_4^ + $$ respectively are
Q4.
mcq single
+4 / 1
Which one of the following compounds shows the presence of intramolecular hydrogen bond?
Q5.
mcq single
+4 / 1
The correct geometry and hybridization for XeF~4~ are
Q6.
mcq single
+4 / 1
Which of the following pairs of ions is isoelectronic and isostructural ?
Q7.
mcq single
+4 / 1
The percentage of pyridine (C~5~H~5~N) that forms pyridinium ion (C~5~H~5~N^(+)H) ina 0.10 M aqueous pyridine solution (K~b~ for C~5~H~5~N = 1.7 $$ \times $$ 10^($$-$$9)) is
Q8.
mcq single
+4 / 1
The solubility of AgCl~(s)~ with solubility product 1.6 $$ \times $$ 10^($$-$$10) in 0.1 M NaCl solution would be
Q9.
mcq single
+4 / 1
Which of the following fluro-compounds is most likely to behave as a Lewis base ?
Q10.
mcq single
+4 / 1
The decomposition of phosphine (PH~3~) on tungsten at low pressure is a first-order reaction. It is because the
Q11.
mcq single
+4 / 1
Hot concentrated sulphate acid is a moderately strong oxidizing agent. Which of the following reactions does not show oxidizing behaviour?
Q12.
mcq single
+4 / 1
Consider the reaction,
CH~3~CH~2~CH~2~Br + NaCN $$ \to $$ CH~3~CH~2~CH~2~CN + NaBr
This reaction will be the fastest in
Q13.
mcq single
+4 / 1
Which one of the following is incorrect for ideal solution?
Q14.
mcq single
+4 / 1
The van't Hoff factor (i) for a dilute aqueous solution of the strong electrolyte barium hydroxide is
Q15.
mcq single
+4 / 1
Suppose the elements X and Y combine to form two compounds XY~2~ and X~3~Y~2~. When 0.1 mole of XY~2~ weights 10 g and 0.05 mole of X~3~Y~2~ weighs 9 g, the atomic weights of X and Y are
Q16.
mcq single
+4 / 1
For a sample of perfect gas when its pressure is changed isothermally from p~i~ to p~f~, the entropy change is given by
Q17.
mcq single
+4 / 1
Boric acid is an acid because its molecule
Q18.
mcq single
+4 / 1
AlF~3~ is soluble in HF only in presence of KF. It is due to the formation of
Q19.
mcq single
+4 / 1
Jahn-Teller effect is not observed in high spin complexes of
Q20.
mcq single
+4 / 1
The correct increasing order of *trans*-effect of the following species is
Q21.
mcq single
+4 / 1
Which among the given molecules can exhibit tautomerism?


Q22.
mcq single
+4 / 1
The correct structure of the product 'A' formed in the reaction


Q23.
mcq single
+4 / 1
The correct order of strengths of the carboxylic acids
 is
is
 is
is
Q24.
mcq single
+4 / 1
In pyrrole the electron density is maximum on


Q25.
mcq single
+4 / 1
The compound that will react most readily with gaseous bromine has the formula
Q26.
mcq single
+4 / 1
Which of the following can be used as the halide component for Friedel-Crafts reaction?
Q27.
mcq single
+4 / 1
Which of the following compounds shall not produce propene by reaction with HBr followed by elimination or direct only elimination reaction ?
Q28.
mcq single
+4 / 1
In which of the following molecules, all atoms are coplanar?
Q29.
mcq single
+4 / 1
In the given reaction,
 the product P is
the product P is
 the product P is
the product P is
Q30.
mcq single
+4 / 1
The central dogma of molecular genetics states that the genetic information flows from
Q31.
mcq single
+4 / 1
The correct corresponding order of names of four aldoses with configuration given below
 respectively, is
respectively, is
 respectively, is
respectively, is
Q32.
mcq single
+4 / 1
Zinc can be coated on iron to produce galvanized iron but the reverse is not possible. It is because
Q33.
mcq single
+4 / 1
The number of electrons delivered at the cathode during electrolysis by a current of 1 ampere in 60 seconds is (charge on electron = 1.60 $$ \times $$ 10^($$-$$19)C)
Q34.
mcq single
+4 / 1
If the E^(o)~cell~ for a given reaction has a negative value, which of the following gives the correct relationships for the values of $$\Delta $$G^(o) and K~eq~ ?
Q35.
mcq single
+4 / 1
The molar conductivity of a 0.5 mol/dm^(3) solution of AgNO~3~ with electrolytic conductivity of 5.76 $$ \times $$ 10^($$-$$3) S cm^($$-$$1) at 298 K is
Q36.
mcq single
+4 / 1
During the electrolysis of molten sodium chloride, the time required to produce 0.10 mol of chlorine gas using a current of 3 amperes is
Q37.
mcq single
+4 / 1
Which one of the following nitro-compounds does not react with nitrous acid?
Q38.
mcq single
+4 / 1
A given nitrogen-containing aromatic compound 'A' reacts with Sn/HCl, followed by HNO~2~ to give instable compound 'B'. 'B', on treatment with phenol, forms a beautiful coloured compound 'C' with the molecular formula C~12~H~10~N~2~O. The structure of compound 'A' is
Q39.
mcq single
+4 / 1
How many electrons can fit in the orbital for which n = 3 and $$l$$ = 1?
Q40.
mcq single
+4 / 1
Which of the following pairs of d-orbitals will have electron density along the axes ?
Q41.
mcq single
+4 / 1
Which one of the following statements related to lanthanoids is incorrect?
Q42.
mcq single
+4 / 1
Among the following, which one is a wrong statement ?
Q43.
mcq single
+4 / 1
The hybridizations of atomic orbitals of nitrogen in NO$$_2^ + $$, NO$$_3^ - $$ and NH$$_4^ + $$ respectively are
Q44.
mcq single
+4 / 1
Which one of the following compounds shows the presence of intramolecular hydrogen bond?
Q45.
mcq single
+4 / 1
The correct geometry and hybridization for XeF~4~ are
Q46.
mcq single
+4 / 1
Which of the following pairs of ions is isoelectronic and isostructural ?
Q47.
mcq single
+4 / 1
The percentage of pyridine (C~5~H~5~N) that forms pyridinium ion (C~5~H~5~N^(+)H) ina 0.10 M aqueous pyridine solution (K~b~ for C~5~H~5~N = 1.7 $$ \times $$ 10^($$-$$9)) is
Q48.
mcq single
+4 / 1
The solubility of AgCl~(s)~ with solubility product 1.6 $$ \times $$ 10^($$-$$10) in 0.1 M NaCl solution would be
Q49.
mcq single
+4 / 1
Which of the following fluro-compounds is most likely to behave as a Lewis base ?
Q50.
mcq single
+4 / 1
The decomposition of phosphine (PH~3~) on tungsten at low pressure is a first-order reaction. It is because the
Q51.
mcq single
+4 / 1
Hot concentrated sulphate acid is a moderately strong oxidizing agent. Which of the following reactions does not show oxidizing behaviour?
Q52.
mcq single
+4 / 1
Consider the reaction,
CH~3~CH~2~CH~2~Br + NaCN $$ \to $$ CH~3~CH~2~CH~2~CN + NaBr
This reaction will be the fastest in
Q53.
mcq single
+4 / 1
Which one of the following is incorrect for ideal solution?
Q54.
mcq single
+4 / 1
The van't Hoff factor (i) for a dilute aqueous solution of the strong electrolyte barium hydroxide is
Q55.
mcq single
+4 / 1
Suppose the elements X and Y combine to form two compounds XY~2~ and X~3~Y~2~. When 0.1 mole of XY~2~ weights 10 g and 0.05 mole of X~3~Y~2~ weighs 9 g, the atomic weights of X and Y are
Q56.
mcq single
+4 / 1
For a sample of perfect gas when its pressure is changed isothermally from p~i~ to p~f~, the entropy change is given by
Q57.
mcq single
+4 / 1
Boric acid is an acid because its molecule
Q58.
mcq single
+4 / 1
AlF~3~ is soluble in HF only in presence of KF. It is due to the formation of
Q59.
mcq single
+4 / 1
Jahn-Teller effect is not observed in high spin complexes of
Q60.
mcq single
+4 / 1
The correct increasing order of *trans*-effect of the following species is
Q61.
mcq single
+4 / 1
Which among the given molecules can exhibit tautomerism?


