neet-2017
NEET 2017
MEDICAL 2017 Previous Year
3 hDuration
656Total Marks
328Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 328 across 3 section(s); maximum marks: 656.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
HgCl~2~ and I~2~ both when dissolved in water containing I^($$-$$) ions, the pair of species formed is
Q2.
mcq single
+4 / 1
The reason for greater range of oxidation states in actinoids is attributed to
Q3.
mcq single
+4 / 1
Name the gas that can readily decolourise acidified KMnO~4~ solution.
Q4.
mcq single
+4 / 1
The species, having bond angles of 120^(o) is
Q5.
mcq multi
+4 / 1
Which of the following pairs of compounds is isostructural ?
Q6.
mcq single
+4 / 1
Which one of the following pairs of species have the same bond order ?
Q7.
mcq single
+4 / 1
Concentration of the Ag^(+) ions in a saturated solution of Ag~2~C~2~O~4~ is 2.2 $$ \times $$ 10^($$-$$4) mol L^($$-$$1). Solubility product of Ag~2~C~2~O~4~ is
Q8.
mcq single
+4 / 1
Mechanism of a hypothetical reaction
X~2~ + Y~2~ $$ \to $$ 2XY, is given below :
(i) X~2~ $$ \to $$ X + X (fast)
(ii) X + Y~2~ $$\rightleftharpoons$$ XY + Y (slow)
(iii) X + Y $$ \to $$ XY (fast)
The overall order of the reaction will be
Q9.
mcq single
+4 / 1
A first order reaction has a specific reaction rate of 10^($$-$$2) sec^($$-$$1). How much time will it take for 20 g of the reactant to reduce to 5 g?
Q10.
mcq single
+4 / 1
Identify $$A$$ and predict the type of reaction.


Q11.
mcq single
+4 / 1
If molality of the dilute solution is doubled, the value of molal depression constant (*K~f~*) will be
Q12.
mcq single
+4 / 1
Which of the following is dependent on temperature?
Q13.
mcq single
+4 / 1
The equilibrium constants of the following are
N~2~ + 3H~2~ $$\rightleftharpoons$$ 2NH~3~; K~1~
N~2~ + O~2~ $$\rightleftharpoons$$ 2NO~~; K~2~
H~2~ + $${1 \over 2}$$O~2~ $$\rightleftharpoons$$
H~2~O; K~3~
The equilibrium constant (K) of the reaction :
2NH~3~ + $${5 \over 2}$$ O~2~ $$\rightleftharpoons$$ 2NO + 3H~2~O will be
Q14.
mcq single
+4 / 1
A 20 litre container at 400 K contains CO~2(g)~ at pressure 0.4 atm and an excess of SrO (neglect the volume of solid SrO). The volume of the container is now decreased by moving the movable piston fitted in the container. The maximum volume of the container, when pressure of CO~2~ attains its maximum value, will be
(Given that : SrCO~3(s)~ $$\rightleftharpoons$$ SrO~(s)~ + CO~2(g)~, K~p~ = 1.6 atm)
Q15.
mcq single
+4 / 1
The element Z = 114 has been discovered recently. It will belong to which of the following family/group and electronic configuration ?
Q16.
mcq single
+4 / 1
A gas is allowed to expand in a well insulated container against a constant external pressure of 2.5 atm from an initial volume of 2.50 L to a final volume of 4.50 L. The change in internal energy $$\Delta $$U of the gas in joules will be
Q17.
mcq single
+4 / 1
For a given reaction, $$\Delta $$H = 35.5 kJ mol^($$-$$1) and $$\Delta $$S = 83.6 J K^($$-$$1) mol^($$-$$1). The reaction is spontaneous at (Assume that $$\Delta $$H and $$\Delta $$S do not vary with temperature.)
Q18.
mcq single
+4 / 1
Match the interhalogen compounds of column-I with the geometry in column-II and assign the correct code.
Column $${\rm I}$$
Column $${\rm I}$$$${\rm I}$$
(A)
XX'
(i)
T-shape
(B)
XX'~3~
(ii)
Pentagonal bipyramidal
(C)
XX'~5~
(iii)
Linear
(D)
XX'~7~
(iv)
Square pyramidal
(v)
Tetrahedral
Q19.
mcq single
+4 / 1
It is because of inability of ns^(2) electrons of the valence shell to participate in bonding that
Q20.
mcq single
+4 / 1
In which pair of ions both the species contain S $$-$$ S bond?
Q21.
mcq single
+4 / 1
Pick out the correct statement with respect to [Mn(CN)~6~]^(3$$-$$).
Q22.
mcq single
+4 / 1
An example of a sigma bonded organometallic compound is
Q23.
mcq single
+4 / 1
Correct increasing order for the wavelengths of absorption in the visible region for the complexes of Co^(3+) is
Q24.
mcq single
+4 / 1
The correct order of the stoichiometries of AgCl formed when AgNO~3~ in excess is treated with the complexes : CoCl~3~.6NH~3~, CoCl~3~.5NH~3~, CoCl~3~.4NH~3~ respectively is
Q25.
mcq single
+4 / 1
The most suitable method of separation of 1 : 1 mixture of ortho and para-nitrophenols is
Q26.
mcq single
+4 / 1
The IUPAC name of the compound


Q27.
mcq single
+4 / 1
The correct statement regarding electrophile is
Q28.
mcq single
+4 / 1
Consider the reactions,
 Identify *A, X, Y* and *Z*.
Identify *A, X, Y* and *Z*.
 Identify *A, X, Y* and *Z*.
Identify *A, X, Y* and *Z*.
Q29.
mcq single
+4 / 1
Of the following, which is the product formed when cyclohexanone undergoes aldol condensation followed by heating?
Q30.
mcq single
+4 / 1
The heating of phenyl methyl ether with HI produces
Q31.
mcq single
+4 / 1
Which one is the most acidic compound?
Q32.
mcq single
+4 / 1
Predict the correct intermediate and product in the following reaction :
$${H_3}C - C \equiv CH\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{HgS{O_4}}^{{H_2}O,{H_2}S{O_4}}} \mathop {Intermediate}\limits_{(A)} \to \mathop {\Pr oduct}\limits_{(B)} $$
Q33.
mcq single
+4 / 1
Which one is the correct order of acidity?
Q34.
mcq single
+4 / 1
With respect to the conformers of ethane, which of the following statements is true?
Q35.
mcq single
+4 / 1
Which of the following statements is not correct?
Q36.
mcq single
+4 / 1
In the electrochemical cell :
$$Zn\left| {ZnS{O_4}\left( {0.01\,M} \right)} \right|$$$$\left| {CuS{O_4}\left( {1.0M} \right)} \right|Cu,$$
the emf of this Daniell cell is E~1~. When the concentration of ZnSO~4~ is changed to 1.0 M and that of CuSO4 changed to 0.01 M, the emf changes to E~2~. From the followings, which one is the relationship between E~1~ and E~2~? (Given, RT/F = 0.059)
Q37.
mcq single
+4 / 1
The correct increasing order of basic strength for the following compounds is


