neet-2024
NEET 2024
MEDICAL 2024 Previous Year
3 hDuration
800Total Marks
400Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 400 across 3 section(s); maximum marks: 800.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
The $$\mathrm{E}^{\circ}$$ value for the $$\mathrm{Mn}^{3+} / \mathrm{Mn}^{2+}$$ couple is more positive than that of $$\mathrm{Cr}^{3+} / \mathrm{Cr}^{2+}$$ or $$\mathrm{Fe}^{3+} / \mathrm{Fe}^{2+}$$ due to change of
Q2.
mcq single
+4 / 1
The pair of lanthanoid ions which are diamagnetic is
Q3.
mcq single
+4 / 1
'Spin only' magnetic moment is same for which of the following ions?
A. $$\mathrm{Ti}^{3+}$$
B. $$\mathrm{Cr}^{2+}$$
C. $$\mathrm{Mn}^{2+}$$
D. $$\mathrm{Fe}^{2+}$$
E. $$\mathrm{Sc}^{3+}$$
Choose the most appropriate answer from the options given below.
Q4.
mcq single
+4 / 1
Given below are two statements:
Statement I: The boiling point of hydrides of Group 16 elements follow the order
$$\mathrm{H}_2 \mathrm{O}>\mathrm{H}_2 \mathrm{Te}>\mathrm{H}_2 \mathrm{Se}>\mathrm{H}_2 \mathrm{S} \text {. }$$
Statement II: On the basis of molecular mass, $$\mathrm{H}_2 \mathrm{O}$$ is expected to have lower boiling point than the other members of the group but due to the presence of extensive $$\mathrm{H}$$-bonding in $$\mathrm{H}_2 \mathrm{O}$$, it has higher boiling point.
In the light of the above statements, choose the correct answer from the options given below:
Q5.
mcq single
+4 / 1
Match List I with List II.
List I
(Molecule)
List II
(Number and types of bond/s between two carbon atoms)
A.
ethane
I.
one $$\sigma$$-bond and two $$\pi$$-bonds
B.
ethene
II.
two $$\pi$$-bonds
C.
carbon molecule, C$$_2$$
III.
one $$\sigma$$-bond
D.
ethyne
IV.
one $$\sigma$$-bond and one $$\pi$$-bond
Choose the correct answer from the options given below :
Q6.
mcq single
+4 / 1
Identify the correct answer.
Q7.
mcq single
+4 / 1
Match List I with List II.
List I
(Compound)
List II
(Shape/geometry)
A.
NH$$_3$$
I.
Trigonal Pyramidal
B.
BrF$$_5$$
II.
Square Planar
C.
XeF$$_4$$
III.
Octahedral
D.
SF$$_6$$
IV.
Square Pyramidal
Choose the correct answer from the options given below:
Q8.
mcq single
+4 / 1
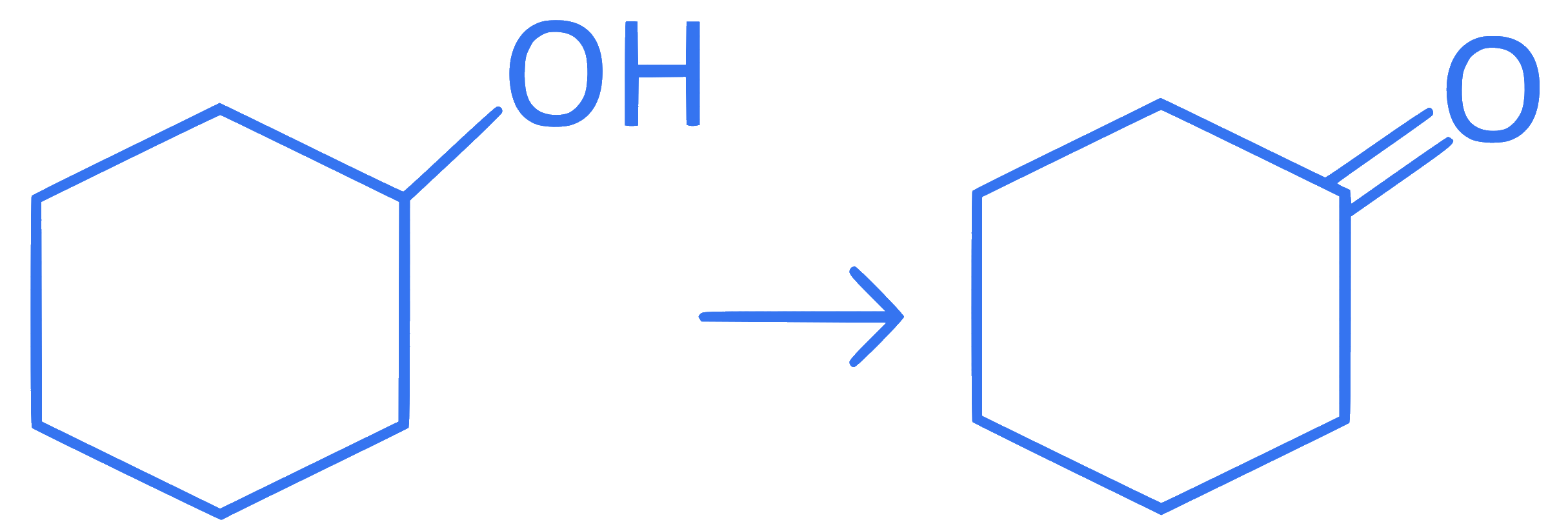
Intramolecular hydrogen bonding is present in
Q9.
mcq single
+4 / 1
Activation energy of any chemical reaction can be calculated if one knows the value of
Q10.
mcq single
+4 / 1
The rate of a reaction quadruples when temperature changes from $$27^{\circ} \mathrm{C}$$ to $$57^{\circ} \mathrm{C}$$. Calculate the energy of activation.
Given $$\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \log 4=0.6021$$
Q11.
mcq single
+4 / 1
Which plot of $$\ln \mathrm{k}$$ vs $$\frac{1}{\mathrm{~T}}$$ is consistent with Arrhenius equation?
Q12.
mcq single
+4 / 1
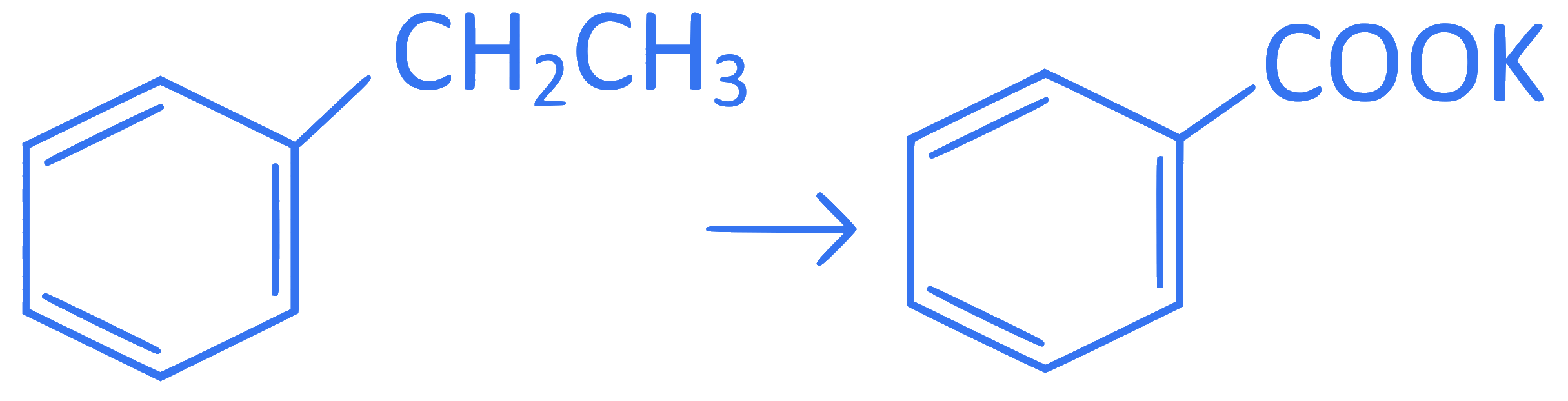
Which reaction is NOT a redox reaction?
Q13.
mcq single
+4 / 1
The compound that will undergo S~N~1 reaction with the fastest rate is
Q14.
mcq single
+4 / 1
Identify the major product C formed in the following reaction sequence :
$$
\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{I} \xrightarrow{\mathrm{NaCN}} \mathrm{A}
$$
$$\mathrm{\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{Partial\,hydrolysis}^{O{H^ - }}} B\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{B{r_2}}^{NaOH}} \mathop C\limits_{(major)}}$$
Q15.
mcq single
+4 / 1
The Henry's law constant $$(\mathrm{K}_{\mathrm{H}})$$ values of three gases $$(\mathrm{A}, \mathrm{B}, \mathrm{C})$$ in water are $$145, 2 \times 10^{-5}$$ and $$35 \mathrm{~kbar}$$ respectively. The solubility of these gases in water follow the order:
Q16.
mcq single
+4 / 1
The plot of osmotic pressure (П) vs concentration $$(\mathrm{mol} \mathrm{~L}^{-1})$$ for a solution gives a straight line with slope $$25.73 \mathrm{~L} \mathrm{~bar} \mathrm{~mol}^{-1}$$. The temperature at which the osmotic pressure measurement is done is
(Use $$\mathrm{R}=0.083 \mathrm{~L} \mathrm{~bar} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$$)
Q17.
mcq single
+4 / 1
A compound X contains $$32 \%$$ of A, $$20 \%$$ of B and remaining percentage of C. Then, the empirical formula of $$\mathrm{X}$$ is :
(Given atomic masses of A=64 ; B=40 ; C=32 u)
Q18.
mcq single
+4 / 1
The highest number of helium atoms is in
Q19.
mcq single
+4 / 1
1 gram of sodium hydroxide was treated with $$25 \mathrm{~mL}$$ of $$0.75 \mathrm{~M} \mathrm{~HCl}$$ solution, the mass of sodium hydroxide left unreacted is equal to
Q20.
mcq single
+4 / 1
For the reaction $$2 \mathrm{~A} \rightleftharpoons \mathrm{B}+\mathrm{C}, \mathrm{K}_{\mathrm{c}}=4 \times 10^{-3}$$. At a given time, the composition of reaction mixture is:
$$[A]=[B]=[C]=2 \times 10^{-3} \mathrm{M} \text {. }$$ Then, which of the following is correct?
Q21.
mcq single
+4 / 1
In which of the following equilibria, $$\mathrm{K}_p$$ and $$\mathrm{K}_{\mathrm{c}}$$ are NOT equal?
Q22.
mcq single
+4 / 1
Consider the following reaction in a sealed vessel at equilibrium with concentrations of $$\mathrm{N}_2=3.0 \times 10^{-3} \mathrm{M}, \mathrm{O}_2=4.2 \times 10^{-3} \mathrm{M}$$ and $$\mathrm{NO}=2.8 \times 10^{-3} \mathrm{M}$$.
$$2 \mathrm{NO}_{(\mathrm{g})} \rightleftharpoons \mathrm{N}_{2(\mathrm{~g})}+\mathrm{O}_{2(\mathrm{~g})}$$
If $$0.1 \mathrm{~mol} \mathrm{~L} \mathrm{~L}^{-1}$$ of $$\mathrm{NO}_{(\mathrm{g})}$$ is taken in a closed vessel, what will be degree of dissociation ($$\alpha$$) of $$\mathrm{NO}_{(\mathrm{g})}$$ at equilibrium?
Q23.
mcq single
+4 / 1
Arrange the following elements in increasing order of electronegativity:
N, O, F, C, Si
Choose the correct answer from the options given below :
Q24.
mcq single
+4 / 1
Arrange the following elements in increasing order of first ionization enthalpy:
$$\mathrm{Li}, \mathrm{Be}, \mathrm{B}, \mathrm{C}, \mathrm{N}$$
Choose the correct answer from the options given below:
Q25.
mcq single
+4 / 1
In which of the following processes entropy increases?
A. A liquid evaporates to vapour.
B. Temperature of a crystalline solid lowered from $$130 \mathrm{~K}$$ to $$0 \mathrm{~K}$$.
C. $$2 \mathrm{NaHCO}_{3(\mathrm{~s})} \rightarrow \mathrm{Na}_2 \mathrm{CO}_{3(\mathrm{~s})}+\mathrm{CO}_{2(\mathrm{~g})}+\mathrm{H}_2 \mathrm{O}_{(\mathrm{g})}$$
D. $$\mathrm{Cl}_{2(g)} \rightarrow 2 \mathrm{Cl}_{(g)}$$
Choose the correct answer from the options given below:
Q26.
mcq single
+4 / 1
The work done during reversible isothermal expansion of one mole of hydrogen gas at $$25^{\circ} \mathrm{C}$$ from pressure of 20 atmosphere to 10 atmosphere is (Given $$\mathrm{R}=2.0 \mathrm{~cal} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$$)
Q27.
mcq single
+4 / 1
Match List I with List II.
List I
(Process)
List II
(Conditions)
A.
Isothermal process
I.
No heat exchange
B.
Isochoric process
II.
Carried out at constant temperature
C.
Isobaric process
III.
Carried out at constant volume
D.
Adiabatic process
IV.
Carried out at constant pressure
Choose the correct answer from the options given below:
Q28.
mcq single
+4 / 1
Among Group 16 elements, which one does NOT show -2 oxidation state?
Q29.
mcq single
+4 / 1
Given below are certain cations. Using inorganic qualitative analysis, arrange them in increasing group number from 0 to $$\mathrm{VI}$$.
A. $$\mathrm{Al}^{3+}$$
B. $$\mathrm{Cu}^{2+}$$
C. $$\mathrm{Ba}^{2+}$$
D. $$\mathrm{Co}^{2+}$$
E. $$\mathrm{Mg}^{2+}$$
Choose the correct answer from the options given below.
Q30.
mcq single
+4 / 1
Match List I with List II.
List I
(Complex)
List II
(Type of isomerism)
A.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5\left(\mathrm{NO}_2\right)\right] \mathrm{Cl}_2
$$
I.
Solvate isomerism
B.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5\left(\mathrm{SO}_4\right)\right] \mathrm{Br}
$$
II.
Linkage isomerism
C.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]\left[\mathrm{Cr}(\mathrm{CN})_6\right]
$$
III.
Ionization isomerism
D.
$$
\left[\mathrm{Co}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{Cl}_3
$$
IV.
Coordination isomerism
Choose the correct answer from the options given below:
Q31.
mcq single
+4 / 1
Given below are two statements :
Statement I: Both $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ and $$[\mathrm{CoF}_6]^{3-}$$ complexes are octahedral but differ in their magnetic behaviour.
Statement II: $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ is diamagnetic whereas $$[\mathrm{CoF}_6]^{3-}$$ is paramagnetic.
In the light of the above statements, choose the correct answer from the options given below:
Q32.
mcq single
+4 / 1
Given below are two statements :
Statement I : $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ is a homoleptic complex whereas $$[\mathrm{Co}(\mathrm{NH}_3)_4 \mathrm{Cl}_2]^{+}$$ is a heteroleptic complex.
Statement II : Complex $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ has only one kind of ligands but $$[\mathrm{Co}(\mathrm{NH}_3)_4 \mathrm{Cl}_2]^{+}$$ has more than one kind of ligands.
In the light of the above statements, choose the correct answer from the options given below.
Q33.
mcq single
+4 / 1
Given below are two statements:
Statement I : The boiling point of three isomeric pentanes follows the order
n-pentane > isopentane > neopentane
Statement II : When branching increases, the molecule attains a shape of sphere. This results in smaller surface area for contact, due to which the intermolecular forces between the spherical molecules are weak, thereby lowering the boiling point.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q34.
mcq single
+4 / 1
During the preparation of Mohr's salt solution (Ferrous ammonium sulphate), which of the following acid is added to prevent hydrolysis of $$\mathrm{Fe}^{2+}$$ ion?
Q35.
mcq single
+4 / 1
On heating, some solid substances change from solid to vapour state without passing through liquid state. The technique used for the purification of such solid substances based on the above principle is known as
Q36.
mcq single
+4 / 1
The most stable carbocation among the following is :
Q37.
mcq single
+4 / 1
A compound with a molecular formula of $$\mathrm{C}_6 \mathrm{H}_{14}$$ has two tertiary carbons. Its IUPAC name is :
Q38.
mcq single
+4 / 1
For the given reaction:
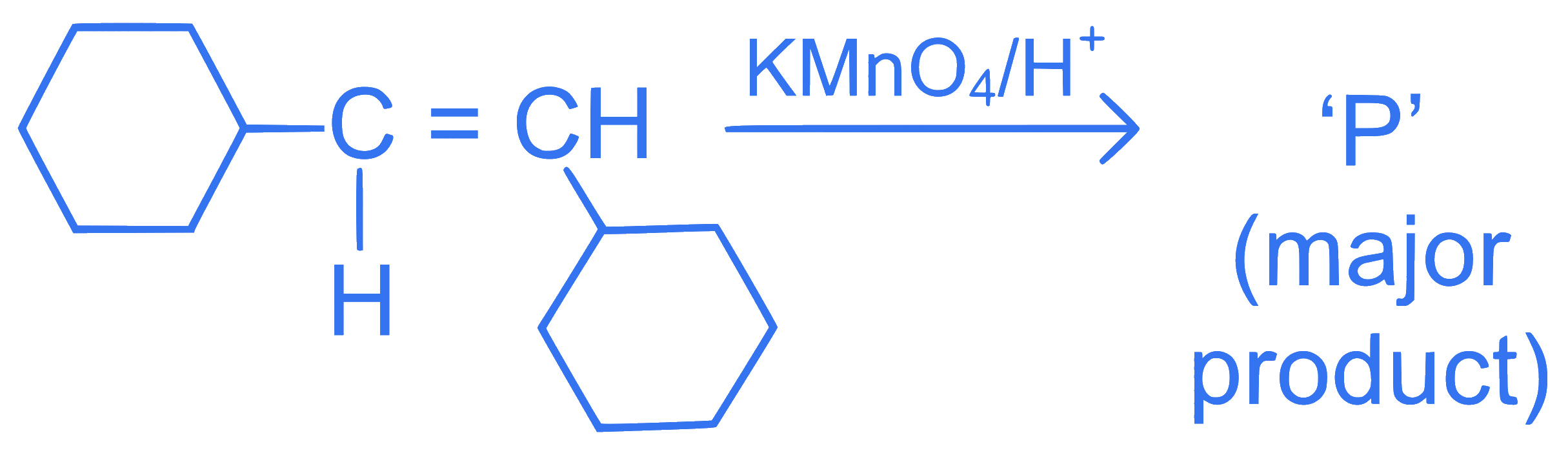 'P' is
'P' is
 'P' is
'P' is
Q39.
mcq single
+4 / 1
Match List I with List II.
List I
(Reaction)
List II
(Reagents/Condition)
A.
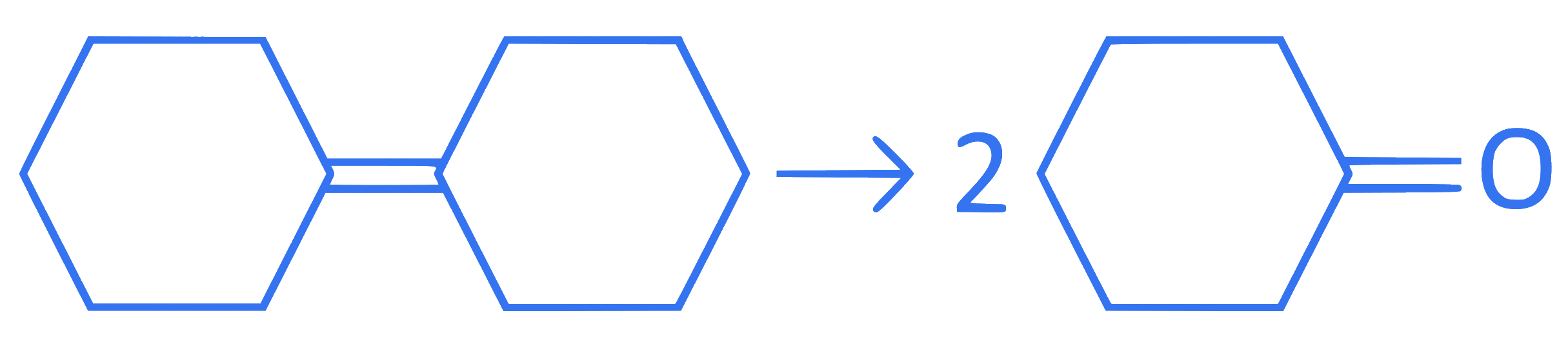 I.
I.
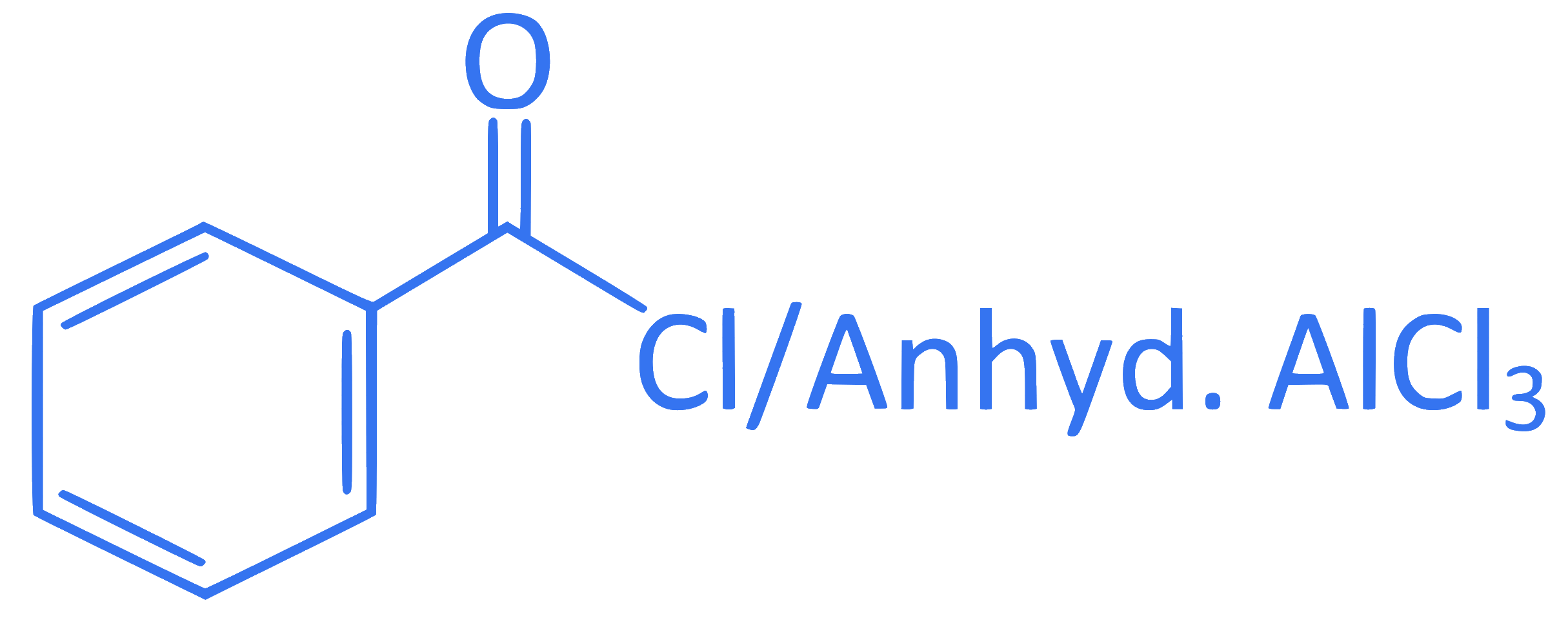 B.
B.
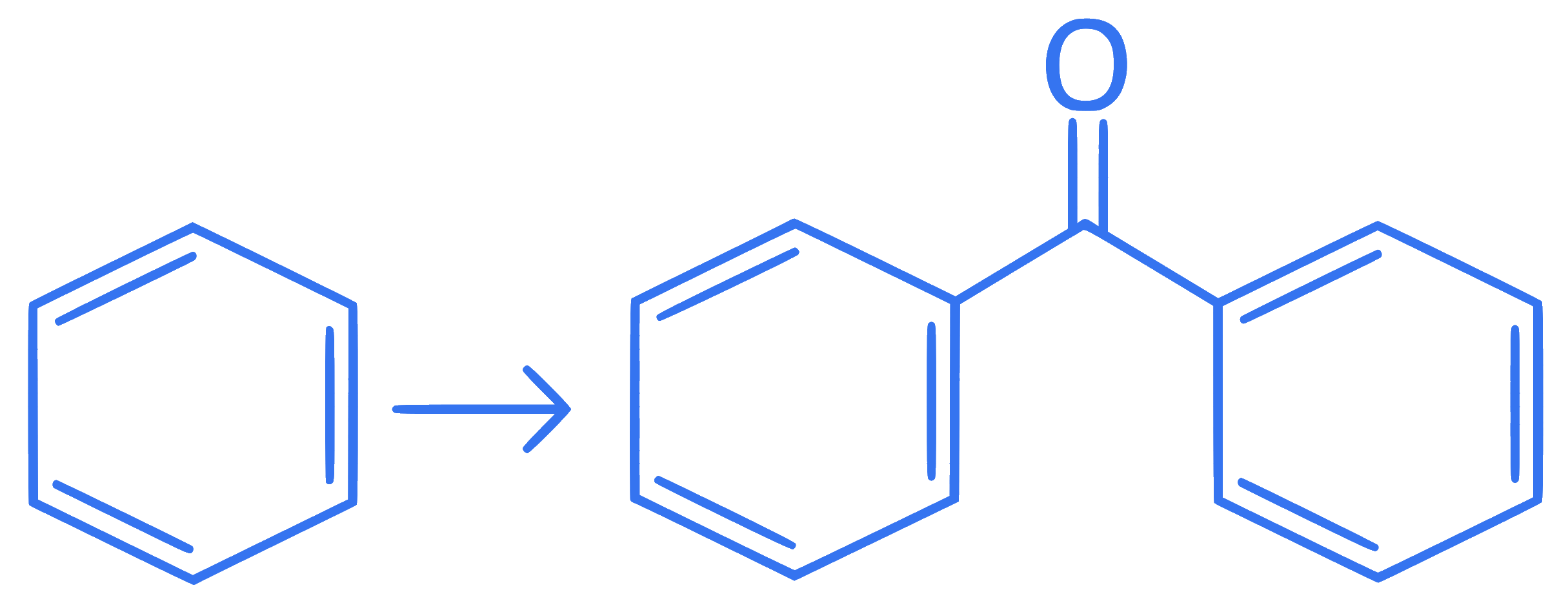 II.
CrO$$_3$$
C.
II.
CrO$$_3$$
C.
 III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
 IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
 I.
I.
 B.
B.
 II.
CrO$$_3$$
C.
II.
CrO$$_3$$
C.
 III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
 IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
Q40.
mcq single
+4 / 1
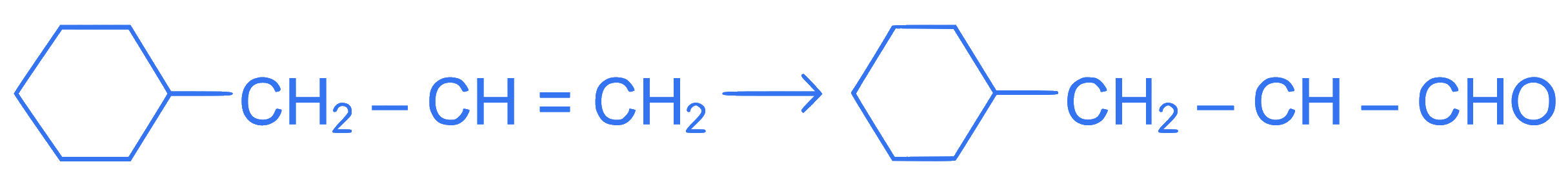
Identify the correct reagents that would bring about the following transformation.

