NEET 2024 (Re-Examination)
MEDICAL 2024 Previous Year
3 hDuration
800Total Marks
400Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 400 across 3 section(s); maximum marks: 800.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Identify the incorrect statement.
Q2.
mcq single
+4 / 1
Which of the following pairs of ions will have same spin only magnetic moment values within the pair?
A. $$\mathrm{Zn}^{2+}, \mathrm{Ti}^{2+}$$
B. $$\mathrm{Cr}^{2+}, \mathrm{Fe}^{2+}$$
C. $$\mathrm{Ti}^{3+}, \mathrm{Cu}^{2+}$$
D. $$\mathrm{V}^{2+}, \mathrm{Cu}^{+}$$
Choose the correct answer from the options given below :
Q3.
mcq single
+4 / 1
The UV-visible absorption bands in the spectra of lanthanoid ions are 'X', probably because of the excitation of electrons involving 'Y'. The 'X' and 'Y', respectively, are :
Narrow and d and f orbitals
Q4.
mcq single
+4 / 1
Baeyer's reagent is :
Q5.
mcq single
+4 / 1
Match List I with List II:
List - I
(Block/group in periodic table)
List - II
(Element)
(A)
Lanthanoid
(I)
Ce
(B)
d-block element
(II)
As
(C)
p-block element
(III)
Cs
(D)
s-block element
(IV)
Mn
Choose the correct answer from the options given below:
Q6.
mcq single
+4 / 1
Which of the following set of ions act as oxidising agents?
Q7.
mcq single
+4 / 1
Identify the incorrect statement about $$\mathrm{PCl}_5$$.
Q8.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Molecule
List - II
Bond enthalpy (kJ mol$$^{-1}$$)
(A)
HCl
(I)
435.8
(B)
N$$_2$$
(II)
498
(C)
H$$_2$$
(III)
946.0
(D)
O$$_2$$
(IV)
431.0
Choose the correct answer from the options given below:
Q9.
mcq single
+4 / 1
Which of the following molecules has "NON ZERO" dipole moment value?
Q10.
mcq single
+4 / 1
Arrange the following compounds in increasing order of their solubilities in chloroform:
$$\mathrm{NaCl}, \mathrm{CH}_3 \mathrm{OH}$$, cyclohexane, $$\mathrm{CH}_3 \mathrm{CN}$$
Q11.
mcq single
+4 / 1
Which indicator is used in the titration of sodium hydroxide against oxalic acid and what is the colour change at the end point?
Q12.
mcq single
+4 / 1
The ratio of solubility of AgCl in 0.1 M KCl solution to the solubility of AgCl in water is:
(Given : Solubility product of AgCl = 10$$^{–10}$$)
Q13.
mcq single
+4 / 1
Which of the following plot represents the variation of $$\ln \mathrm{k}$$ versus $$\frac{1}{\mathrm{~T}}$$ in accordance with Arrhenius equation?
Q14.
mcq single
+4 / 1
Following data is for a reaction between reactants A and B :
Rate
$$\mathrm{mol} \mathrm{~L}^{-1} \mathrm{~s}^{-1}$$
$$\mathrm{[A]}$$
$$\mathrm{[B]}$$
$$
2 \times 10^{-3}
$$
0.1 M
0.1 M
$$
4 \times 10^{-3}
$$
0.2 M
0.1 M
$$
1.6 \times 10^{-2}
$$
0.2 M
0.2 M
$$
\text { The order of the reaction with respect to } \mathrm{A} \text { and } \mathrm{B} \text {, respectively, are }
$$
Q15.
mcq single
+4 / 1
Rate constants of a reaction at $$500 \mathrm{~K}$$ and $$700 \mathrm{~K}$$ are $$0.04 \mathrm{~s}^{-1}$$ and $$0.14 \mathrm{~s}^{-1}$$, respectively; then, activation energy of the reaction is :
(Given: $$\log 3.5=0.5441, \mathrm{R}=8.31 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$$)
Q16.
mcq single
+4 / 1
The oxidation states not shown by Mn in given reaction is :
$$
3 \mathrm{MnO}_4^{2-}+4 \mathrm{H}^{+} \longrightarrow 2 \mathrm{MnO}_4^{-}+\mathrm{MnO}_2+2 \mathrm{H}_2 \mathrm{O}
$$
A. +6
B. +2
C. +4
D. +7
E. +3
Choose the most appropriate answer from the options given below :
Q17.
mcq single
+4 / 1
The following reaction method
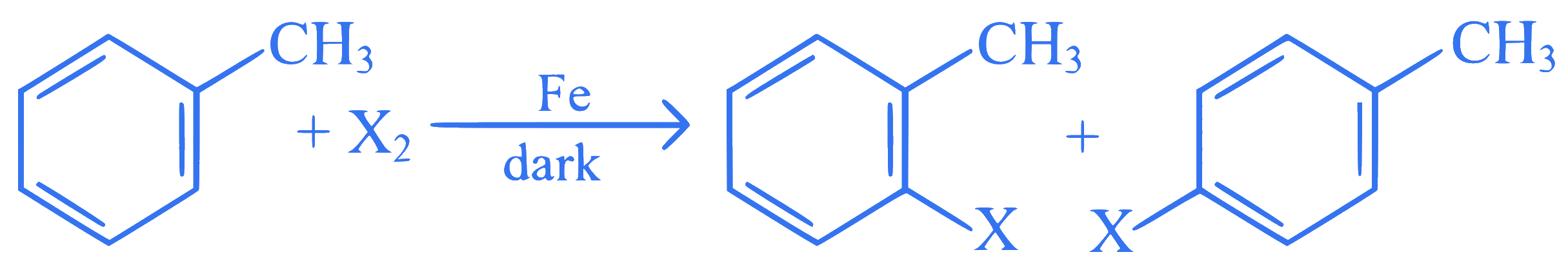 is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
 is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
Q18.
mcq single
+4 / 1
The major product $$\mathrm{C}$$ in the below mentioned reaction is:
$$\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{Br} \xrightarrow[\Delta]{\text { alc. } \mathrm{KOH}} \mathrm{A} \xrightarrow{\mathrm{HBr}} \mathrm{B} \xrightarrow[\Delta]{\text { aq. } \mathrm{KOH}} \mathrm{C}$$
Q19.
mcq single
+4 / 1
Mass of glucose $$(\mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_6)$$ required to be dissolved to prepare one litre of its solution which is isotonic with $$15 \mathrm{~g} \mathrm{~L}^{-1}$$ solution of urea $$(\mathrm{NH}_3 \mathrm{CONH}_2)$$ is (Given: Molar mass in $$\mathrm{g} \mathrm{mol}^{-1} \mathrm{C}: 12, \mathrm{H}: 1, \mathrm{O}: 16, \mathrm{~N}: 14$$)
Q20.
mcq single
+4 / 1
On complete combustion, 0.3 g of an organic compound gave 0.2 g of CO$$_2$$ and 0.1 g of H$$_2$$O. The percentage
composition of carbon and hydrogen in the compound, respectively is:
Q21.
mcq single
+4 / 1
$$1.0 \mathrm{~g}$$ of $$\mathrm{H}_2$$ has same number of molecules as in:
Q22.
mcq single
+4 / 1
The amount of glucose required to prepare $$250 \mathrm{~mL}$$ of $$\frac{\mathrm{M}}{20}$$ aqueous solution is :
(Molar mass of glucose : $$180 \mathrm{~g} \mathrm{~mol}^{-1}$$)
Q23.
mcq single
+4 / 1
At a given temperature and pressure, the equilibrium constant values for the equilibria are given below:
$$\begin{aligned}
& 3 \mathrm{~A}_2+\mathrm{B}_2 \rightleftharpoons 2 \mathrm{~A}_3 \mathrm{~B}, \mathrm{~K}_1 \\
& \mathrm{~A}_3 \mathrm{~B} \rightleftharpoons \frac{3}{2} \mathrm{~A}_2+\frac{1}{2} \mathrm{~B}_2, \mathrm{~K}_2
\end{aligned}$$
The relation between $$\mathrm{K}_1$$ and $$\mathrm{K}_2$$ is :
Q24.
mcq single
+4 / 1
For the reaction in equilibrium
$$\mathrm{N}_2(\mathrm{~g})+3 \mathrm{H}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{NH}_3(\mathrm{~g}), \Delta \mathrm{H}=-\mathrm{Q}$$
Reaction is favoured in forward direction by:
Q25.
mcq single
+4 / 1
The correct decreasing order of atomic radii (pm) of $$\mathrm{Li}, \mathrm{Be}, \mathrm{B}$$ and $$\mathrm{C}$$ is
Q26.
mcq single
+4 / 1
Match List I with List II :
List - I
(Atom/Molecule)
List - II
(Property)
(A)
Nitrogen atom
(I)
Paramagnetic
(B)
Fluorine molecule
(II)
Most relative element in group 18
(C)
Oxygen molecule
(III)
Element with highest ionisation enthalpy in group 15
(D)
Xenon atom
(IV)
Strongest oxidising agent
Identify the correct answer from the options given below :
Q27.
mcq single
+4 / 1
Choose the correct statement for the work done in the expansion and heat absorbed or released when
5 litres of an ideal gas at 10 atmospheric pressure isothermally expands into vacuum until volume is
15 litres :
Q28.
mcq single
+4 / 1
For the following reaction at $$300 \mathrm{~K}$$
$$\mathrm{A}_2(\mathrm{~g})+3 \mathrm{~B}_2(\mathrm{~g}) \rightarrow 2 \mathrm{AB}_3(\mathrm{~g})$$
the enthalpy change is $$+15 \mathrm{~kJ}$$, then the internal energy change is :
Q29.
mcq single
+4 / 1
For an endothermic reaction:
(A) $$\mathrm{q}_{\mathrm{p}}$$ is negative.
(B) $$\Delta_{\mathrm{r}} \mathrm{H}$$ is positive.
(C) $$\Delta_r \mathrm{H}$$ is negative.
(D) $$\mathrm{q}_{\mathrm{p}}$$ is positive.
Choose the correct answer from the options given below:
Q30.
mcq single
+4 / 1
Match List I with List II:
List - I
Solid salt treated with dil. H$$_2$$SO$$_4$$
List - II
Anion detected
(A)
effervescence of colourless gas
(I)
NO$$_2^-$$
(B)
gas with smell of rotten egg
(II)
CO$$_3^{2-}$$
(C)
gas with pungent smell
(III)
S$$^{2-}$$
(D)
brown fumes
(IV)
SO$$_3^{2-}$$
Choose the correct answer from the options given below:
Q31.
mcq single
+4 / 1
Identify the incorrect statement from the following :
Q32.
mcq single
+4 / 1
Which of the following is not an ambidentate ligand?
Q33.
mcq single
+4 / 1
Ethylene diaminetetraacetate ion is a/an:
Q34.
mcq single
+4 / 1
$$\left[\mathrm{Mn}_2(\mathrm{CO})_{10}\right]$$ and $$\left[\mathrm{Co}_2(\mathrm{CO})_8\right]$$ structures have
A. Metal-Metal linkage
B. Terminal CO groups
C. Bridging CO groups
D. Metal in zero oxidation state
Choose the correct answer from the options given below
Q35.
mcq single
+4 / 1
Match List-I with List-II :
List - I
(Test/reagent)
List - II
(Radical identified)
(A)
Lake Test
(I)
NO$$_3^-$$
(B)
Nessler's Reagent
(II)
Fe$$^{3+}$$
(C)
Potassium sulphocyanide
(III)
Al$$^{3+}$$
(D)
Brown Ring Test
(IV)
NH$$_4^+$$
Choose the correct answer from the options given below :
Q36.
mcq single
+4 / 1
Methyl group attached to a positively charged carbon atom stabilizes the carbocation due to
Q37.
mcq single
+4 / 1
The correct IUPAC name of the compound
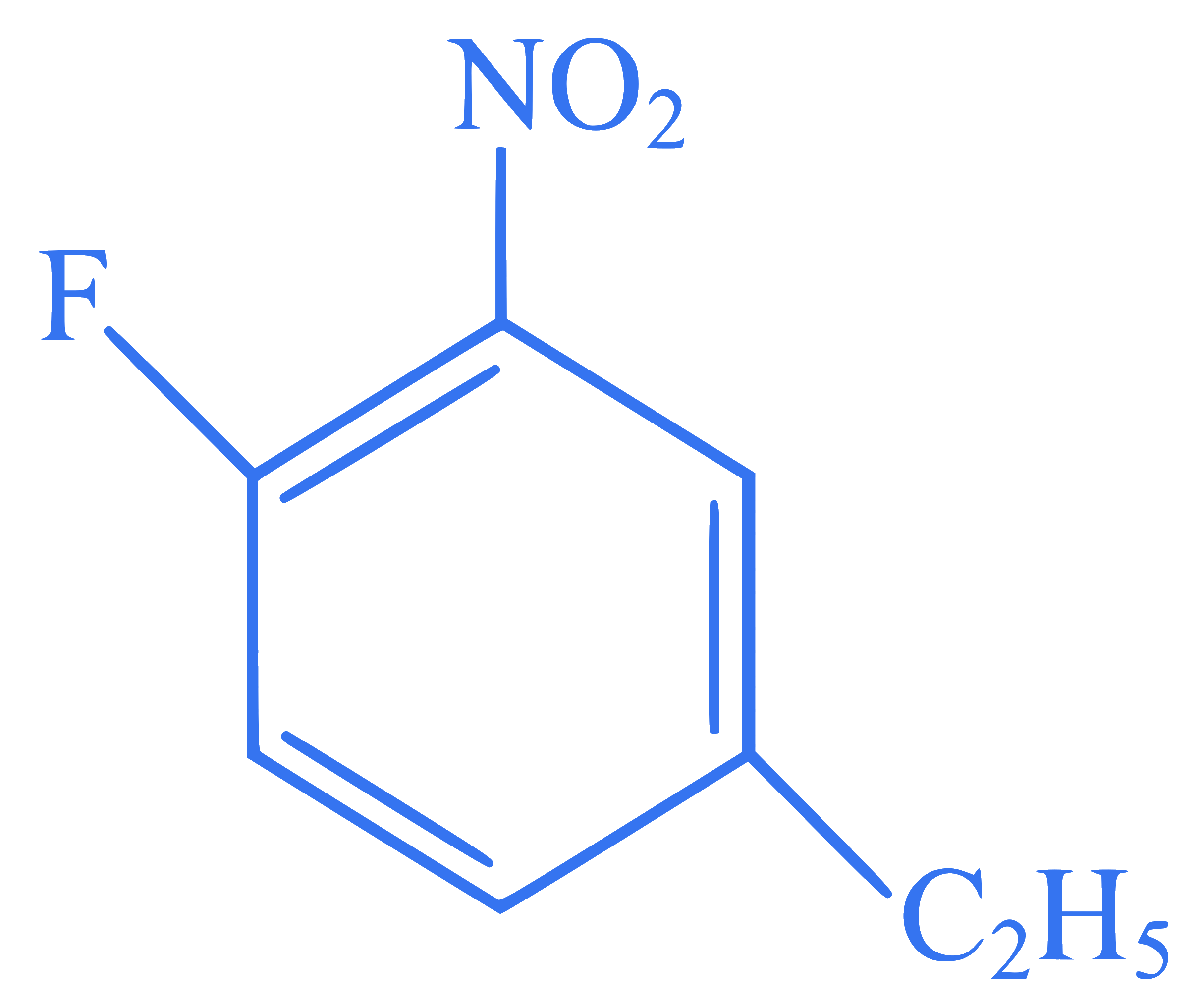 is :
is :
 is :
is :
Q38.
mcq single
+4 / 1
A steam volatile organic compound which is immiscible with water has a boiling point of $$250^{\circ} \mathrm{C}$$. During steam distillation, a mixture of this organic compound and water will boil :
Q39.
mcq single
+4 / 1
Select the incorrect reaction among the following :
Q40.
mcq single
+4 / 1
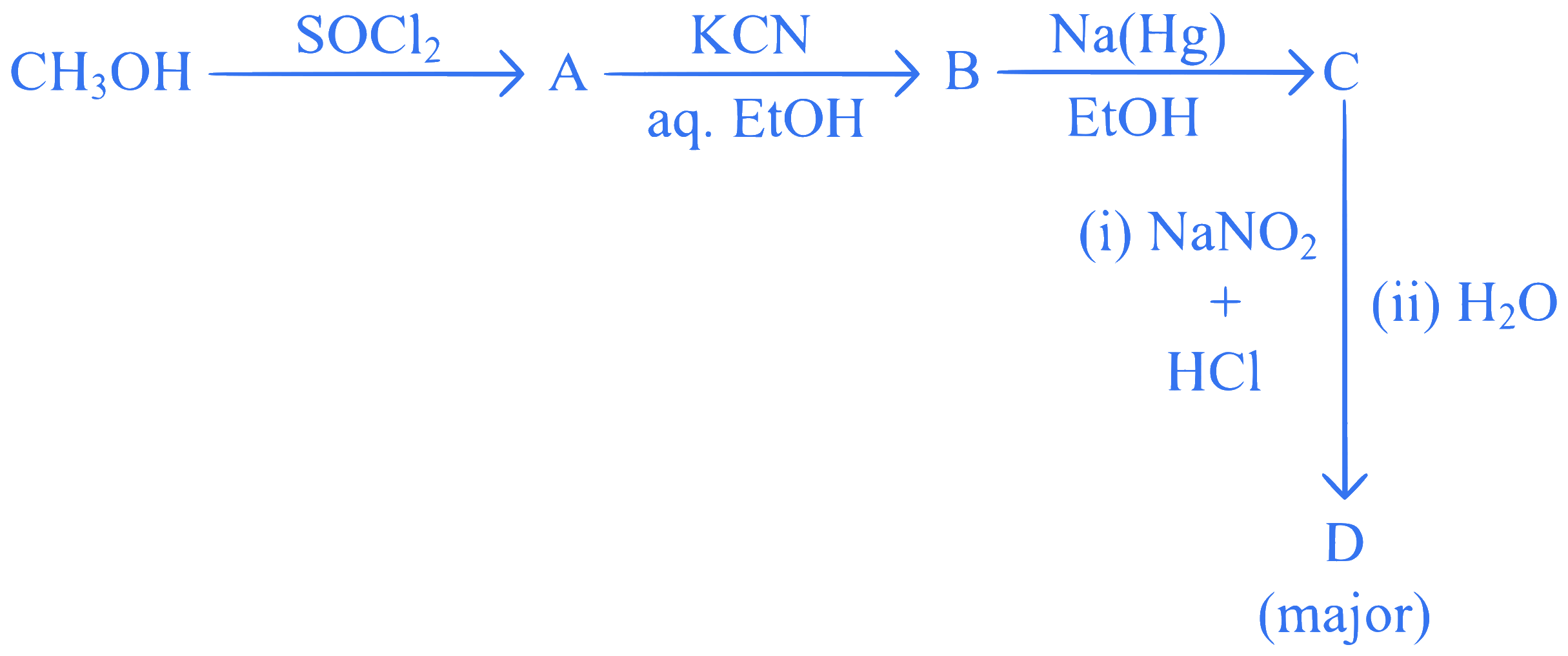
Identify D in the following sequence of reactions:
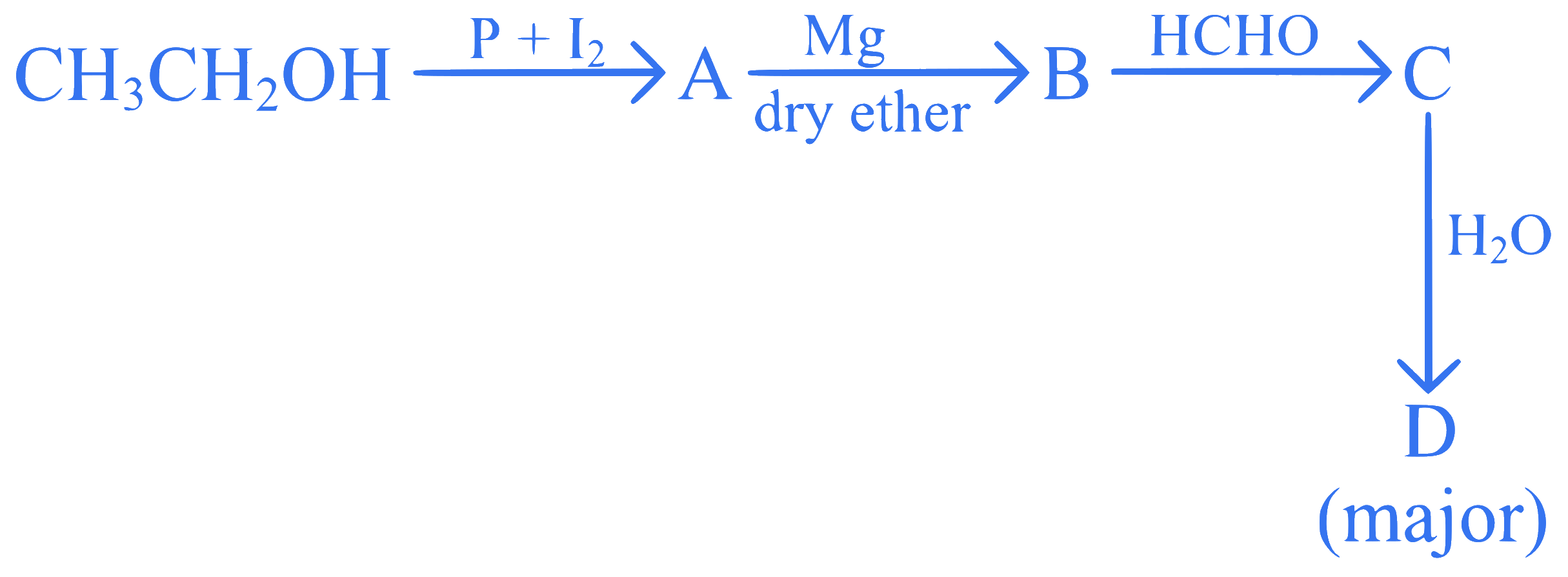
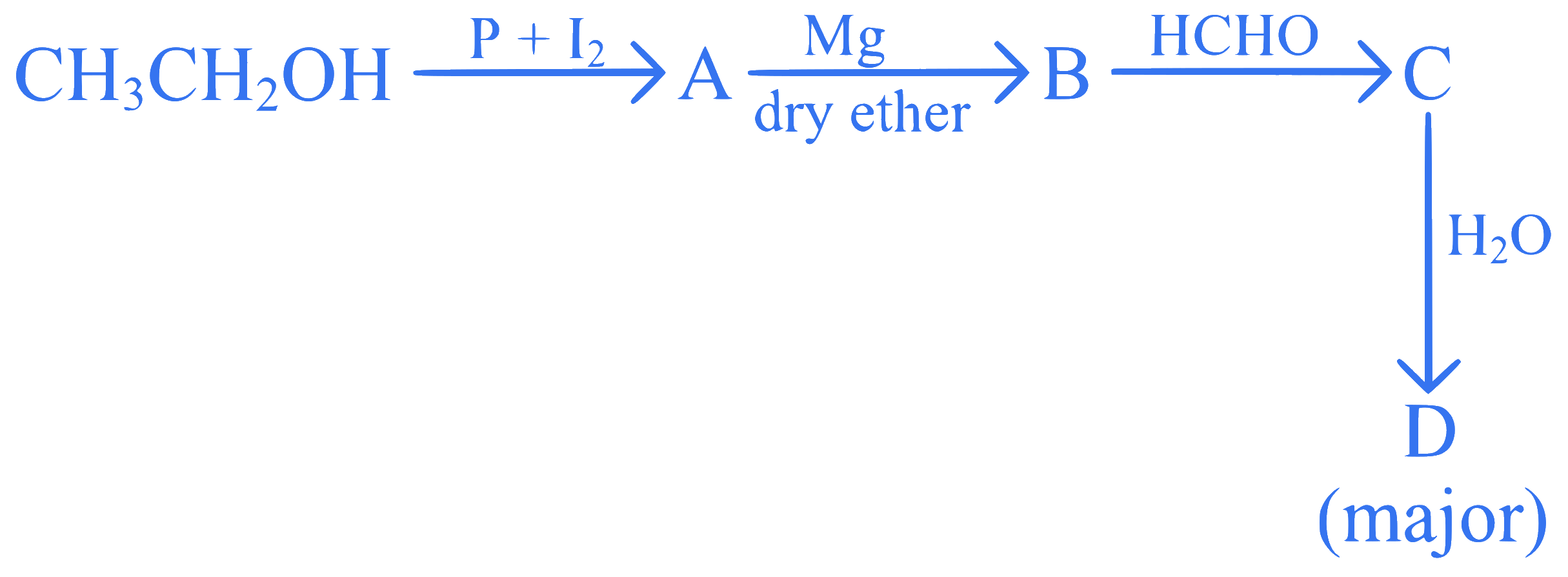
Q41.
mcq single
+4 / 1
The major product D formed in the following reaction sequence is :