Q38.
mcq single
+4 / 1
Which of the following reactions is appropriate for converting acetamide to methanamine?
Q39.
mcq single
+4 / 1
Which one is the wrong statement ?
Q40.
mcq single
+4 / 1
HgCl~2~ and I~2~ both when dissolved in water containing I^($$-$$) ions, the pair of species formed is
Q41.
mcq single
+4 / 1
The reason for greater range of oxidation states in actinoids is attributed to
Q42.
mcq single
+4 / 1
Name the gas that can readily decolourise acidified KMnO~4~ solution.
Q43.
mcq single
+4 / 1
The species, having bond angles of 120^(o) is
Q44.
mcq multi
+4 / 1
Which of the following pairs of compounds is isostructural ?
Q45.
mcq single
+4 / 1
Which one of the following pairs of species have the same bond order ?
Q46.
mcq single
+4 / 1
Concentration of the Ag^(+) ions in a saturated solution of Ag~2~C~2~O~4~ is 2.2 $$ \times $$ 10^($$-$$4) mol L^($$-$$1). Solubility product of Ag~2~C~2~O~4~ is
Q47.
mcq single
+4 / 1
Mechanism of a hypothetical reaction
X~2~ + Y~2~ $$ \to $$ 2XY, is given below :
(i) X~2~ $$ \to $$ X + X (fast)
(ii) X + Y~2~ $$\rightleftharpoons$$ XY + Y (slow)
(iii) X + Y $$ \to $$ XY (fast)
The overall order of the reaction will be
Q48.
mcq single
+4 / 1
A first order reaction has a specific reaction rate of 10^($$-$$2) sec^($$-$$1). How much time will it take for 20 g of the reactant to reduce to 5 g?
Q49.
mcq single
+4 / 1
Identify $$A$$ and predict the type of reaction.
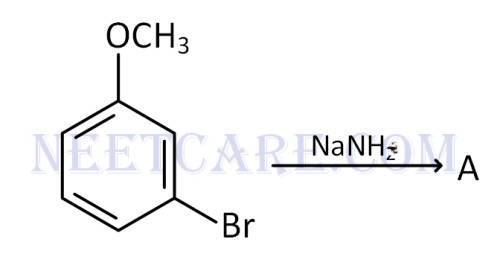

Q50.
mcq single
+4 / 1
If molality of the dilute solution is doubled, the value of molal depression constant (*K~f~*) will be
Q51.
mcq single
+4 / 1
Which of the following is dependent on temperature?
Q52.
mcq single
+4 / 1
The equilibrium constants of the following are
N~2~ + 3H~2~ $$\rightleftharpoons$$ 2NH~3~; K~1~
N~2~ + O~2~ $$\rightleftharpoons$$ 2NO~~; K~2~
H~2~ + $${1 \over 2}$$O~2~ $$\rightleftharpoons$$
H~2~O; K~3~
The equilibrium constant (K) of the reaction :
2NH~3~ + $${5 \over 2}$$ O~2~ $$\rightleftharpoons$$ 2NO + 3H~2~O will be
Q53.
mcq single
+4 / 1
A 20 litre container at 400 K contains CO~2(g)~ at pressure 0.4 atm and an excess of SrO (neglect the volume of solid SrO). The volume of the container is now decreased by moving the movable piston fitted in the container. The maximum volume of the container, when pressure of CO~2~ attains its maximum value, will be
(Given that : SrCO~3(s)~ $$\rightleftharpoons$$ SrO~(s)~ + CO~2(g)~, K~p~ = 1.6 atm)
Q54.
mcq single
+4 / 1
The element Z = 114 has been discovered recently. It will belong to which of the following family/group and electronic configuration ?
Q55.
mcq single
+4 / 1
A gas is allowed to expand in a well insulated container against a constant external pressure of 2.5 atm from an initial volume of 2.50 L to a final volume of 4.50 L. The change in internal energy $$\Delta $$U of the gas in joules will be
Q56.
mcq single
+4 / 1
For a given reaction, $$\Delta $$H = 35.5 kJ mol^($$-$$1) and $$\Delta $$S = 83.6 J K^($$-$$1) mol^($$-$$1). The reaction is spontaneous at (Assume that $$\Delta $$H and $$\Delta $$S do not vary with temperature.)
Q57.
mcq single
+4 / 1
Match the interhalogen compounds of column-I with the geometry in column-II and assign the correct code.
Column $${\rm I}$$
Column $${\rm I}$$$${\rm I}$$
(A)
XX'
(i)
T-shape
(B)
XX'~3~
(ii)
Pentagonal bipyramidal
(C)
XX'~5~
(iii)
Linear
(D)
XX'~7~
(iv)
Square pyramidal
(v)
Tetrahedral
Q58.
mcq single
+4 / 1
It is because of inability of ns^(2) electrons of the valence shell to participate in bonding that
Q59.
mcq single
+4 / 1
In which pair of ions both the species contain S $$-$$ S bond?
Q60.
mcq single
+4 / 1
Pick out the correct statement with respect to [Mn(CN)~6~]^(3$$-$$).
Q61.
mcq single
+4 / 1
An example of a sigma bonded organometallic compound is
Q62.
mcq single
+4 / 1
Correct increasing order for the wavelengths of absorption in the visible region for the complexes of Co^(3+) is
Q63.
mcq single
+4 / 1
The correct order of the stoichiometries of AgCl formed when AgNO~3~ in excess is treated with the complexes : CoCl~3~.6NH~3~, CoCl~3~.5NH~3~, CoCl~3~.4NH~3~ respectively is
Q64.
mcq single
+4 / 1
The most suitable method of separation of 1 : 1 mixture of ortho and para-nitrophenols is
Q65.
mcq single
+4 / 1
The IUPAC name of the compound


Q66.
mcq single
+4 / 1
The correct statement regarding electrophile is
Q67.
mcq single
+4 / 1
Consider the reactions,
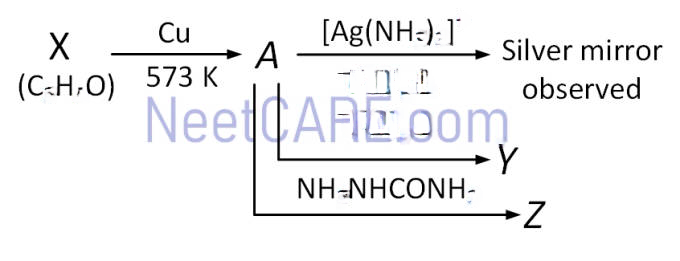 Identify *A, X, Y* and *Z*.
Identify *A, X, Y* and *Z*.
 Identify *A, X, Y* and *Z*.
Identify *A, X, Y* and *Z*.
Q68.
mcq single
+4 / 1
Of the following, which is the product formed when cyclohexanone undergoes aldol condensation followed by heating?
Q69.
mcq single
+4 / 1
The heating of phenyl methyl ether with HI produces
Q70.
mcq single
+4 / 1
Which one is the most acidic compound?
Q71.
mcq single
+4 / 1
Predict the correct intermediate and product in the following reaction :
$${H_3}C - C \equiv CH\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{HgS{O_4}}^{{H_2}O,{H_2}S{O_4}}} \mathop {Intermediate}\limits_{(A)} \to \mathop {\Pr oduct}\limits_{(B)} $$
Q72.
mcq single
+4 / 1
Which one is the correct order of acidity?
Q73.
mcq single
+4 / 1
With respect to the conformers of ethane, which of the following statements is true?
Q74.
mcq single
+4 / 1
Which of the following statements is not correct?
Q75.
mcq single
+4 / 1
In the electrochemical cell :
$$Zn\left| {ZnS{O_4}\left( {0.01\,M} \right)} \right|$$$$\left| {CuS{O_4}\left( {1.0M} \right)} \right|Cu,$$
the emf of this Daniell cell is E~1~. When the concentration of ZnSO~4~ is changed to 1.0 M and that of CuSO4 changed to 0.01 M, the emf changes to E~2~. From the followings, which one is the relationship between E~1~ and E~2~? (Given, RT/F = 0.059)
Q76.
mcq single
+4 / 1
The correct increasing order of basic strength for the following compounds is