Q41.
mcq single
+4 / 1
Which one of the following alcohols reacts instantaneously with Lucas reagent?
Q42.
mcq single
+4 / 1
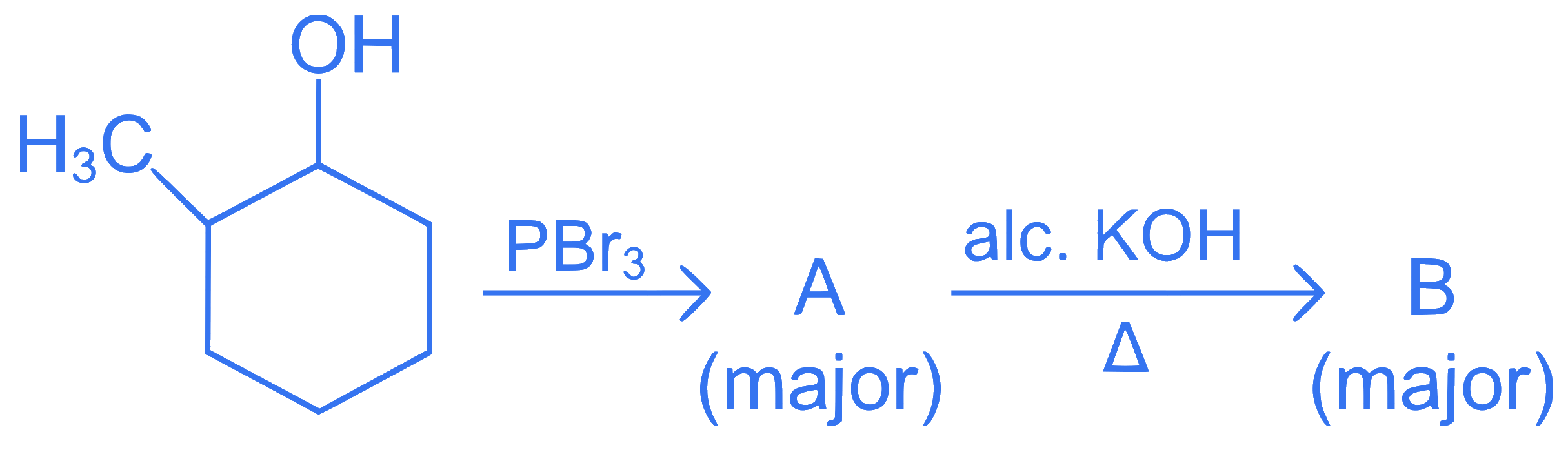
Major products A and B formed in the following reaction sequence, are

Q43.
mcq single
+4 / 1
The products $$\mathrm{A}$$ and $$\mathrm{B}$$ obtained in the following reactions, respectively, are
$$\begin{aligned}
& 3 \mathrm{ROH}+\mathrm{PCl}_3 \rightarrow 3 \mathrm{RCl}+\mathrm{A} \\
& \mathrm{ROH}+\mathrm{PCl}_5 \rightarrow \mathrm{RCl}+\mathrm{HCl}+\mathrm{B}
\end{aligned}$$
Q44.
mcq single
+4 / 1
The reagents with which glucose does not react to give the corresponding tests/products are
A. Tollen's reagent
B. Schiff's reagent
C. HCN
D. $$\mathrm{NH}_2 \mathrm{OH}$$
E. $$\mathrm{NaHSO}_3$$
Choose the correct options from the given below:
Q45.
mcq single
+4 / 1
Fehling's solution 'A' is
Q46.
mcq single
+4 / 1
Match List I with List II.
List I
(Conversion)
List II
(Number of Faraday required)
A.
1 mole of H$$_2$$O to O$$_2$$
I.
3F
B.
1 mol of MnO$$_4^-$$ to Mn$$^{2+}$$
II.
2F
C.
1.5 mol of Ca from molten CaCl$$_2$$
III.
1F
D.
1 mol of FeO to Fe$$_2$$O$$_3$$
IV.
5F
Choose the correct answer from the options given below :
Q47.
mcq single
+4 / 1
Mass in grams of copper deposited by passing 9.6487 A current through a voltmeter containing copper sulphate solution for 100 seconds is (Given : Molar mass of $$\mathrm{Cu}: 63 \mathrm{~g} \mathrm{~mol}^{-1}, 1 \mathrm{~F}=96487 \mathrm{C}$$)
Q48.
mcq single
+4 / 1
Given below are two statements:
Statement I : Aniline does not undergo Friedel-Crafts alkylation reaction.
Statement II : Aniline cannot be prepared through Gabriel synthesis.
In the light of the above statements, choose the correct answer from the options given below:
Q49.
mcq single
+4 / 1
The energy of an electron in the ground state $$\mathrm{(n=1)}$$ for $$\mathrm{He}^{+}$$ ion is $$\mathrm{-x} \mathrm{~J}$$, then that for an electron in $$\mathrm{(n=2)}$$ state for $$\mathrm{Be}^{3+}$$ ion in $$\mathrm{J}$$ is
Q50.
mcq single
+4 / 1
Match List I with List II.
List I
(Quantum Number)
List II
(Information provided)
A.
$$\mathrm{m_l}$$
I.
Shape of orbital
B.
$$\mathrm{m_s}$$
II.
Size of orbital
C.
I
III.
Orientation of orbital
D.
n
IV.
Orientation of spin of electron
Choose the correct answer from the options given below :
Q51.
mcq single
+4 / 1
The $$\mathrm{E}^{\circ}$$ value for the $$\mathrm{Mn}^{3+} / \mathrm{Mn}^{2+}$$ couple is more positive than that of $$\mathrm{Cr}^{3+} / \mathrm{Cr}^{2+}$$ or $$\mathrm{Fe}^{3+} / \mathrm{Fe}^{2+}$$ due to change of
Q52.
mcq single
+4 / 1
The pair of lanthanoid ions which are diamagnetic is
Q53.
mcq single
+4 / 1
'Spin only' magnetic moment is same for which of the following ions?
A. $$\mathrm{Ti}^{3+}$$
B. $$\mathrm{Cr}^{2+}$$
C. $$\mathrm{Mn}^{2+}$$
D. $$\mathrm{Fe}^{2+}$$
E. $$\mathrm{Sc}^{3+}$$
Choose the most appropriate answer from the options given below.
Q54.
mcq single
+4 / 1
Given below are two statements:
Statement I: The boiling point of hydrides of Group 16 elements follow the order
$$\mathrm{H}_2 \mathrm{O}>\mathrm{H}_2 \mathrm{Te}>\mathrm{H}_2 \mathrm{Se}>\mathrm{H}_2 \mathrm{S} \text {. }$$
Statement II: On the basis of molecular mass, $$\mathrm{H}_2 \mathrm{O}$$ is expected to have lower boiling point than the other members of the group but due to the presence of extensive $$\mathrm{H}$$-bonding in $$\mathrm{H}_2 \mathrm{O}$$, it has higher boiling point.
In the light of the above statements, choose the correct answer from the options given below:
Q55.
mcq single
+4 / 1
Match List I with List II.
List I
(Molecule)
List II
(Number and types of bond/s between two carbon atoms)
A.
ethane
I.
one $$\sigma$$-bond and two $$\pi$$-bonds
B.
ethene
II.
two $$\pi$$-bonds
C.
carbon molecule, C$$_2$$
III.
one $$\sigma$$-bond
D.
ethyne
IV.
one $$\sigma$$-bond and one $$\pi$$-bond
Choose the correct answer from the options given below :
Q56.
mcq single
+4 / 1
Identify the correct answer.
Q57.
mcq single
+4 / 1
Match List I with List II.
List I
(Compound)
List II
(Shape/geometry)
A.
NH$$_3$$
I.
Trigonal Pyramidal
B.
BrF$$_5$$
II.
Square Planar
C.
XeF$$_4$$
III.
Octahedral
D.
SF$$_6$$
IV.
Square Pyramidal
Choose the correct answer from the options given below:
Q58.
mcq single
+4 / 1
Intramolecular hydrogen bonding is present in
Q59.
mcq single
+4 / 1
Activation energy of any chemical reaction can be calculated if one knows the value of
Q60.
mcq single
+4 / 1
The rate of a reaction quadruples when temperature changes from $$27^{\circ} \mathrm{C}$$ to $$57^{\circ} \mathrm{C}$$. Calculate the energy of activation.
Given $$\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}, \log 4=0.6021$$
Q61.
mcq single
+4 / 1
Which plot of $$\ln \mathrm{k}$$ vs $$\frac{1}{\mathrm{~T}}$$ is consistent with Arrhenius equation?
Q62.
mcq single
+4 / 1
Which reaction is NOT a redox reaction?
Q63.
mcq single
+4 / 1
The compound that will undergo S~N~1 reaction with the fastest rate is
Q64.
mcq single
+4 / 1
Identify the major product C formed in the following reaction sequence :
$$
\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{I} \xrightarrow{\mathrm{NaCN}} \mathrm{A}
$$
$$\mathrm{\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{Partial\,hydrolysis}^{O{H^ - }}} B\mathrel{\mathop{\kern0pt\longrightarrow}
\limits_{B{r_2}}^{NaOH}} \mathop C\limits_{(major)}}$$
Q65.
mcq single
+4 / 1
The Henry's law constant $$(\mathrm{K}_{\mathrm{H}})$$ values of three gases $$(\mathrm{A}, \mathrm{B}, \mathrm{C})$$ in water are $$145, 2 \times 10^{-5}$$ and $$35 \mathrm{~kbar}$$ respectively. The solubility of these gases in water follow the order:
Q66.
mcq single
+4 / 1
The plot of osmotic pressure (П) vs concentration $$(\mathrm{mol} \mathrm{~L}^{-1})$$ for a solution gives a straight line with slope $$25.73 \mathrm{~L} \mathrm{~bar} \mathrm{~mol}^{-1}$$. The temperature at which the osmotic pressure measurement is done is
(Use $$\mathrm{R}=0.083 \mathrm{~L} \mathrm{~bar} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$$)
Q67.
mcq single
+4 / 1
A compound X contains $$32 \%$$ of A, $$20 \%$$ of B and remaining percentage of C. Then, the empirical formula of $$\mathrm{X}$$ is :
(Given atomic masses of A=64 ; B=40 ; C=32 u)
Q68.
mcq single
+4 / 1
The highest number of helium atoms is in
Q69.
mcq single
+4 / 1
1 gram of sodium hydroxide was treated with $$25 \mathrm{~mL}$$ of $$0.75 \mathrm{~M} \mathrm{~HCl}$$ solution, the mass of sodium hydroxide left unreacted is equal to
Q70.
mcq single
+4 / 1
For the reaction $$2 \mathrm{~A} \rightleftharpoons \mathrm{B}+\mathrm{C}, \mathrm{K}_{\mathrm{c}}=4 \times 10^{-3}$$. At a given time, the composition of reaction mixture is:
$$[A]=[B]=[C]=2 \times 10^{-3} \mathrm{M} \text {. }$$ Then, which of the following is correct?
Q71.
mcq single
+4 / 1
In which of the following equilibria, $$\mathrm{K}_p$$ and $$\mathrm{K}_{\mathrm{c}}$$ are NOT equal?
Q72.
mcq single
+4 / 1
Consider the following reaction in a sealed vessel at equilibrium with concentrations of $$\mathrm{N}_2=3.0 \times 10^{-3} \mathrm{M}, \mathrm{O}_2=4.2 \times 10^{-3} \mathrm{M}$$ and $$\mathrm{NO}=2.8 \times 10^{-3} \mathrm{M}$$.
$$2 \mathrm{NO}_{(\mathrm{g})} \rightleftharpoons \mathrm{N}_{2(\mathrm{~g})}+\mathrm{O}_{2(\mathrm{~g})}$$
If $$0.1 \mathrm{~mol} \mathrm{~L} \mathrm{~L}^{-1}$$ of $$\mathrm{NO}_{(\mathrm{g})}$$ is taken in a closed vessel, what will be degree of dissociation ($$\alpha$$) of $$\mathrm{NO}_{(\mathrm{g})}$$ at equilibrium?
Q73.
mcq single
+4 / 1
Arrange the following elements in increasing order of electronegativity:
N, O, F, C, Si
Choose the correct answer from the options given below :
Q74.
mcq single
+4 / 1
Arrange the following elements in increasing order of first ionization enthalpy:
$$\mathrm{Li}, \mathrm{Be}, \mathrm{B}, \mathrm{C}, \mathrm{N}$$
Choose the correct answer from the options given below:
Q75.
mcq single
+4 / 1
In which of the following processes entropy increases?
A. A liquid evaporates to vapour.
B. Temperature of a crystalline solid lowered from $$130 \mathrm{~K}$$ to $$0 \mathrm{~K}$$.
C. $$2 \mathrm{NaHCO}_{3(\mathrm{~s})} \rightarrow \mathrm{Na}_2 \mathrm{CO}_{3(\mathrm{~s})}+\mathrm{CO}_{2(\mathrm{~g})}+\mathrm{H}_2 \mathrm{O}_{(\mathrm{g})}$$
D. $$\mathrm{Cl}_{2(g)} \rightarrow 2 \mathrm{Cl}_{(g)}$$
Choose the correct answer from the options given below:
Q76.
mcq single
+4 / 1
The work done during reversible isothermal expansion of one mole of hydrogen gas at $$25^{\circ} \mathrm{C}$$ from pressure of 20 atmosphere to 10 atmosphere is (Given $$\mathrm{R}=2.0 \mathrm{~cal} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$$)
Q77.
mcq single
+4 / 1
Match List I with List II.
List I
(Process)
List II
(Conditions)
A.
Isothermal process
I.
No heat exchange
B.
Isochoric process
II.
Carried out at constant temperature
C.
Isobaric process
III.
Carried out at constant volume
D.
Adiabatic process
IV.
Carried out at constant pressure
Choose the correct answer from the options given below:
Q78.
mcq single
+4 / 1
Among Group 16 elements, which one does NOT show -2 oxidation state?
Q79.
mcq single
+4 / 1
Given below are certain cations. Using inorganic qualitative analysis, arrange them in increasing group number from 0 to $$\mathrm{VI}$$.
A. $$\mathrm{Al}^{3+}$$
B. $$\mathrm{Cu}^{2+}$$
C. $$\mathrm{Ba}^{2+}$$
D. $$\mathrm{Co}^{2+}$$
E. $$\mathrm{Mg}^{2+}$$
Choose the correct answer from the options given below.
Q80.
mcq single
+4 / 1
Match List I with List II.
List I
(Complex)
List II
(Type of isomerism)
A.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5\left(\mathrm{NO}_2\right)\right] \mathrm{Cl}_2
$$
I.
Solvate isomerism
B.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5\left(\mathrm{SO}_4\right)\right] \mathrm{Br}
$$
II.
Linkage isomerism
C.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]\left[\mathrm{Cr}(\mathrm{CN})_6\right]
$$
III.
Ionization isomerism
D.
$$
\left[\mathrm{Co}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{Cl}_3
$$
IV.
Coordination isomerism
Choose the correct answer from the options given below:
Q81.
mcq single
+4 / 1
Given below are two statements :
Statement I: Both $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ and $$[\mathrm{CoF}_6]^{3-}$$ complexes are octahedral but differ in their magnetic behaviour.
Statement II: $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ is diamagnetic whereas $$[\mathrm{CoF}_6]^{3-}$$ is paramagnetic.
In the light of the above statements, choose the correct answer from the options given below:
Q82.
mcq single
+4 / 1
Given below are two statements :
Statement I : $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ is a homoleptic complex whereas $$[\mathrm{Co}(\mathrm{NH}_3)_4 \mathrm{Cl}_2]^{+}$$ is a heteroleptic complex.
Statement II : Complex $$[\mathrm{Co}(\mathrm{NH}_3)_6]^{3+}$$ has only one kind of ligands but $$[\mathrm{Co}(\mathrm{NH}_3)_4 \mathrm{Cl}_2]^{+}$$ has more than one kind of ligands.
In the light of the above statements, choose the correct answer from the options given below.
Q83.
mcq single
+4 / 1
Given below are two statements:
Statement I : The boiling point of three isomeric pentanes follows the order
n-pentane > isopentane > neopentane
Statement II : When branching increases, the molecule attains a shape of sphere. This results in smaller surface area for contact, due to which the intermolecular forces between the spherical molecules are weak, thereby lowering the boiling point.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q84.
mcq single
+4 / 1
During the preparation of Mohr's salt solution (Ferrous ammonium sulphate), which of the following acid is added to prevent hydrolysis of $$\mathrm{Fe}^{2+}$$ ion?
Q85.
mcq single
+4 / 1
On heating, some solid substances change from solid to vapour state without passing through liquid state. The technique used for the purification of such solid substances based on the above principle is known as
Q86.
mcq single
+4 / 1
The most stable carbocation among the following is :
Q87.
mcq single
+4 / 1
A compound with a molecular formula of $$\mathrm{C}_6 \mathrm{H}_{14}$$ has two tertiary carbons. Its IUPAC name is :
Q88.
mcq single
+4 / 1
For the given reaction:
 'P' is
'P' is
 'P' is
'P' is
Q89.
mcq single
+4 / 1
Match List I with List II.
List I
(Reaction)
List II
(Reagents/Condition)
A.
 I.
I.
 B.
B.
 II.
CrO$$_3$$
C.
II.
CrO$$_3$$
C.
 III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
 IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
 I.
I.
 B.
B.
 II.
CrO$$_3$$
C.
II.
CrO$$_3$$
C.
 III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
III.
KMnO$$_4$$/KOH, $$\Delta$$
D.
 IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
IV.
(i) O$$_3$$
(ii) Zn-H$$_2$$O
Choose the correct answer from the options given below:
Q90.
mcq single
+4 / 1
Identify the correct reagents that would bring about the following transformation.


