neet-2022
NEET 2022 Phase 1
MEDICAL 2022 Previous Year
3 hDuration
692Total Marks
346Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 346 across 3 section(s); maximum marks: 692.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R)
Assertion (A) : ICI is more reactive than I~2~
Reason (R) : I-CI bond is weaker than I-I bond.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q2.
mcq single
+4 / 1
Which amongst the following is incorrect statement?
Q3.
mcq single
+4 / 1
Amongst the following which one will have maximum 'lone pair - lone pair' electron repulsions?
Q4.
mcq single
+4 / 1
The pH of the solution containing 50 mL each of 0.10 M sodium acetate and 0.01 M acetic acid is [Given pK~a~ of CH~3~COOH = 4.57]
Q5.
mcq single
+4 / 1
The given graph is a representation of kinetics of a reaction
 The y and x axes for zero and first order reactions, respectively are
The y and x axes for zero and first order reactions, respectively are
 The y and x axes for zero and first order reactions, respectively are
The y and x axes for zero and first order reactions, respectively are
Q6.
mcq single
+4 / 1
For a first order reaction A $$\to$$ Products, initial concentration of A is 0.1 M, which becomes 0.001 M after 5 minutes. Rate constant for the reaction in min^($$-$$1) is
Q7.
mcq single
+4 / 1
In one molal solution that contains 0.5 mole of a solute, there is
Q8.
mcq single
+4 / 1
What mass of 95% pure CaCO~3~ will be required to neutralise 50 mL of 0.5 M HCl solution according to the following reaction?
CaCO~3(s)~ + 2HCl~(aq)~ $$\to$$ CaCl~2(aq)~ + Co~2(g)~ + 2H~2~O~(l)~
[Calculate upto second place of decimal point]
Q9.
mcq single
+4 / 1
3O~2~(g) $$\rightleftharpoons$$ 2O~3~(g)
for the above reaction at 298 K, K~c~ is found to be 3.0 $$\times$$ 10^($$-$$59). If the concentration of O~2~ at equilibrium is 0.040 M then concentration of O~3~ in M is
Q10.
mcq single
+4 / 1
Gadolinium has a low value of third ionisation enthalpy because of
Q11.
mcq single
+4 / 1
The IUPAC name of an element with atomic number 119 is
Q12.
mcq single
+4 / 1
Which of the following p-V curve represents maximum work done?
Q13.
mcq single
+4 / 1
Match List-I with List-II.
List - I
(Hydrides)
List - II
(Nature)
(a)
MgH$$_2$$
(i)
Electron precise
(b)
GeH$$_4$$
(ii)
Electron deficient
(c)
B$$_2$$H$$_6$$
(iii)
Electron rich
(d)
HF
(iv)
Ionic
Choose the correct answer from the options given below:
Q14.
mcq single
+4 / 1
Given below are two statements
Statement I : The boiling points of the following hydrides of group 16 elements increases in the order -
H~2~O < H~2~S < H~2~Se < H~2~Te
Statement II : The boiling points of these hydrides increase with increase in molar mass.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q15.
mcq single
+4 / 1
Which of the following statement is not correct about diborane?
Q16.
mcq single
+4 / 1
The order of energy absorbed which is responsible for the color of complexes
(A) [Ni(H~2~O)~2~(en)~2~]^(2+)
(B) [Ni(H~2~O)~4~(en)~~]^(2+) and
(C) [Ni(en)~3~]^(2+)
is
Q17.
mcq single
+4 / 1
The IUPAC name of the complex -
[Ag(H~2~O)~2~][Ag(CN)~2~] is:
Q18.
mcq single
+4 / 1
The incorrect statement regarding chirality is
Q19.
mcq single
+4 / 1
The Kjeldahl's method for the estimation of nitrogen can be used to estimate the amount of nitrogen in which one of the following compounds?
Q20.
mcq single
+4 / 1
Which compound amongst the following is not an aromatic compound?
Q21.
mcq single
+4 / 1
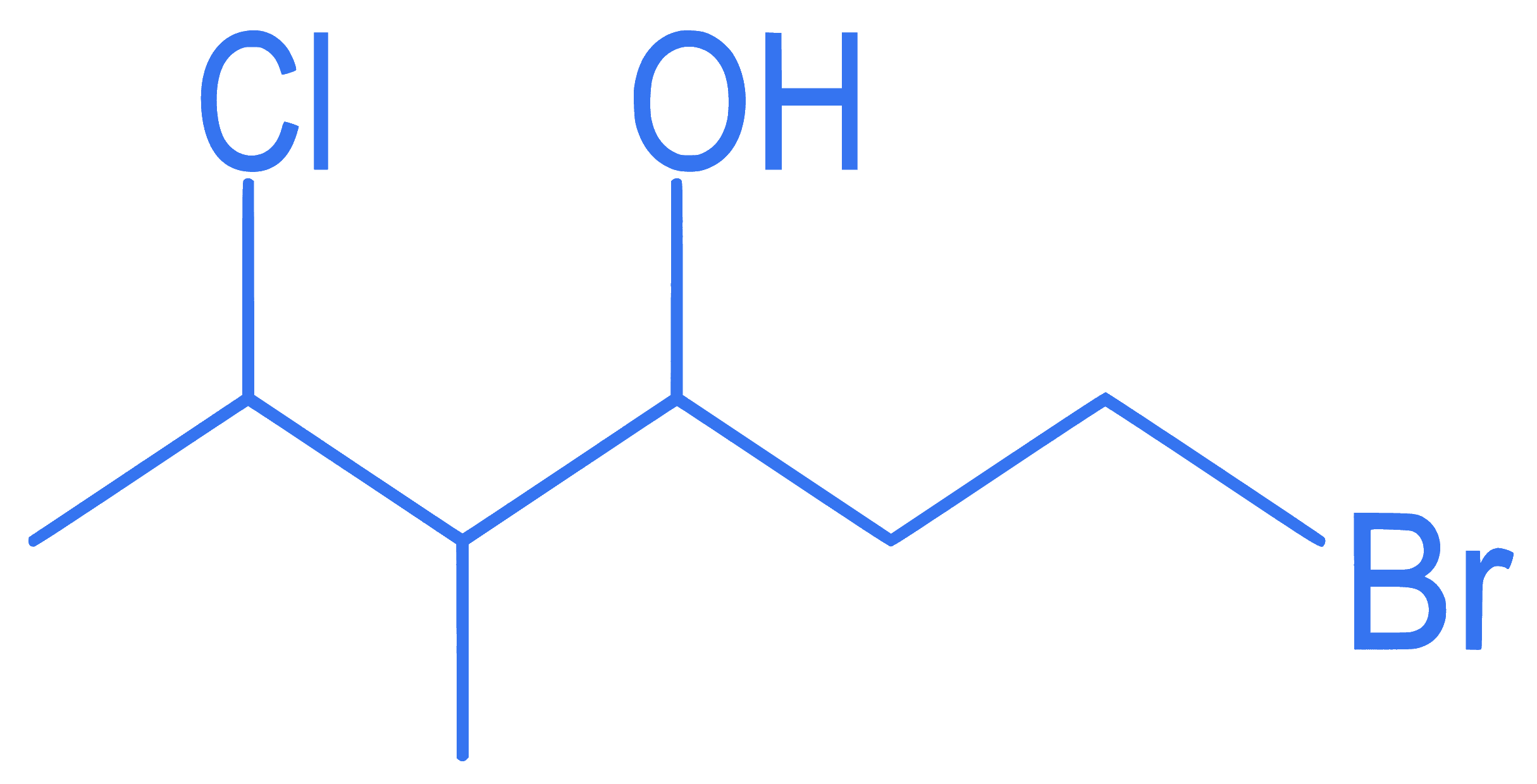
The correct IUPAC name of the following compound is

Q22.
mcq single
+4 / 1
Match List-I with List-II.
List - I
(Products formed)
List - II
(Reaction of carbonyl compound with)
(a)
Cyanohydrin
(i)
NH$$_2$$OH
(b)
Acetal
(ii)
RNH$$_2$$
(c)
Schiff's base
(iii)
Alcohol
(d)
Oxime
(iv)
HCN
Choose the correct answer from the options given below
Q23.
mcq single
+4 / 1
Given below are two statements :
Statement I : The boiling points of aldehydes and ketones are higher than hydrocarbons of comparable molecular masses because of weak molecular association in aldehydes and ketones due to dipole - dipole interactions.
Statement II : The boiling points of aldehydes and ketones are lower than the alcohols of similar molecular masses due to the absence of H-bonding.
In the light of the above statements, choose the most appropriate answer from the given below
Q24.
mcq single
+4 / 1
RMgX + CO~2~ $$\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{ether}^{dry}} $$ Y $$\buildrel {{H_3}{O^ + }} \over
\longrightarrow $$ RCOOH
What is Y in the above reaction?
Q25.
mcq single
+4 / 1
Compound X on reaction with O~3~ followed by Zn/H~2~O gives formaldehyde and 2-methyl propanal as products. The compound X is
Q26.
mcq single
+4 / 1
Which one of the following is not formed when acetone reacts with 2-pentanone in the presence of dilute NaOH followed by heating?
Q27.
mcq single
+4 / 1
Given below are two statements:
Statement I : In Lucas test, primary, secondary and tertiary alcohols are distinguished on the basis of their reactivity with conc. HCl + ZnCl~2~, known as Lucas Reagent.
Statement II : Primary alcohols are most reactive and immediately produce turbidity at room temperature on reaction with Lucas Reagent.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q28.
mcq single
+4 / 1
Given below are two statements:
Statement I: The acidic strength of monosubstituted nitrophenol is higher than phenol because of electron withdrawing nitro group.
Statement II : o-nitrophenol, m-nitrophenol and p-nitrophenol will have same acidic strength as they have one nitro group attached to the phenolic ring.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q29.
mcq single
+4 / 1
The incorrect statement regarding enzymes is
Q30.
mcq single
+4 / 1
Given below are half cell reactions:
$$MnO_4^ - + 8{H^ + } + 5{e^ - } \to M{n^{2 + }} + 4{H_2}O$$,
$$E_{M{n^{2 + }}/MnO_4^ - }^o = - 1.510\,V$$
$${1 \over 2}{O_2} + 2{H^ + } + 2{e^ - } \to {H_2}O$$
$$E_{{O_2}/{H_2}O}^o = + 1.223\,V$$
Will the permanganate ion, $$MnO_4^ - $$ liberate O~2~ from water in the presence of an acid?
Q31.
mcq single
+4 / 1
Find the emf of the cell in which the following reaction takes place at 298 K
Ni(s) + 2Ag^(+) (0.001 M) $$\to$$ Ni^(2+) (0.001 M) + 2Ag(s)
(Given that E$$_{cell}^o$$ = 10.5 V, $${{2.303\,RT} \over F} = 0.059$$ at 298 K)
Q32.
mcq single
+4 / 1
At 298 K, the standard electrode potentials of Cu^(2+) / Cu, Zn^(2+) / Zn, Fe^(2+) / Fe and Ag^(+) / Ag are 0.34 V, $$-$$0.76 V, $$-$$0.44 V V and 0.80 V, respectively.
On the basis of standard electrode potential, predict which of the following reaction cannot occur?
Q33.
mcq single
+4 / 1
Which of the following sequence of reactions is suitable to synthesize chlorobenzene?
Q34.
mcq single
+4 / 1
The product formed from the following reaction sequence is
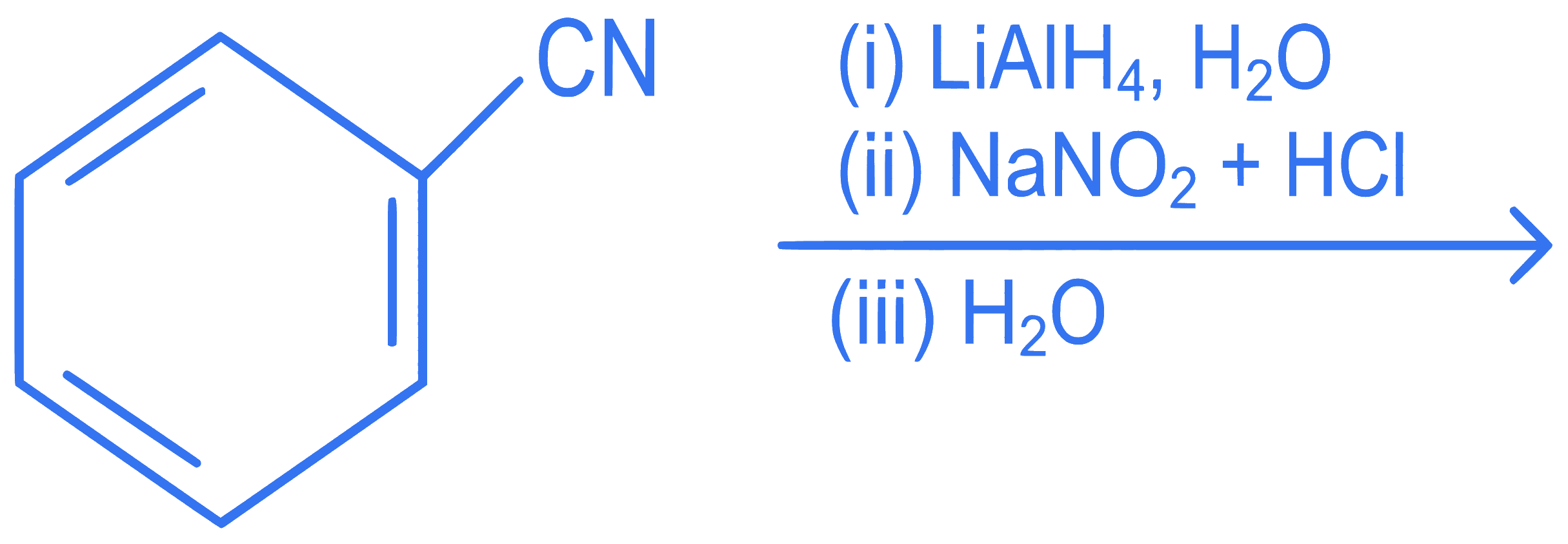
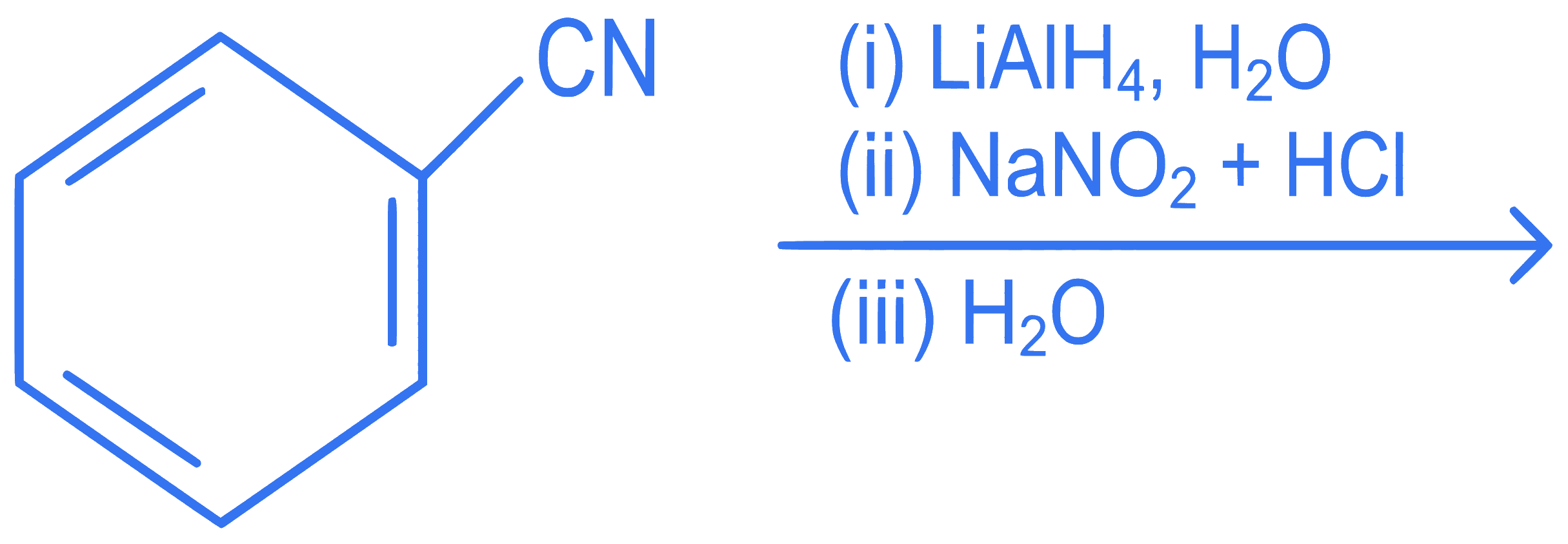
Q35.
