neet-2019
NEET 2019
MEDICAL 2019 Previous Year
3 hDuration
632Total Marks
316Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 316 across 3 section(s); maximum marks: 632.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
The correct structure of tribromooctaoxide is :
Q2.
mcq single
+4 / 1
The manganate and permanganate ions are tetrahedral due to :
Q3.
mcq single
+4 / 1
The number of sigma ($$\sigma $$) and pi ($$\pi $$) bonds in pent-2-en-4-yne is -
Q4.
mcq single
+4 / 1
Which of the following diatomic molecular species has only $$\pi $$ bonds according to Molecular Orbital Theory?
Q5.
mcq single
+4 / 1
pH of a saturated solution of Ca(OH)~2~ is 9. The solubility product (K~sp~) of Ca(OH)~2~ is :
Q6.
mcq single
+4 / 1
Which will make basic buffer?
Q7.
mcq single
+4 / 1
Conjugate base for Bronsted acids H~2~O and HF are :
Q8.
mcq single
+4 / 1
If the rate constant for a first order reaction is k, the time (t) required for the completion of 99 % of the
reaction is given by :
Q9.
mcq single
+4 / 1
For the chemical reaction
N~2~(g) + 3H~2~(g) ⇌ 2NH~3~(g)
the correct option is :
Q10.
mcq single
+4 / 1
Which of the following reactions are disproportionation reaction?
**
(a) 2Cu^(+) $$ \to $$ Cu^(2+)
+ Cu^(0)
(b) 3MnO~4~^(2+) + 4H^(+) $$ \to $$ 2MnO~4~^(-) + MnO~2~ + 2H~2~O
(c) 2KMnO~4~ $$\buildrel \Delta \over
\longrightarrow $$ K~2~MnO~4~ + MnO~2~ + O~2~
(d) 2MnO~4~^(-) + 3Mn^(2+) + 2H~2~O $$ \to $$ 5MnO~2~ + 4$${H^ \oplus }$$
Select the correct** option from the following :
Q11.
mcq single
+4 / 1
For an ideal solution, the **correct** option is :
Q12.
mcq single
+4 / 1
The mixture that forms maximum boiling
azeotrope is :
Q13.
mcq single
+4 / 1
The number of moles of hydrogen molecules required to produce 20 moles of ammonia through Haber’s
process is :
Q14.
mcq single
+4 / 1
For the second period elements the correct increasing order of first ionization enthalpy is :
Q15.
mcq single
+4 / 1
In which case change in entropy is negative?
Q16.
mcq single
+4 / 1
Under isothermal condition, a gas at 300 K expands from 0.1 L to 0.25 L against a constant external pressure
of 2 bar. The work done by the gas is [Given that 1 L bar = 100 J]
Q17.
mcq single
+4 / 1
Identify the incorrect statement related to PCl~5~ from the following :
Q18.
mcq single
+4 / 1
Which is the correct thermal stability order for H~2~E (E = O, S, Se, Te and Po)?
Q19.
mcq single
+4 / 1
Which of the following is **incorrect** statement?
Q20.
mcq single
+4 / 1
Match the following :
(a) Pure nitrogen
(i) Chlorine
(b) Haber process
(ii) Sulphuric acid
(c) Contact process
(iii) Ammonia
(d) Deacon’s process
(iv) Sodium azide or Barium azide
Which of the following is the correct option?
Q21.
mcq single
+4 / 1
Match the Xenon compounds in Column-I with its structure in Column-II and assign the correct code :


Q22.
mcq single
+4 / 1
Which of the following species is **not** stable?
Q23.
mcq single
+4 / 1
What is the **correct** electronic configuration of the central atom in K~4~[Fe(CN)~6~] based on crystal field theory?
Q24.
mcq single
+4 / 1
The compound that is most difficult to protonate is :
Q25.
mcq single
+4 / 1
The structure of intermediate A in the following reaction, is :


Q26.
mcq single
+4 / 1
Among the following, the reaction that proceeds through an electrophilic substitution, is :
Q27.
mcq single
+4 / 1
The most suitable reagent for the following conversion, is :


Q28.
mcq single
+4 / 1
An alkene “A” on reaction with O~3~ and Zn + H~2~O gives propanone and ethanal in equimolar ratio. Addition
of HCl to alkene “A” gives “B” as the major product. The structure of product “B” is :
Q29.
mcq single
+4 / 1
The non-essential amino acid among the following is :
Q30.
mcq single
+4 / 1
For the cell reaction
2Fe^(3+)(aq) + 2I^(–)
(aq) $$ \to $$ 2Fe^(2+)(aq) + I~2~(aq)
$${E_{cell}^\Theta }$$ = 0.24 V at 298 K. The standard Gibbs energy ($$\Delta $$~r~G^(o)) of the cell reaction is :
[Given that Faraday constant F = 96500 C mol^(–1)]
Q31.
mcq single
+4 / 1
For a cell involving one electron $$E_{cell}^\Theta $$ = 0.59 V at 298 K, the equilibrium constant for the cell reaction is :
[Given that $${{2.303RT} \over F}$$ = 0.059 V at T = 298 K ]
Q32.
mcq single
+4 / 1
The correct order of the basic strength of methyl substituted amines in aqueous solution is :
Q33.
mcq single
+4 / 1
The major product of the following reaction is :