Q91.
mcq single
+4 / 1
Which one of the following alcohols reacts instantaneously with Lucas reagent?
Q92.
mcq single
+4 / 1
Major products A and B formed in the following reaction sequence, are


Q93.
mcq single
+4 / 1
The products $$\mathrm{A}$$ and $$\mathrm{B}$$ obtained in the following reactions, respectively, are
$$\begin{aligned}
& 3 \mathrm{ROH}+\mathrm{PCl}_3 \rightarrow 3 \mathrm{RCl}+\mathrm{A} \\
& \mathrm{ROH}+\mathrm{PCl}_5 \rightarrow \mathrm{RCl}+\mathrm{HCl}+\mathrm{B}
\end{aligned}$$
Q94.
mcq single
+4 / 1
The reagents with which glucose does not react to give the corresponding tests/products are
A. Tollen's reagent
B. Schiff's reagent
C. HCN
D. $$\mathrm{NH}_2 \mathrm{OH}$$
E. $$\mathrm{NaHSO}_3$$
Choose the correct options from the given below:
Q95.
mcq single
+4 / 1
Fehling's solution 'A' is
Q96.
mcq single
+4 / 1
Match List I with List II.
List I
(Conversion)
List II
(Number of Faraday required)
A.
1 mole of H$$_2$$O to O$$_2$$
I.
3F
B.
1 mol of MnO$$_4^-$$ to Mn$$^{2+}$$
II.
2F
C.
1.5 mol of Ca from molten CaCl$$_2$$
III.
1F
D.
1 mol of FeO to Fe$$_2$$O$$_3$$
IV.
5F
Choose the correct answer from the options given below :
Q97.
mcq single
+4 / 1
Mass in grams of copper deposited by passing 9.6487 A current through a voltmeter containing copper sulphate solution for 100 seconds is (Given : Molar mass of $$\mathrm{Cu}: 63 \mathrm{~g} \mathrm{~mol}^{-1}, 1 \mathrm{~F}=96487 \mathrm{C}$$)
Q98.
mcq single
+4 / 1
Given below are two statements:
Statement I : Aniline does not undergo Friedel-Crafts alkylation reaction.
Statement II : Aniline cannot be prepared through Gabriel synthesis.
In the light of the above statements, choose the correct answer from the options given below:
Q99.
mcq single
+4 / 1
The energy of an electron in the ground state $$\mathrm{(n=1)}$$ for $$\mathrm{He}^{+}$$ ion is $$\mathrm{-x} \mathrm{~J}$$, then that for an electron in $$\mathrm{(n=2)}$$ state for $$\mathrm{Be}^{3+}$$ ion in $$\mathrm{J}$$ is
Q100.
mcq single
+4 / 1
Match List I with List II.
List I
(Quantum Number)
List II
(Information provided)
A.
$$\mathrm{m_l}$$
I.
Shape of orbital
B.
$$\mathrm{m_s}$$
II.
Size of orbital
C.
I
III.
Orientation of orbital
D.
n
IV.
Orientation of spin of electron
Choose the correct answer from the options given below :
Physics
Physics
Q1.
mcq single
+4 / 1
If the monochromatic source in Young's double slit experiment is replaced by white light, then
Q2.
mcq single
+4 / 1
An unpolarised light beam strikes a glass surface at Brewster's angle. Then
Q3.
mcq single
+4 / 1
A bob is whirled in a horizontal plane by means of a string with an initial speed of $$\omega \mathrm{~rpm}$$. The tension in the string is $$T$$. If speed becomes $$2 \omega$$ while keeping the same radius, the tension in the string becomes:
Q4.
mcq single
+4 / 1
A wheel of a bullock cart is rolling on a level road as shown in the figure below. If its linear speed is $$v$$ in the direction shown, which one of the following options is correct ($$P$$ and $$Q$$ are any highest and lowest points on the wheel, respectively)?
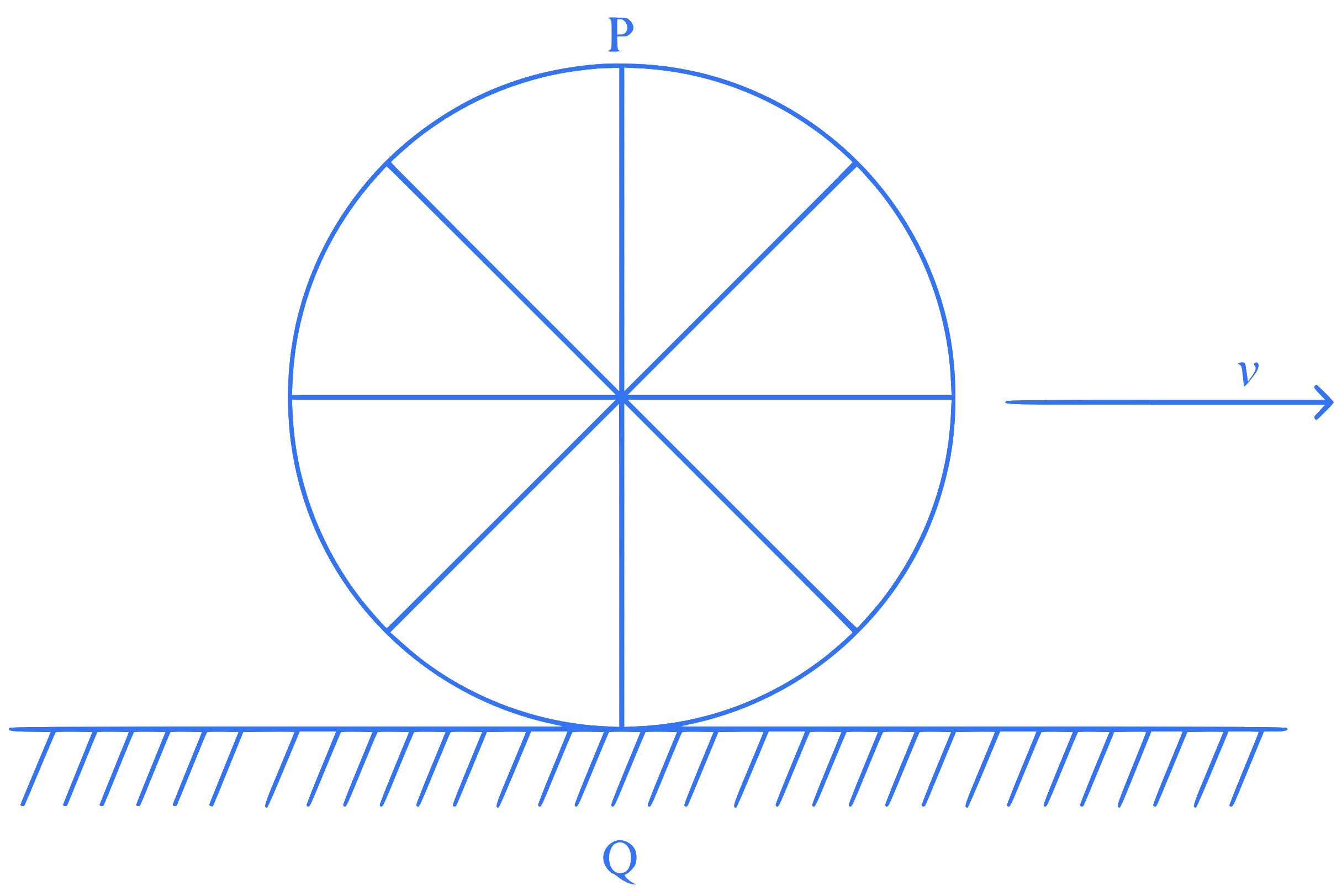

Q5.
mcq single
+4 / 1
The moment of inertia of a thin rod about an axis passing through its mid point and perpendicular to the rod is $$2400 \mathrm{~g} \mathrm{~cm}^2$$. The length of the $$400 \mathrm{~g}$$ rod is nearly:
Q6.
mcq single
+4 / 1
In a vernier callipers, $$(N+1)$$ divisions of vernier scale coincide with $$N$$ divisions of main scale. If $$1 \mathrm{~MSD}$$ represents $$0.1 \mathrm{~mm}$$, the vernier constant (in $$\mathrm{cm}$$) is:
Q7.
mcq single
+4 / 1
The quantities which have the same dimensions as those of solid angle are:
Q8.
mcq single
+4 / 1
A force defined by $$F=\alpha t^2+\beta t$$ acts on a particle at a given time $$t$$. The factor which is dimensionless, if $$\alpha$$ and $$\beta$$ are constants, is:
Q9.
mcq single
+4 / 1
The following graph represents the $$T$$-$$V$$ curves of an ideal gas (where $$T$$ is the temperature and $$V$$ the volume) at three pressures $$P_1, P_2$$ and $$P_3$$ compared with those of Charles's law represented as dotted lines.
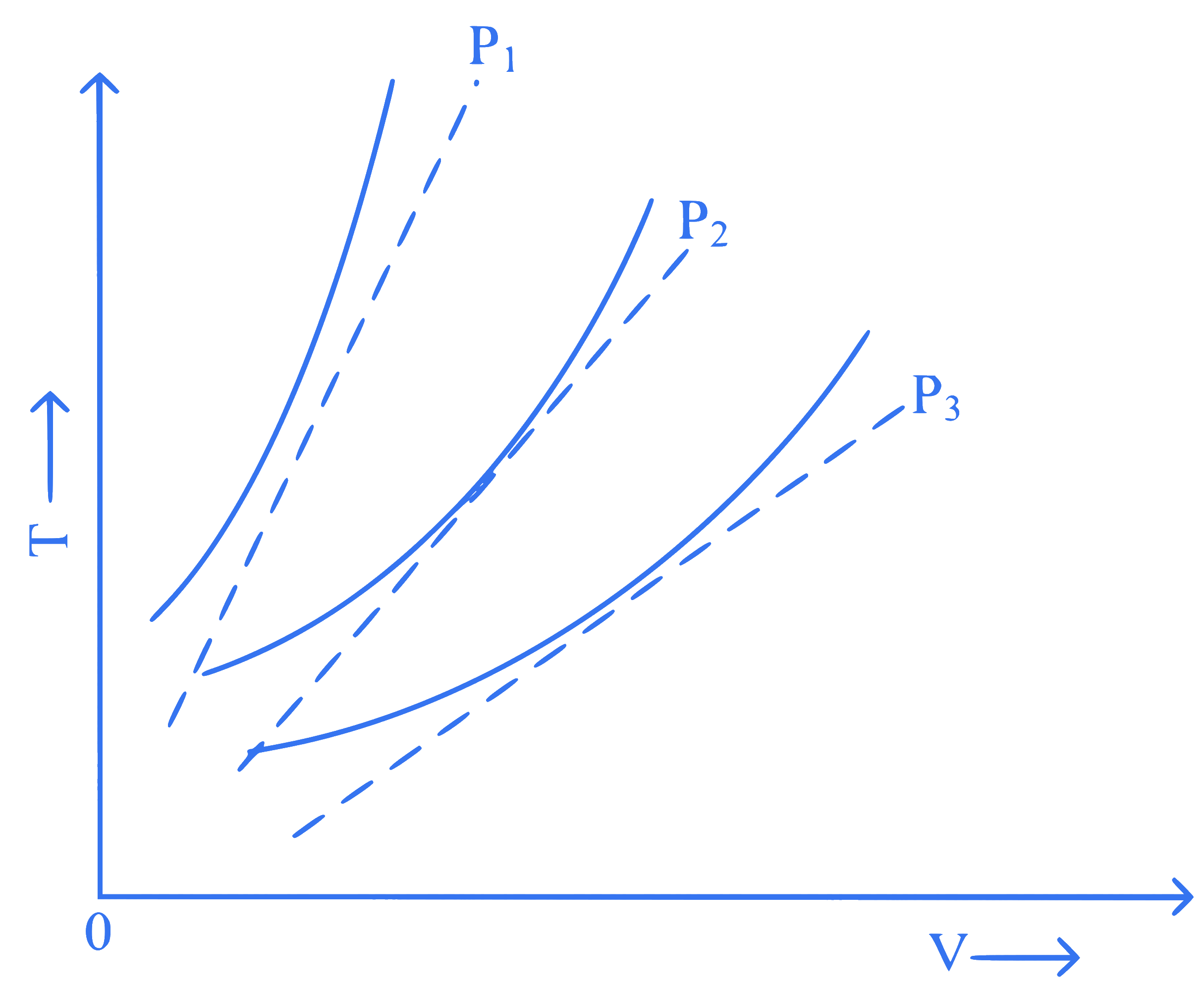 Then the correct relation is :
Then the correct relation is :
 Then the correct relation is :
Then the correct relation is :
Q10.
mcq single
+4 / 1
A thermodynamic system is taken through the cycle $$abcda$$. The work done by the gas along the path $$b c$$ is:
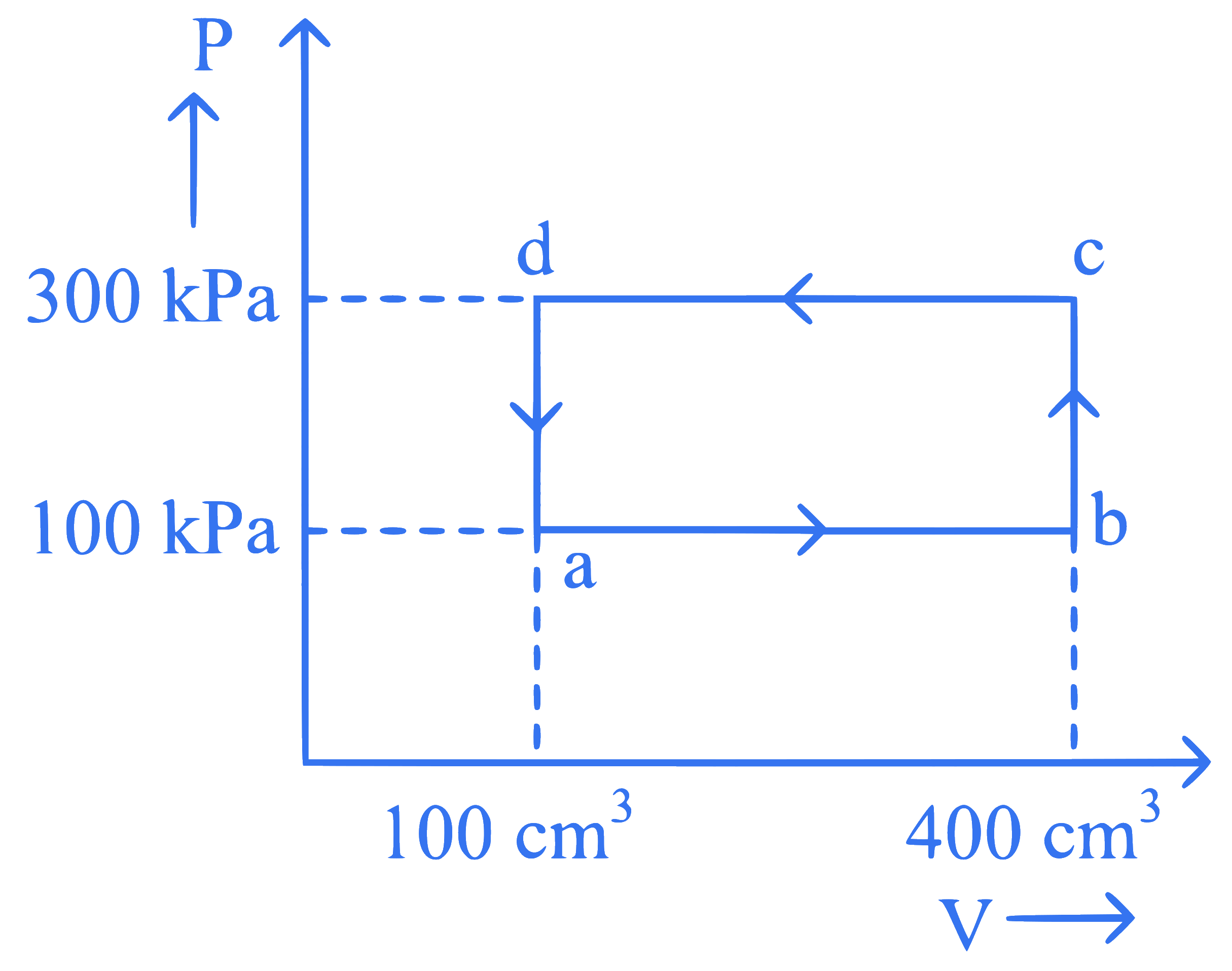

Q11.
mcq single
+4 / 1
A thin flat circular disc of radius $$4.5 \mathrm{~cm}$$ is placed gently over the surface of water. If surface tension of water is $$0.07 \mathrm{~N} \mathrm{~m}^{-1}$$, then the excess force required to take it away from the surface is
Q12.
mcq single
+4 / 1
A metallic bar of Young's modulus, $$0.5 \times 10^{11} \mathrm{~N} \mathrm{~m}^{-2}$$ and coefficient of linear thermal expansion $$10^{-5}{ }^{\circ} \mathrm{C}^{-1}$$, length $$1 \mathrm{~m}$$ and area of cross-section $$10^{-3} \mathrm{~m}^2$$ is heated from $$0^{\circ} \mathrm{C}$$ to $$100^{\circ} \mathrm{C}$$ without expansion or bending. The compressive force developed in it is :
Q13.
mcq single
+4 / 1
The maximum elongation of a steel wire of $$1 \mathrm{~m}$$ length if the elastic limit of steel and its Young's modulus, respectively, are $$8 \times 10^8 \mathrm{~N} \mathrm{~m}^{-2}$$ and $$2 \times 10^{11} \mathrm{~N} \mathrm{~m}^{-2}$$, is:
Q14.
mcq single
+4 / 1
A thin spherical shell is charged by some source. The potential difference between the two points $$C$$ and $$P$$ (in V) shown in the figure is:
(Take $$\frac{1}{4 \pi \varepsilon_0}=9 \times 10^9$$ SI units)
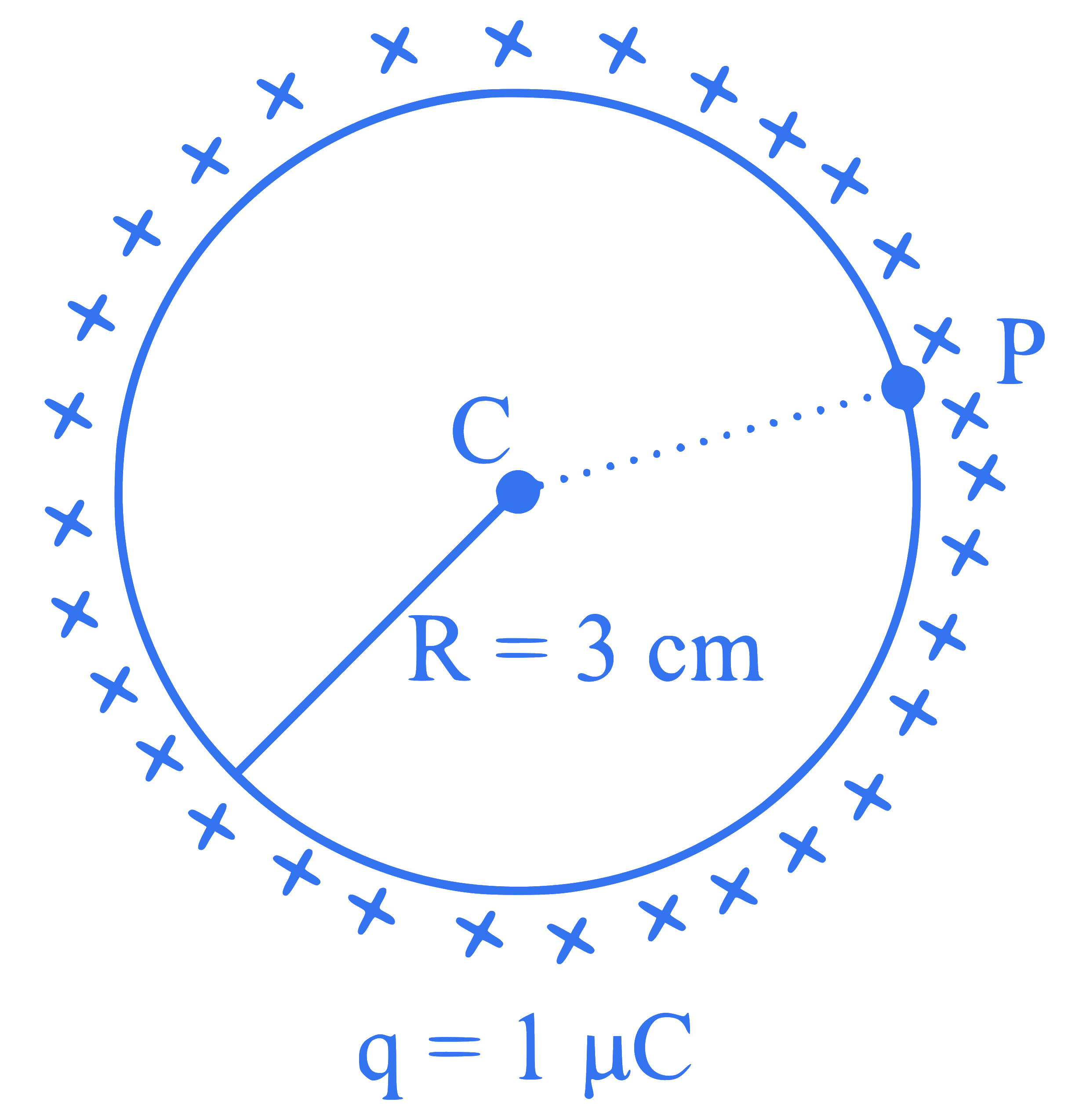

