neet-2026
NEET 2026
MEDICAL 2026 Previous Year
3 hDuration
720Total Marks
360Questions
3Sections
Instructions
General instructions for this test:
- Duration: 3 h. The timer starts as soon as you begin and cannot be paused.
- Total questions: 360 across 3 section(s); maximum marks: 720.
- You are allowed 1 attempt(s) at this test.
- Use the question palette on the right to navigate. Answered questions are highlighted in green; questions marked for review are highlighted in yellow.
- Each question's marking scheme (correct / wrong) is shown on the question card. Unanswered questions receive zero marks.
- Switching tabs, exiting full-screen, or attempting to copy text is monitored. Repeated tab-switching may auto-submit the test.
- Your answers autosave as you navigate. Click Submit Test when you are done. The test will be auto-submitted when the timer expires.
No exam-specific instructions were provided for this paper.
Paper Structure
Chemistry
Chemistry
Q1.
mcq single
+4 / 1
Methane reacts with steam at 1273 K in the presence of nickel catalyst to form :
Q2.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Transition metal/compound/ complex)
List II
(Catalytic Role)
A.
$$\mathrm{V}_2 \mathrm{O}_5$$
I.
Preparation of ammonia from $\mathrm{N}_2 / \mathrm{H}_2$ mixture
B.
Fe
II.
Polymerisation of alkynes
C.
$\mathrm{PdCl}_2$
III.
Preparation of $\mathrm{H}_2 \mathrm{SO}_4$ and $\mathrm{SO}_2$
D.
Ni complex
IV.
Oxidation of ethyne to ethanal
Choose the correct answer from the options given below.
Q3.
mcq single
+4 / 1
Although +3 oxidation state is most common in lanthanoids, cerium still shows +4 oxidation state because:
Q4.
mcq single
+4 / 1
The calculated 'spin-only' magnetic moment $\mathrm{Ti}^{2+}\left(3 \mathrm{~d}^2\right)$ is :
Q5.
mcq single
+4 / 1
Identify the correct statement about $\mathrm{CIF}_3$ from the following options :
Q6.
mcq single
+4 / 1
$$ \text { The correct formal charges on oxygen atoms numbered } 2,1 \text { and } 3 \text { respectively are : } $$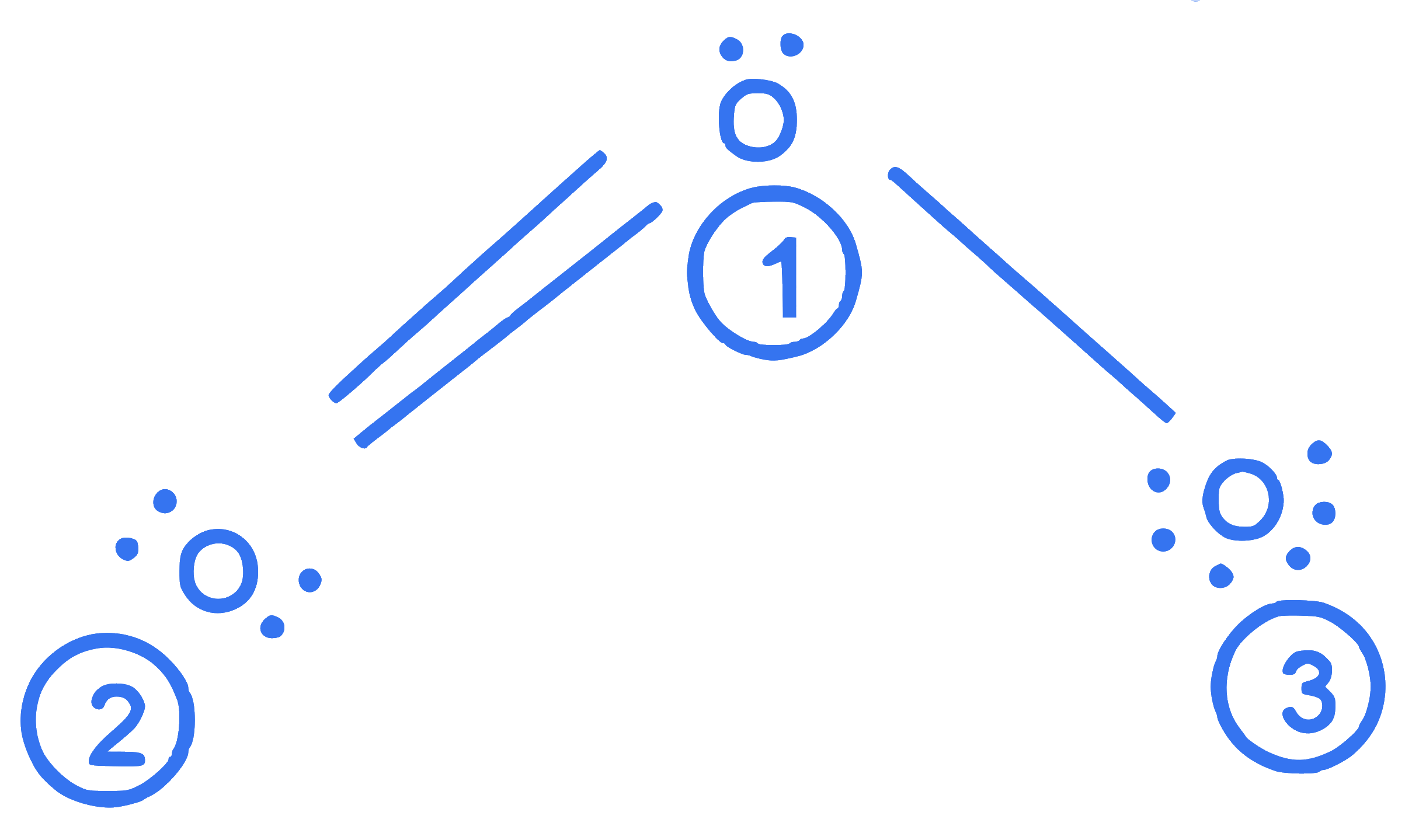

Q7.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
List-II
A.
$\mathrm{C}_2 \mathrm{H}_4$
I.
$3 \sigma$ bonds, $2 \pi$ bonds
B.
$\mathrm{C}_2 \mathrm{H}_2$
II.
$3 \sigma$ bonds, one lone pair
C.
$\mathrm{CH}_4$
III.
$4 \sigma$ bonds
D.
$\mathrm{NH}_3$
IV.
$5 \sigma$ bonds, $1 \pi$ bond
Choose the correct answer from the options given below :
Q8.
mcq single
+4 / 1
Phenolphthalein is used as an indicator for the titration of sodium hydroxide solution against a standard solution of oxalic acid. The colour change that is observed at an alkaline pH close to the equivalence point during this titration is:
Q9.
mcq single
+4 / 1
In a qualitative analysis, $\mathrm{Bi}^{3+}$ is detected by appearance of precipitate of $\mathrm{BiO}(\mathrm{OH})(\mathrm{s})$. Calculate pH when the following equilibrium exists at 298 K .
$$ \mathrm{BiO}(\mathrm{OH})(\mathrm{s}) \rightleftharpoons \mathrm{BiO}^{+}(\mathrm{aq})+\mathrm{OH}^{-}(\mathrm{aq}), \mathrm{K}=4 \times 10^{-10} $$
(Given : $\log 2=0.3010$ )
Q10.
mcq single
+4 / 1
At 298 K , a certain buffer solution contains equal concentrations of $\mathrm{X}^{-}$and $\mathrm{HX}, \mathrm{K}_{\mathrm{b}}$ for $\mathrm{X}^{-}$is $10^{-10}$. What is the pH of this buffer solution?
Q11.
mcq single
+4 / 1
For a certain reaction $R \rightarrow$ Product, the plot of concentration $[R]$ vs time has a negative slope as shown. The order of reaction is :
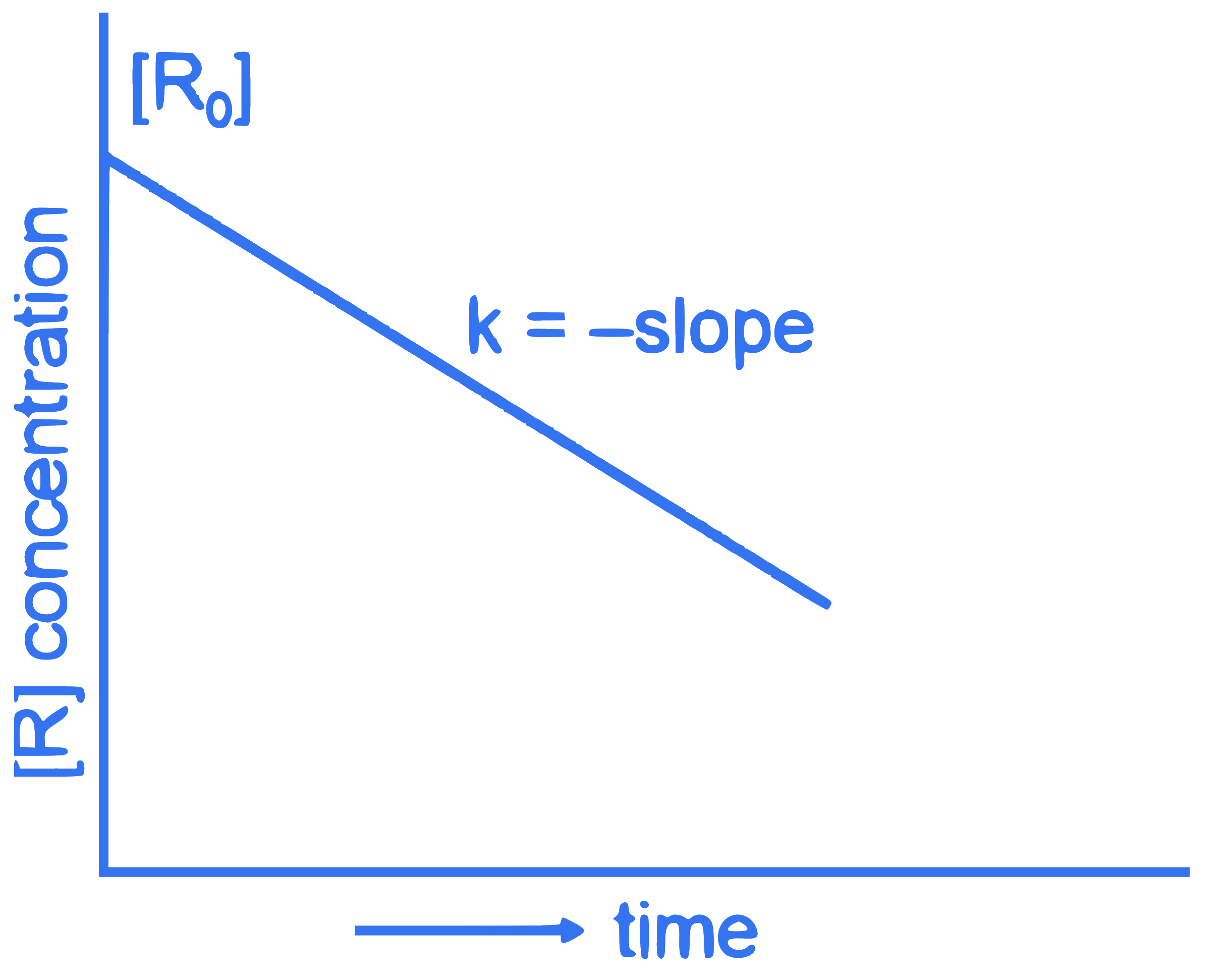

Q12.
mcq single
+4 / 1
Given below is an expression for the rate constant of a first-order reaction occurring at a certain temperature, $\mathrm{T}(\mathrm{K})$.
$$ \operatorname{lnk}=14.34-\frac{1.25 \times 10^4}{T} $$
The energy of activation in $\mathrm{kcal} \mathrm{mol}^{-1}$ for the reaction is :
(Given: $\mathrm{k}^{-1} \mathrm{~s} \mathrm{~s}^{-1}, \mathrm{R}=1.987 \mathrm{cal} \mathrm{mol}^{-1} \mathrm{~K}^{-1}$ )
Q13.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Order of reaction)
List-II
(Unit of rate constant)
A.
Zero order
I.
$\mathrm{mol}^{-1} \mathrm{~L} \mathrm{~s}^{-1}$
B.
First order
II.
$\mathrm{mol}^{-2} \mathrm{~L}^2 \mathrm{~s}^{-1}$
C.
Second order
III.
$\mathrm{s}^{-1}$
D.
Third order
IV.
$\mathrm{mol} \mathrm{L}^{-1} \mathrm{~s}^{-1}$
Choose the correct answer from the options given below :
Q14.
mcq single
+4 / 1
Identify the correct statements :
(A) The molality of 2.5 g of ethanoic acid (Molar mass : $60 \mathrm{~g} \mathrm{~mol}^{-1}$ ) in 75 g of benzene solution is 0.556 m .
(B) The molarity of a solution containing 5 g of NaOH (molar mass : $40 \mathrm{~g} \mathrm{~mol}^{-1}$ ) in 450 mL of solution is 0.278 M at 298 K .
(C) Aquatic species are more comfortable in cold water.
(D) The solubility of gas increases with decrease in pressure.
(E) For a binary mixture of $A$ and $B$, the number of moles of $A$ and $B$ are $n_A$ and $n_B$ respectively. The mole fraction of $B$ will be $x_B=\frac{n_A}{n_A+n_B}$.
Choose the correct answer from the options given below :
Q15.
mcq single
+4 / 1
Mixture of chloroform and acetone forms a solution with negative deviation from Raoult's law due to :
Q16.
mcq single
+4 / 1
The number of hydrogen atoms present in 5.4 g of urea is:
(Given: Molar mass of urea : $60 \mathrm{~g} \mathrm{~mol}^{-1}$
$\mathrm{N}_{\mathrm{A}}: 6.022 \times 10^{23}$ particles $\mathrm{mol}^{-1}$ )
Q17.
mcq single
+4 / 1
When $1 \mathrm{dm}^3$ of CO~2~ gas is passed over hot coke the volume of gaseous mixture after complete reaction at STP becomes $1.4 \mathrm{dm}^3$. The composition of the gaseous mixture at STP is :
Q18.
mcq single
+4 / 1
Given below are certain reactions. Identify the reaction for which $K_P \neq K_C$.
Q19.
mcq single
+4 / 1
The correct order of increasing metallic character of $\mathrm{Na}, \mathrm{Be}, \mathrm{P}, \mathrm{Mg}$ and Si is
Q20.
mcq single
+4 / 1
Identify the incorrect statement from the following :
Q21.
mcq single
+4 / 1
At a certain temperature, $\mathrm{T}(\mathrm{K})$, during a process, 500 J is absorbed by the system and work of 200 J is done by the system. Then change in internal energy of the system is :
Q22.
mcq single
+4 / 1
Consider the following reaction :
$$ \begin{aligned} & 2 \mathrm{~A}(\mathrm{~g})+\mathrm{B}(\mathrm{~g}) \rightarrow 2 \mathrm{D}(\mathrm{~g}) \\ & \Delta \mathrm{U}^{\ominus}=-10 \mathrm{~kJ} \mathrm{~mol}^{-1} \text { and } \Delta \mathrm{S}^{\ominus}=-44 \mathrm{JK}^{-1} \text { at } 298 \mathrm{~K} . \end{aligned} $$
Identify the correct option with $\Delta \mathrm{G}^{\ominus}$ for the reaction and spontaneity of the reaction at 298 K .
(Given : $\mathrm{R}=8.31 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ )
Q23.
mcq single
+4 / 1
Identify the incorrect statement from the following:
Q24.
mcq single
+4 / 1
Identify the incorrect statement from the following:
Q25.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Complex)
List-II
(Type of isomerism)
A.
$$
\left[\mathrm{Pt}\left(\mathrm{NH}_3\right)_2 \mathrm{Cl}_2\right]
$$
I.
Optical
B.
$$
\left[\mathrm{Co}(\mathrm{en})_3\right]^{3+}
$$
II.
Solvate
C.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5 \mathrm{NO}_2\right] \mathrm{Cl}_2
$$
III.
Geometrical
D.
$$
\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{Cl}_3
$$
IV.
Linkage
Choose the correct answer from the options given below :
Q26.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Complex/ion)
List II
(Shape/geometry)
A.
$$
\left[\mathrm{Pt}\left(\mathrm{Cl}_2\right)\left(\mathrm{NH}_3\right)_2\right]
$$
I.
Octahedral
B.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right] \mathrm{Cl}_3
$$
II.
Trigonal bipyramidal
C.
$$
\left[\mathrm{NiCl}_4\right]^{2-}
$$
III.
Square planar
D.
$$
\left[\mathrm{Fe}(\mathrm{CO})_5\right]
$$
IV.
Tetrahedral
Choose the correct answer from the options given below :
Q27.
mcq single
+4 / 1
Which one of the following is an ambidentate ligand?
Q28.
mcq single
+4 / 1
$$ \text { The correct IUPAC name of the following compound is : } $$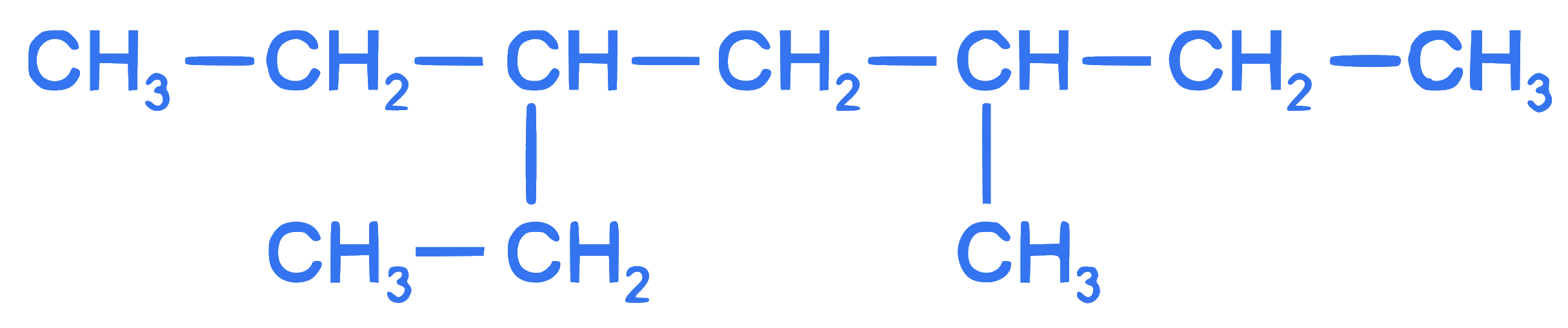
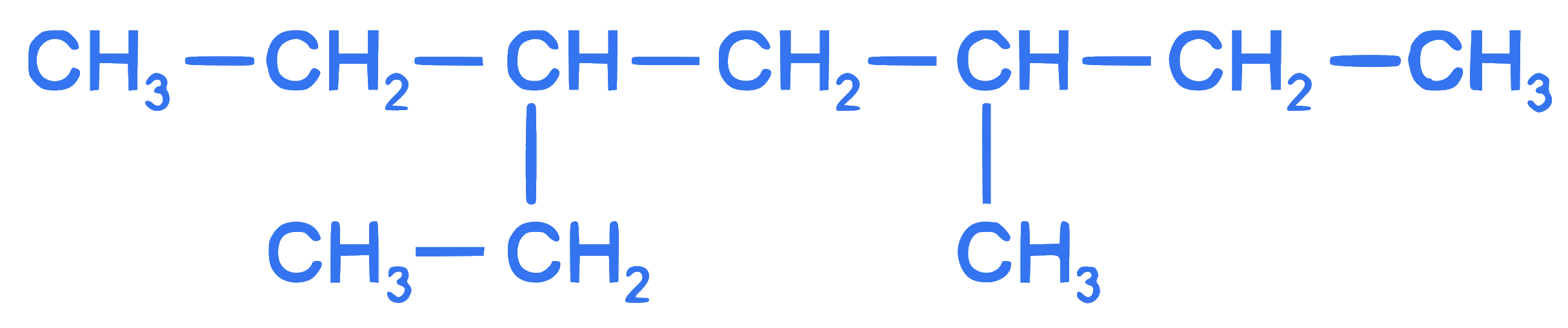
Q29.
mcq single
+4 / 1
During Lassaigne's test, the elements present in an organic compound are converted from :
Q30.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
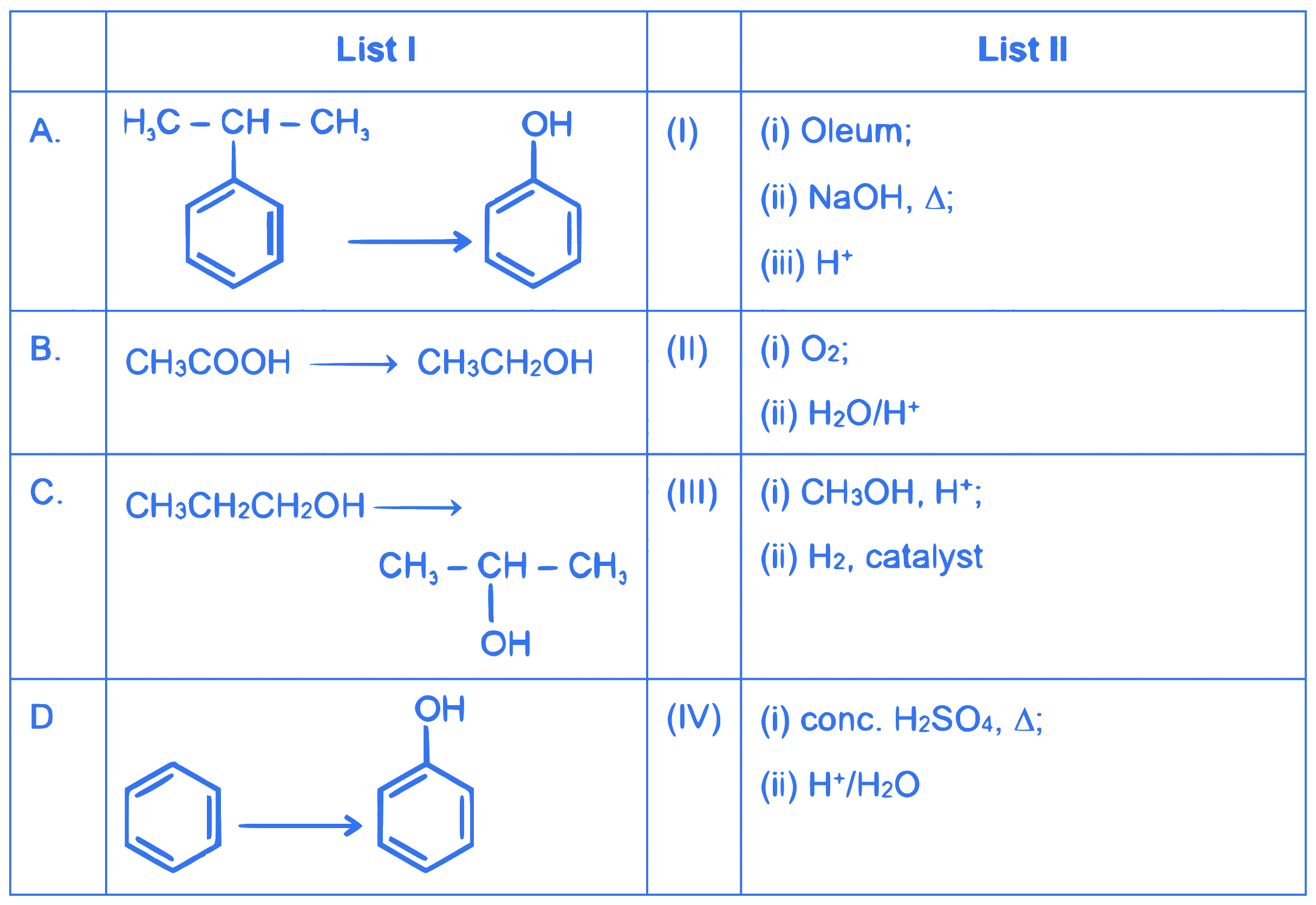 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q31.
mcq single
+4 / 1
In a test tube containing a salt, a few drops of dilute $\mathrm{H}_2 \mathrm{SO}_4$ was added, which gave colourless vapours having the smell of vinegar. The vapours turned the blue litmus paper red.
Identify the correct anion from the following :
Q32.
mcq single
+4 / 1
Compound $\mathrm{P}\left(\mathrm{C}_8 \mathrm{H}_8 \mathrm{O}\right)$ gives a red orange precipitate with 2,4-DNP reagent and it does not reduce Fehling's reagent. On drastic oxidation with chromic acid, $P$ gives an aromatic product $Q$ that produces effervescence on treating with aq. $\mathrm{NaHCO}_3$. Compounds P and Q , respectively, are :
Q33.
mcq single
+4 / 1
$$ \text { In the following reaction sequence, } \mathrm{X} \text { and } \mathrm{Z} \text { respectively are : } $$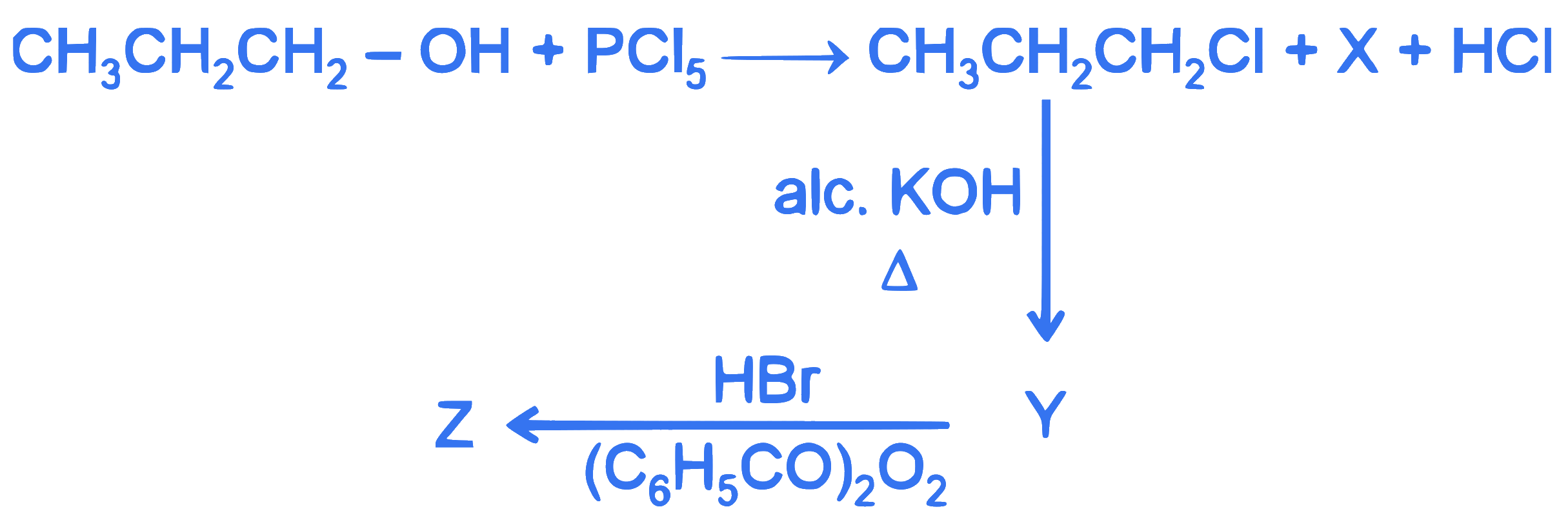