Q62.
mcq single
+4 / 1
The correct structure of the product 'A' formed in the reaction
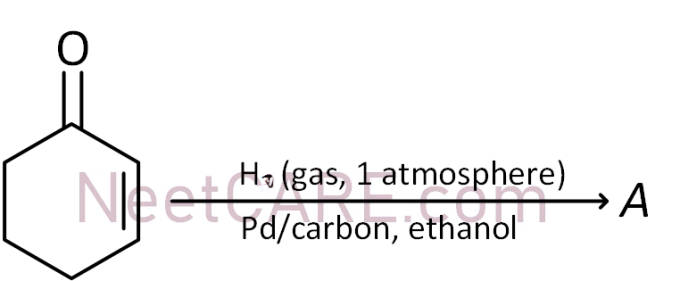
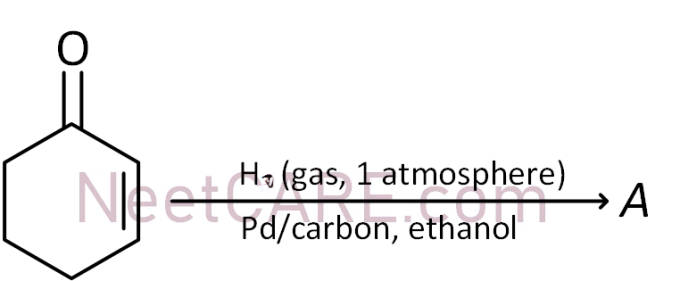
Q63.
mcq single
+4 / 1
The correct order of strengths of the carboxylic acids
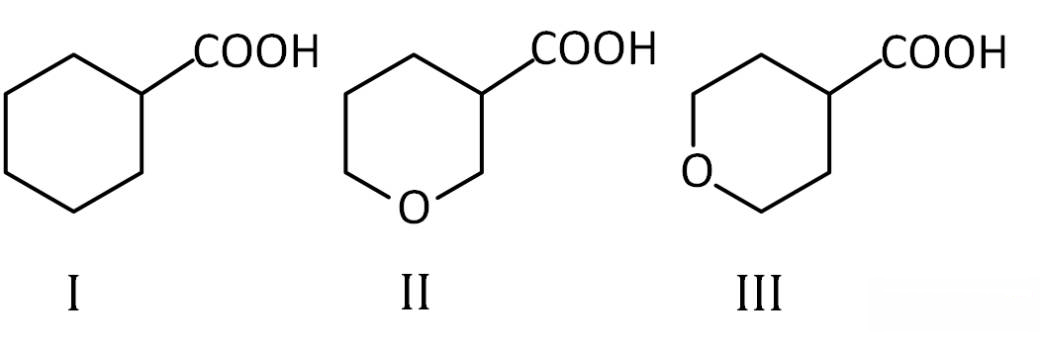 is
is
 is
is
Q64.
mcq single
+4 / 1
In pyrrole the electron density is maximum on


Q65.
mcq single
+4 / 1
The compound that will react most readily with gaseous bromine has the formula
Q66.
mcq single
+4 / 1
Which of the following can be used as the halide component for Friedel-Crafts reaction?
Q67.
mcq single
+4 / 1
Which of the following compounds shall not produce propene by reaction with HBr followed by elimination or direct only elimination reaction ?
Q68.
mcq single
+4 / 1
In which of the following molecules, all atoms are coplanar?
Q69.
mcq single
+4 / 1
In the given reaction,
 the product P is
the product P is
 the product P is
the product P is
Q70.
mcq single
+4 / 1
The central dogma of molecular genetics states that the genetic information flows from
Q71.
mcq single
+4 / 1
The correct corresponding order of names of four aldoses with configuration given below
 respectively, is
respectively, is
 respectively, is
respectively, is
Q72.
mcq single
+4 / 1
Zinc can be coated on iron to produce galvanized iron but the reverse is not possible. It is because
Q73.
mcq single
+4 / 1
The number of electrons delivered at the cathode during electrolysis by a current of 1 ampere in 60 seconds is (charge on electron = 1.60 $$ \times $$ 10^($$-$$19)C)
Q74.
mcq single
+4 / 1
If the E^(o)~cell~ for a given reaction has a negative value, which of the following gives the correct relationships for the values of $$\Delta $$G^(o) and K~eq~ ?
Q75.
mcq single
+4 / 1
The molar conductivity of a 0.5 mol/dm^(3) solution of AgNO~3~ with electrolytic conductivity of 5.76 $$ \times $$ 10^($$-$$3) S cm^($$-$$1) at 298 K is
Q76.
mcq single
+4 / 1
During the electrolysis of molten sodium chloride, the time required to produce 0.10 mol of chlorine gas using a current of 3 amperes is
Q77.
mcq single
+4 / 1
Which one of the following nitro-compounds does not react with nitrous acid?
Q78.
mcq single
+4 / 1
A given nitrogen-containing aromatic compound 'A' reacts with Sn/HCl, followed by HNO~2~ to give instable compound 'B'. 'B', on treatment with phenol, forms a beautiful coloured compound 'C' with the molecular formula C~12~H~10~N~2~O. The structure of compound 'A' is
Q79.
mcq single
+4 / 1
How many electrons can fit in the orbital for which n = 3 and $$l$$ = 1?
Q80.
mcq single
+4 / 1
Which of the following pairs of d-orbitals will have electron density along the axes ?
Physics
Physics
Q1.
mcq single
+4 / 1
The interference pattern is obtained with two coherent light sources of intensity ratio n. In the interference pattern, the ratio $${{{I_{max}} - {I_{\min }}} \over {{I_{max}} + {I_{min}}}}$$ will be
Q2.
mcq single
+4 / 1
A linear aperture whose width is 0.02 cm is placed immediately in front of a lens of focal length 60 cm. The aparture is illuminated normally by a parallel beam of wavelength 5 $$ \times $$ 10^($$-$$5) cm. The distance of the first dark band of the diffraction pattern from the centre of the screen is
Q3.
mcq single
+4 / 1
A light rod of length $$l$$ has two masses m~1~ and m~2~ attached to its two ends. The moment of inertia of the system about an axis perpendicular to the rod and passing through the centre of mass is
Q4.
mcq single
+4 / 1
Two rotating bodies A and B of masses m and 2m with moments of inertia $${I_A}$$ and $${I_B}$$ ($${I_B}$$ > $${I_A}$$) have equal kinetic energy of rotation. If L~A~ and L~B~ be their angular momenta respectively, then
Q5.
mcq single
+4 / 1
A solid sphere of mass m and radius R is rotating about its diameter. A solid cylinder of same mass and same radius is also rotating about its geometrical axis with an angular speed twice that of the sphere. The ratio of their kinetic energies of rotation (E~sphere~ / E~cylinder~) will be
Q6.
mcq single
+4 / 1
A uniform magnetic field is restricted within a region of rafius r. The magnetic field changes with time at a rate $${{d\overrightarrow B } \over {dt}}$$. Loop 1 of radius R > r encloses the region r and loop 2 of radius R is outside the region of magnetic field as shown in the figure. Then the e.m.f. generated is