Q15.
mcq single
+4 / 1
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: The potential (V) at any axial point, at $$2 \mathrm{~m}$$ distance $$(r)$$ from the centre of the dipole of dipole moment vector $$\vec{P}$$ of magnitude, $$4 \times 10^{-6} \mathrm{C} \mathrm{m}$$, is $$\pm 9 \times 10^3 \mathrm{~V}$$.
(Take $$\frac{1}{4 \pi \epsilon_0}=9 \times 10^9 \mathrm{SI}$$ units)
Reason R: $$V= \pm \frac{2 P}{4 \pi \epsilon_0 r^2}$$, where $$r$$ is the distance of any axial point, situated at $$2 \mathrm{~m}$$ from the centre of the dipole.
In the light of the above statements, choose the correct answer from the options given below:
Q16.
mcq single
+4 / 1
The minimum energy required to launch a satellite of mass $$m$$ from the surface of earth of mass $$M$$ and radius $$R$$ in a circular orbit at an altitude of $$2 R$$ from the surface of the earth is:
Q17.
mcq single
+4 / 1
The mass of a planet is $$\frac{1}{10}$$th that of the earth and its diameter is half that of the earth. The acceleration due to gravity on that planet is:
Q18.
mcq single
+4 / 1
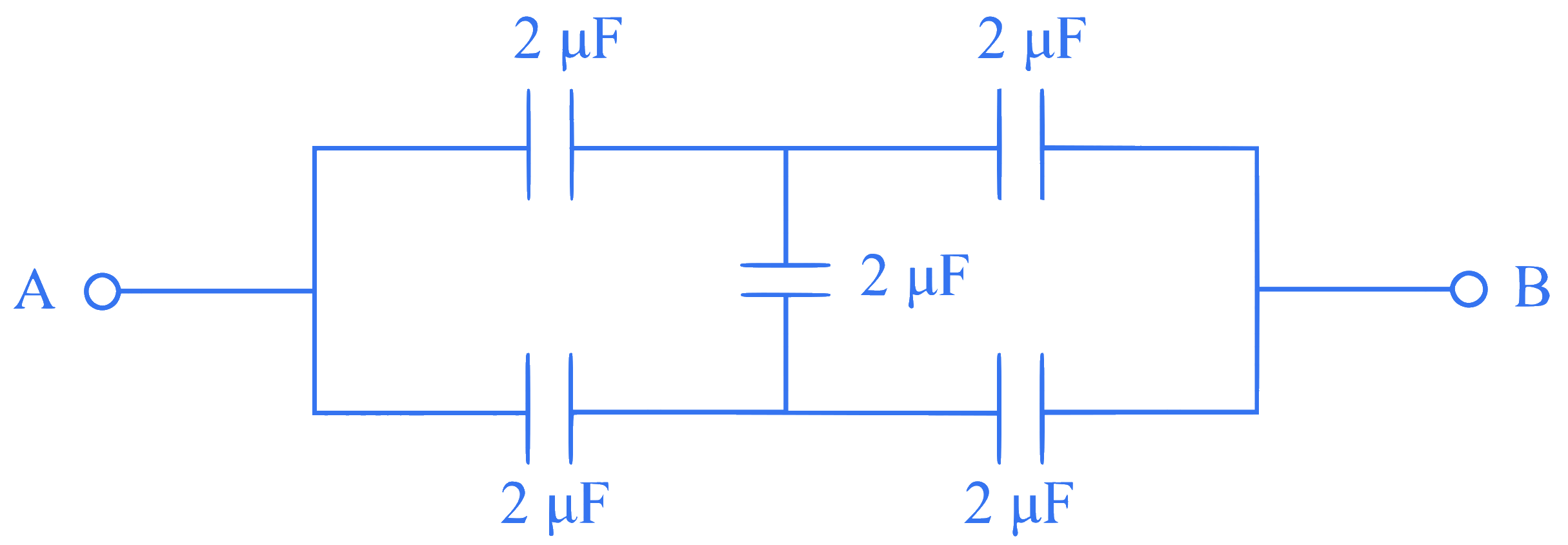
In the following circuit, the equivalent capacitance between terminal A and terminal B is :

Q19.
mcq single
+4 / 1
If the plates of a parallel plate capacitor connected to a battery are moved close to each other, then
A. the charge stored in it, increases.
B. the energy stored in it, decreases.
C. its capacitance increases.
D. the ratio of charge to its potential remains the same.
E. the product of charge and voltage increases.
Choose the most appropriate answer from the options given below:
Q20.
mcq single
+4 / 1
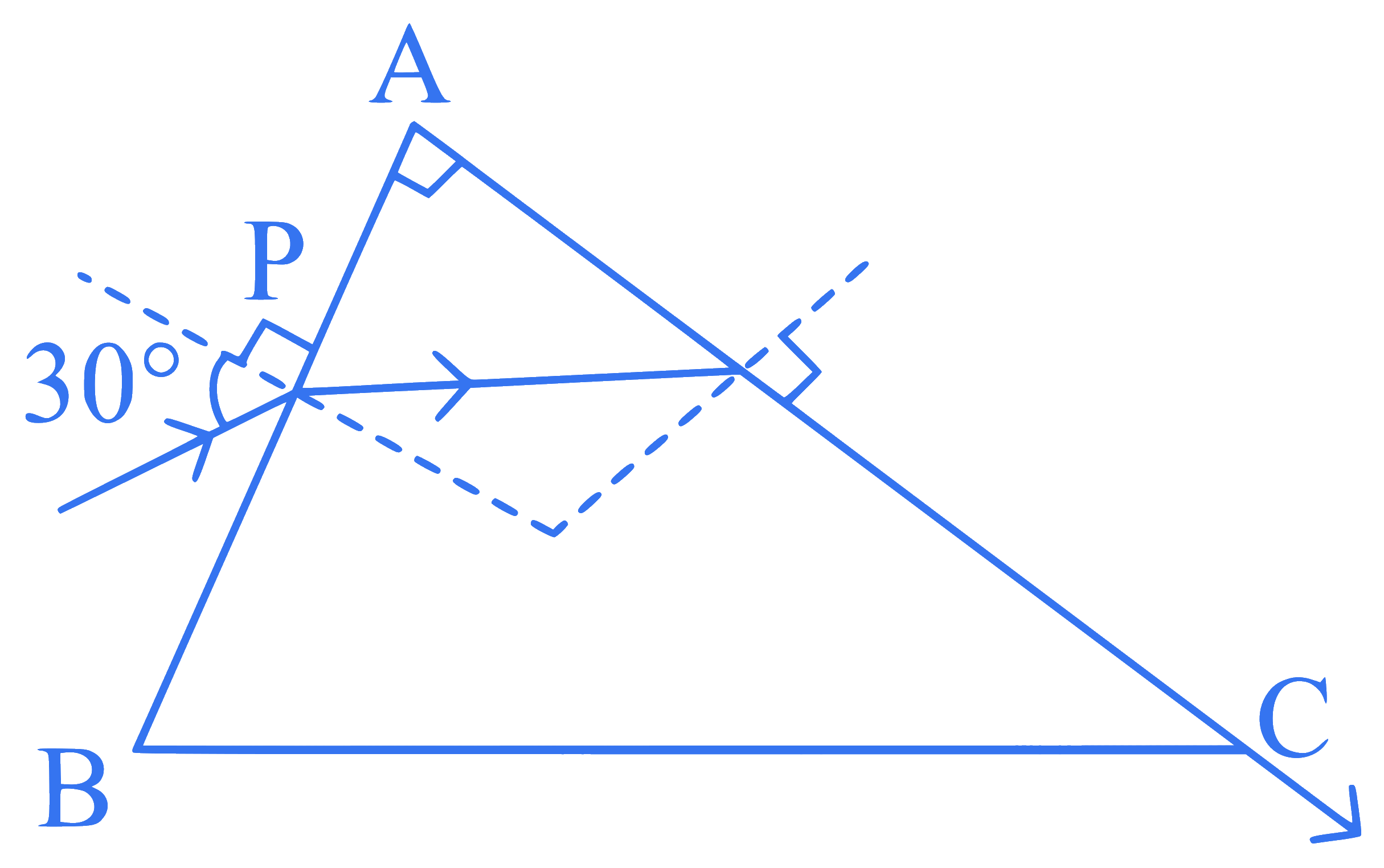
A light ray enters through a right angled prism at point $$P$$ with the angle of incidence $$30^{\circ}$$ as shown in figure. It travels through the prism parallel to its base $$B C$$ and emerges along the face $$A C$$. The refractive index of the prism is:

Q21.
mcq single
+4 / 1
A small telescope has an objective of focal length $$140 \mathrm{~cm}$$ and an eye piece of focal length $$5.0 \mathrm{~cm}$$. The magnifying power of telescope for viewing a distant object is:
Q22.
mcq single
+4 / 1
If $$x=5 \sin \left(\pi t+\frac{\pi}{3}\right) \mathrm{m}$$ represents the motion of a particle executing simple harmonic motion, the amplitude and time period of motion, respectively, are
Q23.
mcq single
+4 / 1
If the mass of the bob in a simple pendulum is increased to thrice its original mass and its length is made half its original length, then the new time period of oscillation is $$\frac{x}{2}$$ times its original time period. Then the value of $$x$$ is:
Q24.
mcq single
+4 / 1
A logic circuit provides the output $$Y$$ as per the following truth table :
 The expression of the output Y is :
The expression of the output Y is :
 The expression of the output Y is :
The expression of the output Y is :
Q25.
mcq single
+4 / 1
Consider the following statements A and B and identify the correct answer :
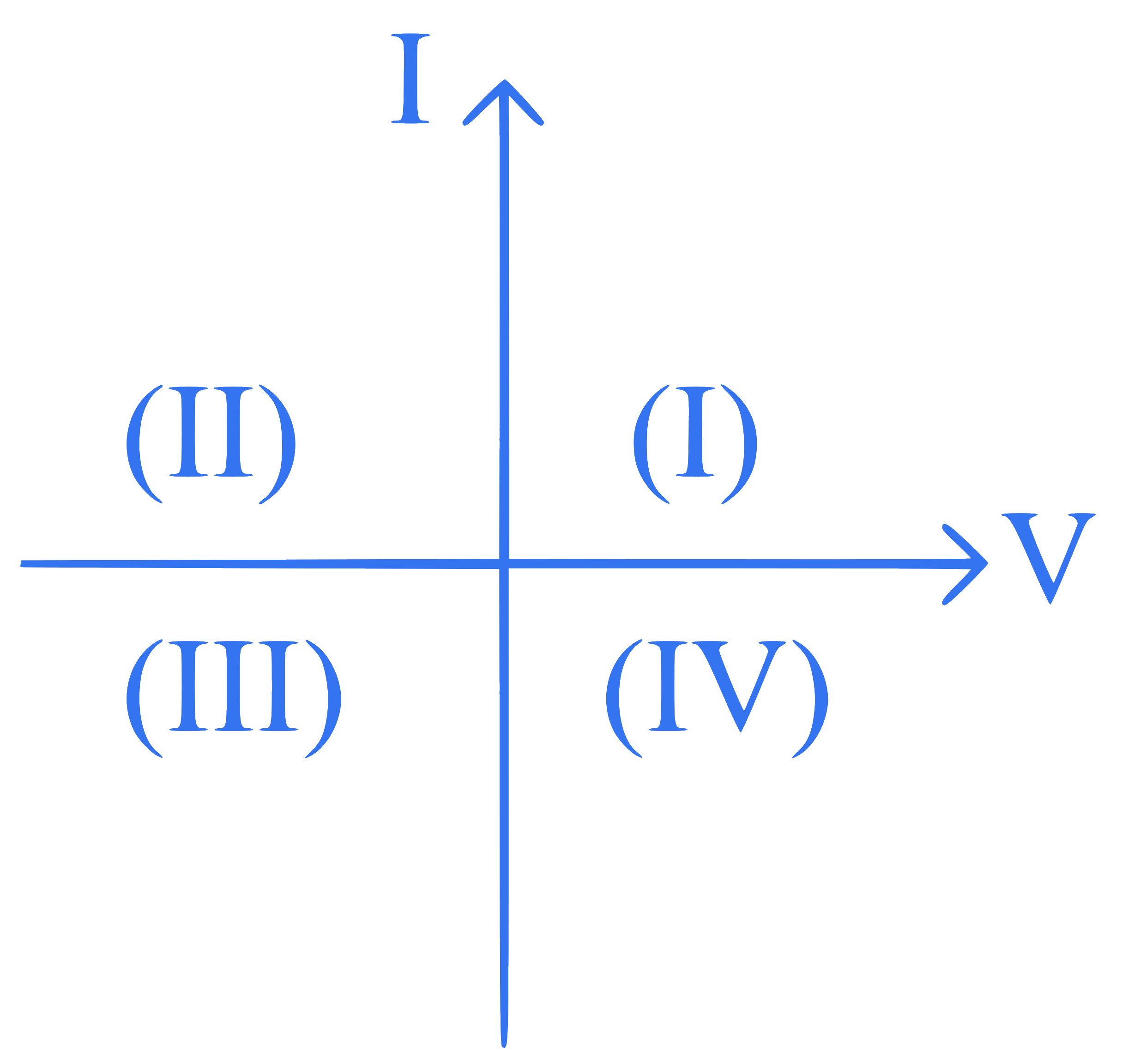 A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
 A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
Q26.
mcq single
+4 / 1
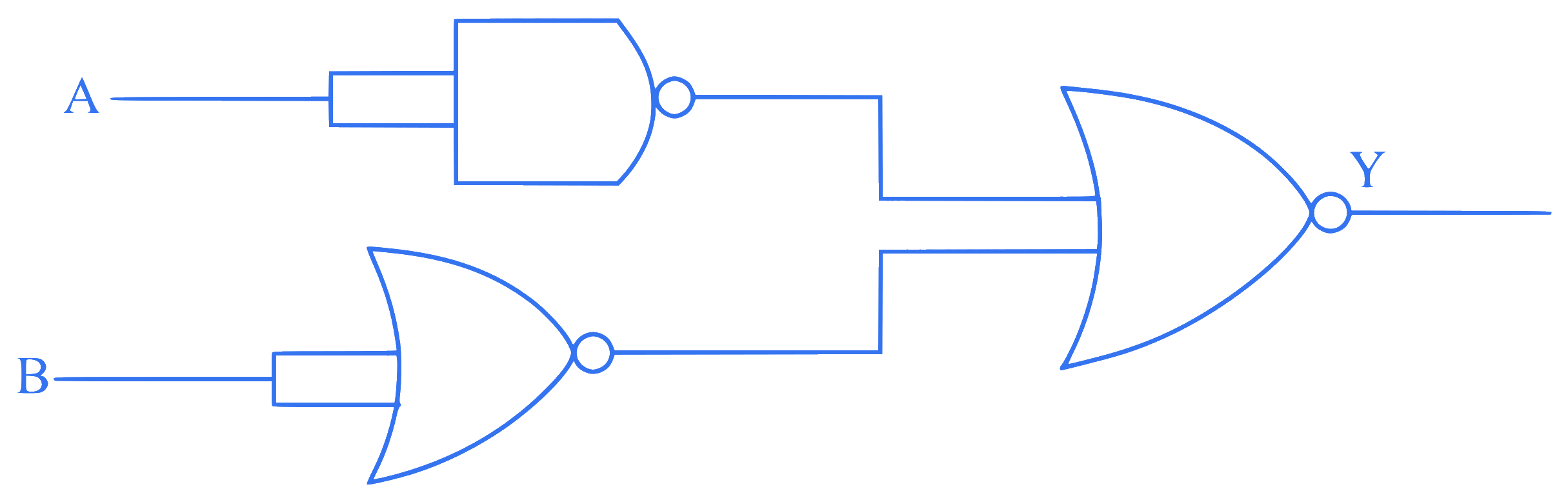
$$
\text { The output ( } Y \text { ) of the given logic gate is similar to the output of an/a }
$$

Q27.
mcq single
+4 / 1
A tightly wound 100 turns coil of radius $$10 \mathrm{~cm}$$ carries a current of $$7 \mathrm{~A}$$. The magnitude of the magnetic field at the centre of the coil is (Take permeability of free space as $$4 \pi \times 10^{-7} \mathrm{SI}$$ units):
Q28.
mcq single
+4 / 1
A parallel plate capacitor is charged by connecting it to a battery through a resistor. If $$I$$ is the current in the circuit, then in the gap between the plates:
Q29.
mcq single
+4 / 1
The graph which shows the variation of $$\left(\frac{1}{\lambda^2}\right)$$ and its kinetic energy, $$E$$ is (where $$\lambda$$ is de Broglie wavelength of a free particle):
Q30.
mcq single
+4 / 1
If $$c$$ is the velocity of light in free space, the correct statements about photon among the following are:
A. The energy of a photon is $$E=h v$$.
B. The velocity of a photon is $$c$$.
C. The momentum of a photon, $$p=\frac{h v}{c}$$.
D. In a photon-electron collision, both total energy and total momentum are conserved.
E. Photon possesses positive charge.
Choose the correct answer from the options given below:
Q31.
mcq single
+4 / 1
The velocity $$(v)-$$ time $$(t)$$ plot of the motion of a body is shown below:
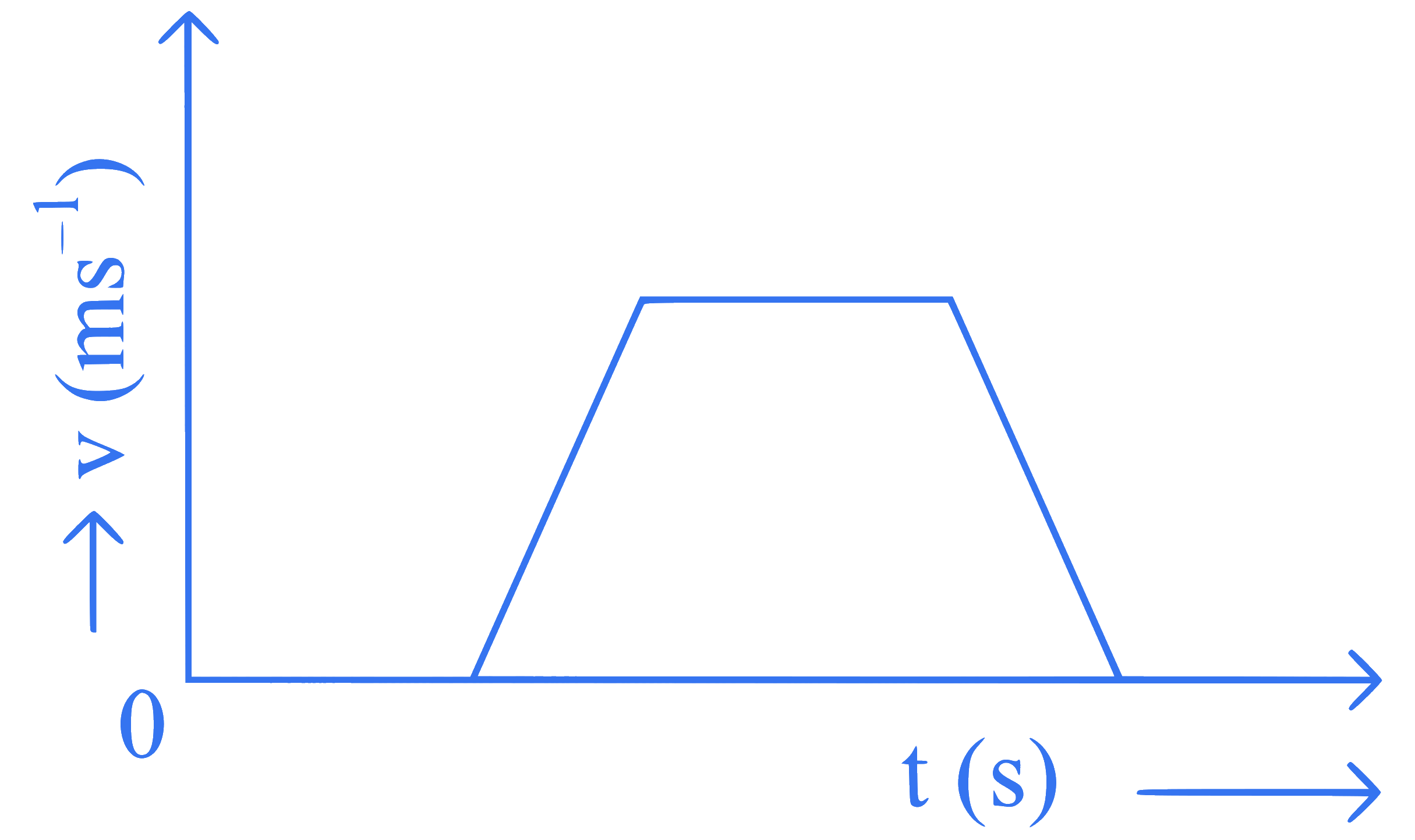 The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
 The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
Q32.
mcq single
+4 / 1
Given below are two statements:
Statement I: Atoms are electrically neutral as they contain equal number of positive and negative charges.
Statement II: Atoms of each element are stable and emit their characteristic spectrum.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q33.
mcq single
+4 / 1
$$
{ }_{82}^{290} X \xrightarrow{\alpha} Y \xrightarrow{e^{+}} Z \xrightarrow{\beta^{-}} P \xrightarrow{e^{-}} Q
$$
In the nuclear emission stated above, the mass number and atomic number of the product $$Q$$ respectively, are
Q34.
mcq single
+4 / 1
Match List I with List II:
List I
(Spectral Lines of Hydrogen for transitions from)
List II
(Wavelengths (nm))
A.
$$
n_2=3 \text { to } n_1=2
$$
I.
410.2
B.
$$
n_2=4 \text { to } n_1=2
$$
II.
434.1
C.
$$
n_2=5 \text { to } n_1=2
$$
III.
656.3
D.
$$
n_2=6 \text { to } n_1=2
$$
IV.
486.1
Choose the correct answer from the options given below:
Q35.
mcq single
+4 / 1
Two bodies $$A$$ and $$B$$ of same mass undergo completely inelastic one dimensional collision. The body $$A$$ moves with velocity $$v_1$$ while body $$B$$ is at rest before collision. The velocity of the system after collision is $$v_2$$. The ratio $$v_1: v_2$$ is
Q36.
mcq single
+4 / 1
A horizontal force $$10 \mathrm{~N}$$ is applied to a block $$A$$ as shown in figure. The mass of blocks $$A$$ and $$B$$ are $$2 \mathrm{~kg}$$ and 3 $$\mathrm{kg}$$ respectively. The blocks slide over a frictionless surface. The force exerted by block $$A$$ on block $$B$$ is :
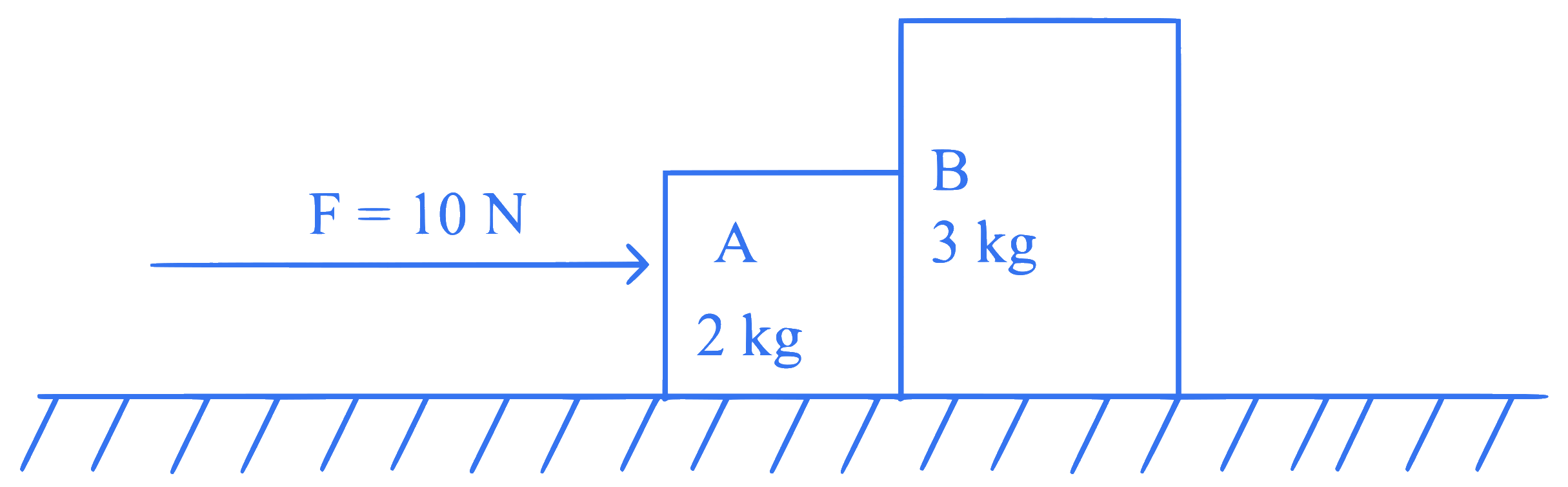