Q34.
mcq single
+4 / 1
4d, 5p, 5f and 6p orbitals are arranged in the order of decreasing energy. The correct option is :
Q35.
mcq single
+4 / 1
Which of the following series of transitions in the spectrum of hydrogen atom falls in visible region?
Q36.
mcq single
+4 / 1
The correct structure of tribromooctaoxide is :
Q37.
mcq single
+4 / 1
The manganate and permanganate ions are tetrahedral due to :
Q38.
mcq single
+4 / 1
The number of sigma ($$\sigma $$) and pi ($$\pi $$) bonds in pent-2-en-4-yne is -
Q39.
mcq single
+4 / 1
Which of the following diatomic molecular species has only $$\pi $$ bonds according to Molecular Orbital Theory?
Q40.
mcq single
+4 / 1
pH of a saturated solution of Ca(OH)~2~ is 9. The solubility product (K~sp~) of Ca(OH)~2~ is :
Q41.
mcq single
+4 / 1
Which will make basic buffer?
Q42.
mcq single
+4 / 1
Conjugate base for Bronsted acids H~2~O and HF are :
Q43.
mcq single
+4 / 1
If the rate constant for a first order reaction is k, the time (t) required for the completion of 99 % of the
reaction is given by :
Q44.
mcq single
+4 / 1
For the chemical reaction
N~2~(g) + 3H~2~(g) ⇌ 2NH~3~(g)
the correct option is :
Q45.
mcq single
+4 / 1
Which of the following reactions are disproportionation reaction?
**
(a) 2Cu^(+) $$ \to $$ Cu^(2+)
+ Cu^(0)
(b) 3MnO~4~^(2+) + 4H^(+) $$ \to $$ 2MnO~4~^(-) + MnO~2~ + 2H~2~O
(c) 2KMnO~4~ $$\buildrel \Delta \over
\longrightarrow $$ K~2~MnO~4~ + MnO~2~ + O~2~
(d) 2MnO~4~^(-) + 3Mn^(2+) + 2H~2~O $$ \to $$ 5MnO~2~ + 4$${H^ \oplus }$$
Select the correct** option from the following :
Q46.
mcq single
+4 / 1
For an ideal solution, the **correct** option is :
Q47.
mcq single
+4 / 1
The mixture that forms maximum boiling
azeotrope is :
Q48.
mcq single
+4 / 1
The number of moles of hydrogen molecules required to produce 20 moles of ammonia through Haber’s
process is :
Q49.
mcq single
+4 / 1
For the second period elements the correct increasing order of first ionization enthalpy is :
Q50.
mcq single
+4 / 1
In which case change in entropy is negative?
Q51.
mcq single
+4 / 1
Under isothermal condition, a gas at 300 K expands from 0.1 L to 0.25 L against a constant external pressure
of 2 bar. The work done by the gas is [Given that 1 L bar = 100 J]
Q52.
mcq single
+4 / 1
Identify the incorrect statement related to PCl~5~ from the following :
Q53.
mcq single
+4 / 1
Which is the correct thermal stability order for H~2~E (E = O, S, Se, Te and Po)?
Q54.
mcq single
+4 / 1
Which of the following is **incorrect** statement?
Q55.
mcq single
+4 / 1
Match the following :
(a) Pure nitrogen
(i) Chlorine
(b) Haber process
(ii) Sulphuric acid
(c) Contact process
(iii) Ammonia
(d) Deacon’s process
(iv) Sodium azide or Barium azide
Which of the following is the correct option?
Q56.
mcq single
+4 / 1
Match the Xenon compounds in Column-I with its structure in Column-II and assign the correct code :


Q57.
mcq single
+4 / 1
Which of the following species is **not** stable?
Q58.
mcq single
+4 / 1
What is the **correct** electronic configuration of the central atom in K~4~[Fe(CN)~6~] based on crystal field theory?
Q59.
mcq single
+4 / 1
The compound that is most difficult to protonate is :
Q60.
mcq single
+4 / 1
The structure of intermediate A in the following reaction, is :


Q61.
mcq single
+4 / 1
Among the following, the reaction that proceeds through an electrophilic substitution, is :
Q62.
mcq single
+4 / 1
The most suitable reagent for the following conversion, is :


Q63.
mcq single
+4 / 1
An alkene “A” on reaction with O~3~ and Zn + H~2~O gives propanone and ethanal in equimolar ratio. Addition
of HCl to alkene “A” gives “B” as the major product. The structure of product “B” is :
Q64.
mcq single
+4 / 1
The non-essential amino acid among the following is :
Q65.
mcq single
+4 / 1
For the cell reaction
2Fe^(3+)(aq) + 2I^(–)
(aq) $$ \to $$ 2Fe^(2+)(aq) + I~2~(aq)
$${E_{cell}^\Theta }$$ = 0.24 V at 298 K. The standard Gibbs energy ($$\Delta $$~r~G^(o)) of the cell reaction is :
[Given that Faraday constant F = 96500 C mol^(–1)]
Q66.
mcq single
+4 / 1
For a cell involving one electron $$E_{cell}^\Theta $$ = 0.59 V at 298 K, the equilibrium constant for the cell reaction is :
[Given that $${{2.303RT} \over F}$$ = 0.059 V at T = 298 K ]
Q67.
mcq single
+4 / 1
The correct order of the basic strength of methyl substituted amines in aqueous solution is :
Q68.
mcq single
+4 / 1
The major product of the following reaction is :


