NEET 2020 Phase 1
MEDICAL 2020 Previous Year
3 hDuration
640Total Marks
320Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 320 across 3 section(s); maximum marks: 640.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Identify the incorrect statement.
Q2.
mcq single
+4 / 1
Which of the following set of molecules will have zero dipole moment?
Q3.
mcq single
+4 / 1
Identify a molecule which does not exist.
Q4.
mcq single
+4 / 1
Find out the solubility of Ni(OH)~2~ in 0.1M NaOH. Given that the ionic product of Ni(OH)~2~ is 2 $$ \times $$ 10^(-15).
Q5.
mcq single
+4 / 1
The rate constant for a first order reaction is 4.606 $$ \times $$ 10^(-3) s^(-1). The time required to reduce 2.0 g of the reactant to 0.2 g is :
Q6.
mcq single
+4 / 1
An increase in the concentration of the reactants of a reaction leads to change in :
Q7.
mcq single
+4 / 1
What is the change in oxidation number of carbon in the following reaction?
CH~4~(g) + 4Cl~2~(g) $$ \to $$ CCl~4~(l) + 4HCl(g)
Q8.
mcq single
+4 / 1
Elimination reaction of 2-Bromo-pentane to form pent-2-ene is :
(A) $$\beta $$-Elimination reaction
(B) Follows Zaitsev rule
(C) Dehydrohalogenation reaction
(D) Dehydration reaction
Q9.
mcq single
+4 / 1
Which of the following alkane cannot be made in good yield by Wurtz reaction?
Q10.
mcq single
+4 / 1
The mixture which shows positive deviation from Raoult's law is :
Q11.
mcq single
+4 / 1
The freezing point depression constant (K~r~) of benzene is 5. 12 K kg mol^(-1). The freezing point depression for the solution of molality 0.078 m containing a non-electrolyte solute in benzene is rounded off upto two decimal places :
Q12.
mcq single
+4 / 1
Which one of the following has maximum number of atoms?
Q13.
mcq single
+4 / 1.33
The number of protons, neutrons and electrons in ^(175)~71~Lu, respectively, are :
Q14.
mcq single
+4 / 1
Match the following :
Oxide
Nature
(A)
CO
(i)
Basic
(B)
BaO
(ii)
Neutral
(C)
Al~2~O~3~
(iii)
Acidic
(D)
Cl~2~O~7~
(iv)
Amphoteric
Which of the following is correct option?
Q15.
mcq single
+4 / 1
Identify the incorrect match.
Name
IUPAC Official Name
(A)
Unnilunium
(i)
Mendelevium
(B)
Unniltrium
(ii)
Lawrencium
(C)
Unnilhexium
(iii)
Seaborgium
(D)
Unununnium
(iv)
Darmstadtium
Q16.
mcq single
+4 / 1
The correct option for free expansion of an ideal gas under adiabatic condition is :
Q17.
mcq single
+4 / 1
Hydrolysis of sucrose is given by the following reaction.
Sucrose + H~2~O ⇌
Glucose + Fructose
If the equilibrium constant (K~c~) is 2 $$ \times $$ 10^(13) at 800 K, the value of $$\Delta r{G^\Theta }$$
at the same temperature will be :
Q18.
mcq single
+4 / 1
For the reaction, 2Cl(g) $$ \to $$ Cl~2~(g), the correct option is :
Q19.
mcq single
+4 / 1
Which of the following is not correct about carbon monoxide?
Q20.
mcq single
+4 / 1
Match the following and identify the correct option.
(A)
CO(g) + H~2~(g)
(i)
Mg(HCO~3~)~2~ + Ca(HCO~3~)~2~
(B)
Temporary hardness of water
(ii)
An electron deficient hydride
(C)
B~2~H~6~
(iii)
Synthesis gas
(D)
H~2~O~2~
(iv)
Non-planar structure
Q21.
mcq single
+4 / 1
Identify the correct statements from the following :
(A) CO~2~(g) is used as refrigerant for ice-cream and frozen food.
(B) The structure of C~60~ contains twelve six-carbon rings and twenty five carbon rings.
(C) ZSM-5, a type of zeolite, is used to convert alcohols into gasoline.
(D) CO is colourless and odourless gas.
Q22.
mcq single
+4 / 1
Which of the following oxoacid of sulphur has $$ - $$
O$$ - $$
O$$ - $$
linkage?
Q23.
mcq single
+4 / 1
Which of the following is the correct order of increasing field strength of ligands to form coordination compounds?
Q24.
mcq single
+4 / 1
The calculated spin only magnetic moment of Cr^(2+) ion is?
Q25.
mcq single
+4 / 1
Urea reacts with water to form A which will decompose to form B. B when passed through Cu^(2+) (aq), deep blue colour solution C is formed. What is the formula of C from the following?
Q26.
mcq single
+4 / 1
A tertiary butyl carbocation is more stable than a secondary butyl carbocation because of which of the following?
Q27.
mcq single
+4 / 1
Paper chromatography is an example of :
Q28.
mcq single
+4 / 1
Reaction between benzaldehyde and acetophenone in presence of dilute NaOH is known as :
Q29.
mcq single
+4 / 1
Reaction between acetone and methylmagnesium chloride followed by hydrolysis will give :
Q30.
mcq single
+4 / 1
Identify compound X in the following sequence of reactions :


Q31.
mcq single
+4 / 1
Anisole on cleavage with Hl gives :
Q32.
mcq single
+4 / 1
As alkene on ozonolysis gives methanal as one of the product, Its structure is :
Q33.
mcq single
+4 / 1
Sucrose on hydrolysis gives :
Q34.
mcq single
+4 / 1
Which of the following is a basic amino acid?
Q35.
mcq single
+4 / 1
On electrolysis of dil. sulphuric acid using Platinum (Pt) electrode, the product obtained at anode will be :
Q36.
mcq single
+4 / 1
The number of Faradays(F) required to produce 20 g of calcium from molten CaCl~2~ (Atomic mass of Ca = 40 g mol^(-1)) is :
Q37.
mcq single
+4 / 1
Which of the following amine will give the carbylamine test?
Q38.
mcq single
+4 / 1
Identify the incorrect statement.
Q39.
mcq single
+4 / 1
Which of the following set of molecules will have zero dipole moment?
Q40.
mcq single
+4 / 1
Identify a molecule which does not exist.
Q41.
mcq single
+4 / 1
Find out the solubility of Ni(OH)~2~ in 0.1M NaOH. Given that the ionic product of Ni(OH)~2~ is 2 $$ \times $$ 10^(-15).
Q42.
mcq single
+4 / 1
The rate constant for a first order reaction is 4.606 $$ \times $$ 10^(-3) s^(-1). The time required to reduce 2.0 g of the reactant to 0.2 g is :
Q43.
mcq single
+4 / 1
An increase in the concentration of the reactants of a reaction leads to change in :
Q44.
mcq single
+4 / 1
What is the change in oxidation number of carbon in the following reaction?