Q42.
mcq single
+4 / 1
The alkane that can be oxidized to the corresponding alcohol by KMnO$$_4$$ as per the equation
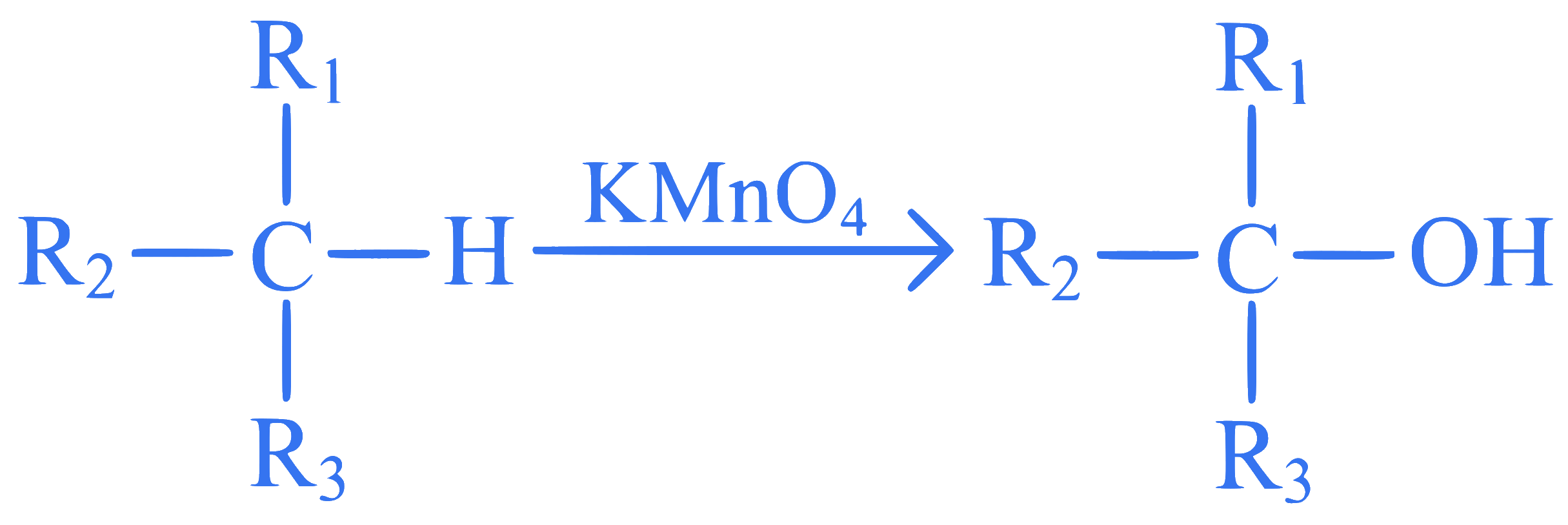 is, when :
is, when :
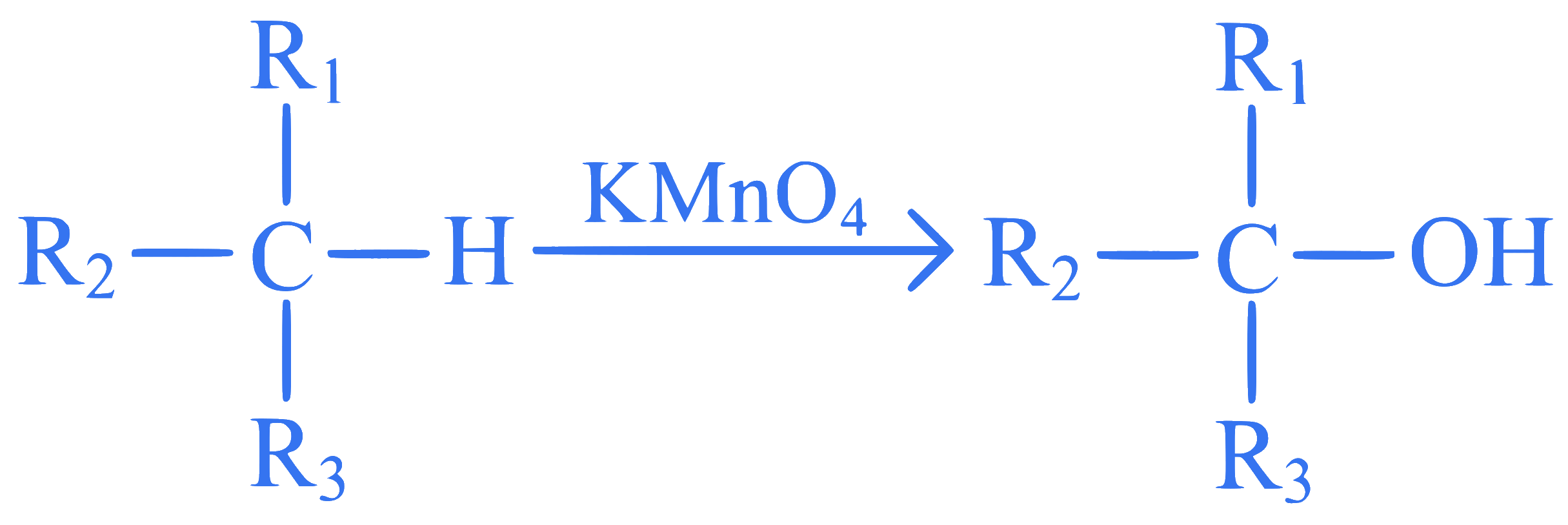 is, when :
is, when :
Q43.
mcq single
+4 / 1
Given below are two statements:
Statement I: Propene on treatment with diborane gives an addition product with the formula
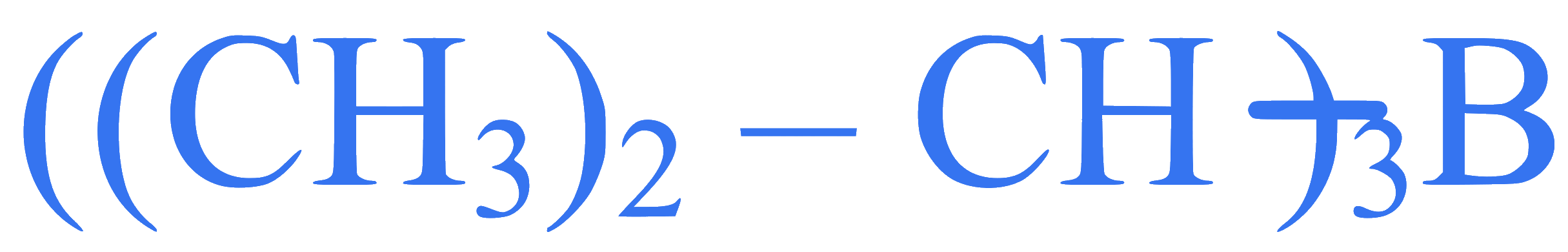 Statement II: Oxidation of
Statement II: Oxidation of
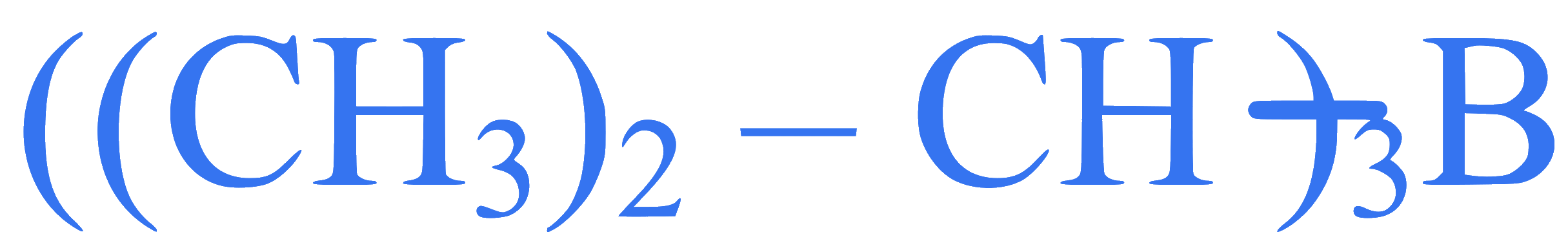 with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
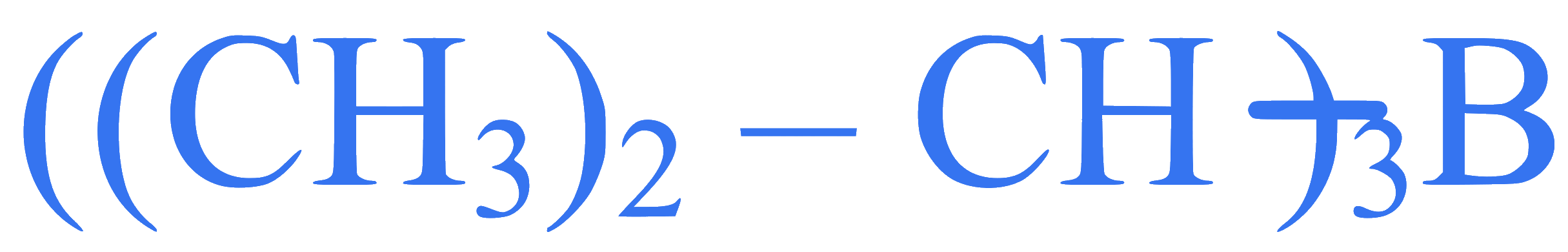 Statement II: Oxidation of
Statement II: Oxidation of
 with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q44.
mcq single
+4 / 1
Given below are two statements :
Statement I : Glycogen is similar to amylose in its structure.
Statement II : Glycogen is found in yeast and fungi also.
In the light of the above statements, choose the correct answer from the options given below :
Q45.
mcq single
+4 / 1
The standard cell potential of the following cell $$\mathrm{Zn}\left|\mathrm{Zn}^{2+}(\mathrm{aq})\right| \mathrm{Fe}^{2+}(\mathrm{aq}) \mid \mathrm{Fe}$$ is $$0.32 \mathrm{~V}$$. Calculate the standard Gibbs energy change for the reaction:
$$\mathrm{Zn}(\mathrm{s})+\mathrm{Fe}^{2+}(\mathrm{aq}) \rightarrow \mathrm{Zn}^{2+}(\mathrm{aq})+\mathrm{Fe}(\mathrm{s})$$
(Given : $$1 \mathrm{~F}=96487 \mathrm{C}$$)
Q46.
mcq single
+4 / 1
From the following select the one which is not an example of corrosion.
Q47.
mcq single
+4 / 1
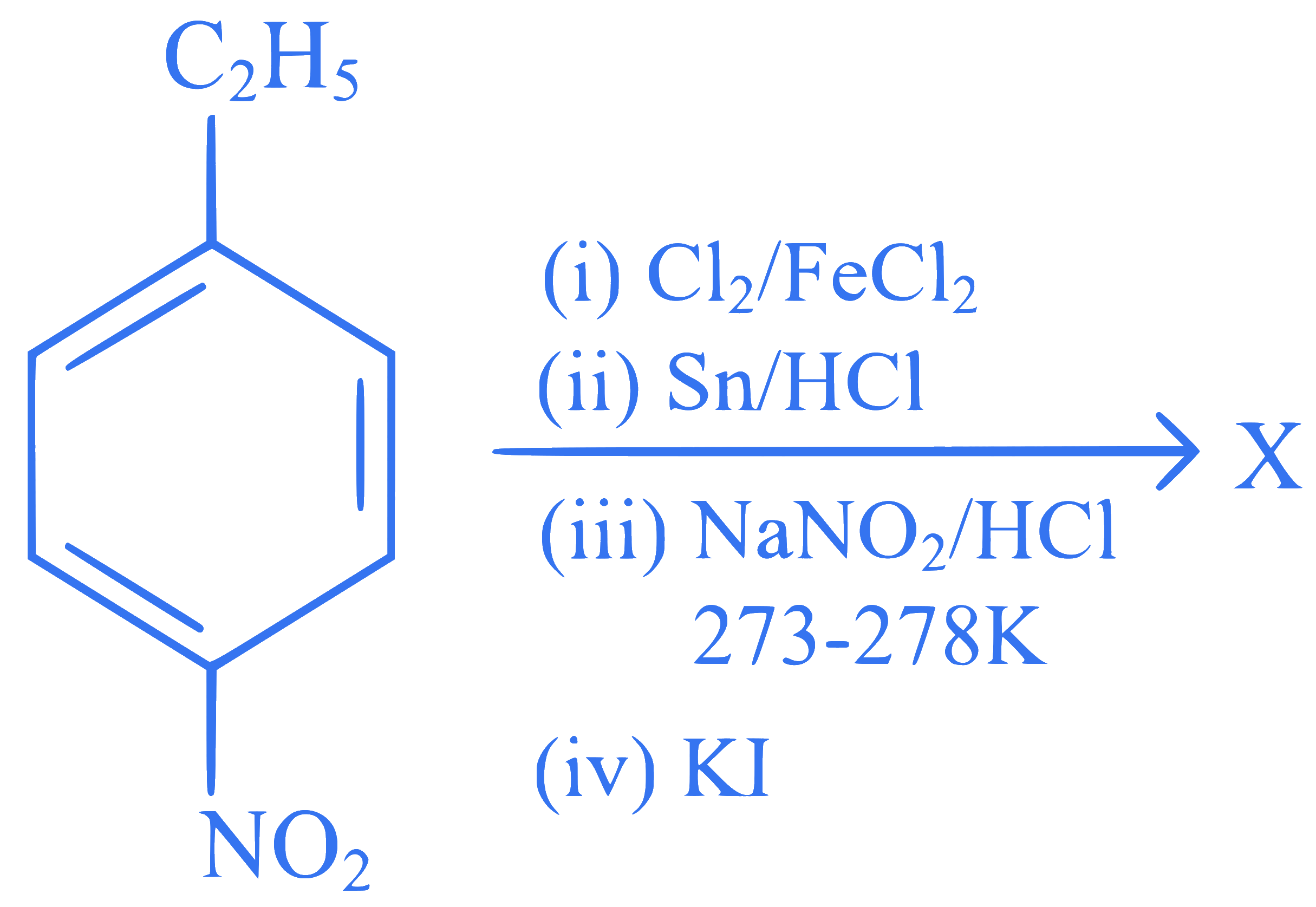
The major product X formed in the following reaction sequence is :

Q48.
mcq single
+4 / 1
The compound that does not undergo Friedel-Crafts alkylation reaction but gives a positive carbylamine test
is :
Q49.
mcq single
+4 / 1
Given below are two statements:
Statement I: The Balmer spectral line for H atom with lowest energy is located at $$\frac{5}{36} R_H \mathrm{~cm}^{-1}$$.
($$\mathrm{R}_{\mathrm{H}}=$$ Rydberg constant)
Statement II: When the temperature of blackbody increases, the maxima of the curve (intensity and wavelength) shifts to shorter wavelength.
In the light of the above statements, choose the correct answer from the options given below:
Q50.
mcq single
+4 / 1
The quantum numbers of four electrons are given below :
I. $$n=4 ; I=2 ; m_1=-2 ; s=-\frac{1}{2}$$
II. $$n=3 ; I=2 ; m_1=1 ; s=+\frac{1}{2}$$
III. $$n=4 ; I=1 ; m_1=0 ; s=+\frac{1}{2}$$
IV. $$n=3 ; I=1 ; m_1=-1 ; s=+\frac{1}{2}$$
The correct decreasing order of energy of these electrons is
Q51.
mcq single
+4 / 1
Identify the incorrect statement.
Q52.
mcq single
+4 / 1
Which of the following pairs of ions will have same spin only magnetic moment values within the pair?
A. $$\mathrm{Zn}^{2+}, \mathrm{Ti}^{2+}$$
B. $$\mathrm{Cr}^{2+}, \mathrm{Fe}^{2+}$$
C. $$\mathrm{Ti}^{3+}, \mathrm{Cu}^{2+}$$
D. $$\mathrm{V}^{2+}, \mathrm{Cu}^{+}$$
Choose the correct answer from the options given below :
Q53.
mcq single
+4 / 1
The UV-visible absorption bands in the spectra of lanthanoid ions are 'X', probably because of the excitation of electrons involving 'Y'. The 'X' and 'Y', respectively, are :
Narrow and d and f orbitals
Q54.
mcq single
+4 / 1
Baeyer's reagent is :
Q55.
mcq single
+4 / 1
Match List I with List II:
List - I
(Block/group in periodic table)
List - II
(Element)
(A)
Lanthanoid
(I)
Ce
(B)
d-block element
(II)
As
(C)
p-block element
(III)
Cs
(D)
s-block element
(IV)
Mn
Choose the correct answer from the options given below:
Q56.
mcq single
+4 / 1
Which of the following set of ions act as oxidising agents?
Q57.
mcq single
+4 / 1
Identify the incorrect statement about $$\mathrm{PCl}_5$$.
Q58.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Molecule
List - II
Bond enthalpy (kJ mol$$^{-1}$$)
(A)
HCl
(I)
435.8
(B)
N$$_2$$
(II)
498
(C)
H$$_2$$
(III)
946.0
(D)
O$$_2$$
(IV)
431.0
Choose the correct answer from the options given below:
Q59.
mcq single
+4 / 1
Which of the following molecules has "NON ZERO" dipole moment value?
Q60.
mcq single
+4 / 1
Arrange the following compounds in increasing order of their solubilities in chloroform:
$$\mathrm{NaCl}, \mathrm{CH}_3 \mathrm{OH}$$, cyclohexane, $$\mathrm{CH}_3 \mathrm{CN}$$
Q61.
mcq single
+4 / 1
Which indicator is used in the titration of sodium hydroxide against oxalic acid and what is the colour change at the end point?
Q62.
mcq single
+4 / 1
The ratio of solubility of AgCl in 0.1 M KCl solution to the solubility of AgCl in water is:
(Given : Solubility product of AgCl = 10$$^{–10}$$)
Q63.
mcq single
+4 / 1
Which of the following plot represents the variation of $$\ln \mathrm{k}$$ versus $$\frac{1}{\mathrm{~T}}$$ in accordance with Arrhenius equation?
Q64.
mcq single
+4 / 1
Following data is for a reaction between reactants A and B :
Rate
$$\mathrm{mol} \mathrm{~L}^{-1} \mathrm{~s}^{-1}$$
$$\mathrm{[A]}$$
$$\mathrm{[B]}$$
$$
2 \times 10^{-3}
$$
0.1 M
0.1 M
$$
4 \times 10^{-3}
$$
0.2 M
0.1 M
$$
1.6 \times 10^{-2}
$$
0.2 M
0.2 M
$$
\text { The order of the reaction with respect to } \mathrm{A} \text { and } \mathrm{B} \text {, respectively, are }
$$
Q65.
mcq single
+4 / 1
Rate constants of a reaction at $$500 \mathrm{~K}$$ and $$700 \mathrm{~K}$$ are $$0.04 \mathrm{~s}^{-1}$$ and $$0.14 \mathrm{~s}^{-1}$$, respectively; then, activation energy of the reaction is :
(Given: $$\log 3.5=0.5441, \mathrm{R}=8.31 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$$)
Q66.
mcq single
+4 / 1
The oxidation states not shown by Mn in given reaction is :
$$
3 \mathrm{MnO}_4^{2-}+4 \mathrm{H}^{+} \longrightarrow 2 \mathrm{MnO}_4^{-}+\mathrm{MnO}_2+2 \mathrm{H}_2 \mathrm{O}
$$
A. +6
B. +2
C. +4
D. +7
E. +3
Choose the most appropriate answer from the options given below :
Q67.
mcq single
+4 / 1
The following reaction method
 is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
 is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
Q68.
mcq single
+4 / 1
The major product $$\mathrm{C}$$ in the below mentioned reaction is:
$$\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{Br} \xrightarrow[\Delta]{\text { alc. } \mathrm{KOH}} \mathrm{A} \xrightarrow{\mathrm{HBr}} \mathrm{B} \xrightarrow[\Delta]{\text { aq. } \mathrm{KOH}} \mathrm{C}$$
Q69.
mcq single
+4 / 1
Mass of glucose $$(\mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_6)$$ required to be dissolved to prepare one litre of its solution which is isotonic with $$15 \mathrm{~g} \mathrm{~L}^{-1}$$ solution of urea $$(\mathrm{NH}_3 \mathrm{CONH}_2)$$ is (Given: Molar mass in $$\mathrm{g} \mathrm{mol}^{-1} \mathrm{C}: 12, \mathrm{H}: 1, \mathrm{O}: 16, \mathrm{~N}: 14$$)
Q70.
mcq single
+4 / 1
On complete combustion, 0.3 g of an organic compound gave 0.2 g of CO$$_2$$ and 0.1 g of H$$_2$$O. The percentage
composition of carbon and hydrogen in the compound, respectively is:
Q71.
mcq single
+4 / 1
$$1.0 \mathrm{~g}$$ of $$\mathrm{H}_2$$ has same number of molecules as in:
Q72.
mcq single
+4 / 1
The amount of glucose required to prepare $$250 \mathrm{~mL}$$ of $$\frac{\mathrm{M}}{20}$$ aqueous solution is :
(Molar mass of glucose : $$180 \mathrm{~g} \mathrm{~mol}^{-1}$$)
Q73.
mcq single
+4 / 1
At a given temperature and pressure, the equilibrium constant values for the equilibria are given below:
$$\begin{aligned}
& 3 \mathrm{~A}_2+\mathrm{B}_2 \rightleftharpoons 2 \mathrm{~A}_3 \mathrm{~B}, \mathrm{~K}_1 \\
& \mathrm{~A}_3 \mathrm{~B} \rightleftharpoons \frac{3}{2} \mathrm{~A}_2+\frac{1}{2} \mathrm{~B}_2, \mathrm{~K}_2
\end{aligned}$$
The relation between $$\mathrm{K}_1$$ and $$\mathrm{K}_2$$ is :
Q74.
mcq single
+4 / 1
For the reaction in equilibrium
$$\mathrm{N}_2(\mathrm{~g})+3 \mathrm{H}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{NH}_3(\mathrm{~g}), \Delta \mathrm{H}=-\mathrm{Q}$$
Reaction is favoured in forward direction by:
Q75.
mcq single
+4 / 1
The correct decreasing order of atomic radii (pm) of $$\mathrm{Li}, \mathrm{Be}, \mathrm{B}$$ and $$\mathrm{C}$$ is
Q76.
mcq single
+4 / 1
Match List I with List II :
List - I
(Atom/Molecule)
List - II
(Property)
(A)
Nitrogen atom
(I)
Paramagnetic
(B)
Fluorine molecule
(II)
Most relative element in group 18
(C)
Oxygen molecule
(III)
Element with highest ionisation enthalpy in group 15
(D)
Xenon atom
(IV)
Strongest oxidising agent
Identify the correct answer from the options given below :
Q77.
mcq single
+4 / 1
Choose the correct statement for the work done in the expansion and heat absorbed or released when
5 litres of an ideal gas at 10 atmospheric pressure isothermally expands into vacuum until volume is
15 litres :
Q78.
mcq single
+4 / 1
For the following reaction at $$300 \mathrm{~K}$$
$$\mathrm{A}_2(\mathrm{~g})+3 \mathrm{~B}_2(\mathrm{~g}) \rightarrow 2 \mathrm{AB}_3(\mathrm{~g})$$
the enthalpy change is $$+15 \mathrm{~kJ}$$, then the internal energy change is :
Q79.
mcq single
+4 / 1
For an endothermic reaction:
(A) $$\mathrm{q}_{\mathrm{p}}$$ is negative.
(B) $$\Delta_{\mathrm{r}} \mathrm{H}$$ is positive.
(C) $$\Delta_r \mathrm{H}$$ is negative.
(D) $$\mathrm{q}_{\mathrm{p}}$$ is positive.
Choose the correct answer from the options given below:
Q80.
mcq single
+4 / 1
Match List I with List II:
List - I
Solid salt treated with dil. H$$_2$$SO$$_4$$
List - II
Anion detected
(A)
effervescence of colourless gas
(I)
NO$$_2^-$$
(B)
gas with smell of rotten egg
(II)
CO$$_3^{2-}$$
(C)
gas with pungent smell
(III)
S$$^{2-}$$
(D)
brown fumes
(IV)
SO$$_3^{2-}$$
Choose the correct answer from the options given below:
Q81.
mcq single
+4 / 1
Identify the incorrect statement from the following :
Q82.
mcq single
+4 / 1
Which of the following is not an ambidentate ligand?
Q83.
mcq single
+4 / 1
Ethylene diaminetetraacetate ion is a/an:
Q84.
mcq single
+4 / 1
$$\left[\mathrm{Mn}_2(\mathrm{CO})_{10}\right]$$ and $$\left[\mathrm{Co}_2(\mathrm{CO})_8\right]$$ structures have
A. Metal-Metal linkage
B. Terminal CO groups
C. Bridging CO groups
D. Metal in zero oxidation state
Choose the correct answer from the options given below
Q85.
mcq single
+4 / 1
Match List-I with List-II :
List - I
(Test/reagent)
List - II
(Radical identified)
(A)
Lake Test
(I)
NO$$_3^-$$
(B)
Nessler's Reagent
(II)
Fe$$^{3+}$$
(C)
Potassium sulphocyanide
(III)
Al$$^{3+}$$
(D)
Brown Ring Test
(IV)
NH$$_4^+$$
Choose the correct answer from the options given below :
Q86.
mcq single
+4 / 1
Methyl group attached to a positively charged carbon atom stabilizes the carbocation due to
Q87.
mcq single
+4 / 1
The correct IUPAC name of the compound
 is :
is :
 is :
is :
Q88.
mcq single
+4 / 1
A steam volatile organic compound which is immiscible with water has a boiling point of $$250^{\circ} \mathrm{C}$$. During steam distillation, a mixture of this organic compound and water will boil :
Q89.
mcq single
+4 / 1
Select the incorrect reaction among the following :
Q90.
mcq single
+4 / 1
Identify D in the following sequence of reactions:
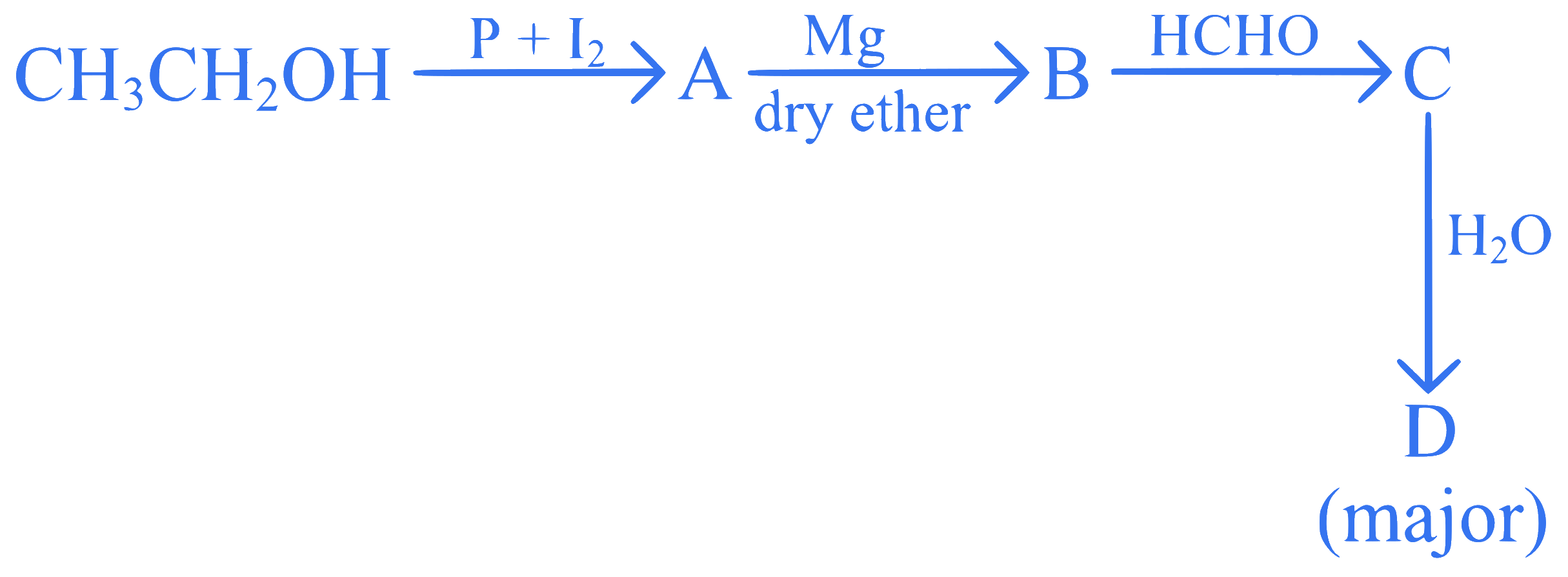
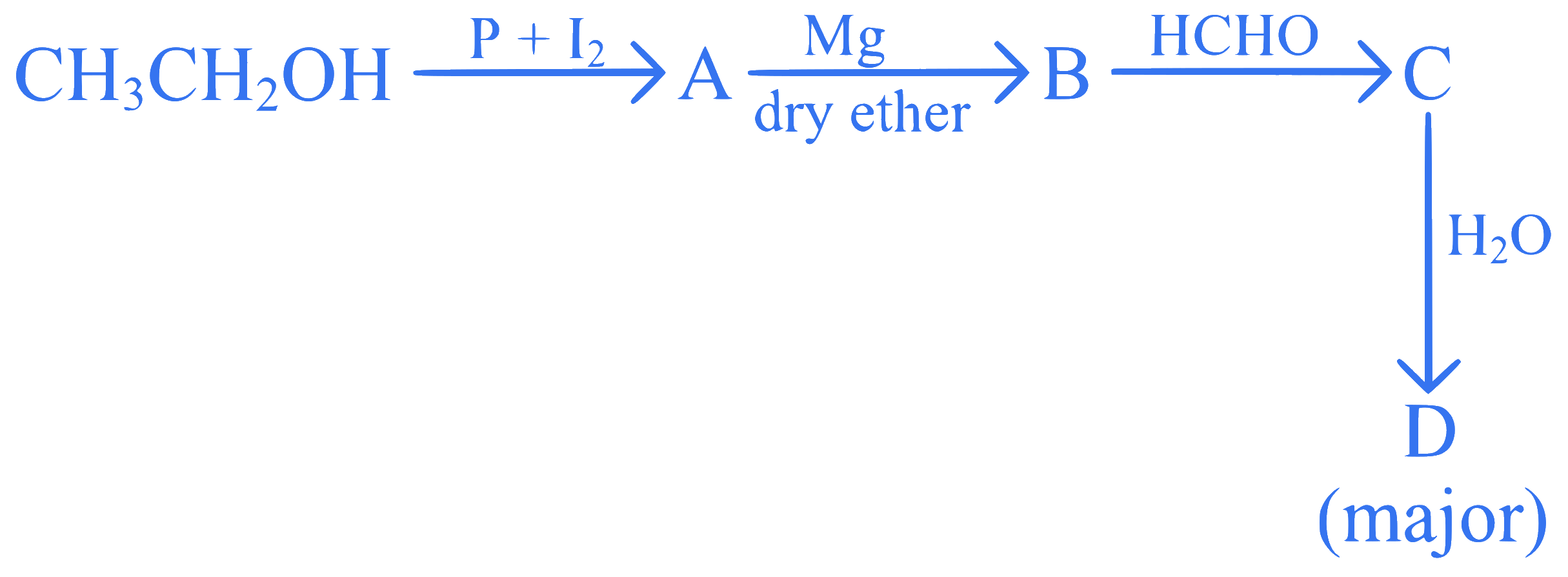
Q91.
mcq single
+4 / 1
The major product D formed in the following reaction sequence is :