Q7.
mcq single
+4 / 1
Planck's constant (h), speed of light in vacuum (c) and Newton's gravitional constant (G) are three fundamental constants. Which of the following combinations of these has the dimension of length ?
Q8.
mcq single
+4 / 1
One mole of an ideal monatomic gas undergoes a process described by the equation PV^(3) = constant. The heat capacity of the gas during this process is
Q9.
mcq single
+4 / 1
The temperature inside a refrigerator is t~2~ ^(o)C. The amount of heat delivered to the room for each joule of electrical energy consumed ideally will be
Q10.
mcq single
+4 / 1
A given sample of an ideal gas occupies a volume V at a pressure P and absolute temperature T. The mass of each molecule of the gas is m. Which of the following gives the density of the gas ?
Q11.
mcq single
+4 / 1
Two identical bodies are made of a material for which the heat capacity increases with temperature. One of these is at 100^(o)C, while the other one is at 0^(o)C. If the two bodies are brought into contact, then, assuming no heat loss, the final common temperature is
Q12.
mcq single
+4 / 1
A body cools from a temperature 3T to 2T in 10 minutes. The room temperature is T. Assume that Newton's law of cooling is applicable. The temperature of the body at the end of next 10 minutes will be
Q13.
mcq single
+4 / 1
Three liquids of densities $$\rho $$~1~, $$\rho $$~2~ and $$\rho $$~3~ (with $$\rho $$~1~ > $$\rho $$~2~ > $$\rho $$~3~), having the same value of surface tension T, rise to the same height in three identical capillaries. The angles of contact $$\theta $$~1~, $$\theta $$~2~ and $$\theta $$~3~ obey
Q14.
mcq single
+4 / 1
A rectangular film of liquid is extended from (4 cm $$ \times $$ 2 cm) to (5 cm $$ \times $$ 4 cm). If the work done is 3 $$ \times $$ 10^($$-$$4) J, the value of the surface tension of the liquid is
Q15.
mcq single
+4 / 1
An electric dipole is placed at an angle of 30^(o) with an electric field intensity 2 $$ \times $$ 10^(5) N C^($$-$$1). It experiences a torque equal to 4 N m. The charge on the dipole, if the dipole length is 2 cm, is
Q16.
mcq single
+4 / 1
A satellite of mass m is orbiting the earth (of radius R) at a height h from its surface. The total energy of the satellite in terms of g~0~, the value of acceleration due to gravity at the earth's surface, is
Q17.
mcq single
+4 / 1
Starting from the centre of the earth having radius R, the variation of g (acceleration due to gravity) is shown by
Q18.
mcq single
+4 / 1
A parallel-plate capacitor of area a, plate separation d and capacitance C is filled with four dielectric materials having dielectric constants k~1~, k~2~, k~3~ and k~4~ as shown in the figure. If a single dielectric material is to be used to have the same capacitance C in this capacitor, then its dielectric constant k is given by


Q19.
mcq single
+4 / 1
An air bubble in a glass slab with refractive index 1.5 (near normal incidence) is 5 cm deep when viewed from one surface and 3 cm deep when viewed from the opposite face. The thickness (in cm) of the slab is
Q20.
mcq single
+4 / 1
Two identical glass ($$\mu $$~g~ = 3/2) equiconvex lenses of focal length $$f$$ each are kept in contact. The space between the two lenses is filled with water $$\left( {{\mu _w} = 4/3} \right)$$. The focal length of the combination is
Q21.
mcq single
+4 / 1
A person can see clearly objects only when they lie between 50 cm and 400 cm from his eyes . In order to increase the maximum distance of distinct vision to infinity, the type and power of the correcting lens, the person has to use. will be
Q22.
mcq single
+4 / 1
A body of mass m is attached to the lower end of a spring whose upper end is fixed. The spring has negligible mass. When the mass m is slightly pulled down and released, it oscillates with a time period of 3s. When the mass m is increased by 1 kg, the time period of oscillations becomes 5 s. The value of m in kg is
Q23.
mcq single
+4 / 1
The given circuit has two ideal diodes connected as shown in the figure. The current flowing through the resistance R~1~ will be


Q24.
mcq single
+4 / 1
What is the output Y in the following circuit, when all the three inputs A, B, C are first 0 and then 1 ?


Q25.
mcq single
+4 / 1
An electron is moving in a circular path under the influence of a transverse magnetic field of 3.57 $$ \times $$ 10^(-2) T. If the value of e/m is 1.76 $$ \times $$ 10^(11) C kg^($$-$$1), the frequency of revoluation of the electron is
Q26.
mcq single
+4 / 1
A long wire carrying a steady current is bent into a circular loop of one turn. The magnetic field at the centre of the loop is B. It is then bent into a circular coil of n turns. The magnetic field at the centre of this coil of n turns will be
Q27.
mcq single
+4 / 1
Electrons of mass m with de-Broglie wavelength $$\lambda $$ fall on the target in an X-ray tube. The cutoff wavelength ($$\lambda $$~0~) of the emitted X-ray is
Q28.
mcq single
+4 / 1
Photons with energy 5 eV are incifent on a cathode C in a photoelectric cell. The maximum energy of emitted photoelectrons is 2 eV. When photons of energy 6 eV are incident on C, no photoelectrons will reach the anode A, if the stopping potential of A relative to C is
Q29.
mcq single
+4 / 1
Two cars P and Q start from a point at the same time in a straight line and their positions are represented by
x~P~(t) = (at + bt^(2)) and x~Q~(t) = (ft $$-$$ t^(2)).
At what time do the cars have the same velocity ?
Q30.
mcq single
+4 / 1
If an electron in a hydrogen atom jumps from the 3^(rd) orbit to the 2^(nd) orbit, it emits a photon of wavelength $$\lambda $$. When it jumps from the 4^(th) orbit to the 3^(rd) orbit, the corresponding wavelength of the photon will be
Q31.
mcq single
+4 / 1
In the given figure, a = 15 m s^($$-$$2) represents the total acceleration of particle moving in the clockwise direction in a circle of radius R = 2.5 m at a given instant of time. The speed of the particle is