Q69.
mcq single
+4 / 1
4d, 5p, 5f and 6p orbitals are arranged in the order of decreasing energy. The correct option is :
Q70.
mcq single
+4 / 1
Which of the following series of transitions in the spectrum of hydrogen atom falls in visible region?
Physics
Physics
Q1.
mcq single
+4 / 1
In a double slit experiment, when light of wavelength 400 nm was used, the angular width of the first minima formed on a screen placed 1m away, was found to be 0.2^(o). What will be the angular width of the first minima, ($$\mu $$~water~ = 4/3) if the entire experimental apparatus is immersed in water?
Q2.
mcq single
+4 / 1
A solid cylinder of mass 2 kg and radius 4 cm is rotating about its axis at the rate of 3 rpm. The torque required to stop after 2$$\pi $$ revolutions is :
Q3.
mcq single
+4 / 1
Two particles A and B are moving in uniform circular motion in concentric circles of radii r~A~ and r~B~ with speed v~A~ and v~B~ respecitively. Their time period of rotation is the same. The ratio of angular speed of A to that of B will be :
Q4.
mcq single
+4 / 1
A disc of radius 2 m and mass 100 kg rolls on a horizontal floor. Its centre of mass has speed of 20 cm/s. How much work is needed to stop it?
Q5.
mcq single
+4 / 1
A 800 turn coil of effective area 0.05 m^(2) is kept. perpendicular to a magnetic filed 5 × 10^(–5) T. When the plane of the coil is rotated by 90^(o) around any of its coplanar axis in 0.1 s, the emf induced in the coil will be :
Q6.
mcq single
+4 / 1
In which of the following devices, the eddy current effect is **not** used?
Q7.
mcq single
+4 / 1
In an experiment, the percentage of error occurred in the measurement of physical quantities A, B, C and D are 1%, 2 %, 3 %and 4 % respectively. Then the maximum percentage of error in the measurement X, where
X = $${{{A^2}{B^{1/2}}} \over {{C^{1/3}}{D^3}}}$$, will be :
Q8.
mcq single
+4 / 1
Increase in tempertaure of a gas filled in a container would lead to :
Q9.
mcq single
+4 / 1
In which of the following processes, heat is neither absorbed nor released by a system?
Q10.
mcq single
+4 / 1
A copper rod of 88 cm and an aluminium rod of unknown length have their increase in length independent of increase in tmperature. The length of aluminium rod is : ($$\alpha $$~Cu~ = 1.7 × 10^(–5) K^(–1) and
$$\alpha $$~Al~ = 2.2 × 10^(–5) K^(–1))
Q11.
mcq single
+4 / 1
When a block of mass M is suspended by a long wire of length L, the length of the wire becomes (L + $$l$$). The elastic potential energy stored in the extended wire is :
Q12.
mcq single
+4 / 1
A small hole of area of cross-section 2 mm^(2) present near the bottom of a fully filled open tank of height 2 m. Taking g = 10 m/s^(2), the rate of flow of water through the open hole would be nearly :
Q13.
mcq single
+4 / 1
The unit of thermal conductivity is :
Q14.
mcq single
+4 / 1
A soap bubble, having radius of 1 mm, is blown from a detergent solution having a surface tension of 2.5 $$ \times $$ 10^(–2) N/m. The pressure inside the bubble equals at a point Z~0~ below the free surface of water in a container. Taking g = 10 m/s^(2), density of water = 10^(3) kg/m^(3), the value of Z~0~ is :
Q15.
mcq single
+4 / 1
Two point charges A and B, having charges +Q and – Q respectively, are placed at certain distance apart and force acting between them is F. If 25% charge of A is transferred to B, then force between the charges becomes :
Q16.
mcq single
+4 / 1
A hollow metal sphere of radius R is uniformly charged. The electric field due to the sphere at a distance r from the centre :
Q17.
mcq single
+4 / 1
Two parallel infinite line charges with linear charge densities +$$\lambda $$ C/m and -$$\lambda $$ C/m are placed at a distance of 2R in free space. What is the electric field mid-way between the two line charges?
Q18.
mcq single
+4 / 1
The work done to raise a mass m from the surface of the earth to a height h, which is equal to the radius of the earth, is :
Q19.
mcq single
+4 / 1
A body weight 200 N on the surface of earth. How much will it weight half way down to the centre of the earth?
Q20.
mcq single
+4 / 1
A parallel plate capacitor of capacitance 20 $$\mu $$F is being charged by a voltage source whose potential is changing at the rate of 3V/s. The conduction current through the connecting wires, and the displacement current through the plates of the capacitor, would be, respectively :
Q21.
mcq single
+4 / 1
Pick the **wrong** answer in the context with rainbow.
Q22.
mcq single
+4 / 1
In total internal reflection when the angle of incidence is equal to the critical angle for the pair of media in contact, what will be angle of refraction?
Q23.
mcq single
+4 / 1
Two similar thin equi-convex lenses, of focal length f each, are kept coaxially in contact with each other such that the focal length of the combination is F~1~. When the space between the two lenses is filled with glycerin (which has the same refractive index (m = 1.5) as that of glass) then the equivalent focal length is F~2~. The ratio F~1~ : F~2~ will be :
Q24.
mcq single
+4 / 1
The displacement of a particle executing simple harmonic motion is given by
y = A~0~ + A sin$$\omega $$t + B cos$$\omega $$t.
Then the amplitude of its oscillation is given by :
Q25.
mcq single
+4 / 1
The radius of circle, the period of revolution, initial position and sense of revolution are indicated is the figure.
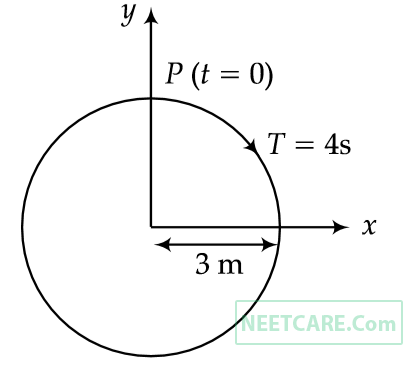 y- projection of the radius vector of rotating particle P is :
y- projection of the radius vector of rotating particle P is :
 y- projection of the radius vector of rotating particle P is :
y- projection of the radius vector of rotating particle P is :
Q26.
mcq single
+4 / 1
Average velocity of a particle executing SHM in one complete vibration is :
Q27.
mcq single
+4 / 1
The **correct** Boolean operation represented by the circuit diagram drawn is :


Q28.
mcq single
+4 / 1
For a p-type semiconductor, which of the following statements is **true**?
Q29.
mcq single
+4 / 1
A cylinderical conductor of radius R is carrying a constant current. The plot of the magnitude of the magnetic field. B with the distane d from the centre of the conductor, is correctly represented by the figure :
Q30.
mcq single
+4 / 1
Ionized hydrogen atoms and $$\alpha $$-particles with same momenta enters perpendicular to a constant magnetic field, B. The ratio of their radii of their paths r~H~ : r~$$\alpha $$~ will be :
Q31.
mcq single
+4 / 1
An electron is accelerated through a potential difference of 10,000 V. Its de Broglie wavelength is (nearly) : (m~e~ = 9 $$ \times $$ 10^(–31) kg)
Q32.
mcq single
+4 / 1
The speed of a swimmer in still water is 20 m/s. The speed of river water is 10 m/s and is flowing due east. If he is standing on the south bank and wishes to cross the river along the shortest path, the angle at which he should make his strokes w.r.t. north is given by :
Q33.
mcq single
+4 / 1
$$\alpha $$-particale consists of :
Q34.
mcq single
+4 / 1
The total energy of an electron in an atom in an orbit is –3.4 eV. Its kinetic and potential energies are, respectively.
Q35.
mcq single
+4 / 1
When an object is shot from the bottom of a long smooth inclined plane kept at an angle 60^(o) with horizontal, it can travel a distance x~1~ along the plane. But when the inclination is decreased to 30^(o) and the same object is shot with the same velocity, it can travel x~2~ distance. Then x~1~ : x~2~ will be :
Q36.
mcq single
+4 / 1
A particle moving with velocity
$$\overrightarrow V $$ is acted by three forces shown by the vector triangle PQR. The velocity of the particle will :


Q37.
mcq single
+4 / 1
Body A of mass 4m moving with speed u collides with another body B of mass 2 m, at rest. The collision is head on and elastic in nature. After the collision the fraction of energy lost by the colliding body A is :
Q38.
mcq single
+4 / 1
A block of mass 10 kg is in contact against the inner wall of a hollow cylindrical drum of radius 1 m. The coefficient of friction between the block and the inner wall of the cylinder is 0.1. The minimum angular velocity needed for the cylinder to keep the block stationary when the cylinder is vertical and rotating about its axis, will be : (g = 10 m/s^(2))
Q39.
mcq single
+4 / 1
A mass m is attached to a thin wire and whirled in a vertical circle. The wire is most likely to break when :
Q40.
mcq single
+4 / 1
At a point A on the earth’s surface the angle of dip, $$\delta $$ = + 25^(o). At a point B on the earth's surface the angle of dip, $$\delta $$ = – 25^(o). We can interpret that :
Q41.
mcq single
+4 / 1
A force F = 20 + 10y acts on a particle in y-direction where F is in newton and y in meter. Work done by this force to move the particle from y = 0 to y = 1 m is :
Q42.
mcq single
+4 / 1
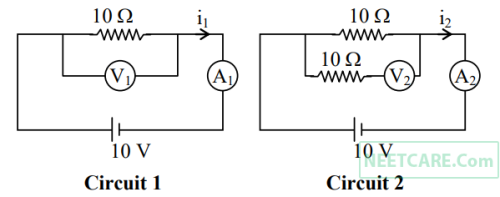
In the circuits shown below, the readings of voltmeters and the ammeters will be :