Q34.
mcq single
+4 / 1
The pair of molecules that are metamers among the following is :
Q35.
mcq single
+4 / 1
The functional group that can be identified through phthalein dye test is :
Q36.
mcq single
+4 / 1
The number of chlorine atoms present in the organic products $X$ and $Y$ of the following reactions, respectively, are :
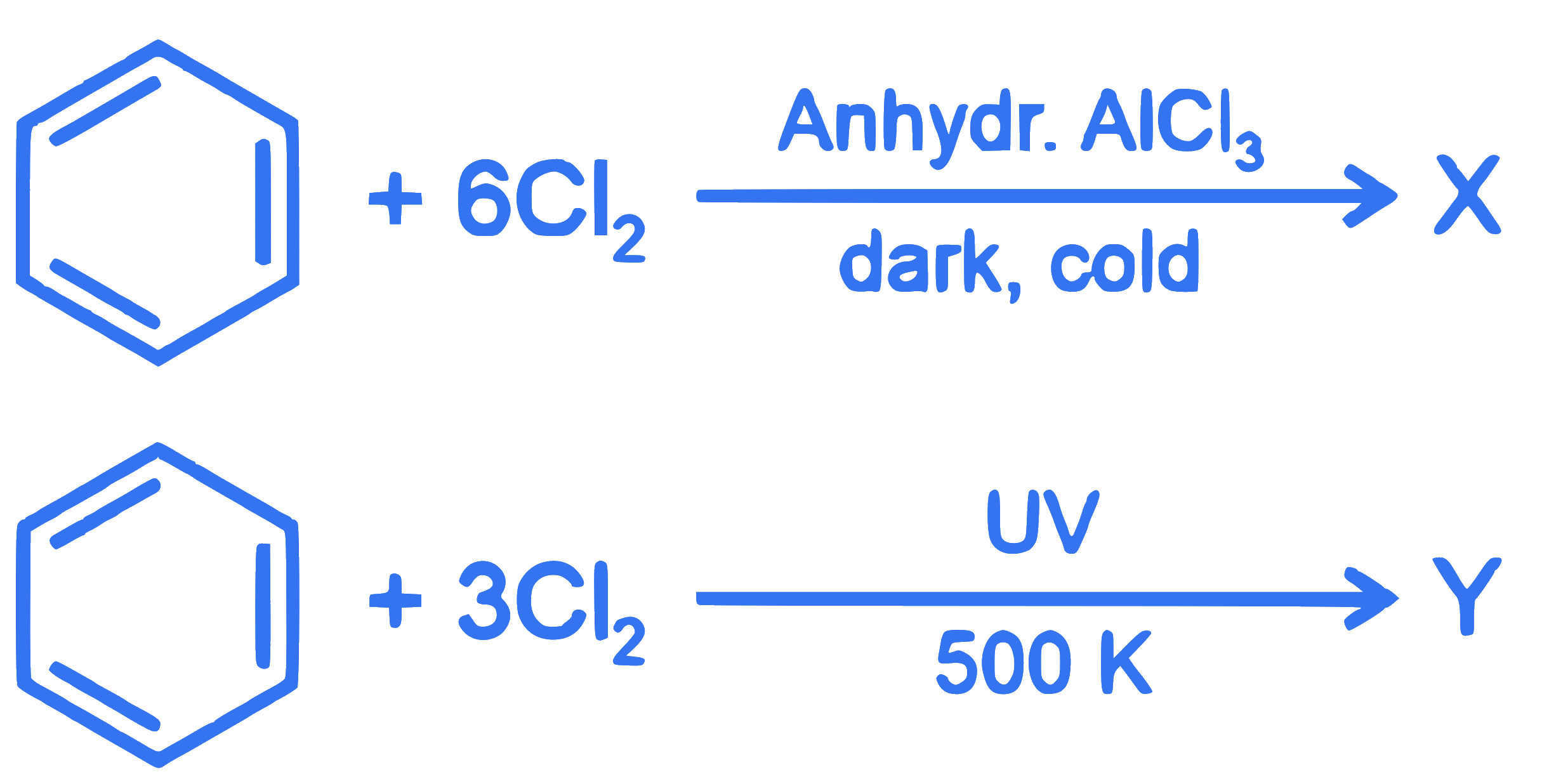

Q37.
mcq single
+4 / 1
The correct statement with regard to the secondary structure of DNA/RNA is
Q38.
mcq single
+4 / 1
A solution of copper sulphate is electrolysed for 10 minutes with a current of 1.5 amperes. The mass of copper deposited at cathode is :
(Given : Molar mass of $\mathrm{Cu}=63 \mathrm{~g} \mathrm{~mol}^{-1}$;
$$ \left.1 \mathrm{~F}=96487 \mathrm{C} \mathrm{~mol}^{-1}\right) $$
Q39.
mcq single
+4 / 1
Calculate emf of the half cell given below :
$$ \begin{aligned} & \mathrm{Pt}(\mathrm{~s})\left|\mathrm{H}_2(\mathrm{~g}, 2 \mathrm{~atm})\right| \mathrm{HCl}(\mathrm{aq}, 0.02 \mathrm{M}) \\ & \mathrm{E}_{\mathrm{H}_2 / \mathrm{H}^{+}}^{\circ}=0 \mathrm{~V} \end{aligned} $$
(Given: $\frac{2.303 R T}{F}=0.059, \log 2=0.3010$ )
Q40.
mcq single
+4 / 1
Select the reagents that reduce nitriles to primary amines.
A. (i) $\mathrm{LiAlH}_4$; (ii) $\mathrm{H}_2 \mathrm{O}$
B. $\mathrm{Sn}+\mathrm{HCl}$
C. $\mathrm{H}_2 / \mathrm{Ni}$
D. $\mathrm{Na}(\mathrm{Hg}) / \mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}$
E. $\mathrm{Br}_2 / \mathrm{aq} . \mathrm{NaOH}$
Choose the correct answer from the options given below.
Q41.
mcq single
+4 / 1
$$ \text { Two products } X \text { and } Y \text { are formed in the following reaction sequence. } $$
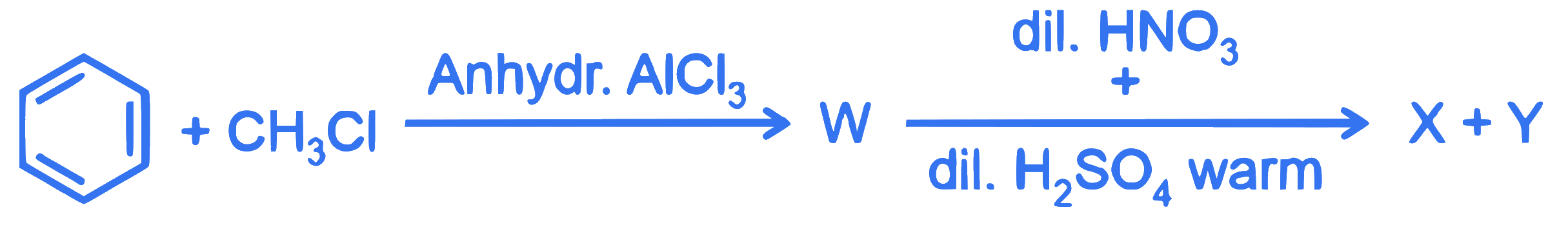 The suitable method that can be used for the separation of products X and Y is :
The suitable method that can be used for the separation of products X and Y is :
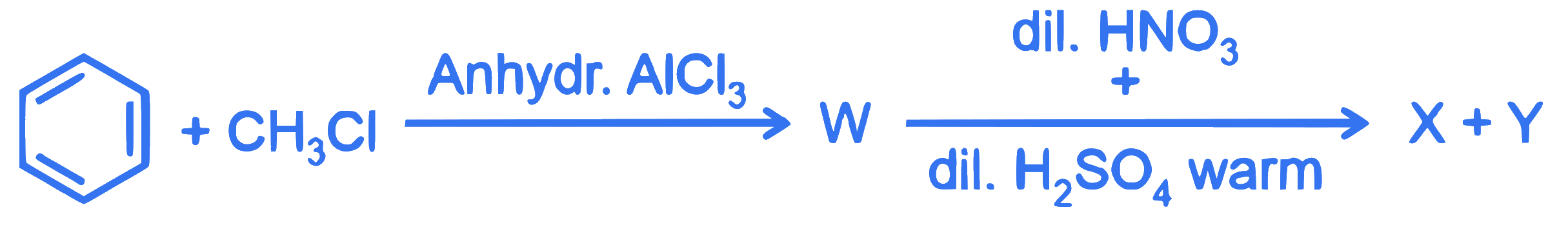 The suitable method that can be used for the separation of products X and Y is :
The suitable method that can be used for the separation of products X and Y is :
Q42.
mcq single
+4 / 1
The major product Z formed in the following sequence of reactions is

Q43.
mcq single
+4 / 1
$$ \begin{aligned} &\text { The following two reactions give the same foul smelling product } Z \text {. }\\ &\mathrm{C}_2 \mathrm{H}_5 \mathrm{Cl} \xrightarrow{\mathrm{X}} \mathrm{Z} \end{aligned} $$
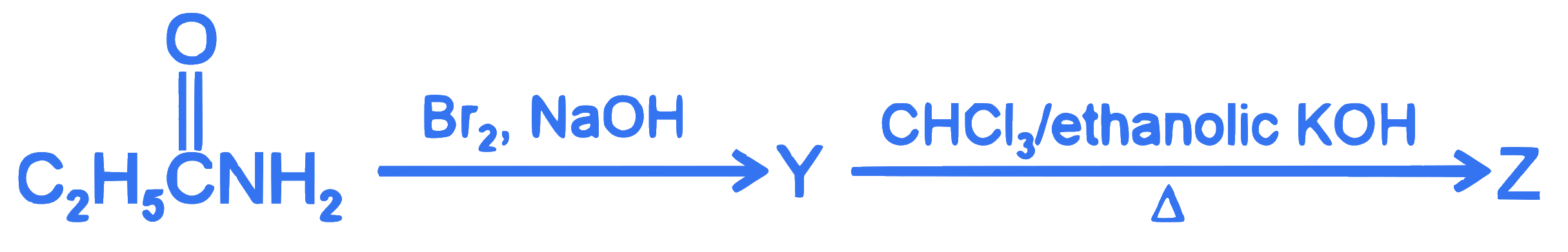 $X$ and $Z$, respectively, are :
$X$ and $Z$, respectively, are :
 $X$ and $Z$, respectively, are :
$X$ and $Z$, respectively, are :
Q44.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Quantum Numbers)
List II (Orbital)
'n'
'I'
A.
2
1
I.
3d
B.
4
0
II.
2p
C.
5
3
III.
4s
D.
3
2
IV.
5f
Choose the correct answer from the options given below.
Q45.
mcq single
+4 / 1
A bulb is rated at 150 watt, converting $8 \%$ energy into light. If energy of one photon is $4.42 \times 10^{-19} \mathrm{~J}$, how many photons are emitted by the bulb per second?
Q46.
mcq single
+4 / 1
Methane reacts with steam at 1273 K in the presence of nickel catalyst to form :
Q47.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Transition metal/compound/ complex)
List II
(Catalytic Role)
A.
$$\mathrm{V}_2 \mathrm{O}_5$$
I.
Preparation of ammonia from $\mathrm{N}_2 / \mathrm{H}_2$ mixture
B.
Fe
II.
Polymerisation of alkynes
C.
$\mathrm{PdCl}_2$
III.
Preparation of $\mathrm{H}_2 \mathrm{SO}_4$ and $\mathrm{SO}_2$
D.
Ni complex
IV.
Oxidation of ethyne to ethanal
Choose the correct answer from the options given below.
Q48.
mcq single
+4 / 1
Although +3 oxidation state is most common in lanthanoids, cerium still shows +4 oxidation state because:
Q49.
mcq single
+4 / 1
The calculated 'spin-only' magnetic moment $\mathrm{Ti}^{2+}\left(3 \mathrm{~d}^2\right)$ is :
Q50.
mcq single
+4 / 1
Identify the correct statement about $\mathrm{CIF}_3$ from the following options :
Q51.
mcq single
+4 / 1
$$ \text { The correct formal charges on oxygen atoms numbered } 2,1 \text { and } 3 \text { respectively are : } $$

Q52.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
List-II
A.
$\mathrm{C}_2 \mathrm{H}_4$
I.
$3 \sigma$ bonds, $2 \pi$ bonds
B.
$\mathrm{C}_2 \mathrm{H}_2$
II.
$3 \sigma$ bonds, one lone pair
C.
$\mathrm{CH}_4$
III.
$4 \sigma$ bonds
D.
$\mathrm{NH}_3$
IV.
$5 \sigma$ bonds, $1 \pi$ bond
Choose the correct answer from the options given below :
Q53.
mcq single
+4 / 1
Phenolphthalein is used as an indicator for the titration of sodium hydroxide solution against a standard solution of oxalic acid. The colour change that is observed at an alkaline pH close to the equivalence point during this titration is:
Q54.
mcq single
+4 / 1
In a qualitative analysis, $\mathrm{Bi}^{3+}$ is detected by appearance of precipitate of $\mathrm{BiO}(\mathrm{OH})(\mathrm{s})$. Calculate pH when the following equilibrium exists at 298 K .
$$ \mathrm{BiO}(\mathrm{OH})(\mathrm{s}) \rightleftharpoons \mathrm{BiO}^{+}(\mathrm{aq})+\mathrm{OH}^{-}(\mathrm{aq}), \mathrm{K}=4 \times 10^{-10} $$
(Given : $\log 2=0.3010$ )
Q55.
mcq single
+4 / 1
At 298 K , a certain buffer solution contains equal concentrations of $\mathrm{X}^{-}$and $\mathrm{HX}, \mathrm{K}_{\mathrm{b}}$ for $\mathrm{X}^{-}$is $10^{-10}$. What is the pH of this buffer solution?
Q56.
mcq single
+4 / 1
For a certain reaction $R \rightarrow$ Product, the plot of concentration $[R]$ vs time has a negative slope as shown. The order of reaction is :