Q77.
mcq single
+4 / 1
Which of the following reactions is appropriate for converting acetamide to methanamine?
Q78.
mcq single
+4 / 1
Which one is the wrong statement ?
Physics
Physics
Q1.
mcq single
+4 / 1
The ratio of resolving powers of an optical microscope for two wavelength $$\lambda $$~1~ = 4000 $$\mathop A\limits^ \circ $$ and $${\lambda _2}$$ = 6000 $$\mathop A\limits^ \circ $$ is
Q2.
mcq single
+4 / 1
Two polaroids P~1~ and P~2~ are placed with their axis perpendicular to each other. Unpolarised light $$I$$~0~ is incident on P~1~. A third polaroid P~3~ is kept in between P~1~ and P~2~ such that its axis makes an angle 45^(o) with that of P~1~. The intensity of transmitted light through P~2~ is
Q3.
mcq single
+4 / 1
Young's double slit experiment is first performed in air and then in a medium other than air. It is found that 8^(th) bright fringe in the medium lies where 5^(th) dark fringe lies in air. The refractive index of the medium is nearly
Q4.
mcq single
+4 / 1
Two discs of same moment of inertia rotating about their regular axis passing through centre and perpendicular to the plane of disc with angular velocities $${\omega _1}$$ and $${\omega _2}$$. They are brought into contact face to face coinciding the axis of rotation. The expression for loss of energy during this process is
Q5.
mcq single
+4 / 1
A rope is wound around a hollow cylinder of mass 3 kg and radius 40 cm. What is the angular acceleration of the cylinder if the rope is pulled with a force of 30 N?
Q6.
mcq single
+4 / 1
A long solenoid of diameter 0.1 m has 2 $$ \times $$ 10^(4) turns per meter. At the centre of the solenoid, a coil of 100 turns and radius 0.01 m is placed with its axis coinciding with the solenoid axis. The current in the solenoid reduces at a constant rate to 0 A from 4 A in 0.05 s. If the resistance of the coil is 10 $$\pi $$^(2) $$\Omega $$, the total charge flowing through the coil during this time is
Q7.
mcq single
+4 / 1
A physical quantity of the dimensions of length that can be formed out of c, G and $${{{e^2}} \over {4\pi {\varepsilon _0}}}$$ is [c is velocity of light, G is the universal constant of gravitation and e is charge]
Q8.
mcq single
+4 / 1
Thermodynamic processes are indicated in the following diagram.
 Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
 Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
Q9.
mcq single
+4 / 1
A carnot engine having an efficiency of $${1 \over {10}}$$ as heat engine, is used as a refrigerator. If the work done on the system is 10 J, the amount of energy absorbed from the reservoir at lower temperature is
Q10.
mcq single
+4 / 1
A gas mixture consists of 2 moles of O~2~ and 4 moles of Ar at temperature T. Neglecting all vibrational modes, the total internal energy of the system is
Q11.
mcq single
+4 / 1
The bulk modulus of a spherical object is 'B'. If it is subjected to uniform pressure 'P', the fractional decrease in radius is
Q12.
mcq single
+4 / 1
A spherical black body with a radius of 12 cm radiates 450 watt power at 500 K. If the radius were halved and the temperature doubled, the power radiated in watt would be
Q13.
mcq single
+4 / 1
Two rods A and B of different materials are welded together as shown in figure. Their thermal conductivities are K~1~ and K~2~. The thermal conductivity of the composite will be


Q14.
mcq single
+4 / 1
A U tube with both ends open to the atmosphere, is partially filled with water. Oil, which is immiscible with water, is poured into one side until it stands at a distance of 10 mm above the water level on the other side. Meanwhile the water rises by 65 mm from its original level (see diagram). The density of the oil is


Q15.
mcq single
+4 / 1
The diagrams below show regions of equipotentials.
 A positive charge is moved from A to B in each diagram.
A positive charge is moved from A to B in each diagram.
 A positive charge is moved from A to B in each diagram.
A positive charge is moved from A to B in each diagram.
Q16.
mcq single
+4 / 1
Suppose the charge of a proton and an electron differ slightly. One of them is $$-$$e, the other is (e + $$\Delta $$e). If the net of electrostatic force and gravitational force between two hydrogen atoms placed at a distance d (musch greater than atomic size) apart is zero, then $$\Delta $$e is of the order of
[Given : mass of hydrogen m~h~ = 1.67 $$ \times $$ 10$$-$$^(27) kg]
Q17.
mcq single
+4 / 1
Two astronauts are floating in gravitational free space after having lost contact with their spaceship. The two will
Q18.
mcq single
+4 / 1
The acceleration due to gravity at a height at a height 1 km above the rearth is the same as at a depth d below the surface of earth. Then
Q19.
mcq single
+4 / 1
A capacitor is charged by a battery. The battery is removed and another identical unchanged capacitor is connected in parallel. The total electrostatic energy of resulting system
Q20.
mcq single
+4 / 1
A thin prism having refracting angle 10^(o) is made of glass of refractive infex 1.42. This prism is combined with another thin prism of glass of refractive index 1.7. This combination producess dispersion without deviation. The refracting angle of second prism should be
Q21.
mcq single
+4 / 1
A beam of light from a source L is incident normally on a plane mirror fixed at a certain distance x from the source. The beam is reflected back as a spot on a scale placed just above the source L. When the mirror is rotated through a small angle $$\theta $$, the spot of the light is found to move through a distance y on the scale. The angle $$\theta $$ is given by
Q22.
mcq single
+4 / 1
A particle executes linear simple harmonic motion with an amplitude of 3 cm. When the particle is at 2 cm from the mean position, the magnitude of its velocity is equal to that of its acceleration. Then its time period in second is
Q23.
mcq single
+4 / 1
A spring of force constant k is cut into lengths of ratio 1 : 2 : 3. They are connected in series and the new force constant is K'. Then they are connected in parallel and force constant is k''. Then k' : k'' is
Q24.
mcq single
+4 / 1
Which one of the following represents forward bias diode ?
Q25.
mcq single
+4 / 1
The given electrical network is equivalent to