Q37.
mcq single
+4 / 1
A particle moving with uniform speed in a circular path maintains:
Q38.
mcq single
+4 / 1
Match List I with List II
List I
(Material)
List II
(Susceptibility ($$\chi))
A.
Diamagnetic
I.
$$\chi=0$$
B.
Ferromagnetic
II.
$$0 > \chi \ge -1$$
C.
Paramagnetic
III.
$$\chi >> 1$$
D.
Non-magnetic
IV.
$$0 < \chi < \varepsilon$$ (a small positive number)
Choose the correct answer from the options given below.
Q39.
mcq single
+4 / 1
A sheet is placed on a horizontal surface in front of a strong magnetic pole. A force is needed to:
A. hold the sheet there if it is magnetic.
B. hold the sheet there if it is non-magnetic.
C. move the sheet away from the pole with uniform velocity if it is conducting.
D. move the sheet away from the pole with uniform velocity if it is both, non-conducting and non-polar.
Choose the correct statement(s) from the options given below:
Q40.
mcq single
+4 / 1
An iron bar of length $$L$$ has magnetic moment $$M$$. It is bent at the middle of its length such that the two arms make an angle $$60^{\circ}$$ with each other. The magnetic moment of this new magnet is :
Q41.
mcq single
+4 / 1
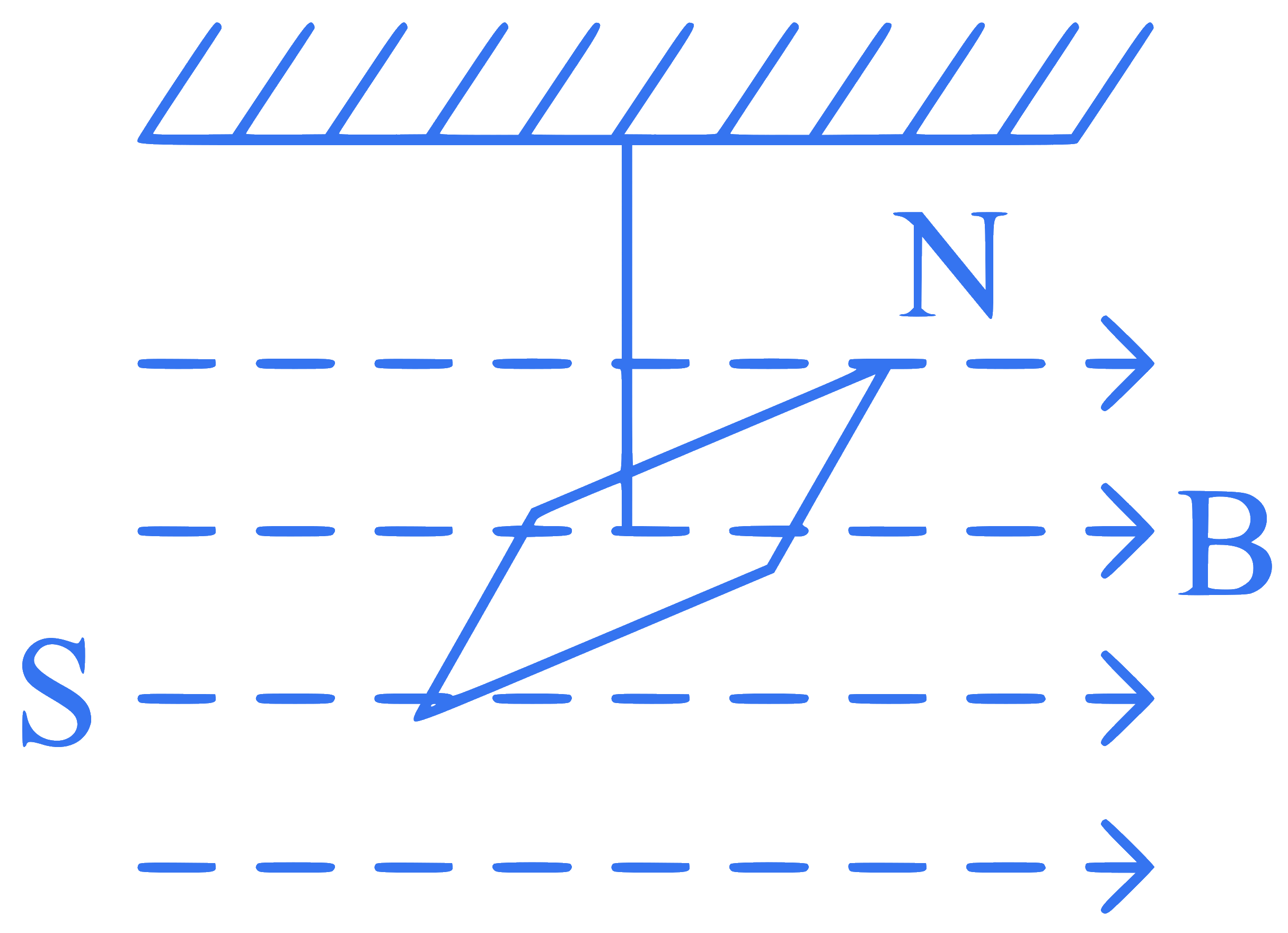
In a uniform magnetic field of $$0.049 \mathrm{~T}$$, a magnetic needle performs 20 complete oscillations in 5 seconds as shown. The moment of inertia of the needle is $$9.8 \times 10^{-6} \mathrm{~kg} \mathrm{~m}^2$$. If the magnitude of magnetic moment of the needle is $$x \times 10^{-5} \mathrm{~Am}^2$$, then the value of '$$x$$' is :

Q42.
mcq single
+4 / 1
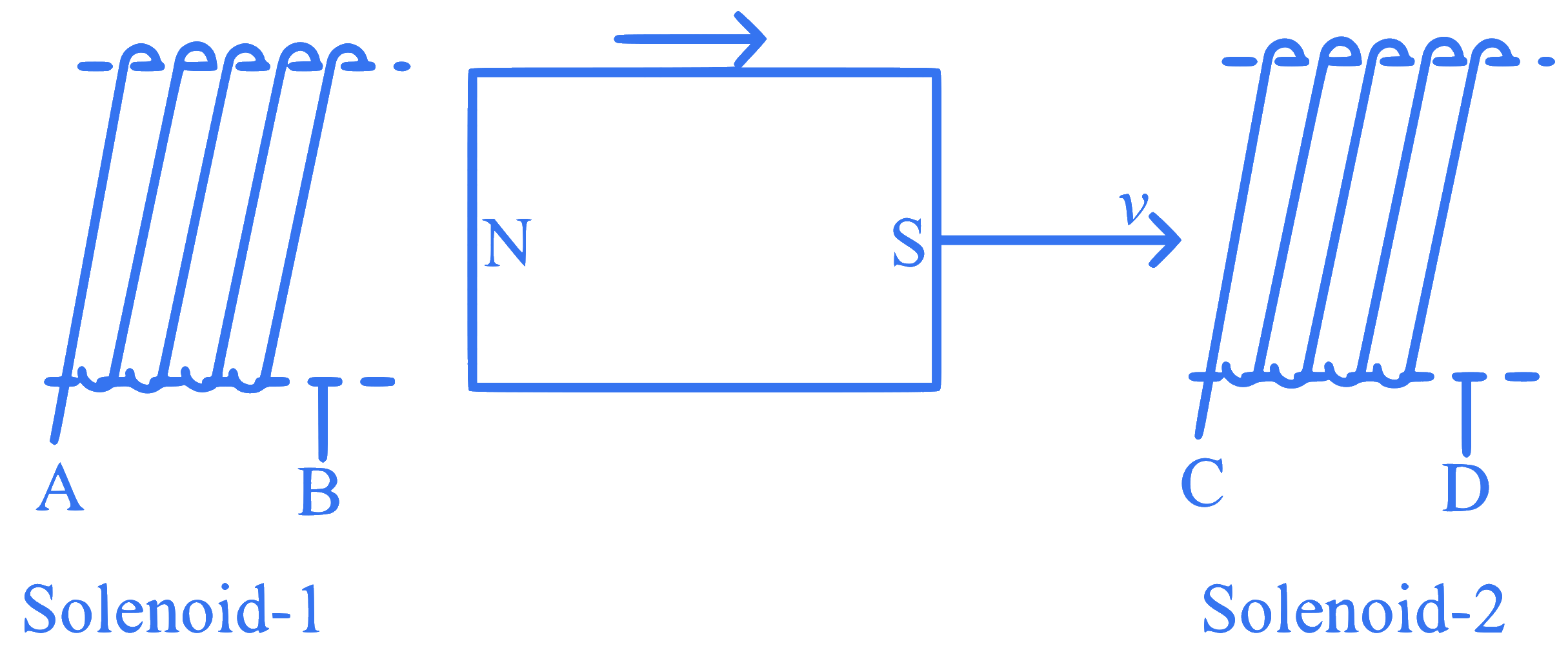 In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
 In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
Q43.
mcq single
+4 / 1
At any instant of time $$t$$, the displacement of any particle is given by $$2 t-1$$ ($$\mathrm{SI}$$ unit) under the influence of force of $$5 \mathrm{~N}$$. The value of instantaneous power is (in $$\mathrm{SI}$$ unit):
Q44.
mcq single
+4 / 1
Two heaters $$A$$ and $$B$$ have power rating of $$1 \mathrm{~kW}$$ and $$2 \mathrm{~kW}$$, respectively. Those two are first connected in series and then in parallel to a fixed power source. The ratio of power outputs for these two cases is:
Q45.
mcq single
+4 / 1
A wire of length '$$l$$' and resistance $$100 \Omega$$ is divided into 10 equal parts. The first 5 parts are connected in series while the next 5 parts are connected in parallel. The two combinations are again connected in series. The resistance of this final combination is:
Q46.
mcq single
+4 / 1
Choose the correct circuit which can achieve the bridge balance.
Q47.
mcq single
+4 / 1
The terminal voltage of the battery, whose emf is $$10 \mathrm{~V}$$ and internal resistance $$1 \Omega$$, when connected through an external resistance of $$4 \Omega$$ as shown in the figure is:
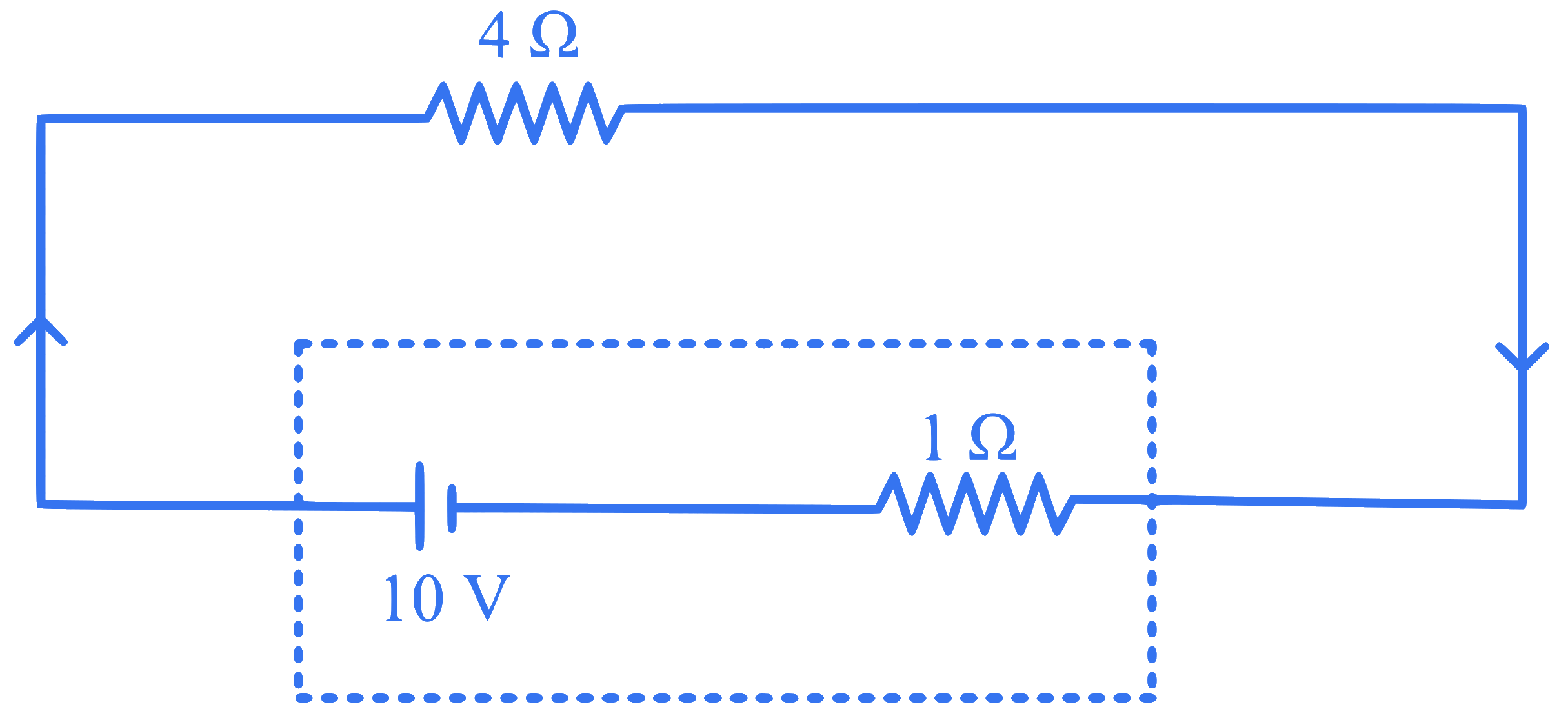

Q48.
mcq single
+4 / 1
The property which is not of an electromagnetic wave travelling in free space is that:
Q49.
mcq single
+4 / 1
In an ideal transformer, the turns ratio is $$\frac{N_P}{N_S}=\frac{1}{2}$$. The ratio $$V_S: V_P$$ is equal to (the symbols carry their usual meaning) :
Q50.
mcq single
+4 / 1
A $$10 \mu \mathrm{F}$$ capacitor is connected to a $$210 \mathrm{~V}, 50 \mathrm{~Hz}$$ source as shown in figure. The peak current in the circuit is nearly $$(\pi=3.14)$$ :
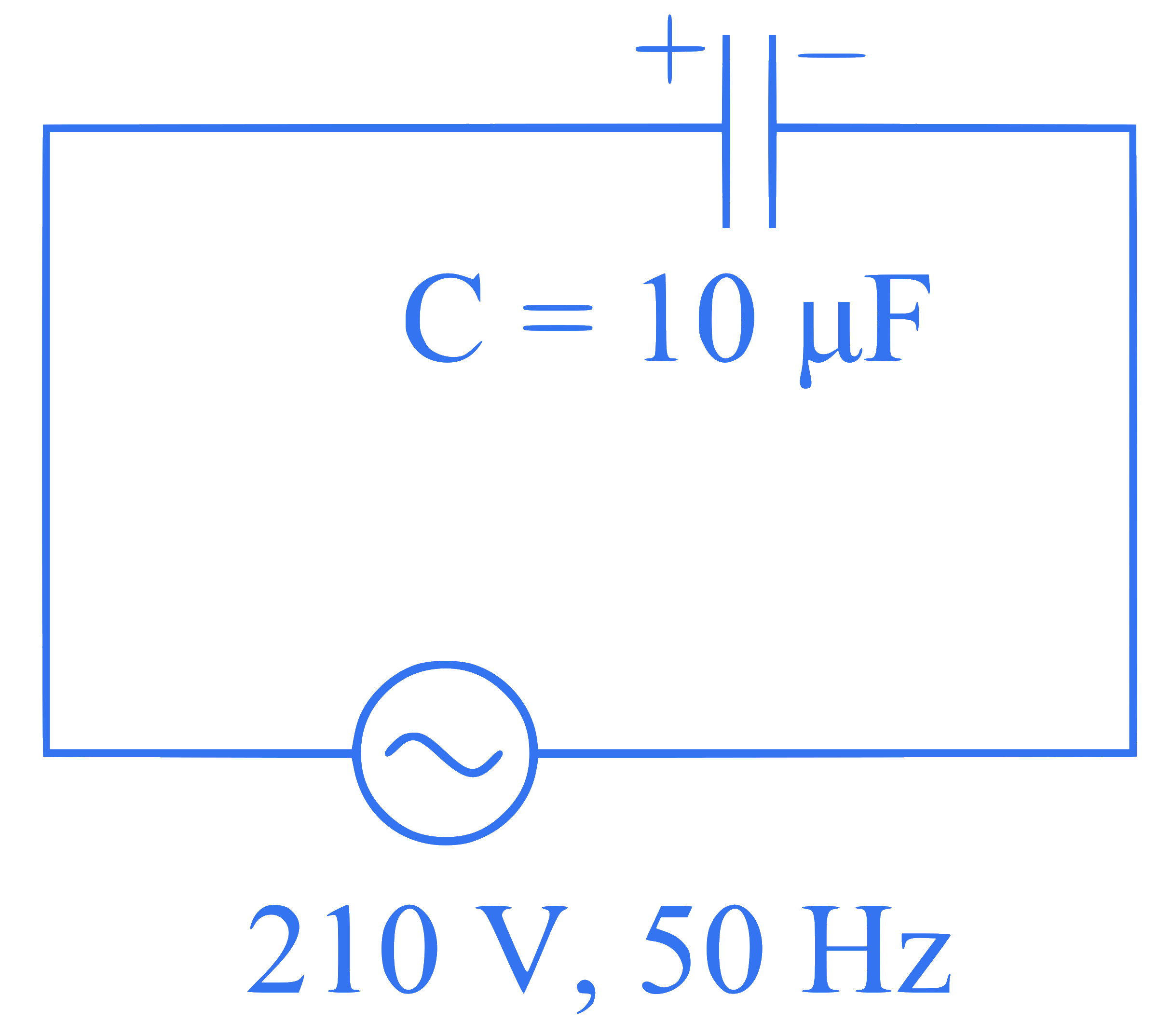

Q51.
mcq single
+4 / 1
If the monochromatic source in Young's double slit experiment is replaced by white light, then
Q52.
mcq single
+4 / 1
An unpolarised light beam strikes a glass surface at Brewster's angle. Then
Q53.
mcq single
+4 / 1
A bob is whirled in a horizontal plane by means of a string with an initial speed of $$\omega \mathrm{~rpm}$$. The tension in the string is $$T$$. If speed becomes $$2 \omega$$ while keeping the same radius, the tension in the string becomes:
Q54.
mcq single
+4 / 1
A wheel of a bullock cart is rolling on a level road as shown in the figure below. If its linear speed is $$v$$ in the direction shown, which one of the following options is correct ($$P$$ and $$Q$$ are any highest and lowest points on the wheel, respectively)?