CH~4~(g) + 4Cl~2~(g) $$ \to $$ CCl~4~(l) + 4HCl(g)
Q45.
mcq single
+4 / 1
Elimination reaction of 2-Bromo-pentane to form pent-2-ene is :
(A) $$\beta $$-Elimination reaction
(B) Follows Zaitsev rule
(C) Dehydrohalogenation reaction
(D) Dehydration reaction
Q46.
mcq single
+4 / 1
Which of the following alkane cannot be made in good yield by Wurtz reaction?
Q47.
mcq single
+4 / 1
The mixture which shows positive deviation from Raoult's law is :
Q48.
mcq single
+4 / 1
The freezing point depression constant (K~r~) of benzene is 5. 12 K kg mol^(-1). The freezing point depression for the solution of molality 0.078 m containing a non-electrolyte solute in benzene is rounded off upto two decimal places :
Q49.
mcq single
+4 / 1
Which one of the following has maximum number of atoms?
Q50.
mcq single
+4 / 1.33
The number of protons, neutrons and electrons in ^(175)~71~Lu, respectively, are :
Q51.
mcq single
+4 / 1
Match the following :
Oxide
Nature
(A)
CO
(i)
Basic
(B)
BaO
(ii)
Neutral
(C)
Al~2~O~3~
(iii)
Acidic
(D)
Cl~2~O~7~
(iv)
Amphoteric
Which of the following is correct option?
Q52.
mcq single
+4 / 1
Identify the incorrect match.
Name
IUPAC Official Name
(A)
Unnilunium
(i)
Mendelevium
(B)
Unniltrium
(ii)
Lawrencium
(C)
Unnilhexium
(iii)
Seaborgium
(D)
Unununnium
(iv)
Darmstadtium
Q53.
mcq single
+4 / 1
The correct option for free expansion of an ideal gas under adiabatic condition is :
Q54.
mcq single
+4 / 1
Hydrolysis of sucrose is given by the following reaction.
Sucrose + H~2~O ⇌
Glucose + Fructose
If the equilibrium constant (K~c~) is 2 $$ \times $$ 10^(13) at 800 K, the value of $$\Delta r{G^\Theta }$$
at the same temperature will be :
Q55.
mcq single
+4 / 1
For the reaction, 2Cl(g) $$ \to $$ Cl~2~(g), the correct option is :
Q56.
mcq single
+4 / 1
Which of the following is not correct about carbon monoxide?
Q57.
mcq single
+4 / 1
Match the following and identify the correct option.
(A)
CO(g) + H~2~(g)
(i)
Mg(HCO~3~)~2~ + Ca(HCO~3~)~2~
(B)
Temporary hardness of water
(ii)
An electron deficient hydride
(C)
B~2~H~6~
(iii)
Synthesis gas
(D)
H~2~O~2~
(iv)
Non-planar structure
Q58.
mcq single
+4 / 1
Identify the correct statements from the following :
(A) CO~2~(g) is used as refrigerant for ice-cream and frozen food.
(B) The structure of C~60~ contains twelve six-carbon rings and twenty five carbon rings.
(C) ZSM-5, a type of zeolite, is used to convert alcohols into gasoline.
(D) CO is colourless and odourless gas.
Q59.
mcq single
+4 / 1
Which of the following oxoacid of sulphur has $$ - $$
O$$ - $$
O$$ - $$
linkage?
Q60.
mcq single
+4 / 1
Which of the following is the correct order of increasing field strength of ligands to form coordination compounds?
Q61.
mcq single
+4 / 1
The calculated spin only magnetic moment of Cr^(2+) ion is?
Q62.
mcq single
+4 / 1
Urea reacts with water to form A which will decompose to form B. B when passed through Cu^(2+) (aq), deep blue colour solution C is formed. What is the formula of C from the following?
Q63.
mcq single
+4 / 1
A tertiary butyl carbocation is more stable than a secondary butyl carbocation because of which of the following?
Q64.
mcq single
+4 / 1
Paper chromatography is an example of :
Q65.
mcq single
+4 / 1
Reaction between benzaldehyde and acetophenone in presence of dilute NaOH is known as :
Q66.
mcq single
+4 / 1
Reaction between acetone and methylmagnesium chloride followed by hydrolysis will give :
Q67.
mcq single
+4 / 1
Identify compound X in the following sequence of reactions :
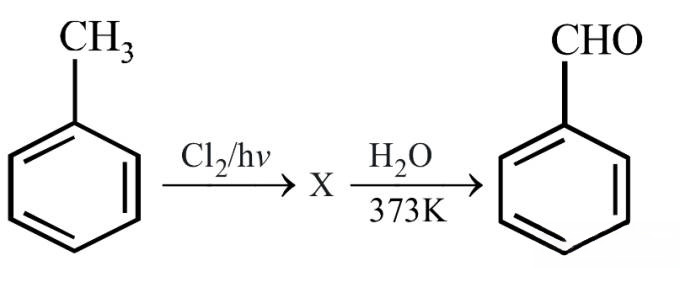

Q68.
mcq single
+4 / 1
Anisole on cleavage with Hl gives :
Q69.
mcq single
+4 / 1
As alkene on ozonolysis gives methanal as one of the product, Its structure is :
Q70.
mcq single
+4 / 1
Sucrose on hydrolysis gives :
Q71.
mcq single
+4 / 1
Which of the following is a basic amino acid?
Q72.
mcq single
+4 / 1
On electrolysis of dil. sulphuric acid using Platinum (Pt) electrode, the product obtained at anode will be :
Q73.
mcq single
+4 / 1
The number of Faradays(F) required to produce 20 g of calcium from molten CaCl~2~ (Atomic mass of Ca = 40 g mol^(-1)) is :
Q74.
mcq single
+4 / 1
Which of the following amine will give the carbylamine test?
Physics
Physics
Q1.
mcq single
+4 / 1
Assume that light of wavelength 600 nm is coming from a star. The limit of resolution of telescope whose objective has a diameter of 2 m is :
Q2.
mcq single
+4 / 1
In Young's double slit experiment, if the separation between coherent sources is halved and the distance of the screen from the coherent sources is doubled, then the fringe width becomes :
Q3.
mcq single
+4 / 1
Find the torque about the origin when a force of 3 $$\hat j$$
N acts on a particle whose position vector is 2 $$\hat k$$ m.
Q4.
mcq single
+4 / 1
The energy required to break one bond in DNA is 10^(-20) J. This value in eV is nearly :
Q5.
mcq single
+4 / 1
Taking into account of the significant figures, what is the value of $$9.99 m - 0.0099m$$?
Q6.
mcq single
+4 / 1
A screw gauge has least count of 0.01 mm and there are 50 divisions in its circular scale.
