NEET 2023 Manipur
MEDICAL 2023 Previous Year
3 hDuration
696Total Marks
348Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 348 across 3 section(s); maximum marks: 696.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Ionisation enthalpy increases along each series of the transition elements from left to right. However, small variations occur.
Reason (R) : There is corresponding increase in nuclear charge which accompanies the filling of electrons in the inner d-orbitals.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q2.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Ionisation enthalpies of early actinoids are lower than for early lanthanoids.
Reason (R) : Electrons are entering 5f orbitals in actinoids which experience greater shielding from nuclear charge.
In the light of the above statements, choose the correct answer from the options given below :
Q3.
mcq single
+4 / 1
Which one of the following represents all isoelectronic species ?
Q4.
mcq single
+4 / 1
Given below are two statements:
Statement I : Hydrated chlorides and bromides of $$\mathrm{Ca}, \mathrm{Sr}$$ and $$\mathrm{Ba}$$ on heating undergo hydrolysis.
Statement II : Hydrated chlorides and bromides of $$\mathrm{Be}$$ and $$\mathrm{Mg}$$ on heating undergo dehydration.
In the light of the above statements, choose the correct answer from the options given below :
Q5.
mcq single
+4 / 1
Which one of the following statements is incorrect related to Molecular Orbital Theory?
Q6.
mcq single
+4 / 1
The correct order of dipole moments for molecules $$\mathrm{NH}_3, \mathrm{H}_2 \mathrm{~S}, \mathrm{CH}_4$$ and $$\mathrm{HF}$$, is:
Q7.
mcq single
+4 / 1
An acidic buffer is prepared by mixing :
Q8.
mcq single
+4 / 1
The correct options for the rate law that corresponds to overall first order reaction is
Q9.
mcq single
+4 / 1
For a reaction $$3 \mathrm{~A} \rightarrow 2 \mathrm{~B}$$
The average rate of appearance of $$\mathrm{B}$$ is given by $$\frac{\Delta[B]}{\Delta t}$$. The correct relation between the average rate of appearance of $$\mathrm{B}$$ with the average rate of disappearance of A is given in option :
Q10.
mcq single
+4 / 1
The correct option for a redox couple is:
Q11.
mcq single
+4 / 1
Identify 'X' in the following reaction.
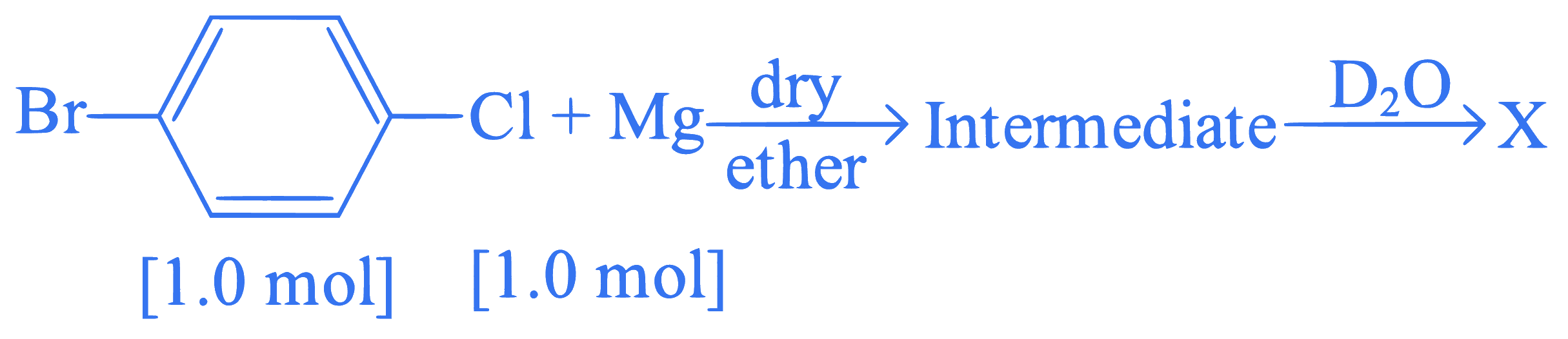
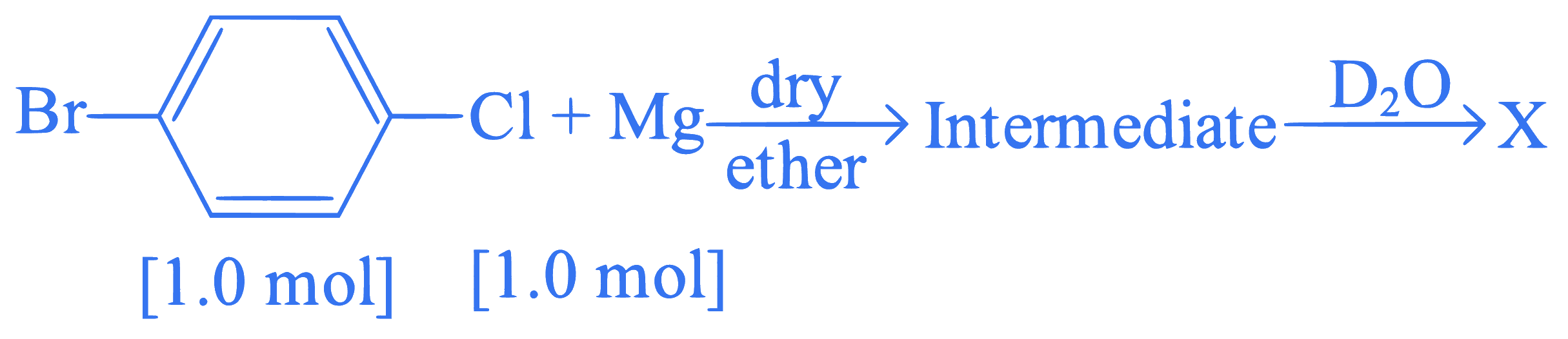
Q12.
mcq single
+4 / 1
Identify the product in the following reaction
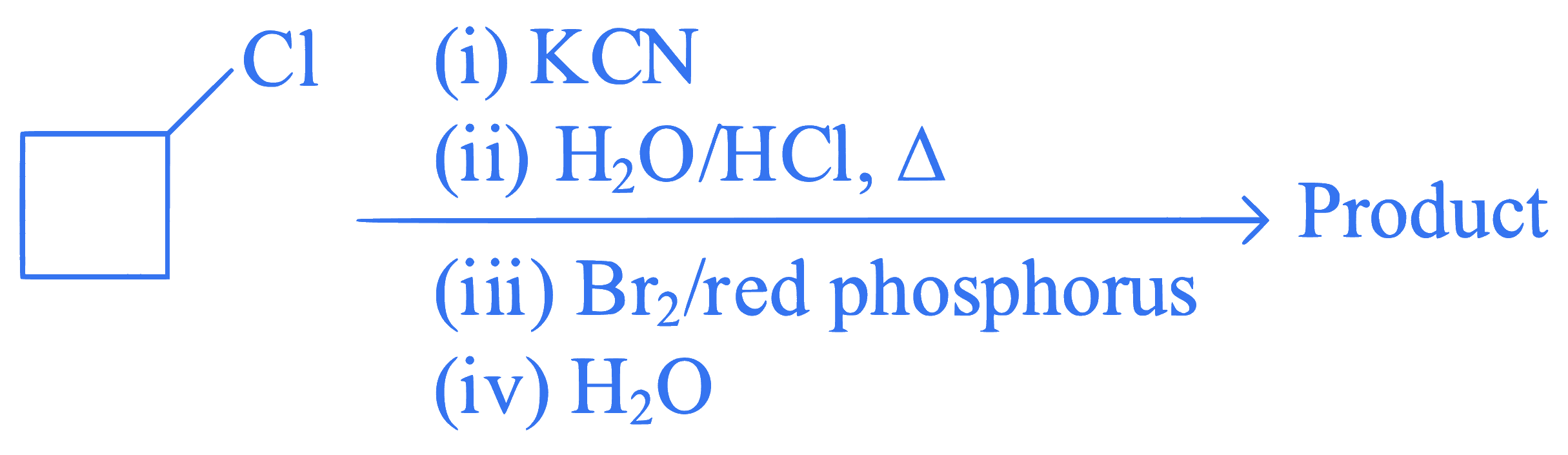
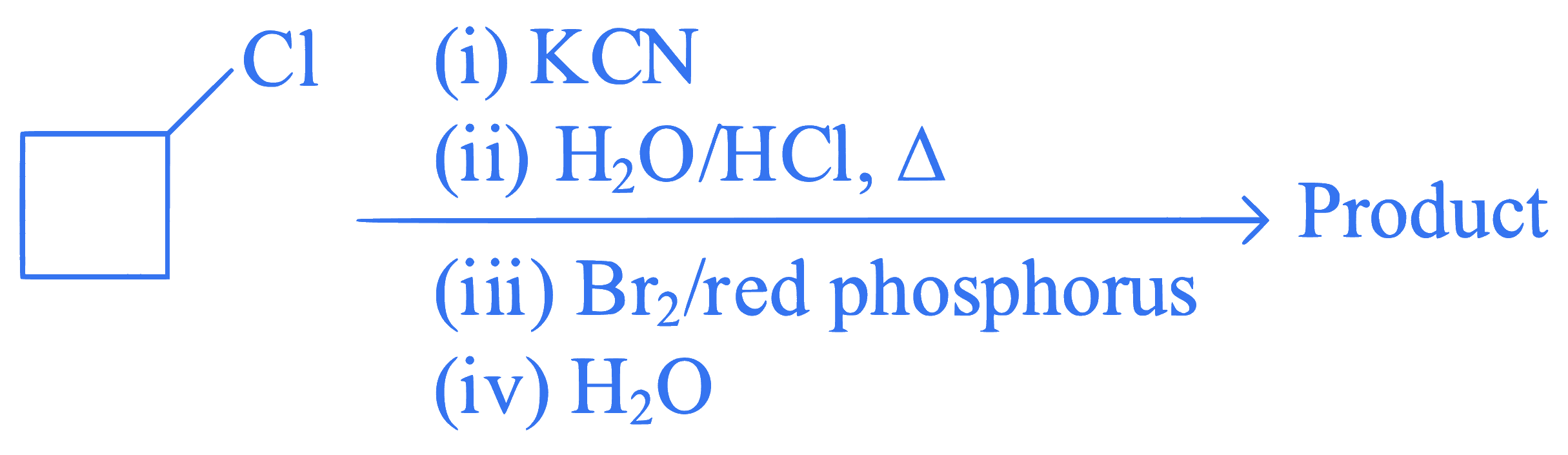
Q13.
mcq single
+4 / 1
Which amongst the following reactions of alkyl halides produces isonitrile as a major product?
(A) $$\mathrm{R-X+HCN\to}$$
(B) $$\mathrm{R-X+AgCN\to}$$
(C) $$\mathrm{R-X+KCN\to}$$
(D) $$\mathrm{R-X+NaCN\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{{C_2}{H_5}OH}^{{H_2}O}}}$$
Choose the most appropriate answer from the options given below:
Q14.
mcq single
+4 / 1
Choose the correct sequence of reagents in the conversion of 4-nitrotoluene to 2-bromotoluene.
Q15.
mcq single
+4 / 1
Which amongst the following aqueous solution of electrolytes will have minimum elevation in boiling point? Choose the correct option :-
Q16.
mcq single
+4 / 1
The density of 1 M solution of a compound 'X' is 1.25 g mL$$^{-1}$$. The correct option for the molality of solution is (Molar mass of compound X = 85 g):
Q17.
mcq single
+4 / 1
For a weak acid HA, the percentage of dissociation is nearly 1% at equilibrium. If the concentration of acid is 0.1 mol L$$^{-1}$$, then the correct option for its K$$_a$$ at the same temperature is :
Q18.
mcq single
+4 / 1
Which of the following is correctly matched?
Q19.
mcq single
+4 / 1
The correct sequence given below containing neutral, acidic, basic and amphoteric oxide each, respectively, is
Q20.
mcq single
+4 / 1
Consider the following reaction :-
$$2 \mathrm{H}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(\mathrm{g}) \Delta_{\mathrm{r}} \mathrm{H}^{\circ}=-483.64 \mathrm{~kJ} \text {. }$$
What is the enthalpy change for decomposition of one mole of water? (Choose the right option).
Q21.
mcq single
+4 / 1
Match the List - I with List - II
List - I
(Hydride)
List - II
(Type of Hydride)
(A)
$$\mathrm{NaH}$$
(I)
Electron precise
(B)
$$\mathrm{PH_3}$$
(II)
Saline
(C)
$$\mathrm{GeH_4}$$
(III)
Metallic
(D)
$$\mathrm{LaH_{2.87}}$$
(IV)
Electron rich
Choose the correct answer from the options given below :
Q22.
mcq single
+4 / 1
Select the element (M) whose trihalides cannot be hydrolysed to produce an ion of the form [M(H$$_2$$O$$_6$$]$$^{3+}$$
Q23.
mcq single
+4 / 1
Which of the following forms a set of complex and a double salt, respectively?
Q24.
mcq single
+4 / 1
Type of isomerism exhibited by compounds
$$
\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_6 \mathrm{Cl}_3,\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_5 \mathrm{Cl}_2 \mathrm{Cl}_2 \cdot \mathrm{H}_2 \mathrm{O}\right.\right.,\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_4 \mathrm{Cl}_2\right] \mathrm{Cl} .2 \mathrm{H}_2 \mathrm{O}$$ and the value of coordination number $$(\mathrm{CN})$$ of central metal ion in all these compounds, respectively is :
Q25.
mcq single
+4 / 1
Which amongst the following compounds/species is least basic?
Q26.
mcq single
+4 / 1
Which amongst the following compounds will show geometrical isomerism?
Q27.
mcq single
+4 / 1
Given below are two statements:
Statement I : In an organic compound, when inductive and electromeric effects operate in opposite directions, the inductive effect predominates.
Statement II : Hyperconjugation is observed in o-xylene.
In the light of the above statements, choose the correct answer from the options given below :
Q28.
mcq single
+4 / 1
The correct order for the rate of $$\alpha,\beta$$-dehydrohalogenation for the following compounds is _________.
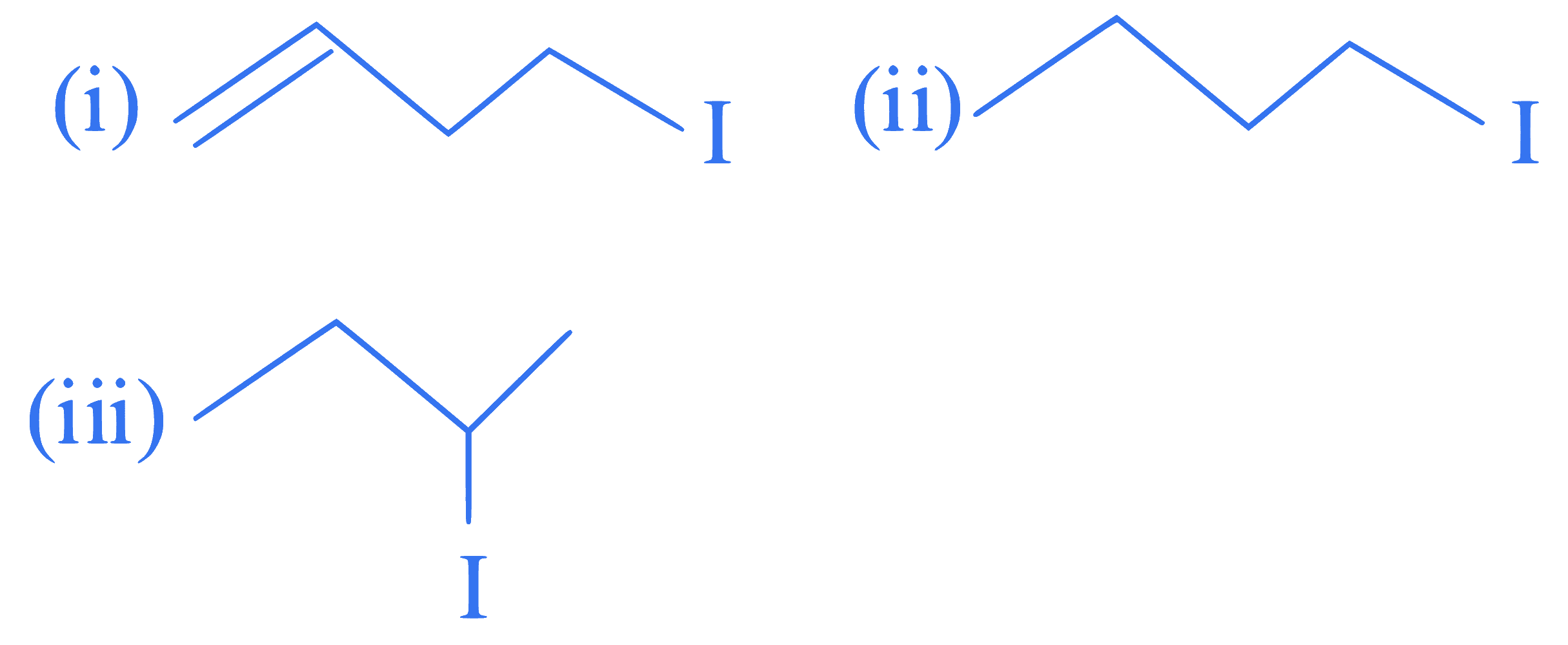
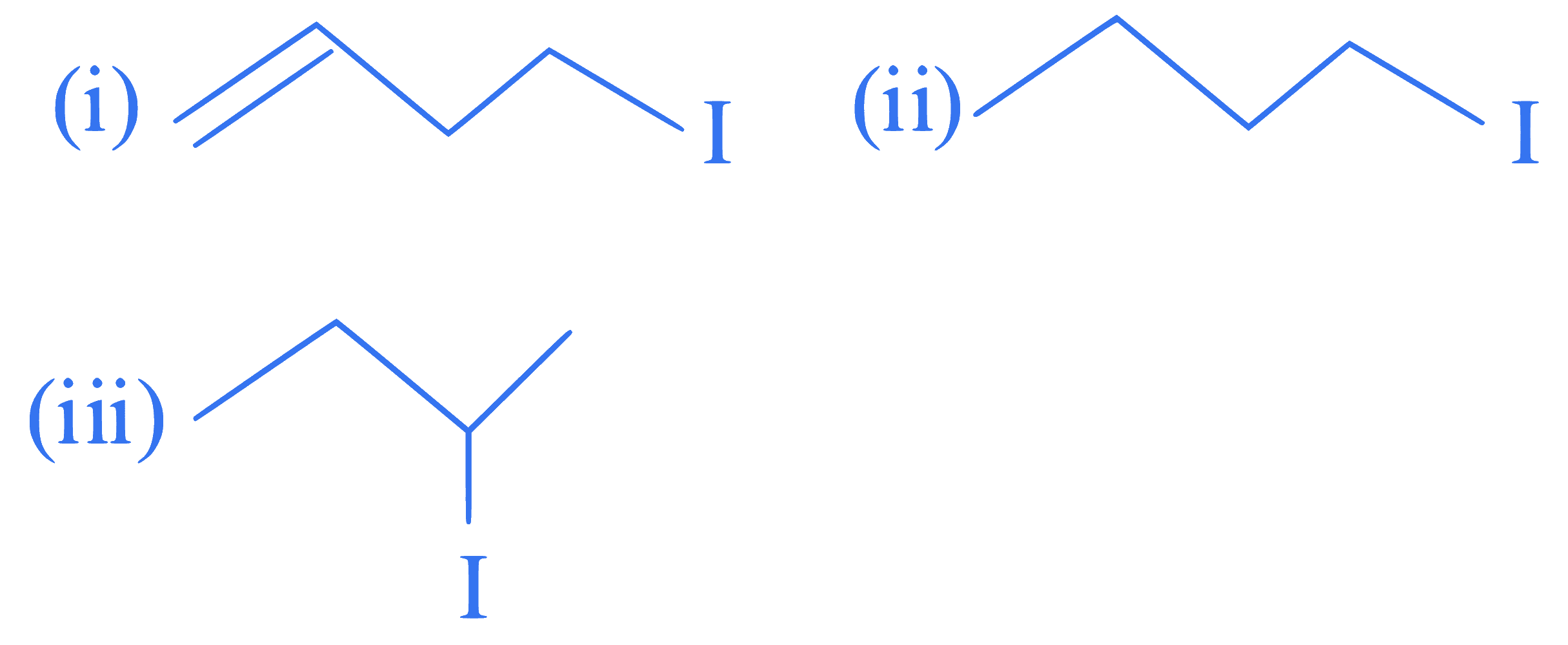
Q29.
mcq single
+4 / 1
Reagents which can be used to convert alcohols to carboxylic acids, are
(A) $$\mathrm{CrO}_3-\mathrm{H}_2 \mathrm{SO}_4$$
(B) $$\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7+\mathrm{H}_2 \mathrm{SO}_4$$
(C) $$\mathrm{KMnO}_4+\mathrm{KOH} / \mathrm{H}_3 \mathrm{O}^{+}$$
(D) $$\mathrm{Cu}, 573 \mathrm{~K}$$
(E) $$\mathrm{CrO}_3,\left(\mathrm{CH}_3 \mathrm{CO}\right)_2 \mathrm{O}$$
Choose the most appropriate answer from the options given below :
Q30.
mcq single
+4 / 1
The major product formed in the following conversion is ___________.
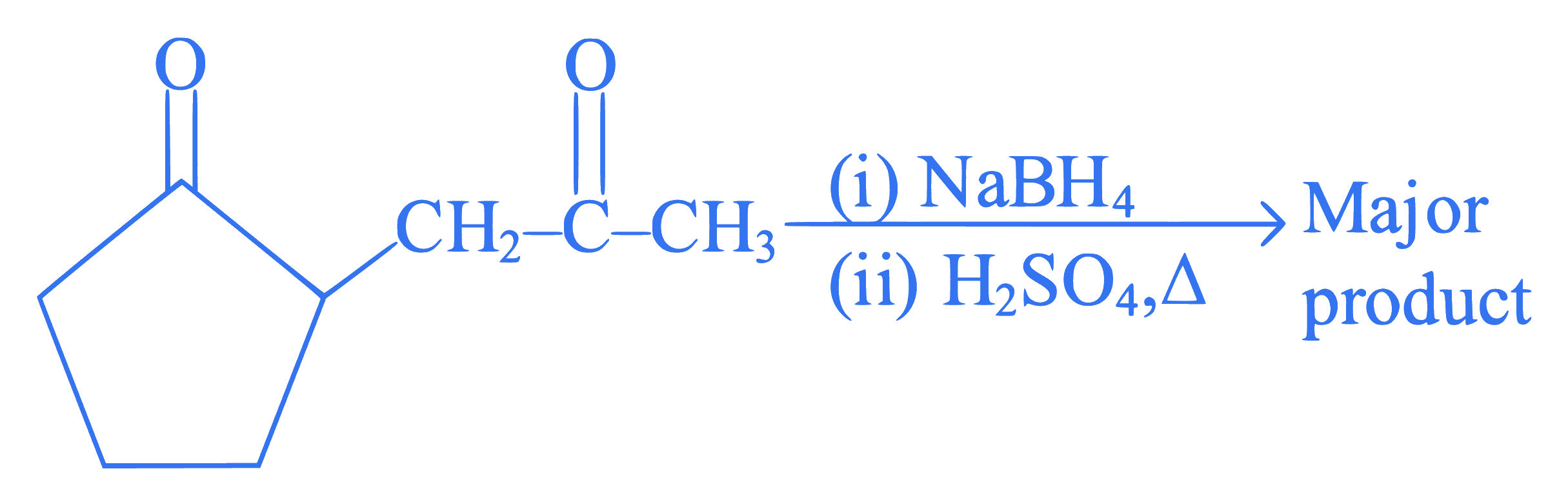
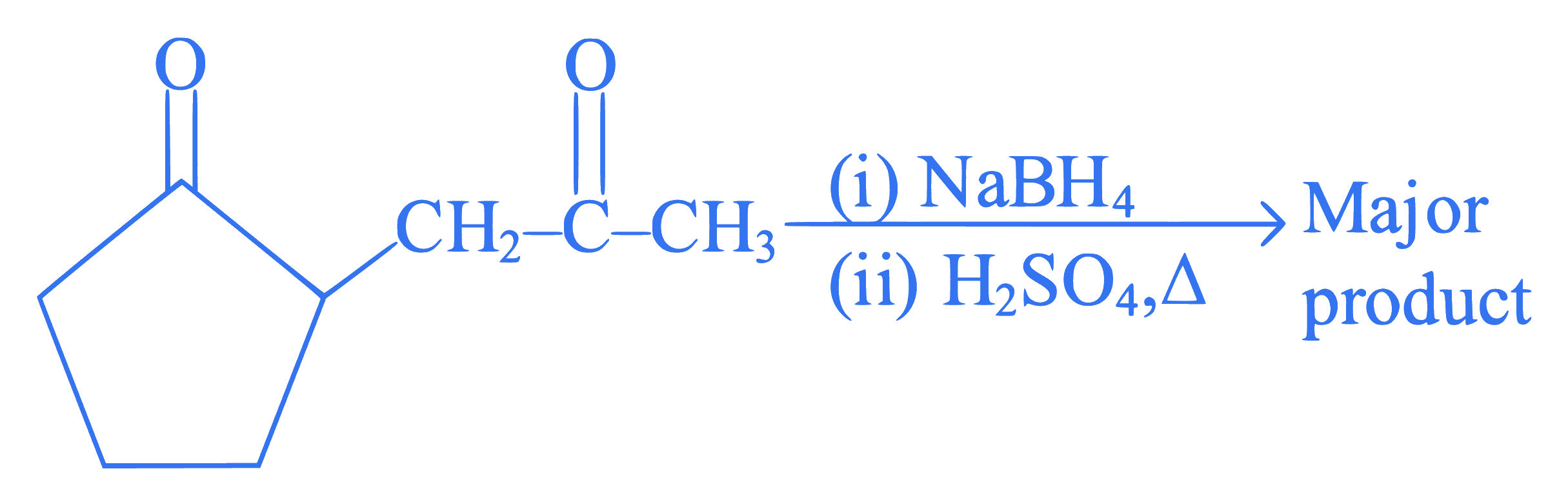
Q31.
mcq single
+4 / 1
The following conversion is known as:
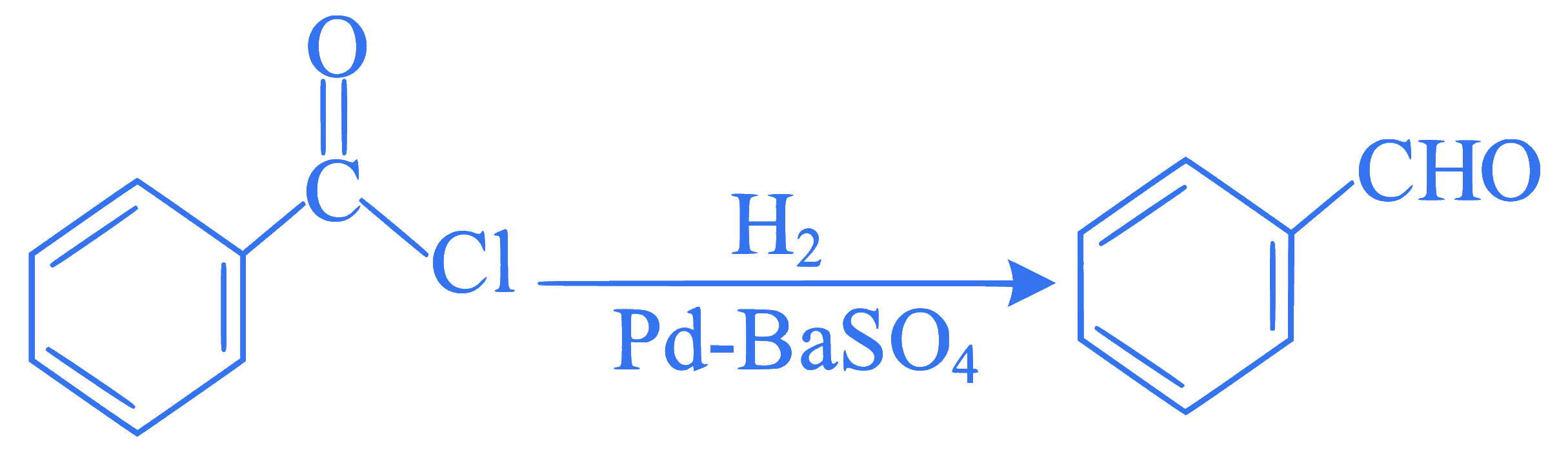
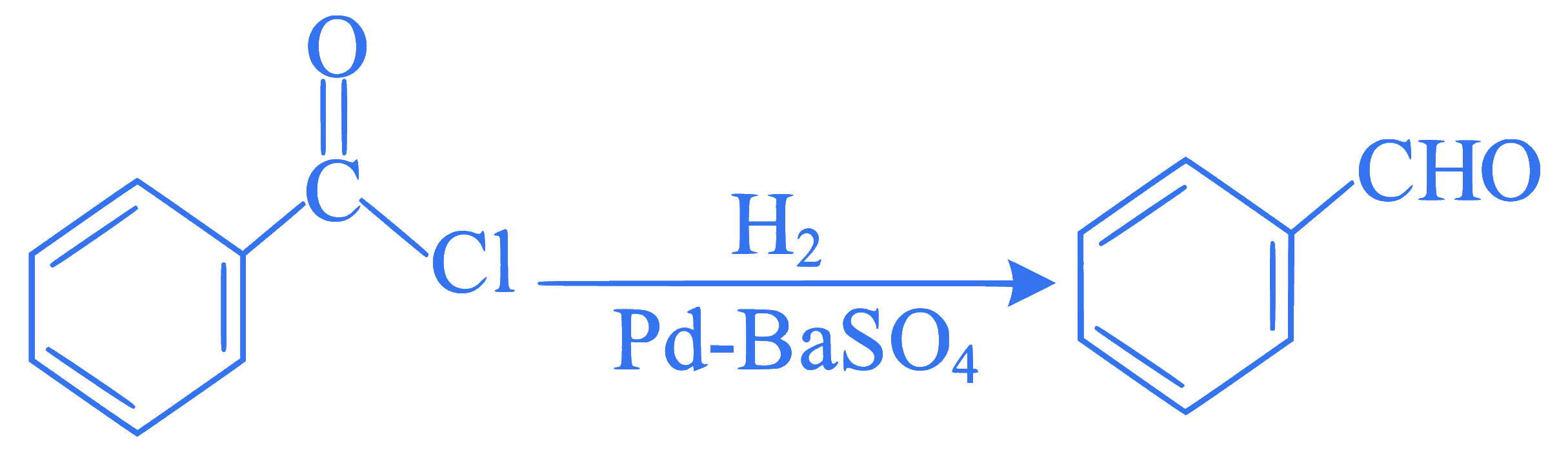
Q32.
mcq single
+4 / 1
Consider the given reaction:
$$\mathrm{CH}_3 \mathrm{COCH}_3 \stackrel{\text { dil } \mathrm{Ba}(\mathrm{OH})_2}{\longrightarrow} \text { ''X" }$$
The functional groups present in compound "$$\mathrm{X}$$" are:
Q33.
mcq single
+4 / 1
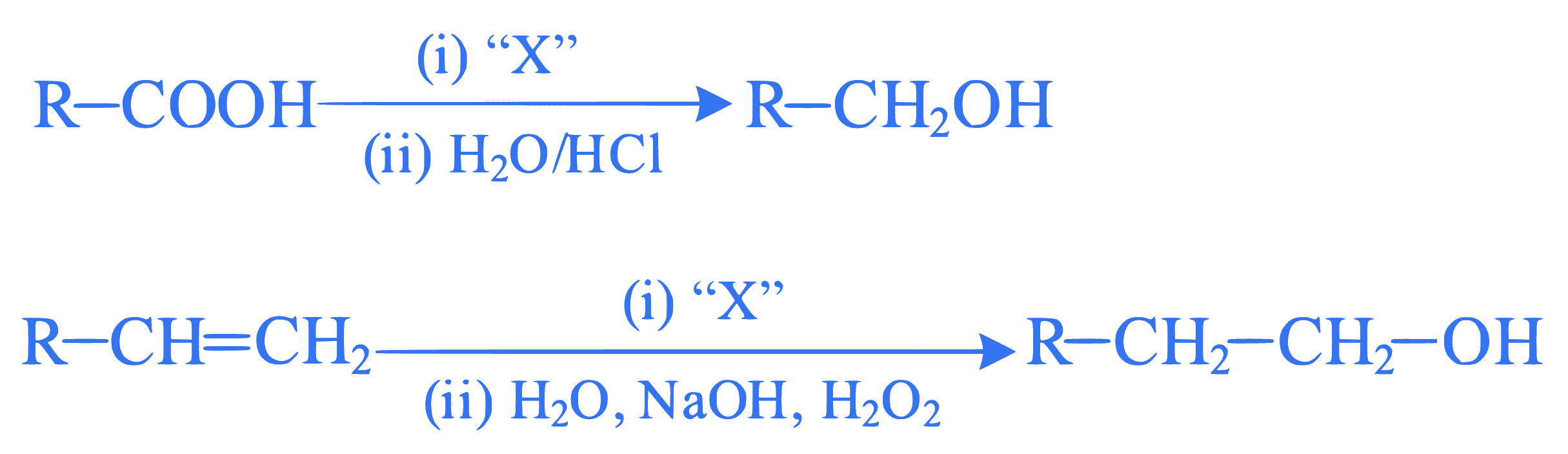 Identify 'X' in above reactions
Identify 'X' in above reactions
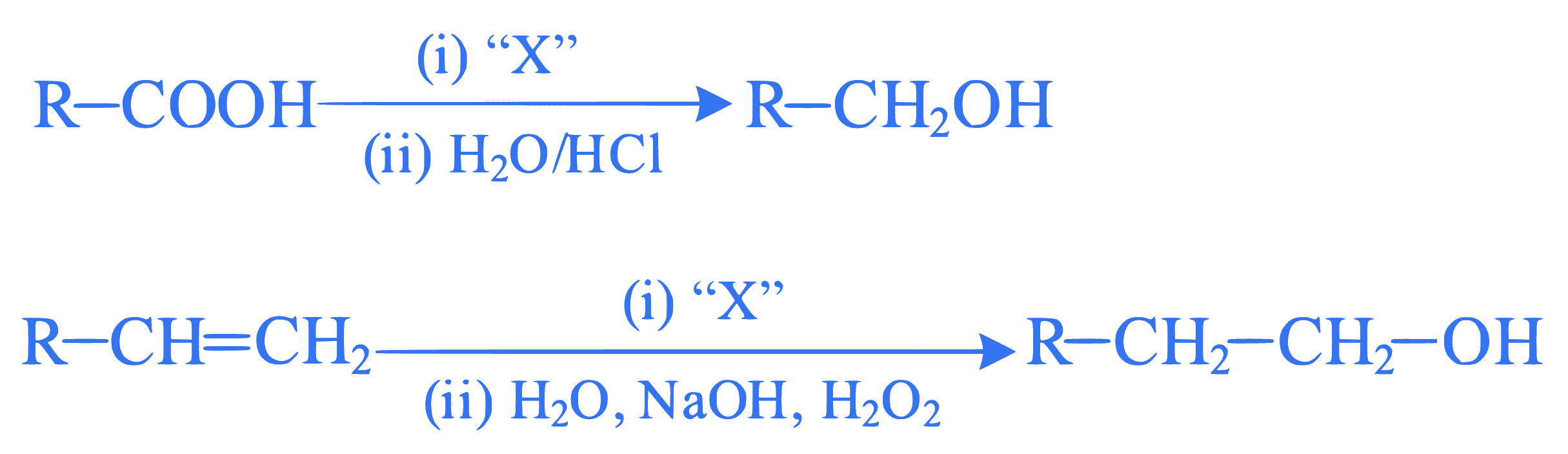 Identify 'X' in above reactions
Identify 'X' in above reactions
Q34.
mcq single
+4 / 1
The $$\mathrm{E}^{\Theta}$$ values for
$$\begin{aligned}
& \mathrm{Al}^{+} / \mathrm{Al}=+0.55 \mathrm{~V} \text { and } \mathrm{Tl}^{+} / \mathrm{Tl}=-0.34 \mathrm{~V} \\
& \mathrm{Al}^{3+} / \mathrm{Al}=-1.66 \mathrm{~V} \text { and } \mathrm{T}^{3+} / \mathrm{Tl}=+1.26 \mathrm{~V}
\end{aligned}$$
Identify the incorrect statement
Q35.
mcq single
+4 / 1
Molar conductance of an electrolyte increase with dilution according to the equation:
$$\Lambda_{\mathrm{m}}=\Lambda_{\mathrm{m}}^{\circ}-\mathrm{A} \sqrt{\mathrm{c}}$$
Which of the following statements are true?
(A) This equation applies to both strong and weak electrolytes.
(B) Value of the constant $$\mathrm{A}$$ depends upon the nature of the solvent.
(C) Value of constant $$\mathrm{A}$$ is same for both $$\mathrm{BaCl}_2$$ and $$\mathrm{MgSO}_4$$
(D) Value of constant $$\mathrm{A}$$ is same for both $$\mathrm{BaCl}_2$$ and $$\mathrm{Mg}(\mathrm{OH})_2$$
Choose the most appropriate answer from the options given below:
Q36.
mcq single
+4 / 1
The correct value of cell potential in volt for the reaction that occurs when the following two half cells are connected, is
$$\begin{aligned}
& \mathrm{Fe}_{(\mathrm{aq})}^{2+}+2 \mathrm{e}^{-} \rightarrow \mathrm{Fe}(\mathrm{s}), \mathrm{E}^{\circ}=-0.44 \mathrm{~V} \\
& \mathrm{Cr}_2 \mathrm{O}_7^{2-} \text { (aq) }+14 \mathrm{H}^{+}+6 e^{-} \rightarrow 2 \mathrm{Cr}^{3+}+7 \mathrm{H}_2 \mathrm{O} \\
& \mathrm{E}^{\circ}=+1.33 \mathrm{~V}
\end{aligned}$$
Q37.
mcq single
+4 / 1
Given below are two statements:
Statement I : The value of wave function, $$\psi $$ depends upon the coordinates of the electron in the atom.
Statement II : The probability of finding an electron at a point within an atom is proportional to the orbital wave function.
In the light of the above statements, choose the correct answer from the options given below:
Q38.
mcq single
+4 / 1
Incorrect set of quantum numbers from the following is :
Q39.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Ionisation enthalpy increases along each series of the transition elements from left to right. However, small variations occur.
Reason (R) : There is corresponding increase in nuclear charge which accompanies the filling of electrons in the inner d-orbitals.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q40.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Ionisation enthalpies of early actinoids are lower than for early lanthanoids.
Reason (R) : Electrons are entering 5f orbitals in actinoids which experience greater shielding from nuclear charge.