Q43.
mcq single
+4 / 1
Which of the following acts as a circuit protection device?
Q44.
mcq single
+4 / 1
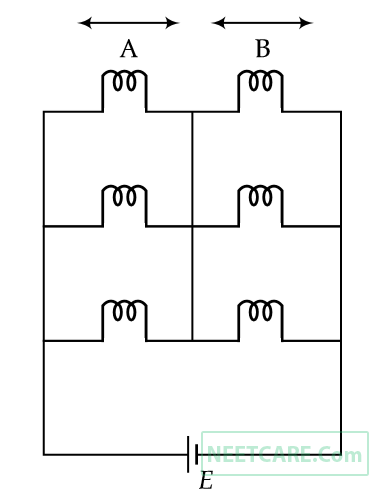
Six similar bulbs are connected as shown in the figure with a DC source of emf E, and zero internal resistance.
The ratio of power consumption by the bulbs when (i) all are glowing and (ii) in the situation when two from section A and one from section B are glowing, will be :

Q45.
mcq single
+4 / 1
Which colour of the light has the longest wavelength?
Q46.
mcq single
+4 / 1
In a double slit experiment, when light of wavelength 400 nm was used, the angular width of the first minima formed on a screen placed 1m away, was found to be 0.2^(o). What will be the angular width of the first minima, ($$\mu $$~water~ = 4/3) if the entire experimental apparatus is immersed in water?
Q47.
mcq single
+4 / 1
A solid cylinder of mass 2 kg and radius 4 cm is rotating about its axis at the rate of 3 rpm. The torque required to stop after 2$$\pi $$ revolutions is :
Q48.
mcq single
+4 / 1
Two particles A and B are moving in uniform circular motion in concentric circles of radii r~A~ and r~B~ with speed v~A~ and v~B~ respecitively. Their time period of rotation is the same. The ratio of angular speed of A to that of B will be :
Q49.
mcq single
+4 / 1
A disc of radius 2 m and mass 100 kg rolls on a horizontal floor. Its centre of mass has speed of 20 cm/s. How much work is needed to stop it?
Q50.
mcq single
+4 / 1
A 800 turn coil of effective area 0.05 m^(2) is kept. perpendicular to a magnetic filed 5 × 10^(–5) T. When the plane of the coil is rotated by 90^(o) around any of its coplanar axis in 0.1 s, the emf induced in the coil will be :
Q51.
mcq single
+4 / 1
In which of the following devices, the eddy current effect is **not** used?
Q52.
mcq single
+4 / 1
In an experiment, the percentage of error occurred in the measurement of physical quantities A, B, C and D are 1%, 2 %, 3 %and 4 % respectively. Then the maximum percentage of error in the measurement X, where
X = $${{{A^2}{B^{1/2}}} \over {{C^{1/3}}{D^3}}}$$, will be :
Q53.
mcq single
+4 / 1
Increase in tempertaure of a gas filled in a container would lead to :
Q54.
mcq single
+4 / 1
In which of the following processes, heat is neither absorbed nor released by a system?
Q55.
mcq single
+4 / 1
A copper rod of 88 cm and an aluminium rod of unknown length have their increase in length independent of increase in tmperature. The length of aluminium rod is : ($$\alpha $$~Cu~ = 1.7 × 10^(–5) K^(–1) and
$$\alpha $$~Al~ = 2.2 × 10^(–5) K^(–1))
Q56.
mcq single
+4 / 1
When a block of mass M is suspended by a long wire of length L, the length of the wire becomes (L + $$l$$). The elastic potential energy stored in the extended wire is :
Q57.
mcq single
+4 / 1
A small hole of area of cross-section 2 mm^(2) present near the bottom of a fully filled open tank of height 2 m. Taking g = 10 m/s^(2), the rate of flow of water through the open hole would be nearly :
Q58.
mcq single
+4 / 1
The unit of thermal conductivity is :
Q59.
mcq single
+4 / 1
A soap bubble, having radius of 1 mm, is blown from a detergent solution having a surface tension of 2.5 $$ \times $$ 10^(–2) N/m. The pressure inside the bubble equals at a point Z~0~ below the free surface of water in a container. Taking g = 10 m/s^(2), density of water = 10^(3) kg/m^(3), the value of Z~0~ is :
Q60.
mcq single
+4 / 1
Two point charges A and B, having charges +Q and – Q respectively, are placed at certain distance apart and force acting between them is F. If 25% charge of A is transferred to B, then force between the charges becomes :
Q61.
mcq single
+4 / 1
A hollow metal sphere of radius R is uniformly charged. The electric field due to the sphere at a distance r from the centre :
Q62.
mcq single
+4 / 1
Two parallel infinite line charges with linear charge densities +$$\lambda $$ C/m and -$$\lambda $$ C/m are placed at a distance of 2R in free space. What is the electric field mid-way between the two line charges?
Q63.
mcq single
+4 / 1
The work done to raise a mass m from the surface of the earth to a height h, which is equal to the radius of the earth, is :
Q64.
mcq single
+4 / 1
A body weight 200 N on the surface of earth. How much will it weight half way down to the centre of the earth?
Q65.
mcq single
+4 / 1
A parallel plate capacitor of capacitance 20 $$\mu $$F is being charged by a voltage source whose potential is changing at the rate of 3V/s. The conduction current through the connecting wires, and the displacement current through the plates of the capacitor, would be, respectively :
Q66.
mcq single
+4 / 1
Pick the **wrong** answer in the context with rainbow.
Q67.
mcq single
+4 / 1
In total internal reflection when the angle of incidence is equal to the critical angle for the pair of media in contact, what will be angle of refraction?
Q68.
mcq single
+4 / 1
Two similar thin equi-convex lenses, of focal length f each, are kept coaxially in contact with each other such that the focal length of the combination is F~1~. When the space between the two lenses is filled with glycerin (which has the same refractive index (m = 1.5) as that of glass) then the equivalent focal length is F~2~. The ratio F~1~ : F~2~ will be :
Q69.
mcq single
+4 / 1
The displacement of a particle executing simple harmonic motion is given by
y = A~0~ + A sin$$\omega $$t + B cos$$\omega $$t.
Then the amplitude of its oscillation is given by :
Q70.
mcq single
+4 / 1
The radius of circle, the period of revolution, initial position and sense of revolution are indicated is the figure.
 y- projection of the radius vector of rotating particle P is :
y- projection of the radius vector of rotating particle P is :
 y- projection of the radius vector of rotating particle P is :
y- projection of the radius vector of rotating particle P is :
Q71.
mcq single
+4 / 1
Average velocity of a particle executing SHM in one complete vibration is :
Q72.
mcq single
+4 / 1
The **correct** Boolean operation represented by the circuit diagram drawn is :