Q57.
mcq single
+4 / 1
Given below is an expression for the rate constant of a first-order reaction occurring at a certain temperature, $\mathrm{T}(\mathrm{K})$.
$$ \operatorname{lnk}=14.34-\frac{1.25 \times 10^4}{T} $$
The energy of activation in $\mathrm{kcal} \mathrm{mol}^{-1}$ for the reaction is :
(Given: $\mathrm{k}^{-1} \mathrm{~s} \mathrm{~s}^{-1}, \mathrm{R}=1.987 \mathrm{cal} \mathrm{mol}^{-1} \mathrm{~K}^{-1}$ )
Q58.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Order of reaction)
List-II
(Unit of rate constant)
A.
Zero order
I.
$\mathrm{mol}^{-1} \mathrm{~L} \mathrm{~s}^{-1}$
B.
First order
II.
$\mathrm{mol}^{-2} \mathrm{~L}^2 \mathrm{~s}^{-1}$
C.
Second order
III.
$\mathrm{s}^{-1}$
D.
Third order
IV.
$\mathrm{mol} \mathrm{L}^{-1} \mathrm{~s}^{-1}$
Choose the correct answer from the options given below :
Q59.
mcq single
+4 / 1
Identify the correct statements :
(A) The molality of 2.5 g of ethanoic acid (Molar mass : $60 \mathrm{~g} \mathrm{~mol}^{-1}$ ) in 75 g of benzene solution is 0.556 m .
(B) The molarity of a solution containing 5 g of NaOH (molar mass : $40 \mathrm{~g} \mathrm{~mol}^{-1}$ ) in 450 mL of solution is 0.278 M at 298 K .
(C) Aquatic species are more comfortable in cold water.
(D) The solubility of gas increases with decrease in pressure.
(E) For a binary mixture of $A$ and $B$, the number of moles of $A$ and $B$ are $n_A$ and $n_B$ respectively. The mole fraction of $B$ will be $x_B=\frac{n_A}{n_A+n_B}$.
Choose the correct answer from the options given below :
Q60.
mcq single
+4 / 1
Mixture of chloroform and acetone forms a solution with negative deviation from Raoult's law due to :
Q61.
mcq single
+4 / 1
The number of hydrogen atoms present in 5.4 g of urea is:
(Given: Molar mass of urea : $60 \mathrm{~g} \mathrm{~mol}^{-1}$
$\mathrm{N}_{\mathrm{A}}: 6.022 \times 10^{23}$ particles $\mathrm{mol}^{-1}$ )
Q62.
mcq single
+4 / 1
When $1 \mathrm{dm}^3$ of CO~2~ gas is passed over hot coke the volume of gaseous mixture after complete reaction at STP becomes $1.4 \mathrm{dm}^3$. The composition of the gaseous mixture at STP is :
Q63.
mcq single
+4 / 1
Given below are certain reactions. Identify the reaction for which $K_P \neq K_C$.
Q64.
mcq single
+4 / 1
The correct order of increasing metallic character of $\mathrm{Na}, \mathrm{Be}, \mathrm{P}, \mathrm{Mg}$ and Si is
Q65.
mcq single
+4 / 1
Identify the incorrect statement from the following :
Q66.
mcq single
+4 / 1
At a certain temperature, $\mathrm{T}(\mathrm{K})$, during a process, 500 J is absorbed by the system and work of 200 J is done by the system. Then change in internal energy of the system is :
Q67.
mcq single
+4 / 1
Consider the following reaction :
$$ \begin{aligned} & 2 \mathrm{~A}(\mathrm{~g})+\mathrm{B}(\mathrm{~g}) \rightarrow 2 \mathrm{D}(\mathrm{~g}) \\ & \Delta \mathrm{U}^{\ominus}=-10 \mathrm{~kJ} \mathrm{~mol}^{-1} \text { and } \Delta \mathrm{S}^{\ominus}=-44 \mathrm{JK}^{-1} \text { at } 298 \mathrm{~K} . \end{aligned} $$
Identify the correct option with $\Delta \mathrm{G}^{\ominus}$ for the reaction and spontaneity of the reaction at 298 K .
(Given : $\mathrm{R}=8.31 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ )
Q68.
mcq single
+4 / 1
Identify the incorrect statement from the following:
Q69.
mcq single
+4 / 1
Identify the incorrect statement from the following:
Q70.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Complex)
List-II
(Type of isomerism)
A.
$$
\left[\mathrm{Pt}\left(\mathrm{NH}_3\right)_2 \mathrm{Cl}_2\right]
$$
I.
Optical
B.
$$
\left[\mathrm{Co}(\mathrm{en})_3\right]^{3+}
$$
II.
Solvate
C.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5 \mathrm{NO}_2\right] \mathrm{Cl}_2
$$
III.
Geometrical
D.
$$
\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right] \mathrm{Cl}_3
$$
IV.
Linkage
Choose the correct answer from the options given below :
Q71.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Complex/ion)
List II
(Shape/geometry)
A.
$$
\left[\mathrm{Pt}\left(\mathrm{Cl}_2\right)\left(\mathrm{NH}_3\right)_2\right]
$$
I.
Octahedral
B.
$$
\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right] \mathrm{Cl}_3
$$
II.
Trigonal bipyramidal
C.
$$
\left[\mathrm{NiCl}_4\right]^{2-}
$$
III.
Square planar
D.
$$
\left[\mathrm{Fe}(\mathrm{CO})_5\right]
$$
IV.
Tetrahedral
Choose the correct answer from the options given below :
Q72.
mcq single
+4 / 1
Which one of the following is an ambidentate ligand?
Q73.
mcq single
+4 / 1
$$ \text { The correct IUPAC name of the following compound is : } $$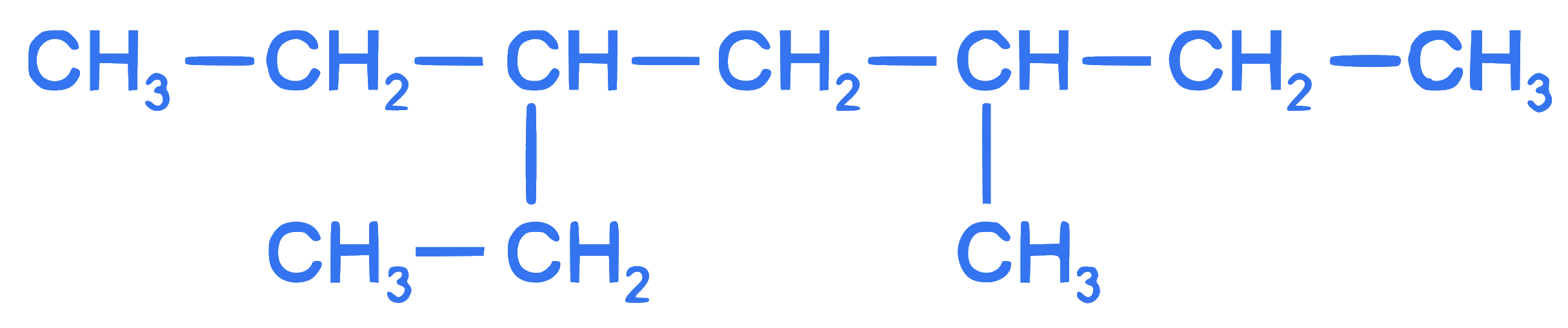
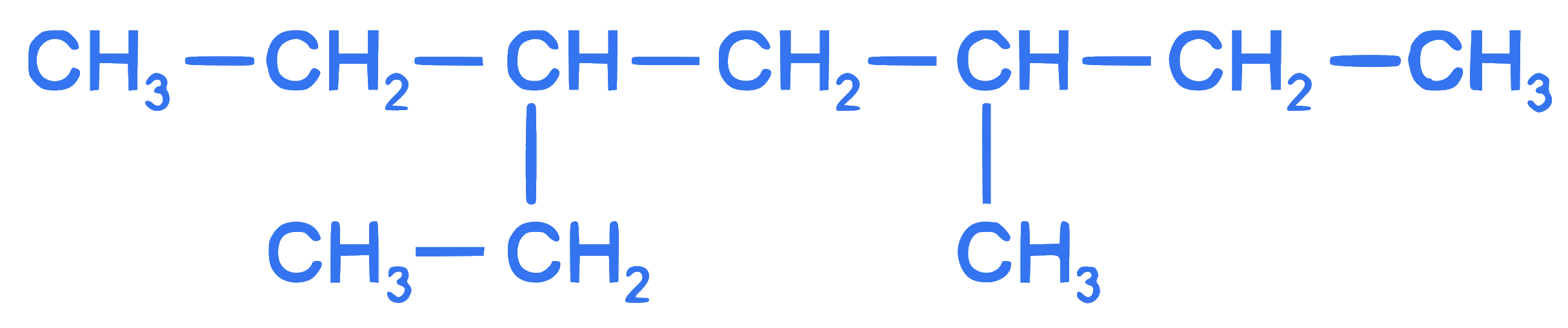
Q74.
mcq single
+4 / 1
During Lassaigne's test, the elements present in an organic compound are converted from :
Q75.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
Q76.
mcq single
+4 / 1
In a test tube containing a salt, a few drops of dilute $\mathrm{H}_2 \mathrm{SO}_4$ was added, which gave colourless vapours having the smell of vinegar. The vapours turned the blue litmus paper red.
Identify the correct anion from the following :
Q77.
mcq single
+4 / 1
Compound $\mathrm{P}\left(\mathrm{C}_8 \mathrm{H}_8 \mathrm{O}\right)$ gives a red orange precipitate with 2,4-DNP reagent and it does not reduce Fehling's reagent. On drastic oxidation with chromic acid, $P$ gives an aromatic product $Q$ that produces effervescence on treating with aq. $\mathrm{NaHCO}_3$. Compounds P and Q , respectively, are :
Q78.
mcq single
+4 / 1
$$ \text { In the following reaction sequence, } \mathrm{X} \text { and } \mathrm{Z} \text { respectively are : } $$

Q79.
mcq single
+4 / 1
The pair of molecules that are metamers among the following is :
Q80.
mcq single
+4 / 1
The functional group that can be identified through phthalein dye test is :
Q81.
mcq single
+4 / 1
The number of chlorine atoms present in the organic products $X$ and $Y$ of the following reactions, respectively, are :


Q82.
mcq single
+4 / 1
The correct statement with regard to the secondary structure of DNA/RNA is
Q83.
mcq single
+4 / 1
A solution of copper sulphate is electrolysed for 10 minutes with a current of 1.5 amperes. The mass of copper deposited at cathode is :
(Given : Molar mass of $\mathrm{Cu}=63 \mathrm{~g} \mathrm{~mol}^{-1}$;
$$ \left.1 \mathrm{~F}=96487 \mathrm{C} \mathrm{~mol}^{-1}\right) $$
Q84.
mcq single
+4 / 1
Calculate emf of the half cell given below :
$$ \begin{aligned} & \mathrm{Pt}(\mathrm{~s})\left|\mathrm{H}_2(\mathrm{~g}, 2 \mathrm{~atm})\right| \mathrm{HCl}(\mathrm{aq}, 0.02 \mathrm{M}) \\ & \mathrm{E}_{\mathrm{H}_2 / \mathrm{H}^{+}}^{\circ}=0 \mathrm{~V} \end{aligned} $$
(Given: $\frac{2.303 R T}{F}=0.059, \log 2=0.3010$ )
Q85.
mcq single
+4 / 1
Select the reagents that reduce nitriles to primary amines.
A. (i) $\mathrm{LiAlH}_4$; (ii) $\mathrm{H}_2 \mathrm{O}$
B. $\mathrm{Sn}+\mathrm{HCl}$
C. $\mathrm{H}_2 / \mathrm{Ni}$
D. $\mathrm{Na}(\mathrm{Hg}) / \mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}$
E. $\mathrm{Br}_2 / \mathrm{aq} . \mathrm{NaOH}$
Choose the correct answer from the options given below.
Q86.
mcq single
+4 / 1
$$ \text { Two products } X \text { and } Y \text { are formed in the following reaction sequence. } $$
 The suitable method that can be used for the separation of products X and Y is :
The suitable method that can be used for the separation of products X and Y is :
 The suitable method that can be used for the separation of products X and Y is :
The suitable method that can be used for the separation of products X and Y is :
Q87.
mcq single
+4 / 1
The major product Z formed in the following sequence of reactions is

Q88.
mcq single
+4 / 1
$$ \begin{aligned} &\text { The following two reactions give the same foul smelling product } Z \text {. }\\ &\mathrm{C}_2 \mathrm{H}_5 \mathrm{Cl} \xrightarrow{\mathrm{X}} \mathrm{Z} \end{aligned} $$
 $X$ and $Z$, respectively, are :
$X$ and $Z$, respectively, are :
 $X$ and $Z$, respectively, are :
$X$ and $Z$, respectively, are :
Q89.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Quantum Numbers)
List II (Orbital)
'n'
'I'
A.
2
1
I.
3d
B.
4
0
II.
2p
C.
5
3
III.
4s
D.
3
2
IV.
5f
Choose the correct answer from the options given below.
Q90.
mcq single
+4 / 1
A bulb is rated at 150 watt, converting $8 \%$ energy into light. If energy of one photon is $4.42 \times 10^{-19} \mathrm{~J}$, how many photons are emitted by the bulb per second?
Physics
Physics
Q1.
mcq single
+4 / 1
In interference and diffraction, the light energy is redistributed. If it reduces in one region, producing a dark fringe, it increases in another region, producing a bright fringe.
A. As there is no gain or loss of energy, these phenomena are consistent with the principle of conservation of energy.
B. Diffraction and interference are characteristics exhibited only by light waves.
Choose the correct answer from the options given below:
Q2.
mcq single
+4 / 1
In Young's double slit experiment, using monochromatic light of wavelength $\lambda$, the intensity of light at a point on the screen where the path difference is $\lambda$, is $K$ units. The intensity of light at a point where the path difference is $\frac{\lambda}{3}$ will be
Q3.
mcq single
+4 / 1
The angular speed of a flywheel is increased from 600 rpm to 1200 rpm in 10 s . The number of revolutions completed by the flywheel during this time is:
Q4.
mcq single
+4 / 1
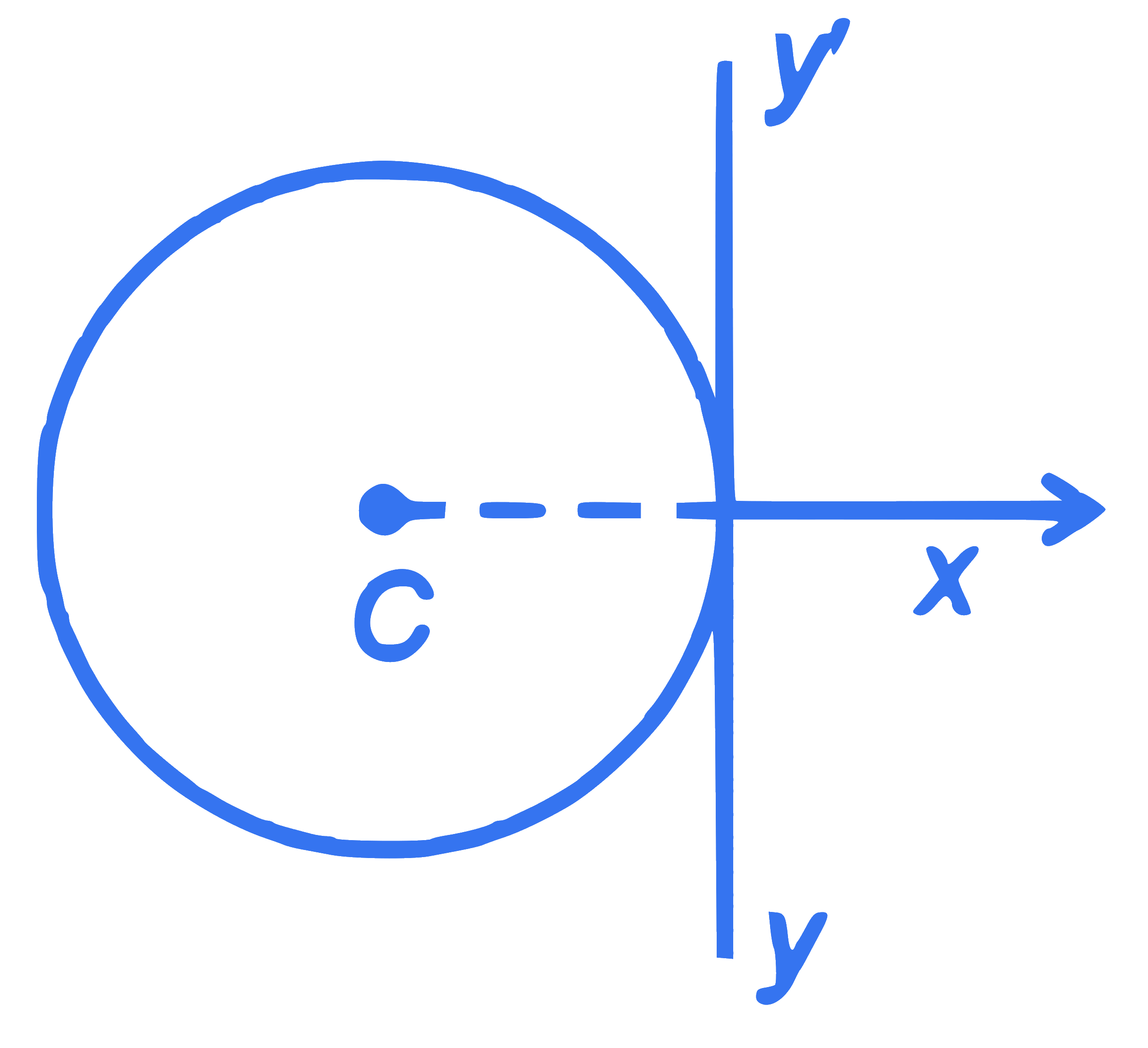
A thin wire of length ' $L$ ' and linear mass density ' $m$ ' is bent into a circular ring (in $x-y$ plane) with centre ' $C$ ' as shown in figure. The moment of inertia of the ring about an axis $y y^{\prime}$ will be:

Q5.
mcq single
+4 / 1
A rectangular wire loop of sides 8 cm and 3 cm with a small cut, is moving out of a region of uniform magnetic field of magnitude 0.3 T directed normal to the plane of the loop. The emf developed across the cut, if the velocity of the loop is $2 \mathrm{~cm} \mathrm{~s}^{-1}$, in a direction normal to the shorter side of the loop, will be :
Q6.
mcq single
+4 / 1
In a vernier calliper, 20 VSD coincide with 16 MSD (each division of length 1 mm ). The least count of the vernier callipers is:
Q7.
mcq single
+4 / 1
Each side of a metallic cube of mass 5.580 kg is measured to the 9.0 cm . Keeping the significant figures in view, the density of the material of the cube can be best expressed as $X \times 10^3 \mathrm{~kg} \mathrm{~m}^{-3}$ where the value of $X$ is:
Q8.
mcq single
+4 / 1
The speed of light in vacuum is taken as unity. If light takes 6 min 40 s to reach the Earth from the Sun, the distance between the Sun and the Earth in new unit is:
Q9.
mcq single
+4 / 1
A flask contains argon and chlorine in the ratio of $2: 1$ by mass. The temperature of the mixture is $27^{\circ} \mathrm{C}$. The ratio of root mean square speed of the molecules of the two gases $\left(\frac{V_{\mathrm{rms}}^{\mathrm{Ar}}}{V_{\mathrm{rms}}^{\mathrm{Cl}}}\right)$ is:
(Atomic mass of argon $=40.0 \mathrm{u}$ and molecular mass of chlorine $=70.0 \mathrm{u}$ )
Q10.
mcq single
+4 / 1
An electric heater supplies heat to a system at a rate of 100 W . If the system performs work at a rate of $75 \mathrm{~J} / \mathrm{s}$, then the rate at which internal energy increases will be:
Q11.
mcq single
+4 / 1
A submarine is designed to withstand an absolute pressure of 100 atm . How deep can it go below the water surface?
(Consider the density of water $=1000 \mathrm{~kg} \mathrm{~m}^{-3}$, $1 \mathrm{~atm}=1 \times 10^5 \mathrm{~Pa}$ and gravitational acceleration $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q12.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I }
$$
$$
\text { List I I}
$$
A.
Young's Modulus
I.
$$
\frac{\Delta d}{\Delta L}\left(\frac{L}{d}\right)
$$
B.
Compressibility
II.
$$
\frac{F L}{A(\Delta L)}
$$
C.
Bulk Modulus
III.
$$
-\frac{1}{\Delta P}\left(\frac{\Delta V}{V}\right)
$$
D.
Poisson's Ratio
IV.
$$
-P\left(\frac{V}{\Delta V}\right)
$$
Choose the correct answer from the options given below:
Q13.
mcq single
+4 / 1
Which of the following statements are correct?
A. Inside a conductor, the electrostatic field is zero.
B. Electric field at the surface of a charged conductor does not depend on its surface charge density.
C. The interior of a charged conductor can have no excess charge in the static situation.
D. At the surface of a charged conductor, the electrostatic field must be normal to the surface at every point.
E. The electrostatic potential is zero everywhere inside a charged conductor.
Choose the correct answer from the options given below:
Q14.
mcq single
+4 / 1
The amount of work done to raise a mass ' $m$ ' from the surface of the Earth to a height equal to the radius of the Earth ' $R$ ' will be
Q15.
mcq single
+4 / 1
Consider two uncharged capacitors of equal capacitance 200 pF . One of them is charged by a 100 V supply and disconnected. Now this capacitor is connected to the uncharged capacitor. The amount of electrostatic energy lost in the process is:
Q16.
mcq single
+4 / 1
Five capacitors of capacitances
$C_1=C_2=C_3=C_4=10 \mu \mathrm{~F}$ and $C_5=2.5 \mu \mathrm{~F}$ are connected as shown, along with a battery of 50 V .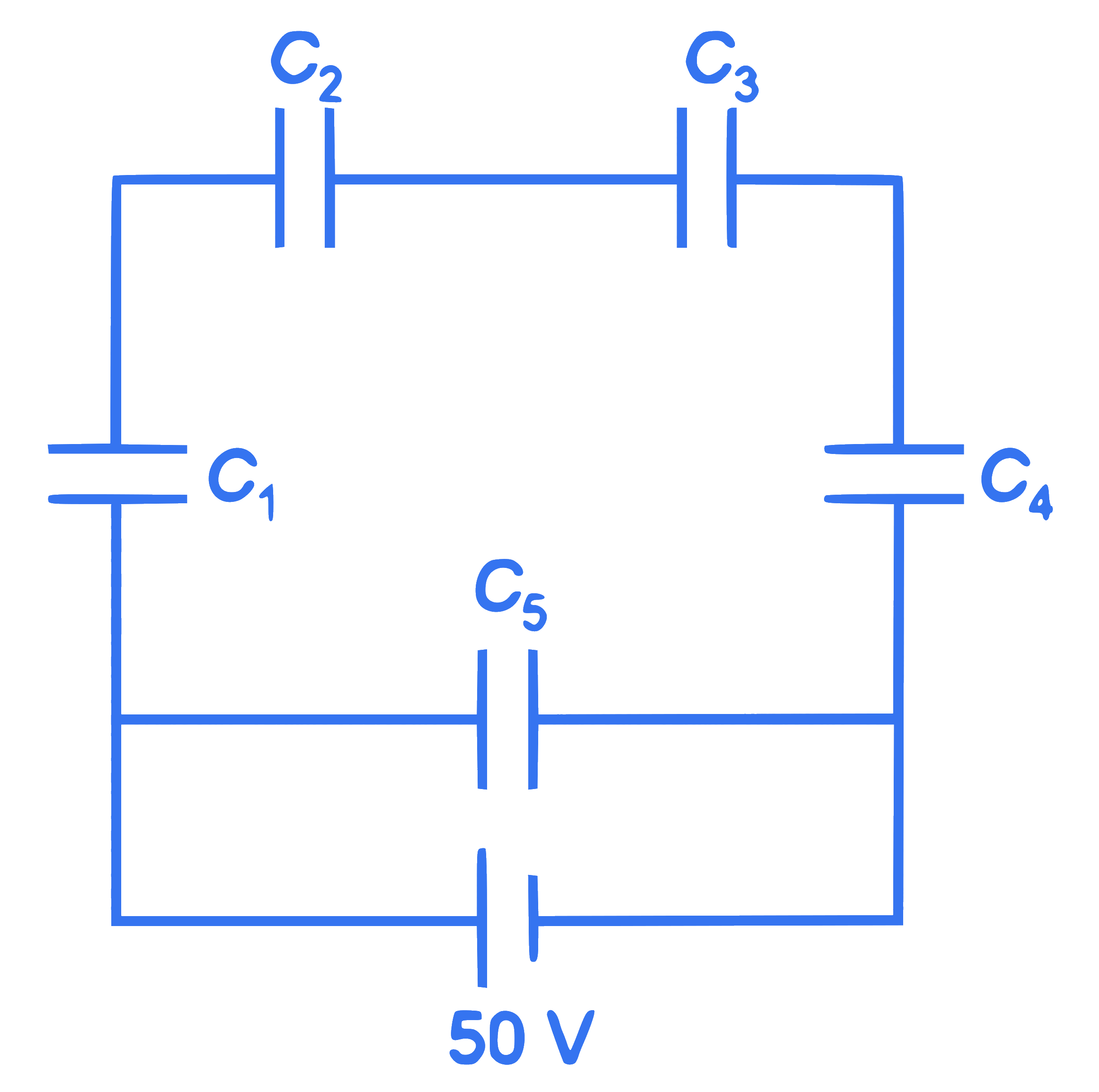 The equivalent capacitance and the charges on each capacitor respectively are:
The equivalent capacitance and the charges on each capacitor respectively are:
 The equivalent capacitance and the charges on each capacitor respectively are:
The equivalent capacitance and the charges on each capacitor respectively are:
Q17.
mcq single
+4 / 1
In a concave lens, a ray of light emanating from the object parallel to the principal axis of the lens after refraction:
Q18.
mcq single
+4 / 1
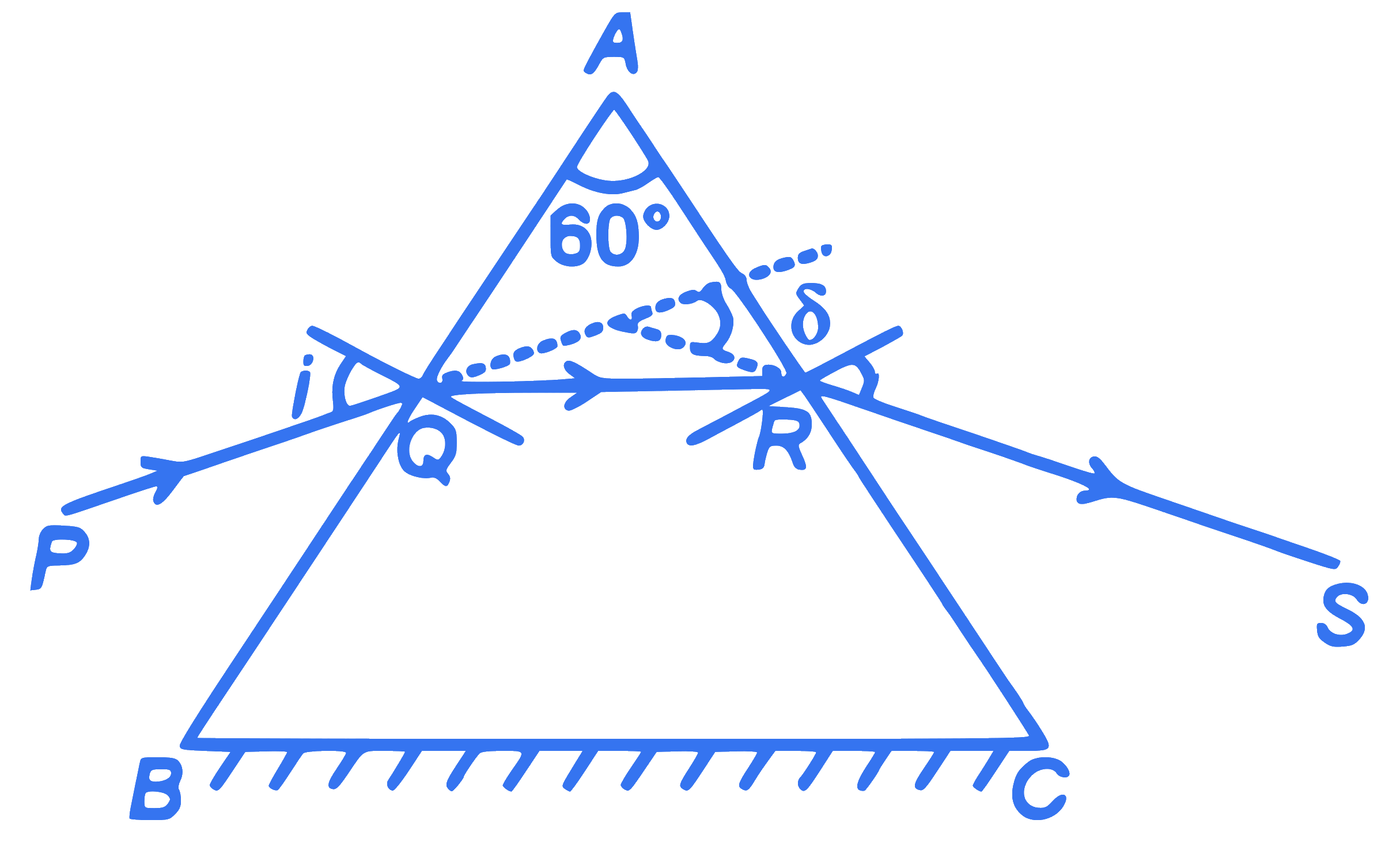
A ray of monochromatic light is passing through an equilateral prism $(A B C)$ as shown in the figure. The refracted ray $(Q R)$ is parallel to its base $(B C)$ and the angle of incidence $(i)$ is $50^{\circ}$. Then the angle of deviation $(\delta)$ is:

Q19.
mcq single
+4 / 1
For a simple pendulum, having time period $T$, the variation of kinetic energy (K.E.) with time $(t)$ is represented by:
Q20.
mcq single
+4 / 1
The sum of kinetic energy and potential energy of a simple pendulum bob is 0.02 joule. The speed of the simple pendulum bob at equilibrium position is approximately:
(Consider mass of the bob $=20 \mathrm{~g}$ )
Q21.
mcq single
+4 / 1
Savitha, a XI standard student, while conducting an experiment to determine the effective length of a simple pendulum $L$, notes down the data of time taken to complete 30 oscillations as 60 s and hence calculates the length of the simple pendulum as: (Take $\pi^2=9.8$, and $g=9.8 \mathrm{~m} / \mathrm{s}^2$ )
Q22.
mcq single
+4 / 1
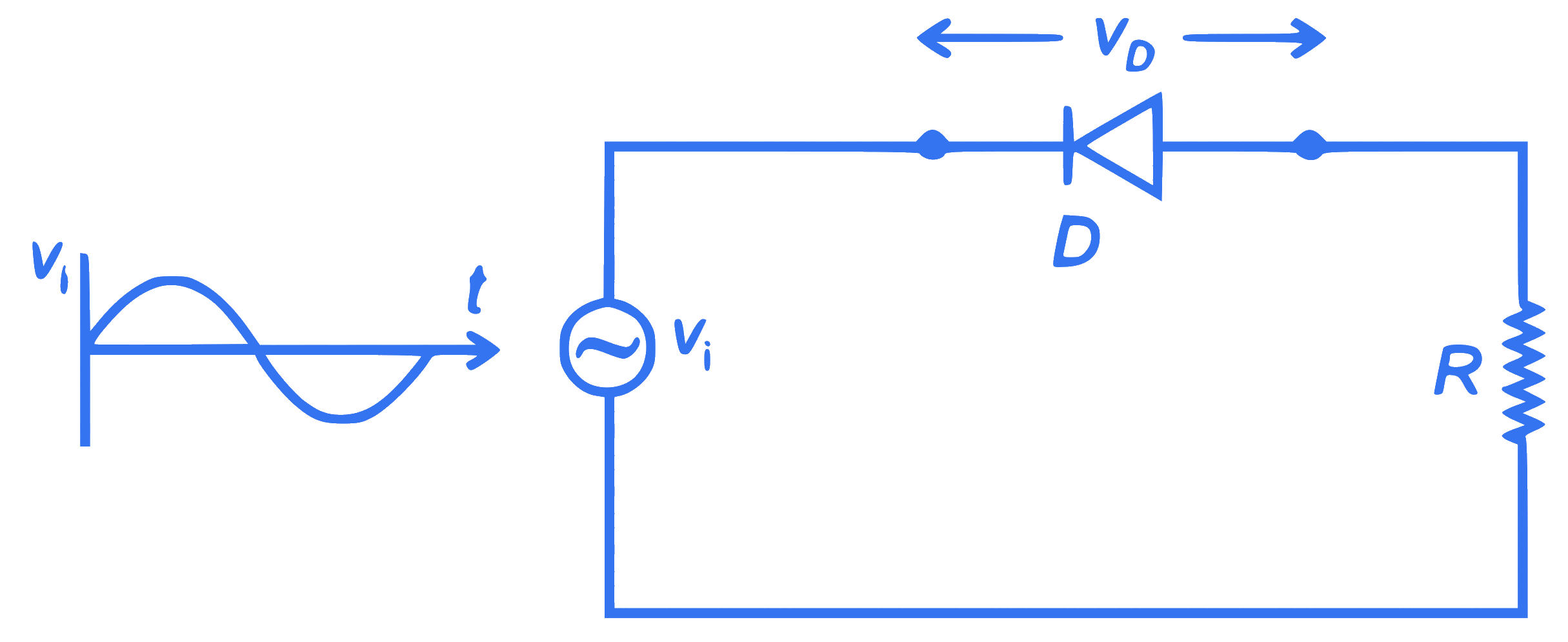
$$ \text { In the circuit shown below, the voltage appearing across the diode } D \text { will be of the form: } $$

Q23.
mcq single
+4 / 1
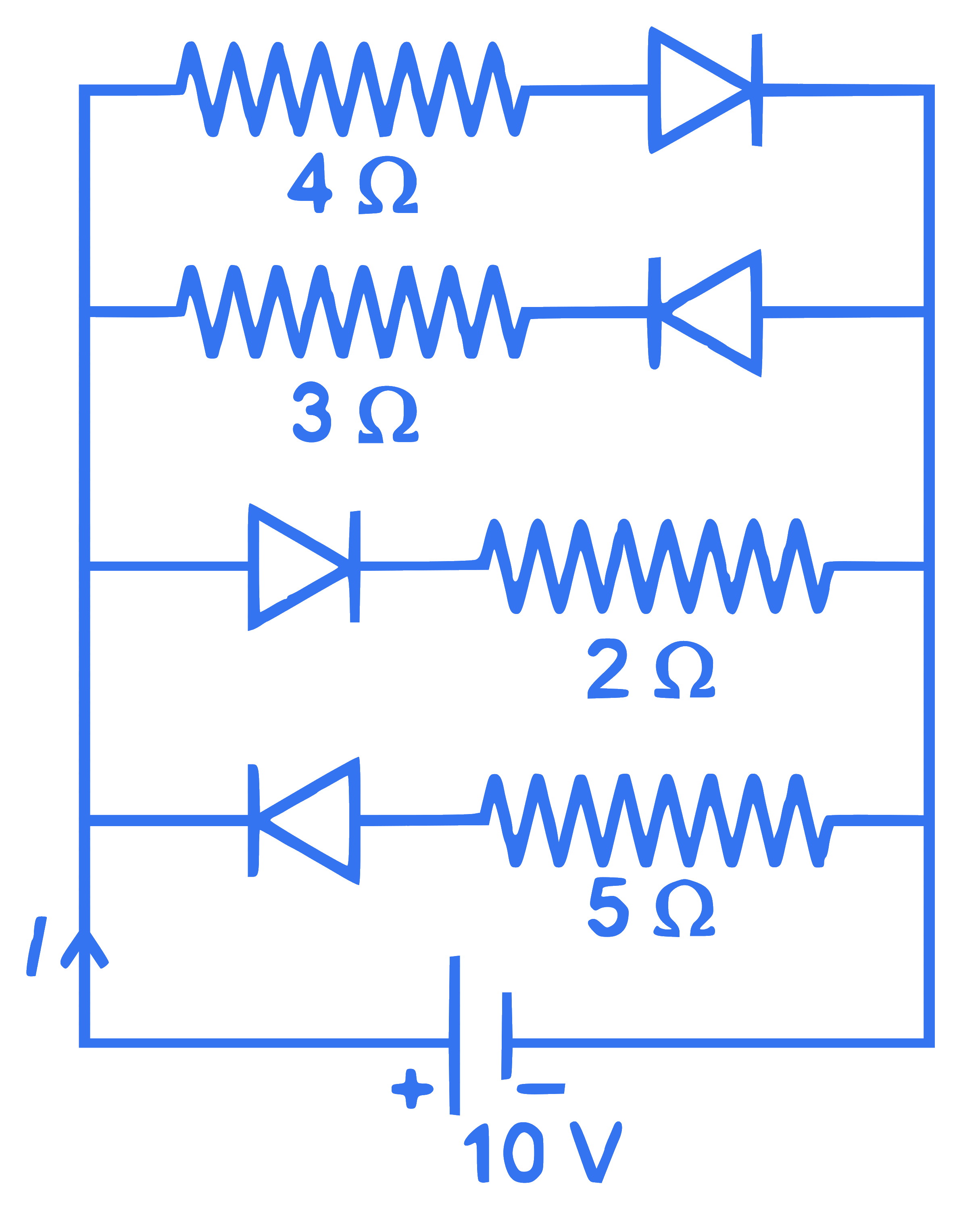
The current / in the circuit shown below is: (All diodes are ideal and identical)

Q24.
mcq single
+4 / 1
Two statements are given below:
A. When the forward bias voltage across a p-n junction diode increases above a certain threshold voltage, the diode current increases significantly.
B. This current is called reverse saturation current.
Choose the correct answer from the options given below:
Q25.
mcq single
+4 / 1
A 100-turn closely wound circular coil of radius 5 cm has a magnetic field of $3.14 \times 10^{-3} \mathrm{~T}$ at its centre. The current flowing through the coil, and the magnitude of the magnetic moment of this coil are, respectively :
(Take $\mu_0=4 \pi \times 10^{-7} \mathrm{~T} \mathrm{~m} / \mathrm{A}$ )
Q26.
mcq single
+4 / 1
The figure given below shows a long straight solid wire of circular cross-section of radius ' $a$ ' carrying steady current $I$. The current $I$ is uniformly distributed across its cross-section. The plot which correctly represents the variation of magnetic field $(B)$ with distance $(r)$ from the axis of the conductor in the region is :