Q32.
mcq single
+4 / 1
A bullet of mass 10 g moving horizontally with a velocity of 400 m s^($$-$$1) strikes a wood block of mass 2 kg which is suspended by light inextensible string of length 5 m. As a result, the centre of gravity of the block found to rise a vertical distance of 10 cm. The speed of the bullet after it emerges out horizontally from the block will be
Q33.
mcq single
+4 / 1
A rigid ball of mass m strikes a rigid wall at 60^(o) and gets reflected without loss of speed as shown in the figure. The value of impulse imparted by the wall on the ball will be :


Q34.
mcq single
+4 / 1
Two identical balls A and B having velocities of 0.5 m s^($$-$$1) and $$-$$0.3 m s^($$-$$1) respectively collide elastically in one dimension. The velocities of B and A after the collision respectively will be :
Q35.
mcq single
+4 / 1
The second overtone of an open organ pipe has the same frquency as the first overtone of a closed pipe L metre long. The length of the open pipe will be
Q36.
mcq single
+4 / 1
Three sound waves of equal amplitudes have frequencies (n $$-$$ 1), n, (n + 1). They superimpose to give beats. The number of beats produced per second will be
Q37.
mcq single
+4 / 1
A bar magnet is hung by a thin cotton thread in a uniform horizontal magnetic field and is in equilibrium state. The energy required to rotate it by 60^(o) is W. Now the torque required to keep the magnet in this new position is
Q38.
mcq single
+4 / 1
A particle moves from a point $$\left( { - 2\widehat i + 5\widehat j} \right)$$ to $$\left( {4\widehat j + 3\widehat k} \right)$$ when a force of $$\left( {4\widehat i + 3\widehat j} \right)$$ N is applied. How much work has been done by the force ?
Q39.
mcq single
+4 / 1
A filament bulb (500 W, 100 V) is to be used in a 230 V main supply. When a resistance R is connected in series, it works perfectly and the bulb consumes 500 W. The value of R is
Q40.
mcq single
+4 / 1
The potential difference (V~A~ $$-$$ V~B~) between the points A and B in the given figure is
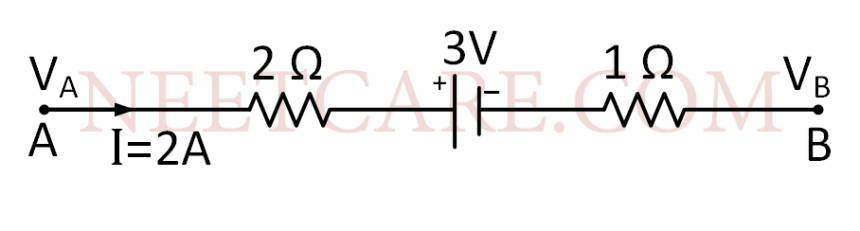
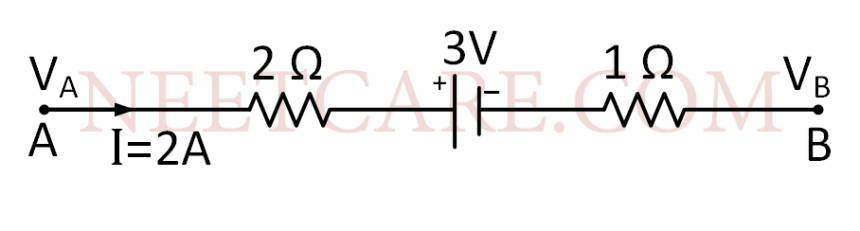
Q41.
mcq single
+4 / 1
Which of the following combinations should be selected for better tuning of an L-C-R circuit used for combination ?
Q42.
mcq single
+4 / 1
The potential differences across the resistance, capacitance and inductance are 80 V, 40 V and 100 V respectively in an L-C-R circuit. The power factor of this circuit is
Q43.
mcq single
+4 / 1
A 100 $$\Omega $$ resistance and a capacitor of 100 $$\Omega $$ reactance are connected in series across a 220 V source. When the capacitor is 50% charged, the peak value of the displacement current is
Q44.
mcq single
+4 / 1
The interference pattern is obtained with two coherent light sources of intensity ratio n. In the interference pattern, the ratio $${{{I_{max}} - {I_{\min }}} \over {{I_{max}} + {I_{min}}}}$$ will be
Q45.
mcq single
+4 / 1
A linear aperture whose width is 0.02 cm is placed immediately in front of a lens of focal length 60 cm. The aparture is illuminated normally by a parallel beam of wavelength 5 $$ \times $$ 10^($$-$$5) cm. The distance of the first dark band of the diffraction pattern from the centre of the screen is
Q46.
mcq single
+4 / 1
A light rod of length $$l$$ has two masses m~1~ and m~2~ attached to its two ends. The moment of inertia of the system about an axis perpendicular to the rod and passing through the centre of mass is
Q47.
mcq single
+4 / 1
Two rotating bodies A and B of masses m and 2m with moments of inertia $${I_A}$$ and $${I_B}$$ ($${I_B}$$ > $${I_A}$$) have equal kinetic energy of rotation. If L~A~ and L~B~ be their angular momenta respectively, then
Q48.
mcq single
+4 / 1
A solid sphere of mass m and radius R is rotating about its diameter. A solid cylinder of same mass and same radius is also rotating about its geometrical axis with an angular speed twice that of the sphere. The ratio of their kinetic energies of rotation (E~sphere~ / E~cylinder~) will be
Q49.
mcq single
+4 / 1
A uniform magnetic field is restricted within a region of rafius r. The magnetic field changes with time at a rate $${{d\overrightarrow B } \over {dt}}$$. Loop 1 of radius R > r encloses the region r and loop 2 of radius R is outside the region of magnetic field as shown in the figure. Then the e.m.f. generated is


Q50.
mcq single
+4 / 1
Planck's constant (h), speed of light in vacuum (c) and Newton's gravitional constant (G) are three fundamental constants. Which of the following combinations of these has the dimension of length ?
Q51.
mcq single
+4 / 1
One mole of an ideal monatomic gas undergoes a process described by the equation PV^(3) = constant. The heat capacity of the gas during this process is
Q52.
mcq single
+4 / 1
The temperature inside a refrigerator is t~2~ ^(o)C. The amount of heat delivered to the room for each joule of electrical energy consumed ideally will be
Q53.
mcq single
+4 / 1
A given sample of an ideal gas occupies a volume V at a pressure P and absolute temperature T. The mass of each molecule of the gas is m. Which of the following gives the density of the gas ?
Q54.
mcq single
+4 / 1
Two identical bodies are made of a material for which the heat capacity increases with temperature. One of these is at 100^(o)C, while the other one is at 0^(o)C. If the two bodies are brought into contact, then, assuming no heat loss, the final common temperature is
Q55.
mcq single
+4 / 1
A body cools from a temperature 3T to 2T in 10 minutes. The room temperature is T. Assume that Newton's law of cooling is applicable. The temperature of the body at the end of next 10 minutes will be
Q56.
mcq single
+4 / 1
Three liquids of densities $$\rho $$~1~, $$\rho $$~2~ and $$\rho $$~3~ (with $$\rho $$~1~ > $$\rho $$~2~ > $$\rho $$~3~), having the same value of surface tension T, rise to the same height in three identical capillaries. The angles of contact $$\theta $$~1~, $$\theta $$~2~ and $$\theta $$~3~ obey
Q57.
mcq single
+4 / 1
A rectangular film of liquid is extended from (4 cm $$ \times $$ 2 cm) to (5 cm $$ \times $$ 4 cm). If the work done is 3 $$ \times $$ 10^($$-$$4) J, the value of the surface tension of the liquid is
Q58.
mcq single
+4 / 1
An electric dipole is placed at an angle of 30^(o) with an electric field intensity 2 $$ \times $$ 10^(5) N C^($$-$$1). It experiences a torque equal to 4 N m. The charge on the dipole, if the dipole length is 2 cm, is
Q59.
mcq single
+4 / 1
A satellite of mass m is orbiting the earth (of radius R) at a height h from its surface. The total energy of the satellite in terms of g~0~, the value of acceleration due to gravity at the earth's surface, is
Q60.
mcq single
+4 / 1
Starting from the centre of the earth having radius R, the variation of g (acceleration due to gravity) is shown by
Q61.
mcq single
+4 / 1
A parallel-plate capacitor of area a, plate separation d and capacitance C is filled with four dielectric materials having dielectric constants k~1~, k~2~, k~3~ and k~4~ as shown in the figure. If a single dielectric material is to be used to have the same capacitance C in this capacitor, then its dielectric constant k is given by