The pitch of the screw gauge is :
Q7.
mcq single
+4 / 1
Dimensions of stress are :
Q8.
mcq single
+4 / 1
The quantities of heat required to raise the temperature of two solid copper spheres of radii r~1~ and r~2~ (r~1~ = 1.5r~2~) through 1 K are in the ratio :
Q9.
mcq single
+4 / 1
Two cylinders A and B of equal capacity are connected to each other via a stop cock. A contains an ideal gas at standard temperature and pressure. B is completely evacuated. The entire systems is thermally insulated. The stop cock is suddenly opened. The Process is :
Q10.
mcq single
+4 / 1
The average thermal energy for a mono-atomic gas is : (k~B~ is Boltzmann constant and T absolute temperature)
Q11.
mcq single
+4 / 1
A cylinder contains hydrogen gas at pressure 249 kPa and temperature 27$$^\circ $$C
Its density is : (R = 8.3 J mol^(-1) K^(-1))
Q12.
mcq single
+4 / 1
The mean free path for a gas, with molecular diameter d and number density n can be expressed as :
Q13.
mcq single
+4 / 1
A wire of length L, are of cross section A is hanging from a fixed support. The length of the wire changed to L~1~ when mass M is suspended from its free end. The expression for Young's modulus is :
Q14.
mcq single
+4 / 1
A capillary tube of radius r is immersed in water and water rises in it to a height h. The mass of the water in the capillary is 5 g. Another capillary tube of radius 2r is immersed in Water. The mass of water that will rise in this tube is :
Q15.
mcq single
+4 / 1
In a certain region of space with volume 0.2 m^(3), the electric potential is found to be 5 V throughout. The magnitude of electric field in this region is :
Q16.
mcq single
+4 / 1
A spherical conductor of radius 10 cm has a charge of 3.2 $$ \times $$ 10^(-7) C distributed uniformly. That is the magnetude of electric field at a point 15 cm from the centre of the sphere?
$$\left( {{1 \over {4\pi {\varepsilon _0}}} = 9 \times {{10}^9}N{m^2}/{c^2}} \right)$$
Q17.
mcq single
+4 / 1
A short electric dipole has a dipole moment of 16 $$ \times $$ 10^(-9) Cm. The electric potential due to the dipole at a point at a distance of 0.6 m from the centre of the dipole, situated on a line making an angle of $$60^\circ $$ with the dipole axis is :
$$\left( {{1 \over {4\pi {\varepsilon _0}}} = 9 \times {{10}^9}N{m^2}/{C^2}} \right)$$
Q18.
mcq single
+4 / 1
A body weighs 72 N on the surface of the earth. What is the gravitational force on it, at a height equal to half the radius of the earth?
Q19.
mcq single
+4 / 1
The capacitance of a parallel plate capacitor with air as medium is 6$$\mu $$F. With the introduction of a dielectric medium, the capacitance become 30 $$\mu $$F The permittivity of the medium is :
$$\left( {{\varepsilon _0} = 8.85 \times {{10}^{ - 12}}{C^2}{N^{ - 1}}{m^{ - 2}}} \right)$$
Q20.
mcq single
+4 / 1
The Brewster's angle i~b~ for an interface should be :
Q21.
mcq single
+4 / 1
A ray is incident at an angle of incidence i on one surface of a small angle prism (with angle of prism A) and emerges normally from the opposite surface. If the refractive index of the material of the prism is $$\mu $$, then the angle of incidence is nearly equal to :
Q22.
mcq single
+4 / 1
The phase difference between displacement and acceleration of a particle in a simple harmonic motion is :
Q23.
mcq single
+4 / 1
The increase in the width of the depletion region in a p-n junction diode is due to :
Q24.
mcq single
+4 / 1
For the logic circuit shown, the truth table is :


Q25.
mcq single
+4 / 1
A long solenoid of 50 cm length having 100 turns carries a current of 2.5 A. The magnetic field at the centre of the solenoid is :
$$\left( {{\mu _0} = 4\pi \times {{10}^{ - 7}}Tm{{A}^{-1}}} \right)$$
Q26.
mcq single
+4 / 1
Light with an average flux of 20 W/cm^(2) falls on a non-reflecting surface at normal incidence having surface area 20 cm^(2). The energy received by the surface during time span of 1 minute is :
Q27.
mcq single
+4 / 1
An electron is accelerated from rest through a potential difference of V volt. If the de-Broglie wavelength of the electron is 1.227 $$ \times $$ 10^(-2) nm, the potential difference is :
Q28.
mcq single
+4 / 1
Light of frequency 1.5 times the threshold frequency is incident on a photosensitive material. What will be the photoelectric current if the frequency is halved and intensity is doubled?
Q29.
mcq single
+4 / 1
A ball is thrown vertically downward with a velocity of 20 m/s from the top of a tower. It hits the ground after some time with a velocity of 80 m/s. The height of the tower is : (g = 10 m/s^(2))
Q30.
mcq single
+4 / 1
For which one of the following, Bohr model is not valid?
Q31.
mcq single
+4 / 1
The energy equivalent of 0.5 g of a substance is :
Q32.
mcq single
+4 / 1
Two particles of mass 5 kg and 10 kg respectively are attached to the two ends of a rigid rod of length 1 m with negligible mass.
The centre of mass of the system from the 5 kg particle is nearly at a distance of :
Q33.
mcq single
+4 / 1
In a guitar, two strings A and B made of same material are slightly out of tune and produce beats of frequency 6 Hz. When tension in B is slightly decreased, the beat frequency of A is 530 Hz, the original freqnency of B will be :
Q34.
mcq single
+4 / 1
Two bodies of mass 4 kg and 6 kg are tied to the ends of a massless string. The string passes over a pulley which is frictionless (see figure). The acceleration of the system in terms of acceleration due to gravity (g) is :


Q35.
mcq single
+4 / 1
An iron rod of susceptibility 599 is subjected to a magnetizing field of 1200 A m^(-1). The permeability of the material of the rod is :
$$\left( {{\mu _0} = 4\pi \times {{10}^{ - 7}}Tm{A^{ - 1}}} \right)$$
Q36.
mcq single
+4 / 1
A resistance wire connected in the left gap of a metre bridge balances a 10 $$\Omega $$
resistance in the right gap at a point which divides the bridge wire in the ratio 3:2. If the length of the resistance wire is 1.5 m, then the length of 1 $$\Omega $$ of the resistance wire is :
Q37.
mcq single
+4 / 1
Which of the following graph represents the variation of resistivity ($$\rho $$) with temperature (T) for copper?
Q38.
mcq single
+4 / 1
The color code of a resistance is given below :
 The values of resistance and tolerance, respectively, are
The values of resistance and tolerance, respectively, are
 The values of resistance and tolerance, respectively, are
The values of resistance and tolerance, respectively, are
Q39.
mcq single
+4 / 1
A charged particle having drift velocity 7.5$$ \times $$10^(-4) ms^(-1) in an electric field of 3$$ \times $$10^(-10) Vm^(-1) has a mobility in m^(2) V^(-1)s^(-1) of :
Q40.
mcq single
+4 / 1
The solids which have the negative temperature coefficient of resistance are :
Q41.
mcq single
+4 / 1
The ratio of contributions made by the electric field and magnetic field components to the intensity of and electromagnetic wave is : (c = speed of electromagnetic waves)
Q42.
mcq single
+4 / 1
A series LCR circuit is connected to an ac voltage source. When L is removed from the circuit, the phase difference between current and voltage is $${\pi \over 3}$$. If instead C is
removed from the circuit, the phase difference is again $${\pi \over 3}$$
between current and voltage. The power factor of the circuit is :
Q43.
mcq single
+4 / 1
A 40 $$\mu $$F capacitor is connected to a 200 V. 50 Hz ac supply. The rms value of the current on the circuit is, nearly :
Q44.
mcq single
+4 / 1
Assume that light of wavelength 600 nm is coming from a star. The limit of resolution of telescope whose objective has a diameter of 2 m is :
Q45.
mcq single
+4 / 1
In Young's double slit experiment, if the separation between coherent sources is halved and the distance of the screen from the coherent sources is doubled, then the fringe width becomes :
Q46.
mcq single
+4 / 1
Find the torque about the origin when a force of 3 $$\hat j$$
N acts on a particle whose position vector is 2 $$\hat k$$ m.