Q26.
mcq single
+4 / 1
An arrangement of three parallel straight wires placed perpendicular to plane of paper carrying same current $$'I'$$ along the same direction as shown in fogure. Magnitude of force per unit length on the middle wire $$'B'$$ is given by


Q27.
mcq single
+4 / 1
The de-Broglie wavelength of a neutron in thermal equilibrium with heavy water at a temperature T (kelvin) and mass m, is
Q28.
mcq multi
+4 / 1
The photoelectric threshold wavelength of silver is 3250 $$ \times $$ 10^($$-$$10) m. The velocity of the electron ejected from a silver surface by ultraviolet light of wavelength 2536 $$ \times $$ 10^($$-$$10) m is
{Given h = 4.14 $$ \times $$ 10^($$-$$15) eV s and c = 3 $$ \times $$ 10^(8) m s^($$-$$1) ]
Q29.
mcq single
+4 / 1
Preeti reached the metro station and found that the escalator was not working. She walked up the sationary escalator in time t~1~. On another days, if she remains stationary on the the moving escalator, then the escalator takes her up in time t~2~. The time taken by her to walk up on the moving escalator will be
Q30.
mcq single
+4 / 1
The ratio of wavelengths of the last line of Balmer series and the last line of Lyman series is
Q31.
mcq single
+4 / 1
The x and y coordinates of the particle at any time are x = 5t $$-$$ 2t^(2) and y = 10t respectively, where x and y are in metres and t in seconds. The acceleration of the particle at t = 2 s is
Q32.
mcq single
+4 / 1
Which of the following statements are correct ?
(1) Centre of mass of a body always coincides with the centre of gravity of the body.
(2) Centre of mass of a body is the point at which the total gravitational torque on the body is zero.
(3) A couple on a body produces both translational and rotational motion in a body
(4) Mechanical advantage greater than one means that small effort can be used to lift a large load.
Q33.
mcq single
+4 / 1
The two nearest harmonics of a tube closed at one end and open at other end are 220 Hz and 260 Hz. What is the fundamental frequency of the system ?
Q34.
mcq single
+4 / 1
One end of string of length $$l$$ is connected to a particle of mass 'm' and the other end is connected to a small peg on a smooth horizontal table. If the particle moves in circle with speed '$$\upsilon $$', the net force on the particle (directed towards centre) will be (T represents the tension in the string)
Q35.
mcq single
+4 / 1
Two blocks A and B of masses 3m and m respectively are connected by a massless and inextensible string. The whole system is suspended by a massless spring as shown in figure. The magnitudes of acceleration of A and B immediately after the string is cut, are respectively


Q36.
mcq single
+4 / 1
If $$\theta $$~1~ and $$\theta $$~2~ be the apparent angles of dip observed in two vertical planes at right angles to each other, then the true angle of dip $$\theta $$ is given by
Q37.
mcq single
+4 / 1
A 250-turn rectangular coil of length 2.1 cm and width 1.25 cm carries a current of 85 $$\mu $$A and subjected to a magnetic field of strength 0.85 T. Work done for rotating the coil by 180^(o) against the torque is
Q38.
mcq single
+4 / 1
Consider a drop of rain water having mass 1 g falling from a height of 1 km. It hits the ground with a speed of 50 m s^($$-$$1). Take 'g' constant with a value 10 m s^($$-$$2). The work done by the (i) gravitational force and the (ii) resistive force of air is
Q39.
mcq single
+4 / 1
The resistance of a wire is 'R' ohm. If it is melted and stretched to 'n' times its original length. its new resistance will be
Q40.
mcq single
+4 / 1
A potentiometer is an accurate and versatile device to make electrical measurements of EMF because the method involves
Q41.
mcq single
+4 / 1
In an electromagnetic wave in free space that root mean square value of the electric field is E~rms~ = 6 V m^($$-$$1). The peak value of the magnetic field is
Q42.
mcq single
+4 / 1
Figure shows a circuit that contains three identical resistors with resistance R = 9.0 $$\Omega $$ each, two identical inductors with inductance $$L$$ = 2.0 mH each, and an ideal battery with emf $$\varepsilon = 18V$$. The current $$i$$ through the battery just after the switch closed is


Q43.
mcq single
+4 / 1
The ratio of resolving powers of an optical microscope for two wavelength $$\lambda $$~1~ = 4000 $$\mathop A\limits^ \circ $$ and $${\lambda _2}$$ = 6000 $$\mathop A\limits^ \circ $$ is
Q44.
mcq single
+4 / 1
Two polaroids P~1~ and P~2~ are placed with their axis perpendicular to each other. Unpolarised light $$I$$~0~ is incident on P~1~. A third polaroid P~3~ is kept in between P~1~ and P~2~ such that its axis makes an angle 45^(o) with that of P~1~. The intensity of transmitted light through P~2~ is
Q45.
mcq single
+4 / 1
Young's double slit experiment is first performed in air and then in a medium other than air. It is found that 8^(th) bright fringe in the medium lies where 5^(th) dark fringe lies in air. The refractive index of the medium is nearly
Q46.
mcq single
+4 / 1
Two discs of same moment of inertia rotating about their regular axis passing through centre and perpendicular to the plane of disc with angular velocities $${\omega _1}$$ and $${\omega _2}$$. They are brought into contact face to face coinciding the axis of rotation. The expression for loss of energy during this process is
Q47.
mcq single
+4 / 1
A rope is wound around a hollow cylinder of mass 3 kg and radius 40 cm. What is the angular acceleration of the cylinder if the rope is pulled with a force of 30 N?
Q48.
mcq single
+4 / 1
A long solenoid of diameter 0.1 m has 2 $$ \times $$ 10^(4) turns per meter. At the centre of the solenoid, a coil of 100 turns and radius 0.01 m is placed with its axis coinciding with the solenoid axis. The current in the solenoid reduces at a constant rate to 0 A from 4 A in 0.05 s. If the resistance of the coil is 10 $$\pi $$^(2) $$\Omega $$, the total charge flowing through the coil during this time is
Q49.
mcq single
+4 / 1
A physical quantity of the dimensions of length that can be formed out of c, G and $${{{e^2}} \over {4\pi {\varepsilon _0}}}$$ is [c is velocity of light, G is the universal constant of gravitation and e is charge]
Q50.
mcq single
+4 / 1
Thermodynamic processes are indicated in the following diagram.
 Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
 Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
Match the following
Column-1
Column-2
P.
Process I
A.
Adiabatic
Q.
Process II
B.
Isobaric
R.
Process III
C.
Isochoric
S.
Process IV
D.
Isothermal
Q51.
mcq single
+4 / 1
A carnot engine having an efficiency of $${1 \over {10}}$$ as heat engine, is used as a refrigerator. If the work done on the system is 10 J, the amount of energy absorbed from the reservoir at lower temperature is
Q52.
mcq single
+4 / 1
A gas mixture consists of 2 moles of O~2~ and 4 moles of Ar at temperature T. Neglecting all vibrational modes, the total internal energy of the system is
Q53.
mcq single
+4 / 1
The bulk modulus of a spherical object is 'B'. If it is subjected to uniform pressure 'P', the fractional decrease in radius is
Q54.
mcq single
+4 / 1
A spherical black body with a radius of 12 cm radiates 450 watt power at 500 K. If the radius were halved and the temperature doubled, the power radiated in watt would be
Q55.
mcq single
+4 / 1
Two rods A and B of different materials are welded together as shown in figure. Their thermal conductivities are K~1~ and K~2~. The thermal conductivity of the composite will be