Q62.
mcq single
+4 / 1
An air bubble in a glass slab with refractive index 1.5 (near normal incidence) is 5 cm deep when viewed from one surface and 3 cm deep when viewed from the opposite face. The thickness (in cm) of the slab is
Q63.
mcq single
+4 / 1
Two identical glass ($$\mu $$~g~ = 3/2) equiconvex lenses of focal length $$f$$ each are kept in contact. The space between the two lenses is filled with water $$\left( {{\mu _w} = 4/3} \right)$$. The focal length of the combination is
Q64.
mcq single
+4 / 1
A person can see clearly objects only when they lie between 50 cm and 400 cm from his eyes . In order to increase the maximum distance of distinct vision to infinity, the type and power of the correcting lens, the person has to use. will be
Q65.
mcq single
+4 / 1
A body of mass m is attached to the lower end of a spring whose upper end is fixed. The spring has negligible mass. When the mass m is slightly pulled down and released, it oscillates with a time period of 3s. When the mass m is increased by 1 kg, the time period of oscillations becomes 5 s. The value of m in kg is
Q66.
mcq single
+4 / 1
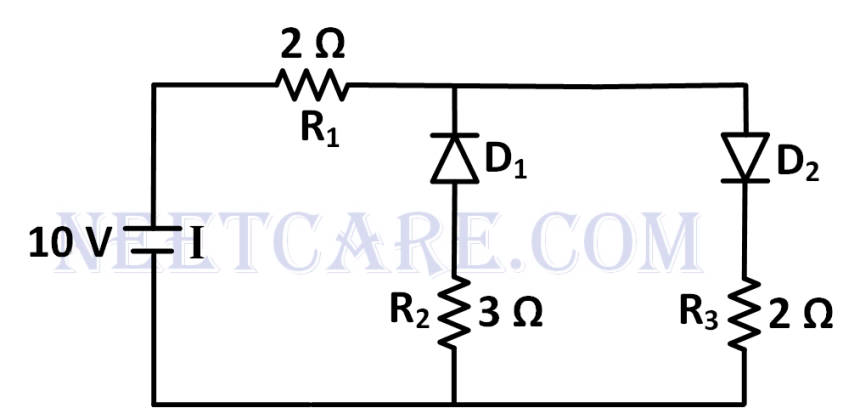
The given circuit has two ideal diodes connected as shown in the figure. The current flowing through the resistance R~1~ will be

Q67.
mcq single
+4 / 1
What is the output Y in the following circuit, when all the three inputs A, B, C are first 0 and then 1 ?


Q68.
mcq single
+4 / 1
An electron is moving in a circular path under the influence of a transverse magnetic field of 3.57 $$ \times $$ 10^(-2) T. If the value of e/m is 1.76 $$ \times $$ 10^(11) C kg^($$-$$1), the frequency of revoluation of the electron is
Q69.
mcq single
+4 / 1
A long wire carrying a steady current is bent into a circular loop of one turn. The magnetic field at the centre of the loop is B. It is then bent into a circular coil of n turns. The magnetic field at the centre of this coil of n turns will be
Q70.
mcq single
+4 / 1
Electrons of mass m with de-Broglie wavelength $$\lambda $$ fall on the target in an X-ray tube. The cutoff wavelength ($$\lambda $$~0~) of the emitted X-ray is
Q71.
mcq single
+4 / 1
Photons with energy 5 eV are incifent on a cathode C in a photoelectric cell. The maximum energy of emitted photoelectrons is 2 eV. When photons of energy 6 eV are incident on C, no photoelectrons will reach the anode A, if the stopping potential of A relative to C is
Q72.
mcq single
+4 / 1
Two cars P and Q start from a point at the same time in a straight line and their positions are represented by
x~P~(t) = (at + bt^(2)) and x~Q~(t) = (ft $$-$$ t^(2)).
At what time do the cars have the same velocity ?
Q73.
mcq single
+4 / 1
If an electron in a hydrogen atom jumps from the 3^(rd) orbit to the 2^(nd) orbit, it emits a photon of wavelength $$\lambda $$. When it jumps from the 4^(th) orbit to the 3^(rd) orbit, the corresponding wavelength of the photon will be
Q74.
mcq single
+4 / 1
In the given figure, a = 15 m s^($$-$$2) represents the total acceleration of particle moving in the clockwise direction in a circle of radius R = 2.5 m at a given instant of time. The speed of the particle is


Q75.
mcq single
+4 / 1
A bullet of mass 10 g moving horizontally with a velocity of 400 m s^($$-$$1) strikes a wood block of mass 2 kg which is suspended by light inextensible string of length 5 m. As a result, the centre of gravity of the block found to rise a vertical distance of 10 cm. The speed of the bullet after it emerges out horizontally from the block will be
Q76.
mcq single
+4 / 1
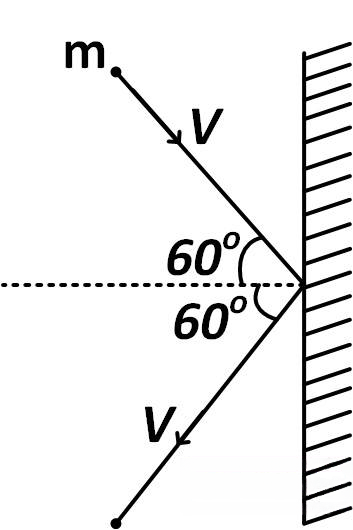
A rigid ball of mass m strikes a rigid wall at 60^(o) and gets reflected without loss of speed as shown in the figure. The value of impulse imparted by the wall on the ball will be :