Q92.
mcq single
+4 / 1
The alkane that can be oxidized to the corresponding alcohol by KMnO$$_4$$ as per the equation
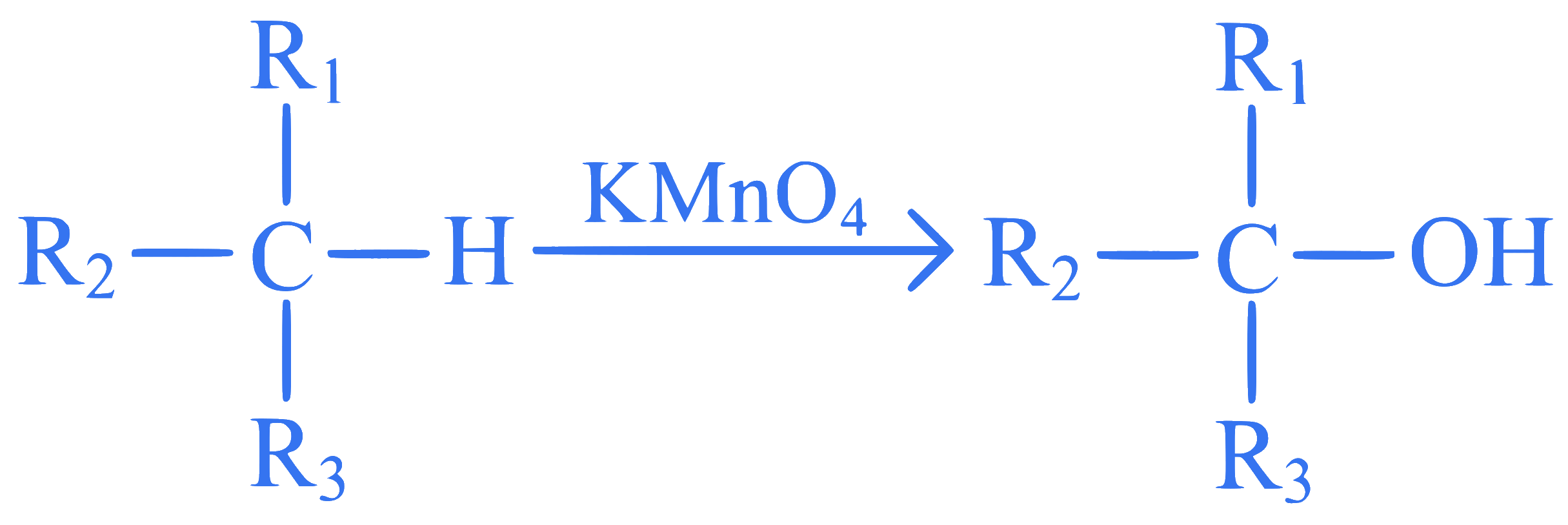 is, when :
is, when :
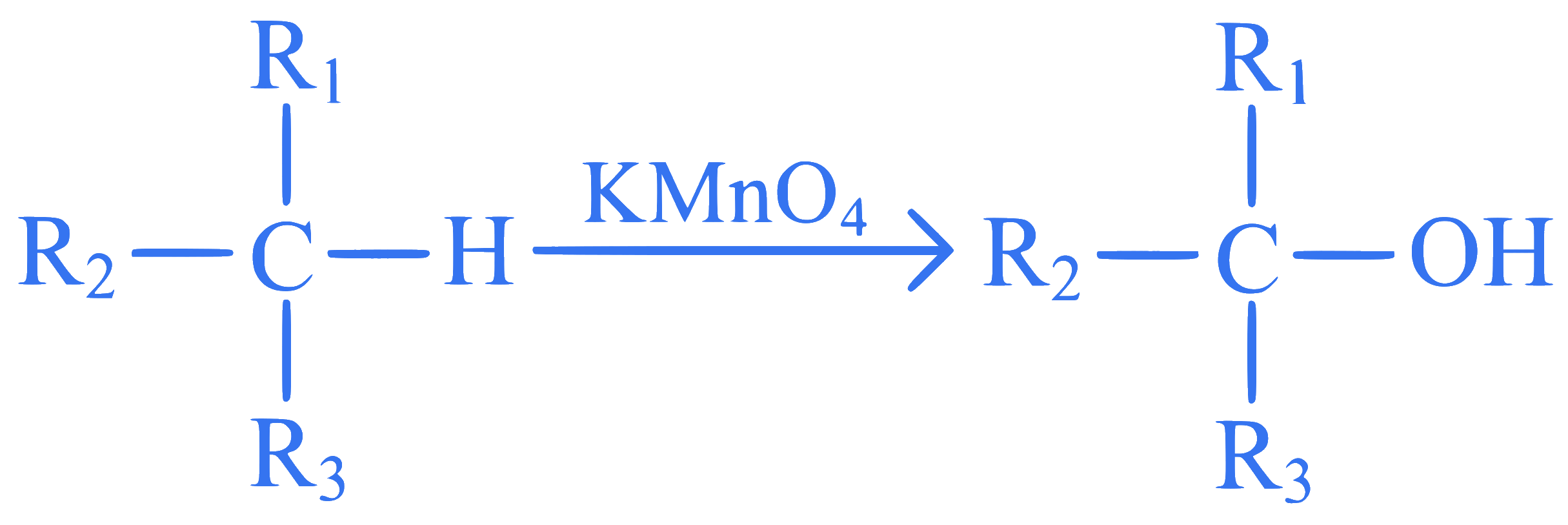 is, when :
is, when :
Q93.
mcq single
+4 / 1
Given below are two statements:
Statement I: Propene on treatment with diborane gives an addition product with the formula
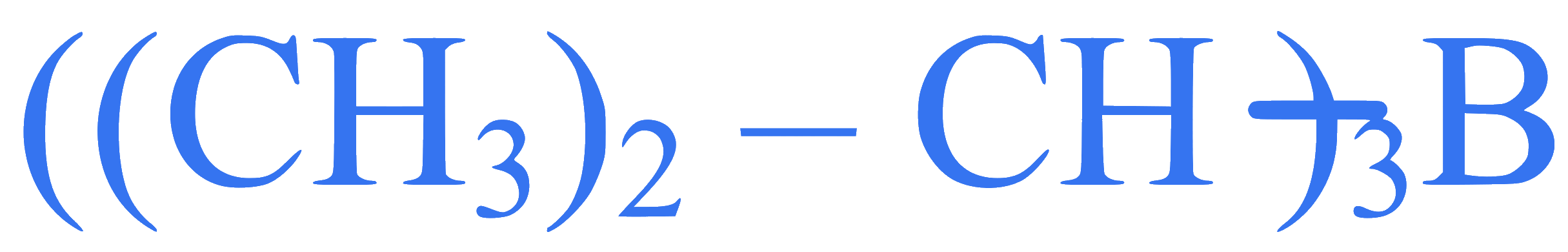 Statement II: Oxidation of
Statement II: Oxidation of
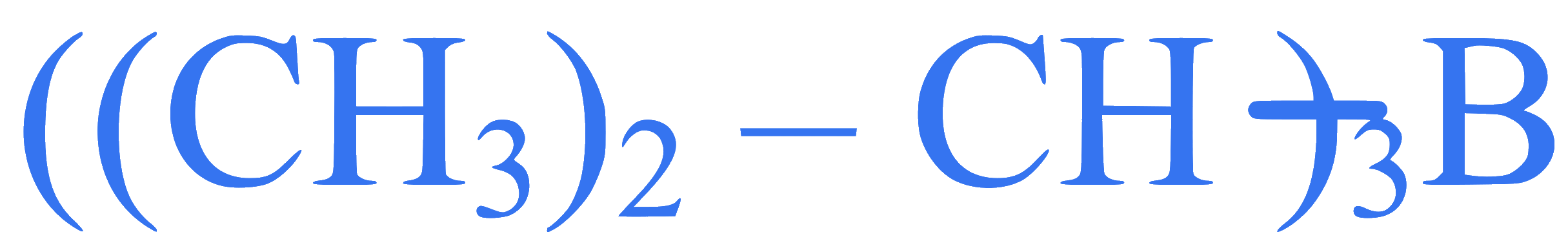 with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
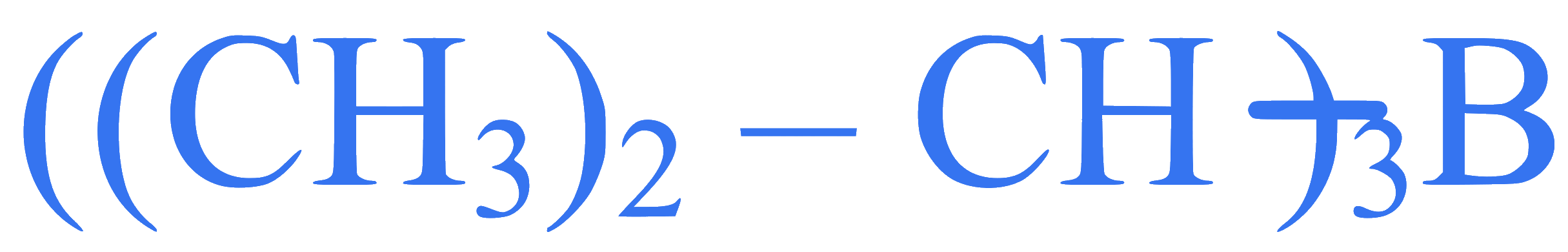 Statement II: Oxidation of
Statement II: Oxidation of
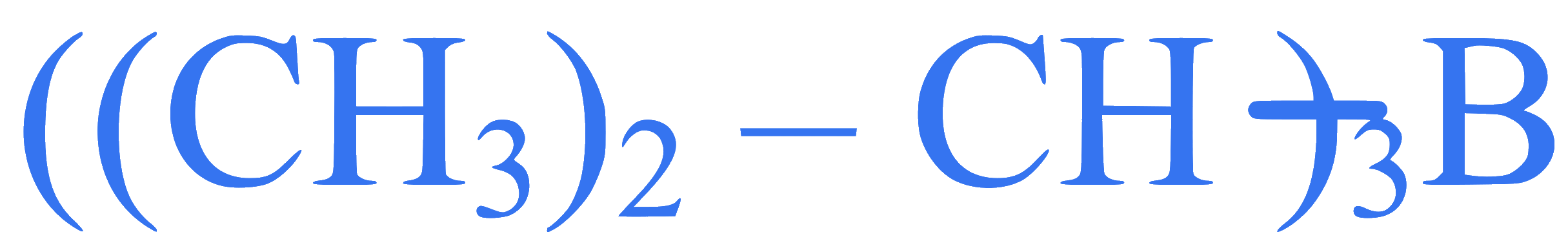 with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
with hydrogen peroxide in presence of $$\mathrm{NaOH}$$ gives propan-2ol.
In the light of the above statements, choose the most appropriate answer from the options given below:
Q94.
mcq single
+4 / 1
Given below are two statements :
Statement I : Glycogen is similar to amylose in its structure.
Statement II : Glycogen is found in yeast and fungi also.
In the light of the above statements, choose the correct answer from the options given below :
Q95.
mcq single
+4 / 1
The standard cell potential of the following cell $$\mathrm{Zn}\left|\mathrm{Zn}^{2+}(\mathrm{aq})\right| \mathrm{Fe}^{2+}(\mathrm{aq}) \mid \mathrm{Fe}$$ is $$0.32 \mathrm{~V}$$. Calculate the standard Gibbs energy change for the reaction:
$$\mathrm{Zn}(\mathrm{s})+\mathrm{Fe}^{2+}(\mathrm{aq}) \rightarrow \mathrm{Zn}^{2+}(\mathrm{aq})+\mathrm{Fe}(\mathrm{s})$$
(Given : $$1 \mathrm{~F}=96487 \mathrm{C}$$)
Q96.
mcq single
+4 / 1
From the following select the one which is not an example of corrosion.
Q97.
mcq single
+4 / 1
The major product X formed in the following reaction sequence is :


Q98.
mcq single
+4 / 1
The compound that does not undergo Friedel-Crafts alkylation reaction but gives a positive carbylamine test
is :
Q99.
mcq single
+4 / 1
Given below are two statements:
Statement I: The Balmer spectral line for H atom with lowest energy is located at $$\frac{5}{36} R_H \mathrm{~cm}^{-1}$$.
($$\mathrm{R}_{\mathrm{H}}=$$ Rydberg constant)
Statement II: When the temperature of blackbody increases, the maxima of the curve (intensity and wavelength) shifts to shorter wavelength.
In the light of the above statements, choose the correct answer from the options given below:
Q100.
mcq single
+4 / 1
The quantum numbers of four electrons are given below :
I. $$n=4 ; I=2 ; m_1=-2 ; s=-\frac{1}{2}$$
II. $$n=3 ; I=2 ; m_1=1 ; s=+\frac{1}{2}$$
III. $$n=4 ; I=1 ; m_1=0 ; s=+\frac{1}{2}$$
IV. $$n=3 ; I=1 ; m_1=-1 ; s=+\frac{1}{2}$$
The correct decreasing order of energy of these electrons is
Physics
Physics
Q1.
mcq single
+4 / 1
Two slits in Young's double slit experiment are $$1.5 \mathrm{~mm}$$ apart and the screen is placed at a distance of $$1 \mathrm{~m}$$ from the slits. If the wavelength of light used is $$600 \times 10^{-9} \mathrm{~m}$$ then the fringe separation is
Q2.
mcq single
+4 / 1
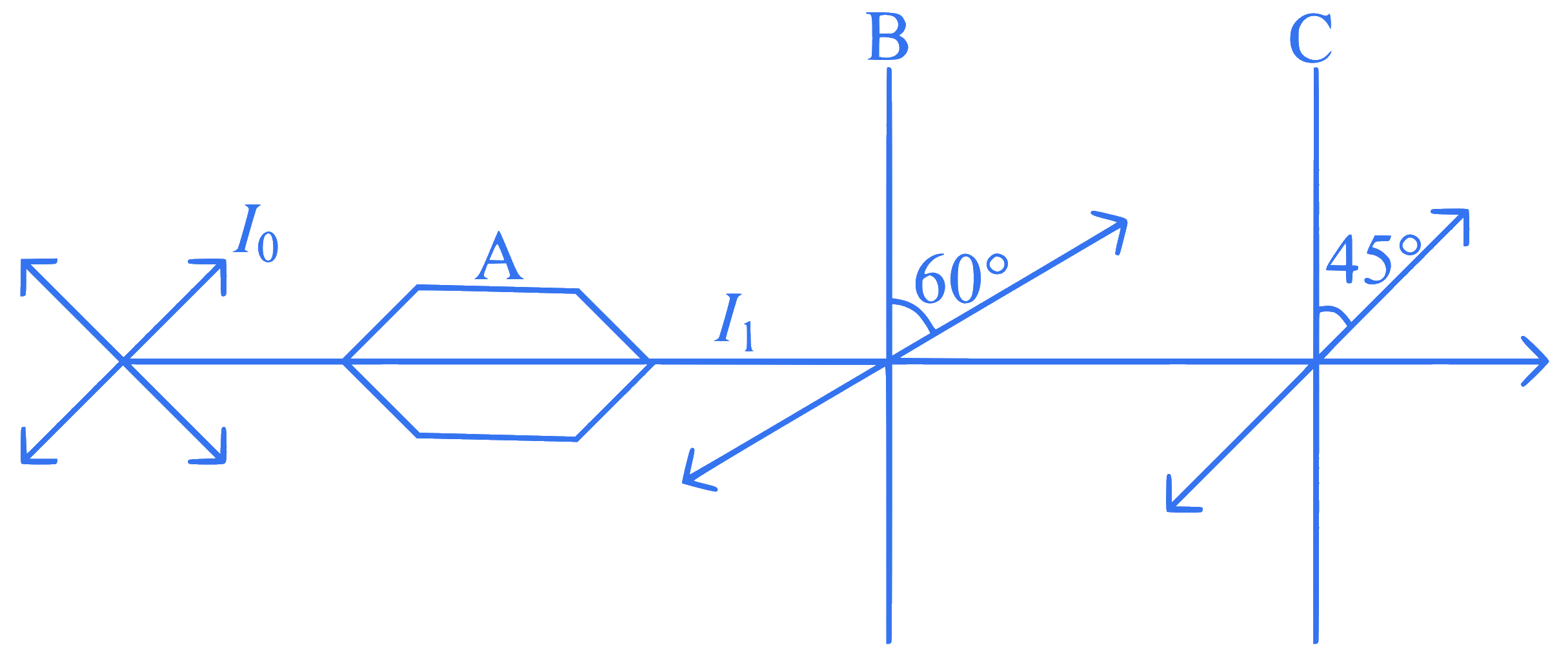
A beam of unpolarized light of intensity I0 is passed through a polaroid A, then through another polaroid B,
oriented at $$60^\circ$$ and finally through another polaroid C, oriented at 45$$^\circ$$ relative to B as shown. The intensity of
emergent light is:

Q3.
mcq single
+4 / 1
Interference pattern can be observed due to superposition of the following waves:
A. $$y=a \sin \omega t$$
B. $$y=a \sin 2 \omega t$$
C. $$y=a \sin (\omega t-\phi)$$
D. $$y=a \sin 3 \omega t$$
Choose the correct answer from the options given below.
Q4.
mcq single
+4 / 1
The radius of gyration of a solid sphere of mass $$5 \mathrm{~kg}$$ about $$X Y$$ is $$5 \mathrm{~m}$$ as shown in figure. The radius of the sphere is $$\frac{5 x}{\sqrt{7}} \mathrm{~m}$$, then the value of $x$ is:


Q5.
mcq single
+4 / 1
Let us consider two solenoids $$A$$ and $$B$$, made from same magnetic material of relative permeability $$\mu_r$$ and equal area of cross-section. Length of $$A$$ is twice that of $$B$$ and the number of turns per unit length in $$A$$ is half that of $$B$$. The ratio of self inductances of the two solenoids, $$L_A: L_B$$ is
Q6.
mcq single
+4 / 1
In an electrical circuit, the voltage is measured as $$V=(200 \pm 4)$$ volt and the current is measured as $$I=(20 \pm 0.2)$$ A. The value of the resistance is:
Q7.
mcq single
+4 / 1
The pitch of an error free screw gauge is $$1 \mathrm{~mm}$$ and there are 100 divisions on the circular scale. While measuring the diameter of a thick wire, the pitch scale reads $$1 \mathrm{~mm}$$ and $$63^{\text {rd }}$$ division on the circular scale coincides with the reference line. The diameter of the wire is:
Q8.
mcq single
+4 / 1
The potential energy of a particle moving along $$x$$-direction varies as $$V=\frac{A x^2}{\sqrt{x}+B}$$. The dimensions of $$\frac{A^2}{B}$$ are:
Q9.
mcq single
+4 / 1
According to the law of equipartition of energy, the number of vibrational modes of a polyatomic gas of constant $$\gamma=\frac{C_p}{C_v}$$ is ($$C_P$$ where $$C_V$$ are the specific heat capacities of the gas at constant pressure and constant volume, respectively):
Q10.
mcq single
+4 / 1
The equilibrium state of a thermodynamic system is described by
A. Pressure
B. Total heat
C. Temperature
D. Volume
E. Work done
Choose the most appropriate answer from the options given below.
Q11.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion $$\mathbf{A}$$ and the other is labelled as Reason $$\mathbf{R}$$.
Assertion A: Houses made of concrete roofs overlaid with foam keep the room hotter during summer.
Reason R: The layer of foam insulation prohibits heat transfer, as it contains air pockets.
In the light of the above statements, choose the correct answer from the options given below.
Q12.
mcq single
+4 / 1
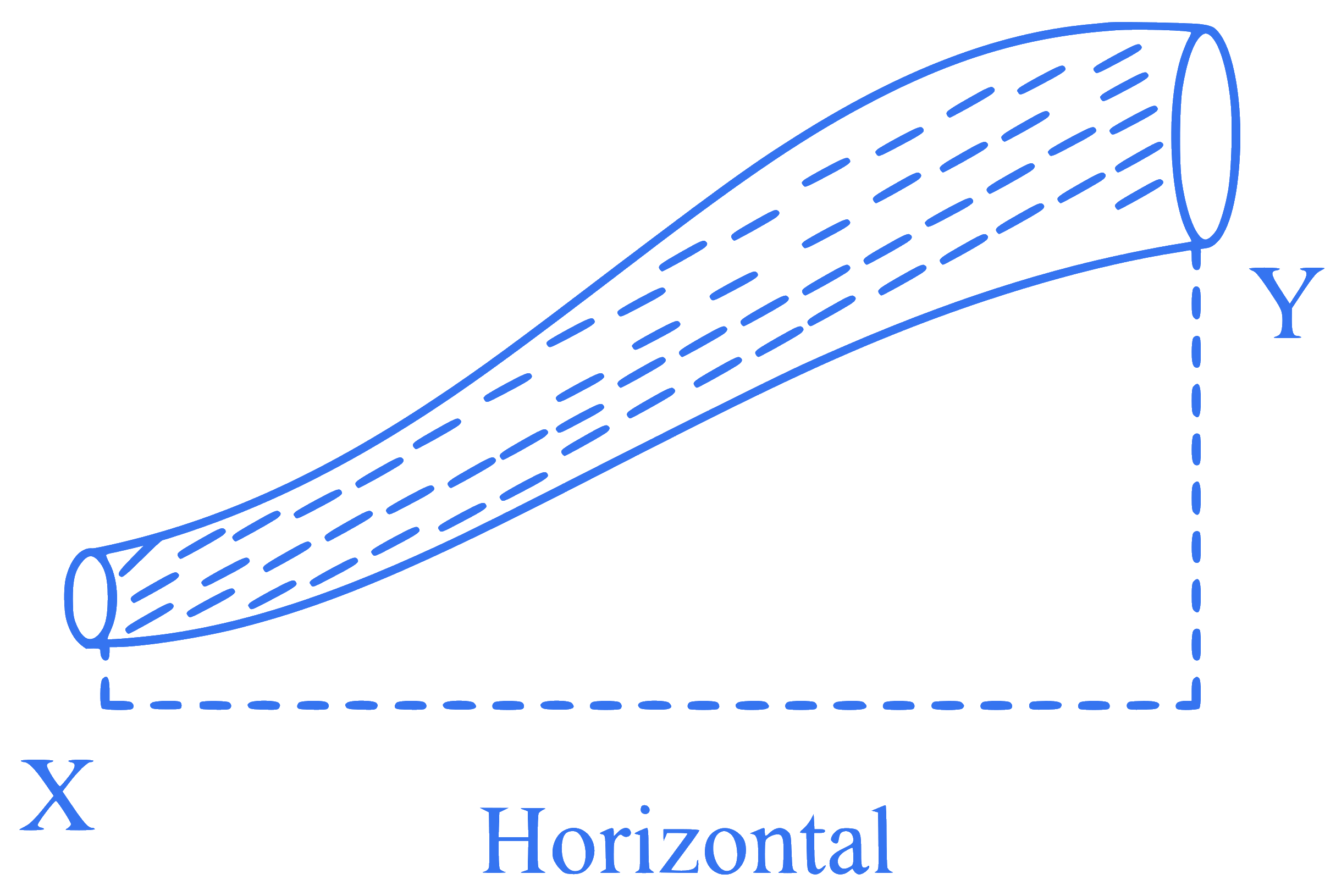
An ideal fluid is flowing in a non-uniform cross-sectional tube $$X Y$$ (as shown in the figure) from end $$X$$ to end $$Y$$. If $$K_1$$ and $$K_2$$ are the kinetic energy per unit volume of the fluid at $$X$$ and $$Y$$ respectively, then the correct option is :

Q13.
mcq single
+4 / 1
The value of electric potential at a distance of $$9 \mathrm{~cm}$$ from the point charge $$4 \times 10^{-7} \mathrm{C}$$ is [Given $$\frac{1}{4 \pi \varepsilon_0}=9 \times 10^9 \mathrm{~N} \mathrm{~m}^2 \mathrm{C}^{-2}$$] :
Q14.
mcq single
+4 / 1
A metal cube of side $$5 \mathrm{~cm}$$ is charged with $$6 \mu \mathrm{C}$$. The surface charge density on the cube is
Q15.
mcq single
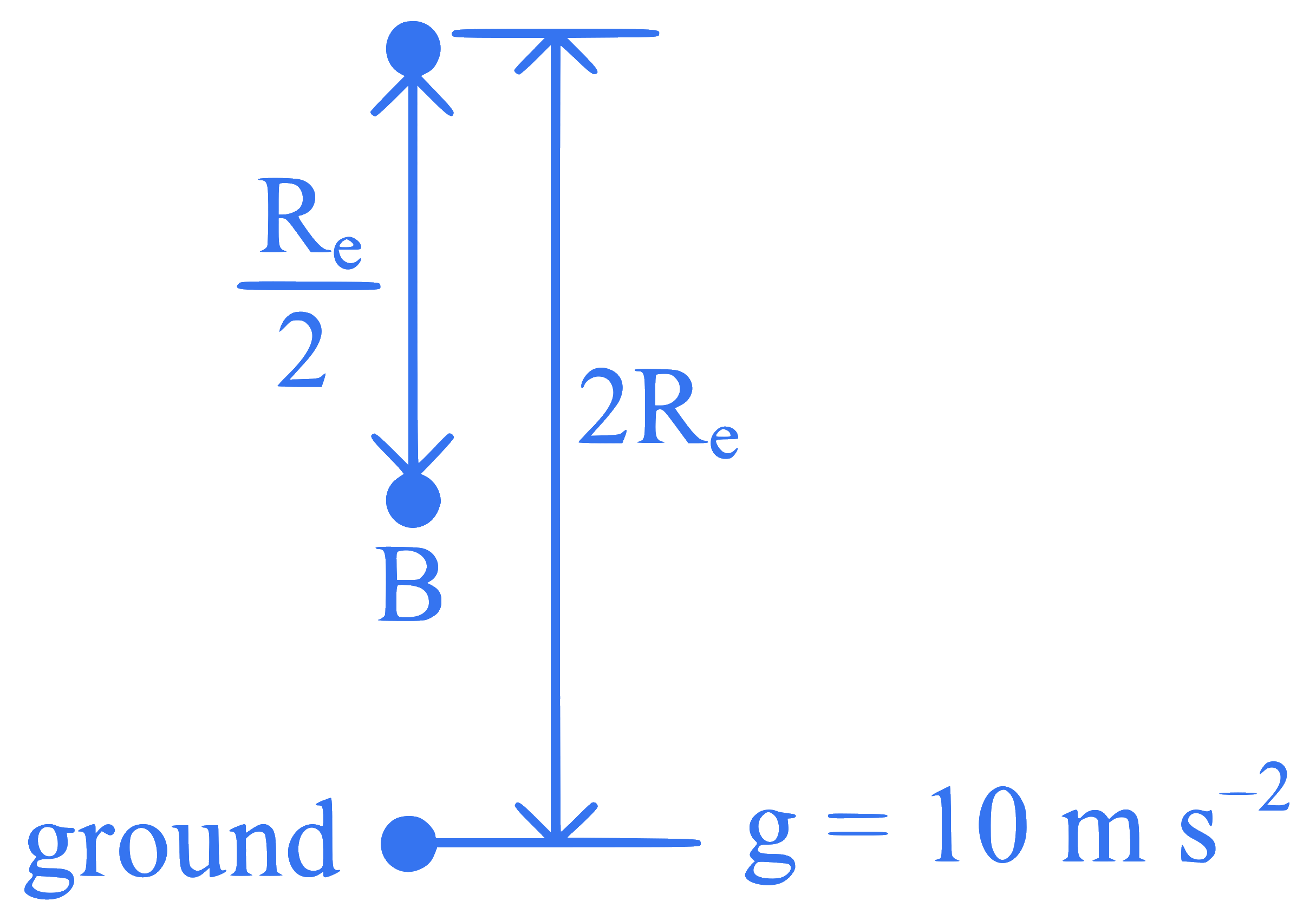
+4 / 1
An object of mass $$100 \mathrm{~kg}$$ falls from point $$A$$ to $$B$$ as shown in figure. The change in its weight, corrected to the nearest integer is ($$R_E$$ is the radius of the earth)

Q16.
mcq single
+4 / 1
The escape velocity for earth is $$v$$. A planet having 9 times mass that of earth and radius, 16 times that of earth, has the escape velocity of:
Q17.
mcq single
+4 / 1
A $$12 \mathrm{~pF}$$ capacitor is connected to a $$50 \mathrm{~V}$$ battery, the electrostatic energy stored in the capacitor in $$\mathrm{nJ}$$ is
Q18.
mcq single
+4 / 1
The capacitance of a capacitor with charge $$q$$ and a potential difference $$V$$ depends on
Q19.
mcq single
+4 / 1
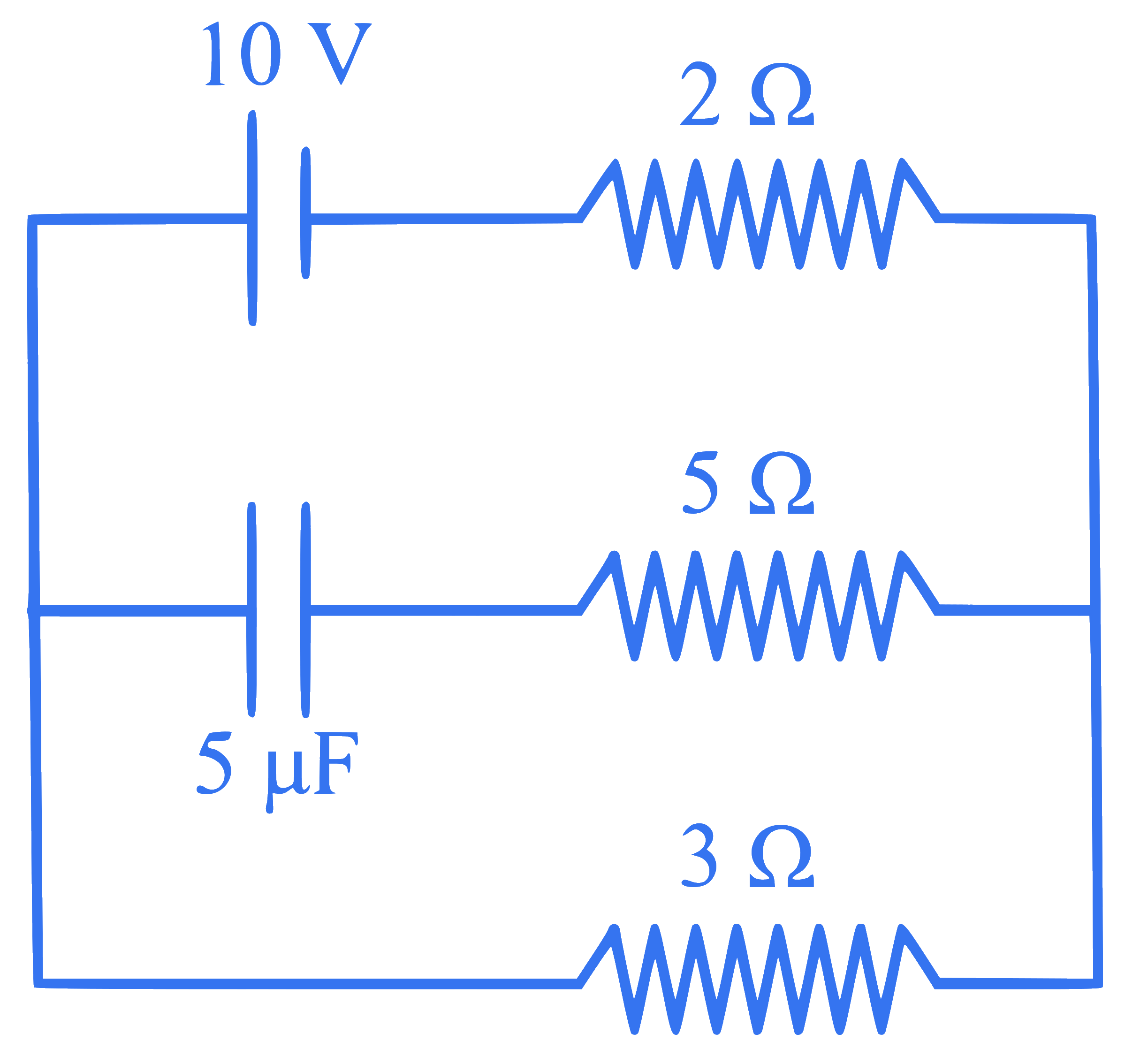
The steady state current in the circuit shown below is :

Q20.
mcq single
+4 / 1
Given below are two statements:
Statement I : Image formation needs regular reflection and/or refraction.
Statement II : The variety in colour of objects we see around us is due to the constituent colours of the light incident on them.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q21.
mcq single
+4 / 1
A particle executing simple harmonic motion with amplitude A has the same potential and kinetic energies at the displacement
Q22.
mcq single
+4 / 1
The two-dimensional motion of a particle, described by $$\vec{r}=(\hat{i}+2 \hat{j}) A \cos \omega t$$ is a/an:
A. parabolic path
B. elliptical path
C. periodic motion
D. simple harmonic motion
Choose the correct answer from the options given below:
Q23.
mcq single
+4 / 1
When the output of an OR gate is applied as input to a NOT gate, then the combination acts as a
Q24.
mcq single
+4 / 1
The output Y for the inputs A and B of the given logic circuit is:
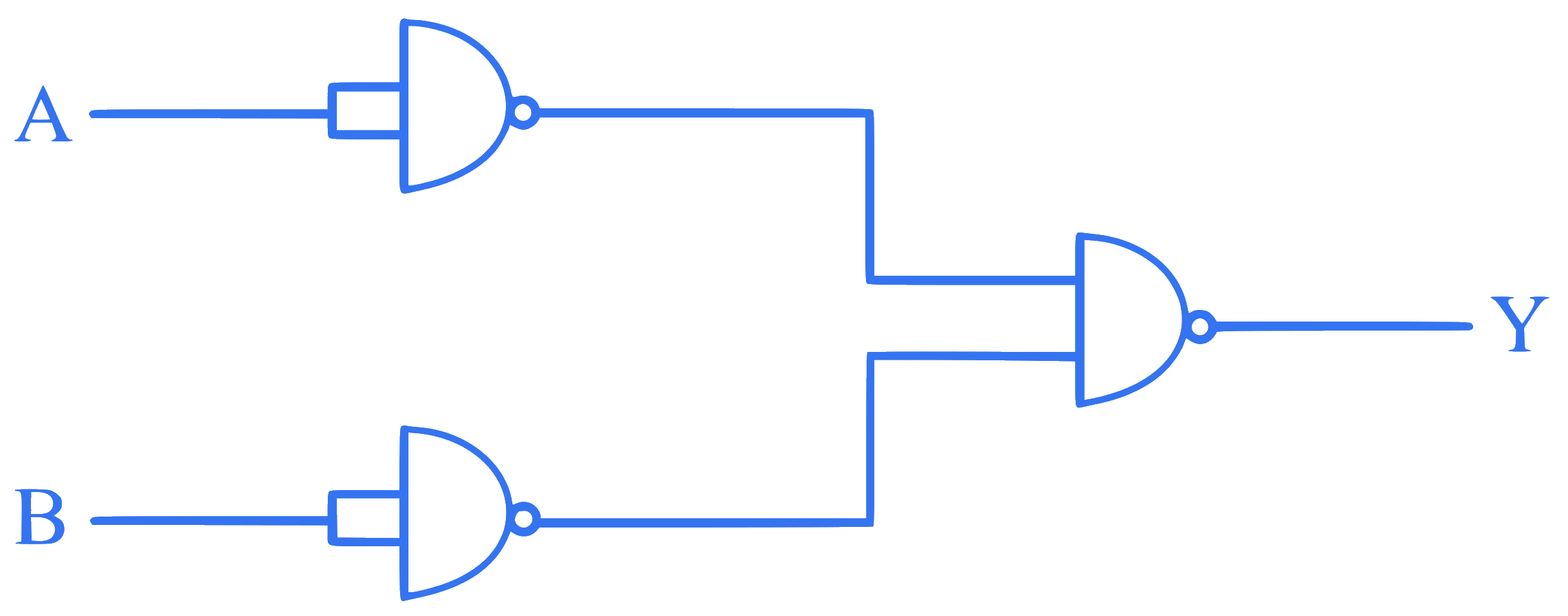