mcq single
+4 / 1
Given below are two statements
Statement I : Primary aliphatic amines react with HNO~2~ to give unstable diazonium salts.
Statement II : Primary aromatic amines react with HNO~2~ to form diazonium salts which are stable even above 300 K.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q36.
mcq single
+4 / 1
Identify the incorrect statement from the following.
Q37.
mcq single
+4 / 1
If radius of second Bohr orbit of the He^(+) ion is 105.8 pm, what is the radius of third Bohr orbit of Li^(2+) ion?
Q38.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R)
Assertion (A) : ICI is more reactive than I~2~
Reason (R) : I-CI bond is weaker than I-I bond.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q39.
mcq single
+4 / 1
Which amongst the following is incorrect statement?
Q40.
mcq single
+4 / 1
Amongst the following which one will have maximum 'lone pair - lone pair' electron repulsions?
Q41.
mcq single
+4 / 1
The pH of the solution containing 50 mL each of 0.10 M sodium acetate and 0.01 M acetic acid is [Given pK~a~ of CH~3~COOH = 4.57]
Q42.
mcq single
+4 / 1
The given graph is a representation of kinetics of a reaction
 The y and x axes for zero and first order reactions, respectively are
The y and x axes for zero and first order reactions, respectively are
 The y and x axes for zero and first order reactions, respectively are
The y and x axes for zero and first order reactions, respectively are
Q43.
mcq single
+4 / 1
For a first order reaction A $$\to$$ Products, initial concentration of A is 0.1 M, which becomes 0.001 M after 5 minutes. Rate constant for the reaction in min^($$-$$1) is
Q44.
mcq single
+4 / 1
In one molal solution that contains 0.5 mole of a solute, there is
Q45.
mcq single
+4 / 1
What mass of 95% pure CaCO~3~ will be required to neutralise 50 mL of 0.5 M HCl solution according to the following reaction?
CaCO~3(s)~ + 2HCl~(aq)~ $$\to$$ CaCl~2(aq)~ + Co~2(g)~ + 2H~2~O~(l)~
[Calculate upto second place of decimal point]
Q46.
mcq single
+4 / 1
3O~2~(g) $$\rightleftharpoons$$ 2O~3~(g)
for the above reaction at 298 K, K~c~ is found to be 3.0 $$\times$$ 10^($$-$$59). If the concentration of O~2~ at equilibrium is 0.040 M then concentration of O~3~ in M is
Q47.
mcq single
+4 / 1
Gadolinium has a low value of third ionisation enthalpy because of
Q48.
mcq single
+4 / 1
The IUPAC name of an element with atomic number 119 is
Q49.
mcq single
+4 / 1
Which of the following p-V curve represents maximum work done?
Q50.
mcq single
+4 / 1
Match List-I with List-II.
List - I
(Hydrides)
List - II
(Nature)
(a)
MgH$$_2$$
(i)
Electron precise
(b)
GeH$$_4$$
(ii)
Electron deficient
(c)
B$$_2$$H$$_6$$
(iii)
Electron rich
(d)
HF
(iv)
Ionic
Choose the correct answer from the options given below:
Q51.
mcq single
+4 / 1
Given below are two statements
Statement I : The boiling points of the following hydrides of group 16 elements increases in the order -
H~2~O < H~2~S < H~2~Se < H~2~Te
Statement II : The boiling points of these hydrides increase with increase in molar mass.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q52.
mcq single
+4 / 1
Which of the following statement is not correct about diborane?
Q53.
mcq single
+4 / 1
The order of energy absorbed which is responsible for the color of complexes
(A) [Ni(H~2~O)~2~(en)~2~]^(2+)
(B) [Ni(H~2~O)~4~(en)~~]^(2+) and
(C) [Ni(en)~3~]^(2+)
is
Q54.
mcq single
+4 / 1
The IUPAC name of the complex -
[Ag(H~2~O)~2~][Ag(CN)~2~] is:
Q55.
mcq single
+4 / 1
The incorrect statement regarding chirality is
Q56.
mcq single
+4 / 1
The Kjeldahl's method for the estimation of nitrogen can be used to estimate the amount of nitrogen in which one of the following compounds?
Q57.
mcq single
+4 / 1
Which compound amongst the following is not an aromatic compound?
Q58.
mcq single
+4 / 1
The correct IUPAC name of the following compound is


Q59.
mcq single
+4 / 1
Match List-I with List-II.
List - I
(Products formed)
List - II
(Reaction of carbonyl compound with)
(a)
Cyanohydrin
(i)
NH$$_2$$OH
(b)
Acetal
(ii)
RNH$$_2$$
(c)
Schiff's base
(iii)
Alcohol
(d)
Oxime
(iv)
HCN
Choose the correct answer from the options given below
Q60.
mcq single
+4 / 1
Given below are two statements :
Statement I : The boiling points of aldehydes and ketones are higher than hydrocarbons of comparable molecular masses because of weak molecular association in aldehydes and ketones due to dipole - dipole interactions.
Statement II : The boiling points of aldehydes and ketones are lower than the alcohols of similar molecular masses due to the absence of H-bonding.
In the light of the above statements, choose the most appropriate answer from the given below
Q61.
mcq single
+4 / 1
RMgX + CO~2~ $$\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{ether}^{dry}} $$ Y $$\buildrel {{H_3}{O^ + }} \over
\longrightarrow $$ RCOOH
What is Y in the above reaction?
Q62.
mcq single
+4 / 1
Compound X on reaction with O~3~ followed by Zn/H~2~O gives formaldehyde and 2-methyl propanal as products. The compound X is
Q63.
mcq single
+4 / 1
Which one of the following is not formed when acetone reacts with 2-pentanone in the presence of dilute NaOH followed by heating?
Q64.
mcq single
+4 / 1
Given below are two statements:
Statement I : In Lucas test, primary, secondary and tertiary alcohols are distinguished on the basis of their reactivity with conc. HCl + ZnCl~2~, known as Lucas Reagent.
Statement II : Primary alcohols are most reactive and immediately produce turbidity at room temperature on reaction with Lucas Reagent.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q65.
mcq single
+4 / 1
Given below are two statements:
Statement I: The acidic strength of monosubstituted nitrophenol is higher than phenol because of electron withdrawing nitro group.
Statement II : o-nitrophenol, m-nitrophenol and p-nitrophenol will have same acidic strength as they have one nitro group attached to the phenolic ring.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q66.
mcq single
+4 / 1
The incorrect statement regarding enzymes is
Q67.
mcq single
+4 / 1
Given below are half cell reactions:
$$MnO_4^ - + 8{H^ + } + 5{e^ - } \to M{n^{2 + }} + 4{H_2}O$$,
$$E_{M{n^{2 + }}/MnO_4^ - }^o = - 1.510\,V$$
$${1 \over 2}{O_2} + 2{H^ + } + 2{e^ - } \to {H_2}O$$
$$E_{{O_2}/{H_2}O}^o = + 1.223\,V$$
Will the permanganate ion, $$MnO_4^ - $$ liberate O~2~ from water in the presence of an acid?
Q68.
mcq single
+4 / 1
Find the emf of the cell in which the following reaction takes place at 298 K
Ni(s) + 2Ag^(+) (0.001 M) $$\to$$ Ni^(2+) (0.001 M) + 2Ag(s)
(Given that E$$_{cell}^o$$ = 10.5 V, $${{2.303\,RT} \over F} = 0.059$$ at 298 K)
Q69.
mcq single
+4 / 1
At 298 K, the standard electrode potentials of Cu^(2+) / Cu, Zn^(2+) / Zn, Fe^(2+) / Fe and Ag^(+) / Ag are 0.34 V, $$-$$0.76 V, $$-$$0.44 V V and 0.80 V, respectively.
On the basis of standard electrode potential, predict which of the following reaction cannot occur?
Q70.
mcq single
+4 / 1
Which of the following sequence of reactions is suitable to synthesize chlorobenzene?
Q71.
mcq single
+4 / 1
The product formed from the following reaction sequence is
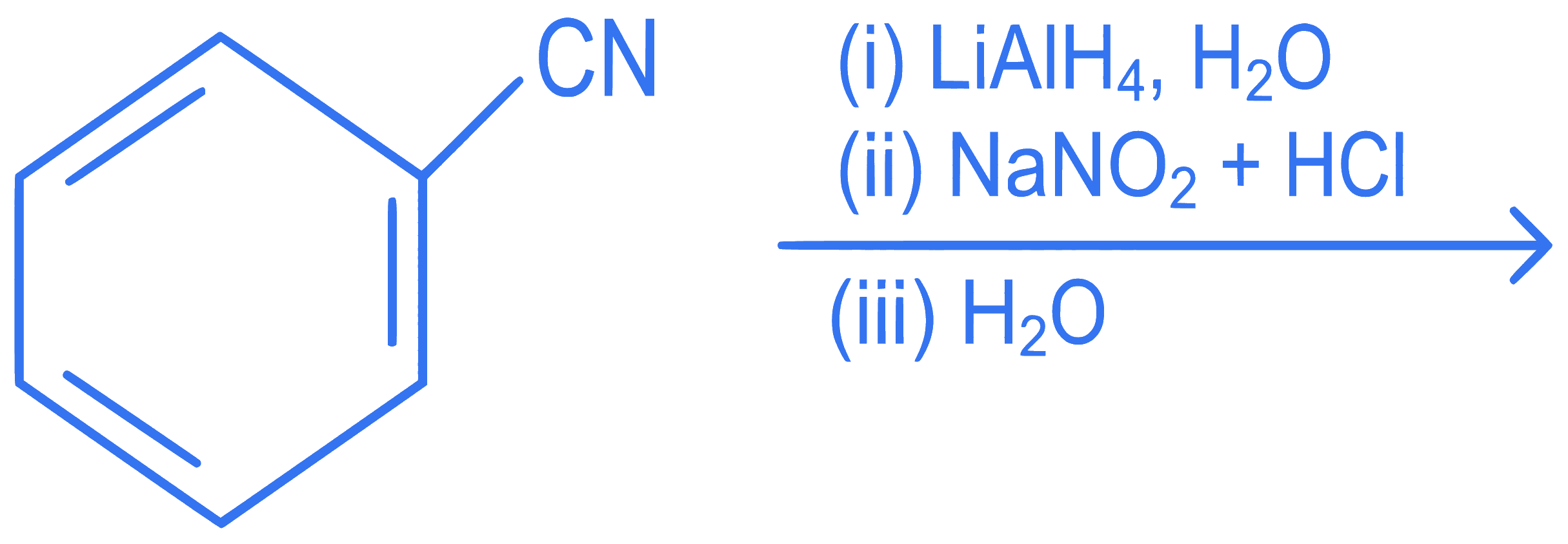
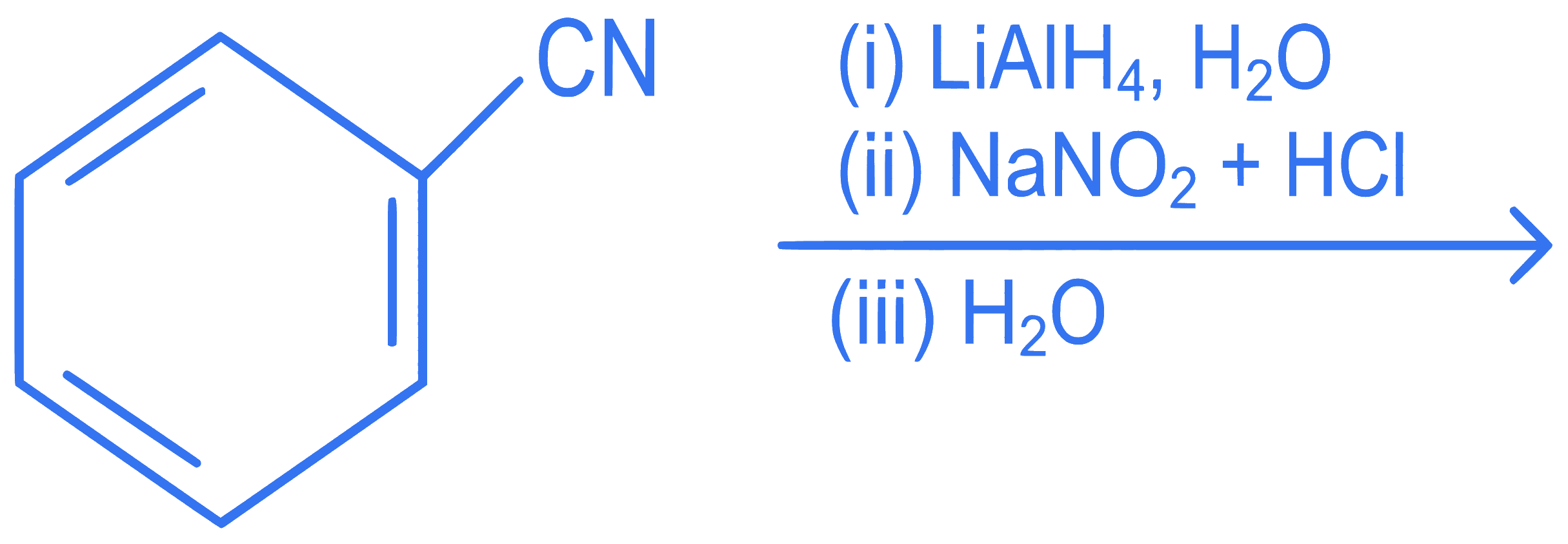
Q72.
mcq single
+4 / 1
Given below are two statements
Statement I : Primary aliphatic amines react with HNO~2~ to give unstable diazonium salts.