Q73.
mcq single
+4 / 1
For a p-type semiconductor, which of the following statements is **true**?
Q74.
mcq single
+4 / 1
A cylinderical conductor of radius R is carrying a constant current. The plot of the magnitude of the magnetic field. B with the distane d from the centre of the conductor, is correctly represented by the figure :
Q75.
mcq single
+4 / 1
Ionized hydrogen atoms and $$\alpha $$-particles with same momenta enters perpendicular to a constant magnetic field, B. The ratio of their radii of their paths r~H~ : r~$$\alpha $$~ will be :
Q76.
mcq single
+4 / 1
An electron is accelerated through a potential difference of 10,000 V. Its de Broglie wavelength is (nearly) : (m~e~ = 9 $$ \times $$ 10^(–31) kg)
Q77.
mcq single
+4 / 1
The speed of a swimmer in still water is 20 m/s. The speed of river water is 10 m/s and is flowing due east. If he is standing on the south bank and wishes to cross the river along the shortest path, the angle at which he should make his strokes w.r.t. north is given by :
Q78.
mcq single
+4 / 1
$$\alpha $$-particale consists of :
Q79.
mcq single
+4 / 1
The total energy of an electron in an atom in an orbit is –3.4 eV. Its kinetic and potential energies are, respectively.
Q80.
mcq single
+4 / 1
When an object is shot from the bottom of a long smooth inclined plane kept at an angle 60^(o) with horizontal, it can travel a distance x~1~ along the plane. But when the inclination is decreased to 30^(o) and the same object is shot with the same velocity, it can travel x~2~ distance. Then x~1~ : x~2~ will be :
Q81.
mcq single
+4 / 1
A particle moving with velocity
$$\overrightarrow V $$ is acted by three forces shown by the vector triangle PQR. The velocity of the particle will :


Q82.
mcq single
+4 / 1
Body A of mass 4m moving with speed u collides with another body B of mass 2 m, at rest. The collision is head on and elastic in nature. After the collision the fraction of energy lost by the colliding body A is :
Q83.
mcq single
+4 / 1
A block of mass 10 kg is in contact against the inner wall of a hollow cylindrical drum of radius 1 m. The coefficient of friction between the block and the inner wall of the cylinder is 0.1. The minimum angular velocity needed for the cylinder to keep the block stationary when the cylinder is vertical and rotating about its axis, will be : (g = 10 m/s^(2))
Q84.
mcq single
+4 / 1
A mass m is attached to a thin wire and whirled in a vertical circle. The wire is most likely to break when :
Q85.
mcq single
+4 / 1
At a point A on the earth’s surface the angle of dip, $$\delta $$ = + 25^(o). At a point B on the earth's surface the angle of dip, $$\delta $$ = – 25^(o). We can interpret that :
Q86.
mcq single
+4 / 1
A force F = 20 + 10y acts on a particle in y-direction where F is in newton and y in meter. Work done by this force to move the particle from y = 0 to y = 1 m is :
Q87.
mcq single
+4 / 1
In the circuits shown below, the readings of voltmeters and the ammeters will be :


Q88.
mcq single
+4 / 1
Which of the following acts as a circuit protection device?
Q89.
mcq single
+4 / 1
Six similar bulbs are connected as shown in the figure with a DC source of emf E, and zero internal resistance.
The ratio of power consumption by the bulbs when (i) all are glowing and (ii) in the situation when two from section A and one from section B are glowing, will be :