Q25.
mcq single
+4 / 1
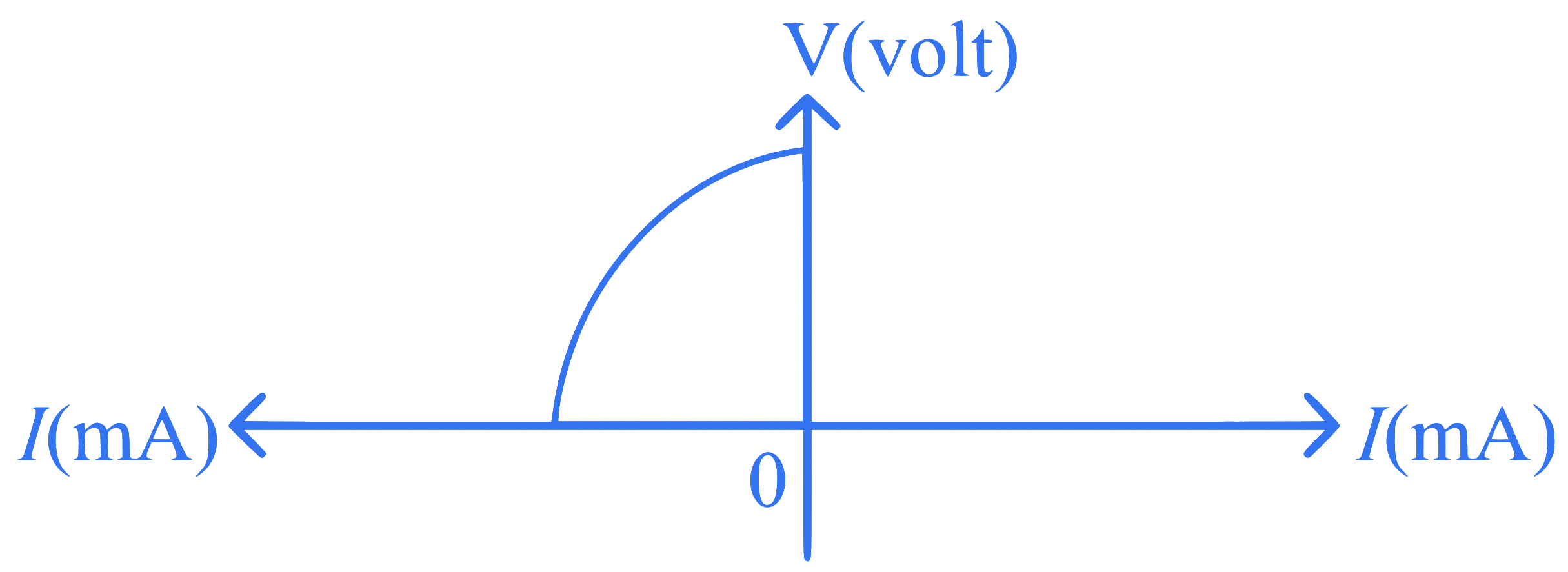 The I-V characteristics shown above are exhibited by a
The I-V characteristics shown above are exhibited by a
 The I-V characteristics shown above are exhibited by a
The I-V characteristics shown above are exhibited by a
Q26.
mcq single
+4 / 1
An electron and an alpha particle are accelerated by the same potential difference. Let $$\lambda_e$$ and $$\lambda_\alpha$$ denote the de-Broglie wavelengths of the electron and the alpha particle, respectively, then:
Q27.
mcq single
+4 / 1
If $$\phi$$ is the work function of photosensitive material in $$\mathrm{eV}$$ and light of wavelength of numerical value $$\lambda=\frac{h c}{e}$$ metre, is incident on it with energy above its threshold value at an instant then the maximum kinetic energy of the photo-electron ejected by it at that instant (Take $$h$$-Plank's constant, $$c$$-velocity of light in free space) is (in SI units):
Q28.
mcq single
+4 / 1
A particle is moving along $$x$$-axis with its position (x) varying with time $$(t)$$ as $$x=\alpha t^4+\beta t^2+\gamma t+\delta$$. The ratio of its initial velocity to its initial acceleration, respectively, is:
Q29.
mcq single
+4 / 1
The spectral series which corresponds to the electronic transition from the levels $$n_2=5,6, \ldots$$ to the level $$n_1=4$$ is
Q30.
mcq single
+4 / 1
Water is used as a coolant in a nuclear reactor because of its
Q31.
mcq single
+4 / 1
Select the correct statements among the following :
A. Slow neutrons can cause fission in $${ }_{92}^{235} \mathrm{U}$$ than fast neutrons.
B. $$\alpha$$-rays are Helium nuclei.
C. $$\beta$$-rays are fast moving electrons or positrons.
D. $$\gamma$$-rays are electromagnetic radiations of wavelengths larger than $$\mathrm{X}$$-rays.
Choose the most appropriate answer from the options given below :
Q32.
mcq single
+4 / 1
Some energy levels of a molecule are shown in the figure with their wavelengths of transitions. Then :
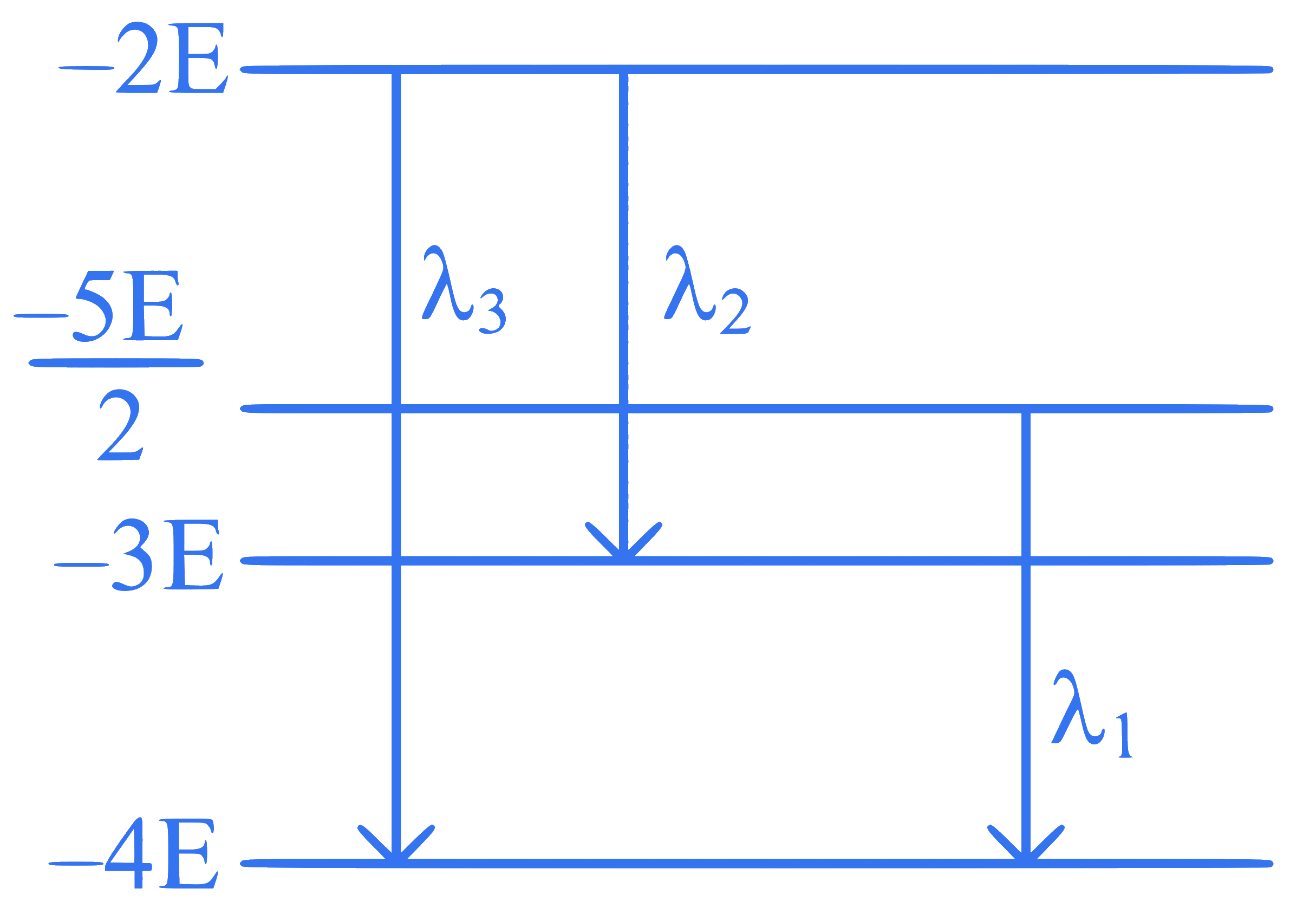

Q33.
mcq single
+4 / 1
Let $$\omega_1, \omega_2$$ and $$\omega_3$$ be the angular speed of the second hand, minute hand and hour hand of a smoothly running analog clock, respectively. If $$x_1, x_2$$ and $$x_3$$ are their respective angular distances in 1 minute then the factor which remains constant $$(k)$$ is
Q34.
mcq single
+4 / 1
A bob is whirled in a horizontal circle by means of a string at an initial speed of $$10 \mathrm{~rpm}$$. If the tension in the string is quadrupled while keeping the radius constant, the new speed is:
Q35.
mcq single
+4 / 1
The displacement of a travelling wave $$y=C \sin \frac{2 \pi}{\lambda}$$ (at $$-x$$) where $$t$$ is time, $$x$$ is distance and $$\lambda$$ is the wavelength, all in S.I. units. Then the frequency of the wave is
Q36.
mcq single
+4 / 1
A box of mass $$5 \mathrm{~kg}$$ is pulled by a cord, up along a frictionless plane inclined at $$30^{\circ}$$ with the horizontal. The tension in the cord is $$30 \mathrm{~N}$$. The acceleration of the box is (Take $$g=10 \mathrm{~m} \mathrm{~s}^{-2}$$)
Q37.
mcq single
+4 / 1
The magnetic moment of an iron bar is $$M$$. It is now bent in such a way that it forms an arc section of a circle subtending an angle of $$60^{\circ}$$ at the centre. The magnetic moment of this arc section is
Q38.
mcq single
+4 / 1
The incorrect relation for a diamagnetic material (all the symbols carry their usual meaning and $$\varepsilon$$ is a small positive number) is
Q39.
mcq single
+4 / 1
The magnetic moment and moment of inertia of a magnetic needle as shown are, respectively, $$1.0 \times 10^{-2} \mathrm{~A} \mathrm{~m}^2$$ and $$\frac{10^{-6}}{\pi^2} \mathrm{~kg} \mathrm{~m}^2$$. If it completes 10 oscillations in $$10 \mathrm{~s}$$, the magnitude of the magnetic field is
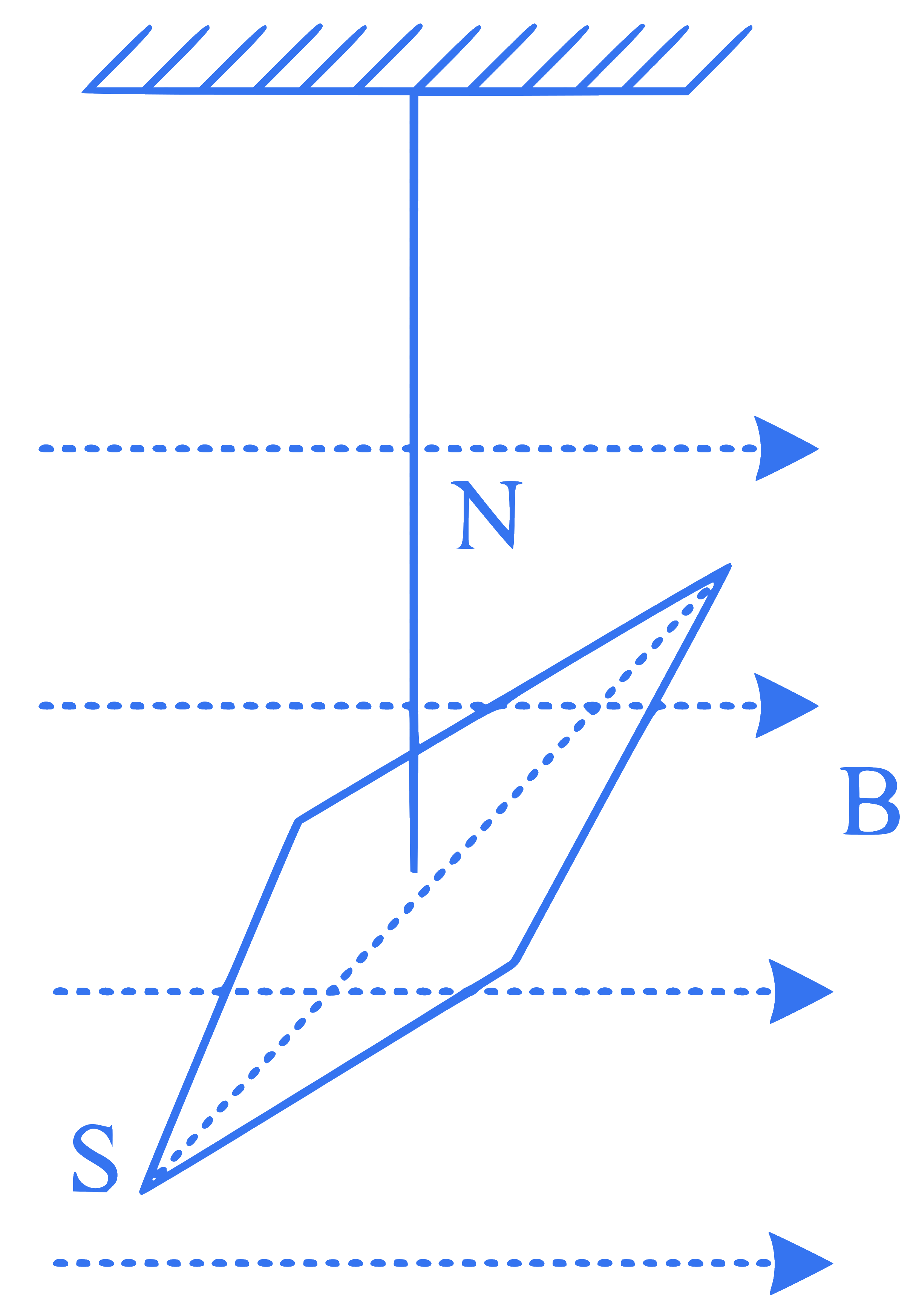

Q40.
mcq single
+4 / 1
The magnetic potential energy, when a magnetic bar of magnetic moment $$\vec{m}$$ is placed perpendicular to the magnetic field $$\vec{B}$$ is
Q41.
mcq single
+4 / 1
An object falls from a height of $$10 \mathrm{~m}$$ above the ground. After striking the ground it loses $$50 \%$$ of its kinetic energy. The height upto which the object can rebounce from the ground is:
Q42.
mcq single
+4 / 1
An object moving along horizontal $$x$$-direction with kinetic energy $$10 \mathrm{~J}$$ is displaced through $$x=(3 \hat{i}) \mathrm{m}$$ by the force $$\vec{F}=(-2 \hat{i}+3 \hat{j}) \mathrm{N}$$. The kinetic energy of the object at the end of the displacement $$x$$ is
Q43.
mcq single
+4 / 1
A uniform wire of diameter $$d$$ carries a current of $$100 \mathrm{~mA}$$ when the mean drift velocity of electrons in the wire is $$v$$. For a wire of diameter $$\frac{d}{2}$$ of the same material to carry a current of $$200 \mathrm{~mA}$$, the mean drift velocity of electrons in the wire is
Q44.
mcq single
+4 / 1
A uniform metal wire of length $$l$$ has $$10 \Omega$$ resistance. Now this wire is stretched to a length $$2l$$ and then bent to form a perfect circle. The equivalent resistance across any arbitrary diameter of that circle is
Q45.
mcq single
+4 / 1
The given circuit shows a uniform straight wire $$A B$$ of $$40 \mathrm{~cm}$$ length fixed at both ends. In order to get zero reading in the galvanometer $$G$$, the free end of $$J$$ is to be placed from $$B$$ at:
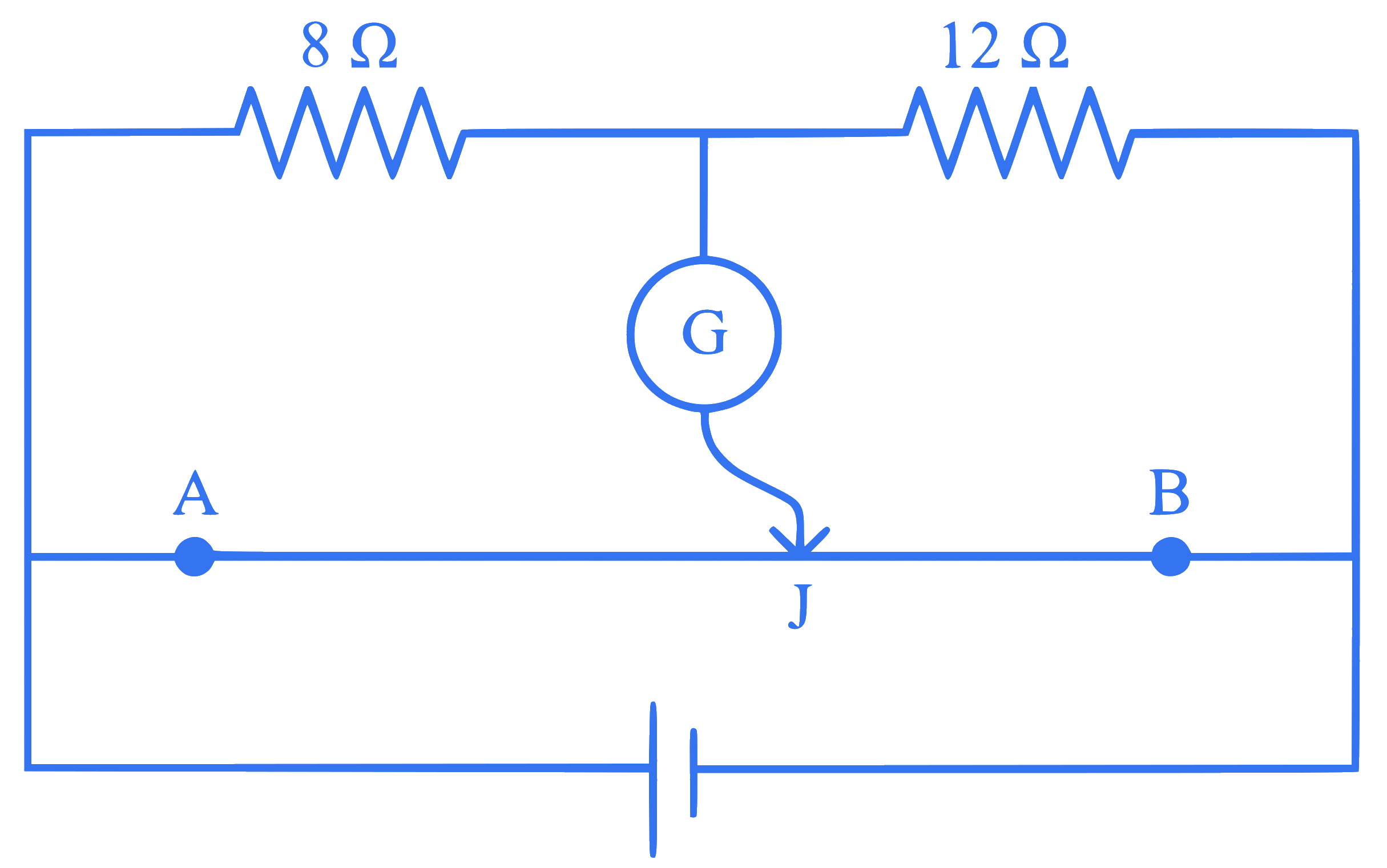

Q46.
mcq single
+4 / 1
If the ratio of relative permeability and relative permittivity of a uniform medium is $$1: 4$$. The ratio of the magnitudes of electric field intensity $$(E)$$ to the magnetic field intensity $$(H)$$ of an EM wave propagating in that medium is
(Given that $$\sqrt{\frac{\mu_0}{\varepsilon_0}}=120 \pi$$):
Q47.
mcq single
+4 / 1
The electromagnetic radiation which has the smallest wavelength are
Q48.
mcq single
+4 / 1
A step up transformer is connected to an ac mains supply of $$220 \mathrm{~V}$$ to operate at $$11000 \mathrm{~V}, 88$$ watt. The current in the secondary circuit, ignoring the power loss in the transformer, is
Q49.
mcq single
+4 / 1
In the circuit shown below, the inductance $$L$$ is connected to an ac source. The current flowing in the circuit is $$I=I_0 \sin \omega t$$. The voltage drop $$\left(V_L\right)$$ across $$L$$ is
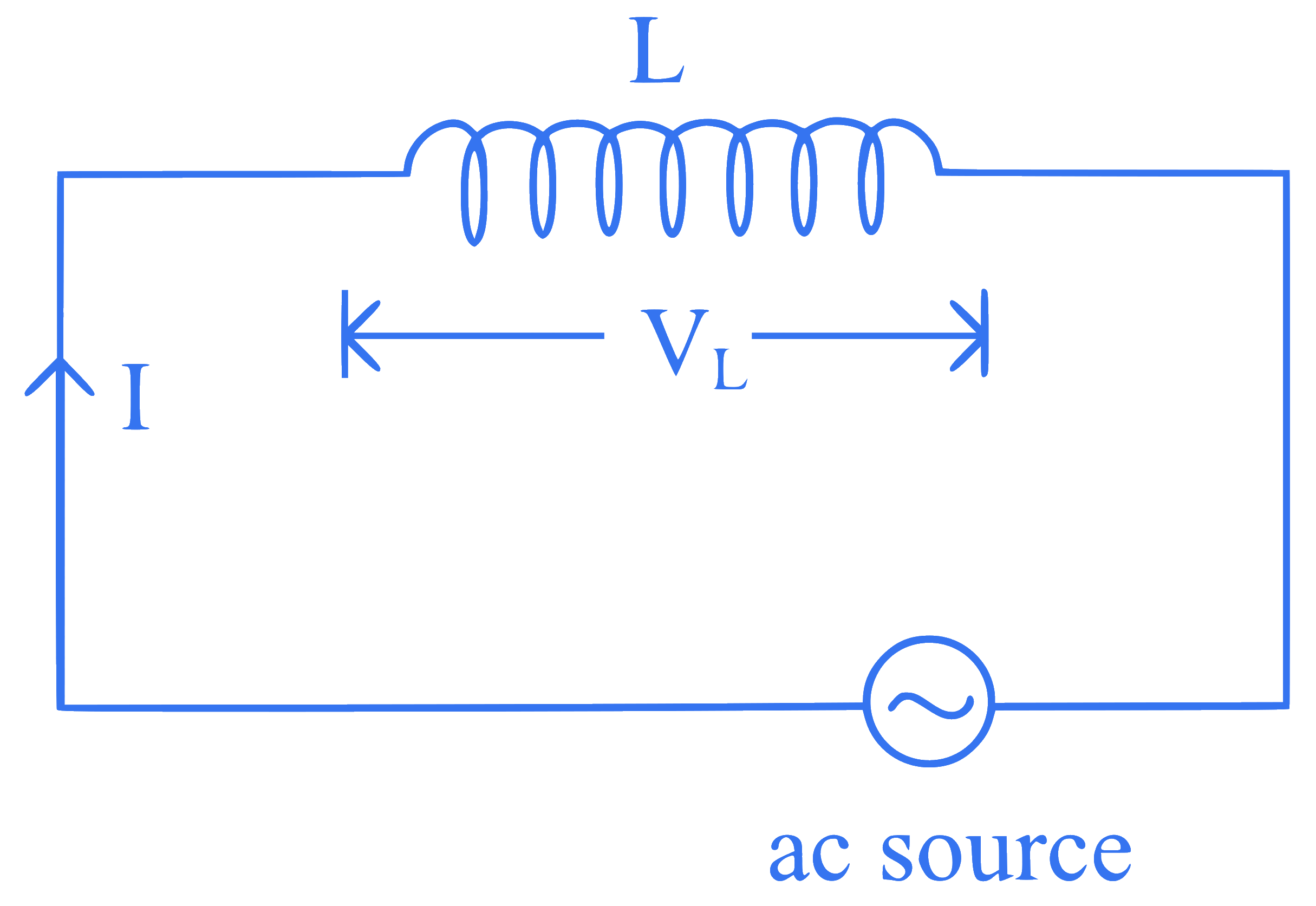

Q50.
mcq single
+4 / 1
The amplitude of the charge oscillating in a circuit decreases exponentially as $$Q=Q_0 e^{-R t/2 L}$$, where $$Q_0$$ is the charge at $$t=0 \mathrm{~s}$$. The time at which charge amplitude decreases to $$0.50 Q_0$$ is nearly:
[Given that $$R=1.5 \Omega, L=12 \mathrm{~mH}, \ln (2)=0.693$$]
Q51.
mcq single
+4 / 1
Two slits in Young's double slit experiment are $$1.5 \mathrm{~mm}$$ apart and the screen is placed at a distance of $$1 \mathrm{~m}$$ from the slits. If the wavelength of light used is $$600 \times 10^{-9} \mathrm{~m}$$ then the fringe separation is
Q52.
mcq single
+4 / 1
A beam of unpolarized light of intensity I0 is passed through a polaroid A, then through another polaroid B,
oriented at $$60^\circ$$ and finally through another polaroid C, oriented at 45$$^\circ$$ relative to B as shown. The intensity of
emergent light is:


Q53.
mcq single
+4 / 1
Interference pattern can be observed due to superposition of the following waves:
A. $$y=a \sin \omega t$$
B. $$y=a \sin 2 \omega t$$
C. $$y=a \sin (\omega t-\phi)$$
D. $$y=a \sin 3 \omega t$$
Choose the correct answer from the options given below.
Q54.
mcq single
+4 / 1
The radius of gyration of a solid sphere of mass $$5 \mathrm{~kg}$$ about $$X Y$$ is $$5 \mathrm{~m}$$ as shown in figure. The radius of the sphere is $$\frac{5 x}{\sqrt{7}} \mathrm{~m}$$, then the value of $x$ is:


Q55.
mcq single
+4 / 1
Let us consider two solenoids $$A$$ and $$B$$, made from same magnetic material of relative permeability $$\mu_r$$ and equal area of cross-section. Length of $$A$$ is twice that of $$B$$ and the number of turns per unit length in $$A$$ is half that of $$B$$. The ratio of self inductances of the two solenoids, $$L_A: L_B$$ is
Q56.
mcq single
+4 / 1
In an electrical circuit, the voltage is measured as $$V=(200 \pm 4)$$ volt and the current is measured as $$I=(20 \pm 0.2)$$ A. The value of the resistance is:
Q57.
mcq single
+4 / 1
The pitch of an error free screw gauge is $$1 \mathrm{~mm}$$ and there are 100 divisions on the circular scale. While measuring the diameter of a thick wire, the pitch scale reads $$1 \mathrm{~mm}$$ and $$63^{\text {rd }}$$ division on the circular scale coincides with the reference line. The diameter of the wire is:
Q58.
mcq single
+4 / 1
The potential energy of a particle moving along $$x$$-direction varies as $$V=\frac{A x^2}{\sqrt{x}+B}$$. The dimensions of $$\frac{A^2}{B}$$ are:
Q59.
mcq single
+4 / 1
According to the law of equipartition of energy, the number of vibrational modes of a polyatomic gas of constant $$\gamma=\frac{C_p}{C_v}$$ is ($$C_P$$ where $$C_V$$ are the specific heat capacities of the gas at constant pressure and constant volume, respectively):
Q60.
mcq single
+4 / 1
The equilibrium state of a thermodynamic system is described by
A. Pressure
B. Total heat
C. Temperature
D. Volume
E. Work done
Choose the most appropriate answer from the options given below.
Q61.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion $$\mathbf{A}$$ and the other is labelled as Reason $$\mathbf{R}$$.
Assertion A: Houses made of concrete roofs overlaid with foam keep the room hotter during summer.
Reason R: The layer of foam insulation prohibits heat transfer, as it contains air pockets.
In the light of the above statements, choose the correct answer from the options given below.
Q62.
mcq single
+4 / 1
An ideal fluid is flowing in a non-uniform cross-sectional tube $$X Y$$ (as shown in the figure) from end $$X$$ to end $$Y$$. If $$K_1$$ and $$K_2$$ are the kinetic energy per unit volume of the fluid at $$X$$ and $$Y$$ respectively, then the correct option is :