Statement II : Primary aromatic amines react with HNO~2~ to form diazonium salts which are stable even above 300 K.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q73.
mcq single
+4 / 1
Identify the incorrect statement from the following.
Q74.
mcq single
+4 / 1
If radius of second Bohr orbit of the He^(+) ion is 105.8 pm, what is the radius of third Bohr orbit of Li^(2+) ion?
Physics
Physics
Q1.
mcq single
+4 / 1
In a Young's double slit experiment, a student observes 8 fringes in a certain segment of screen when a monochromatic light of 600 nm wavelength is used. If the wavelength of light is changed to 400 nm, then the number of fringes he would observe in the same region of the screen is
Q2.
mcq single
+4 / 1
The angular speed of a fly wheel moving with uniform angular acceleration changes from 1200 rpm to 3120 rpm in 16 seconds. The angular acceleration in rad/s^(2) is
Q3.
mcq single
+4 / 1
The ratio of the radius of gyration of a thin uniform disc about an axis passing through its centre and normal to its plane to the radius of gyration of the disc about its diameter is
Q4.
mcq single
+4 / 1
A square loop of side 1 m and resistance 1 $$\Omega$$ is placed in a magnetic field of 0.5 T. If the plane of loop is perpendicular to the direction of magnetic field, the magnetic flux through the loop is
Q5.
mcq single
+4 / 1
Plane angle and solid angle have
Q6.
mcq single
+4 / 1
The area of a rectangular field (in m^(2)) of length 55.3 m and breadth 25 m after rounding off the value for correct significant digits is
Q7.
mcq single
+4 / 1
The dimensions [MLT^($$-$$2)A^($$-$$2)] belong to the
Q8.
mcq single
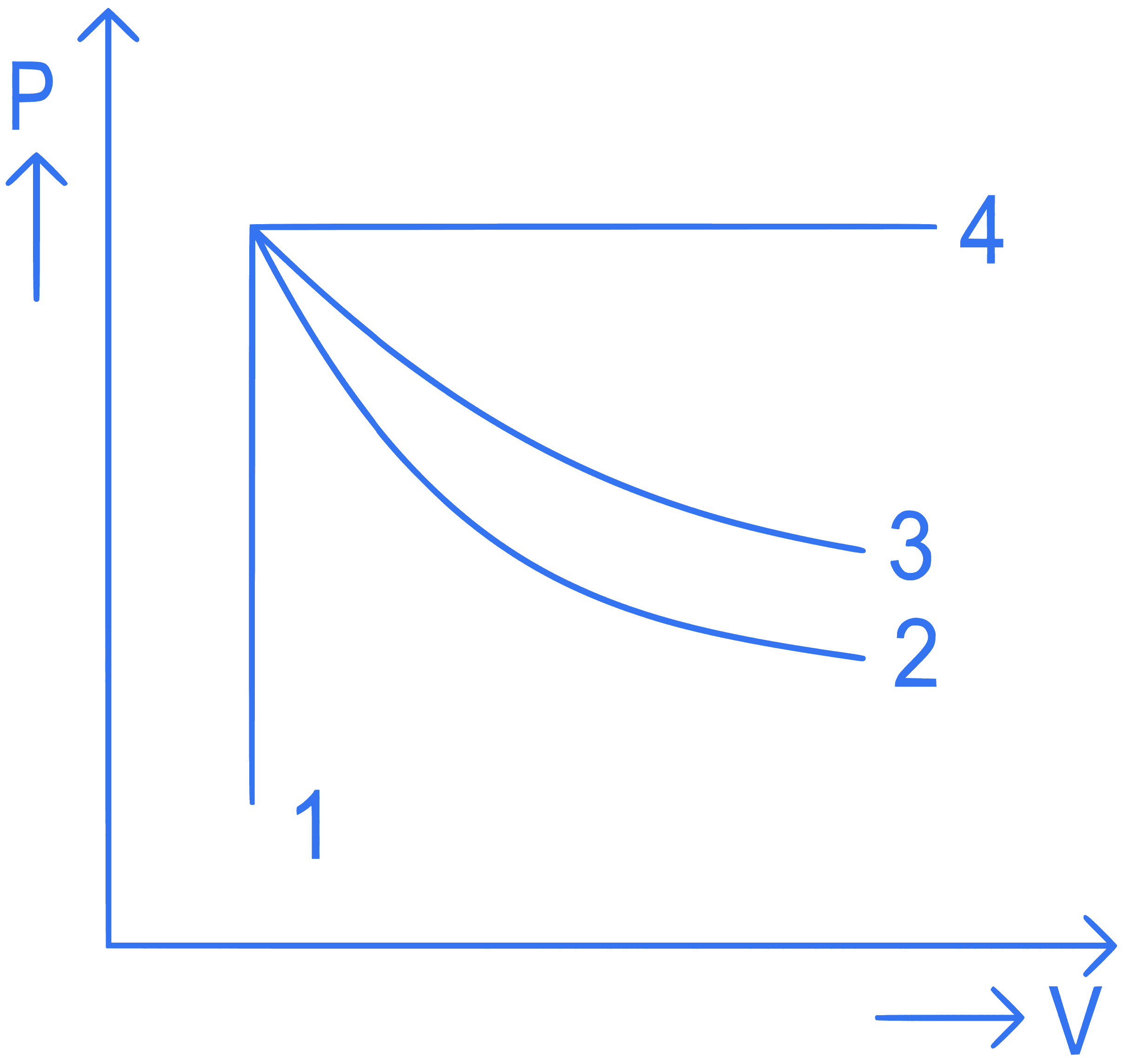
+4 / 1
An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among 1, 2, 3 and 4 is

Q9.
mcq single
+4 / 1
The volume occupied by the molecules contained in 4.5 kg water at STP, if the intermolecular forces vanish away is
Q10.
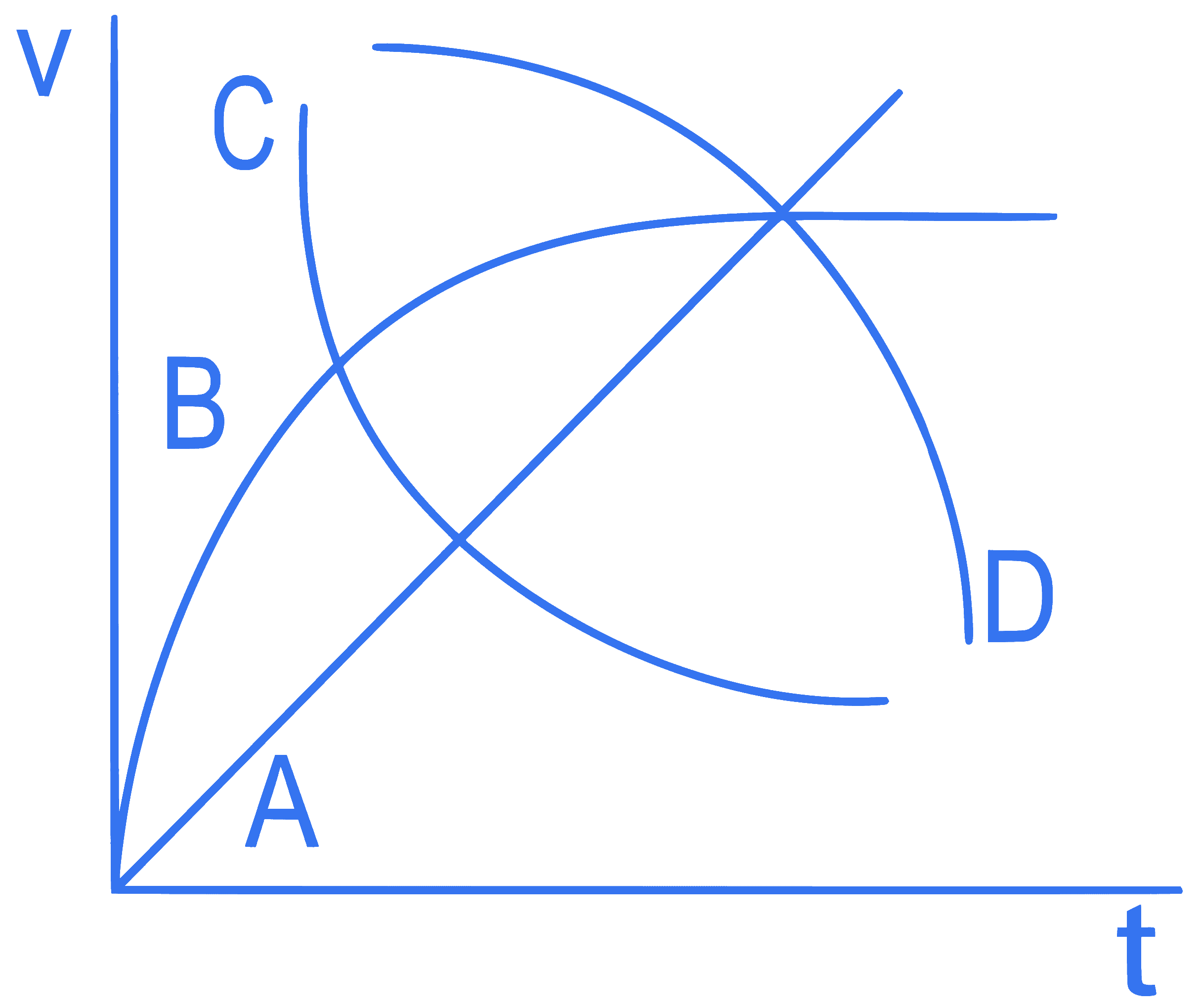
mcq single
+4 / 1
A spherical ball is dropped in a long column of a highly viscous liquid. The curve in the graph shown, which represents the speed of the ball (v) as a function of time (t) is

Q11.
mcq single
+4 / 1
If a soap bubble expands, the pressure inside the bubble
Q12.
mcq single
+4 / 1
Given below are two statements : One is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : The stretching of a spring is determined by the shear modulus of the material of the spring.
Reason (R) : A coil spring of copper has more tensile strength than a steel spring of same dimensions.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q13.
mcq single
+4 / 1
Two point charges $$-$$q and +q are placed at a distance of L, as shown in the figure.
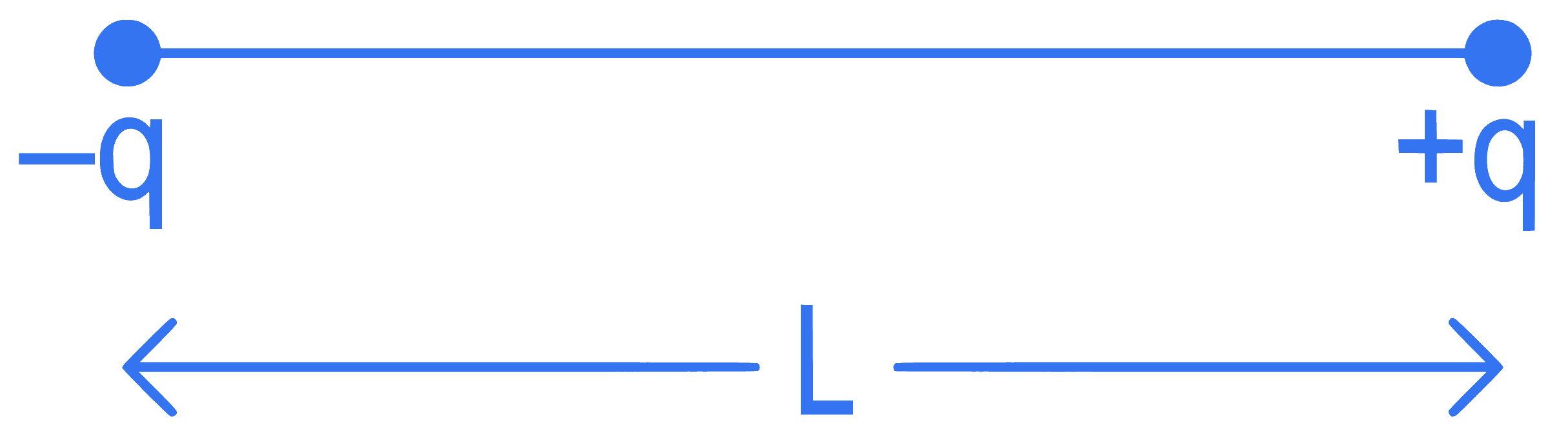 The magnitude of electric field intensity at a distance R(R >> L) varies as:
The magnitude of electric field intensity at a distance R(R >> L) varies as:
 The magnitude of electric field intensity at a distance R(R >> L) varies as:
The magnitude of electric field intensity at a distance R(R >> L) varies as:
Q14.
mcq single
+4 / 1
The angle between the electric lines of force and the equipotential surface is
Q15.
mcq single
+4 / 1
Two hollow conducting spheres of radii R~1~ and R~2~ (R~1~ >> R~2~) have equal charges. The potential would be
Q16.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(a)
Gravitational constant (G)
(i)
$$[{L^2}{T^{ - 2}}]$$
(b)
Gravitational potential energy
(ii)
$$[{M^{ - 1}}{L^3}{T^{ - 2}}]$$
(c)
Gravitational potential
(iii)
$$[L{T^{ - 2}}]$$
(d)
Gravitational intensity
(iv)
$$[M{L^2}{T^{ - 2}}]$$
Choose the correct answer from the options given below
Q17.
mcq single
+4 / 1
A body of mass 60 g experiences a gravitational force of 3.0 N, when placed at a particular point. The magnitude of the gravitational field intensity at that point is
Q18.