Q56.
mcq single
+4 / 1
A U tube with both ends open to the atmosphere, is partially filled with water. Oil, which is immiscible with water, is poured into one side until it stands at a distance of 10 mm above the water level on the other side. Meanwhile the water rises by 65 mm from its original level (see diagram). The density of the oil is
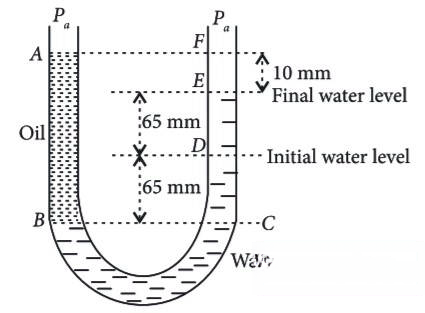

Q57.
mcq single
+4 / 1
The diagrams below show regions of equipotentials.
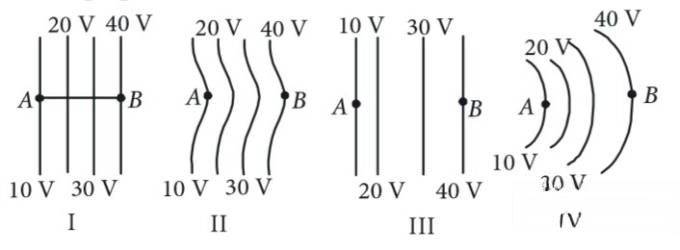 A positive charge is moved from A to B in each diagram.
A positive charge is moved from A to B in each diagram.
 A positive charge is moved from A to B in each diagram.
A positive charge is moved from A to B in each diagram.
Q58.
mcq single
+4 / 1
Suppose the charge of a proton and an electron differ slightly. One of them is $$-$$e, the other is (e + $$\Delta $$e). If the net of electrostatic force and gravitational force between two hydrogen atoms placed at a distance d (musch greater than atomic size) apart is zero, then $$\Delta $$e is of the order of
[Given : mass of hydrogen m~h~ = 1.67 $$ \times $$ 10$$-$$^(27) kg]
Q59.
mcq single
+4 / 1
Two astronauts are floating in gravitational free space after having lost contact with their spaceship. The two will
Q60.
mcq single
+4 / 1
The acceleration due to gravity at a height at a height 1 km above the rearth is the same as at a depth d below the surface of earth. Then
Q61.
mcq single
+4 / 1
A capacitor is charged by a battery. The battery is removed and another identical unchanged capacitor is connected in parallel. The total electrostatic energy of resulting system
Q62.
mcq single
+4 / 1
A thin prism having refracting angle 10^(o) is made of glass of refractive infex 1.42. This prism is combined with another thin prism of glass of refractive index 1.7. This combination producess dispersion without deviation. The refracting angle of second prism should be
Q63.
mcq single
+4 / 1
A beam of light from a source L is incident normally on a plane mirror fixed at a certain distance x from the source. The beam is reflected back as a spot on a scale placed just above the source L. When the mirror is rotated through a small angle $$\theta $$, the spot of the light is found to move through a distance y on the scale. The angle $$\theta $$ is given by
Q64.
mcq single
+4 / 1
A particle executes linear simple harmonic motion with an amplitude of 3 cm. When the particle is at 2 cm from the mean position, the magnitude of its velocity is equal to that of its acceleration. Then its time period in second is
Q65.
mcq single
+4 / 1
A spring of force constant k is cut into lengths of ratio 1 : 2 : 3. They are connected in series and the new force constant is K'. Then they are connected in parallel and force constant is k''. Then k' : k'' is
Q66.
mcq single
+4 / 1
Which one of the following represents forward bias diode ?
Q67.
mcq single
+4 / 1
The given electrical network is equivalent to


Q68.
mcq single
+4 / 1
An arrangement of three parallel straight wires placed perpendicular to plane of paper carrying same current $$'I'$$ along the same direction as shown in fogure. Magnitude of force per unit length on the middle wire $$'B'$$ is given by


Q69.
mcq single
+4 / 1
The de-Broglie wavelength of a neutron in thermal equilibrium with heavy water at a temperature T (kelvin) and mass m, is
Q70.
mcq multi
+4 / 1
The photoelectric threshold wavelength of silver is 3250 $$ \times $$ 10^($$-$$10) m. The velocity of the electron ejected from a silver surface by ultraviolet light of wavelength 2536 $$ \times $$ 10^($$-$$10) m is
{Given h = 4.14 $$ \times $$ 10^($$-$$15) eV s and c = 3 $$ \times $$ 10^(8) m s^($$-$$1) ]
Q71.
mcq single
+4 / 1
Preeti reached the metro station and found that the escalator was not working. She walked up the sationary escalator in time t~1~. On another days, if she remains stationary on the the moving escalator, then the escalator takes her up in time t~2~. The time taken by her to walk up on the moving escalator will be
Q72.
mcq single
+4 / 1
The ratio of wavelengths of the last line of Balmer series and the last line of Lyman series is
Q73.
mcq single
+4 / 1
The x and y coordinates of the particle at any time are x = 5t $$-$$ 2t^(2) and y = 10t respectively, where x and y are in metres and t in seconds. The acceleration of the particle at t = 2 s is
Q74.
mcq single
+4 / 1
Which of the following statements are correct ?
(1) Centre of mass of a body always coincides with the centre of gravity of the body.
(2) Centre of mass of a body is the point at which the total gravitational torque on the body is zero.
(3) A couple on a body produces both translational and rotational motion in a body
(4) Mechanical advantage greater than one means that small effort can be used to lift a large load.
Q75.
mcq single
+4 / 1
The two nearest harmonics of a tube closed at one end and open at other end are 220 Hz and 260 Hz. What is the fundamental frequency of the system ?
Q76.
mcq single
+4 / 1
One end of string of length $$l$$ is connected to a particle of mass 'm' and the other end is connected to a small peg on a smooth horizontal table. If the particle moves in circle with speed '$$\upsilon $$', the net force on the particle (directed towards centre) will be (T represents the tension in the string)
Q77.
mcq single
+4 / 1
Two blocks A and B of masses 3m and m respectively are connected by a massless and inextensible string. The whole system is suspended by a massless spring as shown in figure. The magnitudes of acceleration of A and B immediately after the string is cut, are respectively
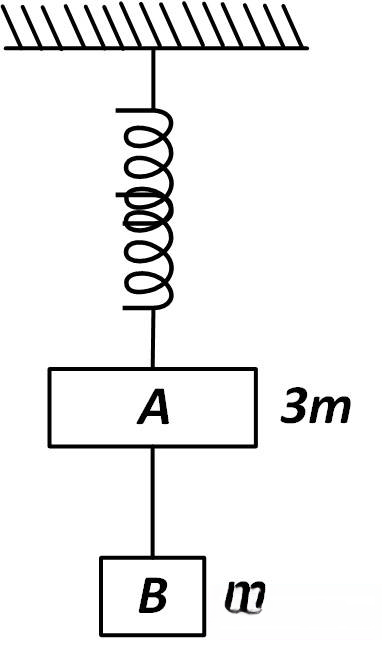