In the light of the above statements, choose the correct answer from the options given below :
Q41.
mcq single
+4 / 1
Which one of the following represents all isoelectronic species ?
Q42.
mcq single
+4 / 1
Given below are two statements:
Statement I : Hydrated chlorides and bromides of $$\mathrm{Ca}, \mathrm{Sr}$$ and $$\mathrm{Ba}$$ on heating undergo hydrolysis.
Statement II : Hydrated chlorides and bromides of $$\mathrm{Be}$$ and $$\mathrm{Mg}$$ on heating undergo dehydration.
In the light of the above statements, choose the correct answer from the options given below :
Q43.
mcq single
+4 / 1
Which one of the following statements is incorrect related to Molecular Orbital Theory?
Q44.
mcq single
+4 / 1
The correct order of dipole moments for molecules $$\mathrm{NH}_3, \mathrm{H}_2 \mathrm{~S}, \mathrm{CH}_4$$ and $$\mathrm{HF}$$, is:
Q45.
mcq single
+4 / 1
An acidic buffer is prepared by mixing :
Q46.
mcq single
+4 / 1
The correct options for the rate law that corresponds to overall first order reaction is
Q47.
mcq single
+4 / 1
For a reaction $$3 \mathrm{~A} \rightarrow 2 \mathrm{~B}$$
The average rate of appearance of $$\mathrm{B}$$ is given by $$\frac{\Delta[B]}{\Delta t}$$. The correct relation between the average rate of appearance of $$\mathrm{B}$$ with the average rate of disappearance of A is given in option :
Q48.
mcq single
+4 / 1
The correct option for a redox couple is:
Q49.
mcq single
+4 / 1
Identify 'X' in the following reaction.
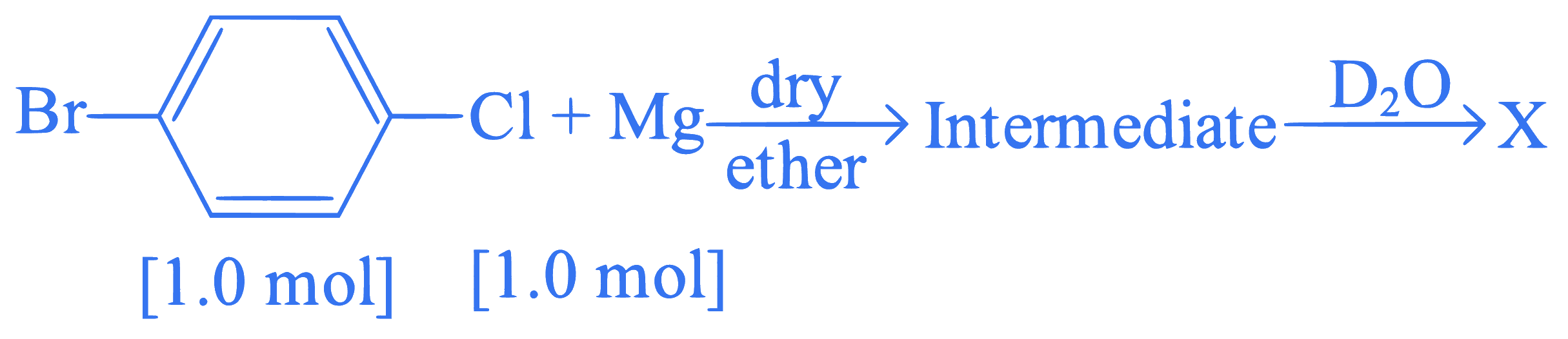
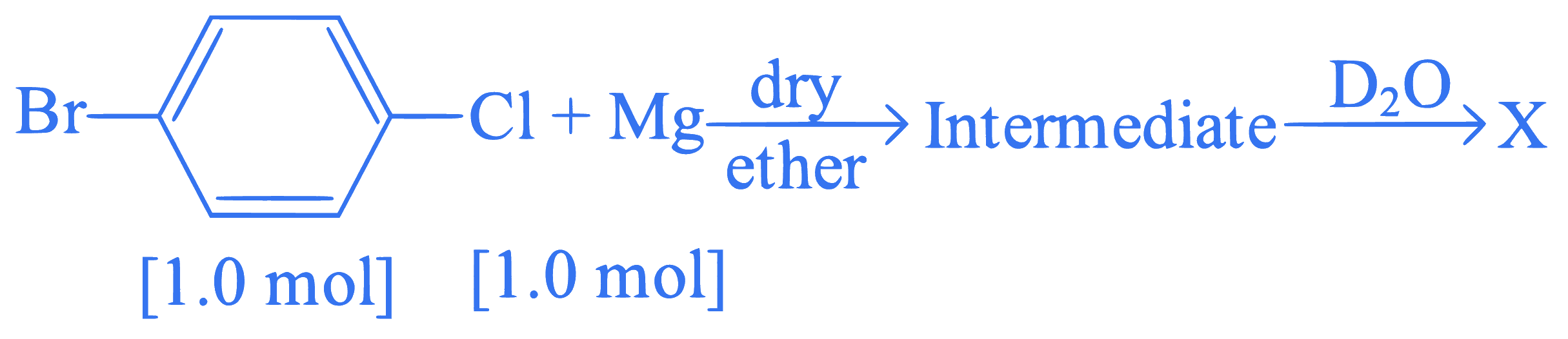
Q50.
mcq single
+4 / 1
Identify the product in the following reaction
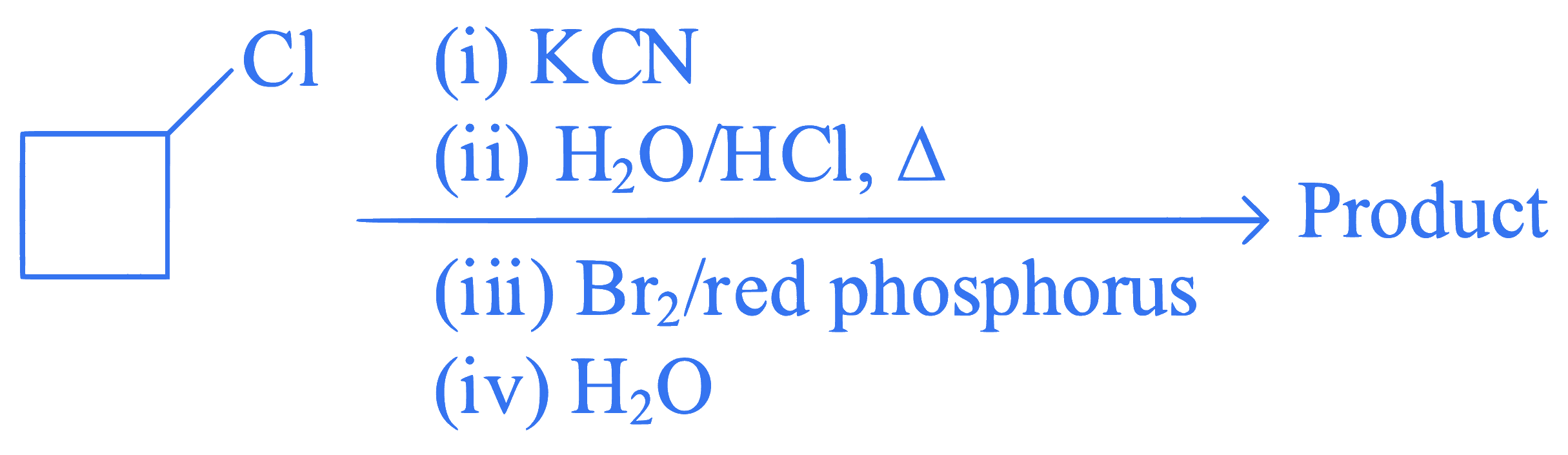
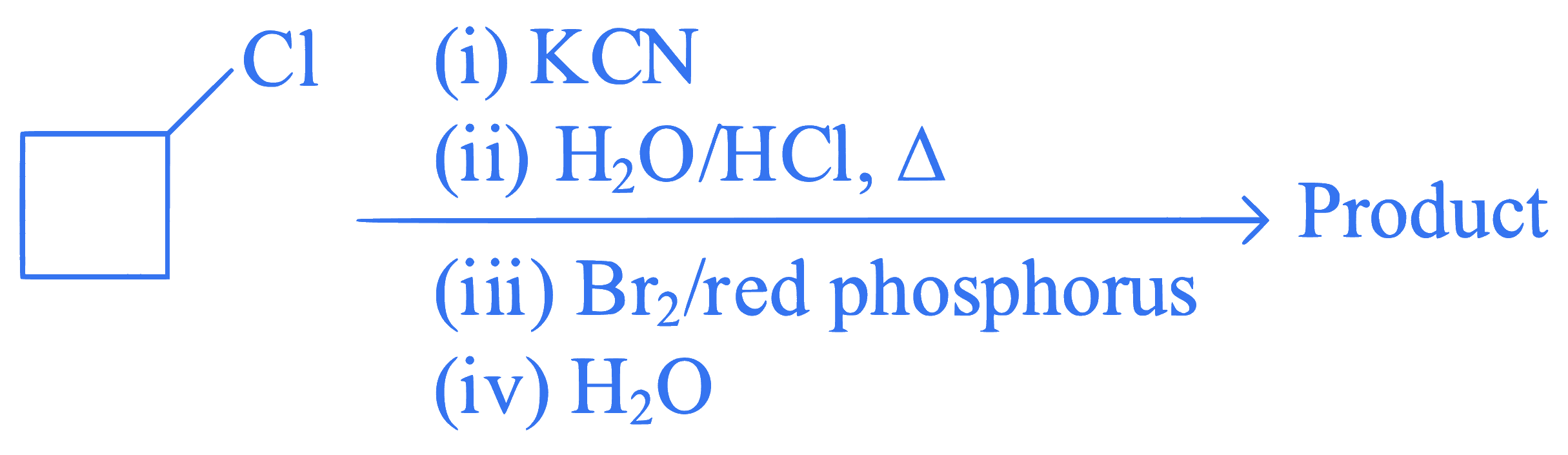
Q51.
mcq single
+4 / 1
Which amongst the following reactions of alkyl halides produces isonitrile as a major product?
(A) $$\mathrm{R-X+HCN\to}$$
(B) $$\mathrm{R-X+AgCN\to}$$
(C) $$\mathrm{R-X+KCN\to}$$
(D) $$\mathrm{R-X+NaCN\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{{C_2}{H_5}OH}^{{H_2}O}}}$$
Choose the most appropriate answer from the options given below:
Q52.
mcq single
+4 / 1
Choose the correct sequence of reagents in the conversion of 4-nitrotoluene to 2-bromotoluene.
Q53.
mcq single
+4 / 1
Which amongst the following aqueous solution of electrolytes will have minimum elevation in boiling point? Choose the correct option :-
Q54.
mcq single
+4 / 1
The density of 1 M solution of a compound 'X' is 1.25 g mL$$^{-1}$$. The correct option for the molality of solution is (Molar mass of compound X = 85 g):
Q55.
mcq single
+4 / 1
For a weak acid HA, the percentage of dissociation is nearly 1% at equilibrium. If the concentration of acid is 0.1 mol L$$^{-1}$$, then the correct option for its K$$_a$$ at the same temperature is :
Q56.
mcq single
+4 / 1
Which of the following is correctly matched?
Q57.
mcq single
+4 / 1
The correct sequence given below containing neutral, acidic, basic and amphoteric oxide each, respectively, is
Q58.
mcq single
+4 / 1
Consider the following reaction :-
$$2 \mathrm{H}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(\mathrm{g}) \Delta_{\mathrm{r}} \mathrm{H}^{\circ}=-483.64 \mathrm{~kJ} \text {. }$$
What is the enthalpy change for decomposition of one mole of water? (Choose the right option).
Q59.
mcq single
+4 / 1
Match the List - I with List - II
List - I
(Hydride)
List - II
(Type of Hydride)
(A)
$$\mathrm{NaH}$$
(I)
Electron precise
(B)
$$\mathrm{PH_3}$$
(II)
Saline
(C)
$$\mathrm{GeH_4}$$
(III)
Metallic
(D)
$$\mathrm{LaH_{2.87}}$$
(IV)
Electron rich
Choose the correct answer from the options given below :
Q60.
mcq single
+4 / 1
Select the element (M) whose trihalides cannot be hydrolysed to produce an ion of the form [M(H$$_2$$O$$_6$$]$$^{3+}$$
Q61.
mcq single
+4 / 1
Which of the following forms a set of complex and a double salt, respectively?
Q62.
mcq single
+4 / 1
Type of isomerism exhibited by compounds
$$
\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_6 \mathrm{Cl}_3,\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_5 \mathrm{Cl}_2 \mathrm{Cl}_2 \cdot \mathrm{H}_2 \mathrm{O}\right.\right.,\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_4 \mathrm{Cl}_2\right] \mathrm{Cl} .2 \mathrm{H}_2 \mathrm{O}$$ and the value of coordination number $$(\mathrm{CN})$$ of central metal ion in all these compounds, respectively is :
Q63.
mcq single
+4 / 1
Which amongst the following compounds/species is least basic?
Q64.
mcq single
+4 / 1
Which amongst the following compounds will show geometrical isomerism?
Q65.
mcq single
+4 / 1
Given below are two statements:
Statement I : In an organic compound, when inductive and electromeric effects operate in opposite directions, the inductive effect predominates.
Statement II : Hyperconjugation is observed in o-xylene.
In the light of the above statements, choose the correct answer from the options given below :
Q66.
mcq single
+4 / 1
The correct order for the rate of $$\alpha,\beta$$-dehydrohalogenation for the following compounds is _________.
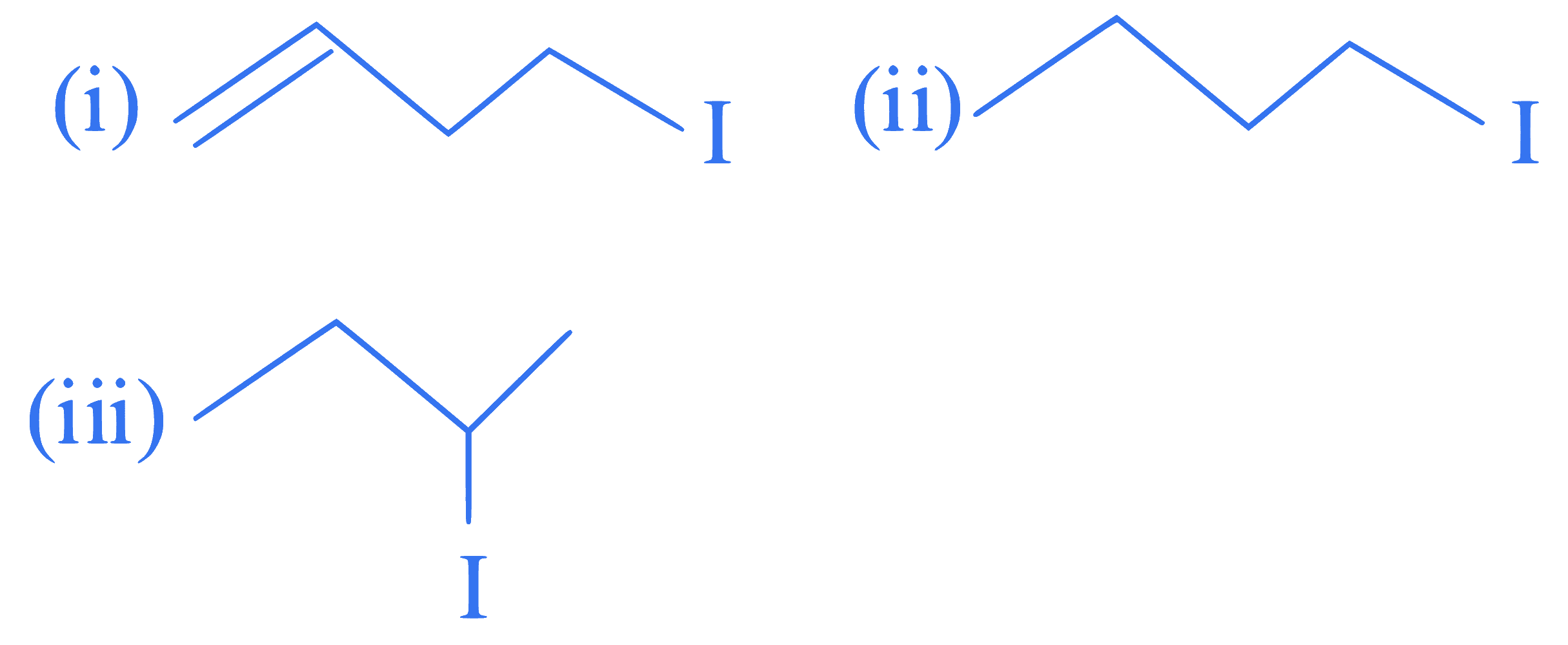
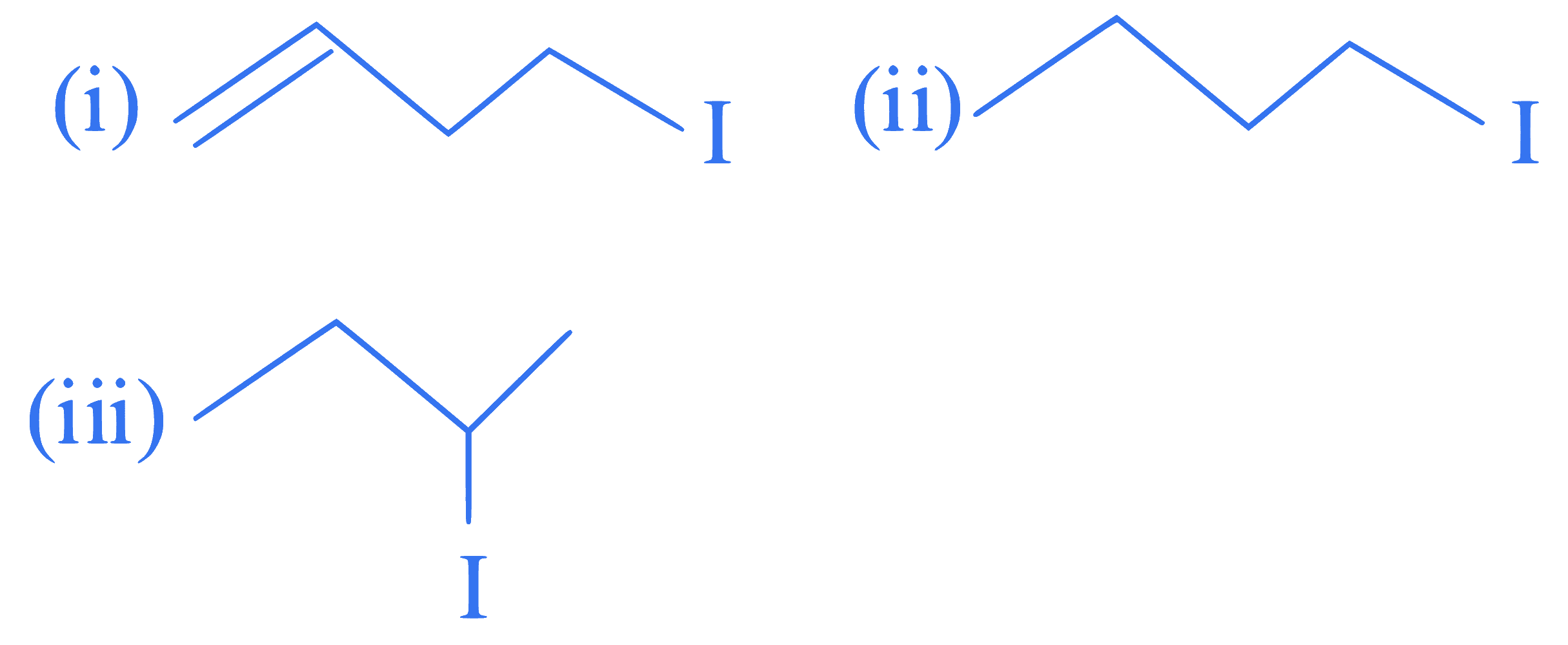
Q67.
mcq single
+4 / 1
Reagents which can be used to convert alcohols to carboxylic acids, are
(A) $$\mathrm{CrO}_3-\mathrm{H}_2 \mathrm{SO}_4$$
(B) $$\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7+\mathrm{H}_2 \mathrm{SO}_4$$
(C) $$\mathrm{KMnO}_4+\mathrm{KOH} / \mathrm{H}_3 \mathrm{O}^{+}$$
(D) $$\mathrm{Cu}, 573 \mathrm{~K}$$
(E) $$\mathrm{CrO}_3,\left(\mathrm{CH}_3 \mathrm{CO}\right)_2 \mathrm{O}$$
Choose the most appropriate answer from the options given below :
Q68.
mcq single
+4 / 1
The major product formed in the following conversion is ___________.
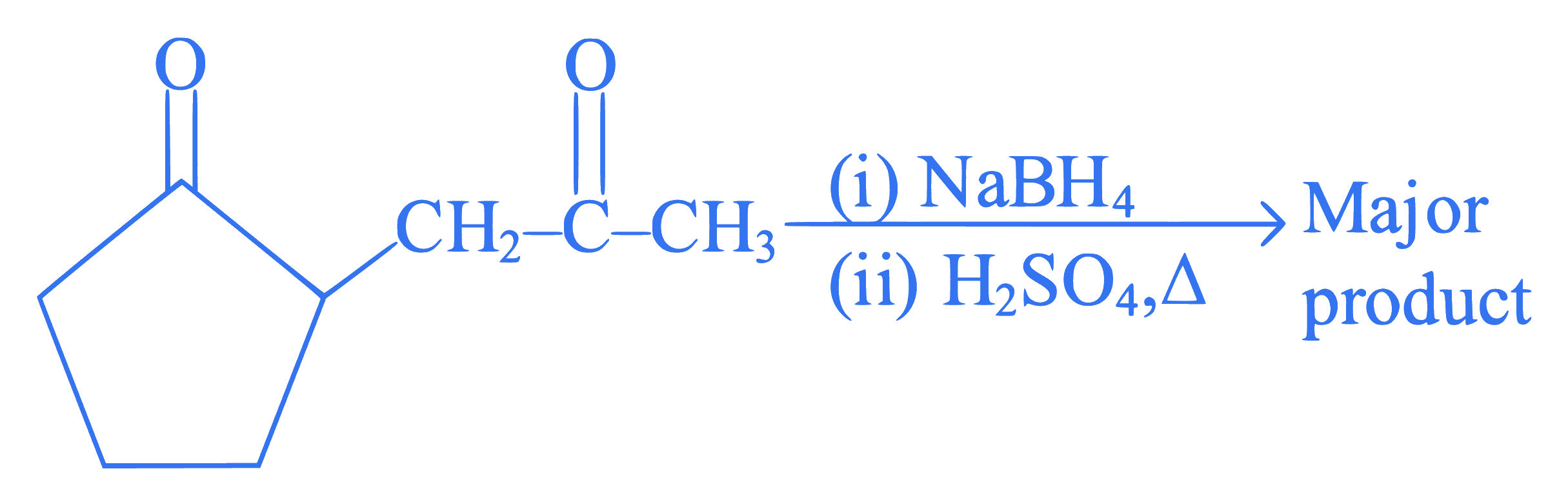
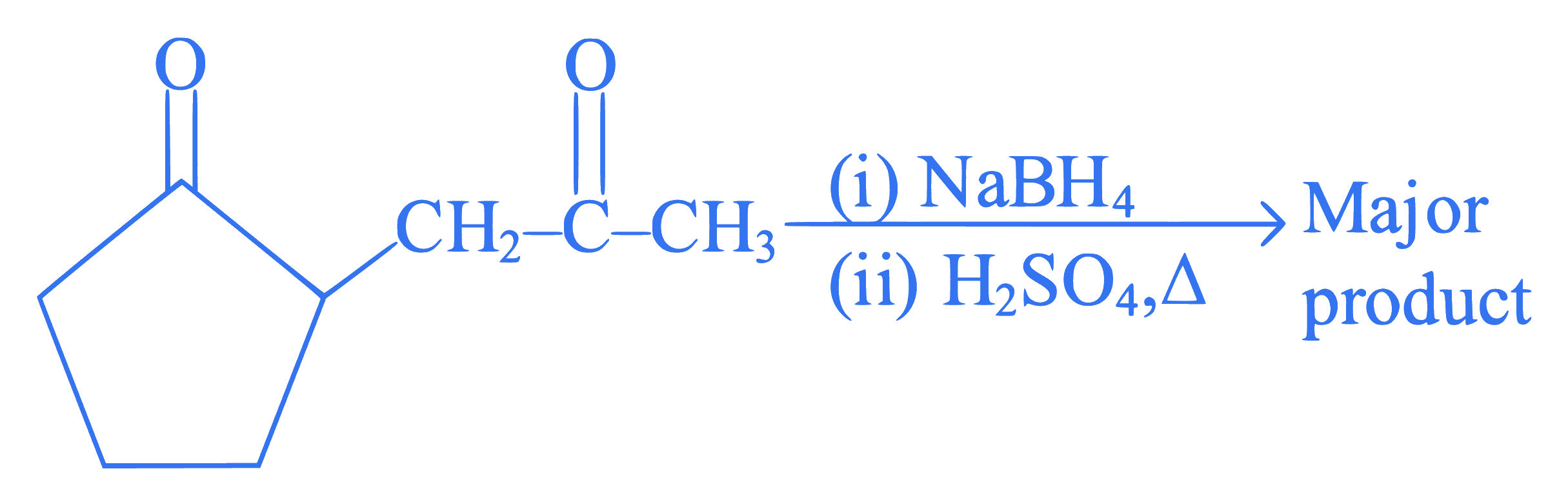
Q69.
mcq single
+4 / 1
The following conversion is known as:
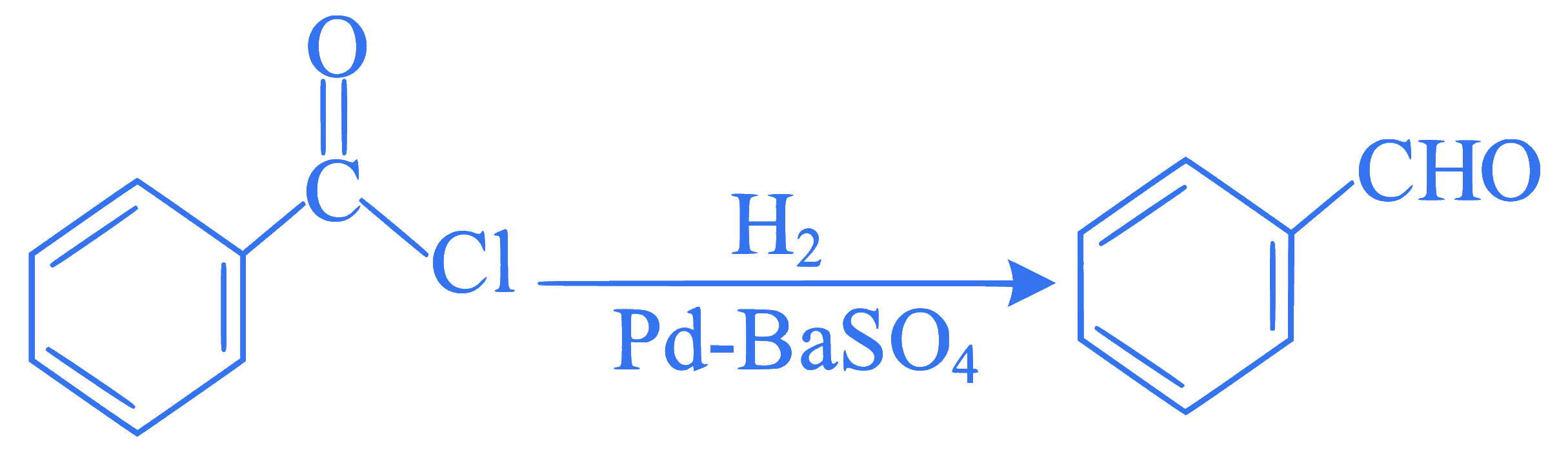
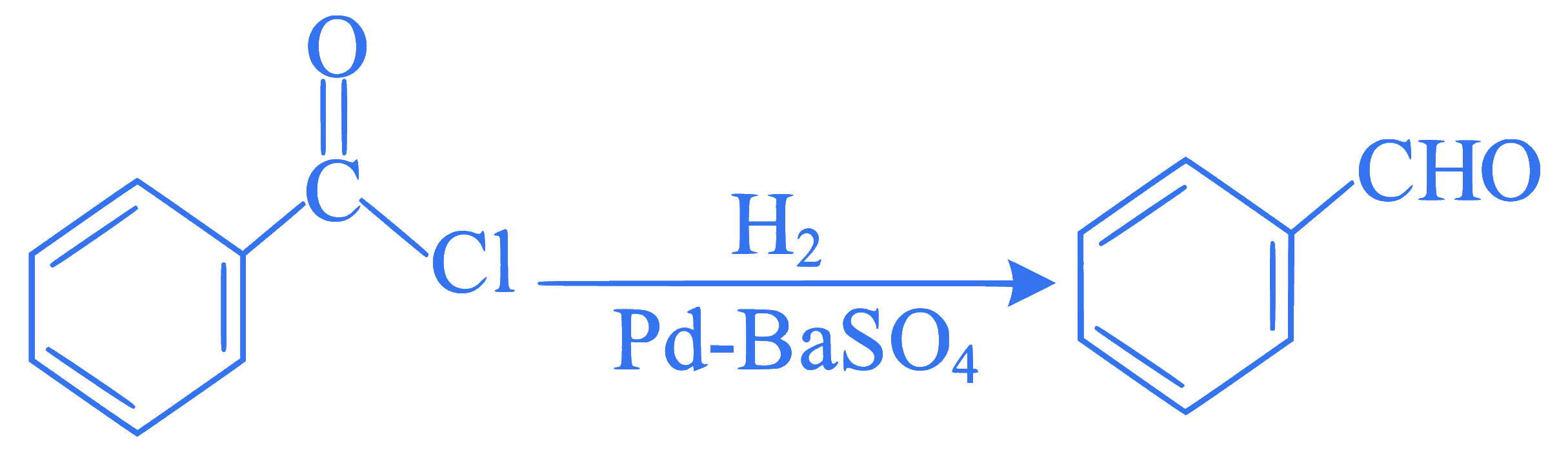
Q70.
mcq single
+4 / 1
Consider the given reaction:
$$\mathrm{CH}_3 \mathrm{COCH}_3 \stackrel{\text { dil } \mathrm{Ba}(\mathrm{OH})_2}{\longrightarrow} \text { ''X" }$$
The functional groups present in compound "$$\mathrm{X}$$" are:
Q71.
mcq single
+4 / 1
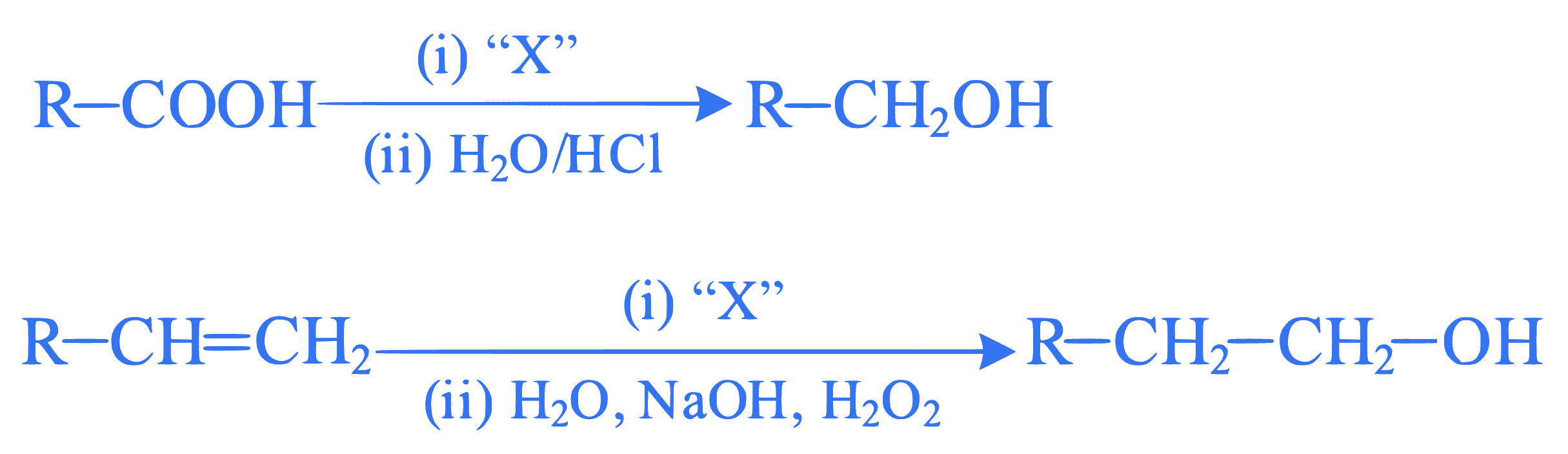 Identify 'X' in above reactions
Identify 'X' in above reactions
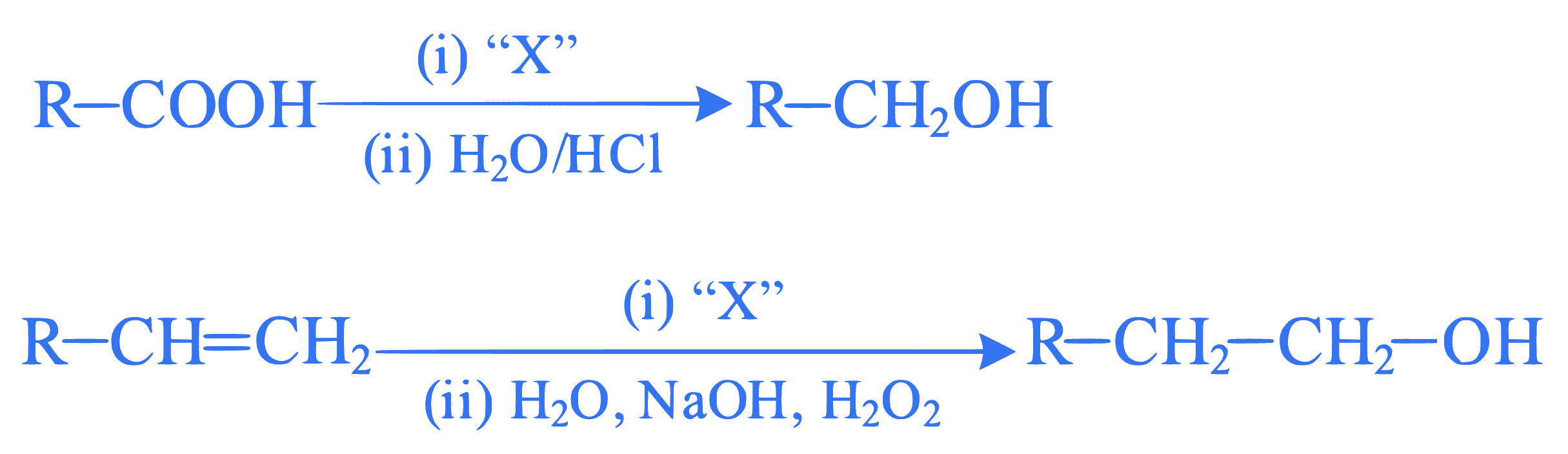 Identify 'X' in above reactions
Identify 'X' in above reactions
Q72.
mcq single
+4 / 1
The $$\mathrm{E}^{\Theta}$$ values for
$$\begin{aligned}
& \mathrm{Al}^{+} / \mathrm{Al}=+0.55 \mathrm{~V} \text { and } \mathrm{Tl}^{+} / \mathrm{Tl}=-0.34 \mathrm{~V} \\
& \mathrm{Al}^{3+} / \mathrm{Al}=-1.66 \mathrm{~V} \text { and } \mathrm{T}^{3+} / \mathrm{Tl}=+1.26 \mathrm{~V}
\end{aligned}$$
Identify the incorrect statement
Q73.
mcq single
+4 / 1
Molar conductance of an electrolyte increase with dilution according to the equation:
$$\Lambda_{\mathrm{m}}=\Lambda_{\mathrm{m}}^{\circ}-\mathrm{A} \sqrt{\mathrm{c}}$$
Which of the following statements are true?
(A) This equation applies to both strong and weak electrolytes.
(B) Value of the constant $$\mathrm{A}$$ depends upon the nature of the solvent.
(C) Value of constant $$\mathrm{A}$$ is same for both $$\mathrm{BaCl}_2$$ and $$\mathrm{MgSO}_4$$
(D) Value of constant $$\mathrm{A}$$ is same for both $$\mathrm{BaCl}_2$$ and $$\mathrm{Mg}(\mathrm{OH})_2$$
Choose the most appropriate answer from the options given below:
Q74.
mcq single
+4 / 1
The correct value of cell potential in volt for the reaction that occurs when the following two half cells are connected, is
$$\begin{aligned}
& \mathrm{Fe}_{(\mathrm{aq})}^{2+}+2 \mathrm{e}^{-} \rightarrow \mathrm{Fe}(\mathrm{s}), \mathrm{E}^{\circ}=-0.44 \mathrm{~V} \\
& \mathrm{Cr}_2 \mathrm{O}_7^{2-} \text { (aq) }+14 \mathrm{H}^{+}+6 e^{-} \rightarrow 2 \mathrm{Cr}^{3+}+7 \mathrm{H}_2 \mathrm{O} \\
& \mathrm{E}^{\circ}=+1.33 \mathrm{~V}
\end{aligned}$$
Q75.
mcq single
+4 / 1
Given below are two statements:
Statement I : The value of wave function, $$\psi $$ depends upon the coordinates of the electron in the atom.
Statement II : The probability of finding an electron at a point within an atom is proportional to the orbital wave function.
In the light of the above statements, choose the correct answer from the options given below:
Q76.
mcq single
+4 / 1
Incorrect set of quantum numbers from the following is :
Physics
Physics
Q1.
mcq single
+4 / 1
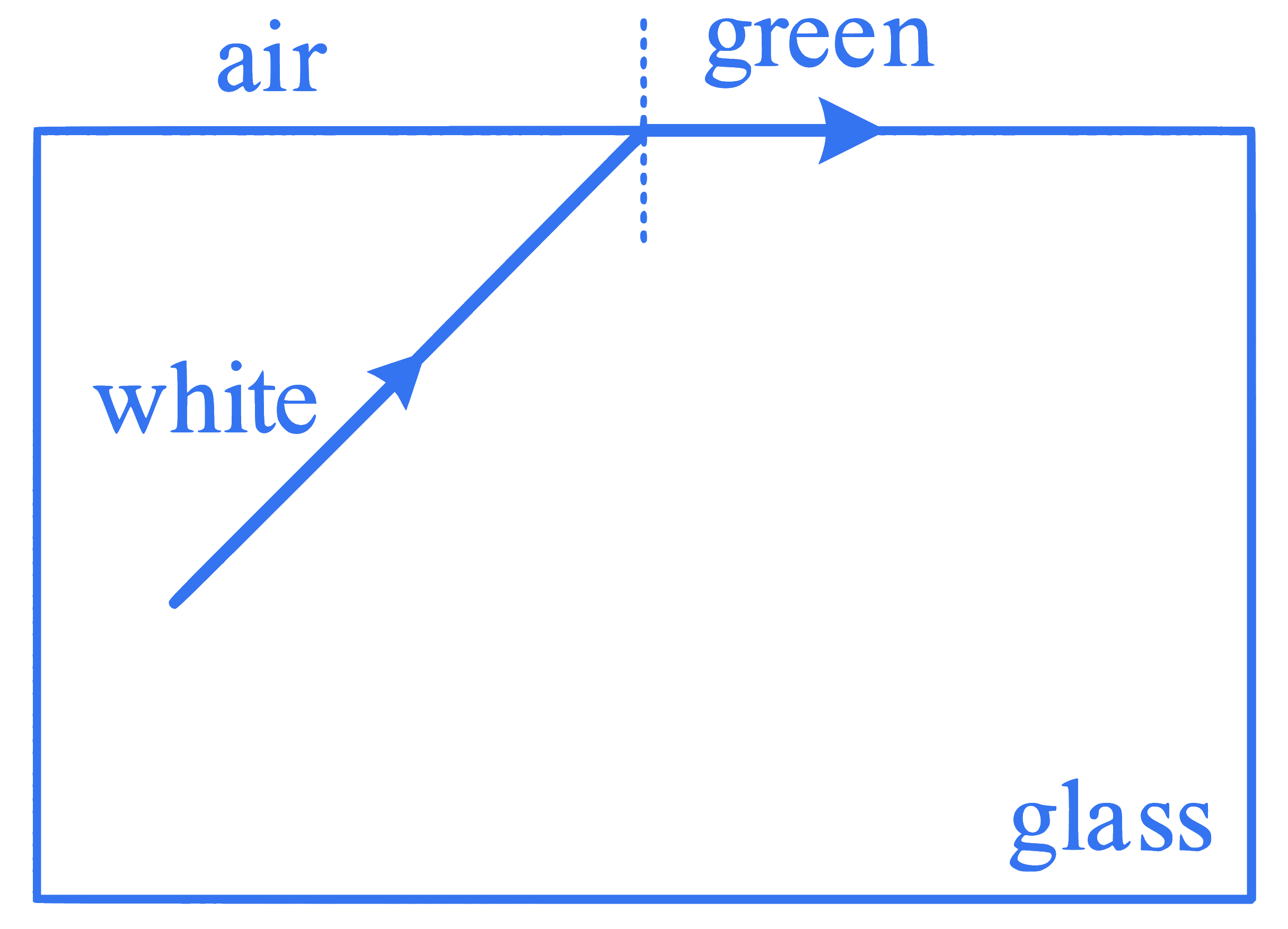 Which set of colours will come out in air for a situation shown in figure?
Which set of colours will come out in air for a situation shown in figure?
 Which set of colours will come out in air for a situation shown in figure?
Which set of colours will come out in air for a situation shown in figure?
Q2.
mcq single
+4 / 1
A constant torque of $$100 \mathrm{~N} \mathrm{~m}$$ turns a wheel of moment of inertia $$300 \mathrm{~kg} \mathrm{~m}^2$$ about an axis passing through its centre. Starting from rest, its angular velocity after $$3 \mathrm{~s}$$ is :-
Q3.
mcq single
+4 / 1
An emf is generated by an ac generator having 100 turn coil, of loop area $$1 \mathrm{~m}^2$$. The coil rotates at a speed of one revolution per second and placed in a uniform magnetic field of $$0.05 \mathrm{~T}$$ perpendicular to the axis of rotation of the coil. The maximum value of emf is :-
Q4.
mcq single
+4 / 1
The mechanical quantity, which has dimensions of reciprocal of mass $$(\mathrm{M}^{-1})$$ is :
Q5.
mcq single
+4 / 1
The diameter of a spherical bob, when measured with vernier callipers yielded the following values : $$3.33 \mathrm{~cm}, 3.32 \mathrm{~cm}, 3.34 \mathrm{~cm}, 3.33 \mathrm{~cm}$$ and $$3.32 \mathrm{~cm}$$. The mean diameter to appropriate significant figures is :
Q6.
mcq single
+4 / 1
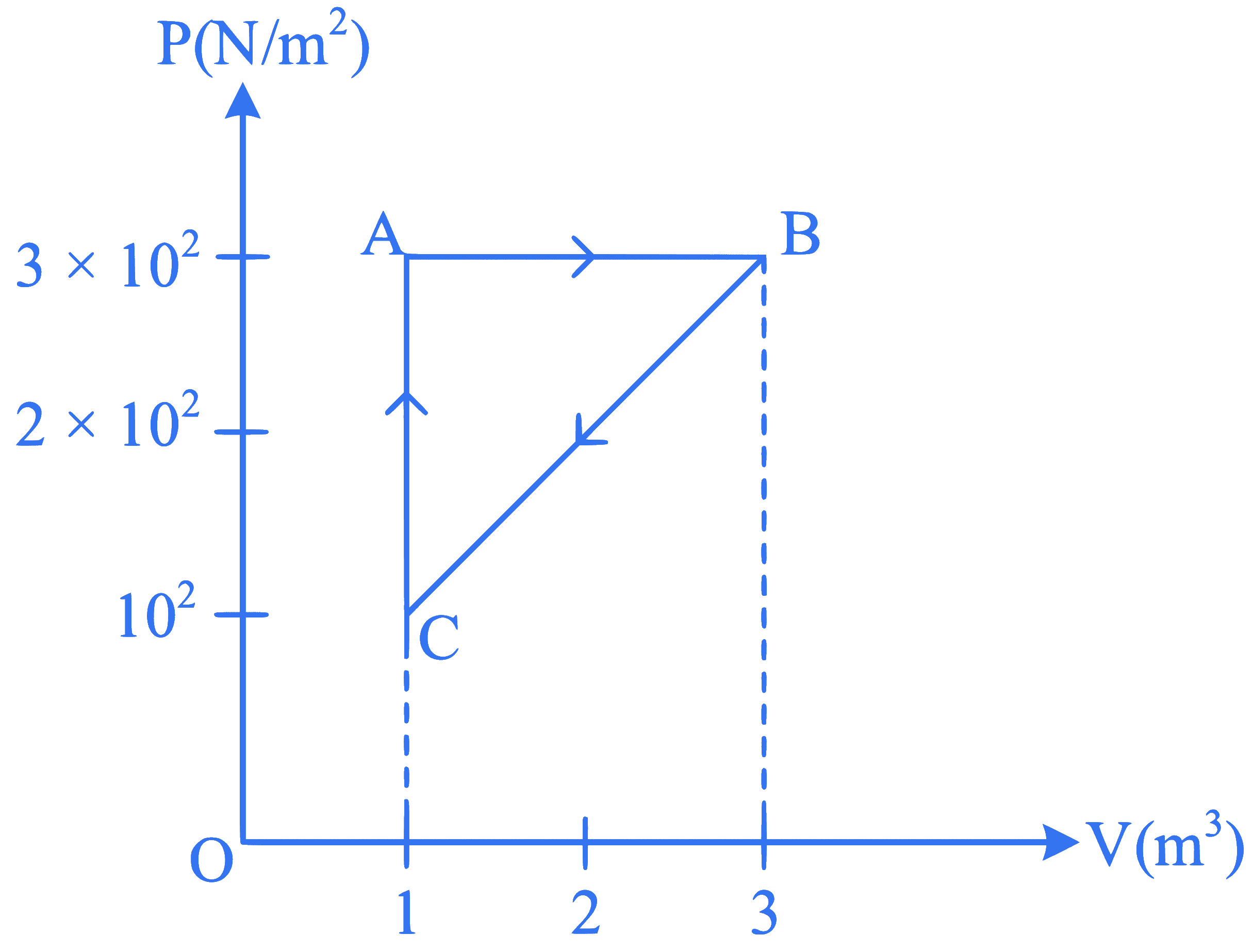
For the given cycle, the work done during isobaric process is:

Q7.
mcq single
+4 / 1
A container of volume $$200 \mathrm{~cm}^3$$ contains 0.2 mole of hydrogen gas and 0.3 mole of argon gas. The pressure of the system at temperature $$200 \mathrm{~K}$$ ($$\mathrm{R}=8.3 \mathrm{JK}^{-1} \mathrm{~mol}^{-1}$$) will be :-
Q8.
mcq single
+4 / 1
Which of the following statement is not true?
Q9.
mcq single
+4 / 1
The amount of elastic potential energy per unit volume (in SI unit) of a steel wire of length $$100 \mathrm{~cm}$$ to stretch it by $$1 \mathrm{~mm}$$ is (if Young's modulus of the wire $$=2.0 \times 10^{11} \mathrm{Nm}^{-2}$$ ) :
Q10.
mcq single
+4 / 1
The viscous drag acting on a metal sphere of diameter $$1 \mathrm{~mm}$$, falling through a fluid of viscosity $$0.8 \mathrm{~Pa}$$ s with a velocity of $$2 \mathrm{~m} \mathrm{~s}^{-1}$$ is equal to :
Q11.
mcq single
+4 / 1
According to Gauss law of electrostatics, electric flux through a closed surface depends on :
Q12.
mcq single
+4 / 1
A charge $$\mathrm{Q} ~\mu \mathrm{C}$$ is placed at the centre of a cube. The flux coming out from any one of its faces will be (in SI unit) :
Q13.
mcq single
+4 / 1
If a conducting sphere of radius $$\mathrm{R}$$ is charged. Then the electric field at a distance $$\mathrm{r}(\mathrm{r} > \mathrm{R})$$ from the centre of the sphere would be, $$(\mathrm{V}=$$ potential on the surface of the sphere)
Q14.
mcq single
+4 / 1
If $$\mathrm{R}$$ is the radius of the earth and $$\mathrm{g}$$ is the acceleration due to gravity on the earth surface. Then the mean density of the earth will be :
Q15.
mcq single
+4 / 1
The escape velocity of a body on the earth surface is $$11.2 \mathrm{~km} / \mathrm{s}$$. If the same body is projected upward with velocity $$22.4 \mathrm{~km} / \mathrm{s}$$, the velocity of this body at infinite distance from the centre of the earth will be:
Q16.
mcq single
+4 / 1
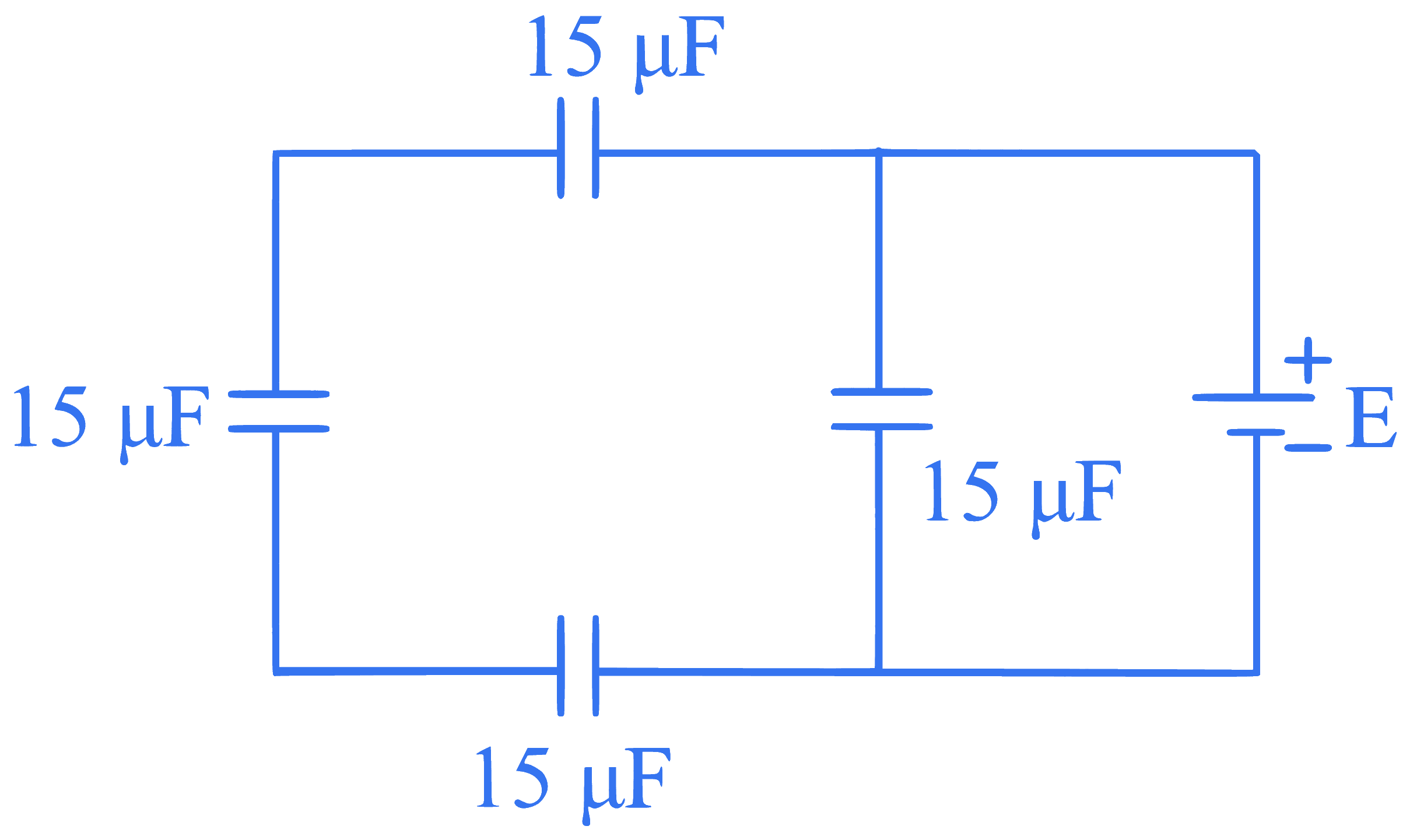
The equivalent capacitance of the arrangement shown in figure is:

Q17.
mcq single
+4 / 1
To produce an instantaneous displacement current of $$2 \mathrm{~mA}$$ in the space between the parallel plates of a capacitor of capacitance $$4 ~\mu \mathrm{F}$$, the rate of change of applied variable potential difference $$\left(\frac{\mathrm{dV}}{\mathrm{dt}}\right)$$ must be :-
Q18.
mcq single
+4 / 1
$$\epsilon_0$$ and $$\mu_0$$ are the electric permittivity and magnetic permeability of free space respectively. If the corresponding quantities of a medium are $$2 ~\epsilon_0$$ and $$1.5 ~\mu_0$$ respectively, the refractive index of the medium will nearly be :
Q19.
mcq single
+4 / 1
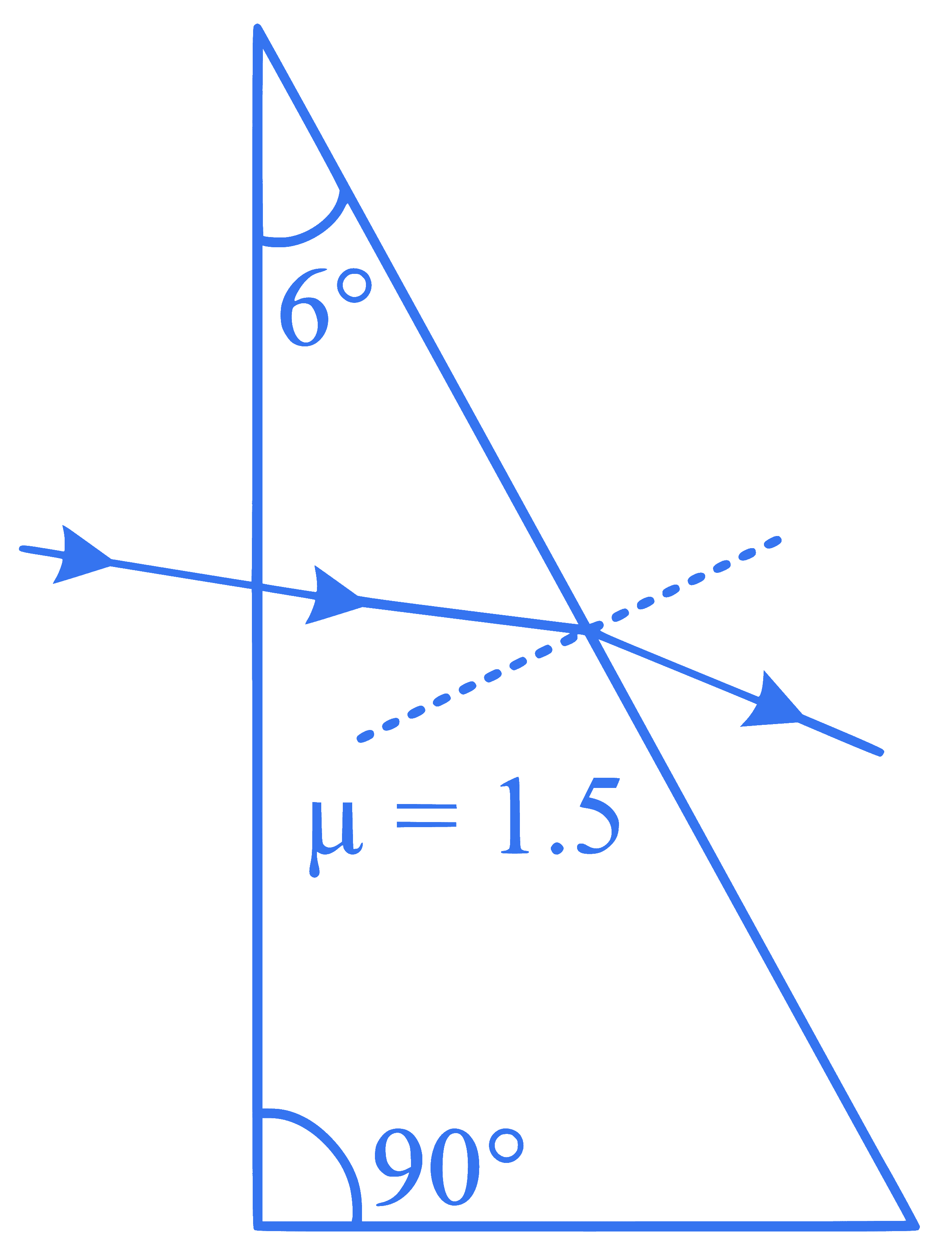
A horizontal ray of light is incident on the right angled prism with prism angle $$6^{\circ}$$. If the refractive index of the material of the prism is 1.5 , then the angle of emergence will be:

Q20.
mcq single
+4 / 1
An object is mounted on a wall. Its image of equal size is to be obtained on a parallel wall with the help of a convex lens placed between these walls. The lens is kept at distance x in front of the second wall. The required focal length of the lens will be :
Q21.
mcq single
+4 / 1
A lens is made up of 3 different transparent media as shown in figure. A point object O is placed on its axis beyond $$2f$$. How many real images will be obtained on the other side?


Q22.
mcq single
+4 / 1
A simple pendulum oscillating in air has a period of $$\sqrt{3} \mathrm{~s}$$. If it is completely immersed in non-viscous liquid, having density $$\left(\frac{1}{4}\right)^{\text {th }}$$ of the material of the bob, the new period will be :-
Q23.
mcq single
+4 / 1
The given circuit is equivalent to:
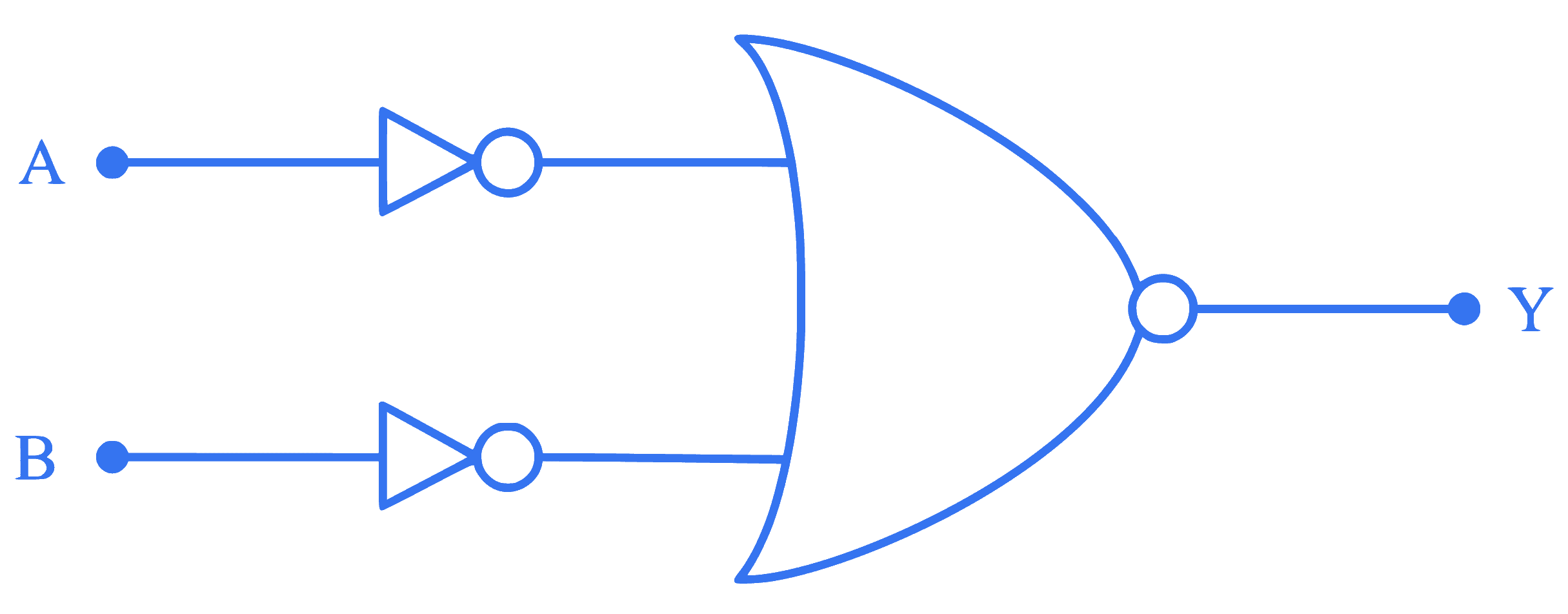

Q24.
mcq single
+4 / 1
On the basis of electrical conductivity, which one of the following material has the smallest resistivity?
Q25.
mcq single
+4 / 1
A p-type extrinsic semiconductor is obtained when Germanium is doped with:
Q26.
mcq single
+4 / 1
A uniform electric field and a uniform magnetic field are acting along the same direction in a certain region. If an electron is projected in the region such that its velocity is pointed along the direction of fields, then the electron:
Q27.
mcq single
+4 / 1
A long straight wire of length $$2 \mathrm{~m}$$ and mass $$250 \mathrm{~g}$$ is suspended horizontally in a uniform horizontal magnetic field of $$0.7 \mathrm{~T}$$. The amount of current flowing through the wire will be $$\left(\mathrm{g}=9.8 \mathrm{~ms}^{-2}\right)$$ :
Q28.
mcq single
+4 / 1
The de Broglie wavelength associated with an electron, accelerated by a potential difference of 81 V is given by:
Q29.
mcq single
+4 / 1
The maximum kinetic energy of the emitted photoelectrons in photoelectric effect is independent of:
Q30.
mcq single
+4 / 1
The position of a particle is given by
$$\vec{r}(t)=4 t \hat{i}+2 t^2 \hat{j}+5 \hat{k}
$$
where $$\mathrm{t}$$ is in seconds and $$\mathrm{r}$$ in metre. Find the magnitude and direction of velocity $$v(t)$$, at $$t=1 \mathrm{~s}$$, with respect to $$\mathrm{x}$$-axis
Q31.
mcq single
+4 / 1
The angular momentum of an electron moving in an orbit of hydrogen atom is $$\mathrm{1.5\left(\frac{h}{\pi}\right)}$$. The energy in the same orbit is nearly.
Q32.
mcq single
+4 / 1
The ground state energy of hydrogen atom is $$-13.6 ~\mathrm{eV}$$. The energy needed to ionize hydrogen atom from its second excited state will be :
Q33.
mcq single
+4 / 1
The wavelength of Lyman series of hydrogen atom appears in:
Q34.
mcq single
+4 / 1
A ball is projected from point A with velocity $$20 \mathrm{~m} \mathrm{~s}^{-1}$$ at an angle $$60^{\circ}$$ to the horizontal direction. At the highest point $$\mathrm{B}$$ of the path (as shown in figure), the velocity $$\mathrm{v} \mathrm{m} \mathrm{s}^{-1}$$ of the ball will be:
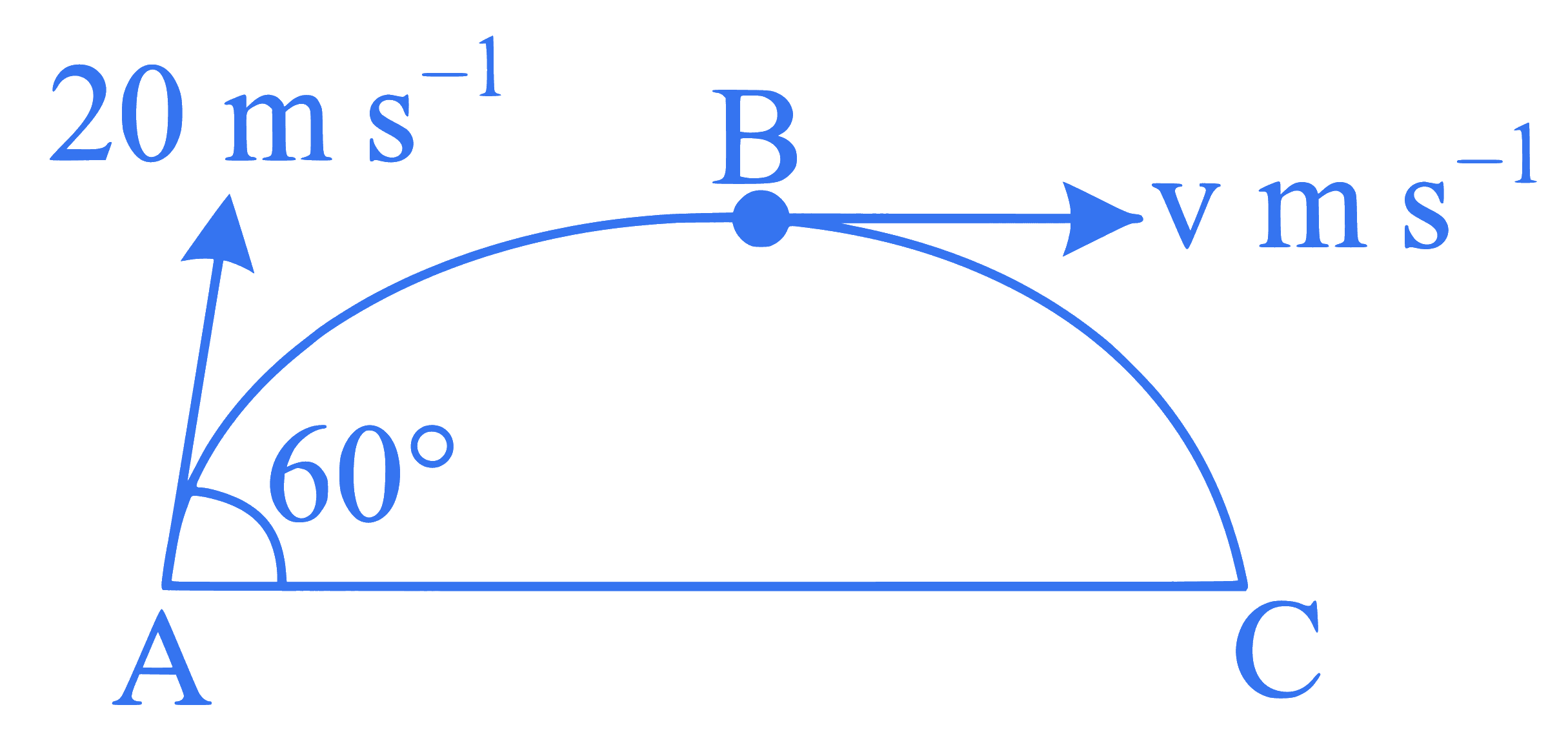

Q35.
mcq single
+4 / 1
A particle is executing uniform circular motion with velocity $$\vec{v}$$ and acceleration $$\vec{a}$$. Which of the following is true?
Q36.
mcq single
+4 / 1
Two particles A and B initially at rest, move towards each other under mutual force of attraction. At an instance when the speed of A is v and speed of B is 3v, the speed of centre of mass is :
Q37.
mcq single
+4 / 1
A bullet of mass $$m$$ hits a block of mass $$M$$ elastically. The transfer of energy is the maximum, when :
Q38.
mcq single
+4 / 1
A $$1 \mathrm{~kg}$$ object strikes a wall with velocity $$1 \mathrm{~m} \mathrm{~s}^{-1}$$ at an angle of $$60^{\circ}$$ with the wall and reflects at the same angle. If it remains in contact with wall for $$0.1 \mathrm{~s}$$, then the force exerted on the wall is :
Q39.
mcq single
+4 / 1
The $$4^{\text {th }}$$ overtone of a closed organ pipe is same as that of $$3^{\text {rd }}$$ overtone of an open pipe. The ratio of the length of the closed pipe to the length of the open pipe is :
Q40.
mcq single
+4 / 1
A block of mass 2 kg is placed on inclined rough surface AC (as shown in figure) of coefficient of friction $$\mu$$. If g = 10 m s$$^{-2}$$, the net force (in N) on the block will be:
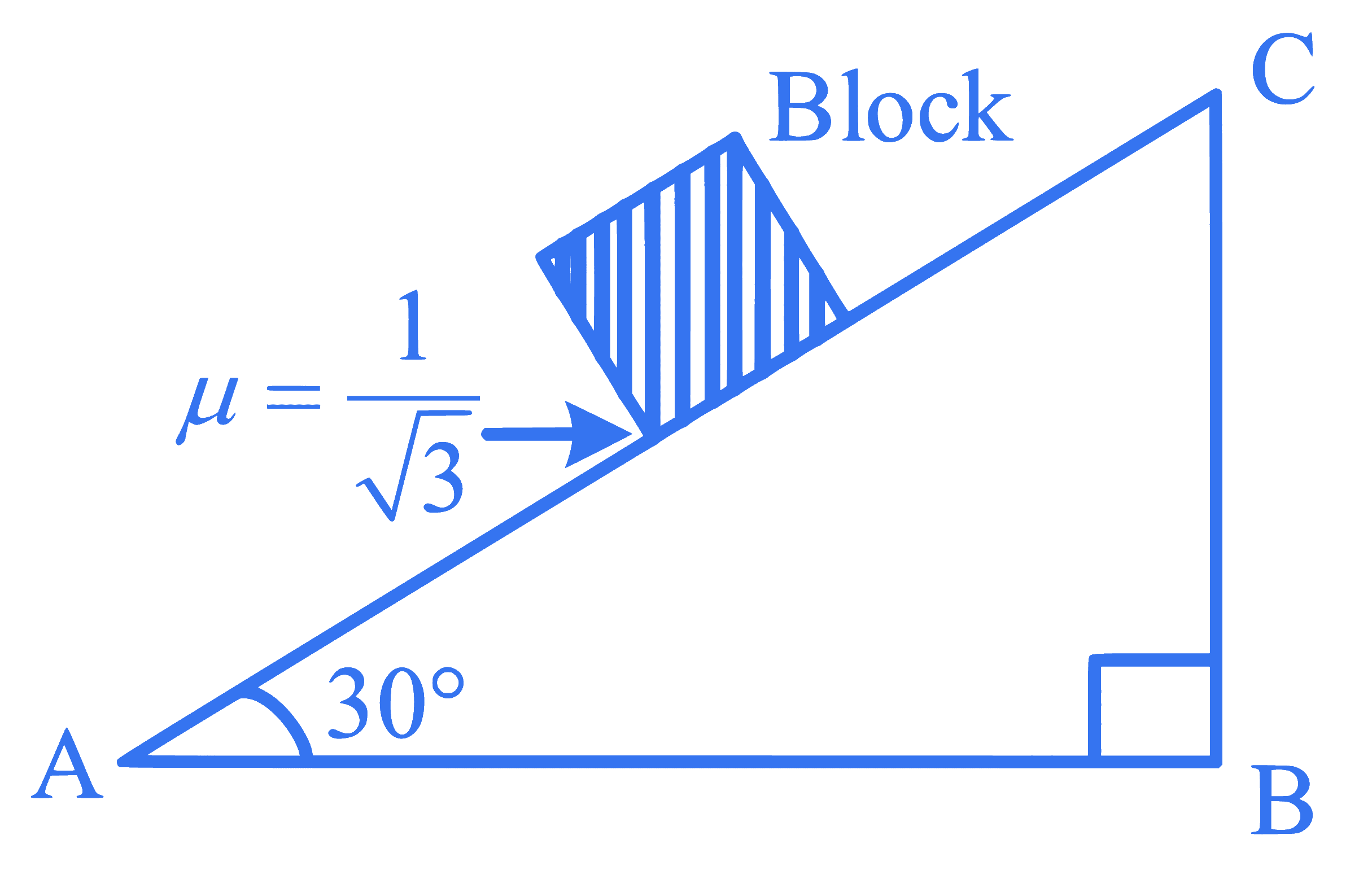