mcq single
+4 / 1
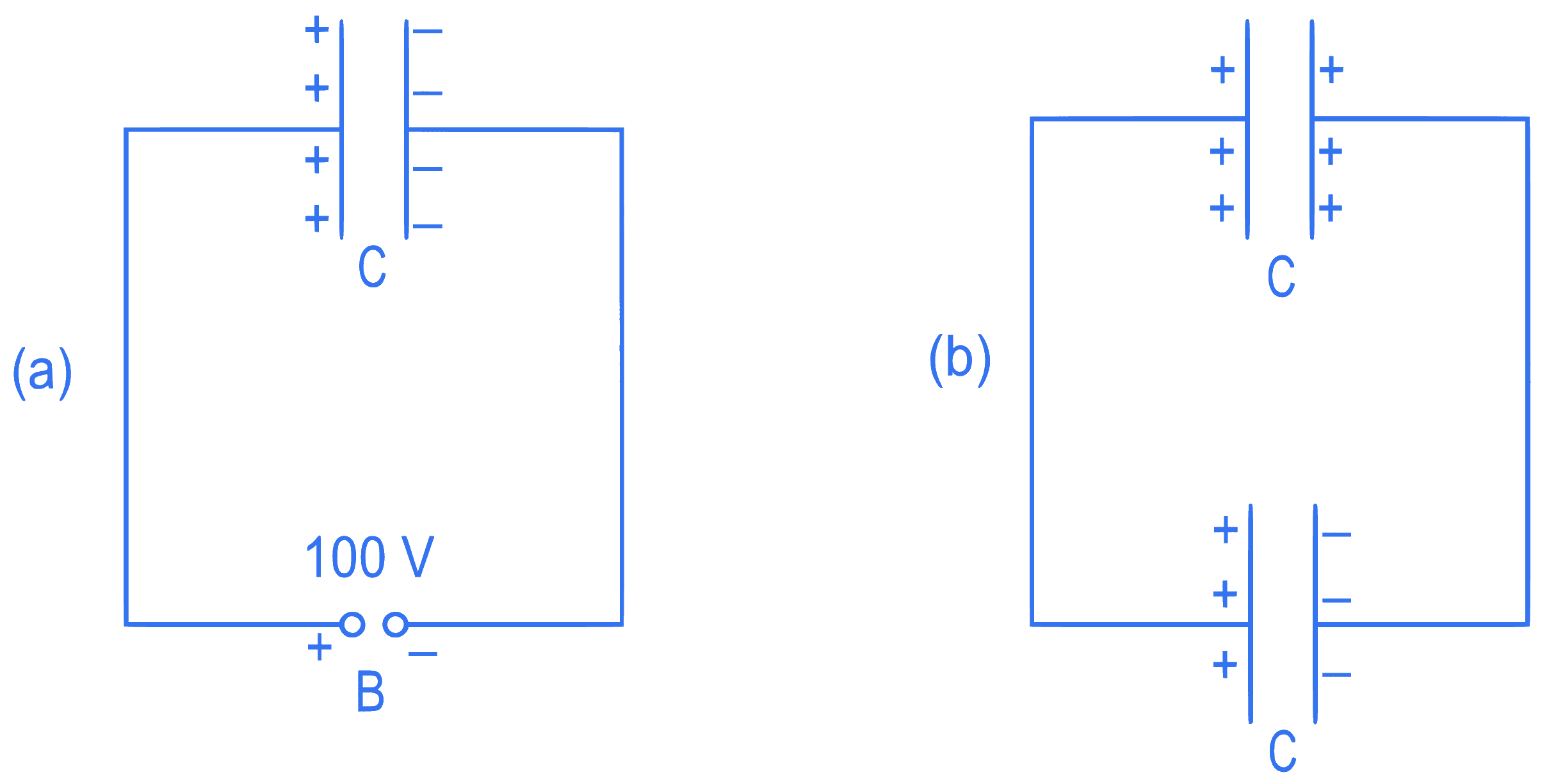
A capacitor of capacitance C = 900 pF is charged fully by 100 V battery B as shown in figure (a). Then it is disconnected from the battery and connected to another uncharged capacitor of capacitance C = 900 pF as shown in figure (b). The electrostatic energy stored by the system (b) is

Q19.
mcq single
+4 / 1
Two transparent media A and B are separated by a plane boundary. The speed of light in those media are 1.5 $$\times$$ 10^(8) m/s and 2.0 $$\times$$ 10^(8) m/s, respectively. The critical angle for a ray of light for these two media is
Q20.
mcq single
+4 / 1
A biconvex lens has radii of curvature, 20 cm each. If the refractive index of the material of the lens is 1.5, the power of the lens is
Q21.
mcq single
+4 / 1
A light ray falls on a glass surface of refractive index $$\sqrt3$$, at an angle 60$$^\circ$$. The angle between the refracted and reflected rays would be
Q22.
mcq single
+4 / 1
Two pendulums of length 121 cm and 100 cm start vibrating in phase. At some instant, the two are at their mean position in the same phase. The minimum number of vibrations of the shorter pendulum after which the two are again in phase at the mean position is :
Q23.
mcq single
+4 / 1
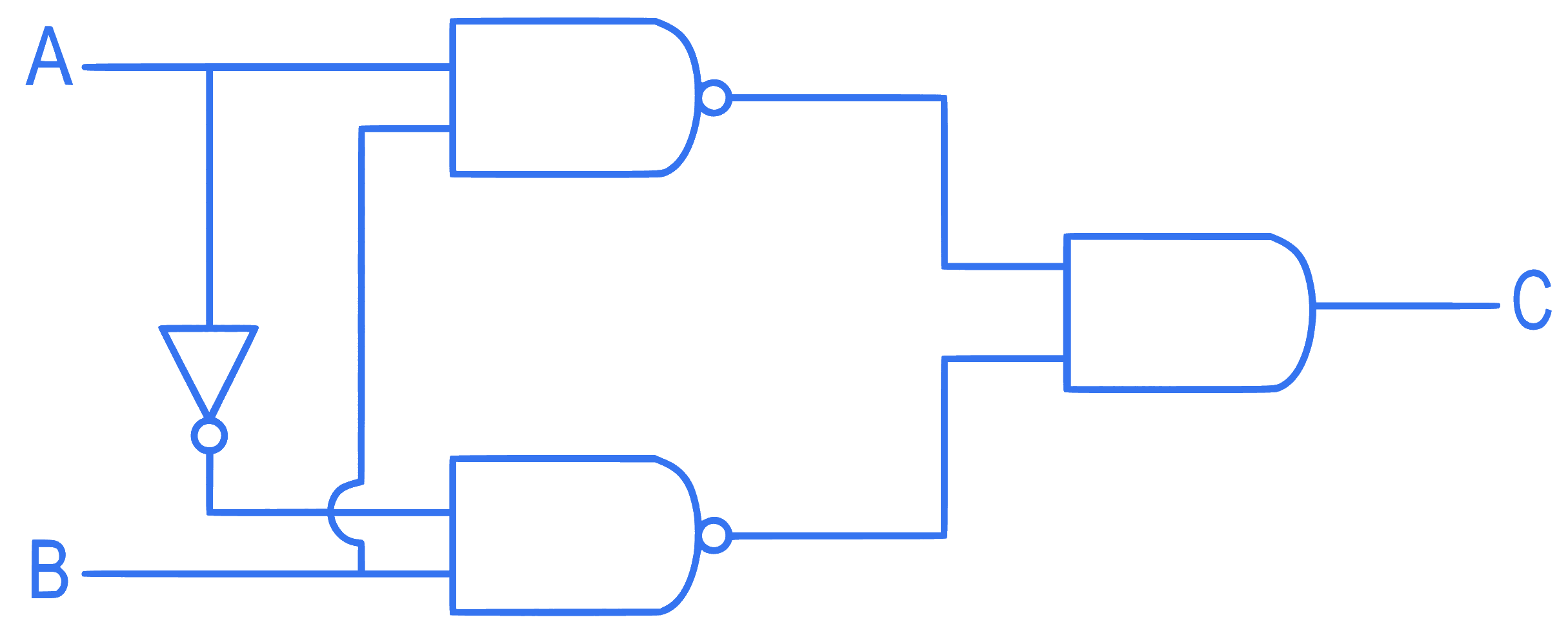 The truth table for the given logic circuit is
The truth table for the given logic circuit is
 The truth table for the given logic circuit is
The truth table for the given logic circuit is
Q24.
mcq single
+4 / 1
As the temperature increases, the electrical resistance
Q25.
mcq single
+4 / 1
In half wave rectification, if the input frequency is 60 Hz, then the output frequency would be
Q26.
mcq single
+4 / 1
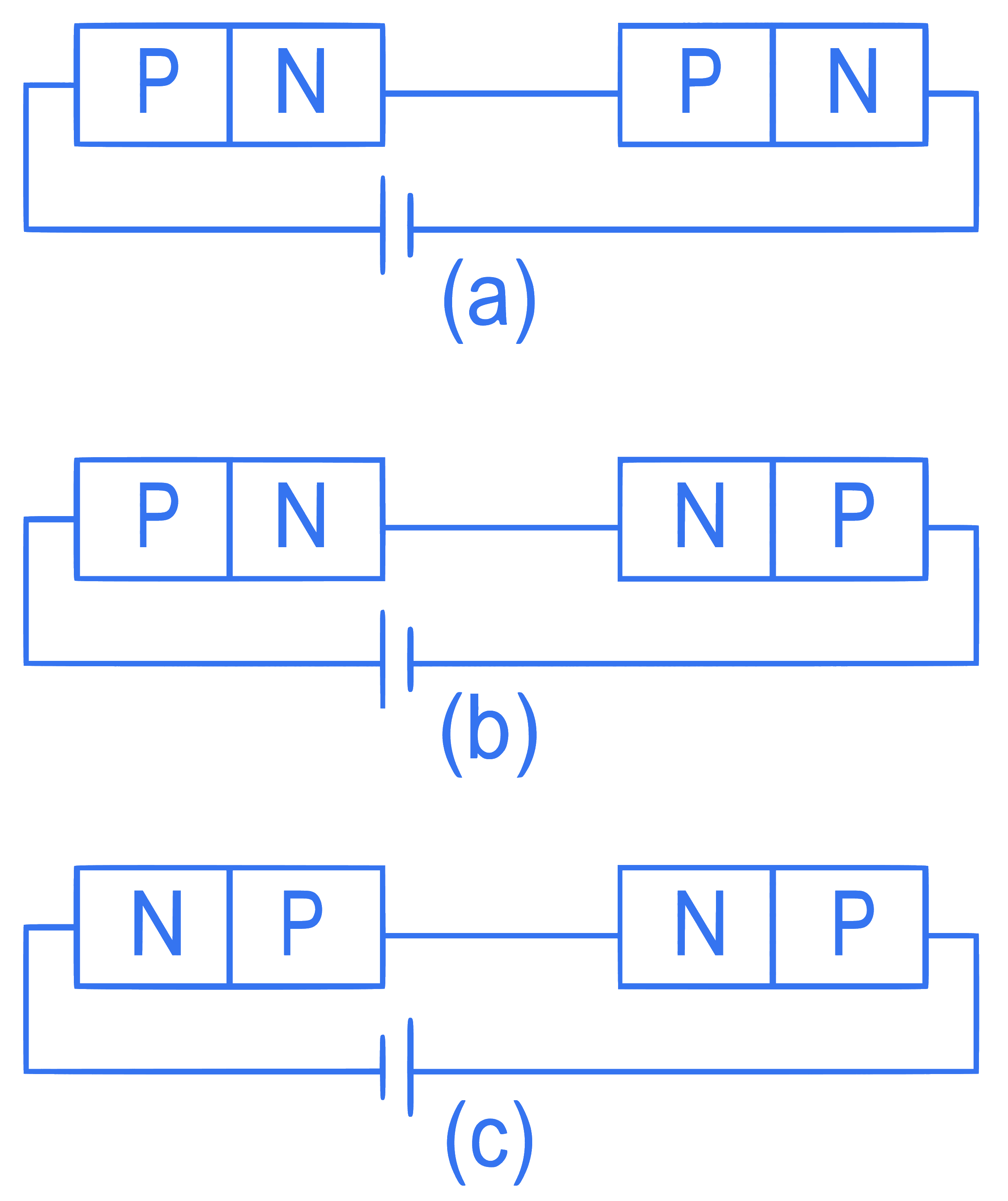 In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
 In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
Q27.
mcq single
+4 / 1
Given below are two statements:
Statement I : Biot-Savart's law gives us the expression for the magnetic field strength of an infinitesimal current element (Idl) of a current carrying conductor only.
Statement II : Biot-Savart's law is analogous to Coulomb's inverse square law of charge q, with the former being related to the field produced by a scalar source, Idl while the latter being produced by a vector source, q.
In light of above statements choose the most appropriate answer from the options given below.