Q55.
mcq single
+4 / 1
The moment of inertia of a thin rod about an axis passing through its mid point and perpendicular to the rod is $$2400 \mathrm{~g} \mathrm{~cm}^2$$. The length of the $$400 \mathrm{~g}$$ rod is nearly:
Q56.
mcq single
+4 / 1
In a vernier callipers, $$(N+1)$$ divisions of vernier scale coincide with $$N$$ divisions of main scale. If $$1 \mathrm{~MSD}$$ represents $$0.1 \mathrm{~mm}$$, the vernier constant (in $$\mathrm{cm}$$) is:
Q57.
mcq single
+4 / 1
The quantities which have the same dimensions as those of solid angle are:
Q58.
mcq single
+4 / 1
A force defined by $$F=\alpha t^2+\beta t$$ acts on a particle at a given time $$t$$. The factor which is dimensionless, if $$\alpha$$ and $$\beta$$ are constants, is:
Q59.
mcq single
+4 / 1
The following graph represents the $$T$$-$$V$$ curves of an ideal gas (where $$T$$ is the temperature and $$V$$ the volume) at three pressures $$P_1, P_2$$ and $$P_3$$ compared with those of Charles's law represented as dotted lines.
 Then the correct relation is :
Then the correct relation is :
 Then the correct relation is :
Then the correct relation is :
Q60.
mcq single
+4 / 1
A thermodynamic system is taken through the cycle $$abcda$$. The work done by the gas along the path $$b c$$ is:


Q61.
mcq single
+4 / 1
A thin flat circular disc of radius $$4.5 \mathrm{~cm}$$ is placed gently over the surface of water. If surface tension of water is $$0.07 \mathrm{~N} \mathrm{~m}^{-1}$$, then the excess force required to take it away from the surface is
Q62.
mcq single
+4 / 1
A metallic bar of Young's modulus, $$0.5 \times 10^{11} \mathrm{~N} \mathrm{~m}^{-2}$$ and coefficient of linear thermal expansion $$10^{-5}{ }^{\circ} \mathrm{C}^{-1}$$, length $$1 \mathrm{~m}$$ and area of cross-section $$10^{-3} \mathrm{~m}^2$$ is heated from $$0^{\circ} \mathrm{C}$$ to $$100^{\circ} \mathrm{C}$$ without expansion or bending. The compressive force developed in it is :
Q63.
mcq single
+4 / 1
The maximum elongation of a steel wire of $$1 \mathrm{~m}$$ length if the elastic limit of steel and its Young's modulus, respectively, are $$8 \times 10^8 \mathrm{~N} \mathrm{~m}^{-2}$$ and $$2 \times 10^{11} \mathrm{~N} \mathrm{~m}^{-2}$$, is:
Q64.
mcq single
+4 / 1
A thin spherical shell is charged by some source. The potential difference between the two points $$C$$ and $$P$$ (in V) shown in the figure is:
(Take $$\frac{1}{4 \pi \varepsilon_0}=9 \times 10^9$$ SI units)


Q65.
mcq single
+4 / 1
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: The potential (V) at any axial point, at $$2 \mathrm{~m}$$ distance $$(r)$$ from the centre of the dipole of dipole moment vector $$\vec{P}$$ of magnitude, $$4 \times 10^{-6} \mathrm{C} \mathrm{m}$$, is $$\pm 9 \times 10^3 \mathrm{~V}$$.
(Take $$\frac{1}{4 \pi \epsilon_0}=9 \times 10^9 \mathrm{SI}$$ units)
Reason R: $$V= \pm \frac{2 P}{4 \pi \epsilon_0 r^2}$$, where $$r$$ is the distance of any axial point, situated at $$2 \mathrm{~m}$$ from the centre of the dipole.
In the light of the above statements, choose the correct answer from the options given below:
Q66.
mcq single
+4 / 1
The minimum energy required to launch a satellite of mass $$m$$ from the surface of earth of mass $$M$$ and radius $$R$$ in a circular orbit at an altitude of $$2 R$$ from the surface of the earth is:
Q67.
mcq single
+4 / 1
The mass of a planet is $$\frac{1}{10}$$th that of the earth and its diameter is half that of the earth. The acceleration due to gravity on that planet is:
Q68.
mcq single
+4 / 1
In the following circuit, the equivalent capacitance between terminal A and terminal B is :


Q69.
mcq single
+4 / 1
If the plates of a parallel plate capacitor connected to a battery are moved close to each other, then
A. the charge stored in it, increases.
B. the energy stored in it, decreases.
C. its capacitance increases.
D. the ratio of charge to its potential remains the same.
E. the product of charge and voltage increases.
Choose the most appropriate answer from the options given below:
Q70.
mcq single
+4 / 1
A light ray enters through a right angled prism at point $$P$$ with the angle of incidence $$30^{\circ}$$ as shown in figure. It travels through the prism parallel to its base $$B C$$ and emerges along the face $$A C$$. The refractive index of the prism is:


Q71.
mcq single
+4 / 1
A small telescope has an objective of focal length $$140 \mathrm{~cm}$$ and an eye piece of focal length $$5.0 \mathrm{~cm}$$. The magnifying power of telescope for viewing a distant object is:
Q72.
mcq single
+4 / 1
If $$x=5 \sin \left(\pi t+\frac{\pi}{3}\right) \mathrm{m}$$ represents the motion of a particle executing simple harmonic motion, the amplitude and time period of motion, respectively, are
Q73.
mcq single
+4 / 1
If the mass of the bob in a simple pendulum is increased to thrice its original mass and its length is made half its original length, then the new time period of oscillation is $$\frac{x}{2}$$ times its original time period. Then the value of $$x$$ is:
Q74.
mcq single
+4 / 1
A logic circuit provides the output $$Y$$ as per the following truth table :
 The expression of the output Y is :
The expression of the output Y is :
 The expression of the output Y is :
The expression of the output Y is :
Q75.
mcq single
+4 / 1
Consider the following statements A and B and identify the correct answer :
 A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
 A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
A. For a solar-cell, the I-V characteristics lies in the IV quadrant of the given graph.
B. In a reverse biased $$p n$$ junction diode, the current measured in $$(\mu \mathrm{A})$$, is due to majority charge carriers.
Q76.
mcq single
+4 / 1
$$
\text { The output ( } Y \text { ) of the given logic gate is similar to the output of an/a }
$$


Q77.
mcq single
+4 / 1
A tightly wound 100 turns coil of radius $$10 \mathrm{~cm}$$ carries a current of $$7 \mathrm{~A}$$. The magnitude of the magnetic field at the centre of the coil is (Take permeability of free space as $$4 \pi \times 10^{-7} \mathrm{SI}$$ units):
Q78.
mcq single
+4 / 1
A parallel plate capacitor is charged by connecting it to a battery through a resistor. If $$I$$ is the current in the circuit, then in the gap between the plates:
Q79.
mcq single
+4 / 1
The graph which shows the variation of $$\left(\frac{1}{\lambda^2}\right)$$ and its kinetic energy, $$E$$ is (where $$\lambda$$ is de Broglie wavelength of a free particle):
Q80.
mcq single
+4 / 1
If $$c$$ is the velocity of light in free space, the correct statements about photon among the following are:
A. The energy of a photon is $$E=h v$$.
B. The velocity of a photon is $$c$$.
C. The momentum of a photon, $$p=\frac{h v}{c}$$.
D. In a photon-electron collision, both total energy and total momentum are conserved.
E. Photon possesses positive charge.
Choose the correct answer from the options given below:
Q81.
mcq single
+4 / 1
The velocity $$(v)-$$ time $$(t)$$ plot of the motion of a body is shown below:
 The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
 The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
The acceleration $$(a)-$$ time $$(t)$$ graph that best suits this motion is :
Q82.
mcq single
+4 / 1
Given below are two statements:
Statement I: Atoms are electrically neutral as they contain equal number of positive and negative charges.
Statement II: Atoms of each element are stable and emit their characteristic spectrum.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q83.
mcq single
+4 / 1
$$
{ }_{82}^{290} X \xrightarrow{\alpha} Y \xrightarrow{e^{+}} Z \xrightarrow{\beta^{-}} P \xrightarrow{e^{-}} Q
$$
In the nuclear emission stated above, the mass number and atomic number of the product $$Q$$ respectively, are
Q84.
mcq single
+4 / 1
Match List I with List II:
List I
(Spectral Lines of Hydrogen for transitions from)
List II
(Wavelengths (nm))
A.
$$
n_2=3 \text { to } n_1=2
$$
I.
410.2
B.
$$
n_2=4 \text { to } n_1=2
$$
II.
434.1
C.
$$
n_2=5 \text { to } n_1=2
$$
III.
656.3
D.
$$
n_2=6 \text { to } n_1=2
$$
IV.
486.1
Choose the correct answer from the options given below:
Q85.
mcq single
+4 / 1
Two bodies $$A$$ and $$B$$ of same mass undergo completely inelastic one dimensional collision. The body $$A$$ moves with velocity $$v_1$$ while body $$B$$ is at rest before collision. The velocity of the system after collision is $$v_2$$. The ratio $$v_1: v_2$$ is
Q86.
mcq single
+4 / 1
A horizontal force $$10 \mathrm{~N}$$ is applied to a block $$A$$ as shown in figure. The mass of blocks $$A$$ and $$B$$ are $$2 \mathrm{~kg}$$ and 3 $$\mathrm{kg}$$ respectively. The blocks slide over a frictionless surface. The force exerted by block $$A$$ on block $$B$$ is :


Q87.
mcq single
+4 / 1
A particle moving with uniform speed in a circular path maintains:
Q88.
mcq single
+4 / 1
Match List I with List II
List I
(Material)
List II
(Susceptibility ($$\chi))
A.
Diamagnetic
I.
$$\chi=0$$
B.
Ferromagnetic
II.
$$0 > \chi \ge -1$$
C.
Paramagnetic
III.
$$\chi >> 1$$
D.
Non-magnetic
IV.
$$0 < \chi < \varepsilon$$ (a small positive number)
Choose the correct answer from the options given below.
Q89.
mcq single
+4 / 1
A sheet is placed on a horizontal surface in front of a strong magnetic pole. A force is needed to:
A. hold the sheet there if it is magnetic.
B. hold the sheet there if it is non-magnetic.
C. move the sheet away from the pole with uniform velocity if it is conducting.
D. move the sheet away from the pole with uniform velocity if it is both, non-conducting and non-polar.
Choose the correct statement(s) from the options given below:
Q90.
mcq single
+4 / 1
An iron bar of length $$L$$ has magnetic moment $$M$$. It is bent at the middle of its length such that the two arms make an angle $$60^{\circ}$$ with each other. The magnetic moment of this new magnet is :
Q91.
mcq single
+4 / 1
In a uniform magnetic field of $$0.049 \mathrm{~T}$$, a magnetic needle performs 20 complete oscillations in 5 seconds as shown. The moment of inertia of the needle is $$9.8 \times 10^{-6} \mathrm{~kg} \mathrm{~m}^2$$. If the magnitude of magnetic moment of the needle is $$x \times 10^{-5} \mathrm{~Am}^2$$, then the value of '$$x$$' is :


Q92.
mcq single
+4 / 1
 In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
 In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
In the above diagram, a strong bar magnet is moving towards solenoid-2 from solenoid-1. The direction of induced current in solenoid-1 and that in solenoid-2, respectively, are through the directions:
Q93.
mcq single
+4 / 1
At any instant of time $$t$$, the displacement of any particle is given by $$2 t-1$$ ($$\mathrm{SI}$$ unit) under the influence of force of $$5 \mathrm{~N}$$. The value of instantaneous power is (in $$\mathrm{SI}$$ unit):
Q94.
mcq single
+4 / 1
Two heaters $$A$$ and $$B$$ have power rating of $$1 \mathrm{~kW}$$ and $$2 \mathrm{~kW}$$, respectively. Those two are first connected in series and then in parallel to a fixed power source. The ratio of power outputs for these two cases is:
Q95.
mcq single
+4 / 1
A wire of length '$$l$$' and resistance $$100 \Omega$$ is divided into 10 equal parts. The first 5 parts are connected in series while the next 5 parts are connected in parallel. The two combinations are again connected in series. The resistance of this final combination is:
Q96.
mcq single
+4 / 1
Choose the correct circuit which can achieve the bridge balance.
Q97.
mcq single
+4 / 1
The terminal voltage of the battery, whose emf is $$10 \mathrm{~V}$$ and internal resistance $$1 \Omega$$, when connected through an external resistance of $$4 \Omega$$ as shown in the figure is:


Q98.
mcq single
+4 / 1
The property which is not of an electromagnetic wave travelling in free space is that:
Q99.
mcq single
+4 / 1
In an ideal transformer, the turns ratio is $$\frac{N_P}{N_S}=\frac{1}{2}$$. The ratio $$V_S: V_P$$ is equal to (the symbols carry their usual meaning) :
Q100.
mcq single
+4 / 1
A $$10 \mu \mathrm{F}$$ capacitor is connected to a $$210 \mathrm{~V}, 50 \mathrm{~Hz}$$ source as shown in figure. The peak current in the circuit is nearly $$(\pi=3.14)$$ :


Biology
Biology
Q1.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Clostridium butylicum
I.
Ethanol
B.
Saccharomyces cerevisiae
II.
Streptokinase
C.
Trichoderma polysporum
III.
Butyric acid
D.
Streptococcus sp.
IV.
Cyclosporin-A
Choose the correct answer from the options given below:
Q2.
mcq single
+4 / 1
In an ecosystem if the Net Primary Productivity (NPP) of first trophic level is $$100 x\left(\mathrm{kcal} \mathrm{~m}^{-2}\right) \mathrm{yr}^{-1}$$, what would be the GPP (Gross Primary Productivity) of the third trophic level of the same ecosystem?
Q3.
mcq single
+4 / 1
In a plant, black seed color $$(\mathrm{BB} / \mathrm{Bb})$$ is dominant over white seed color $$(\mathrm{bb})$$. In order to find out the genotype of the black seed plant, with which of the following genotype will you cross it?
Q4.
mcq single
+4 / 1
As per ABO blood grouping system, the blood group of father is $$\mathrm{B}^{+}$$, mother is $$\mathrm{A}^{+}$$ and child is $$\mathrm{O}^{+}$$. Their respective genotype can be
A. I^(B)i/I^(A)i/ii
B. I^(B)I^(B)/I^(A)I^(A)/ii
C. iI^(B)/iI^(A)/I^(A)IB^()
D. I^(A)i/I^(B)i/I^(A)i
E. iI^(B)/iI^(A)/I^(A)I^(B)
Choose the most appropriate answer from the options given below :
Q5.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Two or more alternative forms of a gene
I.
Back cross
B.
Cross of F$$_1$$ progeny with homozygous recessive parent
II.
Ploidy
C.
Cross of F$$_1$$ progeny with any of the parents
III.
Allele
D.
Number of chromosome sets in plant
IV.
Test cross
Choose the correct answer from the options given below:
Q6.
mcq single
+4 / 1
Which one of the following can be explained on the basis of Mendel's Law of Dominance?
A. Out of one pair of factors one is dominant and the other is recessive.
B. Alleles do not show any expression and both the characters appear as such in F$$_2$$ generation.
C. Factors occur in pairs in normal diploid plants.
D. The discrete unit controlling a particular character is called factor.
E. The expression of only one of the parental characters is found in a monohybrid cross.
Choose the correct answer from the options given below:
Q7.
mcq single
+4 / 1
A pink flowered Snapdragon plant was crossed with a red flowered Snapdragon plant. What type of phenotype/s is/are expected in the progeny?
Q8.
mcq single
+4 / 1
In both sexes of cockroach, a pair of jointed filamentous structures called anal cerci are present on
Q9.
mcq single
+4 / 1
Match List I with List II related to digestive system of cockroach.
List - I
List - II
A.
The structures used for storing of food
I.
Gizzaard
B.
Ring of 6-8 blind tubules at junction of foregut and midgut.
II.
Gastric Caeca
C.
Ring of 100-150 yellow coloured thin filaments at junction of midgut and hindgut.
III.
Malpighian tubules
D.
The structures used for grinding the food.
IV.
Crop
Choose the correct answer from the options given below:
Q10.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Unicellular glandular epithelium
I.
Salivary glands
B.
Compound epithelium
II.
Pancreas
C.
Multicellular glandular epithelium
III.
Goblet cells of alimentary canal
D.
Endocrine glandular epithelium
IV.
Moist surface of buccal cavity
Choose the correct answer from the options given below:
Q11.
mcq single
+4 / 1
Given below are two statements:
Statement I: In $$\mathrm{C}_3$$ plants, some $$\mathrm{O}_2$$ binds to $$\mathrm{RuBisCO}$$, hence $$\mathrm{CO}_2$$ fixation is decreased.
Statement II: In $$\mathrm{C}_4$$ plants, mesophyll cells show very little photorespiration while bundle sheath cells do not show photorespiration.
In the light of the above statements, choose the correct answer from the options given below:
Q12.
mcq single
+4 / 1
Which of the following are required for the dark reaction of photosynthesis?
A. Light
B. Chlorophyll
C. $$\mathrm{CO}_2$$
D. ATP
E. NADPH
Choose the correct answer from the options given below:
Q13.
mcq single
+4 / 1
How many molecules of ATP and NADPH are required for every molecule of $$\mathrm{CO}_2$$ fixed in the Calvin cycle?
Q14.
mcq single
+4 / 1
The following are the statements about non-chordates:
A. Pharynx is perforated by gill slits.
B. Notochord is absent.
C. Central nervous system is dorsal.
D. Heart is dorsal if present.
E. Post anal tail is absent.
Choose the most appropriate answer from the options given below:
Q15.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Pleurobrachia
I.
Mollusca
B.
Radula
II.
Ctenophora
C.
Stomochord
III.
Osteichthyes
D.
Air bladder
IV.
Hemichordata
Choose the correct answer from the options given below :
Q16.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Pterophyllum
I.
Hag fish
B.
Myxine
II.
Saw fish
C.
Pristis
III.
Angel fish
D.
Exocoetus
IV.
Flying fish
Choose the correct answer from the options given below :
Q17.
mcq single
+4 / 1
Consider the following statements :
A. Annelids are true coelomates
B. Poriferans are pseudocoelomates
C. Aschelminthes are acoelomates
D. Platyhelminthes are pseudocoelomates
Choose the correct answer from the options given below :
Q18.
mcq single
+4 / 1
Read the following statements and choose the set of correct statements:
In the members of Phaeophyceae,
A. Asexual reproduction occurs usually by biflagellate zoospores.
B. Sexual reproduction is by oogamous method only.
C. Stored food is in the form of carbohydrates which is either mannitol or laminarin.
D. The major pigments found are chlorophyll a, c and carotenoids and xanthophyll.
E. Vegetative cells have a cellulosic wall, usually covered on the outside by gelatinous coating of algin.
Choose the correct answer from the options given below:
Q19.
mcq single
+4 / 1
Which of the following factors are favourable for the formation of oxyhaemoglobin in alveoli?
Q20.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Expiratory capacity
I.
Expiratory reserve volume + Tidal volume + Inspiratory reserve volume
B.
Functional residual capacity
II.
Tidal volume + Expiratory reserve volume
C.
Vital capacity
III.
Tidal volume + Inspiratory reserve volume
D.
Inspiratory capacity
IV.
Expiratory reserve volume + Residual volume
Choose the correct answer from the options given below :
Q21.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
$$\alpha$$ -I antitrypsin
I.
Cotton bollworm
B.
Cry IAb
II.
ADA deficiency
C.
Cry IAc
III.
Emphysema
D.
Enzyme replacement therapy
IV.
Corn borer
Choose the correct answer form the options given below:
Q22.
mcq single
+4 / 1
The following diagram showing restriction sites in E. coli cloning vector pBR322. Find the role of ‘X’ and ‘Y’ genes :
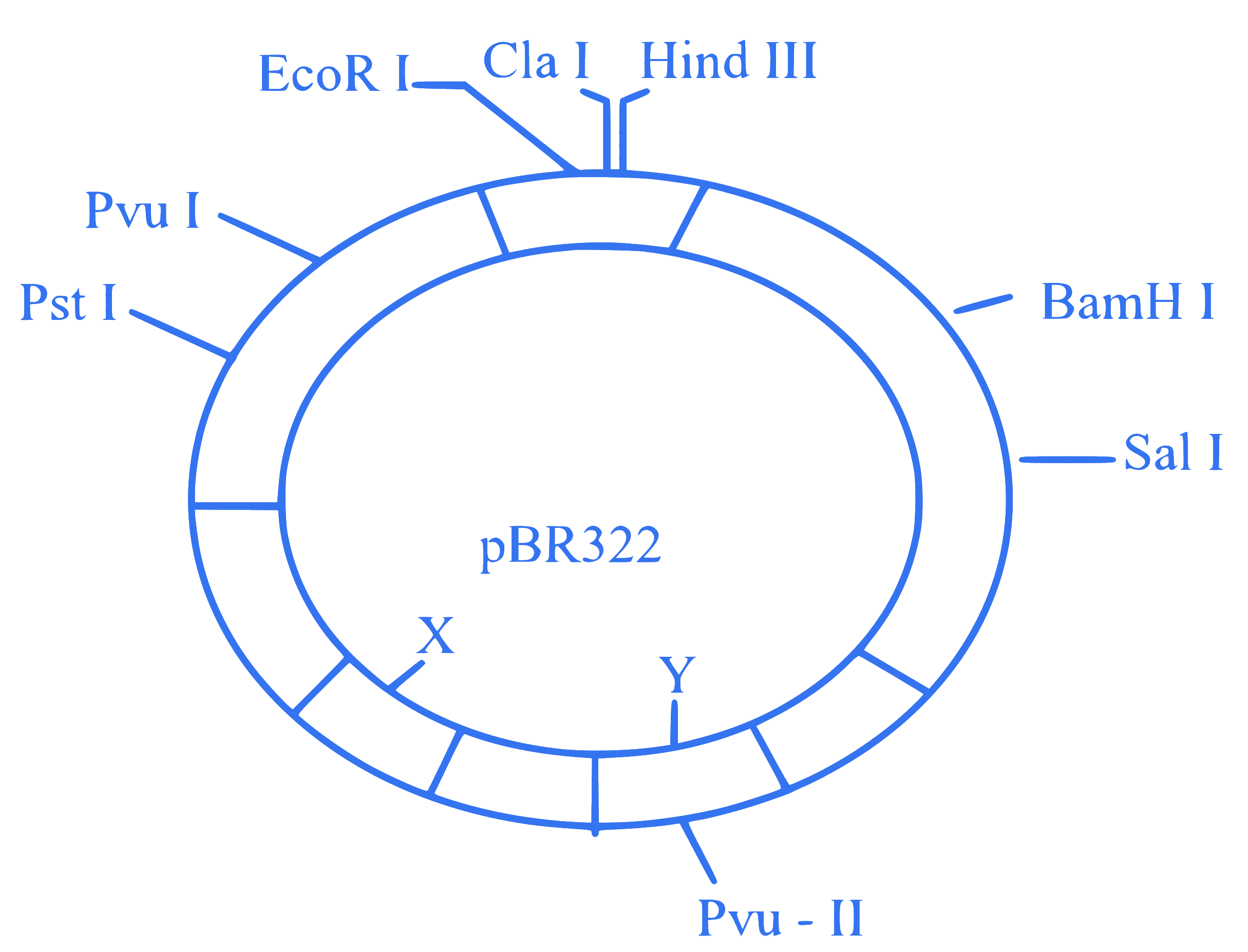