Q63.
mcq single
+4 / 1
The value of electric potential at a distance of $$9 \mathrm{~cm}$$ from the point charge $$4 \times 10^{-7} \mathrm{C}$$ is [Given $$\frac{1}{4 \pi \varepsilon_0}=9 \times 10^9 \mathrm{~N} \mathrm{~m}^2 \mathrm{C}^{-2}$$] :
Q64.
mcq single
+4 / 1
A metal cube of side $$5 \mathrm{~cm}$$ is charged with $$6 \mu \mathrm{C}$$. The surface charge density on the cube is
Q65.
mcq single
+4 / 1
An object of mass $$100 \mathrm{~kg}$$ falls from point $$A$$ to $$B$$ as shown in figure. The change in its weight, corrected to the nearest integer is ($$R_E$$ is the radius of the earth)


Q66.
mcq single
+4 / 1
The escape velocity for earth is $$v$$. A planet having 9 times mass that of earth and radius, 16 times that of earth, has the escape velocity of:
Q67.
mcq single
+4 / 1
A $$12 \mathrm{~pF}$$ capacitor is connected to a $$50 \mathrm{~V}$$ battery, the electrostatic energy stored in the capacitor in $$\mathrm{nJ}$$ is
Q68.
mcq single
+4 / 1
The capacitance of a capacitor with charge $$q$$ and a potential difference $$V$$ depends on
Q69.
mcq single
+4 / 1
The steady state current in the circuit shown below is :


Q70.
mcq single
+4 / 1
Given below are two statements:
Statement I : Image formation needs regular reflection and/or refraction.
Statement II : The variety in colour of objects we see around us is due to the constituent colours of the light incident on them.
In the light of the above statements, choose the most appropriate answer from the options given below :
Q71.
mcq single
+4 / 1
A particle executing simple harmonic motion with amplitude A has the same potential and kinetic energies at the displacement
Q72.
mcq single
+4 / 1
The two-dimensional motion of a particle, described by $$\vec{r}=(\hat{i}+2 \hat{j}) A \cos \omega t$$ is a/an:
A. parabolic path
B. elliptical path
C. periodic motion
D. simple harmonic motion
Choose the correct answer from the options given below:
Q73.
mcq single
+4 / 1
When the output of an OR gate is applied as input to a NOT gate, then the combination acts as a
Q74.
mcq single
+4 / 1
The output Y for the inputs A and B of the given logic circuit is:


Q75.
mcq single
+4 / 1
 The I-V characteristics shown above are exhibited by a
The I-V characteristics shown above are exhibited by a
 The I-V characteristics shown above are exhibited by a
The I-V characteristics shown above are exhibited by a
Q76.
mcq single
+4 / 1
An electron and an alpha particle are accelerated by the same potential difference. Let $$\lambda_e$$ and $$\lambda_\alpha$$ denote the de-Broglie wavelengths of the electron and the alpha particle, respectively, then:
Q77.
mcq single
+4 / 1
If $$\phi$$ is the work function of photosensitive material in $$\mathrm{eV}$$ and light of wavelength of numerical value $$\lambda=\frac{h c}{e}$$ metre, is incident on it with energy above its threshold value at an instant then the maximum kinetic energy of the photo-electron ejected by it at that instant (Take $$h$$-Plank's constant, $$c$$-velocity of light in free space) is (in SI units):
Q78.
mcq single
+4 / 1
A particle is moving along $$x$$-axis with its position (x) varying with time $$(t)$$ as $$x=\alpha t^4+\beta t^2+\gamma t+\delta$$. The ratio of its initial velocity to its initial acceleration, respectively, is:
Q79.
mcq single
+4 / 1
The spectral series which corresponds to the electronic transition from the levels $$n_2=5,6, \ldots$$ to the level $$n_1=4$$ is
Q80.
mcq single
+4 / 1
Water is used as a coolant in a nuclear reactor because of its
Q81.
mcq single
+4 / 1
Select the correct statements among the following :
A. Slow neutrons can cause fission in $${ }_{92}^{235} \mathrm{U}$$ than fast neutrons.
B. $$\alpha$$-rays are Helium nuclei.
C. $$\beta$$-rays are fast moving electrons or positrons.
D. $$\gamma$$-rays are electromagnetic radiations of wavelengths larger than $$\mathrm{X}$$-rays.
Choose the most appropriate answer from the options given below :
Q82.
mcq single
+4 / 1
Some energy levels of a molecule are shown in the figure with their wavelengths of transitions. Then :


Q83.
mcq single
+4 / 1
Let $$\omega_1, \omega_2$$ and $$\omega_3$$ be the angular speed of the second hand, minute hand and hour hand of a smoothly running analog clock, respectively. If $$x_1, x_2$$ and $$x_3$$ are their respective angular distances in 1 minute then the factor which remains constant $$(k)$$ is
Q84.
mcq single
+4 / 1
A bob is whirled in a horizontal circle by means of a string at an initial speed of $$10 \mathrm{~rpm}$$. If the tension in the string is quadrupled while keeping the radius constant, the new speed is:
Q85.
mcq single
+4 / 1
The displacement of a travelling wave $$y=C \sin \frac{2 \pi}{\lambda}$$ (at $$-x$$) where $$t$$ is time, $$x$$ is distance and $$\lambda$$ is the wavelength, all in S.I. units. Then the frequency of the wave is
Q86.
mcq single
+4 / 1
A box of mass $$5 \mathrm{~kg}$$ is pulled by a cord, up along a frictionless plane inclined at $$30^{\circ}$$ with the horizontal. The tension in the cord is $$30 \mathrm{~N}$$. The acceleration of the box is (Take $$g=10 \mathrm{~m} \mathrm{~s}^{-2}$$)
Q87.
mcq single
+4 / 1
The magnetic moment of an iron bar is $$M$$. It is now bent in such a way that it forms an arc section of a circle subtending an angle of $$60^{\circ}$$ at the centre. The magnetic moment of this arc section is
Q88.
mcq single
+4 / 1
The incorrect relation for a diamagnetic material (all the symbols carry their usual meaning and $$\varepsilon$$ is a small positive number) is
Q89.
mcq single
+4 / 1
The magnetic moment and moment of inertia of a magnetic needle as shown are, respectively, $$1.0 \times 10^{-2} \mathrm{~A} \mathrm{~m}^2$$ and $$\frac{10^{-6}}{\pi^2} \mathrm{~kg} \mathrm{~m}^2$$. If it completes 10 oscillations in $$10 \mathrm{~s}$$, the magnitude of the magnetic field is


Q90.
mcq single
+4 / 1
The magnetic potential energy, when a magnetic bar of magnetic moment $$\vec{m}$$ is placed perpendicular to the magnetic field $$\vec{B}$$ is
Q91.
mcq single
+4 / 1
An object falls from a height of $$10 \mathrm{~m}$$ above the ground. After striking the ground it loses $$50 \%$$ of its kinetic energy. The height upto which the object can rebounce from the ground is:
Q92.
mcq single
+4 / 1
An object moving along horizontal $$x$$-direction with kinetic energy $$10 \mathrm{~J}$$ is displaced through $$x=(3 \hat{i}) \mathrm{m}$$ by the force $$\vec{F}=(-2 \hat{i}+3 \hat{j}) \mathrm{N}$$. The kinetic energy of the object at the end of the displacement $$x$$ is
Q93.
mcq single
+4 / 1
A uniform wire of diameter $$d$$ carries a current of $$100 \mathrm{~mA}$$ when the mean drift velocity of electrons in the wire is $$v$$. For a wire of diameter $$\frac{d}{2}$$ of the same material to carry a current of $$200 \mathrm{~mA}$$, the mean drift velocity of electrons in the wire is
Q94.
mcq single
+4 / 1
A uniform metal wire of length $$l$$ has $$10 \Omega$$ resistance. Now this wire is stretched to a length $$2l$$ and then bent to form a perfect circle. The equivalent resistance across any arbitrary diameter of that circle is
Q95.
mcq single
+4 / 1
The given circuit shows a uniform straight wire $$A B$$ of $$40 \mathrm{~cm}$$ length fixed at both ends. In order to get zero reading in the galvanometer $$G$$, the free end of $$J$$ is to be placed from $$B$$ at:


Q96.
mcq single
+4 / 1
If the ratio of relative permeability and relative permittivity of a uniform medium is $$1: 4$$. The ratio of the magnitudes of electric field intensity $$(E)$$ to the magnetic field intensity $$(H)$$ of an EM wave propagating in that medium is
(Given that $$\sqrt{\frac{\mu_0}{\varepsilon_0}}=120 \pi$$):
Q97.
mcq single
+4 / 1
The electromagnetic radiation which has the smallest wavelength are
Q98.
mcq single
+4 / 1
A step up transformer is connected to an ac mains supply of $$220 \mathrm{~V}$$ to operate at $$11000 \mathrm{~V}, 88$$ watt. The current in the secondary circuit, ignoring the power loss in the transformer, is
Q99.
mcq single
+4 / 1
In the circuit shown below, the inductance $$L$$ is connected to an ac source. The current flowing in the circuit is $$I=I_0 \sin \omega t$$. The voltage drop $$\left(V_L\right)$$ across $$L$$ is


Q100.
mcq single
+4 / 1
The amplitude of the charge oscillating in a circuit decreases exponentially as $$Q=Q_0 e^{-R t/2 L}$$, where $$Q_0$$ is the charge at $$t=0 \mathrm{~s}$$. The time at which charge amplitude decreases to $$0.50 Q_0$$ is nearly:
[Given that $$R=1.5 \Omega, L=12 \mathrm{~mH}, \ln (2)=0.693$$]
Biology
Biology
Q1.
mcq single
+4 / 1
Match List-I with List-II relating to microbes and their products:
List - I
(Microbes)
List - II
(Products)
(A)
Streptococcus
(I)
Citric acid
(B)
Trichoderma polysporum
(II)
Clot buster
(C)
Monascus purpureus
(III)
Cyclosporin A
(D)
Aspergillus niger
(IV)
Statins
Choose the correct answer from the options given below:
Q2.
mcq single
+4 / 1
Which one of the following is not a limitation of ecological pyramids?
Q3.
mcq single
+4 / 1
Consider the pyramid of energy of an ecosystem given below:
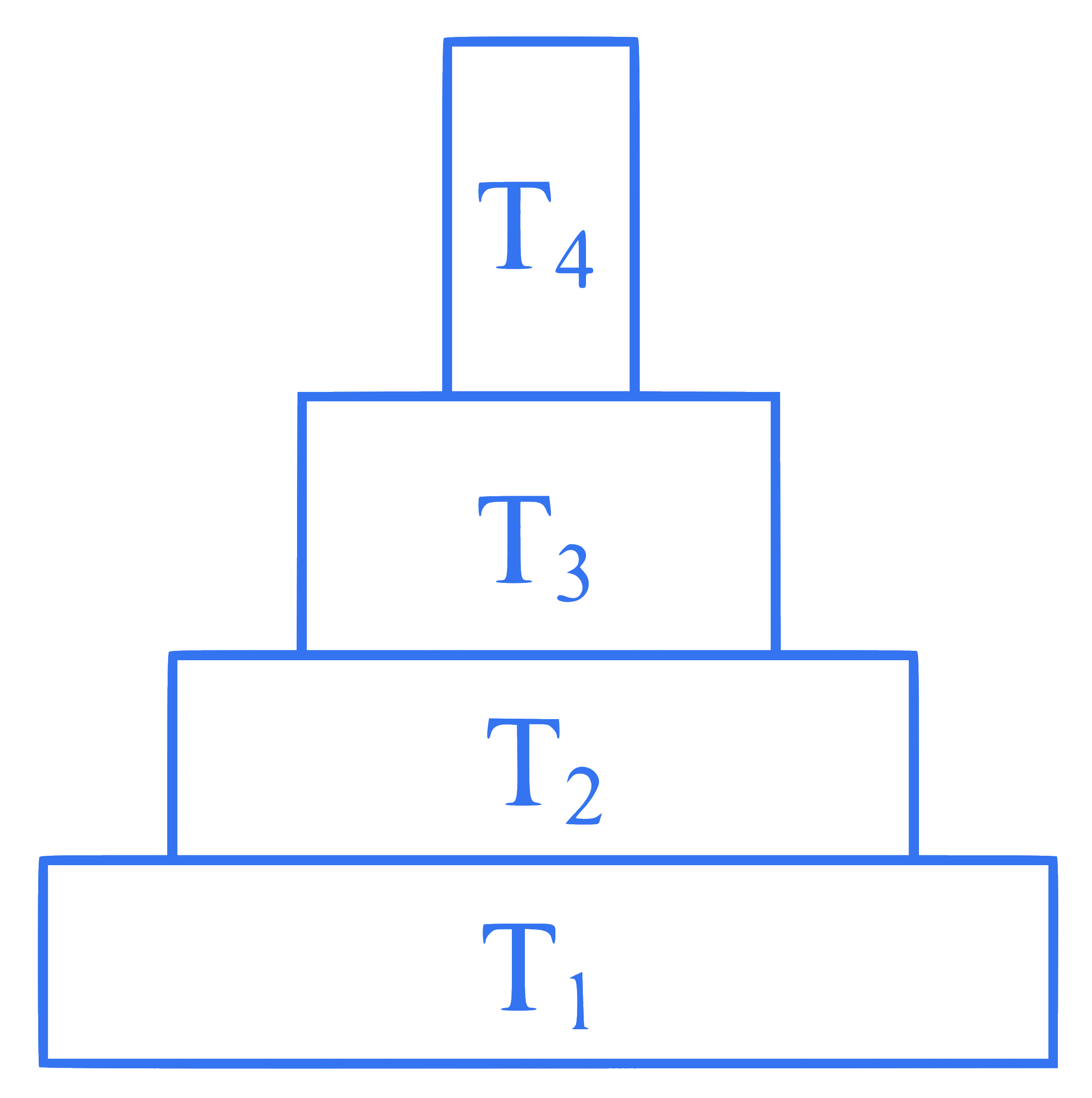 If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
 If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
Q4.
mcq single
+4 / 1
When a tall pea plant with round seeds was selfed, it produced the progeny of :
(a) Tall plants with round seeds and
(b) Tall plants with wrinkled seeds.
Identify the genotype of the parent plant.
Q5.
mcq single
+4 / 1
The mother has $$\mathrm{A}+$$ blood group the father has $$\mathrm{B}+$$ and the child is $$\mathrm{A}+$$. What can be the possibility of genotypes of all three, respectively?
A. $$I^A I^A|I B i| I^B i$$
B. $$I^A I^A\left|I^B i\right| I^A i$$
C. $$I^B i\left|I^A I^A\right| I^A I^B$$
D. $$\left|A \beta^A\right| I^B I^B \mid I^A i$$
E. $$I^A i\left|I^B i\right| I^A i$$
Choose the correct answer from the option given below:
Q6.
mcq single
+4 / 1
Given below are two statements:
Statement I: When many alleles of a single gene govern a character, it is called polygenic inheritance.
Statement II: In Polygenic inheritance, the effect of each allele is additive.
In the light of the above statements, choose the correct answer from the options given below.
Q7.
mcq single
+4 / 1
Aneuploidy is a chromosomal disorder where chromosome number is not the exact copy of its haploid set of chromosomes, due to :
A. Substitution
B. Addition
C. Deletion
D. Translocation
E. Inversion
Choose the most appropriate answer from the options given below :
Q8.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Type of Inheritance
List - II
Example
(A)
Incomplete dominance
(I)
Blood groups in human
(B)
Co-dominance
(II)
Flower colour in Antirrhinum
(C)
Pleiotropy
(III)
Skin colour in human
(D)
Polygenic inheritance
(IV)
Phenylketonuria
Choose the correct answer from the options given below:
Q9.
mcq single
+4 / 1
Which of the following is/are present in female cockroach?
A. Collateral gland
B. Mushroom gland
C. Spermatheca
D. Anal style
E. Phallic gland
Choose the most appropriate answer from the options given below:
Q10.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
(A)
Squamous Epithelium
(I)
Goblet cells of alimentary canal
(B)
Ciliated Epithelium
(II)
Inner lining of pancreatic ducts
(C)
Glandular Epithelium
(III)
Walls of blood vessels
(D)
Compound Epithelium
(IV)
Inner surface of Fallopian tubes
Choose the correct answer from the options given below :
Q11.
mcq single
+4 / 1
In which of the following connective tissues, the cells secrete fibres of collagen or elastin?
A. Cartilage
B. Bone
C. Adipose tissue
D. Blood
E. Areolar tissue
Choose the most appropriate answer from the options given below :
Q12.
mcq single
+4 / 1
Which of the following are required for the light reaction of Photosynthesis?
A. $$\mathrm{CO}_2$$
B. $$\mathrm{O}_2$$
C. $$\mathrm{H}_2 \mathrm{O}$$
D. Chlorophyll
E. Light
Choose the correct answer from the options given below:
Q13.
mcq single
+4 / 1
Observe the given figure. Identify the different stages labelled with alphabets by selecting the correct option.
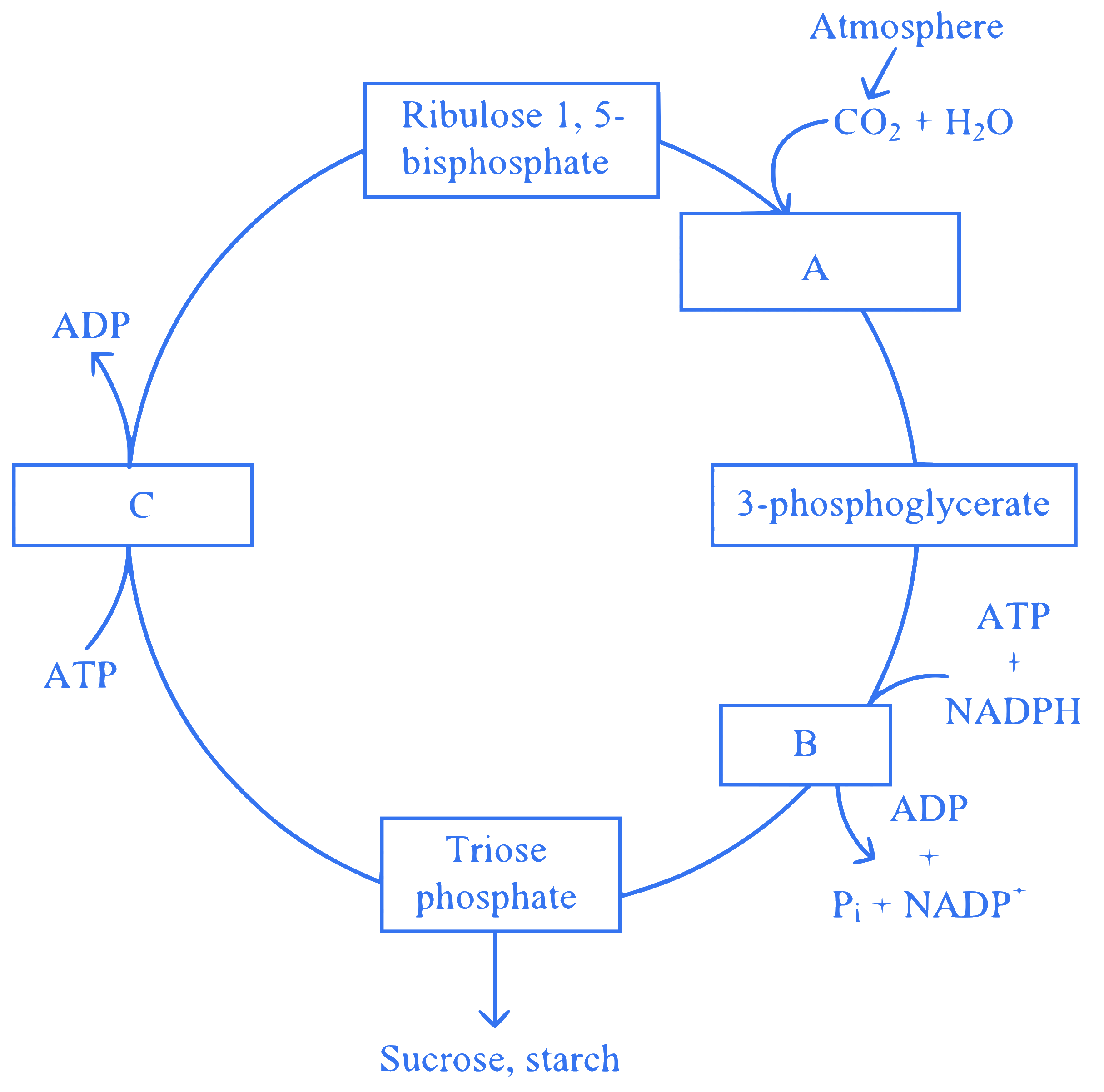