Q28.
mcq single
+4 / 1
From Ampere's circuital law for a long straight wire of circular cross-section carrying a steady current, the variation of magnetic field in the inside and outside region of the wire is
Q29.
mcq single
+4 / 1
A long solenoid of radius 1 mm has 100 turns per mm. If 1 A current flows in the solenoid, the magnetic field strength at the centre of the solenoid is
Q30.
mcq single
+4 / 1
When two monochromatic lights of frequency, v and $${v \over 2}$$ are incident on a photoelectric metal, their stopping potential becomes $${{{V_s}} \over 2}$$ and V~s~ respectively. The threshold frequency for this metal is
Q31.
mcq single
+4 / 1
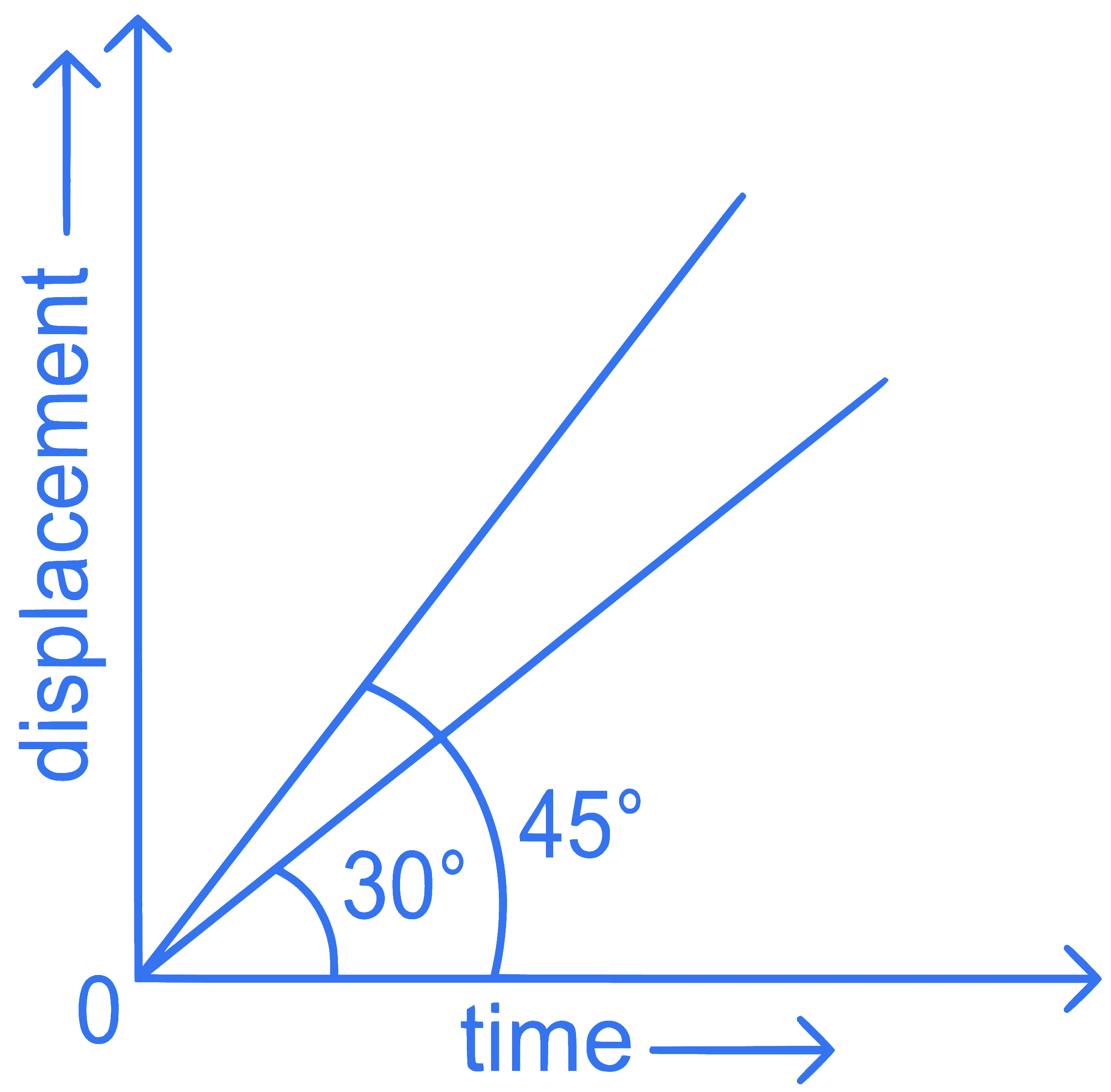
The displacement-time graphs of two moving particles make angles of 30$$^\circ$$ and 45$$^\circ$$ with the x-axis as shown in the figure. The ratio of their respective velocity is

Q32.
mcq single
+4 / 1
The ratio of the distances travelled by a freely falling body in the 1^(st), 2^(nd), 3^(rd) and 4^(th) second
Q33.
mcq single
+4 / 1
A nucleus of mass number 189 splits into two nuclei having mass number 125 and 64. The ratio of radius of two daughter nuclei respectively is
Q34.
mcq single
+4 / 1
Let T~1~ and T~2~ be the energy of an electron in the first and second excited states of hydrogen atoms, respectively. According to the Bohr's model of an atom, the ratio T~1~ : T~2~ is
Q35.
mcq single
+4 / 1
A ball is projected with a velocity, 10 ms^($$-$$1), at an angle of 60$$^\circ$$ with the vertical direction. Its speed at the highest point of its trajectory will be
Q36.
mcq single
+4 / 1
Two objects of mass 10 kg and 20 kg respectively are connected to the two ends of a rigid rod of length 10 m with negligible mass. The distance of the center of mass of the system from the 10 kg mass is :
Q37.
mcq single
+4 / 1
A shell of mass m is at rest initially. It explodes into three fragments having mass in the ratio 2 : 2 : 1. If the fragments having equal mass fly off along mutually perpendicular directions with speed v, the speed of the third (lighter) fragment is :
Q38.
mcq single
+4 / 1
If the initial tension on a stretched string is doubled, then the ratio of the initial and final speeds of a transverse wave along the string is
Q39.
mcq single
+4 / 1
A big circular coil of 1000 turns and average radius 10 m is rotating about its horizontal diameter at 2 rad s^($$-$$1). If the vertical component of earth's magnetic field at that place is 2 $$\times$$ 10^($$-$$5) T and electrical resistance of the coil is 12.56 $$\Omega$$, then the maximum induced current in the coil will be
Q40.
mcq single
+4 / 1
The energy that will be ideally radiated by a 100 kW transmitter in 1 hour is
Q41.
mcq single
+4 / 1
An electric lift with a maximum load of 2000 kg (lift + passengers) is moving up with a constant speed of 1.5 ms^($$-$$1). The frictional force opposing the motion is 3000 N. The minimum power delivered by the motor to the lift in watts is : (g = 10 ms^($$-$$2))
Q42.
mcq single
+4 / 1
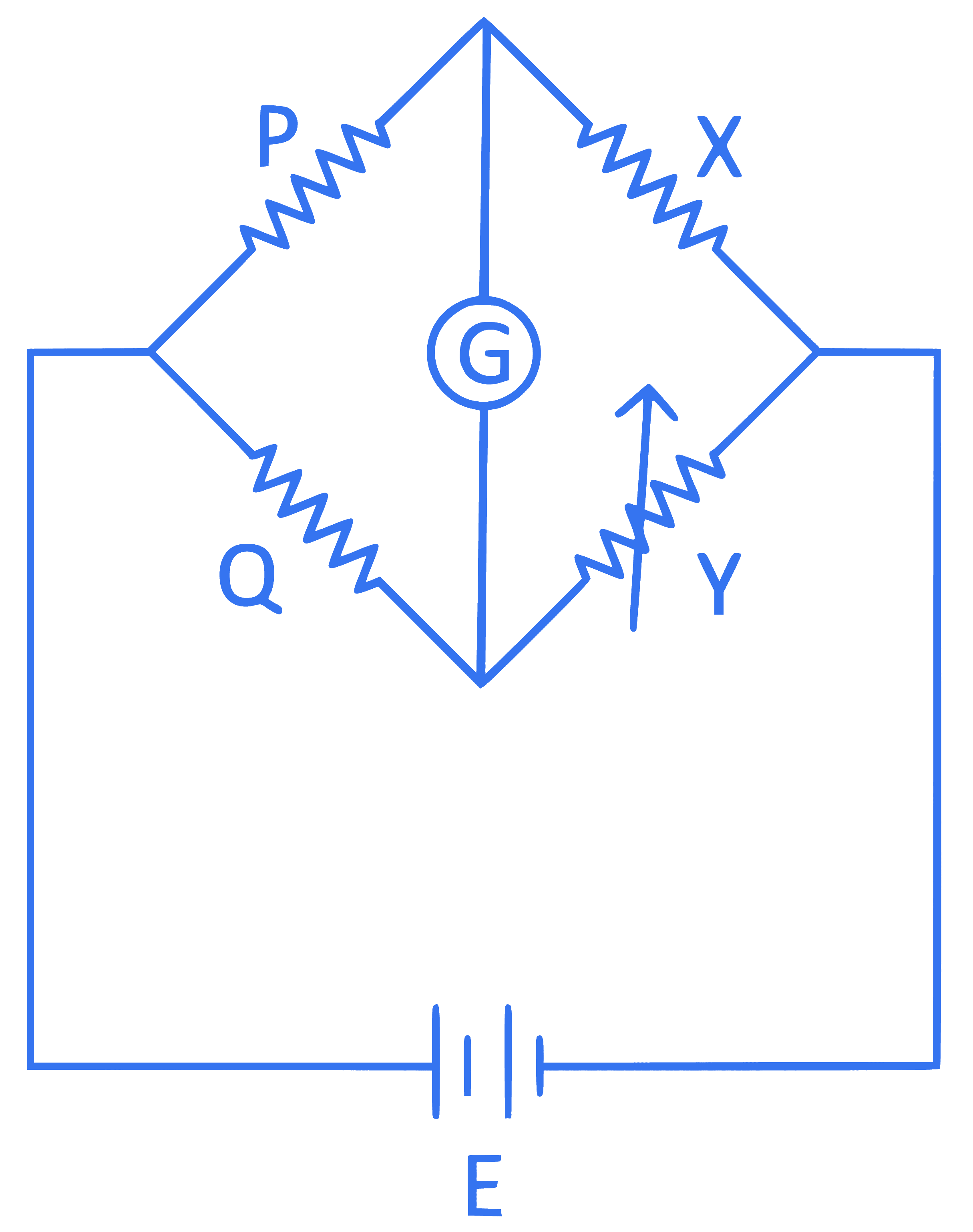
A wheatstone bridge is used to determine the value of unknown resistance X by adjusting the variable resistance Y as shown in the figure. For the most precise measurement of X, the resistance P and Q

Q43.
mcq single
+4 / 1
A copper wire of length 10 m and radius $$\left( {{{{{10}^{ - 2}}} \over {\sqrt \pi }}} \right)$$ m has electrical resistance of 10 $$\Omega$$. The current density in the wire for an electric field strength of 10 (V/m) is
Q44.
mcq single
+4 / 1
Two resistors of resistance, 100 $$\Omega$$ and 200 $$\Omega$$ are connected in parallel in an electrical circuit. The ratio of the thermal energy developed in 100 $$\Omega$$ to that in 200 $$\Omega$$ in a given time is
Q45.
mcq single
+4 / 1
Match List - I with List - II
List - I
(Electromagnetic waves)
List - II
(Wavelength)
(a)
AM radio waves
(i)
$${10^{ - 10}}$$ m
(b)
Microwaves
(ii)
$${10^2}$$ m
(c)
Infrared radiations
(iii)
$${10^{ - 2}}$$ m
(d)
X-rays
(iv)
$${10^{ - 4}}$$ m
Choose the correct answer from the options given below
Q46.
mcq single
+4 / 1
When light propagates through a material medium of relative permittivity $$\varepsilon $$~r~ and relative permeability $$\mu$$~r~, the velocity of light, v is given by (c-velocity of light in vacuum)
Q47.
mcq single
+4 / 1
The peak voltage of the ac source is equal to
Q48.
mcq single
+4 / 1
A series LCR circuit with inductance 10 H, capacitance 10 $$\mu$$F, resistance 50 $$\Omega$$ is connected to an ac source of voltage, V = 200sin(100t) volt. If the resonant frequency of the LCR circuit is v~0~ and the frequency of the ac source is v, then
Q49.
mcq single
+4 / 1
In a Young's double slit experiment, a student observes 8 fringes in a certain segment of screen when a monochromatic light of 600 nm wavelength is used. If the wavelength of light is changed to 400 nm, then the number of fringes he would observe in the same region of the screen is
Q50.
mcq single
+4 / 1
The angular speed of a fly wheel moving with uniform angular acceleration changes from 1200 rpm to 3120 rpm in 16 seconds. The angular acceleration in rad/s^(2) is
Q51.
mcq single
+4 / 1
The ratio of the radius of gyration of a thin uniform disc about an axis passing through its centre and normal to its plane to the radius of gyration of the disc about its diameter is
Q52.
mcq single
+4 / 1
A square loop of side 1 m and resistance 1 $$\Omega$$ is placed in a magnetic field of 0.5 T. If the plane of loop is perpendicular to the direction of magnetic field, the magnetic flux through the loop is
Q53.
mcq single
+4 / 1
Plane angle and solid angle have
Q54.
mcq single
+4 / 1
The area of a rectangular field (in m^(2)) of length 55.3 m and breadth 25 m after rounding off the value for correct significant digits is
Q55.
mcq single
+4 / 1
The dimensions [MLT^($$-$$2)A^($$-$$2)] belong to the
Q56.
mcq single
+4 / 1
An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among 1, 2, 3 and 4 is


Q57.
mcq single
+4 / 1
The volume occupied by the molecules contained in 4.5 kg water at STP, if the intermolecular forces vanish away is
Q58.
mcq single
+4 / 1
A spherical ball is dropped in a long column of a highly viscous liquid. The curve in the graph shown, which represents the speed of the ball (v) as a function of time (t) is


Q59.
mcq single
+4 / 1
If a soap bubble expands, the pressure inside the bubble
Q60.
mcq single
+4 / 1
Given below are two statements : One is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : The stretching of a spring is determined by the shear modulus of the material of the spring.
Reason (R) : A coil spring of copper has more tensile strength than a steel spring of same dimensions.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q61.
mcq single
+4 / 1
Two point charges $$-$$q and +q are placed at a distance of L, as shown in the figure.
 The magnitude of electric field intensity at a distance R(R >> L) varies as:
The magnitude of electric field intensity at a distance R(R >> L) varies as:
 The magnitude of electric field intensity at a distance R(R >> L) varies as:
The magnitude of electric field intensity at a distance R(R >> L) varies as:
Q62.
mcq single
+4 / 1
The angle between the electric lines of force and the equipotential surface is
Q63.
mcq single
+4 / 1
Two hollow conducting spheres of radii R~1~ and R~2~ (R~1~ >> R~2~) have equal charges. The potential would be
Q64.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(a)
Gravitational constant (G)
(i)
$$[{L^2}{T^{ - 2}}]$$
(b)
Gravitational potential energy
(ii)
$$[{M^{ - 1}}{L^3}{T^{ - 2}}]$$
(c)
Gravitational potential
(iii)
$$[L{T^{ - 2}}]$$
(d)
Gravitational intensity
(iv)
$$[M{L^2}{T^{ - 2}}]$$
Choose the correct answer from the options given below
Q65.
mcq single
+4 / 1
A body of mass 60 g experiences a gravitational force of 3.0 N, when placed at a particular point. The magnitude of the gravitational field intensity at that point is
Q66.
mcq single
+4 / 1
A capacitor of capacitance C = 900 pF is charged fully by 100 V battery B as shown in figure (a). Then it is disconnected from the battery and connected to another uncharged capacitor of capacitance C = 900 pF as shown in figure (b). The electrostatic energy stored by the system (b) is


Q67.
mcq single
+4 / 1
Two transparent media A and B are separated by a plane boundary. The speed of light in those media are 1.5 $$\times$$ 10^(8) m/s and 2.0 $$\times$$ 10^(8) m/s, respectively. The critical angle for a ray of light for these two media is
Q68.
mcq single
+4 / 1
A biconvex lens has radii of curvature, 20 cm each. If the refractive index of the material of the lens is 1.5, the power of the lens is
Q69.
mcq single
+4 / 1
A light ray falls on a glass surface of refractive index $$\sqrt3$$, at an angle 60$$^\circ$$. The angle between the refracted and reflected rays would be
Q70.
mcq single
+4 / 1
Two pendulums of length 121 cm and 100 cm start vibrating in phase. At some instant, the two are at their mean position in the same phase. The minimum number of vibrations of the shorter pendulum after which the two are again in phase at the mean position is :
Q71.
mcq single
+4 / 1
 The truth table for the given logic circuit is
The truth table for the given logic circuit is
 The truth table for the given logic circuit is
The truth table for the given logic circuit is
Q72.
mcq single
+4 / 1
As the temperature increases, the electrical resistance
Q73.
mcq single
+4 / 1
In half wave rectification, if the input frequency is 60 Hz, then the output frequency would be
Q74.
mcq single
+4 / 1
 In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
 In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
In the given circuits (a), (b) and (c), the potential drop across the two p-n junctions are equal in
Q75.
mcq single
+4 / 1
Given below are two statements:
Statement I : Biot-Savart's law gives us the expression for the magnetic field strength of an infinitesimal current element (Idl) of a current carrying conductor only.