Q90.
mcq single
+4 / 1
Which colour of the light has the longest wavelength?
Biology
Biology
Q1.
mcq single
+4 / 1
Which of the following is a commercial blood cholesterol lowering agent?
Q2.
mcq single
+4 / 1
Match the following organisms with the products they produce :
**
(a) Lactobacillus
(i) Cheese
(b) Saccharomyces cerevisiae
(ii) Curd
(c) Aspergillus nige
(iii) Citric Acid
(d) Acetobacter aceti
(iv) Bread
(v) Acetic Acid
Select the correct** option.
Q3.
mcq single
+4 / 1
Which of the following can be used as a biocontrol agent in the treatment of plant disease?
Q4.
mcq single
+4 / 1
Select the correct group of biocontrol agents.
Q5.
mcq single
+4 / 1
Thiobacillus is a group of bacteria helpful in carrying out :
Q6.
mcq single
+4 / 1
What triggers activation of protoxin to active Bt toxin of *Bacillus thuringiensis* in boll worm?
Q7.
mcq single
+4 / 1
Which of the following is the most important cause for animals and plants being driven to extinction?
Q8.
mcq single
+4 / 1
Which of the following ecological pyramids is generally inverted?
Q9.
mcq single
+4 / 1
What map unit (Centimorgan) is adopted in the construction of genetic maps?
Q10.
mcq single
+4 / 1
Select the **incorrect** statement.
Q11.
mcq single
+4 / 1
A gene locus has two alleles A, a. If the frequency of dominant allele A is 0.4, then what will be the
frequency of homozygous dominant, heterozygous and homozygous recessive individuals in the population?
Q12.
mcq single
+4 / 1
In *Antirrhinum* (Snapdragon), a red flower was crossed with a white flower and in F~1~ generation, pink
flowers were obtained. When pink flowers were selfed, the F~2~ generation showed white, red and pink
flowers. Choose the **incorrect** statement from the following :
Q13.
mcq single
+4 / 1
What is the genetic disorder in which an individual has an overall masculine development, gynaecomastia,
and is sterile?
Q14.
mcq single
+4 / 1
The frequency of recombination between gene pairs on the same chromosome as a measure of the distance
between genes was explained by :
Q15.
mcq single
+4 / 1
Select the **correct** sequence of organs in the alimentary canal of cockroach starting from mouth :
Q16.
mcq single
+4 / 1
The ciliated epithelial cells are required to move particles or mucus in a specific direction. In humans, these
cells are mainly present in :
Q17.
mcq single
+4 / 1
Consider following features:
(a) Organ system level of organisation
(b) Bilateral symmetry
(c) True coelomates with segmentation of body
Select the correct option of animal groups which possess all the above characteristics
Q18.
mcq single
+4 / 1
Match the following organisms with their respective characteristics:
(a)
Pila
(i)
Flame cells
(b)
Bombyx
(ii)
Comb plates
(c)
Pleurobrachia
(iii)
Radula
(d)
Taenia
(iv)
Malpighian
Select the correct option from the following :
Q19.
mcq single
+4 / 1
From evolutionary point of view, retention of the female gametophyte with developing young embryo on the
parent sporophyte for some time, is first observed in :
Q20.
mcq single
+4 / 1
Tidal Volume and Expiratory Reserve Volume of an athlete is 500 mL and 1000 mL respectively. What will
be his Expiratory Capacity if the Residual Volume is 1200 mL?
Q21.
mcq single
+4 / 1
Which of the following glucose transporters is insulin-dependent?
Q22.
mcq single
+4 / 1
Which of the following statements in correct?
Q23.
mcq single
+4 / 1
Which part of the brain is responsible for thermoregulation?
Q24.
mcq single
+4 / 1
Phloem in gymnosperms lacks :
Q25.
mcq single
+4 / 1
Which of the statements given below is not true about formation of Annual Rings in trees?
Q26.
mcq single
+4 / 1
What is the direction of movement of sugars in phloem?
Q27.
mcq single
+4 / 1
Which of the following statements regarding mitochondria is **incorrect**?
Q28.
mcq single
+4 / 1
Which of the following pair of organelles does not contains DNA?
Q29.
mcq single
+4 / 1
Which of the following statements is **not** correct?
Q30.
mcq single
+4 / 1
The shorter and longer arms of a submetacentric chromosome are referred to as:
Q31.
mcq single
+4 / 1
Which of the following features of genetic code does allow bacteria to produce human insulin by
recombinant DNA technology?
Q32.
mcq single
+4 / 1
Purines found both in DNA and RNA are:
Q33.
mcq single
+4 / 1
Match the following genes of the Lac operon with their respective produces :
**
(a) i gene
(i) $$\beta $$-galactosidase
(b) z gene
(ii) Permease
(c) a gene
(iii) Repressor
(d) y gene
(iv) Transacetylase
Select the correct** option.
Q34.
mcq single
+4 / 1
Expressed Sequence Tags (ESTs) refers to :
Q35.
mcq single
+4 / 1
Under which of the following conditions will there be no change in the reading frame of following mRNA?
5' AACAGCGGUGCUAUU 3'
Q36.
mcq single
+4 / 1
Colostrum, the yellowish fluid, secreted by mother during the initial days of lactation is very essential to
impart immunity to the newborn infants because it contains :
Q37.
mcq single
+4 / 1
Identify the correct pair representing the causative agent of typhoid fever and the confirmatory test for
typhoid.
Q38.
mcq single
+4 / 1
Which of the following sexually transmitted diseases is **not** completely curable ?
Q39.
mcq single
+4 / 1
Which of the following immune responses is responsible for rejection of kidney graft?
Q40.
mcq single
+4 / 1
Drug called ‘Heroin’ is synthesized by :
Q41.
mcq single
+4 / 1
The Earth summit held in Rio de Janeiro in 1992 was called :
Q42.
mcq single
+4 / 1
Match Column-I with Column –II.
**
Column - I
Column - II
(a) Saprophyte
(i) Symbiotic association
or fungi with plant roots
(b) Parasite
(ii) Decomposition of dead
organic materials
(c) Lichens
(iii)Living on living plants of animals
(d) Mycorrhiza
(iv) Symbiotic association of algae and fungi
Choose the correct** answer from the options given below :
Q43.
mcq single
+4 / 1
What would be the heart rate of a person if the cardiac output is 5L, blood volume in the ventricles at the end
of diastole is 100 mL and at the end of ventricular systole is 50 mL ?
Q44.
mcq single
+4 / 1
Match the Column-I with Column-II :
 Select the correct option.
Select the correct option.
 Select the correct option.
Select the correct option.
Q45.
mcq single
+4 / 1
Cells in G~0~ phase :
Q46.
mcq single
+4 / 1
The correct sequence of phases of cell cycle is :
Q47.
mcq single
+4 / 1
Select the correct sequence for transport of sperm cells in male reproductive system :
Q48.
mcq single
+4 / 1
Extrusion of second polar body from egg nucleus occurs :
Q49.
mcq single
+4 / 1
Match the following hormones with the respective disease :
(a) Insulin
(i) Addison’s disease
(b) Thyroxin
(ii) Diabetes insipidus
(c) Corticoids
(iii) Acromegaly
(d) Growth Hormones
(iv) Goitre
(v) Diabetes mellitus
Select the correct option.
Q50.
mcq single
+4 / 1
How does steroid hormone influence the cellular activities?
Q51.
mcq single
+4 / 1
Which one of the following is **not** a method of in situ conservation of biodiversity?