Q47.
mcq single
+4 / 1
The energy required to break one bond in DNA is 10^(-20) J. This value in eV is nearly :
Q48.
mcq single
+4 / 1
Taking into account of the significant figures, what is the value of $$9.99 m - 0.0099m$$?
Q49.
mcq single
+4 / 1
A screw gauge has least count of 0.01 mm and there are 50 divisions in its circular scale.
The pitch of the screw gauge is :
Q50.
mcq single
+4 / 1
Dimensions of stress are :
Q51.
mcq single
+4 / 1
The quantities of heat required to raise the temperature of two solid copper spheres of radii r~1~ and r~2~ (r~1~ = 1.5r~2~) through 1 K are in the ratio :
Q52.
mcq single
+4 / 1
Two cylinders A and B of equal capacity are connected to each other via a stop cock. A contains an ideal gas at standard temperature and pressure. B is completely evacuated. The entire systems is thermally insulated. The stop cock is suddenly opened. The Process is :
Q53.
mcq single
+4 / 1
The average thermal energy for a mono-atomic gas is : (k~B~ is Boltzmann constant and T absolute temperature)
Q54.
mcq single
+4 / 1
A cylinder contains hydrogen gas at pressure 249 kPa and temperature 27$$^\circ $$C
Its density is : (R = 8.3 J mol^(-1) K^(-1))
Q55.
mcq single
+4 / 1
The mean free path for a gas, with molecular diameter d and number density n can be expressed as :
Q56.
mcq single
+4 / 1
A wire of length L, are of cross section A is hanging from a fixed support. The length of the wire changed to L~1~ when mass M is suspended from its free end. The expression for Young's modulus is :
Q57.
mcq single
+4 / 1
A capillary tube of radius r is immersed in water and water rises in it to a height h. The mass of the water in the capillary is 5 g. Another capillary tube of radius 2r is immersed in Water. The mass of water that will rise in this tube is :
Q58.
mcq single
+4 / 1
In a certain region of space with volume 0.2 m^(3), the electric potential is found to be 5 V throughout. The magnitude of electric field in this region is :
Q59.
mcq single
+4 / 1
A spherical conductor of radius 10 cm has a charge of 3.2 $$ \times $$ 10^(-7) C distributed uniformly. That is the magnetude of electric field at a point 15 cm from the centre of the sphere?
$$\left( {{1 \over {4\pi {\varepsilon _0}}} = 9 \times {{10}^9}N{m^2}/{c^2}} \right)$$
Q60.
mcq single
+4 / 1
A short electric dipole has a dipole moment of 16 $$ \times $$ 10^(-9) Cm. The electric potential due to the dipole at a point at a distance of 0.6 m from the centre of the dipole, situated on a line making an angle of $$60^\circ $$ with the dipole axis is :
$$\left( {{1 \over {4\pi {\varepsilon _0}}} = 9 \times {{10}^9}N{m^2}/{C^2}} \right)$$
Q61.
mcq single
+4 / 1
A body weighs 72 N on the surface of the earth. What is the gravitational force on it, at a height equal to half the radius of the earth?
Q62.
mcq single
+4 / 1
The capacitance of a parallel plate capacitor with air as medium is 6$$\mu $$F. With the introduction of a dielectric medium, the capacitance become 30 $$\mu $$F The permittivity of the medium is :
$$\left( {{\varepsilon _0} = 8.85 \times {{10}^{ - 12}}{C^2}{N^{ - 1}}{m^{ - 2}}} \right)$$
Q63.
mcq single
+4 / 1
The Brewster's angle i~b~ for an interface should be :
Q64.
mcq single
+4 / 1
A ray is incident at an angle of incidence i on one surface of a small angle prism (with angle of prism A) and emerges normally from the opposite surface. If the refractive index of the material of the prism is $$\mu $$, then the angle of incidence is nearly equal to :
Q65.
mcq single
+4 / 1
The phase difference between displacement and acceleration of a particle in a simple harmonic motion is :
Q66.
mcq single
+4 / 1
The increase in the width of the depletion region in a p-n junction diode is due to :
Q67.
mcq single
+4 / 1
For the logic circuit shown, the truth table is :
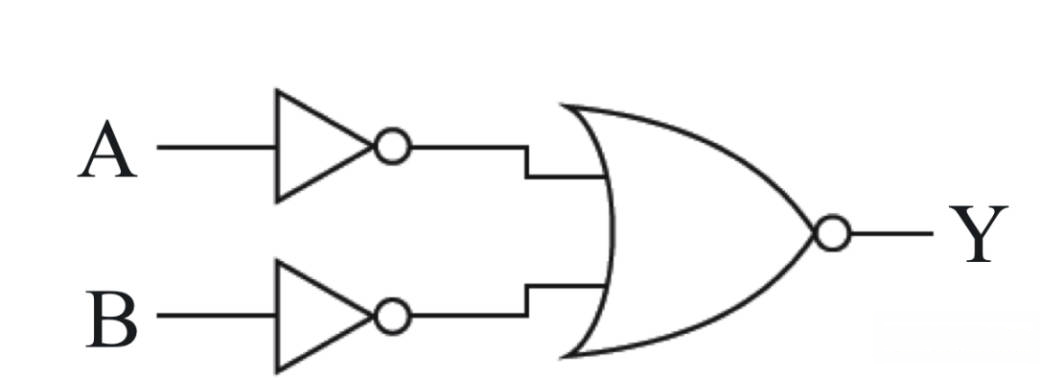

Q68.
mcq single
+4 / 1
A long solenoid of 50 cm length having 100 turns carries a current of 2.5 A. The magnetic field at the centre of the solenoid is :
$$\left( {{\mu _0} = 4\pi \times {{10}^{ - 7}}Tm{{A}^{-1}}} \right)$$
Q69.
mcq single
+4 / 1
Light with an average flux of 20 W/cm^(2) falls on a non-reflecting surface at normal incidence having surface area 20 cm^(2). The energy received by the surface during time span of 1 minute is :
Q70.
mcq single
+4 / 1
An electron is accelerated from rest through a potential difference of V volt. If the de-Broglie wavelength of the electron is 1.227 $$ \times $$ 10^(-2) nm, the potential difference is :
Q71.
mcq single
+4 / 1
Light of frequency 1.5 times the threshold frequency is incident on a photosensitive material. What will be the photoelectric current if the frequency is halved and intensity is doubled?
Q72.
mcq single
+4 / 1
A ball is thrown vertically downward with a velocity of 20 m/s from the top of a tower. It hits the ground after some time with a velocity of 80 m/s. The height of the tower is : (g = 10 m/s^(2))
Q73.
mcq single
+4 / 1
For which one of the following, Bohr model is not valid?