Statement II : Biot-Savart's law is analogous to Coulomb's inverse square law of charge q, with the former being related to the field produced by a scalar source, Idl while the latter being produced by a vector source, q.
In light of above statements choose the most appropriate answer from the options given below.
Q76.
mcq single
+4 / 1
From Ampere's circuital law for a long straight wire of circular cross-section carrying a steady current, the variation of magnetic field in the inside and outside region of the wire is
Q77.
mcq single
+4 / 1
A long solenoid of radius 1 mm has 100 turns per mm. If 1 A current flows in the solenoid, the magnetic field strength at the centre of the solenoid is
Q78.
mcq single
+4 / 1
When two monochromatic lights of frequency, v and $${v \over 2}$$ are incident on a photoelectric metal, their stopping potential becomes $${{{V_s}} \over 2}$$ and V~s~ respectively. The threshold frequency for this metal is
Q79.
mcq single
+4 / 1
The displacement-time graphs of two moving particles make angles of 30$$^\circ$$ and 45$$^\circ$$ with the x-axis as shown in the figure. The ratio of their respective velocity is


Q80.
mcq single
+4 / 1
The ratio of the distances travelled by a freely falling body in the 1^(st), 2^(nd), 3^(rd) and 4^(th) second
Q81.
mcq single
+4 / 1
A nucleus of mass number 189 splits into two nuclei having mass number 125 and 64. The ratio of radius of two daughter nuclei respectively is
Q82.
mcq single
+4 / 1
Let T~1~ and T~2~ be the energy of an electron in the first and second excited states of hydrogen atoms, respectively. According to the Bohr's model of an atom, the ratio T~1~ : T~2~ is
Q83.
mcq single
+4 / 1
A ball is projected with a velocity, 10 ms^($$-$$1), at an angle of 60$$^\circ$$ with the vertical direction. Its speed at the highest point of its trajectory will be
Q84.
mcq single
+4 / 1
Two objects of mass 10 kg and 20 kg respectively are connected to the two ends of a rigid rod of length 10 m with negligible mass. The distance of the center of mass of the system from the 10 kg mass is :
Q85.
mcq single
+4 / 1
A shell of mass m is at rest initially. It explodes into three fragments having mass in the ratio 2 : 2 : 1. If the fragments having equal mass fly off along mutually perpendicular directions with speed v, the speed of the third (lighter) fragment is :
Q86.
mcq single
+4 / 1
If the initial tension on a stretched string is doubled, then the ratio of the initial and final speeds of a transverse wave along the string is
Q87.
mcq single
+4 / 1
A big circular coil of 1000 turns and average radius 10 m is rotating about its horizontal diameter at 2 rad s^($$-$$1). If the vertical component of earth's magnetic field at that place is 2 $$\times$$ 10^($$-$$5) T and electrical resistance of the coil is 12.56 $$\Omega$$, then the maximum induced current in the coil will be
Q88.
mcq single
+4 / 1
The energy that will be ideally radiated by a 100 kW transmitter in 1 hour is
Q89.
mcq single
+4 / 1
An electric lift with a maximum load of 2000 kg (lift + passengers) is moving up with a constant speed of 1.5 ms^($$-$$1). The frictional force opposing the motion is 3000 N. The minimum power delivered by the motor to the lift in watts is : (g = 10 ms^($$-$$2))
Q90.
mcq single
+4 / 1
A wheatstone bridge is used to determine the value of unknown resistance X by adjusting the variable resistance Y as shown in the figure. For the most precise measurement of X, the resistance P and Q


Q91.
mcq single
+4 / 1
A copper wire of length 10 m and radius $$\left( {{{{{10}^{ - 2}}} \over {\sqrt \pi }}} \right)$$ m has electrical resistance of 10 $$\Omega$$. The current density in the wire for an electric field strength of 10 (V/m) is
Q92.
mcq single
+4 / 1
Two resistors of resistance, 100 $$\Omega$$ and 200 $$\Omega$$ are connected in parallel in an electrical circuit. The ratio of the thermal energy developed in 100 $$\Omega$$ to that in 200 $$\Omega$$ in a given time is
Q93.
mcq single
+4 / 1
Match List - I with List - II
List - I
(Electromagnetic waves)
List - II
(Wavelength)
(a)
AM radio waves
(i)
$${10^{ - 10}}$$ m
(b)
Microwaves
(ii)
$${10^2}$$ m
(c)
Infrared radiations
(iii)
$${10^{ - 2}}$$ m
(d)
X-rays
(iv)
$${10^{ - 4}}$$ m
Choose the correct answer from the options given below
Q94.
mcq single
+4 / 1
When light propagates through a material medium of relative permittivity $$\varepsilon $$~r~ and relative permeability $$\mu$$~r~, the velocity of light, v is given by (c-velocity of light in vacuum)
Q95.
mcq single
+4 / 1
The peak voltage of the ac source is equal to
Q96.
mcq single
+4 / 1
A series LCR circuit with inductance 10 H, capacitance 10 $$\mu$$F, resistance 50 $$\Omega$$ is connected to an ac source of voltage, V = 200sin(100t) volt. If the resonant frequency of the LCR circuit is v~0~ and the frequency of the ac source is v, then
Biology
Biology
Q1.
mcq single
+4 / 1
Identify the microorganism which is responsible for the production of an immunosuppressive molecule cyclosporin A :
Q2.
mcq single
+4 / 1
Which one of the following will accelerate phosphorus cycle?
Q3.
mcq single
+4 / 1
Detritivores breakdown detritus into smaller particles. This process is called :
Q4.
mcq single
+4 / 1
Given below are two statements:
Statement I : Decomposition is a process in which the detritus is degraded into simpler substances by microbes.
Statement II : Decomposition is faster if the detritus is rich in lignin and chitin.
In the light of the above statements, choose the correct answer from the options given below :
Q5.
mcq single
+4 / 1
Given below are two statements:
Statement I : Mendel studied seven pairs of contrasting traits in pea plants and proposed the Laws of Inheritance.
Statement II : Seven characters examined by Mendel in his experiment on pea plants were seed shape and colour, flower colour, pod shape and colour, flower position and stem height.
In the light of the above statements, choose the correct answer from the options given below :
Q6.
mcq single
+4 / 1
If a colour blind female marries a man whose mother was also colour blind, what are the chances of her progeny having colour blindness?
Q7.
mcq single
+4 / 1
XO type of sex determination can be found in :
Q8.
mcq single
+4 / 1
Which of the following occurs due to the presence of autosome linked dominant trait?
Q9.
mcq single
+4 / 1
The recombination frequency between the genes a & c 5&, b & c is 15%, b & d is 9%, a & b is 20%, c & d is 25% and a & d is 29%. What will be the sequence of these genes on a linear chromosome?
Q10.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Mendel's law of independent assortment does not hold good for the genes that are located closely on the same chromosome.
Reason (R) : Closely located genes assort independently.
In the light of the above statements, choose the correct answer from the options given below :
Q11.
mcq single
+4 / 1
Which of the following is not a connective tissue?
Q12.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(a)
Bronchioles
(i)
Dense Regular Connective Tissue
(b)
Goblet Cell
(ii)
Loose Connective Tissue
(c)
Tendons
(iii)
Glandular Tissue
(d)
Adipose Tissue
(iv)
Ciliated Epithelium
Choose the correct answer from the options given below:
Q13.
mcq single
+4 / 1
Tegmina in cockroach, arises from
Q14.
mcq single
+4 / 1
In which of the following animals, digestive tract has additional chambers like crop and gizzard?
Q15.
mcq single
+4 / 1
Which one of the following is not true regarding the release of energy during ATP synthesis through chemiosmosis? It involves:
Q16.
mcq single
+4 / 1
Given below are two statements:
Statement I : The primary CO~2~ acceptor in C~4~ plants is phosphoenolpyruvate and is found in the mesophyll cells.
Statement II : Mesophyll cells of C~4~ plants lack RuBisCo enzyme.
In the light of the above statements, choose the correct answer from the options given below:
Q17.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : All vertebrates are chordates but all chordates are not vertebrates.
Reason (R) : Notochord is replaced by vertebral column in the adult vertebrates.
In the light of the above statements, choose the most appropriate answer from the option given below:
Q18.
mcq single
+4 / 1
Exoskeleton of arthropods is composed of:
Q19.
mcq single
+4 / 1
Hydrocolloid carrageen is obtained form :
Q20.
mcq single
+4 / 1
Which of the following is incorrectly matched?
Q21.
mcq single
+4 / 1
Match the plant with the kind of life cycle it exhibits:
List - I
List - II
(a)
Spirogyra
(i)
Dominant diploid sporophyte vascular plant, with highly reduced male or female gametophyte
(b)
Fern
(ii)
Dominant haploid free-living gametophyte
(c)
Funaria
(iii)
Dominant diploid sporophyte alternating with reduced gametophyte called prothallus
(d)
Cycas
(iv)
Dominant haploid leafy gametophyte alternating with partially dependent multicellular sporophyte
Choose the correct answer from the options given below :
Q22.
mcq single
+4 / 1
Which of the following is not the function of conducting part of respiratory system?
Q23.
mcq single
+4 / 1
Under normal physiological conditions in human being every 100 ml of oxygenated blood can deliver ____________ ml of O~2~ to the tissues.
Q24.
mcq single
+4 / 1
In the taxonomic categories which hierarchical arrangement in ascending order is correct in case of animals?
Q25.
mcq single
+4 / 1
In gene therapy of Adenosine Deaminase (ADA) deficiency, the patient requires periodic infusion of genetically engineered lymphocytes because:
Q26.
mcq single
+4 / 1
In the following palindromic base sequences of DNA, which one can be cut easily by particular restriction enzyme?
Q27.
mcq single
+4 / 1
Statements related to human Insulin are given below.
Which statements is/are correct about genetically engineered Insulin?
(a) Pro-hormone insulin contain extra stretch of C-peptide
(b) A-peptide and B-peptide chains of insulin were produced separately in E.coli, extracted and combined by creating disulphide bond between them.
(c) Insulin used for treating Diabetes was extracted from Cattles and Pigs.
(d) Pro-hormone Insulin needs to be processed for converting into a mature and functional hormone.
(e) Some patients develop allergic reactions to the foreign insulin.
Choose the most appropriate answer from the options given below :
Q28.
mcq single
+4 / 1
Select the incorrect statement regarding synapses :
Q29.
mcq single
+4 / 1
The anatomy of springwood shows some peculiar features. Identify the correct set of statements about springwood.
(a) It is also called as the earlywood
(b) In spring season cambium produces xylem elements with narrow vessels
(c) It is lighter in colour
(d) The springwood along with autumn wood shows alternate concentric rings forming annual rings
(e) It has lower density
Choose the correct answer from the options given below:
Q30.
mcq single
+4 / 1
what is the role of large bundle sheath cells found around the vascular bundles in C~4~ plants?
Q31.
mcq single
+4 / 1
Read the following statements about the vascular bundles:
(a) In roots, xylem and phloem in a vascular bundle are arranged in an alternate manner along the different radii.
(b) Conjoint closed vascular bundles do not possess cambium
(c) In open vascular bundles, cambium is present in between xylem and phloem
(d) The vascular bundles of dicotyledonous stem possess endarch protoxylem
(e) In monocotyledonous root, usually there are more than six xylem bundles present
Choose the correct answer from the options given below :
Q32.
mcq single
+4 / 1
"Girdling Experiment" was performed by Plant Physiologists to identify the plant tissue through which:
Q33.
mcq single
+4 / 1
In old trees the greater part of secondary xylem is dark brown and resistant to insect attack due to :
(a) secretion of secondary metabolities and their deposition in the lumen of vessels.
(b) deposition of organic compounds like tannins and resins in the central layers of stem.
(c) deposition of suberin and aromatic substances in the outer layer of stem.
(d) deposition of tannins, gum, resin and aromatic substances in the peripheral layers of stem.
(e) presence of parenchyma cells, functionally active xylem elements and essential oils.
Choose the correct answer from the options given below :
Q34.
mcq single
+4 / 1
Which of the following statements with respect to Endoplasmic Reticulum is incorrect?
Q35.
mcq single
+4 / 1
Read the following statements and choose the set of correct statements:
(a) Euchromatin is loosely packed chromatin
(b) Heterochromatin is transcriptionally active
(c) Histone octamer is wrapped by negatively charged DNA in nucleosome
(d) Histones are rich in lysine and arginine
(e) A typical nucleosome contains 400 bp of DNA helix
Choose the correct answer from the options given below :
Q36.
mcq single
+4 / 1
The process of translation of mRNA to proteins begins as soon as:
Q37.
mcq single
+4 / 1
DNA polymorphism forms the basis of:
Q38.
mcq single
+4 / 1
In an E. Coli strain i gene gets mutated and its product can not bind the inducer molecule. If growth medium is provided with lactose, what will be the outcome?
Q39.
mcq single
+4 / 1
If a geneticist uses the blind approach for sequencing the whole genome of an organism, followed by assignment of function to different segments, the methodology adopted by him is called as :
Q40.
mcq single
+4 / 1
Ten E.coli cells with ^(15)N - dsDNA are incubated in medium containing ^(14)N nucleotide. After 60 minutes, how many E.coli cells will have DNA totally free from ^(15)N?
Q41.
mcq single
+4 / 1
Transposons can be used during which one of the following?