Q23.
mcq single
+4 / 1
Given below are two statements:
Statement I : Bt toxins are insect group specific and coded by a gene cry IAc.
Statement II : Bt toxin exists as inactive protoxin in B. thuringiensis. However, after ingestion by the insect the inactive protoxin gets converted into active form due to acidic $$\mathrm{pH}$$ of the insect gut.
In the light of the above statements, choose the correct answer from the options given below:
Q24.
mcq single
+4 / 1
The capacity to generate a whole plant from any cell of the plant is called:
Q25.
mcq single
+4 / 1
What is the fate of a piece of DNA carrying only gene of interest which is transferred into an alien organism?
A. The piece of DNA would be able to multiply itself independently in the progeny cells of the organism.
B. It may get integrated into the genome of the recipient.
C. It may multiply and be inherited along with the host DNA.
D. The alien piece of DNA is not an integral part of chromosome.
E. It shows ability to replicate.
Choose the correct answer from the options given below:
Q26.
mcq single
+4 / 1
Which of the following statements is incorrect?
Q27.
mcq single
+4 / 1
Which of the following are fused in somatic hybridization involving two varieties of plants?
Q28.
mcq single
+4 / 1
Given below are two statements:
Statement I: The cerebral hemispheres are connected by nerve tract known as corpus callosum.
Statement II: The brain stem consists of the medulla oblongata, pons and cerebrum.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q29.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Pons
I.
Provides additional space for Neurons, regulates posture and balance.
B.
Hypothalamus
II.
Controls respiration and gastric secretions.
C.
Medulla
III.
Connects different regions of the brain.
D.
Cerebellum
IV.
Neuro secretory cells
Choose the correct answer from the options given below :
Q30.
mcq single
+4 / 1
In the given figure, which component has thin outer walls and highly thickened inner walls?
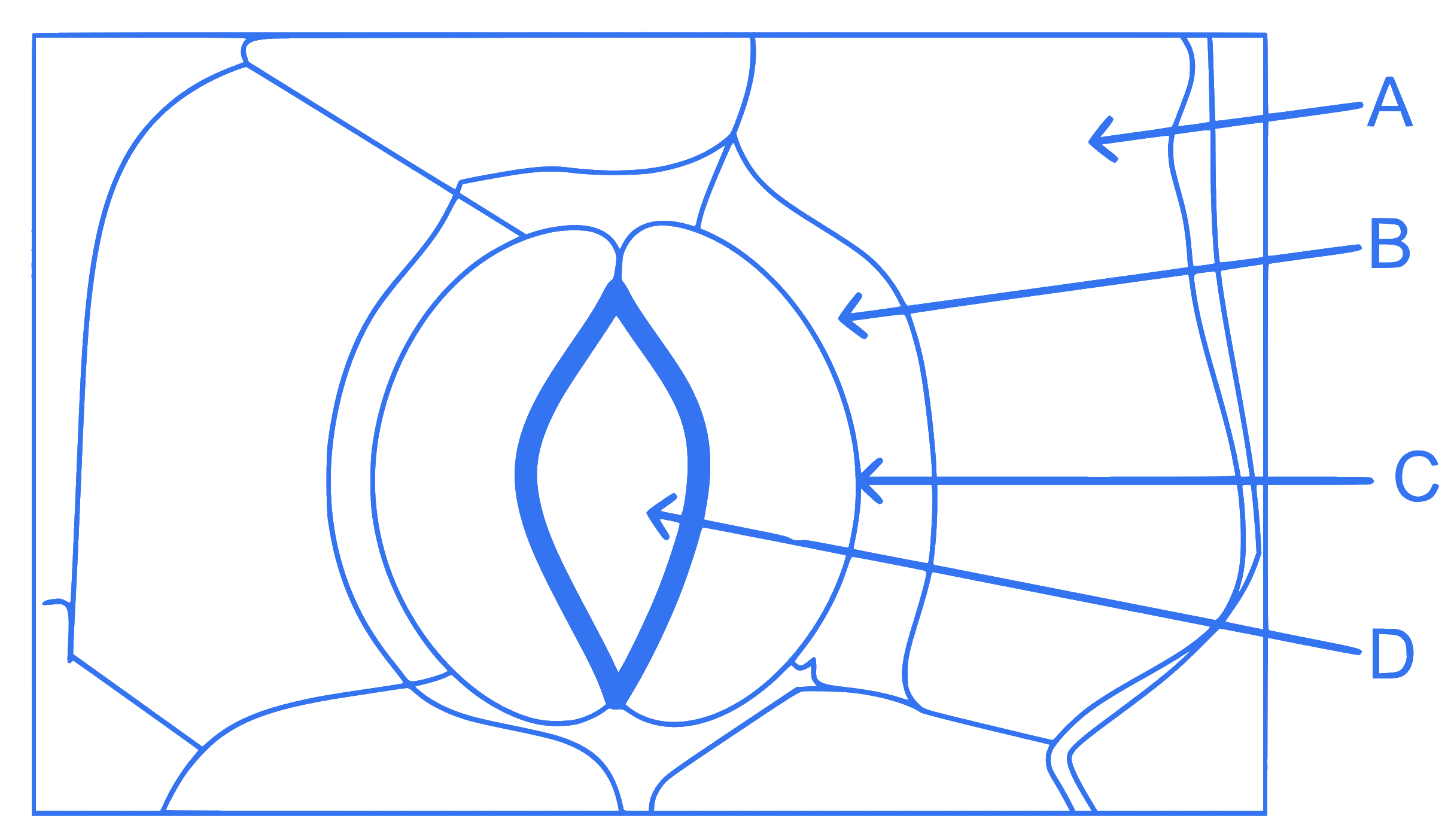

Q31.
mcq single
+4 / 1
Given below are two statements:
Statement I : Parenchyma is living but collenchyma is dead tissue.
Statement II : Gymnosperms lack xylem vessels but presence of xylem vessels is the characteristic of angiosperms.
In the light of the above statements, choose the correct answer from the options given below:
Q32.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Axoneme
I.
Centriole
B.
Cartwheel pattern
II.
Cilia and flagella
C.
Crista
III.
Chromosome
D.
Satellite
IV.
Mitochondria
Choose the correct answer from the options given below :
Q33.
mcq single
+4 / 1
Given below are two statements:
Statement I: Mitochondria and chloroplasts both double membranes bound organelles.
Statement II: Inner membrane of mitochondria is relatively less permeable, as compared chloroplast.
In the light of the above statements, choose the mis appropriate answer from the options given below:
Q34.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Nucleolus
I.
Site of formation of glycolipid
B.
Centriole
II.
Organization like the cartwheel
C.
Leucoplasts
III.
Site of active ribosomal RNA synthesis
D.
Golgi apparatus
IV.
For storing nutrients
Choose the correct answer from the options given below:
Q35.
mcq single
+4 / 1
The DNA present in chloroplast is:
Q36.
mcq single
+4 / 1
The lactose present in the growth medium of bacteria is transported to the cell by the action of
Q37.
mcq single
+4 / 1
Which one is the correct product of DNA dependent RNA polymerase to the given template?
3'TACATGGCAAATATCCATTCA5'
Q38.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Frederick Griffith
I.
Genetic code
B.
Francois Jacob & Jacque Monod
II.
Semi-conservative mode of DNA replication
C.
Har Gobind Khorana
III.
Tranformation
D.
Meselson & Stahl
IV.
Lac operon
Choose the correct answer from the options given below:
Q39.
mcq single
+4 / 1
A transcription unit in DNA is defined primarily by the three regions in DNA and these are with respect to upstream and down stream end;
Q40.
mcq single
+4 / 1
Which of the following statement is correct regarding the process of replication in E.coli?
Q41.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Down's syndrome
I.
11$$^\mathrm{th}$$ chromosome
B.
$$\alpha$$-thalassemia
II.
'X' chromosome
C.
$$\beta$$-thalassemia
III.
21$$^\mathrm{st}$$ chromosome
D.
Klinefelter's syndrome
IV.
16$$^\mathrm{th}$$ chromosome
Choose the correct answer from the options given below :
Q42.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
RNA polymerase III
I.
snRNPs
B.
Termination of transcription
II.
Promotor
C.
Splicing of Exons
III.
Rho factor
D.
TATA box
IV.
SnRNAs, tRNA
Choose the correct answer from the options given below :
Q43.
mcq single
+4 / 1
Given below are two statements :
Statement I : Bone marrow is the main lymphoid organ where all blood cells including lymphocytes are produced.
Statement II : Both bone marrow and thymus provide micro environments for the development and maturation of T-lymphocytes.
In the light of above statements, choose the most appropriate answer from the options given below
Q44.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Cocaine
I.
Effective sedative in surgery
B.
Heroin
II.
Cannabis sativa
C.
Morphine
III.
Erythroxylum
D.
Marijuana
IV.
Papaver somniferum
Choose the correct answer from the options given below:
Q45.
mcq single
+4 / 1
Which of the following are Autoimmune disorders?
A. Myasthenia gravis
B. Rheumatoid arthritis
C. Gout
D. Muscular dystrophy
E. Systemic Lupus Erythematosus (SLE)
Choose the most appropriate answer from the options given below:
Q46.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Typhoid
I.
Fungus
B.
Leishmaniasis
II.
Nematode
C.
Ringworm
III.
Protozoa
D.
Filariasis
IV.
Bacteria
Choose the correct answer from the options given below:
Q47.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Common cold
I.
Plasmodium
B.
Haemozoin
II.
Typhoid
C.
Widal test
III.
Rhinoviruses
D.
Allergy
IV.
Dust mites
Choose the correct answer from the options given below :
Q48.
mcq single
+4 / 1
Which one of the following factors will not affect the Hardy-Weinberg equilibrium?
Q49.
mcq single
+4 / 1
Given below are two statements:
Statement I: Gause's competitive exclusion principle states that two closely related species competing for different resources cannot exist indefinitely.
Statement II: According to Gause's principle, during competition, the inferior will be eliminated. This may be true if resources are limiting.
In the light of the above statements, choose the correct answer from the options given below :
Q50.
mcq single
+4 / 1
The equation of Verhulst-Pearl logistic growth is $$\frac{\mathrm{dN}}{\mathrm{dt}}=\mathrm{rN}\left[\frac{\mathrm{K}-\mathrm{N}}{\mathrm{K}}\right]$$.
From this equation, $$\mathrm{K}$$ indicates:
Q51.
mcq single
+4 / 1
Following are the stages of pathway for conduction of an action potential through the heart
A. AV bundle
B. Purkinje fibres
C. AV node
D. Bundle branches
E. SA node
Choose the correct sequence of pathway from the options given below
Q52.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
P wave
(i)
Heart muscles are electrically silent.
B.
QRS complex
(ii)
Depolarisation of ventricles.
C.
T wave
(iii)
Depolarisation of atria.
D.
T-P gap
(iv)
Repolarisation of ventricles.
Choose the correct answer from the options given below :
Q53.
mcq single
+4 / 1
Following are the stages of cell division :
A. Gap 2 phase
B. Cytokinesis
C. Synthesis phase
D. Karyokinesis
E. Gap 1 phase
Choose the correct sequence of stages from the options given below :
Q54.
mcq single
+4 / 1
Spindle fibers attach to kinetochores of chromosomes during
Q55.
mcq single
+4 / 1
Match List I with List II :
List - I
(Sub Phases of Prophase I)
List - II
(Specific Characters)
A.
Diakinesis
I.
Synaptonemal complex formation
B.
Pachytene
II.
Completion of terminalisation of chiasmata
C.
Zygotene
III.
Chromosomes look like thin threads
D.
Leptotene
IV.
Appearance of recombination nodules
Choose the correct answer from the options given below
Q56.
mcq single
+4 / 1
Given below are two statements:
Statement I : Chromosomes become gradually visible under light microscope during leptotene stage.
Statement II : The beginning of diplotene stage is recognized by dissolution of synaptonemal complex.
In the light of the above statements, choose the correct answer from the options given below:
Q57.
mcq single
+4 / 1
Identify the correct option (A), (B), (C), (D) with respect to spermatogenesis.
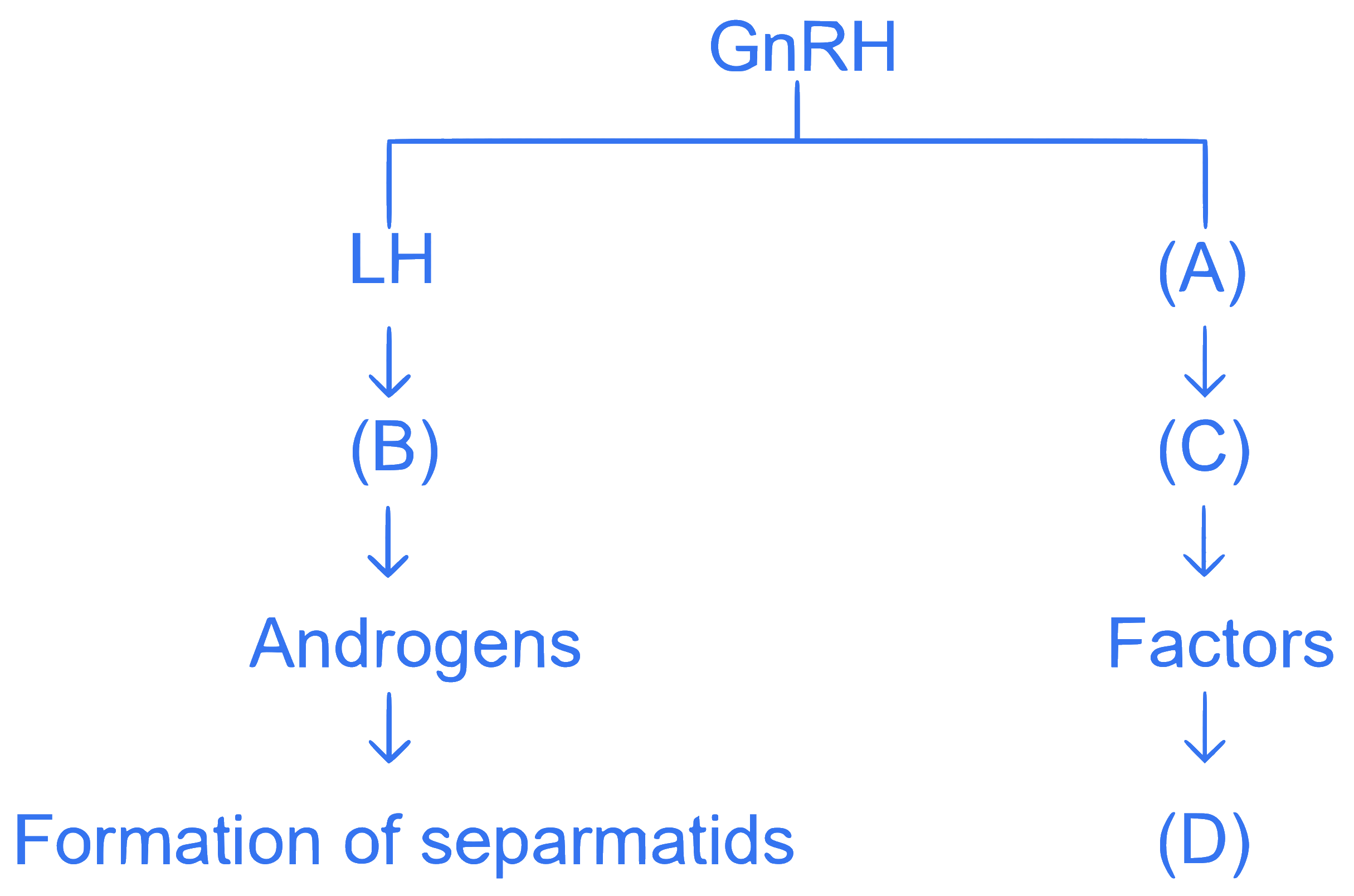

Q58.
mcq single
+4 / 1
Given below are two statements:
Statement I: The presence or absence of hymen is not a reliable indicator of virginity.
Statement II: The hymen is torn during the first coitus only.
In the light of the above above statements, choose the correct answer from the options given below :
Q59.
mcq single
+4 / 1
Which of the following is not a component of Fallopian tube?
Q60.
mcq single
+4 / 1
Which of the following is not a steroid hormone?
Q61.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason R :
Assertion A : FSH acts upon ovarian follicles in female and Leydig cells in male.
Reason R : Growing ovarian follicles secrete estrogen in female while interstitial cells secrete androgen in male human being.
In the light of the above statements, choose the correct answer from the options given below :
Q62.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Exophthalmic goiter
I.
Excess secretion of cortisol, moon face \& hypergylcemia.
B.
Acromegaly
II.
Hypo-secretion of thyroid hormone and stunted growth.
C.
Cushing's syndrome
III.
Hyper secretion of thyroid hormone \& protruding eye balls.
D.
Cretinism
IV.
Excessive secretion of growth hormone.
Choose the correct answer from the options given below :
Q63.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Robert May
I.
Species-Area relationship
B.
Alexander von Humboldt
II.
Long term ecosystem experiment using out door plots
C.
Paul Ehrlich
III.
Global species diversity at about 7 million
D.
David Tilman
IV.
Rivet popper hypothesis
Choose the correct answer from the options given below:
Q64.
mcq single
+4 / 1
Tropical regions show greatest level of species richness because
A. Tropical latitudes have remained relatively undisturbed for millions of years, hence more time was available for species diversification.
B. Tropical environments are more seasonal.
C. More solar energy is available in tropics.
D. Constant environments promote niche specialization.
E. Tropical environments are constant and predictable.
Choose the correct answer from the options given below.
Q65.
mcq single
+4 / 1
The type of conservation in which the threatened species are taken out from their natural habitat and placed in special setting where they can be protected and given special care is called
Q66.
mcq single
+4 / 1
List of endangered species was released by
Q67.
mcq single
+4 / 1
These are regarded as major causes of biodiversity loss:
A. Over exploitation
B. Co-extinction
C. Mutation
D. Habitat loss and fragmentation
E. Migration
Choose the correct option:
Q68.
mcq single
+4 / 1
Spraying sugarcane crop with which of the following plant growth regulators, increases the length of stem, thus, increasing the yield?
Q69.
mcq single
+4 / 1
Auxin is used by gardeners to prepare weed-free lawns. But no damage is caused to grass as auxin
Q70.
mcq single
+4 / 1
Formation of interfascicular cambium from fully developed parenchyma cells is an example for
Q71.
mcq single
+4 / 1
Identify the step in tricarboxylic acid cycle, which does not involve oxidation of substrate.
Q72.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Citric acid cycle
I.
Cytoplasm
B.
Glycolysis
II.
Mitochondrial matrix
C.
Electron transport system
III.
Intermembrane space of mitochondria
D.
Proton gradient
IV.
Inner mitochondrial membrane
Choose the correct answer from the options given below:
Q73.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Fibrous joints
I.
Adjacent vertebrae, limited movement
B.
Cartilaginous joints
II.
Humerus and Pectoral girdle, rotational movement
C.
Hinge joints
III.
Skull, don't allow any movement
D.
Ball and socket joints
IV.
Knee, help in locomotion
Choose the correct answer from the options given below :
Q74.
mcq single
+4 / 1
Three types of muscles are given as a, b and c. Identify the correct matching pair along with their location in human body:
 Name of muscle/location
Name of muscle/location
 Name of muscle/location
Name of muscle/location
Q75.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Rhizopus
I.
Mushroom
B.
Ustilago
II.
Smut fungus
C.
Puccinia
III.
Bread mould
D.
Agaricis
IV.
Rust fungus
Choose the correct answer from the options given below:
Q76.
mcq single
+4 / 1
Which one of the following is not a criterion for classification of fungi?
Q77.
mcq single
+4 / 1
The flippers of the Penguins and Dolphins are the example of the
Q78.
mcq single
+4 / 1
Given below are some stages of human evolution.
Arrange them in correct sequence. (Past to Recent)
A. Homo habilis
B. Homo sapiens
C. Homo neanderthalensis
D. Homo erectus
Choose the correct sequence of human evolution from the options given below:
Q79.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Mesozoic Era
I.
Lower invertebrates
B.
Proterozoic Era
II.
Fish & Amphibia
C.
Cenozoic Era
III.
Birds & Reptiles
D.
Paleozoic Era
IV.
Mammals
Choose the correct answer from the options given below :
Q80.
mcq single
+4 / 1
Given below are two statements :
Statement I : In the nephron, the descending limb of loop of Henle is impermeable to water and permeable to electrolytes.
Statement II : The proximal convoluted tubule is lined by simple columnar brush border epithelium and increases the surface area for reabsorption.
In the light of the above statements, choose the correct answer from the option given below :
Q81.
mcq single
+4 / 1
Choose the correct statement given below regarding juxta medullary nephron.
Q82.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Non-medicated IUD
I.
Multiload 375
B.
Copper releasing IUD
II.
Progestogens
C.
Hormone releasing IUD
III.
Lippes loop
D.
Implants
IV.
LNG-20
Choose the correct answer from the option given below:
Q83.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A : Breast-feeding during initial period of infant growth is recommended by doctors for bringing a healthy baby.
Reason R : Colostrum contains several antibodies absolutely essential to develop resistance for the new born baby.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q84.
mcq single
+4 / 1
Which of the following is not a natural/traditional contraceptive method?
Q85.
mcq single
+4 / 1
Identify the correct description about the given figure :


Q86.
mcq single
+4 / 1
Identify the set of correct statements:
A. The flowers of Vallisneria are colourful and produce nectar.
B. The flowers of water lily are not pollinated by water.
C. In most of water-pollinated species, the pollen grains are protected from wetting.
D. Pollen grains of some hydrophytes are long and ribbon like.
E. In some hydrophytes, the pollen grains are carried passively inside water.
Choose the correct answer from the options given below.
Q87.
mcq single
+4 / 1
Identify the part of the seed from the given figure which is destined to form root when the seed germinates.