Q41.
mcq single
+4 / 1
The variation of susceptibility $$(\chi)$$ with absolute temperature ( $$\mathrm{T}$$ ) for a paramagnetic material is represented as :
Q42.
mcq single
+4 / 1
A particle moves with a velocity $$(5 \hat{i}-3 \hat{j}+6 \hat{k}) ~\mathrm{ms}^{-1}$$ horizontally under the action of constant force $$(10 \hat{\mathrm{i}}+10 \hat{\mathrm{j}}+20 \hat{\mathrm{k}}) \mathrm{N}$$. The instantaneous power supplied to the particle is :
Q43.
mcq single
+4 / 1
A certain wire $$\mathrm{A}$$ has resistance $$81 ~\Omega$$. The resistance of another wire $$\mathrm{B}$$ of same material and equal length but of diameter thrice the diameter of A will be :
Q44.
mcq single
+4 / 1
A copper wire of radius $$1 \mathrm{~mm}$$ contains $$10^{22}$$ free electrons per cubic metre. The drift velocity for free electrons when $$10 \mathrm{~A}$$ current flows through the wire will be (Given, charge on electron $$=1.6 \times 10^{-19} \mathrm{C}$$ ) :
Q45.
mcq single
+4 / 1
The emf of a cell having internal resistance $$1 \Omega$$ is balanced against a length of $$330 \mathrm{~cm}$$ on a potentiometer wire. When an external resistance of $$2 \Omega$$ is connected across the cell, the balancing length will be :
Q46.
mcq single
+4 / 1
If Z$$_1$$ and Z$$_2$$ are the impedances of the given circuits (a) and (b) as shown in figures, then choose the correct option
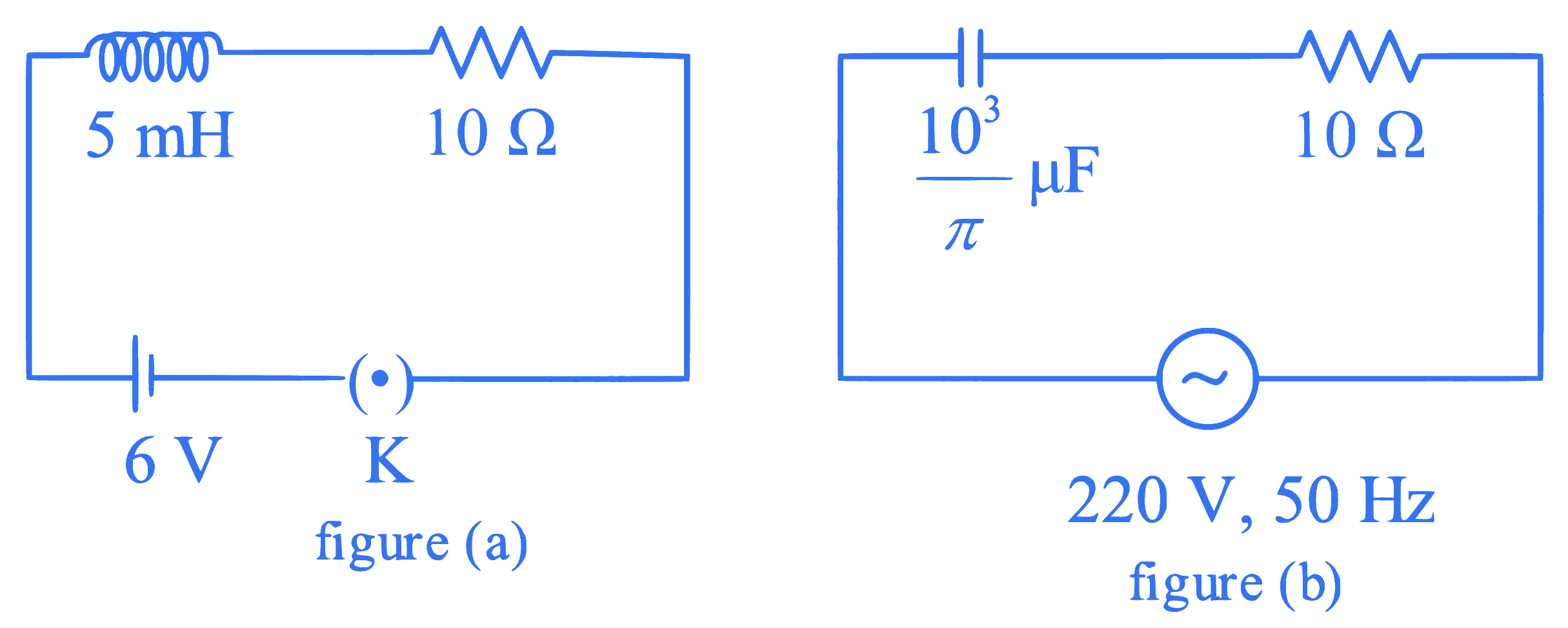

Q47.
mcq single
+4 / 1
The maximum power is dissipated for an ac in a/an:
Q48.
mcq single
+4 / 1
For very high frequencies, the effective impedance of the circuit (shown in the figure) will be:-
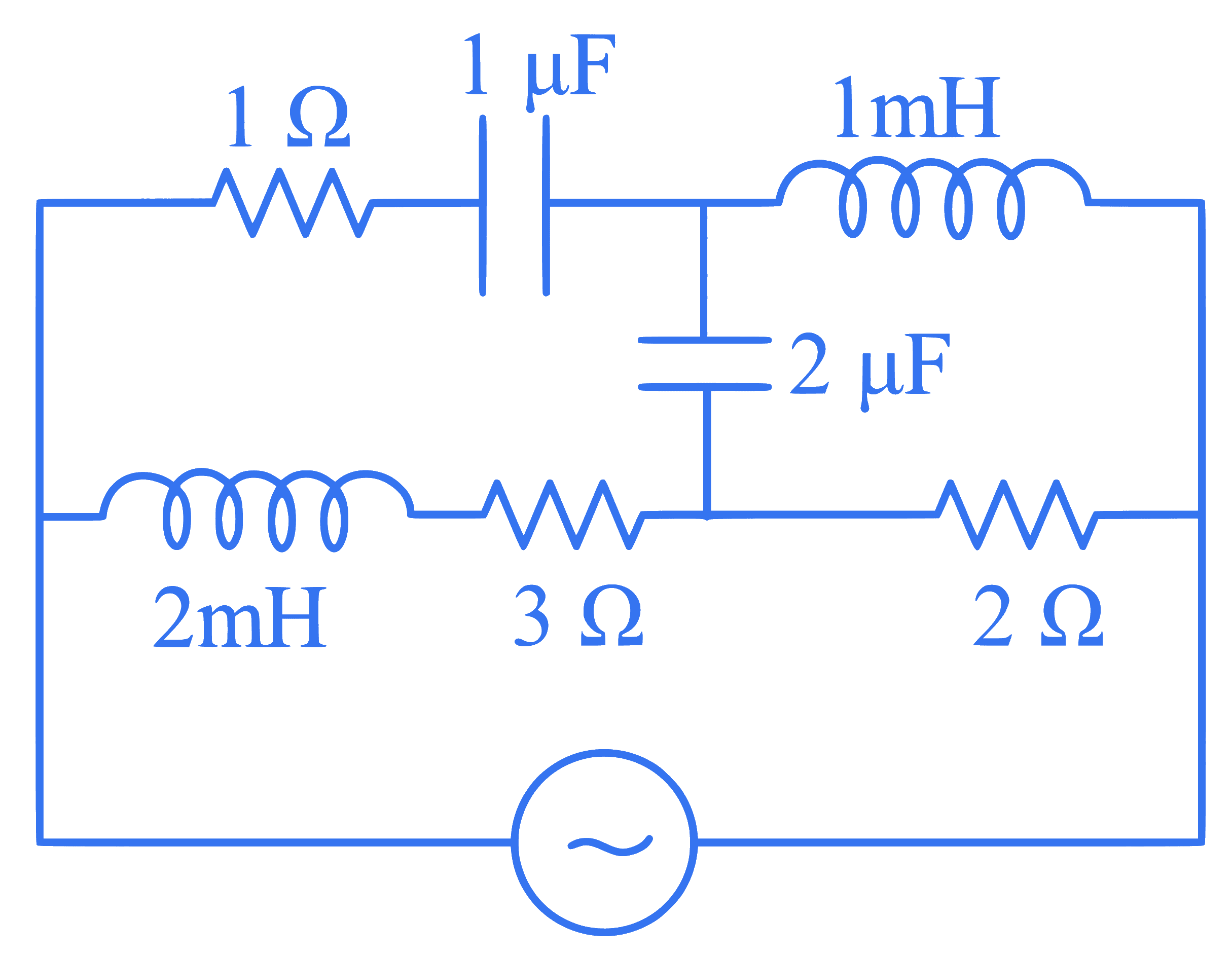

Q49.
mcq single
+4 / 1
An ac source is connected in the given circuit. The value of $\phi$ will be :
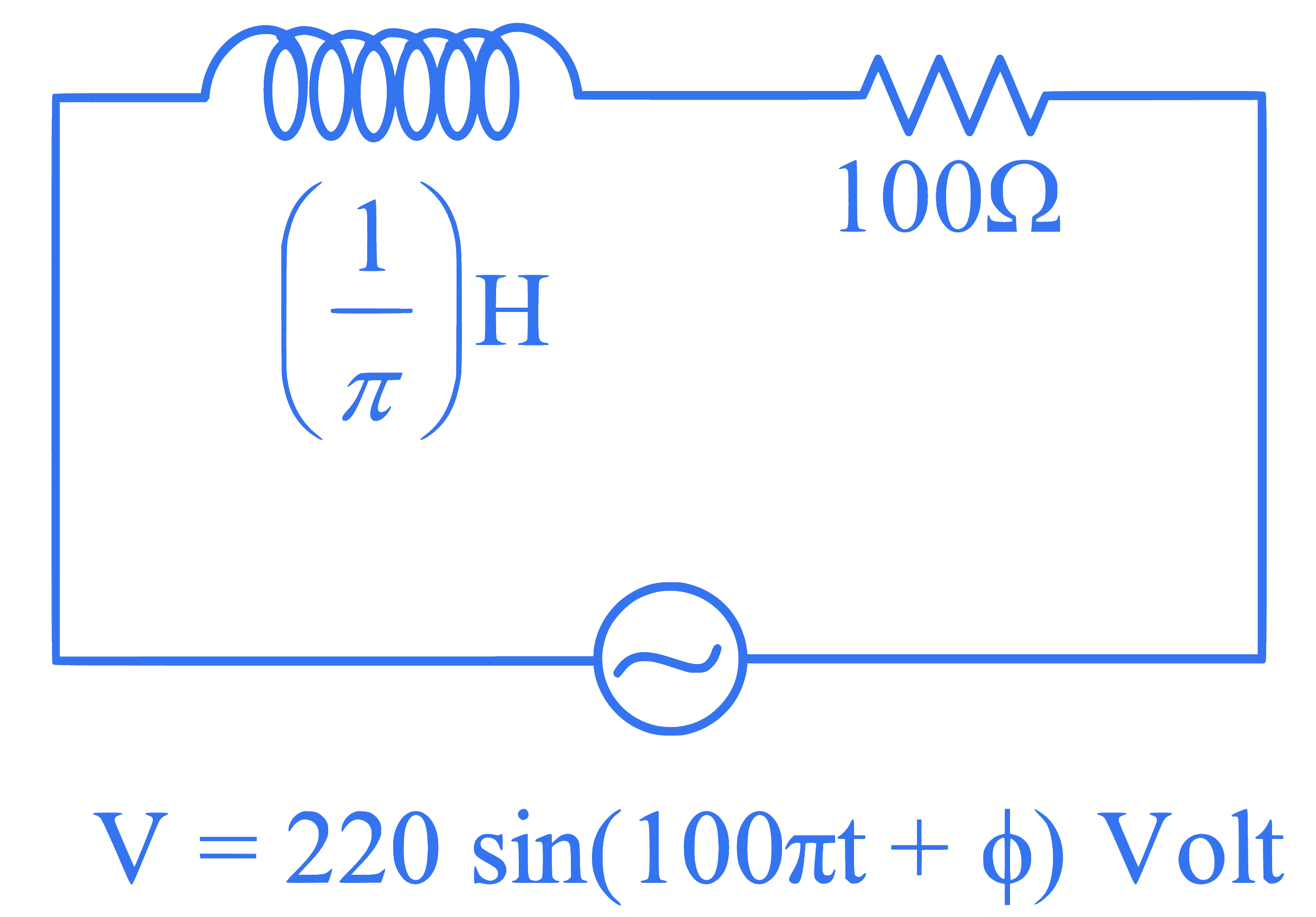

Q50.
mcq single
+4 / 1
 Which set of colours will come out in air for a situation shown in figure?
Which set of colours will come out in air for a situation shown in figure?
 Which set of colours will come out in air for a situation shown in figure?
Which set of colours will come out in air for a situation shown in figure?
Q51.
mcq single
+4 / 1
A constant torque of $$100 \mathrm{~N} \mathrm{~m}$$ turns a wheel of moment of inertia $$300 \mathrm{~kg} \mathrm{~m}^2$$ about an axis passing through its centre. Starting from rest, its angular velocity after $$3 \mathrm{~s}$$ is :-
Q52.
mcq single
+4 / 1
An emf is generated by an ac generator having 100 turn coil, of loop area $$1 \mathrm{~m}^2$$. The coil rotates at a speed of one revolution per second and placed in a uniform magnetic field of $$0.05 \mathrm{~T}$$ perpendicular to the axis of rotation of the coil. The maximum value of emf is :-
Q53.
mcq single
+4 / 1
The mechanical quantity, which has dimensions of reciprocal of mass $$(\mathrm{M}^{-1})$$ is :
Q54.
mcq single
+4 / 1
The diameter of a spherical bob, when measured with vernier callipers yielded the following values : $$3.33 \mathrm{~cm}, 3.32 \mathrm{~cm}, 3.34 \mathrm{~cm}, 3.33 \mathrm{~cm}$$ and $$3.32 \mathrm{~cm}$$. The mean diameter to appropriate significant figures is :
Q55.
mcq single
+4 / 1
For the given cycle, the work done during isobaric process is:


Q56.
mcq single
+4 / 1
A container of volume $$200 \mathrm{~cm}^3$$ contains 0.2 mole of hydrogen gas and 0.3 mole of argon gas. The pressure of the system at temperature $$200 \mathrm{~K}$$ ($$\mathrm{R}=8.3 \mathrm{JK}^{-1} \mathrm{~mol}^{-1}$$) will be :-
Q57.
mcq single
+4 / 1
Which of the following statement is not true?
Q58.
mcq single
+4 / 1
The amount of elastic potential energy per unit volume (in SI unit) of a steel wire of length $$100 \mathrm{~cm}$$ to stretch it by $$1 \mathrm{~mm}$$ is (if Young's modulus of the wire $$=2.0 \times 10^{11} \mathrm{Nm}^{-2}$$ ) :
Q59.
mcq single
+4 / 1
The viscous drag acting on a metal sphere of diameter $$1 \mathrm{~mm}$$, falling through a fluid of viscosity $$0.8 \mathrm{~Pa}$$ s with a velocity of $$2 \mathrm{~m} \mathrm{~s}^{-1}$$ is equal to :
Q60.
mcq single
+4 / 1
According to Gauss law of electrostatics, electric flux through a closed surface depends on :
Q61.
mcq single
+4 / 1
A charge $$\mathrm{Q} ~\mu \mathrm{C}$$ is placed at the centre of a cube. The flux coming out from any one of its faces will be (in SI unit) :
Q62.
mcq single
+4 / 1
If a conducting sphere of radius $$\mathrm{R}$$ is charged. Then the electric field at a distance $$\mathrm{r}(\mathrm{r} > \mathrm{R})$$ from the centre of the sphere would be, $$(\mathrm{V}=$$ potential on the surface of the sphere)
Q63.
mcq single
+4 / 1
If $$\mathrm{R}$$ is the radius of the earth and $$\mathrm{g}$$ is the acceleration due to gravity on the earth surface. Then the mean density of the earth will be :
Q64.
mcq single
+4 / 1
The escape velocity of a body on the earth surface is $$11.2 \mathrm{~km} / \mathrm{s}$$. If the same body is projected upward with velocity $$22.4 \mathrm{~km} / \mathrm{s}$$, the velocity of this body at infinite distance from the centre of the earth will be:
Q65.
mcq single
+4 / 1
The equivalent capacitance of the arrangement shown in figure is:


Q66.
mcq single
+4 / 1
To produce an instantaneous displacement current of $$2 \mathrm{~mA}$$ in the space between the parallel plates of a capacitor of capacitance $$4 ~\mu \mathrm{F}$$, the rate of change of applied variable potential difference $$\left(\frac{\mathrm{dV}}{\mathrm{dt}}\right)$$ must be :-
Q67.
mcq single
+4 / 1
$$\epsilon_0$$ and $$\mu_0$$ are the electric permittivity and magnetic permeability of free space respectively. If the corresponding quantities of a medium are $$2 ~\epsilon_0$$ and $$1.5 ~\mu_0$$ respectively, the refractive index of the medium will nearly be :
Q68.
mcq single
+4 / 1
A horizontal ray of light is incident on the right angled prism with prism angle $$6^{\circ}$$. If the refractive index of the material of the prism is 1.5 , then the angle of emergence will be:


Q69.
mcq single
+4 / 1
An object is mounted on a wall. Its image of equal size is to be obtained on a parallel wall with the help of a convex lens placed between these walls. The lens is kept at distance x in front of the second wall. The required focal length of the lens will be :
Q70.
mcq single
+4 / 1
A lens is made up of 3 different transparent media as shown in figure. A point object O is placed on its axis beyond $$2f$$. How many real images will be obtained on the other side?