Q78.
mcq single
+4 / 1
If $$\theta $$~1~ and $$\theta $$~2~ be the apparent angles of dip observed in two vertical planes at right angles to each other, then the true angle of dip $$\theta $$ is given by
Q79.
mcq single
+4 / 1
A 250-turn rectangular coil of length 2.1 cm and width 1.25 cm carries a current of 85 $$\mu $$A and subjected to a magnetic field of strength 0.85 T. Work done for rotating the coil by 180^(o) against the torque is
Q80.
mcq single
+4 / 1
Consider a drop of rain water having mass 1 g falling from a height of 1 km. It hits the ground with a speed of 50 m s^($$-$$1). Take 'g' constant with a value 10 m s^($$-$$2). The work done by the (i) gravitational force and the (ii) resistive force of air is
Q81.
mcq single
+4 / 1
The resistance of a wire is 'R' ohm. If it is melted and stretched to 'n' times its original length. its new resistance will be
Q82.
mcq single
+4 / 1
A potentiometer is an accurate and versatile device to make electrical measurements of EMF because the method involves
Q83.
mcq single
+4 / 1
In an electromagnetic wave in free space that root mean square value of the electric field is E~rms~ = 6 V m^($$-$$1). The peak value of the magnetic field is
Q84.
mcq single
+4 / 1
Figure shows a circuit that contains three identical resistors with resistance R = 9.0 $$\Omega $$ each, two identical inductors with inductance $$L$$ = 2.0 mH each, and an ideal battery with emf $$\varepsilon = 18V$$. The current $$i$$ through the battery just after the switch closed is