Q52.
mcq single
+4 / 1
What is the site of perception of photoperiod necessary for induction of flowering in plants?
Q53.
mcq single
+4 / 1
Respiratory Quotient (RQ) value of tripalmitin is :
Q54.
mcq single
+4 / 1
Conversion of glucose to glucose-6 phosphate, the first irreversible reaction of glycolysis, is catalyzed by
Q55.
mcq single
+4 / 1
Select the correct option :
Q56.
mcq single
+4 / 1
Which of the following muscular disorders is inherited?
Q57.
mcq single
+4 / 1
Which of the following statements is **incorrect**?
Q58.
mcq single
+4 / 1
Which of the following statement is **incorrect**?
Q59.
mcq single
+4 / 1
Select the **correctly** written scientific name of Mango which was first described by Carolus Linnaeus :
Q60.
mcq single
+4 / 1
Variations caused by mutation, as proposed by Hugo de Vries, are :
Q61.
mcq single
+4 / 1
In a species, the weight of newborn ranges from 2 to 5 kg. 97% of the newborn with an average weight
between 3 to 3.3 kg survive whereas 99% of the infants born with weights from 2 to 2.5 kg or 4.5 to 5kg die.
Which type of selection process is taking place?
Q62.
mcq single
+4 / 1
The concept of ''*Omnis cellula-e cellula*'' regarding cell division was first proposed by :
Q63.
mcq single
+4 / 1
Match the hominids with their correct brain size :
(a) Homo habilis
(i) 900 cc
(b) Homo neanderthalensis
(ii) 1350 cc
(c) Homo erectus
(iii) 650-800 cc
(d) Homo sapiens
(iv) 1400 cc
Select the correct option.
Q64.
mcq single
+4 / 1
Use of an artificial kindney during hemodialysis may result in :
**
(a) Nitrogenous waste build-up in the body
(b) Non –elimination of excess potassium ions
(c) Reduced absorption of calcium ions from gastro-intestinal tract
(d) Reduced RBC production
Which of the following options is the most appropriate**?
Q65.
mcq single
+4 / 1
Which of the following factors is responsible for the formation of concentrated urine?
Q66.
mcq single
+4 / 1
Which of the following contraceptive methods do involve a role of hormone?
Q67.
mcq single
+4 / 1
Select the horomone-releasing Intra-Uterine Devices.
Q68.
mcq single
+4 / 1
Pinus seed **cannot** germinate and establish without fungal association. This is because :
Q69.
mcq single
+4 / 1
What is the fate of the male gametes discharged in the synergid?
Q70.
mcq single
+4 / 1
Persistent nucellus in the seed is known as
Q71.
mcq single
+4 / 1
Which one of the following statements regarding post-fertilization development in flowering plants is
incorrect?
Q72.
mcq single
+4 / 1
In some plants, the female gamete develops into embryo without fertilization. This phenomenon is known as :
Q73.
mcq single
+4 / 1
Placentation, in which ovules develop on the inner wall of the ovary or in peripheral part, is :
Q74.
mcq single
+4 / 1
Grass leaves curl inward during very dry weather. Select the most appropriate reason from the following :
Q75.
mcq single
+4 / 1
Consider the following statements :
**
(A)** Coenzyme of metal ion that is tightly bound to enzyme protein is called prosthetic group.
**(B)** A complete catalytic active enzyme with its bound prosthetic group is called apoenzyme
**
Select the correct** option
Q76.
mcq single
+4 / 1
Concanavalin A is :
Q77.
mcq single
+4 / 1
DNA precipitation out of a mixture of bimolecules can be achieved by treatment with :
Q78.
mcq single
+4 / 1
Following statements describe the characteristics of the enzyme Restriction Endonuclease. Identify the
**incorrect** statement.
Q79.
mcq single
+4 / 1
Which of the following is a commercial blood cholesterol lowering agent?
Q80.
mcq single
+4 / 1
Match the following organisms with the products they produce :
**
(a) Lactobacillus
(i) Cheese
(b) Saccharomyces cerevisiae
(ii) Curd
(c) Aspergillus nige
(iii) Citric Acid
(d) Acetobacter aceti
(iv) Bread
(v) Acetic Acid
Select the correct** option.
Q81.
mcq single
+4 / 1
Which of the following can be used as a biocontrol agent in the treatment of plant disease?
Q82.
mcq single
+4 / 1
Select the correct group of biocontrol agents.
Q83.
mcq single
+4 / 1
Thiobacillus is a group of bacteria helpful in carrying out :
Q84.
mcq single
+4 / 1
What triggers activation of protoxin to active Bt toxin of *Bacillus thuringiensis* in boll worm?
Q85.
mcq single
+4 / 1
Which of the following is the most important cause for animals and plants being driven to extinction?
Q86.
mcq single
+4 / 1
Which of the following ecological pyramids is generally inverted?
Q87.
mcq single
+4 / 1
What map unit (Centimorgan) is adopted in the construction of genetic maps?
Q88.
mcq single
+4 / 1
Select the **incorrect** statement.
Q89.
mcq single
+4 / 1
A gene locus has two alleles A, a. If the frequency of dominant allele A is 0.4, then what will be the
frequency of homozygous dominant, heterozygous and homozygous recessive individuals in the population?
Q90.
mcq single
+4 / 1
In *Antirrhinum* (Snapdragon), a red flower was crossed with a white flower and in F~1~ generation, pink
flowers were obtained. When pink flowers were selfed, the F~2~ generation showed white, red and pink
flowers. Choose the **incorrect** statement from the following :
Q91.
mcq single
+4 / 1
What is the genetic disorder in which an individual has an overall masculine development, gynaecomastia,
and is sterile?
Q92.
mcq single
+4 / 1
The frequency of recombination between gene pairs on the same chromosome as a measure of the distance
between genes was explained by :
Q93.
mcq single
+4 / 1
Select the **correct** sequence of organs in the alimentary canal of cockroach starting from mouth :
Q94.
mcq single
+4 / 1
The ciliated epithelial cells are required to move particles or mucus in a specific direction. In humans, these
cells are mainly present in :
Q95.
mcq single
+4 / 1
Consider following features:
(a) Organ system level of organisation
(b) Bilateral symmetry
(c) True coelomates with segmentation of body
Select the correct option of animal groups which possess all the above characteristics
Q96.
mcq single
+4 / 1
Match the following organisms with their respective characteristics:
(a)
Pila
(i)
Flame cells
(b)
Bombyx
(ii)
Comb plates
(c)
Pleurobrachia
(iii)
Radula
(d)
Taenia
(iv)
Malpighian
Select the correct option from the following :
Q97.
mcq single
+4 / 1
From evolutionary point of view, retention of the female gametophyte with developing young embryo on the
parent sporophyte for some time, is first observed in :
Q98.
mcq single
+4 / 1
Tidal Volume and Expiratory Reserve Volume of an athlete is 500 mL and 1000 mL respectively. What will
be his Expiratory Capacity if the Residual Volume is 1200 mL?
Q99.
mcq single
+4 / 1
Which of the following glucose transporters is insulin-dependent?
Q100.
mcq single
+4 / 1
Which of the following statements in correct?
Q101.
mcq single
+4 / 1
Which part of the brain is responsible for thermoregulation?
Q102.
mcq single
+4 / 1
Phloem in gymnosperms lacks :
Q103.
mcq single
+4 / 1