Q71.
mcq single
+4 / 1
A simple pendulum oscillating in air has a period of $$\sqrt{3} \mathrm{~s}$$. If it is completely immersed in non-viscous liquid, having density $$\left(\frac{1}{4}\right)^{\text {th }}$$ of the material of the bob, the new period will be :-
Q72.
mcq single
+4 / 1
The given circuit is equivalent to:


Q73.
mcq single
+4 / 1
On the basis of electrical conductivity, which one of the following material has the smallest resistivity?
Q74.
mcq single
+4 / 1
A p-type extrinsic semiconductor is obtained when Germanium is doped with:
Q75.
mcq single
+4 / 1
A uniform electric field and a uniform magnetic field are acting along the same direction in a certain region. If an electron is projected in the region such that its velocity is pointed along the direction of fields, then the electron:
Q76.
mcq single
+4 / 1
A long straight wire of length $$2 \mathrm{~m}$$ and mass $$250 \mathrm{~g}$$ is suspended horizontally in a uniform horizontal magnetic field of $$0.7 \mathrm{~T}$$. The amount of current flowing through the wire will be $$\left(\mathrm{g}=9.8 \mathrm{~ms}^{-2}\right)$$ :
Q77.
mcq single
+4 / 1
The de Broglie wavelength associated with an electron, accelerated by a potential difference of 81 V is given by:
Q78.
mcq single
+4 / 1
The maximum kinetic energy of the emitted photoelectrons in photoelectric effect is independent of:
Q79.
mcq single
+4 / 1
The position of a particle is given by
$$\vec{r}(t)=4 t \hat{i}+2 t^2 \hat{j}+5 \hat{k}
$$
where $$\mathrm{t}$$ is in seconds and $$\mathrm{r}$$ in metre. Find the magnitude and direction of velocity $$v(t)$$, at $$t=1 \mathrm{~s}$$, with respect to $$\mathrm{x}$$-axis
Q80.
mcq single
+4 / 1
The angular momentum of an electron moving in an orbit of hydrogen atom is $$\mathrm{1.5\left(\frac{h}{\pi}\right)}$$. The energy in the same orbit is nearly.
Q81.
mcq single
+4 / 1
The ground state energy of hydrogen atom is $$-13.6 ~\mathrm{eV}$$. The energy needed to ionize hydrogen atom from its second excited state will be :
Q82.
mcq single
+4 / 1
The wavelength of Lyman series of hydrogen atom appears in:
Q83.
mcq single
+4 / 1
A ball is projected from point A with velocity $$20 \mathrm{~m} \mathrm{~s}^{-1}$$ at an angle $$60^{\circ}$$ to the horizontal direction. At the highest point $$\mathrm{B}$$ of the path (as shown in figure), the velocity $$\mathrm{v} \mathrm{m} \mathrm{s}^{-1}$$ of the ball will be:


Q84.
mcq single
+4 / 1
A particle is executing uniform circular motion with velocity $$\vec{v}$$ and acceleration $$\vec{a}$$. Which of the following is true?
Q85.
mcq single
+4 / 1
Two particles A and B initially at rest, move towards each other under mutual force of attraction. At an instance when the speed of A is v and speed of B is 3v, the speed of centre of mass is :
Q86.
mcq single
+4 / 1
A bullet of mass $$m$$ hits a block of mass $$M$$ elastically. The transfer of energy is the maximum, when :
Q87.
mcq single
+4 / 1
A $$1 \mathrm{~kg}$$ object strikes a wall with velocity $$1 \mathrm{~m} \mathrm{~s}^{-1}$$ at an angle of $$60^{\circ}$$ with the wall and reflects at the same angle. If it remains in contact with wall for $$0.1 \mathrm{~s}$$, then the force exerted on the wall is :
Q88.
mcq single
+4 / 1
The $$4^{\text {th }}$$ overtone of a closed organ pipe is same as that of $$3^{\text {rd }}$$ overtone of an open pipe. The ratio of the length of the closed pipe to the length of the open pipe is :
Q89.
mcq single
+4 / 1
A block of mass 2 kg is placed on inclined rough surface AC (as shown in figure) of coefficient of friction $$\mu$$. If g = 10 m s$$^{-2}$$, the net force (in N) on the block will be:


Q90.
mcq single
+4 / 1
The variation of susceptibility $$(\chi)$$ with absolute temperature ( $$\mathrm{T}$$ ) for a paramagnetic material is represented as :
Q91.
mcq single
+4 / 1
A particle moves with a velocity $$(5 \hat{i}-3 \hat{j}+6 \hat{k}) ~\mathrm{ms}^{-1}$$ horizontally under the action of constant force $$(10 \hat{\mathrm{i}}+10 \hat{\mathrm{j}}+20 \hat{\mathrm{k}}) \mathrm{N}$$. The instantaneous power supplied to the particle is :
Q92.
mcq single
+4 / 1
A certain wire $$\mathrm{A}$$ has resistance $$81 ~\Omega$$. The resistance of another wire $$\mathrm{B}$$ of same material and equal length but of diameter thrice the diameter of A will be :
Q93.
mcq single
+4 / 1
A copper wire of radius $$1 \mathrm{~mm}$$ contains $$10^{22}$$ free electrons per cubic metre. The drift velocity for free electrons when $$10 \mathrm{~A}$$ current flows through the wire will be (Given, charge on electron $$=1.6 \times 10^{-19} \mathrm{C}$$ ) :
Q94.
mcq single
+4 / 1
The emf of a cell having internal resistance $$1 \Omega$$ is balanced against a length of $$330 \mathrm{~cm}$$ on a potentiometer wire. When an external resistance of $$2 \Omega$$ is connected across the cell, the balancing length will be :
Q95.
mcq single
+4 / 1
If Z$$_1$$ and Z$$_2$$ are the impedances of the given circuits (a) and (b) as shown in figures, then choose the correct option


Q96.
mcq single
+4 / 1
The maximum power is dissipated for an ac in a/an:
Q97.
mcq single
+4 / 1
For very high frequencies, the effective impedance of the circuit (shown in the figure) will be:-


Q98.
mcq single
+4 / 1
An ac source is connected in the given circuit. The value of $\phi$ will be :