Q42.
mcq single
+4 / 1
If the length of a DNA molecule is 1.1 metres, what will b the approximate number of base pairs?
Q43.
mcq single
+4 / 1
Given below are two statements:
Statement I : Autoimmune disorder is a condition where body defense mechanism recognizes its own cells as foreign bodies.
Statement II : Rheumatoid arthritis is a condition where body does not attack self cells.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q44.
mcq single
+4 / 1
Select the incorrect statement with respect to acquired immunity.
Q45.
mcq single
+4 / 1
Which one of the following statements cannot be connected to Predation?
Q46.
mcq single
+4 / 1
While explaining interspecific interaction of population, (+) sign is assigned for beneficial interaction, ($$-$$) sign is assigned for detrimental interaction and (0) for neutral interaction. Which of the following interactions can be assigned (+) for one specifies and ($$-$$) for another specifies involved in the interaction?
Q47.
mcq single
+4 / 1
If '8' Drosophila in a laboratory population of '80' died during a week, the death rate in the population is ___________ individuals per Drosophila per week.
Q48.
mcq single
+4 / 1
Given below are two statements:
Statement I : The coagulum is formed of network of threads called thrombins.
Statement II : Spleen is the graveyard of erythrocytes.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q49.
mcq single
+4 / 1
Which one of the following statements is correct?
Q50.
mcq single
+4 / 1
Which one of the following never occurs during mitotic cell division?
Q51.
mcq single
+4 / 1
Regarding Meiosis, which of the statements is incorrect?
Q52.
mcq single
+4 / 1
Select the incorrect statement with reference to mitosis :
Q53.
mcq single
+4 / 1
The appearance of recombination nodules on homologous chromosomes during meiosis characterizes:
Q54.
mcq single
+4 / 1
Match List I with List II.
List - I
List - II
(a)
Metacentric chromosome
(i)
Centromere situated close to the end forming one extremely short and one very long arms
(b)
Acrocentric chromosome
(ii)
Centromere at the terminal end
(c)
Submetacentric
(iii)
Centromere in the middle forming two equal arms of chromosomes
(d)
Telocentric chromosome
(iv)
Centromere slightly away from the middle forming one shorter arm and one longer arm
Choose the correct answer from the options given below:
Q55.
mcq single
+4 / 1
Given below are two statements :
Statement I : The release of sperms into the seminiferous tubules is called spermiation.
Statement II : Spermiogenesis is the process of formation of sperms from spermatogonia.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q56.
mcq single
+4 / 1
At which stage of life the oogenesis process is initiated?
Q57.
mcq single
+4 / 1
Which of the following statements are true for spermatogenesis but do not hold true for Oogenesis?
(a) It results in the formation of haploid gametes
(b) Differentiation of gamete occurs after the completion of meiosis
(c) Meiosis occurs continuously in a mitotically dividing stem cell population
(d) It is controlled by the Luteinising Hormone (LH) and Follicle Stimulating Hormone (FSH) secreted by the anterior pituitary
(e) It is initiated at puberty
Choose the most appropriate answer from the options given below :
Q58.
mcq single
+4 / 1
Which of the following are not the effects of Parathyroid hormone?
(a) Stimulates the process of bone resorption
(b) Decreases Ca^(2+) level in blood
(c) Reabsorption of Ca^(2+) by renal tubules
(d) Decreases the absorption of Ca^(2+) from digested food
(e) Increases metabolism of carbohydrates
Choose the most appropriate answer from the options given below:
Q59.
mcq single
+4 / 1
Habitat loss and fragmentation, over exploitation, alien species invasion and co-extinction are causes for :
Q60.
mcq single
+4 / 1
In-situ conservation refers to :
Q61.
mcq single
+4 / 1
Which of the following is not a method of ex situ conservation?
Q62.
mcq single
+4 / 1
Production of Cucumber has increased manifold in recent years. Application of which of the following phytohormones has resulted in this increased yield as the hormone is known to produce female flowers in the plants:
Q63.
mcq single
+4 / 1
The gaseous plant growth regulator is used in plants to:
Q64.
mcq single
+4 / 1
Which one of the following plants does not show plasticity?
Q65.
mcq single
+4 / 1
What is the net gain of ATP when each molecule of glucose is converted to two molecules of pyruvic acid?
Q66.
mcq single
+4 / 1
What amount of energy is released from glucose during lactic acid fermentation?
Q67.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Osteoporosis is characterised by decreased bone mass and increased chance of fractures.
Reason (R) : Common cause of osteoporosis is increased levels of estrogen.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q68.
mcq single
+4 / 1
Which of the following is a correct match for disease and its symptoms?
Q69.
mcq single
+4 / 1
Which of the following is a correct statement?
Q70.
mcq single
+4 / 1
Given below are two statements:
Statement I : Mycoplasma can pass through less than 1 micron filter size.
Statement II : Mycoplasma are bacteria with cell wall.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q71.
mcq single
+4 / 1
Natural selection where more individuals acquire specific character value other than the mean character value, leads to
Q72.
mcq single
+4 / 1
Which of the following statements is not true?
Q73.
mcq single
+4 / 1
Nitrogenous waste is excreted in the form of pellet or paste by :
Q74.
mcq single
+4 / 1
Lippe's loop is a type of contraceptive used as:
Q75.
mcq single
+4 / 1
Match List-I with List-II with respect to methods of Contraception and their respective actions.
List - I
List - II
(a)
Diaphragms
(i)
Inhibit ovulation and implantation
(b)
Contraceptive Pills
(ii)
Increase phagocytosis of sperm within Uterus
(c)
Intra Uterine Devices
(iii)
Absence of Menstrual cycle and ovulation following parturition
(d)
Lactational Amenorrhea
(iv)
They cover the cervix blocking the entry of sperms
Choose the correct answer from the options given below :
Q76.
mcq single
+4 / 1
Given below are two statements:
Statement I : Cleistogamous flowers are invariably autogamous
Statement II : Cleistogamy is disadvantageous as there is no chance for cross pollination
In the light of the above statements, choose the correct answer from the options given below :
Q77.
mcq single
+4 / 1
Identify the incorrect statement related to Pollination:
Q78.
mcq single
+4 / 1
The flowers are Zygomorphic in :
(a) Mustard (b) Gulmohar (c) Cassia (d) Datura (e) Chilly
Choose the correct answer from the options given below :
Q79.
mcq single
+4 / 1
Identify the correct set of statements:
(a) The leaflets are modified into pointed hard thorns in Citrus and Bougainvillea
(b) Axillary buds form slender and spirally coiled tendrils in cucumber and pumpkin
(c) Stem is flattened and fleshy in Opuntia and modified to perform the function of leaves
(d) Rhizophora shows vertically upward growing roots that help to get oxygen for respiration
(e) Subaerially growing stems in grasses and strawberry help in vegetative propagation
Choose the correct answer from the options given below:
Q80.
mcq single
+4 / 1
Which one of the following plants shows vexillary aestivation and diadelphous stamens?
Q81.
mcq single
+4 / 1
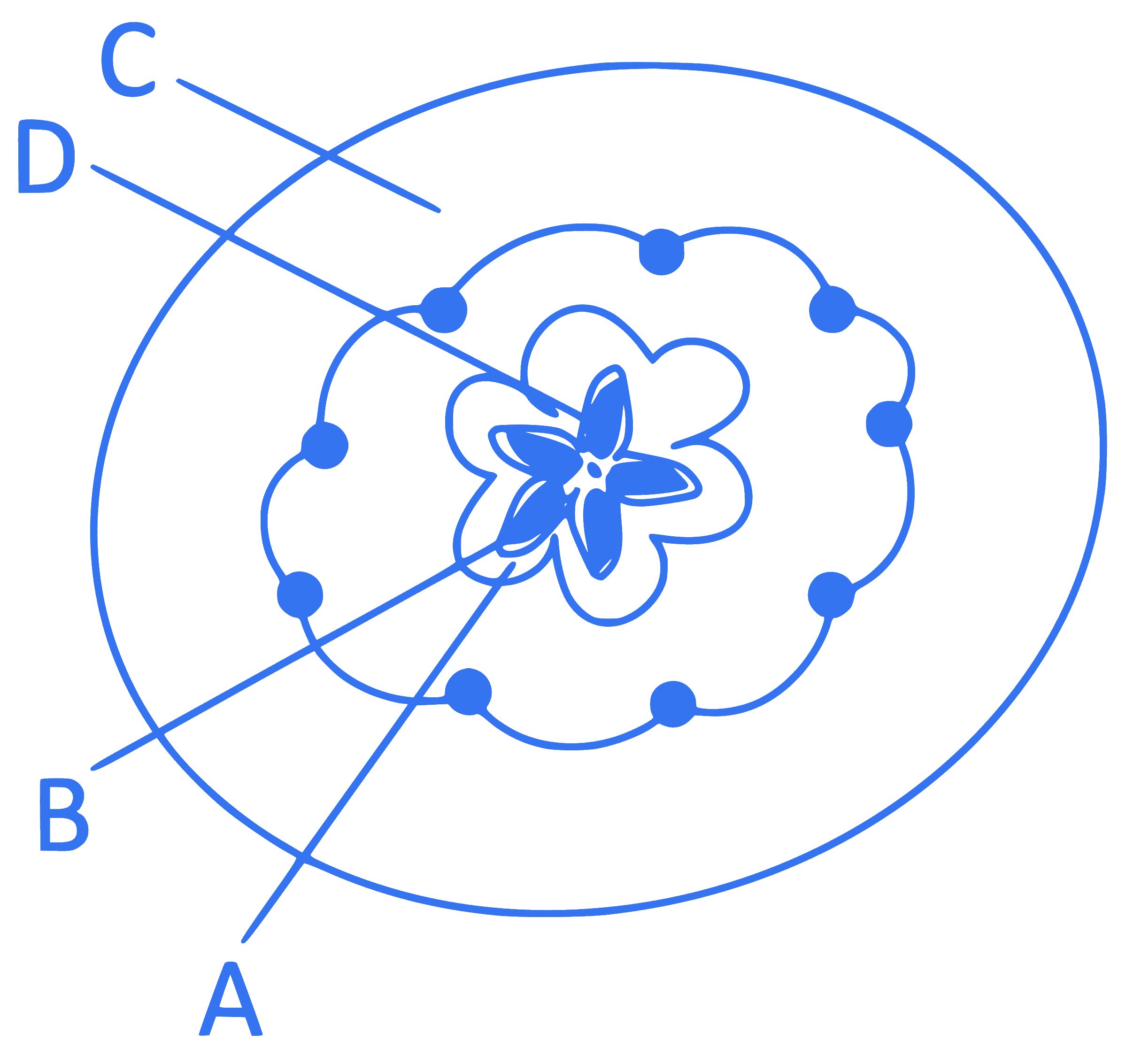
Which part of the fruit, labelled in the given figure makes it a false fruit?

Q82.
mcq single
+4 / 1
Match List-I with List-II:
List - I
(Biological Molecules)
List - II
(Biological functions)
(a)
Glycogen
(i)
Hormone
(b)
Globulin
(ii)
Biocatalyst
(c)
Steroids
(iii)
Antibody
(d)
Thrombin
(iv)
Storage product
Choose the correct answer from the options given below:
Q83.
mcq single
+4 / 1
A dehydration reaction links two glucose molecules to product maltose. If the formula for glucose is C~6~H~12~O~6~ then what is the formula for maltose?
Q84.
mcq single
+4 / 1
Read the following statements on lipids and find out correct set of statements :
(a) Lecithin found in the plasma membrane is a glycolipid
(b) Saturated fatty acids possess one or more c = c bonds
(c) Gingely oil has lower melting point, hence remains as oil in winter
(d) Lipids are generally insoluble in water but soluble in some organic solvents
(e) When fatty acid is esterified with glycerol, monoglycerides are formed
Choose the correct answer from the option given below:
Q85.
mcq single
+4 / 1
Given below are two statements:
Statement I : Restriction endonucleases recognise specific sequence to cut DNA known as palindromic nucleotide sequence.
Statement II : Restriction endonucleases cut the DNA strand a little away from the centre of the palindromic site.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q86.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Polymerase chain reaction is used in DNA amplification.
Reason (R) : The ampicillin resistant gene is used as a selectable marker to check transformation.
In the light of the above statements, choose the correct answer from the options given below :
Q87.
mcq single
+4 / 1
Which of the following is not a desirable feature of a cloning vector?
Q88.
mcq single
+4 / 1
Which one of the following statement is not true regarding gel electrophoresis technique?
Q89.
mcq single
+4 / 1
Identify the microorganism which is responsible for the production of an immunosuppressive molecule cyclosporin A :
Q90.
mcq single
+4 / 1
Which one of the following will accelerate phosphorus cycle?
Q91.
mcq single
+4 / 1
Detritivores breakdown detritus into smaller particles. This process is called :
Q92.
mcq single
+4 / 1
Given below are two statements:
Statement I : Decomposition is a process in which the detritus is degraded into simpler substances by microbes.
Statement II : Decomposition is faster if the detritus is rich in lignin and chitin.
In the light of the above statements, choose the correct answer from the options given below :
Q93.
mcq single
+4 / 1
Given below are two statements:
Statement I : Mendel studied seven pairs of contrasting traits in pea plants and proposed the Laws of Inheritance.
Statement II : Seven characters examined by Mendel in his experiment on pea plants were seed shape and colour, flower colour, pod shape and colour, flower position and stem height.
In the light of the above statements, choose the correct answer from the options given below :
Q94.
mcq single
+4 / 1
If a colour blind female marries a man whose mother was also colour blind, what are the chances of her progeny having colour blindness?
Q95.
mcq single
+4 / 1
XO type of sex determination can be found in :
Q96.
mcq single
+4 / 1
Which of the following occurs due to the presence of autosome linked dominant trait?
Q97.
mcq single
+4 / 1
The recombination frequency between the genes a & c 5&, b & c is 15%, b & d is 9%, a & b is 20%, c & d is 25% and a & d is 29%. What will be the sequence of these genes on a linear chromosome?
Q98.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Mendel's law of independent assortment does not hold good for the genes that are located closely on the same chromosome.
Reason (R) : Closely located genes assort independently.
In the light of the above statements, choose the correct answer from the options given below :
Q99.
mcq single
+4 / 1
Which of the following is not a connective tissue?
Q100.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(a)
Bronchioles
(i)
Dense Regular Connective Tissue
(b)
Goblet Cell
(ii)
Loose Connective Tissue
(c)
Tendons
(iii)
Glandular Tissue
(d)
Adipose Tissue
(iv)
Ciliated Epithelium
Choose the correct answer from the options given below:
Q101.
mcq single
+4 / 1
Tegmina in cockroach, arises from
Q102.
mcq single
+4 / 1
In which of the following animals, digestive tract has additional chambers like crop and gizzard?