Biology
Biology
Q1.
mcq single
+4 / 1
Select the mismatch :
Q2.
mcq single
+4 / 1
Which of the following in sewage treatment removes suspended solids ?
Q3.
mcq single
+4 / 1
Which of the following is correctly matched for the product produced by them ?
Q4.
mcq single
+4 / 1
Which ecosystem has the maximum biomass ?
Q5.
mcq single
+4 / 1
Which one from those given below is the period for Mendel’s hybridisation experiments ?
Q6.
mcq single
+4 / 1
The genotypes of a Husband and Wife are I^(A)I^(B) and I^(A)i.
Among the blood types of their children, how many different genotypes and phenotypes are possible?
Q7.
mcq single
+4 / 1
A disease caused by an autosomal primary non-disjunction is :
Q8.
mcq single
+4 / 1
Thalassemia and sickle cell anemia are caused due to a problem in globin molecule synthesis. Select the correct statement.
Q9.
mcq single
+4 / 1
Among the following characters, which one was
not considered by Mendel in his experiments on
pea?
Q10.
mcq single
+4 / 1
Frog’s heart when taken out of the body continues to beat for sometime.
Select the best option from the following statements.
(a) Frog is a poikilotherm
(b) Frog does not have any coronary circulation
(c) Heart is “myogenic” in nature
(d) Heart is autoexcitable
Q11.
mcq single
+4 / 1
Select the correct route for the passage of sperms in male frogs :
Q12.
mcq single
+4 / 1
Phosphoenol pyruvate (PEP) is the primary CO~2~ acceptor in
Q13.
mcq single
+4 / 1
With reference to factors affecting the rate of photosynthesis, which of the following statements is not correct?
Q14.
mcq single
+4 / 1
Which among these is the correct combination of aquatic mammals?
Q15.
mcq single
+4 / 1
Which of the following represents order of 'Horse'?
Q16.
mcq single
+4 / 1
In case of poriferans, the spongocoel is lined
with flagellated cells called
Q17.
mcq single
+4 / 1
Important characteristic that hemichordates share with chordates is
Q18.
mcq single
+4 / 1
In case of proiferans, the spongocoel is lined with flagellated cells called
Q19.
mcq single
+4 / 1
Zygotic meiosis is characteristic of
Q20.
mcq single
+4 / 1
An example of colonial alga is
Q21.
mcq single
+4 / 1
Life cycles of *Ectocarpus* and *Fucus* respectively are
Q22.
mcq single
+4 / 1
Select the mismatch
Q23.
mcq single
+4 / 1
Lungs are made up of air-filed sacs, the alveoli. The do not collapse even after forceful expiration, because of
Q24.
mcq single
+4 / 1
Receptor sites for neurotransmitters are present on
Q25.
mcq single
+4 / 1
Myelin sheath is produced by
Q26.
mcq single
+4 / 1
Good vishion depends on adequate intake of carotene rich food.
Select the best option from the following statements.
(1) Vitamins A derivatives are formed from carotene,
(2) The photopigments are embedded in the membrane discs of the inner segment.
(3) Retinal is a derivative of vitamin A.
(4) Retinal is a light absorbing part of all the visual photopigments.
Q27.
mcq single
+4 / 1
Root hairs develop from the region of :
Q28.
mcq single
+4 / 1
The vascular cambium normally gives rise to :
Q29.
mcq single
+4 / 1
Identify the wrong statement in context of heartwood :
Q30.
mcq single
+4 / 1
Which of the following is made up of dead cells ?
Q31.
mcq single
+4 / 1
Which of the following cell organelles is responsible for extracting energy from carbohydrates to form ATP?
Q32.
mcq single
+4 / 1
If there are 999 bases in an RNA that codes for a protein with 333 amino acids, and the base at position 901 is
deleted such that the length of the RNA becomes 998 bases, how many codons will be altered ?
Q33.
mcq single
+4 / 1
Which of the following RNAs should be most abundant in animal cell ?
Q34.
mcq single
+4 / 1
During DNA replication, Okazaki fragments are used to elongate
Q35.
mcq single
+4 / 1
The association of histone H~1~ with a nucleosome indicates :
Q36.
mcq single
+4 / 1
The final proof for DNA as the genetic material came from the experiments of
Q37.
mcq single
+4 / 1
Spliceosomes are not found in cells of :
Q38.
mcq single
+4 / 1
Transplantation of tissues/ organs fails often due to non-acceptance by the patient's body. Which type of immuneresponse is responsible for such rejections ?
Q39.
mcq single
+4 / 1
MALT constitutes about ________ percent of the lymphoid tissue in human body
Q40.
mcq single
+4 / 1
Plants which produce characteristic pneumatophores and show vivipary belong to :
Q41.
mcq single
+4 / 1
Asymptote in a logistic growth curve is obtained when :
Q42.
mcq single
+4 / 1
Presence of plants arranged into well defined vertical layers depending on their height can be seen best in :
Q43.
mcq single
+4 / 1
Mycorrhizae are the example of :
Q44.
mcq single
+4 / 1
The hepatic portal vein drains blood to liver from
Q45.
mcq single
+4 / 1
Adult human RBCs are enucleate. Which of the following statement(s) is/are most appropriate explanation for this feature?
(1) They do not need to reproduce.
(2) They are somatic cells.
(3) They do not metabolise.
(4) All their internal space is available for oxygen transport.
Q46.
mcq single
+4 / 1
Anaphase Promoting Complex (APC) is a protein degradation machinery necessary for proper mitosis of animal cell. If APC is defective in a human cell, which of the following is expected to occur?
Q47.
mcq single
+4 / 1
Which of the following options gives the correct sequence of events during mitosis?
Q48.
mcq single
+4 / 1
Capacitation occurs in
Q49.
mcq single
+4 / 1
Hypersecretion of growth hormone in adults does not cause further increase in height, because
Q50.
mcq single
+4 / 1
GnRH, a hypothalamic hormone, needed in reproduction, acts on
Q51.
mcq single
+4 / 1
A temporary endocrine gland in the human body is
Q52.
mcq single
+4 / 1
Which one of the following is related to Ex-situ conservation of threatened animals and plants ?
Q53.
mcq single
+4 / 1
Alexander Von Humbolt described for the first time :
Q54.
mcq single
+4 / 1
The region of Biosphere Reserve which is legally protected and where no human activity is allowed is known as :
Q55.
mcq single
+4 / 1
Fruit and leaf drop at early stages can be prevented by the application of
Q56.
mcq single
+4 / 1
Which statement is wrong for Krebs' cycle?
Q57.
mcq single
+4 / 1
Out of 'X' pairs of ribs in humans only 'Y' pairs are true ribs. Select the option that correctly represents values of X and Y and provides their explanation.
Q58.
mcq single
+4 / 1
The pivot joint between atlas and axis is a type of
Q59.
mcq single
+4 / 1
Viroids differ from viruses in having
Q60.
mcq single
+4 / 1
Which among the following are the smallest living cells, known without a definite cell wall, pathogenic to plants as well as animals and can survise without oxygen?
Q61.
mcq single
+4 / 1
Which of the following are found in extreme saline conditions?
Q62.
mcq single
+4 / 1
DNA replication in bacteria occurs
Q63.
mcq single
+4 / 1
Which of the following components provides sticky character to the bacterial cell?
Q64.
mcq single
+4 / 1
A decrease in blood pressure/volume will not cause the release of
Q65.
mcq single
+4 / 1
Which of the following statements is correct?
Q66.
mcq single
+4 / 1
Match the following sexually transmitted diseases (**Column – I**) with their causative agent (**Column – II**) and
select the correct option
Column – I
Column – II
(a) Gonorrhea
(i) HIV
(b) Syphilis
(ii) Neisseria
(c) Genital Warts
(iii) Treponema
(d) AIDS
(iv) Human papilloma – virus
Q67.
mcq single
+4 / 1
The function of copper ions in copper releasing IUD's is :
Q68.
mcq multi
+4 / 1
In case of a couple where the male is having a very low sperm count, which technique will be suitable for fertilization ?
Q69.
mcq single
+4 / 1
Double fertilisation is exhibited by
Q70.
mcq single
+4 / 1
Functional megaspore in an angiosperm develops into an
Q71.
mcq single
+4 / 1
Flowers which have single ovule in the ovary and are packed into inflorescence are usualy pollinated by
Q72.
mcq single
+4 / 1
Attractants and rewards are required for
Q73.
mcq single
+4 / 1
A dioecious flowering plant prevents both
Q74.
mcq single
+4 / 1
In *Bougainvillea*, throns are the modifications of
Q75.
mcq single
+4 / 1
Coconut fruit is a
Q76.
mcq single
+4 / 1
The morphological nature of the edible part of coconut is
Q77.
mcq single
+4 / 1
Which of the following are not polymeric?
Q78.
mcq single
+4 / 1
Which of the following statements is correct with reference to enzymes?
Q79.
mcq single
+4 / 1
The process of separation and purification of expressed protein before marketing is called :
Q80.
mcq single
+4 / 1
What is the criterion for DNA fragments movement on agarose gel during gel electrophoresis ?
Q81.
mcq single
+4 / 1
The DNA fragments separated on an agarose gel can be visualised after staining with
Q82.
mcq single
+4 / 1
DNA fragments are
Q83.
mcq single
+4 / 1
A gene whose expression helps to identify transformed cell is known as
Q84.
mcq single
+4 / 1