Q74.
mcq single
+4 / 1
The energy equivalent of 0.5 g of a substance is :
Q75.
mcq single
+4 / 1
Two particles of mass 5 kg and 10 kg respectively are attached to the two ends of a rigid rod of length 1 m with negligible mass.
The centre of mass of the system from the 5 kg particle is nearly at a distance of :
Q76.
mcq single
+4 / 1
In a guitar, two strings A and B made of same material are slightly out of tune and produce beats of frequency 6 Hz. When tension in B is slightly decreased, the beat frequency of A is 530 Hz, the original freqnency of B will be :
Q77.
mcq single
+4 / 1
Two bodies of mass 4 kg and 6 kg are tied to the ends of a massless string. The string passes over a pulley which is frictionless (see figure). The acceleration of the system in terms of acceleration due to gravity (g) is :
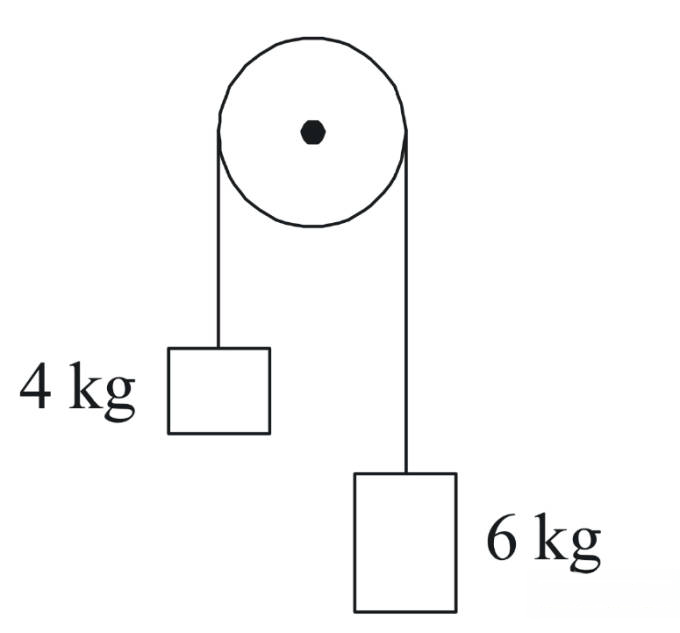

Q78.
mcq single
+4 / 1
An iron rod of susceptibility 599 is subjected to a magnetizing field of 1200 A m^(-1). The permeability of the material of the rod is :
$$\left( {{\mu _0} = 4\pi \times {{10}^{ - 7}}Tm{A^{ - 1}}} \right)$$
Q79.
mcq single
+4 / 1
A resistance wire connected in the left gap of a metre bridge balances a 10 $$\Omega $$
resistance in the right gap at a point which divides the bridge wire in the ratio 3:2. If the length of the resistance wire is 1.5 m, then the length of 1 $$\Omega $$ of the resistance wire is :
Q80.
mcq single
+4 / 1
Which of the following graph represents the variation of resistivity ($$\rho $$) with temperature (T) for copper?
Q81.
mcq single
+4 / 1
The color code of a resistance is given below :
 The values of resistance and tolerance, respectively, are
The values of resistance and tolerance, respectively, are
 The values of resistance and tolerance, respectively, are
The values of resistance and tolerance, respectively, are
Q82.
mcq single
+4 / 1
A charged particle having drift velocity 7.5$$ \times $$10^(-4) ms^(-1) in an electric field of 3$$ \times $$10^(-10) Vm^(-1) has a mobility in m^(2) V^(-1)s^(-1) of :
Q83.
mcq single
+4 / 1
The solids which have the negative temperature coefficient of resistance are :
Q84.
mcq single
+4 / 1
The ratio of contributions made by the electric field and magnetic field components to the intensity of and electromagnetic wave is : (c = speed of electromagnetic waves)
Q85.
mcq single
+4 / 1
A series LCR circuit is connected to an ac voltage source. When L is removed from the circuit, the phase difference between current and voltage is $${\pi \over 3}$$. If instead C is
removed from the circuit, the phase difference is again $${\pi \over 3}$$
between current and voltage. The power factor of the circuit is :
Q86.
mcq single
+4 / 1
A 40 $$\mu $$F capacitor is connected to a 200 V. 50 Hz ac supply. The rms value of the current on the circuit is, nearly :
Biology
Biology
Q1.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Clostridium butylicum
(i) Cyclosporin-A
(b) Trichoderma polysporum
(ii) Butyric Acid
(c) Monascus purpureus
(iii) Citric Acid
(d) Aspergillus niger
(iv) Blood cholesterol lowering agent
Q2.
mcq single
+4 / 1
Which of the following is put into Anaerobic
sludge digester for further sewage treatment?
Q3.
mcq single
+4 / 1
In relation to Gross primary productivity and
Net primary productivity of an ecosystem,
which one of the following statements is
correct?
Q4.
mcq single
+4 / 1
Match the trophic levels with their correct species
examples in grassland ecosystem.
(A) Fourth trophic level (i) Crow
(B) Second trophic level (ii) Vulture
(C) First trophic level (iii) Rabbit
(D) Third trophic level (iv) Grass
Select the correct option
Q5.
mcq single
+4 / 1
Experimental verification of the chromosomal
theory of inheritance was done by
Q6.
mcq single
+4 / 1
Select the correct match
Q7.
mcq single
+4 / 1
How many true breeding pea plant varieties
did Mendel select as pairs, which were similar
except in one character with contrasting
traits?
Q8.
mcq single
+4 / 1
Cuboidal epithelium with brush border of microvilli is found in
Q9.
mcq single
+4 / 1
The oxygenation activity of RuBisCo enzyme
in photorespiration leads to the formation of
Q10.
mcq single
+4 / 1
In light reaction, plastoquinone facilitates the
transfer of electrons from
Q11.
mcq single
+4 / 1
Match the following columns and select the correct option :
Column-I
Column-II
(a) Gregarious,
polyphagous pest
(i) Asterias
(b) Adult with radial
symmetry and larva
with bilateral symmetry
(ii) Scorpion
(c) Book lungs
(iii) Ctenoplana
(d) Bioluminescence
(iv) Locusta
Q12.
mcq single
+4 / 1
Match the following columns and select the correct option :
Column-I
Column-II
(a) 6-15 pairs of gill slits
(i) Trygon
(b) Heterocercal
(ii) Cyclostomes
(c) Air Bladder
(iii) Chondrichthyes
(d) Poison sting
(iv) Osteichthyes
Q13.
mcq single
+4 / 1
If the head of cockroach is removed, it may live for few days because
Q14.
mcq single
+4 / 1
Bilaterally symmetrical and acoelomate animals are exemplified by
Q15.
mcq single
+4 / 1
Which of the following statements are true for the phylum-Chordata
(i) In Urochordata notochord extends from head to tail and it is present throughout their life.
(ii) In Vertebrata notochord is present during the embryonic period only.
(iii) Central nervous system is dorsal and hollow.
(iv) Chordata is divided into 3 subphyla: Hemichordata, Tunicata and Cephalochordata.