Biology
Biology
Q1.
mcq single
+4 / 1
The amount of nutrients such as carbon, nitrogen, potassium and calcium present in the soil at any given time is referred to as :
Q2.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(A)
Hydrarch succession
(I)
Gradual change in the species composition
(B)
Xerarch succession
(II)
Faster and climax reached quickly
(C)
Ecological succession
(III)
Lichens to mesic conditions
(D)
Secondary succession
(IV)
Phytoplankton to mesic conditions
Choose the correct answer from the options given below :
Q3.
mcq single
+4 / 1
The species of plants that plays a vital role in controlling the relative abundance of other species in a community is called _________.
Q4.
mcq single
+4 / 1
Select the correct statement/s with respect to
mechanism of sex determination in Grasshopper.
(A) It is an example of female heterogamety.
(B) Male produces two different types of gametes
either with or without X chromosome.
(C) Total number of chromosomes (autosomes and
sex chromosomes) is same in both males and females.
(D) All eggs bear an additional X chromosome
besides the autosomes.
Choose the **correct** answer from the options given
below :
Q5.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Monohybrid Cross
(I)
1 : 1
(B)
Dihybrid Cross
(II)
1 : 2 : 1
(C)
Incomplete dominance
(III)
3 : 1
(D)
Test Cross
(IV)
9 : 3 : 3 : 1
Choose the correct answer from the options given below :
Q6.
mcq single
+4 / 1
In which disorder change of single base pair in the
gene for beta globin chain results in change of
glutamic acid to valine ?
Q7.
mcq single
+4 / 1
A certain plant homozygous for yellow seeds and
red flowers was crossed with a plant homozygous
for green seeds and white flowers. The F~1~ plants had yellow seeds and pink flowers. The F~1~ plants were selfed to get F~2~ progeny. Assuming independent assortment of the two characters, how many phenotypic categories are expected for these characters in the F~2~ generation ?
Q8.
mcq single
+4 / 1
A heterozygous pea plant with violet flowers was crossed with homozygous pea plant with white flower. Violet is dominant over white. Which one of the following represents the expected combinations among 40 progenies formed?
Q9.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Columnar epithelium
(I)
Ducts of glands
(B)
Ciliated epithelium
(II)
Inner lining of stomach and intestine
(C)
Squamous epithelium
(III)
Inner lining of bronchioles
(D)
Cuboidal epithelium
(IV)
Endothelium
Choose the correct answer from the options given
below :
Q10.
mcq single
+4 / 1
The Cockroach is :
Q11.
mcq single
+4 / 1
Given below are two statements :
Statement I : In cockroach, the forewings are transparent and prothoracic in origin.
Statement II : In cockroach, the hind wings are opaque, leathery and mesothoracic in origin.
In the light of the above statements, choose the
correct answer from the options given below :
Q12.
mcq single
+4 / 1
Which out of the following statements is incorrect?
Q13.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Chlorophyll a
(I)
Yellow to yellow orange
(B)
Chlorophyll b
(II)
Yellow green
(C)
Xanthophyll
(III)
Blue green
(D)
Carotenoid
(IV)
Yellow
Choose the correct answer from the options given below :
Q14.
mcq single
+4 / 1
Given below are two statements :
Statement I : RuBisCO is the most abundant enzyme in the world.
Statement II : Photorespiration does not occur in C$$_4$$ plants.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q15.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Contractile vacuole
(I)
Asterias
(B)
Water vascular system
(II)
Amoeba
(C)
Canal system
(III)
Spongilla
(D)
Flame cells
(IV)
Taenia
Choose the correct answer from the options given
below :
Q16.
mcq single
+4 / 1
Select the correct statements :
(A) Platyhelminthes are triploblastic
pseudocoelomate and bilaterally symmetrical
organisms.
(B) Ctenophores reproduce only sexually and
fertilization is external.
(C) In tapeworm, fertilization is internal but sexes
are not separate.
(D) Ctenophores are exclusively marine, diploblastic
and bioluminescent organisms.
(E) In sponges, fertilization is external and
development is direct.
Choose the correct answer from the options given
below :
Q17.
mcq single
+4 / 1
Which of the following statements is **true**?
Q18.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Pteropsida
(I)
Psilotum
(B)
Lycopsida
(II)
Equisetum
(C)
Psilopsida
(III)
Adiantum
(D)
Sphenopsida
(IV)
Selaginella
Choose the correct answer from the options given below :
Q19.
mcq single
+4 / 1
Which classes of algae possess pigment fucoxanthin
and pigment phycoerythrin, respectively ?
Q20.
mcq single
+4 / 1
Select the sequence of steps in Respiration.
(A) Diffusion of gases (O~2~ and CO~2~) across alveolar
membrane.
(B) Diffusion of O~2~ and CO~2~ between blood and tissues.
(C) Transport of gases by the blood
(D) Pulmonary ventilation by which atmospheric air
is drawn in and CO~2~ rich alveolar air is released out.
(E) Utilisation of O~2~ by the cells for catabolic reactions are resultant release of CO~2~
Choose the correct answer from the options given
below :
Q21.
mcq single
+4 / 1
Given below are two statements : one is labelled as
Assertion (A) and the other is labelled as Reason
(R).
Assertion (A) : A person goes to high altitude and experiences **"Altitude Sickness"** with symptoms like breathing difficulty and heart palpitations.
Reason (R) : Due to low atmospheric pressure at high altitude, the body does not get sufficient oxygen.
In the light of the above statements, choose the
correct answer from the options given below :
Q22.
mcq single
+4 / 1
Which of the following statement is incorrect about Agrobacterium tumifaciens?
Q23.
mcq single
+4 / 1
Which of the following can act as molecular
scissors?
Q24.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Gene therapy
(I)
Separation of DNA fragments
(B)
RNA interference
(II)
Diagnostic test for AIDS
(C)
ELISA
(III)
Cellular defence
(D)
Gel Electrophoresis
(IV)
Allows correction of a gene defect.
Choose the correct answer from the options given
below :
Q25.
mcq single
+4 / 1
Brainstem of human brain consists of :
Q26.
mcq single
+4 / 1
Given below are two statements :
Statement I :- The nose contains mucus – coated receptors which are specialised for receiving the sense of smell and are called olfactory receptors.
Statement II :- Wall of the eye ball has three layers. The external layer is called choroid (dense connective tissue), middle layer is sclera (thin pigmented layer) and
internal layer is retina (ganglion cells, bipolar cells and photoreceptor cells).
In the light of the above statements, choose the
correct answer from the options given below:
Q27.
mcq single
+4 / 1
The transverse section of a plant part showed polyarch, radial and exarch xylem, with endodermis and pericycle. The plant part is identified as :
Q28.
mcq single
+4 / 1
Consider the following tissues in the stelar region of a stem showing secondary growth.
(A) Primary xylem
(B) Secondary xylem
(C) Primary phloem
(D) Secondary phloem
Arrange these in the correct sequence of their position from pith towards corts.
Q29.
mcq single
+4 / 1
Consider the following plant tissues :
(A) Axillary buds
(B) Fascicular vascular cambium
(C) Interfascicular cambium
(D) Cork cambium
(E) Intercalary meristem
Identify the lateral meristems among the above.
Q30.
mcq single
+4 / 1
Given below are two statements :
Statement I :- In bacteria, the mesosomes are formed by the extensions of plasma membrane.
Statement II :- The mesosomes, in bacteria, help in DNA replication and cell wall formation.
In the light of the above statements, choose the
most appropriate answer from the options given
below:
Q31.
mcq single
+4 / 1
Which of the following statements are correct with
respect of Golgi apparatus ?
(A) It is the important site of formation of
glycoprotein and glycolipids.
(B) It produces cellular energy in the form of ATP.
(C) It modifies the protein synthesized by ribosomes
on ER.
(D) It facilitates the transport of ions.
(E) It provides mechanical support.
Choose the most appropriate answer from the
options given below :
Q32.
mcq single
+4 / 1
Name the component that binds to the operator region of an operon and prevents RNA polymerase from transcribing the operon.
Q33.
mcq single
+4 / 1
Which one of the following acts as an inducer for
lac operon ?
Q34.
mcq single
+4 / 1
The last chromosome sequenced in Human Genome Project was:
Q35.
mcq single
+4 / 1
The salient features of genetic code are :
(A) The code is palindromic
(B) UGA act as initiator codon
(C) The code is unambiguous and specific
(D) The code is nearly universal
Choose the most appropriate answer from the
options given below :
Q36.
mcq single
+4 / 1
With reference to Hershey and Chase experiments.
Select the correct statements.
(A) Viruses grown in the presence of radioactive
phosphorus contained radioactive DNA.
(B) Viruses grown on radioactive sulphur contained
radioactive proteins.
(C) Viruses grown on radioactive phosphorus
contained radioactive protein.
(D) Viruses grown on radioactive sulphur contained
radioactive DNA.
(E) Viruses grown on radioactive protein contained
radioactive DNA.
Choose the most appropriate answer from the
options given below :
Q37.
mcq single
+4 / 1
Given below are two statements :
Statement I : RNA being unstable, mutate at a faster rate.
Statement II : RNA can directly code for synthesis of proteins hence can easily express the characters.
In the light of the above statements, choose the
correct answer from the options given below :
Q38.
mcq single
+4 / 1
Select the correct statements about sickle cell
anaemia.
(A) There is a change in gene for beta globin.
(B) In the beta globin, there is valine in the place of
Lysine.
(C) It is an example of point mutation.
(D) In the normal gene U is replaced by A.
Choose the correct answer from the options given
below :
Q39.
mcq single
+4 / 1
Which scientist conducted an experiment with ^(32)P
and ^(35)S labelled phages for demonstrating that DNA is the genetic material?
Q40.
mcq single
+4 / 1
Given below are two statements :
Statement I : The process of copying genetic information from one strand of the DNA into RNA is termed as transcription.
Statement II : A transcription unit in DNA is defined primarily by the three regions in the DNA i.e., a promotor, the structural gene and a terminator.
In the light of the above statements, choose the
correct answer from the options given below :
Q41.
mcq single
+4 / 1
Which of the following sexually transmitted
infections are completely curable ?
Q42.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Typhoid
(I)
Protozoan
(B)
Elephantiasis
(II)
Salmonella
(C)
Ringworm
(III)
Aschelminthes
(D)
Malaria
(IV)
Microsporum
Choose the correct answer from the options given
below :
Q43.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Cytokine barriers
(I)
Mucus coating of respiratory tract
(B)
Cellular barriers
(II)
Interferons
(C)
Physiological barriers
(III)
Neutrophils and Macrophages
(D)
Physical barriers
(IV)
Tears and Saliva
Choose the correct answer from the options given
below :
Q44.
mcq single
+4 / 1
Plants offer rewards to animals in the form of pollen and nectar and the animals facilitate the pollination process. This is an example of :
Q45.
mcq single
+4 / 1
If there are 250 snails in a pond, and within a year
their number increases to 2500 by reproduction.
What should be their birth rate per snail per year ?
Q46.
mcq single
+4 / 1
Match List-I with List-II.
List - I
(ECG)
List - II
(Electrical activity of heart)
(A)
P-wave
(I)
Depolarisation of ventricles
(B)
QRS complex
(II)
End of systole
(C)
T wave
(III)
Depolarisation of atria
(D)
End of T wave
(IV)
Repolarisation of ventricles
Choose the correct answer from the options given
below :
Q47.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Eosinophils
(I)
6 - 8%
(B)
Lymphocytes
(II)
2 - 3%
(C)
Neutrophils
(III)
20 - 25%
(D)
Monocytes
(IV)
60 - 65%
Choose the correct answer from the options given
below :
Q48.
mcq single
+4 / 1
The dissolution of synaptonemal complex occurs during :
Q49.
mcq single
+4 / 1
During which stages of mitosis and meiosis,
respectively does the centromere of each
chromosome split ?
Q50.
mcq single
+4 / 1
Doubling of the number of chromosomes can be achieved by disrupting mitotic cell division soon after :
Q51.
mcq single
+4 / 1
Select correct sequence of substages of Prophase-I
of Meiotic division :
(A) Zygotene
(B) Pachytene
(C) Diakinesis
(D) Leptotene
(E) Diplotene
Choose the correct answer from the options given
below :
Q52.
mcq single
+4 / 1
Which one of the following is the quiescent stage of
cell cycle?
Q53.
mcq single
+4 / 1
Given below are two statements regarding
oogenesis:
Statement I :- The primary follicles get surrounded by more layers of granulosa cells, a theca and shows fluid filled cavity antrum. Now it is called secondary follicle.
Statement II :- Graffian follicle ruptures to release the secondary oocyte from the ovary by the process called
ovulation.
In the light of the above statements, choose the
correct answer from the options given below:
Q54.
mcq single
+4 / 1
Which of the following statements are correct with
respect to the hormone and its function?
(A) Thyrocalcitonin (TCT) regulates the blood
calcium level.
(B) In males, FSH and androgens regulate
spermatogenesis.
(C) Hyperthyroidism can lead to goitre.
(D) Glucocorticoids are secreted in Adrenal Medulla.
(E) Parathyroid hormone is regulated by circulating
levels of sodium ions.
Choose the most appropriate answer from the
options given below :
Q55.
mcq single
+4 / 1
Given below are two statements :
Statement I : Parathyroid hormone acts on bones and stimulates the process of bone resorption.
Statement II : Parathyroid hormone along with Thyrocalcitonin plays a significant role in carbohydrate metabolism.
In the light of the above statements, choose the
correct answer from the options given below :
Q56.
mcq single
+4 / 1
Select incorrect statement, regarding chemical
structure of insulin.
Q57.
mcq single
+4 / 1
For chemical defence against herbivores, Calotropis
has :
Q58.
mcq single
+4 / 1
In 'rivet popper hypothesis', Paul Ehrlich compared the rivets in an airplane to :
Q59.
mcq single
+4 / 1
Which of the following is/are cause(s) of biodiversity
losses ?
Q60.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Auxin
(I)
Promotes female flower formation in cucumber
(B)
Gibberellin
(II)
Overcoming apical dominance
(C)
Cytokinin
(III)
Increase in the length of grape stalks
(D)
Ethylene
(IV)
Promotes flowering in pineapple
Choose the **correct** answer from the options given below :
Q61.
mcq single
+4 / 1
Which of the following statements is not correct ?
Q62.
mcq single
+4 / 1
The phenomenon which is influenced by auxin and also played a major role in its discovery :
Q63.
mcq single
+4 / 1
How many times decarboxylation occurs during each TCA cycle?
Q64.
mcq single
+4 / 1
Fatty acids are connected with the respiratory pathway through :
Q65.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Palm bones
(I)
Phalanges
(B)
Wrist bones
(II)
Metacarpals
(C)
Ankle bones
(III)
Carpals
(D)
Digit bones
(IV)
Tarsals
Choose the correct answer from the options given
below :
Q66.
mcq single
+4 / 1
'X' and 'Y' are the components of Binomial
nomenclature. This naming system was proposed by
'Z' :
Q67.
mcq single
+4 / 1
House fly belongs to _____ family.
Q68.
mcq single
+4 / 1
Identify the fossil of man who showed the following
characteristics.
(A) Brain capacity of 1400 cc
(B) Used hides to protect their body
(C) Buried their dead bodies
In the light of above statements, choose the correct
answer from the options given below :
Q69.
mcq single
+4 / 1
Arrange the events of Renin - Angiotensin
mechanism in correct sequence.
(A) Activation of JG cells and release of renin.
(B) Angiotensin II activates release of aldosterone.
(C) Fall in glomerular blood pressure.
(D) Reabsorption of Na^(+) and water from distal
convoluted tubule.
(E) Angiotensinogen is converted to Angiotensin I
and then to Angiotensin II.
Choose the correct answer from the options given
below :
Q70.
mcq single
+4 / 1
Given below are two statements :
Statement I :- Goblet cells are unicellular glands.
Statement II :- Earwax is the secretion of exocrine gland.
In the light of the above statements, choose the
correct answer from the options given below:
Q71.
mcq single
+4 / 1
Given below are two statements : one is labelled as
Assertion (A) and the other is labelled as Reason
(R)
Assertion (A) : Ascending limb of loop of Henle is impermeable to water and allows transport of electrolytes actively or passively.
Reason (R) : Dilution of filtrate takes place due to efflux of electrolytes in the medullary fluid.
In the light of the above statements, choose the
correct answer from the options given below :
Q72.
mcq single
+4 / 1
Which of the following statements are correct ?
(A) Reproductive health refers to total well-being in
all aspects of reproduction.
(B) Amniocentesis is legally banned for sex
determination in India.
(C) "Saheli" – a new oral contraceptive for females
was developed in collaboration with ICMR (New
Delhi).
(D) Amniocentesis is used to determine genetic
disorders and survivability of foetus.
Choose the most appropriate answer from the
options given below :
Q73.
mcq single
+4 / 1
Given below are two statements :
Statement I :- Intra Cytoplasmic Sperm Injection (ICSI) is another specialised procedure of in-vivo fertilisation.
Statement II :- Infertility cases due to inability of the male partner to inseminate female can be corrected by artificial insemination (AI).
In the light of the above statements, choose the
correct answer from the options given below:
Q74.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Non-medicated IUDs
(I)
Multiload 375
(B)
Copper releasing IUDs
(II)
Rubber barrier
(C)
Hormone releasing IUDs
(III)
Lippes loop
(D)
Vaults
(IV)
LNG-20
Choose the correct answer from the options given
below :
Q75.
mcq single
+4 / 1
In which of the following sets of families, the pollen grains are viable for months?
Q76.
mcq single
+4 / 1
In angiosperms the correct sequence of events in formation of female gametophyte in the ovule is :
(A) 3 successive free nuclear divisions functional megaspore.
(B) Degeneration of 3 megaspores.
(C) Meiotic division in megaspore mother cell.
(D) Migration of 3 nuclei towards each pole.
(E) Formation of wall resulting in seven celled embryosac.
Choose the correct answer from the options given below :
Q77.
mcq single
+4 / 1
Transfer of pollen grains from anther to stigma of another flower of same plant is known as :
Q78.
mcq single
+4 / 1
Match the following :
Type of flower
Example
(A)
Zygomorphic
(I)
Mustard
(B)
Hypogynous
(II)
Plum
(C)
Perigynous
(III)
Cassia
(D)
Epigynous
(IV)
Cucumber
Select the correct option :
Q79.
mcq single
+4 / 1
In Calotropis, aestivation is :
Q80.
mcq single
+4 / 1
In a pea flower, five petals are arranged in a specialized manner with one posterior, two lateral and two anterior. These are named as ___________, __________ and _________ respectively.
Q81.
mcq single
+4 / 1
Inulin is a polymer of :
Q82.
mcq single
+4 / 1
Which of the following is not a secondary
metabolite?
Q83.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(A)
Protein
(I)
C=C double bonds
(B)
Unsaturated fatty acid
(II)
Phosphodiester bond
(C)
Nucleic acid
(III)
Glycosidic bonds
(D)
Polysaccharide
(IV)
Peptide bonds
Choose the **correct** answer from the options given below :
Q84.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Terpenoides
(I)
Codeine
(B)
Lectins
(II)
Diterpenes
(C)
Alkaloids
(III)
Ricin
(D)
Toxins
(IV)
Concanavalin A
Choose the correct answer from the options given
below :
Q85.
mcq single
+4 / 1
Thermostable DNA polymerase used in PCR was isolated from :
Q86.
mcq single
+4 / 1
Ligation of foreign DNA at which of the following site will result in loss of tetracyclin resistance of pBR322 :
Q87.
mcq single
+4 / 1
Match List I with List II.
List - I
List - II
(A)
Kanamycin
(I)
Delivers genes into animal cells
(B)
ClaI
(II)
Selectable marker
(C)
Disarmed retroviruses
(III)
Restriction site
(D)
Kanamycin $$^\mathrm{R}$$gene
(IV)
Antibiotic resistance
Choose the **correct** answer from the options given below :
Q88.
mcq single
+4 / 1
The amount of nutrients such as carbon, nitrogen, potassium and calcium present in the soil at any given time is referred to as :
Q89.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(A)
Hydrarch succession
(I)
Gradual change in the species composition
(B)
Xerarch succession
(II)
Faster and climax reached quickly
(C)
Ecological succession
(III)
Lichens to mesic conditions
(D)
Secondary succession
(IV)
Phytoplankton to mesic conditions
Choose the correct answer from the options given below :
Q90.
mcq single
+4 / 1
The species of plants that plays a vital role in controlling the relative abundance of other species in a community is called _________.
Q91.
mcq single
+4 / 1
Select the correct statement/s with respect to
mechanism of sex determination in Grasshopper.
(A) It is an example of female heterogamety.
(B) Male produces two different types of gametes
either with or without X chromosome.
(C) Total number of chromosomes (autosomes and
sex chromosomes) is same in both males and females.
(D) All eggs bear an additional X chromosome
besides the autosomes.
Choose the **correct** answer from the options given
below :
Q92.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Monohybrid Cross
(I)
1 : 1
(B)
Dihybrid Cross
(II)
1 : 2 : 1
(C)
Incomplete dominance
(III)
3 : 1
(D)
Test Cross
(IV)
9 : 3 : 3 : 1
Choose the correct answer from the options given below :
Q93.
mcq single
+4 / 1
In which disorder change of single base pair in the
gene for beta globin chain results in change of
glutamic acid to valine ?
Q94.
mcq single
+4 / 1
A certain plant homozygous for yellow seeds and
red flowers was crossed with a plant homozygous
for green seeds and white flowers. The F~1~ plants had yellow seeds and pink flowers. The F~1~ plants were selfed to get F~2~ progeny. Assuming independent assortment of the two characters, how many phenotypic categories are expected for these characters in the F~2~ generation ?
Q95.
mcq single
+4 / 1
A heterozygous pea plant with violet flowers was crossed with homozygous pea plant with white flower. Violet is dominant over white. Which one of the following represents the expected combinations among 40 progenies formed?
Q96.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Columnar epithelium
(I)
Ducts of glands
(B)
Ciliated epithelium
(II)
Inner lining of stomach and intestine
(C)
Squamous epithelium
(III)
Inner lining of bronchioles
(D)
Cuboidal epithelium
(IV)
Endothelium
Choose the correct answer from the options given
below :
Q97.
mcq single
+4 / 1
The Cockroach is :
Q98.
mcq single
+4 / 1
Given below are two statements :
Statement I : In cockroach, the forewings are transparent and prothoracic in origin.
Statement II : In cockroach, the hind wings are opaque, leathery and mesothoracic in origin.
In the light of the above statements, choose the
correct answer from the options given below :
Q99.
mcq single
+4 / 1
Which out of the following statements is incorrect?
Q100.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Chlorophyll a
(I)
Yellow to yellow orange
(B)
Chlorophyll b
(II)
Yellow green
(C)
Xanthophyll
(III)
Blue green
(D)
Carotenoid
(IV)
Yellow
Choose the correct answer from the options given below :
Q101.
mcq single
+4 / 1
Given below are two statements :
Statement I : RuBisCO is the most abundant enzyme in the world.
Statement II : Photorespiration does not occur in C$$_4$$ plants.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q102.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Contractile vacuole
(I)
Asterias
(B)
Water vascular system
(II)
Amoeba
(C)
Canal system
(III)
Spongilla
(D)
Flame cells
(IV)
Taenia
Choose the correct answer from the options given
below :
Q103.
mcq single
+4 / 1
Select the correct statements :
(A) Platyhelminthes are triploblastic
pseudocoelomate and bilaterally symmetrical
organisms.
(B) Ctenophores reproduce only sexually and
fertilization is external.
(C) In tapeworm, fertilization is internal but sexes
are not separate.
(D) Ctenophores are exclusively marine, diploblastic
and bioluminescent organisms.
(E) In sponges, fertilization is external and
development is direct.
Choose the correct answer from the options given
below :
Q104.
mcq single
+4 / 1
Which of the following statements is **true**?
Q105.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Pteropsida
(I)
Psilotum
(B)
Lycopsida
(II)
Equisetum
(C)
Psilopsida
(III)
Adiantum
(D)
Sphenopsida
(IV)
Selaginella
Choose the correct answer from the options given below :
Q106.
mcq single
+4 / 1
Which classes of algae possess pigment fucoxanthin
and pigment phycoerythrin, respectively ?
Q107.
mcq single
+4 / 1
Select the sequence of steps in Respiration.
(A) Diffusion of gases (O~2~ and CO~2~) across alveolar
membrane.
(B) Diffusion of O~2~ and CO~2~ between blood and tissues.
(C) Transport of gases by the blood
(D) Pulmonary ventilation by which atmospheric air
is drawn in and CO~2~ rich alveolar air is released out.
(E) Utilisation of O~2~ by the cells for catabolic reactions are resultant release of CO~2~
Choose the correct answer from the options given
below :
Q108.
mcq single
+4 / 1
Given below are two statements : one is labelled as
Assertion (A) and the other is labelled as Reason
(R).
Assertion (A) : A person goes to high altitude and experiences **"Altitude Sickness"** with symptoms like breathing difficulty and heart palpitations.
Reason (R) : Due to low atmospheric pressure at high altitude, the body does not get sufficient oxygen.
In the light of the above statements, choose the
correct answer from the options given below :
Q109.
mcq single
+4 / 1
Which of the following statement is incorrect about Agrobacterium tumifaciens?
Q110.
mcq single
+4 / 1
Which of the following can act as molecular
scissors?
Q111.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Gene therapy
(I)
Separation of DNA fragments
(B)
RNA interference
(II)
Diagnostic test for AIDS
(C)
ELISA
(III)
Cellular defence
(D)
Gel Electrophoresis
(IV)
Allows correction of a gene defect.
Choose the correct answer from the options given
below :
Q112.
mcq single
+4 / 1
Brainstem of human brain consists of :
Q113.
mcq single
+4 / 1
Given below are two statements :
Statement I :- The nose contains mucus – coated receptors which are specialised for receiving the sense of smell and are called olfactory receptors.
Statement II :- Wall of the eye ball has three layers. The external layer is called choroid (dense connective tissue), middle layer is sclera (thin pigmented layer) and
internal layer is retina (ganglion cells, bipolar cells and photoreceptor cells).
In the light of the above statements, choose the
correct answer from the options given below:
Q114.
mcq single
+4 / 1
The transverse section of a plant part showed polyarch, radial and exarch xylem, with endodermis and pericycle. The plant part is identified as :
Q115.
mcq single
+4 / 1
Consider the following tissues in the stelar region of a stem showing secondary growth.
(A) Primary xylem
(B) Secondary xylem
(C) Primary phloem
(D) Secondary phloem
Arrange these in the correct sequence of their position from pith towards corts.
Q116.
mcq single
+4 / 1
Consider the following plant tissues :
(A) Axillary buds
(B) Fascicular vascular cambium
(C) Interfascicular cambium
(D) Cork cambium
(E) Intercalary meristem
Identify the lateral meristems among the above.
Q117.
mcq single
+4 / 1
Given below are two statements :
Statement I :- In bacteria, the mesosomes are formed by the extensions of plasma membrane.
Statement II :- The mesosomes, in bacteria, help in DNA replication and cell wall formation.
In the light of the above statements, choose the
most appropriate answer from the options given
below:
Q118.
mcq single
+4 / 1
Which of the following statements are correct with
respect of Golgi apparatus ?
(A) It is the important site of formation of
glycoprotein and glycolipids.
(B) It produces cellular energy in the form of ATP.
(C) It modifies the protein synthesized by ribosomes
on ER.
(D) It facilitates the transport of ions.
(E) It provides mechanical support.
Choose the most appropriate answer from the
options given below :
Q119.
mcq single
+4 / 1
Name the component that binds to the operator region of an operon and prevents RNA polymerase from transcribing the operon.
Q120.
mcq single
+4 / 1
Which one of the following acts as an inducer for
lac operon ?
Q121.
mcq single
+4 / 1
The last chromosome sequenced in Human Genome Project was:
Q122.
mcq single
+4 / 1
The salient features of genetic code are :
(A) The code is palindromic
(B) UGA act as initiator codon
(C) The code is unambiguous and specific
(D) The code is nearly universal
Choose the most appropriate answer from the
options given below :
Q123.
mcq single
+4 / 1
With reference to Hershey and Chase experiments.
Select the correct statements.
(A) Viruses grown in the presence of radioactive
phosphorus contained radioactive DNA.
(B) Viruses grown on radioactive sulphur contained
radioactive proteins.
(C) Viruses grown on radioactive phosphorus
contained radioactive protein.
(D) Viruses grown on radioactive sulphur contained
radioactive DNA.
(E) Viruses grown on radioactive protein contained
radioactive DNA.
Choose the most appropriate answer from the
options given below :
Q124.
mcq single
+4 / 1
Given below are two statements :
Statement I : RNA being unstable, mutate at a faster rate.
Statement II : RNA can directly code for synthesis of proteins hence can easily express the characters.
In the light of the above statements, choose the
correct answer from the options given below :
Q125.
mcq single
+4 / 1
Select the correct statements about sickle cell
anaemia.
(A) There is a change in gene for beta globin.
(B) In the beta globin, there is valine in the place of
Lysine.
(C) It is an example of point mutation.
(D) In the normal gene U is replaced by A.
Choose the correct answer from the options given
below :
Q126.
mcq single
+4 / 1
Which scientist conducted an experiment with ^(32)P
and ^(35)S labelled phages for demonstrating that DNA is the genetic material?
Q127.
mcq single
+4 / 1
Given below are two statements :
Statement I : The process of copying genetic information from one strand of the DNA into RNA is termed as transcription.
Statement II : A transcription unit in DNA is defined primarily by the three regions in the DNA i.e., a promotor, the structural gene and a terminator.
In the light of the above statements, choose the
correct answer from the options given below :
Q128.
mcq single
+4 / 1
Which of the following sexually transmitted
infections are completely curable ?
Q129.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Typhoid
(I)
Protozoan
(B)
Elephantiasis
(II)
Salmonella
(C)
Ringworm
(III)
Aschelminthes
(D)
Malaria
(IV)
Microsporum
Choose the correct answer from the options given
below :
Q130.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Cytokine barriers
(I)
Mucus coating of respiratory tract
(B)
Cellular barriers
(II)
Interferons
(C)
Physiological barriers
(III)
Neutrophils and Macrophages
(D)
Physical barriers
(IV)
Tears and Saliva
Choose the correct answer from the options given
below :
Q131.
mcq single
+4 / 1
Plants offer rewards to animals in the form of pollen and nectar and the animals facilitate the pollination process. This is an example of :
Q132.
mcq single
+4 / 1
If there are 250 snails in a pond, and within a year
their number increases to 2500 by reproduction.
What should be their birth rate per snail per year ?
Q133.
mcq single
+4 / 1
Match List-I with List-II.
List - I
(ECG)
List - II
(Electrical activity of heart)
(A)
P-wave
(I)
Depolarisation of ventricles
(B)
QRS complex
(II)
End of systole
(C)
T wave
(III)
Depolarisation of atria
(D)
End of T wave
(IV)
Repolarisation of ventricles
Choose the correct answer from the options given
below :
Q134.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Eosinophils
(I)
6 - 8%
(B)
Lymphocytes
(II)
2 - 3%
(C)
Neutrophils
(III)
20 - 25%
(D)
Monocytes
(IV)
60 - 65%
Choose the correct answer from the options given
below :
Q135.
mcq single
+4 / 1
The dissolution of synaptonemal complex occurs during :
Q136.
mcq single
+4 / 1
During which stages of mitosis and meiosis,
respectively does the centromere of each
chromosome split ?
Q137.
mcq single
+4 / 1
Doubling of the number of chromosomes can be achieved by disrupting mitotic cell division soon after :
Q138.
mcq single
+4 / 1
Select correct sequence of substages of Prophase-I
of Meiotic division :
(A) Zygotene
(B) Pachytene
(C) Diakinesis
(D) Leptotene
(E) Diplotene
Choose the correct answer from the options given
below :
Q139.
mcq single
+4 / 1
Which one of the following is the quiescent stage of
cell cycle?
Q140.
mcq single
+4 / 1
Given below are two statements regarding
oogenesis:
Statement I :- The primary follicles get surrounded by more layers of granulosa cells, a theca and shows fluid filled cavity antrum. Now it is called secondary follicle.
Statement II :- Graffian follicle ruptures to release the secondary oocyte from the ovary by the process called
ovulation.
In the light of the above statements, choose the
correct answer from the options given below:
Q141.
mcq single
+4 / 1
Which of the following statements are correct with
respect to the hormone and its function?
(A) Thyrocalcitonin (TCT) regulates the blood
calcium level.
(B) In males, FSH and androgens regulate
spermatogenesis.
(C) Hyperthyroidism can lead to goitre.
(D) Glucocorticoids are secreted in Adrenal Medulla.
(E) Parathyroid hormone is regulated by circulating
levels of sodium ions.
Choose the most appropriate answer from the
options given below :
Q142.
mcq single
+4 / 1
Given below are two statements :
Statement I : Parathyroid hormone acts on bones and stimulates the process of bone resorption.
Statement II : Parathyroid hormone along with Thyrocalcitonin plays a significant role in carbohydrate metabolism.
In the light of the above statements, choose the
correct answer from the options given below :
Q143.
mcq single
+4 / 1
Select incorrect statement, regarding chemical
structure of insulin.
Q144.
mcq single
+4 / 1
For chemical defence against herbivores, Calotropis
has :
Q145.
mcq single
+4 / 1
In 'rivet popper hypothesis', Paul Ehrlich compared the rivets in an airplane to :
Q146.
mcq single
+4 / 1
Which of the following is/are cause(s) of biodiversity
losses ?
Q147.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Auxin
(I)
Promotes female flower formation in cucumber
(B)
Gibberellin
(II)
Overcoming apical dominance
(C)
Cytokinin
(III)
Increase in the length of grape stalks
(D)
Ethylene
(IV)
Promotes flowering in pineapple
Choose the **correct** answer from the options given below :
Q148.
mcq single
+4 / 1
Which of the following statements is not correct ?
Q149.
mcq single
+4 / 1
The phenomenon which is influenced by auxin and also played a major role in its discovery :
Q150.
mcq single
+4 / 1
How many times decarboxylation occurs during each TCA cycle?
Q151.
mcq single
+4 / 1
Fatty acids are connected with the respiratory pathway through :
Q152.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Palm bones
(I)
Phalanges
(B)
Wrist bones
(II)
Metacarpals
(C)
Ankle bones
(III)
Carpals
(D)
Digit bones
(IV)
Tarsals
Choose the correct answer from the options given
below :
Q153.
mcq single
+4 / 1
'X' and 'Y' are the components of Binomial
nomenclature. This naming system was proposed by
'Z' :
Q154.
mcq single
+4 / 1
House fly belongs to _____ family.
Q155.
mcq single
+4 / 1
Identify the fossil of man who showed the following
characteristics.
(A) Brain capacity of 1400 cc
(B) Used hides to protect their body
(C) Buried their dead bodies
In the light of above statements, choose the correct
answer from the options given below :
Q156.
mcq single
+4 / 1
Arrange the events of Renin - Angiotensin
mechanism in correct sequence.
(A) Activation of JG cells and release of renin.
(B) Angiotensin II activates release of aldosterone.
(C) Fall in glomerular blood pressure.
(D) Reabsorption of Na^(+) and water from distal
convoluted tubule.
(E) Angiotensinogen is converted to Angiotensin I
and then to Angiotensin II.
Choose the correct answer from the options given
below :
Q157.
mcq single
+4 / 1
Given below are two statements :
Statement I :- Goblet cells are unicellular glands.
Statement II :- Earwax is the secretion of exocrine gland.
In the light of the above statements, choose the
correct answer from the options given below:
Q158.
mcq single
+4 / 1
Given below are two statements : one is labelled as
Assertion (A) and the other is labelled as Reason
(R)
Assertion (A) : Ascending limb of loop of Henle is impermeable to water and allows transport of electrolytes actively or passively.
Reason (R) : Dilution of filtrate takes place due to efflux of electrolytes in the medullary fluid.
In the light of the above statements, choose the
correct answer from the options given below :
Q159.
mcq single
+4 / 1
Which of the following statements are correct ?
(A) Reproductive health refers to total well-being in
all aspects of reproduction.
(B) Amniocentesis is legally banned for sex
determination in India.
(C) "Saheli" – a new oral contraceptive for females
was developed in collaboration with ICMR (New
Delhi).
(D) Amniocentesis is used to determine genetic
disorders and survivability of foetus.
Choose the most appropriate answer from the
options given below :
Q160.
mcq single
+4 / 1
Given below are two statements :
Statement I :- Intra Cytoplasmic Sperm Injection (ICSI) is another specialised procedure of in-vivo fertilisation.
Statement II :- Infertility cases due to inability of the male partner to inseminate female can be corrected by artificial insemination (AI).
In the light of the above statements, choose the
correct answer from the options given below:
Q161.
mcq single
+4 / 1
Match List - I with List - II.
List - I
List - II
(A)
Non-medicated IUDs
(I)
Multiload 375
(B)
Copper releasing IUDs
(II)
Rubber barrier
(C)
Hormone releasing IUDs
(III)
Lippes loop
(D)
Vaults
(IV)
LNG-20
Choose the correct answer from the options given
below :
Q162.
mcq single
+4 / 1
In which of the following sets of families, the pollen grains are viable for months?
Q163.
mcq single
+4 / 1
In angiosperms the correct sequence of events in formation of female gametophyte in the ovule is :
(A) 3 successive free nuclear divisions functional megaspore.
(B) Degeneration of 3 megaspores.
(C) Meiotic division in megaspore mother cell.
(D) Migration of 3 nuclei towards each pole.
(E) Formation of wall resulting in seven celled embryosac.
Choose the correct answer from the options given below :
Q164.
mcq single
+4 / 1
Transfer of pollen grains from anther to stigma of another flower of same plant is known as :
Q165.
mcq single
+4 / 1
Match the following :
Type of flower
Example
(A)
Zygomorphic
(I)
Mustard
(B)
Hypogynous
(II)
Plum
(C)
Perigynous
(III)
Cassia
(D)
Epigynous
(IV)
Cucumber
Select the correct option :
Q166.
mcq single
+4 / 1
In Calotropis, aestivation is :
Q167.
mcq single
+4 / 1
In a pea flower, five petals are arranged in a specialized manner with one posterior, two lateral and two anterior. These are named as ___________, __________ and _________ respectively.
Q168.
mcq single
+4 / 1
Inulin is a polymer of :
Q169.
mcq single
+4 / 1
Which of the following is not a secondary
metabolite?
Q170.
mcq single
+4 / 1
Match List - I with List - II
List - I
List - II
(A)
Protein
(I)
C=C double bonds
(B)
Unsaturated fatty acid
(II)
Phosphodiester bond
(C)
Nucleic acid
(III)
Glycosidic bonds
(D)
Polysaccharide
(IV)
Peptide bonds
Choose the **correct** answer from the options given below :
Q171.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Terpenoides
(I)
Codeine
(B)
Lectins
(II)
Diterpenes
(C)
Alkaloids
(III)
Ricin
(D)
Toxins
(IV)
Concanavalin A
Choose the correct answer from the options given
below :
Q172.
mcq single
+4 / 1
Thermostable DNA polymerase used in PCR was isolated from :
Q173.
mcq single
+4 / 1
Ligation of foreign DNA at which of the following site will result in loss of tetracyclin resistance of pBR322 :
Q174.
mcq single
+4 / 1
Match List I with List II.
List - I
List - II
(A)
Kanamycin
(I)
Delivers genes into animal cells
(B)
ClaI
(II)
Selectable marker
(C)
Disarmed retroviruses
(III)
Restriction site
(D)
Kanamycin $$^\mathrm{R}$$gene
(IV)
Antibiotic resistance
Choose the **correct** answer from the options given below :