Select the mismatch :
Q85.
mcq single
+4 / 1
Which of the following in sewage treatment removes suspended solids ?
Q86.
mcq single
+4 / 1
Which of the following is correctly matched for the product produced by them ?
Q87.
mcq single
+4 / 1
Which ecosystem has the maximum biomass ?
Q88.
mcq single
+4 / 1
Which one from those given below is the period for Mendel’s hybridisation experiments ?
Q89.
mcq single
+4 / 1
The genotypes of a Husband and Wife are I^(A)I^(B) and I^(A)i.
Among the blood types of their children, how many different genotypes and phenotypes are possible?
Q90.
mcq single
+4 / 1
A disease caused by an autosomal primary non-disjunction is :
Q91.
mcq single
+4 / 1
Thalassemia and sickle cell anemia are caused due to a problem in globin molecule synthesis. Select the correct statement.
Q92.
mcq single
+4 / 1
Among the following characters, which one was
not considered by Mendel in his experiments on
pea?
Q93.
mcq single
+4 / 1
Frog’s heart when taken out of the body continues to beat for sometime.
Select the best option from the following statements.
(a) Frog is a poikilotherm
(b) Frog does not have any coronary circulation
(c) Heart is “myogenic” in nature
(d) Heart is autoexcitable
Q94.
mcq single
+4 / 1
Select the correct route for the passage of sperms in male frogs :
Q95.
mcq single
+4 / 1
Phosphoenol pyruvate (PEP) is the primary CO~2~ acceptor in
Q96.
mcq single
+4 / 1
With reference to factors affecting the rate of photosynthesis, which of the following statements is not correct?
Q97.
mcq single
+4 / 1
Which among these is the correct combination of aquatic mammals?
Q98.
mcq single
+4 / 1
Which of the following represents order of 'Horse'?
Q99.
mcq single
+4 / 1
In case of poriferans, the spongocoel is lined
with flagellated cells called
Q100.
mcq single
+4 / 1
Important characteristic that hemichordates share with chordates is
Q101.
mcq single
+4 / 1
In case of proiferans, the spongocoel is lined with flagellated cells called
Q102.
mcq single
+4 / 1
Zygotic meiosis is characteristic of
Q103.
mcq single
+4 / 1
An example of colonial alga is
Q104.
mcq single
+4 / 1
Life cycles of *Ectocarpus* and *Fucus* respectively are
Q105.
mcq single
+4 / 1
Select the mismatch
Q106.
mcq single
+4 / 1
Lungs are made up of air-filed sacs, the alveoli. The do not collapse even after forceful expiration, because of
Q107.
mcq single
+4 / 1
Receptor sites for neurotransmitters are present on
Q108.
mcq single
+4 / 1
Myelin sheath is produced by
Q109.
mcq single
+4 / 1
Good vishion depends on adequate intake of carotene rich food.
Select the best option from the following statements.
(1) Vitamins A derivatives are formed from carotene,
(2) The photopigments are embedded in the membrane discs of the inner segment.
(3) Retinal is a derivative of vitamin A.
(4) Retinal is a light absorbing part of all the visual photopigments.
Q110.
mcq single
+4 / 1
Root hairs develop from the region of :
Q111.
mcq single
+4 / 1
The vascular cambium normally gives rise to :
Q112.
mcq single
+4 / 1
Identify the wrong statement in context of heartwood :
Q113.
mcq single
+4 / 1
Which of the following is made up of dead cells ?
Q114.
mcq single
+4 / 1
Which of the following cell organelles is responsible for extracting energy from carbohydrates to form ATP?
Q115.
mcq single
+4 / 1
If there are 999 bases in an RNA that codes for a protein with 333 amino acids, and the base at position 901 is
deleted such that the length of the RNA becomes 998 bases, how many codons will be altered ?
Q116.
mcq single
+4 / 1
Which of the following RNAs should be most abundant in animal cell ?
Q117.
mcq single
+4 / 1
During DNA replication, Okazaki fragments are used to elongate
Q118.
mcq single
+4 / 1
The association of histone H~1~ with a nucleosome indicates :
Q119.
mcq single
+4 / 1
The final proof for DNA as the genetic material came from the experiments of
Q120.
mcq single
+4 / 1
Spliceosomes are not found in cells of :
Q121.
mcq single
+4 / 1
Transplantation of tissues/ organs fails often due to non-acceptance by the patient's body. Which type of immuneresponse is responsible for such rejections ?
Q122.
mcq single
+4 / 1
MALT constitutes about ________ percent of the lymphoid tissue in human body
Q123.
mcq single
+4 / 1
Plants which produce characteristic pneumatophores and show vivipary belong to :
Q124.
mcq single
+4 / 1
Asymptote in a logistic growth curve is obtained when :
Q125.
mcq single
+4 / 1
Presence of plants arranged into well defined vertical layers depending on their height can be seen best in :
Q126.
mcq single
+4 / 1
Mycorrhizae are the example of :
Q127.
mcq single
+4 / 1
The hepatic portal vein drains blood to liver from
Q128.
mcq single
+4 / 1
Adult human RBCs are enucleate. Which of the following statement(s) is/are most appropriate explanation for this feature?
(1) They do not need to reproduce.
(2) They are somatic cells.
(3) They do not metabolise.
(4) All their internal space is available for oxygen transport.
Q129.
mcq single
+4 / 1
Anaphase Promoting Complex (APC) is a protein degradation machinery necessary for proper mitosis of animal cell. If APC is defective in a human cell, which of the following is expected to occur?
Q130.
mcq single
+4 / 1
Which of the following options gives the correct sequence of events during mitosis?
Q131.
mcq single
+4 / 1
Capacitation occurs in
Q132.
mcq single
+4 / 1
Hypersecretion of growth hormone in adults does not cause further increase in height, because
Q133.
mcq single
+4 / 1
GnRH, a hypothalamic hormone, needed in reproduction, acts on
Q134.
mcq single
+4 / 1
A temporary endocrine gland in the human body is
Q135.
mcq single
+4 / 1
Which one of the following is related to Ex-situ conservation of threatened animals and plants ?
Q136.
mcq single
+4 / 1
Alexander Von Humbolt described for the first time :
Q137.
mcq single
+4 / 1
The region of Biosphere Reserve which is legally protected and where no human activity is allowed is known as :
Q138.
mcq single
+4 / 1
Fruit and leaf drop at early stages can be prevented by the application of
Q139.
mcq single
+4 / 1
Which statement is wrong for Krebs' cycle?
Q140.
mcq single
+4 / 1
Out of 'X' pairs of ribs in humans only 'Y' pairs are true ribs. Select the option that correctly represents values of X and Y and provides their explanation.
Q141.
mcq single
+4 / 1
The pivot joint between atlas and axis is a type of
Q142.
mcq single
+4 / 1
Viroids differ from viruses in having
Q143.
mcq single
+4 / 1
Which among the following are the smallest living cells, known without a definite cell wall, pathogenic to plants as well as animals and can survise without oxygen?
Q144.
mcq single
+4 / 1
Which of the following are found in extreme saline conditions?
Q145.
mcq single
+4 / 1
DNA replication in bacteria occurs
Q146.
mcq single
+4 / 1
Which of the following components provides sticky character to the bacterial cell?
Q147.
mcq single
+4 / 1
A decrease in blood pressure/volume will not cause the release of
Q148.
mcq single
+4 / 1
Which of the following statements is correct?
Q149.
mcq single
+4 / 1
Match the following sexually transmitted diseases (**Column – I**) with their causative agent (**Column – II**) and
select the correct option
Column – I
Column – II
(a) Gonorrhea
(i) HIV
(b) Syphilis
(ii) Neisseria
(c) Genital Warts
(iii) Treponema
(d) AIDS
(iv) Human papilloma – virus
Q150.
mcq single
+4 / 1
The function of copper ions in copper releasing IUD's is :
Q151.
mcq multi
+4 / 1
In case of a couple where the male is having a very low sperm count, which technique will be suitable for fertilization ?
Q152.
mcq single
+4 / 1
Double fertilisation is exhibited by
Q153.
mcq single
+4 / 1
Functional megaspore in an angiosperm develops into an
Q154.
mcq single
+4 / 1
Flowers which have single ovule in the ovary and are packed into inflorescence are usualy pollinated by
Q155.
mcq single
+4 / 1
Attractants and rewards are required for
Q156.
mcq single
+4 / 1
A dioecious flowering plant prevents both
Q157.
mcq single
+4 / 1
In *Bougainvillea*, throns are the modifications of
Q158.
mcq single
+4 / 1
Coconut fruit is a
Q159.
mcq single
+4 / 1
The morphological nature of the edible part of coconut is
Q160.
mcq single
+4 / 1
Which of the following are not polymeric?
Q161.
mcq single
+4 / 1
Which of the following statements is correct with reference to enzymes?
Q162.
mcq single
+4 / 1
The process of separation and purification of expressed protein before marketing is called :
Q163.
mcq single
+4 / 1
What is the criterion for DNA fragments movement on agarose gel during gel electrophoresis ?
Q164.
mcq single
+4 / 1
The DNA fragments separated on an agarose gel can be visualised after staining with
Q165.
mcq single
+4 / 1
DNA fragments are
Q166.
mcq single
+4 / 1
A gene whose expression helps to identify transformed cell is known as