Q77.
mcq single
+4 / 1
Two identical balls A and B having velocities of 0.5 m s^($$-$$1) and $$-$$0.3 m s^($$-$$1) respectively collide elastically in one dimension. The velocities of B and A after the collision respectively will be :
Q78.
mcq single
+4 / 1
The second overtone of an open organ pipe has the same frquency as the first overtone of a closed pipe L metre long. The length of the open pipe will be
Q79.
mcq single
+4 / 1
Three sound waves of equal amplitudes have frequencies (n $$-$$ 1), n, (n + 1). They superimpose to give beats. The number of beats produced per second will be
Q80.
mcq single
+4 / 1
A bar magnet is hung by a thin cotton thread in a uniform horizontal magnetic field and is in equilibrium state. The energy required to rotate it by 60^(o) is W. Now the torque required to keep the magnet in this new position is
Q81.
mcq single
+4 / 1
A particle moves from a point $$\left( { - 2\widehat i + 5\widehat j} \right)$$ to $$\left( {4\widehat j + 3\widehat k} \right)$$ when a force of $$\left( {4\widehat i + 3\widehat j} \right)$$ N is applied. How much work has been done by the force ?
Q82.
mcq single
+4 / 1
A filament bulb (500 W, 100 V) is to be used in a 230 V main supply. When a resistance R is connected in series, it works perfectly and the bulb consumes 500 W. The value of R is
Q83.
mcq single
+4 / 1
The potential difference (V~A~ $$-$$ V~B~) between the points A and B in the given figure is
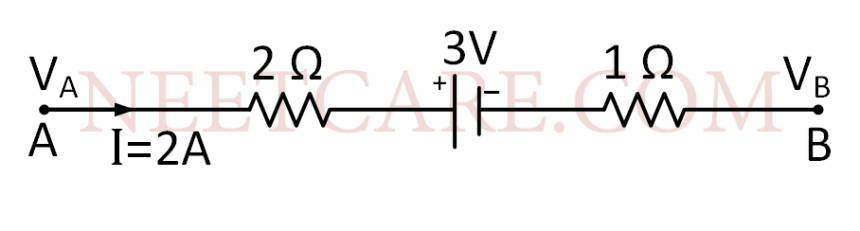
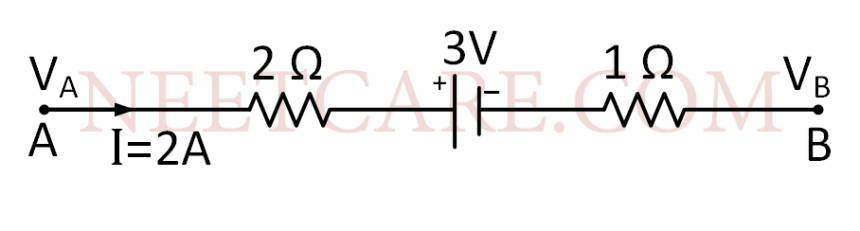
Q84.
mcq single
+4 / 1
Which of the following combinations should be selected for better tuning of an L-C-R circuit used for combination ?
Q85.
mcq single
+4 / 1
The potential differences across the resistance, capacitance and inductance are 80 V, 40 V and 100 V respectively in an L-C-R circuit. The power factor of this circuit is
Q86.
mcq single
+4 / 1
A 100 $$\Omega $$ resistance and a capacitor of 100 $$\Omega $$ reactance are connected in series across a 220 V source. When the capacitor is 50% charged, the peak value of the displacement current is
Biology
Biology
Q1.
mcq single
+4 / 1
Match **Column-I** with **Column-II** and select the correct option using the codes given below –
Column I
Column II
(a) Pistils fused together
(i) Gametogenesis
(b) Formation of gametes
(ii) Pistillate
(c) Hyphae of higher Ascomycetes
(iii) Syncarpous
(d) Unisexual female flower
(iv) Dikaryotic
Q2.
mcq single
+4 / 1
The primary producers of the deep-sea hydrothermal vent ecosystem are
Q3.
mcq single
+4 / 1
If a colour-blind man marries a woman who is homozygous for normal colour vision, the probability of their
son being colour-blind is
Q4.
mcq single
+4 / 1
The mechanism that causes a gene to move from one linkage group to another is called
Q5.
mcq single
+4 / 1
In male cockroaches, sperms are stored in which part of the reproductive system ?
Q6.
mcq single
+4 / 1
Smooth muscles are
Q7.
mcq single
+4 / 1
The process which makes major difference between C~3~ and C~4~ plants is
Q8.
mcq single
+4 / 1
Choose the correct statement
Q9.
mcq single
+4 / 1
Match column I with column II for housefly classification and select the correct option using the codes given below.
Column $${\rm I}$$
Column $${\rm I}$$$${\rm I}$$
(A)
Family
(i)
Diptera
(B)
Order
(ii)
Arthropoda
(C)
Class
(iii)
Muscidae
(D)
Phylum
(iv)
Insecta
Q10.
mcq single
+4 / 1
Conifers are adapted to tolerate extreme environmental conditions because of
Q11.
mcq single
+4 / 1
Which one of the following statements is wrong?
Q12.
mcq single
+4 / 1
The partial pressure of oxygen in the alveoli of the lungs is
Q13.
mcq single
+4 / 1
Lungs do not collapse between breaths and some air always remains in the lungs which can never be expelled because
Q14.
mcq single
+4 / 1
The label of a herbarium sheet does not carry information on
Q15.
mcq single
+4 / 1
Study the four statements (A-D) given below and select the two correct ones out of them.
A. Definition of biological species was given by Ernst Mayr.
B. Photoperiod does not affect reproduction in plants.
C. Binomial nomenclature system was given by R.H. Whittaker.
D. In unicellular organisms, reproduction is synonymous with growth.
The two correct statements are
Q16.
mcq single
+4 / 1
Which kind of therapy was given in 1990 to a four-year-old girl with adenosine deaminase [ADA] deficiency ?
Q17.
mcq single
+4 / 1
Choose the correct statement.
Q18.
mcq single
+4 / 1
Cortex is the region found between -
Q19.
mcq single
+4 / 1
The balloon-shaped structures called tyloses
Q20.
mcq single
+4 / 1
Select the mismatch.
Q21.
mcq single
+4 / 1
A cell organelle containing hydrolytic enzymes is
Q22.
mcq single
+4 / 1
Select the wrong statement-
Q23.
mcq single
+4 / 1
A molecule that can act as a genetic material must fulfill the traits given below, except
Q24.
mcq single
+4 / 1
DNA-dependent RNA polymerase catalyzes transcription on one strand of the DNA which is called the
Q25.
mcq single
+4 / 1
Taylor conducted the experiments to prove semiconservative mode of chromosome replication on
Q26.
mcq single
+4 / 1
Which of the following rRNAs acts as structural RNA as well as ribozyme in bacteria ?
Q27.