Which of the statements given below is not true about formation of Annual Rings in trees?
Q104.
mcq single
+4 / 1
What is the direction of movement of sugars in phloem?
Q105.
mcq single
+4 / 1
Which of the following statements regarding mitochondria is **incorrect**?
Q106.
mcq single
+4 / 1
Which of the following pair of organelles does not contains DNA?
Q107.
mcq single
+4 / 1
Which of the following statements is **not** correct?
Q108.
mcq single
+4 / 1
The shorter and longer arms of a submetacentric chromosome are referred to as:
Q109.
mcq single
+4 / 1
Which of the following features of genetic code does allow bacteria to produce human insulin by
recombinant DNA technology?
Q110.
mcq single
+4 / 1
Purines found both in DNA and RNA are:
Q111.
mcq single
+4 / 1
Match the following genes of the Lac operon with their respective produces :
**
(a) i gene
(i) $$\beta $$-galactosidase
(b) z gene
(ii) Permease
(c) a gene
(iii) Repressor
(d) y gene
(iv) Transacetylase
Select the correct** option.
Q112.
mcq single
+4 / 1
Expressed Sequence Tags (ESTs) refers to :
Q113.
mcq single
+4 / 1
Under which of the following conditions will there be no change in the reading frame of following mRNA?
5' AACAGCGGUGCUAUU 3'
Q114.
mcq single
+4 / 1
Colostrum, the yellowish fluid, secreted by mother during the initial days of lactation is very essential to
impart immunity to the newborn infants because it contains :
Q115.
mcq single
+4 / 1
Identify the correct pair representing the causative agent of typhoid fever and the confirmatory test for
typhoid.
Q116.
mcq single
+4 / 1
Which of the following sexually transmitted diseases is **not** completely curable ?
Q117.
mcq single
+4 / 1
Which of the following immune responses is responsible for rejection of kidney graft?
Q118.
mcq single
+4 / 1
Drug called ‘Heroin’ is synthesized by :
Q119.
mcq single
+4 / 1
The Earth summit held in Rio de Janeiro in 1992 was called :
Q120.
mcq single
+4 / 1
Match Column-I with Column –II.
**
Column - I
Column - II
(a) Saprophyte
(i) Symbiotic association
or fungi with plant roots
(b) Parasite
(ii) Decomposition of dead
organic materials
(c) Lichens
(iii)Living on living plants of animals
(d) Mycorrhiza
(iv) Symbiotic association of algae and fungi
Choose the correct** answer from the options given below :
Q121.
mcq single
+4 / 1
What would be the heart rate of a person if the cardiac output is 5L, blood volume in the ventricles at the end
of diastole is 100 mL and at the end of ventricular systole is 50 mL ?
Q122.
mcq single
+4 / 1
Match the Column-I with Column-II :
 Select the correct option.
Select the correct option.
 Select the correct option.
Select the correct option.
Q123.
mcq single
+4 / 1
Cells in G~0~ phase :
Q124.
mcq single
+4 / 1
The correct sequence of phases of cell cycle is :
Q125.
mcq single
+4 / 1
Select the correct sequence for transport of sperm cells in male reproductive system :
Q126.
mcq single
+4 / 1
Extrusion of second polar body from egg nucleus occurs :
Q127.
mcq single
+4 / 1
Match the following hormones with the respective disease :
(a) Insulin
(i) Addison’s disease
(b) Thyroxin
(ii) Diabetes insipidus
(c) Corticoids
(iii) Acromegaly
(d) Growth Hormones
(iv) Goitre
(v) Diabetes mellitus
Select the correct option.
Q128.
mcq single
+4 / 1
How does steroid hormone influence the cellular activities?
Q129.
mcq single
+4 / 1
Which one of the following is **not** a method of in situ conservation of biodiversity?
Q130.
mcq single
+4 / 1
What is the site of perception of photoperiod necessary for induction of flowering in plants?
Q131.
mcq single
+4 / 1
Respiratory Quotient (RQ) value of tripalmitin is :
Q132.
mcq single
+4 / 1
Conversion of glucose to glucose-6 phosphate, the first irreversible reaction of glycolysis, is catalyzed by
Q133.
mcq single
+4 / 1
Select the correct option :
Q134.
mcq single
+4 / 1
Which of the following muscular disorders is inherited?
Q135.
mcq single
+4 / 1
Which of the following statements is **incorrect**?
Q136.
mcq single
+4 / 1
Which of the following statement is **incorrect**?
Q137.
mcq single
+4 / 1
Select the **correctly** written scientific name of Mango which was first described by Carolus Linnaeus :
Q138.
mcq single
+4 / 1
Variations caused by mutation, as proposed by Hugo de Vries, are :
Q139.
mcq single
+4 / 1
In a species, the weight of newborn ranges from 2 to 5 kg. 97% of the newborn with an average weight
between 3 to 3.3 kg survive whereas 99% of the infants born with weights from 2 to 2.5 kg or 4.5 to 5kg die.
Which type of selection process is taking place?
Q140.
mcq single
+4 / 1
The concept of ''*Omnis cellula-e cellula*'' regarding cell division was first proposed by :
Q141.
mcq single
+4 / 1
Match the hominids with their correct brain size :
(a) Homo habilis
(i) 900 cc
(b) Homo neanderthalensis
(ii) 1350 cc
(c) Homo erectus
(iii) 650-800 cc
(d) Homo sapiens
(iv) 1400 cc
Select the correct option.
Q142.
mcq single
+4 / 1
Use of an artificial kindney during hemodialysis may result in :
**
(a) Nitrogenous waste build-up in the body
(b) Non –elimination of excess potassium ions
(c) Reduced absorption of calcium ions from gastro-intestinal tract
(d) Reduced RBC production
Which of the following options is the most appropriate**?
Q143.
mcq single
+4 / 1
Which of the following factors is responsible for the formation of concentrated urine?
Q144.
mcq single
+4 / 1
Which of the following contraceptive methods do involve a role of hormone?
Q145.
mcq single
+4 / 1
Select the horomone-releasing Intra-Uterine Devices.
Q146.
mcq single
+4 / 1
Pinus seed **cannot** germinate and establish without fungal association. This is because :
Q147.
mcq single
+4 / 1
What is the fate of the male gametes discharged in the synergid?
Q148.
mcq single
+4 / 1
Persistent nucellus in the seed is known as
Q149.
mcq single
+4 / 1
Which one of the following statements regarding post-fertilization development in flowering plants is
incorrect?
Q150.
mcq single
+4 / 1
In some plants, the female gamete develops into embryo without fertilization. This phenomenon is known as :
Q151.
mcq single
+4 / 1
Placentation, in which ovules develop on the inner wall of the ovary or in peripheral part, is :
Q152.
mcq single
+4 / 1
Grass leaves curl inward during very dry weather. Select the most appropriate reason from the following :
Q153.
mcq single
+4 / 1
Consider the following statements :
**
(A)** Coenzyme of metal ion that is tightly bound to enzyme protein is called prosthetic group.
**(B)** A complete catalytic active enzyme with its bound prosthetic group is called apoenzyme
**
Select the correct** option
Q154.
mcq single
+4 / 1
Concanavalin A is :
Q155.
mcq single
+4 / 1
DNA precipitation out of a mixture of bimolecules can be achieved by treatment with :
Q156.
mcq single
+4 / 1
Following statements describe the characteristics of the enzyme Restriction Endonuclease. Identify the
**incorrect** statement.