Q27.
mcq single
+4 / 1
$$ \text { Match List I with List II. } $$
$$
\text { List-I }
$$
$$
\text { List-II }
$$
A.
$E=h v$
I.
de Broglie wavelength
B.
Diffraction and Interference
II.
Particle nature of light
C.
$\lambda=h / p$
III.
Wave nature of light
D.
Compton effect
IV.
Energy of photon
Q28.
mcq single
+4 / 1
For a metal of work function 6.6 eV , which of the following wavelengths of incident radiation does not give rise to the photoelectric effect?
(Take Planck's constant as $6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$ )
Q29.
mcq single
+4 / 1
When a ruler falls vertically, 5 different persons catch it with different reaction times. ( $g=9.8 \mathrm{~m} \mathrm{~s}^{-2}$ )
A. Person A has reaction time of 0.20 s .
B. Person $B$ has reaction time of 0.22 s .
C. Person C has reaction time of 0.18 s .
D. Person D has reaction time of 0.19 s .
E. Person E has reaction time of 0.21 s .
What is the correct order of the distance travelled by the ruler for each person?
Q30.
mcq single
+4 / 1
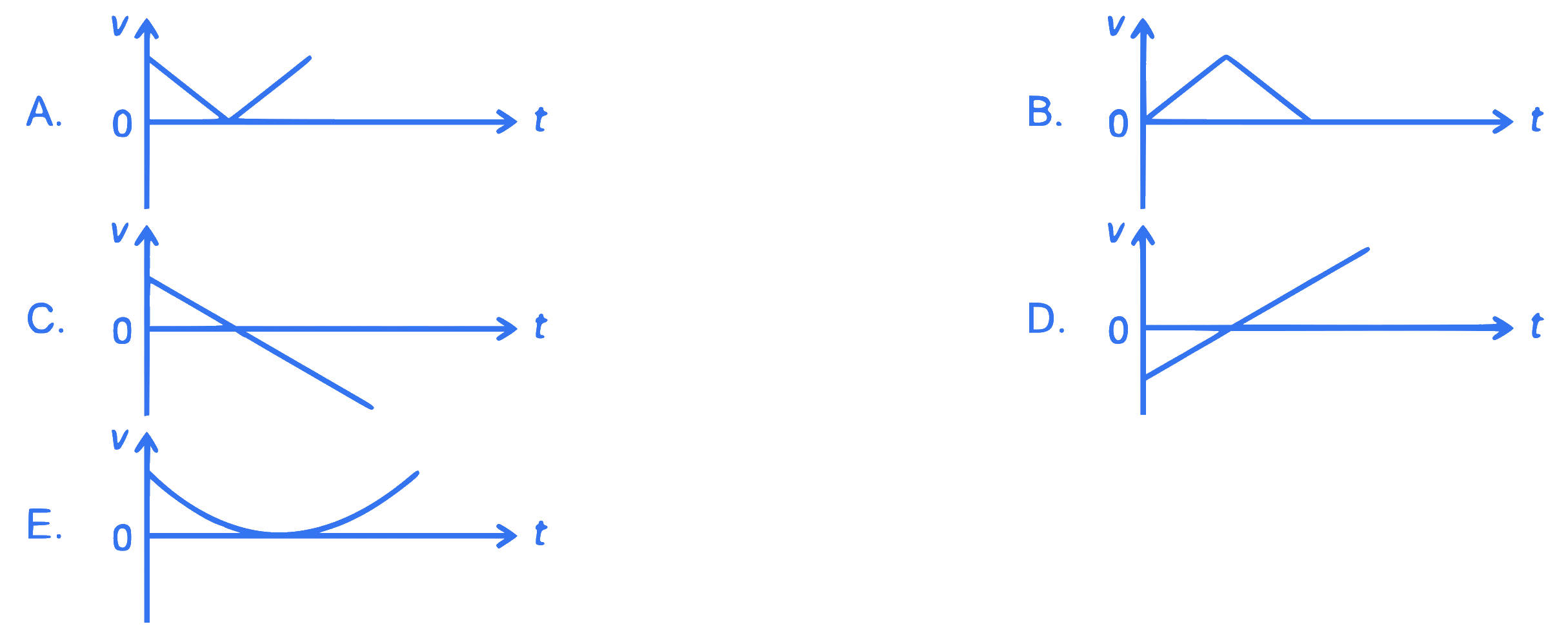
The following plots show variation of velocity $(v)$ with time $(t)$ of a ball thrown vertically upward, and falling back. Which of the following plots is/are correct?

Q31.
mcq single
+4 / 1
In the first excited state of hydrogen atom, the energy of its electron is -3.4 eV . The radial distance of the electron from the hydrogen nucleus in this case is approximately:
(Take $1 \mathrm{eV}=1.6 \times 10^{-19} \mathrm{~J}, \mathrm{e}=1.6 \times 10^{-19} \mathrm{C}$ and $\frac{1}{4 \pi \varepsilon_0}=9 \times 10^9 \mathrm{~N} \mathrm{~m}^2 / \mathrm{C}^2$ )
Q32.
mcq single
+4 / 1
Four statements are given ( $A$ is mass number):
A. The volume of a nucleus is proportional to $A^{1 / 3}$.
B. The volume of a nucleus is proportional to $A$.
C. The difference in mass of an atom and its nucleus is called the mass defect.
D. The difference in mass of a nucleus and its constituents is called the mass defect.
Choose the correct answer from the options given below:
Q33.
mcq single
+4 / 1
An unknown nucleus has a nuclear density of $2.29 \times 10^{17} \mathrm{~kg} / \mathrm{m}^3$ and mass of $19.926 \times 10^{-27} \mathrm{~kg}$. Its mass number $A$ is approximately:
(Take $R_0=1.2 \times 10^{-15} \mathrm{~m}, 4 \pi=12.56$ )
Q34.
mcq single
+4 / 1
For a travelling harmonic wave
$y(x, t)=2.0 \cos 2 \pi(10 t-0.0080 x+0.35)$, where $x$ and $y$ are in cm and $t$ in $s$. The phase difference between oscillatory motion of two points separated by a distance of 0.5 m is:
Q35.
mcq single
+4 / 1
A box of mass 15 kg is kept on the floor of a stationary trolley. The coefficient of static friction between the box and the trolley is 0.12 . Keeping the box in stationary state over the trolley, the maximum acceleration with which the trolley can be moved horizontally in $\mathrm{m} \mathrm{s}^{-2}$ is:
$$ \left(g=10 \mathrm{~m} / \mathrm{s}^2\right) $$
Q36.
mcq single
+4 / 1
The magnitude and direction of the acceleration produced in a body of mass 5 kg when two mutually perpendicular forces 8 N and 6 N act on it, are respectively:
Q37.
mcq single
+4 / 1
The power of a crane, which lifts a mass of 1000 kg to a height of 20 m in 10 s is: $\left(g=9.8 \mathrm{~m} / \mathrm{s}^2\right)$
Q38.
mcq single
+4 / 1
A galvanometer of resistance $100 \Omega$ gives full scale deflection for a current of 1 mA . It is converted into an ammeter of range $0-10 \mathrm{~A}$. The shunt required is:
Q39.
mcq single
+4 / 1
A resistor is connected to a battery of 12 V emf and internal resistance $2 \Omega$. If the current in the circuit is 0.6 A , the terminal voltage of the battery is:
Q40.
mcq single
+4 / 1
In a metre bridge experiment (see figure), the positions of the cell, $E$, and galvanometer, $G$, are interchanged. We shall observe in the galvanometer:
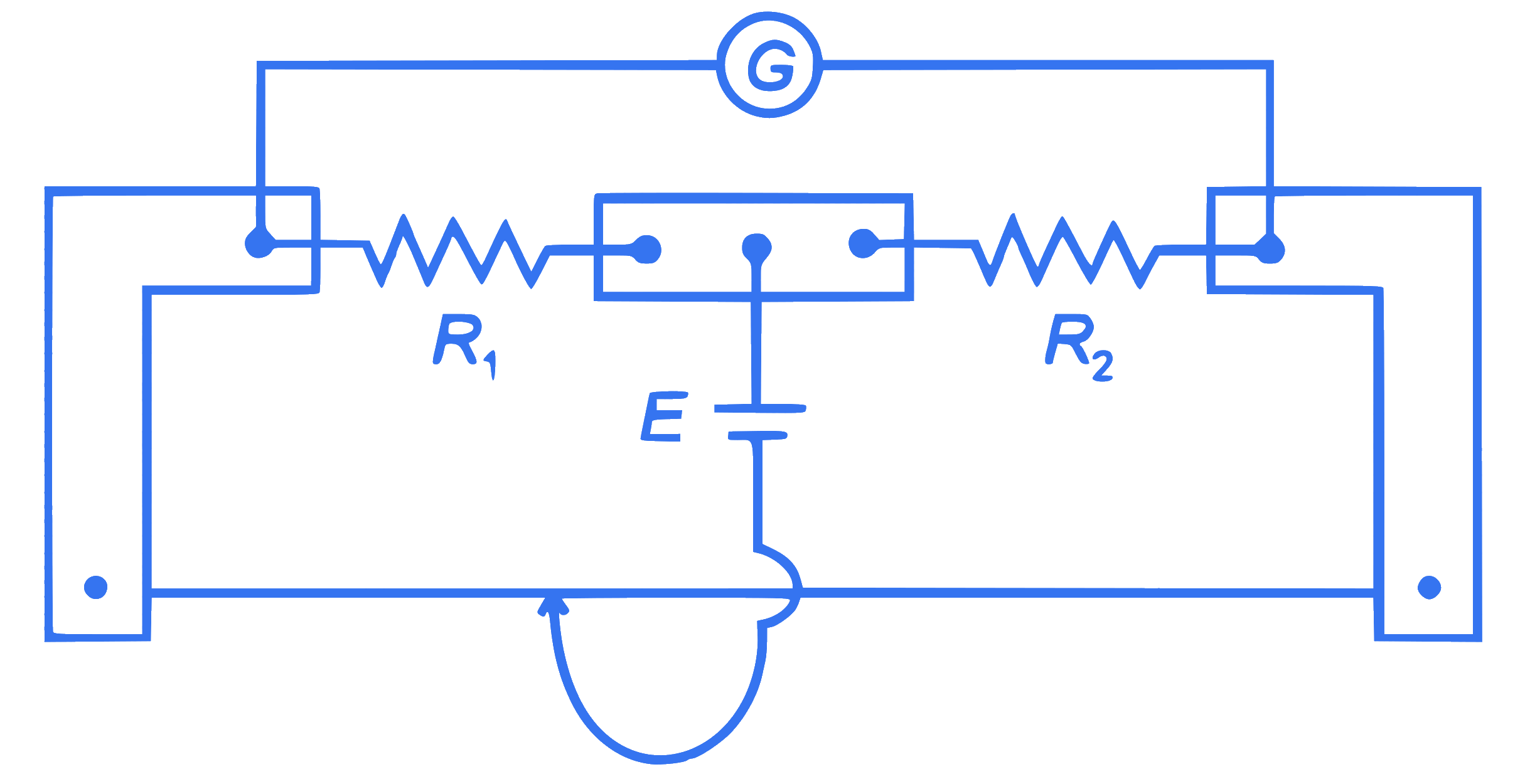

Q41.
mcq single
+4 / 1
A uniform metallic wire having resistance $4 \Omega$ is bent to form a square loop (ABCD) (see figure). A resistance of $2 \Omega$ is connected between points $B$ and $D$ and a battery of 2 V is connected across points $A$ and $C$ as shown in the figure. Now the value of current $(l)$ is :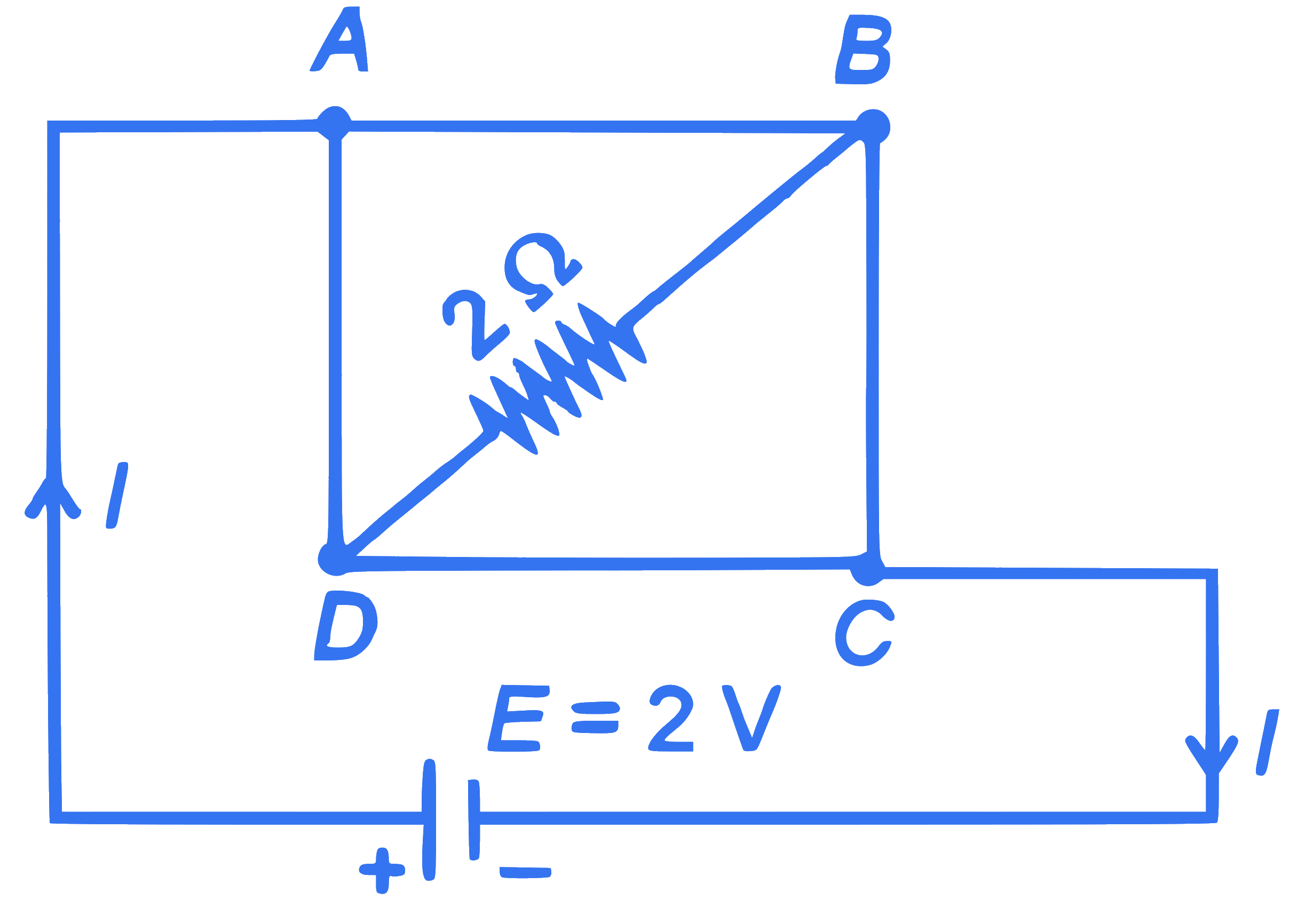

Q42.
mcq single
+4 / 1
A room heater is rated $400 \mathrm{~W}, 220 \mathrm{~V}$. If the supply voltage drops to 200 V , what will be the power consumed (approximately)?
Q43.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
List-I
(Electromagnetic wave)
List-II
(Production)
A.
Microwave
I.
Electrons in atoms emit light when they move from a higher energy level to a lower energy level
B.
Visible light
II.
Radioactive decay of nucleus
C.
Gamma rays
III.
Vibration of atoms and molecules
D.
Infra-red rays
IV.
Klystron valve or magnetron valve
Choose the correct answer from the options given below:
Q44.
mcq single
+4 / 1
The peak value of an alternating current is 5 A and frequency is 60 Hz . How long will the current, starting from zero, take to reach the peak value?
Q45.
mcq single
+4 / 1
An ac circuit contains a resistance of $1 \mathrm{k} \Omega$, a capacitor of $0.1 \mu \mathrm{~F}$ and an inductor of 1 mH connected in series. The resonance frequency of the circuit is approximately:
Q46.
mcq single
+4 / 1
In interference and diffraction, the light energy is redistributed. If it reduces in one region, producing a dark fringe, it increases in another region, producing a bright fringe.
A. As there is no gain or loss of energy, these phenomena are consistent with the principle of conservation of energy.
B. Diffraction and interference are characteristics exhibited only by light waves.
Choose the correct answer from the options given below:
Q47.
mcq single
+4 / 1
In Young's double slit experiment, using monochromatic light of wavelength $\lambda$, the intensity of light at a point on the screen where the path difference is $\lambda$, is $K$ units. The intensity of light at a point where the path difference is $\frac{\lambda}{3}$ will be
Q48.
mcq single
+4 / 1
The angular speed of a flywheel is increased from 600 rpm to 1200 rpm in 10 s . The number of revolutions completed by the flywheel during this time is:
Q49.
mcq single
+4 / 1
A thin wire of length ' $L$ ' and linear mass density ' $m$ ' is bent into a circular ring (in $x-y$ plane) with centre ' $C$ ' as shown in figure. The moment of inertia of the ring about an axis $y y^{\prime}$ will be:


Q50.
mcq single
+4 / 1
A rectangular wire loop of sides 8 cm and 3 cm with a small cut, is moving out of a region of uniform magnetic field of magnitude 0.3 T directed normal to the plane of the loop. The emf developed across the cut, if the velocity of the loop is $2 \mathrm{~cm} \mathrm{~s}^{-1}$, in a direction normal to the shorter side of the loop, will be :
Q51.
mcq single
+4 / 1
In a vernier calliper, 20 VSD coincide with 16 MSD (each division of length 1 mm ). The least count of the vernier callipers is:
Q52.
mcq single
+4 / 1
Each side of a metallic cube of mass 5.580 kg is measured to the 9.0 cm . Keeping the significant figures in view, the density of the material of the cube can be best expressed as $X \times 10^3 \mathrm{~kg} \mathrm{~m}^{-3}$ where the value of $X$ is:
Q53.
mcq single
+4 / 1
The speed of light in vacuum is taken as unity. If light takes 6 min 40 s to reach the Earth from the Sun, the distance between the Sun and the Earth in new unit is:
Q54.
mcq single
+4 / 1
A flask contains argon and chlorine in the ratio of $2: 1$ by mass. The temperature of the mixture is $27^{\circ} \mathrm{C}$. The ratio of root mean square speed of the molecules of the two gases $\left(\frac{V_{\mathrm{rms}}^{\mathrm{Ar}}}{V_{\mathrm{rms}}^{\mathrm{Cl}}}\right)$ is:
(Atomic mass of argon $=40.0 \mathrm{u}$ and molecular mass of chlorine $=70.0 \mathrm{u}$ )
Q55.
mcq single
+4 / 1
An electric heater supplies heat to a system at a rate of 100 W . If the system performs work at a rate of $75 \mathrm{~J} / \mathrm{s}$, then the rate at which internal energy increases will be:
Q56.
mcq single
+4 / 1
A submarine is designed to withstand an absolute pressure of 100 atm . How deep can it go below the water surface?
(Consider the density of water $=1000 \mathrm{~kg} \mathrm{~m}^{-3}$, $1 \mathrm{~atm}=1 \times 10^5 \mathrm{~Pa}$ and gravitational acceleration $g=10 \mathrm{~m} / \mathrm{s}^2$ )
Q57.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I }
$$
$$
\text { List I I}
$$
A.
Young's Modulus
I.
$$
\frac{\Delta d}{\Delta L}\left(\frac{L}{d}\right)
$$
B.
Compressibility
II.
$$
\frac{F L}{A(\Delta L)}
$$
C.
Bulk Modulus
III.
$$
-\frac{1}{\Delta P}\left(\frac{\Delta V}{V}\right)
$$
D.
Poisson's Ratio
IV.
$$
-P\left(\frac{V}{\Delta V}\right)
$$
Choose the correct answer from the options given below:
Q58.
mcq single
+4 / 1
Which of the following statements are correct?
A. Inside a conductor, the electrostatic field is zero.
B. Electric field at the surface of a charged conductor does not depend on its surface charge density.
C. The interior of a charged conductor can have no excess charge in the static situation.
D. At the surface of a charged conductor, the electrostatic field must be normal to the surface at every point.
E. The electrostatic potential is zero everywhere inside a charged conductor.
Choose the correct answer from the options given below:
Q59.
mcq single
+4 / 1
The amount of work done to raise a mass ' $m$ ' from the surface of the Earth to a height equal to the radius of the Earth ' $R$ ' will be
Q60.
mcq single
+4 / 1
Consider two uncharged capacitors of equal capacitance 200 pF . One of them is charged by a 100 V supply and disconnected. Now this capacitor is connected to the uncharged capacitor. The amount of electrostatic energy lost in the process is:
Q61.
mcq single
+4 / 1
Five capacitors of capacitances
$C_1=C_2=C_3=C_4=10 \mu \mathrm{~F}$ and $C_5=2.5 \mu \mathrm{~F}$ are connected as shown, along with a battery of 50 V . The equivalent capacitance and the charges on each capacitor respectively are:
The equivalent capacitance and the charges on each capacitor respectively are:
 The equivalent capacitance and the charges on each capacitor respectively are:
The equivalent capacitance and the charges on each capacitor respectively are:
Q62.
mcq single
+4 / 1
In a concave lens, a ray of light emanating from the object parallel to the principal axis of the lens after refraction:
Q63.
mcq single
+4 / 1
A ray of monochromatic light is passing through an equilateral prism $(A B C)$ as shown in the figure. The refracted ray $(Q R)$ is parallel to its base $(B C)$ and the angle of incidence $(i)$ is $50^{\circ}$. Then the angle of deviation $(\delta)$ is:


Q64.
mcq single
+4 / 1
For a simple pendulum, having time period $T$, the variation of kinetic energy (K.E.) with time $(t)$ is represented by:
Q65.
mcq single
+4 / 1
The sum of kinetic energy and potential energy of a simple pendulum bob is 0.02 joule. The speed of the simple pendulum bob at equilibrium position is approximately:
(Consider mass of the bob $=20 \mathrm{~g}$ )
Q66.
mcq single
+4 / 1
Savitha, a XI standard student, while conducting an experiment to determine the effective length of a simple pendulum $L$, notes down the data of time taken to complete 30 oscillations as 60 s and hence calculates the length of the simple pendulum as: (Take $\pi^2=9.8$, and $g=9.8 \mathrm{~m} / \mathrm{s}^2$ )
Q67.
mcq single
+4 / 1
$$ \text { In the circuit shown below, the voltage appearing across the diode } D \text { will be of the form: } $$


Q68.
mcq single
+4 / 1
The current / in the circuit shown below is: (All diodes are ideal and identical)


Q69.
mcq single
+4 / 1
Two statements are given below:
A. When the forward bias voltage across a p-n junction diode increases above a certain threshold voltage, the diode current increases significantly.
B. This current is called reverse saturation current.
Choose the correct answer from the options given below:
Q70.
mcq single
+4 / 1
A 100-turn closely wound circular coil of radius 5 cm has a magnetic field of $3.14 \times 10^{-3} \mathrm{~T}$ at its centre. The current flowing through the coil, and the magnitude of the magnetic moment of this coil are, respectively :
(Take $\mu_0=4 \pi \times 10^{-7} \mathrm{~T} \mathrm{~m} / \mathrm{A}$ )
Q71.
mcq single
+4 / 1
The figure given below shows a long straight solid wire of circular cross-section of radius ' $a$ ' carrying steady current $I$. The current $I$ is uniformly distributed across its cross-section. The plot which correctly represents the variation of magnetic field $(B)$ with distance $(r)$ from the axis of the conductor in the region is :


Q72.
mcq single
+4 / 1
$$ \text { Match List I with List II. } $$
$$
\text { List-I }
$$
$$
\text { List-II }
$$
A.
$E=h v$
I.
de Broglie wavelength
B.
Diffraction and Interference
II.
Particle nature of light
C.
$\lambda=h / p$
III.
Wave nature of light
D.
Compton effect
IV.
Energy of photon
Q73.
mcq single
+4 / 1
For a metal of work function 6.6 eV , which of the following wavelengths of incident radiation does not give rise to the photoelectric effect?
(Take Planck's constant as $6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$ )
Q74.
mcq single
+4 / 1
When a ruler falls vertically, 5 different persons catch it with different reaction times. ( $g=9.8 \mathrm{~m} \mathrm{~s}^{-2}$ )
A. Person A has reaction time of 0.20 s .
B. Person $B$ has reaction time of 0.22 s .
C. Person C has reaction time of 0.18 s .
D. Person D has reaction time of 0.19 s .
E. Person E has reaction time of 0.21 s .
What is the correct order of the distance travelled by the ruler for each person?
Q75.
mcq single
+4 / 1
The following plots show variation of velocity $(v)$ with time $(t)$ of a ball thrown vertically upward, and falling back. Which of the following plots is/are correct?


Q76.
mcq single
+4 / 1
In the first excited state of hydrogen atom, the energy of its electron is -3.4 eV . The radial distance of the electron from the hydrogen nucleus in this case is approximately:
(Take $1 \mathrm{eV}=1.6 \times 10^{-19} \mathrm{~J}, \mathrm{e}=1.6 \times 10^{-19} \mathrm{C}$ and $\frac{1}{4 \pi \varepsilon_0}=9 \times 10^9 \mathrm{~N} \mathrm{~m}^2 / \mathrm{C}^2$ )
Q77.
mcq single
+4 / 1
Four statements are given ( $A$ is mass number):
A. The volume of a nucleus is proportional to $A^{1 / 3}$.
B. The volume of a nucleus is proportional to $A$.
C. The difference in mass of an atom and its nucleus is called the mass defect.
D. The difference in mass of a nucleus and its constituents is called the mass defect.
Choose the correct answer from the options given below:
Q78.
mcq single
+4 / 1
An unknown nucleus has a nuclear density of $2.29 \times 10^{17} \mathrm{~kg} / \mathrm{m}^3$ and mass of $19.926 \times 10^{-27} \mathrm{~kg}$. Its mass number $A$ is approximately:
(Take $R_0=1.2 \times 10^{-15} \mathrm{~m}, 4 \pi=12.56$ )
Q79.
mcq single
+4 / 1
For a travelling harmonic wave
$y(x, t)=2.0 \cos 2 \pi(10 t-0.0080 x+0.35)$, where $x$ and $y$ are in cm and $t$ in $s$. The phase difference between oscillatory motion of two points separated by a distance of 0.5 m is:
Q80.
mcq single
+4 / 1
A box of mass 15 kg is kept on the floor of a stationary trolley. The coefficient of static friction between the box and the trolley is 0.12 . Keeping the box in stationary state over the trolley, the maximum acceleration with which the trolley can be moved horizontally in $\mathrm{m} \mathrm{s}^{-2}$ is:
$$ \left(g=10 \mathrm{~m} / \mathrm{s}^2\right) $$
Q81.
mcq single
+4 / 1
The magnitude and direction of the acceleration produced in a body of mass 5 kg when two mutually perpendicular forces 8 N and 6 N act on it, are respectively:
Q82.
mcq single
+4 / 1
The power of a crane, which lifts a mass of 1000 kg to a height of 20 m in 10 s is: $\left(g=9.8 \mathrm{~m} / \mathrm{s}^2\right)$
Q83.
mcq single
+4 / 1
A galvanometer of resistance $100 \Omega$ gives full scale deflection for a current of 1 mA . It is converted into an ammeter of range $0-10 \mathrm{~A}$. The shunt required is:
Q84.
mcq single
+4 / 1
A resistor is connected to a battery of 12 V emf and internal resistance $2 \Omega$. If the current in the circuit is 0.6 A , the terminal voltage of the battery is:
Q85.
mcq single
+4 / 1
In a metre bridge experiment (see figure), the positions of the cell, $E$, and galvanometer, $G$, are interchanged. We shall observe in the galvanometer:


Q86.
mcq single
+4 / 1
A uniform metallic wire having resistance $4 \Omega$ is bent to form a square loop (ABCD) (see figure). A resistance of $2 \Omega$ is connected between points $B$ and $D$ and a battery of 2 V is connected across points $A$ and $C$ as shown in the figure. Now the value of current $(l)$ is :