mcq single
+4 / 1
The equivalent of a structural gene is -
Q28.
mcq single
+4 / 1
Which of the following is correct regarding AIDS causative agent HIV ?
Q29.
mcq single
+4 / 1
Which of the following sets of diseases is caused by bacteria ?
Q30.
mcq single
+4 / 1
If '+' sign is assigned to beneficial interaction, '–' sign to detrimental and '0' sign to neutral interaction, then the population interaction represented by '+' '–' refers to
Q31.
mcq single
+4 / 1
Which of the following is correct for r-selected species ?
Q32.
mcq single
+4 / 1
The principle of competitive exclusion was stated by
Q33.
mcq single
+4 / 1
Serum differs from blood in
Q34.
mcq single
+4 / 1
Name the blood cells, whose reduction in number can cause clotting disorder, leading to excessive loss of blood from the body.
Q35.
mcq single
+4 / 1
During cell growth, DNA synthesis takes place on
Q36.
mcq single
+4 / 1
Match the stages of meiosis in column I to their characteristic features in column II and select the correct option using the codes given below.
**
Column** $${\rm I}$$
**Column** $${\rm I}$$$${\rm I}$$
A.
Pachytene
(i)
Pairing of homologous
chromosomes
B.
Metaphase $${\rm I}$$
(ii)
Terminalisation of
chiasmata
C.
Diakinesis
(iii)
Crossing-over takes
place
D.
Zygotene
(iv)
Chromosomes align
at equatorial plate
Q37.
mcq single
+4 / 1
When cell has stalled DNA replication fork, which checkpoint should be predominantly activated?
Q38.
mcq single
+4 / 1
Match column I with column II and select the correct option using the codes given below.
Column I
Column II
(A)
Mons pubis
(i)
Embryo formation
(B)
Antrum
(ii)
Sperm
(C)
Trophectoderm
(iii)
Female external genitalia
(D)
Nebenkern
(iv)
Graafian follicle
Q39.
mcq single
+4 / 1
Which of the following depicts the correct pathway of transport of sperms?
Q40.
mcq single
+4 / 1
Several hormones like hCG, hPL, estrogen, progesterone are produced by
Q41.
mcq single
+4 / 1
Graves' disease is caused due to
Q42.
mcq single
+4 / 1
The posterior pituitary gland is not a 'true' endocrine gland because
Q43.
mcq single
+4 / 1
Name a peptide hormone which acts mainly on hepatocytes, adipocytes and enhances cellular glucose uptake and utilisation.
Q44.
mcq single
+4 / 1
How many hot spots of biodiversity in the world have been identified till date by Norman Myers ?
Q45.
mcq single
+4 / 1
Which of the following is correctly matched ?
Q46.
mcq single
+4 / 1
Red List contains data or information on
Q47.
mcq single
+4 / 1
Which of the following National Parks is home to the famous musk deer or hangul ?
Q48.
mcq single
+4 / 1
You are given a tissue with its potential for differentiation in an artificial culture. Which of the following pairs of hormones would you add to the medium to secure shoots as well as roots?
Q49.
mcq single
+4 / 1
Phytochrome is a
Q50.
mcq single
+4 / 1
Oxidative phosphorylation is
Q51.
mcq single
+4 / 1
Which of the following biomolecules is common to respiration-mediated breakdown of fats, carbohydrates and proteins?
Q52.
mcq single
+4 / 1
Osteoporosis, an age-related disease of skeletal system, may occur due to
Q53.
mcq single
+4 / 1
Name the ion responsible for unmasking of active sites for myosin for cross-bridge activity during muscle contraction.
Q54.
mcq single
+4 / 1
Which one of the following is wrong for fungi?
Q55.
mcq single
+4 / 1
Select the wrong statement.
Q56.
mcq single
+4 / 1
Methanogens belong to
Q57.
mcq single
+4 / 1
In Hardy-Weinberg equation, the frequency of heterozygous individual is represented by
Q58.
mcq single
+4 / 1
Which of the following is the correct sequence of events in the origin of life ?
I. Formation of protobionts
II. Synthesis of organic monomers
III. Synthesis of organic polymers
IV. Formation of DNA-based genetic systems
Q59.
mcq single
+4 / 1
The chronological order of human evolution from early to the recent is
Q60.
mcq single
+4 / 1
Genetic drift operates in
Q61.
mcq single
+4 / 1
The part of nephron involved in active reabsorption of sodium is
Q62.
mcq single
+4 / 1
Embryo with more than 16 blastgomeres formed due to in vitro fertilization is transferred into
Q63.
mcq single
+4 / 1
Which of the following is hormone releasing IUD ?
Q64.
mcq single
+4 / 1
Which of the following is incorrect regarding vasectomy ?
Q65.
mcq single
+4 / 1
The ovule of an angiosperm is technically equivalent to
Q66.
mcq single
+4 / 1
Pollination in water hyacinth and water lily is brought about by the agency of
Q67.
mcq single
+4 / 1
In majority of angiosperms
Q68.
mcq single
+4 / 1
How many plants among *Indigofera., Sesbania,* *Salvia, Allium, Aloe,* mustard, groundnut, radish, gram and turnip have stamens with different lengths in their flowers?
Q69.
mcq single
+4 / 1
The term 'polyadelphous' is related to
Q70.
mcq single
+4 / 1
Radial symmetry is found in the flowers of
Q71.
mcq single
+4 / 1
Free-central placentation is found in
Q72.
mcq single
+4 / 1
A non-proteinaceous enzyme is
Q73.
mcq single
+4 / 1
Which of the following is the least likely to be involved in stablishing the three-dimensional folding of most proteins?
Q74.
mcq single
+4 / 1
Which of the following describes the given graph correctly?