Q16.
mcq single
+4 / 1
Strobili or cones are found in
Q17.
mcq single
+4 / 1
Which of the following pairs is of unicellular algae?
Q18.
mcq single
+4 / 1
Floridean starch has structure similar to
Q19.
mcq single
+4 / 1
Identify the wrong statement with reference to
transport of oxygen.
Q20.
mcq single
+4 / 1
Select the correct events that occur during
inspiration.
(i) Contraction of diaphragm
(ii) Contraction of external inter-costal muscles
(iii) Pulmonary volume decreases
(iv) Intra pulmonary pressure increases
Q21.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Bt cotton
(i) Gene therapy
(b) Adenosine
deaminase
deficiency
(ii) Cellular defence
(c) RNAi
(iii) Detection of
HIV infection
(d) PCR
(iv) Bacillus
thuringiensis
Q22.
mcq single
+4 / 1
Which of the following statements is not
correct?
Q23.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Organ of Corti
(i) Connects middle
ear and pharynx
(b) Cochlea
(ii) Coiled part of
the labyrinth
(c) Eustachian tube
(iii) Attached to the
oval window
(d) Stapes
(iv) Located on the
basilar
membrane
Q24.
mcq single
+4 / 1
The transverse section of a plant shows following anatomical features :
(i) Large number of scattered vascular bundles surrounded by bundle sheath
(ii) Large conspicuous parenchymatous ground tissue
(iii) Vascular bundles conjoint and closed
(iv) Phloem parenchyma absent
Identify the category of plant and its part:
Q25.
mcq single
+4 / 1
The roots that originate from the base of the stem are
Q26.
mcq single
+4 / 1
Identify the incorrect statement.
Q27.
mcq single
+4 / 1
Which of the following statements about inclusion bodies is incorrect?
Q28.
mcq single
+4 / 1
Which is the important site of formation of glycoproteins and glycolipids in eukaryotic cells?
Q29.
mcq single
+4 / 1
If the distance between two consecutive base
pairs is 0.34 nm and the total number of base
pairs of a DNA double helix in a typical
mammalian cell is 6.6 × 109 bp, then the length
of the DNA is approximately
Q30.
mcq single
+4 / 1
Which of the following statements is correct?
Q31.
mcq single
+4 / 1
Name the enzyme that facilitates opening of
DNA helix during transcription.
Q32.
mcq single
+4 / 1
The first phase of translation is
Q33.
mcq single
+4 / 1
Identify the wrong statement with reference to
immunity
Q34.
mcq single
+4 / 1
Match the following diseases with the causative
organism and select the correct option
Column-I
Column-II
(a) Typhoid
(i) Wuchereria
(b) Pneumonia
(ii) Plasmodium
(c) Filariasis
(iii) Salmonella
(d) Malaria
(iv) Haemophilus
Q35.
mcq single
+4 / 1
The infectious stage of Plasmodium that enters
the human body is
Q36.
mcq single
+4 / 1
The process of growth is maximum during
Q37.
mcq single
+4 / 1
Secondary metabolites such as nicotine,
strychnine and caffeine are produced by plants
for their
Q38.
mcq single
+4 / 1
Which of the following is not an attribute of a
population?
Q39.
mcq single
+4 / 1
The QRS complex in a standard ECG represents
Q40.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Eosinophils
(i) Immune response
(b) Basophils
(ii) Phagocytosis
(c) Neutrophils
(iii) Release
histaminase,
destructive enzymes
(d) Lymphocytes
(iv) Release granules
containing
histamine
Q41.
mcq single
+4 / 1
Match the following with respect to meiosis
(A) Zygotene (i) Terminalization
(B) Pachytene (ii) Chiasmata
(C) Diplotene (iii) Crossing over
(D) Diakinesis (iv) Synapsis
Select the correct option from the following
Q42.
mcq single
+4 / 1
Identify the correct statement with regard to
G~1~ phase (Gap 1) of interphase
Q43.
mcq single
+4 / 1
Some dividing cells exit the cell cycle and enter vegetative inactive stage. This is called quiescent stage (G~0~). This process occurs at the end of
Q44.
mcq single
+4 / 1
Dissolution of the synaptonemal complex
occurs during
Q45.
mcq single
+4 / 1
Which of the following hormone levels will
cause release of ovum (ovulation) from the
graffian follicle?
Q46.
mcq single
+4 / 1
Meiotic division of the secondary oocyte is
completed
Q47.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(A) Placenta
(i) Androgens
(B) Zona pellucida
(ii) Human Chorionic
Gonadotropin
(hCG)
(C) Bulbo-urethral
(iii) Layer of the ovum
(D) Leydig cells
(iv) Lubrication of
the Penis
Q48.
mcq single
+4 / 1
Select the correct statement
Q49.
mcq single
+4 / 1
Match the following columns and select the
correct option
Column-I
Column-II
(a) Pituitary gland
(i) Grave’s disease
(b) Thyroid gland
(ii) Diabetes mellitus
(c) Adrenal gland
(iii) Diabetes insipidus
(d) Pancreas
(iv) Addison’s disease
Q50.
mcq single
+4 / 1
Which of the following regions of the globe
exhibits highest species diversity?
Q51.
mcq single
+4 / 1
According to Robert May, the global species
diversity is about
Q52.
mcq single
+4 / 1
Name the plant growth regulator which upon
spraying on sugarcane crop, increases the
length of stem, thus increasing the yield of
sugarcane crop
Q53.
mcq single
+4 / 1
Which of the following is not an inhibitory
substance governing seed dormancy?
Q54.
mcq single
+4 / 1
The number of substrate level phosphorylations
in one turn of citric acid cycle is
Q55.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Floating Ribs
(i) Located between
second and
seventh ribs
(b) Acromion
(ii) Head of the
Humerus
(c) Scapula
(iii) Clavicle
(d) Glenoid cavity
(iv) Do not connect
with the sternum
Q56.
mcq single
+4 / 1
Which of the following is correct about viroids?
Q57.
mcq single
+4 / 1
From his experiments, S.L. Miller produced
amino acids by mixing the following in a
closed flask
Q58.
mcq single
+4 / 1
Embryological support for evolution was
disapproved by
Q59.
mcq single
+4 / 1
Flippers of Penguins and Dolphins are
examples of
Q60.
mcq single
+4 / 1
Which of the following refer to correct
example(s) of organisms which have evolved
due to changes in environment brought about
by anthropogenic action?
(a) Darwin’s Finches of Galapagos islands.
(b) Herbicide resistant weeds.
(c) Drug resistant eukaryotes.
(d) Man-created breeds of domesticated
animals like dogs.
Q61.
mcq single
+4 / 1
Which of the following would help in
prevention of diuresis?
Q62.
mcq single
+4 / 1
Presence of which of the following conditions
in urine are indicative of Diabetes Mellitus?
Q63.
mcq single
+4 / 1
In which of the following techniques, the
embryos are transferred to assist those females
who cannot conceive?
Q64.
mcq single
+4 / 1
Select the option including all sexually
transmitted diseases.