Q87.
mcq single
+4 / 1
A room heater is rated $400 \mathrm{~W}, 220 \mathrm{~V}$. If the supply voltage drops to 200 V , what will be the power consumed (approximately)?
Q88.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
List-I
(Electromagnetic wave)
List-II
(Production)
A.
Microwave
I.
Electrons in atoms emit light when they move from a higher energy level to a lower energy level
B.
Visible light
II.
Radioactive decay of nucleus
C.
Gamma rays
III.
Vibration of atoms and molecules
D.
Infra-red rays
IV.
Klystron valve or magnetron valve
Choose the correct answer from the options given below:
Q89.
mcq single
+4 / 1
The peak value of an alternating current is 5 A and frequency is 60 Hz . How long will the current, starting from zero, take to reach the peak value?
Q90.
mcq single
+4 / 1
An ac circuit contains a resistance of $1 \mathrm{k} \Omega$, a capacitor of $0.1 \mu \mathrm{~F}$ and an inductor of 1 mH connected in series. The resonance frequency of the circuit is approximately:
Biology
Biology
Q1.
mcq single
+4 / 1
What is the reason behind production of large holes in 'Swiss Cheese'?
Q2.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I (Bioactive molecules) }
$$
$$
\text { List II (Importance) }
$$
A.
Streptokinase
I.
Immunosuppressive agent
B.
Statins
II.
Removal of clots from the blood vessels
C.
Lipases
III.
Blood cholesterol-lowering agent
D.
Cyclosporin A
IV.
Detergent formulations
Choose the correct answer from the options given below:
Q3.
mcq single
+4 / 1
Ecological pyramids represent the relationship between the organisms at different trophic levels and they are generally inverted for:
Q4.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
$$
\text { List-I }
$$
$$
\text { List-II }
$$
A.
Productivity
I.
Gross primary productivity minus respiration losses
B.
Net primary productivity
II.
Rate of formation of new organic matter by consumers
C.
Gross primary productivity
III.
Rate of biomass production
D.
Secondary productivity
IV.
Rate of production of organic matter during photosynthesis
Choose the correct answer from the options given below :
Q5.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
Decomposition
I.
Accumulation of dark coloured amorphous colloidal substance
B.
Detritus
II.
Release of inorganic nutrients by the activity of microbes in soil
C.
Mineralisation
III.
Breaking down of complex organic matter into inorganic substances.
D.
Humification
IV.
Dead remains of plants and animals including fecal matter
Choose the correct answer from the options given below:
Q6.
mcq single
+4 / 1
What is the probability of having children with 'O' blood group, where both mother and father are heterozygous for ' $\underline{\mathrm{A}}$ ' and ' $\underline{\mathrm{B}}$ ' blood group, respectively?
Q7.
mcq single
+4 / 1
In a population of a grasshopper species, the chromosome number of some members is 23 and some other members possess 24 chromosomes. The 23 and 24 chromosome-bearing members in this species are $\_\_\_\_$ .
Q8.
mcq single
+4 / 1
Which of the following statements are true with reference to the sex-determination in honeybees?
A. An offspring formed from the union of a sperm and an egg, develops as a female (queen or worker).
B. An unfertilized egg develops as a male by parthenogenesis.
C. A male has half the number of chromosomes than that of a female.
D. Males produce sperms by meiosis.
E. Honeybees have a haplodiploid sex-determination system.
Choose the correct answer from the options given below :
Q9.
mcq single
+4 / 1
Which one of the following disorders is caused by the substitution of Glutamic acid (Glu) by Valine (Val) at the sixth position of the beta globin chain of the haemoglobin molecule?
Q10.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
Incomplete dominance
I.
Human skin colour
B.
Co-dominance
II.
Inheritance of flower colour in Antirrhinum sp.
C.
Pleiotropy
III.
Phenylketonuria disease in humans
D.
Polygenic inheritance
IV.
ABO blood groups
Choose the correct answer from the options given below :
Q11.
mcq single
+4 / 1
Male frogs can be distinguished from female frogs due to the presence of
A. Bulging eyes
B. Vocal sacs
C. Webbed digits in feet
D. Copulatory pad on first digit of fore limbs
E. Olive green-coloured skin with dark irregular spots
Choose the correct answer from the options given below
Q12.
mcq single
+4 / 1
Choose the correct statements regarding frog's anatomy:
A. Hepatic portal system is the special venous connection between liver and intestine.
B. There are twelve pairs of cranial nerves arising from the brain.
C. The ureters and oviducts open separately into the cloaca in female frogs.
D. Hind-brain consists of cerebellum, medulla oblongata and optic lobes.
E. Sinus venosus joins the right atrium of heart.
Choose the correct answer from the options given below:
Q13.
mcq single
+4 / 1
The enzyme required for carboxylation in the Calvin cycle is
Q14.
mcq single
+4 / 1
How many ATP and NADPH molecules are required to make one molecule of glucose through the Calvin pathway?
Q15.
mcq single
+4 / 1
Find the incorrect statement(s) about photosynthesis from the following:
A. The water splitting complex is associated with PSI.
B. $\mathrm{C}_4$ plants use the $\mathrm{C}_3$ pathway of $\mathrm{CO}_2$ fixation as the main biosynthetic pathway.
C. In $\mathrm{C}_4$ plants, photorespiration does not occur.
D. $\mathrm{C}_3$ plants exhibit 'Kranz' anatomy.
E. ATP synthesis in chloroplast occurs through chemiosmosis.
Choose the answer from the options given below:
Q16.
mcq single
+4 / 1
Select the set of fishes which belong to the class Osteichthyes:
Q17.
mcq single
+4 / 1
Select the incorrect statements from the following:
A. Digestive system in Platyhelminthes is incomplete.
B. Bilateral symmetry is a characteristic feature of adult Echinoderms.
C. Pseudocoelom is possessed by Aschelminthes.
D. Notochord is persistent throughout life in the class Chondrichthyes.
E. Members of class Reptilia maintain a constant body temperature.
Choose the answer from the options given below:
Q18.
mcq single
+4 / 1
The flightless bird with forelimbs modified as paddle-like structures suited for swimming is known as:
Q19.
mcq single
+4 / 1
A group of researchers procured some fish like animals and upon investigation the following characters were observed:
A. Endoskeleton was made of cartilage.
B. Ectoparasitic; as they were found attached on fish skin with their circular sucking mouth.
C. Paired fins and scales were absent, but 7 pairs of gill slits were present.
Which of the following species of animals did they consider to fit best with these characters?
Q20.
mcq single
+4 / 1
In which one of the following, the ovules are not enclosed by an ovary wall and remain exposed?
Q21.
mcq single
+4 / 1
$$ \text { Match List-I with List-II. } $$
List-I
(Respiratory Volume)
List-II
(Capacity in mL )
A.
ERV (Expiratory Reserve Volume)
I.
$2500-3000 \mathrm{~mL}$
B.
RV (Residual Volume)
II.
$500 \,\,mL$
C.
IRV (Inspiratory Reserve Volume)
III.
$1000-1100 \mathrm{~mL}$
D.
TV (Tidal Volume)
IV.
$1100-1200 \mathrm{~mL}$
Choose the correct answer from the options given below :
Q22.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Molluscs
I.
Pulmonary respiration only
B.
Reptiles
II.
Branchial respiration
C.
Adult amphibians
III.
Cellular respiration
D.
Amoeba
IV.
Pulmonary and cutaneous respiration
Choose the correct answer from the options given below:
Q23.
mcq single
+4 / 1
In humans, respiration occurs in the following steps. Arrange these steps in the correct order.
A. Diffusion of $\mathrm{O}_2$ and $\mathrm{CO}_2$ between blood and tissues
B. Diffusion of $\mathrm{O}_2$ and $\mathrm{CO}_2$ across alveolar membrane
C. Pulmonary ventilation by which atmospheric air is drawn in and $\mathrm{CO}_2$ rich alveolar air is released out
D. Cellular respiration
E. Transport of gases by the blood
Choose the correct answer from the options given below
Q24.
mcq single
+4 / 1
Which one of the following statements is not true about the universal rules of binomial nomenclature?
Q25.
mcq single
+4 / 1
The toxin proteins isolated from Bacillus thuringiensis, coded by which of the following genes would contrl cotton bollworms and corn borer, respectively?
Q26.
mcq single
+4 / 1
The human protein named $\boldsymbol{\alpha}$-1-antitrypsin, obtained from transgenic animals, is used for the treatment of $\_\_\_\_$ .
Q27.
mcq single
+4 / 1
The specific receptors for neurotransmitters in a synapse are present on $\_\_\_\_$ .
Q28.
mcq single
+4 / 1
$$ \text { Match List-I with List-II : } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
Conjunctive tissue
I.
Specialised cells in the vicinity of guard cells
B.
Casparian strips
II.
Endodermal cells rich in starch
C.
Subsidiary cells
III.
Tissue between xylem and phloem
D.
Starch sheath
IV.
Endodermal cells with suberin deposition
Choose the correct answer from the options given below :
Q29.
mcq single
+4 / 1
The main function of bulliform cells in grasses is :
Q30.
mcq single
+4 / 1
Non-membrane bound cell organelles found in both prokaryotic and eukaryotic cells are $\_\_\_\_$ .
Q31.
mcq single
+4 / 1
Choose the correct statements regarding cell organelles and their inclusions.
A. The endomembrane system includes Golgi complex, endoplasmic reticulum and mitochondria.
B. Rough endoplasmic reticulum bears ribosomes on its surface.
C. Both mitochondria and plastids have circular DNA.
D. A network of microtubules, microfilaments and intermediate filaments present in the cytoplasm is called cytoskeleton.
E. Mitochondrion is a single membrane-bound structure.
Choose the correct answer from the options given below :
Q32.
mcq single
+4 / 1
Select the correct statements regarding cell membrane in eukaryotic cell.
A. Membrane of human RBCs has approximately $52 \%$ protein.
B. Major phospholipids are arranged in a bilayer.
C. Extensions of the plasma membrane into the cell form mesosomes.
D. Tails towards the inner part of lipids are hydrophobic and thus protected from aqueous medium.
E. Glycocalyx is present on the outer surface of the plasma membrane.
Choose the correct answer from the options given below:
Q33.
mcq single
+4 / 1
Which one of the following is the site for active ribosomal RNA synthesis?
Q34.
mcq single
+4 / 1
Which of the following statements are correct with reference to a transcription unit?
A. A transcription unit in DNA is defined primarily by three regions : promoter, structural gene and terminator.
B. The promoter is said to be located towards the 5'-end of the structural gene.
C. The promoter is a DNA sequence that provides binding site for RNA polymerase.
D. The promoter defines the template and coding strands.
E. The terminator is located towards the $3^{\prime}$-end of the coding strand and it defines the end of the process of transcription.
Choose the correct answer from the options given below:
Q35.
mcq single
+4 / 1
In the lac operon, the $\boldsymbol{z}$ gene codes for
Q36.
mcq single
+4 / 1
Which of the following statements are correct with reference to packaging of DNA helix ?
A. Histones are organized to form a unit of eight molecules called histone octamer.
B. Histones are negatively charged basic proteins.
C. Histones are rich in the basic amino acid residues - lysine and arginine.
D. The positively charged DNA is wrapped around the histone octamer to form nucleosome.
E. The packaging of chromatin at higher levels requires an additional set of proteins called non-histone chromosomal proteins.
Choose the correct answer from the options given below :
Q37.
mcq single
+4 / 1
The sixth mutant codon of beta globin gene causing polymerization of Haemoglobin and change in RBC shape is $\_\_\_\_$
Q38.
mcq single
+4 / 1
Arrange the following steps of DNA fingerprinting in a correct sequence.
A. Isolation of DNA and its digestion by restriction endonucleases.
B. Hybridisation using a labelled VNTR probe.
C. Transferring of separated DNA fragments to synthetic membranes.
D. Detection of hybridised DNA fragments by autoradiography.
E. Separation of DNA fragments by electrophoresis.
Choose the correct answer from the options given below :
Q39.
mcq single
+4 / 1
The following are the stages of life cycle of Plasmodium. Arrange the stages in the proper order.
A. The parasites reproduce asexually in RBCs, bursting the cells.
B. The parasites reproduce asexually in liver cells, bursting the cells and releasing into blood.
C. Gametocytes develop in RBCs.
D. Sporozoites reach the liver through the blood.
E. Female mosquito injects sporozoites into humans during bite.
Choose the correct answer from the options given below:
Q40.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I (Drug) }
$$
$$
\text { List II (Effect) }
$$
A.
Nicotine
I.
Causes sense of euphoria and increased energy
B.
Morphine
II.
Stimulates adrenal gland to release catecholamines into blood circulation
C.
Heroin
III.
Effective sedative and painkiller
D.
Cocaine
IV.
A depressant; slows down body function
Choose the correct answer from the options given below:
Q41.
mcq single
+4 / 1
In which animal do haploid cells divide mitotically to produce gametes?
Q42.
mcq single
+4 / 1
Which one of the following is an appropriate example of sexual deceit?
Q43.
mcq single
+4 / 1
Choose the correct statements regarding population interactions between two species.
A. In both parasitism and commensalism, only one species benefits and the other species is harmed.
B. Both species benefit in mutualism.
C. Both species benefit in commensalism.
D. In parasitism, only one species benefits and the other species is harmed.
E. In amensalism, one species is harmed and the other is unaffected.
Choose the correct answer from the options given below:
Q44.
mcq single
+4 / 1
Which of the following equations depicts Verhulst-Pearl logistic population growth?
Q45.
mcq single
+4 / 1
The WBC count of a person's blood sample is $8000 / \mathrm{cu} \mathrm{mm}$. How many eosinophils and lymphocytes would be in the same blood sample approximately?
Q46.
mcq single
+4 / 1
Select the incorrect statement with reference to Rh grouping.
A. Erythroblastosis foetalis is a condition observed having foetus with $\mathrm{Rh}^{-\mathrm{ve}}$ blood and mother with $\mathrm{Rh}^{+\mathrm{ve}}$ blood.
B. Rh antigen is observed on RBCs in the majority of human beings.
C. Before blood transfusion, Rh group should also be matched.
D. Rh incompatibility is observed when a pregnant mother is $\mathrm{Rh}^{-\mathrm{ve}}$ and the foetus is $\mathrm{Rh}^{+\mathrm{ve}}$.
E. Erythroblastosis foetalis can be avoided by administering anti-Rh antibodies to the mother immediately after the delivery of the second child.
Choose the answer from the options given below :
(1) C and D only
(2) A and B only
(3) A and E only
(4) B and C onlySelect the incorrect statement with reference to Rh grouping.
A. Erythroblastosis foetalis is a condition observed having foetus with $\mathrm{Rh}^{-\mathrm{ve}}$ blood and mother with $\mathrm{Rh}^{+\mathrm{ve}}$ blood.
B. Rh antigen is observed on RBCs in the majority of human beings.
C. Before blood transfusion, Rh group should also be matched.
D. Rh incompatibility is observed when a pregnant mother is $\mathrm{Rh}^{-\mathrm{ve}}$ and the foetus is $\mathrm{Rh}^{+\mathrm{ve}}$.
E. Erythroblastosis foetalis can be avoided by administering anti-Rh antibodies to the mother immediately after the delivery of the second child.
Choose the answer from the options given below :
Q47.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
$\mathrm{G}_1$ phase
I.
Actual cell division occurs
B.
S phase
II.
Cell is metabolically active and continuously grows but does not replicate its DNA
C.
$\mathrm{G}_2$ Phase
III.
Synthesis of DNA occurs and the amount of DNA per cell doubles
D.
M phase
IV.
Proteins are synthesized while cell growth continues
Choose the correct answer from the options given below :
Q48.
mcq single
+4 / 1
$$ \text { Match List I with List II related to embryonic development at various months of pregnancy: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
The foetus movement starts and hair appears on the head
I.
24 weeks of pregnancy
B.
The foetus develops limbs and digits
II.
20 weeks of pregnancy
C.
The foetus develops external genital organs
III.
8 weeks of pregnancy
D.
The foetus body is covered with fine hair; eyelids separate and eyelashes are formed
IV.
12 weeks of pregnancy
Choose the correct answer from the options given below:
Q49.
mcq single
+4 / 1
Spermatogonia undergo a series of cell divisions statements to produce sperms. Select the correct from the following :
A. Spermatogonia always undergo meiotic cell division.
B. Primary spermatocytes divide mitotically to produce secondary spermatocytes.
C. Secondary spermatocytes, through their second meiotic division, produce haploid spermatids.
D. Spermatids produce spermatozoa through mitosis.
E. Spermatids transform into spermatozoa by spermiogenesis.
Choose the correct answer from the options given below:
Q50.
mcq single
+4 / 1
Arrange the following cell layers/structures around the female gamete, from outer to inner side :
A. Zona pellucida
B. Perivitelline space
C. Corona radiata
D. Plasma membrane of ovum
Choose the correct answer from the options given below :
Q51.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Cortisol
I.
Stimulates the formation of alveoli in mammary glands
B.
Aldosterone
II.
Produces anti-inflammatory reactions
C.
Cholecystokinin
III.
Stimulates reabsorption of $\mathrm{Na}^{+}$ and water from renal tubule
D.
Progesterone
IV.
Stimulates secretion of pancreatic enzymes and bile juice
Choose the correct answer from the options given below:
Q52.
mcq single
+4 / 1
Arrange the following steps of somatic hybridisation in a correct sequence.
A. Digestion of cell walls.
B. Isolation of naked protoplasts.
C. Fusion of protoplasts to get hybrid protoplast.
D. Isolation of single cells from two different varieites of plants.
E. Growing of hybrid protoplast to form a new plant.
Choose the correct answer from the options given below:
Q53.
mcq single
+4 / 1
Which of the following is an in situ conservation method?
Q54.
mcq single
+4 / 1
Which of the following statements are correct?
A. The Amazon rainforest being cut and cleared for cultivation of soyabeans is an example of habitat loss.
B. Steller's sea cow and passenger pigeon became extinct due to over-exploitation by humans.
C. The Nile perch introduced into Lake Victoria in East Africa helped in population growth of cichlid fish in the lake.
D. Water hyacinth is an invasive species.
E. When a species becomes extinct, the plant and animal species associated with it are not affected.
Choose the correct answer from the options given below:
Q55.
mcq single
+4 / 1
"The Evil Quartet" of biodiversity loss includes which of the following?
Q56.
mcq single
+4 / 1
Since the origin and diversification of life on Earth, there have been five episodes of mass extinction of species. How is the sixth extinction, which is in progress, different from the previous episodes?
Q57.
mcq single
+4 / 1
Exploring molecular, genetic and species-level diversity for products of economic importance is called
Q58.
mcq single
+4 / 1
$$ \text { Match List-I with List-II } $$
List-I
(Growth Regulator)
List-II
(Function/Effect)
A.
$$
\text { 2,4-D }
$$
I.
Brewing industry
B.
$$
\mathrm{GA}_3
$$
II.
Stimulation of stomatal closure
C.
Kinetin
III.
Herbicide
D.
ABA
IV.
Nutrient mobilisation
Choose the correct answer from the options given below :
Q59.
mcq single
+4 / 1
Heterophyllous development in response to environment is an example of which of the following phenomena?
Q60.
mcq single
+4 / 1
Which one of the following is not a characteristic of plant cells in the phase of elongation?
Q61.
mcq single
+4 / 1
$$ 2\left(\mathrm{C}_{51} \mathrm{H}_{98} \mathrm{C}_6\right)+145 \mathrm{O}_2 \rightarrow 102 \mathrm{CO}_2+98 \mathrm{H}_2 \mathrm{O}+\text { energy } $$
The Respiratory Quotient (RQ) of a biomolecule used for respiration, as per the above equation would be :
Q62.
mcq single
+4 / 1
$$ \text { Match List-I with List-II: } $$
$$
\text { List-I (Process) }
$$
$$
\text { List-II (Location) }
$$
A.
Glycolysis
I.
Inner mitochondrial membrane
B.
ETS
II.
Mitochondrial matrix
C.
Accumulation of protons
III.
Cytoplasm
D.
Krebs' cycle
IV.
Intermembrane space
Choose the correct answer from the options given below:
Q63.
mcq single
+4 / 1
Which of the following statements are correct with reference to human endoskeleton?
A. Human skull is monocondylic.
B. The joint between any two adjoining vertebrae is a cartilaginous joint.
C. In human beings, the number of cervical vertebrae is seven.
D. All ribs except the last 2 pairs are bicephalic.
E. The occipital bone of skull is articulated with atlas vertebra.
Choose the correct answer from the options given below:
Q64.
mcq single
+4 / 1
$$ \text { Match List I with List II related to muscular/skeletal system: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Tetany
I.
Inflammation of joints
B.
Arthritis
II.
Autoimmune disorder affecting neuromuscular junction
C.
Myasthenia gravis
III.
Wild contraction in muscle due to low $\mathrm{Ca}^{++}$in body fluid
D.
Muscular dystrophy
IV.
Progressive degeneration of skeletal muscle
Choose the correct answer from the options given below:
Q65.
mcq single
+4 / 1
Choose the correct statements regarding muscle contraction.
A. A motor neuron carries a signal sent by the Central Nervous System (CNS) to the sarcolemma of the muscle fibre.
B. The neural signal generates an action potential which causes the release of $\mathrm{Ca}^{++}$into sarcoplasm.
C. Increase in $\mathrm{Ca}^{++}$inactivates the actin for breaking cross bridges.
D. Actin binds to the myosin head to form a cross bridge.
E. Shortening of sarcomere takes place, by pulling actin filaments towards the centre of ' A ' band.
Choose the correct answer from the options given below :
Q66.
mcq single
+4 / 1
The main criteria used for Five Kingdom Classification proposed By R.H. Whittaker (1969) included :
A. Cell structure
B. Body organization
C. Presence of flagellum
D. Reproduction
E. Phylogenetic relationships
Choose the correct answer from the options given below :
Q67.
mcq single
+4 / 1
$$ \text { Match List I with List II with respect to chronology of evolution of life forms } $$
List-I
List-II
A.
About 65 mya
I.
Jawless fish probably evolved
B.
About 500 mya
II.
The dinosaurs suddenly disappeared from the earth
C.
About 350 mya
III.
Seaweeds and few plants probably existed
D.
About 320 mya
IV.
Invertebrates were formed and became active
Choose the correct answer from the options given below:
Q68.
mcq single
+4 / 1
Which of the following is not an example of convergent evolution?
Q69.
mcq single
+4 / 1
Evolution of human appears parallel to the progressive development of brain and language skills. As such, the evolution of individual species in the sequence of their appearance is:
Q70.
mcq single
+4 / 1
Arrange the following events occuring in Renin-Angiotensin mechanism in the correct order:
A. Increase in blood pressure and Glomerular filtration rate
B. Reabsorption of $\mathrm{Na}^{+}$and water from distal parts of tubule due to Aldosterone
C. Fall in Glomerular filtration rate
D. Vasoconstriction by Angiotensin II and release of Aldosterone.
E. Renin converts Angiotensinogen into Angiotensin I, followed by Angiotensin II.
Choose the correct answer from the options given below:
Q71.
mcq single
+4 / 1
The JGA (Juxta Glomerular Apparatus) is a special sensitive region formed by cellular modifications in
$\_\_\_\_$ related to the same nephron.
Q72.
mcq single
+4 / 1
Choose the correct statement regarding GIFT to overcome infertility.
Q73.
mcq single
+4 / 1
$$ \text { Match List I and List II } $$
List-I
List-II
A.
Progestasert
I.
Barrier made of rubber used by females
B.
Multiload 375
II.
Oral contraceptive
C.
Diaphragm
III.
Hormone releasing IUD
D.
Saheli
IV.
Copper releasing IUD
Choose the correct answer from the options given below:
Q74.
mcq single
+4 / 1
Which one of the following is a triploid cell?
Q75.
mcq single
+4 / 1
Which one of the following types of pollination brings genetically different types of pollen grains to the stigma?
Q76.
mcq single
+4 / 1
Arrange the following in the correct developmental sequence related to microsporogenesis :
A. Microspore tetrads
B. Sporogenous tissue
C. Pollen grains
D. Pollen mother cells
Choose the correct answer from the options given below :
Q77.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Placentation)
$$
\begin{gathered}
\text { List II } \\
\text { (Example) }
\end{gathered}
$$
A.
Marginal
I.
Mustard
B.
Axile
II.
Pea
C.
Parietal
III.
Marigold
D.
Basal
IV.
Lemon
Choose the correct answer from the options given below :
Q78.
mcq single
+4 / 1
In angiosperms, root hairs arise from which one of the following regions of the root?
Q79.
mcq single
+4 / 1
$$ \text { Which of the following floral formula is the correct floral formula of Solanaceae family? } $$
Q80.
mcq single
+4 / 1
In racemose inflorescence, $\_\_\_\_$ .
Q81.
mcq single
+4 / 1
Which of the following statements are correct regarding amino acids?
A. They are substituted methanes.
B. Serine is an aromatic amino acid.
C. Valine is a neutral amino acid.
D. Lysine is an acidic amino acid.
Choose the correct answer from the options given below:
Q82.
mcq single
+4 / 1
Alpha-helix is found in which level of protein structure?
Q83.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Trypsin
I.
Intercellular ground substance
B.
Morphine
II.
Lectin
C.
Concanavalin A
III.
Enzyme
D.
Collagen
IV.
Alkaloid
Choose the correct answer from the options given below :
Q84.
mcq single
+4 / 1
The following reaction depicts the activity of a particular class of enzymes : Identify the enzymes class ' $E$ ' from the following options :
Identify the enzymes class ' $E$ ' from the following options :
 Identify the enzymes class ' $E$ ' from the following options :
Identify the enzymes class ' $E$ ' from the following options :
Q85.
mcq single
+4 / 1
Identify the correct statements about biomolecules.
A. Lipids are generally water soluble.
B. Proteins are polypeptides.
C. Polysaccharides are long chains of sugars.
D. Adenine and guanine are substituted pyrimidines.
E. Almost all enzymes are proteins.
Choose the correct answer from the options given below :
Q86.
mcq single
+4 / 1
Which of the following statements are correct with respect to DNA separation, isolation and visualization?
A. The cutting of DNA is done by molecular scissors.
B. The DNA fragments separate according to their size in an agarose gel, upon electrophoresis.
C. The separated DNA fragments can be seen without staining when exposed to UV light.
D. The separated DNA fragments, when stained with ethidium bromide, can be seen in visible light.
Choose the correct answer from the options given below :
Q87.
mcq single
+4 / 1
Which of the following statements are not true regarding restriction endonucleases?
A. They are called molecular scissors.
B. These are the enzymes responsible for restricting the growth of bacteriophages in E. coli.
C. They cut the DNA only at the centre of the palindromic sites.
D. They remove nucleotides only from the ends of DNA fragments.
E. They recognise specific palindromic base-pair sequences.
Choose the answer from the options given below :
Q88.
mcq single
+4 / 1
Insertion of a foreign DNA at BamHI site in an E.coli cloning vector pBR322 results in the loss of antibiotic resistance towards:
Q89.
mcq single
+4 / 1
Identify the correct sequence of steps in each cycle of Polymerase Chain Reaction :
Q90.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List-I }
$$
$$
\text { List-II }
$$
A.
Genetically modified organism
I.
Agrobacterium tumefaciens
B.
Thermostable DNA polymerase
II.
Bt cotton
C.
Ti plasmid
III.
Thermus aquaticus
D.
pBR322
IV.
Escherichia coli
Q91.
mcq single
+4 / 1
What is the reason behind production of large holes in 'Swiss Cheese'?
Q92.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I (Bioactive molecules) }
$$
$$
\text { List II (Importance) }
$$
A.
Streptokinase
I.
Immunosuppressive agent
B.
Statins
II.
Removal of clots from the blood vessels
C.
Lipases
III.
Blood cholesterol-lowering agent
D.
Cyclosporin A
IV.
Detergent formulations
Choose the correct answer from the options given below:
Q93.
mcq single
+4 / 1
Ecological pyramids represent the relationship between the organisms at different trophic levels and they are generally inverted for:
Q94.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
$$
\text { List-I }
$$
$$
\text { List-II }
$$
A.
Productivity
I.
Gross primary productivity minus respiration losses
B.
Net primary productivity
II.
Rate of formation of new organic matter by consumers
C.
Gross primary productivity
III.
Rate of biomass production
D.
Secondary productivity
IV.
Rate of production of organic matter during photosynthesis
Choose the correct answer from the options given below :
Q95.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
Decomposition
I.
Accumulation of dark coloured amorphous colloidal substance
B.
Detritus
II.
Release of inorganic nutrients by the activity of microbes in soil
C.
Mineralisation
III.
Breaking down of complex organic matter into inorganic substances.
D.
Humification
IV.
Dead remains of plants and animals including fecal matter
Choose the correct answer from the options given below:
Q96.
mcq single
+4 / 1
What is the probability of having children with 'O' blood group, where both mother and father are heterozygous for ' $\underline{\mathrm{A}}$ ' and ' $\underline{\mathrm{B}}$ ' blood group, respectively?
Q97.
mcq single
+4 / 1
In a population of a grasshopper species, the chromosome number of some members is 23 and some other members possess 24 chromosomes. The 23 and 24 chromosome-bearing members in this species are $\_\_\_\_$ .
Q98.
mcq single
+4 / 1
Which of the following statements are true with reference to the sex-determination in honeybees?
A. An offspring formed from the union of a sperm and an egg, develops as a female (queen or worker).
B. An unfertilized egg develops as a male by parthenogenesis.
C. A male has half the number of chromosomes than that of a female.
D. Males produce sperms by meiosis.
E. Honeybees have a haplodiploid sex-determination system.
Choose the correct answer from the options given below :
Q99.
mcq single
+4 / 1
Which one of the following disorders is caused by the substitution of Glutamic acid (Glu) by Valine (Val) at the sixth position of the beta globin chain of the haemoglobin molecule?
Q100.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
Incomplete dominance
I.
Human skin colour
B.
Co-dominance
II.
Inheritance of flower colour in Antirrhinum sp.
C.
Pleiotropy
III.
Phenylketonuria disease in humans
D.
Polygenic inheritance
IV.
ABO blood groups
Choose the correct answer from the options given below :
Q101.
mcq single
+4 / 1
Male frogs can be distinguished from female frogs due to the presence of
A. Bulging eyes
B. Vocal sacs
C. Webbed digits in feet
D. Copulatory pad on first digit of fore limbs
E. Olive green-coloured skin with dark irregular spots
Choose the correct answer from the options given below
Q102.
mcq single
+4 / 1
Choose the correct statements regarding frog's anatomy:
A. Hepatic portal system is the special venous connection between liver and intestine.
B. There are twelve pairs of cranial nerves arising from the brain.
C. The ureters and oviducts open separately into the cloaca in female frogs.
D. Hind-brain consists of cerebellum, medulla oblongata and optic lobes.
E. Sinus venosus joins the right atrium of heart.
Choose the correct answer from the options given below:
Q103.
mcq single
+4 / 1
The enzyme required for carboxylation in the Calvin cycle is
Q104.
mcq single
+4 / 1
How many ATP and NADPH molecules are required to make one molecule of glucose through the Calvin pathway?
Q105.
mcq single
+4 / 1
Find the incorrect statement(s) about photosynthesis from the following:
A. The water splitting complex is associated with PSI.
B. $\mathrm{C}_4$ plants use the $\mathrm{C}_3$ pathway of $\mathrm{CO}_2$ fixation as the main biosynthetic pathway.
C. In $\mathrm{C}_4$ plants, photorespiration does not occur.
D. $\mathrm{C}_3$ plants exhibit 'Kranz' anatomy.
E. ATP synthesis in chloroplast occurs through chemiosmosis.
Choose the answer from the options given below:
Q106.
mcq single
+4 / 1
Select the set of fishes which belong to the class Osteichthyes:
Q107.
mcq single
+4 / 1
Select the incorrect statements from the following:
A. Digestive system in Platyhelminthes is incomplete.
B. Bilateral symmetry is a characteristic feature of adult Echinoderms.
C. Pseudocoelom is possessed by Aschelminthes.
D. Notochord is persistent throughout life in the class Chondrichthyes.
E. Members of class Reptilia maintain a constant body temperature.
Choose the answer from the options given below:
Q108.
mcq single
+4 / 1
The flightless bird with forelimbs modified as paddle-like structures suited for swimming is known as:
Q109.
mcq single
+4 / 1
A group of researchers procured some fish like animals and upon investigation the following characters were observed:
A. Endoskeleton was made of cartilage.
B. Ectoparasitic; as they were found attached on fish skin with their circular sucking mouth.
C. Paired fins and scales were absent, but 7 pairs of gill slits were present.
Which of the following species of animals did they consider to fit best with these characters?
Q110.
mcq single
+4 / 1
In which one of the following, the ovules are not enclosed by an ovary wall and remain exposed?
Q111.
mcq single
+4 / 1
$$ \text { Match List-I with List-II. } $$
List-I
(Respiratory Volume)
List-II
(Capacity in mL )
A.
ERV (Expiratory Reserve Volume)
I.
$2500-3000 \mathrm{~mL}$
B.
RV (Residual Volume)
II.
$500 \,\,mL$
C.
IRV (Inspiratory Reserve Volume)
III.
$1000-1100 \mathrm{~mL}$
D.
TV (Tidal Volume)
IV.
$1100-1200 \mathrm{~mL}$
Choose the correct answer from the options given below :
Q112.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Molluscs
I.
Pulmonary respiration only
B.
Reptiles
II.
Branchial respiration
C.
Adult amphibians
III.
Cellular respiration
D.
Amoeba
IV.
Pulmonary and cutaneous respiration
Choose the correct answer from the options given below:
Q113.
mcq single
+4 / 1
In humans, respiration occurs in the following steps. Arrange these steps in the correct order.
A. Diffusion of $\mathrm{O}_2$ and $\mathrm{CO}_2$ between blood and tissues
B. Diffusion of $\mathrm{O}_2$ and $\mathrm{CO}_2$ across alveolar membrane
C. Pulmonary ventilation by which atmospheric air is drawn in and $\mathrm{CO}_2$ rich alveolar air is released out
D. Cellular respiration
E. Transport of gases by the blood
Choose the correct answer from the options given below
Q114.
mcq single
+4 / 1
Which one of the following statements is not true about the universal rules of binomial nomenclature?
Q115.
mcq single
+4 / 1
The toxin proteins isolated from Bacillus thuringiensis, coded by which of the following genes would contrl cotton bollworms and corn borer, respectively?
Q116.
mcq single
+4 / 1
The human protein named $\boldsymbol{\alpha}$-1-antitrypsin, obtained from transgenic animals, is used for the treatment of $\_\_\_\_$ .
Q117.
mcq single
+4 / 1
The specific receptors for neurotransmitters in a synapse are present on $\_\_\_\_$ .
Q118.
mcq single
+4 / 1
$$ \text { Match List-I with List-II : } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
Conjunctive tissue
I.
Specialised cells in the vicinity of guard cells
B.
Casparian strips
II.
Endodermal cells rich in starch
C.
Subsidiary cells
III.
Tissue between xylem and phloem
D.
Starch sheath
IV.
Endodermal cells with suberin deposition
Choose the correct answer from the options given below :
Q119.
mcq single
+4 / 1
The main function of bulliform cells in grasses is :
Q120.
mcq single
+4 / 1
Non-membrane bound cell organelles found in both prokaryotic and eukaryotic cells are $\_\_\_\_$ .
Q121.
mcq single
+4 / 1
Choose the correct statements regarding cell organelles and their inclusions.
A. The endomembrane system includes Golgi complex, endoplasmic reticulum and mitochondria.
B. Rough endoplasmic reticulum bears ribosomes on its surface.
C. Both mitochondria and plastids have circular DNA.
D. A network of microtubules, microfilaments and intermediate filaments present in the cytoplasm is called cytoskeleton.
E. Mitochondrion is a single membrane-bound structure.
Choose the correct answer from the options given below :
Q122.
mcq single
+4 / 1
Select the correct statements regarding cell membrane in eukaryotic cell.
A. Membrane of human RBCs has approximately $52 \%$ protein.
B. Major phospholipids are arranged in a bilayer.
C. Extensions of the plasma membrane into the cell form mesosomes.
D. Tails towards the inner part of lipids are hydrophobic and thus protected from aqueous medium.
E. Glycocalyx is present on the outer surface of the plasma membrane.
Choose the correct answer from the options given below:
Q123.
mcq single
+4 / 1
Which one of the following is the site for active ribosomal RNA synthesis?
Q124.
mcq single
+4 / 1
Which of the following statements are correct with reference to a transcription unit?
A. A transcription unit in DNA is defined primarily by three regions : promoter, structural gene and terminator.
B. The promoter is said to be located towards the 5'-end of the structural gene.
C. The promoter is a DNA sequence that provides binding site for RNA polymerase.
D. The promoter defines the template and coding strands.
E. The terminator is located towards the $3^{\prime}$-end of the coding strand and it defines the end of the process of transcription.
Choose the correct answer from the options given below:
Q125.
mcq single
+4 / 1
In the lac operon, the $\boldsymbol{z}$ gene codes for
Q126.
mcq single
+4 / 1
Which of the following statements are correct with reference to packaging of DNA helix ?
A. Histones are organized to form a unit of eight molecules called histone octamer.
B. Histones are negatively charged basic proteins.
C. Histones are rich in the basic amino acid residues - lysine and arginine.
D. The positively charged DNA is wrapped around the histone octamer to form nucleosome.
E. The packaging of chromatin at higher levels requires an additional set of proteins called non-histone chromosomal proteins.
Choose the correct answer from the options given below :
Q127.
mcq single
+4 / 1
The sixth mutant codon of beta globin gene causing polymerization of Haemoglobin and change in RBC shape is $\_\_\_\_$
Q128.
mcq single
+4 / 1
Arrange the following steps of DNA fingerprinting in a correct sequence.
A. Isolation of DNA and its digestion by restriction endonucleases.
B. Hybridisation using a labelled VNTR probe.
C. Transferring of separated DNA fragments to synthetic membranes.
D. Detection of hybridised DNA fragments by autoradiography.
E. Separation of DNA fragments by electrophoresis.
Choose the correct answer from the options given below :
Q129.
mcq single
+4 / 1
The following are the stages of life cycle of Plasmodium. Arrange the stages in the proper order.
A. The parasites reproduce asexually in RBCs, bursting the cells.
B. The parasites reproduce asexually in liver cells, bursting the cells and releasing into blood.
C. Gametocytes develop in RBCs.
D. Sporozoites reach the liver through the blood.
E. Female mosquito injects sporozoites into humans during bite.
Choose the correct answer from the options given below:
Q130.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I (Drug) }
$$
$$
\text { List II (Effect) }
$$
A.
Nicotine
I.
Causes sense of euphoria and increased energy
B.
Morphine
II.
Stimulates adrenal gland to release catecholamines into blood circulation
C.
Heroin
III.
Effective sedative and painkiller
D.
Cocaine
IV.
A depressant; slows down body function
Choose the correct answer from the options given below:
Q131.
mcq single
+4 / 1
In which animal do haploid cells divide mitotically to produce gametes?
Q132.
mcq single
+4 / 1
Which one of the following is an appropriate example of sexual deceit?
Q133.
mcq single
+4 / 1
Choose the correct statements regarding population interactions between two species.
A. In both parasitism and commensalism, only one species benefits and the other species is harmed.
B. Both species benefit in mutualism.
C. Both species benefit in commensalism.
D. In parasitism, only one species benefits and the other species is harmed.
E. In amensalism, one species is harmed and the other is unaffected.
Choose the correct answer from the options given below:
Q134.
mcq single
+4 / 1
Which of the following equations depicts Verhulst-Pearl logistic population growth?
Q135.
mcq single
+4 / 1
The WBC count of a person's blood sample is $8000 / \mathrm{cu} \mathrm{mm}$. How many eosinophils and lymphocytes would be in the same blood sample approximately?
Q136.
mcq single
+4 / 1
Select the incorrect statement with reference to Rh grouping.
A. Erythroblastosis foetalis is a condition observed having foetus with $\mathrm{Rh}^{-\mathrm{ve}}$ blood and mother with $\mathrm{Rh}^{+\mathrm{ve}}$ blood.
B. Rh antigen is observed on RBCs in the majority of human beings.
C. Before blood transfusion, Rh group should also be matched.
D. Rh incompatibility is observed when a pregnant mother is $\mathrm{Rh}^{-\mathrm{ve}}$ and the foetus is $\mathrm{Rh}^{+\mathrm{ve}}$.
E. Erythroblastosis foetalis can be avoided by administering anti-Rh antibodies to the mother immediately after the delivery of the second child.
Choose the answer from the options given below :
(1) C and D only
(2) A and B only
(3) A and E only
(4) B and C onlySelect the incorrect statement with reference to Rh grouping.
A. Erythroblastosis foetalis is a condition observed having foetus with $\mathrm{Rh}^{-\mathrm{ve}}$ blood and mother with $\mathrm{Rh}^{+\mathrm{ve}}$ blood.
B. Rh antigen is observed on RBCs in the majority of human beings.
C. Before blood transfusion, Rh group should also be matched.
D. Rh incompatibility is observed when a pregnant mother is $\mathrm{Rh}^{-\mathrm{ve}}$ and the foetus is $\mathrm{Rh}^{+\mathrm{ve}}$.
E. Erythroblastosis foetalis can be avoided by administering anti-Rh antibodies to the mother immediately after the delivery of the second child.
Choose the answer from the options given below :
Q137.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List-I
(Phase of cell cycle)
List-II
(Activity)
A.
$\mathrm{G}_1$ phase
I.
Actual cell division occurs
B.
S phase
II.
Cell is metabolically active and continuously grows but does not replicate its DNA
C.
$\mathrm{G}_2$ Phase
III.
Synthesis of DNA occurs and the amount of DNA per cell doubles
D.
M phase
IV.
Proteins are synthesized while cell growth continues
Choose the correct answer from the options given below :
Q138.
mcq single
+4 / 1
$$ \text { Match List I with List II related to embryonic development at various months of pregnancy: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
The foetus movement starts and hair appears on the head
I.
24 weeks of pregnancy
B.
The foetus develops limbs and digits
II.
20 weeks of pregnancy
C.
The foetus develops external genital organs
III.
8 weeks of pregnancy
D.
The foetus body is covered with fine hair; eyelids separate and eyelashes are formed
IV.
12 weeks of pregnancy
Choose the correct answer from the options given below:
Q139.
mcq single
+4 / 1
Spermatogonia undergo a series of cell divisions statements to produce sperms. Select the correct from the following :
A. Spermatogonia always undergo meiotic cell division.
B. Primary spermatocytes divide mitotically to produce secondary spermatocytes.
C. Secondary spermatocytes, through their second meiotic division, produce haploid spermatids.
D. Spermatids produce spermatozoa through mitosis.
E. Spermatids transform into spermatozoa by spermiogenesis.
Choose the correct answer from the options given below:
Q140.
mcq single
+4 / 1
Arrange the following cell layers/structures around the female gamete, from outer to inner side :
A. Zona pellucida
B. Perivitelline space
C. Corona radiata
D. Plasma membrane of ovum
Choose the correct answer from the options given below :
Q141.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Cortisol
I.
Stimulates the formation of alveoli in mammary glands
B.
Aldosterone
II.
Produces anti-inflammatory reactions
C.
Cholecystokinin
III.
Stimulates reabsorption of $\mathrm{Na}^{+}$ and water from renal tubule
D.
Progesterone
IV.
Stimulates secretion of pancreatic enzymes and bile juice
Choose the correct answer from the options given below:
Q142.
mcq single
+4 / 1
Arrange the following steps of somatic hybridisation in a correct sequence.
A. Digestion of cell walls.
B. Isolation of naked protoplasts.
C. Fusion of protoplasts to get hybrid protoplast.
D. Isolation of single cells from two different varieites of plants.
E. Growing of hybrid protoplast to form a new plant.
Choose the correct answer from the options given below:
Q143.
mcq single
+4 / 1
Which of the following is an in situ conservation method?
Q144.
mcq single
+4 / 1
Which of the following statements are correct?
A. The Amazon rainforest being cut and cleared for cultivation of soyabeans is an example of habitat loss.
B. Steller's sea cow and passenger pigeon became extinct due to over-exploitation by humans.
C. The Nile perch introduced into Lake Victoria in East Africa helped in population growth of cichlid fish in the lake.
D. Water hyacinth is an invasive species.
E. When a species becomes extinct, the plant and animal species associated with it are not affected.
Choose the correct answer from the options given below:
Q145.
mcq single
+4 / 1
"The Evil Quartet" of biodiversity loss includes which of the following?
Q146.
mcq single
+4 / 1
Since the origin and diversification of life on Earth, there have been five episodes of mass extinction of species. How is the sixth extinction, which is in progress, different from the previous episodes?
Q147.
mcq single
+4 / 1
Exploring molecular, genetic and species-level diversity for products of economic importance is called
Q148.
mcq single
+4 / 1
$$ \text { Match List-I with List-II } $$
List-I
(Growth Regulator)
List-II
(Function/Effect)
A.
$$
\text { 2,4-D }
$$
I.
Brewing industry
B.
$$
\mathrm{GA}_3
$$
II.
Stimulation of stomatal closure
C.
Kinetin
III.
Herbicide
D.
ABA
IV.
Nutrient mobilisation
Choose the correct answer from the options given below :
Q149.
mcq single
+4 / 1
Heterophyllous development in response to environment is an example of which of the following phenomena?
Q150.
mcq single
+4 / 1
Which one of the following is not a characteristic of plant cells in the phase of elongation?
Q151.
mcq single
+4 / 1
$$ 2\left(\mathrm{C}_{51} \mathrm{H}_{98} \mathrm{C}_6\right)+145 \mathrm{O}_2 \rightarrow 102 \mathrm{CO}_2+98 \mathrm{H}_2 \mathrm{O}+\text { energy } $$
The Respiratory Quotient (RQ) of a biomolecule used for respiration, as per the above equation would be :
Q152.
mcq single
+4 / 1
$$ \text { Match List-I with List-II: } $$
$$
\text { List-I (Process) }
$$
$$
\text { List-II (Location) }
$$
A.
Glycolysis
I.
Inner mitochondrial membrane
B.
ETS
II.
Mitochondrial matrix
C.
Accumulation of protons
III.
Cytoplasm
D.
Krebs' cycle
IV.
Intermembrane space
Choose the correct answer from the options given below:
Q153.
mcq single
+4 / 1
Which of the following statements are correct with reference to human endoskeleton?
A. Human skull is monocondylic.
B. The joint between any two adjoining vertebrae is a cartilaginous joint.
C. In human beings, the number of cervical vertebrae is seven.
D. All ribs except the last 2 pairs are bicephalic.
E. The occipital bone of skull is articulated with atlas vertebra.
Choose the correct answer from the options given below:
Q154.
mcq single
+4 / 1
$$ \text { Match List I with List II related to muscular/skeletal system: } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Tetany
I.
Inflammation of joints
B.
Arthritis
II.
Autoimmune disorder affecting neuromuscular junction
C.
Myasthenia gravis
III.
Wild contraction in muscle due to low $\mathrm{Ca}^{++}$in body fluid
D.
Muscular dystrophy
IV.
Progressive degeneration of skeletal muscle
Choose the correct answer from the options given below:
Q155.
mcq single
+4 / 1
Choose the correct statements regarding muscle contraction.
A. A motor neuron carries a signal sent by the Central Nervous System (CNS) to the sarcolemma of the muscle fibre.
B. The neural signal generates an action potential which causes the release of $\mathrm{Ca}^{++}$into sarcoplasm.
C. Increase in $\mathrm{Ca}^{++}$inactivates the actin for breaking cross bridges.
D. Actin binds to the myosin head to form a cross bridge.
E. Shortening of sarcomere takes place, by pulling actin filaments towards the centre of ' A ' band.
Choose the correct answer from the options given below :
Q156.
mcq single
+4 / 1
The main criteria used for Five Kingdom Classification proposed By R.H. Whittaker (1969) included :
A. Cell structure
B. Body organization
C. Presence of flagellum
D. Reproduction
E. Phylogenetic relationships
Choose the correct answer from the options given below :
Q157.
mcq single
+4 / 1
$$ \text { Match List I with List II with respect to chronology of evolution of life forms } $$
List-I
List-II
A.
About 65 mya
I.
Jawless fish probably evolved
B.
About 500 mya
II.
The dinosaurs suddenly disappeared from the earth
C.
About 350 mya
III.
Seaweeds and few plants probably existed
D.
About 320 mya
IV.
Invertebrates were formed and became active
Choose the correct answer from the options given below:
Q158.
mcq single
+4 / 1
Which of the following is not an example of convergent evolution?
Q159.
mcq single
+4 / 1
Evolution of human appears parallel to the progressive development of brain and language skills. As such, the evolution of individual species in the sequence of their appearance is:
Q160.
mcq single
+4 / 1
Arrange the following events occuring in Renin-Angiotensin mechanism in the correct order:
A. Increase in blood pressure and Glomerular filtration rate
B. Reabsorption of $\mathrm{Na}^{+}$and water from distal parts of tubule due to Aldosterone
C. Fall in Glomerular filtration rate
D. Vasoconstriction by Angiotensin II and release of Aldosterone.
E. Renin converts Angiotensinogen into Angiotensin I, followed by Angiotensin II.
Choose the correct answer from the options given below:
Q161.
mcq single
+4 / 1
The JGA (Juxta Glomerular Apparatus) is a special sensitive region formed by cellular modifications in
$\_\_\_\_$ related to the same nephron.
Q162.
mcq single
+4 / 1
Choose the correct statement regarding GIFT to overcome infertility.
Q163.
mcq single
+4 / 1
$$ \text { Match List I and List II } $$
List-I
List-II
A.
Progestasert
I.
Barrier made of rubber used by females
B.
Multiload 375
II.
Oral contraceptive
C.
Diaphragm
III.
Hormone releasing IUD
D.
Saheli
IV.
Copper releasing IUD
Choose the correct answer from the options given below:
Q164.
mcq single
+4 / 1
Which one of the following is a triploid cell?
Q165.
mcq single
+4 / 1
Which one of the following types of pollination brings genetically different types of pollen grains to the stigma?
Q166.
mcq single
+4 / 1
Arrange the following in the correct developmental sequence related to microsporogenesis :
A. Microspore tetrads
B. Sporogenous tissue
C. Pollen grains
D. Pollen mother cells
Choose the correct answer from the options given below :
Q167.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
List I
(Placentation)
$$
\begin{gathered}
\text { List II } \\
\text { (Example) }
\end{gathered}
$$
A.
Marginal
I.
Mustard
B.
Axile
II.
Pea
C.
Parietal
III.
Marigold
D.
Basal
IV.
Lemon
Choose the correct answer from the options given below :
Q168.
mcq single
+4 / 1
In angiosperms, root hairs arise from which one of the following regions of the root?
Q169.
mcq single
+4 / 1
$$ \text { Which of the following floral formula is the correct floral formula of Solanaceae family? } $$
Q170.
mcq single
+4 / 1
In racemose inflorescence, $\_\_\_\_$ .
Q171.
mcq single
+4 / 1
Which of the following statements are correct regarding amino acids?
A. They are substituted methanes.
B. Serine is an aromatic amino acid.
C. Valine is a neutral amino acid.
D. Lysine is an acidic amino acid.
Choose the correct answer from the options given below:
Q172.
mcq single
+4 / 1
Alpha-helix is found in which level of protein structure?
Q173.
mcq single
+4 / 1
$$ \text { Match List I with List II : } $$
$$
\text { List I }
$$
$$
\text { List II }
$$
A.
Trypsin
I.
Intercellular ground substance
B.
Morphine
II.
Lectin
C.
Concanavalin A
III.
Enzyme
D.
Collagen
IV.
Alkaloid
Choose the correct answer from the options given below :
Q174.
mcq single
+4 / 1
The following reaction depicts the activity of a particular class of enzymes : Identify the enzymes class ' $E$ ' from the following options :
Identify the enzymes class ' $E$ ' from the following options :
 Identify the enzymes class ' $E$ ' from the following options :
Identify the enzymes class ' $E$ ' from the following options :
Q175.
mcq single
+4 / 1
Identify the correct statements about biomolecules.
A. Lipids are generally water soluble.
B. Proteins are polypeptides.
C. Polysaccharides are long chains of sugars.
D. Adenine and guanine are substituted pyrimidines.
E. Almost all enzymes are proteins.
Choose the correct answer from the options given below :
Q176.
mcq single
+4 / 1
Which of the following statements are correct with respect to DNA separation, isolation and visualization?
A. The cutting of DNA is done by molecular scissors.
B. The DNA fragments separate according to their size in an agarose gel, upon electrophoresis.
C. The separated DNA fragments can be seen without staining when exposed to UV light.
D. The separated DNA fragments, when stained with ethidium bromide, can be seen in visible light.
Choose the correct answer from the options given below :
Q177.
mcq single
+4 / 1
Which of the following statements are not true regarding restriction endonucleases?
A. They are called molecular scissors.
B. These are the enzymes responsible for restricting the growth of bacteriophages in E. coli.
C. They cut the DNA only at the centre of the palindromic sites.
D. They remove nucleotides only from the ends of DNA fragments.
E. They recognise specific palindromic base-pair sequences.
Choose the answer from the options given below :
Q178.
mcq single
+4 / 1
Insertion of a foreign DNA at BamHI site in an E.coli cloning vector pBR322 results in the loss of antibiotic resistance towards:
Q179.
mcq single
+4 / 1
Identify the correct sequence of steps in each cycle of Polymerase Chain Reaction :
Q180.
mcq single
+4 / 1
$$ \text { Match List I with List II: } $$
$$
\text { List-I }
$$
$$
\text { List-II }
$$
A.
Genetically modified organism
I.
Agrobacterium tumefaciens
B.
Thermostable DNA polymerase
II.
Bt cotton
C.
Ti plasmid
III.
Thermus aquaticus
D.
pBR322
IV.
Escherichia coli