Q14.
mcq single
+4 / 1
Which one of the following products diffuses out of the chloroplast during photosynthesis?
Q15.
mcq single
+4 / 1
Which of the following pairs is an incorrect match?
Q16.
mcq single
+4 / 1
Open Circulatory system is present in :
Q17.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Chondrichthyes
(I)
Clarias
(B)
Cyclostomata
(II)
Carcharodon
(C)
Osteichthyes
(III)
Myxine
(D)
Amphibia
(IV)
Ichthyophis
Choose the correct answer from the options given below :
Q18.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Members of subphylum vertebrata possess notochord during the embryonic period. The notochord is replaced by a cartilaginous or bony vertebral column in the adult.
Reason R: Thus all chordates are vertebrates not all vertebrates are chordates.
In the light of the above statements choose the correct answer from the option given below.
Q19.
mcq single
+4 / 1
Which of the following is the correct match?
Q20.
mcq single
+4 / 1
Identify the incorrect pair :
Q21.
mcq single
+4 / 1
Which one of the following is not found in Gymnosperms?
Q22.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: During the transportation of gases, about 20-25 percent of CO$$_2$$ is carried by Haemoglobin as carbamino-haemoglobin.
Reason R: This binding is related to high pCO$$_2$$ and low pO$$_2$$ in tissues.
In the light of the above statements, choose the correct answer from the options given below.
Q23.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Residual Volume
(I)
Maximum volume of air that can be breathed in after forced expiration
(B)
Vital Capacity
(II)
Volume of air inspired or expired during normal respiration
(C)
Expiratory Capacity
(III)
Volume of air remaining in lungs after forcible expiration
(D)
Tidal Volume
(IV)
Total volume of air expired after normal inspiration
Choose the correct answer from the options given below:
Q24.
mcq single
+4 / 1
Following are the steps involved in action of toxin in Bt. Cotton
A. The inactive toxin converted into active form due to alkaline pH of gut of insect.
B. Bacillus thuringiensis produce crystals with toxic insecticidal proteins.
C. The alkaline pH solubilises the crystals.
D. The activated toxin binds to the surface of midgut cells, creates pores and causes death of the
insect.
E. The toxin proteins exist as inactive protoxins in bacteria.
Choose the correct sequence of steps from the options given below:
Q25.
mcq single
+4 / 1
Match List-I with List -II
List - I
List - II
(A)
Schwann cells
(I)
Neurotransmitter
(B)
Synaptic knob
(II)
Cerebral cortex
(C)
Bipolar neurons
(III)
Myelin sheath
(D)
Multipolar neurons
(IV)
Retina
Choose the correct answer from the options given below:
Q26.
mcq single
+4 / 1
Given below are two statements:
Statement I: In a dicotyledonous leaf, the adaxial epidermis generally bears more stomata than the abaxial
epidermis.
Statement II: In a dicotyledonous leaf, the adaxially placed palisade parenchyma is made up of elongated
cells, which are arranged vertically and parallel to each other.
In the light of the above statements, choose the correct answer from the options given below:
Q27.
mcq single
+4 / 1
Which of the following simple tissues are commonly found in the fruit walls of nuts and pulp of pear?
Q28.
mcq single
+4 / 1
Which of the following statements is correct about the type of junction and their role in our body?
Q29.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
F$$_1$$ Particles
(I)
Chromosomes
(B)
Histones
(II)
Cillia
(C)
Axoneme
(III)
Golgi apparatus
(D)
Cisternae
(IV)
Mitochondria
Choose the correct answer from the options given below:
Q30.
mcq single
+4 / 1
Mesosome in a cell is a :
Q31.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Histones
(I)
Loosely packed chromatin
(B)
Nucleosome
(II)
Densely packed chromatin
(C)
Euchromatin
(III)
Positively charged basic proteins
(D)
Heterochromatin
(IV)
DNA wrapped around histone octamer
Choose the correct answer from the options given below:
Q32.
mcq single
+4 / 1
Given below are two statements :
Statement I : Concentrically arranged cisternae of Golgi complex are arranged near the nucleus with distinct convex cis or maturing and concave trans or forming face.
Statement II : A number of proteins are modified in the cisternae of Golgi complex before they are released from cis face.
In the light of the above statements, choose the correct answer from the option given below.
Q33.
mcq single
+4 / 1
List - I
List - II
(A)
Fleming
(I)
Disc shaped sacs or cisternae near cell nucleus
(B)
Robert Brown
(II)
Chromatin
(C)
George Palade
(III)
Ribosomes
(D)
Camillo Golgi
(IV)
Nucleus
Choose the correct answer from the options given below:
Q34.
mcq single
+4 / 1
Given below are two statements :
Statement I : RNA interference takes place in all Eukaryotic organisms as method of cellular defense.
Statement II : RNAi involves the silencing of a specific mRNA due to a complementary single-stranded RNA molecule that binds and prevents translation of mRNA
In the light of the above statements, choose the correct answer from the options given below.
Q35.
mcq single
+4 / 1
Given below are two statements :
Statement I: In the lac operon, the z gene codes for beta-galactosidase which is primarily responsible for
the hydrolysis of lactose into galactose and glucose.
Statement II: In addition to lactose, glucose or galactose can also induce lac operon.
In the light of the above statements, choose the correct answer from the options given below :
Q36.
mcq single
+4 / 1
Given below are two statements regarding RNA polymerase in prokaryotes.
Statement I : In prokaryotes, RNA polymerase is capable of catalysing the process of elongation during
transcription.
Statement II : RNA polymerase associate transiently with ‘Rho’ factor to initiate transcription.
In the light of the above statements, choose the correct answer from the options given below :
Q37.
mcq single
+4 / 1
Given below are two statements:
Statement I: In eukaryotes there are three RNA polymerases in the nucleus in addition to the RNA
polymerase found in the organelles.
Statement II: All the three RNA polymerases in eukaryotic nucleus have different roles.
In the light of the above statements, choose the correct answer from the options given below:
Q38.
mcq single
+4 / 1
In a chromosome, there is a specific DNA sequence, responsible for initiating replication. It is called as:
Q39.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
(A)
B-Lymphocytes
(I)
Passive immunity
(B)
Interferons
(II)
Cell mediated immunity
(C)
T-Lymphocytes
(III)
Produce an army of proteins in response to pathogens
(D)
Colostrum
(IV)
Innate immunity
Choose the correct answer from the options given below :
Q40.
mcq single
+4 / 1
Math List-I with List-II:
List - I
List - II
(A)
Malignant tumors
(I)
Destroy tumors
(B)
MALT
(II)
AIDS
(C)
NACO
(III)
Metastasis
(D)
$$\alpha$$-Interferons
(IV)
Lymphoid tissue
Choose the correct answer from the options given below:
Q41.
mcq single
+4 / 1
Given below are two statements:
Statement I: Antibiotics are chemicals produced by microbes that kill other microbes.
Statement II: Antibodies are chemicals formed in body that eliminate microbes.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q42.
mcq single
+4 / 1
Which of the following helps in maintenance of the pressure gradient in sieve tubes?
Q43.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Predator
(I)
Ophrys
(B)
Mutualism
(II)
Pisaster
(C)
Parasitism
(III)
Female wasp and fig
(D)
Sexual deceit
(IV)
Plasmodium
Chose the correct answer from the options given below:
Q44.
mcq single
+4 / 1
When will the population density increase, under special conditions?
When the number of :
Q45.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Migratory flamingoes and resident fish in South American lakes
(I)
Interference competition
(B)
Abingdon tortoise became extinct after introduction of goats in their habitat
(II)
Competitive release
(C)
Chathamalus expands its distributional range in the absence of Balanus
(III)
Resource Partitioning
(D)
Five closely related species of Warblers feeding in different locations on same tree
(IV)
Interspecific competition
Choose the correct answer from the options given below:
Q46.
mcq single
+4 / 1
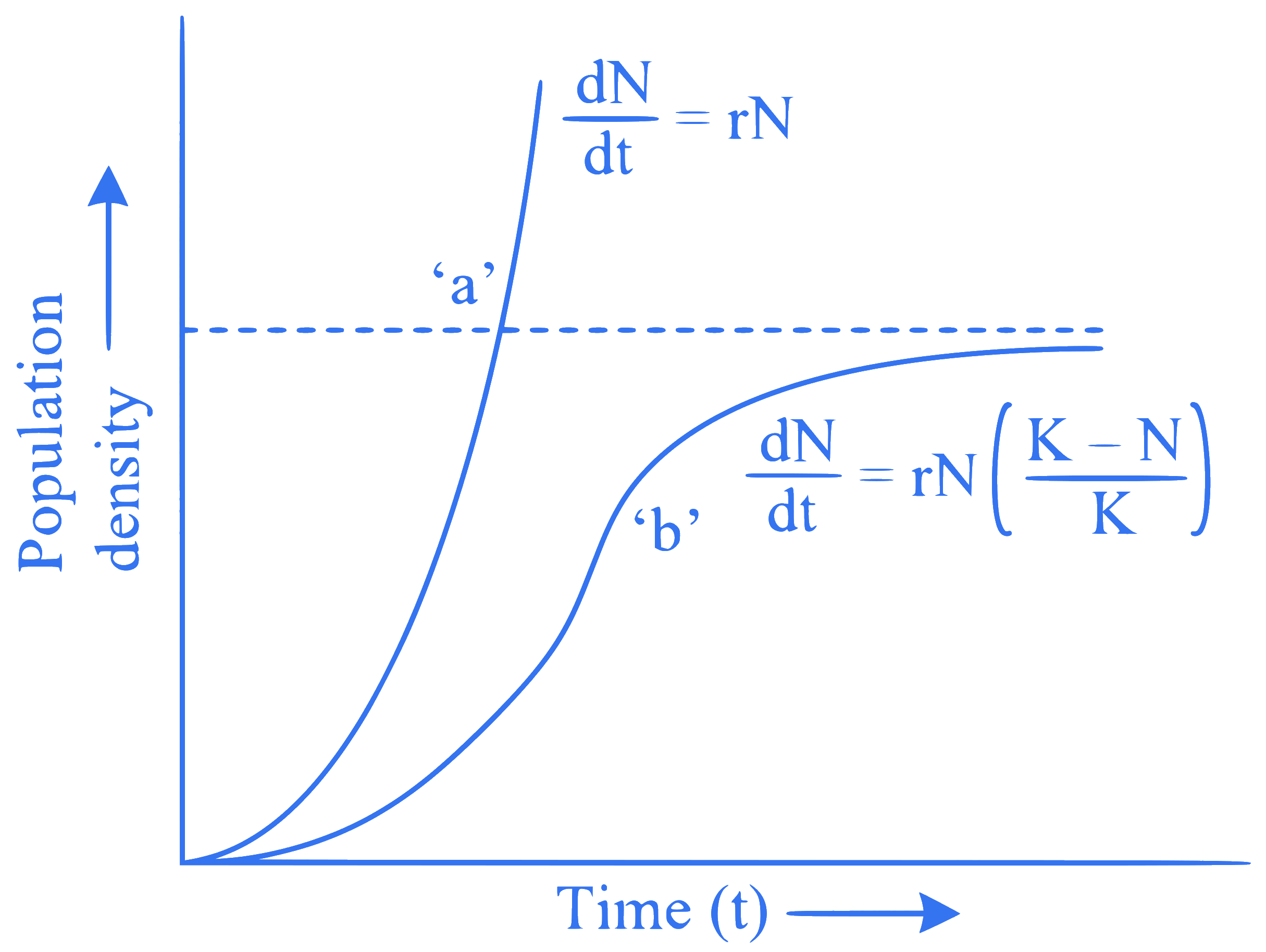
What do 'a' and 'b' represent in the following population growth curve?

Q47.
mcq single
+4 / 1
Diuresis is prevented by:
Q48.
mcq single
+4 / 1
'Lub' sound of Heart is caused by the _________.
Q49.
mcq single
+4 / 1
A person with blood group ARh– can receive the blood transfusion from which of the following types?
A. BRh$$^–$$
B. ABRh$$^–$$
C. ORh$$^–$$
D. ARh$$^–$$
E. ARh$$^+$$
Choose the correct answer from the options given below :
Q50.
mcq single
+4 / 1
Match List-I with List-II.
List - I
Event
List - II
Stage of Prophase-I (Meiosis-I)
(A)
Chiasmata formation
(I)
Pachytene
(B)
Crossing over
(II)
Diakinesis
(C)
Synaptonemal complex formation
(III)
Diplotene
(D)
Terminalisation of chiasmata
(IV)
Zygotene
Choose the correct answer from the options given below :
Q51.
mcq single
+4 / 1
Given below are two statements:
Statement I: Failure of segregation of chromatids during cell cycle resulting in the gain or loss of whole set of chromosome in an organism is known as aneuploidy.
Statement II: Failure of cytokinesis after anaphase stage of cell division results in the gain or loss of a chromosome is called polyploidy.
In the light of the above statements, choose the correct answer from the options given below:
Q52.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Metacentric chromosome
(I)
Chromosome has a terminal centromere
(B)
Sub-metacentric chromosome
(II)
Middle centromere forming two equal arms of chromosome
(C)
Acrocentric chromosome
(III)
Centromere is slightly away from the middle of chromosome resulting into two unequal arms
(D)
Telocentric chromosome
(IV)
Centromere is situated close to its end forming one extremely short and one very long arm
Choose the correct answer from the options given below:
Q53.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Cells are metabolically active and proliferate
(I)
G$$_2$$ phase
(B)
DNA replication takes place
(II)
G$$_1$$ phase
(C)
Proteins are synthesised
(III)
G$$_0$$ phase
(D)
Quiescent stage with metabolically active cells
(IV)
S phase
Choose the correct answer from the options given below:
Q54.
mcq single
+4 / 1
Recombination between homologous chromosomes is completed by the end of
Q55.
mcq single
+4 / 1
Arrange the following parts in human Mammary gland, traversing the route of milk ejection.
A. Mammary duct
B. Lactiferous duct
C. Mammary alveolus
D. Ampulla
E. Mammary tubule
Choose the correct answer from the options given below:
Q56.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: During menstrual cycle, the ovulation takes place approximately on 14th day.
Reason R: Rapid secretion of LH in the middle of menstrual cycle induces rupture of Graafian follicle and thereby the release of ovum.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q57.
mcq single
+4 / 1
Match List-I with List-II relating to human female external genitalia.
List - I
List - II
(A)
Mons pubis
(I)
A fleshy fold of tissue surrounding the vaginal opening
(B)
Clitoris
(II)
Fatty cushion of cells covered by skin and hair
(C)
Hymen
(III)
Tiny finger-like structure above labia minora
(D)
Labina majora
(IV)
A thin membrane-like structure covering vaginal opening
Choose the correct answer from the option given below :
Q58.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Parturition
(I)
Several antibodies for new-born babies
(B)
Placenta
(II)
Collection of ovum after ovulation
(C)
Colostrum
(III)
Foetal ejection reflex
(D)
Fimbriae
(IV)
Secretion of the hormone hCG
Choose the correct answer from the option given below:
Q59.
mcq single
+4 / 1
Identify the wrong statements :
A. Erythropoietin is produced by juxtaglomerular cells of the kidney
B. Leydig cells produce Androgens
C. Atrial Natriuretic factor, a peptide hormone is secreted by the seminiferous tubules of the testes
D. Cholecystokinin is produced by gastrointestinal tract
E. Gastrin acts on intestinal wall and helps in the production of pepsinogen
Choose the most appropriate answer from the options given below :
Q60.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Epinephrine
(I)
Hyperglycemia
(B)
Thyroxine
(II)
Smooth muscle contraction
(C)
Oxytocin
(III)
Basal metabolic rate
(D)
Glucagon
(IV)
Emergency hormone
Choose the correct answer from the options given below :
Q61.
mcq single
+4 / 1
Cryopreservation technique is used for :
Q62.
mcq single
+4 / 1
Which one of the following is not included under in-situ conservation?
Q63.
mcq single
+4 / 1
The regions with high level of species richness, high degree of endemism and a loss of $$70 \%$$ of the species and habitat are identified as:
Q64.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Biodiversity hotspot
(I)
Khasi and Jantia hills in Meghalaya
(B)
Sacred groves
(II)
World Summit on Sustainable Development 2002
(C)
Johannesburg, South Africa
(III)
Parthenium
(D)
Alien species invasion
(IV)
Western Ghats
Choose the correct answer from the options given below:
Q65.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Abscisic acid
(I)
Promotes female flowers in cucumber
(B)
Ethylene
(II)
Helps seeds to withstand desiccation
(C)
Gibberellin
(III)
Helps in nutrient mobilisation
(D)
Cytokinin
(IV)
Promotes bolting in beet, cabbage etc.
Choose the correct answer from the options given below:
Q66.
mcq single
+4 / 1
Given below are some statements about plant growth regulators.
A. All GAs are acidic in nature.
B. Auxins are antagonists to GAs.
C. Zeatin was isolated from coconut milk.
D. Ethylene induces flowering in Mango.
E. Abscisic acid induces parthenocarpy.
Choose the correct set of statements from the options given below :
Q67.
mcq single
+4 / 1
F. Skoog observed that callus proliferated from the internodal segments of tobacco stem when auxin was
supplied with one of the following except :
Q68.
mcq single
+4 / 1
Which of the following are correct about cellular respiration?
A. Cellular respiration is the breaking of C-C bonds of complex organic molecules by oxidation.
B. The entire cellular respiration takes place in Mitochondria.
C. Fermentation takes place under anaerobic condition in germinating seeds.
D. The fate of pyruvate formed during glycolysis depends on the type of organism also.
E. Water is formed during respiration as a result of O$$_2$$ accepting electrons and getting reduced.
Choose the correct answer from the options given below :
Q69.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
ETS Complex I
(I)
NADH Dehydrogenase
(B)
ETS Complex II
(II)
Cytochrome bC$$_1$$
(C)
ETS Complex III
(III)
Cytochrome C oxidase
(D)
ETS Complex IV
(IV)
Succinate Dehydrogenase
Choose the correct answer from the options given below :
Q70.
mcq single
+4 / 1
Select the correct statements regarding mechanism of muscle contraction.
A. It is initiated by a signal sent by CNS via sensory neuron.
B. Neurotransmitter generates action potential in the sarcolemma.
C. Increased Ca$$^{++}$$ level leads to the binding of calcium with troponin on action filaments.
D. Masking of active site for actin is activated.
E. Utilising the energy from ATP hydrolysis to form cross bridge.
Choose the most appropriate answer from the options given below:
Q71.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Location of Joint
List - II
Type of Joint
(A)
Joint between humerus and pectoral girdle
(I)
Gliding joint
(B)
Knee joint
(II)
Ball and Socket joint
(C)
Joint between atlas and axis
(III)
Hinge joint
(D)
Joint between carpals
(IV)
Pivot joint
Chose the correct answer from the options given below:
Q72.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Organisms
List - II
Mode of Nutrition
(A)
Euglenoid
(I)
Parasitic
(B)
Dinoflagellate
(II)
Saprophytic
(C)
Slime mould
(III)
Photosynthetic
(D)
Plasmodium
(IV)
Switching between photosynthetic and heterotrophic mode
Choose the correct answer from the options given below:
Q73.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Gene pool
(I)
Stable within a generation
(B)
Genetic drift
(II)
Change in gene frequency by chance
(C)
Gene flow
(III)
Transfer of genes into or out of population
(D)
Gene frequency
(IV)
Total number of genes and their alleles
Choose the correct answer from the options given below :
Q74.
mcq single
+4 / 1
Which evolutionary phenomenon is depicted by the sketch given in figure?


Q75.
mcq single
+4 / 1
Match List-I with List-II with respect to convergent evolution:
List - I
List - II
(A)
Lemur
(I)
Flying phalanger
(B)
Bobcat
(II)
Numbat
(C)
Anteater
(III)
Spotted cuscus
(D)
Flying squirrels
(IV)
Tasmanian tiger cat
Choose the correct answer from the options given below:
Q76.
mcq single
+4 / 1
What is the correct order (old to recent) of periods in Paleozoic era?
Q77.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
(A)
Living Fossil
(I)
Elongated canine teeth
(B)
Connecting Link
(II)
Vermiform appendix
(C)
Vestigial Organ
(III)
Echidna
(D)
Atavism
(IV)
Latimeria
Choose the correct answer from the options given below:
Q78.
mcq single
+4 / 1
Given below are two statements:
Statements I: Concentrated urine is formed due to counter current mechanism in nephron.
Statement II: Counter current mechanism helps to maintain osmotic gradient in the medullary interstitium.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q79.
mcq single
+4 / 1
Following is the list of STDs. Select the diseases which are not completely curable.
A. Genital warts
B. Genital herpes
C. Syphilis
D. Hepatitis-B
E. Trichomoniasis
Choose the correct answer from the options given below:
Q80.
mcq single
+4 / 1
Match List-I with List-II relating to examples of various kind of IUDs and barrier:
List - I
List - II
(A)
Copper releasing IUD
(I)
Vaults
(B)
Non-medicated IUD
(II)
Multiload 375
(C)
Contraceptive barrier
(III)
LNG-20
(D)
Hormone releasing IUD
(IV)
Lippes loop
Choose the correct answer from the options given below:
Q81.
mcq single
+4 / 1
Pollen grains remain preserved as fossils due to the presence of :
Q82.
mcq single
+4 / 1
The part marked as ‘x’ in the given figure is

Q83.
mcq single
+4 / 1
Which part of the ovule stores reserve food materials?
Q84.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
China rose
(I)
Free central
(B)
Mustard
(II)
Basal
(C)
Primrose
(III)
Axile
(D)
Marigold
(IV)
Parietal
Choose the correct answer from the options given below :
Q85.
mcq single
+4 / 1
Which of the following examples show monocarpellary, unilocular ovary with many ovules?
A. Sesbania
B. Brinjal
C. Indigofera
D. Tobacco
E. Asparagus
Choose the correct answer from the options given below :
Q86.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Vexillary aestivation
(I)
Brinjal
(B)
Epipetalous stamens
(II)
Peach
(C)
Epiphyllous stamens
(III)
Pea
(D)
Perigynous flower
(IV)
Lily
Choose the correct answer from the options given below :
Q87.
mcq single
+4 / 1
Which of the following is a nucleotide?
Q88.
mcq single
+4 / 1
Ligases is a class of enzymes responsible for catalysing the linking together of two compounds.
Which of the following bonds is not catalysed by it?
Q89.
mcq single
+4 / 1
Enzymes that catalyse the removal of groups from substrates by mechanisms other than hydrolysis leaving double bonds, are known as :
Q90.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Primary structure of protein
(I)
Human haemoglobin
(B)
Secondary structure of protein
(II)
Disulphide bonds
(C)
Tertiary structure of protein
(III)
Polypeptide chain
(D)
Quanternary structure of protein
(IV)
Alpha helix and $$\beta$$ sheet
Choose the correct answer from the options given below :
Q91.
mcq single
+4 / 1
Which of the following graphs depicts the effect of substrate concentration on velocity of enzyme catalysed reaction?
Q92.
mcq single
+4 / 1
Which of the following are not fatty acids?
A. Glutamic acid
B. Arachidonic acid
C. Palmitic acid
D. Lecithin
E. Aspartic acid
Choose the correct answer from the options given below :
Q93.
mcq single
+4 / 1
Recombinant DNA molecule can be created normally by cutting the vector DNA and source DNA
respectively with:
Q94.
mcq single
+4 / 1
Which of the following are correct about EcoRI?
A. Cut the DNA with blunt end
B. Cut the DNA with sticky end
C. Recognise a specific palindromic sequence
D. Cut the DNA between the base $$\mathrm{G}$$ and $$\mathrm{A}$$ when encounters the DNA sequence 'GAATTC'
E. Exonuclease
Choose the correct answer from the options given below:
Q95.
mcq single
+4 / 1
The Bt toxin in genetically engineered Bt cotton kills the pest by:
Q96.
mcq single
+4 / 1
Match List-I with List - II :
List - I
List - II
(A)
Genetically engineered Human Insulin
(I)
Gene therapy
(B)
GM Cotton
(II)
E. coli
(C)
ADA Deficiency
(III)
Antigen-antibody interaction
(D)
ELISA
(IV)
Bacillus thuringiensis
Choose the correct answer from the options given below:
Q97.
mcq single
+4 / 1
Identify the incorrect statement related to gel electrophoresis.
Q98.
mcq single
+4 / 1
Given below are two statements:
Statement I : The Indian Government has set up GEAC, which will make decisions regarding the validity of GM research.
Statement II : Biopiracy is the term used to refer to the use of bio-resources by native people.
In the light of the above statements, choose the correct answer from the options given below :
Q99.
mcq single
+4 / 1
Select the restriction endonuclease enzymes whose restriction sites are present for the tetracycline resistance (tet$$^{\mathrm{R}}$$) gene in the pBR322 cloning vector.
Q100.
mcq single
+4 / 1
Following are the steps involved in the process of PCR.
A. Annealing
B. Amplification (~1 billion times)
C. Denaturation
D. Treatment with Taq polymerase and deoxynucleotides
E. Extension
Choose the correct sequence of steps of PCR from the options given below :
Q101.
mcq single
+4 / 1
Match List-I with List-II relating to microbes and their products:
List - I
(Microbes)
List - II
(Products)
(A)
Streptococcus
(I)
Citric acid
(B)
Trichoderma polysporum
(II)
Clot buster
(C)
Monascus purpureus
(III)
Cyclosporin A
(D)
Aspergillus niger
(IV)
Statins
Choose the correct answer from the options given below:
Q102.
mcq single
+4 / 1
Which one of the following is not a limitation of ecological pyramids?
Q103.
mcq single
+4 / 1
Consider the pyramid of energy of an ecosystem given below:
 If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
 If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
If T$$_4$$ is equivalent to $$1000 \mathrm{~J}$$, what is the value at $$\mathrm{T}_1$$ ?
Q104.
mcq single
+4 / 1
When a tall pea plant with round seeds was selfed, it produced the progeny of :
(a) Tall plants with round seeds and
(b) Tall plants with wrinkled seeds.
Identify the genotype of the parent plant.
Q105.
mcq single
+4 / 1
The mother has $$\mathrm{A}+$$ blood group the father has $$\mathrm{B}+$$ and the child is $$\mathrm{A}+$$. What can be the possibility of genotypes of all three, respectively?
A. $$I^A I^A|I B i| I^B i$$
B. $$I^A I^A\left|I^B i\right| I^A i$$
C. $$I^B i\left|I^A I^A\right| I^A I^B$$
D. $$\left|A \beta^A\right| I^B I^B \mid I^A i$$
E. $$I^A i\left|I^B i\right| I^A i$$
Choose the correct answer from the option given below:
Q106.
mcq single
+4 / 1
Given below are two statements:
Statement I: When many alleles of a single gene govern a character, it is called polygenic inheritance.
Statement II: In Polygenic inheritance, the effect of each allele is additive.
In the light of the above statements, choose the correct answer from the options given below.
Q107.
mcq single
+4 / 1
Aneuploidy is a chromosomal disorder where chromosome number is not the exact copy of its haploid set of chromosomes, due to :
A. Substitution
B. Addition
C. Deletion
D. Translocation
E. Inversion
Choose the most appropriate answer from the options given below :
Q108.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Type of Inheritance
List - II
Example
(A)
Incomplete dominance
(I)
Blood groups in human
(B)
Co-dominance
(II)
Flower colour in Antirrhinum
(C)
Pleiotropy
(III)
Skin colour in human
(D)
Polygenic inheritance
(IV)
Phenylketonuria
Choose the correct answer from the options given below:
Q109.
mcq single
+4 / 1
Which of the following is/are present in female cockroach?
A. Collateral gland
B. Mushroom gland
C. Spermatheca
D. Anal style
E. Phallic gland
Choose the most appropriate answer from the options given below:
Q110.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
(A)
Squamous Epithelium
(I)
Goblet cells of alimentary canal
(B)
Ciliated Epithelium
(II)
Inner lining of pancreatic ducts
(C)
Glandular Epithelium
(III)
Walls of blood vessels
(D)
Compound Epithelium
(IV)
Inner surface of Fallopian tubes
Choose the correct answer from the options given below :
Q111.
mcq single
+4 / 1
In which of the following connective tissues, the cells secrete fibres of collagen or elastin?
A. Cartilage
B. Bone
C. Adipose tissue
D. Blood
E. Areolar tissue
Choose the most appropriate answer from the options given below :
Q112.
mcq single
+4 / 1
Which of the following are required for the light reaction of Photosynthesis?
A. $$\mathrm{CO}_2$$
B. $$\mathrm{O}_2$$
C. $$\mathrm{H}_2 \mathrm{O}$$
D. Chlorophyll
E. Light
Choose the correct answer from the options given below:
Q113.
mcq single
+4 / 1
Observe the given figure. Identify the different stages labelled with alphabets by selecting the correct option.