Q65.
mcq single
+4 / 1
The plant parts which consist of two generations
- one within the other
(i) Pollen grains inside the anther
(ii) Germinated pollen grain with two male
gametes
(iii) Seed inside the fruit
(iv) Embryo sac inside the ovule
Q66.
mcq single
+4 / 1
The body of the ovule is fused within the
funicle at
Q67.
mcq single
+4 / 1
The ovary is half inferior in:
Q68.
mcq single
+4 / 1
Ray florets have
Q69.
mcq single
+4 / 1
Identify the substances having glycosidic bond and peptide bond, respectively in their structure
Q70.
mcq single
+4 / 1
Which one of the following is the most abundant protein in the animals?
Q71.
mcq single
+4 / 1
Identify the wrong statement with reference to
the gene ‘I’ that controls ABO blood groups.
Q72.
mcq single
+4 / 1
Match the following
(a) Inhibitor of
catalytic activity
(i) Ricin
(b) Possess peptide bonds
(ii) Malonate
(c) Cell wall material
in fungi
(iii) Chitin
(d) Secondary
metabolite
(iv) Collagen
Choose the correct option from the following
Q73.
mcq single
+4 / 1
Identify the basic amino acid from the
following
Q74.
mcq single
+4 / 1
In gel electrophoresis, separated DNA fragments
can be visualized with the help of
Q75.
mcq single
+4 / 1
The specific palindromic sequence which is
recognized by EcoRl is
Q76.
mcq single
+4 / 1
Match the organism with its use in biotechnology
(a) Bacillus thuringiensis
(i) Cloning vector
(b) Thermus aquaticus
(ii) Construction of first rDNA molecule
(c) Agrobacterium tumefaciens
(iii) DNA polymerase
(d) Salmonella typhimurium
(iv) Cry proteins
Select the correct option from the following:
Q77.
mcq single
+4 / 1
Bt cotton variety that was developed by
the introduction of toxin gene of Bacillus
thuringiensis (Bt) is resistant to
Q78.
mcq single
+4 / 1
Identify the wrong statement with regard to
Restriction Enzymes
Q79.
mcq single
+4 / 1
The sequence that controls the copy number of
the linked DNA in the vector, is termed
Q80.
mcq single
+4 / 1
Choose the correct pair from the following
Q81.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Clostridium butylicum
(i) Cyclosporin-A
(b) Trichoderma polysporum
(ii) Butyric Acid
(c) Monascus purpureus
(iii) Citric Acid
(d) Aspergillus niger
(iv) Blood cholesterol lowering agent
Q82.
mcq single
+4 / 1
Which of the following is put into Anaerobic
sludge digester for further sewage treatment?
Q83.
mcq single
+4 / 1
In relation to Gross primary productivity and
Net primary productivity of an ecosystem,
which one of the following statements is
correct?
Q84.
mcq single
+4 / 1
Match the trophic levels with their correct species
examples in grassland ecosystem.
(A) Fourth trophic level (i) Crow
(B) Second trophic level (ii) Vulture
(C) First trophic level (iii) Rabbit
(D) Third trophic level (iv) Grass
Select the correct option
Q85.
mcq single
+4 / 1
Experimental verification of the chromosomal
theory of inheritance was done by
Q86.
mcq single
+4 / 1
Select the correct match
Q87.
mcq single
+4 / 1
How many true breeding pea plant varieties
did Mendel select as pairs, which were similar
except in one character with contrasting
traits?
Q88.
mcq single
+4 / 1
Cuboidal epithelium with brush border of microvilli is found in
Q89.
mcq single
+4 / 1
The oxygenation activity of RuBisCo enzyme
in photorespiration leads to the formation of
Q90.
mcq single
+4 / 1
In light reaction, plastoquinone facilitates the
transfer of electrons from
Q91.
mcq single
+4 / 1
Match the following columns and select the correct option :
Column-I
Column-II
(a) Gregarious,
polyphagous pest
(i) Asterias
(b) Adult with radial
symmetry and larva
with bilateral symmetry
(ii) Scorpion
(c) Book lungs
(iii) Ctenoplana
(d) Bioluminescence
(iv) Locusta
Q92.
mcq single
+4 / 1
Match the following columns and select the correct option :
Column-I
Column-II
(a) 6-15 pairs of gill slits
(i) Trygon
(b) Heterocercal
(ii) Cyclostomes
(c) Air Bladder
(iii) Chondrichthyes
(d) Poison sting
(iv) Osteichthyes
Q93.
mcq single
+4 / 1
If the head of cockroach is removed, it may live for few days because
Q94.
mcq single
+4 / 1
Bilaterally symmetrical and acoelomate animals are exemplified by
Q95.
mcq single
+4 / 1
Which of the following statements are true for the phylum-Chordata
(i) In Urochordata notochord extends from head to tail and it is present throughout their life.
(ii) In Vertebrata notochord is present during the embryonic period only.
(iii) Central nervous system is dorsal and hollow.
(iv) Chordata is divided into 3 subphyla: Hemichordata, Tunicata and Cephalochordata.
Q96.
mcq single
+4 / 1
Strobili or cones are found in
Q97.
mcq single
+4 / 1
Which of the following pairs is of unicellular algae?
Q98.
mcq single
+4 / 1
Floridean starch has structure similar to
Q99.
mcq single
+4 / 1
Identify the wrong statement with reference to
transport of oxygen.
Q100.
mcq single
+4 / 1
Select the correct events that occur during
inspiration.
(i) Contraction of diaphragm
(ii) Contraction of external inter-costal muscles
(iii) Pulmonary volume decreases
(iv) Intra pulmonary pressure increases
Q101.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Bt cotton
(i) Gene therapy
(b) Adenosine
deaminase
deficiency
(ii) Cellular defence
(c) RNAi
(iii) Detection of
HIV infection
(d) PCR
(iv) Bacillus
thuringiensis
Q102.
mcq single
+4 / 1
Which of the following statements is not
correct?
Q103.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Organ of Corti
(i) Connects middle
ear and pharynx
(b) Cochlea
(ii) Coiled part of
the labyrinth
(c) Eustachian tube
(iii) Attached to the
oval window
(d) Stapes
(iv) Located on the
basilar
membrane
Q104.
mcq single
+4 / 1
The transverse section of a plant shows following anatomical features :
(i) Large number of scattered vascular bundles surrounded by bundle sheath
(ii) Large conspicuous parenchymatous ground tissue
(iii) Vascular bundles conjoint and closed
(iv) Phloem parenchyma absent
Identify the category of plant and its part:
Q105.
mcq single
+4 / 1
The roots that originate from the base of the stem are
Q106.
mcq single
+4 / 1
Identify the incorrect statement.
Q107.
mcq single
+4 / 1
Which of the following statements about inclusion bodies is incorrect?
Q108.
mcq single
+4 / 1
Which is the important site of formation of glycoproteins and glycolipids in eukaryotic cells?
Q109.
mcq single
+4 / 1
If the distance between two consecutive base
pairs is 0.34 nm and the total number of base
pairs of a DNA double helix in a typical
mammalian cell is 6.6 × 109 bp, then the length
of the DNA is approximately
Q110.
mcq single
+4 / 1
Which of the following statements is correct?