Q88.
mcq single
+4 / 1
Match List I with List II
List - I
(Types of Stamens)
List - II
(Example)
A.
Monoadelphous
I.
Citrus
B.
Diadelphous
II.
Pea
C.
Polyadelphous
III.
Lily
D.
Epiphyllous
IV.
China-rose
Choose the correct answer from the options given below:
Q89.
mcq single
+4 / 1
Bulliform cells are responsible for
Q90.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Rose
I.
Twisted aestivation
B.
Pea
II.
Perigynous flower
C.
Cotton
III.
Drupe
D.
Mango
IV.
Marginal placentation
Choose the correct answer from the options given below :
Q91.
mcq single
+4 / 1
Which of the following is an example of actinomorphic flower?
Q92.
mcq single
+4 / 1
Identify the type of flowers based on the position of calyx, corolla and androecium with respect to the ovary from the given figures (a) and (b)

Q93.
mcq single
+4 / 1
Inhibition of Succinic dehydrogenase enzyme by malonate is a classical example of:
Q94.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
GLUT-4
I.
Hormone
B.
Insulin
II.
Enzyme
C.
Trypsin
III.
Intercellular ground substance
D.
Collagen
IV.
Enables glucose transport into cells
Choose the correct answer from the options given below.
Q95.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Lipase
I.
Peptide bond
B.
Nuclease
II.
Ester bond
C.
Protease
III.
Glycosidic bond
D.
Amylase
IV.
Phosphodiester bond
Choose the correct answer from the options given below :
Q96.
mcq single
+4 / 1
The cofactor of the enzyme carboxypeptidase is:
Q97.
mcq single
+4 / 1
Regarding catalytic cycle of an enzyme action, select the correct sequential steps :
A. Substrate enzyme complex formation.
B. Free enzyme ready to bind with another substrate.
C. Release of products.
D. Chemical bonds of the substrate broken.
E. Substrate binding to active site.
Choose the correct answer from the options given below :
Q98.
mcq single
+4 / 1
Lecithin, a small molecular weight organic compound found in living tissues, is an example of:
Q99.
mcq single
+4 / 1
Hind II always cuts DNA molecules at a particular point called recognition sequence and it consists of:
Q100.
mcq single
+4 / 1
The "Ti plasmid" of Agrobacterium tumefaciens stands for
Q101.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Clostridium butylicum
I.
Ethanol
B.
Saccharomyces cerevisiae
II.
Streptokinase
C.
Trichoderma polysporum
III.
Butyric acid
D.
Streptococcus sp.
IV.
Cyclosporin-A
Choose the correct answer from the options given below:
Q102.
mcq single
+4 / 1
In an ecosystem if the Net Primary Productivity (NPP) of first trophic level is $$100 x\left(\mathrm{kcal} \mathrm{~m}^{-2}\right) \mathrm{yr}^{-1}$$, what would be the GPP (Gross Primary Productivity) of the third trophic level of the same ecosystem?
Q103.
mcq single
+4 / 1
In a plant, black seed color $$(\mathrm{BB} / \mathrm{Bb})$$ is dominant over white seed color $$(\mathrm{bb})$$. In order to find out the genotype of the black seed plant, with which of the following genotype will you cross it?
Q104.
mcq single
+4 / 1
As per ABO blood grouping system, the blood group of father is $$\mathrm{B}^{+}$$, mother is $$\mathrm{A}^{+}$$ and child is $$\mathrm{O}^{+}$$. Their respective genotype can be
A. I^(B)i/I^(A)i/ii
B. I^(B)I^(B)/I^(A)I^(A)/ii
C. iI^(B)/iI^(A)/I^(A)IB^()
D. I^(A)i/I^(B)i/I^(A)i
E. iI^(B)/iI^(A)/I^(A)I^(B)
Choose the most appropriate answer from the options given below :
Q105.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Two or more alternative forms of a gene
I.
Back cross
B.
Cross of F$$_1$$ progeny with homozygous recessive parent
II.
Ploidy
C.
Cross of F$$_1$$ progeny with any of the parents
III.
Allele
D.
Number of chromosome sets in plant
IV.
Test cross
Choose the correct answer from the options given below:
Q106.
mcq single
+4 / 1
Which one of the following can be explained on the basis of Mendel's Law of Dominance?
A. Out of one pair of factors one is dominant and the other is recessive.
B. Alleles do not show any expression and both the characters appear as such in F$$_2$$ generation.
C. Factors occur in pairs in normal diploid plants.
D. The discrete unit controlling a particular character is called factor.
E. The expression of only one of the parental characters is found in a monohybrid cross.
Choose the correct answer from the options given below:
Q107.
mcq single
+4 / 1
A pink flowered Snapdragon plant was crossed with a red flowered Snapdragon plant. What type of phenotype/s is/are expected in the progeny?
Q108.
mcq single
+4 / 1
In both sexes of cockroach, a pair of jointed filamentous structures called anal cerci are present on
Q109.
mcq single
+4 / 1
Match List I with List II related to digestive system of cockroach.
List - I
List - II
A.
The structures used for storing of food
I.
Gizzaard
B.
Ring of 6-8 blind tubules at junction of foregut and midgut.
II.
Gastric Caeca
C.
Ring of 100-150 yellow coloured thin filaments at junction of midgut and hindgut.
III.
Malpighian tubules
D.
The structures used for grinding the food.
IV.
Crop
Choose the correct answer from the options given below:
Q110.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Unicellular glandular epithelium
I.
Salivary glands
B.
Compound epithelium
II.
Pancreas
C.
Multicellular glandular epithelium
III.
Goblet cells of alimentary canal
D.
Endocrine glandular epithelium
IV.
Moist surface of buccal cavity
Choose the correct answer from the options given below:
Q111.
mcq single
+4 / 1
Given below are two statements:
Statement I: In $$\mathrm{C}_3$$ plants, some $$\mathrm{O}_2$$ binds to $$\mathrm{RuBisCO}$$, hence $$\mathrm{CO}_2$$ fixation is decreased.
Statement II: In $$\mathrm{C}_4$$ plants, mesophyll cells show very little photorespiration while bundle sheath cells do not show photorespiration.
In the light of the above statements, choose the correct answer from the options given below:
Q112.
mcq single
+4 / 1
Which of the following are required for the dark reaction of photosynthesis?
A. Light
B. Chlorophyll
C. $$\mathrm{CO}_2$$
D. ATP
E. NADPH
Choose the correct answer from the options given below:
Q113.
mcq single
+4 / 1
How many molecules of ATP and NADPH are required for every molecule of $$\mathrm{CO}_2$$ fixed in the Calvin cycle?
Q114.
mcq single
+4 / 1
The following are the statements about non-chordates:
A. Pharynx is perforated by gill slits.
B. Notochord is absent.
C. Central nervous system is dorsal.
D. Heart is dorsal if present.
E. Post anal tail is absent.
Choose the most appropriate answer from the options given below:
Q115.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Pleurobrachia
I.
Mollusca
B.
Radula
II.
Ctenophora
C.
Stomochord
III.
Osteichthyes
D.
Air bladder
IV.
Hemichordata
Choose the correct answer from the options given below :
Q116.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Pterophyllum
I.
Hag fish
B.
Myxine
II.
Saw fish
C.
Pristis
III.
Angel fish
D.
Exocoetus
IV.
Flying fish
Choose the correct answer from the options given below :
Q117.
mcq single
+4 / 1
Consider the following statements :
A. Annelids are true coelomates
B. Poriferans are pseudocoelomates
C. Aschelminthes are acoelomates
D. Platyhelminthes are pseudocoelomates
Choose the correct answer from the options given below :
Q118.
mcq single
+4 / 1
Read the following statements and choose the set of correct statements:
In the members of Phaeophyceae,
A. Asexual reproduction occurs usually by biflagellate zoospores.
B. Sexual reproduction is by oogamous method only.
C. Stored food is in the form of carbohydrates which is either mannitol or laminarin.
D. The major pigments found are chlorophyll a, c and carotenoids and xanthophyll.
E. Vegetative cells have a cellulosic wall, usually covered on the outside by gelatinous coating of algin.
Choose the correct answer from the options given below:
Q119.
mcq single
+4 / 1
Which of the following factors are favourable for the formation of oxyhaemoglobin in alveoli?
Q120.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Expiratory capacity
I.
Expiratory reserve volume + Tidal volume + Inspiratory reserve volume
B.
Functional residual capacity
II.
Tidal volume + Expiratory reserve volume
C.
Vital capacity
III.
Tidal volume + Inspiratory reserve volume
D.
Inspiratory capacity
IV.
Expiratory reserve volume + Residual volume
Choose the correct answer from the options given below :
Q121.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
$$\alpha$$ -I antitrypsin
I.
Cotton bollworm
B.
Cry IAb
II.
ADA deficiency
C.
Cry IAc
III.
Emphysema
D.
Enzyme replacement therapy
IV.
Corn borer
Choose the correct answer form the options given below:
Q122.
mcq single
+4 / 1
The following diagram showing restriction sites in E. coli cloning vector pBR322. Find the role of ‘X’ and ‘Y’ genes :


Q123.
mcq single
+4 / 1
Given below are two statements:
Statement I : Bt toxins are insect group specific and coded by a gene cry IAc.
Statement II : Bt toxin exists as inactive protoxin in B. thuringiensis. However, after ingestion by the insect the inactive protoxin gets converted into active form due to acidic $$\mathrm{pH}$$ of the insect gut.
In the light of the above statements, choose the correct answer from the options given below:
Q124.
mcq single
+4 / 1
The capacity to generate a whole plant from any cell of the plant is called:
Q125.
mcq single
+4 / 1
What is the fate of a piece of DNA carrying only gene of interest which is transferred into an alien organism?
A. The piece of DNA would be able to multiply itself independently in the progeny cells of the organism.
B. It may get integrated into the genome of the recipient.
C. It may multiply and be inherited along with the host DNA.
D. The alien piece of DNA is not an integral part of chromosome.
E. It shows ability to replicate.
Choose the correct answer from the options given below:
Q126.
mcq single
+4 / 1
Which of the following statements is incorrect?
Q127.
mcq single
+4 / 1
Which of the following are fused in somatic hybridization involving two varieties of plants?
Q128.
mcq single
+4 / 1
Given below are two statements:
Statement I: The cerebral hemispheres are connected by nerve tract known as corpus callosum.
Statement II: The brain stem consists of the medulla oblongata, pons and cerebrum.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q129.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Pons
I.
Provides additional space for Neurons, regulates posture and balance.
B.
Hypothalamus
II.
Controls respiration and gastric secretions.
C.
Medulla
III.
Connects different regions of the brain.
D.
Cerebellum
IV.
Neuro secretory cells
Choose the correct answer from the options given below :
Q130.
mcq single
+4 / 1
In the given figure, which component has thin outer walls and highly thickened inner walls?


Q131.
mcq single
+4 / 1
Given below are two statements:
Statement I : Parenchyma is living but collenchyma is dead tissue.
Statement II : Gymnosperms lack xylem vessels but presence of xylem vessels is the characteristic of angiosperms.
In the light of the above statements, choose the correct answer from the options given below:
Q132.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Axoneme
I.
Centriole
B.
Cartwheel pattern
II.
Cilia and flagella
C.
Crista
III.
Chromosome
D.
Satellite
IV.
Mitochondria
Choose the correct answer from the options given below :
Q133.
mcq single
+4 / 1
Given below are two statements:
Statement I: Mitochondria and chloroplasts both double membranes bound organelles.
Statement II: Inner membrane of mitochondria is relatively less permeable, as compared chloroplast.
In the light of the above statements, choose the mis appropriate answer from the options given below:
Q134.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Nucleolus
I.
Site of formation of glycolipid
B.
Centriole
II.
Organization like the cartwheel
C.
Leucoplasts
III.
Site of active ribosomal RNA synthesis
D.
Golgi apparatus
IV.
For storing nutrients
Choose the correct answer from the options given below:
Q135.
mcq single
+4 / 1
The DNA present in chloroplast is:
Q136.
mcq single
+4 / 1
The lactose present in the growth medium of bacteria is transported to the cell by the action of
Q137.
mcq single
+4 / 1
Which one is the correct product of DNA dependent RNA polymerase to the given template?
3'TACATGGCAAATATCCATTCA5'
Q138.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Frederick Griffith
I.
Genetic code
B.
Francois Jacob & Jacque Monod
II.
Semi-conservative mode of DNA replication
C.
Har Gobind Khorana
III.
Tranformation
D.
Meselson & Stahl
IV.
Lac operon
Choose the correct answer from the options given below:
Q139.
mcq single
+4 / 1
A transcription unit in DNA is defined primarily by the three regions in DNA and these are with respect to upstream and down stream end;
Q140.
mcq single
+4 / 1
Which of the following statement is correct regarding the process of replication in E.coli?
Q141.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Down's syndrome
I.
11$$^\mathrm{th}$$ chromosome
B.
$$\alpha$$-thalassemia
II.
'X' chromosome
C.
$$\beta$$-thalassemia
III.
21$$^\mathrm{st}$$ chromosome
D.
Klinefelter's syndrome
IV.
16$$^\mathrm{th}$$ chromosome
Choose the correct answer from the options given below :
Q142.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
RNA polymerase III
I.
snRNPs
B.
Termination of transcription
II.
Promotor
C.
Splicing of Exons
III.
Rho factor
D.
TATA box
IV.
SnRNAs, tRNA
Choose the correct answer from the options given below :
Q143.
mcq single
+4 / 1
Given below are two statements :
Statement I : Bone marrow is the main lymphoid organ where all blood cells including lymphocytes are produced.
Statement II : Both bone marrow and thymus provide micro environments for the development and maturation of T-lymphocytes.
In the light of above statements, choose the most appropriate answer from the options given below
Q144.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Cocaine
I.
Effective sedative in surgery
B.
Heroin
II.
Cannabis sativa
C.
Morphine
III.
Erythroxylum
D.
Marijuana
IV.
Papaver somniferum
Choose the correct answer from the options given below:
Q145.
mcq single
+4 / 1
Which of the following are Autoimmune disorders?
A. Myasthenia gravis
B. Rheumatoid arthritis
C. Gout
D. Muscular dystrophy
E. Systemic Lupus Erythematosus (SLE)
Choose the most appropriate answer from the options given below:
Q146.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Typhoid
I.
Fungus
B.
Leishmaniasis
II.
Nematode
C.
Ringworm
III.
Protozoa
D.
Filariasis
IV.
Bacteria
Choose the correct answer from the options given below:
Q147.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Common cold
I.
Plasmodium
B.
Haemozoin
II.
Typhoid
C.
Widal test
III.
Rhinoviruses
D.
Allergy
IV.
Dust mites
Choose the correct answer from the options given below :
Q148.
mcq single
+4 / 1
Which one of the following factors will not affect the Hardy-Weinberg equilibrium?
Q149.
mcq single
+4 / 1
Given below are two statements:
Statement I: Gause's competitive exclusion principle states that two closely related species competing for different resources cannot exist indefinitely.
Statement II: According to Gause's principle, during competition, the inferior will be eliminated. This may be true if resources are limiting.
In the light of the above statements, choose the correct answer from the options given below :
Q150.
mcq single
+4 / 1
The equation of Verhulst-Pearl logistic growth is $$\frac{\mathrm{dN}}{\mathrm{dt}}=\mathrm{rN}\left[\frac{\mathrm{K}-\mathrm{N}}{\mathrm{K}}\right]$$.
From this equation, $$\mathrm{K}$$ indicates:
Q151.
mcq single
+4 / 1
Following are the stages of pathway for conduction of an action potential through the heart
A. AV bundle
B. Purkinje fibres
C. AV node
D. Bundle branches
E. SA node
Choose the correct sequence of pathway from the options given below
Q152.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
P wave
(i)
Heart muscles are electrically silent.
B.
QRS complex
(ii)
Depolarisation of ventricles.
C.
T wave
(iii)
Depolarisation of atria.
D.
T-P gap
(iv)
Repolarisation of ventricles.
Choose the correct answer from the options given below :
Q153.
mcq single
+4 / 1
Following are the stages of cell division :
A. Gap 2 phase
B. Cytokinesis
C. Synthesis phase
D. Karyokinesis
E. Gap 1 phase
Choose the correct sequence of stages from the options given below :
Q154.
mcq single
+4 / 1
Spindle fibers attach to kinetochores of chromosomes during
Q155.
mcq single
+4 / 1
Match List I with List II :
List - I
(Sub Phases of Prophase I)
List - II
(Specific Characters)
A.
Diakinesis
I.
Synaptonemal complex formation
B.
Pachytene
II.
Completion of terminalisation of chiasmata
C.
Zygotene
III.
Chromosomes look like thin threads
D.
Leptotene
IV.
Appearance of recombination nodules
Choose the correct answer from the options given below
Q156.
mcq single
+4 / 1
Given below are two statements:
Statement I : Chromosomes become gradually visible under light microscope during leptotene stage.
Statement II : The beginning of diplotene stage is recognized by dissolution of synaptonemal complex.
In the light of the above statements, choose the correct answer from the options given below:
Q157.
mcq single
+4 / 1
Identify the correct option (A), (B), (C), (D) with respect to spermatogenesis.


Q158.
mcq single
+4 / 1
Given below are two statements:
Statement I: The presence or absence of hymen is not a reliable indicator of virginity.
Statement II: The hymen is torn during the first coitus only.
In the light of the above above statements, choose the correct answer from the options given below :
Q159.
mcq single
+4 / 1
Which of the following is not a component of Fallopian tube?
Q160.
mcq single
+4 / 1
Which of the following is not a steroid hormone?
Q161.
mcq single
+4 / 1
Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason R :
Assertion A : FSH acts upon ovarian follicles in female and Leydig cells in male.
Reason R : Growing ovarian follicles secrete estrogen in female while interstitial cells secrete androgen in male human being.
In the light of the above statements, choose the correct answer from the options given below :
Q162.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Exophthalmic goiter
I.
Excess secretion of cortisol, moon face \& hypergylcemia.
B.
Acromegaly
II.
Hypo-secretion of thyroid hormone and stunted growth.
C.
Cushing's syndrome
III.
Hyper secretion of thyroid hormone \& protruding eye balls.
D.
Cretinism
IV.
Excessive secretion of growth hormone.
Choose the correct answer from the options given below :
Q163.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Robert May
I.
Species-Area relationship
B.
Alexander von Humboldt
II.
Long term ecosystem experiment using out door plots
C.
Paul Ehrlich
III.
Global species diversity at about 7 million
D.
David Tilman
IV.
Rivet popper hypothesis
Choose the correct answer from the options given below:
Q164.
mcq single
+4 / 1
Tropical regions show greatest level of species richness because
A. Tropical latitudes have remained relatively undisturbed for millions of years, hence more time was available for species diversification.
B. Tropical environments are more seasonal.
C. More solar energy is available in tropics.
D. Constant environments promote niche specialization.
E. Tropical environments are constant and predictable.
Choose the correct answer from the options given below.
Q165.
mcq single
+4 / 1
The type of conservation in which the threatened species are taken out from their natural habitat and placed in special setting where they can be protected and given special care is called
Q166.
mcq single
+4 / 1
List of endangered species was released by
Q167.
mcq single
+4 / 1
These are regarded as major causes of biodiversity loss:
A. Over exploitation
B. Co-extinction
C. Mutation
D. Habitat loss and fragmentation
E. Migration
Choose the correct option:
Q168.
mcq single
+4 / 1
Spraying sugarcane crop with which of the following plant growth regulators, increases the length of stem, thus, increasing the yield?
Q169.
mcq single
+4 / 1
Auxin is used by gardeners to prepare weed-free lawns. But no damage is caused to grass as auxin
Q170.
mcq single
+4 / 1
Formation of interfascicular cambium from fully developed parenchyma cells is an example for
Q171.
mcq single
+4 / 1
Identify the step in tricarboxylic acid cycle, which does not involve oxidation of substrate.
Q172.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Citric acid cycle
I.
Cytoplasm
B.
Glycolysis
II.
Mitochondrial matrix
C.
Electron transport system
III.
Intermembrane space of mitochondria
D.
Proton gradient
IV.
Inner mitochondrial membrane
Choose the correct answer from the options given below:
Q173.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
A.
Fibrous joints
I.
Adjacent vertebrae, limited movement
B.
Cartilaginous joints
II.
Humerus and Pectoral girdle, rotational movement
C.
Hinge joints
III.
Skull, don't allow any movement
D.
Ball and socket joints
IV.
Knee, help in locomotion
Choose the correct answer from the options given below :
Q174.
mcq single
+4 / 1
Three types of muscles are given as a, b and c. Identify the correct matching pair along with their location in human body:
 Name of muscle/location
Name of muscle/location
 Name of muscle/location
Name of muscle/location
Q175.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Rhizopus
I.
Mushroom
B.
Ustilago
II.
Smut fungus
C.
Puccinia
III.
Bread mould
D.
Agaricis
IV.
Rust fungus
Choose the correct answer from the options given below:
Q176.
mcq single
+4 / 1
Which one of the following is not a criterion for classification of fungi?
Q177.
mcq single
+4 / 1
The flippers of the Penguins and Dolphins are the example of the
Q178.
mcq single
+4 / 1
Given below are some stages of human evolution.
Arrange them in correct sequence. (Past to Recent)
A. Homo habilis
B. Homo sapiens
C. Homo neanderthalensis
D. Homo erectus
Choose the correct sequence of human evolution from the options given below:
Q179.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Mesozoic Era
I.
Lower invertebrates
B.
Proterozoic Era
II.
Fish & Amphibia
C.
Cenozoic Era
III.
Birds & Reptiles
D.
Paleozoic Era
IV.
Mammals
Choose the correct answer from the options given below :
Q180.
mcq single
+4 / 1
Given below are two statements :
Statement I : In the nephron, the descending limb of loop of Henle is impermeable to water and permeable to electrolytes.
Statement II : The proximal convoluted tubule is lined by simple columnar brush border epithelium and increases the surface area for reabsorption.
In the light of the above statements, choose the correct answer from the option given below :
Q181.
mcq single
+4 / 1
Choose the correct statement given below regarding juxta medullary nephron.
Q182.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Non-medicated IUD
I.
Multiload 375
B.
Copper releasing IUD
II.
Progestogens
C.
Hormone releasing IUD
III.
Lippes loop
D.
Implants
IV.
LNG-20
Choose the correct answer from the option given below:
Q183.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A : Breast-feeding during initial period of infant growth is recommended by doctors for bringing a healthy baby.
Reason R : Colostrum contains several antibodies absolutely essential to develop resistance for the new born baby.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q184.
mcq single
+4 / 1
Which of the following is not a natural/traditional contraceptive method?
Q185.
mcq single
+4 / 1
Identify the correct description about the given figure :


Q186.
mcq single
+4 / 1
Identify the set of correct statements:
A. The flowers of Vallisneria are colourful and produce nectar.
B. The flowers of water lily are not pollinated by water.
C. In most of water-pollinated species, the pollen grains are protected from wetting.
D. Pollen grains of some hydrophytes are long and ribbon like.
E. In some hydrophytes, the pollen grains are carried passively inside water.
Choose the correct answer from the options given below.
Q187.
mcq single
+4 / 1
Identify the part of the seed from the given figure which is destined to form root when the seed germinates.


Q188.
mcq single
+4 / 1
Match List I with List II
List - I
(Types of Stamens)
List - II
(Example)
A.
Monoadelphous
I.
Citrus
B.
Diadelphous
II.
Pea
C.
Polyadelphous
III.
Lily
D.
Epiphyllous
IV.
China-rose
Choose the correct answer from the options given below:
Q189.
mcq single
+4 / 1
Bulliform cells are responsible for
Q190.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
Rose
I.
Twisted aestivation
B.
Pea
II.
Perigynous flower
C.
Cotton
III.
Drupe
D.
Mango
IV.
Marginal placentation
Choose the correct answer from the options given below :
Q191.
mcq single
+4 / 1
Which of the following is an example of actinomorphic flower?
Q192.
mcq single
+4 / 1
Identify the type of flowers based on the position of calyx, corolla and androecium with respect to the ovary from the given figures (a) and (b)


Q193.
mcq single
+4 / 1
Inhibition of Succinic dehydrogenase enzyme by malonate is a classical example of:
Q194.
mcq single
+4 / 1
Match List I with List II
List - I
List - II
A.
GLUT-4
I.
Hormone
B.
Insulin
II.
Enzyme
C.
Trypsin
III.
Intercellular ground substance
D.
Collagen
IV.
Enables glucose transport into cells
Choose the correct answer from the options given below.
Q195.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
A.
Lipase
I.
Peptide bond
B.
Nuclease
II.
Ester bond
C.
Protease
III.
Glycosidic bond
D.
Amylase
IV.
Phosphodiester bond
Choose the correct answer from the options given below :
Q196.
mcq single
+4 / 1
The cofactor of the enzyme carboxypeptidase is:
Q197.
mcq single
+4 / 1
Regarding catalytic cycle of an enzyme action, select the correct sequential steps :
A. Substrate enzyme complex formation.
B. Free enzyme ready to bind with another substrate.
C. Release of products.
D. Chemical bonds of the substrate broken.
E. Substrate binding to active site.
Choose the correct answer from the options given below :
Q198.
mcq single
+4 / 1
Lecithin, a small molecular weight organic compound found in living tissues, is an example of:
Q199.
mcq single
+4 / 1
Hind II always cuts DNA molecules at a particular point called recognition sequence and it consists of:
Q200.
mcq single
+4 / 1
The "Ti plasmid" of Agrobacterium tumefaciens stands for