Q75.
mcq single
+4 / 1
Which of the following restriction enzymes produces blunt ends ?
Q76.
mcq single
+4 / 1
Stirred-tank bioreactors have been designed for -
Q77.
mcq single
+4 / 1
Which of the following is not a component of downstream processing ?
Q78.
mcq single
+4 / 1
A foreign DNA and plasmid cut by the same restriction endonuclease can be joined to form a recombinant plasmid using
Q79.
mcq single
+4 / 1
Match **Column-I** with **Column-II** and select the correct option using the codes given below –
Column I
Column II
(a) Pistils fused together
(i) Gametogenesis
(b) Formation of gametes
(ii) Pistillate
(c) Hyphae of higher Ascomycetes
(iii) Syncarpous
(d) Unisexual female flower
(iv) Dikaryotic
Q80.
mcq single
+4 / 1
The primary producers of the deep-sea hydrothermal vent ecosystem are
Q81.
mcq single
+4 / 1
If a colour-blind man marries a woman who is homozygous for normal colour vision, the probability of their
son being colour-blind is
Q82.
mcq single
+4 / 1
The mechanism that causes a gene to move from one linkage group to another is called
Q83.
mcq single
+4 / 1
In male cockroaches, sperms are stored in which part of the reproductive system ?
Q84.
mcq single
+4 / 1
Smooth muscles are
Q85.
mcq single
+4 / 1
The process which makes major difference between C~3~ and C~4~ plants is
Q86.
mcq single
+4 / 1
Choose the correct statement
Q87.
mcq single
+4 / 1
Match column I with column II for housefly classification and select the correct option using the codes given below.
Column $${\rm I}$$
Column $${\rm I}$$$${\rm I}$$
(A)
Family
(i)
Diptera
(B)
Order
(ii)
Arthropoda
(C)
Class
(iii)
Muscidae
(D)
Phylum
(iv)
Insecta
Q88.
mcq single
+4 / 1
Conifers are adapted to tolerate extreme environmental conditions because of
Q89.
mcq single
+4 / 1
Which one of the following statements is wrong?
Q90.
mcq single
+4 / 1
The partial pressure of oxygen in the alveoli of the lungs is
Q91.
mcq single
+4 / 1
Lungs do not collapse between breaths and some air always remains in the lungs which can never be expelled because
Q92.
mcq single
+4 / 1
The label of a herbarium sheet does not carry information on
Q93.
mcq single
+4 / 1
Study the four statements (A-D) given below and select the two correct ones out of them.
A. Definition of biological species was given by Ernst Mayr.
B. Photoperiod does not affect reproduction in plants.
C. Binomial nomenclature system was given by R.H. Whittaker.
D. In unicellular organisms, reproduction is synonymous with growth.
The two correct statements are
Q94.
mcq single
+4 / 1
Which kind of therapy was given in 1990 to a four-year-old girl with adenosine deaminase [ADA] deficiency ?
Q95.
mcq single
+4 / 1
Choose the correct statement.
Q96.
mcq single
+4 / 1
Cortex is the region found between -
Q97.
mcq single
+4 / 1
The balloon-shaped structures called tyloses
Q98.
mcq single
+4 / 1
Select the mismatch.
Q99.
mcq single
+4 / 1
A cell organelle containing hydrolytic enzymes is
Q100.
mcq single
+4 / 1
Select the wrong statement-
Q101.
mcq single
+4 / 1
A molecule that can act as a genetic material must fulfill the traits given below, except
Q102.
mcq single
+4 / 1
DNA-dependent RNA polymerase catalyzes transcription on one strand of the DNA which is called the
Q103.
mcq single
+4 / 1
Taylor conducted the experiments to prove semiconservative mode of chromosome replication on
Q104.
mcq single
+4 / 1
Which of the following rRNAs acts as structural RNA as well as ribozyme in bacteria ?
Q105.
mcq single
+4 / 1
The equivalent of a structural gene is -
Q106.
mcq single
+4 / 1
Which of the following is correct regarding AIDS causative agent HIV ?
Q107.
mcq single
+4 / 1
Which of the following sets of diseases is caused by bacteria ?
Q108.
mcq single
+4 / 1
If '+' sign is assigned to beneficial interaction, '–' sign to detrimental and '0' sign to neutral interaction, then the population interaction represented by '+' '–' refers to
Q109.
mcq single
+4 / 1
Which of the following is correct for r-selected species ?
Q110.
mcq single
+4 / 1
The principle of competitive exclusion was stated by
Q111.
mcq single
+4 / 1
Serum differs from blood in
Q112.
mcq single
+4 / 1
Name the blood cells, whose reduction in number can cause clotting disorder, leading to excessive loss of blood from the body.
Q113.
mcq single
+4 / 1
During cell growth, DNA synthesis takes place on
Q114.
mcq single
+4 / 1
Match the stages of meiosis in column I to their characteristic features in column II and select the correct option using the codes given below.
**
Column** $${\rm I}$$
**Column** $${\rm I}$$$${\rm I}$$
A.
Pachytene
(i)
Pairing of homologous
chromosomes
B.
Metaphase $${\rm I}$$
(ii)
Terminalisation of
chiasmata
C.
Diakinesis
(iii)
Crossing-over takes
place
D.
Zygotene
(iv)
Chromosomes align
at equatorial plate
Q115.
mcq single
+4 / 1
When cell has stalled DNA replication fork, which checkpoint should be predominantly activated?
Q116.
mcq single
+4 / 1
Match column I with column II and select the correct option using the codes given below.
Column I
Column II
(A)
Mons pubis
(i)
Embryo formation
(B)
Antrum
(ii)
Sperm
(C)
Trophectoderm
(iii)
Female external genitalia
(D)
Nebenkern
(iv)
Graafian follicle
Q117.
mcq single
+4 / 1
Which of the following depicts the correct pathway of transport of sperms?
Q118.
mcq single
+4 / 1
Several hormones like hCG, hPL, estrogen, progesterone are produced by
Q119.
mcq single
+4 / 1
Graves' disease is caused due to
Q120.
mcq single
+4 / 1
The posterior pituitary gland is not a 'true' endocrine gland because
Q121.
mcq single
+4 / 1
Name a peptide hormone which acts mainly on hepatocytes, adipocytes and enhances cellular glucose uptake and utilisation.
Q122.
mcq single
+4 / 1
How many hot spots of biodiversity in the world have been identified till date by Norman Myers ?
Q123.
mcq single
+4 / 1
Which of the following is correctly matched ?
Q124.
mcq single
+4 / 1
Red List contains data or information on
Q125.
mcq single
+4 / 1
Which of the following National Parks is home to the famous musk deer or hangul ?
Q126.
mcq single
+4 / 1
You are given a tissue with its potential for differentiation in an artificial culture. Which of the following pairs of hormones would you add to the medium to secure shoots as well as roots?
Q127.
mcq single
+4 / 1
Phytochrome is a
Q128.
mcq single
+4 / 1
Oxidative phosphorylation is
Q129.
mcq single
+4 / 1
Which of the following biomolecules is common to respiration-mediated breakdown of fats, carbohydrates and proteins?
Q130.
mcq single
+4 / 1
Osteoporosis, an age-related disease of skeletal system, may occur due to
Q131.
mcq single
+4 / 1
Name the ion responsible for unmasking of active sites for myosin for cross-bridge activity during muscle contraction.
Q132.
mcq single
+4 / 1
Which one of the following is wrong for fungi?
Q133.
mcq single
+4 / 1
Select the wrong statement.
Q134.
mcq single
+4 / 1
Methanogens belong to
Q135.
mcq single
+4 / 1
In Hardy-Weinberg equation, the frequency of heterozygous individual is represented by
Q136.
mcq single
+4 / 1
Which of the following is the correct sequence of events in the origin of life ?
I. Formation of protobionts
II. Synthesis of organic monomers
III. Synthesis of organic polymers
IV. Formation of DNA-based genetic systems
Q137.
mcq single
+4 / 1
The chronological order of human evolution from early to the recent is
Q138.
mcq single
+4 / 1
Genetic drift operates in
Q139.
mcq single
+4 / 1
The part of nephron involved in active reabsorption of sodium is
Q140.
mcq single
+4 / 1
Embryo with more than 16 blastgomeres formed due to in vitro fertilization is transferred into
Q141.
mcq single
+4 / 1
Which of the following is hormone releasing IUD ?
Q142.
mcq single
+4 / 1
Which of the following is incorrect regarding vasectomy ?
Q143.
mcq single
+4 / 1
The ovule of an angiosperm is technically equivalent to
Q144.
mcq single
+4 / 1
Pollination in water hyacinth and water lily is brought about by the agency of
Q145.
mcq single
+4 / 1
In majority of angiosperms
Q146.
mcq single
+4 / 1
How many plants among *Indigofera., Sesbania,* *Salvia, Allium, Aloe,* mustard, groundnut, radish, gram and turnip have stamens with different lengths in their flowers?
Q147.
mcq single
+4 / 1
The term 'polyadelphous' is related to
Q148.
mcq single
+4 / 1
Radial symmetry is found in the flowers of
Q149.
mcq single
+4 / 1
Free-central placentation is found in
Q150.
mcq single
+4 / 1
A non-proteinaceous enzyme is
Q151.
mcq single
+4 / 1
Which of the following is the least likely to be involved in stablishing the three-dimensional folding of most proteins?
Q152.
mcq single
+4 / 1
Which of the following describes the given graph correctly?


Q153.
mcq single
+4 / 1
Which of the following restriction enzymes produces blunt ends ?
Q154.
mcq single
+4 / 1
Stirred-tank bioreactors have been designed for -
Q155.
mcq single
+4 / 1
Which of the following is not a component of downstream processing ?
Q156.
mcq single
+4 / 1
A foreign DNA and plasmid cut by the same restriction endonuclease can be joined to form a recombinant plasmid using