Q111.
mcq single
+4 / 1
Name the enzyme that facilitates opening of
DNA helix during transcription.
Q112.
mcq single
+4 / 1
The first phase of translation is
Q113.
mcq single
+4 / 1
Identify the wrong statement with reference to
immunity
Q114.
mcq single
+4 / 1
Match the following diseases with the causative
organism and select the correct option
Column-I
Column-II
(a) Typhoid
(i) Wuchereria
(b) Pneumonia
(ii) Plasmodium
(c) Filariasis
(iii) Salmonella
(d) Malaria
(iv) Haemophilus
Q115.
mcq single
+4 / 1
The infectious stage of Plasmodium that enters
the human body is
Q116.
mcq single
+4 / 1
The process of growth is maximum during
Q117.
mcq single
+4 / 1
Secondary metabolites such as nicotine,
strychnine and caffeine are produced by plants
for their
Q118.
mcq single
+4 / 1
Which of the following is not an attribute of a
population?
Q119.
mcq single
+4 / 1
The QRS complex in a standard ECG represents
Q120.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Eosinophils
(i) Immune response
(b) Basophils
(ii) Phagocytosis
(c) Neutrophils
(iii) Release
histaminase,
destructive enzymes
(d) Lymphocytes
(iv) Release granules
containing
histamine
Q121.
mcq single
+4 / 1
Match the following with respect to meiosis
(A) Zygotene (i) Terminalization
(B) Pachytene (ii) Chiasmata
(C) Diplotene (iii) Crossing over
(D) Diakinesis (iv) Synapsis
Select the correct option from the following
Q122.
mcq single
+4 / 1
Identify the correct statement with regard to
G~1~ phase (Gap 1) of interphase
Q123.
mcq single
+4 / 1
Some dividing cells exit the cell cycle and enter vegetative inactive stage. This is called quiescent stage (G~0~). This process occurs at the end of
Q124.
mcq single
+4 / 1
Dissolution of the synaptonemal complex
occurs during
Q125.
mcq single
+4 / 1
Which of the following hormone levels will
cause release of ovum (ovulation) from the
graffian follicle?
Q126.
mcq single
+4 / 1
Meiotic division of the secondary oocyte is
completed
Q127.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(A) Placenta
(i) Androgens
(B) Zona pellucida
(ii) Human Chorionic
Gonadotropin
(hCG)
(C) Bulbo-urethral
(iii) Layer of the ovum
(D) Leydig cells
(iv) Lubrication of
the Penis
Q128.
mcq single
+4 / 1
Select the correct statement
Q129.
mcq single
+4 / 1
Match the following columns and select the
correct option
Column-I
Column-II
(a) Pituitary gland
(i) Grave’s disease
(b) Thyroid gland
(ii) Diabetes mellitus
(c) Adrenal gland
(iii) Diabetes insipidus
(d) Pancreas
(iv) Addison’s disease
Q130.
mcq single
+4 / 1
Which of the following regions of the globe
exhibits highest species diversity?
Q131.
mcq single
+4 / 1
According to Robert May, the global species
diversity is about
Q132.
mcq single
+4 / 1
Name the plant growth regulator which upon
spraying on sugarcane crop, increases the
length of stem, thus increasing the yield of
sugarcane crop
Q133.
mcq single
+4 / 1
Which of the following is not an inhibitory
substance governing seed dormancy?
Q134.
mcq single
+4 / 1
The number of substrate level phosphorylations
in one turn of citric acid cycle is
Q135.
mcq single
+4 / 1
Match the following columns and select the
correct option.
Column-I
Column-II
(a) Floating Ribs
(i) Located between
second and
seventh ribs
(b) Acromion
(ii) Head of the
Humerus
(c) Scapula
(iii) Clavicle
(d) Glenoid cavity
(iv) Do not connect
with the sternum
Q136.
mcq single
+4 / 1
Which of the following is correct about viroids?
Q137.
mcq single
+4 / 1
From his experiments, S.L. Miller produced
amino acids by mixing the following in a
closed flask
Q138.
mcq single
+4 / 1
Embryological support for evolution was
disapproved by
Q139.
mcq single
+4 / 1
Flippers of Penguins and Dolphins are
examples of
Q140.
mcq single
+4 / 1
Which of the following refer to correct
example(s) of organisms which have evolved
due to changes in environment brought about
by anthropogenic action?
(a) Darwin’s Finches of Galapagos islands.
(b) Herbicide resistant weeds.
(c) Drug resistant eukaryotes.
(d) Man-created breeds of domesticated
animals like dogs.
Q141.
mcq single
+4 / 1
Which of the following would help in
prevention of diuresis?
Q142.
mcq single
+4 / 1
Presence of which of the following conditions
in urine are indicative of Diabetes Mellitus?
Q143.
mcq single
+4 / 1
In which of the following techniques, the
embryos are transferred to assist those females
who cannot conceive?
Q144.
mcq single
+4 / 1
Select the option including all sexually
transmitted diseases.
Q145.
mcq single
+4 / 1
The plant parts which consist of two generations
- one within the other
(i) Pollen grains inside the anther
(ii) Germinated pollen grain with two male
gametes
(iii) Seed inside the fruit
(iv) Embryo sac inside the ovule
Q146.
mcq single
+4 / 1
The body of the ovule is fused within the
funicle at
Q147.
mcq single
+4 / 1
The ovary is half inferior in:
Q148.
mcq single
+4 / 1
Ray florets have
Q149.
mcq single
+4 / 1
Identify the substances having glycosidic bond and peptide bond, respectively in their structure
Q150.
mcq single
+4 / 1
Which one of the following is the most abundant protein in the animals?
Q151.
mcq single
+4 / 1
Identify the wrong statement with reference to
the gene ‘I’ that controls ABO blood groups.
Q152.
mcq single
+4 / 1
Match the following
(a) Inhibitor of
catalytic activity
(i) Ricin
(b) Possess peptide bonds
(ii) Malonate
(c) Cell wall material
in fungi
(iii) Chitin
(d) Secondary
metabolite
(iv) Collagen
Choose the correct option from the following
Q153.
mcq single
+4 / 1
Identify the basic amino acid from the
following
Q154.
mcq single
+4 / 1
In gel electrophoresis, separated DNA fragments
can be visualized with the help of
Q155.
mcq single
+4 / 1
The specific palindromic sequence which is
recognized by EcoRl is
Q156.
mcq single
+4 / 1
Match the organism with its use in biotechnology
(a) Bacillus thuringiensis
(i) Cloning vector
(b) Thermus aquaticus
(ii) Construction of first rDNA molecule
(c) Agrobacterium tumefaciens
(iii) DNA polymerase
(d) Salmonella typhimurium
(iv) Cry proteins
Select the correct option from the following:
Q157.
mcq single
+4 / 1
Bt cotton variety that was developed by
the introduction of toxin gene of Bacillus
thuringiensis (Bt) is resistant to
Q158.
mcq single
+4 / 1
Identify the wrong statement with regard to
Restriction Enzymes
Q159.
mcq single
+4 / 1
The sequence that controls the copy number of
the linked DNA in the vector, is termed
Q160.
mcq single
+4 / 1
Choose the correct pair from the following