Q114.
mcq single
+4 / 1
Which one of the following products diffuses out of the chloroplast during photosynthesis?
Q115.
mcq single
+4 / 1
Which of the following pairs is an incorrect match?
Q116.
mcq single
+4 / 1
Open Circulatory system is present in :
Q117.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Chondrichthyes
(I)
Clarias
(B)
Cyclostomata
(II)
Carcharodon
(C)
Osteichthyes
(III)
Myxine
(D)
Amphibia
(IV)
Ichthyophis
Choose the correct answer from the options given below :
Q118.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Members of subphylum vertebrata possess notochord during the embryonic period. The notochord is replaced by a cartilaginous or bony vertebral column in the adult.
Reason R: Thus all chordates are vertebrates not all vertebrates are chordates.
In the light of the above statements choose the correct answer from the option given below.
Q119.
mcq single
+4 / 1
Which of the following is the correct match?
Q120.
mcq single
+4 / 1
Identify the incorrect pair :
Q121.
mcq single
+4 / 1
Which one of the following is not found in Gymnosperms?
Q122.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: During the transportation of gases, about 20-25 percent of CO$$_2$$ is carried by Haemoglobin as carbamino-haemoglobin.
Reason R: This binding is related to high pCO$$_2$$ and low pO$$_2$$ in tissues.
In the light of the above statements, choose the correct answer from the options given below.
Q123.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Residual Volume
(I)
Maximum volume of air that can be breathed in after forced expiration
(B)
Vital Capacity
(II)
Volume of air inspired or expired during normal respiration
(C)
Expiratory Capacity
(III)
Volume of air remaining in lungs after forcible expiration
(D)
Tidal Volume
(IV)
Total volume of air expired after normal inspiration
Choose the correct answer from the options given below:
Q124.
mcq single
+4 / 1
Following are the steps involved in action of toxin in Bt. Cotton
A. The inactive toxin converted into active form due to alkaline pH of gut of insect.
B. Bacillus thuringiensis produce crystals with toxic insecticidal proteins.
C. The alkaline pH solubilises the crystals.
D. The activated toxin binds to the surface of midgut cells, creates pores and causes death of the
insect.
E. The toxin proteins exist as inactive protoxins in bacteria.
Choose the correct sequence of steps from the options given below:
Q125.
mcq single
+4 / 1
Match List-I with List -II
List - I
List - II
(A)
Schwann cells
(I)
Neurotransmitter
(B)
Synaptic knob
(II)
Cerebral cortex
(C)
Bipolar neurons
(III)
Myelin sheath
(D)
Multipolar neurons
(IV)
Retina
Choose the correct answer from the options given below:
Q126.
mcq single
+4 / 1
Given below are two statements:
Statement I: In a dicotyledonous leaf, the adaxial epidermis generally bears more stomata than the abaxial
epidermis.
Statement II: In a dicotyledonous leaf, the adaxially placed palisade parenchyma is made up of elongated
cells, which are arranged vertically and parallel to each other.
In the light of the above statements, choose the correct answer from the options given below:
Q127.
mcq single
+4 / 1
Which of the following simple tissues are commonly found in the fruit walls of nuts and pulp of pear?
Q128.
mcq single
+4 / 1
Which of the following statements is correct about the type of junction and their role in our body?
Q129.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
F$$_1$$ Particles
(I)
Chromosomes
(B)
Histones
(II)
Cillia
(C)
Axoneme
(III)
Golgi apparatus
(D)
Cisternae
(IV)
Mitochondria
Choose the correct answer from the options given below:
Q130.
mcq single
+4 / 1
Mesosome in a cell is a :
Q131.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Histones
(I)
Loosely packed chromatin
(B)
Nucleosome
(II)
Densely packed chromatin
(C)
Euchromatin
(III)
Positively charged basic proteins
(D)
Heterochromatin
(IV)
DNA wrapped around histone octamer
Choose the correct answer from the options given below:
Q132.
mcq single
+4 / 1
Given below are two statements :
Statement I : Concentrically arranged cisternae of Golgi complex are arranged near the nucleus with distinct convex cis or maturing and concave trans or forming face.
Statement II : A number of proteins are modified in the cisternae of Golgi complex before they are released from cis face.
In the light of the above statements, choose the correct answer from the option given below.
Q133.
mcq single
+4 / 1
List - I
List - II
(A)
Fleming
(I)
Disc shaped sacs or cisternae near cell nucleus
(B)
Robert Brown
(II)
Chromatin
(C)
George Palade
(III)
Ribosomes
(D)
Camillo Golgi
(IV)
Nucleus
Choose the correct answer from the options given below:
Q134.
mcq single
+4 / 1
Given below are two statements :
Statement I : RNA interference takes place in all Eukaryotic organisms as method of cellular defense.
Statement II : RNAi involves the silencing of a specific mRNA due to a complementary single-stranded RNA molecule that binds and prevents translation of mRNA
In the light of the above statements, choose the correct answer from the options given below.
Q135.
mcq single
+4 / 1
Given below are two statements :
Statement I: In the lac operon, the z gene codes for beta-galactosidase which is primarily responsible for
the hydrolysis of lactose into galactose and glucose.
Statement II: In addition to lactose, glucose or galactose can also induce lac operon.
In the light of the above statements, choose the correct answer from the options given below :
Q136.
mcq single
+4 / 1
Given below are two statements regarding RNA polymerase in prokaryotes.
Statement I : In prokaryotes, RNA polymerase is capable of catalysing the process of elongation during
transcription.
Statement II : RNA polymerase associate transiently with ‘Rho’ factor to initiate transcription.
In the light of the above statements, choose the correct answer from the options given below :
Q137.
mcq single
+4 / 1
Given below are two statements:
Statement I: In eukaryotes there are three RNA polymerases in the nucleus in addition to the RNA
polymerase found in the organelles.
Statement II: All the three RNA polymerases in eukaryotic nucleus have different roles.
In the light of the above statements, choose the correct answer from the options given below:
Q138.
mcq single
+4 / 1
In a chromosome, there is a specific DNA sequence, responsible for initiating replication. It is called as:
Q139.
mcq single
+4 / 1
Match List I with List II :
List - I
List - II
(A)
B-Lymphocytes
(I)
Passive immunity
(B)
Interferons
(II)
Cell mediated immunity
(C)
T-Lymphocytes
(III)
Produce an army of proteins in response to pathogens
(D)
Colostrum
(IV)
Innate immunity
Choose the correct answer from the options given below :
Q140.
mcq single
+4 / 1
Math List-I with List-II:
List - I
List - II
(A)
Malignant tumors
(I)
Destroy tumors
(B)
MALT
(II)
AIDS
(C)
NACO
(III)
Metastasis
(D)
$$\alpha$$-Interferons
(IV)
Lymphoid tissue
Choose the correct answer from the options given below:
Q141.
mcq single
+4 / 1
Given below are two statements:
Statement I: Antibiotics are chemicals produced by microbes that kill other microbes.
Statement II: Antibodies are chemicals formed in body that eliminate microbes.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q142.
mcq single
+4 / 1
Which of the following helps in maintenance of the pressure gradient in sieve tubes?
Q143.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Predator
(I)
Ophrys
(B)
Mutualism
(II)
Pisaster
(C)
Parasitism
(III)
Female wasp and fig
(D)
Sexual deceit
(IV)
Plasmodium
Chose the correct answer from the options given below:
Q144.
mcq single
+4 / 1
When will the population density increase, under special conditions?
When the number of :
Q145.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Migratory flamingoes and resident fish in South American lakes
(I)
Interference competition
(B)
Abingdon tortoise became extinct after introduction of goats in their habitat
(II)
Competitive release
(C)
Chathamalus expands its distributional range in the absence of Balanus
(III)
Resource Partitioning
(D)
Five closely related species of Warblers feeding in different locations on same tree
(IV)
Interspecific competition
Choose the correct answer from the options given below:
Q146.
mcq single
+4 / 1
What do 'a' and 'b' represent in the following population growth curve?


Q147.
mcq single
+4 / 1
Diuresis is prevented by:
Q148.
mcq single
+4 / 1
'Lub' sound of Heart is caused by the _________.
Q149.
mcq single
+4 / 1
A person with blood group ARh– can receive the blood transfusion from which of the following types?
A. BRh$$^–$$
B. ABRh$$^–$$
C. ORh$$^–$$
D. ARh$$^–$$
E. ARh$$^+$$
Choose the correct answer from the options given below :
Q150.
mcq single
+4 / 1
Match List-I with List-II.
List - I
Event
List - II
Stage of Prophase-I (Meiosis-I)
(A)
Chiasmata formation
(I)
Pachytene
(B)
Crossing over
(II)
Diakinesis
(C)
Synaptonemal complex formation
(III)
Diplotene
(D)
Terminalisation of chiasmata
(IV)
Zygotene
Choose the correct answer from the options given below :
Q151.
mcq single
+4 / 1
Given below are two statements:
Statement I: Failure of segregation of chromatids during cell cycle resulting in the gain or loss of whole set of chromosome in an organism is known as aneuploidy.
Statement II: Failure of cytokinesis after anaphase stage of cell division results in the gain or loss of a chromosome is called polyploidy.
In the light of the above statements, choose the correct answer from the options given below:
Q152.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Metacentric chromosome
(I)
Chromosome has a terminal centromere
(B)
Sub-metacentric chromosome
(II)
Middle centromere forming two equal arms of chromosome
(C)
Acrocentric chromosome
(III)
Centromere is slightly away from the middle of chromosome resulting into two unequal arms
(D)
Telocentric chromosome
(IV)
Centromere is situated close to its end forming one extremely short and one very long arm
Choose the correct answer from the options given below:
Q153.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Cells are metabolically active and proliferate
(I)
G$$_2$$ phase
(B)
DNA replication takes place
(II)
G$$_1$$ phase
(C)
Proteins are synthesised
(III)
G$$_0$$ phase
(D)
Quiescent stage with metabolically active cells
(IV)
S phase
Choose the correct answer from the options given below:
Q154.
mcq single
+4 / 1
Recombination between homologous chromosomes is completed by the end of
Q155.
mcq single
+4 / 1
Arrange the following parts in human Mammary gland, traversing the route of milk ejection.
A. Mammary duct
B. Lactiferous duct
C. Mammary alveolus
D. Ampulla
E. Mammary tubule
Choose the correct answer from the options given below:
Q156.
mcq single
+4 / 1
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: During menstrual cycle, the ovulation takes place approximately on 14th day.
Reason R: Rapid secretion of LH in the middle of menstrual cycle induces rupture of Graafian follicle and thereby the release of ovum.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q157.
mcq single
+4 / 1
Match List-I with List-II relating to human female external genitalia.
List - I
List - II
(A)
Mons pubis
(I)
A fleshy fold of tissue surrounding the vaginal opening
(B)
Clitoris
(II)
Fatty cushion of cells covered by skin and hair
(C)
Hymen
(III)
Tiny finger-like structure above labia minora
(D)
Labina majora
(IV)
A thin membrane-like structure covering vaginal opening
Choose the correct answer from the option given below :
Q158.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Parturition
(I)
Several antibodies for new-born babies
(B)
Placenta
(II)
Collection of ovum after ovulation
(C)
Colostrum
(III)
Foetal ejection reflex
(D)
Fimbriae
(IV)
Secretion of the hormone hCG
Choose the correct answer from the option given below:
Q159.
mcq single
+4 / 1
Identify the wrong statements :
A. Erythropoietin is produced by juxtaglomerular cells of the kidney
B. Leydig cells produce Androgens
C. Atrial Natriuretic factor, a peptide hormone is secreted by the seminiferous tubules of the testes
D. Cholecystokinin is produced by gastrointestinal tract
E. Gastrin acts on intestinal wall and helps in the production of pepsinogen
Choose the most appropriate answer from the options given below :
Q160.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Epinephrine
(I)
Hyperglycemia
(B)
Thyroxine
(II)
Smooth muscle contraction
(C)
Oxytocin
(III)
Basal metabolic rate
(D)
Glucagon
(IV)
Emergency hormone
Choose the correct answer from the options given below :
Q161.
mcq single
+4 / 1
Cryopreservation technique is used for :
Q162.
mcq single
+4 / 1
Which one of the following is not included under in-situ conservation?
Q163.
mcq single
+4 / 1
The regions with high level of species richness, high degree of endemism and a loss of $$70 \%$$ of the species and habitat are identified as:
Q164.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
Biodiversity hotspot
(I)
Khasi and Jantia hills in Meghalaya
(B)
Sacred groves
(II)
World Summit on Sustainable Development 2002
(C)
Johannesburg, South Africa
(III)
Parthenium
(D)
Alien species invasion
(IV)
Western Ghats
Choose the correct answer from the options given below:
Q165.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Abscisic acid
(I)
Promotes female flowers in cucumber
(B)
Ethylene
(II)
Helps seeds to withstand desiccation
(C)
Gibberellin
(III)
Helps in nutrient mobilisation
(D)
Cytokinin
(IV)
Promotes bolting in beet, cabbage etc.
Choose the correct answer from the options given below:
Q166.
mcq single
+4 / 1
Given below are some statements about plant growth regulators.
A. All GAs are acidic in nature.
B. Auxins are antagonists to GAs.
C. Zeatin was isolated from coconut milk.
D. Ethylene induces flowering in Mango.
E. Abscisic acid induces parthenocarpy.
Choose the correct set of statements from the options given below :
Q167.
mcq single
+4 / 1
F. Skoog observed that callus proliferated from the internodal segments of tobacco stem when auxin was
supplied with one of the following except :
Q168.
mcq single
+4 / 1
Which of the following are correct about cellular respiration?
A. Cellular respiration is the breaking of C-C bonds of complex organic molecules by oxidation.
B. The entire cellular respiration takes place in Mitochondria.
C. Fermentation takes place under anaerobic condition in germinating seeds.
D. The fate of pyruvate formed during glycolysis depends on the type of organism also.
E. Water is formed during respiration as a result of O$$_2$$ accepting electrons and getting reduced.
Choose the correct answer from the options given below :
Q169.
mcq single
+4 / 1
Match List-I with List-II:
List - I
List - II
(A)
ETS Complex I
(I)
NADH Dehydrogenase
(B)
ETS Complex II
(II)
Cytochrome bC$$_1$$
(C)
ETS Complex III
(III)
Cytochrome C oxidase
(D)
ETS Complex IV
(IV)
Succinate Dehydrogenase
Choose the correct answer from the options given below :
Q170.
mcq single
+4 / 1
Select the correct statements regarding mechanism of muscle contraction.
A. It is initiated by a signal sent by CNS via sensory neuron.
B. Neurotransmitter generates action potential in the sarcolemma.
C. Increased Ca$$^{++}$$ level leads to the binding of calcium with troponin on action filaments.
D. Masking of active site for actin is activated.
E. Utilising the energy from ATP hydrolysis to form cross bridge.
Choose the most appropriate answer from the options given below:
Q171.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Location of Joint
List - II
Type of Joint
(A)
Joint between humerus and pectoral girdle
(I)
Gliding joint
(B)
Knee joint
(II)
Ball and Socket joint
(C)
Joint between atlas and axis
(III)
Hinge joint
(D)
Joint between carpals
(IV)
Pivot joint
Chose the correct answer from the options given below:
Q172.
mcq single
+4 / 1
Match List-I with List-II:
List - I
Organisms
List - II
Mode of Nutrition
(A)
Euglenoid
(I)
Parasitic
(B)
Dinoflagellate
(II)
Saprophytic
(C)
Slime mould
(III)
Photosynthetic
(D)
Plasmodium
(IV)
Switching between photosynthetic and heterotrophic mode
Choose the correct answer from the options given below:
Q173.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Gene pool
(I)
Stable within a generation
(B)
Genetic drift
(II)
Change in gene frequency by chance
(C)
Gene flow
(III)
Transfer of genes into or out of population
(D)
Gene frequency
(IV)
Total number of genes and their alleles
Choose the correct answer from the options given below :
Q174.
mcq single
+4 / 1
Which evolutionary phenomenon is depicted by the sketch given in figure?


Q175.
mcq single
+4 / 1
Match List-I with List-II with respect to convergent evolution:
List - I
List - II
(A)
Lemur
(I)
Flying phalanger
(B)
Bobcat
(II)
Numbat
(C)
Anteater
(III)
Spotted cuscus
(D)
Flying squirrels
(IV)
Tasmanian tiger cat
Choose the correct answer from the options given below:
Q176.
mcq single
+4 / 1
What is the correct order (old to recent) of periods in Paleozoic era?
Q177.
mcq single
+4 / 1
Match List I with List II:
List - I
List - II
(A)
Living Fossil
(I)
Elongated canine teeth
(B)
Connecting Link
(II)
Vermiform appendix
(C)
Vestigial Organ
(III)
Echidna
(D)
Atavism
(IV)
Latimeria
Choose the correct answer from the options given below:
Q178.
mcq single
+4 / 1
Given below are two statements:
Statements I: Concentrated urine is formed due to counter current mechanism in nephron.
Statement II: Counter current mechanism helps to maintain osmotic gradient in the medullary interstitium.
In the light of the above statements, choose the most appropriate answer from the options given below.
Q179.
mcq single
+4 / 1
Following is the list of STDs. Select the diseases which are not completely curable.
A. Genital warts
B. Genital herpes
C. Syphilis
D. Hepatitis-B
E. Trichomoniasis
Choose the correct answer from the options given below:
Q180.
mcq single
+4 / 1
Match List-I with List-II relating to examples of various kind of IUDs and barrier:
List - I
List - II
(A)
Copper releasing IUD
(I)
Vaults
(B)
Non-medicated IUD
(II)
Multiload 375
(C)
Contraceptive barrier
(III)
LNG-20
(D)
Hormone releasing IUD
(IV)
Lippes loop
Choose the correct answer from the options given below:
Q181.
mcq single
+4 / 1
Pollen grains remain preserved as fossils due to the presence of :
Q182.
mcq single
+4 / 1
The part marked as ‘x’ in the given figure is


Q183.
mcq single
+4 / 1
Which part of the ovule stores reserve food materials?
Q184.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
China rose
(I)
Free central
(B)
Mustard
(II)
Basal
(C)
Primrose
(III)
Axile
(D)
Marigold
(IV)
Parietal
Choose the correct answer from the options given below :
Q185.
mcq single
+4 / 1
Which of the following examples show monocarpellary, unilocular ovary with many ovules?
A. Sesbania
B. Brinjal
C. Indigofera
D. Tobacco
E. Asparagus
Choose the correct answer from the options given below :
Q186.
mcq single
+4 / 1
Match List-I with List-II :
List - I
List - II
(A)
Vexillary aestivation
(I)
Brinjal
(B)
Epipetalous stamens
(II)
Peach
(C)
Epiphyllous stamens
(III)
Pea
(D)
Perigynous flower
(IV)
Lily
Choose the correct answer from the options given below :
Q187.
mcq single
+4 / 1
Which of the following is a nucleotide?
Q188.
mcq single
+4 / 1
Ligases is a class of enzymes responsible for catalysing the linking together of two compounds.
Which of the following bonds is not catalysed by it?
Q189.
mcq single
+4 / 1
Enzymes that catalyse the removal of groups from substrates by mechanisms other than hydrolysis leaving double bonds, are known as :
Q190.
mcq single
+4 / 1
Match List-I with List-II.
List - I
List - II
(A)
Primary structure of protein
(I)
Human haemoglobin
(B)
Secondary structure of protein
(II)
Disulphide bonds
(C)
Tertiary structure of protein
(III)
Polypeptide chain
(D)
Quanternary structure of protein
(IV)
Alpha helix and $$\beta$$ sheet
Choose the correct answer from the options given below :
Q191.
mcq single
+4 / 1
Which of the following graphs depicts the effect of substrate concentration on velocity of enzyme catalysed reaction?
Q192.
mcq single
+4 / 1
Which of the following are not fatty acids?
A. Glutamic acid
B. Arachidonic acid
C. Palmitic acid
D. Lecithin
E. Aspartic acid
Choose the correct answer from the options given below :
Q193.
mcq single
+4 / 1
Recombinant DNA molecule can be created normally by cutting the vector DNA and source DNA
respectively with:
Q194.
mcq single
+4 / 1
Which of the following are correct about EcoRI?
A. Cut the DNA with blunt end
B. Cut the DNA with sticky end
C. Recognise a specific palindromic sequence
D. Cut the DNA between the base $$\mathrm{G}$$ and $$\mathrm{A}$$ when encounters the DNA sequence 'GAATTC'
E. Exonuclease
Choose the correct answer from the options given below:
Q195.
mcq single
+4 / 1
The Bt toxin in genetically engineered Bt cotton kills the pest by:
Q196.
mcq single
+4 / 1
Match List-I with List - II :
List - I
List - II
(A)
Genetically engineered Human Insulin
(I)
Gene therapy
(B)
GM Cotton
(II)
E. coli
(C)
ADA Deficiency
(III)
Antigen-antibody interaction
(D)
ELISA
(IV)
Bacillus thuringiensis
Choose the correct answer from the options given below:
Q197.
mcq single
+4 / 1
Identify the incorrect statement related to gel electrophoresis.
Q198.
mcq single
+4 / 1
Given below are two statements:
Statement I : The Indian Government has set up GEAC, which will make decisions regarding the validity of GM research.
Statement II : Biopiracy is the term used to refer to the use of bio-resources by native people.
In the light of the above statements, choose the correct answer from the options given below :
Q199.
mcq single
+4 / 1
Select the restriction endonuclease enzymes whose restriction sites are present for the tetracycline resistance (tet$$^{\mathrm{R}}$$) gene in the pBR322 cloning vector.
Q200.
mcq single
+4 / 1
Following are the steps involved in the process of PCR.
A. Annealing
B. Amplification (~1 billion times)
C. Denaturation
D. Treatment with Taq polymerase and deoxynucleotides
E. Extension
Choose the correct sequence of steps of PCR from the options given below :