Q103.
mcq single
+4 / 1
Which one of the following is not true regarding the release of energy during ATP synthesis through chemiosmosis? It involves:
Q104.
mcq single
+4 / 1
Given below are two statements:
Statement I : The primary CO~2~ acceptor in C~4~ plants is phosphoenolpyruvate and is found in the mesophyll cells.
Statement II : Mesophyll cells of C~4~ plants lack RuBisCo enzyme.
In the light of the above statements, choose the correct answer from the options given below:
Q105.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : All vertebrates are chordates but all chordates are not vertebrates.
Reason (R) : Notochord is replaced by vertebral column in the adult vertebrates.
In the light of the above statements, choose the most appropriate answer from the option given below:
Q106.
mcq single
+4 / 1
Exoskeleton of arthropods is composed of:
Q107.
mcq single
+4 / 1
Hydrocolloid carrageen is obtained form :
Q108.
mcq single
+4 / 1
Which of the following is incorrectly matched?
Q109.
mcq single
+4 / 1
Match the plant with the kind of life cycle it exhibits:
List - I
List - II
(a)
Spirogyra
(i)
Dominant diploid sporophyte vascular plant, with highly reduced male or female gametophyte
(b)
Fern
(ii)
Dominant haploid free-living gametophyte
(c)
Funaria
(iii)
Dominant diploid sporophyte alternating with reduced gametophyte called prothallus
(d)
Cycas
(iv)
Dominant haploid leafy gametophyte alternating with partially dependent multicellular sporophyte
Choose the correct answer from the options given below :
Q110.
mcq single
+4 / 1
Which of the following is not the function of conducting part of respiratory system?
Q111.
mcq single
+4 / 1
Under normal physiological conditions in human being every 100 ml of oxygenated blood can deliver ____________ ml of O~2~ to the tissues.
Q112.
mcq single
+4 / 1
In the taxonomic categories which hierarchical arrangement in ascending order is correct in case of animals?
Q113.
mcq single
+4 / 1
In gene therapy of Adenosine Deaminase (ADA) deficiency, the patient requires periodic infusion of genetically engineered lymphocytes because:
Q114.
mcq single
+4 / 1
In the following palindromic base sequences of DNA, which one can be cut easily by particular restriction enzyme?
Q115.
mcq single
+4 / 1
Statements related to human Insulin are given below.
Which statements is/are correct about genetically engineered Insulin?
(a) Pro-hormone insulin contain extra stretch of C-peptide
(b) A-peptide and B-peptide chains of insulin were produced separately in E.coli, extracted and combined by creating disulphide bond between them.
(c) Insulin used for treating Diabetes was extracted from Cattles and Pigs.
(d) Pro-hormone Insulin needs to be processed for converting into a mature and functional hormone.
(e) Some patients develop allergic reactions to the foreign insulin.
Choose the most appropriate answer from the options given below :
Q116.
mcq single
+4 / 1
Select the incorrect statement regarding synapses :
Q117.
mcq single
+4 / 1
The anatomy of springwood shows some peculiar features. Identify the correct set of statements about springwood.
(a) It is also called as the earlywood
(b) In spring season cambium produces xylem elements with narrow vessels
(c) It is lighter in colour
(d) The springwood along with autumn wood shows alternate concentric rings forming annual rings
(e) It has lower density
Choose the correct answer from the options given below:
Q118.
mcq single
+4 / 1
what is the role of large bundle sheath cells found around the vascular bundles in C~4~ plants?
Q119.
mcq single
+4 / 1
Read the following statements about the vascular bundles:
(a) In roots, xylem and phloem in a vascular bundle are arranged in an alternate manner along the different radii.
(b) Conjoint closed vascular bundles do not possess cambium
(c) In open vascular bundles, cambium is present in between xylem and phloem
(d) The vascular bundles of dicotyledonous stem possess endarch protoxylem
(e) In monocotyledonous root, usually there are more than six xylem bundles present
Choose the correct answer from the options given below :
Q120.
mcq single
+4 / 1
"Girdling Experiment" was performed by Plant Physiologists to identify the plant tissue through which:
Q121.
mcq single
+4 / 1
In old trees the greater part of secondary xylem is dark brown and resistant to insect attack due to :
(a) secretion of secondary metabolities and their deposition in the lumen of vessels.
(b) deposition of organic compounds like tannins and resins in the central layers of stem.
(c) deposition of suberin and aromatic substances in the outer layer of stem.
(d) deposition of tannins, gum, resin and aromatic substances in the peripheral layers of stem.
(e) presence of parenchyma cells, functionally active xylem elements and essential oils.
Choose the correct answer from the options given below :
Q122.
mcq single
+4 / 1
Which of the following statements with respect to Endoplasmic Reticulum is incorrect?
Q123.
mcq single
+4 / 1
Read the following statements and choose the set of correct statements:
(a) Euchromatin is loosely packed chromatin
(b) Heterochromatin is transcriptionally active
(c) Histone octamer is wrapped by negatively charged DNA in nucleosome
(d) Histones are rich in lysine and arginine
(e) A typical nucleosome contains 400 bp of DNA helix
Choose the correct answer from the options given below :
Q124.
mcq single
+4 / 1
The process of translation of mRNA to proteins begins as soon as:
Q125.
mcq single
+4 / 1
DNA polymorphism forms the basis of:
Q126.
mcq single
+4 / 1
In an E. Coli strain i gene gets mutated and its product can not bind the inducer molecule. If growth medium is provided with lactose, what will be the outcome?
Q127.
mcq single
+4 / 1
If a geneticist uses the blind approach for sequencing the whole genome of an organism, followed by assignment of function to different segments, the methodology adopted by him is called as :
Q128.
mcq single
+4 / 1
Ten E.coli cells with ^(15)N - dsDNA are incubated in medium containing ^(14)N nucleotide. After 60 minutes, how many E.coli cells will have DNA totally free from ^(15)N?
Q129.
mcq single
+4 / 1
Transposons can be used during which one of the following?
Q130.
mcq single
+4 / 1
If the length of a DNA molecule is 1.1 metres, what will b the approximate number of base pairs?
Q131.
mcq single
+4 / 1
Given below are two statements:
Statement I : Autoimmune disorder is a condition where body defense mechanism recognizes its own cells as foreign bodies.
Statement II : Rheumatoid arthritis is a condition where body does not attack self cells.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q132.
mcq single
+4 / 1
Select the incorrect statement with respect to acquired immunity.
Q133.
mcq single
+4 / 1
Which one of the following statements cannot be connected to Predation?
Q134.
mcq single
+4 / 1
While explaining interspecific interaction of population, (+) sign is assigned for beneficial interaction, ($$-$$) sign is assigned for detrimental interaction and (0) for neutral interaction. Which of the following interactions can be assigned (+) for one specifies and ($$-$$) for another specifies involved in the interaction?
Q135.
mcq single
+4 / 1
If '8' Drosophila in a laboratory population of '80' died during a week, the death rate in the population is ___________ individuals per Drosophila per week.
Q136.
mcq single
+4 / 1
Given below are two statements:
Statement I : The coagulum is formed of network of threads called thrombins.
Statement II : Spleen is the graveyard of erythrocytes.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q137.
mcq single
+4 / 1
Which one of the following statements is correct?
Q138.
mcq single
+4 / 1
Which one of the following never occurs during mitotic cell division?
Q139.
mcq single
+4 / 1
Regarding Meiosis, which of the statements is incorrect?
Q140.
mcq single
+4 / 1
Select the incorrect statement with reference to mitosis :
Q141.
mcq single
+4 / 1
The appearance of recombination nodules on homologous chromosomes during meiosis characterizes:
Q142.
mcq single
+4 / 1
Match List I with List II.
List - I
List - II
(a)
Metacentric chromosome
(i)
Centromere situated close to the end forming one extremely short and one very long arms
(b)
Acrocentric chromosome
(ii)
Centromere at the terminal end
(c)
Submetacentric
(iii)
Centromere in the middle forming two equal arms of chromosomes
(d)
Telocentric chromosome
(iv)
Centromere slightly away from the middle forming one shorter arm and one longer arm
Choose the correct answer from the options given below:
Q143.
mcq single
+4 / 1
Given below are two statements :
Statement I : The release of sperms into the seminiferous tubules is called spermiation.
Statement II : Spermiogenesis is the process of formation of sperms from spermatogonia.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q144.
mcq single
+4 / 1
At which stage of life the oogenesis process is initiated?
Q145.
mcq single
+4 / 1
Which of the following statements are true for spermatogenesis but do not hold true for Oogenesis?
(a) It results in the formation of haploid gametes
(b) Differentiation of gamete occurs after the completion of meiosis
(c) Meiosis occurs continuously in a mitotically dividing stem cell population
(d) It is controlled by the Luteinising Hormone (LH) and Follicle Stimulating Hormone (FSH) secreted by the anterior pituitary
(e) It is initiated at puberty
Choose the most appropriate answer from the options given below :
Q146.
mcq single
+4 / 1
Which of the following are not the effects of Parathyroid hormone?
(a) Stimulates the process of bone resorption
(b) Decreases Ca^(2+) level in blood
(c) Reabsorption of Ca^(2+) by renal tubules
(d) Decreases the absorption of Ca^(2+) from digested food
(e) Increases metabolism of carbohydrates
Choose the most appropriate answer from the options given below:
Q147.
mcq single
+4 / 1
Habitat loss and fragmentation, over exploitation, alien species invasion and co-extinction are causes for :
Q148.
mcq single
+4 / 1
In-situ conservation refers to :
Q149.
mcq single
+4 / 1
Which of the following is not a method of ex situ conservation?
Q150.
mcq single
+4 / 1
Production of Cucumber has increased manifold in recent years. Application of which of the following phytohormones has resulted in this increased yield as the hormone is known to produce female flowers in the plants:
Q151.
mcq single
+4 / 1
The gaseous plant growth regulator is used in plants to:
Q152.
mcq single
+4 / 1
Which one of the following plants does not show plasticity?
Q153.
mcq single
+4 / 1
What is the net gain of ATP when each molecule of glucose is converted to two molecules of pyruvic acid?
Q154.
mcq single
+4 / 1
What amount of energy is released from glucose during lactic acid fermentation?
Q155.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Osteoporosis is characterised by decreased bone mass and increased chance of fractures.
Reason (R) : Common cause of osteoporosis is increased levels of estrogen.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q156.
mcq single
+4 / 1
Which of the following is a correct match for disease and its symptoms?
Q157.
mcq single
+4 / 1
Which of the following is a correct statement?
Q158.
mcq single
+4 / 1
Given below are two statements:
Statement I : Mycoplasma can pass through less than 1 micron filter size.
Statement II : Mycoplasma are bacteria with cell wall.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q159.
mcq single
+4 / 1
Natural selection where more individuals acquire specific character value other than the mean character value, leads to
Q160.
mcq single
+4 / 1
Which of the following statements is not true?
Q161.
mcq single
+4 / 1
Nitrogenous waste is excreted in the form of pellet or paste by :
Q162.
mcq single
+4 / 1
Lippe's loop is a type of contraceptive used as:
Q163.
mcq single
+4 / 1
Match List-I with List-II with respect to methods of Contraception and their respective actions.
List - I
List - II
(a)
Diaphragms
(i)
Inhibit ovulation and implantation
(b)
Contraceptive Pills
(ii)
Increase phagocytosis of sperm within Uterus
(c)
Intra Uterine Devices
(iii)
Absence of Menstrual cycle and ovulation following parturition
(d)
Lactational Amenorrhea
(iv)
They cover the cervix blocking the entry of sperms
Choose the correct answer from the options given below :
Q164.
mcq single
+4 / 1
Given below are two statements:
Statement I : Cleistogamous flowers are invariably autogamous
Statement II : Cleistogamy is disadvantageous as there is no chance for cross pollination
In the light of the above statements, choose the correct answer from the options given below :
Q165.
mcq single
+4 / 1
Identify the incorrect statement related to Pollination:
Q166.
mcq single
+4 / 1
The flowers are Zygomorphic in :
(a) Mustard (b) Gulmohar (c) Cassia (d) Datura (e) Chilly
Choose the correct answer from the options given below :
Q167.
mcq single
+4 / 1
Identify the correct set of statements:
(a) The leaflets are modified into pointed hard thorns in Citrus and Bougainvillea
(b) Axillary buds form slender and spirally coiled tendrils in cucumber and pumpkin
(c) Stem is flattened and fleshy in Opuntia and modified to perform the function of leaves
(d) Rhizophora shows vertically upward growing roots that help to get oxygen for respiration
(e) Subaerially growing stems in grasses and strawberry help in vegetative propagation
Choose the correct answer from the options given below:
Q168.
mcq single
+4 / 1
Which one of the following plants shows vexillary aestivation and diadelphous stamens?
Q169.
mcq single
+4 / 1
Which part of the fruit, labelled in the given figure makes it a false fruit?


Q170.
mcq single
+4 / 1
Match List-I with List-II:
List - I
(Biological Molecules)
List - II
(Biological functions)
(a)
Glycogen
(i)
Hormone
(b)
Globulin
(ii)
Biocatalyst
(c)
Steroids
(iii)
Antibody
(d)
Thrombin
(iv)
Storage product
Choose the correct answer from the options given below:
Q171.
mcq single
+4 / 1
A dehydration reaction links two glucose molecules to product maltose. If the formula for glucose is C~6~H~12~O~6~ then what is the formula for maltose?
Q172.
mcq single
+4 / 1
Read the following statements on lipids and find out correct set of statements :
(a) Lecithin found in the plasma membrane is a glycolipid
(b) Saturated fatty acids possess one or more c = c bonds
(c) Gingely oil has lower melting point, hence remains as oil in winter
(d) Lipids are generally insoluble in water but soluble in some organic solvents
(e) When fatty acid is esterified with glycerol, monoglycerides are formed
Choose the correct answer from the option given below:
Q173.
mcq single
+4 / 1
Given below are two statements:
Statement I : Restriction endonucleases recognise specific sequence to cut DNA known as palindromic nucleotide sequence.
Statement II : Restriction endonucleases cut the DNA strand a little away from the centre of the palindromic site.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q174.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Polymerase chain reaction is used in DNA amplification.
Reason (R) : The ampicillin resistant gene is used as a selectable marker to check transformation.
In the light of the above statements, choose the correct answer from the options given below :
Q175.
mcq single
+4 / 1
Which of the following is not a desirable feature of a cloning vector?
Q176.
mcq single
+4 / 1
Which one of the following statement is not true regarding gel electrophoresis technique?